Abstract
Ventilatory parameters obtained during exercise predict survival in several chronic diseases; however, long-term changes in exercise ventilatory parameters in patients with CF have yet to be examined and potential differences between sexes in CF are unknown.
Purpose:
we sought to examine the change in exercise ventilatory parameters over time in patients with CF and determine if the change is different between sexes.
Methods:
exercise capacity (VO2 peak) and exercise ventilatory parameters (VE/VO2 peak, VE/VCO2 peak, and VE/VCO2 slope) were determined from a maximal cardio-pulmonary test on a cycle ergometer on two visits separated by 39 ± 16 months in 20 patients with CF (10 female, 10 male).
Results:
no differences between sexes were observed at visit 1 (all p>0.05). Overall, exercise ventilatory parameters significantly (p<0.05) deteriorated between visits, with no change (p>0.05) in VO2 peak. Moreover, compared with males, female patients exhibited greater deteriorations in VE/VO2 peak (p=0.001), VE/VCO2 peak (p=0.002), and VE/VCO2 slope (p=0.016) between visits.
Conclusions:
these data in patients with CF indicate that exercise ventilatory parameters decline over time despite no change in VO2 peak, and female patients exhibit a more rapid deterioration compared with males.
Keywords: exercise capacity, pulmonary function, ventilatory efficiency
Introduction
A hallmark characteristic of cystic fibrosis (CF) is exercise intolerance, exhibited by a reduction in exercise capacity (VO2 peak) (Lands et al. 1992; Troosters et al. 2009) that predicts mortality, independent of lung function (Nixon et al. 1992; Pianosi et al. 2005). Several factors contribute to exercise intolerance in CF including skeletal muscle dysfunction, dysfunctional oxygen uptake kinetics, and abnormal pulmonary mechanics (Fielding et al. 2015; Gruet et al. 2017; Troosters et al. 2009; McKone et al. 2005). However, while the accelerated long-term decline in lung function and exercise capacity in patients with CF has been well-documented (Corey et al. 1976; Penketh et al. 1987; Cantin 1995; Pianosi et al. 2005), long-term changes in ventilatory responses to exercise in CF remain unclear.
The slope of minute ventilation to ventilatory CO2 output during exercise (VE/VCO2) has important prognostic value in patients with heart failure and pulmonary hypertension, even outperforming VO2 peak as a predictor of survival (Kleber et al. 2000; MacGowan and Murali 2000; Schwaiblmair et al. 2012). The high prognostic value of the VE/VCO2 slope in other clinical populations may be partly explained by the fact that it is effort-independent, and thus less susceptible to false interpretation due to early test termination either by the patient or investigator. Indeed, achieving maximal effort during a CPET is dependent on a range of factors including the testing protocol used, intrinsic motivation of the participant, and the participant’s perceived exertion (Takken et al. 2017). Routine exercise testing in CF is now recommended (Hebestreit et al. 2015); therefore, monitoring changes in exercise ventilatory parameters may provide enhanced prognostic value in CF compared with traditional exercise outcomes such as VO2 peak.
A substantial body of evidence demonstrates higher mortality in female patients with CF compared with their male counterparts (Dodge et al. 2007; MacKenzie et al. 2014; Rosenfeld et al. 1997); a phenomenon which is not explained by factors including nutritional status, pulmonary function, or airway microbiology (FitzSimmons 1993). Across varying levels of disease severity (classified by forced expiratory volume in one second [FEV1]), lower absolute peak workload and relative VO2 peak were both observed in female patients with CF compared with males (Orenstein and Nixon 1991). In healthy children, sex has been implicated as a potential modifier of exercise ventilatory dynamics. Smaller females exhibit a higher VE for a given VO2 compared to larger females, while no such trend exists in males (Godfrey et al. 1971). In CF, longitudinal data in young adults suggest a similar degree of change between sexes in lung function, exercise capacity, and VE peak (Moorcroft et al. 1997). While several indices of ventilatory efficiency during exercise have been shown to be similar between sexes at a single time point (Orenstein and Nixon 1991), it is unknown how sex influences long-term changes in exercise ventilatory parameters, particularly as disease severity worsens. Therefore, the purpose of this study was to examine longitudinal changes in exercise ventilatory parameters in patients with CF and compare the magnitude of change between sexes.
Methods
Participants
Data from 20 patients with CF (10 males, 10 females) age 8 – 42 years old were available for this retrospective analysis. Participants were excluded if they 1) had a FEV1 < 50% of predicted, 2) had a resting oxygen saturation (SpO2) < 85%, 3) self-reported to be a smoker, 4) were diagnosed with pulmonary hypertension, 5) were diagnosed with sleep apnea or sleep disorders, 6) had a clinical diagnosis of cardiovascular disease, hypertension, or CF-related diabetes, or 7) were prescribed any vaso-active medications (i.e., nitrates, beta blockers, ACE inhibitors, etc.).
All data were routinely collected in the Laboratory of Exercise and Vascular Physiology at the Georgia Prevention Institute from patients who were participating in various ongoing research studies. Non-interventional patient data (i.e., observational data collection only) on two separate visits (visit 1 [baseline] and visit 2), were required for the present analysis. Patients were only tested if they had been clinically stable (no exacerbations, need for antibiotic treatment, or major change in medical status) for at least two weeks prior to testing. In addition, data were included from patients who were taking standard CF therapy (i.e. bronchodilators, daily multivitamin supplements (e.g., AquADEK), and pancreatic enzyme supplements). Data from patients who were on CFTR modulators (e.g., lumacaftor, ivacaftor); however, were not included in the analysis. Data were collected over a six year period between December 2010 and November 2016. All protocols were approved by the Institutional Review Board at Augusta University and all methods were performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki. Informed assent/consent was obtained by all participants and parents of minors prior to participation.
