Abstract
Plant extracts are a potential source of new compounds for nematode control and may be an excellent alternative for the control gastrointestinal nematodes that are resistant to conventional anthelmintics. However, research involving natural products is a complex process. The main challenge is the identification of bioactive compounds. Online analytical techniques with universal detectors, such as high-performance liquid chromatography-mass spectrometry (HPLC-MS), together with metabolomics could enable the fast, accurate evaluation of a massive amount of data, constituting a viable option for the identification of active compounds in plant extracts. This study focused on the evaluation of the ovicidal activity of ethanol extracts from 17 plants collected from the Pantanal wetland in the state of Mato Grosso do Sul, Brazil, against eggs of Haemonchus placei using the egg hatchability test. The ethanol extracts were obtained using accelerated solvent extraction. The data on ovicidal activity, mass spectrometry and metabolomics were evaluated using HPLC-DAD-MS, partial least squares regression analysis (PLS-DA) and a correlation map (univariate correlation analyses) to detect compounds that have a positive correlation with biological activity. Among the ten metabolites with the best correlation coefficients, six were phenylpropanoids, two were triterpene saponins, one was a brevipolide, and one was a flavonoid. Combinations of metabolites with high ovicidal action were also identified, such as phenylpropanoids combined with the triterpene saponins and the flavonoid, flavonoids combined with iridoid and phenylpropanoids, and saponins combined with phenylpropanoid. The positive correlation between classes of compounds in plants belonging to different genera and biological activity (as previously identified in the literature) reinforces the robustness of the statistical data and demonstrates the efficacy of this method for the selection of bioactive compounds without the need for isolation and reevaluation. The proposed method also enables the determination of synergism among the classes, which would be impracticable using traditional methods. The present investigation demonstrates that the metabolomic technique was efficient at detecting secondary metabolites with ovicidal activity against H. placei. Thus, the use of metabolomics can be a tool to accelerate and simplify bioprospecting research with plant extracts in veterinary parasitology.
Introduction
Gastrointestinal nematodiasis (GIN) substantially affects cattle health, particularly in cases of co-infection by infectious and parasitic agents [1]. Among parasitic species of veterinary concern, the hematophagous nematode Haemonchus placei is particularly pathogenic to cattle in tropical regions, causing hypoproteinemia, anemia, and anorexia in host animals [2], often with considerable economic losses.
Given the economic impact of GIN on cattle [3,4] and considering the increasing difficulty in controlling these parasitic nematodes with traditional anthelmintic drugs, especially H. placei [5,6,7], plant-based agents constitute a promising alternative [8,9,10,11]. Plants extracts are a potential source of new compounds for the control of nematodes [12] and the use of plant-based drugs with anthelmintic activity may be an excellent alternative for controlling GIN in ruminants [9, 11]. However, research involving natural products is a complex task. The main challenge is the identification of bioactive compounds in plant extracts [11].
Online analytical techniques with universal detectors, such as high-performance liquid chromatography coupled to mass spectrometry (HPLC-MS), can speed up the discovery of new compounds from plants as well as identify those that have previously been reported in the literature [13]. When combined with metabolomic tools, this strategy could enable the fast, accurate evaluation of a massive amount of data, constituting a viable option for the identification of active compounds in plant extracts [14,15].
Therefore, the present study aimed to evaluate the ovicidal activity of extracts from 17 plants collected from the Pantanal wetland in Brazil against eggs of H. placei using accelerated solvent extraction, mass spectrometry and metabolomic tools to determine bioactive compounds.
Materials and methods
The biological samples used in this study (plants and parasite) were registered with the SisGen plataform (code: A63DB37). All activities that were performed with animals respected the ethical principles adopted by the National Council for the Control of Animal Experimentation (NCCAE), and were approved by the Committee on Ethics in the Use of Animals / CEUA / UFMS with Protocol n°. 475/2012.
2.1. Haemonchus placei isolate
HpIBR1 is an isolate of H. placei cryopreserved at the Laboratory of Veterinary Parasitology of the Federal University of Mato Grosso do Sul (UFMS). This isolate has been phenotypically characterized as resistant to ivermectin [16].
For maintenance of the isolate, a calf was dewormed for three consecutive days with oral albendazole sulfoxide (Ricobendazole, Ourofino, Brazil) at a concentration of 7.5 mg/kg. The animal was evaluated daily for GIN infection by fecal egg counts [17] and was considered free of helminth infection when 30 days had elapsed with no fecal helminth detected, at which point the animal was orally inoculated with 10,000 third-stage larvae of H. placei. From the 30th day post-inoculation, fecal samples were collected daily to obtain eggs for use in the in vitro tests of the plant extracts.
2.2. Plant material
Seventeen plant species (Table 1) were randomly selected from the Corumbá River–Abobral region (São Miguel farm, Estrada Parque, UFMS, Pantanal Field Station and São Bento farm) in the rainy season (December 2011 to March 2012). A voucher specimen of each species was deposited at the CGMS Herbarium of the UFMS.
Table 1. Families, species, code, collection sites, parts collected and popular names of plants evaluated for ovicidal activity against Haemonchus placei.
| Family | Species | Code | Collection site | Part collected | Popular name |
|---|---|---|---|---|---|
| Alismataceae | Echinodorus paniculatus Micheli | E_panic | 19°34’36”S; 57°1’11”W | AP, FL | Chapéu-de-couro |
| Alismataceae | Melanthera latifolia(Gardner) Cabrera | M_latif | 19°34’36”S; 57°1’11”W | AP, FL | – |
| Asteraceae | Aspilia latissima Malme | A_latis | 19°34’36”S; 57°1’11”W | LE | Fumeiro |
| Asteraceae | Centratherum punctatum Cass. | C_punct | 19°37’5” S; 57°2’4”W | AP, FL | Balaio-de-velho |
| Bignoniaceae | Handroanthus serratifolius (Vahl) S.O. Grose | H_serra | 19°34’36”S; 57° 1’11”W | FL | Ipê-amarelo |
| Convolvulaceae | Ipomoea chiliantha Hallier f. | I_chili | 19°37’5” S; 57°2’4”W | AP | Cipó-de-leite |
| Euphorbiaceae | Sebastiana hispida (Mart.) Pax | S_hispi | 19°34’7”S; 57°1’15”W | AP | Mercúrio |
| Hippocrateaceae | Hippocratea volubilis L. | I_volub | 19°34’7”S; 57°1’15”W | TW, LE | Fava-de-arara |
| Lamiaceae | Hyptis brevipes Poit | H_brevi | 19°34’7”S; 57°1’15”W | WP, FL | Alfavaca-do-mato |
| Lamiaceae | Hyptis mutabilis (Rich.) Briq. | H_mutab | 19°34’7”S; 57°1’15”W | AP, FL | Sambacuité, cheirosa, betônica-brava |
| Lauraceae | Ocotea diospyrifolia (Meisn.) Mez | O_diosp | 19°36’30”S; 57°2’8”W | TW, LE | Canela-preta |
| Rubiaceae | Dioidia kuntzei K. Schum. | D_kuntz | 19°34’7”S; 57°1’15”W | WP, FL | – |
| Rubiaceae | Tocoynea formosa (Cham. & Schltdl.) K. Schum. | T_form | 19°34’7”S; 57°1’15”W | TW, LE | Jenipapinho |
| Salicaceae | Casearia aculeata Jacq. | C_acule | 19°34’7”S; 57°1’15”W | TW, LE, FL | Cruzeiro |
| Scrophulareaceae | Angelonia hirta Cham. | A_hirta | 19°34’7”S; 57°1’15”W | AP, FL, FR | – |
| Verbenaceae | Lantana canescens Kunth | L_canes | 19°37’5” S; 57°2’4”W | AP, FL | Camara, cidreira |
| Verbenaceae | Vitex cymosa Bertero ex Spreng. | V_cymos | 19°34’7”S; 57°1’15”W | TW, LE | Azeitona-do-mato, jaramantaia, tarumã |
TW: twigs; LE: leaves; FR: fruit; AP: aerial parts (including leaves, twigs and stems); FL: flowers; RO: roots; WP: whole plant (including roots, leaves and twigs.
