Abstract
Background:
We aimed to evaluate the correlation of neutrophil-to-lymphocyte ratio (NLR) with pathological response, disease-free survival (DFS), and overall survival (OS) in patients with breast cancer and under neoadjuvant chemotherapy (NAC).
Materials and methods:
We performed a systematical search using Cochrane Library, ScienceDirect, PubMed, Embase, and Web of Science up to May 2018. On the basis of the data directly obtained from the available studies, the odds ratios (ORs) and their 95% confidence intervals (95% CIs) were pooled on the basis of higher or lower NLR levels.
Results:
The meta-analysis showed that high NLR was significantly associated with poor NAC response (OR = 2.27, 95% CI: 1.46–3.53, P < .001) but not with the DFS (OR = 1.18, 95% CI: 0.78–1.78, P = .435) and OS (OR = 2.781, 95% CI: 0.54–14.32, P = .221).
Conclusion:
Although high NLR was significantly associated with poor pathological response, we were unable to demonstrate the prognostic value of NLR for DFS and OS in patients with breast cancer who were undergoing NAC.
Keywords: breast cancer, neoadjuvant chemotherapy, neutrophil-to-lymphocyte ratio, pathological response, prognosis
1. Introduction
Currently, neoadjuvant chemotherapy (NAC) is widely used in managing locally advanced breast cancer primarily because of its ability to downsize large tumors. NAC increases the rate of breast-conserving surgery if the breast cancer cell is sensitive to chemotherapy. Previous studies demonstrated that patients with breast cancer in pathologic complete remission (pCR) who underwent NAC hold a significantly better prognosis than patients without pCP.[1,2] The accurate assessment of a response to NAC before surgery is crucial in breast cancer management. The early and accurate prediction of tumor response to NAC may contribute to the personalization of a treatment regimen and prevention of ineffective chemotherapy.[3,4]
The presence of an elevated peripheral neutrophil-to-lymphocyte ratio (NLR), an indicator of systemic inflammation, has been recognized as a poor prognostic factor in various cancers. Some articles[5–14] showed that high NLR is associated with poor NAC response in patients with breast cancer, and other investigations failed.[15] To address this controversy, we evaluated the NLR as a potential prognostic marker for breast cancer and determine the relationship between NLR and clinicopathologic features through meta-analysis.
2. Methods
The study report is based on the guidelines of Preferred Reporting Items for Systematic Reviews and Meta-Analyses[16] and Meta-Analysis of Observational Studies in Epidemiology. All results and analyses were based on previous ethically approved studies; thus, no further ethical approval and patient consent are required.
2.1. Literature search
Cochrane Library, ScienceDirect, PubMed, Embase, and Web of Science were searched for studies up to May 2018 by using the following search terms: “breast cancer,” “neutrophil-to-lymphocyte ratio,” and “neoadjuvant chemotherapy.” The bibliographies from these relevant articles were also manually searched for additional eligible studies.
The following studies were included: studies concerning NAC response of cancer patients with high NRLs versus that of cancer patients with low NRLs; studies with complete information for the assessment of hazard ratios (HRs) or risk ratios (RRs) and their 95% confidence intervals (95% CIs) for pathological response, disease-free survival (DFS), and overall survival (OS); and studies with duplicate data and those that reported the largest sample. The exclusion criteria were insufficient data and letters, editorials, case reports, reviews, comments, or meeting abstracts.
2.2. Data extraction and quality assessment
Two investigators (LBX and YHL) independently reviewed all the identified studies and extracted data by using a predefined form. The results were confirmed by a third reviewer (JL). The following information was abstracted from each eligible study: first author's name, publication year, study location, cutoff value, and patient quantity. The odds ratios (ORs) and 95% CIs obtained directly from published articles were integrated in the meta-analysis in accordance with the study conducted.
