Abstract
Chronic kidney disease (CKD) is an important cause of morbidity and mortality in HIV-infected individuals, even in the antiretroviral therapy (ART) era. Inflammatory cytokines and adipokines have been suggested to play a role in the development of CKD. The aim of the present study was to examine the circulating levels of a novel proinflammatory cytokine, angiopoietin-like protein 2 (ANGPTL2), in a cohort of 72 HIV-positive subjects on ART. HIV-positive patients were on cART for at least one year. Urine and blood samples were collected. Various factors were analyzed including body mass index (BMI), smoking, and presence/treatment for comorbidities such as diabetes. The estimated glomerular filtration rate was calculated by using the Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) equation. Plasma samples obtained were stored and used to measure sCD14 and ANGPTL2 levels. Data were presented as mean (± standard deviation) or median (interquartile range) for continuous variables. Categorical variables were expressed as number (%). Variables were compared using Student's t-test, Mann-Whitney test, or χ2 test. The results showed an independent negative association between plasma ANGPTL2 and CKD-EPI values. Further prospective studies on larger cohorts are needed to evaluate the pathogenetic role of ANGPTL2 as well as its use as a diagnostic marker of renal dysfunction.
Keywords: angiopoietin-like protein 2, cART, CKD-EPI, inflammation, renal function
Introduction
Chronic kidney disease (CKD) is a public health problem, and a major risk factor for end-stage kidney disease, cardiovascular disease and premature death. In HIV-infected subjects, CKD is the fourth cause of death for non-AIDS-related diseases, after malignancies, cardiovascular and liver disease (1–20). With the prolonged survival of HIV-infected individuals, the prevalence of CKD is expected to increase along with the burden of comorbidities, such as diabetes and hypertension (21).
In recent years, it has been suggested that inflammatory cytokines play a role in the pathogenesis of CKD (22). Angiopoietin-like protein 2 (ANGPTL2) is an adipokine belonging to the recently characterized family of ANGPTLs (23). Although structurally similar to angiopoietin, ANGPTLs do not bind to the classical angiopoietin receptors, and several members of the family are involved, not only in the regulation of angiogenesis, but also in glucose and lipid metabolism (23). ANGPTL2 is abundantly produced in fat tissue and its levels are increased in the setting of insulin resistance, obesity, and diabetes (23,24). Excess ANGPTL2 secretion leads to chronic inflammation and endothelial dysfunction. Interestingly, in a recent report of Usui et al elevated circulating levels of ANGPTL2 were associated with the likelihood of CKD in a large Japanese cohort of uninfected individuals (25). To date, however, no studies have investigated the link between serum ANGPTL2 levels and renal function in HIV-positive individuals.
The aim of the present study was to assess the relationship between circulating ANGPTL2 levels and renal function in a cohort of HIV-positive subjects on combination antiretroviral therapy (cART).
Subjects and methods
Study population
In this cross-sectional study, we consecutively enrolled 72 HIV-positive patients attending the HIV Outpatient Clinic of the Division of Infectious Diseases in Catania, Italy, who were on cART for at least one year. All the participants provided written informed consent to participate in the study, which was conducted in accordance with the Declaration of Helsinki.
We extracted the following parameters from medical records: patient demographics, time since HIV diagnosis and initiation of cART, type of cART, HCV and/or HBV coinfection, body mass index (BMI), smoking, and presence/treatment for comorbidities, such as diabetes mellitus, hypertension, and dyslipidemia. At the time of enrollment, urine and blood samples were collected to evaluate CD4+ and CD8+ T cell count, plasma HIV RNA, highly sensitive (hs)-C reactive protein (CRP), presence of proteinuria and microalbuminuria. Estimated glomerular filtration rate (eGFR) was calculated by using the Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) equation. Plasma samples were stored at −80°C and then used to measure sCD14 (Quantikine sCD14 Immunoassay; R&D Systems, Inc., Minneapolis, MN, USA) with a detection range of 40–3,200 ng/ml and ANGPTL2 (Uscn Life Science Inc., Wuhan, China) with a detection range of 0.313–20 ng/ml levels, according to manufacturer's protocol.
