ABSTRACT
Emergence of drug resistance among the causative organisms for respiratory tract infections represents a critical challenge to the global health care community. Further, although vaccination can prevent disease, vaccine development is impeded by several factors. Therefore, novel approaches to treat and manage respiratory infections are urgently needed. Passive immunization represents a possible alternative to meet this need. Immunoglobulin Y antibodies (IgYs) from the yolk of chicken eggs have previously been used against bacterial and viral infections in human and animals. Their advantages include lack of reaction with mammalian Fc receptors, low production cost, and ease of extraction. Compared to mammalian IgGs, they have higher target specificity and greater binding avidity. They also possess remarkable pathogen-neutralizing activity in the respiratory tract and lungs. In this review, we provide an overview of avian IgYs and describe their potential therapeutic applications for the prevention and treatment of respiratory infections.
KEYWORDS: immunotherapy, passive immunization, IgY, respiratory infection
Introduction
Respiratory tract infections (RTIs) represent one of the most common illnesses encountered in clinical medicine. In developed countries, RTIs account for the majority of antibiotic prescriptions, more than 30% of lost work-days, and 20% of all medical consultations.1 About 1.3 million children under 5 years die from acute respiratory infections (ARI) worldwide every year2 and represents the cause of death in one third of children under five years in developing countries.3
Multidrug-resistant (MDR) or difficult-to-treat pathogens are an enormous challenge to the global health care community because of the high morbidity and mortality associated with them as well as the economic burden that they place on patients and the health care system. Antibiotics are a major tool in managing infections caused by these pathogens,4–8 but the emergence of antibiotic resistance has undermined their usefulness in some cases. Furthermore, a substantial proportion of respiratory infections are acute viral infections. The management of such infections includes use of antivirals and treatment to relieve disease symptoms. However, most antiviral agents target conserved viral proteins, which places selective pressure on the virus, leading to the development of antiviral resistance.9 Viral infections can be prevented by vaccination, but vaccine development faces multiple challenges, including antigenic variations in viral strains, low efficacy or short-term immune responses. In the case of outbreaks, rapid vaccine development may not be possible to control the spread of infection.10 Therefore, an urgent need exists to develop novel approaches to treat and manage the respiratory infections
Passive immunization offers an alternative approach for treating infections in immune-compromised individuals and avoiding any side effects that might result from vaccination.11 Antibody preparations used for passive immunization mainly contain polyclonal antibodies derived from the sera of immunized animals, immunized humans, and in some cases convalescing patients.12,13 The effective use of polyclonal antibodies faces several challenges, including standardization and patient safety. Monoclonal antibodies (mAbs) have been suggested as an alternative to polyclonal antibodies, but their use is limited by high production cost (about US$100/g).14 In addition, the possibility of viral escape mutants requires production of numerous mAbs targeting several antigens. This strategy would increase the cost and require additional efficacy and safety studies.15
Immunoglobulin (Ig)Ys are produced by chickens and other birds, reptiles, and amphibians. The function of IgYs is similar to that of mammalian IgGs.16 IgYs are present in the sera of chickens and are passed to the embryo through egg yolk.17 Egg IgY antibodies have previously been used against bacterial18 and viral infections.19
IgY is well tolerated because chicken eggs are a natural part of the human diet. In addition, it can be used in patients with egg allergies because the purified IgY does not contain egg albumin, the common trigger for allergic reactions to eggs.20 Moreover, the immunogenicity of IgY has been tested previously in both pigs and mice. Vega et al21 and Torche et al22have both demonstrated that administration of IgY to pigs via both systemic and local routes induced an anti-IgY antibody response, primarily consisting of the IgG subclass. These data suggest that IgY is antigenic and although the biochemical properties of this antibody molecule do not facilitate considerable binding to mammalian Fc receptors, serum sickness is a theoretical possibility if IgY is administered in large amounts. Whether or not IgY elicits an allergic response in pigs is unknown, however Akita et al23 demonstrated that administration of egg yolk containing IgY, purified IgY, and IgY Fab’ to mice failed to induce an IgE response. They further determined that there was very little cross reactivity between egg white protein, which is highly allergenic, and purified IgY.
Because IgY does not react with the human complement system or Fc receptors, the risk of further inflammation is minimal.24 IgY is thought to work by binding to the bacteria or viruses, leading to their elimination through the gut and prevention of bacterial replication or virus spread.25 In some cases, passive immunization using IgY antibodies has rapid and local onset of action, and can be given to patients with active infection. It can also be used in immature or impaired immune response, such as infants and immuno-compromised adults.26,27
An additional advantage of IgYs is their high content of sialic acid,28 which is reported to increase the half-life of the drug29 compared with those with lower sialic acid content. This finding suggests that IgY-based therapy could have a longer circulating half-life, which could increase its efficacy against infections.
The aim of this review is to highlight the potential use of specific IgYs in immunotherapy for the prevention and treatment of respiratory tract infections.
Passive immunization discovery and use
Passive immunization is the administration of preformed antibodies or immunoglobulins to treat various infectious diseases.24,25,30 Passive immunity can be either natural or acquired.24,31 Maternal antibodies transferred to the offspring deliver protection against pathogens, which represents natural passive immunity. In birds, the egg yolk is the reservoir of the circulating IgYs, which then enter the embryonic circulation of the developing embryo.32,33 The maternal IgYs provide protection for a very short time (about 2 weeks), and their levels in the chicks’ blood decrease rapidly after 1 week from hatching, when the young birds begin to synthesize their own antibodies.34
Passive immunity can also be artificially induced by transferring antibodies or serum derived from immune subjects to non-immune subjects by systemic, intravenous, or oral routes. Owing to the short-term nature of passive immunity, a continuous supply of the preformed antibodies is needed, which requires that they be produced on a large scale.35 Such production of large quantities of IgY antibodies from egg yolk can be achieved via hyperimmunization of chickens. The process includes the immunization of hens with specific antigens at regular intervals for continuous production of IgYs in the egg yolks. Extraction of these IgYs from egg yolks ensures a constant supply of the antibodies.36
Passive immunization was first introduced more than 100 years ago by Albert Calmette and others.37 The passive immunization principle has since been extensively used for treating and preventing diseases in humans and animals.31,38,39 An expected advantage of antibody-based treatments is the absence of off-target resistance or altered distribution of the host flora because the treatments target specific disease-causing pathogens.40