The quality of the eligible studies in this present meta-analysis was assessed independently by 2 reviewers (YHL and YFY) in accordance with the Newcastle–Ottawa Scale (NOS).[17] Studies with scores of ≥5 were considered of high quality. Disagreements on the quality assessment of studies were solved through discussion.
2.3. Statistical analysis
All analyses were conducted using STATA/MP 14.2 (StataCorp, College Station, TX). The prognostic value of NLR in this meta-analysis was determined with the pooled ORs and corresponding 95% CIs. Heterogeneity between studies was evaluated with Cochran Q test and I2 test. Fixed-effect models were adopted only for a P > .1 or I2 < 50%. Otherwise, the random-effect models were applied for the calculation of the pooled HR. To assess a small study effect, we used Egger and Begg test to detect asymmetry in the funnel plot. Because the number of included studies reported DFS and OS was too small, we did not assess meta-regression analyses, publication bias, and sensitivity analysis. All statistical tests were 2-sided, and a P value of < .05 was considered statistically significant.
3. Results
3.1. Literature search
The initial search returned 122 studies, and additional 6 studies were identified by manual search. After 22 duplicated studies were removed, 106 studies were screened, and 91 studies were excluded because of their irrelevant titles or abstracts. Of the remaining 15 potential full-text studies, 2 studies had incomplete data, and 2 were conference abstracts. Eventually, 11 publications were determined eligible for the present pooled analysis. The literature search process was summarized in a flow diagram on the basis of PRISMA (Fig. 1).
Figure 1.
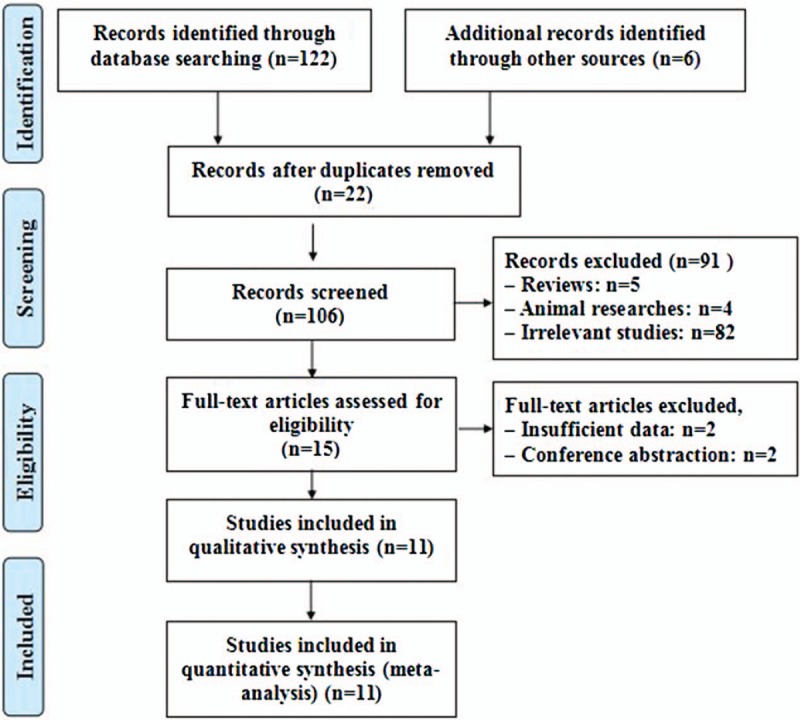
Flow diagram of the inclusion and exclusion of studies.
3.2. Characteristics of the included studies
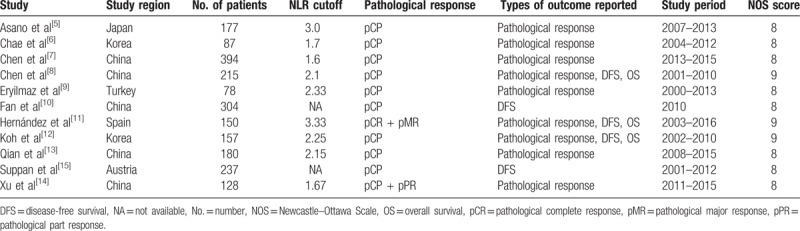
The characteristics of the included studies are listed in Table 1. The 11 studies included 2107 patients with breast cancer reported worldwide: 5 from China, 2 from Korea, 1 from Turkey, 1 from Spain, 1 from Japan and 1 from Austria, and these studies were published from 2011 to 2018. The subject number of studies varied from 78 to 394, with a mean size of 191. Nine of the studies reported the pathological response of patients on the basis of NLR levels, 5 also reported the DFS, and 3 studies reported the OS.