2.3. Plant extracts
The collected material was initially stabilized and dried in a forced-draft oven at 40 °C. Extraction was performed in an ASE 150 accelerated solvent extraction (Dionex) with ethanol:water (7:3) as the solvent at 100 °C and 1600 psi in a single cycle with a 5-min static time, 60% rinse volume and 50 s purge time. The extracts were concentrated in a rotary evaporator, stored in 15-mL Falcon tubes, labeled, sealed and kept at –20 °C until testing.
2.4 Egg hatchability test
Eggs were immediately recovered from fresh fecal samples using the procedure described by Coles et al. [18], adapted by Bizimenyera et al. [19] and further modified as follows: A 500-μL aliquot of water containing approximately 100 eggs was pipetted into each plate well, followed by the addition of 500 μL of either the treatment solution or water (control). The plates were incubated at 27 °C for 24 h. The assays were performed in triplicate for each extract concentration using 24-well cell culture plates.
2.5. Ovicidal evaluation
Two egg hatchability tests were used to evaluate the effect of the extracts against H. placei eggs. In experiment I (screening test), the extracts were weighed, diluted in distilled water using a vortex (IKA) and subsequently evaluated at concentrations of 1, 10, 50, and 100 mg/mL (Table 2). In experiment II, the 13 species that demonstrated efficacy in the screening test were evaluated further: Angelonia hirta, Aspilia latissima, Centratherum punctatum, Dioidia kuntzei, Echinodorus paniculatus, Hyptis brevipes, Hyptis mutabilis, Ipomoea chiliantha, Lantana canescens, Sebastiana hispida, Handroanthus serratifolius, Tocoynea formosa and Vitex cymosa. As small amounts of sediment were found in assays with H. serratifolius and T. formosa, the EC90 and EC50 values for these species were obtained from data collected after the removal of the residue. The concentration that led to the lowest hatching rate in experiment I was employed as the high dose, from which nine other dilutions at 1:2 ratios were serially prepared.
Table 2. Plant species, extract concentrations screened, hatching rates and p-values (water/extract concentration).
| Species | Hatching rate (%) | p-value | ||||
|---|---|---|---|---|---|---|
| Water | 1 mg/mL | 10 mg/mL | 50 mg/mL | 100 mg/mL | ||
| Angelonia hirta | 78.4b | 79.3b | 58.5a | 40.3a | N.E. | 0.0001 |
| Aspilia latissimi | 73.4b | 80.1b | 5.7a | 0.0a | N.E. | <0.0001 |
| Casearia aculeate | 89.7a | 88.7a | 92.7a | 89.8a | N.E. | 0.0789 |
| Centratherum punctatum | 86.5c | N.E. | 59.5b | 57.0ab | 2.3a | <0.0001 |
| Diodia kuntzei | 79.2c | 58.6b | 18.5a | 18.0a | N.E. | <0.0001 |
| Echinodorus paniculatus | 78.0c | 78.5bc | 0.6ab | 21.4ab | N.E. | <0.0001 |
| Hippocratea volubilis | 80.7b | 84.9ab | 74.9a | 83.9ab | N.E. | 0.0155 |
| Hyptis brevipes | 76.7c | 73.7bc | 57.3bc | 43.3ab | N.E. | 0.0199 |
| Hyptis mutabilis | 81.0b | 78.9b | 12.6a | 8.4a | N.E. | <0.0001 |
| Ipomoea chiliantha | 95.3b | N.E. | 4.2a | 0.7a | 0.6a | <0.0001 |
| Lantana canescens | 83.4b | N.E. | 0.0a | 19.3a | 7.9a | 0.0024 |
| Melanthera latifolia | 74.0a | 86.0b | 88.0b | 80.0ab | N.E. | 0.0023 |
| Ocotea diospyrifolia | 75.4a | 89.2b | 88.9b | 84.4ab | N.E. | 0.0143 |
| Sebastiana hispida | 93.2b | N.E. | 0.7a | 2.7a | 2.9a | <0.0001 |
| Handroanthus serratifolius | 84.0d | 95.4d | 40.9c | 18.5b | 0.0a | <0.0001 |
| Tocoyena formosa | 81.0b | 83.9b | 53.8a | 72.5b | N.E. | 0.0001 |
| Vitex cymosa | 83.0c | N.E. | 57.4b | 1.4a | 0.0a | <0.0001 |
Different letters on the same row indicate significant differences (α = 0.05, one-way ANOVA followed by Bonferroni’s post-hoc test); N.E.: Not Evaluated.
The concentration ranges tested in the experiment II were as follows: A. hirta, 100–0.19 mg/mL; A. latissima, 50–0.10 mg/mL; C. punctatum, 120–0.23 mg/mL; D. kuntzei, 100–0.19 mg/mL; E. paniculatus, 100–0.19 mg/mL; H. mutabilis, 80–0.16 mg/mL; H. brevipes, 100–0.19 mg/mL; I. chiliantha, 50–0.10 mg/mL; L. canescens, 10–0.02 mg/mL; S. hispida, 50–0.10 mg/mL; H. serratifolius, 3.3–0.006 mg/mL; T. formosa, 120–0.23 mg/mL; and V. cymosa, 50–0.10 mg/mL. The positive control (thiabendazole) was tested in the range of 5 × 10−4 to 1 × 10−6 mg/mL.
2.6. HPLC-DAD-MS analyses
The extracts were analyzed in triplicate by random sampling in an HPLC coupled to a diode array detector (DAD) (Shimadzu) and a mass spectrometer ESI-qTOF microTOF-Q III (Bruker Daltonics). Separation of the compounds was performed by a Kinetex C-18 (2.6 μ, 150 x 2.2 mm, Phenomenex) chromatographic column protected by a pre-column. The mobile phase was ultrapure water (solvent A) and acetonitrile (solvent B), both with 0.1% formic acid (v/v). The following was the gradient elution: 0–2 min 3% B; 2-25min 3–25% B; 25–40 min 25–80% B, followed by column washing and reconditioning (8 minutes). The flow rate was 0.3 mL/min. The column oven was 50 °C and the injection volume was 1 μL. The UV analyses were performed in the wavelength range of 240–800 nm, with the mass spectrometer operating in negative and positive mode (m/z 120–1200). The samples used for the quality control of the metabolic analyses consisted of a pool produced by the addition of 50 μL of each sample, which was injected in every six analyses. The pool data were not considered in the statistical analysis. The identification of the compounds was based on mass spectrometry (accurate mass and ion fragmentation pathway) and UV data compared to information reported in the literature. The molecular formula of each compound was determined based on the mass errors within ± 5 ppm and mSigma below 30.
2.7. Statistical analysis
In the screening test, hatching rates were expressed as percentages [18]:
The data were subjected to one-way analysis of variance (ANOVA) with hatchability as the single factor, followed by Bonferroni’s post-hoc test (α = 0.05). All treatments were compared to the controls (water) and to each other. Extracts were considered active when at least one of the concentrations led to lower hatching rates than those observed in the controls (p < 0.05).
Dose vs. response sigmoid curves were constructed by non-linear regression for the active extracts based on 10 concentrations. The concentrations were log-transformed (X = log X) and effectiveness values (for each repetition) were expressed as percentages. The EC50 (mean effective concentration) and EC90 were calculated using the following equations: Y = 100 / (1 + 10^((Log EC50 –X) * HillSlope)) and log EC50 = log EC90 –(1 / HillSlope) * log (90 / (100–90)) where Y = Bottom + (Top–Bottom) / (1 + 10^((Log EC50 –X) * HillSlope)), in which X = concentration log, Y = effectiveness (%), Bottom = minimum effectiveness, Top = maximum effectiveness, and HillSlope = the dose vs. response curve slope. EC90/EC50 ratios were expressed as mg/mL.