Intranasal antibody prophylaxis, which is based on the passive immunity principle, was reported to show promise against viral respiratory infections in animal models, including respiratory syncytial virus (RSV), influenza virus, and Sendai virus, and it was expected to show good efficacy in human clinical trials for influenza A and B viruses, Coxsackie virus, and rhinoviruses. However, studies showed low antiviral efficacy of the antibodies used and suggested that this result might be attributable to low levels of viral-specific antibodies in the immunoglobulin (Ig) preparations used.41
Immunoglobulin y antibodies
Evidence for maternal IgY transfer from chicken to egg yolk for embryo protection was first reported more than 100 years ago.42
Immunizing hens for the production of egg yolk IgY antibodies could permit efficient production of large quantities of antibodies (≈ 100 mg of total IgY/egg).24 These antibodies could in turn provide useful biological alternatives for specific antiviral therapy against several respiratory infections.43 IgYs are highly stable at pH 4–9 and up to 65°C in aqueous conditions, and they retain antigen-binding activity in the presence of pepsin at pH 4–6. These characteristics make them very good candidates for most types of processing and applications.43 The large-scale, relatively simply production of IgYs together with the ease of transportation and storage make them a good candidate for global therapeutic use in the time of a pandemic.44
Use of IgY antibodies has been an accepted practice since 1996,45 also, the Veterinary Office of the Swiss Government (Office Vétérinaire Fédéral) approved the practice in 1999.36 In humans, IgY has been shown to be effective for the prevention and treatment of acne and other dermatological infections.46 It has also been used against candidiasis, dental caries, and periodontitis of the oral cavity47–49; gastritis and Helicobacter pylori50,51; intestinal disorders such as celiac disease, cholera, and diarrhea52–54; metabolic syndrome55; and illness caused by environmental factors, such as norovirus, dust mites, and snake venom.56–58
Our group previously developed a reliable murine model for H. pylori infection.59 In addition, we developed IgY antibodies in chickens that target a pathogenic H. pylori strain and used these antibodies to treat infected mice. Passively immunized mice had a significantly lower degree of infection and gastritis than unimmunized animals.59
Oral administration of anti-H. pylori IgY can be used a complementary therapy combined with routine antibiotic therapy.60
Mode of action
Agglutination
IgYs cause agglutination of pathogens (virus, bacterial, fungal), leading to their immobilization, which facilitates their removal from the gut.61
Adherence-blockade
Inhibition of adhesion has been shown to be the main mechanism of IgY action against pathogens in vitro.62,63 Experiments in vivo demonstrated that IgY prevented Escherichia coli K88 from adhering to the intestinal mucus of piglets.62 IgY was also found to bind to exposed factors on the surface of gram-negative bacteria, such as fimbriae (or pili), flagella, lipopolysaccharides, and outer membrane proteins.25 Binding of the IgYs blocked or impaired the function of these growth-related components of the bacteria. It is also possible that specific IgY binding to bacteria could alter cellular signaling processes, leading to decreased toxin production and release.25
Opsonization followed by phagocytosis
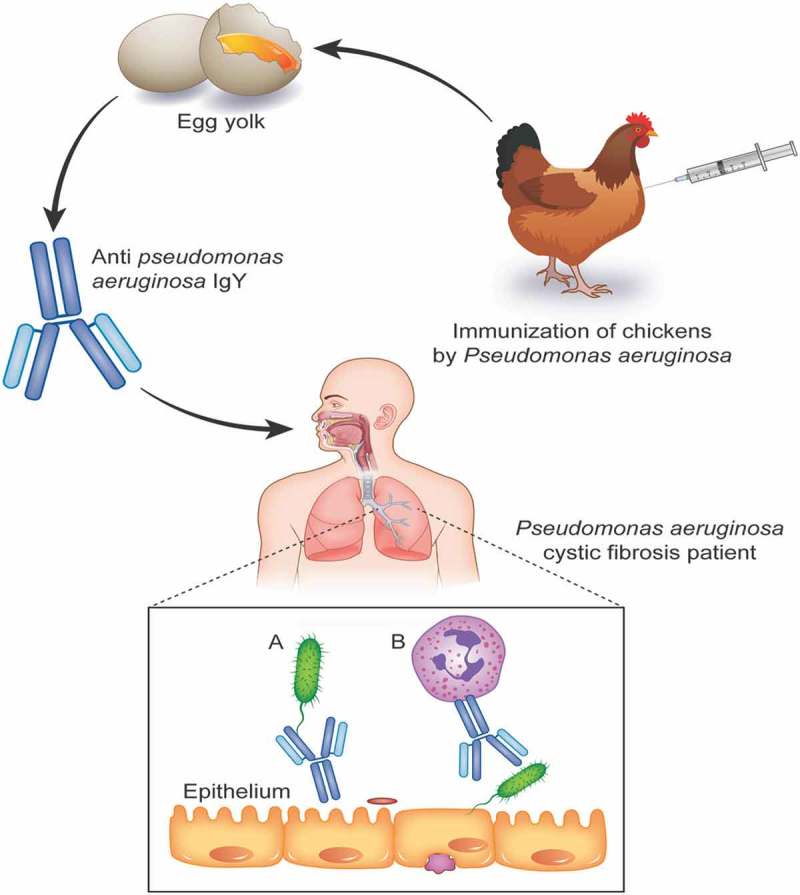
Several studies have suggested that IgY can enhance phagocytic activity against invading pathogens. For example, IgY improved phagocytosis of Staphylococcus aureus by neutrophils64 as well as the phagocytic activity against E. coli by milk macrophages and polymorphonuclear neutrophil leukocytes.65 Studies have also shown that binding of specific IgY on the surface of Salmonella typhimurium63 and E. coli O11165 leads to structural alterations that can be detected using electron cloud and/or electric field on the bacterial surface.63 Such changes can increase the susceptibility of bacterial cells to phagocytosis. A recent in vitro study showed that IgY opsonization of P. aeruginosa augments the PMN-mediated respiratory burst and subsequent bacterial elimination.66 The researchers found that viable bacteria are reduced by 87% when anti-P. aeruginosa IgY is used to opsonize the bacteria prior to phagocytosis (Figure 1A). Adding IgYs to a bacterial suspension caused bacterial clumping and immobilization within minutes of opsonization, as shown by indirect immunofluorescence. The addition of IgY enlarged the target geometry through bacterial aggregation, which augmented phagocytic efficiency.66
Figure 1.
Structural comparison between mammalian IgG and avian IgY.
Neutralization
IgY can prevent internalization of S. aureus by mammary epithelial cells, leading to neutralization of bacterial toxins.67 On the other hand, IgYs can inhibit cell-to-cell spread of virus particles, thus suppressing viral colonization.61
Advantages of passive immunization with IgY
IgY antibodies have numerous advantages.20 First, they can be naturally produced, avoiding environmental contamination because they are not synthetic drugs. In addition, they do not induce specific resistance because they are directed against multiple antigenic targets. Because IgY antibodies specifically target a particular pathogen, they do not affect the microbial population of the host. Consequently, they do not induce the unpleasant side effects that are typical of antibiotics. Finally, IgY antibodies are not deposited in meat, hence avoiding potential violations of regulations in countries that forbid the use of antibiotics for poultry and livestock.68
IgY antibodies used for passive immunotherapy also have advantages in addition to having local, rapid activity to the pathogen, they can be given to a wide range of individuals of any age, ranging from infants to adults and including immunodeficient patients and pregnant women.20
As natural components of eggs, the IgY antibodies tend to be nontoxic. Further, they can be stored for months in powder form at low moisture without the need for refrigeration, and they are cheaper and faster to produce than vaccines.61
Use of IgY antibodies also has general advantages in the context of immunotherapy. Egg collection is pain-free and animal friendly, which is beneficial from the perspective of animal welfare. Further, IgY has greater binding avidity to target antigens than mammalian IgG.69 IgY antibodies can also be produced against conserved mammalian proteins more easily and more successfully than IgG antibodies can be produced in other mammals due to evolutionary distance between mammals and birds.70 They also need lower antigen quantities to induce an efficient immune response.68 IgY can be stored in eggs for at least 1 year at 4°C.71 Large quantities of IgY antibodies can be produced by one chicken (about 22 g/year), and 2% to 10% of these antibodies are target specific,25,72 which exceeds the annual production from four rabbits.68 Furthermore, the production of eggs for human consumption is already carried out on an industrial scale, making low cost production of IgY in large quantities technically feasible.36,73
IgY structure
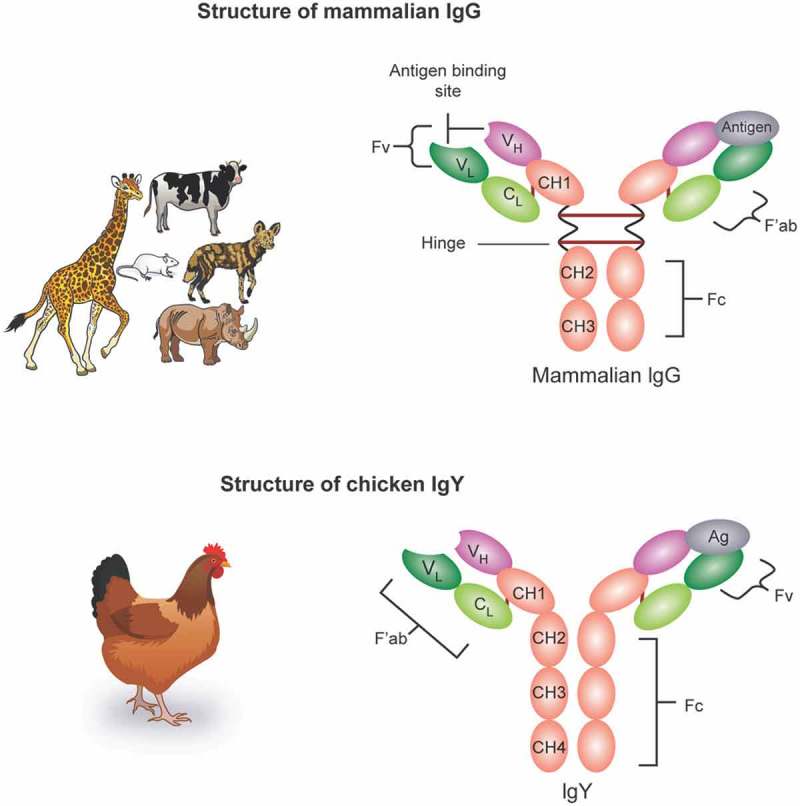
Chicken IgA and IgM are structurally and functionally analogous to mammalian IgA and IgM, while IgY differs from mammalian IgG despite their similar function. IgY is the predominant serum antibody in birds, reptiles, amphibians, and lungfish,16,74 and its serum concentrations far exceed those of IgA or IgM.33 IgYs are concentrated in the egg yolk, while IgA and IgM are found in the egg white. Although functional similarity exists between IgY and IgG, their structures are markedly different, as shown in (Figure 2).25
Figure 2.