Statistical analysis
Data were analyzed using the Statistical Package for Social Sciences version 22.0 (SPSS, Inc., Chicago, IL, USA). Data are presented as mean ± standard deviation for continuous variables with normal distribution or median (interquartile range) for continuous variables with skewed distribution. Categorical variables are expressed as number (%). Variables that were normally distributed, continuous but not normally distributed, or categorical were compared using Student's t-test, Mann-Whitney test, or χ2 test, respectively. Pearson's correlation test was used to assess the correlation of CKD-EPI values and demographic, biochemical and viro-immunological variables. Statistically significant variables in the univariate analysis (P<0.1) were included in the multivariate regression analysis, the dependent variable being CKD-EPI. In the multivariate regression model, both sCD14 and ANGPTL2 were log-transformed (ln) to approximate normality. Presence of multicollinearity was checked by examining the variance inflation factor (VIF) and variables included in the final model if VIF was <5.
Results
Subject characteristics
The main demographic and clinical characteristics of the study population are reported in Table I. In total, 72 patients were enrolled, and all were Caucasian, with a median age of 47 (range, 36–53) years. As for risk factors for HIV acquisition, 26 individuals (36.1%) were heterosexual, and 40 (55.6%) were men having sex with men. In addition, 14 (19.4%) women were included in the study. Median time since HIV diagnosis was 122 (range, 59–200) months, and median time since cART initiation was 72 (31–191) months. Median current CD4+ T cell count was 605 (409–789) cells/µl, while HIV viral load was <50 copies/ml in 59 patients (82%). Of the 72 patients, 27 patients (37.5%) received a protease inhibitor (PI)-based regimen, 57 (79.2%) were on tenofovir (TDF), and 10 (13.9%) on raltegravir. In addition, 26 (36.1%) were smokers, and 6 patients (8.3%) were co-infected with hepatitis C. In terms of comorbidities, 34 subjects (47.2%) had a BMI >25 kg/m2, 6.9% of the study population had diabetes, 18.1% hypertension, 30.5% dyslipidemia. A total of 10 individuals (13.9%) had a high cardiovascular risk (i.e., Framingham risk score >20%), and 13.9% were taking lipid-lowering drugs.
Table I.
Demographics and clinical characteristics of the study population.
| Variable | No. (n=72) |
|---|---|
| Age (years) | 47 (36–53) |
| Sex, female [n (%)] | 14 (19.4) |
| MSM [n (%)] | 40 (56) |
| Time since HIV diagnosis (months) | 122 (59–200) |
| Current CD4+ T-cell count (cells/µl) | 605 (409–789) |
| HIV viral load <50 copies/ml [n (%)] | 59 (82) |
| Time on cART (months) | 72 (31–191) |
| Current use of PI [n (%)] | 27 (37.5) |
| Current use of TDF [n (%)] | 57 (79.2) |
| Current use of raltegravir [n (%)] | 10 (13.9) |
| BMI (kg/m2) | 24 (22–26.6) |
| Current smoking [n (%)] | 26 (36.1) |
| Hepatitis C coinfection [n (%)] | 6 (8.3) |
| Diabetes mellitus [n (%)] | 5 (6.9) |
| Hypertension [n (%)] | 13 (18.1) |
| Dyslipidemia [n (%)] | 22 (30.6) |
| Framingham risk score (%) | 4.5 (1–11.2) |
| eGFR (ml/min/1.73 m2), based on CKD-EPI formula | 96.9±18.5 |
| Proteinuria [n (%)] | 23 (32) |
| Microalbuminuria [n (%)] | 18 (25) |
| hs-CRP (ng/ml) | 0.18 (0.07–0.47) |
| ANGPTL2 (ng/ml) | 2.09 (1.45–3.03) |
| sCD14 (ng/ml) | 1,969 (1,747–2,292) |
Data are mean ± standard deviation or median (interquartile range), unless otherwise specified. ANGPTL2, angiopoietin-like protein 2; BMI, body mass index; cART, combination antiretroviral therapy; CKD-EPI, Chronic Kidney Disease Epidemiology Collaboration; eGFR, estimated glomerular filtration rate; HIV, human immunodeficiency virus; hs-CRP, highly sensitive-C reactive protein; MSM, men having sex with men; PI, protease inhibitor; sCD14, soluble CD14; TDF, tenofovir.
