Abstract
Oral cancer is a major health problem in India, and in certain parts, it represents more than 50% of all cancers. Since almost all of these patients receive chemo-radiotherapy with or without surgery for treatment, a vast majority of them also develop oral mucositis, a debilitating adverse effect of chemo-radiation. There have been various reports in the literature regarding the beneficial role of honey in the management of oral mucositis. The objective of this study was to investigate whether the application of honey in mucositis confers any significant improvement in lesions of mucositis and more specifically whether application of honey brings about any improvement in the quality of life of patients suffering from chemo-radiation-induced oral mucositis. If found to be beneficial, honey could provide a simple, elegant and cost-effective solution to a troublesome health problem, thus benefiting a large number of patients.
Keywords: Oral squamous cell carcinoma, Radiotherapy, Mucositis, Honey, Quality of life
Introduction and Review of Literature
Oral cancer is one of the leading causes of morbidity and mortality in the world. It profoundly alters the lives of both patients and their family and imposes a huge demand on healthcare services.
Oral cancer constitutes a group of neoplasms affecting any region of the oral cavity, pharynx and salivary glands. The term oral cancer can, however, be used interchangeably with oral squamous cell carcinoma (OSCC), which according to estimates constitutes more than 90% of all oral neoplasms [1]. OSCC is currently the 10th most common human malignancy affecting more than 378,500 individuals per year worldwide with three quarters of cases belonging to the developing world. In countries of the Indian subcontinent, especially in parts of India, oral cancer can represent more than 50% of all cancers and is therefore a major public health problem [2].
In addition to surgery, radiation and chemotherapy are important modalities in the treatment of cancers. These therapies, being largely non-specific, in addition to tumor cells, act on normal tissues with a high cell turnover, such as those of the oral and gastrointestinal mucosa and the bone marrow.
Oral mucositis is inflammation of the oral mucosa resulting from cancer therapy typically manifesting as atrophy, swelling, erythema and ulceration. Mucositis may be chemotherapy- or radiation-induced.
Prospective studies indicate that the majority of patients having cancer of, or receiving high-dose chemo-radiotherapy to the head and neck, develop severe oral mucositis with pain that is not always palliated by opioid analgesics and approximately 15% will require hospitalization for treatment-related complications. In addition, severe oral mucositis may potentially interfere with the ability to deliver the intended course of therapy, leading to unplanned interruptions in treatment, impacting on loco-regional tumor control, patient survival and quality of life [3, 4].
Radiation mucositis can be described in five overlapping stages: initiation, up-regulation, amplification, ulceration and healing.
Initiation involves radiation exposure and consequential DNA strand breakage and most importantly, generation of Reactive Oxygen Species (ROS), direct damage to cells, tissues and blood vessels, and the start of other biological events that create a cascade of reactions contributing to tissue damage. Activation of transcription factors such as nuclear factor-kappa B, Wnt, p53 and their associated pathways leads to a local increase in pro-inflammatory cytokines including interleukin (IL)-6 and tumor necrosis factor (TNF).
The nuclear factor-kappa B (NF-kB) pathway is the most significant in the context of oral mucositis. Apart from being directly activated by radiation and chemotherapy, NF-kB can be induced indirectly by ROS. Among the numerous genes governed by NF-kB are the ones responsible for production of molecules known to be involved in mucositis, such as cytokines, and cytokine modulators, such as COX-2, inducible NO-synthase, superoxide dismutase. In normal conditions a major consequence of the effects of NF-kB in normal cells is apoptosis.
Positive feedback mechanisms result in amplification and acceleration of the process and further activation of transcription factors, which finally leads to ulceration, the major clinical manifestation of mucositis. Oral bacteria colonize denuded connective tissues, and in neutropenic patients, bacteria may invade into the systemic circulation causing bacteraemia and sepsis [5].
