Abstract
Eosinophils normally constitute only a few per cent of circulating leukocytes, though they are more numerous in tissues vulnerable to attack by environmental microorganisms. Eosinophils can kill invasive parasites, but they also have immunoregulatory functions and may be involved in, for example, the connective tissue remodeling that occurs in conjunction with inflammation. Although their effects may be beneficial to the host, for instance in the event of helminthic infestation, they may also cause tissue damage, for example in allergy and asthma. Recent years have witnessed significant advances in our knowledge of these fascinating but still enigmatic cells.
The eosinophil granulocyte was first described in 1879 by Paul Ehrlich, who discovered a blood cell that had high affinity for acid dyes and, in particular, eosin (1) (Figure 1). Eosin, which gives the cells their characteristic red-orange color, is named after Eos, the goddess of dawn in Greek mythology. Paul Ehrlich also put forward the hypothesis that eosinophils develop in the bone marrow (2) and exert their functions in the peripheral tissues.
All vertebrates seem to have eosinophils in their blood, but these cells have also been described in more primitive organisms such as sharks, turtles and snakes (3). However. there are differences in morphology, and a comparison of eosinophils from different species also reveals considerable differences in the protein content of their characteristic cytoplasmic granules. For example, in contrast to many vertebrates, eosinophils from the cat, rhino, hyena and okapi lack peroxidase (3).
Origin in the bone marrow
Eosinophils mature from the myeloid lineage of developing blood cells in the bone marrow. Multipotent stem cells differentiate due to stimulation by specific growth factors, leading to expression of specific genes which result in the production of the different proteins, e. g. The granule proteins, that give the eosinophils their characteristic features.
The differentiation of an immature stem cell into an eosinophil granulocyte is dependent on several growth factors, including granulocyte/macrophage colony-stimulating factor (GM-CSF), interleukin-3 (IL-3) and, especially, IL-5 (4). Each of these three binds to a specific alpha chain on the surface of an eosinophil granulocyte. The respective alpha chains subsequently compete for a common beta chain and the resulting heterodimer is responsible for transducing signals into the cell.
GM-CSF, IL-3 and IL-S also have effects on mature eosinophils, e.g. activating them in different ways and delaying apoptosis (programmed cell death) (5-7).
Granule proteins
As mentioned earlier, eosinophils have characteristic staining properties, with an affinity for acid dyes. This is due to a high content of positively charged (cationic) proteins in the cytoplasmic granules. These so-called specific granules contain crystalloid structures, giving eosinophils a characteristic appearance in the electron microscope (Figure 2 a). Four different cationic proteins stored in specific granules have been isolated and characterized (8) (Figure 2b).
Figure 2a.
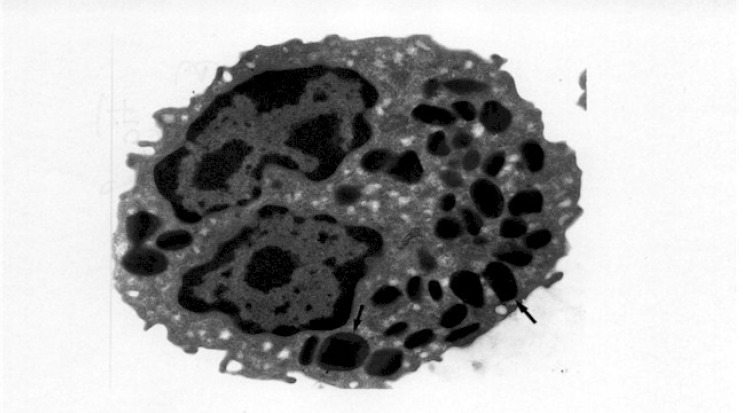
Electron micrograph of an eosinophil granulocyte. The cell has a typical bibbed nucleus (n). In the cytoplasm the characteristic specific granules are seen (arrows). containing crystalloid core.
Figure 2b.

Schematic drawing of the different compartments in the specific granules. Major basic protein (MBP) constitutes the crystalloid core while eosinophil cationic protein (ECP), eosinophilderived neurotoxin (EDN), eosinophil peroxidase (EPO) and cytokines are stored in the matrix of granules.
Major basic protein (MBP) constitutes the crystalloid core of eosinophil granules (9). MBP is cytotoxic to several helminths, protozoa and bacteria in vitro. Instillation of MBP in the airways of rabbits has been shown to induce hyperreactivity and contraction of bronchial smooth muscle, in analogy with asthma in humans (8).