Participant characteristics and pulmonary function testing
Participant testing included assessments of height, weight, and calculated BMI. Body composition (fat percent and fat-free mass [FFM]) was determined using dual-energy X-ray absorptiometry (DXA; QDR-4500W, Hologic, Waltham, MA). Routine spirometric function testing was performed according to the American Thoracic Society standards (American Thoracic Society 1995). Percent predicted data were determined using the current reference standards available at the time of testing (National Health and Nutrition Examination Survey [NHANES] III (Hankinson et al. 1999) or the 2012 European Respiratory Society Task Force guidelines (Quanjer et al. 2012)) .
Cardio-pulmonary exercise test (CPET)
At each visit, a CPET was performed on a cycle ergometer using the Godfrey protocol to determine exercise capacity as previously described by our group (Tucker et al. 2017). Briefly, expired gases were analyzed in a mixing chamber by a TruOne® 2400 metabolic cart (ParvoMedics, Sandy, UT) and analyzed as 30 second averages to obtain VO2 peak and peak minute ventilation (VE peak). Ventilatory threshold (VT) was determined using the v-slope method (Beaver et al. 1986) for calculation of percent VO2 peak at VT. Additional ventilatory parameters including the ratio of VE to both VO2 and carbon dioxide output (VCO2) at peak exercise (VE/VO2 peak and VE/VCO2 peak, respectively), end-tidal CO2 (PETCO2), and the VE/VCO2 slope, were determined according to previously published methodology (Schwaiblmair et al. 2012; Fielding et al. 2015). Maximum voluntary ventilation (MVV) was calculated as 27.7(FEV1) + 8.8(predicted FEV1) for patients aged ≤18 years (Stein et al. 2003) or FEV1 × 40 for patients aged >18 years (Wasserman et al. 2005) and expressed relative to peak VE (VE/MVV). Respiratory rate (RR), tidal volume (VT, corrected for dead space of the breathing apparatus), and fractional expired CO2 (FeCO2) were determined from the metabolic cart at rest and during exercise.
Statistical analyses
All analyses were performed using SPSS version 24 (IBM Corporation, Somers, NY). Descriptive statistics were generated and range as well as normality checks performed. Two-way repeated measures ANOVA (sex by time) were used to determine group differences in the change over time for all outcomes. Preliminary analyses revealed non-significant associations between the time between visits 1 and 2 and the primary outcome variables (changes in lung function, VO2 peak, and the VE/VCO2 slope). Therefore, time between visits was not included as a covariate in the regression model. The Greenhouse-Geisser correction was applied when the assumption of sphericity was violated. Effect sizes (partial eta squared []) are reported for the interaction terms of the ANOVA, where values of 0.01, 0.06, and 0.14 correspond to small, medium, and large effects, respectively (Cohen 1988). Pearson correlations were used to examine associations between baseline disease severity (FEV1, % predicted) and changes in exercise ventilatory parameters. Comparisons of baseline (visit 1) participant characteristics were performed using independent group t-tests. DXA scans were unable to be completed on two female patients, therefore values of FFM, body fat %, and VO2 relative to FFM are presented with an n=8 in this group. An alpha <0.05 was considered statistically significant for all analyses. Data are presented as mean ± standard deviation (SD) unless stated otherwise.
Results
Participant characteristics
The majority (15/20) of patients were homozygous for the ΔF508 CFTR mutation (seven males, eight females), two were ΔF508/G551D (one male, one female), two were ΔF508/621+1G->T (one male, one female), and one male was homozygous for (TG)11-5T. Participant characteristics are presented in Table 1. At visit 1, severity of lung disease was normal in 12 patients (seven males, five females), mild in six patients (two males, four females), and moderate in two patients (one male, one female). At visit 2, disease severity was normal in seven patients (five males, two females), mild in seven patients (two males, five females), and moderate in six patients (three male, three female). There were no differences in age (p=0.217), BMI (p=0.228), FEV1 (p=0.215), or FEV1/FVC (p=0.733) between males and females. At visit 1, height (p=0.004), weight (p=0.013), FFM (p<0.001), and FVC (p=0.001) were all higher in males versus females, whereas % body fat was lower in males (p<0.001). Overall, the average collection time between visits was 39 ± 16 (range 7 – 71) months. Importantly, the average time between visits was similar (p=0.222) between males and females (35 ± 20 [range 7 – 71] vs. 43 ± 10 [range 26 – 54] months, respectively).
Table 1.
