Abstract
Background:
Surgeons are increasingly asked to operate on patients with residual disease after immunotherapy. The safety and utility of lung resection in this setting are unknown.
Methods:
We performed a retrospective review of patients who underwent lung resection within 6 months of treatment with checkpoint blockade agents for metastatic or unresectable cancer. Survival was estimated from first resection using the Kaplan-Meier approach.
Results:
Database query identified 19 patients who underwent 22 resections for suspected residual disease with therapeutic intent following immunotherapy between 2012 and 2016. Lung cancer was the most common diagnosis (47%), followed by metastatic melanoma (37%). The most frequently used agents were nivolumab (32%), pembrolizumab (32%), and ipilimumab (16%). Patients received a mean of 21 doses (range, 1–70); the final dose was administered, on average, 75 days before surgery (range, 7–183). Anatomic resection (lobectomy or greater) was performed in 11 cases (50%). Four lobectomies were attempted minimally invasively; 1 required conversion to thoracotomy. Of the resected patients, 68% had viable tumor remaining. R0 resection was achieved in 95% of cases. Mean operative time for lobectomy was 227 minutes (range, 150–394). Complications occurred in 32% of cases; all but 1 were minor (grade 1/2). Two-year overall and disease-free survival were 77% and 42%.
Conclusions:
In patients with previously metastatic or unresectable cancer, lung resection for suspected residual disease following immunotherapy is feasible, with high rates of R0 resection. Operations can be technically challenging, but significant morbidity appears to be rare. Outcomes are encouraging, with reasonable survivals during short-interval follow-up.
Introduction
Recent advances in cancer biology have begun to elucidate the mechanisms by which tumors evade immune recognition. As such, strategies to exploit these mechanisms—such as the use of monoclonal antibodies targeting the immune regulatory proteins programmed death-ligand 1 (PD-L1) and cytotoxic T -lymphocyte-associated protein 4 (CTLA-4)—are now viable treatment options for patients with metastatic cancer. Results of recent clinical trials have demonstrated durable treatment responses in subsets of patients with metastatic solid tumors (1, 2). As a result, and with the expansion of clinical guidelines to include larger patient subsets, FDA approval of additional drugs, and aggressive direct-to-consumer marketing strategies, the popularity of immunotherapy agents is rapidly increasing.
It is notable that the diseases for which these agents are commonly prescribed, such as non-small cell lung cancer (NSCLC) and cutaneous melanoma, are often associated with a significant intrathoracic disease burden. Therefore, patients are frequently referred for thoracic surgical consultation to address the possibility of residual disease in the setting of partial or complete treatment response.
Although the therapeutic value of resecting residual primary or metastatic foci remains controversial in general, the safety and feasibility of pulmonary resection following treatment with immunotherapy agents in particular have not yet been studied. Previous clinical trials have suggested a small but not insignificant risk of complications, such as pneumonitis, thyroiditis, and adrenal insufficiency, in patients treated with these agents (3–5). Although many of these complications are of low severity, their cumulative effect on the perioperative care of patients is unclear. Similarly, the effect of the inflammatory response that is critical to the mechanism of action of these agents on the technical aspects of lung resection remains to be determined. Our institution previously published a case series on surgical resection in 5 initial patients receiving checkpoint inhibitors for advanced lung cancer (6); here, we incorporate those patients into a larger, more diverse cohort. The goal of this study was to analyze clinical, surgical, and outcomes data on patients referred for pulmonary resection following treatment with immune checkpoint inhibitors for previously metastatic or unresectable tumors at our quaternary care center.
