Abstract
Salvianolic acid B (SB) is an antioxidant derived from Salvia militarize, and is one of the most widely used herbs in traditional Chinese medicine. SB is a potent antioxidant that has been well documented as a scavenger of oxygen free radicals, and has been used for the prevention and treatment of atherosclerosis-associated disorders. To explore its potential therapeutic effects in treating radiation damage, in this study, mice were treated with SB at different doses of 5, 12.5 and 20 mg/kg, subsequent to receiving γ-irradiation. The effects of SB on peripheral blood, bone marrow nucleated cells, spleen and thymus indices, and oxidation resistance were evaluated in both radiated mice and control groups. The results indicated that SB significantly increased the counts of peripheral white blood cells, red blood cells and platelets. The number of nucleated cells in the bone marrow and the level of protein increased as well. In addition, improved spleen and thymus indices in the bone marrow were observed. SB treatment additionally reversed the deterioration of both the thymus and spleen indices, which is associated with increased serum superoxide dismutase activity and decreasing malondialdehyde levels via nuclear factor (erythroid-derived 2)-like 2 protein/BTB and CNC homology 1 mediated antioxidant effect. Furthermore, ROS levels and Bax protein expression were also suppressed by SB. The data suggested that SB is effective in protecting mice from γ-radiation injury, and could potentially be applicable for clinical use. Notably, the present study identified a promising candidate drug for enhancing the hematopoietic and immune systems.
Keywords: γ-radiation, salvianolic acid B, peripheral blood, oxidative damage, Nrf2, Bach1
Introduction
Radiation is an important environmental factor that affects human health. Previous studies have suggested that ionizing radiation causes direct and indirect cell death via DNA strand breaks and free radical formation (1). Free radicals destroy cellular membranes, which are made of polyunsaturated fatty acids and are highly susceptible to oxidative damage, eventually resulting in programmed cell death (2,3). In addition, oxidative damage leading to lipid peroxidation activates cellular components that may have serious effects on cells, and results in various of diseases (4–6). Due to developments in and application of nuclear technology in industry, agriculture and medicine, exposure to ionizing radiation may occur in nuclear accidents and cancer treatment, thus, the likelihood of people suffering from radiation damage has increased. This harmful environment can potentially cause genetic mutations and damage the human hematopoietic, immune, reproductive, digestive and nervous systems (7,8).
In an attempt to find effective, reliable and inexpensive anti-radiation drugs, research has been conducted into identifying plant-derived radio-protectors. At present, numerous natural plant-based compounds have displayed a diverse array of biological activities that may be relevant to the mitigation of ionizing radiation-induced damage in mammalian systems. These include puerarin, tea polyphenols, emodin and quercetin (9–13), which could potentially protect against γ-radiation-induced toxicity by inhibiting DNA damage and oxidative stress.
Salvia militarize, also known as Danshen, is one of the most widely used herbs in traditional Chinese medicine. It has been demonstrated to have positive effects on stasis, blood flow activation and menstruation, thus has been used to treat a variety of diseases (14). Modern pharmacological studies have demonstrated that Salvia protects vascular endothelial cells, acts as an anti-arrhythmic, works to combat atherosclerosis, improves microcirculation, protects the heart, increases coronary blood flow, improves myocardial ischemia, has an anti-inflammatory act against lipid peroxidation and pulmonary fibrosis, scavenges radicals and protects liver cells (15–20). It has also been used in the treatment of coronary artery disease and other cardiovascular disorders (21).
The primary ingredients of Salvia militarize include fat-soluble components, such as tanshinone, cryptotanshinone and water-soluble components, including danshensu, salvianolic acid A, B, and C. The most abundant constituent among the water-soluble compounds is, Salvianolic acid B (SB; Fig. 1) which is a potent antioxidant and acts as a scavenger of oxygen free radicals to protect against ischemia-reperfusion injuries in heart (22), spinal cord I/R injury (23) and 6-hydroxydopamine induced apoptosis in SH-SY5Y cells (24). However, no study investigating the protective effects of SB on radiation-induced oxidative damage has been reported. Therefore, the aim of the current study was to evaluate if SB treatment had a protective effect on mice injured by ionizing irradiation and to elucidate its potential pharmacological mechanism.
Figure 1.
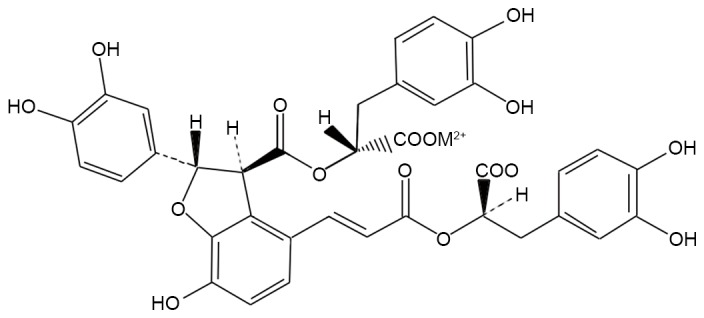
Chemical structure of salvianolic acid B.
Materials and methods
Reagents
Salvianolic acid B (purity >98%) was purchased from the National Institute for the Control of Pharmaceutical and Biological Products (Beijing, China) and dissolved in saline. Ethinylestradiol (purity >98%), used as a positive control drug for treating radiation induced damage, was purchased from Adamas Reagent Ltd. (Shanghai, China) and dissolved in oil for injection. The superoxide dismutase (SOD) and malondialdehyde (MDA) kits were purchased from Nanjing Jiancheng Bioengineering Institute (Nanjing, China). Cell lysis buffer was obtained from Cell Signaling Technology, Inc. (Shanghai, China), the bicinchoninic acid (BCA) protein quantification kit (cat. no. W041), erythrocyte diluents, platelet diluents and leukocytes diluents were all purchased from Nanjing Jiancheng Bioengineering Institute (Nanjing, China). The other chemicals used were of reagent grade from Sinopharm Chemical Reagent Co., Ltd (Shanghai, China).
Animals
A total of 30 male and 30 female Kuming (KM) mice of SPF grade, weighing 18–20 g were obtained from the Laboratory Animal Services Center, Guangzhou University of Chinese Medicine (Guangzhou, China). They were acclimatized in an air-conditioned room maintained at a temperature between 20–25°C, humidity of 55±5%, 12-h light/dark cycle and with free access to food and water. The animal studies were approved by the Animal Ethics Committee of Guangzhou University of Chinese Medicine.