Participant demographics and parameters of exercise capacity collected from the CPET at visits 1 and 2.
| Visit 1 |
Visit 2 |
Change |
||||
|---|---|---|---|---|---|---|
| Variable | Males | Females | Males | Females | Males | Females |
| Demographics | ||||||
| Age (y) | 20.4 ± 11.4 | 15.1 ± 6.9 | 23.2 ± 10.9† | 18.7 ± 6.0† | 2.8 ± 1.6 | 3.6 ± 1.3 |
| Height (cm) | 169 ± 17 | 148 ± 11 * | 171 ± 14† | 156 ± 5† | 2 ± 4 | 9 ± 9 |
| Weight (kg) | 60.6 ± 16.2 | 42.6 ± 12.8* | 67.3 ± 15.3† | 49.3 ± 8.9† | 6.7 ± 5.0 | 6.8 ± 8.8 |
| BMI (kg/m2) | 20.8 ± 3.0 | 19.1 ± 3.1 | 22.7 ± 2.9† | 19.6 ± 3.9† | 1.9 ± 1.5 | 0.5 ± 2.6 |
| Fat-free mass (kg)a | 48.5 ± 12.5 | 30.4 ± 5.9* | 48.7 ± 11.7 | 31.5 ± 2.9 | 0.2 ± 3.2 | 1.1 ± 4.6 |
| Body fat (%)a | 14.5 ± 3.0 | 27.0 ± 7.2* | 22.3 ± 5.8† | 33.3 ± 4.7† | 7.8 ± 4.8 | 6.3 ± 4.7 |
| FVC (L) | 4.69 ± 1.47 | 2.74 ± 0.58* | 4.60 ± 1.62 | 3.01 ± 0.52 | −0.10 ± 0.47 | 0.27 ± 0.35 |
| FEV1 (% predicted) | 96.1 ± 20.8 | 85.4 ± 16.1 | 85.6 ± 24.3† | 77.6 ± 13.5† | −10.5 ± 9.1 | −7.8 ± 8.3 |
| FEV1/FVC | 0.78 ± 0.10 | 0.77 ± 0.06 | 0.76 ± 0.1 | 0.77 ± 0.05 | −0.02 ± 0.07 | 0.01 ± 0.04 |
| Exercise Parameters | ||||||
| VO2 peak (L/min) | 2.08 ± 0.72 | 1.20 ± 0.21* | 2.15 ± 0.64 | 1.34 ± 0.18 | 0.07 ± 0.38 | 0.14 ± 0.16 |
| VO2 peak (ml/min/kg) | 34.1 ± 6.2 | 29.4 ± 6.0 | 32.3 ± 5.8 | 27.5 ± 4.0 | −1.9 ± 5.6 | −1.9 ± 3.8 |
| VO2 peak (ml/min/kgFFM)a | 42.4 ± 7.2 | 41.4 ± 6.0 | 44.6 ± 8.0 | 43.1 ± 4.4 | 2.2 ± 9.2 | 1.8 ± 4.3 |
| VO2 peak (% predicted) | 79.6 ± 14.1 | 67.6 ± 7.2* | 79.9 ± 15.9 | 74.3 ± 8.3 | 0.3 ± 11.4 | 6.7 ± 7.8 |
| VO2 peak at VT (%) | 55.4 ± 9.2 | 56.5 ± 5.6 | 53.7 ± 13.7 | 61.2 ± 6.2 | −1.7 ± 12.0 | 4.7 ± 9.5 |
| VE (L/min) | 74.2 ± 30.1 | 41.3 ± 13.7* | 83.6 ± 18.4† | 64.9 ± 10.0† | 9.4 ± 27.6 | 23.6 ± 9.9 |
| VE/MVV (%) | 63.7 ± 23.1 | 59.9 ± 17.2 | 80.5 ± 28.6† | 88.9 ± 22.2† | 16.8 ± 29.8 | 29.0 ± 12.6 |
| VE/VCO2 slope at VT | 21.1 ± 4.6 | 21.7 ± 2.0 | 21.0 ± 3.1 | 23.0 ± 2.9 | 0.0 ± 5.2 | 1.3 ± 2.9 |
| Workload at VT (W) | 83 ± 38 | 41 ± 14* | 80 ± 30 | 57 ± 16 | −6 ± 19 | 13 ± 20 |
| Peak work (W) | 157 ± 48 | 95 ± 27* | 169 ±39† | 117 ± 19† | 12 ± 21 | 22 ± 17 |
| Peak work (W/kg) | 2.6 ± 0.3 | 2.4 ± 0.6 | 2.5 ± 0.3 | 2.4 ± 0.3 | 0.0 ± 0.4 | 0.0 ± 0.6 |
| Max heart rate (bpm) | 165 ± 23 | 168 ± 14 | 169 ± 17 | 172 ± 13 | 4 ± 20 | 5 ± 20 |
Values are mean ± SD. BMI = body mass index; FEV1 = forced expiratory volume in 1 second; FFM = fat-free mass; FVC = forced vital capacity; MVV = maximum voluntary ventilation; VT = ventilatory threshold; VE = peak ventilation. an=8 in females.
Significant difference versus males at visit 1 (p<0.05).
Significant change from visit 1, independent of sex (p<0.05).
Age, height, weight, BMI, and % body fat all increased between visits, independent of sex (all p<0.05). Overall, FEV1 (% predicted) decreased over time (F(1,18)=22.1, p<0.001); however, FFM (F(1,18)=39.7, p=0.496), FVC (F(1,18)=39.7, p=0.357), and FVC/FEV1 (F(1,18)=39.7, p=0.548) did not change. No differences between sexes were observed for changes in any other variable (all p>0.05).