Eosinophil Cationic Protein (ECP) is a granule protein with ribonuclease activity. It is cytotoxic not only to bacteria and helminths but also to human cells (8). At least the bactericidal effect seems to be independent of the ribonuclease activity (10), indicating that other mechanisms of action may also be involved.
Eosinophil-Derived Neurotoxin (EDN) is structurally closely related to ECP and like ECP it has ribonuclease activity (8). Recently, it has been suggested that EDN has a role in host defense against RNA viruses, such as the respiratory syncytial virus (I I). The EDN and ECP genes evolved as the result of a gene duplication that occurred at the time when the New and Old World monkeys were separated (12). Since then, both these genes have accumulated new mutations at a rate exceeding that of all so far studied primate genes. Despite this, both ECP and EDN have conserved their ribonuclease activity. This indicates that both genes are under high evolutionary pressure and that the ribonuclease activity is an important property of both ECP and EDN (12).
The Eosinophil Peroxidase (EPO) molecule displays 60 per cent identity with neutrophil myeloperoxidase at the amino acid level. EPO is cytotoxic, among other things, to certain helminths. EPO also induces mast cell degranulation (8).
Charcot-Leyden Crystal (CLC) protein has for many years been associated with allergic inflammation since it makes up the characteristic bipyramidal, hexagonal microscopic crystals seen in sputa from patients with asthma (fig. 2 c). It is a hydrophobic protein and, in contrast to the cationic proteins, it has a neutral isoelectric point. Both eosinophils and basophils contain large amounts of this protein. CLC has lysophospholipase activity that may help to protect the cell from its own cytotoxic by-products, generated during the production of lipid-derived inflammatory mediators, such as platelet activating factor (PAF) and leukotriene C4(13). CLC protein lacks a typical signal peptide to direct the newly synthesized molecule into the endoplasmic reticulum for subsequent processing in the Golgi apparatus and storage in granules. As a consequence CLC protein is found mainly in the cytoplasm and nucleus of the cells. However, it is also stored in a small subpopulation of granules, and in such a high concentration that it sometimes crystallizes (14).
Figure 2c.

Electron micrograph showing a Charcot-Leyden crystal with a typical hexagonal shape. The ultra thin section was incubated with antibody against CLC protein. The antibody was visualised with protein A-colloidal gold and appears as small black dots scattered over the surface of the crystalloid structure, indicating the presence of CLC protein
Recruitment to sites of inflammation
In the mid 1980s, Mosmann, Coffman and colleagues showed that T helper (Th) cells could be divided into subgroups, Thl and Th2, based on the pattern of cytokines they synthesize (15). Since then, monocytes, NK cells, mastcells, eosinophils and basophils have also been shown to also produce the cytokines typical of Thl and Th2 cells (16). An alteration in the nomenclature to type 1 and type 2 cytokines has therefore been suggested (16). In allergic inflammation, where eosinophils participate, a type-2 pattern of cytokines is seen, which includes the synthesis and release of IL-4, IL-5. IL-6, IL-10 and IL-13. Interferon-g and IL-12 (from cells with type-1 activity) inhibit the activity of cells with a type-2 profile, whereas IL-4 and IL-10 inhibit type-1 activity. In this way a delicate balance between the two systems can be maintained (16) (Figure 3).
Figure 3.

Comparison of type 1 (Thl) and type 2 (Th2) cytokines. Type I cytokines mainly transmit cell-mediated immunity whereas type 2 cytokines transmit humoral immunity. The two systems balance each other by crosswise inhibition, mediated by IFNy IL-12 and IL-4 IL-10, respectively (artist: Piroska von Gegerfeldt).
How can a selective recruitment of eosinophils to the site of allergic inflammation be attained? In recent years, a possible mechanism has emerged whereby type-2 cytokines, such as IL-4 and IL-13, are released locally by, for example, macrophages or lymphocytes in peripheral tissues (17). These cytokines stimulate endothelial cells in post capillary venules, making them express specific adhesion molecules on their surface to which eosinophils, but not neutrophils, have the corresponding ligands (17). Eosinophils interact with the activated endothelial cells in a sequence of events involving several different adhesion molecules (Figure 4).
Figure 4.

Schematic drawing of eosinophil recruitment to the site of an inflammatory reaction.