γ-irradiation and drug administration
Following a week of acclimation, the KM mice were randomly divided into six groups with ten mice in each group: Normal group, model group (radiation injury only), ethinylestradiol-positive control group, and three SB dosage groups (low, SB-L; medium, SB-M; and high, SB-H). The mice in the positive control group were injected intraperitoneally with 5 mg/kg of ethynylestradiol, the mice in the SB groups were treated with 5, 12.5, 20 mg/kg of SB, respectively, while mice in normal and model groups were injected with 0.1 ml/10 g of body weight/day of saline. To achieve bone marrow suppression, all mice, except those in the normal group, were exposed to 60Co-γ ray radiation (Foshan Plastics Group Co., Ltd., Foshan, China) at a total dose of 6 Gy (0.1 Gy/min, for 185 sec). Drug administration started from day 1 post-irradiation, and lasted for a total of 14 days, with one dose each day.
Determination of peripheral blood cell counts
Blood was drawn from the tail vein of all the groups of mice pre-irradiation and on days 3, 7 and 14 post-irradiation and then mixed with erythrocyte diluents, leukocyte diluents and platelet diluents, respectively. The peripheral white blood cell (WBC), red blood cell (RBC) and platelet (PLT) counts were then determined.
Bone marrow nucleated cell counts
A total of 1 h after the final drug administration on day 14, all mice were sacrificed by cervical dislocation and the bone marrows were flushed from the whole femoral bone with 1 ml phosphate-buffered saline (PBS) and mixed homogenously to make a cell suspension. The cell suspension was added to a solution of 60% lymphocyte separation medium (density 1.077 g/ml, Cat. no. 17-829E, Lonza Group, Ltd., Basel, Switzerland), and centrifuged using the Heraeus Laboratory Centrifuge (Model Biofuge Pico, Heraeus, Germany) at 18°C at 300 × g for 30 min. The white ring layer was separated by a sterile plastic pipette after the plasma fluid was deprived. The nucleated cells were diluted with PBS and centrifuged under the same conditions. The cell pellets were then suspended homogenously, and were used to count nucleated cells under the optical microscope.
Determination of protein content of bone marrow cells
Lysis buffer was added to the bone marrow cell suspension, incubated on ice for 30 min, and centrifuged using the Heraeus Laboratory Centrifuge at 4°C at 13,000 × g for 10 min. The supernatant was separated, dispensed, stored at −80°C. The protein concentration was determined by the BCA method.
Thymus and spleen indices
On day 14 post-radiation, all animals were sacrificed and the spleen and thymus were removed and weighed. The thymus and spleen indices were calculated by dividing organ weight by body weight according to the following formulae. Thymus index=thymus weight (mg)/body weight (g)*10; spleen index=spleen weight (mg)/body weight (g)*10.
Measures of antioxidant capacity
On day 14 post-radiation, blood samples were obtained from the post-ocular venous plex from mice that were fasted overnight. They were anesthetized with diethyl ether. The blood was allowed to clot at room temperature for 1 h prior to centrifugation using the Heraeus Laboratory Centrifuge at 4°C at 1,200 × g for 15 min to separate the serum. Serum SOD and MDA levels were detected by ELISA test kits (Nanjing Jiancheng Bioengineering Institute, Nanjing, China) according to the manufacturer's instructions.
Reverse transcription-quantitative polymerase chain reaction (RT-qPCR)
The expression levels of BTB and CNC homology 1 (Bach1) and nuclear factor (erythroid-derived 2)-like 2 protein (Nrf2) were determined using RT-qPCR. RNA extraction was prepared using TRIzol reagent (Takara Biotechnology Co., Ltd., Dalian, China) according to the manufacturer's instructions. A Total RNA (1 mg) was used to synthesize the first strand of cDNA using Bestar qPCR RT kit (DBI Bioscience, Heidelberg, Germany). The mRNA expression was evaluated by qPCR on Stratagene Mx3000P Real time PCR platform (Agilent Technologies, Inc., Santa Clara, CA, USA) with SYBR Green PCR core reagents. The PCR reaction system (20 µl) contained qPCR master mix 10 µl, plus forward and reverse primers (10 µM) 1.0 µl, cDNA template 1 µl and ddH2O 8 µl. β-actin was applied as the internal reference. The following primers were synthesized and applied: Nrf2, forward 5′-GGTTGCCCACATTCCCAAAT-3′ and reverse 5′-AGCAATGAAGACTGGGCTCT3′; Bach1, forward 5′-TAGTGTGGAGCGAGAAGTGG-3′ and reverse 5′-ACCTAACCACGGACACTCAG-3′; β-actin, forward 5′-CATTGCTGACAGGATGCAGA-3′ and reverse 5′-CTGCTGGAAGGTGGACAGTGA-3′. The reaction procedure was initiated with denaturation at 94°C for 2 min and followed by 40 repeated cycles (denaturation at 94°C for 20 sec, annealing at 58°C for 20 sec and extension at 72°C for 20 sec). The Ct-value for each sample was calculated with the ΔΔCq-method (25), and the results were expressed as 2−ΔΔCq to analyze the fold change.
Western blotting
Western blot analysis was performed as described previously (26). Briefly, portions of liver were homogenized for each group and nucleoprotein was extracted from samples by using Nuclear Protein Extraction kit (Beyotime Institute of Biotechnology, Wuhan, China) according to the manufacturer's instructions. Proteins (20–40 µg) were loaded in each lane. Membranes were incubated overnight at 4°C with anti-Nrf2 (cat. no. ab89443; 1:1,000; Abcam, Cambridge, UK), anti-Bach1 (cat. no. NBP1-88722; 1:500; Novus Biologicals Ltd., Cambridge, UK) and anti-Bax (cat. no. 2772; 1:1,000; Cell Signaling Technology, Inc., Danvers, MA, USA) antibodies. Blots were subsequently incubated with horseradish peroxidase-conjugated secondary antibodies (cat nos. A4416 and A6154; 1:10,000; Sigma-Aldrich; Merck KGaA) for 1 h at room temperature. Histone 3 (Sigma-Aldrich; Merck KGaA) served as a control.
Cellular ROS detection assay
ROS were detected with 2,7-dichlorofluorescein diacetate (DCFH-DA; Beyotime Institute of Biotechnology) according to the manufacturers' instructions. Livers were obtained from the mice of each group. Collagenase and trypsin were added to enzymolyze liver tissues into single-cell suspension. Cells were incubated with DCFH-DA at a final concentration of 10 mM for 30 min and washed 3 times with HEPES buffer in the dark. Subsequently cells were washed with PBS three times. Following centrifugation, cell pellets were suspended in PBS for immediate analysis by flow cytometry.