There is no general consensus on the best management of radiation mucositis currently. Antibiotics, NSAIDS, colony-stimulating factors, mucosal coating agents, topical steroids, antiseptic mouthwashes, parenteral amifostine, low-energy laser, glutamine, prostaglandin E analogues and other remedies have been used with mixed success, tarnished mainly by problems such as lack of efficacy, difficulty in using and toxicity [4, 5].
Honey has been a well-known medicament since ancient times, but recently there has been a resurgence of interest in using honey in wound management. A detailed account of the antimicrobial properties of honey is beyond the scope of this discussion, but its efficacy against various Gram-positive and Gram-negative bacteria and fungi has been reported. Also honey, unlike other antimicrobials, is not cytotoxic to host cells even at high concentrations [6, 7].
Honey has potent anti-inflammatory properties. High concentration of the free radicals during inflammation can damage and break down lipids, proteins and nucleic acids. Honey can reduce the damage caused by free radicals and thus prevents further tissue necrosis. Consumption of honey has been demonstrated to improve significantly the antioxidant profile of the individual’s serum, thus acting to eliminate oxidative stress in tissues [8, 9].
In their landmark animal study, Hussein et al. [10] conclusively demonstrated the molecular mechanism of the anti-inflammatory action of Gelam honey in a murine model. They demonstrated that Gelam honey inhibited the nuclear translocation and activation of the NF-kB pathway, which plays a key role in radiation-induced mucositis.
The application of honey for amelioration of chemo-radiation-induced mucositis has undergone a fair bit of scrutiny. Motallebnejad et al. [11] reported a statistically significant reduction in mucositis among honey-receiving patients given chemo-radiotherapy (CRT) compared with controls (p = 0.000). In their trial Rashad et al. [12] reported that only 15% of the study group receiving topical honey with concomitant CRT developed grade 3 mucositis as opposed to 65% of the control group. Khanal et al. [13] (2010), comparing the mucositis-limiting qualities of honey with lignocaine, reported that a significantly lower proportion of patients developed intolerable oral mucositis in the honey group. Samdariya et al. [14] in their RCT reported honey significantly reduced the severity of mucositis associated pain and lessened treatment gaps. Similar findings have been reported elsewhere in the literature [15–19].
In the light of these results, honey may be a viable adjunct treatment for oral mucositis.
Aim of the Study
The beneficial influence of honey in mucositis has been investigated adequately by in vitro and in vivo studies, but the assessment of any intervention is incomplete without an assessment of its actual impact on the quality of human life. The aim of this study therefore was to assess the effect of topical application and systemic (oral) intake of honey in chemo-radiation-induced mucositis and to assess its clinical benefits in improving quality of life in patients of head and neck cancer.
Material and Method
A prospective single blind randomized control trial was carried out on patients of biopsy-proven head and neck squamous cell carcinoma undergoing neoadjuvant chemotherapy followed by concurrent chemo-radiation. A total of forty patients (stage III and IVa TNM) receiving radiation to the head and neck included in the study were randomized (by computer generated random numbers) into two groups, a case group and a control group each containing twenty patients.
Both groups were given two cycles of Taxol-based induction chemotherapy at intervals of 3 weeks and then subjected to radiation along with cisplatin based chemotherapy 4 weeks from completion of induction chemotherapy. Radiation was given by external beam conventional method to primary tumor site and neck. All patients were given standard oral hygiene instructions. Dental prophylaxis was done for all patients by a periodontist.
Ethical clearance was obtained from the Institutional Ethics Committee, and informed consent of all patients was taken in a standard format as advised by the research cell, King George’s Medical University, Lucknow.
The parameters evaluated in the study were:
Mucositis was assessed by the Common Terminology Criteria for Adverse Events (CTCAE) version 4, weekly (after radiation was started) during the course of radiotherapy and once more four weeks after completion of radiation.
To assess the effect on quality of life, we used the FACT-H&N (version 4) questionnaire with the kind permission of FACIT.org. The patient was interviewed weekly (after radiation was started) during the course of radiotherapy and once more 4 weeks after completion of radiation. All patients were interviewed using the Hindi version of the questionnaire provided by FACIT.org through e-mail correspondence. The QOL scores were calculated by the FACIT Administration and Scoring Guidelines.
