Summary
There is evidence that Janus kinase (JAK)–signal transducer and activator of transcription (STAT) signaling plays a role in the pathogenesis of sarcoidosis. We treated a patient with cutaneous sarcoidosis with the JAK inhibitor tofacitinib; the patient had not previously had a response to medications and had not received systemic glucocorticoids. This treatment resulted in clinical and histologic remission of her skin disease. Sequencing of RNA and immunohistochemical examination of skin-lesion samples obtained from the patient before and during therapy and immunohistochemical testing of lesion samples obtained from other patients with cutaneous sarcoidosis support a role for JAK-STAT signaling in cutaneous sarcoidosis. (Funded by the Ranjini and Ajay Poddar Resource Fund for Dermatologic Diseases Research and others.)
Sarcoidosis is an inflammatory disease that is associated with the formation of noncaseating granulomas in one or multiple organ systems. Skin involvement is seen in approximately 25% of patients with sarcoidosis.1 Systemic glucocorticoids are the initial treatment for sarcoidosis with systemic involvement and may be used for the treatment of cutaneous sarcoidosis.2,3 Granulomas in patients with sarcoidosis are composed primarily of macrophages and T cells.4 The activation of macrophages in granulomas is considered to be dependent on helper T cells and mediated in part by interferon-γ.5,6 Interferon-γ activates the Janus kinase (JAK)–signal transducer and activator of transcription (STAT) signaling pathway, resulting in the up-regulation of STAT1 transcriptional targets. Several studies have shown that JAK-STAT pathway activation signatures, especially STAT1-dependent transcripts, are characteristic of the transcriptome in both peripheral-blood mononuclear cells and other tissues in patients with sarcoidosis.7–10
We treated a patient who had refractory cutaneous sarcoidosis with the oral JAK inhibitor tofacitinib, which resulted in clinical and histologic remission of skin lesions. We also performed molecular characterization of the response using global gene-expression profiling of skin-lesion samples obtained from this patient, and we analyzed a series of biopsy samples obtained from other patients with cutaneous sarcoidosis.
Methods
Clinical Data and Specimen Collection
The patient provided written informed consent that indicated that she understood that the skin biopsies were being performed for research purposes. Comparative deidentified skin-lesion samples from other patients with cutaneous sarcoidosis were obtained from archival material. The two components of the Cutaneous Sarcoidosis Activity and Morphology Instrument (CSAMI)11 were used to gauge the severity of cutaneous sarcoidosis; the disease-activity score ranges from 0 to 165 and the tissue-damage score ranges from 0 to 22, with higher scores indicating greater disease activity and tissue damage, respectively. For statistical comparisons we used unpaired Student’s t-tests in Prism 7 software (GraphPad).
Histologic and Immunohistochemical Testing
Skin-lesion samples were obtained from the index patient just before treatment with tofacitinib and again 10 months later while the patient was receiving treatment. Samples from both periods were processed for histopathological evaluation with the use of hematoxylin and eosin staining and with immunohistochemical testing to stain macrophages (with CD68) and to detect activated JAK-STAT signaling (with phosphorylated STAT1 [pSTAT1] and phosphorylated STAT3 [pSTAT3]). Details are provided in Supplementary Appendix 1, available with the full text of this article at NEJM.org.
RNA Extraction and Sequencing
Portions of flash-frozen skin-lesion samples that were obtained before treatment and again during treatment with tofacitinib underwent RNA sequencing. Technical details of the library preparation, sequencing, and data analysis, including gene-set enrichment analysis, are described in Supplementary Appendix 1.
Histologic Case Series for Comparison with Index Patient
We assembled a set of deidentified, archival skin-lesion samples that had been obtained from 21 patients with cutaneous sarcoidosis and 10 patients with xanthelasma as well as skin samples from 5 healthy controls (Table S1 in Supplementary Appendix 1). Immunohistochemical testing with the use of pSTAT1 (Tyr701 58D6, Cell Signaling Technology) and pSTAT3 (Tyr705 D3A7, Cell Signaling Technology) to detect JAK-STAT pathway activation was performed and quantified with the use of Fiji ImageJ software (see Supplementary Appendix 1). The resulting immunohistochemical test score for each sample represents the percentage of the tissue area that was stained positively for the marker.