Statistical analysis
All data were expressed as the mean ± standard deviation. SPSS 17.0 software (SPSS, Inc., Chicago, IL, USA) was used for statistical analysis. One-way analysis of variance was followed by Tukey's post-hoc test. Student's t-test analysis was used for assessing significant differences between the normal and model groups. P<0.05 was considered to indicate a statistically significant difference.
Results
Effect of SB on peripheral blood count of irradiated mice
Bone marrow is highly sensitive to radiation and exposure often damages the hematopoietic cells. Thus, it was investigated whether treatment with SB had beneficial effects on hematopoietic function of irradiated mice. To this end, the cell counts of peripheral WBC, RBC and PLT were measured with and without treatment of SB from radiated mice. As presented in Fig. 2, the trend of peripheral WBC, RBC and PLT in mice were in the normal range and no significant differences were observed between the groups prior to radiation. However, following exposure to 60Co-γ radiation at a dose of 60 Gy, the peripheral WBC, RBC and PLT counts decreased significantly after γ radiation comparing with the normal group (P<0.01). Although cell counts in the model group exhibited a marginal auto-recovery on days 7 and 14, SB treatment significantly raised the count of WBC, RBC and PLT on days 3, 7 and 14, compared with the model group. These results suggest that SB alleviates the reduction in the number of peripheral WBC, RBC and PLT induced by radiation and resulted in less damage to the mice.
Figure 2.
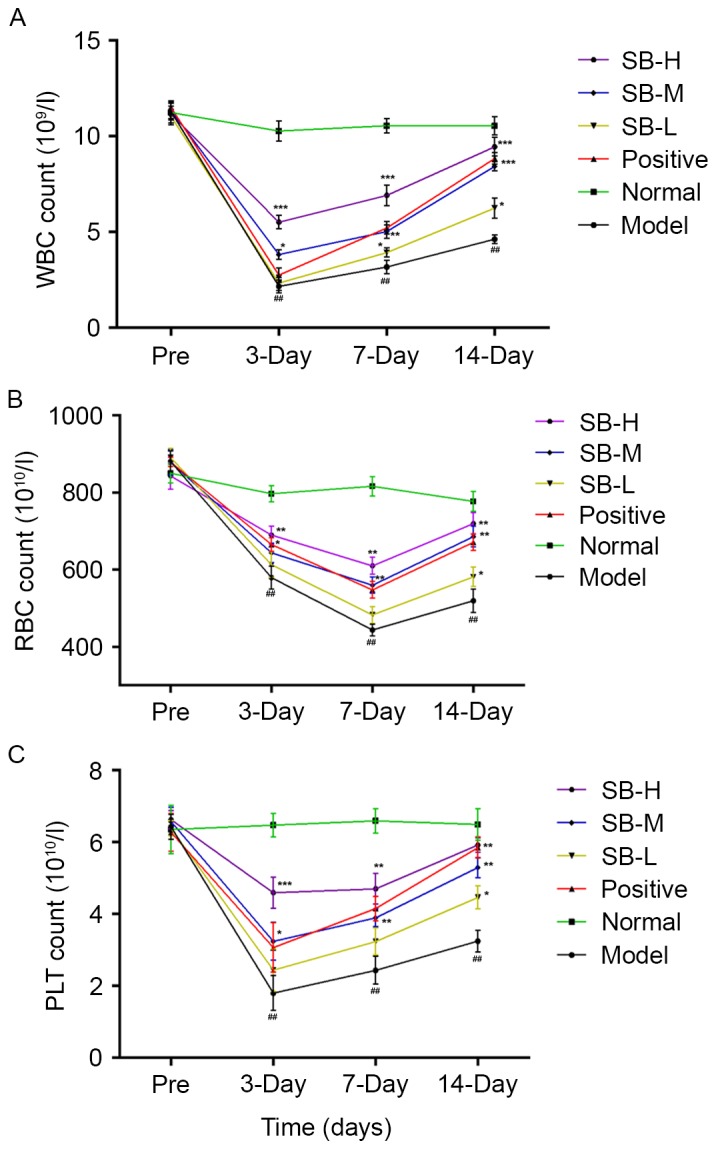
Effect of SB on peripheral (A) WBC, (B) RBC and (C) PLT counts of irradiated mice induced by 60Co-γ radiation with n=10 mice in each group pre-irradiation and on days 3, 7 and 14 post-irradiation. Data were expressed as the mean ± standard error; with n=10 mice in each group. ##P<0.01 (model vs. normal), *P<0.05, **P<0.01 and ***P<0.001 SB treatment group vs. the model group. SB, salvianolic acid B; WBC, white blood cell; RBC, red blood cell; PLT, platelet.
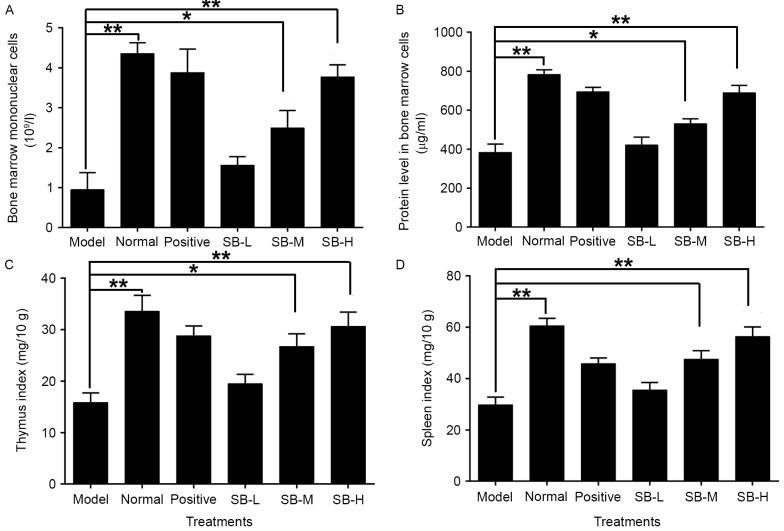
Effect of SB on nucleated cells in bone marrow of radiated mice
Radiation destroys the microenvironment of the bone marrow, thereby affecting hematopoietic function. The decrease in nucleated cells of the bone marrow indicates disorders of hematopoietic system. The number of nucleated cells in the bone marrow decreased significantly (P<0.01) in the model group as compared with the normal group (Fig. 3A). Administering the mice with three different doses of SB noticeably increased the number of nucleated cells in the bone marrow suggesting that treatment with SB may have a protective effect on bone marrow hematopoietic cells.
Figure 3.
