Abstract
There are relatively few investigations of the emotion expressivity of children at risk for the later development of schizophrenia and schizophrenia-spectrum disorders. Using data from the New York High-Risk Project, we compared children’s emotional expressivity during a semi-structured videotaped interview. Data were coded for 173 child subjects: 61 with schizophrenic parents (HRSz); 54 with affectively ill parents (HRAff); and 58 with psychiatrically “normal” parents (NC). A child’s affective responses were rated for the presence of discrete positive, negative, or neutral emotions by coders naive to group membership. Responses were also rated for anxiety, flat affect, inappropriate affect, and emotional withdrawal/disengagement. Compared with the two other two groups, HRSz children displayed significantly more negative affect in response to questions regarding their most negative experiences and, when questioned about their self-concept, they displayed significantly less positive affect. Both HRSz and HRAff children showed more inappropriate affect than NC children. Significantly more HRSz children were rated as demonstrating a lack of emotional engagement. Children making inappropriate displays of positive affect while discussing a negative topic were most likely to manifest a psychiatric disorder as an adult. These findings suggest that inappropriate affect may be a nonspecific indicator of risk for psychopathology. Emotional withdrawal in childhood may be a potential indicator of risk for schizophrenia.
Keywords: genetic high-risk, schizophrenia, affect, emotional expressivity
1. INTRODUCTION
There is longstanding interest in children at risk for schizophrenia. This is largely due to recognition that the years prior to schizophrenic psychoses manifestation in adulthood are characterized by atypical and/or abnormal development [1]. The notion that adulthood psychopathology is preceded by signs of childhood behavioral and/or emotional disturbance is consistent with conceptualizations of schizophrenia-related psychoses as neurodevelopmental in origin [2]. Since Kraepelin [3], affective disturbances have been considered central to schizophrenia. Affective disturbances in schizophrenia include affective experience, and expression, impairments such as anhedonia and flat, or blunted, affect, increased emotional arousal and reactivity, and anxiety symptoms [4]. As emotional disturbances and affective dysregulation are observed in various forms of psychopathology, it is useful to examine whether specific aspects of emotional dysfunction associate with heightened risk for schizophrenia.
There are relatively few investigations into the emotional expressivity of children at risk for the later development of schizophrenia and schizophrenia-spectrum disorders. Using childhood home movies of schizophrenia patients and their healthy siblings, Walker, and her colleagues [5,6], compared the facial expressions of emotions from infancy through adolescence. This archival-observational method proved quite fruitful in that it revealed that both male and female pre-schizophrenic children showed greater negative emotion than same-gender siblings who did not later develop schizophrenia. Comparative analyses also revealed that pre-schizophrenic girls, but not boys, displayed a significantly lower proportional duration of joy responses than their healthy siblings.
Videotaped data from the Copenhagen High-Risk Study [7] revealed that individuals who developed schizophrenia in adulthood could be discriminated from those who did not on the basis of their lunchtime eating behavior. Specifically, children who later developed schizophrenia smiled and vocalized less, whether initiated by themselves or another child, than children who either developed a non-schizophrenic form of psychopathology, or did not develop a mental illness.
There are also relevant prior findings from the New York High-Risk Project (NYHRP). The NYHRP is a prospective, longitudinal, high-risk study of initially unaffected and untreated offspring of individuals with schizophrenia, major affective disorders, and those without psychiatric disorders. The project consisted of two independent samples—Sample A and Sample B—who were followed continuously since recruitment through the period of greatest lifetime risk for schizophrenia, up through a mean age of 42 years. Based upon a subset of Sample B, Dworkin et al. [8] found that ratings of the videotaped interviews of the high-risk offspring revealed significant differences in social competence, such as emotional withdrawal, as defined by the Premorbid Adjustment Scale [9]; at ages 12 and 15, and affective flattening at 15. In another analysis of Sample B [10], the children of schizophrenia patients showed significantly fewer smiles per minute during adolescence and greater affective flattening. It is noteworthy that the reports by Dworkin and colleagues were based on a subset of the NYHRP dataset, and before all the children had passed through the period of greatest risk.
The present study is an extension of the earlier analyses using the NYHRP interview data. In addition to comparing the offspring of parents with psychiatric disorders in terms of their emotional expressions in response to questions during a semi-structured interview, we also compared offspring groups in terms of the affective quality of the child/interviewer interaction. The degree of child emotional engagement versus emotionally withdrawal was examined. The present report extends earlier work by following the subjects through the period of greatest lifetime risk for schizophrenia.
We were interested in exploring whether an association between increased negative displays and risk for the later development of schizophrenia would be found. Adult-onset schizophrenia patients display fewer negative facial displays compared to healthy controls despite experiencing the same amount of emotion [11]. When comparing young offspring of schizophrenic, depressed, and well mothers, Goodman [12] observed less negative affect among the offspring of mothers with schizophrenia. Walker et al. observed that pre-schizophrenic children exhibited more negative emotional facial expressions than their unaffected siblings. We hypothesized that children of schizophrenia patients would display more negative affect than the other children based upon prior NYHRP findings based on a subset of the sample and findings based of the Walker’s archival-observational study.
We hypothesized that we would find an association between reduced positive displays and the risk for the later development of schizophrenia. The basis for this hypothesis is both conceptual and empirical. First, schizophrenia patients display fewer positive facial displays compared with non-schizophrenia patients in response to a range of emotion-eliciting stimuli [11]. Secondly, we reasoned that the offspring of schizophrenia patients might show similar expressive affectivity as their mothers with schizophrenia, due to genetic, social learning, and/or transactional developmental processes. Finally, fewer positive facial displays among those at heightened risk for the later development of schizophrenia would be consistent with the earlier findings of Walker and colleagues.
We also included other affective indicators that could be viewed as interpersonally maladaptive, such as low positive affect, anxiety, inappropriate affect, and/or flat affect. We examined whether any of these affective indicators of risk for the later development of psychopathology would be relatively specific for schizophrenia. As children of mothers with depression are at risk for the later development of psychopathology [13,14], that offspring group served as a psychiatric comparison.
We hypothesized that both groups of children of psychiatric patients would display higher rates of anxiety relative to children of psychiatrically healthy women. Extant research literature provides additional support for our a priori hypothesis. Birth cohort studies such as [15] indicate that anxiety is a childhood predictor of later adult psychiatric disorder. Epidemiological studies [16] identify anxiety symptoms as a risk factor in the development of adult schizophrenia. Children and adolescents at genetic risk for schizophrenia had significantly higher anxiety scores than healthy comparison peers [17]. Children of unipolar depressed women are reported as showing significantly more internalizing behavior problems, including anxiety, than other groups of children such as children of bipolar women [18] and children of psychiatrically healthy women [19,20].
We also reasoned that the increased rates of anxiety might partly reflect the additional stress associated with living with a parent suffering from a chronic illness and dealing with functional impairments.
