Abstract
The normal function of ovaries, along with the secretion of sex hormones, is among the most important endocrine factors that maintain the female sexual characteristics and promote follicular development and ovulation. Premature ovarian insufficiency (POI) is a common cause in the etiology of female infertility. It is defined as the loss of ovarian function before the age of 40. The characteristics of POI are menstrual disorders, including amenorrhea and delayed menstruation, accompanied by a raised gonadotrophin level and decreased estradiol level. Inflammatory aging is a new concept in the research field of aging. It refers to a chronic and low-degree proinflammatory state which occurs with increasing age. Inflammatory aging is closely associated with multiple diseases, as excessive inflammation can induce the inflammatory lesions in certain organs of the body. In recent years, studies have shown that inflammatory aging plays a significant role in the pathogenesis of POI. This paper begins with the pathogenesis of inflammatory aging and summarizes the relationship between inflammatory aging and premature ovarian insufficiency in a comprehensive way, as well as discussing the new diagnostic and therapeutic methods of POI.
1. Introduction
Due to environmental pollution, huge living and working pressures, and other factors, female infertility and premature ovarian insufficiency (POI) have become global issues receiving great attention. POI is a disease defined as the cessation of ovarian function with decreased estrogen levels and elevated gonadotropin levels before the age of 40 [1]. Previous studies have shown that the incidence of POI in women before the age of 40 is approximately 1%, and its incidence in women before 30 is 1‰. Moreover, approximately 10% to 28% of women experienced primary amenorrhea and approximately 4% to 18% of women exhibited secondary amenorrhea [2, 3]. In recent years, studies have shown that inflammatory aging is closely related to POI. Additionally, ovarian biopsy from patients with POI have shown lymphocytic infiltration and other ovarian immune responses [4–8]. Therefore, the role inflammation plays in ovarian function degeneration, as well as the therapeutic methods to delay ovarian aging and improve ovarian function, are currently heavily researched topics in the field of reproduction. The paper will discuss the mechanism of inflammatory aging and its relationship with POI.
2. Mechanisms of Inflamm-Aging
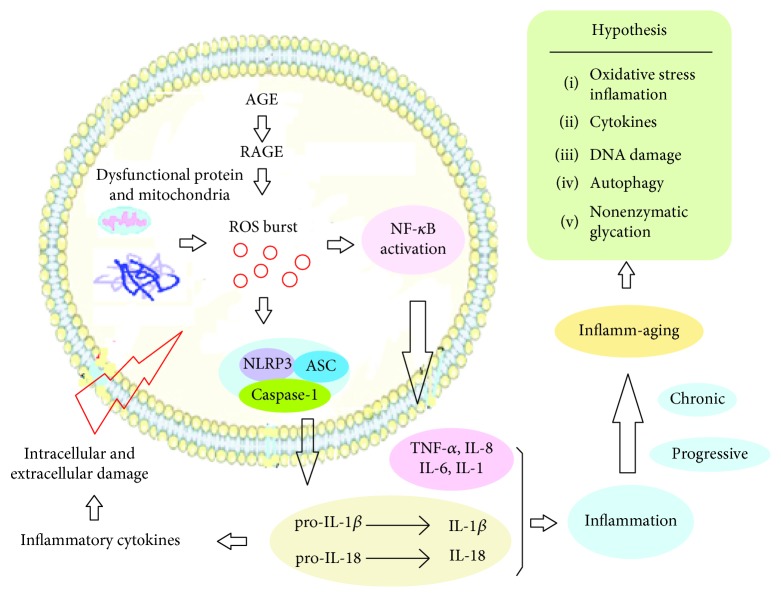
The natural aging of the body is a long and complex biological process, which results from the interaction of intrinsic and extrinsic factors during the body's degeneration period. Aging is characterized by the degeneration of structures; the imbalance of the internal environment; the decline of function; and the loss of adaptability, resilience, and resistance [9]. With the rapid development of science, the study of aging is coming into a new research stage; however, the definite mechanism of aging has not been thoroughly elucidated to date. In recent years, an increasing number of studies have found that inflammation is associated with the occurrence of aging; as in the process of aging, the tissues and organs of the body are accompanied by a chronic, progressive proinflammatory state [10–12]. Inflamm-aging [13] was a term first coined by Franceschi et al. in 2000. At present, the mechanism of inflammatory aging can be illustrated with the hypothesis presented in Figure 1.