Renal function and predictors of reduced CKD-EPI
Mean CKD-EPI was 96.9±18.5 ml/min; 25 patients (34.7%) had CKD-EPI <90 ml/min, while 2.8% had values <60 ml/min. In addition, 23 individuals (32%) had proteinuria, while 18 (25%) had microalbuminuria.
Individuals with high cardiovascular risk had decreased renal function compared to low-risk subjects (85 vs. 99 ml/min, P=0.03). As expected, patients on treatment for hypertension had lower CKD-EPI values (87 vs. 99 ml/min, P=0.07); similarly, individuals taking lipid-lowering drugs had lower CKD-EPI values when compared to those without dyslipidemia (86 vs. 98 ml/min, P=0.06).
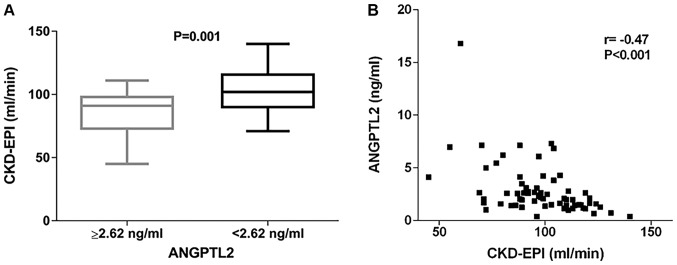
All individuals had detectable levels of ANGPTL2. Median ANGPTL2 circulating level was 2.09 (1.45–3.03) ng/ml. Median sCD14 levels was 1,969 (1,747–2,292) ng/ml. Interestingly, we found that patients with ANGPTL2 values in the highest tertile (i.e., ANGPTL2 values ≥2.62 ng/ml) had significantly lower CKD-EPI values when compared to the rest of the cohort (86 vs. 103 ml/min, P=0.001) (Fig. 1A). Analogously, individuals with sCD14 values in the upper tertile (i.e., sCD14 values ≥2,072 ng/ml) had lower CKD-EPI values (89 vs. 101 ml/min, P=0.02) (data not shown).
Figure 1.
(A) Distribution of Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) values according to circulating angiopoietin-like protein 2 (ANGPTL2) levels. CKD-EPI values were significantly lower in individuals with ANGPTL2 ≥2.62 ng/ml, corresponding to the upper tertile of ANGPTL2 distribution (86 vs. 103 ml/min, P=0.001). (B) Correlation between CKD-EPI and ANGPTL2 circulating levels in the study population (r=−0.47, P<0.001).
As expected, CKD-EPI values were negatively correlated with age (r=−0.56, P<0.001). Moreover, they were negatively correlated with Framingham risk score (r=−0.37, P=0.001), sCD14 levels (r=−0.37, P=0.002) and ANGPTL2 (r=−0.47, P<0.001; Fig. 1B).
In multivariate analysis, only older age (β=−0.42, P=0.013) and higher circulating ANGPTL2 (β=−0.29, P=0.024) remained significantly associated with reduced CKD-EPI values (Table II).
Table II.
Multivariate regression model for CKD-EPI, including factors associated with reduced renal function in univariate analysis.
| Variables | β | B (SE) | P-value |
|---|---|---|---|
| Age (per year) | −0.42 | −0.67 (0.26) | 0.013 |
| ANGPTL2 (ln, ng/ml) | −0.29 | −7.7 (3.3) | 0.024 |
| sCD14 (ln, ng/ml) | −0.19 | −21.9 (12.9) | 0.095 |
| Framingham risk score (%) | 0.15 | 0.31 (0.29) | 0.29 |
| Antihypertensive therapy, yes | −0.03 | −1.1 (5.3) | 0.83 |
| Lipid-lowering drugs, yes | 0.002 | 0.11 (5.9) | 0.98 |
ANGPTL2, angiopoietin-like protein 2; β, standardized coefficient; B, unstandardized coefficient; sCD14, soluble CD14; SE, standard error.