The affective system, thought to be composed of neural processes, expressive behavior, and subjective experience, interacts and is coordinated with the cognitive system and affective-cognitive control structures during typical development [21]. If schizophrenia is a disorder of “pandysmaturation” [22] or “cognitive dysmetria” [23], then individuals at heightened genetic risk for the disorder may be expected to display less control over the processes regulating the output of their emotional system. Individuals with less control over such processes, as well as less control over the coordination between their affective and developing cognitive systems, are more likely to manifest disturbances such as faulty connection between their emotional display and the emotions expressed, i.e., inappropriate affect. We hypothesized that individuals with a genetic diathesis for schizophrenia (i.e., the offspring of schizophrenic patients) might be more likely to display inappropriate affect.
Blunted, or flat, affect, a reduction in the intensity of expressed feeling, is considered a core schizophrenia symptom [4]. In the New York Infant Study, one of the earliest indications of an association between emotional abnormalities and a schizophrenia diathesis, four out of five of the offspring of a mother with schizophrenia displayed affect described as flat, detached, or distant [24].
Earlier investigators [25–27] reported associations between affective deficits and neuromotor dysfunction. More recently, Lee et al. [28] used functional brain imaging to demonstrate how schizophrenia patients with flat affect showed functional disturbance in the mirror neuron system. We expected that a larger proportion of HRSz offspring would display flat affect compared to the other offspring groups.
We were interested in examining whether high-risk offspring displayed emotional withdrawal. Social withdrawal was previously observed among school-age children—especially males who were at genetic risk for schizophrenia—in both the Jerusalem Infant Development Study and the Israeli High-Risk Study [29,30]. Prior investigations, based on the National Child Development Study using teacher ratings [31], and a prospective followup study of offspring of mothers with psychotic disorders [32] suggested that childhood social inhibition and emotional withdrawal might be an antecedent to schizophrenia. Retrospective parental and caregiver reports [33,34] indicate that emotional and social withdrawal in mid-childhood associates with heightened risk for a adulthood schizophrenia-related outcome. Moreover, prodromal studies [35,36] indicate that individuals with non-affective psychosis typically display social withdrawal in the years preceding their first psychotic episode. We thought it likely that a greater proportion of the schizophrenia offspring group would display emotional withdrawal compared to the other offspring groups.
2. MATERIALS AND METHODS
2.1. New York high-risk project
All participants were drawn from the New York High-Risk Project (NYHRP). The NYHRP was a prospective, longitudinal study of the offspring of schizophrenic, affectively ill, and psychiatrically normal parents (HRSz, HRAff, and NC offspring groups, respectively). Recruitment procedures, parental diagnoses, and longitudinal follow-up details appear elsewhere [37–39]. Mentally ill parents were identified through consecutive admissions at several large New York State psychiatric facilities. They were recruited if they met the study criteria, including, but not limited to, having at least one biological child aged 7–12 inclusive. All offspring were Caucasian, English-speaking, and free of mental retardation/intellectual disability, major psychiatric disorders or treatment for emotional problems at the time of recruitment in 1971–1972 (Sample A) or 1977–1979 (Sample B) at ages 9.5 ± 1.7 and 9.0 ± 1.8 (mean ± SD), respectively.
Two independent samples were recruited. The addition of a second sample allowed a test of the replicability of findings from the first sample as well as a way of testing additional hypotheses suggested by data generated from Sample A. Since inception the study has had 7 rounds of assessments, with telephone interviews at least once annually. Offspring were followed from mid-childhood (7–12) through mid-adulthood. After a complete description of the study, written informed consent was obtained from the parents for themselves and their children starting in round 1 and individually from the children who had reached 18 in subsequent rounds. Institutional Review Board approval was obtained for each phase of the NYHRP.
2.2. Participants
Our subjects were drawn from both independent samples of the New York High-Risk Project. We had the usable videotaped interviews from both mid-childhood and adolescence and diagnostic interviews from adulthood for 173 offspring. The combined sample consisted of 61 offspring of schizophrenic parents (HRSz group); 54 offspring of affectively ill parents (HRAff group); and 58 offspring of psychiatrically normal parents (NC group). Child mean age at the time of the videotaped interviews was 10.65 years (range, 7–15).
2.3. Videotaped childhood interviews
During the first, second, and third testing rounds, child psychiatrists who were naive to subject group membership administered 30 min semi-structured videotaped interviews pertaining to subject family and home life, school and extracurricular activities, fantasy life, and self-concept. These interviews were subsequently rated for psychopathology and adaptive functioning using the Mental Health Assessment Form (MHAF) [40]. These videotapes were later transferred to DVD and coded by trained raters who were naive to group membership and/or adult outcome. A child’s affect was rated during selected segments of the interviews.
2.4. Selection of coding segments
The primary author, who was naive to group membership and adulthood outcome diagnoses, was responsible selecting which segments were to be coded. Before actual rating had begun, the project coordinator provided the primary author with a representative sampling of videotaped interviews. These videotapes consisted mostly of target subjects’ siblings (n = 20) who were not included in the final sample. The objective was to select questions that would be the most likely to elicit positive, or negative, emotional responses from the child and that were queried consistently and frequently enough to insure that there would be sufficient data to code. We selected certain questions that were expected to elicit positive affect (e.g., “What was the best thing that ever happened to you? What was the happiest thing that ever happened to you?”) or negative affect (e.g., “What was the worst thing that ever happened to you? What’s the saddest thing that ever happened to you?”). We also selected questions that were likely to elicit a positive, negative, or neutral, affective response. These types of questions tended to vary by age. Younger children were asked (“Are you happy or are you sad most of the time” whereas older children were asked (“What’s your general mood like?”). All children were asked “What would you change about yourself?” or “Is there anything that you would change about yourself?” and all children were asked either about day dreaming/mind-wandering or dreaming at night. The number of subjects who were rated on each question varied somewhat as a result of videotape deterioration, as well as differences in quality of the available recording [41]. Only one interview session for each child was used. The session used was the session that had the greatest number of codable segments (i.e., questions). It is noteworthy that mental health professionals administering the MHAF were querying the children about their experiences, while the research assistants were rating the child’s affective expressions as they were recounting those experiences.
2.5. Coding system and training in coding procedures
After careful consideration of extant coding systems for rating affective responses in children based primarily upon micro-analysis of musculature, we combined that information with vocal prosody ratings and overall behavioral observations. Research assistants who rated the offspring’s emotional expressivity received extensive training with Ekman faces, as well as additional consultation with an expert in child emotions. Further training was conducted using videotapes of study participant siblings. Inter-rater reliability was determined by having three coders independently code 50% of these video records. After attaining an average 0.85 agreement, participant video coding proceeded. In order to assess inter-rater reliability, 10% of all interviews were randomly selected and recoded by a second observer. Calibration meetings were held at regular intervals with the primary trainer. Inter-rater reliability for specific emotions and qualitative ratings was assessed by calculating intra-class correlations and kappa statistics, respectively [42].