Effect of SB on bone marrow (A) mononuclear cells counts, (B) protein content of bone marrow cells, (C) thymus index and (D) spleen index in mice exposed to 60Co-γ radiation. Data were expressed as mean ± standard error; with n=10 mice in each group. **P<0.01 model vs. normal, *P<0.05 and **P<0.01 SB treatment group vs. the model group.
Impact of SB on protein synthesis in bone marrow cells of radiated mice
A significant proportion of damage resulting from ionizing radiation is associated with the production of reactive oxygen species (ROS), which oxidize DNA, proteins, lipids and cofactors. Ionizing radiation breaks DNA strands and indirectly impact protein synthesis of bone marrow cells. In addition, radiation directly damages protein, including ROS-induced protein chemical alterations via carbonization and nitrosylation (27). Thus, protein synthesis was assessed in mice exposed to radiation. Ionizing radiation markedly decreased the protein content in bone marrow cells as compared with the normal group (P<0.01; Fig. 3B), whereas the treatment of SB significantly increased the protein levels in bone marrow cells. This suggests that SB may protect bone marrow cells from oxidative damage of protein.
Effect of SB on the spleen and thymus of radiated mice
Radiation damage to the thymus and spleen destroys the immune system, increasing the probability of complications. To investigate the effect of SB on the thymus and spleen of irradiated mice, the thymus and spleen indices were evaluated. As presented in Fig. 3C and D, the thymus and spleen indices were both reduced significantly as compared with the normal group (P<0.01), whereas treatment with three different doses of SB significantly increased the thymus and spleen indices of irradiated mice in a dose-dependent manner. This suggests that SB treatment can significantly neutralize the damage caused by radiation in the spleen and thymus of radiated mice.
Effect of SB on oxidation resistance in radiated mice
Ionizing radiation affects the formation of free oxygen radicals. These act on the cellular membrane and cause lipid peroxidation, resulting in functional and metabolic changes of the membrane. To evaluate the effect of SB on oxidative stress, the levels of serum SOD and MDA were assessed. SOD activity of radiated mice in serum was significantly increased (P<0.01), whereas MDA levels were significantly reduced comparing with the normal group (P<0.01; Fig. 4). SB treatment markedly increased MDA levels and down-regulated SOD levels, in a dose-dependent manner comparing with the model group. Meanwhile, the expression level of Nrf2 in radiated mice decreased and Bach1, which competed with Nrf2 for binding to antioxidant response element, was markedly increased (P<0.001, Fig. 5A and B). Notably, this changing tendency was completely reversed following treatment with SB compared with the model group (Fig. 5A and B), which indicates that SB has a protective effect on ionizing radiation injury in mice by activating Nrf2-mediated antioxidant effect. These tendencies were confirmed by western blotting (Fig. 5C).
Figure 4.
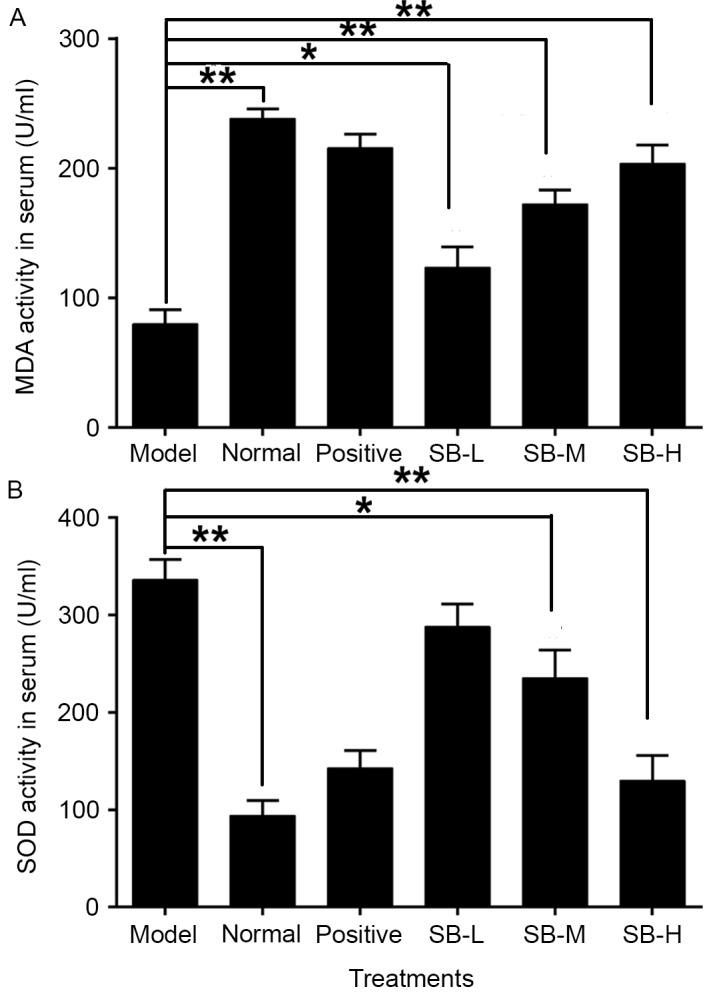
Effect of SB on serum (A) MDA and (B) SOD levels in ionizing radiation injured mice. Data were expressed as mean ± standard error; with n=10 mice in each group. **P<0.01 model group vs. normal group, *P<0.05 and **P<0.01 SB treatment group vs. the model group. SB, salvianolic acid B; MDA, malondialdehyde; SOD, superoxide dismutase.
Figure 5.