Ventilatory parameters during exercise and exercise capacity
At visit 1, VE/VO2 peak (33.4 ± 6.9 vs. 36.1 ± 8.1, p=0.432), VE/VCO2 peak (30.5 ± 3.9 vs. 30.4 ± 3.7, p=0.954), and the VE/VCO2 slope (28.5 ± 3.8 vs. 27.8 ± 3.5, p=0.705) were not different between females and males, respectively. Overall, VE/VO2 peak (Δ10.0 ± 8.1, F(1,18)=54.6, p<0.001), VE/VCO2 peak (Δ4.9 ± 4.4, F(1,18)=39.7, p<0.001), and the VE/VCO2 slope (Δ4.2 ± 3.6, F(1,18)=36.8, p<0.001) all significantly increased over time. Females, however, exhibited a significantly greater change in VE/VO2 peak (F(1,18)=16.4, p=0.001, =0.48), VE/VCO2 peak (F(1,18)=12.8, p=0.002, =0.42), and the VE/VCO2 slope (F(1,18)=7.0, p=0.016, =0.28) compared to males (Figure 1). In contrast, absolute VO2 peak (F(1,18)=2.4, p=0.138), VO2 peak relative to body mass (F(1,18)=3.0, p=0.100) and FFM (F(1,16)=1.3, p=0.276) did not change over time, nor were there any differences in the magnitude of change between sexes (p=0.614, 0.996, and 0.903, respectively). At visit 1, no differences in RR were present between males and females at rest (21 ± 4 vs. 20 ± 6 bpm, p=0.585), during VT (23 ± 8 vs. 24 ± 6 bpm, p=0.728), or at peak exercise (38 ± 10 vs. 40 ± 8 bpm, p=0.612). However, RR increased significantly between visits in females relative to males at rest (Δ6 ± 4 vs. −2 ± 5 bpm, F(1,18)=13.9, p=0.002, =0.44) and during VT (Δ3 ± 5 vs. −3 ± 5 bpm, F(1,18)=7.2, p=0.015, =0.29), but not at peak exercise (Δ8 ± 7 vs. 5 ± 13 bpm, F(1,18)=0.5, p=0.494, =0.03).
Figure 1.
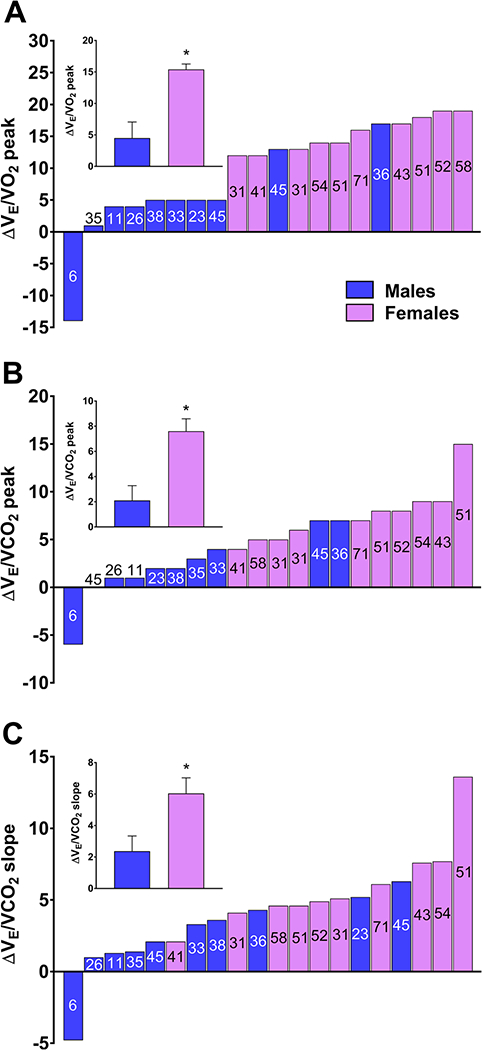
Individual and mean (inset) changes in the ratio of minute ventilation (VE) to oxygen uptake (VO2) at peak exercise (A), VE to carbon dioxide output (VCO2) at peak exercise (B), and the VE to VCO2 slope (C) in male and female patients between visits 1 and 2. The time between visits (months) are presented with each column for each patient. *significantly greater in females versus males (p≤0.016).
As illustrated in Figure 2, while FeCO2 at peak exercise was similar between groups at visit 1 (p=0.725), there was a significantly greater reduction (F(1,18)=6.4, p=0.021, =0.26) between visits in female patients, although FeCO2 was not significantly different (p=0.073) between groups at visit 2 (Figure 2A). Similarly, there were no differences in peak PETCO2 between groups at visit 1 (p=0.715), however, females exhibited a significantly greater decline (F(1,18)=12.9, p=0.002, =0.42) and lower peak PETCO2 at visit 2 (p=0.017; Figure 2B).
Figure 2.
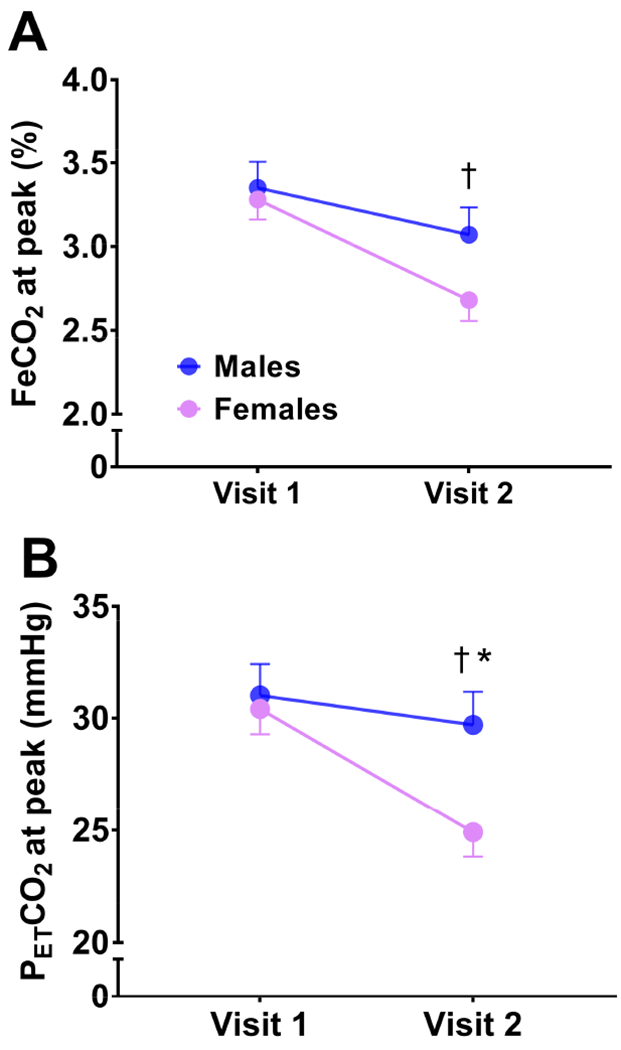
Group changes between visits in FeCO2 at peak exercise (A) and PETCO2 at peak exercise (B). †significant group by time interaction; *significant difference between groups at visit 2 (p=0.017).
