Abstract
The MarketScan databases are a family of administrative claims databases that contain data on inpatient and outpatient claims, outpatient prescription claims, clinical utilization records, and healthcare expenditures. The three main databases available for use are each composed of a convenience sample for one of the following patient populations: (1) patients with employer-based health insurance from contributing employers, (2) Medicare beneficiaries who possess supplemental insurance paid by their employers, and (3) patients with Medicaid in one of eleven participating states. Eleven supplemental databases are available, which are utilized to overcome the limited clinical data available in the core MarketScan databases. There are several limitations to this database, primarily related to the fact that individuals or their family members within two of the core databases mandatorily possess some form of employer-based health insurance, which prevents the dataset from being nationally representative. Nonetheless, this database provides detailed and rigorously maintained claims data to identify healthcare utilization patterns among this cohort of patients.
Keywords: Truven MarketScan, health insurance, colorectal surgery
Overview
The MarketScan databases, which are compiled and maintained by Truven Health Analytics, are a family of administrative claims-based databases that contain data on almost 230 million unique patients in the United States to date. 1 Studies using information captured by these databases have resulted in over 900 peer-reviewed publications since its launch in 1995, and its use in published studies has drastically increased in recent years ( Fig. 1 ). 2 With over 20 years of data, MarketScan databases offer the potential to analyze national trends in healthcare utilization, medication usage, and even the natural history of diseases. 3 Trend analyses are also made possible by the high longitudinal integrity and patient retention contained within MarketScan, which is a major strength offered by these datasets. The current MarketScan family of databases is composed of three main databases and eleven supplemental databases ( Table 1 ), thirteen of which are claims based and one of which is hospital discharge based. 2 After selecting the appropriate database to answer a particular clinical question, the MarketScan system provides a comprehensive avenue for exploring the strengths and weaknesses of the current U.S. healthcare system. In this review, we will present the structure, strengths, and weaknesses of the MarketScan family of databases, followed by a brief review of studies that have used these databases to address research specific to the field of colon and rectal surgery.
Fig. 1.
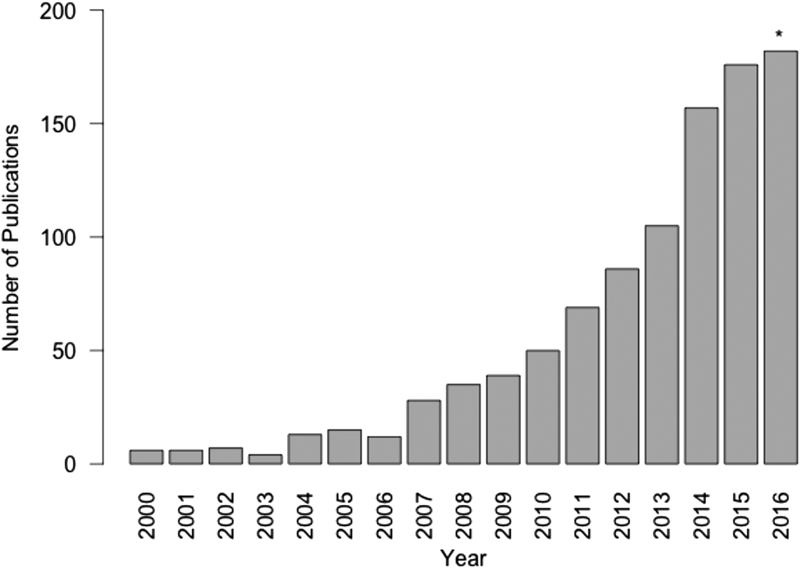
Number of articles per year in PubMed identified using the search term “MarketScan,” from 2000 to 2015. *The year 2016 only includes January through November estimates.