Patients and Methods
We performed a retrospective review of our prospectively maintained thoracic surgical database to identify patients who underwent pulmonary resection within 6 months of treatment with T -cell checkpoint inhibitors. Eligible patients were previously diagnosed with biopsy-proven NSCLC or metastatic cancer from another site. At the time of presentation, lung cancer patients were deemed to be unresectable on the basis of presence of distant metastatic disease or extensive mediastinal lymphadenopathy. Patients undergoing immunotherapy as part of a planned induction strategy were specifically excluded. Patients were typically referred for surgery if they had radiographic findings suspicious for persistent or progressive disease in the lung following checkpoint blockade therapy. Patients with metastatic NSCLC referred for surgery generally had clinical or pathologic resolution of their extrathoracic metastatic sites following either checkpoint blockade therapy alone or checkpoint blockade therapy in conjunction with a local modality, such as stereotactic radiation or surgical resection. Institutional review board approval was previously obtained. Patient charts were individually reviewed, and data regarding demographic, clinical, and pathologic variables and outcomes were abstracted.
Immunotherapy treatment included anti–PD-1 agents (nivolumab and pembrolizumab), anti–CTLA-4 agents (ipilimumab), or anti–PD-L1 agents (durvalumab and atezolizumab). NSCLC was staged according to the 7th edition of the American Joint Committee on Cancer’s TNM staging criteria. Operative time was extracted from the operating room documentation and was defined as the time from incision to wound closure. Surgical complications were defined according to the Society of Thoracic Surgeons database criteria and were recorded prospectively. Estimates of survival were determined according to the Kaplan-Meier method. Overall survival was calculated from the date of the initial resection to the date of death, or patients were censored at the date of the last follow-up. Disease-free survival was calculated in the same manner, but instead patients were censored at the date of death, disease recurrence, or last follow-up. Patients were followed postoperatively with routine CT scans every 3 to 6 months, at the discretion of the treating physician. Recurrence was determined either radiographically or pathologically by the treating physician. Tissue biopsy was not required to document recurrence. We compared observed complication rates with the historical control using the one-sample proportion test with continuity correction. Statistical calculations were performed using SPSS Statistics (version 24, IBM, Armonk, NY).
Results
Database query identified 19 patients who underwent 22 resections between 2012 and 2016. Demographic and clinical information for the cohort is listed in Table 1. Mean age at surgery was 58 years (range, 37–73 years). Lung cancer was the most common diagnosis (n= 9, 47%), followed by metastatic melanoma (n= 7, 37%). Two patients had breast cancer, and 1 had sarcoma. Of the patients with NSCLC, the majority had stage IV disease at the time of presentation (n=7, 78%). Most patients with metastatic disease had disease outside of the chest on initial presentation (brain [n=3], adrenal [n=2], omentum [n=1], soft tissue [n=1]). One patient had bilateral lung metastases. These patients were referred for surgery if metastatic sites were completely treated but if findings suspicious for persistent disease remained in the lung. The remaining 2 patients had stage IIIA disease but were initially deemed to be unresectable owing to extensive mediastinal lymphadenopathy. All patients with histologic subtypes other than NSCLC had lung metastases.
Table 1.
Demographic and clinical information for patients undergoing pulmonary resection following immunotherapy
| Characteristic | All Patients (n=19) |
|---|---|
| Age, years, mean (range) | 58 (37–73) |
| Diagnosis | |
| Lung cancer | 9 (47) |
| Melanoma | 7 (37) |
| Other | 3 (16) |
| Drug regimen | |
| Nivolumab | 6 (32) |
| Pembrolizumab | 6 (32) |
| Ipilimumab | 3 (16) |
| Combined ipilimumab and nivolumab | 1 (5) |
| Other | 3 (16) |
| Doses before resection, mean (range) | 21 (1–70) |
| Time from last treatment to surgery, days, mean (range) | 75 (7–183) |
Unless otherwise noted, data are no. (%).
Of the patients with lung cancer, 5 (55%) received systemic chemotherapy before initiation of checkpoint blockade. No patients received thoracic radiation in the preoperative setting. Four patients with lung cancer received checkpoint blockade as first-line therapy as part of a clinical trial. Among patients with primary malignancies other than lung cancer, 7 of 10 (70%) received immunotherapy as first-line treatment. One received interferon-alpha, and two received systemic chemotherapy before beginning treatment.