Independent of sex, VE peak (F(1,18)=12.7, p=0.002), VE/MVV (F(1,18)=20.0, p<0.001), and absolute peak workload (F(1,18)=15.6, p=0.001) significantly increased over time (Table 1). There were no significant changes over time in % predicted VO2 peak (F(1,18)=2.6, p=0.126), VO2 peak at VT (F(1,18)=0.4, p=0.542), workload at VT (F(1,17)=0.7, p=0.409), VE/VCO2 slope at VT (F(1,18)=0.4, p=0.512), relative peak workload (F(1,18)=0.0, p=0.964), or max heart rate during exercise (F(1,18)=0.9, p=0.364). In addition, the magnitude of change for these variables was not different between sexes (all p>0.05).
Relationships between disease severity and FeCO2 and changes in ventilatory parameters during exercise
As illustrated in Figure 3, in the overall sample (males and females combined), there were significant associations between baseline disease severity (FEV1, % predicted) and changes in exercise ventilatory parameters. Specifically, baseline FEV1 was negatively associated with changes in VE/VCO2 peak (r=−0.545, p=0.013; Figure 3A) and the VE/VCO2 slope (r=−0.469, p=0.037; Figure 3B) but not VE/VO2 peak (r=−0.404, p=0.077; Figure 3C). Changes in FEV1 were not significantly associated with concomitant changes in exercise ventilatory parameters (all p>0.05). Significant negative associations were also present between changes in FeCO2 at peak (%) and changes in VE/VO2 peak (r=−0.842, p<0.001; Figure 3D), VE/VCO2 peak (r=−0.772, p<0.001; Figure 3E) and the VE/VCO2 slope (r=−0.742, p<0.001; Figure 3F).
Figure 3.
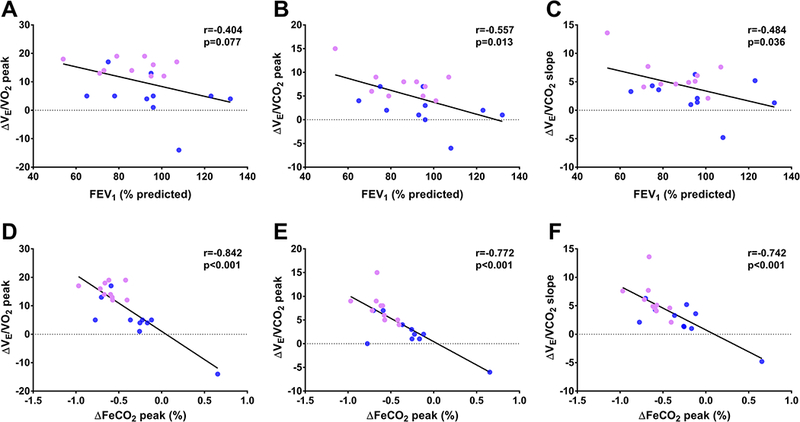
Associations between baseline FEV1 (% predicted) and changes in exercise ventilatory parameters (A, B, and C) and between changes in FeCO2 at peak and exercise ventilatory parameters (D, E, and F). Lilac and blue dots indicate female and male patients, respectively.
Discussion
Exercise capacity (VO2 peak) predicts survival in patients with CF. Exercise ventilator parameters provide prognostic utility in other diseases (Guazzi et al. 2003; MacGowan and Murali 2000; Schwaiblmair et al. 2012) and data from the present study indicate that patients with CF exhibit a deterioration in exercise ventilatory parameters over a period of ∼3 years, despite non-significant changes in VO2 peak. In addition, female patients exhibited a more pronounced deterioration in exercise ventilatory parameters compared with males.