The patients in the study group were instructed to rinse their mouth slowly with 20 ml honey and then swallow it slowly, 15 min before radiation exposure, and 15 min after exposure and 6 h post-radiation therapy. (For the purposes of this study, fresh, organic and unprocessed honey was sourced from the National Botanical Research Institute).
Additionally in keeping with the findings of previous studies [17, 18], the study group was instructed to consume a total of 100 ml (1.2–1.5 gm/kg/day of) honey per day in divided doses to maintain adequate serum antioxidant levels to protect against oxidative stress.
The control group was instructed to rinse their mouth with normal saline 15 min before radiation exposure and 15 min after radiation exposure.
Results
A total of 20 cases and 20 age (± 3 years)- and sex-matched controls were included in the study. There was no significant (p > 0.05) difference in regard to the age, gender and primary site of carcinoma between cases and controls showing comparability of the groups. SCC of the Buccal mucosa was the most common diagnosis in both cases (40%) and controls (25%). SCC of the supraglottic larynx was the second most common diagnosis in both cases (15%) and controls (20%) (Fig. 1).
Fig. 1.
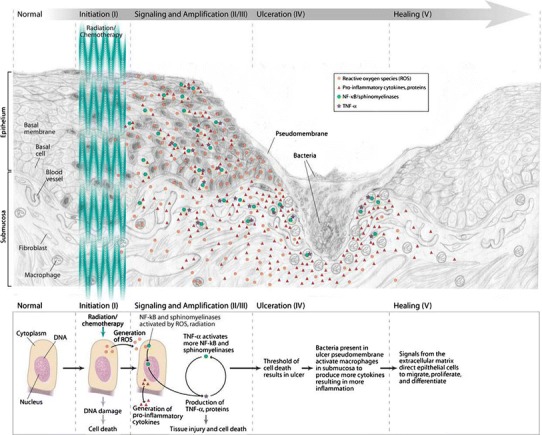
The pathogenesis of oral mucositis given by Dr Stephen T. Sonis [5]
The quality of life (QOL) was insignificantly (p > 0.05) lower in cases (92.90 ± 21.56) than controls (99.34 ± 14.78) at 1st week. There was no significant (p > 0.05) difference in QOL at 2nd, 3rd and 4th weeks between cases and controls. However, QOL became significantly (p = 0.0001) higher in cases (102.55 ± 11.03) compared to controls (82.4 ± 12.00) 4th week post-radiotherapy (Fig. 2, Table 1).
Fig. 2.

Comparison of QOL between cases and controls at time intervals
Table 1.
Comparison of QOL between cases and controls at time intervals
| Cases (n = 20) | Controls (n = 20) | p valuea | |
|---|---|---|---|
| 1st week | 92.90 ± 21.56 | 99.34 ± 14.78 | 0.27 |
| 2nd week | 90.38 ± 16.63 | 90.67 ± 14.84 | 0.95 |
| 3rd week | 83.53 ± 15.24 | 82.69 ± 14.38 | 0.85 |
| 4th week | 75.73 ± 16.62 | 76.55 ± 13.96 | 0.86 |
| 4th week post-therapy | 102.55 ± 11.03 | 82.4 ± 12.00 | 0.0001* |
aUnpaired t test, *Significant
There was no significant (p > 0.05) difference in mucositis grade at 1st, 2nd, 3rd and 4th weeks between cases and controls. However, mucositis grade was significantly (p = 0.0001) different between cases and controls at 4th week post-therapy (Fig. 3, Table 2).
Fig. 3.

Comparison of mucositis grade between cases and controls
Table 2.