Mode of action of Anti- Pseudomonas aeruginosa IgY from immunized chicken for protection of respiratory tract of cystic fibrosis patients. A) IgY binds to flagella of the Pseudomonas aeruginosa and inhibits the bacterial adhesion. B) IgY opsonization of Pseudomonas aeruginosa augments the PMN-mediated respiratory burst and subsequent bacterial elimination.
Among the differences, the H chain of IgYs have one variable region (VH) and four constant regions (CH) regions, while mammalian IgGs have only three CH regions. Also, IgY lacks a hinge region between the CH1 and CH2 domains, making it less flexible than IgG.75
Stability of IgY in the manufacturing processes
One of the most useful characteristics of IgYs is their stability during processing steps and under physiological conditions after administration.24 IgYs were found to be stable during storage at room temperature or at 4°C for 6 months without losing their activity.76 Furthermore, IgY antibodies can be stored for 5–10 years at 4°C without significant loss of their activity; they can even retain their activity after storage for 6 months at room temperature or for 1 month at 37°C.77 IgYs are preferably stored at −20°C for long-term storage; storage at −70°C can cause up to 50% loss of their activity.78 The stability of IgY is not affected by freeze-drying, although repeated freeze–thaw cycles might compromise its activity.79 One report80 showed that the thermal stability of IgYs was not affected by lyophilization at 90°C for 15 minutes. Long-term storage of IgY is better achieved by spray drying to avoid losing antibody potency.81
Acid stability of IgY antibodies is reported to be between pH 4 and 11, while activity decreases at lower pH values.75,82–85 The loss of activity at lower pH values might be due to conformational changes that distort the antigen-binding site.75,84 IgY antibodies are stable in alkaline conditions up to pH 11.0, while they lose activity at pH values ≥ 12.75,82
Safety
Safety is a major way in which IgY is superior to mammalian IgG. IgYs are safer than IgGs because they do not bind to human Fc receptors or fix mammalian complement components, hence they do not trigger potentially dangerous immune responses.17 Kubickova et al86 exposed immortalized human lung epithelial cells to IgY, using lipopolysaccharide as a positive control and phosphate-buffered saline as a negative control. Treatments also included exposure to human and goat IgG, and exposure lasted 24 hours for all treatments. The researchers found that the levels of pro-inflammatory cytokines tumor necrosis factor (TNF)-α, interleukin (IL)-1β, IL-6, and granulocyte-macrophage colony-stimulating factor (GM-CSF) were very low in cell cultures treated with IgY compared with the high levels of TNF-α and GM-CSF in cells treated with lipopolysaccharides, indicating that IgYs do not cause inflammatory responses in lung cells and can thus be safely used for prevention of airway infections.86 Additionally, oral IgY antibodies have been applied to treat rotavirus infections in humans54 and to treat pulmonary Pseudomonas aeruginosa infections, and no negative side effects of IgY treatment have been observed in up to 10 years of use.87
The safety of intranasal IgY delivery has been documented for the treatment of influenza virus19 and acute and chronic pharyngitis for human.88
Use of IgY for the treatment of respiratory infections
Influenza
Worldwide, influenza viruses cause 3–5 million hospitalizations and 250,000–500,000 deaths annually.89,90
Vaccination and antiviral drugs are the current approaches for the prevention and treatment for this infection.91 The long time needed to prepare a new influenza vaccine (about 6 months) for the new strains leave the population vulnerable to infection.92 The available antiviral drugs have limited efficacy because the virus can develop resistant variants with different active target binding sites. Consequently, the drugs are not effective when taken beyond the third day of infection.93
The rapid development of new treatments against influenza is a critical need that would intensify in a pandemic situation. Passive immunization has the potential to deliver prophylactic as well as therapeutic effects.12
Chicken IgYs have been tested for their use as passive immunotherapeutic or prophylactic agents against influenza viruses. Yang et al94 produced large quantities of egg yolk IgYs (9.18 mg/mL egg yolk) after 8 weeks immunization with an inactivated H1N1 virus. Hemagglutination inhibition and Western blotting assays showed the specific binding of the IgY antibodies that were produced to the hemagglutinin and neuraminidase of H1N1. Plaque reduction assays showed the reduction of H1N1 infection by IgY antibodies. In vivo studies in a mouse model showed that the anti-H1N1 IgYs provided protection against the virus by reducing the infectious titer of the virus in the lung, with no changes to the normal structure and weight of the mouse lung tissue. Besides the antiviral effect of the anti-H1N1 IgYs, the researchers also showed a protective effect comparable to that of the neuraminidase inhibitor oseltamivir. These results indicate that IgYs can provide a highly effective alternative approach for the treatment of influenza (Table 1).
Table 1.
Summary of studies performed to evaluate the efficacy of IgY antibodies against respiratory pathogens.
| Pathogen/antigen | in vitro/in vivo study | Human/animal model | Mode of IgY delivery | Effects of IgY | Reference |
|---|---|---|---|---|---|
| Influenza Virus | |||||
| H1N1 | in vivo | Mice | intranasal once a day for 4 days post-infection or 2 h prior to infection and once a day for four days post-infection | Protection against the virus by reducing the infectious titer of the virus in the lung comparable to that of the neuraminidase inhibitor | 95 |
| influenza B virus | in vivo | Mice | intranasal 2h before infection or 2, 26, 50, and 74 h post-infection. | reducing viral replication in the lungs | 43 |
| H1N1, H3N2, and H5N1 | in vivo | Mice | Intranasal administration of H5N1-specific IgYs 1 hour prior to infection Intranasal before and after lethal infection with H5N1 and H5N2 |
100% protection against lethal challenge with H5N1and protect against A/Puerto Rico/8/34 H1N1 Complete recover of the infection |
44 |
| H5N1, H1N1 | in vivo | Mice | Intranasal before or after lethal infection | Prevent the infection or significantly reduce viral replication resulting in complete recovery from the disease in mice | 96 |
| A/H1N1 2009 | In vitro | Inhibit the cytopathological effects of H1N1 | 97 | ||
| Pseudomonas aeruginosa | |||||
| In vitro | IgY opsonization augments the PMN-mediated respiratory burst and subsequent bacterial elimination | 66 | |||
| In vitro | Bind to the P. aeruginosa protein flagellin reducing the adherence of the bacteria to host cells of CF patients | 110 | |||
| in vivo | Mice | Intranasal 1hour before or 6 hours after infection | Significant reduction of bacterial load and inflammatory cytokines | 18 | |
| in vivo | human | Oral administration for more than 14 years | Decrease or prevent colonization | 87,104,111 | |
| Severe acute respiratory syndrome (SARS) | In vitro | IgY antibody was able to neutralize the SARS coronavirus | 80 | ||
| Mycobacterium tuberculosis (TB) | In vitro | Specific IgY against TB increased starting 2 weeks after first immunization and persist 200 days after last immunization | 120 | ||
| Bovine respiratory syncytial virus (BRSV) | In vitro | Purified IgY preparations, obtained from hens immunized with BRSV, neutralized BRSV in vitro | 125 |
The pathogen-specific titer of IgY typically starts to increase in eggs from the second week after immunization, peaking in the fifth week. Wen et al43 isolated egg yolk IgY against influenza B virus after immunization of the hens with an inactivated virus. The IgY yield was 76.5 mg per yolk, with a purity of 98.2%. The specific binding of the IgY to the viral proteins was demonstrated by Western blotting and hemagglutination inhibition test. The researchers used plaque reduction assays to demonstrate the efficacy of the specific IgY in neutralizing the influenza infection in MDCK cells. In vivo studies showed that intranasal treatment of mice prior to or after influenza B virus infection with virus-specific IgY had a protective effect by reducing viral replication in the lungs. The work of Wen et al showed that IgY specific to influenza B can readily provide a good alternative for influenza B prevention and treatment.43 Influenza-specific IgYs can be administered to humans (either intranasally or orally) and could serve as a quick and safe strategy in the fight against a pandemic influenza (Table 1).44
Pathogen-specific IgYs can remain in the sera and eggs of immunized hens for at least 2 months. In vitro investigation of IgYs produced from hens immunized with inactivated H1N1, H3N2, and H5N1 influenza viruses showed that the IgYs that were produced inhibited homologous as well as heterologous influenza viral strains.44 In vivo studies in a mouse model showed that intranasal administration of H5N1-specific IgYs 1 hour prior to infection had a 100% protection against lethal challenge with H5N1. Interestingly, IgY to H5N1 was found to also protect against A/Puerto Rico/8/34 H1N1.44 Another study found that intranasal administration of H5N1-specific IgYs in mice before and after lethal infection with H5N1 and H5N2 resulted in complete recover of the infection. Another interesting finding from this study was the presence of anti-H5N1 IgY antibodies in eggs bought directly from the market.19
Another study tested the efficacy of avian IgY neutralizing antibodies against the pandemic influenza virus A/H1N1. The antibodies were derived from ostrich eggs immunized with a swine influenza virus vaccine strain.95 The quantity of the IgY produced was very large and was found to have strong cross-reactivity with pandemic influenza H1N1 and swine influenza virus as shown by ELISA and immunocytochemistry. Ostrich IgY antibodies were found to inhibit the hemaggregation activity of erythrocytes that was induced by pandemic influenza A/H1N1 virus. The IgYs generated were found to inhibit the cytopathological effects of H1N1 on MDCK cells upon co-cultures with the antibodies, confirming the viral neutralization in the cells. The large amount of IgY generated from only one female ostrich makes this approach a cost-effective way to produce IgY antibodies against pandemic influenza virus A/H1N1(Table 1).