Discussion
In the present study, we found an independent association between reduced renal function and elevated levels of ANGPTL2 in a cohort of HIV-positive individuals on cART. ANGPTL2 is a recently characterized adipokine, which has been associated with several metabolic disorders, including insulin resistance, obesity and diabetes (23,24). Existing studies have shown that ANGPTL2 can act as a mediator of chronic inflammation (23). ANGPTL2 is mainly produced in visceral adipose tissue, where it promotes macrophage recruitment and neoangiogenesis. In obese mice, elevated levels of ANGPTL2 led to chronic adipose tissue inflammation and remodeling, and finally to systemic insulin resistance (24). Similarly, in obese humans, elevated levels of ANGPTL2 have been associated with the presence of insulin resistance as well as the risk of developing diabetes (24,26). Moreover, both in animal and human studies, elevated ANGPTL2 levels have been positively correlated with vascular inflammation and atherosclerosis progression (24). Inflammatory cytokines and adipokines, such as resistin, visfatin and adiponectin, have been suggested to play a role in the development of CKD. In a large population-based study recently conducted in Japan, the authors found that circulating ANGPTL2 levels were independently associated with the likelihood of CKD (25). In this study, CKD was defined as the presence of albuminuria or eGFR <60 ml/min. The authors reported that the odds ratio (OR) for CKD increased with elevated circulating ANGPTL2. The OR for CKD was analyzed across quintiles and found to be significantly higher in the second quintile [ANGPTL2 values 2.01–2.48 ng/ml, OR=1.67, 95% confidence interval (CI): 1.24–2.24], and thereafter to plateau to the fifth quintile (OR=1.79, 95% CI: 1.32–2.43). The association remained significant after correcting for known cardiovascular risk factors and hs-CRP levels. However, when separating eGFR and albuminuria as markers of reduced renal function, only the latter remained significantly associated with circulating ANGPTL2 levels. In our study, the limited sample size did not allow us to evaluate whether ANGPTL2 was a predictor of albuminuria or not. By immunochemistry, ANGPT2 has been shown to be upregulated in the glomeruli of subjects with diabetes (25). Moreover, ANGPTL2 has been reported to activate the NF-κB pathway in endothelial cells. As NF-κB upregulation in the glomerulus has been correlated with albuminuria, it can be hypothesized that increased ANGPTL2 levels may represent a marker of endothelial dysfunction and chronic inflammation in multiple organs, including the kidney (24,27,28).
The presence of abnormalities in renal function is frequent in HIV-positive individuals (21). Up to 30% of HIV-positive individuals have proteinuria and 4% of them can develop CKD over time. In addition to traditional risk factors, such as aging, diabetes, chronic hepatitis, hypertension, and black ethnicity, several HIV-related factors can increase the risk of CKD, including low CD4+ T cell nadir, elevated HIV viral load, and exposure to some antiretroviral drugs, especially TDF and PIs (21). HIV-positive individuals are known to have increased levels of chronic inflammation and immune activation compared to the general population (29). We hypothesize therefore that elevated levels of ANGPTL2 may reflect this proinflammatory status. Although the design of the study is not appropriate to assess causality, ANGPTL2 may be a multiorgan marker of endothelial dysfunction and, more specifically, a marker of renal impairment in this case. With the increase in life expectancy and comorbidities in HIV-infected individuals, identification of a novel marker of renal dysfunction can help design more effective strategies to prevent or delay the progression towards CKD.
Our study has several limitations: first, the small sample size, which limits the statistical power of our analysis; second, the lack of a control group of uninfected individuals; third, its cross-sectional design, which does not allow to establish causal relationships. In fact, we cannot exclude that increased ANGPTL2 results from reduced renal function, although ANGPTL2 should not be filtered by the glomerulus, considering its molecular weight of 57 kDa (25). On the basis of our findings, we are currently enrolling a larger cohort of individuals on cART and a matched group of HIV-negative subjects in order to validate the results of this pilot study. Moreover, we are currently interested in evaluating ANGPTL2 levels in untreated HIV infection and establishing whether circulating ANGPTL2 levels change over time after starting cART.