Comparison of mucositis grade between cases and controls at time intervals
| Mucositis grade | Cases (n = 20) | Controls < n = 20) | p valuea | ||
|---|---|---|---|---|---|
| No. | % | No. | % | ||
| 1st week | |||||
| Grade 1 | 14 | 70.0 | 16 | 80.0 | 0.53 |
| Grade 2 | 5 | 25.0 | 4 | 20.0 | |
| Grade 3 | 1 | 5.0 | 0 | 0.0 | |
| 2nd week | |||||
| Grade 1 | 11 | 55.0 | 9 | 45.0 | 0.60 |
| Grade 2 | 6 | 30.0 | 9 | 45.0 | |
| Grade 3 | 3 | 15.0 | 2 | 10.0 | |
| 3rd week | |||||
| Grade 1 | 5 | 55.0 | 5 | 25.0 | 0.92 |
| Grade 2 | 11 | 30.0 | 10 | 50.0 | |
| Grade 3 | 4 | 15.0 | 5 | 25.0 | |
| 4th week | |||||
| Grade 1 | 2 | 10.0 | 4 | 20.0 | 0.16 |
| Grade 2 | 12 | 60.0 | 6 | 30.0 | |
| Grade 3 | 6 | 30.0 | 10 | 50.0 | |
| 4th week post-radiotherapy | |||||
| Grade 1 | 19 | 95.0 | 7 | 35.0 | 0.0001* |
| Grade 2 | 1 | 5.0 | 12 | 60.0 | |
| Grade 3 | 0 | 0.0 | 1 | 5.0 | |
aUnpaired t test, *Significant
There was significant decrease (p < 0.05) in both cases and controls from 1st week to 3rd and 4th week. But, there was significant increase (p < 0.05) in QOL in cases from 1st, 2nd, 3rd and 4th to 4th week post-therapy. QOL was significantly (p = 0.0001) increased only from 4th week to 4th week post-therapy in both cases and controls. However, the mean improvement of QOL was much better in cases than in controls (Fig. 4, Table 3).
Fig. 4.
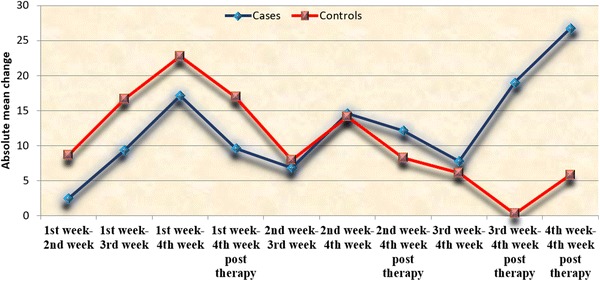
Comparison of absolute mean change in QOL in cases and controls from 1st week to subsequent time intervals
Table 3.
Comparison of mean change in QOL in cases and controls from 1st week to subsequent time intervals
| Time intervals | Cases (n = 20) | Controls (n = 20) | ||
|---|---|---|---|---|
| Mean change | p valuea | Mean change | p valuea | |
| 1st week–2nd week | − 2.52 ± 17.21 | 0.52 | − 8.66 ± 8.73 | 0.0001* |
| 1st week–3rd week | − 9.37 ± 18.66 | 0.03* | − 16.64 ± 9.44 | 0.0001* |
| 1st week–4th week | − 17.17 ± 23.10 | 0.004* | − 22.79 ± 10.88 | 0.0001* |
| 1st week–4th week post-therapy | 9.64 ± 19.86 | 0.04* | − 16.94 ± 11.73 | 0.0001* |
| 2nd week–3rd week | − 6.84 ± 8.06 | 0.001* | − 7.98 ± 5.22 | 0.0001* |
| 2nd week–4th week | − 14.64 ± 17.63 | 0.001* | − 14.12 ± 7.39 | 0.0001* |
| 2nd week–4th week post-therapy | 12.16 ± 14.62 | 0.001* | − 8.27 ± 9.02 | 0.001* |
| 3rd week–4th week | − 7.8 ± 11.94 | 0.009* | − 6.14 ± 6.02 | 0.0001* |
| 3rd week–4th week post-therapy | 19.01 ± 14.49 | 0.0001* | − 0.29 ± 7.61 | 0.86 |
| 4th week–4th week post-therapy | 26.81 ± 17.80 | 0.0001* | 5.85 ± 5.09 | 0.0001* |
aUnpaired t test
The pre- and post-intervention clinical picture (Figs. 5, 6).