Figure 1.
The mechanisms of inflamm-aging. In the process of aging, with the activation of inflammatory factors, the body appears to be in a chronic, progressively elevated proinflammatory state called inflammatory aging. ROS in the body is increased due to several factors. It causes oxidative stress and a series of inflammatory reactions activated by NLPR3 and NF-κB. It is now summarized as follows: oxidative stress inflammation, cytokines, DNA damage, autophagy, and nonenzymatic glycation.
2.1. Oxidative Stress and Inflammation
Oxidative stress refers to an imbalance between oxidation and antioxidation in vivo, which tends to be oxidative and leads to neutrophil inflammatory infiltration. Oxidative stress is caused by excessive reactive oxygen species (including oxygen free radicals) and plays an important role in the inflammatory aging of the body caused by chronic inflammation [14, 15]. In the 1990s, Sohal and Weindruch [16] noted the defects of the free radical theory and proposed the concept of oxidative stress based on the close relationship between oxidative stress and aging. Ottaviani and Franceschi [17] investigated a series of different species, ranging from invertebrates to humans, and found that immune-stress-inflammation formed a defense network of the body, confirming that stress was one of the causes of inflammatory aging. In addition, antioxidants have been successfully applied to reduce oxidative stress damage and improve the longevity of individuals which also confirm the hypothesis of oxidative stress inflammation [18, 19].
2.2. Proinflammatory Cytokines and Aging
Excessive expression of the inflammatory factors will cause a high proinflammatory state in the body, which is an important element in inflammatory aging. A large number of studies have shown that the serum levels of inflammatory factors, such as IL-6, IL-8, TNF-α, and PGE2, are significantly higher in aging organs [20–22]. Salvioli et al. [23] examined normal individuals of all ages and patients with diseases related to aging (such as Alzheimer's disease and Type 2 diabetes) and determined that elevated levels of proinflammatory cytokines play an important role in aging.
2.3. DNA Damage
The DNA repair mechanism of the normal body can repair damaged DNA, but with increasing age, DNA damage continues to accumulate, eventually leading to cell death. Bonafe et al.'s study proved that the inflammatory aging of the body is due to DNA damage; with stem cells and stromal fibroblasts differentiating, the overexpression of proinflammatory cytokines is triggered, which disintegrates the multishell cytokine network [24].
2.4. Autophagy
Professor Christian de Duve proposed the concept of autophagy in the 1960s, and accumulated evidence suggests that autophagy is prevalent in eukaryotic cells and plays a vital role in maintaining cell homeostasis and delaying aging [18, 25, 26]. Salminen et al. [27] found that in the course of aging, the autophagic cleansing capacity declines gradually and dysfunctional protein and mitochondria accumulate, leading to an increased level of reactive oxygen species (ROS) and oxidative stress. ROS activates NOD-like receptor 3 (NLRP3) and causes a series of inflammatory reactions, such as the increased secretion levels of IL-1β and IL-18. In turn, these cytokines also accelerate the aging process through inflammation caused by inhibiting autophagy [28].
2.5. Glycation
Saccharification is one of the endogenous aging mechanisms. Advanced glycosylation end products (AGEs) are the end products of nonenzymatic glycosylation that accumulate in the body with increasing age [29]. The glycosylation aging theory accepted by many scholars argues that glycosylation will cause the crosslinking damage of proteins, which can convert proteins with normal structures into abnormal aging ones. Studies have shown that the AGE receptor RAGE (receptor for end products of advanced glycation) regulates inflammation, apoptosis, autophagy, senescence, and other important cellular processes by combining and transducing various inflammatory gametes, such as AGEs, S100 calmodulin, and high mobility group box-1 protein (HMGB.1) [30, 31]. Nakashima et al. [32] noted that when comparing the number of glycosylated protein and membrane protein on the erythrocyte membrane which were labelled with fluorescent substances, the fluorescence degree of erythrocytes from older individuals were significantly higher than those from younger individuals. Nonenzymatic glycosylation products can also accelerate body aging by a range of changes, such as the enhancement of lipid peroxidation.
