Abstract
Introduction
Anti-interferon-gamma (IFN-γ) autoantibodies are increasingly recognized as a cause of adult-onset immunodeficiency (AOID) worldwide. These patients are susceptible to various intracellular pathogens especially nontuberculous mycobacteria. Most of the patients have a refractory clinical course. Herein, we report the use of immunotherapy with pulse intravenous cyclophosphamide (IVCY) in patients who had progressive, refractory Mycobacterium abscessus infection.
Method
We included patients, seen at Srinagarind Hospital, Thailand, infected with M. abscessus, who had received ≥3 courses of parenteral antibiotics within the last 12 months and who received pulse IVCY with a tapering dose of prednisolone.
Results
There were 8 AOID patients who met the criteria and received pulse IVCY between January 2011 and December 2015. One patient was lost to follow-up after 5 courses of IVCY: he had died at home 3 months later. Five patients had favorable outcomes: 2 were able to discontinue NTM therapy, and 3 had stable disease and were on NTM treatment without hospitalization for parenteral antibiotics. Two patients relapsed and needed hospitalization. The IFN-γ Ab titers among the 7 patients were significantly decreased during treatment, and the median initial antibody titer started at 200,000 and then decreased to 5,000 after 2 years of treatment (P < 0.0001). The antibody titer reduction among responsive vs. nonresponsive patient was significantly different after 6 months of treatment: the median antibody titer was 5,000 and 100,000, respectively (P = 0.0467).
Conclusion
IVCY therapy might be an alternative treatment for AOID patients infected with M. abscessus and refractory to antimycobacterial therapy.
1. Introduction
Anti-interferon-γ (IFN-γ) autoantibodies are increasingly recognized as a cause of adult-onset immunodeficiency (AOID) worldwide [1–19]. These patients are susceptible to various intracellular pathogens especially nontuberculous mycobacteria (NTM). The majority of patients have a persistent NTM infection and require long-term antimycobacterial therapy [20]. Some patients infected with rapidly growing mycobacteria (RGM) have recurrent disease despite continuous oral antimycobacterial treatment and need hospitalization for parenteral imipenem. Various adjunctive therapies have been used to treat patients with anti-IFN-γ autoantibodies [3, 4, 21–25], including B cell depletion therapy with rituximab which has attracted much attention. Owing to economic constraints, rituximab is not accessible through the Thai National Health Insurance program for off-label treatment.
Cyclophosphamide is an alkylating agent affecting depletion of both B and T cells, hence reducing the production of pathogenic autoantibodies. Intravenous cyclophosphamide (IVCY) has been used successfully as an induction therapy for severe lupus nephritis, either 6 monthly pulses of IVCY (NIH regimen) [26] or 6 pulses of lower-dose IVCY every 2 weeks (Euro-Lupus Nephritis Trail) [27]. The low-dose regimen was found to be associated with half as many severe infections as compared to the high-dose regimen, and long-term outcomes did not differ at the 10-year follow-up [28]. Currently, IVCY has been used as a standard regimen particularly for severe autoimmune and autoinflammatory diseases such as systemic lupus erythematosus (SLE) [29–32], active alveolitis from systemic sclerosis [33–35], antineutrophil cytoplasmic antibodies- (ANCA-) associated primary vasculitis syndrome [36–38], and other autoimmune diseases [39–44].
Plasmapheresis followed by pulse cyclophosphamide has been successfully treated in an AOID patient with recurrent MAC infection [21]. Herein, we report the use of immunotherapy with intravenous cyclophosphamide (IVCY) in 8 AOID patients with a high titer of anti-IFN-γ autoantibodies who had progressive refractory M. abscessus infection needing frequent hospitalization for parenteral antibiotics.
2. Methods
2.1. Ethics
This study is a retrospective study and has been approved by our ethics committee (HE601199).
2.2. Patients and Immunotherapy
All patients were seen at Srinagarind Hospital, a tertiary care university hospital. The standard regimen for treating AOID patients infected with RGM is a combination of oral antimycobacterial agents, mostly macrolides and fluoroquinolones. If they have progressive disease—despite being continuously on oral antimycobacterial treatment—they will be hospitalized for parenteral antibiotic treatment (i.e., particularly imipenem for 2–4 weeks).
We included patients who received ≥3 courses of parenteral antibiotics within 12 months and who received pulse IVCY. The regimen consists of methylprednisolone 1,000 mg intravenous on the last day of parenteral antibiotics followed by oral prednisolone 30 mg/day and IVCY 400 mg every 2 weeks for 6 cycles, then IVCY 400 mg every 4–6 weeks for 3 cycles plus oral prednisolone 15 mg/day, then IVCY 400 mg every 8–12 weeks for 3 cycles and oral prednisolone 10 mg/day, and then IVCY 400 mg every 12 weeks and oral prednisolone 5 mg/day until 2 years of therapy was completed. All patients received a combination of oral antimycobacterial treatment continuously.