Table 1. Overview of available databases within the MarketScan family 2 .
| MarketScan family of databases | |
|---|---|
| Core databases | Supplemental databases |
| • Commercial Claims and Encounters (CCAE) ○ Includes inpatient and outpatient medical, procedural, and outpatient pharmaceutical claims for millions of individuals who possess employer-sponsored health insurance, as well as their spouses and dependents • Medicare Supplemental ○ Contains similar data to the CCAE database, but on retirees who possess employer-sponsored Medicare supplemental insurance, including both claims from the Medicare-paid portion and the employer-covered portion • Medicaid Multi-State ○ Includes similar data to the CCAE database on individuals enrolled in Medicaid in 11 states, contains additional demographic data on race and disability status |
• Health and productivity management ○ Integrates data on workplace absence, workers' compensation, and short- and long-term disability to ascertain direct and indirect costs of disease states • Benefit plan design database ○ Includes data on home healthcare, physical therapy, hospice, and skilled nursing facility utilization • Laboratory database ○ Contains inpatient and outpatient laboratory test results since 2005 • Health risk assessment database ○ Provides data on health behaviors (e.g., smoking status, alcohol use, body mass index, exercise) commonly associated with disease • Dental database ○ Links dental and medical claims to allow correlation between various medical and oral disease states • Hospital drug database ○ Provides more detailed data on hospital drug use, including length of therapy and route of administration • Inpatient drug link file ○ Provides link between main MarketScan data and inpatient pharmaceutical claims • EMR-claims linked data ○ Expands clinical data available through MarketScan by patient-level deterministic and probabilistic linking to available EMR data • Mortality file ○ Includes mortality data on subset of patients for whom mortality was identified through linkage with SSA records • Oncology EMR database ○ Includes data on histology, tumor type/stage, and cancer treatments • Oncology EMR-claims linked dataset ○ Expands data available through MarketScan by patient-level linking to available clinical oncology data |
Abbreviations: EMR, electronic medical record; SSA, Social Security Administration.
Commercial Claims and Encounters Database
The three main databases that comprise MarketScan include data on patients utilizing one of the two largest sources of health insurance in the United States: commercial employer-based insurance and government-based insurance. 4 Data found in the Commercial Claims and Encounters (CCAE) database contains de-identified individual-specific details for those covered by 1 of over 300 contributing employers that sponsor private health insurance, as well as data on both the spouses and the dependents of the employees. 2 Data included within this database are obtained from inpatient and outpatient medical claims, outpatient prescription claims, clinical utilization records, and healthcare expenditures. This comprehensive collection of data allows for analysis of diagnoses (using International Classification of Disease, 9th edition, Clinical Modifications [ICD-9-CM] codes) and procedures (using Current Procedural Terminology [CPT] codes) for which claims are submitted, as well as the resultant costs, reimbursements, and out-of-pocket expenses borne by patients, provider systems, and payers. 3 5 6 Certain patient demographic and hospital characteristics, however, such as race, income, and hospital or physician volume, are not included in the database. Nonetheless, for studies that focus on health states of working-age individuals or their dependents, this database provides high-quality claims data to identify healthcare utilization patterns among this cohort of patients.
Medicare Supplemental and Medicaid Multi-State Databases
To evaluate trends in health and healthcare utilization among older Americans or those without employer-sponsored health insurance, other databases that are available through MarketScan include the Medicare Supplemental (MDCR) database and the Medicaid Multi-State (MMS) database. The scope and depth of health-related data contained in these databases is equivalent to that available within the CCAE database, although, as expected (since Medicare and Medicaid are used by a smaller proportion of the U.S. population), the patients represent a smaller proportion of the population covered under these plans. The MDCR captures information on the subset of Medicare beneficiaries who possess supplemental insurance paid by their employers, which in 2010 represented approximately 14% of the 46 million retirees with Medicare benefits. 2 Although it does not include data for all Medicare beneficiaries, for those who are included within the database, MarketScan contains data on both the employer-paid portion and the Medicare-paid portion of the payments, as well as any out-of-pocket expenses, thus capturing a fairly complete account of healthcare coverage patterns in this population. 2 The MMS database contains health-related data for the Medicaid populations of 11 states with a wide geographic dispersal, offering data on over 36 million individuals in the United States. 7 Although the Medicaid database has a more limited population, it has additional utility for clinical research due to the availability of additional demographic details—such as race and disability status—that enable researchers to address issues in health disparities and special populations. 2 Although not representative of all beneficiaries in the Medicare and Medicaid populations, these two MarketScan databases provide the ability to obtain granular healthcare utilization data regarding two at-risk populations that may be of particular interest to health services researchers.