In conclusion, to the best of our knowledge, this is the first study to identify an association between renal dysfunction and increased circulating ANGPTL2. Further research is needed to clarify the potential pathogenetic mechanisms underlying this association as well as assess whether ANGPTL2 may represent a novel marker of declining renal function in HIV infection.
Acknowledgements
We would like to thank all the patients who participated to the study.
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Authors' contributions
GN and MRP designed the study. MM, RB, BMC and BC recruited the patients and gathered data and informed consent modules. MDR, MRP and EVR statistically analyzed the patients' clinical and laboratory data. GN, MRP and MC wrote the manuscript, and GFP, FC, EF, GM and AC revised the manuscript.
Ethics approval and consent to participate
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Patient consent for publication
Written informed consent was obtained from the individual participants included in the study.
Competing interests
The authors declare that they have no conflict of interest.
References
- 1.Pomerantz RJ, Nunnari G. HIV and GB virus C - can two viruses be better than one? N Engl J Med. 2004;350:963–965. doi: 10.1056/NEJMp048004. [DOI] [PubMed] [Google Scholar]
- 2.Nunnari G, Coco C, Pinzone MR, Pavone P, Berretta M, Di Rosa M, Schnell M, Calabrese G, Cacopardo B. The role of micronutrients in the diet of HIV-1-infected individuals. Front Biosci (Elite Ed) 2012;4:2442–2456. doi: 10.2741/e556. [DOI] [PubMed] [Google Scholar]
- 3.Pinzone MR, Berretta M, Cacopardo B, Nunnari G. Epstein-barr virus- and Kaposi sarcoma-associated herpesvirus-related malignancies in the setting of human immunodeficiency virus infection. Semin Oncol. 2015;42:258–271. doi: 10.1053/j.seminoncol.2014.12.026. [DOI] [PubMed] [Google Scholar]
- 4.Pinzone MR, Di Rosa M, Celesia BM, Condorelli F, Malaguarnera M, Madeddu G, Martellotta F, Castronuovo D, Gussio M, Coco C, et al. LPS and HIV gp120 modulate monocyte/macrophage CYP27B1 and CYP24A1 expression leading to vitamin D consumption and hypovitaminosis D in HIV-infected individuals. Eur Rev Med Pharmacol Sci. 2013;17:1938–1950. [PubMed] [Google Scholar]
- 5.Pinzone MR, Cacopardo B, Condorelli F, Di Rosa M, Nunnari G. Sirtuin-1 and HIV-1: An overview. Curr Drug Targets. 2013;14:648–652. doi: 10.2174/1389450111314060005. [DOI] [PubMed] [Google Scholar]
- 6.Capetti A, Landonio S, Meraviglia P, Di Biagio A, Lo Caputo S, Sterrantino G, Ammassari A, Menzaghi B, Franzetti M, De Socio GV, et al. 96 Week follow-up of HIV-infected patients in rescue with raltegravir plus optimized backbone regimens: A multicentre Italian experience. PLoS One. 2012;7:e39222. doi: 10.1371/journal.pone.0039222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Visalli G, Paiardini M, Chirico C, Cervasi B, Currò M, Ferlazzo N, Bertuccio MP, Favaloro A, Pellicanò G, Sturniolo G, et al. Intracellular accumulation of cell cycle regulatory proteins and nucleolin re-localization are associated with pre-lethal ultrastructural lesions in circulating T lymphocytes: The HIV-induced cell cycle dysregulation revisited. Cell Cycle. 2010;9:2130–2140. doi: 10.4161/cc.9.11.11754. [DOI] [PubMed] [Google Scholar]