Table 1.
Characteristics of the included studies.
3.3. High NLR and pathological response or prognosis
A meta-analysis showed that high NLR is significantly associated with poor NAC response (OR = 2.27, 95% CI: 1.46–3.53, P < .001; I2 = 58.6%, P = .013; Fig. 2A) but not with DFS (OR = 1.18, 95% CI: 0.78–1.78, P = .435; I2 = 72.1%, P = .006; Fig. 2B) and OS (OR = 2.781, 95% CI: 0.54–14.32, P = .221; I2 = 73.3%, P = .024; Fig. 2C).
Figure 2.
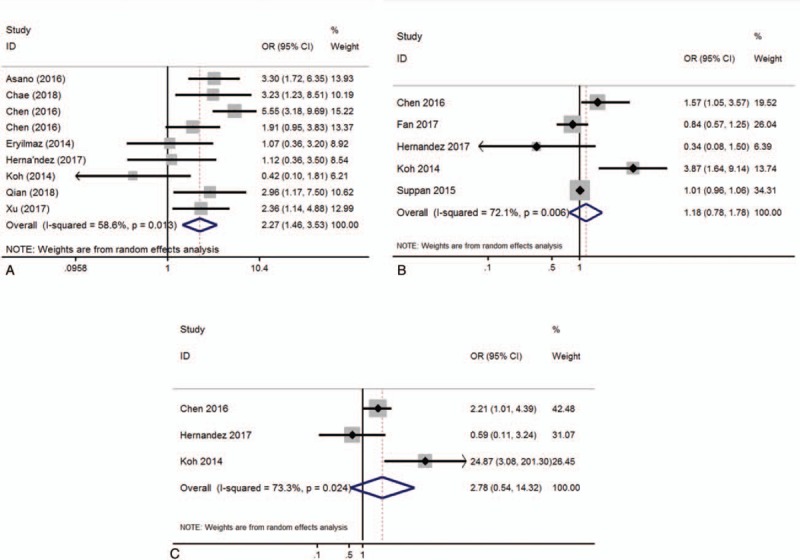
Forest plot of the association of high NLR and prognostic role. (A) Pathological response, (B) DFS, and (C) OS.
3.4. Meta-regression analyses
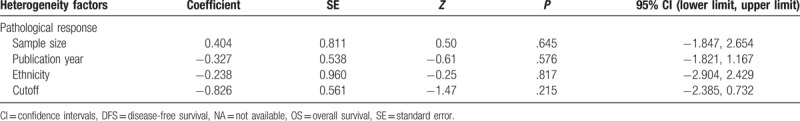
To address the heterogeneity among analyzed studies, we performed meta-regression analyses by sample size (≤100 or >100), ethnicity (Asian or Non-Asian), publication year (before 2016 or after 2016), and cutoff value (≤2.0 or >2.0), but did not account for the heterogeneity source (Table 2).
Table 2.
Meta-regression analysis of potential sources of heterogeneity.
3.5. Sensitivity analysis
Given that heterogeneity was observed in the meta-analysis, sensitivity analysis was performed for the studies included in this work. Figure 3 shows that the results of most of the included studies are close to the central line without obvious deviation.
Figure 3.