Case Report
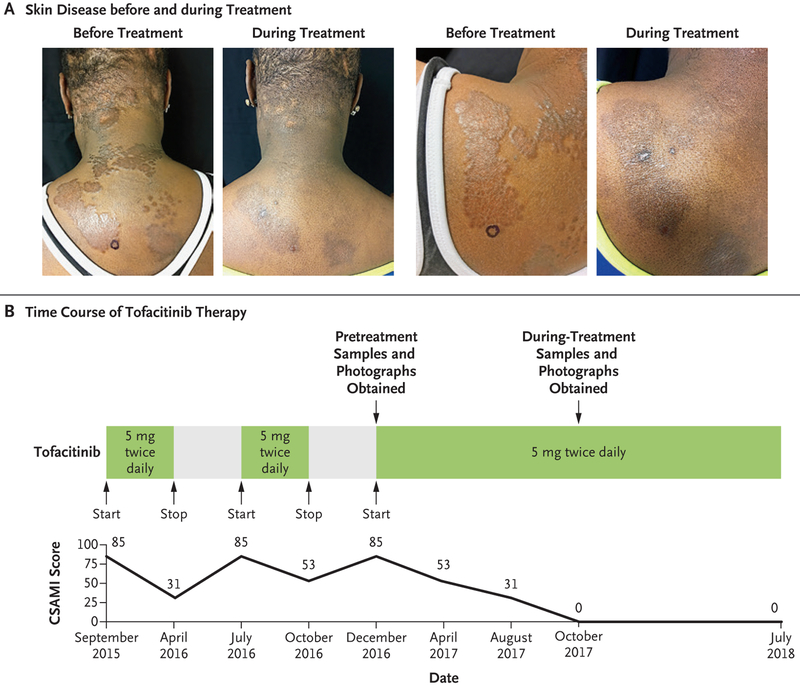
A 48-year-old woman who had an 8-year history of cutaneous and pulmonary sarcoidosis was evaluated for the management of treatment-resistant skin lesions. Computed tomography (CT) of the chest that was performed 8 years before presentation revealed mediastinal and hilar adenopathy with peribronchovascular and perilymphatic nodules in both lungs, which were most prominent in the upper lobes; transbronchial lung-biopsy samples showed noncaseating granulomas. The patient had no pulmonary symptoms, and results on spirometry were normal; she was not treated for her pulmonary disease. The results of baseline and follow-up pulmonary-function tests are shown in Table S3 in Supplementary Appendix 1. The ophthalmologic examination was unremarkable, and there was no palpable adenopathy. Skin examination showed numerous pink–brown, indurated papules and plaques, many of which were annular and measured up to 20 cm in the greatest dimension on her scalp, neck, torso, arms, and legs (Fig. 1A, and Fig. S1A in Supplementary Appendix 1). Alopecia was evident in areas of scalp involvement. Skin-lesion samples from the neck and leg showed noncaseating granulomas — findings that were consistent with sarcoidosis.
Figure 1. Clinical Remission of Sarcoidosis with Tofacitinib Therapy.
Panel A shows photographs of skin disease before treatment and during treatment. The before-treatment sample was obtained from the circled area. Skin-lesion samples that were obtained during treatment were obtained after 10 months of therapy. Panel B shows the time course of tofacitinib therapy. The clinical response with therapy and the clinical worsening when the patient was not receiving therapy are indicated by the change in the Cutaneous Sarcoidosis Activity and Morphology Instrument (CSAMI)11 score for disease activity. The CSAMI was used to gauge the severity of cutaneous sarcoidosis; scores range from 0 to 165 for disease activity, with higher scores indicating greater disease activity. Although the disease activity score extends to 165, the patient’s maximum disease activity score never exceeded 85; hence the y axis for this graph extends only to 100.
Over a period of 8 years, her cutaneous disease had not responded to topical glucocorticoids, minocycline at a dose of 100 mg twice daily, hydroxychloroquine at a dose of 200 mg twice daily, methotrexate at a dose of 15 to 20 mg weekly, adalimumab at a dose of 40 mg every other week, tacrolimus at a dose of 0.085 mg per kilogram of body weight per day, and apremilast at a dose of 30 mg twice daily, each given for various durations. In 2015, her CSAMI score was 85 for disease activity and 13 for tissue damage; both scores were in the middle range of possible scores. Owing to the patient’s hypertension, hyperlipidemia, and type 2 diabetes, in addition to the chronic nature of her cutaneous disease and likely need for long-term treatment, systemic glucocorticoids were not administered.