Pseudomonas aeruginosa in patients with cystic fibrosis (CF)
CF is a multisystem autosomal recessive disorder affecting approximately 70,000 people worldwide.96 CF is caused by dysfunction of the cystic fibrosis transmembrane conductance regulator (CFTR) protein. This CFTR dysfunction results in very thick secretions in the airways, leading to difficult mucociliary clearance.96 Pseudomonas aeruginosa is very common in CF patient, resulting in a decline in lung function with higher morbidity and mortality.97,98 Eradication of P. aeruginosa is difficult and patients usually experience chronic infection.4,99 A characteristic change in the bacteria during the course of infection is alginate production, which leads to greater difficulty in pathogen eradication.4,99 Antibiotic therapy is currently the best management approach in these cases.100 Early interventions targeting the bacterial colonization step can prevent the chronic stage from developing,66 while later interventions that fail to eradicate P. aeruginosa in CF patients will increase the risk of exacerbation.101
Binding of IgY to P. aeruginosa early in the course of infection prevents adhesion of the bacteria to oropharynx, which can prevent bacterial colonization.102 IgA in the respiratory mucosa appears to have a role in the initial host response by helping to reduce bacterial colonization.103 Consequently, CF patients with IgA deficiency could have greater susceptibility to bacterial colonization.104 Passive immunization with IgY could prophylactically augment mucosal IgA immunity, thus increasing the resistance to P. aeruginosa colonization.105
IgY was found to induce rapid and competent bacterial clearance in a murine pneumonia model.18 This study found that the pulmonary bacterial load in anti-P. aeruginosa IgY-treated mice was lower than in the controls by more than 2-log after 24 hours of infection. The IgY was found to induce a rapid decline in the bacterial load within the first hours of infection. The IgY-treated mice had a better clinical state compared to controls, which may be attributable to a lack of disseminated infection since anti-P. aeruginosa IgY protected against bacteremia.18 Production of inflammatory cytokines accompanied the faster bacterial clearance in treated mice. IL-1β and TNF-α, potent mediators of inflammation, were significantly reduced in anti-P. aeruginosa IgY-treated mice compared to controls after 24 hours of infection. Accordingly, the PMN mobilizer granulocyte-colony stimulating factor (G-CSF) and the PMN chemoattractant and murine IL-8 analog macrophage inflammatory protein-2 were significantly decreased at 24 hours post infection in the anti-P. aeruginosa IgY-treated group compared to controls, suggesting a moderation of neutropoiesis consistent with reduced numbers of the bacteria. Lung pathology caused by P. aeruginosa was reduced by the antibacterial activity of IgY immunotherapy (Table 1).18
The prophylactic effects of intranasal administration of IgY antibodies were more notable than intranasal spray 6 hours post infection. The superiority of the prophylactic treatment could have been due to the presence of IgY antibodies in the airways, which prepared the mucosal surface for the opsonophagocytic process and reduced its interaction with the IgY opsonized pathogens.105
Anti-Pseudomonas IgY was shown to bind the P. aeruginosa protein flagellin, which is the main component of the flagellum and is required for the motility and chemotaxis of the bacteria106 as well as the invasion and establishment of infection in the host.107 The activity of anti-Pseudomonas IgY was studied using 2D electrophoresis of P. aeruginosa strains, immunoblotting, and MALDI-TOF-MS. The results revealed that IgY had antibacterial immunoreactivity against all of the studied strains, increasing its potential for use as a prophylactic therapy.108 The researchers found that the binding of IgY with flagellin hindered host invasion by reducing the adherence of the bacteria to host cells of CF patients (Figure 1A). They concluded that IgY might directly hinder adherence or indirectly reduce motility. The binding of IgY to flagellin might reduce the inflammatory response to P. aeruginosa in CF patients (Table 1).108
Clinical evaluations
In a Phase I feasibility study, CF patients were asked to gargle with an antipseudomonal IgY antibody every night after brushing their teeth. This practice prolonged the time from first to next colonization. The specific antipseudomonal IgY prophylaxis experiments showed that 4.4% of the cultures were positive for P. aeruginosa, but none of the patients became chronically colonized. However, in the control group, in which 18.7% of the cultures were positive, 5 of 21 patients became chronically colonized.109 Another study demonstrated that oral administration of IgY against P. aeruginosa prevented pulmonary P. aeruginosa infections in patients with CF. None of the IgY-treated patients in this study became chronically colonized with P. aeruginosa, compared to 24% in the control group. No undesirable side effects were reported for the IgY treatment during the 10-year study period (Table 1).87
Nilsson et al102 put 17 Swedish CFs patients on oral prophylactic therapy with IgY antibodies against P. aeruginosa for up to 12 years. During the course of the study, continuous administration of azithromycin was used in several of the patients. Only 29 cultures tested positive for P. aeruginosa (not including cultures after chronic colonization) in the antibiotic-treated group (P = 0.028). In the IgY group (n = 17), only two siblings were chronically colonized with P. aeruginosa compared to seven patients in the control group (n = 23). There was no decrease in pulmonary function or the body mass index in the IgY-treated patients. These results suggest that the combined use of IgY and antibiotics for the treatment and prophylaxis of P. aeruginosa have the potential to prevent P. aeruginosa colonization and delay or avoid chronic P. aeruginosa infection (Table 1). From this study, Anti-Pseudomonas IgY has the possibility to be complement to antibiotics, when they are insufficient for the treatment of P. aeruginosa infections in CF lungs.
PMN phagocytosis is a fundamental determinant of an appropriate innate immune response against bacterial infections. Dysfunction in this pathway cause excessive bacterial colonization, leading to parenchymal infection that might develop into sepsis.105 IgY was observed to be more hydrophobic than mammalian IgG.75 This hydrophobicity helps the Fc portion to be oriented opposite the antigen, exposing it to Fc-receptor interaction. This feature of increased surface hydrophobicity of the IgY suggests increased phagocytic killing of P. aeruginosa through opsonization (Figure 1B) (Table 1).66
A recent clinical study investigating the use of polyclonal anti-Pseudomonas IgY antibodies to prevent P. aeruginosa recurrence in CF patients is underway (ClinicalTrials.gov identifier NCT01455675).110
Severe acute respiratory syndrome
Coronaviruses (CoVs) were only known to cause the common cold in vertebrates until 2002, when severe acute respiratory syndrome (SARS)-CoV emerged in China.111 The disease rapidly spread worldwide, causing approximately 8000 infections with a 10% mortality rate.112,113
During the SARS outbreak in China, passive immunization using sera from recovered SARS patients showed positive results.80 Anti-SARS coronavirus IgYs were evaluated after isolation from egg yolk of pathogen-free chickens immunized with SARS coronavirus antigen.80 The generated IgY had a high purity and good biological activity, and IgY antibody was able to neutralize the SARS coronavirus at a dilution of up to 1:640. The results of the neutralization experiments showed high concordance with ELISA results. The efficiency of IgY was not altered after lyophilization, potentially making transport and handling easy for passive immunization and short-term protection.80 After evaluation using experimentally infected animal models, anti-SARS IgY produced in this study might be a good candidate for mass production as a SARS-CoV immunotherapeutic (Table 1).