2.6. Ratings of emotional expressivity
Initially, although individual emotions were identified and coded, they were classified as positive, negative, or neutral [43]. The following emotions were coded: positive (happiness, joy/pleasure, relief, satisfaction/pride, excitement, confidence, surprise, humor, and interest); neutral (calmness); and, negative (tension, worry, hostility/antipathy, boredom/apathy, annoyance/irritation, frustration/ anger, sadness/anguish, pain/distress, and disgust). Behavioral anchors at each point of emotional intensity were provided.
All non-neutral emotional expressions were rated on 3 point scales, which were coded as mild, moderate, and severe expressions, as ±1, ±2, and ±3, respectively. The positive and negative signs denoting emotional valence. For example, moderate affect was rated as “2”; if it was positive, it was a “+2” and if negative, it was a “−2”.
2.7. Ratings for the presence of maladaptive affective and social behaviors
A child’s affective, and social, behaviors during selected interview segments were also rated for the presence of possible indicators of later psychopathological risk, such as anxiety, flat affect, inappropriate affect, and emotional withdrawal. If the behavior was rated as “present”, it was also rated in terms of intensity. Anxiety behavioral indices were operationally defined as including excessive fidgeting, lip- and/or nail biting, and playing with the hair or the microphone. Anxiety could therefore be rated as mild, moderate, or severe. We relied upon 3 of the 4 SANS indices [44] for the operational definition of flat affect, i.e., a child was rated as displaying flat affect when they displayed unchanging facial expression, affective non-responsivity, and/or lack of vocal inflection. A child was determined to demonstrate inappropriate affect if there was a discrepancy between their facial expression and the topic discussed. The most frequent type of inappropriate affect was the display of positive affect while discussing a negatively-toned topic, e.g., smiling while talking about riding one’s bicycle into a concrete wall. Emotional withdrawal was operationalized as behavioral evidence of a lack of emotional engagement during the interviews, including, but not limited to, poor eye contact, or the child appearing distant, inattentive, cold, or aloof. An extreme example of emotional withdrawal would be a total lack of communication. A child’s expressiveness affect ratings during the interview were related to the topic discussed and the range of feelings expressed. Emotional withdrawal ratings were based upon the quality of child-interviewer relatedness during the interview.
Emotional withdrawal was rated on a dichotomous scale, “0” or “1”, for the presence or absence, respectively, of emotional engagement. If a child was observed to display emotional withdrawal, then they were also rated for the degree to which they displayed lack of emotional engagement, ranging from mild to severe.
2.8. Assessment of Outcome psychiatric diagnoses
During rounds 4 through 6, the Schedule for Affective Disorders and Schizophrenia Lifetime Version (SADS-L) [45] was administered to all participants 18, and older, by trained clinical psychologists and psychiatric social workers naive to parental diagnostic group and offspring outcome diagnoses in adulthood in order to assess Axis I disorders based on the Research Diagnostic Criteria [46]. Final diagnostic evaluations were conducted in 2002, at mean ages 39.4 ± 1.8 (Sample A) and 34.1 ± 2.3 (Sample B). Detailed diagnostic evaluation descriptions were given previously [37–39]. In addition to the SADS-L, the diagnostic evaluation included all other clinical data: research interviews, psychiatric hospital records, and when relevant, therapist notes and comments.
Adulthood Axis I disorders were categorized according to the following hierarchy : (a) schizophrenia-related psychoses (SRP; including schizophrenia, unspecified functional psychosis, and schizoaffective disorder, mainly schizophrenia, as defined in the RDC); (b) affective psychosis (psychotic major depression, bipolar I with psychosis, bipolar II with psychosis, manic psychosis, and schizoaffective disorder, mainly affective, as defined in the RDC); (c) non-psychotic affective disorders (e.g., major depression); (d) other major Axis I disorders (e.g., anxiety disorders, substance abuse disorders); (e) drug-related psychosis; and, (f) no disorder. Participants were also evaluated for the presence of schizotypal features using the Personality Disorders Examination (PDE) [47], a semi-structured clinical interview designed to assess all DSM-III-R Axis II disorders. The diagnostic algorithm provided in the PDE was used [48].
2.9. Data reduction and statistical analysis
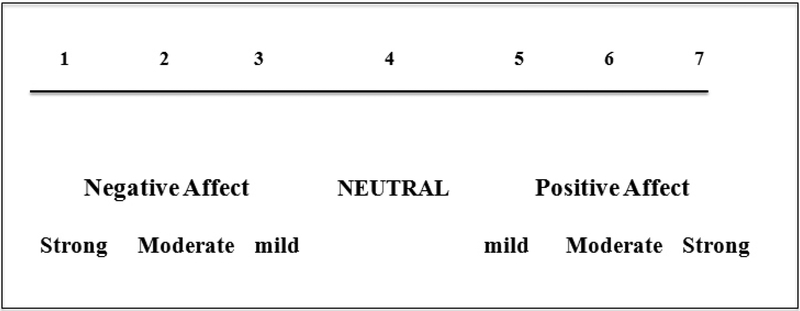
After affective expressivity ratings were made, they were recoded onto a single 7-point rating scale to facilitate cross-question comparisons (Fig. 1). On this 7-point scale, the recoded scores ranged from 1, denoting a strongly negative emotional expression to 7, signifying a strongly positive emotional expression. This method allows for simultaneously conveying valence and intensity of the child’s response to each of the questions posed.
Fig. 1.
Positive and negative affect were initially rated separately, on a scale of 1 (mild) to 3 (strong) for each, with the positive or negative sign denoting the valence. The coding ratings were subsequently placed on a singular scale to facilitate comparisons across questions. As can be seen, 1 was strongly negative, 4 was neutral, and 7 was strongly positive.
Interrater agreement for the video ratings was calculated using Cohen’s kappa [42]. We used univariate analysis of variance (ANOVA) tests to compare the groups in terms of mean levels of affective expressivity in responses to questions. Tukey’s tests were used for follow-up analyses. In order to compare groups in terms of the presence of anxiety, flat affect, inappropriate affect, and/or emotional withdrawal, we used chi-square analysis. All statistical tests of group differences were two-tailed, with the significance (alpha) levels set at p = 0.05.
3. RESULTS
3.1. Demographic characteristics
Age and gender breakdowns for each offspring group appear in Table 1. Mean age among three groups did not differ, F (2, 170) = 0.19, n.s.. The mean age of the sample was 10.65 (±1.8) years. The HRAff group had a higher proportion of females than the other two groups, Χ2 (2) = 14.58, p < 0. 01 [49].
Table 1.
Demographic characteristics of the three offspring groups.
| Characteristic | All | HRSz | HRAff | NC |
|---|---|---|---|---|
| N | 173 | 61 | 54 | 58 |
| Gender | ||||
| Male | 89 | 33 | 17 | 39 |
| Female | 84 | 28 | 37 | 19 |
| Agea | 10.65 ± 1.8 | 10.69 ± 1.9 | 10.74 ± 1.7 | 10.53 ± 1.9 |
Mean ± SD.