The most commonly used single-agent checkpoint inhibitors were nivolumab, pembrolizumab, and ipilimumab (n= 6, 32%, n= 6, 32%, and n=3, 16%, of cases, respectively) (Table 1). One patient (5%) received a combination of nivolumab and ipilimumab. Patients received an average of 21 doses before resection (median, 16; range, 1–70 doses). The mean duration from final treatment to surgery was 75 days (range, 7–183 days).
Surgical details for the patient cohort are listed in Table 2. Anatomic resection (lobectomy or greater) was performed in 11 cases (50%). Anatomic resections included lobectomy (n=8), bilobectomy (n=2), and pneumonectomy (n=1). Of the 11 wedge resections, 10 were minimally invasive wedge resections, and 1 was a combined wedge and chest wall resection. A minimally invasive approach was attempted in 5 of the 11 anatomic resections (45%; 4 lobectomies and 1 bilobectomy), and 1 of the lobectomies (20%) required conversion to thoracotomy. Mean operative time for lobectomy was 227 minutes (median, 214 minutes) with substantial variability in operative time (range, 150–394 minutes). Mean estimated blood loss was 98 mL (range, 3–500 mL).
Table 2.
Operative details of 22 pulmonary resections following immunotherapy
| Operative Detail | All Procedures (n=22) |
|---|---|
| Extent of resection | |
| Lobectomy | 8 (36) |
| Bilobectomy | 2 (9) |
| Pneumonectomy | 1 (5) |
| Wedge | 11 (50) |
| Initial approach | |
| VATS | 12 (55) |
| Robotic | 3 (14) |
| Open | 7 (32) |
| Laterality | |
| Right | 11 (50) |
| Left | 11 (50) |
| Complete (R0) resection | 21 (95) |
| Operative time, minutes, mean (range) | 168 (45–394) |
| Operative time for lobectomy, minutes, mean (range) | 227 (150–394) |
| Estimated blood loss, mL, mean (range) | 98 (3–500) |
| Viable tumor on final pathologic assessment | 15 (68) |
Unless otherwise noted, data are no. (%). VATS, video-assisted thoracic surgery.
Of the 22 resections, 15 (68%) involved viable tumor on final pathologic assessment. A positive margin (R1 or R2) was encountered in only 1 case (5%), which involved a positive R1 (<1 mm) margin following a wedge resection. Of these 22 procedures, 7 were associated with 1 or more complications (overall morbidity, 32%) (Table 3). Two procedures were associated with 2 complications (pneumonia and hyponatremia following a lobectomy and hypotension and nausea following a lobectomy). Most complications were minor in severity, with the exception of a single grade 4 complication involving postoperative pneumonitis (5%) in the contralateral lung approximately 2 weeks after a VATS wedge resection. This patient required mechanical ventilation but ultimately made a full recovery and returned to the operating room for a lobectomy of the affected side to treat his remaining disease 3 months after his initial procedure. Regarding the remaining complications, the most common was prolonged air leak (n=2, 9%), followed by arrhythmia (n=1, 5%), pneumonia (n=1, 5%), and urinary retention (n=1, 5%). There were no deaths at 30 or 90 days. There was no significant difference in the rates of complications between patients who had a complete response and those who had viable tumor remaining on final pathologic assessment.
Table 3.
