Key messages.
What is already known about this subject?
Mammalian/mechanistic target of rapamycin (mTOR) dysregulation plays a key role in lymphocyte function and immunopathology.
What does this study add?
This is the first study demonstrating increased mTOR activity in T and B lymphocytes in salivary glands of patients with Sjögren’s syndrome, which is associated with B cell hyperactivity.
How might this impact on clinical practice?
mTOR targeting prevents activation of T and B cells of patients with Sjögren’s and thus may be pursued as a clinical target.
Introduction
Primary Sjögren’s syndrome (pSS) is a systemic autoimmune disorder characterised by lymphocytic infiltration and dysfunction of exocrine glands. The lymphocytic infiltrates in exocrine glands mainly consist of Th cells and B cells. A hallmark feature of pSS is B cell hyperactivity, including formation of autoantibodies, elevated serum IgG levels, increased numbers of B cells and IgG+/IgM+ plasma cells in salivary glands, and formation of germinal centre-like structures in the salivary glands associated with an increased risk of lymphoma development.1 2 Th cells and in particular T follicular helper (Tfh) cells play an important role in activation of B cells and formation of germinal centre-like structures.1
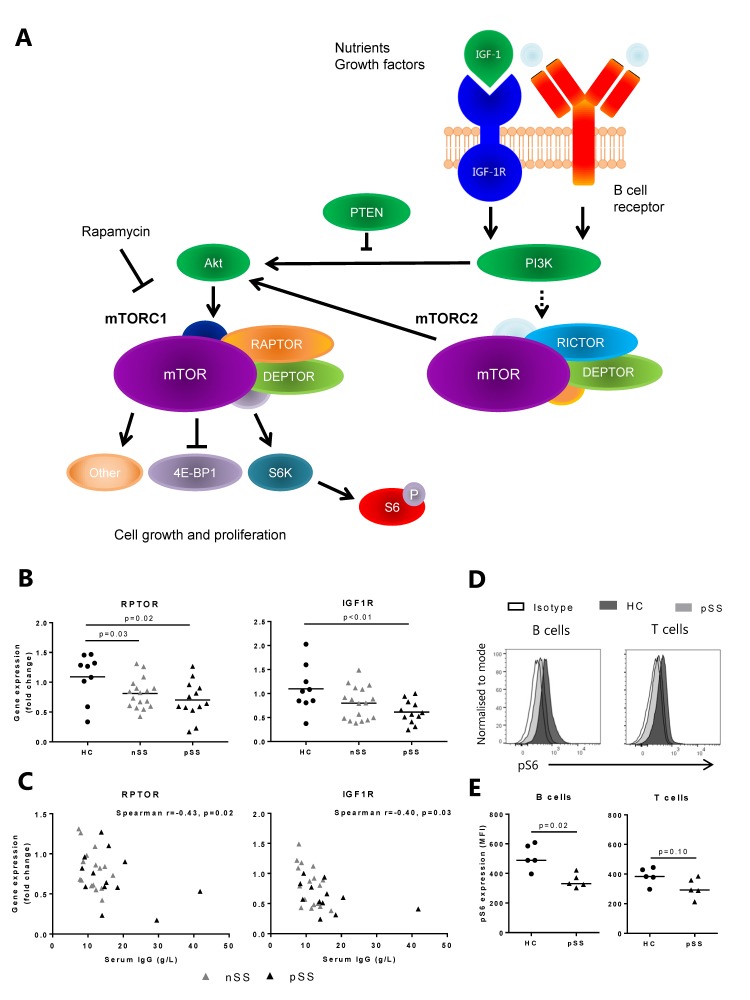
The mammalian/mechanistic target of rapamycin (mTOR) pathway is essential for growth, survival and proliferation of T and B cells and integrates multiple signals from the immune microenvironment, including growth factors, nutrients, and T cell receptor (TCR)/B cell receptor (BCR) engagement.3 4 The serine/threonine kinase mTOR is the catalytic subunit of two distinct complexes: mTOR complex 1 (mTORC1) and mTOR complex 2 (mTORC2), characterised by the incorporation of the proteins Raptor and Rictor, respectively. mTORC1 is generally described to play a role in protein translation, cell growth, proliferation and metabolism, whereas mTORC2 regulates metabolism, cell survival, rearrangement of the cytoskeleton and cell cycle progression (figure 1A).4
Figure 1.