Statistical analysis was performed using GraphPad Prism 6.0 software (GraphPad Software, San Diego, CA, USA, http://www.graphpad.com). For each extract concentration, the hatchability inhibition rate was calculated as follows [18]:
HPLC-DAD-MS analyses were processed in DataAnalysis 4.2 in negative mode, which shows better ionization and a larger number of peaks. Data were aligned using the Metalign software [20], resulting in 1029 entries, which were reduced by MSclust [21]. The entries were regrouped, resulting in 98 reconstituted metabolites from the assembled signals (different ions of the same molecules, such as isotopes, fragments, and adducts). The replicates and pool samples were compared to evaluate the reproducibility of the equipment and processing. The exported data were analyzed with the aid of the MetaboAnalyst 4.0 platform [22], using univariate analysis tools to correlate the active extracts and main compounds of the extracts with Pattern Hunter. In the correlation map between the chemical composition and biological activity determined to apply Pearson’s correlations (significant at p ≤ 0.05), the metabolomic study with the final data referring to EC90 was performed using base 1/EC90, since lower ECs constitute the best results. Compounds with minimum intensity peaks of 20000 in the mass spectrometric analysis were considered for the correlation map between chemical composition and biological activity.
The plants were grouped based on the results of egg hatchability test (experiment I) and analyzed by discriminated partial least squares regression analysis (PLS-DA). The aim of this analysis, which is based primarily on the chemical composition of the extracts, was to characterize the metabolism of active and inactive extracts. The goodness and robustness of the PLS-DA model were estimated from calculations of R2 and Q2, respectively. R2 is the fraction of variance explained by a component and Q2 describes the total fraction predicted by a component. A Q2 value > 0.4 characterizes a model as good and Q2 > 0.7 characterizes the model as robust [23]. A correlation map was used to detect compounds with a positive correlation to the EC90.
Results
Experiment I
Thirteen plants exhibited significant activity during the screening test. Melanthera latifolia, Casearia aculeata, and Ocotea diospyrifolia were ineffective during the screening test (Table 2), with respective hatchability inhibition rates of 19.85%, 10.35% and 15.9% for the highest concentration tested. Higher hatching rates (p = 0.0023) were found when exposing the eggs to the M. latifolia (p = 0.0023) and O. diospyrifolia (p = 0.0143) extracts at concentrations of 1 to 10 mg/mL compared to exposure to the negative control (water). Hippocratea volubilis exhibited activity only at 10 mg/mL, reducing the hatching rate (p = 0.0155). However, based on the need for a high dose, we do not consider it to be a promising species.
3.2. Experiment II
All 13 extracts evaluated at 10 serial concentrations (A. latissima, C. punctatum, D. kuntzei, E. paniculatus, H. mutabilis, H. brevipes, I. chiliantha, L. canescens, S. hispida, H. serratifolius, V. cymosa, A. hirta and T. formosa) exhibited dose-dependent behavior, with a gradual increase in effectiveness with the increase in concentration. EC50 values ranged from 0.5 to 13.13 mg/mL and EC90 values ranged from 1.18 to 174.6 mg/mL (Table 3).
Table 3. Plant species, mean effective concentrations inhibiting hatchability of Haemonchus placei eggs by 50% (EC50) and 90% (EC90), confidence intervals, curve slopes (HillSlope) and coefficient of determination (R2) obtained from serial evaluations of 10 concentrations.
| Species | EC50 (mg/mL) | 95% CI | HillSlope | R2 | EC90 (mg/mL) | 95% CI | HillSlope | R2 |
|---|---|---|---|---|---|---|---|---|
| Angelonia hirta | 2.61 | 1.98–3.43 | 1.08 | 0.89 | 6.87 | 5.34–8.85 | 4.90 | 0.95 |
| Aspilia latissima | 1.28 | 0.99–1.66 | 1.56 | 0.88 | 2.48 | 1.83–3.34 | 7.03 | 0.98 |
| Centratherum punctatum | 12.42 | 10.19–15.13 | 1.54 | 0.93 | 42.07 | 23.17–76.36 | 1.99 | 0.95 |
| Dioidia kuntzei | 2.60 | 2.33–2.91 | 2.92 | 0.96 | 4.75 | 3.90–5.77 | 4.56 | 0.96 |
| Echinodorus paniculatus | 6.43 | 5.36–7.72 | 1.30 | 0.94 | 21.45 | 14.36–32.04 | 2.07 | 0.97 |
| Hyptis brevipes | 3.34 | 3.04–3.66 | 3.03 | 0.97 | 5.71 | 5.10–6.40 | 4.70 | 0.99 |
| Hyptis mutabilis | 2.43 | 1.98–2.98 | 1.59 | 0.91 | 5.51 | 4.69–6.47 | 4.04 | 0.98 |
| Ipomoea chiliantha | 0.54 | 0.35–0.82 | 1.03 | 0.73 | 1.19 | 0.19–7.11 | 15.49 | 0.98 |
| Lantana canescens | 0.75 | 0.46–1.22 | 1.46 | 0.43 | 1.70 | 0.71–4.09 | 8.35 | 0.98 |
| Sebastiana hispida | 1.88 | 1.64–2.15 | 3.86 | 0.93 | 2.72 | 2.13–3.46 | 7.07 | 0.98 |
| Handroanthus serratifolius | 0.83 | 0.72–0.96 | 2.88 | 0.83 | 1.94 | 1.25–3.01 | 3.16 | 0.88 |
| Tocoynea formosa | 10.08 | 5.55–18.18 | 0.58 | 0.64 | 14.52 | 4.98–42.34 | 2.99 | 0.74 |
| Vitex cymosa | 13.13 | 6.53–26.38 | 0.45 | 0.57 | 174.60 | 1.61–18915 | 1.29 | 0.78 |
| Thiabendazole | 2 × 10−6 | 1.9 × 10−6–2.2 × 10−6 | 1.68 | 0.98 | 6 × 10−6 | 4 × 10−6–9 × 10−6 | 1.26 | 0.98 |
Complete inhibition was achieved with extracts from A. hirta (100 mg/mL), D. kuntzei (25 mg/mL), H. brevipes (25 mg/mL), A. latissima (6.25 mg/mL), T. formosa (15 mg/mL), I. chiliantha (6.25 mg/mL), L. canescens (5 mg/mL) and H. serratifolius (2.33 mg/mL). Eggs treated with the S. hispida extract exhibited an amorphous coating and viable larvae inside the eggs at the end of the incubation period, but hatching was low or not observed.
3.3. HPLC-DAD-MS analyses
Six classes of secondary metabolites were positively correlated to ovicidal action against H. placei. The active classes were identified as iridoids (peaks 2, 3, 4, 5, 6, 8 and 9), phenylpropanoids (peaks 7, 12, 18, 20, 23, 25 and 26), hydrolysable tannins (peaks 10 and 11), flavonoids (13, 14, 15, 16, 17, 21, 24 and 30), triterpene saponins (peaks 28, 31, 32, 34, 35, 36 and 37) and brevipolides (peaks 29 and 33) (Table 4).