- Tissue cells, e.g. lymphocytes (Ly) or macrophages (Mø), stimulate endothelial cells in postcapillary venules to express surface adhesion molecules on their surface.
- Initially the cell is ”marginated” in the blood vessel and starts ”rolling” along the endothelium, a process mediated by adhesion molecules of the selectin type.
- The eosinophil granulocytes are activated, in particular by inflammatory mediators released by the endothelial cells, and subsequently bind strongly to the endothelial cells. This firm binding is mediated through ß1- and ß2-integrins. The activation of ß2-integrins also cause a shape change and the eosinophil becomes flattened.
- Several inflammatory mediators produced at the site of inflammation stimulate the eosinophil granulocytes to leave the blood vessel, using its ß1- and ß2-integrins, and enter the tissue where it exerts its function.
(artist: Piroska von Gegerfeldt)
Concentration gradients of substances produced at the site of inflammation guide the cell (chemotaxis) out into the tissues. The combination of substances with chemotactic effects can also result in a selective recruitment of eosinophils. In addition, eosinophils can leave the tissues through the epithelium or mesothelium in order to exert their effect on the inside surface of a lumen, for example in a bronchus, or in the gastrointestinal or urinary tract.
Main function is host defense
It is thought that the main function of eosinophils is in the body’s defense against parasites. Eosinophils can kill several different parasites in vitro, especially in their larval stage (8). Release of the cationic proteins onto the surface of the parasite is likely to be important in this context. Eosinophils have therefore been regarded as primary secretory cells, releasing their granular content into the external environment in contrast to neutrophils which phagocytose microbes and degranulate into the intracellular phagosome containing the microorganism. However, eosinophils are capable of using both mechanisms (Figure 5).
Figure 5.

- The cell phagocytoses bacteria with immunoglobulins and complement molecules on their surface. The bacteria are enclosed in an invagination of cell membrane that ñnally becomes a phagosome of their cytoplasm. Specific granules are transported to the phagosome (1), and after fusion (2) the granular content is released (3) and the cytotoxic process started.
- The eosinophil granulocyte attacks a parasite too big to phagocytose, resulting in ”frustrated” phagocytosis. The granules are transported to the part of the cell membrane that is in contact with the parasite (1). The membranes of the granule and the eosinophil plasma membrane fuse (2) followed by release of the granule content onto the surface of the parasite (3) where the cytotoxic effects are exerted. (artist: Piroska von Gegerfeldt)
The process of degranulation is dependent on cellular activation and the binding of specific receptors to the surface of target cells that are covered with immunoglobulins and complement products. In particular, secretory IgA, IgG and the complement fragment iC3b, immobilized on a surface, seem to be potent inducers of degranulation (18-20). However, it is still a matter of controversy whether eosinophils have receptors for IgE a typical component of allergic inflammation (21).
Inflammatory mediators
Beside the cytotoxic effects of the cationic proteins MBP, ECP, and EPO, eosinophils can, similarly to neutrophils, kill target cells with the oxidase-system present in the cell membrane (8).
In recent years it has been shown that eosinophil granules contain several cytokines and chemokines and that these can be synthesized by activated eosinophils in tissues (22). Among these are cytokines with activating effects on eosinophils (IL-3, IL-5 and GM--CSF), proinflammatory cytokines and chemokines (IL-I, IL-6, IL-8, RANTES [Regulation upon Activation Normal T-cell Expressed and Secreted], MIP-la [Macrophage Inflammatory Protein -1a] and TNF-a [Tumor Necrosis Factor-a] and immunoregulatory cytokines (IL-2, IL-4, IL-10 and interferon-g).
It is of particular interest is that preformed growth factors such as Transforming Growth Factor (TGF)-a [23, 24] and TGF-b1 [25] can be synthesized by eosinophils and carried in the eosinophil granules. The presence of these can explain the fibrosis often seen in conjunction with long-standing eosinophilia but could also point to a possible interaction between eosinophils and cells that have undergone malignant transformation. For example in Hodgkin’s disease (in particular the nodular sclerosing type) and adenocarcinoma of the colon, infiltrating eosinophils are often seen in the tumor tissue (3).
Eosinophils can also synthesize several lipid-derived proinflammatory substances such as prostaglandin E2, thromboxane A2, leukotriene C4, I 5-HETE (15-hydroxyeicosatetraenoic) acid and lipoxin A 4 [26]. The production of these takes place mainly in association with lipid bodies in the cytoplasm [27]. The lipid-derived proinflammatory substances may be of pathophysiological significance in the allergic inflammatory reaction [26].