3.2. Group comparisons by parental risk status
ICC’s for the ratings of positive, neutral, and negative emotional displays were 0.96, 0.97, and 0.80, respectively. Emotion expressivity mean ratings by offspring group and interview question appear in Table 2. Children tended to show more positive emotions in response to questions pertaining to their best, or happiest, experiences. The groups did not differ in positive emotion demonstrations in response to questions regarding their most positive experiences, F (2, 93) = 1.96, n.s. All groups exhibited more negative emotions in response to questions pertaining to their worst, or saddest, experience. HRSz children displayed significantly more negative affect than the other children in response to questions regarding their most negative experiences, F (2, 136) = 4.35, p = 0.02, η2 = 0.06. The HRSz group displayed more negative affect than the HRAff group [t (82) = −2.10, p = 0.04] and the NC group [t (93) = −3.05, p = 0.003].
Table 2.
Mean ratings of affective expressivity by offspring groupa.
| Interview questionb | HRSz | HRAff | NC | p |
|---|---|---|---|---|
| Best/Happiestc | 5.22 ± 1.3 | 5.67 ± 0.8 | 5.59 ± 0.75 | n.s. |
| Worst/Saddestc | 3.50 ± 1.46 | 4.23 ± 1.83 | 4.38 ± 1.36 | * |
| Change in Selfc | 4.67 ± 1.47 | 5.58 ± 0.84 | 5.45 ± 1.12 | * |
| General Moodc | 4.85 ± 1.00 | 5.24 ± 1.23 | 5.11 ± 1.16 | n.s. |
| DayDreamingc | 4.96 ± 1.54 | 5.20 ± 1.15 | 5.27 ± 0.77 | n.s. |
| Dreams at Nightc | 5.31 ± 1.05 | 5.48 ± 1.37 | 5.15 ± 1.17 | n.s |
The ratings are on a 7-point scale. 1 denotes the most strongly negative emotional expression and 7 denotes the most strongly positive emotional expression.
A child’s emotional expressions were rated during their response to semi-structured interview probes by child psychiatrists. See the text for questions. HRSz = offspring at risk for schizophrenia; HRAff = offspring at risk for affective disorders; and NC = offspring of psychiatrically-healthy parents.
The n’s varied slightly in terms of the availability of useable data for analyses of discrete emotions. The n’s ranged from 25–48 for the HRSz group, from 19–44 for the HRAff group, and from 22–47 for the NC group.
p < 0.05.
We also observed a significant group difference when the children were questioned about their self-concept, i.e., how much did they like themselves or what would they change about themselves. The HRSz group showed significantly less positive affect than the other two groups, F (2, 78) = 4.59, p = 0.01, η2 = 0.11. The HRSz children displayed less positive affect than either the HRAff children [t (50) = −2.85, p = 0.01] or the NC children [t (60) = −2.33, p = 0.02].
The three groups did not differ significantly in affective display while describing their general mood, F (2, 92) = 0.94, n.s. The three risk groups did not differ in their affective responses to non-emotionally-charged questions (e.g., describing day dreaming or mind-wandering [F (2, 66) = 0.43, n.s.]), or when describing dream content [F (2, 115) = 0.74, n.s.].
Table 3 provides the number of participants in each offspring group who evidenced anxiety, flat affect, inappropriate affect, and/or emotional withdrawal during the MHAF coded segments. Kappa value for the anxiety ratings was 0.87. Over 75% of the children in each offspring group showed some signs of anxiety while responding to at least one of the coded questions. Overall, the groups did not differ in terms of the proportion of children evidencing anxiety. However, we observed a trend in the two high risk groups to display moderate anxiety during the interview compared with the NC children, Χ2 (4) = 5.55, p = 0.06.
Table 3.
Number (and percent) displaying affective behavior by offspring group.
| Behaviora, b | HRSz | HRAff | NC |
|---|---|---|---|
| Anxiety | |||
| No | 10 (16.4) | 8 (14.8) | 13 (22.4) |
| Mild | 24 (39.3) | 25 (46.3) | 31 (53.4) |
| Moderate | 27 (44.3) | 21 (38.9) | 14 (24.1) |
| Any anxiety | 51 (83.6) | 46 (85.2) | 45 (77.6) |
| Flat Affect | 11 (18.0) | 6 (11.1) | 8 (13.8) |
| Inappropriate Affect | 23 (37.7) | 21 (38.9) | 3 (15.8) |
| Emotional Withdrawal | 32 (52.5) | 10 (18.5) | 16 (27.6) |
Operational definitions for these behaviors appear in Methods.
Cohen’s kappa values for interrater agreement appear in Results. HRSz = offspring at risk for schizophrenia; HRAff = offspring at risk for affective disorder; and, NC = offspring of psychiatrically healthy parents.
Inter-rater reliability for flat affect ratings, as calculated by kappa, was 0.68. In this offspring sample, flat affect was less frequently observed than anxiety, inappropriate affect, or emotional withdrawal. The three offspring groups did not differ significantly in terms of the proportion of children displaying flat affect, Χ2 (2) = 1.14, p = 0.57 and there was no gender difference in this regard.
The kappa value for inter-rater reliability for inappropriate affect was 0.85. HRSz and HRAff children were both more likely to display inappropriate affect than NC children, Χ2 (2) = 21.35, p < 0.001, and Cramer’s V = 0.35. The HRSz group did not show gender differences in terms of inappropriate affect, Χ2 (1) = 0.05, n.s. Similarly, there were no gender differences in the HRAff, or NC, group, Fisher’s Exact Test, p = 0.14 and 0.25, respectively.
Inter-rater reliability for emotional withdrawal ratings, as measured by Cohen’s kappa, was 0.79. A significantly larger proportion of the HRSz children displayed emotional withdrawal than HRAff and NC children, Χ2 (2) = 16.19, p < 0.001, and Cramer’s V = 0.31. Males in the NC offspring group were more likely to be rated as showing emotional withdrawal than the females, Χ2 (1) = 4.12, p < 0.05. This was not the case with either the HRSz group [Χ2 (1) = 1.91, n.s.], or the HRAff group [Fisher’s exact test, p = 0.48].
3.3. Group comparisons by adulthood diagnostic Outcome
Table 4 presents the distribution of participants by offspring group and adulthood outcome diagnoses. Of the twenty offspring who developed either schizophrenia-related psychoses or affective psychoses in adulthood, 65% (13) were from the HRSz group and 30% (n = 6) were from the HRAff group.
Table 4.
Frequency of participants by offspring group and adulthood diagnostic outcome.
| Adult diagnostic outcome | Offspring group |
|||
|---|---|---|---|---|
| HRSz | HRAff | NC | Total | |
| Any psychotic diagnosisa | 13 | 6 | 1 | 20 |
| Any nonpsychotic Axis I diagnosis | 27 | 35 | 15 | 77 |
| No diagnosis (Healthy) | 21 | 13 | 42 | 76 |
| Total | 61 | 54 | 58 | 173 |
HRSz = high-risk for schizophrenia; HRAff = high-risk for affective disorders; NC = normal controls.