2.3. Clinical Monitoring
Patients underwent routine safety monitoring during the IVCY therapy, including complete blood count with differentials, urine analysis, and renal and hepatic function chemistries. We also monitored their anti-IFN-γ titer. Disease activity was assessed by observing clinical signs and evidence of active infection on computed tomography, pathology, culture, or smear as indicated. Treatment and clinical data were collected by review of chart records.
2.4. Determination of IFN-γ Autoantibody Titers
A method for determining anti-IFN-γ autoantibody titer was previously described [20, 45]. Briefly, a 96-well polystyrene plate (Nunc) was precoated with 100 μl of anti-human IFN-γ capture antibody (BD Biosciences) overnight at 4°C. On the day of the experiment, the precoated plate was washed 3 times with 0.05% Tween20 in phosphate-buffered saline (PBS) and then blocked with 200 μl of 10% fetal bovine serum in PBS for 2 h at room temperature. Meanwhile, plasma samples were diluted with 300 pg/ml of recombinant human IFN-γ at 1 : 100, 1 : 1,000, 1 : 5,000, 1 : 10,000, 1 : 50,000, 1 : 100,000, 1 : 200,000, 1 : 400,000, 1 : 800,000, and 1 : 1,600,000 before being incubated at 37°C for 1 h. The preincubated diluted plasma samples (100 μl) were added to the plate and incubated at room temperature for 1 h. After washing 5 times with 0.05% Tween20-PBS, a 100 μl mixture of horseradish peroxidase (HRP), tagged streptavidin, and biotinylated anti-human IFN-γ detection antibody (BD Biosciences) was added to the plate and incubated at room temperature for 1 h. The plate was washed 7 times before adding 100 μl of tetramethylbenzidine (TMB) substrate (BD Biosciences), which was incubated at room temperature for 15 min. The reaction was stopped by adding 25 μl of 2 N H2SO4. Absorbance was measured at 450/570 nm by spectrophotometry.
The level of detectable human IFN-γ was calculated according to the standard curve from each plate. The percentage of neutralizing IFN-γ from each diluted plasma sample was calculated according to the equation below. The highest plasma dilution that resulted in ≥50% neutralization of the IFN-γ was considered a positive titer.
| (1) |
GraphPad Prism version 6 (GraphPad) was used for the statistical analysis. Statistical significance was determined using ANOVA with Dunnett's multiple comparisons test.
All the data used to support the findings of this study are included within the article.
3. Results
3.1. Patients
There were 8 AOID patients who met the criteria and received pulse IVCY between January 2011 and December 2015. All of the patients had M. abscessus lymphadenitis along with other organ involvements. All but two patients were coinfected with other opportunistic infections. All of the patients had had progressive mycobacterial infection for over 10–48 months. The median NTM infection was 17 months before IVCY therapy. The patients received 3-6 courses of parenteral antimicrobial (mostly imipenem) within 12 months prior to the IVCY.
Table 1 summarizes the clinical data and outcomes of disseminated M. abscessus-infected patients treated with IVCY. Patients received 5–25 cycles of IVCY (median, 17 cycles). There were no serious laboratory test results related to cyclophosphamide during the IVCY therapy. Patient #1 received 5 courses of IVCY and then was lost to follow-up: he had died at home some 3 months after we lost contact with him. (Patient #1 was censored from the analysis.) Among the 7 evaluable cases, 5 had favorable outcomes: 2 (patients #2 and #3) had complete remission and were able to discontinue the NTM therapy while 3 (patients #4, #5, and #6) had stable disease but remained on NTM treatment without the need of hospitalization for parenteral antibiotics. Two (patients #7 and #8) relapsed and needed hospitalization. Patient #7 had enlarged lymph nodes after 12 cycles of IVCY and needed hospitalization for imipenem; after which, he resumed IVCY for another 3 cycles but developed Herpes zoster ophthalmicus which required admission for administration of parenteral acyclovir. He had a persistent enlarged epitrochlear lymph node after 25 cycles of IVCY. Patient #8 had 2 relapses requiring hospitalization for parenteral imipenem.
Table 1.