Programmed cell death
Eosinophils can die in tissues due to environmental factors, i. e. necrosis. In addition, eosinophils like many other cells have a built-in mechanism of programmed cell death, i. e. apoptosis. Apoptosis is characterized by fragmentation of the nuclear DNA caused by activation of an endonuclease. Upon electrophoretic separation of DNA from apoptotic cells a typical ladder pattern is seen, where the ‘steps’ are made up of DNA fragments. Apoptosis is probably a mechanism by which the body avoids the accumulation of eosinophils, since they are mainly effector cells and probably can not renew their store of granule proteins. Eosinophils isolated from peripheral blood die after 24-48 hours in culture due to apoptosis. If GM-CSF, IL-3 or IL-5 is added, their life-span increases dramatically and can reach 2-3 weeks when maintained in culture [5-7]. It is therefore reasonable to believe that the eosinophils in the tissues have a fairly long life-span. Apoptotic eosinophils are rapidly recognized by macrophages, which phagocytose the dead cells without any accompanying inflammatory reaction [28].
The role of eosinophils in disease
To study only the concentration of eosinophils in the blood can be misleading since this number rather reflects the balance between production in the bone marrow and recruitment to the tissues, and not the total amount of eosinophils in the body.
Eosinophils are mainly tissue-dwelling cells and there are 100-1000 eosinophils in the different tissues for every eosinophil found in the bloodstream. In adults less than 0.4 × l09 eosinophils per liter is considered normal, while newborns have higher levels [29]. In approximate numbers it can be said that 0.4 - 1.5 × 109 per liter represents mild eosinophilia, 1.5-5.0 × 109 per liter, moderate eosinophilia and >5.0 × 109 per liter, severe eosinophilia [29].
From a global perspective parasitic infection is the most common cause of eosinophilia and the resulting eosinophilia is often severe. In the industrialized countries allergy and asthma are the diseases most commonly associated with eosinophilia, however, usually only in a mild form. Severe eosinophilia in patients with asthma should give rise to the suspicion that a complication, e.g. the Churg-Strauss syndrome or allergic bronchopulmonary aspergillosis, could be present.
The idiopathic hypereosinophilic syndrome Of the whole plethora of conditions associated with blood or tissue eosinophilia (Table I) a condition that in recent years has been treated in a new way deserves special comment. A persistent idiopathic proliferation of eosinophils accompanied by organ damage is designated the idiopathic hypereosinophilic syndrome (HES). It is probably a heterogeneous condition, with several etiologies [30].
HES can be divided into two subgroups, a non-malignant and a malignant form. The non-malignant form is often accompanied by a high IgE level and angio oedema and responds to treatment with steroids. The malignant form has myelodysplastic characteristics such as splenomegaly, a more pronounced eosinophilia and cytogenetic abnormalities and is therefore reminiscent of chronic myeloid leukemia. Complications seen in HES include endocardial fibrosis, neuropathy and thromboembolism. In recent years, interferon-a has been shown to have a good therapeutic effect in the treatment of HES. This is probably due to a direct antiproliferative effect on eosinophil progenitors in the bone marrow [31]. Previously, the conventional therapy consisted of steroids or hydroxyurea.
Conclusion
From a global perspective, eosinophilia is common. In less developed countries, where chronic parasitic infections are common, eosinophils may help the body to encapsulate and fight invading parasites. In the industrialized countries the situation is quite different; eosinophilia is most often seen in the allergic inflammation and is triggered by antigens that do not necessarily need to be fought against in this way. Consequently we often only see the negative side of the allergic inflammation as allergy and asthma. Eosinophils probably have several different functions in the body and may be of importance in the connective tissue remodeling seen in wound healing, fibrosis and the
Figure 1.

Eosinophils in blood from a patient with the idiopathic hypereosinophilic syndrome. The picture was obtained using light microscopy and May-Grunwald-Giemsa staining on a smear from peripheral blood.
TABLE 1.