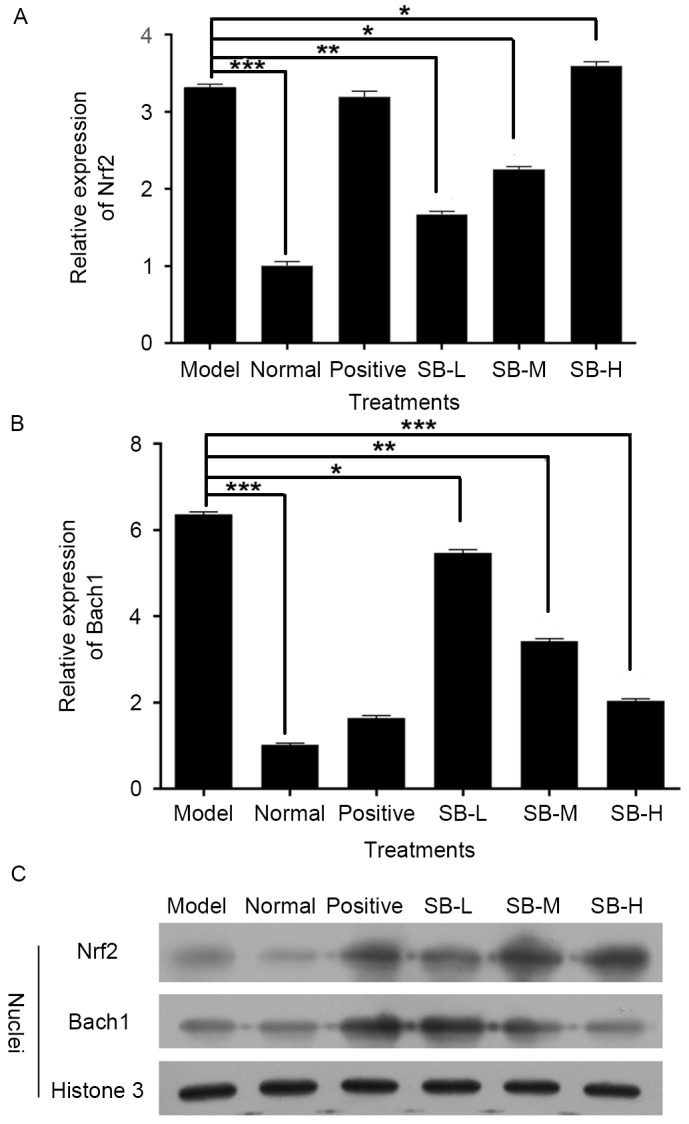
Effect of SB on the (A and B) Mrna (C) and nuclear protein levels of Nrf2 and Bach1 in mice. Data were expressed as the mean ± standard error; with n=10 mice in each group. ***P<0.001 model group vs. normal group, *P<0.05, **P<0.01 and ***P<0.001 SB treatment group vs. the model group. SB, salvianolic acid B; Nrf2, nuclear factor (erythroid-derived 2)-like 2 protein; Bach1, BTB and CNC homology 1.
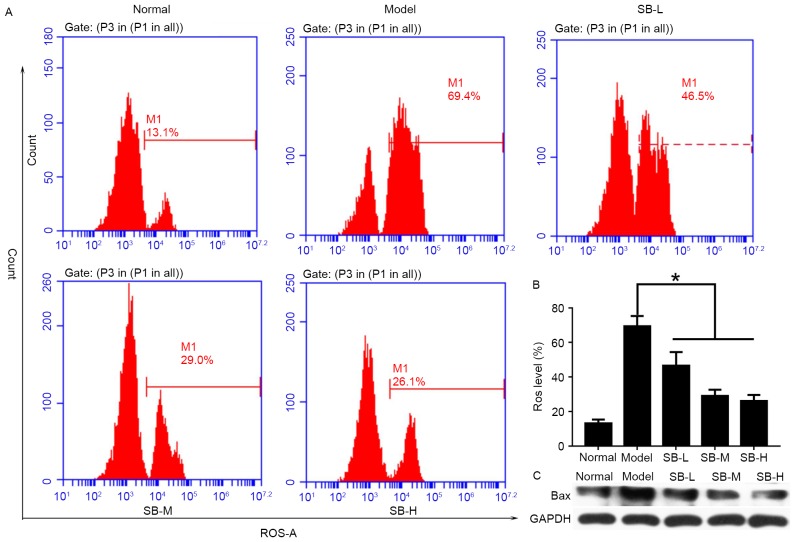
SB reduced ROS level and suppressed Bax expression
To explore the protective mechanism of SB in vivo, ROS levels in the liver were detected by flow cytometry (Fig. 6A). As presented in Fig. 6, mice in the model group exhibited the highest levels of ROS, while lower ROS levels was detected in SB-treated groups. In addition, this inhibitory effect exhibited a dose-dependent manner (Fig. 6B). Bax protein expression was suppressed in liver tissue after SB treatment (Fig. 6C). Therefore, it was suggested that SB could protect mouse livers through reducing ROS levels and inhibiting the relative apoptotic protein levels.
Figure 6.
SB reduced ROS level and inhibited Bax protein expression. (A) Representative images of flow cytometry measurement of ROS levels in mouse liver; (B) quantification of ROS levels in mouse liver from each group. (C) Bax expression in mouse liver measured by western blotting. *P<0.05 vs. the model group. SB, salvianolic acid B; ROS, reactive oxygen species.
Discussion
Ionizing radiation exerts adverse biological effects through direct and indirect processes: Breaks in DNA strands and production of oxygen free radicals. These oxygen free radicals in turn damage and mutate cellular DNA, leading to an increased rate of malignancy with radiation exposure. In addition, free radicals destroy supporting cellular structures, including organelles and cell membranes, resulting in programmed cell death (28). Furthermore, generation of free radicals result in an imbalance of the body's antioxidant system as they attack polyunsaturated fatty acids of the biofilm, ultimately leading to a series of damage to the hematopoietic, immune, nervous and endocrine systems (29). Due to the limitations and adverse effects of synthetic chemical anti-radiation drugs, there is a requirement for anti-radiation ingredients from natural sources, such as plants. Previous studies have indicated that polysaccharides, alkaloids, coumarins, flavonoids, saponins and other compounds possess anti-radiation effects (30–32). The mechanism of this effect may be attributed to protecting DNA, inhibiting immune injury, protecting the hematopoietic system and scavenging free radicals.
Radix Salviae miltiorrhizae has been widely used for thousands of years in traditional Chinese medicine with little reported toxicity (33). Salvianolic acid is the main water-soluble component in Radix Salviae miltiorrhizae. SB is the most abundant component of Salvianolic acid and is valued for both nutritional and medicinal purposes (34). SB exhibits higher scavenging activities than vitamin C against free hydroxyl radicals [HO(−)], superoxide anion radicals [O2(−)], and 1, 1-diphenyl-2-picryl-hydrazyl radicals and 2-azino-bis (3-ethylbenzthiazoline-6-sulfonic acid) radicals (35). As presented in Fig. 1, the compound SB contains multiple phenolic hydroxyl groups, which act as hydrogen donors to provide protons that combine with the oxygen free radicals generated by radiation. This eliminates excessive free radicals in the body and reduces the production of oxygen free radicals. In addition, it interrupts the free radical oxidation chain reaction, removes free radicals, prevents macromolecular damage and serves a role in radiation protection. Hemograms reflect the functional state of the body's hematopoietic system, which is particularly sensitive to radiation injury. Patients exposed to ionizing radiation often appear to show a sharp decline in the number of peripheral RBC, WBC and PLT (36). Therefore, protecting the hematopoietic system or improving the peripheral blood counts is an important biochemical indicator to evaluate the protective effect of novel drug candidates against radiation damage on the body (37). The present study indicated that radiated mice had a damaged hematopoietic system and the number of peripheral RBC, WBC and PLT decreased significantly. SB can significantly improve the peripheral blood counts of radiated mice, potentially by relieving the injury of ionizing radiation on hematopoietic system.