Table 4. Retention time, UV spectrum, mass, molecular formula, chemical class and plants species in which compounds were detected of active peaks with relative intensity of masses higher than 20000 and were positively correlated to EC90.
| Peak |
Ret (min) |
UV | [M-H]- | Molecular formula | MS/MS | Class | Compounds | Plants |
| 1 | 1.1 | --- | 665.2114 | --- | --- | Unknown | Unknown | H_brevi; A_latis; L_canes; V_cymos; H_mutab; M_latif |
| 2 | 2.0 | --- | 389.1076 | C16H22O11 | 389: 227 (C10H11O6); 209 (C10H9O5); 183 (C9H11O4) | Iridoid | Hexosyl iridoid derivative | D_kuntz |
| 3 | 3.0 | --- | 389.1080 | C16H22O11 | 389: 227 (C10H11O6); 209 (C10H9O5); 183 (C9H11O4) | Iridoid | Hexosyl iridoid derivative | D_kuntz |
| 4 | 4.7 | --- | 363.1297 | C15H23O10 | 363:201(C9H13O5) | Iridoid | Hexosyl iridoid derivative | A_hirta |
| 5 | 6.5 | --- | 345.1191 | C15H22O9 | 345: 207 (C11H11O4); 189 (C11H9O3); 183 (C9H11O4) | Iridoid | Hexosyl iridoid derivative | A_hirta |
| 6 | 6.6 | --- | 345.1190 | C15H22O9 | 345: 207 (C11H11O4); 189 (C11H9O3); 183 (C9H11O4) | Iridoid | Hexosyl iridoid derivative | A_hirta |
| 7 | 9.0 | 301/322 | 353.0891 | C16H18O9 | 353: 191 (C7H11O6) | Phenylpropanoid | chlorogenic acid | I_chili; A_latis; S_hispi; D_kuntz; C_punct; V_cymos; M_latif |
| 8 | 10.4 | --- | 413.1085 | C18H22O11 | 413: 371 (C16H19O10); 251 (C12H11O6); 191 (C10H7O4) | Iridoid | Asperuloside | D_kuntz |
| 9 | 11.5 | --- | 405.1401 | C17H26O11 | 405: 387 (C17H23O10); 243 (C11H15O6); 225 (C11H13O5) | Iridoid | Sanshiside methyl ester | L_canes |
| 10 | 11.5 | 268 | 633.0719 | C27H22O18 | 633: 463 (C20H15O13); 301 (C14H5O8); 275 (C13H7O7) | Hydrolized tannin | Corilagin | S_hispi |
| 11 | 12.6 | 268 | 951.0787 | C41H28O27 | 951: 933(C41H25O26); 765 (C34H21O21); 463 (C20H15O13); 301 (C14H5O8); 275 (C13H7O7) | Hydrolized tannin | Geraniin | S_hispi |
| 12 | 12.8 | 301/327 | 515.1180 | C25H24O12 | 515: 191 (C7H11O6); 179 (C9H7O4) | Phenylpropanoid | 1,3 Dicaffeoylquinic | I_chili; A_latis |
| 13 | 16.0 | 281/344 | 463.0859 | C21H20O12 | 463: 301 (C15H9O7) | Flavonoid | Isoquercitrin | A_hirta |
| 14 | 16.4 | 282 | 463.0900 | C21H20O12 | --- | Flavonoid | Flavanone derivative | A_hirta; C_punct |
| Peak | Ret (min) |
UV | [M-H]- | Molecular formula | MS/MS | Class | Compounds | Plants |
| 15 | 17.4 | 270/345 | 739.2096 | C33H39O19 | --- | Flavonoid | Flavonol-hexosyl-dideoxyhexosyl | T_form; V_cymos |
| 16 | 17.9 | 270/344 | 477.1059 | C22H22O12 | 477: 314 (C16H10O7); 299 (C15H7O7) | Flavonoid | Isorhamnetin-O-glucoside | A_hirta |
| 17 | 18.0 | 270/346 | 461.0740 | C21H18O12 | 461: 285 (C15H9O6) | Flavonoid | Kaempferol-O-glucuronide | L_canes; A_hirta; C_punct; V_cymos |
| 18 | 19.1 | 296/327 | 623.1958 | C29H36O15 | 623: 461 (C20H29O12); 315 (C14H19O8); 179 (C9H7O4); 161 (C9H5O3) | Phenylpropanoid | Verbascosideo | L_canes; H_serra |
| 19 | 19.1 | 301/327 | 515.1190 | C25H24O12 | 515: 191 (C7H11O6); 179 (C9H7O4); 173 (C7H9O5) | Phenylpropanoid | 3,4 Dicaffeoylquinic | I_chili; A_latis; C_punct; V_cymos |
| 20 | 19.8 | 301/327 | 515.1193 | C25H24O12 | 515: 191 (C7H11O6); 179 (C9H7O4) | Phenylpropanoid | 3,5 Dicaffeoylquinic | I_chili; A_latis; C_punct; V_cymos |
| 21 | 19.9 | 283/343 | 593.1489 | C27H30O15 | 593: 285 (C15H9O6) | Flavonoid | Kaempferol-O-rutinoside | I_chili; S_hispi; D_kuntz; H_mutab; H_brevi; C_acule |
| 22 | 20.0 | 270 | 419.0970 | C20H20O10 | 419: 179 (C9H7O4) | Unknown | Unknown | H_serra |
| 23 | 20.3 | 290/327 | 623.1973 | C29H36O15 | 623: 461 (C20H29O12); 315 (C14H19O8); 179 (C9H7O4); 161 (C9H5O3) | Phenylpropanoid | Isoverbascosideo | L_canes |
| 24 | 20.5 | 266/338 | 445.0753 | C21H18O11 | 445: 269 (C15H9O5) | Flavonoid | Apigenin-O-glucuronide | L_canes |
| Peak | Ret (min) |
UV | [M-H]- | Molecular formula | MS/MS | Class | Compounds | Plants |
| 25 | 20.6 | 292/326 | 359.0760 | C18H16O8 | 359: 197 (C9H9O5); 179 (C9H7O4); 161 (C9H5O3) | Phenylpropanoid | Rosmarinic acid | H_mutab; H_brevi |
| 26 | 23.5 | 300/326 | 307.0462 | C14H12O8 | --- | Phenylpropanoid | Unknown | E_panic |
| 27 | 23.6 | 287/330 | 717.1420 | C36H30O16 | 717: 519 (C27H19O11); 339 (C18H11O7); 321 (C18H9O6) | Unknown | Unknown | H_mutab; H_brevi |
| 28 | 28.2 | --- | 809.4308 | C42H66O15 | 809: 603 (C35H55O8) | Triterpene saponin | quinovic acid derivative | T_formo |
| 29 | 28.7 | 300/326 | 403.1380 | C21H24O8 | 403: 359 (C20H23O6); 241 (C12H17O5); 197 (C11H17O3); 179 (C9H7O4) | Brevipolide | Dihydro-Brevipolide C | H_brevi |
| 30 | 29.5 | 281/346 | 359.0759 | C18H16O8 | 359: 329 (C16H9O8); 301 (C15H9O7); 286 (C14H8O7); | Flavonoid | 5,6,3'-trihydroxy-3,7,4'-trimethoxyflavone | H_brevi |
| 31 | 30.4 | --- | 955.4908 | C48H76O19 | --- | Triterpene saponin | Calenduloside derivative | A_latis |
| 32 | 30.6 | --- | 793.4358 | C35H55O7 | 793:587 () | Triterpene saponin | --- | T_formo; V_cymos |
| 33 | 30.8 | 300/313 | 387.1445 | C21H24O7 | 403: 343 (C20H23O5); 241 (C12H17O5); 197 (C11H17O3); 179 (C9H7O4) | Brevipolide | Dihydro-Brevipolide F | H_brevi |
| 34 | 31.8 | --- | 955.4911 | C48H76O19 | --- | Triterpene saponin | Calenduloside H | I_chili; A_latis |
| 35 | 32.6 | --- | 939.4971 | C48H76O18 | --- | Triterpene saponin | Triterpene saponin derivative | I_chili; A_latis |
| 36 | 34.8 | --- | 793.4362 | C42H66O14 | --- | Triterpene saponin | Ladyginoside B | I_chili |
| 37 | 36.0 | --- | 777.4416 | C42H66O13 | --- | Triterpene saponin | Triterpene saponin derivative | I_chili; A_latis |
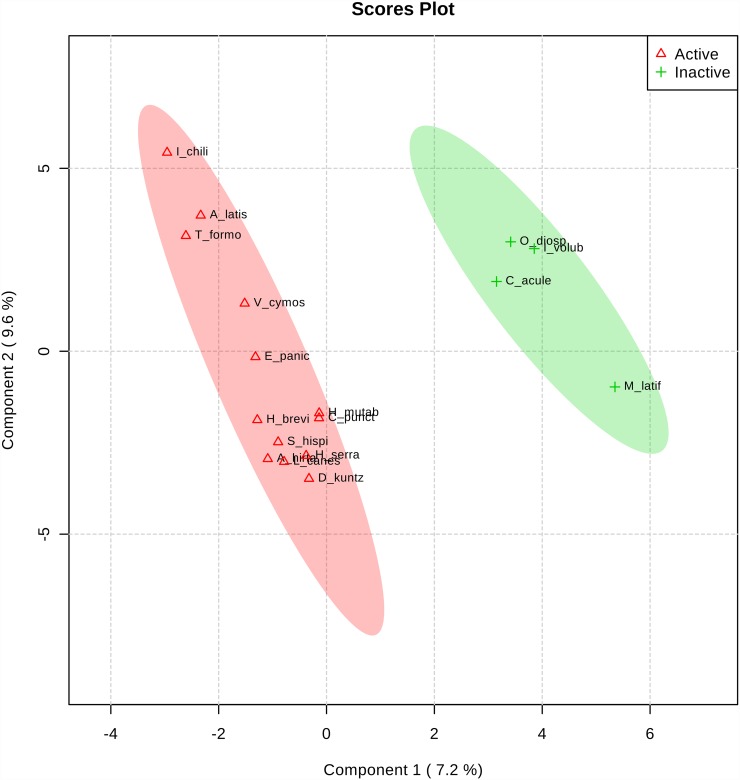
The PLS-DA separated active and inactive species (Fig 1). It is noteworthy that some classes of metabolites found in active plants were similar and not found in the inactive plants. The PLS-DA was a robust method, with values above 0.7 for both R2 and Q2, which proves that the results were reliable.