MarketScan Supplemental Databases
The 11 supplemental databases within the MarketScan family are listed in Table 1 . These databases are designed to augment the information garnered from the core databases described earlier by providing more clinically oriented data, in areas that may be of specific interest to economists, health services researchers, and market analysts. Several of these supplementary databases may be of interest to clinical health services researchers, particularly in the fields of gastroenterology and colorectal surgery. The MarketScan Health Risk Assessment database contains patient self-reported data on behaviors (e.g., smoking, dietary habits) that are otherwise absent in the core databases, and may be of particular use to researchers interested in preoperative risk stratification of patients undergoing elective procedures. 8 The MarketScan Benefit Plan Design database offers important advantages that are typically inaccessible to large national databases, containing data on home healthcare utilization, physical therapy services, and extended care or skilled nursing facility information. 2 These data allow health services researchers to evaluate the longitudinal economic impact of services rendered on inpatient or outpatient bases, rather than a more limited evaluation of the costs of the index encounters alone. The MarketScan Laboratory database allows for the integration of laboratory testing results with medical claims, to provide information about any diagnostic testing performed before and after courses of drug therapy. The MarketScan Oncology Electronic Medical Records (EMR) database captures data relevant to oncologic studies (such as tumor type and stage, histology, cancer treatments, and outcomes) and represents data from over 170 contributing oncology practices in the United States. Moreover, it includes all age groups, with over half of represented individuals over the age of 65 years. 2 Given the large proportion of patients with colorectal malignancy who present over the age of 65 years, this supplementary database is of particular utility when studying this population. 9 Lastly, the MarketScan Mortality File is available and includes data on the subset of individuals within the CCAE and MDCR databases for whom a death could be identified within the Social Security Administration database. 3 While this mortality data are admittedly incomplete, it can potentially offer the added dimension of studying the economic implications surrounding end-of-life care in patients with colorectal diseases. 2 These supplemental databases can be linked to one another to track the clinical utilization of patients throughout a broad range of healthcare encounters.
Strengths
The primary benefit of MarketScan is that it offers a detailed overview of “real-world” healthcare utilization and outcomes for large subset of the U.S. population. While continuous enrollment in an insurance plan is necessary to link patient information across encounters, loss of data due to a change in health plans is attenuated by using employer-provided data to track patients across plans, since people change insurance plans more frequently than they change employers. Additional advantages of this particular family of databases, which are also outlined in Fig. 2 , include the full integration of inpatient and outpatient care, emergency care services, along with outpatient pharmaceutical data, on a per-enrollee level. 3 With this extensive tracking scheme, MarketScan follows patient encounters from a prehospitalization setting, through any inpatient care that was administered, and then through any posthospitalization clinic visits or outpatient pharmaceutical claims. 6 10 In studying patients with colorectal diseases, for whom care pathways often traverse the full spectrum of healthcare settings, accessing complete data for all of the settings is essential for building a complete picture of a particular disease process. Additionally, rather than relying upon prescription practices as a proxy for medication usage, MarketScan identifies prescriptions that were actually filled by the patients, and provides a breakdown of reimbursement and out-of-pocket expenses. 5 Using the Health Plan Benefit Design supplemental database, researchers can also track claims for visiting nurses, rehabilitation facilities, and other aspects of care that are essential for evaluating the full economic impact of diseases. Furthermore, the data present in these databases are based only on claims that have been fully paid or adjudicated, thereby increasing the accuracy of claims data. Finally, despite the extensive linkage and continuity of patient-level data present within the databases, these databases maintain HIPAA (Health Insurance Portability and Accountability Act) compliance through rigorous measures to ensure protection of patient privacy, and all data are de-identified to users who obtain access to the database.
Fig. 2.

Overview of strengths and limitations of the MarketScan family of databases.
Limitations
Despite many strengths, the MarketScan databases do have several limitations that are important to consider when designing potential studies. Perhaps their most considerable limitation is the fact that the CCAE database represents working-age patients covered by private insurance that is supplied by employers who contribute to the database, rather than a random sample from the U.S. population, and therefore are not representative of the U.S. population. With the exception of the MMS database, individuals or their family members within the CCAE and the MDCR mandatorily possess some form of employer-based health insurance, which makes the dataset not nationally representative. Moreover, the employers that contribute to MarketScan data represent large employers, with weaker representation from small or medium size firms. As a result of these weaknesses, these data cannot be weighted to reflect a national sample, nor are results obtained from this database always generalizable to the U.S. population as a whole.