In case of outbreaks, there is a need for a rapid intervention. While the production of vaccines take a longer time than the production of IgY antibodies(about 6 weeks44 from hens vaccination to IgY production), the use of IgY Abs in outbreaks instead of vaccines is quite unlikely most of the time. Fu et al80 reported that, a combination of vaccine, passive immunizations and drug therapy will be required for effective control of SARS.
Mycobacterium tuberculosis
Tuberculosis is a major global concern in both human and animal populations. The disease is caused by a group of highly related intracellular pathogens, Mycobacterium tuberculosis complex (MTBC), that cause human morbidity and mortality worldwide.114 Although a third of the world’s population is latently infected with MTB, only 5–10% develop active tuberculosis (TB).115 In 2016, TB was the leading cause of death, with about 1.3 million human deaths.116 The combination of TB with HIV/AIDS and the development of multidrug-resistant strains have increased the disease burden.117 Immunotherapy might present a new option for treating drug-resistant TB strains, with promising outcomes and better quality of life for patients.118 Intramuscular immunization of Lohmann laying hens showed an increase of anti-MTBC IgY antibodies in egg yolk, with peak concentration at 4 weeks after immunization. The specific antibodies were persist 200 days after the last immunization. The study suggested that, specific anti-MTBC IgY could be an effective approach as immunotherapy of TB (Table 1).
Bovine respiratory syncytial virus (BRSV)
BRSV is an enveloped, negative-stranded, nonsegmented RNA virus that is the main cause of respiratory disease in young calves.119 BRSV is closely related to human RSV (HRSV), which is the most common cause of lower respiratory tract infections in children worldwide. HRSV results in more than 3 million hospitalizations and an annual mortality rate of 66,000–199,000 in children under 5 years.120 It has no licensed vaccine or effective treatment.121 The similarity between the two viruses makes BRSV infection in calves a good animal model for studying HRSV.122 Purified IgY preparations, obtained from hens immunized with BRSV, neutralized BRSV in vitro. First, a group of hens were immunized with six doses of immunogen containing 105 TCID50/ml BRSV at days 0, 18, 32, 56, 85, and 106. The second group were immunized with two doses containing 107 TCID50/ml BRSV at days 0 and 42. Vaccines were prepared with Freund’s complete adjuvant for the first dose and Freund’s incomplete adjuvant for the following doses. In the first group, birds began to respond after the third immunization and antibody titers increased to the maximum after the fifth immunization. In the second group, the antibodies were detectable after the first immunization, and high titers were detected after the second immunization. Notably, only two doses were needed to induce specific neutralizing antibodies in the second group, reducing suffering and stress of birds caused by repeated inoculation.123 The specificity of the IgY obtained against BRSV in the second group, which presented the highest serum neutralizing antibody level, was evaluated by a dot blot assay. IgY antibodies were able to specifically recognize the virus at dilutions up to 1:20,480. Moreover, from the in vitro neutralization test, the highest neutralizing anti-IgY antibody titer was detected after the fifth immunization in the first group and after the second immunization in the second group. These findings suggest that the immune response could be improved by increasing the amount of the antigen used for inoculation. The study concluded that IgY technology is an attractive tool that could potentially be used for prophylaxis and/or treatment of respiratory disease caused by BRSV infection (Table 1).123
Conclusion/future perspectives
The promise of IgY antibodies underscores the need for continuing research to improve different aspects of production, including chicken immunization protocols and adjuvant use, IgY extraction techniques, and methods to increase antibody yield. Development of monoclonal IgY antibodies will combine the advantages of both mAbs and avian IgY. Chicken mAbs are antibodies derived from a single B-cell lineage that has undergone repetitive rounds of somatic hypermutation and clonal selection and are usually of the IgY isotype recognizing a single unique epitope.124 Thus, chicken mAbs tend to be more specific and of higher affinity compared to polyclonal antibodies counterparts.125 Genetically engineered single-chain fragment variable IgY (IgY-scFv) has been successfully generated based on phage display technologies by different research groups.26 Spillner et al126 have previously provided a comprehensive review on established chicken scFvs and their diagnostics and therapeutic applications. These results also open the field for new research for the development of IgY antibodies against other respiratory viruses.
Funding Statement
Authors are grateful for financial support from the King Abdulaziz City for Science and Technology (KACST), Riyadh, Saudi Arabia for providing the grant on MERS-CoV special program (project number 10-1).
Disclosure of potential conflicts of interest
No potential conflict of interest was reported by the authors.
References
- 1.Bryce J, Boschi-Pinto C, Shibuya K, Black RE.. WHO estimates of the causes of death in children. Lancet (London, England). 2005;365:1147–1152. doi: 10.1016/S0140-6736(05)71877-8. [DOI] [PubMed] [Google Scholar]
- 2.Qazi S, Aboubaker S, MacLean R, Fontaine O, Mantel C, Goodman T, Young M, Henderson P, Cherian T. Ending preventable child deaths from pneumonia and diarrhoea by 2025. Development of the integrated global action plan for the prevention and control of pneumonia and diarrhoea. Arch Dis Child. 2015;100(Suppl 1):S23–8. doi: 10.1136/archdischild-2013-305429. [DOI] [PubMed] [Google Scholar]
- 3.Ujunwa F, Ezeonu C. Risk factors for acute respiratory tract infections in under-five children in enugu Southeast Nigeria. Ann Med Health Sci Res. 2014;4:95–99. doi: 10.4103/2141-9248.126610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Mogayzel PJ Jr., Naureckas ET, Robinson KA, Brady C, Guill M, Lahiri T, Lubsch L, Matsui J, Oermann CM, Ratjen F, et al. Cystic Fibrosis Foundation pulmonary guideline. pharmacologic approaches to prevention and eradication of initial Pseudomonas aeruginosa infection. Ann Am Thorac Soc. 2014;11:1640–1650. doi: 10.1513/AnnalsATS.201404-166OC. [DOI] [PubMed] [Google Scholar]
- 5.Flume PA, Mogayzel PJ Jr., Robinson KA, Goss CH, Rosenblatt RL, Kuhn RJ, Marshall BC. Cystic fibrosis pulmonary guidelines: treatment of pulmonary exacerbations. Am J Respir Crit Care Med. 2009;180:802–808. doi: 10.1164/rccm.200812-1845PP. [DOI] [PubMed] [Google Scholar]
- 6.Griffith DE, Aksamit T, Brown-Elliott BA, Catanzaro A, Daley C, Gordin F, Holland SM, Horsburgh R, Huitt G, Iademarco MF, et al. An official ATS/IDSA statement: diagnosis, treatment, and prevention of nontuberculous mycobacterial diseases. Am J Respir Crit Care Med. 2007;175:367–416. doi: 10.1164/rccm.200604-571ST. [DOI] [PubMed] [Google Scholar]
- 7.Mogayzel PJ Jr., Naureckas ET, Robinson KA, Mueller G, Hadjiliadis D, Hoag JB, Lubsch L, Hazle L, Sabadosa K, Marshall B. Cystic fibrosis pulmonary guidelines. Chronic medications for maintenance of lung health. Am J Respir Crit Care Med. 2013;187:680–689. [DOI] [PubMed] [Google Scholar]