Clinical data and outcomes among disseminated M. abscessus-infected patients treated with intravenous cyclophosphamide (IVCY).
| Patient no. | Age/sex | Organ involvement | Treatment duration before IVCY (month) | Other opportunistic infection | Oral antibiotic | Parenteral antibiotic prior to IVCY/duration (week) | No. of IVCY cycle received | Initial IFN-γ Ab titer | IFN-γ Ab titer at the end of treatment | Follow-up after IVCY (month) | Outcome |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 52/M | Lymph nodes, lung, spleen | 19 | Tuberculosis lung, Herpes zoster, penicilliosis | Clarithromycin, ofloxacin | (1) Cefoxitin/2 (2) Imipenem/2 (3) Imipenem/4 (4) Imipenem/4 |
5 | 1 : 100,000 | 1 : 50,000 | Not applicable | Unevaluable: lost to follow-up and died 3 months later |
|
| |||||||||||
| 2 | 54/M | Lymph nodes, lung | 15 | None | Clarithromycin, ciprofloxacin | (1) Imipenem/2 (2) Imipenem/4 (3) Imipenem/4 |
14 | 1 : 200,000 | 1 : 5,000 | 57 | Response: cured with discontinued NTM treatment |
|
| |||||||||||
| 3 | 57/M | Lymph nodes, lung, liver | 10 | Tuberculosis lung | Azithromycin, ciprofloxacin | (1) Imipenem/4 (2) Imipenem/4 (3) Imipenem/4 |
16 | 1 : 200,000 | 1 : 1,000 | 44 | Response: cured with discontinued NTM treatment |
|
| |||||||||||
| 4 | 41/M | Lymph nodes, nasal septum | 19 | Melioidosis | Clarithromycin, ofloxacin | (1) Imipenem/2 (2) Imipenem/4 (3) Imipenem/4 (4) Imipenem/4 |
17 | 1 : 200,000 | 1 : 1,000 | 60 | Response: stable on NTM treatment without hospitalization for parenteral antibiotic |
|
| |||||||||||
| 5 | 54/F | Lymph nodes, skin | 12 | None | Clarithromycin, ciprofloxacin | (1) Imipenem/4 (2) Imipenem/4 (3) Imipenem/4 (4) Imipenem/4 |
17 | 1 : 100,000 | 1 : 1,000 | 28 | Response: stable on NTM treatment without hospitalization for parenteral antibiotic |
|
| |||||||||||
| 6 | 65/M | Lymph nodes, lung, femur | 48 | Salmonellosis | Clarithromycin, ciprofloxacin | (1) Imipenem/4 (2) Imipenem/4 (3) Imipenem/4 |
17 | 1 : 100,000 | 1 : 5,000 | 44 | Response: stable on NTM treatment without hospitalization for parenteral antibiotic |
|
| |||||||||||
| 7 | 34/M | Lymph nodes, lung, multiple bones | 15 | Cryptococcosis, tuberculosis bone | Clarithromycin, ciprofloxacin, linezolid | (1) Imipenem/2 (2) Imipenem/2 (3) Imipenem/4 (4) Imipenem/4 (5) Imipenem/4 (6) Imipenem/4 |
25 | 1 : 200,000 | 1 : 50,000 | 45 | Not response: relapsed with hospitalization for parenteral antibiotic |
|
| |||||||||||
| 8 | 20/F | Lymph nodes, skin, lung, liver, spleen, pancreas, humerus | 26 | Cryptococcosis, Herpes zoster | Azithromycin, ofloxacin, doxycycline, linezolid | (1) Imipenem/4 (2) Imipenem/4 (3) Imipenem/4 |
20 | 1 : 400,000 | 1 : 50,000 | 41 | Not response: relapsed twice with hospitalization for parenteral antibiotic |
3.2. Anti-IFN-γ Autoantibody Titers
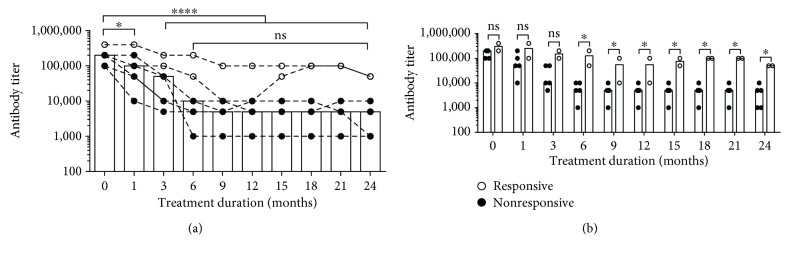
The anti-IFN-γ Ab titers among the 7 evaluable patients were significantly decreased during treatment. The median initial antibody titer starting at 200,000 then was decreased to 5,000 after 2 years of treatment (P < 0.0001). The median antibody titers started to be significantly reduced 1 month after receiving IVCY (Figure 1). Antibody titer reduction among responsive patients (n = 5) vs. nonresponsive patients (n = 2) was significantly different after 6 months of treatment (median antibody titer was 5,000 and 100,000, respectively) (P = 0.0467). Antibody titer among the responsive patients started at between 100,000 and 200,000 and was reduced to around 5,000 (range, 1,000–10,000). The antibody titer of nonresponsive patients started at between 200,000 and 400,000 and was reduced to between 10,000 and 200,000 after the first year of treatment and to 100,000 after the second year of treatment.