Bone marrow tissue is highly sensitive to radiation, and radiation-induced bone marrow damage leads to changes in bone marrow cells that are closely associated with the hematopoietic/stem cell microenvironment damage (38). Mitotically active cells are more susceptible to the detrimental effects of ionizing radiation, therefore, anatomic structures with high mitotic rates, such as red marrow, are more susceptible to the deleterious effects of radiation exposure (39). Ionizing radiation breaks DNA strands, and the number of nucleated cells and protein synthesis in bone marrow cells is affected, however in addition, radiation causes direct damage to the protein, including ROS-induced protein chemical alterations such as carbonylation and nitrosylation (39). The present study demonstrated that SB protects protein damage caused by ionizing radiation.
The spleen and the thymus are important immune organs in mammals, in which immune cells and several immune factors are produced. They constitute the body's defense system and serve an important role in providing resistance to a variety of germs or physical and chemical injuries. Secondly, due to the fact that they are particularly sensitive to radiation damage, patients often exhibit a decline in immunity after receiving radiation therapy, a major side effect of radiation therapy (40). The present study identified that the thymus and spleen indices of ionizing irradiated mice were markedly reduced. Administration of SB alleviates the declining trend caused by ionizing radiation, implying that SB may confer certain protective effects on radiation-damaged organs.
A significant proportion of the damage from ionizing radiation primarily results from the production of ROS that oxidize DNA, proteins, lipids and cofactors (41–43). When normal tissue is exposed to ionizing radiation, the atoms of water molecules or other oxygen-containing molecules are attacked by the photon. This results in the excitation and emission of an electrons from that atom (44). In addition, the ROS generated attack polyunsaturated fatty acids of biofilm phospholipids and causes lipid peroxidation (45). MDA is a lipid peroxidation product of biofilm phospholipid molecules, and its levels reflect the degree of biofilm damaged by ROS (46). SOD is the key enzyme that removes ROS intracellularly, and is widely distributed in various tissues and organs of the body, SOD activity decreases due to aging, ionizing radiation and environmental pollution, which results in the destruction of the body's oxygen metabolism homeostasis (47). Nrf2-mediated antioxidant induction is a cellular adaptive response to oxidative stress challenge and it can activate a series of antioxidant enzymes, including SOD and heme oxygenase and therefore serves a central role in the protection of cells against oxidative damage (19). The current study demonstrated that ionizing radiation led to decreased SOD activity and increased MDA levels in serum. In addition, it suppressed the Nrf2 pathway by reducing Nrf2 expression levels and elevating Bach1 expression levels. However, SB supplementation not only restores decreased SOD activity and increased MDA in radiation injured mice but also increased Nrf2 expression level and reduced Bach1 expression levels. In addition, SB reduced ROS level and suppressed Bax expression in mouse liver. These observations suggest that SB acts as an anti-oxidant by activating Nrf2 pathway in the serum of radiation-exposed mice, which may be associated with reducing oxidative stress and inhibiting apoptosis. The results are in agreement with a previous study, which observed that SB attenuated toxin-induced neuronal damage via Nrf2 (48). However, further investigation using Nrf2 knockout/knockdown mice or specific inhibition of Nrf2 expression is required to confirm the involvement of the Nrf2 pathway in the protective role of SB against radiation damage.
Taken together, it was identified that supplementation of SB to mice exposed to radiation suppressed the MDA levels induced by radiation, increased peripheral RBC, WBC and PLT levels, thymus and spleen indices, and activated the Nrf2-mediated antioxidant pathway. In conclusion, the results, at least in part, indicated that SB has a protective effect against radiation damage and may have valid antioxidant activity in enhancing immunity and the function of the hematopoietic system. Therefore, it is inferred that SB could serve as a promising candidate for adjuvant therapy to alleviate radiation-induced injuries in humans affected by ionizing radiation. The mechanisms underlying the anti-radiation effect of SB require further investigation.
Acknowledgements
Not applicable.
Funding
The present study was supported by grants from Guangdong Province Construction of Traditional Chinese Medicine Strong Province Project (grant no. 20132103), Guangdong Province Universities and Colleges Pearl River Scholar Funded Scheme (grant no. 2011), Science & Technology Planning Project of Guangdong Province Office of Education (grant no. 2014GKXM032) and Science & Technology Planning Project of Nansha District (grant no. 2016CX003).
Availability of data and materials
All data generated or analyzed during this study are included in this published article.
Authors' contributions
RZ and GL designed the experiments. RZ, HL and BZ performed the majority of the experiments. ZL, QZ, TW, YZ, QW and XL assisted with the experiments. RZ, LZL and HL collected the data and completed the data analysis. RZ and LZL drafted the manuscript, and GL and LZL revised the drafts. All authors read and approved the final manuscript.