Decreased mammalian/mechanistic target of rapamycin (mTOR) pathway-related gene and protein expression in circulating B cells from patients with pSS. (A) The mTOR pathway is activated by growth factor and B cell receptor signalling and consists of two complexes: mTOR complex 1 (mTORC1) and mTOR complex 2 (mTORC2). mTORC1 activation leads to phosphorylation of S6 ribosomal protein. (B) Decreased expression of IGF1R and RPTOR in circulating B cells from patients with pSS (C) correlates with serum IgG levels. (D) Representative histograms. (E) Phosphorylation of S6 ribosomal protein (indicating mTOR activity) is decreased in circulating B cells from patients with pSS as compared with healthy controls. A similar trend is seen in circulating T cells. Medians are shown. HC, healthy controls; IGF, insulin-like growth factor; nSS, non-Sjögren’s sicca; pSS, primary Sjögren’s syndrome; pS6, phosphorylated S6.
Inhibition of mTORC1 by rapamycin (sirolimus) and its analogues effectively suppresses T and B cells in kidney transplant patients and multiple trials show promising results in treatment of lymphoid malignancies.5 6 Immunosuppression by mTOR inhibition also has been shown in animal models of rheumatic diseases and patients with rheumatoid arthritis and systemic lupus erythematosus (SLE).4 7 8 However, mTOR activity in T and B cells has not been studied in patients with pSS. In this study, we assessed whether the mTOR pathway plays a role in the activation of B cells and T cells of patients with pSS and whether mTOR targeting has potential in pSS treatment.
Patients and methods
Patients and controls
Patients with pSS (n=13 B cells, n=12 labial salivary gland (LSG) tissues), patients with non-Sjögren’s sicca (nSS, n=17 B cells, n=6 LSG tissues) and healthy subjects (n=9 B cells) were included. All patients with pSS were diagnosed by a rheumatologist and fulfilled the American-European Consensus Group (AECG) criteria (2002).9 When applied in retrospect, all patients with pSS fulfilled the 2016 European League Against Rheumatism/American College of Rheumatology criteria.10 The patients with nSS presented with dryness complaints without a known cause, were not clinically considered to have any generalised autoimmune disease including pSS and did not fulfil the AECG classification criteria. Patient characteristics are shown in online supplementary table 1. At the time the biopsies of the immunofluorescence cohort were taken, EULAR Sjögren's Syndrome Disease Activity Index and EULAR Sjögren's Syndrome Patient Reported Index scores were not available.
rmdopen-2018-000701supp009.pdf (52.9KB, pdf)
Gene expression of isolated B cells
CD19+ B cells were isolated from fresh peripheral blood mononuclear cells (PBMC) using magnetic-activated cell sorting. The expression of the following mTOR pathway-related genes was assessed using a custom qPCR-based array: MTOR, RPTOR, RICTOR, DEPTOR, AKT1, IGF1R, IGF1 and PTEN.
Flow cytometry
Antibodies against CD45, CD19, CD20, CD27, CD38, IgG, CD3, CD4, CD14 and CCR9 were used for ex vivo staining and PBMC culture experiments. Activation of the mTORC1 pathway was assessed by intracellular detection of phosphorylated S6 (pS6) ribosomal protein expression, indicating kinase (S6K) activity downstream of mTORC1. Viability and cell proliferation were assessed.
Immunofluorescence
To study local mTOR activation in salivary glands, immunofluorescence staining was performed on frozen tissue sections from patients with pSS and nSS using anti-CD20, anti-CD138, anti-CD3 or anti-CCR9 in combination with anti-pS6 antibodies.
Cell culture
PBMCs (0.5×106) were cultured in the presence or absence of 1–100 nM rapamycin to study the effect of mTOR inhibition on T and B cell proliferation (6 days) and production of interferon gamma (IFN-γ) (measured by Luminex) and IgG (measured by ELISA) (9 days). T and B cell activation was induced with a combination of 0.1 ng/mL superantigen Staphylococcal enterotoxin B (SEB) and 3.3 µg/mL of TLR9 agonist CpG-C.
Statistical analysis
Statistical analyses were performed in Prism V.6 and SPSS V.21 software. Student’s t-test, paired sample t-test, Mann-Whitney U test and Wilcoxon signed-rank test were used where appropriate. For correlations with disease parameters, Pearson’s and Spearman’s correlation were used where appropriate. Differences and correlations were considered statistically significant at p<0.05.