Fig 1. Geometric profile of active (triangle) and inactive (plus sign) fraction.
Dispersion graph of PLS-DA points (each point represents ethanol extract of plant species).
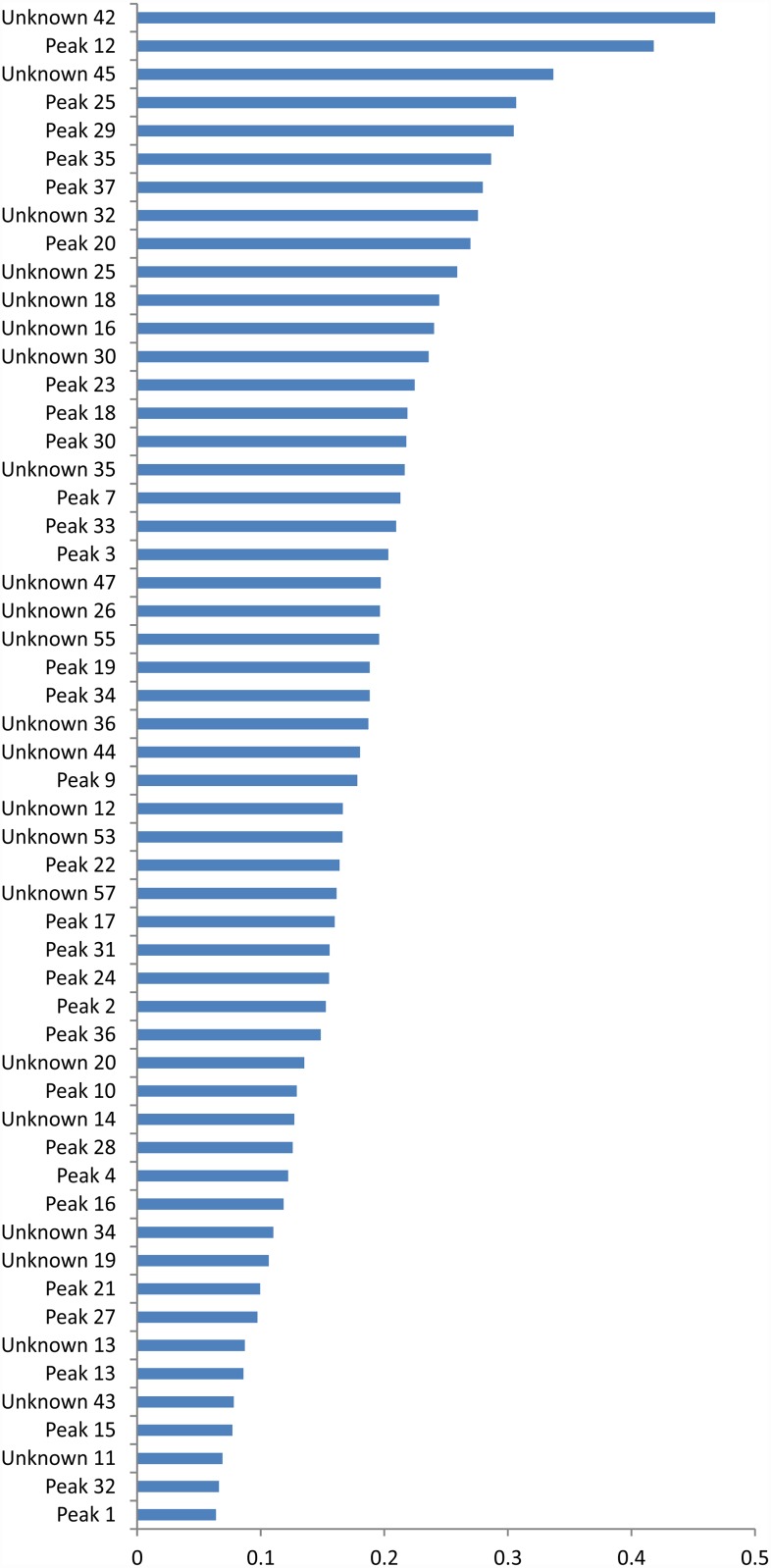
The correlation analysis between compounds and EC90 revealed compounds that were positively correlated to ovicidal activity. Among the ten metabolites with better correlation coefficients, six were phenylpropanoids, two were triterpene saponins, one was a brevipolide and one was a flavonoid (Fig 2). Peak 12 and 20 (exhibiting an intense ion at m/z 515 [M-H] − compatible with C25H24O12), peak 25 (m/z 359.0760 [M-H]− compatible with C18H16O8), peaks 18 and 23 (m/z 623 [M-H]− compatible with C29H36O15) and peak 7 (m/z 353.0891 [M-H]− compatible with C16H18O9) were the phenylpropanoids with the best correlation to biology activity (EC90) and were putatively identified as 1,3 dicaffeoylquinic, 3,5 dicaffeoylquinic, rosmarinic acid, verbascoside, isoverbascoside and chlorogenic acid, respectively. Peak 29 exhibited intense ions at m/z 403.1380 [M-H]− compatible with C21H24O8 and was putatively identified as dihydro-brevipolide C. Peak 4 (m/z 939.4971 [M-H]− compatible with C48H76O18) and 5 (intense ions at m/z 777.4416 [M-H]− compatible with C42H66O13) were putatively identified as triterpene saponin derivatives. Peak 9 (m/z 359. 0759 [M-H]− compatible with C18H16O8) was putatively identified as 5, 6, 3’-trihydroxy-3,7,4’trimethoxyflavone. All compounds listed as unknown in Fig 2 showed a very low intensity (lower than 20000) in the mass spectrometry analysis and was not possible to identify these compounds. They listed the information of this lower intensity compounds separately as supplementary material (S1 Table) to not overload Table 4. Also, chromatograms and MS/MS spectral data that allowed the identification of studied metabolites are presented as supplementary data (S1 and S2 Figs, respectively).
Fig 2. Correlation between secondary metabolites represented by peak observed in mass spectrometry and biological activity of extracts of origin characterized based on EC90.