Figure 1.
Anti-IFN-γ autoantibody titer changes in plasma from responsive patients (n = 5, close circle (●)) and nonresponsive patients (n = 2, open circle (○)) at different time points after receiving intravenous cyclophosphamide (IVCY) pulse therapy. (a) Align dot plot with bar graph represents the median anti-IFN-γ autoantibody titer changing dynamic at each time point in all 7 cases. (b) Scatter dot plot with bar graph compares the median titer between responsive and nonresponsive patients. Statistical significance was determined using ANOVA with Dunnett's multiple comparisons test; ns: nonsignificant; ∗P < 0.05 and ∗∗∗∗P < 0.0001.
Among the 5 responsive patients, their anti-IFN-γ autoantibody titers were followed for 1 year after completing the IVCY pulse therapy (Figure 2). The median anti-IFN-γ autoantibody titer had increased from 5,000 to 10,000 three months after discontinuation of treatment, albeit the difference was not statistically significant. The median titers remained stable for up to 12 months.
Figure 2.
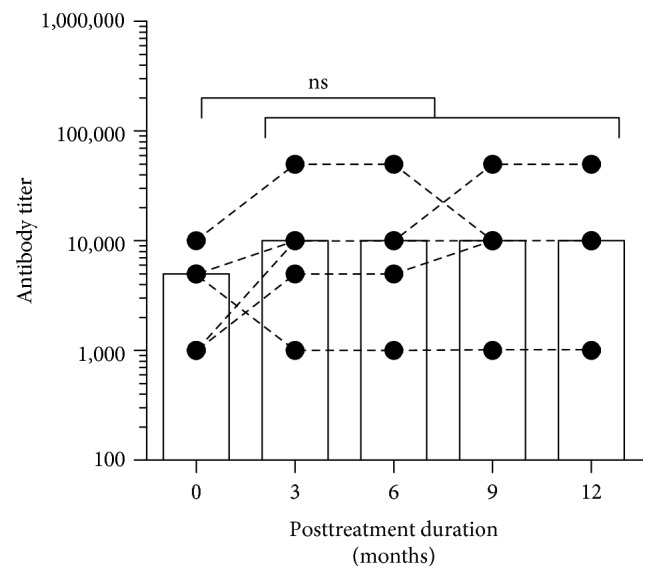
Anti-IFN-γ autoantibody titers of responsive patients after stopping IVCY pulse therapy. Median anti-IFN-γ autoantibody titers in plasma from responsive patients (N = 5) were followed for 1 year at different time points after stopping IVCY pulse therapy. Statistical significance was determined using ANOVA with Dunnett's multiple comparisons test; ns: nonsignificant.
4. Discussion
Our patients had extensive disease as a result of M. abscessus infection, which progressed over the years: the median duration of infection was 17 months. All of the patients had lymphadenitis along with other sites of M. abscessus infection (Table 1). All but two had coinfection with other opportunistic infections. The patients were refractory to antimycobacterial treatment, so we sought an adjunctive immunotherapy. Rituximab has been used in AOID patients associated with mycobacterial infection with clinical improvement [22–25]; among 7 patients reported, they received 4-18 cycles of rituximab over 1-5 years of treatment. Since rituximab is not accessible in our setting, we tried to treat our AOID patients with an available immunosuppressant drug. We used an IVCY regimen resembling the Euro-Lupus protocol for lupus nephritis in combination with a single pulse of methyl prednisolone followed by a moderate dose of prednisolone for the first 3 months and maintenance with low-dose corticosteroid until the end of study. We treated only patients who had a protracted clinical course, defined as having received ≥3 courses of parenteral antibiotic within 12 months. We started the IVCY treatment regimen at the end of the parenteral therapy after the disease was controlled.
Among the 7 evaluable cases, 5 had a clinical response. To date, in two of our patients, the M. abscessus infection was cleared, and the patients were able to discontinue NTM treatment. Three of the remaining patients had stable disease and continued treatment for M. abscessus without needing to be hospitalized for parenteral therapy. Only 2 patients did not respond; they relapsed and needed hospitalization for imipenem therapy.