- 8.Pasteur MC, Bilton D, At H. British thoracic Soc B. British thoracic society guideline for non-CF bronchiectasis. Thorax. 2010;65:I1–I58. doi: 10.1136/thx.2010.136119. [DOI] [PubMed] [Google Scholar]
- 9.McCaskill JL, Ressel S, Alber A, Redford J, Power UF, Schwarze J, Dutia BM, Buck AH. Broad-spectrum inhibition of respiratory virus infection by MicroRNA mimics targeting p38 MAPK signaling. Mol Ther Nucl Acids. 2017;7:256–266. doi: 10.1016/j.omtn.2017.03.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Corti D, Passini N, Lanzavecchia A, Zambon M. Rapid generation of a human monoclonal antibody to combat Middle East respiratory syndrome. J Infect Public Health. 2016;9:231–235. doi: 10.1016/j.jiph.2016.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Sparrow E, Friede M, Sheikh M, Torvaldsen S. Therapeutic antibodies for infectious diseases. Bull World Health Organ. 2017;95:235–237. doi: 10.2471/BLT.16.178061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Keller MA, Stiehm ER. Passive immunity in prevention and treatment of infectious diseases. Clin Microbiol Rev. 2000;13:602–614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Graham BS, Ambrosino DM. History of passive antibody administration for prevention and treatment of infectious diseases. Curr Opin HIV AIDS. 2015;10:129–134. doi: 10.1097/COH.0000000000000154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Kelley B. Industrialization of mAb production technology: the bioprocessing industry at a crossroads. mAbs. 2009;1:443–452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Casadevall A, Dadachova E, Pirofski LA. Passive antibody therapy for infectious diseases. Nat Rev Microbiol. 2004;2:695–703. doi: 10.1038/nrmicro974. [DOI] [PubMed] [Google Scholar]
- 16.Warr GW, Magor KE, Higgins DA. IgY: clues to the origins of modern antibodies. Immunol Today. 1995;16:392–398. doi: 10.1016/0167-5699(95)80008-5. [DOI] [PubMed] [Google Scholar]
- 17.Carlander D, Stalberg J, Larsson A. Chicken antibodies: a clinical chemistry perspective. Ups J Med Sci. 1999;104:179–189. [DOI] [PubMed] [Google Scholar]
- 18.Thomsen K, Christophersen L, Bjarnsholt T, Jensen PO, Moser C, Hoiby N. Anti-Pseudomonas aeruginosa IgY antibodies augment bacterial clearance in a murine pneumonia model. J Cyst Fibr. 2016;15:171–178. doi: 10.1016/j.jcf.2015.08.002. [DOI] [PubMed] [Google Scholar]
- 19.Nguyen HH, Tumpey TM, Park HJ, Byun Y-H, Tran LD, Nguyen VD, Kilgore PE, Czerkinsky C, Katz JM, Seong BL, et al. Prophylactic and therapeutic efficacy of avian antibodies against influenza virus H5N1 and H1N1 in mice. PLoS One. 2010;5:e10152. doi: 10.1371/journal.pone.0010152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Rahman S, Van Nguyen S, Icatlo FC Jr., Umeda K, Kodama Y. Oral passive IgY-based immunotherapeutics: a novel solution for prevention and treatment of alimentary tract diseases. Hum Vaccin Immunother. 2013;9:1039–1048. doi: 10.4161/hv.23383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Vega CG, Bok M, Vlasova AN, Chattha KS, Fernández FM, Wigdorovitz A, Parreño VG, Saif LJ, Salmon H. IgY antibodies protect against human Rotavirus induced diarrhea in the neonatal gnotobiotic piglet disease model. PLoS One. 2012;7:e42788. doi: 10.1371/journal.pone.0042788. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Torche AM, Le Dimna M, Le Corre P, Mesplede A, Le Gal S, Cariolet R, Le Potier MF. Immune responses after local administration of IgY loaded-PLGA microspheres in gut-associated lymphoid tissue in pigs. Vet Immunol Immunopathol. 2006;109:209–217. doi: 10.1016/j.vetimm.2005.08.016. [DOI] [PubMed] [Google Scholar]
- 23.Akita E, Jang C, Kitts D, Nakai S. Evaluation of allergenicity of egg yolk immunoglobulin Y and other egg proteins by passive cutaneous anaphylaxis. Food Agric Immunol. 1999;11:191–201. doi: 10.1080/09540109999861. [DOI] [Google Scholar]
- 24.Kovacs-Nolan J, Mine Y. Egg yolk antibodies for passive immunity. Annu Rev Food Sci Technol. 2012;3:163–182. doi: 10.1146/annurev-food-022811-101137. [DOI] [PubMed] [Google Scholar]
- 25.Xu Y, Li X, Jin L, Zhen Y, Lu Y, Li S, You J, Wang L. Application of chicken egg yolk immunoglobulins in the control of terrestrial and aquatic animal diseases: a review. Biotechnol Adv. 2011;29:860–868. doi: 10.1016/j.biotechadv.2011.07.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Zhang X, Calvert RA, Sutton BJ, Dore KA. IgY: a key isotype in antibody evolution. Biol Rev Camb Philos Soc. 2017;92:2144–2156. doi: 10.1111/brv.12325. [DOI] [PubMed] [Google Scholar]
- 27.Muller S, Schubert A, Zajac J, Dyck T, Oelkrug C. IgY antibodies in human nutrition for disease prevention. Nutr J. 2015;14:109. doi: 10.1186/s12937-015-0067-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Gilgunn S, Millan Martin S, Wormald MR, Zapatero-Rodríguez J, Conroy PJ, O’Kennedy RJ, Rudd PM, Saldova R, Mondelli MU. Comprehensive N-Glycan profiling of avian immunoglobulin Y. PLoS One. 2016;11:e0159859. doi: 10.1371/journal.pone.0159859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Liu L. Antibody glycosylation and its impact on the pharmacokinetics and pharmacodynamics of monoclonal antibodies and Fc-fusion proteins. J Pharm Sci. 2015;104:1866–1884. doi: 10.1002/jps.24444. [DOI] [PubMed] [Google Scholar]
- 30.Casadevall A, Scharff MD. Return to the past: the case for antibody-based therapies in infectious diseases. Clin Infect Dis. 1995;21:150–161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Baxter D. Active and passive immunity, vaccine types, excipients and licensing. Occup Med (Lond). 2007;57:552–556. doi: 10.1093/occmed/kqm110. [DOI] [PubMed] [Google Scholar]
- 32.Patterson R, Youngner JS, Weigle WO, Dixon FJ. The metabolism of serum proteins in the hen and chick and secretion of serum proteins by the ovary of the hen. J Gen Physiol. 1962;45:501–513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Rose ME, Orlans E, Buttress N. Immunoglobulin classes in the hen’s egg: their segregation in yolk and white. Eur J Immunol. 1974;4:521–523. doi: 10.1002/eji.1830040715. [DOI] [PubMed] [Google Scholar]
- 34.Hamal KR, Burgess SC, Pevzner IY, Erf GF. Maternal antibody transfer from dams to their egg yolks, egg whites, and chicks in meat lines of chickens. Poult Sci. 2006;85:1364–1372. doi: 10.1093/ps/85.8.1364. [DOI] [PubMed] [Google Scholar]
- 35.Chalghoumi R, Marcq C, Thewis A, Portetelle D, Beckers Y. Effects of feed supplementation with specific hen egg yolk antibody (immunoglobin Y) on Salmonella species cecal colonization and growth performances of challenged broiler chickens. Poult Sci. 2009;88:2081–2092. doi: 10.3382/ps.2009-00173. [DOI] [PubMed] [Google Scholar]
- 36.Gadde U, Rathinam T, Lillehoj HS. Passive immunization with hyperimmune egg-yolk IgY as prophylaxis and therapy for poultry diseases–A review. Animal Health Res Rev. 2015;16:163–176. doi: 10.1017/S1466252315000195. [DOI] [PubMed] [Google Scholar]
- 37.Calmette A. The treatment of animals poisoned with snake venom by the injection of antivenomous serum. Br Med J. 1896;2:399–400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Eibl MM. History of immunoglobulin replacement. Immunol Allergy Clin North Am. 2008;28:737–64, viii. doi: 10.1016/j.iac.2008.06.004. [DOI] [PubMed] [Google Scholar]
- 39.Hsu JL, Safdar N. Polyclonal immunoglobulins and hyperimmune globulins in prevention and management of infectious diseases. Infect Dis Clin North Am. 2011;25:773–788. doi: 10.1016/j.idc.2011.07.005. [DOI] [PubMed] [Google Scholar]