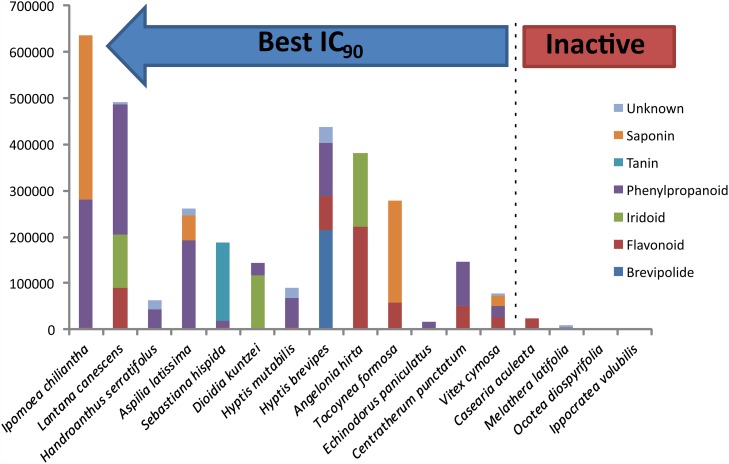
The plant with the greatest ovicidal activity (I. chiliantha) was composed of phenylpropanoids (chlorogenic acid, 1,3 dicaffeoylquinic, 3,4 dicaffeoylquinic and 3,5 dicaffeoylquinic), triterpene saponins (calenduloside H, Triterpene saponin derivative and ladygenoside B) and a flavonoid (kaempferol-O-rutinoside) as the main metabolites (Fig 3). Other combinations of metabolites that had considerable ovidal activity were flavonoids (kaempferol-O-glucuronide and apigenin-O-glucuronide), iridoid (sanshiside methyl ester) and phenylpropanoids (verbascoside and isoverbascoside) combined in the same extract (L. canescens) and saponins (calenduloside derivative, calenduloside H and Triterpene saponin derivative) combined with phenylpropanoids (chlorogenic acid, 1,3 dicaffeoylquinic, 3,4 dicaffeoylquinic and 3,5 dicaffeoylquinic) in the extract from A. latissima. Ethanolic extracts of the species I. chiliantha, L. canescens and A. latissima were among the most active (lowest EC90).
Fig 3. Chemical constitution (classes of major compounds) of ethanol extracts from 17 plant species evaluated for ovicidal action against H. placei.
Plant species ordered based on ovidical activity characterized by EC90 (calculated for active plants plotted in I_chili–V_cymos range) from right (less active) to left (more active).
Discussion
Despite the complex chemical profile of the species studied, it was possible to align the data with the aid of a pool of samples. The choice of univariate correlation methods is justified by the fact that we can directly list the main determinant compounds for biological activity and thus direct future research or studies aimed at the production of extracts enriched with these substances. A recent study has demonstrated the efficacy of this technique for the determination of active compounds from propolis extracts on biofilms of Staphylococcus aureus and Trichomonas vaginalis [24]. In the present work, the positive correlation of the same classes of compounds in plants belonging to different genera and families reinforces the robustness of the statistical data and demonstrates the efficacy of the proposed tool for the selection of bioactive compounds without the need for the isolation and reevaluation. This method also enables determining synergism among classes of compounds, which would be impracticable using traditional methods.
Some of the metabolites identified in the present study have been associated with nematicidal action. Flavonoids (flavones, flavanones, and flavonols) and their aglycone and glycosidic derivatives have been described as having nematicidal action against different species and stages of the lifecycle of ruminant GINs [25,26,27]. Ovicidal activity against Haemonchus contortus has been described for flavonoids from Spiglia anthelmia [12] and the flavonol derivatives quercetin and flavone apigenin extracted from Artemisia campestres (reviewed by [27]). A homoisoflavonoid from Agave sisalana significantly reduced the larval hatchability of nematode parasites of goats (Haemonchus spp., Oesophagostomum and Trichostrongylus spp.) [25]. Moreover, flavonoids have been also associated with the inhibition of larval development [28], inhibition of larval exsheathment (naringenin aglycone, kaempferol, myricetin, luteolin and quercetin) ([29], reviewed by [27]), inhibition of larval migration [30] and a reduction in the motility of H. contortus adults [27].
While the anthelmintic action attributed to the class of flavonoids has been previously reported, not all flavonoids are associated with antiparasitic action, as there is no clear relationship between structure and anthelmintic activity [27]. Besides the molecular structure, other important factors that may influence the activity of flavonoids include the concentration in contact with the parasite and the concomitant presence of other secondary metabolites, which can act in a complementary manner through by different mechanisms of action in the improvement of the solubility (surfactant properties) or stability (antioxidant effect) of other compounds. Klongsiriwet et al. [26] reported the synergistic effect between tannins and flavonoids, as evidenced by an increase in the effect of procyanidins and prodelphinidins (condensed polyphenol derivatives) on the inhibition of larval exsheathment in H. contortus when associated with quercetin or luteolin [12].
Our results suggest that the presence of more than one class of compounds is necessary for high ovicidal activity. This can be seen in C. aculeata, which, despite accumulating flavonoid 21, was shown to be inactive, which may be related to the absence of other classes of compounds. Reinforcing this observation, the most active species (based on EC90) were those with more than one class of metabolites (Ipomoea chiliantha, Lantana canescens, and Handroanthus serratifolius), demonstrating an evident joint action.
The mechanism of action of flavonoids with regard to anthelmintic performance is not known. However, these compounds seem to act in a similar way to tannins, likely interacting with proteins from non-covalent hydrophobic bonds, which may result in disruption and inhibition. Unlike tannins, flavonoids can be easily internalized by helminths and can reach active sites that are inaccessible to tannins [27].
Tannins are polyphenolic compounds with known anthelmintic action and are classified according to their chemical structure as condensed tannins, which are the most widely studied metabolites and are mainly those contained in tannin-rich forages [31], and hydrolyzable tannins. In the present study, only hydrolyzable tannins were detected (ethanol extract from S. hispida). It is noteworthy that although the method used for the detection of secondary metabolites (HPLC-MS) is not the most suitable for detecting high molecular weight compounds, such as condensed tannins, we believe that this class was not present in any extract, since the precursors (catechin and isocatechin monomers) were not found.
The ability of tannins to bind to proteins is well known (reviewed by [11]) and this characteristic has been implicated in the anthelmintic action of these compounds. The ovicidal effect and reduction in the motility of adult parasites of the genus Haemonchus had been demonstrated for ellagic acid present in ellagitannins [32]. The ovicidal action of ellagitannins is associated with the ability of tannins to interact with and precipitate proteins. Particularly in eggs, a coating of tannin-protein complexes forms that is bound to the eggshell and prevents hatching [33]. In the present study, corilagin and geraniin were detected in the extract from Sebastiana hispida and were associated with the ovicidal action of the ethanol extract. The eggs treated with the plant extract exhibited an amorphous coating, with the presence of viable larvae at the end of the incubation period, but no hatching of these larvae was observed, which confirms with the previously described action for hydrolysable tannins in preventing larval hatchability [34]. Corilagin and geraniin have hexahydroxydiphenic acid in their chemical structure and release ellagic acid into the medium when undergoing hydrolysis [32].
Studies reporting the anthelmintic activity of saponins are scarce [34]. Saponins have been identified as part of the chemical composition of extracts that have nematicidal action against parasites of the genus Haemonchus [32,30]. Therefore, these compounds have been recognized for working together on tannins and flavonoids [30], facilitating the action of phenolic derivatives on proteins due to the increased permeability of cell membranes, which are destabilized by the action of saponins (reviewed by [34]). Specific studies confirming the nematicidal action of saponins were only conducted with the phytonematodes Xiphinema index, Meloidogyne incognita and Globodera rostochiensis [34,35]. In the present study, triterpene saponins from Ipomoea chiliantha (peaks 28, 34, 35, 36 and 37), Tocoiena formosa (peak 32) and Aspilia latissima (peaks 28, 31, 34, 35 and 37) were associated with ovicidal action. The extracts from I. chiliantha and A. latissima have saponins and phenylpropanoids (peaks 7, 12, 19 and 20) as the main metabolites, lending support to the notion that these two classes together are related to high ovicidal activity. A. latissima, which was one of the species with the highest activity, also accumulates phenylpropanoids and triterpene saponins.