Operative mortality and complications in 22 pulmonary resections following immunotherapy
| Complication | No. (%) | Highest Grade |
|---|---|---|
| 30-day mortality | 0 (0) | - |
| 90-day mortality | 0 (0) | - |
| Any grade 3 or higher | 1 (5) | - |
| Any grade 2 | 3 (14) | - |
| Any grade 1 | 5 (23) | - |
| Pneumonitis | 1 (5) | 4 |
| Prolonged air leak | 2 (9) | 2 |
| Arrhythmia | 1 (5) | 2 |
| Pneumonia | 1 (5) | 2 |
| Urinary retention | 1 (5) | 1 |
| Hypotension | 1 (5) | 1 |
| Nausea | 1 (5) | 1 |
| Hyponatremia | 1 (5) | 1 |
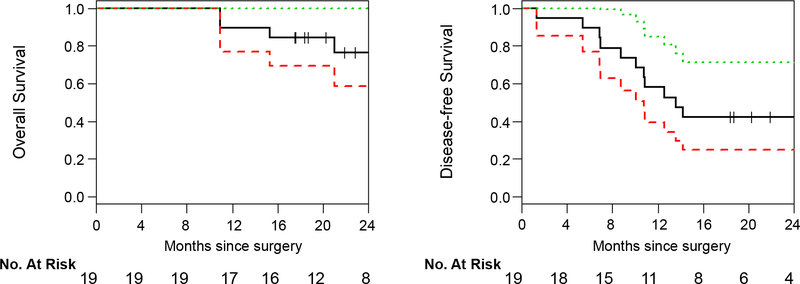
Kaplan-Meier curves for overall survival (OS) and disease-free survival (DFS) are shown in Figure 1. Two-year OS and DFS were 77% (95% CI, 58%−100%) and 42% (95% CI, 25%−71%), respectively. Median (25th, 75th percentile) duration of follow-up was 21.8 months (17.9, 30.9), with a mean of 26.3 months and a range of 10.9 to 66.1 months. Complete information regarding clinical information, treatment details, and follow-up for the study cohort is displayed in Supplemental Table 1.
Figure 1.
Kaplan-Meier curves displaying 2-year overall (left) and disease-free (right) survival in 19 patients undergoing pulmonary resection following immunotherapy (black line). Red and green lines demonstrate the 95% confidence intervals for the curves.
Postoperatively, of the 9 patients with lung cancer, 3 (33%) received some form of adjuvant treatment following completion of surgical therapy. One patient underwent postoperative radiotherapy (PORT), 1 resumed checkpoint blockade, and 1 received systemic chemotherapy. The remaining 6 patients underwent active surveillance. Of the 10 patients with primary malignancies other than lung cancer, 5 (50%) received additional treatment. Two patients received additional postoperative chemotherapy, 2 resumed checkpoint blockade, and 1 received kinase inhibitor therapy. In total, 16 of 19 patients in this study (84%) discontinued checkpoint inhibitor treatment postoperatively.
Comment
The emergence of immune checkpoint inhibitors has rapidly changed the treatment of patients with advanced solid tumors, such as NSCLC and melanoma. While studies on patient selection and effective biomarkers are ongoing, the preliminary finding of a durable benefit of immune checkpoint inhibitors in patients with advanced disease is encouraging (1, 2, 7). These favorable outcomes have resulted in a remarkable increase in the popularity of these agents among both patients and physicians alike. However, patients with a dramatic response to treatment frequently have resolution of several metastatic sites, with only a small burden of suspected or confirmed residual disease. The management of these lesions poses new and challenging questions for the clinician.
Owing to the high frequency of intrathoracic involvement in many of the malignancies immunotherapy is currently used to treat, thoracic surgical consultation is increasingly requested for the evaluation of residual pulmonary lesions following immunotherapy. Whereas surgical resection of these sites may provide important diagnostic information, and potentially even therapeutic benefit, there is limited published information regarding perioperative safety in this patient population. In 2013, Gyorki et al. published a series of 23 patients undergoing general surgical procedures following ipilimumab treatment for melanoma (8). The authors of this study, which consisted mainly of skin resections and laparotomies, observed no major morbidity but did observe a wound complication rate of 22%. To our knowledge, aside from our initial case series of 5 patients, no study has examined the safety of pulmonary resection in patients who have received immunotherapy (6).
Whereas all patients in this study received checkpoint inhibitors due to the presence of initially unresectable disease, and not due to medical inoperability, checkpoint inhibitors will likely become a more attractive treatment option for older patients or for those with comorbidities who are not optimal surgical candidates from the outset. Thus subsequent referral of these patients for resection following treatment may create a high-risk population as the use of these agents increases. Furthermore, such high-risk patients may have received multiple lines of treatment (chemotherapy, targeted therapy, etc.), as well as multiple courses of immunotherapy (the mean number of doses in this series was 21), before resection is considered. In addition, recent studies demonstrating impressive outcomes in patients with stage III NSCLC undergoing chemoradiation followed by immunotherapy will likely result in further application of these agents to patients with operable disease (9, 10).