Any psychotic diagnosis included schizophrenia-related psychotic disorders and affective psychoses; Nonpsychotic Axis I diagnosis included nonpsychotic major affective disorders, anxiety disorders, and substance abuse disorders.
Given the relatively small size of some of the outcome diagnostic groups, we compared the groups in terms of psychotic disorders versus nonpsychotic disorders, and psychiatric disorder versus no psychiatric disorder. The mean ratings of emotion expressivity by adult diagnostic outcome and interview question appear in Table 5. None of the displays of discrete emotions during mid-childhood were associated with adult diagnostic outcomes.
Table 5.
Emotion expressivity mean ratings by adult diagnostic outcome group.
| Interview question | Any psychotic disorder | Any nonpsychotic disorder | No psychiatric disorder | p |
|---|---|---|---|---|
| Best/Happiest | 5.42 ± 1.0 | 5.57 ± 0.8 | 5.40 ± 1.2 | n.s. |
| Worst/Saddestc | 3.93 ± 1.75 | 3.84 ± 1.55 | 4.25 ± 1.60 | n.s. |
| Change in Self | 5.14 ± 1.22 | 5.33 ± 1.18 | 4.97 ± 1.40 | n.s. |
| General Moodc | 4.80 ± 1.10 | 4.98 ± 1.28 | 5.16 ± 0.98 | n.s. |
| Daydreaming | 5.71 ± 0.95 | 5.09 ± 1.20 | 5.04 ± 1.27 | n.s. |
| Dreams at nightc | 5.82 ± 0.87 | 5.28 ± 1.37 | 5.22 ± 1.03 | n.s. |
The ratings are on a 7 point scale, where 1 denotes the most strongly negative emotional expression and 7 denotes the most strongly positive emotional expression. The child’s emotional expressions were rated during their response to semi-structured interview probes by child psychiatrists. See text for questions. The n’s varied slightly in terms of the availability of useable data for analyses of discrete emotions. n’s ranged from 93 for the question regarding a child’s general mood to 139 for the query regarding the child’s worst, or saddest, experience.
p < 0.05.
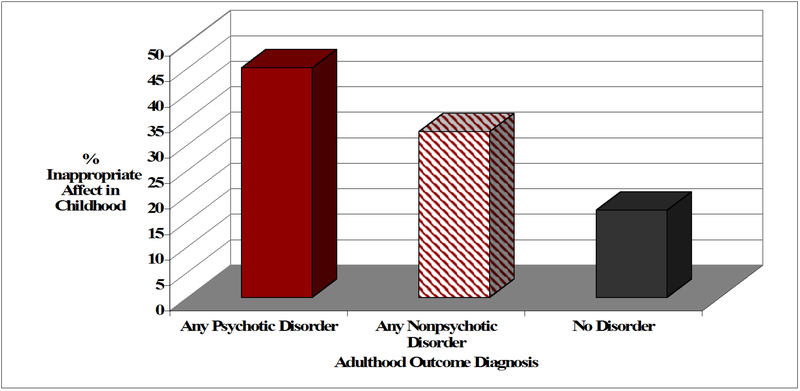
We observed a significant association between a display of inappropriate affect during mid-childhood and adulthood psychiatric outcome. As indicatedin Fig. 2, children having a later psychiatric outcome were significantly more likely to exhibit inappropriate affect during the interview, Χ2 (2) = 8.20, p = 0.02, and Cramer’s V = 0.20. Children showing flat affect during the mid-childhood MHAF were not more likely to develop a psychiatric disorder in adulthood, Χ2 (2) = 0.01, p = 0.99. The association between ratings of childhood emotional withdrawal and adulthood psychiatric outcome did not reach significance, Χ2 (2) = 3.92, n.s. Children manifesting moderate anxiety during the interviews appeared more likely to receive an adulthood psychiatric diagnosis than children manifesting either no,or mild, anxiety during the interviews, Χ2 (2) = 5.38, p = 0.07. The difference was not statistically significant.
Fig. 2.
The percentage of participants who displayed inappropriate affect during the MHAF when interviewed during mid-childhood who later had a psychiatric outcome as determined by a structured clinical interview in adulthood.
4. DISCUSSION
4.1. Advantages and significance of present study
This is the first study to investigate the emotional expressivity of at-risk individuals from childhood through the period of greatest risk for schizophrenia that also includes a non-psychotic psychiatric comparison group. One of the advantages of this study is the focus on a fine-grained analysis of discrete emotions as well as inclusion of molar ratings of emotional engagement. Our ratings of affective and social behaviors in the context of a social encounter provide further indications that individuals at genetic risk for schizophrenia display social and affective deficits early in their development.
We have identified differences in a child’s emotional expressivity during a semi-structured interview that are associated with their genetic risk for psychiatric disorder. Specifically, the HRSz group showed more negative affect and less positive affect in response to questions about their worst experience and their self-concept, respectively. This suggests that HRSz children may genuinely experience more negative affect than children of healthy controls or children of parents with affective disorders.
Studies [50–53] have indicated that social anhedonia is a significant risk factor for schizophrenia-spectrum disorders. The finding that HRSz individuals display less positive affect in response to questions about their self-concept may be relevant to the developmental ontogeny of social anhedonia in schizotypal individuals. The HRSz offspring group showed significantly less positive affect when questioned about their self-concept, i.e., how much did they like themselves or what would they change about themselves. If individuals at risk for schizophrenia experience an altered hedonic response to their own self-concept, this may negatively impact the development of hedonic associations related to social stimuli. More research is needed to clarify the potential role of a reduced hedonic representation of one’s self-concept in the development of social anhedonia.
These findings of impaired affective functioning in HRSz children are consistent with earlier reports based upon the genetic high-risk method [7] as well as the archival-observational method [5,6] indicating greater negative emotion being associated with preschizophrenic children. In contrast to Walker and colleagues [5,6], we did not find that adulthood schizophrenia outcome could be predicted on the basis of facial expressions of comparatively less positive affect, or greater negative affect, during childhood. While we observed significant differences in the frequency of affective displays in response to affect-eliciting questions, these differences did not predict later psychiatric outcomes. We attribute the differences in the findings in part to a different methodology (i.e., the archival-observational method using forced-choice versus prospective longitudinal method using a coding system that provides for assessment of several emotions simultaneously).
More of the high-risk children displayed inappropriate affect than the normal control children. It is noteworthy that in the present study we used a measure of inappropriate affect that combined both positive and negative affect. In future investigations, it would be useful to differentiate the types of inappropriate affect displayed by the child. It is possible that the HRSz and the HRAff groups exhibited inappropriate affect in different ways and for different reasons. For example, one might speculate that the HRAff children may display inappropriate positive affect as a learned response in an attempt to comfort their parents [54]. In contrast, HRSz children may display inappropriate negative affect as a manifestation of neural dysregulation [55].