Conditions frequently associated with blood or tissue eosinophilia (3).
| Disease | Comment |
|---|---|
| Infectious diseases | |
| Helminthic infections | |
| Ceswdes | Rarely eosinophilia |
| Echinococcosis | Eosinophilia in 25-50% of patients with hydatid cyst |
| (Taenia) | |
| Nematodes | High eosinophilia, especially in children. |
| Ascaris | Eosinophilia, especially in children under age 9. |
| Toxocara canis | Invariably high eosinophilia. |
| Filariasis | |
| Anchylostomiasis | |
| Trichinosis | |
| Strongvloidiasis | |
| Trematodes | |
| Fascioliasis | The most common cause of eosinophilia globally. |
| Schistosomiasis | |
| Fungal Infections | Rarely associated with eosinophilia. |
| Aspergillosis | |
| Coceidioidomycosis | |
| Histoplasmosis | |
| Viral Infections | Seldom associated with eosinophilia. |
| Respiratory syncytial virus | Mainly eosinophilia of tissues. |
| Dermatologic diseases | |
| Atopic dermatitis | Common cause of eosinophilia, especially in children. |
| Episodic angiooedema with eosinophilia | |
| Bullous pemfigoid | |
| Immunologic and | |
| rheumatologic diseases | Pronounced eosinophilia. |
| Eosinophil fasciitis | Seldom eosinophilia. |
| Rheumatoid arthritis | |
| Eosinophil myositis | |
| Neoplastic diseases | Rare. Approximately 100 cases reported. |
| eosinophil leukemia | Especially the nodular sclerosing form. |
| Chronic myeloid leukemia | |
| Hodgkin’s disease | |
| Histiocytosis-X | |
| Angiolymphoid hyperplasia with | |
| eosinophilia (Kimura’s disease) | |
| Adenocarcinoma of the gastrointestinal | |
| tract | |
| Eosinophilia from toxic effect (?) | |
| The Spanish Toxic Oil Syndrome | Probably caused by contaminated tryptophane |
| Tryptophan | |
| Diseases of the gastrointestinal tract | Mainly tissue-eosinophilia |
| Eosinophilic gastroenteritis | Mainly tissue-eosinophilia |
| Coeliac disease | Mainly tissue-eosinophilia |
| Food-allergy | |
| Ulcerative colitis | |
| Diseases of the airways | Eosinophil vasculitis and asthma. Moderate to pronounced |
| Asthma, allergic and non-allergic | eosinophilia |
| Churg-Strauss’ syndrome | Common cause of mild eosinophilia. |
| Allergic rhinitis | |
| Loftier syndrome | |
| Chronic eosinophilic pneumonia | |
| Nasal polyposis | |
| Diseases of the nervous system | |
| Eosinophil meningitis | |
| Idiopathic eosinophilia | |
| The Idiopathic Hypereosinophilic Syndrome (VIES) | |
| Other | |
| Therapy with IL-2 |
TABLE 1.
Conditions frequently associated with blood or tissue eosinophilia (3).
| Disease | Comment |
|---|---|
| Infectious diseases | |
| Helminthic infections | |
| Ceswdes | Rarely eosinophilia |
| Echinococcosis | Eosinophilia in 25-50% of patients with hydatid cyst |
| (Taenia) | |
| Nematodes | High eosinophilia, especially in children. |
| Ascaris | Eosinophilia, especially in children under age 9. |
| Toxocara canis | Invariably high eosinophilia. |
| Filariasis | |
| Anchylostomiasis | |
| Trichinosis | |
| Strongvloidiasis | |
| Trematodes | |
| Fascioliasis | The most common cause of eosinophilia globally. |
| Schistosomiasis | |
| Fungal Infections | Rarely associated with eosinophilia. |
| Aspergillosis | |
| Coceidioidomycosis | |
| Histoplasmosis | |
| Viral Infections | Seldom associated with eosinophilia. |
| Respiratory syncytial virus | Mainly eosinophilia of tissues. |
| Dermatologic diseases | |
| Atopic dermatitis | Common cause of eosinophilia, especially in children. |
| Episodic angiooedema with eosinophilia | |
| Bullous pemfigoid | |
| Immunologic and | |
| rheumatologic diseases | Pronounced eosinophilia. |
| Eosinophil fasciitis | Seldom eosinophilia. |
| Rheumatoid arthritis | |
| Eosinophil myositis | |
| Neoplastic diseases | Rare. Approximately 100 cases reported. |
| eosinophil leukemia | Especially the nodular sclerosing form. |