3. Inflammatory Aging and Premature Ovarian Insufficiency (POI)
According to the new guidelines of the European Society of Human Reproduction and Embryology, POI is defined as the loss of ovarian activity in a woman before the age of 40 with elevated gonadotrophin and decreased estrogen [3, 33, 34]. The diagnosis of POI requires simultaneous abnormal menstruation and biochemical abnormalities: oligomenorrhea/amenorrhea for at least 4 months and an elevated FSH level > 25 IU/L on two occasions (4 weeks apart) [35]. The patients include women younger than 40 years (which includes Turner Syndrome patients) and women older than 40 years, but older patients must be affected before the age of 40 [1, 36]. The main clinical manifestation is amenorrhea, which is also accompanied by hot flashes, sweating, loss of libido, and other menopausal symptoms. Severe cases can even lead to infertility, greatly affecting the female reproductive function [37].The etiology and pathogenesis of POI are very complex. A mass of exogenous factors are involved, such as surgery, drugs, and the environment, as well as endogenous factors, such as chromosomal linkage defects [38, 39], autoimmune reactions [40, 41], psychological stress [42], genetic predisposition [1, 43], and congenital enzyme deficiencies. With plenty of domestic and foreign research confirming that TNF-α and IL-6 may play a role in ovarian function [44, 45], it follows that inflammatory factors may be a vital cause of POI. Thus, this review will discuss the relationship between inflammatory aging and POI from the following perspectives.
3.1. Inflammatory Cytokines and Anti-Inflammatory Factors Cause Ovarian Change
In recent years, the role that inflammatory aging plays in ovarian disease has raised great concern, in which follicular rupture is considered as an inflammatory response, and IL-1 and TNF-α are the major cytokines involved in this process.
It has been reported that the abnormal performance of xanthogranulomatous inflammation in the female genital tract is a POI [46]. This suggests that inflammatory aging may be one of the causes of POI. Th1 cell- (a type of T helper cells) mediated immune response is often associated with the inflammatory response. The targeting action of inhibin-alpha on the experimental autoimmune ovarian inflammation is initiated by CD4 (+) Th1 T cells, by stimulating B cells to produce inhibin-alpha neutralizing Abs, which directly mediates POI and transfers the disease to the naive receptor [47].
Many molecules involved in the immune response and inflammatory response are regulated by nuclear factor-κ gene binding (NF-κB), which includes TNF-α, IL-1β, IL-2, IL-6, and colony stimulating factor. In addition, zinc finger protein A20 (tumor necrosis factor alpha-induced protein 3), heme oxygenase (HO-1), and many other anti-inflammatory factors are also regulated by NF-κB. NF-κB is a transcription factor that can turn on the genes related to the inflammation and immune responses. Furthermore, this factor can be activated when the proinflammatory cytokines are present. In this process, there are expressions of inflammatory markers, such as IL-6 and IL-8, and the level of anti-inflammatory factor IL-10 mRNA decreased [48]. Scientists detected NF-κB activity in the mouse brain with aging progression, which indicated that the protein has little activity in the hypothalamus of young mice, and with mouse aging, the protein becomes increasingly active.
Thus, elevated levels of inflammatory cytokines and decreased levels of anti-inflammatory cytokines play a critical role in POI (Figure 2).
Figure 2.
Regulation of proinflammatory cytokines and anti-inflammatory cytokines on premature ovarian insufficiency. Proinflammatory cytokines and anti-inflammatory cytokines maintain a dynamic balance in the normal body. Pathological inflammation in premature ovarian insufficiency is caused by an imbalance of the inflammatory cytokine network.
3.2. Changes of Inflammatory Factors in Ovarian Senescence and Reversal
Naz et al. [44] detected the levels of inflammatory factors in patients with POI and normal women using ELISA, and they found that the value of TNF-α in patients with POI was lower than that in normal women. Consistent with this result, Wang et al. [49] demonstrated that the level of antizona pellucid antibodies (AzpAb) in POI patients was significantly higher than that in normal controls, and the TNF-α and IL-2 levels decreased significantly, whereas INF-γ increased significantly. The levels of TNF-α and IL-2 in the serum of the POI group were significantly lower than those of the normal group, probably as TNF-α was secreted by lymphocytes and granulocytes, while POI patients experience a decreased serum level of the associated inflammatory factors, due to ovarian tissue atrophy and granulocyte reduction. After Sundaresan et al. [50] studied birds, they found that the expression levels of cytokines (IL-1β, IL-6, IL-10, and TGF-β2) and chemokines are elevated in POI, which confirmed the relationship between POI and inflammatory aging.