The ventilatory response during exercise evaluates ventilatory efficiency and can be influenced by factors such as CO2 production and the ventilatory dead space to tidal volume (VD/VT) ratio (Guazzi et al. 2003). In the present study, there was an overall worsening in exercise ventilatory parameters over time, with no significant change in VO2 peak. The second novel finding is that female patients exhibited a significantly greater deterioration in exercise ventilatory parameters compared with males (Figure 1), despite similar rates of decline in lung function and VO2 peak. Importantly, the average time between visits was not significantly different between males and females (p=0.222). Moreover, even when controlling for this factor by including it as a covariate in the regression model, the significant sex by time interaction remained for changes in exercise ventilatory parameters, suggesting that the time between visits did not significantly influence our findings. This sex disparity may be explained, in part, by the observed reduction in CO2 production and/or reduced chemosensitivity to arterial PCO2 (PaCO2) during exercise in females relative to males. In the present study, RR increased between visits in females compared to males at rest (Δ6 vs. −2 bpm) and during VT (Δ3 vs. −3 bpm), and females exhibited a significantly lower FeCO2 at rest (2.23 vs. 2.75%) and tendency for lower FeCO2 at peak exercise (2.67 vs. 3.07%, p=0.073) compared to males at visit 2 (Figure 2A). Previous work in children and young adults with CF suggests that as disease severity worsens (demonstrated via declining FEV1) there is an increase in the respiratory muscle load that is predominated by decreased lung compliance (Hart et al. 2002). As a compensatory mechanism, respiratory rate increases to maintain ventilation but is unable to efficiently clear CO2. While not significantly different at either visit 1 or visit 2, FEV1 (% predicted) was lower in female patients by ∼9-10 percentage points. This raises the possibility that disease severity progressed to a point in female patients between visits where lung compliance was compromised enough to impair ventilatory mechanics during exercise, possibly due to changes in airway obstruction and/or mucus plugging. Indeed, increased airflow accompanying the increased RR in females may have also contributed to greater airway dead space (Thin et al. 2004). Unfortunately, using end-tidal PCO2 to estimate PaCO2 is unreliable in pulmonary diseases such as CF (Pianosi and Hochman 1996) and therefore VD/VT could not be accurately assessed non-invasively in the present investigation. Future studies examining longitudinal changes in lung compliance/resistance and airway dead space in combination with exercise ventilatory parameters are warranted to further our understanding of sex-mediated changes in exercise ventilatory efficiency in CF.
Previous work suggests that long-term maintenance of exercise capacity in CF may be attributed to a number of factors including improved nutritional status (Moorcroft et al. 1997), typically evidenced via weight gain. Indeed, both groups exhibited significant increases in body mass and BMI over time; however, the change was not different between sexes. Exercise capacity was maintained between visits, yet a steep decline in exercise ventilatory parameters was observed. Accordingly, the decline in ventilatory efficiency during exercise may precede reductions in exercise capacity, suggesting that ventilatory dynamics during exercise may be a more sensitive prognostic indicator of changes in exercise tolerance in CF compared to VO2 peak. In addition, while the longitudinal changes in lung function and exercise capacity in the present study are consistent with a previous report (Moorcroft et al. 1997), the observed sex disparity in exercise ventilation changes is unique to this investigation and may provide further insight into the increased mortality rate that is observed in female patients with CF compared with males (Dodge et al. 2007; MacKenzie et al. 2014).
Longitudinal data from Pianosi et al. indicate that disease severity (FEV1, % predicted) contributes to the rate of decline in exercise capacity (Pianosi et al. 2005). Indeed, findings from the present study follow a similar trend to this previous observation as baseline FEV1 was significantly associated with changes in exercise ventilatory parameters (Figure 3). Accordingly, the present data suggest that a greater rate of decline in exercise ventilatory parameters occurs in patients with greater disease severity, suggesting a role of disease severity as a contributing factor towards the deterioration in ventilatory efficiency during exercise. However, changes in FEV1 over time were not significantly associated with concomitant changes in exercise ventilatory parameters.
Experimental Considerations
Physical activity level has been shown to independently predict exercise capacity in patients with CF (Hebestreit et al. 2006). Given the importance of maintaining VO2 peak in this population (Nixon et al. 1992; Pianosi et al. 2005), regular physical activity is recommended for patients with CF (Philpott et al. 2010). In the present study, concomitant changes in exercise training status and/or habitual physical activity levels were not monitored over the observation period. Thus, it is unclear how potential changes in these variables may have contributed to the observed changes in exercise ventilatory parameters. Moreover, habitual physical activity patterns can be different between sexes, particularly during childhood, as boys tend to be more physically active than girls (Schneiderman-Walker et al. 2005). Future studies should seek to examine longitudinal changes in exercise ventilatory parameters in patients with CF while simultaneously controlling for the added influence of changes in physical activity and/or exercise training status.
Independent of sex, age has been shown to be a major determinant of exercise ventilatory control. Not only do healthy children demonstrate greater CO2 chemosensitivity during exercise compared with adults (Gratas-Delamarche et al. 1993), an age-related decline in ventilatory responses to exercise has also been reported in patients with CF throughout childhood (Pianosi and Wolstein 1996). In the present study, while the difference in age between males and females was not statistically significant at visit 1 (20 vs. 15 years, p=0.217), it could be reasoned that the accelerated decline in ventilatory efficiency in females may have been due to differences in physical maturation compared to their male counterparts. However, there was no association between age at visit 1 and changes in VE/VCO2 slope (r=0.031, p=0.897) suggesting that group differences in maturation was likely not a significant contributing factor.
Conclusions
In summary, data from this study in patients with CF suggest that 1) the worsening in exercise ventilatory parameters may precede the decline in VO2 peak, and 2) females exhibit a greater deterioration in exercise ventilatory parameters over time compared with males, despite similar changes in VO2 peak and lung function. Given the disproportionate mortality risk between sexes in CF, these data support further investigation into the potential prognostic value of exercise ventilatory parameters in patients with CF. Future studies using a larger cohort, coupled with multiple experimental visits and a longer observation are warranted to further our understanding of the interrelationships between sex, disease severity, and long-term changes in exercise ventilatory parameters in patients with CF.