With the patient’s agreement, off-label treatment with tofacitinib, an inhibitor of JAK1 and JAK3, was initiated at a dose of 5 mg twice daily. The treatment resulted in abatement of her skin lesions. Because of administrative insurance issues, treatment lapsed for 3 months, during which time the skin lesions enlarged in size. Treatment with tofacitinib was restarted, and again, the skin lesions abated. Subsequently, another lapse of treatment for 3 months led to increased skin disease. Skin-lesion samples were obtained, and tofacitinib therapy was restarted at that time. The results described below are from this third cycle of treatment.
Results
Clinical Course with Tofacitinib Treatment
The patient’s CSAMI disease activity score was 85 before the third cycle of tofacitinib treatment. A skin-lesion sample from the patient’s upper back (Fig. 1A) was obtained 1 week before tofacitinib therapy was restarted, and it showed noncaseating granulomas in the dermis (Fig. 2A). After 4 months of therapy, there was visible thinning of the cutaneous lesions, and the CSAMI disease activity score declined to 53. After 8 months, the CSAMI disease activity score was 31. At 10 months, we noted clinical remission of her cutaneous disease, and the CSAMI disease activity score was 0 (Fig. 1A and 1B, and Fig. S1A and S1B in Supplementary Appendix 1), with only scarring and postinflammatory pigmentary alteration remaining. The CSAMI tissue damage score, 13, was unchanged from before treatment. At the time of this report, the patient’s skin had remained clear for 9 months while she was receiving tofacitinib at a dose of 5 mg twice daily. She had not had adverse effects during 19 months of continuous tofacitinib treatment.
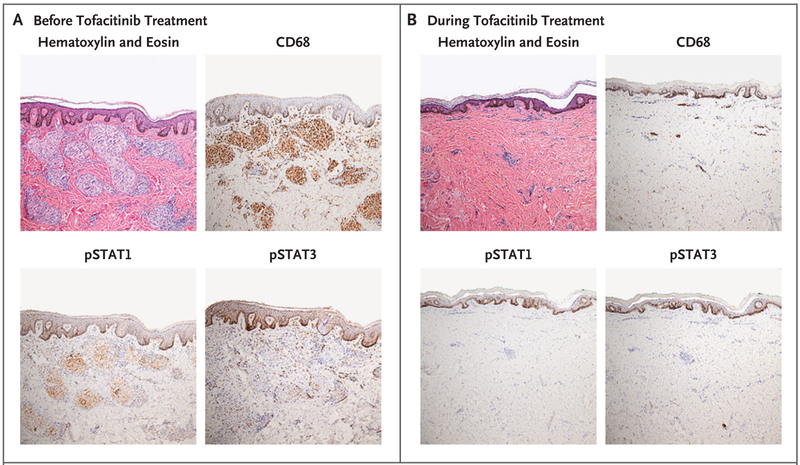
Figure 2. Histologic Remission of Sarcoidosis with Tofacitinib Therapy.
Shown are the results of immunohistochemical testing of skin-lesion samples obtained before and during treatment. Hematoxylin and eosin staining on the sample obtained before treatment showed well-organized, noncaseating granulomas and mild patchy lymphocytic inflammation in the dermis — findings that are typical of sarcoidosis. Staining was also performed with CD68, phosphorylated signal transducer and activator of transcription (STAT) 1 (pSTAT1), and phosphorylated STAT3 (pSTAT3).
Immunohistochemical Testing in the Patient
After 10 months of treatment, two skin-lesion samples were obtained from locations that had been previously involved with disease; these samples showed histologic resolution of granulomas. A few histiocytes and lymphocytes were still present, but granulomas were absent (Fig. 2A and 2B, and Fig. S2A and S2B in Supplementary Appendix 1). Immunohistochemical stains that had shown pSTAT1 and pSTAT3 in the pretreatment skin-lesion samples indicated a negligible presence of these markers in the skin-lesion samples that were obtained during treatment (immunohistochemical test score for pSTAT1, 18.67 vs. 0.32; immunohistochemical test score for pSTAT3, 3.49 vs. 0.73).