| Chronic myeloid leukemia | |
| Hodgkin’s disease | |
| Histiocytosis-X | |
| Angiolymphoid hyperplasia with | |
| eosinophilia (Kimura’s disease) | |
| Adenocarcinoma of the gastrointestinal | |
| tract | |
| Eosinophilia from toxic effect (?) | |
| The Spanish Toxic Oil Syndrome | Probably caused by contaminated tryptophane |
| Tryptophan | |
|
Diseases of the gastrointestinal tract Eosinophilic gastroenteritis Coeliac disease Food-allergy Ulcerative colitis |
Mainly tissue-eosinophilia Mainly tissue-eosinophilia Mainly tissue-eosinophilia |
| Diseases of the airways | Eosinophil vasculitis and asthma. Moderate to pronounced |
| Asthma, allergic and non-allergic | eosinophilia |
| Churg-Strauss’ syndrome | Common cause of mild eosinophilia. |
| Allergic rhinitis | |
| Loftier syndrome | |
| Chronic eosinophilic pneumonia | |
| Nasal polyposis | |
| Diseases of the nervous system | |
| Eosinophil meningitis | |
| Idiopathic eosinophilia | |
| The Idiopathic Hypereosinophilic Syndrome (VIES) | |
| Other | Treatment of hypernephroma. |
| Therapy with IL-2 |
Footnote
This article has in part been previously published in The Swedish Medical Journal (Läkartidningen) 1998; 95: 850-859
We are grateful to Dr. Bengt Johansson, Lund, Sweden, for linguistic revision of the manuscript
LITERATURE CITED
- 1.Ehrlich P. Ueber die specifischen granulationen des Blutes. Arch Anat Physiol Lpz. 1879; 3 Physiol Abt: 571-579. [Google Scholar]
- 2.Ehrlich P. Beitrage zur kenntniss der granulierten bindergewebszellen und der eosinophilen leukocyten. Arch Anat Physiol Lpz. 1879; 3 Physiol Abt: 166-169. [Google Scholar]
- 3.Spry CJF. Eosinophils. A comprehensive review and guide to the scientific and medical literature. Oxford: Oxford University Press, 1988. [Google Scholar]
- 4.Sanderson CJ. Interleukin-5, eosinophils, and disease. Blood 1992; 79: 3101-3109. [PubMed] [Google Scholar]
- 5.Lopez AF, Williamson DJ, Gamble JR, Begley CG, Harlan JM, Klebanoff SJ, et al. Recombinant human granulocyte-macrophage colony stimulating factor stimulates in vitro mature human neutrophil and eosinophil function, surface receptor expression, and survival. J Clin Invest 1986; 78: 1220-1228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Lopez AF, To LB., Yang Y-C, Gamble JR, Shannon MF, Burns GF, et al. Stimulation of proliferation, differentiation, and function of human cells by primate interleukin 3. Proc Natl Acad Sci USA 1987; 84: 2761-2765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Yamaguchi Y, Hayashi Y, Sugama Y, Miura Y, Kashahara T, Kitamura S, et al. Highly purified murine interleukin 5 (IL-5) stimulates eosinophil function and prolongs in vitro survival. IL-S as an eosinophil chemotactic factor. J Exp Med 1988; 167: 1737-1742. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Gleich GJ, Adolphson CR, Leiferman KM. The eosinophil. Gallin JI, Goldstein IM, Snyderman R, Eds. Inflammation: basic principles and clinical correlates. Boca Raton: Raven, 1992: 663-700. [Google Scholar]
- 9.Egesten A, Alumets J, von Mecklenburg C, Palmegren M, Olsson I. Localization of eosinophil cationic protein, major basic protein, and eosinophil peroxidase by immunoelectron microscopic technique. J Histochem Cytochem 1986: 34: 1399-1403. [DOI] [PubMed] [Google Scholar]
- 10.Rosenberg HF. Recombinant human eosinophil cationic protein. Ribonuclease activity is not essential for cytotoxicity. J Biol Chem 1995; 270: 7876-7881. [DOI] [PubMed] [Google Scholar]
- 11.Domachowske JB, Dyer KD, Bonville CA, Rosenberg HF. Recombinant human eosinophil-derived neurotoxin/RNase 2 functions as an effective antiviral agent against respiratory syncytial virus. J Infect Dis 1998; 177:1458-1464. [DOI] [PubMed] [Google Scholar]
- 12.Rosenberg HF, Dyer KD, Tiffany HL, Gonzales M. Rapid evolution of a unique family of primate ribonuclease genes. Nature Genetics 1995; 10: 219-223. [DOI] [PubMed] [Google Scholar]