Acknowledgments
Source of Funding: Supported in part by a Vertex Pharmaceuticals Incorporated IIS Grant and NIH/NIDDK R21DK100783 (R.A.H.).
Abbreviations:
- BMI
body mass index
- CF
cystic fibrosis
- CPET
cardio-pulmonary exercise test
- DXA
dual-energy X-ray absorptiometry
- FeCO2
fraction of expired CO2
- FEV1
forced expiratory volume in 1 second
- FFM
fat-free mass
- FVC
forced vital capacity
- MVV
maximum voluntary ventilation
- PaCO2
partial pressure of arterial CO2
- RR
respiratory rate
- VCO2
volume of carbon dioxide output
- VD/VT
dead space to tidal volume ratio
- VE
minute ventilation
- VO2
volume of oxygen consumption
- VT
tidal volume
- VT
ventilatory threshold
Footnotes
Competing Interest
None to declare.
References
- American Thoracic Society (1995) Standardization of Spirometry, 1994 Update. American Thoracic Society. American journal of respiratory and critical care medicine 152 (3):1107–1136 [DOI] [PubMed] [Google Scholar]
- Beaver WL, Wasserman K, Whipp BJ (1986) A new method for detecting anaerobic threshold by gas exchange. Journal of applied physiology 60 (6):2020–2027 [DOI] [PubMed] [Google Scholar]
- Cantin A (1995) Cystic fibrosis lung inflammation: early, sustained, and severe. American journal of respiratory and critical care medicine 151 (4):939–941 [DOI] [PubMed] [Google Scholar]
- Cohen J (1988) Statistical power analysis for the behavioral sciences . Hilsdale. NJ: Lawrence Earlbaum Associates 2 [Google Scholar]
- Corey M, Levison H, Crozier D (1976) Five-to Seven-Year Course of Pulmonary Function in Cystic Fibrosis 1–3. American Review of Respiratory Disease 114 (6):1085–1092 [DOI] [PubMed] [Google Scholar]
- Dodge J, Lewis P, Stanton M, Wilsher J (2007) Cystic fibrosis mortality and survival in the UK: 1947–2003. European Respiratory Journal 29 (3):522–526 [DOI] [PubMed] [Google Scholar]
- Fielding J, Brantley L, Seigler N, McKie KT, Davison GW, Harris RA (2015) Oxygen uptake kinetics and exercise capacity in children with cystic fibrosis. Pediatric pulmonology 50 (7):647–654 [DOI] [PubMed] [Google Scholar]
- FitzSimmons SC (1993) The changing epidemiology of cystic fibrosis. The Journal of pediatrics 122 (1):1–9 [DOI] [PubMed] [Google Scholar]
- Godfrey S, Davies C, Wozniak E, Barnes CA (1971) Cardio-respiratory response to exercise in normal children. Clinical science 40 (5):419–431 [DOI] [PubMed] [Google Scholar]
- Gratas-Delamarche A, Mercier J, Ramonatxo M, Dassonville J, Prefaut C (1993) Ventilatory response of prepubertal boys and adults to carbon dioxide at rest and during exercise. European journal of applied physiology and occupational physiology 66 (1):25–30 [DOI] [PubMed] [Google Scholar]
- Gruet M, Troosters T, Verges S (2017) Peripheral muscle abnormalities in cystic fibrosis: Etiology, clinical implications and response to therapeutic interventions. Journal of Cystic Fibrosis [DOI] [PubMed] [Google Scholar]
- Guazzi M, De Vita S, Cardano P, Barlera S, Guazzi MD (2003) Normalization for peak oxygen uptake increases the prognostic power of the ventilatory response to exercise in patients with chronic heart failure. American heart journal 146 (3):542–548 [DOI] [PubMed] [Google Scholar]
- Hankinson J, Odencrantz J, Fedan K (1999) Spirometric reference values from a sample of the general U.S. population. Am J Respir Crit Care Med 159:179–187 [DOI] [PubMed] [Google Scholar]
- Hart N, Polkey MI, Clement A, Boule M, Moxham J, Lofaso F, Fauroux B (2002) Changes in pulmonary mechanics with increasing disease severity in children and young adults with cystic fibrosis. American journal of respiratory and critical care medicine 166 (1):61–66 [DOI] [PubMed] [Google Scholar]
- Hebestreit H, Arets HG, Aurora P, Boas S, Cerny F, Hulzebos EH, Karila C, Lands LC, Lowman JD, Swisher A (2015) Statement on exercise testing in cystic fibrosis. Respiration 90 (4):332–351 [DOI] [PubMed] [Google Scholar]
- Hebestreit H, Kieser S, Rüdiger S, Schenk T, Junge S, Hebestreit A, Ballmann M, Posselt H- G, Kriemler S (2006) Physical activity is independently related to aerobic capacity in cystic fibrosis. European Respiratory Journal 28 (4):734–739 [DOI] [PubMed] [Google Scholar]
- Kleber F, Vietzke G, Wernecke K, Bauer U, Opitz C, Wensel R, Sperfeld A, Glaser S (2000) Impairment of ventilatory efficiency in heart failure. Circulation 101 (24):2803–2809 [DOI] [PubMed] [Google Scholar]
- Lands LC, Heigenhauser GJ, Jones NL (1992) Analysis of factors limiting maximal exercise performance in cystic fibrosis. Clinical Science 83 (4):391–397 [DOI] [PubMed] [Google Scholar]
- MacGowan GA, Murali S (2000) Ventilatory and heart rate responses to exercise: better predictors of heart failure mortality than peak exercise oxygen consumption. Circulation 102 (24):e182–e182 [DOI] [PubMed] [Google Scholar]