The ethanol extract from I. chiliantha exhibited the same classes of metabolites as those found observed in A. latissima, but at higher concentrations and there was also an increase in flavonoid 21. This chemical difference may help explain the reduction in the EC50 from 1.28 mg/mL (95% CI 0.99–1.66) in A. latissima to 0.54 mg/mL (95% CI 0.35–0.82) in I. chiliantha. The data suggest increased ovicidal activity in the presence of higher concentrations of saponins (peaks 28, 34, 35, 36 and 37), phenylpropanoids (peaks 7, 12, 19 and 20) and flavonoid 21.
Few investigations have been carried out to determine the effect of phenylpropanoids on parasitic nematodes and data on nematicidal action are restricted to a small number of studies on fish, dog and swine parasites [27]. Recently, some phenylpropanoids have been evaluated with regard to the effects on nematodes of ruminant parasites; caffeic acid, p-coumaric acid, ferulic acid, methyl caffeate, methyl p-coumarate and methyl ferulate were identified as having ovicidal action [36] and chlorogenic acid was identified as having both ovicidal and larvicidal action [37] against H. contortus. The ovicidal effect of chlorogenic acid and its derivatives was also observed in the present study. This is the first report of the ovicidal effect of extracts containing these phenylpropanoids on H. placei. The concentration of monomeric and dimeric chlorogenic acid derivatives that enter into contact with the eggs of the parasite seems to be determinant for the improvement in activity, since this class was not able to inhibit larval hatchability satisfactorily at low concentrations, as verified for the ethanol extract from Melanthera latifolia, which was considered inactive in the present study. Other phenylpropanoids identified (rosmarinic acid, verbascoside and isoverbascoside) were also associated with ovicidal action for the first time. A similar condition was attributed to iridoids, which were evaluated for the first time on H. placei, and brevipolide, evaluated for the first time on nematodes. The evaluation of the anthelmintic potential of iridoids on ruminant GINs is restricted and only the iridoid aucubin has been cataloged for its transient inhibition of motility in third instar larvae of the bovine abomasum parasite Ostertagia ostertagi [38].
Conclusion
The present investigation demonstrates that the metabolomic technique was efficient at detecting secondary metabolites with ovicidal activity against H. placei. Thus, the use of metabolomics can be a tool to accelerate and simplify bioprospecting research with plant extracts in veterinary parasitology. Moreover, it was demonstrated that phenylpropanoids were the compounds with a higher correlation to biological activity. Therefore, these metabolites are implicated in the ovicidal action against H. placei. Other classes, such as flavonoids and triterpene saponins, may act synergistically, increasing the activity of phenylpropanoids.
Supporting information
Number 1–37 were related to the compounds identified in the Table 4.
(PDF)
(PDF)
*[M-2H]-2.
(PDF)
Acknowledgments
The authors wish to thank Professor Fernando Paiva for the technical assistance provided in the implementation of the cryopreservation protocol for the H. placei isolate.
Data Availability
All relevant data are within the manuscript and its Supporting Information files.
Funding Statement
The authors declare that we have received financial support from the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior - Brasil (CAPES), in the form of a master’s degree post-graduate scholarship; of the Fundação de Apoio ao Desenvolvimento do Ensino, Ciência e Tecnologia do Estado de Mato Grosso do Sul (FUNDECT) and the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), that made available of funds to purchase equipment used in the metabolomics studies. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1.Van Wyk IC, Goddard A, Bronsvoort BMC, Coetzer JAW, Handel IG, Hanotte O. et al. The impact of co-infection on the haematological profile of East African Short-Horn Zebu calves. Parasitology. 2014;141:374–388. 10.1017/S0031182013001625 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Gennari SM, Bressan MCRV, Rogero JR, MacLean JM, Duncan JL. Pathophysiology of Haemonchus placei infection in calves. Veterinary Parasitology. 1991; 38:163–172. [DOI] [PubMed] [Google Scholar]
- 3.Dimander SO, Höglund J, Spöorndly E, Waller PJ. The impact of internal parasites on the productivity of young cattle organically reared on semi-natural pastures in Sweden. Veterinary Parasitology. 2000; 90: 271–284. 10.1016/S0304-4017(00)00256-9 [DOI] [PubMed] [Google Scholar]
- 4.Borges FA, Almeida GD, Heckler RP, Lemes RT, Onizuka MKV, Borges DGL. Impact on tropical beef cattle productivity: effect on weight gain of weaned calves. Tropical Animal Health Production. 2013;45:723–727. 10.1007/s11250-012-0280-4 [DOI] [PubMed] [Google Scholar]
- 5.Kaplan RM, Vidyashankar AN. An inconvenient truth: Global worming and anthelmintic resistance. Veterinary Parasitology. 2012; 186: 70–78. 10.1016/j.vetpar.2011.11.048 [DOI] [PubMed] [Google Scholar]
- 6.Lopes WDZ, Teixeira WFP, Felippelli G, Cruz BC, Maciel WG, Soares VE, et al. Assessing resistance of ivermectin and moxidectin against nematode in cattle naturally infected using three different methodologies. Research in Veterinary Science. 2014; 96:133–138. 10.1016/j.rvsc.2013.11.001 [DOI] [PubMed] [Google Scholar]
- 7.Ramos F, Portella LP, Rodrigues FS, Reginato CZ, Potter L, Cezar AS, et al. Anthelmintic resistance in gastrointestinal nematodes of beef cattle in the state of Rio Grande do Sul, Brazil. International Journal for Parasitology: Drugs and Drug Resistance. 2016; 6: 93–101. 10.1016/j.ijpddr.2016.02.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Molento MB, Fortes F, Pondelek D, Borges FA, Chagas ACS, Torres-Acosta JFJ. Challenge of nematode control in ruminants: Focus on Latin America. Veterinary Parasitology. 2011;180:126–132. 10.1016/j.vetpar.2011.05.033 [DOI] [PubMed] [Google Scholar]
- 9.Sandoval-Castro CA, Torres-Acosta JFJ, Hoste H, Salem AZM, Chan-Pérez JI. Using plant bioactive materials to control gastrointestinal tract helminths in livestock. Animal Feed Science and Technology. 2012; 176:192–201. [Google Scholar]
- 10.Zurmic Z, Blache D. Bioactive plants and plant products: Effects on animal function, health and welfare. Animal Feed Science and Technology. 2012;176:150–162. 10.1016/j.anifeedsci.2012.07.018 [DOI] [Google Scholar]
- 11.Borges DGL, Borges FA. Plants and their medicinal potential for controlling gastrointestinal nematodes in ruminants. Nematoda. 2016; 3. [Google Scholar]
- 12.Ribeiro WLC, Andrea WPP, Cavalcante GS, Araújo-Filho JV, Santos JML, Macedo ITF. Effects of Spigelia anthelmia decoction on sheep gastrointestinal nematodes. Small Ruminant Research. 2017; 153: 146–152. [Google Scholar]
- 13.Gaudêncio SP, Pereira F. Dereplication: racing to speed up the natural products discovery process. Natural product reports. 2015; 32(6): 779–810. 10.1039/c4np00134f [DOI] [PubMed] [Google Scholar]
- 14.Ayouni K, Borboucha-Rohmani M, Kim HK, Atmani D, Verpoorte R, Choi YH. Metabolomic tool to identify antioxidant compounds of Fraxinus angustifolia leaf and stem bark extracts. Industrial Crops and Products. 2016; 88: 65–77. 10.1016/j.indcrop.2016.01.001 [DOI] [Google Scholar]
- 15.Chassagne F, Haddad M, Amiel A, Phakeovilay C, Manithip C, Bourdy G, et al. A metabolomic approach to identify anti-hepatocarcinogenic compounds from plants used traditionally in the treatment of liver diseases. Fitoterapia. 2018. 10.1016/j.fitote.2018.02.021 [DOI] [PubMed] [Google Scholar]
- 16.Feliz DC. Resistência a anti-helmínticos em nematodas gastrintestinais de bovinos de corte, no Mato Grosso do Sul, Brasil. 2011. 41f. Dissertação (Mestrado)—Universidade Federal de Mato Grosso do Sul, Campo Grande.