Fig. 5.

Study group
Fig. 6.

Control group
Discussion
In recent times there has been a lot of focus on interventions that positively impact quality of human life. The main objective of this trial was to investigate the impact of honey on improvement in quality of life in patients receiving chemo-radiation.
During the course of the trial, both groups demonstrated steady deterioration in their QOL scores with each week of radiotherapy. But the mean reduction in QOL scores was more for controls than cases. Also improvement following cessation of radiotherapy was significantly more in the case group.
Although swallowing and dietary patterns were not independently assessed, the patients in the study group showed less impairment in swallowing and reported less mucosal pain and hence required less dietary restriction to liquid foods.
From these results, it can be postulated with confidence that isolation of the active therapeutic agents in honey and their targeted delivery could provide significant and quantifiable therapeutic benefit to patients of chemo-radiation-induced mucositis and is therefore a promising field of future research.
Conclusion
Honey not only seems to help combat the ill effects of cumulative radiation damage pertaining to mucositis like mucosal drying, irritation and dysphagia but also speeds up recovery, expediting the process of return to a normal quality of life.
Natural honey is a product with rich nutritional qualities that could be a pleasant, simple and economic modality for prevention and management of chemo-radiation-induced mucositis.
Author Contribution
D.H contributed to data collection and processing, V.S helped in conception of idea and study design, S.M. revised the intellectual content, S.G. involved in study design and study parameters, U.S.P. made critical review, and M.P. participated in logistic support.
Contributor Information
Debraj Howlader, Email: drdebrajhowlader@gmail.com.
Vibha Singh, Email: vibhasinghraghuvanshi@gmail.com, Email: drvibha66@yahoo.com.
Shadab Mohammad, Email: shadab31aug@yahoo.com.
Seema Gupta, Email: seemaguptart@gmail.com.
U. S. Pal, Email: uspalkgmc@gmail.com
Mahesh Pal, Email: m.pal@nbri.res.in.
References
- 1.Anastasios K. Markopoulos, current aspects on oral squamous cell carcinoma. Open Dent J. 2012;6:126–130. doi: 10.2174/1874210601206010126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Crispian Scully, Cancers of the Oral Mucosa. http://emedicine.medscape. com/article/1075729-overview#a6
- 3.Raber-Durlacher JE, Elad S, Barasch A. Oral mucositis. Oral Oncol. 2010;46:452–456. doi: 10.1016/j.oraloncology.2010.03.012. [DOI] [PubMed] [Google Scholar]
- 4.Parulekar W, Mackenzie R, Bjarnason G, Jordan RCK. Scoring oral mucositis. Oral Oncol. 1998;34:63–71. doi: 10.1016/S1368-8375(97)00065-1. [DOI] [PubMed] [Google Scholar]
- 5.Sonis ST. Mucositis: the impact, biology and therapeutic opportunities of oral mucositis. Oral Oncol. 2009;45:015–1020. doi: 10.1016/j.oraloncology.2009.08.006. [DOI] [PubMed] [Google Scholar]
- 6.Jeddar A, Kharsany A, Ramsaroop UG, Bhamjee A, Haffejee IE, Moosa A-S. The antibacterial action of honey. An in vitro study. S Afr Med J. 1985;67(7):257–258. [PubMed] [Google Scholar]
- 7.Ansari MJ, Al-Ghamdi A, Usmani S, Al-Waili NS, Sharma D, Nuru A, Al-Attal Y. Effect of jujube honey on Candida albicans growth and biofilm formation. Arch Med Res. 2013;44:352e360. doi: 10.1016/j.arcmed.2013.06.003. [DOI] [PubMed] [Google Scholar]