The potential toxicity profiles of immunotherapy agents may also pose concerns that are unique to lung resection.(11, 12) In the Keynote trial of pembrolizumab for metastatic NSCLC, pneumonitis was seen in 4% of patients. Half of these cases were grade 3 or higher, and 1 case was fatal (13). Similarly, in the Checkmate 012 trial, which examined the combination of ipilimumab and nivolumab in a similar patient population, pneumonitis was seen in 10% of patients (14). Emerging evidence also suggests that previously unanticipated side effects, such as adrenal insufficiency and transient hyperthyroidism, might pose additional risks in the perioperative period (1, 15). As these drugs are increasingly moved into neoadjuvant trials, operative feasibility and safety are of the utmost significance.
In the current study, we examined a cohort of 19 patients with metastatic or advanced malignancy who underwent 22 pulmonary resections following treatment with immune checkpoint inhibitors. Surgical complications occurred in 32% of cases, and there was a single case of major (grade 3 or higher) morbidity. Interestingly, this involved pneumonitis of the contralateral lung 2 weeks after a VATS wedge resection. The patient had previously received 19 doses of nivolumab without evidence of respiratory compromise, and the most recent drug dose was administered nearly 1 month before the episode. In general, overall complication rates appear to be similar to those in other published series describing postinduction lung resection. For instance, a previous series from our institution, describing a cohort of 549 patients undergoing lung resection following chemotherapy or chemoradiation, identified an overall morbidity rate of 46% (16). This is statistically comparable to the overall complication rate in the current series (32%, p=0.26). Whereas 23% of the patients in that series experienced grade 3 or higher complications (compared with 5% in this series), a higher percentage of patients underwent anatomic resection (84% underwent lobectomy or greater). Overall morbidity for major resection (lobectomy or greater) in the current analysis was 41.7%. Although individual complications are difficult to compare, due to their infrequency, the rate of prolonged air leak in our previous study was 7%, which is comparable to that in the current series (9%). Similarly, Petersen et al. reported on 97 patients undergoing lobectomy following induction chemotherapy or chemoradiation (17). They noted rates of atrial fibrillation (10%) and pneumonia (8%) that are similar to those in the current analysis (5% for both).
The effect of immunotherapy on the technical aspects of lung resection, particularly anatomic resection, is difficult to quantitatively assess. Commonly used surrogates that are indicative of a difficult operation include operative time, blood loss, conversion to thoracotomy, and perioperative complications. In our series, the rate of complications was low, and blood loss was minimal in most cases. There was substantial variability in operative time, and our mean duration of 227 minutes for lobectomy is on par with the majority of postinduction cases in the literature. For instance, Glover et al. report an operative time for lobectomy of 264 minutes in patients who have undergone chemotherapy and 257 minutes in patients who have undergone chemoradiation, compared with 203 minutes in patients who did not receive induction treatment (18). In the current study, conversion to thoracotomy was required in 1 of the 5 anatomic resections attempted via a minimally invasive approach (20%). In this case (a robotic-assisted right upper lobectomy), dense hilar fibrosis necessitated conversion. In comparison, in the previously mentioned study from Petersen et al., 12 patients underwent postinduction VATS lobectomy, 1 of whom (8%) required conversion (17). It should also be noted that, of the 8 lobectomies in our series, 4 were performed through a planned thoracotomy, and the operative reports for several of these cases commented on dense adhesions at either the hilum or the chest wall. Review of operative notes and discussions among surgeons revealed fibrotic adhesions to be an issue in several instances. In one case involving a right upper lobectomy, the right upper lobe bronchus and truncus branch of the pulmonary artery were essentially fused and, despite a tedious attempt at separation, needed to be divided with a single staple fire. Although these extreme challenges appear to be rare, some degree of inflammatory adhesions was common, although not universal.