- 40.Casadevall A, Dadachova E, Pirofski L. Passive antibody therapy for infectious diseases. Nat Rev Microbiol. 2004;2:695–703. doi: 10.1038/nrmicro974. [DOI] [PubMed] [Google Scholar]
- 41.Weltzin R, Monath TP. Intranasal antibody prophylaxis for protection against viral disease. Clin Microbiol Rev. 1999;12:383-+. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Klemperer F. Ueber natürliche Immunitätund ihre Verwerthung für die immunisirungstherapie. Archiv Für Experimentelle Pathologie Und Pharmakologie. 1893;31:356–382. [Google Scholar]
- 43.Wen JL, Zhao SQ, He DG, Yang YN, Li YM, Zhu SS. Preparation and characterization of egg yolk immunoglobulin Y specific to influenza B virus. Antiviral Res. 2012;93:154–159. doi: 10.1016/j.antiviral.2011.11.005. [DOI] [PubMed] [Google Scholar]
- 44.Wallach MG, Webby RJ, Islam F, Walkden-Brown S, Emmoth E, Feinstein R, Gronvik K-O. Cross-protection of chicken immunoglobulin Y antibodies against H5N1 and H1N1 viruses passively administered in mice. Clin Vaccine Immunol. 2011;18:1083–1090. doi: 10.1128/CVI.05075-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Schade R, Hlinak A. Egg yolk antibodies, state of the art and future prospects. Altex. 1996;13:5–9. [PubMed] [Google Scholar]
- 46.Selvan K, Sentila R, Michael A. Generation and characterization of chicken egg yolk antibodies against propionibacterium acnes for the prevention of acne vulgaris. Indian J Dermatol. 2012;57:15–19. doi: 10.4103/0019-5154.92669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Ibrahim El SM, Rahman AK, Isoda R, Umeda K, Van Sa N, Kodama Y. vitro and in vivo effectiveness of egg yolk antibody against Candida albicans (anti-CA IgY). Vaccine. 2008;26:2073–2080. doi: 10.1016/j.vaccine.2008.02.046. [DOI] [PubMed] [Google Scholar]
- 48.Nguyen SV, Icatlo FC Jr., Nakano T, Isogai E, Hirose K, Mizugai H, Kobayashi-Sakamoto M, Isogai H, Chiba I. Anti-cell-associated glucosyltransferase immunoglobulin Y suppression of salivary mutans streptococci in healthy young adults. J Am Dent Assoc. 2011;142:943–949. [DOI] [PubMed] [Google Scholar]
- 49.Yokoyama K, Sugano N, Shimada T, Shofiqur RAKM, Ibrahim E-SM, Isoda R, Umeda K, Sa NV, Kodama Y, Ito K. Effects of egg yolk antibody against Porphyromonas gingivalis gingipains in periodontitis patients. J Oral Sci. 2007;49:201–206. [DOI] [PubMed] [Google Scholar]
- 50.Suzuki H, Nomura S, Masaoka T, Goshima H, Kamata N, Kodama Y, Ishii H, Kitajima M, Nomoto K, Hibi T. Effect of dietary anti-Helicobacter pylori-urease immunoglobulin Y on Helicobacter pylori infection. Aliment Pharmacol Ther. 2004;20(Suppl 1):185–192. doi: 10.1111/apt.2004.20.issue-s1. [DOI] [PubMed] [Google Scholar]
- 51.Hong KS, Ki MR, Ullah HMA, Lee E-J, Kim YD, Chung M-J, Elfadl AK, Park J-K, Jeong K-S. Preventive effect of anti-VacA egg yolk immunoglobulin (IgY) on Helicobacter pylori-infected mice. Vaccine. 2018;36:371–380. doi: 10.1016/j.vaccine.2017.11.082. [DOI] [PubMed] [Google Scholar]
- 52.Gujral N, Lobenberg R, Suresh M, Sunwoo H. In-vitro and in-vivo binding activity of chicken egg yolk immunoglobulin Y (IgY) against gliadin in food matrix. J Agric Food Chem. 2012;60:3166–3172. doi: 10.1021/jf205319s. [DOI] [PubMed] [Google Scholar]
- 53.Hirai K, Arimitsu H, Umeda K, Yokota K, Shen L, Ayada K, Kodama Y, Tsuji T, Hirai Y, Oguma K. Passive oral immunization by egg yolk immunoglobulin (IgY) to Vibrio cholerae effectively prevents cholera. Acta Med Okayama. 2010;64:163–170. doi: 10.18926/AMO/40008. [DOI] [PubMed] [Google Scholar]
- 54.Rahman S, Higo-Moriguchi K, Htun KW, Taniguchi K, Icatlo FC, Tsuji T, Kodama Y, Van Nguyen S, Umeda K, Oo HN, et al. Randomized placebo-controlled clinical trial of immunoglobulin Y as adjunct to standard supportive therapy for rotavirus-associated diarrhea among pediatric patients. Vaccine. 2012;30:4661–4669. doi: 10.1016/j.vaccine.2012.04.091. [DOI] [PubMed] [Google Scholar]
- 55.Hirose M, Ando T, Shofiqur R, Umeda K, Kodama Y, Nguyen S, Goto T, Shimada M, Nagaoka S. Anti-obesity activity of hen egg anti-lipase immunoglobulin yolk, a novel pancreatic lipase inhibitor. Nutr Metab (Lond). 2013;10:70. doi: 10.1186/1743-7075-10-70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Dai YC, Wang YY, Zhang XF, Tan M, Xia M, Wu X-B, Jiang X, Nie J. Evaluation of anti-norovirus IgY from egg yolk of chickens immunized with norovirus P particles. J Virol Methods. 2012;186:126–131. doi: 10.1016/j.jviromet.2012.07.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Lee KE, Han BK, Han JY, Hong JY, Kim MN, Heo WI, Sohn MH, Kim KW, Kim KE. Production of egg yolk antibodies specific to house dust mite proteins. Yonsei Med J. 2014;55:999–1004. doi: 10.3349/ymj.2014.55.4.999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Lee CH, Lee YC, Liang MH, Leu S-J, Lin L-T, Chiang J-R, Yang -Y-Y. Antibodies against venom of the snake deinagkistrodon acutus. Appl Environ Microbiol. 2015;82:71–80. doi: 10.1128/AEM.02608-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Attallah AM, Abbas AT, Ismail H, Abdel-Raouf M, El-Dosoky I. Efficacy of passive immunization with IgY antibodies to a 58-kDa H. pylori antigen on severe gastritis in BALB/c mouse model. J Immunoassay Immunochem. 2009;30:359–377. doi: 10.1080/15321810903187922. [DOI] [PubMed] [Google Scholar]
- 60.Najdi S, Nikbakht Brujeni G, Sheikhi N, Chakhkar S. Development of anti-Helicobacter pylori immunoglobulins Y (IgYs) in quail. Iranian J Veteri Res. 2016;17:106–110. [PMC free article] [PubMed] [Google Scholar]
- 61.Thu HM, Myat TW, Win MM, Thant KZ, Rahman S, Umeda K, Nguyen SV, Icatlo FC, Higo-Moriguchi K, Taniguchi K, et al. Chicken egg yolk antibodies (IgY) for prophylaxis and treatment of rotavirus diarrhea in human and animal neonates: a concise review. Korean J Food Sci Animal Res. 2017;37:1–9. doi: 10.5851/kosfa.2017.37.1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Jin LZ, Baidoo SK, Marquardt RR, Frohlich AA. In vitro inhibition of adhesion of enterotoxigenic Escherichia coli K88 to piglet intestinal mucus by egg-yolk antibodies. FEMS Immunol Med Microbiol. 1998;21:313–321. [DOI] [PubMed] [Google Scholar]
- 63.Lee EN, Sunwoo HH, Menninen K, Sim JS. In vitro studies of chicken egg yolk antibody (IgY) against Salmonella enteritidis and Salmonella typhimurium. Poult Sci. 2002;81:632–641. doi: 10.1093/ps/81.5.632. [DOI] [PubMed] [Google Scholar]
- 64.Nie R, Wu D, Hu G, Zhang J, Yang H, Wen Z. Effect of specific egg yolk immunoglobulins on phagocytosis by neutrophils. Chin J Vet Med. 2004;12:23–25. [Google Scholar]
- 65.Zhen YH, Jin LJ, Guo J, Li X-Y, Lu Y-N, Chen J, Xu Y-P. Characterization of specific egg yolk immunoglobulin (IgY) against mastitis-causing Escherichia coli. Vet Microbiol. 2008;130:126–133. doi: 10.1016/j.vetmic.2007.12.014. [DOI] [PubMed] [Google Scholar]
- 66.Thomsen K, Christophersen L, Bjarnsholt T, Jensen PO, Moser C, Hoiby N. Anti-pseudomonas aeruginosa IgY antibodies induce specific bacterial aggregation and internalization in human polymorphonuclear neutrophils. Infect Immun. 2015;83:2686–2693. doi: 10.1128/IAI.02970-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Wang LH, Li XY, Jin LJ, You J-S, Zhou Y, Li S-Y, Xu Y-P. Characterization of chicken egg yolk immunoglobulins (IgYs) specific for the most prevalent capsular serotypes of mastitis-causing Staphylococcus aureus. Vet Microbiol. 2011;149:415–421. doi: 10.1016/j.vetmic.2010.11.029. [DOI] [PubMed] [Google Scholar]