Accordingly, the level of inflammatory factors should decrease after repairing the damaged ovarian function. Recently, studies have shown that synovial mesenchymal stem cells (SMSCs) may play a role in restoring the damaged ovary in the ovarian follicular microenvironment [51]. When SMSCs were used for the treatment of mice, RT-PCR results showed that the expression level of proinflammatory cytokines, such as TNF-α, TGF-β, IL-8, IL-6, IL-1, and IFNγ, is significantly lower than that of the untreated control group of ovaries. When POI occurs, the expression level of certain inflammatory factors increases, while when the damaged ovarian function is repaired, the level of inflammatory factors decreases accordingly, which is sufficient to show that inflammatory aging and POI are closely related.
3.3. The Cure of Premature Ovarian Insufficiency by the Treatment of Chronic Inflammation
It is evident that inflammatory aging plays an important part in the pathogenesis of POI, which suggests that we can prevent ovarian insufficiency through anti-inflammation. Said et al. [48] found that resveratrol restores ovarian function by increasing serum levels of the anti-Miller hormone (AMH) and reducing ovarian inflammation, mainly through upregulating the expression of peroxisome proliferator-activated receptors and SIRT1 (sirtuin-1) to inhibit NF-κB-induced inflammatory cytokines. These findings indicate the expression of inflammatory factors such as IL-6 and IL-8. Professor Cai's research team injected mice with GnRH, a hormone produced by the hypothalamus, and mice showed a reduction in signs of aging, which indicated that the inflammatory protein nuclear factor (NF-κB) and the upstream activator IKK-β (IκB kinase) inhibited GnRH expression [52]. The antiaging mechanism of GnRH is the hormonal stimulation of the entire cerebral nerve, thereby causing cascading benefits to the entire body. This mechanism may be a new way to regulate aging signals: using anti-inflammatory compounds to treat the brain, which may be able to slow down ovarian aging-related degradation.
He et al. [53] studied the effects and mechanisms of ginsenoside Rg1 acting on the POI induced by D-galactose (D-gal) and found that Rg1 can enhance anti-inflammation and antioxidation. It also reduces the expression of senescence signaling pathway protein, which can reduce damage to the ovary and improve the fertility of POI mice. This study suggests that controlling the development of inflammatory aging may be one of the methods to cure POI. Currently, many inflammatory markers have been identified, such as the plasma tumor necrosis factor, interleukin family, and plasma inflammatory protein which can be used to monitor the ovarian function and cure POI.
However, since there are many other reasons for the occurrence of POI, the pathogenesis is unclear. What anti-inflammatory treatment or other therapeutic methods can make a more remarkable effect? How efficacious is anti-inflammatory treatment on individuals with genetic susceptibility to POI? Can POI be prevented by anti-inflammatory treatment? All these questions have to be answered by further studies.
4. Conclusions
POI not only leads to the decline (or even loss) of the female reproductive endocrine function but may also affect women's psychological conditions and the function of other organs. Achievements have been made by experts in the area of inflammation and reproduction, which have improved our understanding of the etiology and pathogenesis of POI, as well as provided useful information for protecting ovarian function, delaying aging, and improving female fertility. However, the regulatory mechanisms of inflammation-mediated aging are still unclear, and further research is needed to address several issues, especially the mechanisms underlying some antiaging substances, like GnRH.
In summary, future research should focus on addressing several problems: (1) How can the specific regulatory mechanism of inflammatory aging be determined? (2) How can the regulation network and key nodes of POI caused by inflammatory aging be determined? (3) How can inflammatory aging be prevented from happening? How can the therapeutic methods be made specific to treat chronic inflammation and prevent ovarian premature aging? (4) Can the genetic susceptibility of POI be treated by anti-inflammatory treatment? (5) How effective are anti-inflammatory agents in the prevention of POI? Are there any adverse effects?