More detailed information on methods is described in online supplementary file 1.
rmdopen-2018-000701supp008.htm (8.8KB, htm)
Results
Decreased expression of mTOR pathway-related genes and mTOR activity in circulating B cells from patients with pSS correlates with B cell hyperactivity
Contemplating the prominent B cell hyperactivity in pSS we investigated mTOR-associated genes in circulating B cells. Unexpectedly, RPTOR, an important component of the mTORC1, and IGF1R, cellular survival receptor activating the mTOR pathway, expression were significantly decreased in B cells from patients with pSS as compared with healthy controls (HC) (figure 1B). In the B cells from patients with nSS, RPTOR expression was significantly decreased. These results were technically validated by RT-qPCR (online supplementary figure 1). Decreased expression of RPTOR and IGF1R in all patients with sicca significantly correlated with increased serum IgG (figure 1C). Correlations of the separate groups were as follows: pSS: RPTOR r=−0.44, p=0.15, IGF1R r=−0.55, p=0.08; nSS: RPTOR r=−0.49, p=0.03, IGF1R r=−0.43, p=0.08. No significant correlations with other clinical parameters were found. The remaining mTOR-related genes measured were not differentially expressed between the groups. Since RPTOR but not RICTOR was decreased, we further investigated the mTORC1 pathway. Downregulation of mTORC1 activation in circulating B cells from patients with pSS was confirmed by decreased pS6 protein expression (figure 1D,E), which was found in both naïve (CD27−) and memory (CD27+) B cells (online supplementary figure 2). A similar trend was seen in T cells (figure 1D,E). Gating strategy is shown in online supplementary figure 3. We hypothesised that reduced mTOR activity in circulating B cells may reflect migration of activated B cells to the salivary glands and next studied LSG mTOR activity.
rmdopen-2018-000701supp001.JPEG (196.6KB, JPEG)
rmdopen-2018-000701supp002.JPEG (45.1KB, JPEG)
rmdopen-2018-000701supp003.JPEG (87KB, JPEG)
Increased numbers of B cells and T cells with mTORC1 activation in the salivary gland of patients with pSS correlate with B cell hyperactivity
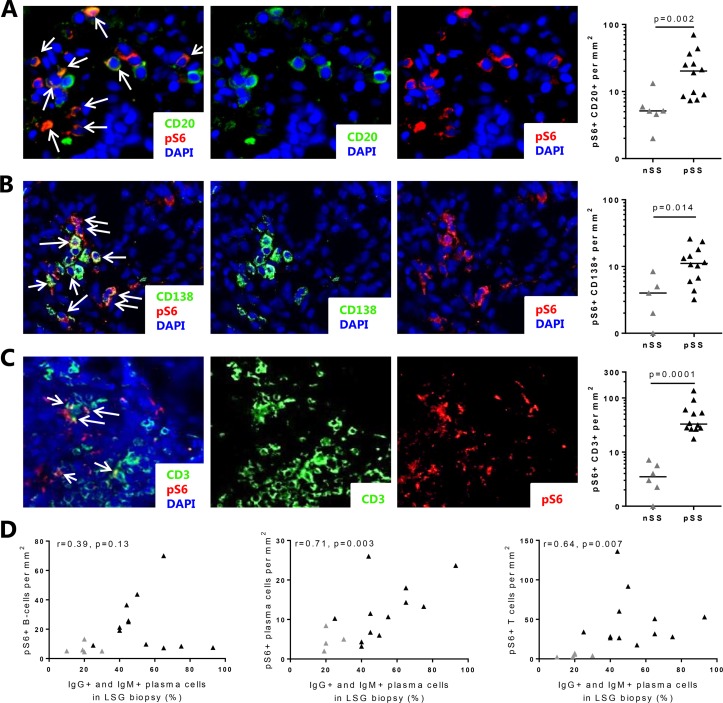
Immunofluorescent colocalisation showed presence of increased numbers of B cells and plasma cells with mTORC1 activation (pS6 expression) in the LSG from patients with pSS (figure 2A,B, nSS shown in online supplementary figure 4). Observing that numerous non-B cells expressed pS6, we next identified CD3+ T cells as another major cell type expressing mTORC1 activity (figure 2C, nSS shown in online supplementary figure 4). In light of our recent identification of the CCL25/CCR9-axis as a potential driver of B cell activity,11 we also assessed mTORC1 activity in CCR9+ cells (this includes T cells and other non-T cells that can express CCR9 including B cells and plasma cells) and observed an increase in patients with pSS (mean±SD 13.7±24.5 cells/mm2 in pSS vs 1.1±0.2 cells/mm2 in nSS). Numbers of T cells and plasma cells, but not B cells or CCR9+ cells with mTORC1 activity, correlated with increased percentages of IgM and IgG-expressing plasma cells (figure 2D). In addition, percentages of T cells, B cells and plasma cells expressing pS6 were calculated. The percentage of pS6+ T cells, but not B cells nor plasma cells, was elevated in patients with pSS (online supplementary figure 5). The percentages did not correlate with clinical parameters including lymphocytic focus score (not shown).