Complete (R0) resection was obtained in the vast majority of cases in our series, which underlines the feasibility and utility of resection in this population. Of note, 32% of patients selected for surgery had no viable tumor found in their final resection specimen. As clinical suspicion of residual disease likely affects selection for surgery, imaging characteristics or diagnostic biomarkers that allow for improved preoperative differentiation of persistent disease from posttreatment scarring would be of great clinical value.
One-year DFS in our cohort was 58%, and 42% remained disease free at 2 years. Although it is beyond the scope of this study to definitively state whether surgical resection alters the natural history of disease in this population, it is nonetheless reassuring that a substantial proportion of patients treated with this strategy do not experience rapid recurrence in additional metastatic sites. Long-term follow-up will be necessary to determine whether this disease control is durable.
The optimal timing from completion of checkpoint blockade to resection remains unclear. In the absence of data-driven algorithms, practice patterns in this study can provide some insight. Most patients in this study were heavily treated (mean doses before resection, 21), and the average time from completion of treatment to surgery was substantial (75 days). According to our data, patients were treated with checkpoint blockade until either their disease progressed through treatment, toxicity necessitated treatment cessation, or, in other instances, optimal radiographic response was achieved but imaging suggested the possibility of residual cancer. In the latter scenario, patients were often observed for a period to ensure they did not manifest additional metastatic sites before resection.
Criteria for identifying patients who would benefit from this approach also need to be clarified. In this series, patients with lung cancer generally had oligometastatic disease, and their metastatic sites outside the lung were clinically resolved following either systemic therapy alone or systemic therapy in combination with a local modality, such as stereotactic radiation or surgical resection. Patients undergoing resection for lung metastases from cancers such as melanoma generally had a small burden of disease limited to the lung. More-specific selection criteria will need to be elucidated in future studies.
There are several limitations to the current study. Although data were extracted from our prospectively maintained database, the study remains retrospective in nature and, therefore, subject to bias in regard to data collection. Second, although clinical situations regarding surgery following immunotherapy are increasing in frequency, the experience with surgery in this setting is still limited. Although this is, to our knowledge, the largest study to date on this topic in the literature, the number of patients in this analysis is relatively small. We would expect that, as additional prospective trials progress, particularly with regard to using these agents as planned induction therapy for resectable disease, the true incidence of morbidity and mortality will become clearer.
In conclusion, surgical resection following immunotherapy for previously metastatic or unresectable solid tumors appears to be safe, with mostly minor complications experienced in this small, retrospective cohort. Complete resection appears to be feasible in most cases, and patients selected for surgery have reasonable DFS and OS during short-interval follow-up. Operations may be challenging, owing to posttreatment adhesions, but, on the basis of the available data, this appears to be similar to treatment with other induction agents. We anticipate that additional information from ongoing trials, particularly in the neoadjuvant setting, will provide further insight.
Supplementary Material
Acknowledgments
This study was supported, in part, by the Memorial Sloan Kettering Cancer Center Support Grant/Core Grant (P30 CA008748). The funding source played no role in any aspect of the research or writing of the manuscript.
Footnotes
Presented at: The Southern Thoracic Surgery Association 64th Annual Meeting, San Antonio, TX, November 8–11, 2017.
Classifications: Lung Cancer, Immunotherapy, Lung resection, Surgery, Complications
COI statement: Dr. Bott is a consultant for AstraZeneca Pharmaceuticals. All other authors declare no conflicts.