One of the advantages of the prospective high-risk design is that it permits the study of behavioral antecedents of adulthood psychiatric outcomes. Our observation of an association between inappropriate affect during childhood and later adulthood diagnostic outcome implies a role for inappropriate affect as a psychopathology risk-related factor. Individuals who developed a psychiatric disorder in adulthood, whether psychotic or nonpsychotic, showed inappropriate affect more frequently than children who did not develop psychiatric illness. This suggests that inappropriate affect in the offspring of psychiatrically ill parents may be a nonspecific indication of risk for the development of adulthood psychopathology.
Furthermore, we observed that HRSz children were more likely to exhibit emotional withdrawal than the children in the other two groups. This suggests that emotional withdrawal may be specific indicator of risk for the later development of schizophrenia. The New York High Risk Project is the first study in which HRSz children have been independently observed to display significantly more emotional withdrawal compared to other groups of at-risk children. Our finding of increased emotional withdrawal being associated with genetic risk for schizophrenia shows considerable consistency with the results of prior investigations of emotional problems as antecedents to schizophrenia [16,31,56,57].
Our findings are consistent with earlier indications that individuals at genetic risk for schizophrenia manifest observable signs of schizotypy, such as emotional withdrawal [58,59] and inappropriate affect [58] at relatively early ages. Our findings extend the literature by suggesting that some of these signs may be specifically related to a schizophrenia diathesis. Despite differences in methodology, the NYHRP data are consistent with findings based upon retrospective parental reports [33] which indicate that emotional withdrawal in mid-childhood is associated with heightened risk for a schizophrenia-related outcomes in adulthood. Our findings regarding emotional withdrawal are also in line with data from prodromal studies [35,36] in which individuals with non-affective psychosis typically display social withdrawal in the years preceding their first episode of psychosis.
4.2. Limitations, strengths, and future directions
One limitation of the present study is that it was unable to explore gender differences within the at-risk groups. The relationship between gender and genetic risk in terms of emotional expressivity deserves further study, given the related findings of Walker and colleagues [5,6] as well as observations that children display gender-linked roles early in development, with gender differences in socio-emotional development appearing in childhood [60]. Nonwhite children were not included in the original samples in the NYHRP, which started in the early 1970s. Conclusions are thus limited to Caucasian populations.
Sample sizes varied according to the question. Due to the quality of the recordings as well as some variations in the semi-structured interviews, not all children were asked all the same questions. This investigation is a secondary analysis of a well-known data set. As expected, when data are used to explore questions that were not salient at the time of the study design, we could not expect that all the relevant data would be available.
We did not measure parent-child attachment, so it is possible that increased negative affect and emotional withdrawal could be related to poorer attachment, rather than simply a genetic endophenotype. We did not assess the level of trauma experienced by the children. Prior work suggests that traumatic experiences during childhood may contribute to the development and/ or maintenance of psychosis [34]. Future research in this area would benefit from measuring level and severity of childhood traumatic events as well as quality of parent-child attachment. Finally, we cannot rule out environmental factor effects that may have contributed to group differences in affective expressivity that we observed. Given the presence of serious psychopathology in the families of the atrisk offspring, it would not have been unreasonable to see increased displays of negative affect overall. However, that was not the case. Rather, we observed that a disproportionate number of children in both high-risk groups exhibited inappropriate affect, relative to the children in the NC group.
5. CONCLUSION
The present study demonstrated that childhood emotional expressivity is associated with later psychiatric outcomes. These data are significant for two reasons: first, they provide convergent evidence for affective deficits being a key feature of schizophrenia-spectrum disorders; and second, they suggest that the genetic diathesis associated with schizophrenia may become manifest early in the life span, years prior to the onset of an overt psychiatric disorder.
ACKNOWLEDGMENTS
Funding support for this project came from the National Institute of Mental Health at the National Institutes of Health (grant MH 19560–01-3 to L.E.K.); a Wisconsin Alumni Research Association Foundation grant and mid-career Vilas Fellowship to D.C.G. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript. The authors thank the NYHRP participants in the study. The authors also gratefully acknowledge: Ulla Adamo, M.A., and Simone Roberts for their work on the project; Alex Bellantuono for his technical assistance with videotape editing and preparation; Carly Thanhouser, B.A., and Elysia Sotiriou, B.A. for their assistance with data coding. Part of these data were presented at the 2013 meetings of the Society for Research in Psychopathology and the International Congress on Schizophrenia Research. We are also grateful to two anonymous reviewers whose incisive comments helped refine the presentation of our work.
ABBREVIATIONS
- NYHRP
New York High-Risk Project
- HRSz
high risk for Schizophrenia
- HRAff
high risk for Affective disorders
- NC
normal comparison
- MHAF
Mental Health Assessment Form
- SADS-L
Schedule for Affective Disorders and Schizophrenia, Lifetime version
- PDE
Personality Disorders Examination
- ANOVA
Analysis of Variance
- ICC
Intra-class correlation
Footnotes
CONFLICTS OF INTEREST
The authors declare that there is no conflict of interest regarding the publication of these findings or this paper.