- 8.Celesia BM, Nigro L, Pinzone MR, Coco C, La Rosa R, Bisicchia F, Mavilla S, Gussio M, Pellicanò G, Milioni V, et al. High prevalence of undiagnosed anxiety symptoms among HIV-positive individuals on cART: A cross-sectional study. Eur Rev Med Pharmacol Sci. 2013;17:2040–2046. [PubMed] [Google Scholar]
- 9.Visalli G, Bertuccio MP, Currò M, Pellicanò G, Sturniolo G, Carnevali A, Spataro P, Ientile R, Picerno I, Cavallari V, et al. Bioenergetics of T cell activation and death in HIV type 1 infection. AIDS Res Hum Retroviruses. 2012;28:1110–1118. doi: 10.1089/aid.2011.0197. [DOI] [PubMed] [Google Scholar]
- 10.Trovato M, Ruggeri RM, Sciacchitano S, Vicchio TM, Picerno I, Pellicanò G, Valenti A, Visalli G. Serum interleukin-6 levels are increased in HIV-infected patients that develop autoimmune disease during long-term follow-up. Immunobiology. 2018;223:264–268. doi: 10.1016/j.imbio.2017.10.039. [DOI] [PubMed] [Google Scholar]
- 11.D'Aleo F, Ceccarelli M, Venanzi Rullo E, Facciolà A, Di Rosa M, Pinzone MR, Condorelli F, Visalli G, Picerno I, Berretta M, et al. Hepatitis C-related hepatocellular carcinoma: Diagnostic and therapeutic management in HIV-patients. Eur Rev Med Pharmacol Sci. 2017;21:5859–5867. doi: 10.26355/eurrev_201712_14035. [DOI] [PubMed] [Google Scholar]
- 12.Squillace N, Ricci E, Quirino T, Gori A, Bandera A, Carenzi L, De Socio GV, Orofino G, Martinelli C, Madeddu G, et al. CISAI Study Group: Safety and tolerability of Elvitegravir/Cobicistat/Emtricitabine/Tenofovir Disoproxil fumarate in a real life setting: Data from surveillance cohort long-term toxicity antiretrovirals/antivirals (SCOLTA) project. PLoS One. 2017;12:e0179254. doi: 10.1371/journal.pone.0179254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Facciolà A, Venanzi Rullo E, Ceccarelli M, D'Aleo F, Di Rosa M, Pinzone MR, Condorelli F, Visalli G, Picerno I, Fisichella R, et al. Kaposi's sarcoma in HIV-infected patients in the era of new antiretrovirals. Eur Rev Med Pharmacol Sci. 2017;21:5868–5869. doi: 10.26355/eurrev_201712_14036. [DOI] [PubMed] [Google Scholar]
- 14.Nunnari G, Sullivan J, Xu Y, Nyirjesy P, Kulkosky J, Cavert W, Frank I, Pomerantz RJ. HIV type 1 cervicovaginal reservoirs in the era of HAART. AIDS Res Hum Retroviruses. 2005;21:714–718. doi: 10.1089/aid.2005.21.714. [DOI] [PubMed] [Google Scholar]
- 15.Nunnari G, Leto D, Sullivan J, Xu Y, Mehlman KE, Kulkosky J, Pomerantz RJ. Seminal reservoirs during an HIV type 1 eradication trial. AIDS Res Hum Retroviruses. 2005;21:768–775. doi: 10.1089/aid.2005.21.768. [DOI] [PubMed] [Google Scholar]
- 16.Martellotta F, Berretta M, Cacopardo B, Fisichella R, Schioppa O, Zanghì A, Spartà D, Cappellani A, Talamini R, Izzi I, et al. Clinical presentation and outcome of squamous cell carcinoma of the anus in HIV-infected patients in the HAART-era: A GICAT experience. Eur Rev Med Pharmacol Sci. 2012;16:1283–1291. [PubMed] [Google Scholar]
- 17.Celesia BM, Castronuovo D, Pinzone MR, Bellissimo F, Mughini MT, Lupo G, Scarpino MR, Gussio M, Palermo F, Cosentino S, et al. Late presentation of HIV infection: Predictors of delayed diagnosis and survival in Eastern Sicily. Eur Rev Med Pharmacol Sci. 2013;17:2218–2224. [PubMed] [Google Scholar]