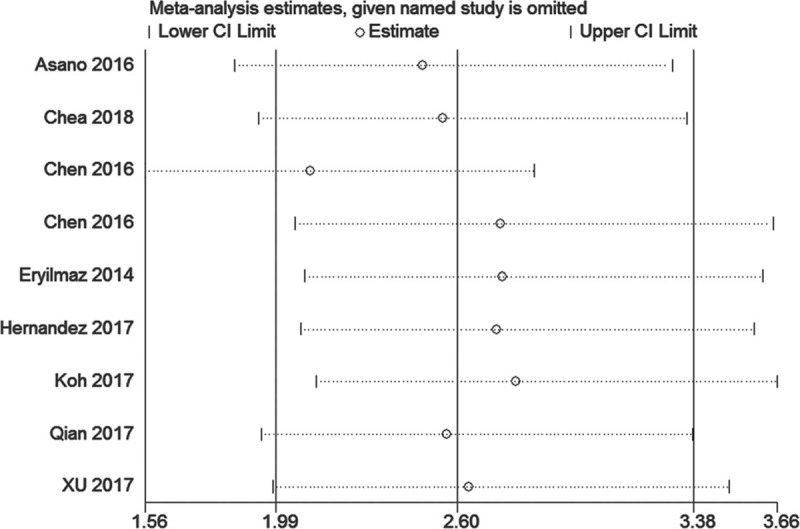
Funnel plots of pathological response for publication bias.
3.6. Publication bias
No evidence of publication bias was observed on the basis of visual inspection of the funnel plots or Begg test for pathological response (P = .917; Fig. 4).
Figure 4.
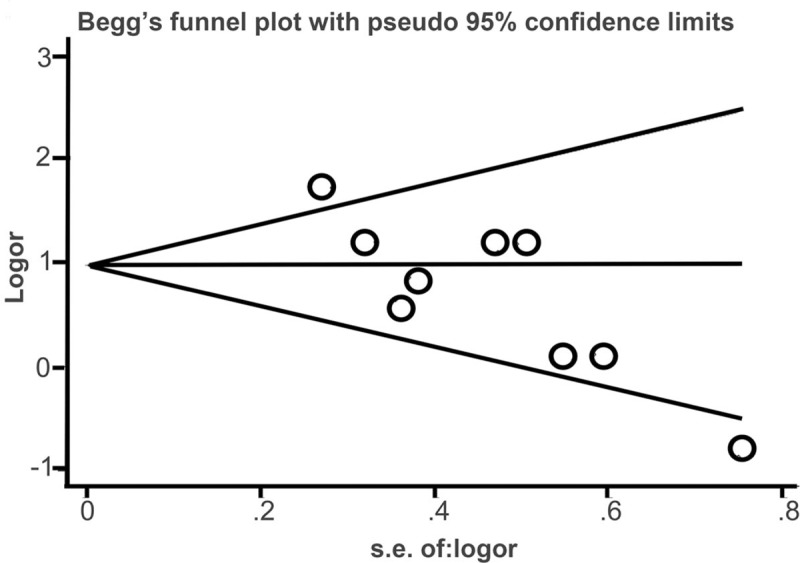
Sensitivity analysis of the relationships between high NLR and the pathological response.
4. Discussion
Systemic inflammation plays an important role in promoting tumor progression. Most cancers develop at infection regions, such as chronic irritation and inflammation. Tumor microenvironments regulated by inflammatory cells clearly play a basic role in the neoplastic process, stimulation of proliferation, and in migration and survival. Numerous studies showed that elevated inflammatory markers, such as NLR and platelet/lymphocyte ratio, are associated with poor prognosis in patients with breast cancer, but the exact results are still undefined.