We demonstrated that IVCY reduced IFN-γ Ab titers significantly. Among the 7 patients studied, the median initial titers decreased after 2 years of treatment (P < 0.0001) and the titers started to decline within one month of receiving IVCY (Figure 1(a)). Comparing between responders and nonresponders, the reduction in Ab titers among responders was significantly greater than that among nonresponders after 6 months of therapy throughout the end of 2 years of therapy (Figure 1(b)). The IFN-γ titers among responders at the end of treatment ranged between 1,000 and 5,000 while for nonresponders' titers, they averaged 50,000. Our findings suggest that IFN-γ Ab titers are correlated to clinical response; thus, an early drop in IFN-γ titer is a predictor of a good outcome of IVCY treatment. The use of Ab titers to monitor disease activity or to determine when to discontinue antimycobacterial treatment needs further investigation.
Patients #7 and #8 received an extended course of IVCY (25 and 20 cycles, respectively). The lack of clinical improvement suggests that extension of IVCY treatment does not help nonresponders to reduce Ab titers to the level of a responsive titer. Among IVCY nonresponders, switching to other immunosuppressive drugs (i.e., mycophenolate mofetil, azathioprine, or calcineurin inhibitor) may be considered. Importantly, Ab titers among responders were stable up to one year after stopping IVCY therapy.
The present study had some limitations. First, it was a retrospective study, so some information might have been lost. Second, the study had a small sample size but that is partly because AOID is uncommon, and not all cases of anti-IFN-γ autoantibody-mediated disseminated NTM require immunotherapy. Third, we did not identify the isolates of M. abscessus to subspecies level nor identify an inducible macrolide resistance gene which might have effect on the treatment outcomes. Fourth, we did not perform plasma-mediated inhibition of IFN-γ-stimulated STAT-1 phosphorylation; notwithstanding, we previously demonstrated that plasma specimens containing anti-interferon-γ autoantibodies inhibited interferon-γ-induced STAT1 phosphorylation [1]. The Ab titers were correlated to clinical response; thus, we hypothesize that the IFN-γ antibodies in our patients were functioning. Despite these limitations, our results offer valuable clinical information vis-à-vis patients with anti-IFN-γ autoantibody-associated M. abscessus infection who had refractory disease in a resource-limited setting.
In conclusion, IVCY therapy might be an alternative adjunctive immunotherapy for AOID patients infected with M. abscessus and refractory to antimycobacterial treatment since it improved clinical outcomes and reduced the titer of anti-IFN-γ autoantibodies. A rapid decline in the titer of anti-IFN-γ antibody after 6 months of immunosuppressive treatment predicted a good outcome. In cases of a persistently high anti-IFN-γ antibody titer, rather than extending the use of IVCY, the clinician should consider switching to other immunosuppressive drug(s) with different target(s) of action. To avoid serious infection related to the use of long-term cyclophosphamide, a better outcome may be achieved by using a less toxic immunosuppressive drug.
Acknowledgments
The authors thank Mr. Bryan Hamman for the assistance with the English-language presentation of the manuscript under the aegis of the Publication Clinic KKU, Thailand. This work has received scholarship under the Post-Doctoral Training Program from Research Affairs and Graduate School, Khon Kaen University, Thailand (Grant no. 59260).
Data Availability
The data used to support the findings of this study are included within the article.
Disclosure
This study has been presented as conference abstract in 28th ECCMID according to the following link: https://www.escmid.org/escmid_publications/escmid_elibrary/material/?mid=64583.
Conflicts of Interest
None of the authors declared conflict of interests relevant for the present work.