- 18.Nuvoli S, Caruana G, Babudieri S, Solinas P, Pellicanò G, Piras B, Fiore V, Bagella P, Calia GM, Yue M, et al. Body fat changes in HIV patients on highly active antiretroviral therapy (HAART): A longitudinal DEXA study. Eur Rev Med Pharmacol Sci. 2018;22:1852–1859. doi: 10.26355/eurrev_201803_14606. [DOI] [PubMed] [Google Scholar]
- 19.Ceccarelli M, Condorelli F, Venanzi Rullo E, Pellicanò GF. Editorial - Improving access and adherence to screening tests for cancers: A new, though old, challenge in the HIV epidemics. WCRJ. 2018;5:e1030. [Google Scholar]
- 20.Ceccarelli M, Rullo EV, Facciolà A, Madeddu G, Cacopardo B, Taibi R, D'Aleo F, Pinzone MR, Picerno I, di Rosa M, et al. Head and neck squamous cell carcinoma and its correlation with human papillomavirus in people living with HIV: A systematic review. Oncotarget. 2018;9:17171–17180. doi: 10.18632/oncotarget.24660. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Scarpino M, Pinzone MR, Di Rosa M, Madeddu G, Focà E, Martellotta F, Schioppa O, Ceccarelli G, Celesia BM, d'Ettorre G, et al. Kidney disease in HIV-infected patients. Eur Rev Med Pharmacol Sci. 2013;17:2660–2667. [PubMed] [Google Scholar]
- 22.Fried L, Solomon C, Shlipak M, Seliger S, Stehman-Breen C, Bleyer AJ, Chaves P, Furberg C, Kuller L, Newman A. Inflammatory and prothrombotic markers and the progression of renal disease in elderly individuals. J Am Soc Nephrol. 2004;15:3184–3191. doi: 10.1097/01.ASN.0000146422.45434.35. [DOI] [PubMed] [Google Scholar]
- 23.Kadomatsu T, Endo M, Miyata K, Oike Y. Diverse roles of ANGPTL2 in physiology and pathophysiology. Trends Endocrinol Metab. 2014;25:245–254. doi: 10.1016/j.tem.2014.03.012. [DOI] [PubMed] [Google Scholar]
- 24.Tabata M, Kadomatsu T, Fukuhara S, Miyata K, Ito Y, Endo M, Urano T, Zhu HJ, Tsukano H, Tazume H, et al. Angiopoietin-like protein 2 promotes chronic adipose tissue inflammation and obesity-related systemic insulin resistance. Cell Metab. 2009;10:178–188. doi: 10.1016/j.cmet.2009.08.003. [DOI] [PubMed] [Google Scholar]
- 25.Usui T, Ninomiya T, Nagata M, Takahashi O, Doi Y, Hata J, Fukuhara M, Kitazono T, Oike Y, Kiyohara Y. Angiopoietin-like protein 2 is associated with chronic kidney disease in a general Japanese population: The Hisayama Study. Circ J. 2013;77:2311–2317. doi: 10.1253/circj.CJ-12-1548. [DOI] [PubMed] [Google Scholar]
- 26.Doi Y, Ninomiya T, Hirakawa Y, Takahashi O, Mukai N, Hata J, Iwase M, Kitazono T, Oike Y, Kiyohara Y. Angiopoietin-like protein 2 and risk of type 2 diabetes in a general Japanese population: The Hisayama study. Diabetes Care. 2013;36:98–100. doi: 10.2337/dc12-0166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Sanz AB, Sanchez-Niño MD, Ramos AM, Moreno JA, Santamaria B, Ruiz-Ortega M, Egido J, Ortiz A. NF-kappaB in renal inflammation. J Am Soc Nephrol. 2010;21:1254–1262. doi: 10.1681/ASN.2010020218. [DOI] [PubMed] [Google Scholar]
- 28.Mezzano S, Aros C, Droguett A, Burgos ME, Ardiles L, Flores C, Schneider H, Ruiz-Ortega M, Egido J. NF-kappaB activation and overexpression of regulated genes in human diabetic nephropathy. Nephrol Dial Transplant. 2004;19:2505–2512. doi: 10.1093/ndt/gfh207. [DOI] [PubMed] [Google Scholar]
- 29.Deeks SG. HIV infection, inflammation, immunosenescence, and aging. Annu Rev Med. 2011;62:141–155. doi: 10.1146/annurev-med-042909-093756. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.