Other limitations derive from the fact that, similar to other claims-based databases, the validity of any claims-based database is reliant upon the accuracy of the medical coding and billing data that it contains. Additionally, the scope of encounters captured by MarketScan is limited to only those for which a claim was submitted; thus, certain comorbidities, medications, or procedures may not be identified. For example, data on over-the-counter medication purchases is not contained in the MarketScan database. Furthermore, while data on prescriptions fills are available, this does not necessarily translate into patient adherence with the prescribed dosing schedules. A study by Lauffenburger et al identified an approximately 10% discrepancy in capturing warfarin utilization by using claims data from this database, potentially due to some patients having prescriptions filled outside of the context of their standard prescription benefit. 11 Lastly, within the core databases, many important demographic and outcomes details—such as race, smoking status, and mortality—are not available without the purchase of supplemental databases, thereby limiting the scope of studies that can be performed with the core datasets.
Obtaining Access to the MarketScan Databases
Currently, there are several ways in which researchers can access the MarketScan databases for clinical research projects. The decision for which option to pursue depends primarily on the funding sources and the specific clinical questions posed by the researcher. Currently, academic institutions may qualify for an academic license from Truven Health Analytics, which typically includes access to both the CCAE and the MDCR databases (containing data on 40–50 million patients per available year). This yearly license may then be utilized by multiple faculty members at a single institution. For researchers who would prefer to conduct a single specific study, external funding can be used to obtain a single study license through Truven Health Analytics to answer a particular research question.
Data that are available within the 11 supplemental databases (e.g., Health Risk Assessment Database, Laboratory Database, etc.) include additional clinical information on various subsets of patients within the core databases. However, for researchers who want to limit their analysis to a particular subset of patients with complete data (i.e., only patients who have data present in the Laboratory Database), Truven offers smaller licenses that give researchers access to the specialized dataset of interest, as well as the patients' corresponding medical and pharmaceutical claims data that are present within the core databases, without having to obtain access to the entire population within the core databases.
After a license is obtained, the data are provided in SAS-formatted data files, which researchers can then access and use to analyze the licensed data. Further information can be obtained from https://marketscan.truvenhealth.com/marketscanportal/ , and specific inquiries regarding the use of Truven MarketScan databases may be directed via email to marketscan@truvenhealth.com.
Colorectal and Gastrointestinal Clinical Research Using MarketScan
Within the fields of colorectal surgery and gastroenterology, the MarketScan databases have been utilized to address a variety of epidemiologic and health services research questions. Below are several illustrative examples of these studies, from a variety of institutions, which utilize these data to answer a wide scope of research queries.
Epidemiology
Given the detailed pharmaceutical data available in MarketScan databases, epidemiologic studies focusing on medication exposures and potential outcomes can be assessed. A good example of this was a case–control study performed by Sehdev et al at the University of Chicago: from 2005 to 2010, individuals with a prior diagnosis of diabetes mellitus (DM) and a new claim for colorectal cancer (CRC) within a 12-month period were identified within the MarketScan CCAE database. 12 These were matched 2:1 with controls, who had a diagnosis of DM but no diagnosis of CRC, by age, sex, and geographical region. The exposure of interest in this study was metformin use within 12 months prior to the diagnosis of CRC (suggesting long-term or chronic use), and the authors hypothesized that prior metformin use would be associated with a reduction in CRC development. Using conditional logistic regression and controlling for available comorbidities and concurrent medication use, the authors identified a significant reduction in the odds (odds ratio [OR]: 0.85, 95% confidence interval [CI]: 0.76–0.95, p < 0.007) of developing CRC among those with prior metformin use. However, when the authors subsequently controlled for healthcare utilization (including outpatient visits and number of hospitalizations), the reduction in odds of developing CRC was no longer significantly. Given that CRS is relatively slow growing neoplasm over the course of years, and the exposure to metformin was assessed for only 1 year, the lack of association may be secondary to study design, or there may be a true lack of association. The authors' ability to control for healthcare utilization in this study represents one of the unique adjuncts in this database that can be taken advantage of within epidemiologic research.