Ethics approval and consent to participate
The animal studies were approved by the Animal Ethics Committee of Guangzhou University of Chinese Medicine.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Pacheco R, Stock H. Effects of Radiation on Bone. Curr Osteoporos Rep. 2013;11:299–304. doi: 10.1007/s11914-013-0174-z. [DOI] [PubMed] [Google Scholar]
- 2.Giusti AM, Raimondi M, Ravagnan G, Sapora O, Parasassi T. Human cell membrane oxidative damage induced by single and fractionated doses of ionizing radiation: A fluorescence spectroscopic study. Int J Radiat Biol. 1998;74:595–605. doi: 10.1080/095530098141177. [DOI] [PubMed] [Google Scholar]
- 3.Zhao W, Diz DI, Robbins ME. Oxidative damage pathways in relation to normal tissue injury. Br J Radiol. 2007;80:S23–S31. doi: 10.1259/bjr/18237646. [DOI] [PubMed] [Google Scholar]
- 4.Rice-Evans C, Burdon R. Free radical-lipid interactions and their pathological consequences. Prog Lipid Res. 1993;32:71–110. doi: 10.1016/0163-7827(93)90006-I. [DOI] [PubMed] [Google Scholar]
- 5.Esterbauer H. Estimation of peroxidative damage. A critical review. Pathol Biol (Paris) 1996;44:25–28. [PubMed] [Google Scholar]
- 6.Packer L, Ong ASH. AOCS Press; Champaign, IL: 1998. Biological Oxidants and Antioxidants. Molecular Mechanisms and Health Effects. [Google Scholar]
- 7.Moulder JE. Radiobiology of nuclear terrorism: Report on an interagency workshop (Bethesda, MD, December 17–18, 2001) Int J Radiat Oncol Biol Phys. 2002;54:327–328. doi: 10.1016/S0360-3016(02)02956-5. [DOI] [PubMed] [Google Scholar]
- 8.Arora R, Gupta D, Chawla R, Sagar R, Sharma A, Kumar R, Prasad J, Singh S, Samanta N, Sharma RK. Radioprotection by plant products: Present status and future prospects. Phytother Res. 2005;19:1–22. doi: 10.1002/ptr.1605. [DOI] [PubMed] [Google Scholar]
- 9.Jin LH, Liu CF, Zeng Y. Protective effects of puerarin on radiation injury of experimental rats. Zhong Xi Yi Jie He Xue Bao. 2005;3:43–45. doi: 10.3736/jcim20050113. (In Chinese) [DOI] [PubMed] [Google Scholar]
- 10.Guo S, Hu Y, Liu P, Wang Y, Guo D, Wang D, Liao H. Protective activity of different concentration of tea polyphenols and its major compound EGCG against whole body irradiation-induced injury in mice. Zhongguo Zhong Yao Za Zhi. 2010;35:1328–1332. doi: 10.4268/cjcmm20101024. (In Chinese) [DOI] [PubMed] [Google Scholar]
- 11.Sharma R, Tiku AB. Emodin, an anthraquinone derivative, protects against gamma radiation-induced toxicity by inhibiting DNA damage and oxidative stress. Int J Radiat Biol. 2014;90:275–283. doi: 10.3109/09553002.2014.884292. [DOI] [PubMed] [Google Scholar]
- 12.Sun XC, Li WB, Li QJ, Li SQ, Zhang M, Xian XH. Spantide inhibits up-regulation of NOS in the pericentral canal region of the spinal cord in the rat formalin test. Chin J Pathophysiol. 2005;21:24222426. [Google Scholar]
- 13.Özyurt H, Çevik Ö, Özgen Z, Özden AS, Çadırcı S, Elmas MA, Ercan F, Gören MZ, Şener G. Quercetin protects radiation-induced DNA damage and apoptosis in kidney and bladder tissues of rats. Free Radic Res. 2014;48:1247–1255. doi: 10.3109/10715762.2014.945925. [DOI] [PubMed] [Google Scholar]
- 14.Su CY, Ming QL, Rahman K, Han T, Qin LP. Salvia miltiorrhiza: Traditional medicinal uses, chemistry and pharmacology. Chin J Nat Med. 2015;13:163–182. doi: 10.1016/S1875-5364(15)30002-9. [DOI] [PubMed] [Google Scholar]
- 15.Ji XY, Tan BK, Zhu YZ. Salvia miltiorrhiza and ischemic diseases. Acta Pharmacol Sin. 2000;21:1089–1094. [PubMed] [Google Scholar]
- 16.Chan K, Chui SH, Wong DY, Ha WY, Chan CL, Wong RN. Protective effects of Danshensu from the aqueous extract of Salvia miltiorrhiza (Danshen) against homocysteine-induced endothelial dysfunction. Life Sci. 2004;75:3157–3171. doi: 10.1016/j.lfs.2004.06.010. [DOI] [PubMed] [Google Scholar]
- 17.Liu HB, Xu J, Peng Y. Targets of Danshen's active components for activating blood circulation activities. Acta Phys-Chim Sin. 2010;26:199–205. [Google Scholar]
- 18.Zhang N, Zou H, Jin L, Wang J, Zhong MF, Huang P, Gu BQ, Mao SL, Zhang C, Chen H. Biphasic effects of sodium danshensu on vessel function in isolated rat aorta. Acta Pharmacol Sin. 2010;31:421–428. doi: 10.1038/aps.2010.24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ji W, Gong B. Hypolipidemic activity and mechanism of purified herbal extract of Salvia miltiorrhiza in hyperlipidemic rats. J Ethnopharmacol. 2008;119:291–298. doi: 10.1016/j.jep.2008.07.013. [DOI] [PubMed] [Google Scholar]
- 20.Paik YH, Yoon YJ, Lee HC, Jung MK, Kang SH, Chung SI, Kim JK, Cho JY, Lee KS, Han KH. Antifibrotic effects of magnesium lithospermate B on hepatic stellate cells and thioacetamide-induced cirrhotic rats. Exp Mol Med. 2011;43:341–349. doi: 10.3858/emm.2011.43.6.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Ji XY, Tan BK, Zhu YZ. Salvia miltiorrhiza and ischemic diseases. Acta Pharmacol Sin. 2000;21:1089–1094. [PubMed] [Google Scholar]
- 22.Zhao GF, Zhang HX, Fan YC. The protecting effect of Salvianolic acid B on rats with myocardial ischemia reperfusion injury. J Liaoning Coll Trad Chin Med. 2004;1:55–56. [Google Scholar]
- 23.Fu J, Fan HB, Guo Z, Wang Z, Li XD, Li J, Pei GX. Salvianolic acid B attenuates spinal cord ischemia-reperfusion-induced neuronal injury and oxidative stress by activating the extracellular signal-regulated kinase pathway in rats. J Surg Res. 2014;188:222–230. doi: 10.1016/j.jss.2013.11.1118. [DOI] [PubMed] [Google Scholar]
- 24.Tian LL, Wang XJ, Sun YN, Li CR, Xing YL, Zhao HB, Duan M, Zhou Z, Wang SQ. Salvianolic acid B, an antioxidant from Salvia miltiorrhiza, prevents 6-hydroxydopamine induced apoptosis in SH-SY5Y cells. Int J Biochem Cell Biol. 2008;40:409–422. doi: 10.1016/j.biocel.2007.08.005. [DOI] [PubMed] [Google Scholar]
- 25.Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods. 2001;25:402–408. doi: 10.1006/meth.2001.1262. [DOI] [PubMed] [Google Scholar]