Figure 2.
Elevated numbers of B cells, plasma cells and T cells with activated mTOR complex 1 (mTORC1) in salivary glands of patients with pSS correlate with B cell hyperactivity. In the salivary glands of patients with pSS, increased numbers of B cells (A), plasma cells (B) and T cells (C) with activated mTORC1 were observed as compared with patients with nSS. (D) Numbers of T cells and plasma cells with activated mTORC1 (pS6+) as assessed by immunofluorescence correlate with IgG+ and IgM+ plasma cells. Medians are shown. Magnification: 400×. nSS, non-Sjögren’s sicca; pSS, primary Sjögren’s syndrome; pS6, phosphorylated S6.
rmdopen-2018-000701supp004.JPEG (190.8KB, JPEG)
rmdopen-2018-000701supp005.JPEG (88.8KB, JPEG)
mTOR inhibition robustly decreases B cell and T cell activation
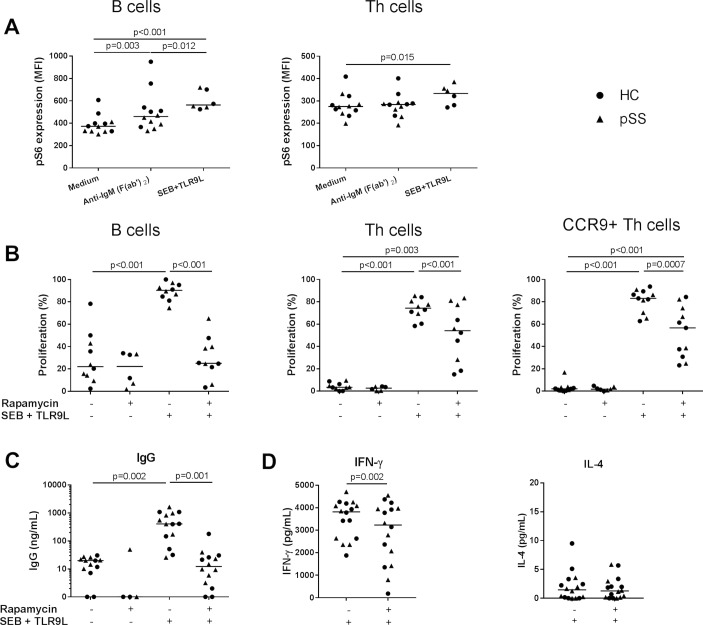
Activation of PBMCs with a combination of TCR cross-linking superantigen SEB and TLR9 ligand resulted in increased phosphorylation of S6 in CD4+ T helper (Th) cells and B cells (figure 3A), associated with Th cell and B cell proliferation (figure 3B) and production of IFN-γ and IgG (figure 3C,D) in both HC and pSS. BCR cross-linking by anti-IgM was used as a control condition and induced mTORC1 activation in B cells but not in Th cells (figure 3A).12 Rapamycin most optimally inhibited T and B cell proliferation at 100 nM (online supplementary figure 6). At this concentration, rapamycin inhibited mTOR activity and reduced Th cell and B cell proliferation (figure 3B) and production of IFN-γ and IgG (figure 3C,D) of both HC and patients with pSS. All inhibitions by rapamycin were significant in both HC and pSS (p<0.05), for Th cells and CCR9+ Th cells in pSS (both p=0.08) and IFN-γ in HC (p=0.09), similar trends were found.
Figure 3.
B cell and T cell proliferation and production of IgG and IFN-γ are inhibited by mammalian/mechanistic target of rapamycin (mTOR) targeting in vitro. (A) B cell receptor cross-linking results in increased mTOR complex 1 (mTORC1) activation (phosphorylation of S6) in B cells. Activation of T cells, including CCR9+ Th cells and B cells by a combination of superantigen SEB and TLR9-ligand CpG-C induces mTORC1 activation and is associated with proliferation of these cells (B) and IgG (C) and IFN-γ production (D), which is inhibited by rapamycin (100 nM). For all graphs: healthy controls (HC, circles), patients with primary Sjögren’s syndrome (pSS, triangles). Medians are shown. IFN-γ, interferon gamma; IL, interleukin; SEB, Staphylococcal enterotoxin B; Th, T helper.