Time Trends
When researching a particular type of diagnostic or therapeutic encounter within the healthcare field, the MarketScan databases allow for the identification of trends in utilization over time. Peery and colleagues used the three main MarketScan databases (Commercial, Medicare, and Medicaid) to identify the incidence and costs of three types of gastrointestinal endoscopic procedures in 2009: upper endoscopy (including esophagogastroduodenoscopy, endoscopic ultrasonography, and enteroscopy), lower endoscopy (including colonoscopy, flexible sigmoidoscopy, endorectal ultrasound, and pouchoscopy), and endoscopic retrograde cholangiopancreatography (ERCP). 13 The incidence of these procedures was standardized to the age and gender distribution of the entire U.S. population, and found to be 6.9 million for upper endoscopies, 11.5 million for lower endoscopies, and 228,000 for biliary endoscopies. Estimates for total outpatient costs were $12.3 billion, $19.2 billion, and $900 million, respectively. Then, using the MarketScan CCAE database, trends in time over a 10-year period (2000–2009) for each of the three types of endoscopic procedures were identified (shown in Fig. 3 ). Among commercially insured patients, the authors identified a significant increase for both upper and lower endoscopy over that period of time (54 and 17%, respectively), but an overall decrease (by 16%) in the use of ERCP.
Fig. 3.
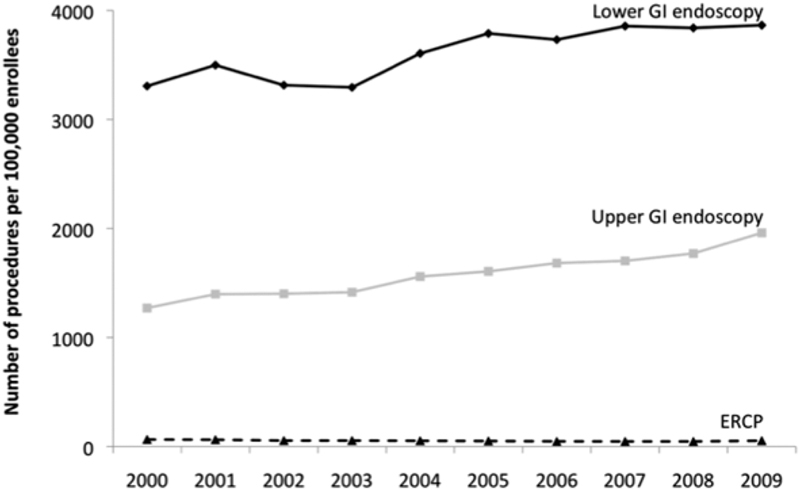
Time trends in endoscopy procedure rates in commercially insured patients in the United States, 2000–2009. (Reprinted from Peery et al, 13 © Peery 2012, with permission from Elsevier.)
Rare Diseases/Occurrences
With the large numbers of patients present within the databases, the MarketScan databases are able to capture relatively large numbers of diagnoses and outcomes that are otherwise infrequent or rare. Foley et al utilized the MarketScan CCAE database and the MarketScan Medicare Supplemental and Coordination of Benefits Database to identify cases of infusion reactions (IR) associated with the use of cetuximab in patients with CRC. 14 Risk factors for severe IRs that require treatment are poorly understandable, and studying this event requires a large cohort of these infrequent occurrences. 15 Using these databases, the authors were able to identify 1,122 patients with colorectal cancer who received cetuximab. Within this cohort, IRs were identified within MarketScan by using a combination of outpatient claims, inpatient claims, and medication-related data, and were found in 94 of the 1,122 patients (8.4%) comprising the study cohort. Logistic regression was used to model the occurrence of an IR as a function of covariates. Both residence in a pollen state (OR: 2.67, 95% CI: 1.27–5.62) and initial cetuximab administration (OR: 1.84, 95% CI: 1.26–2.71) were significantly associated with the occurrence of IRs, whereas increasing age was protective (OR: 0.96, 95% CI: 0.94–0.98). Using cost data, the authors were also able to identify the additional costs associated with management of IRs, which were found to be $1,725 for IRs requiring outpatient treatment and $9,308 for severe IRs requiring either emergency department visits or inpatient hospitalizations (total adjusted costs shown in Fig. 4 ).
Fig. 4.