- 8.Al-Waili NS. Effects of daily consumption of honey solution on haematological indices and blood levels of minerals and enzymes in normal individuals. J Med Food. 2003;6:135–140. doi: 10.1089/109662003322233549. [DOI] [PubMed] [Google Scholar]
- 9.Schramm DD, Karim M, Schrader HR, Holt RR, Cardetti M, Keen CL. Honey with high levels of antioxidants can provide protection to healthy human subjects. J Agric Food Chem. 2003;51:1732–1735. doi: 10.1021/jf025928k. [DOI] [PubMed] [Google Scholar]
- 10.Hussein SZ, Mohd Yusoff K, Makpol S, Mohd Yusof YA. Gelam honey attenuates carrageenan-induced rat paw inflammation via NF-kB pathway. PLoS ONE. 2013;8(8):e72365. doi: 10.1371/journal.pone.0072365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Motallebnejad M, Akram S, Moghadamnia A, Moulana Z, Omidi S. The effect of topical application of pure honey on radiation-induced mucositis: a randomized clinical trial. J Contemp Dent Pract. 2008;9(3):40–47. [PubMed] [Google Scholar]
- 12.Rashad UM, Al-Gezawy SM, El-Gezawy E, Azzaz AN. Honey as topical prophylaxis against radiochemotherapy-induced mucositis in head and neck cancer. J Laryngol Otol. 2009;123(2):223–228. doi: 10.1017/S0022215108002478. [DOI] [PubMed] [Google Scholar]
- 13.Khanal B, Baliga M, Uppal N. Effect of topical honey on limitation of radiationinduced oral mucositis: an intervention study. Int J Oral Maxillofac Surg. 2010;39:1181–1185. doi: 10.1016/j.ijom.2010.05.014. [DOI] [PubMed] [Google Scholar]
- 14.Samdariya S, Lewis S, Kauser H, Ahmed I, Kumar D. A randomized controlled trial evaluating the role of honey in reducing pain due to radiation induced mucositis in head and neck cancer patients. Indian J Palliat Care. 2015;21(3):268–273. doi: 10.4103/0973-1075.164892. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Charalambous M, Raftopoulos V, Lambrinou E, Charalambous E. The effectiveness of honey for the management of radiotherapy-induced oral mucositis in head and neck cancer patients: a systematic review of clinical trials. Eur J Integr Med. 2013;5(2013):217–225. doi: 10.1016/j.eujim.2013.01.003. [DOI] [Google Scholar]
- 16.Cho HK, Jeong YM, Lee HS, Lee YJ, Hwang SH. Effects of honey on oral mucositis in patients with head and neck cancer: a meta-analysis. Laryngoscope. 2005;125:2085–2092. doi: 10.1002/lary.25233. [DOI] [PubMed] [Google Scholar]
- 17.Song JJ, Twumasi-Ankrah P, Salcido R. Systematic review and meta-analysis on the use of honey to protect from the effects of radiation induced oral mucositis. Adv Skin Wound Care. 2012;25(1):23–28. doi: 10.1097/01.ASW.0000410687.14363.a3. [DOI] [PubMed] [Google Scholar]
- 18.Co JL, Mejia MBA, Que JC, Dizon JMR. Effectiveness of honey on radiation-induced oral mucositis, time to mucositis, weight loss, and treatment interruptions among patients with head and neck malignancies: a meta-analysis and systematic review of literature. Head Neck. 2016;38(7):1119–1128. doi: 10.1002/hed.24431. [DOI] [PubMed] [Google Scholar]
- 19.Xu J-L, Xia R, Sun Z-H, Sun L, Min X, Liu C, Zhang H, Zhu Y-M. Effects of honey use on the management of radio/chemotherapy-induced mucositis: a metaanalysis of randomized controlled trials. Int J Oral Maxillofac Surg. 2016;45:1618–1625. doi: 10.1016/j.ijom.2016.04.023. [DOI] [PubMed] [Google Scholar]