With the development of reproductive medicine, the pathogenesis and mechanism of POI will be better understood. Furthermore, the prevention of POI and restoration of the female reproductive function will become hot topics for researchers both domestic and foreign. In any event, anti-inflammatory aging will become an important strategy in the prevention and treatment of POI.
Acknowledgments
The authors would like to thank Dr. Yuehui Zheng for article mentoring. We also thank the members in our lab who participated in this article. This work was supported by the National Nature Science Foundation of China (Nos. 81671455 and 81360100), the Natural Science Foundation of Jiangxi Province (Nos. 20161BAB205207 and 20161BAB205213), and the Graduate Innovation Special Fund of Nanchang University (No. CX2017229).
Conflicts of Interest
The authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors' Contributions
Yaoqi Huang was involved in the initial literature search, in the acquisition and interpretation of data, and in the design of the first draft of the article. Chuan Hu, Haifeng Ye, Ruichen Luo, and Xinxin Fu revised the synthesis and made the final revisions. Jian Huang, Xiaoyan Li, and Weiyun Chen were involved in the review of the manuscript and gave some advice on some concerns. Yuehui Zheng approved the final manuscript. All authors read and approved the final manuscript.
References
- 1.Qin C., Chen Y., Lin Q., Yao J., Wu W., Xie J. The significance of polymorphism and expression of oestrogen metabolism-related genes in Chinese women with premature ovarian insufficiency. Reproductive Biomedicine Online. 2017;35(5):609–615. doi: 10.1016/j.rbmo.2017.07.007. [DOI] [PubMed] [Google Scholar]
- 2.Laml T., Schulz-Lobmeyr I., Obruca A., Huber J. C., Hartmann B. W. Premature ovarian failure: etiology and prospects. Gynecological Endocrinology. 2000;14(4):292–302. doi: 10.3109/09513590009167696. [DOI] [PubMed] [Google Scholar]
- 3.Webber L., Davies M., Anderson R., et al. ESHRE guideline: management of women with premature ovarian insufficiency. Human Reproduction. 2016;31(5):926–937. doi: 10.1093/humrep/dew027. [DOI] [PubMed] [Google Scholar]
- 4.Sen A., Kushnir V. A., Barad D. H., Gleicher N. Endocrine autoimmune diseases and female infertility. Nature Reviews Endocrinology. 2014;10(1):37–50. doi: 10.1038/nrendo.2013.212. [DOI] [PubMed] [Google Scholar]
- 5.Sammaritano L. R. Menopause in patients with autoimmune diseases. Autoimmunity Reviews. 2012;11(6-7):A430–A436. doi: 10.1016/j.autrev.2011.11.006. [DOI] [PubMed] [Google Scholar]
- 6.Petrikova J., Lazurova I. Ovarian failure and polycystic ovary syndrome. Autoimmunity Reviews. 2012;11(6-7):A471–A478. doi: 10.1016/j.autrev.2011.11.010. [DOI] [PubMed] [Google Scholar]
- 7.Carp H. J. A., Selmi C., Shoenfeld Y. The autoimmune bases of infertility and pregnancy loss. Journal of Autoimmunity. 2012;38(2-3):J266–J274. doi: 10.1016/j.jaut.2011.11.016. [DOI] [PubMed] [Google Scholar]
- 8.Reato G., Morlin L., Chen S., et al. Premature ovarian failure in patients with autoimmune Addison's disease: clinical, genetic, and immunological evaluation. The Journal of Clinical Endocrinology and Metabolism. 2011;96(8):E1255–E1261. doi: 10.1210/jc.2011-0414. [DOI] [PubMed] [Google Scholar]
- 9.Vohra B. P., Sharma S. P., Kansal V. K. Age-dependent variations in mitochondrial and cytosolic antioxidant enzymes and lipid peroxidation in different regions of central nervous system of guinea pigs. Indian Journal of Biochemistry & Biophysics. 2001;38(5):321–326. [PubMed] [Google Scholar]
- 10.Prattichizzo F., Micolucci L., Cricca M., et al. Exosome-based immunomodulation during aging: a nano-perspective on inflamm-aging. Mechanisms of Ageing and Development. 2017;168:44–53. doi: 10.1016/j.mad.2017.02.008. [DOI] [PubMed] [Google Scholar]