References
- 1.Browne S. K., Burbelo P. D., Chetchotisakd P., et al. Adult-onset immunodeficiency in Thailand and Taiwan. The New England Journal of Medicine. 2012;367(8):725–734. doi: 10.1056/NEJMoa1111160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Höflich C., Sabat R., Rosseau S., et al. Naturally occurring anti-IFN-γ autoantibody and severe infections with Mycobacterium cheloneae and Burkholderia cocovenenans. Blood. 2004;103(2):673–675. doi: 10.1182/blood-2003-04-1065. [DOI] [PubMed] [Google Scholar]
- 3.Döffinger R., Helbert M. R., Barcenas-Morales G., et al. Autoantibodies to interferon-γ in a patient with selective susceptibility to mycobacterial infection and organ-specific autoimmunity. Clinical Infectious Diseases. 2004;38(1):e10–e14. doi: 10.1086/380453. [DOI] [PubMed] [Google Scholar]
- 4.Kampmann B., Hemingway C., Stephens A., et al. Acquired predisposition to mycobacterial disease due to autoantibodies to IFN-γ. The Journal of Clinical Investigation. 2005;115(9):2480–2488. doi: 10.1172/JCI19316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Patel S. Y., Ding L., Brown M. R., et al. Anti-IFN-γ autoantibodies in disseminated nontuberculous mycobacterial infections. Journal of Immunology. 2005;175(7):4769–4776. doi: 10.4049/jimmunol.175.7.4769. [DOI] [PubMed] [Google Scholar]
- 6.Tanaka Y., Hori T., Ito K., Fujita T., Ishikawa T., Uchiyama T. Disseminated Mycobacterium avium complex infection in a patient with autoantibody to interferon-gamma. Internal Medicine. 2007;46(13):1005–1009. doi: 10.2169/internalmedicine.46.6452. [DOI] [PubMed] [Google Scholar]
- 7.Koya T., Tsubata C., Kagamu H., et al. Anti-interferon-gamma autoantibody in a patient with disseminated Mycobacterium avium complex. Journal of Infection and Chemotherapy. 2009;15(2):118–122. doi: 10.1007/s10156-008-0662-8. [DOI] [PubMed] [Google Scholar]
- 8.Tang B. S. F., Chan J. F. W., Chen M., et al. Disseminated penicilliosis, recurrent bacteremic nontyphoidal salmonellosis, and burkholderiosis associated with acquired immunodeficiency due to autoantibody against gamma interferon. Clinical and Vaccine Immunology. 2010;17(7):1132–1138. doi: 10.1128/CVI.00053-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kampitak T., Suwanpimolkul G., Browne S., Suankratay C. Anti-interferon-γ autoantibody and opportunistic infections: case series and review of the literature. Infection. 2011;39(1):65–71. doi: 10.1007/s15010-010-0067-3. [DOI] [PubMed] [Google Scholar]
- 10.Lee W. I., Huang J. L., Wu T. S., et al. Patients with inhibitory and neutralizing auto-antibodies to interferon-γ resemble the sporadic adult-onset phenotype of mendelian susceptibility to mycobacterial disease (MSMD) lacking Bacille Calmette-Guerin (BCG)-induced diseases. Immunobiology. 2013;218(5):762–771. doi: 10.1016/j.imbio.2012.08.281. [DOI] [PubMed] [Google Scholar]
- 11.Ishii T., Tamura A., Matsui H., et al. Disseminated Mycobacterium avium complex infection in a patient carrying autoantibody to interferon-γ. Journal of Infection and Chemotherapy. 2013;19(6):1152–1157. doi: 10.1007/s10156-013-0572-2. [DOI] [PubMed] [Google Scholar]
- 12.Wongkulab P., Wipasa J., Chaiwarith R., Supparatpinyo K. Autoantibody to interferon-gamma associated with adult-onset immunodeficiency in non-HIV individuals in northern Thailand. PLoS One. 2013;8(9, article e76371) doi: 10.1371/journal.pone.0076371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Chaisathaphol T., Jitmuang A. Disseminated Mycobacterium avium and recurrent Salmonella group D infection in a patient with autoantibodies to interferon-gamma. The Southeast Asian Journal of Tropical Medicine and Public Health. 2013;44(3):460–467. [PubMed] [Google Scholar]
- 14.Suzuki K., Terada J., Sasaki Y., et al. Pulmonary Mycobacterium fortuitum infection with cervical lymphadenitis in a patient carrying autoantibodies to interferon-γ. Internal Medicine. 2014;53(12):1361–1364. doi: 10.2169/internalmedicine.53.1931. [DOI] [PubMed] [Google Scholar]