RNA Sequencing
RNA sequencing was performed on flash-frozen skin-lesion samples, both before and during treatment. These data were compared with publicly available RNA sequencing data from normal human skin. Principal component analysis of the samples is shown in Figure S3A in Supplementary Appendix 1. Gene-set enrichment analysis showed activation of interferon-γ and tumor necrosis factor (TNF) α signaling as well as interleukin-6–STAT3 and mammalian target of rapamycin complex 1 (mTORC1) signaling in the pretreatment skin-lesion samples; in contrast, in skin-lesion samples obtained during treatment and in normal skin samples, these pathways did not appear to be activated. While the patient was taking tofacitinib, there was down-regulation of the messenger RNA (mRNA) in the JAK-STAT–dependent pathways (interferon-γ and interleukin-6) as well as of mRNA in pathways that are not directly regulated by JAK-STAT (TNFα and mTORC1) (Figs. S3B through S3E and S4A through S4F in Supplementary Appendix 1 and Table S2 in Supplementary Appendix 2, available at NEJM.org). Other proinflammatory transcripts that have been implicated in sarcoidosis pathogenesis, including TLR2, TLR8, IL1B, IL12B, IL18, IL27, CXCL9, and CXCL10, were reduced during tofacitinib therapy (Fig. S4E and S4F in Supplementary Appendix 1).
Immunohistochemical Testing in Other Patients with Sarcoidosis
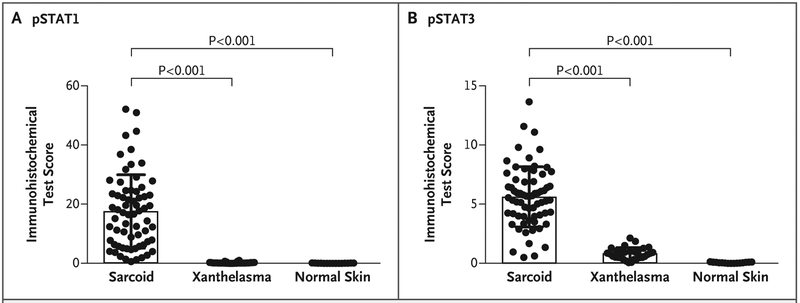
To further evaluate JAK-STAT activation in cutaneous sarcoidosis, we performed immunohistochemical testing for pSTAT1 and pSTAT3 on 21 archival skin-lesion samples (Fig. 3A and 3B, and Fig. S5A and S5B and Table S1 in Supplementary Appendix 1). All these skin-lesion samples were strongly positive for both pSTAT1 (mean [±SE] immunohistochemical test score, 17.58±1.58) and pSTAT3 (mean score, 5.62±0.21), with pSTAT1 staining tending to be strongest in the center of the granuloma and more prominent in macrophages than in T cells. Furthermore, pSTAT3 staining was strongest in the nuclei of lymphocytes within and between granulomas (Fig. S5A in Supplementary Appendix 1). Staining for pSTAT1 and pSTAT3 was significantly higher in samples obtained from patients with sarcoidosis than in control samples of normal skin. In control samples, the mean immunohistochemical test score was 0.06±0.01 for both pSTAT1 and pSTAT3 (P<0.001 for both comparisons with samples from patients with sarcoidosis). Staining for pSTAT1 and pSTAT3 was also significantly higher in samples obtained from patients with sarcoidosis than in samples obtained from patients with xanthelasma, another cutaneous granulomatous disorder. In patients with xanthelasma, the mean scores were 0.21±0.04 for pSTAT1 and 0.83±0.09 for pSTAT3 (P<0.001 for both comparisons with samples from patients with sarcoidosis).
Figure 3. Levels of pSTAT1 and pSTAT3 in Skin-Lesion Samples from Patients with Cutaneous Sarcoidosis, Xanthelasma, or Normal Skin.
The immunohistochemical test score represents the percentage of the tissue area that was stained positively for the marker (pSTAT1 in Panel A and pSTAT3 in Panel B). Each black dot represents the score for one 100× microscopic field from each skin-lesion sample (three fields quantified per sample). The bar graph shows the mean score, and I bars the standard deviation.
Effect of Tofacitinib on Pulmonary Disease
During this patient’s 8-year course of different immunomodulatory therapies for her cutaneous sarcoidosis, she underwent interval radiography of the chest that revealed progression of her pulmonary sarcoidosis. Before the initiation of tofacitinib therapy, she had upper lobe–predominant fibrotic lung disease in both lungs and scattered nodules. After 12 months of tofacitinib treatment, CT of the chest revealed persistence of her upper lobe–predominant fibrotic lung disease with peribronchovascular and perilymphatic nodules. At the time of this report, the patient had remained without pulmonary symptoms and has had largely unchanged results on spirometry. A mild impairment in diffusion that was present after 12 months of tofacitinib therapy improved after 18 months of therapy (Table S3 in Supplementary Appendix 1); however, the clinical meaningfulness of this finding is not clear. The upper lobe–predominant fibrotic lung disease that was present in this patient before the initiation of tofacitinib therapy persisted during therapy.