- 11.Thevaranjan N., Puchta A., Schulz C., et al. Age-associated microbial dysbiosis promotes intestinal permeability, systemic inflammation, and macrophage dysfunction. Cell Host & Microbe. 2017;21(4):455–466.e4. doi: 10.1016/j.chom.2017.03.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Salminen A., Huuskonen J., Ojala J., Kauppinen A., Kaarniranta K., Suuronen T. Activation of innate immunity system during aging: NF-κB signaling is the molecular culprit of inflamm-aging. Ageing Research Reviews. 2008;7(2):83–105. doi: 10.1016/j.arr.2007.09.002. [DOI] [PubMed] [Google Scholar]
- 13.Franceschi C., Bonafè M., Valensin S., et al. Inflamm-aging: an evolutionary perspective on immunosenescence. Annals of the New York Academy of Sciences. 2000;908(1):244–254. doi: 10.1111/j.1749-6632.2000.tb06651.x. [DOI] [PubMed] [Google Scholar]
- 14.Vida C., de Toda I. M., Cruces J., Garrido A., Gonzalez-Sanchez M., de la Fuente M. Role of macrophages in age-related oxidative stress and lipofuscin accumulation in mice. Redox Biology. 2017;12:423–437. doi: 10.1016/j.redox.2017.03.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Picca A., Lezza A. M. S., Leeuwenburgh C., et al. Fueling inflamm-aging through mitochondrial dysfunction: mechanisms and molecular targets. International Journal of Molecular Sciences. 2017;18(5) doi: 10.3390/ijms18050933. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Sohal R. S., Weindruch R. Oxidative stress, caloric restriction, and aging. Science. 1996;273(5271):59–63. doi: 10.1126/science.273.5271.59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Ottaviani E., Franceschi C. The neuroimmunology of stress from invertebrates to man. Progress in Neurobiology. 1996;48(4-5):421–440. doi: 10.1016/0301-0082(95)00049-6. [DOI] [PubMed] [Google Scholar]
- 18.Xia S., Zhang X., Zheng S., et al. An update on inflamm-aging: mechanisms, prevention, and treatment. Journal of Immunology Research. 2016;2016:12. doi: 10.1155/2016/8426874.8426874 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Pangrazzi L., Meryk A., Naismith E., et al. “Inflamm-aging” influences immune cell survival factors in human bone marrow. European Journal of Immunology. 2017;47(3):481–492. doi: 10.1002/eji.201646570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Mei C., Zheng F. Chronic inflammation potentiates kidney aging. Seminars in Nephrology. 2009;29(6):555–568. doi: 10.1016/j.semnephrol.2009.07.002. [DOI] [PubMed] [Google Scholar]
- 21.Freund A., Orjalo A. V., Desprez P. Y., Campisi J. Inflammatory networks during cellular senescence: causes and consequences. Trends in Molecular Medicine. 2010;16(5):238–246. doi: 10.1016/j.molmed.2010.03.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Bruunsgaard H., Andersen-Ranberg K., Hjelmborg J. . B., Pedersen B. K., Jeune B. Elevated levels of tumor necrosis factor alpha and mortality in centenarians. The American Journal of Medicine. 2003;115(4):278–283. doi: 10.1016/S0002-9343(03)00329-2. [DOI] [PubMed] [Google Scholar]
- 23.Salvioli S., Capri M., Valensin S., et al. Inflamm-aging, cytokines and aging: state of the art, new hypotheses on the role of mitochondria and new perspectives from systems biology. Current Pharmaceutical Design. 2006;12(24):3161–3171. doi: 10.2174/138161206777947470. [DOI] [PubMed] [Google Scholar]
- 24.Bonafe M., Storci G., Franceschi C. Inflamm-aging of the stem cell niche: breast cancer as a paradigmatic example: breakdown of the multi-shell cytokine network fuels cancer in aged people. BioEssays. 2012;34(1):40–49. doi: 10.1002/bies.201100104. [DOI] [PubMed] [Google Scholar]