REFERENCES
- 1.Rutter M Psychopathology and development: I. Childhood antecedents of adult psychiatric disorder. Aust N Z J Psychiatry. 1984; 18(3): 225–234. [DOI] [PubMed] [Google Scholar]
- 2.Weinberger DR. Implications of normal brain development for the pathogenesis of schizophrenia. Arch Gen Psychiatry. 1987; 44: 660–669. [DOI] [PubMed] [Google Scholar]
- 3.Kraepelin E Dementia Praecox and Paraphrenia. Chicago: Chicago Medical Book Company; 1896. [Google Scholar]
- 4.Tandon R, Nasrallah HA, Keshavan MS. Schizophrenia, “just the facts” 4. Clinical features and conceptualization. Schizophr Res. 2009; 110: 1–23. [DOI] [PubMed] [Google Scholar]
- 5.Walker EF, Grimes KE, Davis DM, Smith AJ. Childhood precursors of schizophrenia: Facial expressions of emotion. Am J Psychiatry. 1993; 150(11): 1654–1660. [DOI] [PubMed] [Google Scholar]
- 6.Walker EF, Lewine RJ. Prediction of adult-onset schizophrenia from childhood home movies of the patients. Am J Psychiatry. 1990; 147: 1052–1056. [DOI] [PubMed] [Google Scholar]
- 7.Schiffman J, Walker EF, Ekstrom M, Schulsinger F, Sorensen H, Mednick S. Childhhood videotaped social and neuromtor precursors of schizophrenia: A prospective investigation. Am J Psychiatry. 2004; 161: 2021–2027. [DOI] [PubMed] [Google Scholar]
- 8.Dworkin RH, Bernstein G, Kaplansky LM, Lipsitz JD, Rinaldi A, Slater SL, Cornblatt BA, Erlenmeyer-Kimling L. Social competence and positive and negative symptoms: A longitudinal study of children and adolescents at risk for schizophrenia and affective disorder. Am J Psychiatry. 1991; 148: 1182–1188. [DOI] [PubMed] [Google Scholar]
- 9.Cannon-Spoor HE, Potkin SG, Wyatt RJ. Measurement of premorbid adjustment in chronic schizophrenia. Schizophr Bull. 1982; 8(3): 470–484. [DOI] [PubMed] [Google Scholar]
- 10.Dworkin RH, Cornblatt BA, Friedmann R, Kaplansky LM, Lewis JA, Rinaldi A, Shilliday C, Erlenmeyer-Kimling L. Childhood precursors of affective vs. social deficits in adolescents at risk for schizophrenia. Schizophr Bull. 1993; 19(3): 563–577. [DOI] [PubMed] [Google Scholar]
- 11.Kring AM. Emotion in schizophrenia: Old mystery, new understanding. Curr Dir Psychol Sci. 1999; 8(5): 160–163. [Google Scholar]
- 12.Goodman SH. Emory University Project on children of disturbed parents. Schizophr Bull. 1987; 13(3): 411–423. [DOI] [PubMed] [Google Scholar]
- 13.Goodman SH,Gotlib IH. Risk for psychopathology in the children of depressed mothers: A developmental model for understanding mechanisms of transmission. Psychol Rev. 1999; 106(3): 458–490. [DOI] [PubMed] [Google Scholar]
- 14.Jacobs RH, Talati A, Wickramaratne P, Warner V. The influence of paternal and maternal major depressive disorder on offspring psychiatric disorder. J Child Fam Stud. 2015; 24: 2345–2351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Gyllenberg D, Sourander A, Niemelä S, Helenius H, Sillanmäki L, Piha J, Kumpulainen K, Tamminen T, Moilanen I, Almqvist F. Childhood predictors of later psychiatric hospital treatment: findings from the Finnish 1981 birth cohort study. Eur Child Adolesc Psychiatry. 2010; 19: 823–833. [DOI] [PubMed] [Google Scholar]
- 16.Tien AY, Eaton WW. Psychopathologic precursors and sociodemographic risk factors for the schizophrenic syndrome. Arch Gen Psychiatry. 1992; 49: 37–46. [DOI] [PubMed] [Google Scholar]
- 17.Owens DG, Miller P, Lawrie SM, Johnstone EC. Pathogenesis of schizophrenia: a psychopathological perspective. Br J Psychiatry. 2005; 186: 386–393. [DOI] [PubMed] [Google Scholar]
- 18.Anderson CA, Hammen CL. Psychosocial outcomes of children of unipolar depressed, bipolar, medically ill, and normal women: A longitudinal study. J Consult Clin Psychol. 1993; 61(3): 448–454. [DOI] [PubMed] [Google Scholar]
- 19.Weissman MM, Warner V, Wickramaratne P, Moreau D, Olfson M. Offspring of depressed parents 10 years later. Arch Gen Psychiatry. 1997; 54(10): 932–940. [DOI] [PubMed] [Google Scholar]
- 20.Zahn-Waxler C, Van Hulle C. Empathy, guilt, and depression: when caring for others becomes costly to children Oakley B, Knafo A, Madhavan G, Wilson DS, editors. Pathological Altruism. New York: Oxford University Press; 2012. p. 310–346. [Google Scholar]
- 21.Cicchetti D, Ackerman BP, Izard CE. Emotions and emotion regulation in developmental psychopathology. Dev Psychopathol. 1995; 7: 1–10. [Google Scholar]
- 22.Fish B Neurobiologic antecedents of schizophrenia in children: Evidence for an inhereited, congenital neurointegrative defect. Arch Gen Psychiatry. 1977; 34: 1297–1313. [DOI] [PubMed] [Google Scholar]
- 23.Andreasen NC. A unitary model of schizophrenia. Bleuler’s “fragmented phrene” as schizencephaly. Arch Gen Psychiatry. 1999; 56: 781–787. [DOI] [PubMed] [Google Scholar]
- 24.Carlson GA, Fish B. Longitudinal course of schizophrenia spectrum symptoms in offspring of psychiatrically hospitalized mothers. J Child Adolesc Psychopharmacol. 2005; 15(3): 362–383. [DOI] [PubMed] [Google Scholar]
- 25.Manschreck TC, Maher BA, Waller NC, Ames D, Latham C. Deficient motor synchrony in schizophrenic disorders: Clinical correlates. Biol Psychiatry. 1985; 20: 990–1002. [DOI] [PubMed] [Google Scholar]
- 26.Manschreck TC, Maher BA, Ruckos ME, Vereen DR. Disturbed voluntary motor activity in schizophrenic disorder. Psychol Med. 1982; 12: 73–84. [DOI] [PubMed] [Google Scholar]
- 27.Dworkin RH, Clark SC, Amador XF, Gorman JM. Does affective blunting in schizophrenia reflect affective deficit or neuromotor dysfunction? Schizophr Res. 1996; 20(3): 301–306. [DOI] [PubMed] [Google Scholar]
- 28.Lee JS, Chun JW, Yoon SY, Park HJ, Kim JJ. Involvement of the mirror neuron system in blunted affect in schizophrenia. Schizophr Res. 2014; 152: 268–274. [DOI] [PubMed] [Google Scholar]
- 29.Hans SL, Marcus J, Henson L, Auerbach JG, Mirsky AF. Interpersonal behavior of children at risk for schizophrenia. Psychiatry. 1992; 55: 314–335. [DOI] [PubMed] [Google Scholar]
- 30.Sohlberg S, Yaniv S. Social adjustment and cognitive performance of high-risk children. Schizophr Bull. 1985; 11: 61–65. [DOI] [PubMed] [Google Scholar]
- 31.Done DJ, Crow TJ, Johnstone EC, Sacker A. Childhood antecedents of schizophrenia and affective illness: social adjustment at ages 7 and 11. BMJ. 1994; 309: 695–699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Niemi LT, Suvisaari JM, Haukka JK, Lonnqvist JK. Childhood predictors of future psychiatric morbidity in offspring of mothers with psychotic disorder: Results from the Helsinki High-Risk Study. Br J Psychiatry. 2005; 186: 108–114. [DOI] [PubMed] [Google Scholar]
- 33.Baum KM, Walker EF. Childhood behavioral precursors of adult symptom dimensions in schizophrenia. Schizophr Res. 1995; 16: 111–120. [DOI] [PubMed] [Google Scholar]
- 34.Matheson SL, Vijayan H, Dickson H, Shepherd AM, Carr VJ, Laurens KR. Systematic metaanalysis of childhood social withdrawal in schizophrenia, and comparison with data from at-risk children aged 9–14 years. J Psychiatr Res. 2013; 47(8): 1061–1068. [DOI] [PubMed] [Google Scholar]
- 35.Yung AR, McGorry PD. The initial prodrome in psychosis: Descriptive and qualitative aspects. Aust N Z J Psychiatry. 1996; 30: 587–599. [DOI] [PubMed] [Google Scholar]
- 36.Corcoran C, Gerson R, Sills-Shahar R, Nickou C, McGlashan T, Malaspina D, Davidson L. Trajectory to a first episode of psychosis: a qualitative research study with families. Early Interv Psychiatry. 2007; 1: 308–315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Erlenmeyer-Kimling L, Adamo UH, Rock D, Roberts SA, Bassett AS, Squires-Wheeler E, Cornblatt BA, Endicott J, Pape S, Gottesman II. The New York High-Risk Project: Prevalence and comorbidity of Axis I disorders in offspring of schizophrenic parents at 25-Year follow-up. Arch Gen Psychiatry. 1997; 54(12): 1096–1102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Erlenmeyer-Kimling L, Rock D, Roberts SA, Janal M, Kestenbaum C, Cornblatt B, Adamo UH, Gottesman II. Attention, memory, and motor skills as childhood predictors of schizophrenia-related psychoses: The New York High-Risk Project. Am J Psychiatry. 2000; 157(9): 1416–1422. [DOI] [PubMed] [Google Scholar]
- 39.Erlenmeyer-Kimling L, Squires-Wheeler E, Adamo UH, Bassett AS, Cornblatt BA, Kestenbaum CJ, Rock D, Roberts SA, Gottesman II. The New York High-Risk Project: Psychoses and Cluster A personality disorders in offspring of schizophrenic parents at 23 years of follow-up. Arch Gen Psychiatry. 1995; 52(10): 857–865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Kestenbaum CJ, Bird HR. A reliability study of the Mental HealthAssessment Form for schoolage chidlren. J Am Acad Child Psychiatry. 1978; 17(2): 338–347. [DOI] [PubMed] [Google Scholar]
- 41.Video recording quality affected the ability to have a close-up view of the child’s face in order to rate expressions at the micro-level of analysis for that particular question. We also needed to be able to hear the child’s answer. If either of these criteria was not met, the particular question was not rated.