Discussion
There are limited therapies for chronic, severe cutaneous sarcoidosis.1,2 Glucocorticoids, which have been approved by the Food and Drug Administration for the treatment of pulmonary sarcoidosis, are often used in patients with systemic sarcoidosis and may be used to treat cutaneous disease.2,3,12 The response of a patient with cutaneous sarcoidosis to prednisone is often incomplete, and even when a good response occurs, there are adverse effects associated with long-term use. Evidence for the effectiveness of other therapies for cutaneous sarcoidosis, including methotrexate, TNFα blockers, antimalarial drugs, tetracycline antibiotic agents, thalidomide, and other immunomodulatory agents, is limited and has been derived from small, uncontrolled case series.2,13 Our patient’s cutaneous disease did not respond to most of these medications. Treatment with tofacitinib (an inhibitor of JAK1 and JAK3), however, resulted in clinical and histologic remission of cutaneous disease. The withdrawal of treatment resulted in relapse of disease, cutaneous lesions abated after treatment was restarted, and stable clinical remission of her skin disease was observed with ongoing treatment. There was no definite effect on her pulmonary sarcoid.
Several studies have shown transcriptional JAK-STAT activation, particularly for STAT1, in sarcoidosis lesions.7–10 This was detected in our patient, in whom up-regulation of STAT1 transcripts was seen in the pretreatment skin-lesion sample, and this finding is consistent with the role that is attributed to T-cell–derived interferon-γ in driving macrophage activation in granulomas. The direct inhibition of the interleukin-6–STAT3 pathway, which has also been implicated in sarcoidosis pathogenesis,14,15 may also have a role in the response to tofacitinib therapy, on the basis of the RNA sequencing data. We also found that the transcriptional activity of non–JAK-STAT–dependent pathways that have been implicated in sarcoidosis pathogenesis, including mTORC1 and TNFα, was down-regulated by tofacitinib.16,17 Furthermore, levels of additional proinflammatory gene products, including toll-like receptors, interleukin-1β, interleukin-12, interleukin-18, interleukin-27, CXCL9, and CXCL10, which have previously been shown to be activated in patients with sarcoidosis, were reduced with tofacitinib treatment.16,18 The improvement in both JAK-STAT–dependent and JAK-STAT–independent pathways and transcripts suggests that the dysregulation of JAK-STAT–dependent cytokines (e.g., interferon-γ) is pathogenically involved in cutaneous sarcoidosis and, probably, in sarcoidosis in general.
The response of cutaneous sarcoidosis to tofacitinib therapy that was observed in our patient needs to be replicated in other patients. Additional studies will be necessary to understand the efficacy and safety of tofacitinib treatment in patients with sarcoidosis and may provide more detailed information about the molecular pathogenesis of this disease.
Supplementary Material
JOURNAL ARCHIVE AT NEJM.ORG.
Every article published by the Journal is now available at NEJM.org, beginning with the first article published in January 1812. The entire archive is fully searchable, and browsing of titles and tables of contents is easy and available to all. Individual subscribers are entitled to free 24-hour access to 50 archive articles per year. Access to content in the archive is available on a per-article basis and is also being provided through many institutional subscriptions.
Acknowledgments
Supported by a grant (to Dr. King) from the Ranjini and Ajay Poddar Resource Fund for Dermatologic Diseases Research, a training grant (T32AR007016, to Dr. Damsky) from the National Institutes of Health (NIH), a Research Fellowship and Career Development Award (to Dr. Damsky) from the Dermatology Foundation, and a Medical Science Training Program grant (T32GM007205, to Ms. Thakral) from the NIH.
We thank V. Klump and M. Napoletano and the staff of the Yale dermatopathology laboratory; M. Zhong and E. Cheng for RNA sequencing performed at the Yale Stem Cell Center Genomics Core Facility, which was supported by the Connecticut Regenerative Medicine Research Fund and the Li Ka Shing Foundation; M. Gulati and N. Kaminski of the Department of Internal Medicine, Section of Pulmonary, Critical Care, and Sleep Medicine at the Yale School of Medicine; and the staff of the Yale Center for Research Computing for guidance and use of the research computing infrastructure.