rmdopen-2018-000701supp006.JPEG (333.8KB, JPEG)
Interleukin-4 production was low and unaffected by rapamycin (figure 3D). This corroborates the large body of literature in which inhibition of T and B cell activity by rapamycin is shown.5 6 13 14 Proliferation of CCR9-expressing Th cells and CD3+ CD4− (CD8/γδT/NKT) cells was inhibited similarly to total Th cells (figure 3B and online supplementary figure 7). Rapamycin did not significantly affect viability of lymphocytes (online supplementary figure 6).
rmdopen-2018-000701supp007.JPEG (40.2KB, JPEG)
Discussion
We here for the first time studied mTOR activity in circulating and salivary gland lymphocytes from patients with pSS and found increased mTORC1 activity in salivary gland B cells and T cells, which was associated with local and systemic B cell hyperactivity. Proliferation of B cells, Th cells, Tc cells and CCR9+ Th cells and production of IgG and IFN-γ could be effectively halted in vitro with mTOR inhibition using rapamycin, affecting proliferation and IgG production more strongly than IFN-γ production
The downregulation of mTOR pathway-related genes in circulating B cells of patients with pSS inversely correlated with increased serum IgG levels. As elevated numbers of B cells with activated mTORC1 pathway were found in salivary glands of patients with pSS, this could indicate that activated B cells with higher mTOR activity have migrated to involved organs. Supporting this hypothesis, decreased numbers of memory B cells have been found in the peripheral blood of patients with pSS.15 In our small cohort we did not observe significant reduction of frequencies of memory B cells (not shown), but we did observe reduced pS6 expression in both naïve and memory B cells in patients with pSS as compared with HC. Reduced expression of mTOR-related genes in all circulating B cells might additionally be caused by systemic mediators causing active downregulation. The factors inducing such downregulation have not been identified, but may include Galectin-9 (Gal9). Recently, in addition to patients with SLE,16 we have documented strongly increased systemic levels of Gal9 in patients with pSS as compared with HC. Gal9 levels correlate with IFN-induced gene and protein expression as well as disease activity in patients with pSS (van Roon et al, manuscript in preparation). Gal9 provides negative feedback and inhibits phenotypic maturation of plasmacytoid dendritic cells (pDCs) and B cells and abrogates their ability to mount cytokine responses to TLR7/TLR9 ligands. Importantly, immune complex-mediated and neutrophil extracellular trap-mediated pDC activation is inhibited by Gal9. Additionally, the mTOR/pS6 pathway which is recruited by both pDCs and B cells for TLR-mediated IFN secretion and autoantibody generation is attenuated by Gal9.17
Also in patients with nSS, downregulation of RPTOR in circulating B cells was found, possibly since some of these patients show signs of immune activation. In this respect, we observed local B cell hyperactivity in some patients with nSS, like patients with pSS. In addition, although patients with pSS show increased absolute pS6-expressing T and B cells our data demonstrate that patients with nSS show substantial proportions of pS6-expressing T and B cells. Whether this represents enhanced activation in patients with nSS or naturally occurring immunity at this barrier site remains to be studied. However, recently we demonstrated that in serum and circulating antigen-presenting cells like classical/conventional dendritic cells and pDCs molecular aberrances were significantly overlapping with patients with pSS (Hillen et al, manuscript in preparation). Alternatively, B cells (and T cells) with reduced pS6 expression in patients with nSS may also be caused by increased migration of B and T cells to the salivary gland. Recently, using a novel method to count cells in salivary gland tissue (epigenetic cell counting) we demonstrated that patients with nSS have equal numbers of B and T cells in their salivary glands as a large proportion (~50%) of patients with pSS (in both groups ~20% of all cells).18 In this respect, patients with nSS may represent a group with signs of inflammation, but lacking lymphoid organisation (1 focus/4 mm2) that is associated with significant less mTOR activation in the glands. Future studies on the molecular and cellular characteristics of patients with nSS may add to characterisation of this poorly defined group both in the clinics and in research.
Insulin-like growth factor (IGF)-1 receptor triggering previously has been demonstrated as one of the upstream activators of mTOR.19–22 Furthermore, IGF-1 has shown to play role in B cell activation and function.23 Interestingly, we found IGFR1 to be expressed in B cells from patients with pSS at lower levels than in B cells from HC. This was in line with decreased activation of the mTOR pathway. Downregulation of IGF1R may be one of the mechanisms by which decreased mTOR activation is found in peripheral blood B cells of patients with pSS. Other receptors upstream of mTOR potentially involved in downregulation in pSS still remain to be studied.