- 17.Gordon HM, Whitlock HV. A new technique for counting nematode eggs in sheep faeces. Journal of Council for Scientific Industrial Research. 1939; 12(1): 50–52. [Google Scholar]
- 18.Coles CG, Bauer C, Borgsteede FHM, Geerts S, Klei TR, Taylor MA, Waller PJW. World Association for the Advancement of Veterinary Parasitology (W.A.A.V.P) methods for the detection of anthelmintic resistance in nematodes of veterinary importance. Veterinary Parasitology. 1992;44:35–44. [DOI] [PubMed] [Google Scholar]
- 19.Bizimenyera ES, Githiori JB, Eloff JN, Swan GE. In vitro activity of Peltophorum africanum Sond. (Fabaceae) extracts on the egg hatching and larval development of the parasitic nematode Trichostrongylus colubriformis. Veterinary Parasitology. 2006; 142:336–343. 10.1016/j.vetpar.2006.06.013 [DOI] [PubMed] [Google Scholar]
- 20.Lommen A. MetAlign: interface-driven, versatile metabolomics tool for hyphenated full-scan mass spectrometry data preprocessing. Analytical Chemistry. 2009; 81(8): 3079–3086. 10.1021/ac900036d [DOI] [PubMed] [Google Scholar]
- 21.Tikunov YM, Laptenok S, Hall RD, Bovy A, De Vos RCH. MSClust: a tool for unsupervised mass spectra extraction of chromatography-mass spectrometry ion-wise aligned data. Metabolomics. 2012; 8 (4):714–718. 10.1007/s11306-011-0368-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Xia J, Wishart DS. Using MetaboAnalyst 3.0 for Comprehensive Metabolomics Data Analysis. Current Protocols in Bioinformatics. 2016. 10.1002/cpbi.11 [DOI] [PubMed] [Google Scholar]
- 23.Titman CM, Downs JA, Oliver SG, Carmichael PL, Scott AD, Griffin JL. A metabolomic and multivariate statistical process to assess the effectsof genotoxins in Saccharomyces cerevisiae. Molecular BioSystems. 2009; 5: 1913–1924. 10.1039/b907754e [DOI] [PubMed] [Google Scholar]
- 24.de Oliveira Dembogurski DS, Trentin DS, Boaretto AG, Rigo GV, Silva RC, Tasca T, et al. Brown propolis-metabolomic innovative approach to determine compounds capable of killing Staphylococcus aureus biofilm and Trichomonas vaginalis. Food Research International. 2018;111:661–673. 10.1016/j.foodres.2018.05.033 [DOI] [PubMed] [Google Scholar]
- 25.Botura MB, Santos JDG, Silva GD, Lima HG, Oliveira JVA, Almeida MA, et al. In vitro ovicidal and larvicidal activity of Agave sisalana Perr. (sisal) on gastrointestinal nematodes of goats. Veterinary Parasitology. 2013; 192: 211–217. 10.1016/j.vetpar.2012.10.012 [DOI] [PubMed] [Google Scholar]
- 26.Klongsiriwet C, Quijada J, Williams AR, Mueller-Harvey I, Williamson EM, Hoste H. Synergistic inhibition of Haemonchus contortus exsheathment by flavonoid monomers and condensed tannins. International Journal for Parasitology: Drugs and Drug Resistance. 2015; 5:127–134. 10.1016/j.ijpddr.2015.06.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Spiegler V, Liebau E, Hensel A. Medicinal plant extracts and plant-derived polyphenols with anthelmintic activity against intestinal nematodes. Natural Products Report. 2017; 34:627–643. 10.1039/c6np00126b [DOI] [PubMed] [Google Scholar]
- 28.Morais-Costa F, Soares ACM, Bastos GA, Nunes YRF, Geraseev LC, Braga FC, et al. Plants of the Cerrado naturally selected by grazing sheep may have potential for inhibiting development of Haemonchus contortus larva. Tropical Animal Health Production.2015; 47:1321–1328. 10.1007/s11250-015-0866-8 [DOI] [PubMed] [Google Scholar]
- 29.Mengistu G, Hoste H, Karonene M, Salminene JP, Hendriks WH, Pellikaan WF. The in vitro anthelmintic properties of browse plant species against Haemonchus contortus is determined by the polyphenol content and composition. Veterinary Parasitology. 2017; 237:110–116. 10.1016/j.vetpar.2016.12.020 [DOI] [PubMed] [Google Scholar]
- 30.Féboli A, Laurentiz AC, Soares SCS, Augusto JG, Anjos LA, Magalhães LG, et al. Ovicidal and larvicidal activity of extracts of Opuntia ficus-indica against gastrointestinal nematodes of naturally infected sheep. Veterinary Parasitology. 2016; 226:65–68. 10.1016/j.vetpar.2016.06.030 [DOI] [PubMed] [Google Scholar]
- 31.Hoste H, Torres-Acosta JFJ. Non chemical control of helminths in ruminants: Adapting solution for changing worms in a changing world. Veterinary Parasitology. 2011;180:144–154. 10.1016/j.vetpar.2011.05.035 [DOI] [PubMed] [Google Scholar]
- 32.Mondal H, Hossain H, Awang K, Saha S, Mamun-Ur-Rashid S, Islam MK, et al. Anthelmintic Activity of Ellagic Acid, a Major Constituent of Alternanthera sessilis Against Haemonchus contortus. Pakistan Veterinary Journal.2015; 35:58–62. [Google Scholar]
- 33.Engstrom MT, Karonen M, Ahern JR, Baert N, Payre B, Hoste H, Salminen JP. Chemical Structures of Plant Hydrolyzable Tannins Reveal Their in Vitro Activity against Egg Hatching and Motility of Haemonchus contortus Nematodes. Journal of Agricultural and Food Chemistry. 2016; 64:840–851. 10.1021/acs.jafc.5b05691 [DOI] [PubMed] [Google Scholar]
- 34.Argentiniere MP, D’Addabbo T, Tava A, Agostinelli A, Jurzysta M, Avato P. Evaluation of nematicidal properties of saponins from Medicago spp. European Journal of Plant Pathology. 2008; 120: 189–197. [Google Scholar]
- 35.D’Addabbo T, Carbonara T, Leonetti P, Radicci V, Tava A, Avato P. Control of plant parasitic nematodes with active saponins and biomass from Medicago sativa. Phytochemistry Reviews. 2011; 10: 503–519. 10.1007/s11101-010-9180-2 [DOI] [Google Scholar]
- 36.Castillo-Mitre GF, Olmedo-Juárez A, Rojo-Rubio R, González-Cortázar M, Mendoza-de Gives P, Hernández-Beteta EE, et al. Caffeoyl and coumaroyl derivatives from Acacia cochliacantha exhibit ovicidal activity against Haemonchus contortus. Journal of Ethnopharmacology. 2017; 204:125–131. 10.1016/j.jep.2017.04.010 [DOI] [PubMed] [Google Scholar]
- 37.Díaz GJ, Hernandez GT, Zamilpa A, Perez MBC, Bribiesca JER, Mendo OH. In vitro assessment of Argemone mexicana, Taraxacum officinale, Ruta chalepensis and Tagetes filifolia against Haemonchus contortus nematode eggs and infective (L3) larvae. Microbial Pathogenesis. 2017; 109: 162–168. 10.1016/j.micpath.2017.05.048 [DOI] [PubMed] [Google Scholar]
- 38.Gustine DL, Sanderson MA, Getzie J, Donner S, Gueldner R, Jennings N. 2010. A strategy for detecting natural anthelmintic constituents of the grassland species Plantago lanceolata. < http://www.internationalgrasslands.org/publications/pdfs/id1108.pdf > (accessed 08.09.17).
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Number 1–37 were related to the compounds identified in the Table 4.
(PDF)
(PDF)
*[M-2H]-2.
(PDF)
Data Availability Statement
All relevant data are within the manuscript and its Supporting Information files.