- MacKenzie T, Gifford AH, Sabadosa KA, Quinton HB, Knapp EA, Goss CH, Marshall BC (2014) Longevity of patients with cystic fibrosis in 2000 to 2010 and beyond: survival analysis of the cystic fibrosis foundation patient registry. Annals of internal medicine 161 (4):233–241 [DOI] [PMC free article] [PubMed] [Google Scholar]
- McKone EF, Barry SC, FitzGerald MX, Gallagher CG (2005) Role of arterial hypoxemia and pulmonary mechanics in exercise limitation in adults with cystic fibrosis. Journal of Applied Physiology 99 (3):1012–1018 [DOI] [PubMed] [Google Scholar]
- Moorcroft AJ, Dodd ME, Webb AK (1997) Long-term change in exercise capacity, body mass, and pulmonary function in adults with cystic fibrosis. Chest 111 (2):338–343 [DOI] [PubMed] [Google Scholar]
- Nixon PA, Orenstein DM, Kelsey SF, Doershuk CF (1992) The prognostic value of exercise testing in patients with cystic fibrosis. New England Journal of Medicine 327 (25):1785–1788 [DOI] [PubMed] [Google Scholar]
- Orenstein DM, Nixon PA (1991) Exercise performance and breathing patterns in cystic fibrosis: Male-female differences and influence of resting pulmonary function. Pediatric pulmonology 10 (2):101–105 [DOI] [PubMed] [Google Scholar]
- Penketh A, Wise A, Mearns M, Hodson M, Batten J (1987) Cystic fibrosis in adolescents and adults. Thorax 42 (7):526–532 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Philpott J, Houghton K, Luke A (2010) Physical activity recommendations for children with specific chronic health conditions: Juvenile idiopathic arthritis, hemophilia, asthma and cystic fibrosis. Paediatrics & child health 15 (4):213–218 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pianosi P, Hochman J (1996) End-tidal estimates of arterial PCO2 for cardiac output measurement by CO2 rebreathing: A study in patients with cystic fibrosis and healthy controls. Pediatric pulmonology 22 (3):154–160 [DOI] [PubMed] [Google Scholar]
- Pianosi P, Leblanc J, Almudevar A (2005) Peak oxygen uptake and mortality in children with cystic fibrosis. Thorax 60 (1):50–54 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pianosi P, Wolstein R (1996) Carbon dioxide chemosensitivity and exercise ventilation in healthy children and in children with cystic fibrosis. Pediatric research 40 (3):508. [DOI] [PubMed] [Google Scholar]
- Quanjer PH, Stanojevic S, Cole TJ, Baur X, Hall GL, Culver BH, Enright PL, Hankinson JL, Ip MS, Zheng J (2012) Multi-ethnic reference values for spirometry for the 3-95-yr age range: the global lung function 2012 equations. European Respiratory Journal 40 (6):1324–1343 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rosenfeld M, Davis R, FitzSimmons S, Pepe M, Ramsey B (1997) Gender gap in cystic fibrosis mortality. American journal of epidemiology 145 (9):794–803 [DOI] [PubMed] [Google Scholar]
- Schneiderman-Walker J, Wilkes D, Strug L, Lands L, Pollock S, Selvadurai H, Hay J, Coates A, Corey M (2005) Sex differences in habitual physical activity and lung function decline in children with cystic fibrosis. The Journal of pediatrics 147 (3):321–326 [DOI] [PubMed] [Google Scholar]
- Schwaiblmair M, Faul C, von Scheidt W, Berghaus TM (2012) Ventilatory efficiency testing as prognostic value in patients with pulmonary hypertension. BMC pulmonary medicine 12 (1):23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stein R, Selvadurai H, Coates A, Wilkes DL, Schneiderman-Walker J, Corey M (2003) Determination of maximal voluntary ventilation in children with cystic fibrosis. Pediatric pulmonology 35 (6):467–471 [DOI] [PubMed] [Google Scholar]
- Takken T, Bongers BC, van Brussel M, Haapala EA, Hulzebos EH (2017) Cardiopulmonary Exercise Testing in Pediatrics. Annals of the American Thoracic Society (ja) [DOI] [PubMed] [Google Scholar]
- Thin A, Dodd J, Gallagher C, Fitzgerald M, Mcloughlin P (2004) Effect of respiratory rate on airway deadspace ventilation during exercise in cystic fibrosis. Respiratory medicine 98 (11 ):1063–1070 [DOI] [PubMed] [Google Scholar]
- Troosters T, Langer D, Vrijsen B, Segers J, Wouters K, Janssens W, Gosselink R, Decramer M, Dupont L (2009) Skeletal muscle weakness, exercise tolerance and physical activity in adults with cystic fibrosis. European Respiratory Journal 33 (1):99–106 [DOI] [PubMed] [Google Scholar]
- Tucker MA, Crandall R, Seigler N, Rodriguez-Miguelez P, McKie KT, Forseen C, Thomas J, Harris RA (2017) A single bout of maximal exercise improves lung function in patients with cystic fibrosis. Journal of Cystic Fibrosis 16 (6):752–758 [DOI] [PubMed] [Google Scholar]
- Wasserman K, Hansen J, Sue D, Stringer W, Whipp B (2005) Clinical exercise testing. Principles of exercise testing and interpretation including pathophysiology and clinical applications. Lippincott Williams & Wilkins, [Google Scholar]