- 26.Lu J, Zhang F, Zhao D, Hong L, Min J, Zhang L, Li F, Yan Y, Li H, Ma Y, Li Q. ATRA-inhibited proliferation in glioma cells is associated with subcellular redistribution of β-catenin via up-regulation of Axin. J Neurooncol. 2008;87:271–277. doi: 10.1007/s11060-008-9518-4. [DOI] [PubMed] [Google Scholar]
- 27.Yamamori T, Yasui H, Yamazumi M, Wada Y, Nakamura Y, Nakamura H, Inanami O. Ionizing radiation induces mitochondrial reactive oxygen species production accompanied by upregulation of mitochondrial electron transport chain function and mitochondrial content under control of the cell cycle checkpoint. Free Radic Biol Med. 2012;53:260–270. doi: 10.1016/j.freeradbiomed.2012.04.033. [DOI] [PubMed] [Google Scholar]
- 28.Robbins ME, Zhao W. Chronic oxidative stress and radiation-induced late normal tissue injury: A review. Int J Radiat Biol. 2004;80:251–259. doi: 10.1080/09553000410001692726. [DOI] [PubMed] [Google Scholar]
- 29.Epperly M, Bray J, Kraeger S, Zwacka R, Engelhardt J, Travis E, Greenberger J. Prevention of late effects of irradiation lung damage by manganese superoxide dismutase gene therapy. Gene Ther. 1998;5:196–208. doi: 10.1038/sj.gt.3300580. [DOI] [PubMed] [Google Scholar]
- 30.Shi J, Cheng C, Zhao H, Jing J, Gong N, Lu W. In vivo anti-radiation activities of the Ulva pertusa polysaccharides and polysaccharide-iron(III) complex. Int J Biol Macromol. 2013;60:341–346. doi: 10.1016/j.ijbiomac.2013.06.001. [DOI] [PubMed] [Google Scholar]
- 31.Ji R, Zhong K, Li Y, Li Y. Effects of tea polyphenols on mice survival rate and white blood cell count after radiation. Wei Sheng Yan Jiu. 2002;31:394–395. (In Chinese) [PubMed] [Google Scholar]
- 32.Zhang XD, Ma C, Sun X. Advances in mechanism of natural anti radiation drug. Chin J Radiol Health. 2004;13:228–230. [Google Scholar]
- 33.Ho JH, Hong CY. Salvianolic acids: Small compounds with multiple mechanisms for cardiovascular protection. J Biomed Sci. 2011;18:30. doi: 10.1186/1423-0127-18-30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Wang ZS, Luo P, Dai SH, Liu ZB, Zheng XR, Chen T. Salvianolic acid B induces apoptosis in human glioma U87 cells through p38-mediated ROS generation. Cell Mol Neurobiol. 2013;33:921–928. doi: 10.1007/s10571-013-9958-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Zhao GR, Zhang HM, Ye TX, Xiang ZJ, Yuan YJ, Guo ZX, Zhao LB. Characterization of the radical scavenging and antioxidant activities of danshensu and salvianolic acid B. Food Chem Toxicol. 2008;46:73–81. doi: 10.1016/j.fct.2007.06.034. [DOI] [PubMed] [Google Scholar]
- 36.Geoffrey P, Jacobs a. A review on the effects of ionizing radiation on blood and blood components. Radiat Phys Chem. 1998;53:511–523. doi: 10.1016/S0969-806X(98)00185-6. [DOI] [Google Scholar]
- 37.Edwards JC, Chapman D, Cramp WA, Yatvin MB. The effects of ionizing radiation on biomembrane structure and function. Prog Biophys Mol Biol. 1984;43:71–93. doi: 10.1016/0079-6107(84)90004-X. [DOI] [PubMed] [Google Scholar]
- 38.Zheng K, Wu W, Yang S, Huang L, Chen J, Gong C, Fu Z, Lin R, Tan J. Treatment of radiation-induced acute intestinal injury with bone marrow-derived mesenchymal stem cells. Exp Ther Med. 2016;11:2425–2431. doi: 10.3892/etm.2016.3248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Kokošová N, Tomášová L, Kisková T, Šmajda B. Neuronal analysis and behaviour in prenatally gamma-irradiated rats. Cell Mol Neurobiol. 2015;35:45–55. doi: 10.1007/s10571-014-0144-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Szumiel I. Ionizing radiation-induced cell death. Int J Radiat Biol. 1994;66:329–341. doi: 10.1080/09553009414551271. [DOI] [PubMed] [Google Scholar]
- 41.Kitamura T, Suzuki M, Nishimatsu H, Kurosaki T, Enomoto Y, Fukuhara H, Kume H, Takeuchi T, Miao L, Jiangang H, Xiaoqiang L. Final report on low-dose estramustine phosphate (EMP) monotherapy and very low-dose EMP therapy combined with LH-RH agonist for previously untreated advanced prostate cancer. Aktuelle Urol. 2010;41(Suppl 1):S34–S40. doi: 10.1055/s-0029-1224657. [DOI] [PubMed] [Google Scholar]
- 42.Wong CM, Marcocci L, Liu L, Suzuki YJ. Cell signaling by protein carbonylation and decarbonylation. Antioxid Redox Signal. 2010;12:393–404. doi: 10.1089/ars.2009.2805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Breen AP, Murphy JA. Reactions of oxyl radicals with DNA. Free Radic Biol Med. 1995;18:1033–1077. doi: 10.1016/0891-5849(94)00209-3. [DOI] [PubMed] [Google Scholar]
- 44.Ilya Obodovskiy: Effect of ionizing radiation on biological structures. Fundam Radiat Chem Saf. 2015:87–131. [Google Scholar]
- 45.Pamplona R. Membrane phospholipids, lipoxidative damage and molecular integrity: A causal role in aging and longevity. Biochim Biophys Acta. 2008;1777:1249–1262. doi: 10.1016/j.bbabio.2008.07.003. [DOI] [PubMed] [Google Scholar]
- 46.Kwiecien S, Jasnos K, Magierowski M, Sliwowski Z, Pajdo R, Brzozowski B, Mach T, Wojcik D, Brzozowski T. Lipid peroxidation, reactive oxygen species and antioxidative factors in the pathogenesis of gastric mucosal lesions and mechanism of protection against oxidative stress-induced gastric injury. J Physiol Pharmacol. 2014;65:613–622. [PubMed] [Google Scholar]
- 47.Holley AK, Miao L, St Clair DK, St Clair WH. Redox-modulated phenomena and radiation therapy: The central role of superoxide dismutases. Antioxid Redox Signal. 2014;20:1567–1589. doi: 10.1089/ars.2012.5000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Zhou J, Qu XD, Li ZY, Wei-Ji, Liu Q, Ma YH, He JJ. Salvianolic acid B attenuates toxin-induced neuronal damage via Nrf2-dependent glial cells-mediated protective activity in Parkinson's disease models. PLoS One. 2014;9:e101668. doi: 10.1371/journal.pone.0101668. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All data generated or analyzed during this study are included in this published article.