To indicate mTOR pathway activation, we measured the phosphorylation of ribosomal protein S6 by flow cytometry in circulating lymphocytes and immunofluorescence microscopy in salivary gland tissues. Antibodies against two different positions of S6 phosphorylation for the two techniques were measured for technical reasons. Although both indicators of mTOR activity, phosphorylation of S6 at serine 240/244 (fluorescence activated cell sorting) is considered to be more specific mTOR activation marker than at serine 235/236 (immunofluorescence) as also kinases RSK1 and RSK2 downstream of the extracellular-signal-regulated kinase pathway can in some conditions phosphorylate the latter position.24 Nonetheless, the phosphorylation status of both positions of S6 strongly correlates. Furthermore, B cells from mice deficient for Rptor, an essential component of mTORC1,25 26 have distinguished loss of S6 phosphorylation at serine 235/236. Furthermore, phosphorylation of S6 at serine 235/236 is sensitive to rapamycin in B cell activating factor belonging to the TNF family (BAFF)-stimulated primary B cells13 indicating that S6 phosphorylation at serine 235/236 largely reflects mTOR pathway activation in B cells.
The mTOR pathway plays an important role in the development and function of B cells, including in germinal centres.3 In addition, mTOR is critical for differentiation of Tfh cells, which are essential for germinal centre reactions.3 Development of germinal centre-like structures in the exocrine glands is associated with an increased risk of developing B cell lymphoma.2 Although we did not investigate the association of mTOR and germinal centres we did find a clear correlation of CD3 T cells and B cell hyperactivity in the salivary glands. This clear correlation was not observed for CD20 B cells and B cell hyperactivity, which supports the fact that T cell-driven B cell activation and mTOR activation play a crucial role in pSS immunopathology.
We recently described increased CCR9-expressing Th cells and their ligand CCL25 in pSS salivary glands.11 CCR9+ Th cells are potent drivers of B cell hyperactivity and in mice were shown to induce salivary gland inflammation and CD8+ T cell-dependent immunopathology.27 Hence, CCR9+ Th cells that have Tfh-like characteristics and possibly play a role in germinal centre formation may play a significant role in pSS pathogenesis.11 27 In this study, we found increased CCR9+ cells with activated mTORC1 pathway in the LSG of patients with pSS and demonstrated inhibition of proliferation of blood CCR9+ Th cells by rapamycin. This, next to inhibition of mTOR activity of B cells and Tc cells, indicates that mTOR inhibition may limit immunopathology induced by both Tfh and CCR9+ Tfh-like cells. CCR9-expressing cells, other than Th cells, that migrate to the salivary glands in response to increased CCL25 may include B cells, plasma cells, CD8 T cells and pDCs. Corroborating this we demonstrated considerable proportions of pS6-expressing cells to coexpress CCR9 (~22%).
Corroborating the potential of mTOR targeting for pSS, Shah et al reported that local administration of rapamycin inhibits infiltration of lymphocytes in the exocrine glands in the non-obese diabetic mouse model and restores tear production.28 29 This was associated with suppression of many inflammatory mediators, including CXCL13, CCL19 and CCL20, chemokines associated with formation of ectopic lymphoid structures. Treatment with rapamycin has been shown to be safe and effective in a phase II trial with patients with SLE, decreasing disease activity without withdrawals due to adverse effects.8 However, adverse effects due to rapamycin treatment, including leucopenia and infections, are known. Interestingly, the antidiabetic drug metformin, which has a more favourable safety profile, inhibits mTOR and reduced B cell differentiation into autoreactive plasma cells and formation of germinal centres in a murine SLE model.30
Thus, our data indicate a role for mTOR activity in B cell hyperactivity in pSS and identify mTOR inhibition as a novel potential therapeutic strategy for this disease. As such, studying the efficacy of (combination) therapy using mTOR inhibitors with favourable toxicity profiles in patients with pSS should be pursued.
Acknowledgments
We thank Professor Dr R Goldschmeding and R Broekhuizen from the Pathology Department of the University Medical Center Utrecht for advice and supply of reagents for immunofluorescence experiments, EHM Otten-van der Heijden for advice on culture experiments and Dr CPJ Bekker for performing RT-qPCR experiments.
Footnotes
JCAB and JAGvR contributed equally.
Contributors: SLMB, MRH, CGKW, MZ, AAK, TRDJR, JCAB and JAGvR made substantial contributions to conception and design. SLMB, MRH and CGKW made acquisition of data. All authors contributed to analysis and/or interpretation of data, drafting the article or revising it critically for important intellectual content, and gave approval.
Funding: The authors have not declared a specific grant for this research from any funding agency in the public, commercial or not-for-profit sectors.
Competing interests: None declared.
Patient consent for publication: Not required.
Ethics approval: The study was approved by the hospital’s medical ethics committee.