- 15.O’Connell E., Rosen L. B., LaRue R. W., et al. The first US domestic report of disseminated Mycobacterium avium complex and anti-interferon-γ autoantibodies. Journal of Clinical Immunology. 2014;34(8):928–932. doi: 10.1007/s10875-014-0073-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Nishimura T., Fujita-Suzuki Y., Yonemaru M., et al. Recurrence of disseminated Mycobacterium avium complex disease in a patient with anti-gamma interferon autoantibodies by reinfection. Journal of Clinical Microbiology. 2015;53(4):1436–1438. doi: 10.1128/JCM.03339-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Hase I., Morimoto K., Sakagami T., Kazumi Y., Ishii Y., van Ingen J. Disseminated Mycobacterium gordonae and Mycobacterium mantenii infection with elevated anti-IFN-γ neutralizing autoantibodies. Journal of Infection and Chemotherapy. 2015;21(6):468–472. doi: 10.1016/j.jiac.2015.02.003. [DOI] [PubMed] [Google Scholar]
- 18.Kobayashi T., Morino E., Takasaki J., Nagahara Y., Sugiyama H. Nontuberculous mycobacterial osteomyelitis in human immunodeficiency virus-negative patients: a case series. Japanese Journal of Infectious Diseases. 2016;69(2):149–150. doi: 10.7883/yoken.JJID.2015.100. [DOI] [PubMed] [Google Scholar]
- 19.Aoki A., Sakagami T., Yoshizawa K., et al. Clinical significance of interferon-γ neutralizing autoantibodies against disseminated nontuberculous mycobacterial disease. Clinical Infectious Diseases. 2018;66(8):1239–1245. doi: 10.1093/cid/cix996. [DOI] [PubMed] [Google Scholar]
- 20.Chi C. Y., Lin C. H., Ho M. W., et al. Clinical manifestations, course, and outcome of patients with neutralizing anti-interferon-γ autoantibodies and disseminated nontuberculous mycobacterial infections. Medicine. 2016;95(25, article e3927) doi: 10.1097/MD.0000000000003927. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Baerlecken N., Jacobs R., Stoll M., Schmidt R. E., Witte T. Recurrent, multifocal Mycobacterium avium-intercellulare infection in a patient with interferon-γ autoantibody. Clinical Infectious Diseases. 2009;49(7):e76–e78. doi: 10.1086/605581. [DOI] [PubMed] [Google Scholar]
- 22.Browne S. K., Zaman R., Sampaio E. P., et al. Anti-CD20 (rituximab) therapy for anti-IFN-γ autoantibody-associated nontuberculous mycobacterial infection. Blood. 2012;119(17):3933–3939. doi: 10.1182/blood-2011-12-395707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Czaja C. A., Merkel P. A., Chan E. D., et al. Rituximab as successful adjunct treatment in a patient with disseminated nontuberculous mycobacterial infection due to acquired anti-interferon-γ autoantibody. Clinical Infectious Diseases. 2014;58(6):e115–e118. doi: 10.1093/cid/cit809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Pruetpongpun N., Khawcharoenporn T., Damronglerd P., et al. Disseminated Talaromyces marneffei and Mycobacterium abscessus in a patient with anti-interferon-γ autoantibodies. Open Forum Infectious Diseases. 2016;3(2, article ofw093) doi: 10.1093/ofid/ofw093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Koizumi Y., Sakagami T., Nishiyama N., et al. Rituximab restores IFN-γ-STAT1 function and ameliorates disseminated Mycobacterium avium infection in a patient with anti-interferon-γ autoantibody. Journal of Clinical Immunology. 2017;37(7):644–649. doi: 10.1007/s10875-017-0425-3. [DOI] [PubMed] [Google Scholar]
- 26.Austin H. A., III, Klippel J. H., Balow J. E., et al. Therapy of lupus nephritis. Controlled trial of prednisone and cytotoxic drugs. The New England Journal of Medicine. 1986;314(10):614–619. doi: 10.1056/NEJM198603063141004. [DOI] [PubMed] [Google Scholar]
- 27.Houssiau F. A., Vasconcelos C., D'Cruz D., et al. Immunosuppressive therapy in lupus nephritis: the Euro-Lupus Nephritis Trial, a randomized trial of low-dose versus high-dose intravenous cyclophosphamide. Arthritis and Rheumatism. 2002;46(8):2121–2131. doi: 10.1002/art.10461. [DOI] [PubMed] [Google Scholar]
- 28.Houssiau F. A., Vasconcelos C., D'Cruz D., et al. The 10-year follow-up data of the Euro-Lupus Nephritis Trial comparing low-dose and high-dose intravenous cyclophosphamide. Annals of the Rheumatic Diseases. 2009;69(01):61–64. doi: 10.1136/ard.2008.102533. [DOI] [PubMed] [Google Scholar]
- 29.Urowitz M. B. Is “aggressive” therapy necessary for systemic lupus erythematosus? Rheumatic Diseases Clinics of North America. 1993;19(1):263–270. [PubMed] [Google Scholar]