References
- 1.Horn L, Spigel DR, Vokes EE et al. Nivolumab versus docetaxel in previously treated patients with advanced non-small-cell lung cancer: Two-year outcomes from two randomized, open-label, phase iii trials (checkmate 017 and checkmate 057). J Clin Oncol 2017: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Callahan MK, Kluger H, Postow MA et al. Nivolumab plus ipilimumab in patients with advanced melanoma: Updated survival, response, and safety data in a phase i dose-escalation study. J Clin Oncol 2017: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Brahmer J, Reckamp KL, Baas P et al. Nivolumab versus docetaxel in advanced squamous-cell non-small-cell lung cancer. N Engl J Med 2015;373(2):123–135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Carbone DP, Reck M, Paz-Ares L et al. First-line nivolumab in stage iv or recurrent non-small-cell lung cancer. N Engl J Med 2017;376(25):2415–2426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Borghaei H, Paz-Ares L, Horn L et al. Nivolumab versus docetaxel in advanced nonsquamous non-small-cell lung cancer. N Engl J Med 2015;373(17):1627–1639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Chaft JE, Hellmann MD, Velez MJ, Travis WD, Rusch VW. Initial experience with lung cancer resection after treatment with t-cell checkpoint inhibitors. Ann Thorac Surg 2017;104(3):e217–e218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Iwata T, Yamamoto A, Hara K. Complete response achieved 1 year after a single administration of nivolumab in a patient with lung adenocarcinoma with negative expression of pd-l 1. J Thorac Oncol 2017;12(12):e205–e207. [DOI] [PubMed] [Google Scholar]
- 8.Gyorki DE, Yuan J, Mu Z et al. Immunological insights from patients undergoing surgery on ipilimumab for metastatic melanoma. Ann Surg Oncol 2013;20(9):3106–3111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Antonia SJ, Villegas A, Daniel D et al. Durvalumab after chemoradiotherapy in stage iii non-small-cell lung cancer. N Engl J Med 2017;377(20):1919–1929. [DOI] [PubMed] [Google Scholar]
- 10.McCall NS, Dicker AP, Lu B. Beyond concurrent chemoradiation: The emerging role of pd-1/pd-l1 inhibitors in stage iii lung cancer. Clin Cancer Res 2018. [DOI] [PubMed] [Google Scholar]
- 11.Khunger M, Rakshit S, Pasupuleti V et al. Incidence of pneumonitis with use of programmed death 1 and programmed death-ligand 1 inhibitors in non-small cell lung cancer: A systematic review and meta-analysis of trials. Chest 2017;152(2):271–281. [DOI] [PubMed] [Google Scholar]
- 12.Hu YB, Zhang Q, Li HJ et al. Evaluation of rare but severe immune related adverse effects in pd-1 and pd-l1 inhibitors in non-small cell lung cancer: A meta-analysis. Transl Lung Cancer Res 2017;6(Suppl 1):S8–S20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Garon EB, Rizvi NA, Hui R et al. Pembrolizumab for the treatment of non-small-cell lung cancer. N Engl J Med 2015;372(21):2018–2028. [DOI] [PubMed] [Google Scholar]
- 14.Hellmann MD, Rizvi NA, Goldman JW et al. Nivolumab plus ipilimumab as first-line treatment for advanced non-small-cell lung cancer (checkmate 012): Results of an open-label, phase 1, multicohort study. Lancet Oncol 2017;18(1):31–41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Osorio JC, Ni A, Chaft JE et al. Antibody-mediated thyroid dysfunction during t-cell checkpoint blockade in patients with non-small-cell lung cancer. Ann Oncol 2017;28(3):583–589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Barnett SA, Rusch VW, Zheng J et al. Contemporary results of surgical resection of non-small cell lung cancer after induction therapy: A review of 549 consecutive cases. J Thorac Oncol 2011;6(9): 1530–1536. [DOI] [PubMed] [Google Scholar]
- 17.Petersen RP, Pham D, Toloza EM et al. Thoracoscopic lobectomy: A safe and effective strategy for patients receiving induction therapy for non-small cell lung cancer. Ann Thorac Surg 2006;82(1):214–218; [DOI] [PubMed] [Google Scholar]
- 18.Glover J, Velez-Cubian FO, Toosi K et al. Perioperative outcomes and lymph node assessment after induction therapy in patients with clinical n1 or n2 non-small cell lung cancer. J Thorac Dis 2016;8(8):2165–2174. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.