Provenance and peer review: Not commissioned; externally peer reviewed.
Data sharing statement: No additional data are available.
References
- 1. Nocturne G, Mariette X. B cells in the pathogenesis of primary Sjögren syndrome. Nat Rev Rheumatol 2018;14:133–45. 10.1038/nrrheum.2018.1 [DOI] [PubMed] [Google Scholar]
- 2. Risselada AP, Looije MF, Kruize AA, et al. The role of ectopic germinal centers in the immunopathology of primary Sjögren's syndrome: a systematic review. Semin Arthritis Rheum 2013;42:368–76. 10.1016/j.semarthrit.2012.07.003 [DOI] [PubMed] [Google Scholar]
- 3. Li B, Li Z, Wang P. mTORC1 signaling is essential for germinal center reaction. Immunology 2017;152:276–86. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Perl A. Activation of mTOR (mechanistic target of rapamycin) in rheumatic diseases. Nat Rev Rheumatol 2016;12:169–82. 10.1038/nrrheum.2015.172 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Lee JS, Vo TT, Fruman DA. Targeting mTOR for the treatment of B cell malignancies. Br J Clin Pharmacol 2016;82:1213–28. 10.1111/bcp.12888 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Yost SE, Byrne R, Kaplan B. Transplantation: mTOR inhibition in kidney transplant recipients. Nat Rev Nephrol 2011;7:553–5. 10.1038/nrneph.2011.125 [DOI] [PubMed] [Google Scholar]
- 7. Bruyn GA, Tate G, Caeiro F, et al. Everolimus in patients with rheumatoid arthritis receiving concomitant methotrexate: a 3-month, double-blind, randomised, placebo-controlled, parallel-group, proof-of-concept study. Ann Rheum Dis 2008;67:1090–5. 10.1136/ard.2007.078808 [DOI] [PubMed] [Google Scholar]
- 8. Fernandez D, Bonilla E, Mirza N, et al. Rapamycin reduces disease activity and normalizes T cell activation-induced calcium fluxing in patients with systemic lupus erythematosus. Arthritis Rheum 2006;54:2983–8. 10.1002/art.22085 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Vitali C, Bombardieri S, Jonsson R, et al. Classification criteria for Sjögren's syndrome: a revised version of the European criteria proposed by the American-European consensus group. Ann Rheum Dis 2002;61:554–8. 10.1136/ard.61.6.554 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Shiboski CH, Shiboski SC, Seror R, et al. 2016 American College of Rheumatology/European League Against Rheumatism Classification Criteria for Primary Sjögren's Syndrome: a consensus and data-driven methodology involving three international patient cohorts. Arthritis Rheumatol 2017;69:35–45. 10.1002/art.39859 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Blokland SLM, Hillen MR, Kruize AA. Elevated CCL25 and CCR9-expExpressing T helper cells in salivary glands of primary Sjogren's syndrome patients: potential new axis in lymphoid neogenesis. Arthritis Rheumatol 2017;69:2038–51. [DOI] [PubMed] [Google Scholar]
- 12. Donahue AC, Fruman DA. Proliferation and survival of activated B cells requires sustained antigen receptor engagement and phosphoinositide 3-kinase activation. J Immunol 2003;170:5851–60. 10.4049/jimmunol.170.12.5851 [DOI] [PubMed] [Google Scholar]
- 13. Zeng Q, Qin S, Zhang H, et al. Rapamycin attenuates BAFF-extended proliferation and survival via disruption of mTORC1/2 signaling in normal and neoplastic B-lymphoid cells. J Cell Physiol 2018;233:516–29. 10.1002/jcp.25913 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Limon JJ, Fruman DA. Akt and mTOR in B cell activation and differentiation. Front Immunol 2012;3:228 10.3389/fimmu.2012.00228 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Hansen A, Odendahl M, Reiter K, et al. Diminished peripheral blood memory B cells and accumulation of memory B cells in the salivary glands of patients with Sjögren's syndrome. Arthritis Rheum 2002;46:2160–71. 10.1002/art.10445 [DOI] [PubMed] [Google Scholar]
- 16. van den Hoogen LL, van Roon JAG, Mertens JS, et al. Galectin-9 is an easy to measure biomarker for the interferon signature in systemic lupus erythematosus and antiphospholipid syndrome. Ann Rheum Dis 2018;77:1810–4. 10.1136/annrheumdis-2018-213497 [DOI] [PubMed] [Google Scholar]