- 25.Nakatogawa H., Ohsumi Y. Autophagy: close contact keeps out the uninvited. Current Biology. 2014;24(12):R560–R562. doi: 10.1016/j.cub.2014.05.013. [DOI] [PubMed] [Google Scholar]
- 26.Mizushima N., Yoshimori T., Ohsumi Y. The role of Atg proteins in autophagosome formation. Annual Review of Cell and Developmental Biology. 2011;27(1):107–132. doi: 10.1146/annurev-cellbio-092910-154005. [DOI] [PubMed] [Google Scholar]
- 27.Salminen A., Kaarniranta K., Kauppinen A. Inflammaging: disturbed interplay between autophagy and inflammasomes. Aging. 2012;4(3):166–175. doi: 10.18632/aging.100444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Rippo M. R., Olivieri F., Monsurrò V., Prattichizzo F., Albertini M. C., Procopio A. D. MitomiRs in human inflamm-aging: a hypothesis involving miR-181a, miR-34a and miR-146a. Experimental Gerontology. 2014;56:154–163. doi: 10.1016/j.exger.2014.03.002. [DOI] [PubMed] [Google Scholar]
- 29.Schmidt F. N., Zimmermann E. A., Campbell G. M., et al. Assessment of collagen quality associated with non-enzymatic cross-links in human bone using Fourier-transform infrared imaging. Bone. 2017;97:243–251. doi: 10.1016/j.bone.2017.01.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Roca F., Grossin N., Chassagne P., Puisieux F., Boulanger E. Glycation: the angiogenic paradox in aging and age-related disorders and diseases. Ageing Research Reviews. 2014;15:146–160. doi: 10.1016/j.arr.2014.03.009. [DOI] [PubMed] [Google Scholar]
- 31.Watanabe M., Toyomura T., Wake H., et al. Advanced glycation end products attenuate the function of tumor necrosis factor-like weak inducer of apoptosis to regulate the inflammatory response. Molecular and Cellular Biochemistry. 2017;434(1-2):153–162. doi: 10.1007/s11010-017-3045-6. [DOI] [PubMed] [Google Scholar]
- 32.Nakashima K., Nishizaki O., Andoh Y. Acceleration of hemoglobin glycation with aging. Clinica Chimica Acta. 1993;215(1):111–118. doi: 10.1016/0009-8981(93)90254-2. [DOI] [PubMed] [Google Scholar]
- 33.Kumar R., Alwani M., Kosta S., Kaur R., Agarwal S. BMP15 and GDF9 gene mutations in premature ovarian failure. Journal of Reproduction & Infertility. 2017;18(1):185–189. [PMC free article] [PubMed] [Google Scholar]
- 34.Komorowska B. Autoimmune premature ovarian failure. Menopausal Review. 2016;4(4):210–214. doi: 10.5114/pm.2016.65666. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Bricaire L., Laroche E., Bourcigaux N., Donadille B., Christin-Maitre S. Insuffisances ovariennes prématurées. Presse Médicale. 2013;42(11):1500–1507. doi: 10.1016/j.lpm.2013.04.018. [DOI] [PubMed] [Google Scholar]
- 36.Ghaddhab C., Morin C., Brunel-Guitton C., Mitchell G. A., van Vliet G., Huot C. Premature ovarian failure in French Canadian Leigh syndrome. The Journal of Pediatrics. 2017;184:227–229.e1. doi: 10.1016/j.jpeds.2017.02.008. [DOI] [PubMed] [Google Scholar]
- 37.Kovanci E., Schutt A. K. Premature ovarian failure: clinical presentation and treatment. Obstetrics and Gynecology Clinics of North America. 2015;42(1):153–161. doi: 10.1016/j.ogc.2014.10.004. [DOI] [PubMed] [Google Scholar]
- 38.Rossetti R., Ferrari I., Bonomi M., Persani L. Genetics of primary ovarian insufficiency. Clinical Genetics. 2017;91(2):183–198. doi: 10.1111/cge.12921. [DOI] [PubMed] [Google Scholar]
- 39.Chapman C., Cree L., Shelling A. N. The genetics of premature ovarian failure: current perspectives. International Journal of Women's Health. 2015;7:799–810. doi: 10.2147/IJWH.S64024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Conway G. S. Clinical manifestations of genetic defects affecting gonadotrophins and their receptors. Clinical Endocrinology. 1996;45(6):657–663. doi: 10.1046/j.1365-2265.1996.8680879.x. [DOI] [PubMed] [Google Scholar]