Footnotes
Disclosure forms provided by the authors are available with the full text of this article at NEJM.org.
References
- 1.Noe MH, Rosenbach M. Cutaneous sarcoidosis. Curr Opin Pulm Med 2017; 23: 482–6. [DOI] [PubMed] [Google Scholar]
- 2.Al-Kofahi K, Korsten P, Ascoli C, et al. Management of extrapulmonary sarcoidosis: challenges and solutions. Ther Clin Risk Manag 2016; 12: 1623–34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Spagnolo P, Rossi G, Trisolini R, Sverzellati N, Baughman RP, Wells AU. Pulmonary sarcoidosis. Lancet Respir Med 2018; 6: 389–402. [DOI] [PubMed] [Google Scholar]
- 4.Zissel G, Müller-Quernheim J. Cellular players in the immunopathogenesis of sarcoidosis. Clin Chest Med 2015; 36: 549–60. [DOI] [PubMed] [Google Scholar]
- 5.Broos CE, Hendriks RW, Kool M. T-cell immunology in sarcoidosis: disruption of a delicate balance between helper and regulatory T-cells. Curr Opin Pulm Med 2016; 22: 476–83. [DOI] [PubMed] [Google Scholar]
- 6.Ramstein J, Broos CE, Simpson LJ, et al. IFN-γ-producing T-helper 17.1 cells are increased in sarcoidosis and are more prevalent than T-helper type 1 cells. Am J Respir Crit Care Med 2016; 193: 1281–91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Zhou T, Casanova N, Pouladi N, et al. Identification of Jak-STAT signaling involvement in sarcoidosis severity via a novel microRNA-regulated peripheral blood mono nuclear cell gene signature. Sci Rep 2017; 7: 4237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Zhou T, Zhang W, Sweiss NJ, et al. Peripheral blood gene expression as a novel genomic biomarker in complicated sarcoidosis. PLoS One 2012; 7(9): e44818. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Li H, Zhao X, Wang J, Zong M, Yang H. Bioinformatics analysis of gene expression profile data to screen key genes involved in pulmonary sarcoidosis. Gene 2017; 596: 98–104. [DOI] [PubMed] [Google Scholar]
- 10.Rosenbaum JT, Pasadhika S, Crouser ED, et al. Hypothesis: sarcoidosis is a STAT1-mediated disease. Clin Immunol 2009; 132: 174–83. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Rosenbach M, Yeung H, Chu EY, et al. Reliability and convergent validity of the Cutaneous Sarcoidosis Activity and Morphology Instrument for assessing cutaneous sarcoidosis. JAMA Dermatol 2013; 149: 550–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Stagaki E, Mountford WK, Lackland DT, Judson MA. The treatment of lupus pernio: results of 116 treatment courses in 54 patients. Chest 2009; 135: 468–76. [DOI] [PubMed] [Google Scholar]
- 13.Wanat KA, Rosenbach M. A practical approach to cutaneous sarcoidosis. Am J Clin Dermatol 2014; 15: 283–97. [DOI] [PubMed] [Google Scholar]
- 14.Talreja J, Talwar H, Ahmad N, Rastogi R, Samavati L. Dual inhibition of Rip2 and IRAK1/4 regulates IL-1β and IL-6 in sarcoidosis alveolar macrophages and peripheral blood mononuclear cells. J Immunol 2016; 197: 1368–78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Bihl MP, Laule-Kilian K, Bubendorf L, et al. Progressive pulmonary sarcoidosis — a fibroproliferative process potentially triggered by EGR-1 and IL-6. Sarcoidosis Vasc Diffuse Lung Dis 2006; 23: 38–50. [PubMed] [Google Scholar]
- 16.Linke M, Pham HT, Katholnig K, et al. Chronic signaling via the metabolic checkpoint kinase mTORC1 induces macrophage granuloma formation and marks sarcoidosis progression. Nat Immunol 2017; 18: 293–302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Ziegenhagen MW, Rothe ME, Zissel G, Müller-Quernheim J. Exaggerated TNFalpha release of alveolar macrophages in corticosteroid resistant sarcoidosis. Sarcoidosis Vasc Diffuse Lung Dis 2002; 19: 185–90. [PubMed] [Google Scholar]
- 18.Sakthivel P, Bruder D. Mechanism of granuloma formation in sarcoidosis. Curr Opin Hematol 2017; 24: 59–65. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.