- 13.Weller PF, Bach DS, Austen KR. Biochemical characterization of human eosinophil Charcot-Leyden crystal protein (lysophospholipase). J Biol Chem 1984; 259: 15100-15105 [PubMed] [Google Scholar]
- 14.Calafat J, Janssen H, Knol EF, Weller PF, Egesten A. Ultrastructural localization of Charcot-Leyden crystal protein in human eosinophils and basophils. Eur J Haematol 1997; 58: 58-66. [DOI] [PubMed] [Google Scholar]
- 15.Mosmann TR, Coffman RL. Th I and Th 2 cells: different patterns of lymphokine secretion lead to different functional properties. Ann Rev Immunol 1989; 7:145-173. [DOI] [PubMed] [Google Scholar]
- 16.Lucey DR, Clerici M, Shearer GM. Type I and type 2 cytokine dysregulation in human infectious, neoplastic, and inflammatory diseases. CIin Microbiol Rev 1996; 9:532-562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Bochner BS, Schleimer RP. The role of adhesion molecules in human eosinophil and basophil recruitment. J Allergy Clin Immunol 1994; 94: 427-438. [DOI] [PubMed] [Google Scholar]
- 18.Egesten A, Gullberg U, Olsson I, Richter I. Phorbol ester-induced degranulation in adherent human eosinophil granulocytes is dependent on CDI 1/CD 18 leukocyte integrins. J Leukocyte Biol 1993; 53: 287-293. [DOI] [PubMed] [Google Scholar]
- 19.Hone 5, Kita H. CD1 lb/CD18 (Mac-i) is required for degranulation of human eosinophils induced by human recombinant granulocyte-macrophage colony-stimulating factor and platelet-activating factor. J Immunol 1994; 152: 5457-5467. [PubMed] [Google Scholar]
- 20.Abu-Ghazaleh RI, Fujizawa T, Mestecky J, Kyle RA, Gleich Gi. IgA-induced eosinophil degranulation. J Immunol 1989; 142: 2393-2400. [PubMed] [Google Scholar]
- 21.Kita H, Gleich GJ. Eosinophils and IgE receptors: A continuing controversy. Blood 1997; 89: 3497-3501. [PubMed] [Google Scholar]
- 22.Weller PF. Human eosinophils. J Allergy Immunol 1997; 100: 283-287. [DOI] [PubMed] [Google Scholar]
- 23.Wong DTW, Weller PF, Galli SJ, Elovic A, Rand TH, Gallagher GT, et al. Human eosinophils express transforming growth factor a. J Exp Med 1990; 172: 673-681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Egesten A, Calafat J, Knot EF, Janssen H, Walz TM. Subcellular localization of transforming growth factor-a in human eosinophil granulocytes. Blood 1996; 87: 3910-3918. [PubMed] [Google Scholar]
- 25.Wong DTW, Elovic A, Matossian K, Nagura N, McBride I, Chou MY, et al. Eosinophils from patients with blood eosinophilia express transforming growth factor b1. Blood 1991; 78: 2702-2707. [PubMed] [Google Scholar]
- 26.Weller PF. Lipid, peptide, and cytokine mediators elaborated by eosinophils. Smith H, Cook M. Eds. Immunopharmacology of eosinophils: The handbook of immunopharmacology. London: Academic Press Ltd., 1993: 25-42. [Google Scholar]
- 27.Weller PF, Moynihan-Earley RA, Dvorak HF, Dvorak AM. Cytoplasmic lipid bodies of human eosinophils. Subcellular isolation and analysis of arachidonate incorporation. Am J Pathol 1991; 138: 141-148. [PMC free article] [PubMed] [Google Scholar]
- 28.Stern M, Meagher L, Savill J, Haslett C. Apoptosis in human eosinophils. Programmed cell death in the eosinophil leads to phagocytosis by macrophages and is modulated by IL-5. J Immunol 1992; 148: 3543-3549. [PubMed] [Google Scholar]
- 29.Wardlaw AJ. Eosinophils in the 1990’s: new perspectives on their role in health and disease. Pstgrad Med J 1994; 79: 536-552. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Weller PF, Bubley J. The idiopathic hypereosinophilic syndrome. Blood 1994; 83: 2759-2779. [PubMed] [Google Scholar]
- 31.Butterfield JH, Gleich GJ. Response of six patients with idiopathic hypereosinophilic syndrome to interferon alpha. J Allergy Clin Immunol 1994; 94: 1318-1326. [DOI] [PubMed] [Google Scholar]