- 41.Ye H., Li X., Zheng T., et al. The effect of the immune system on ovarian function and features of ovarian germline stem cells. Springerplus. 2016;5(1):p. 990. doi: 10.1186/s40064-016-2390-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Wang X. F., Zhang L., Wu Q. H., Min J. X., Ma N., Luo L. C. Biological mechanisms of premature ovarian failure caused by psychological stress based on support vector regression. International Journal of Clinical and Experimental Medicine. 2015;8(11):21393–21399. [PMC free article] [PubMed] [Google Scholar]
- 43.Bouali N., Francou B., Bouligand J., et al. New MCM8 mutation associated with premature ovarian insufficiency and chromosomal instability in a highly consanguineous Tunisian family. Fertility and Sterility. 2017;108(4):694–702. doi: 10.1016/j.fertnstert.2017.07.015. [DOI] [PubMed] [Google Scholar]
- 44.Naz R. K., Thurston D., Santoro N. Circulating tumor necrosis factor (TNF)-α in normally cycling women and patients with premature ovarian failure and polycystic ovaries. American Journal of Reproductive Immunology. 1995;34(3):170–175. doi: 10.1111/j.1600-0897.1995.tb00934.x. [DOI] [PubMed] [Google Scholar]
- 45.RVS V., Téllez V. S., CJC H., RMJ O., OME C., Reyes F. A. Serum levels of IL-1beta, IL-6 and TNF-alpha in infertile patients with ovarian dysfunction. Ginecologia y obstetricia de Mexico. 2005;73:604–610. [PubMed] [Google Scholar]
- 46.Singh N., Dadhwal V., Sharma K. A., Mittal S. Xanthogranulomatous inflammation: a rare cause of premature ovarian failure. Archives of Gynecology and Obstetrics. 2009;279(5):729–731. doi: 10.1007/s00404-008-0791-4. [DOI] [PubMed] [Google Scholar]
- 47.Altuntas C. Z., Johnson J. M., Tuohy V. K. Autoimmune targeted disruption of the pituitary-ovarian axis causes premature ovarian failure. Journal of Immunology. 2006;177(3):1988–1996. doi: 10.4049/jimmunol.177.3.1988. [DOI] [PubMed] [Google Scholar]
- 48.Said R. S., el-Demerdash E., Nada A. S., Kamal M. M. Resveratrol inhibits inflammatory signaling implicated in ionizing radiation-induced premature ovarian failure through antagonistic crosstalk between silencing information regulator 1 (SIRT1) and poly(ADP-ribose) polymerase 1 (PARP-1) Biochemical Pharmacology. 2016;103:140–150. doi: 10.1016/j.bcp.2016.01.019. [DOI] [PubMed] [Google Scholar]
- 49.Wang Y. F., Han L. Y., Ou R. Q. Analysis of anti-zona pellucida antibody and tumor necrosis factor-α, γ-interferon and interleukin-2 in sera from patients with premature ovarian failure. Journal of Reproductive Medicine. 2003;12:47–50. [Google Scholar]
- 50.Sundaresan N. R., Saxena V. K., Sastry K. V. H., et al. Cytokines and chemokines in postovulatory follicle regression of domestic chicken (Gallus gallus domesticus) Developmental & Comparative Immunology. 2008;32(3):253–264. doi: 10.1016/j.dci.2007.05.011. [DOI] [PubMed] [Google Scholar]
- 51.Lai D., Wang F., Dong Z., Zhang Q. Skin-derived mesenchymal stem cells help restore function to ovaries in a premature ovarian failure mouse model. PLoS One. 2014;9(5, article e98749) doi: 10.1371/journal.pone.0098749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Zhang G., Li J., Purkayastha S., et al. Hypothalamic programming of systemic ageing involving IKK-β, NF-κB and GnRH. Nature. 2013;497(7448):211–216. doi: 10.1038/nature12143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.He L., Ling L., Wei T., Wang Y., Xiong Z. Ginsenoside Rg1 improves fertility and reduces ovarian pathological damages in premature ovarian failure model of mice. Experimental Biology and Medicine. 2017;242(7):683–691. doi: 10.1177/1535370217693323. [DOI] [PMC free article] [PubMed] [Google Scholar]