- 30.Yee C., Gordon C., Dostal C., et al. EULAR randomized controlled trial of pulse cyclophosphamide and methylprednisolone versus continuous cyclophosphamide and prednisolone followed by azathioprine and prednisolone in lupus nephritis. Annals of the Rheumatic Diseases. 2004;63(5):525–529. doi: 10.1136/ard.2002.003574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Barile-Fabris L., Ariza-Andraca R., Olguín-Ortega L., et al. Controlled clinical trial of IV cyclophosphamide versus IV methylprednisolone in severe neurological manifestations in systemic lupus erythematosus. Annals of the Rheumatic Diseases. 2005;64(4):620–625. doi: 10.1136/ard.2004.025528. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Calguneri M., Ozbalkan Z., Ozturk M. A., Apras S., Ertenli A. I., Kiraz S. Intensified, intermittent, low-dose intravenous cyclophosphamide together with oral alternate-day steroid therapy in lupus nephritis (long-term outcome) Clinical Rheumatology. 2006;25(6):782–788. doi: 10.1007/s10067-006-0217-2. [DOI] [PubMed] [Google Scholar]
- 33.Giacomelli R., Valentini G., Salsano F., et al. Cyclophosphamide pulse regimen in the treatment of alveolitis in systemic sclerosis. The Journal of Rheumatology. 2002;29(4):731–736. [PubMed] [Google Scholar]
- 34.Latsi P. I., Wells A. U. Evaluation and management of alveolitis and interstitial lung disease in scleroderma. Current Opinion in Rheumatology. 2003;15(6):748–755. doi: 10.1097/00002281-200311000-00011. [DOI] [PubMed] [Google Scholar]
- 35.Simeón-Aznar C. P., Fonollosa-Plá V., Tolosa-Vilella C., et al. Intravenous cyclophosphamide pulse therapy in the treatment of systemic sclerosis-related interstitial lung disease: a long term study. The Open Respiratory Medicine Journal. 2008;2(1):39–45. doi: 10.2174/1874306400802010039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Harper L., Morgan M. D., Walsh M., et al. Pulse versus daily oral cyclophosphamide for induction of remission in ANCA-associated vasculitis: long-term follow-up. Annals of the Rheumatic Diseases. 2012;71(6):955–960. doi: 10.1136/annrheumdis-2011-200477. [DOI] [PubMed] [Google Scholar]
- 37.de Groot K., Harper L., Jayne D. R., et al. Pulse versus daily oral cyclophosphamide for induction of remission in antineutrophil cytoplasmic antibody-associated vasculitis: a randomized trial. Annals of Internal Medicine. 2009;150(10):670–680. doi: 10.7326/0003-4819-150-10-200905190-00004. [DOI] [PubMed] [Google Scholar]
- 38.Groot K. d., Adu D., Savage C. O. S. The value of pulse cyclophosphamide in ANCA-associated vasculitis: meta-analysis and critical review. Nephrology Dialysis Transplantation. 2001;16(10):2018–2027. doi: 10.1093/ndt/16.10.2018. [DOI] [PubMed] [Google Scholar]
- 39.Fleischli M. E., Valek R. H., Pandya A. G. Pulse intravenous cyclophosphamide therapy in pemphigus. Archives of Dermatology. 1999;135(1):57–61. doi: 10.1001/archderm.135.1.57. [DOI] [PubMed] [Google Scholar]
- 40.Wallace C. A., Sherry D. D. Trial of intravenous pulse cyclophosphamide and methylprednisolone in the treatment of severe systemic-onset juvenile rheumatoid arthritis. Arthritis and Rheumatism. 1997;40(10):1852–1855. doi: 10.1002/art.1780401019. [DOI] [PubMed] [Google Scholar]
- 41.Gobbini M. I., Smith M. E., Richert N. D., Frank J. A., McFarland H. F. Effect of open label pulse cyclophosphamide therapy on MRI measures of disease activity in five patients with refractory relapsing–remitting multiple sclerosis. Journal of Neuroimmunology. 1999;99(1):142–149. doi: 10.1016/S0165-5728(99)00039-9. [DOI] [PubMed] [Google Scholar]
- 42.Sanders J. S. F., Huitma M. G., Kallenberg C. G. M., Stegeman C. A. Prediction of relapses in PR3-ANCA-associated vasculitis by assessing responses of ANCA titres to treatment. Rheumatology. 2006;45(6):724–729. doi: 10.1093/rheumatology/kei272. [DOI] [PubMed] [Google Scholar]
- 43.Sistigu A., Viaud S., Chaput N., Bracci L., Proietti E., Zitvogel L. Immunomodulatory effects of cyclophosphamide and implementations for vaccine design. Seminars in Immunopathology. 2011;33(4):369–383. doi: 10.1007/s00281-011-0245-0. [DOI] [PubMed] [Google Scholar]
- 44.McCune W. J., Clowse M. B. General principles of the use of cyclophosphamide in rheumatic diseases. In: Post T. W., editor. UpToDate. Waltham, MA, USA: UpToDate; 2016. [Google Scholar]
- 45.Chi C. Y., Chu C. C., Liu J. P., et al. Anti-IFN-γ autoantibodies in adults with disseminated nontuberculous mycobacterial infections are associated with HLA-DRB1∗16:02 and HLA-DQB1∗05:02 and the reactivation of latent varicella-zoster virus infection. Blood. 2013;121(8):1357–1366. doi: 10.1182/blood-2012-08-452482. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The data used to support the findings of this study are included within the article.