In this meta-analysis, we evaluate the correlation of NLR with pathological response by analyzing 9 related researches. The result revealed that higher NLR was significantly associated with poor pathological response in patients with breast cancer; this finding validates several previous studies.[5–14] By contrast, our results differed from those of Li et al,[18] who found high NLR not to associate with poor NAC response. There are 3 potential reasons for this result that is different from the previous systematic review. First, this discrepancy may be due to that we directly extracted HRs and 95% CIs from the original study and did not calculate HRs and 95% CIs from the Kaplan–Meier curves. Second, the previous meta-analysis[18] combined full-text studies with abstracts, but our meta-analysis synthesized full-text studies only. Third, we included 2 latest studies published in 2018, which can lead to conflict with research by Li et al.[18] We then evaluated the relationship between NLR and prognostic significance in patients with breast cancer undergoing NAC. Contrary to our expectations, the pooled data indicated that high NLR was not associated with FS (P = .435) and OS (P = .221). This observation coincides with the results of Li et al.[18] These studies showed that NLR alone without any other inflammatory markers is insufficient to provide sufficient information to clinicians as a long-term prognostic marker.
Heterogeneity is a potential problem that can influence the incorporation effect and the interpretation of results. Because significant heterogeneity was present in this analysis, our study also explored factors that may be a source of heterogeneity by meta-regression analysis. Although the specific covariates of patient and study were examined, none that can affect NLR accuracy was found. In addition, considering the restriction of the number of included studies, we were unable to perform a meta-regression analysis for different clinical characteristics. Thus, the findings presented herein should be interpreted cautiously.
This meta-analysis holds some limitations that need to be addressed in future studies. First, the obvious limitation of this study was a heavy dominance of Asian studies. Given the racial differences in breast cancer, studies from various countries are necessary. Second, the ability to assess the accuracy of NLR may be decreased and may result in publication bias and heterogeneity considering the relatively few number of studies (i.e., 9 for pathological response, 5 for DFS, and 3 for OS). Finally, the presence of other diseases in addition to cancer, such as coronary artery disease, hepatic disease, metabolic syndrome, and any inflammation-related diseases, can alter the level of the NLR. This alteration may have affected our results.
5. Conclusion
Our data support high NLR as a useful predictive factor for assessing treatment response to NAC in patients with breast cancer but do not show association with DFS and OS. Future prospective studies with large sample sizes and enhanced study designs are required to confirm our findings.
Acknowledgment
The authors desire to thank Shunyi Feng for aiding in the statistical analysis.
Author contributions
Conceptualization: Ling Bo Xue, Yong Hong Liu, Bo Zhang, Yan Fang Yang, Jian Jin, Jie Li.
Data curation: Bo Zhang, Yan Fang Yang.
Formal analysis: Li Wei Zhang, Jian Jin.
Investigation: Dong Yang.
Methodology: Dong Yang, Li Wei Zhang, Jian Jin.
Project administration: Ling Bo Xue, Bo Zhang, Yan Fang Yang.
Writing – original draft: Yong Hong Liu.
Writing – review & editing: Jie Li.
Jie Li orcid: 0000-0002-0216-4995.
Footnotes
Abbreviations: CI = confidence interval, NAC = neoadjuvant chemotherapy, NLR = neutrophil-to-lymphocyte ratio, OR = odds ratio.
The authors have no competing interest.