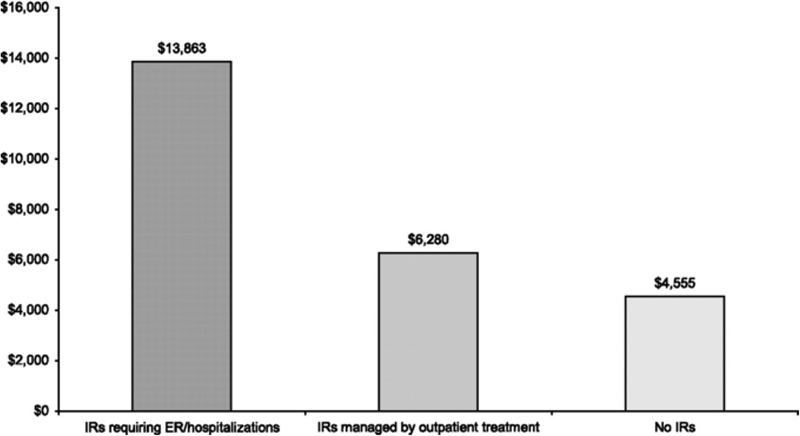
Adjusted costs of cetuximab administration by IR status. ER, emergency room; IR, infusion reaction. (Reproduced with permission from Foley et al. 14 © Foley 2010, Published by Oxford University Press on behalf of the European Society for Medical Oncology.
Costs and Healthcare Utilization
The longitudinal health care data present within the MarketScan Databases allow cumulative health care resource utilization and expenditures (i.e., direct costs) to be determined over years or even decades, rather than in single encounters. Crawshaw and colleagues 16 from the University Hospitals Case Medical Center used the MarketScan CCAE database to identify elective laparoscopic and open colectomies for benign disease performed in 2011. Health care utilization (outpatient, emergency, and inpatient services) and expenditures within 1 year postoperatively were totaled and compared between the two approaches. The authors determined that patients undergoing laparoscopic approaches had significantly lower health care utilization and cumulative costs, both at 90 days postoperatively and 1 year postoperatively, than those undergoing open approaches. The health care cost differences between the two groups were estimated to be approximately 1.26 times higher among patients undergoing open approaches at 90 days (estimated $1715, 95% CI: $338–$2853), and approximately 1.18 times higher at 1 year (95% CI: 1.08–1.23). Furthermore, based on the number and type of encounters present for each individual, the authors were able to estimate the number of days off of work that patients would miss due to health care encounters. This estimate was also significantly lower among patients undergoing laparoscopic approaches (2.78 days fewer, 95% CI: 1.93–3.59 days).
Health Economic Modeling
The MarketScan database was used by Malone et al from the University of Arizona in developing a Markov model for determining the health care costs associated with varying degrees of Crohn's disease severity. 17 The Markov modeling is a method that can be utilized to determine cost-effectiveness or comparative effectiveness of two different hypothetical populations, based on imputation of a copious amount of best-available real-world data. Data from both the MarketScan CCAE database and the MarketScan Medicare Supplemental and Coordination of Benefits database were used as data sources for this study. Because of the detailed data obtained through procedural claims, diagnostic codes (to identify both symptoms and complications), and medication usage, the authors were able to use the American College of Gastroenterology definitions to identify the disease states of remission, mild-moderate, moderate-severe, and severe fulminant. Healthcare costs per 6-month interval were identified within the database and used to estimate the monthly costs associated with each level of disease severity. More importantly, since the data within MarketScan are longitudinal, the authors were able to determine probabilities associated with transitioning from one severity state to another (e.g., progressing from a less severe state to a more severe state, or vice versa, or remaining in the same severity state), based on the observed patterns of disease severity identified within the study population over time. These probabilities were used to parameterize the model, as were the associated costs estimated from the study. Finally, by using values from the literature regarding quality-adjusted life years (QALYs)—the most common metric used in comparative effectiveness research—the authors demonstrated that the costs of care required to gain QALYs were significantly higher among more severe levels of disease: for example, among male patients, costs per QALY were $2,896 for disease in remission, $8,428 for mild-moderate disease, $11,518 for moderate-severe disease, and $69,277 for severe-fulminant disease.
Conclusion
Despite its limitations, the MarketScan family of databases offers unique features that may be of particular interest to clinical researchers. Because of its detail regarding pharmaceutical data—particularly that of outpatient claims—research questions that are traditionally inaccessible by large administrative datasets may be addressed by the data within MarketScan. Evaluating these national trends and patterns of health care utilization in a comprehensive manner is essential for navigating future changes in health care delivery and policy.