- 17. Panda SK, Facchinetti V, Voynova E, et al. Galectin-9 inhibits TLR7-mediated autoimmunity in murine lupus models. J Clin Invest 2018;128:1873–87. 10.1172/JCI97333 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Antonioli L, Pacher P, Vizi ES, et al. CD39 and CD73 in immunity and inflammation. Trends Mol Med 2013;19:355–67. 10.1016/j.molmed.2013.03.005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Weng QP, Andrabi K, Kozlowski MT, et al. Multiple independent inputs are required for activation of the p70 S6 kinase. Mol Cell Biol 1995;15:2333–40. 10.1128/MCB.15.5.2333 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Somwar R, Sumitani S, Taha C, et al. Temporal activation of p70 S6 kinase and Akt1 by insulin: PI 3-kinase-dependent and -independent mechanisms. Am J Physiol 1998;275:E618–E625. 10.1152/ajpendo.1998.275.4.E618 [DOI] [PubMed] [Google Scholar]
- 21. Latres E, Amini AR, Amini AA, et al. Insulin-like growth factor-1 (IGF-1) inversely regulates atrophy-induced genes via the phosphatidylinositol 3-kinase/Akt/mammalian target of rapamycin (PI3K/Akt/mTOR) pathway. J Biol Chem 2005;280:2737–44. 10.1074/jbc.M407517200 [DOI] [PubMed] [Google Scholar]
- 22. von Manteuffel SR, Gingras AC, Ming XF, et al. 4E-BP1 phosphorylation is mediated by the FRAP-p70s6k pathway and is independent of mitogen-activated protein kinase. Proc Natl Acad Sci U S A 1996;93:4076–80. 10.1073/pnas.93.9.4076 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Kim JH, Park HH, Lee CE. IGF-1 potentiation of IL-4-induced CD23/Fc(epsilon)RII expression in human B cells. Mol Cells 2003;15:307–12. [PubMed] [Google Scholar]
- 24. Roux PP, Shahbazian D, Vu H, et al. RAS/ERK signaling promotes site-specific ribosomal protein S6 phosphorylation via RSK and stimulates cap-dependent translation. J Biol Chem 2007;282:14056–64. 10.1074/jbc.M700906200 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Hara K, Maruki Y, Long X, et al. Raptor, a binding partner of target of rapamycin (TOR), mediates TOR action. Cell 2002;110:177–89. 10.1016/S0092-8674(02)00833-4 [DOI] [PubMed] [Google Scholar]
- 26. Kim DH, Sarbassov DD, Ali SM, et al. mTOR interacts with raptor to form a nutrient-sensitive complex that signals to the cell growth machinery. Cell 2002;110:163–75. 10.1016/S0092-8674(02)00808-5 [DOI] [PubMed] [Google Scholar]
- 27. McGuire HM, Vogelzang A, Ma CS, et al. A subset of interleukin-21+ chemokine receptor CCR9+ T helper cells target accessory organs of the digestive system in autoimmunity. Immunity 2011;34:602–15. 10.1016/j.immuni.2011.01.021 [DOI] [PubMed] [Google Scholar]
- 28. Shah M, Edman MC, Janga SR, et al. A rapamycin-binding protein polymer nanoparticle shows potent therapeutic activity in suppressing autoimmune dacryoadenitis in a mouse model of Sjögren's syndrome. J Control Release 2013;171:269–79. 10.1016/j.jconrel.2013.07.016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Shah M, Edman MC, Reddy Janga S, et al. Rapamycin Eye Drops Suppress Lacrimal Gland Inflammation In a Murine Model of Sjögren's Syndrome. Invest Ophthalmol Vis Sci 2017;58:372–85. 10.1167/iovs.16-19159 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Lee SY, Moon SJ, Kim EK, et al. Metformin suppresses systemic autoimmunity in Roquinsan/san mice through inhibiting B cell differentiation into plasma cells via regulation of AMPK/mTOR/STAT3. J Immunol 2017;198:2661–70. 10.4049/jimmunol.1403088 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
rmdopen-2018-000701supp009.pdf (52.9KB, pdf)
rmdopen-2018-000701supp008.htm (8.8KB, htm)
rmdopen-2018-000701supp001.JPEG (196.6KB, JPEG)
rmdopen-2018-000701supp002.JPEG (45.1KB, JPEG)
rmdopen-2018-000701supp003.JPEG (87KB, JPEG)
rmdopen-2018-000701supp004.JPEG (190.8KB, JPEG)
rmdopen-2018-000701supp005.JPEG (88.8KB, JPEG)
rmdopen-2018-000701supp006.JPEG (333.8KB, JPEG)
rmdopen-2018-000701supp007.JPEG (40.2KB, JPEG)