References
- [1].Mersin H, Yildirim E, Berberoglu U, et al. The prognostic importance of triple negative breast carcinoma. Breast 2008;17:341–6. [DOI] [PubMed] [Google Scholar]
- [2].Chaudry M, Lei X, Gonzalezangulo AM, et al. Recurrence and survival among breast cancer patients achieving a pathological complete response to neoadjuvant chemotherapy. Breast Cancer Res Treat 2015;153:417–23. [DOI] [PubMed] [Google Scholar]
- [3].Montagna E, Bagnardi V, Rotmensz N, et al. Pathological complete response after preoperative systemic therapy and outcome: relevance of clinical and biologic baseline features. Breast Cancer Res Treat 2010;124:689–99. [DOI] [PubMed] [Google Scholar]
- [4].Cheng X, Li Y, Liu B, et al. 18F-FDG PET/CT and PET for evaluation of pathological response to neoadjuvant chemotherapy in breast cancer: a meta-analysis. Acta Radiol 2012;53:615–27. [DOI] [PubMed] [Google Scholar]
- [5].Asano Y, Kashiwagi S, Onoda N, et al. Predictive value of neutrophil/lymphocyte ratio for efficacy of preoperative chemotherapy in triple-negative breast cancer. Ann Surg Oncol 2016;23:1104–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [6].Chae S, Kang KM, Kim HJ, et al. Neutrophil-lymphocyte ratio predicts response to chemotherapy in triple-negative breast cancer. Curr Oncol 2018;25:e113–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [7].Chen MS, Zhang YL, Yang HW, et al. Predictive value of peripheral blood neutrophil to lymphocyte ratio and platelet to lymphocyte ratio in breast cancer patients with neoadjuvant chemotherapy. J Chin Oncol 2017;23:180–4. [Google Scholar]
- [8].Chen Y, Chen K, Xiao X, et al. Pretreatment neutrophil-to-lymphocyte ratio is correlated with response to neoadjuvant chemotherapy as an independent prognostic indicator in breast cancer patients: a retrospective study. BMC Cancer 2016;16:320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [9].Eryilmaz MK, Mutlu H, Salim DK, et al. The neutrophil to lymphocyte ratio has a high negative predictive value for pathologic complete response in locally advanced breast cancer patients receiving neoadjuvant chemotherapy. Asian Pac J Cancer Prev 2014;15:7737–40. [DOI] [PubMed] [Google Scholar]
- [10].Fan M, Ouyang T, He Y, et al. Correlation of baseline neutrophil-to-lymphocyte ratio with the curative effect and prognosis of breast cancer patients after neoadjuvant chemotherapy. Chin J Clin Lab Sci 2017;35:114–7. [Google Scholar]
- [11].Hernández CM, Madrona AP, Vázquez PJG, et al. Usefulness of lymphocyte-to-monocyte, neutrophil-to-monocyte and neutrophil-to-lymphocyte ratios as prognostic markers in breast cancer patients treated with neoadjuvant chemotherapy. Clin Transl Oncol 2017;20:476–83. [DOI] [PubMed] [Google Scholar]
- [12].Koh YW, Lee HJ, Ahn JH, et al. Prognostic significance of the ratio of absolute neutrophil to lymphocyte counts for breast cancer patients with ER/PR-positivity and HER2-negativity in neoadjuvant setting. Tumor Biol 2014;35:9823–30. [DOI] [PubMed] [Google Scholar]
- [13].Qian Y, Tao J, Li X, et al. Peripheral inflammation/immune indicators of chemosensitivity and prognosis in breast cancer patients treated with neoadjuvant chemotherapy. Onco Targets Ther 2018;11:1423–32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [14].Xu J, Ni C, Ma C, et al. Association of neutrophil/lymphocyte ratio and platelet/lymphocyte ratio with ER and PR in breast cancer patients and their changes after neoadjuvant chemotherapy. Clin Transl Oncol 2017;19:989–96. [DOI] [PubMed] [Google Scholar]
- [15].Suppan C, Bjelic-Radisic V, Garde ML, et al. Neutrophil/lymphocyte ratio has no predictive or prognostic value in breast cancer patients undergoing preoperative systemic therapy. BMC Cancer 2015;15:1027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [16].Elm E, Altman D, Egger M, et al. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: guidelines for reporting observational studies. Bull World Health Organ 2007;85:867–72. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [17].Oremus M, Oremus C, Hall GB, et al. Team ECSR. Inter-rater and test-retest reliability of quality assessments by novice student raters using the Jadad and Newcastle-Ottawa Scales. BMJ Open 2012;2:e001368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [18].Li X, Dai D, Chen B, et al. The value of neutrophil-to-lymphocyte ratio for response and prognostic effect of neoadjuvant chemotherapy in solid tumors: a systematic review and meta-analysis. J Cancer 2018;9:861–71. [DOI] [PMC free article] [PubMed] [Google Scholar]