References
- 1.Putting Research Data into Your Hands with the MarketScan Databases. 2016. Available at:http://truvenhealth.com/markets/life-sciences/products/data-tools/marketscan-databases. Accessed December 3, 2016
- 2.Quint J. Ann Arbor, MI: Truven Health Analytics Inc.; 2015. Health Research Data for the Real World: The MarketScan Databases. [Google Scholar]
- 3.Miller J. Ann Arbor, MI: Truven Health Analytics Inc.; 2015. Using MarketScan Data for Health Economic Modeling Studies. [Google Scholar]
- 4.Smith J C, Medialia C. Washington, DC: U.S. Government Printing Office; 2015. Health Insurance Coverage in the United States: 2014. [Google Scholar]
- 5.Kulaylat A N, Schubart J R, Schaefer E W, Hollenbeak C S, Cooper A B, Gusani N J. Costs of hepato-pancreato-biliary surgery and readmissions in privately insured US patients. J Surg Res. 2015;199(02):478–486. doi: 10.1016/j.jss.2015.05.002. [DOI] [PubMed] [Google Scholar]
- 6.Stack B C, Bodenner D. Springer International Publishing; 2016. Medical and Surgical Treatment of Parathyroid Diseases: An Evidence-Based Approach. [Google Scholar]
- 7.Carney P I, Lin J, Xia F, Law A. Temporal trend in the use of hysteroscopic vs laparoscopic sterilization and the characteristics of commercially insured and Medicaid-insured females in the US who have had the procedures. Int J Womens Health. 2016;8:137–144. doi: 10.2147/IJWH.S105906. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Huo J, Smith B D, Giordano S H, Reece G P, Tina Shih Y C. A comparison of patient-centered economic and clinical outcomes of post-mastectomy breast reconstruction between obese and non-obese patients. Breast. 2016;30:118–124. doi: 10.1016/j.breast.2016.09.004. [DOI] [PubMed] [Google Scholar]
- 9.Esteva M, Ruiz A, Ramos M et al. Age differences in presentation, diagnosis pathway and management of colorectal cancer. Cancer Epidemiol. 2014;38(04):346–353. doi: 10.1016/j.canep.2014.05.002. [DOI] [PubMed] [Google Scholar]
- 10.Kulaylat A S, Kulaylat A N, Schaefer E W et al. Association of preoperative anti-tumor necrosis factor therapy with adverse postoperative outcomes in patients undergoing abdominal surgery for ulcerative colitis. JAMA Surg. 2017;152(08):e171538. doi: 10.1001/jamasurg.2017.1538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Lauffenburger J C, Balasubramanian A, Farley J F et al. Completeness of prescription information in US commercial claims databases. Pharmacoepidemiol Drug Saf. 2013;22(08):899–906. doi: 10.1002/pds.3458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Sehdev A, Shih Y C, Vekhter B, Bissonnette M B, Olopade O I, Polite B N. Metformin for primary colorectal cancer prevention in patients with diabetes: a case-control study in a US population. Cancer. 2015;121(07):1071–1078. doi: 10.1002/cncr.29165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Peery A F, Dellon E S, Lund J et al. Burden of gastrointestinal disease in the United States: 2012 update. Gastroenterology. 2012;143(05):1179–1.187E6. doi: 10.1053/j.gastro.2012.08.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Foley K A, Wang P F, Barber B L et al. Clinical and economic impact of infusion reactions in patients with colorectal cancer treated with cetuximab. Ann Oncol. 2010;21(07):1455–1461. doi: 10.1093/annonc/mdp535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Erbitux © (cetiximab) [package insert]. New York: ImClone Systems Incorporated and Princeton: NJ: Bristol-Myers Squibb Company; 2006
- 16.Crawshaw B P, Chien H L, Augestad K M, Delaney C P. Effect of laparoscopic surgery on health care utilization and costs in patients who undergo colectomy. JAMA Surg. 2015;150(05):410–415. doi: 10.1001/jamasurg.2014.3171. [DOI] [PubMed] [Google Scholar]
- 17.Malone D C, Waters H C, Van Den Bos J, Popp J, Draaghtel K, Rahman M I. A claims-based Markov model for Crohn's disease. Aliment Pharmacol Ther. 2010;32(03):448–458. doi: 10.1111/j.1365-2036.2010.04356.x. [DOI] [PubMed] [Google Scholar]