- 42.Cohen JA. A coefficient of agreement for nominal scales. Educ Psychol Meas. 1960; 20: 37–46. [Google Scholar]
- 43.In order to insure a sufficient frequency of responses, individual types of emotions were grouped together within the three larger categories.
- 44.Andreasen NC. Negative symptoms in schizophrenia: Definitions and reliability. Arch Gen Psychiatry. 1982; 39: 784–748. [DOI] [PubMed] [Google Scholar]
- 45.Endicott J, Spitzer RL. A diagnostic interview: The Schedule for Affective Disorders and Schizophrenia. Arch Gen Psychiatry. 1978; 35: 837–844. [DOI] [PubMed] [Google Scholar]
- 46.Spitzer RL, Endicott J, Robins E. Research Diagnostic Criteria: Rationale and reliability. Arch Gen Psychiatry. 1978; 35: 773–782. [DOI] [PubMed] [Google Scholar]
- 47.Loranger AW, Susman VL, Oldham JM, Russakoff LM. The personality disorder examination: a preliminary report. J Personal Disord. 1987; 1: 1–13. [Google Scholar]
- 48.Squires-Wheeler E, Skodol AE, Hilldoff Adamo U, Bassett AS, Gewirtz GR, Honer WG, Cornblatt BA, Roberts SA, Erlenmeyer-Kimling L. Personality features and disorder in the subjects in the New York High-Risk Project. J Psychiatr Res. 1993; 27(4): 379–393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Initially we compared males and females within each offspring group in order to detect gender differences. Within the HRSz and NC groups, there were no significant gender differences observed in terms of the display of any of the discrete emotions. Within the HRAff group, the only gender difference observed was in response to the question regarding self-concept; the female children (n = 3) were significantly less positive than their male counterparts (n = 15), t (15) = 2.24, p < 0.05. Because there were so few significant gender differences within each offspring group, and because in some cases, the N’s in the cells were less than 10, we chose to analyze males and females together in order to maximize statistical power. We report the analyses for affective behaviors in the text, in which we used Fisher’s exact tests in cases where any of the cells had an expected count less than 5.
- 50.Kwapil TR. Social anhedonia as a predictor of the development of schizophrenia-spectrum disorders. J Abnorm Psychol. 1998; 107: 558–565. [DOI] [PubMed] [Google Scholar]
- 51.Gooding DC, Tallent KA, Matts CW. Clinical status of at-risk individuals 5 years later: Further validation of the psychometric high-risk strategy. J Abnorm Psychol. 2005; 114(1): 170–175. [DOI] [PubMed] [Google Scholar]
- 52.Gooding DC, Tallent KA, Matts CW. Rates of avoidant, schizotypal, schizoid and paranoid personality disorders in psychometric highrisk groups at 5-year follow-up. Schizophr Res. 2007; 94(1–3): 373–374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Miettunen J, Veijola J, Isohanni M, Paunio T, Freimer N, Jääskeläinen E, Taanila A, Ekelund J, Järvelin MR, Peltonen L, Joukamaa M, Lichtermann D. Identifying schizophrenia and other psychoses with psychological scales in the general population. J Nerv Ment Dis. 2011; 199: 230–238. [DOI] [PubMed] [Google Scholar]
- 54.Zahn-Waxler C, Van Hulle CA. Empathy, guilt, and depression. When caring for others becomes costly to children In: Oakley B, Knafo A, Madhavan G, Wilson DS, editors. Pathological altruism. New York: Oxford University Press; 2011. p. 243–259. [Google Scholar]
- 55.Meehl PE. Schizotaxia, schizotypy, schizophrenia. Am Psychol. 1962; 17(12): 827–838. [Google Scholar]
- 56.Tarbox SI, Pogue-Geile MF. Development of social functioning in preschizophrenia children and adolescents: a systematic review. Psychol Bull. 2008; 134(4): 561–583. [DOI] [PubMed] [Google Scholar]
- 57.Niemi LT, Suvisaari JM, Tuulio-Henriksson A, Lonnqvist JK. Childhood developmental abnormalities in schizophrenia: evidence from high-risk studies. Schizophr Res. 2003; 60: 239–258. [DOI] [PubMed] [Google Scholar]
- 58.Parnas J, Schulsinger F, Schulsinger H, Mednick SA, Teasdale TW. Behavioral precursors of schizophrenia spectrum. Arch Gen Psychiatry. 1982; 39: 658–664. [DOI] [PubMed] [Google Scholar]
- 59.Carlson GA, Fish B. Longitudinal course of schizophrenia spectrum symptoms in offspring of psychiatrically hospitalized mothers. Journal of Child and Adolescent Psychopharmacology. 2005; 15(3): 362–383. [DOI] [PubMed] [Google Scholar]
- 60.Zahn-Waxler C, Shirtlcliff EA, Marceau K. Disorders of childhood and adolescence: Gender and psychopathology. Annu Rev Clin Psychol. 2008; 4: 275–303. [DOI] [PubMed] [Google Scholar]