- 68.Li X, Wang L, Zhen Y, Li S, Xu Y. Chicken egg yolk antibodies (IgY) as non-antibiotic production enhancers for use in swine production: a review. J Anim Sci Biotechnol. 2015;6:40. doi: 10.1186/s40104-015-0038-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Ikemori Y, Peralta RC, Kuroki M, Yokoyama H, Kodama Y. Research note: avidity of chicken yolk antibodies to enterotoxigenic Escherichia coli fimbriae. Poult Sci. 1993;72:2361–2365. doi: 10.3382/ps.0722361. [DOI] [PubMed] [Google Scholar]
- 70.Gassmann M, Thommes P, Weiser T, Hubscher U. Efficient production of chicken egg yolk antibodies against a conserved mammalian protein. FASEB J. 1990;4:2528–2532. [DOI] [PubMed] [Google Scholar]
- 71.Jensenius JC, Andersen I, Hau J, Crone M, Koch C. Eggs: conveniently packaged antibodies. Methods for purification of yolk IgG. J Immunol Methods. 1981;46:63–68. [DOI] [PubMed] [Google Scholar]
- 72.Pauly D, Dorner M, Zhang X, Hlinak A, Dorner B, Schade R. Monitoring of laying capacity, immunoglobulin Y concentration, and antibody titer development in chickens immunized with ricin and botulinum toxins over a two-year period. Poult Sci. 2009;88:281–290. doi: 10.3382/ps.2008-00323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Sharma JM. Introduction to poultry vaccines and immunity. Adv Vet Med. 1999;41:481–494. [DOI] [PubMed] [Google Scholar]
- 74.Leslie GA, Clem LW. Phylogen of immunoglobulin structure and function. 3. Immunoglobulins of the chicken. J Exp Med. 1969;130:1337–1352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Shimizu M, Nagashima H, Sano K, Hashimoto K, Ozeki M, Tsuda K, Hatta H. Molecular stability of chicken and rabbit immunoglobulin G. Biosci Biotechnol Biochem. 1992;56:270–274. [DOI] [PubMed] [Google Scholar]
- 76.Nilsson E, Stalberg J, Larsson A. IgY stability in eggs stored at room temperature or at +4 degrees C. Br Poult Sci. 2012;53:42–46. doi: 10.1080/00071668.2011.646951. [DOI] [PubMed] [Google Scholar]
- 77.Larsson A, Balow RM, Lindahl TL, Forsberg PO. Chicken antibodies: taking advantage of evolution–a review. Poult Sci. 1993;72:1807–1812. doi: 10.3382/ps.0721807. [DOI] [PubMed] [Google Scholar]
- 78.Staak C, Schwarzkopf C, Behn I, Hommel U, Hlinak A, Schade R, Erhard M. Isolation of IgY from Yolk In: Schade R, Behn I, Erhard M, Hlinak A, Staak C, editors Chicken egg yolk antibodies, production and application: IgY-technology. Berlin (Heidelberg): Springer Berlin Heidelberg; 2001. p. 65–107. [Google Scholar]
- 79.Shimizu M, Fitzsimmons RC, Nakai S. Anti-E. coli lmmunoglobulin Y isolated from egg yolk of immunized chickens as a potential food ingredient. J Food Sci. 1988;53:1360–1368. doi: 10.1111/jfds.1988.53.issue-5. [DOI] [Google Scholar]
- 80.Fu CY, Huang H, Wang XM, Liu Y-G, Wang Z-G, Cui S-J, Gao H-L, Li Z, Li J-P, Kong X-G. Preparation and evaluation of anti-SARS coronavirus IgY from yolks of immunized SPF chickens. J Virol Methods. 2006;133:112–115. doi: 10.1016/j.jviromet.2005.10.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Yokoyama H, Peralta RC, Diaz R, Sendo S, Ikemori Y, Kodama Y. Passive protective effect of chicken egg yolk immunoglobulins against experimental enterotoxigenic Escherichia coli infection in neonatal piglets. Infect Immun. 1992;60:998–1007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Losch U, Schranner I, Wanke R, Jurgens L. The chicken egg, an antibody source. Zentralblatt Fur Veterinarmedizin Reihe B J Vet Med Series B. 1986;33:609–619. [DOI] [PubMed] [Google Scholar]
- 83.Hatta H, Tsuda K, Akachi S, Kim M, Yamamoto T. Productivity and some properties of egg yolk antibody (IgY) against human rotavirus compared with rabbit IgG. Biosci Biotechnol Biochem. 1993;57:450–454. [DOI] [PubMed] [Google Scholar]
- 84.Shimizu M, Nagashima H, Hashimoto K. Comparative studies in molecular stability of immunoglobulin G from different species. Comp Biochem Phys B. 1993;106:255–261. [DOI] [PubMed] [Google Scholar]
- 85.Lee KA, Chang SK, Lee YJ, Lee JH, Koo NS. Acid stability of anti-Helicobacter pyroli IgY in aqueous polyol solution. J Biochem Mol Biol. 2002;35:488–493. [DOI] [PubMed] [Google Scholar]
- 86.Kubickova B, Majerova B, Hadrabova J, Noskova L, Stiborova M, Hodek P. Effect of chicken antibodies on inflammation in human lung epithelial cell lines. Neuro Endocrinol Lett. 2014;35(Suppl 2):99–104. [PubMed] [Google Scholar]
- 87.Nilsson E, Kollberg H, Johannesson M, Wejaker PE, Carlander D, Larsson A. More than 10 years’ continuous oral treatment with specific immunoglobulin Y for the prevention of Pseudomonas aeruginosa infections: a case report. J Med Food. 2007;10:375–378. doi: 10.1089/jmf.2006.214. [DOI] [PubMed] [Google Scholar]
- 88.Xie MQ, Meng YX, Li ZH, Li Y-Q, Zhang K-X, Zhao X-M, Xiong G-J, Chen Y-M. [Effect of specific immunoglobulin Y in the treatment of acute and chronic pharyngitis]. Zhonghua Er Bi Yan Hou Ke Za Zhi. 2004;39:112–115. [PubMed] [Google Scholar]
- 89.Lafond KE, Nair H, Rasooly MH, Valente F, Booy R, Rahman M, Kitsutani P, Yu H, Guzman G, Coulibaly D, et al. Global role and burden of influenza in pediatric respiratory hospitalizations, 1982-2012: a systematic analysis. PLoS Med. 2016;13:e1001977. doi: 10.1371/journal.pmed.1001977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Vos T, Barber RM, Bell B, Bertozzi-Villa A, Biryukov S, Bolliger I, Charlson F, Davis A, Degenhardt L, Dicker D, et al. Global, regional, and national incidence, prevalence, and years lived with disability for 301 acute and chronic diseases and injuries in 188 countries, 1990-2013: a systematic analysis for the global burden of disease study 2013. Lancet (London, England). 2015;386:743–800. doi: 10.1016/S0140-6736(15)60692-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Bassaganya-Riera J, Song R, Roberts PC, Hontecillas R. PPAR-gamma activation as an anti-inflammatory therapy for respiratory virus infections. Viral Immunol. 2010;23:343–352. doi: 10.1089/vim.2010.0016. [DOI] [PubMed] [Google Scholar]
- 92.Quigley E. Influenza therapies: vaccines and antiviral drugs. Drug Discov Today. 2006;11:478–480. doi: 10.1016/j.drudis.2006.04.010. [DOI] [PubMed] [Google Scholar]
- 93.Berry CM. Antibody immunoprophylaxis and immunotherapy for influenza virus infection: utilization of monoclonal or polyclonal antibodies?. Hum Vaccin Immunother. 2017;14(3):1–4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Yang YE, Wen JL, Zhao SQ, Zhang K, Zhou YL. Prophylaxis and therapy of pandemic H1N1 virus infection using egg yolk antibody. J Virol Methods. 2014;206:19–26. doi: 10.1016/j.jviromet.2014.05.016. [DOI] [PubMed] [Google Scholar]
- 95.Tsukamoto M, Hiroi S, Adachi K, Kato H, Inai M, Konishi I, Tanaka M, Yamamoto R, Sawa M, Handharyani E, et al. Antibodies against swine influenza virus neutralize the pandemic influenza virus A/H1N1. Mol Med Rep. 2011;4:209–214. doi: 10.3892/mmr.2011.410. [DOI] [PubMed] [Google Scholar]
- 96.Maselli DJ, Keyt H, Restrepo MI. Inhaled antibiotic therapy in chronic respiratory diseases. Int J Mol Sci. 2017;18:1062. doi: 10.3390/ijms18051062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Emerson J, Rosenfeld M, McNamara S, Ramsey B, Gibson RL. Pseudomonas aeruginosa and other predictors of mortality and morbidity in young children with cystic fibrosis. Pediatr Pulmonol. 2002;34:91–100. doi: 10.1002/ppul.10127. [DOI] [PubMed] [Google Scholar]
- 98.Nixon GM, Armstrong DS, Carzino R, Carlin JB, Olinsky A, Robertson CF, Grimwood K. Clinical outcome after early Pseudomonas aeruginosa infection in cystic fibrosis. J Pediatr. 2001;138:699–704. doi: 10.1067/mpd.2001.112897. [DOI] [PubMed] [Google Scholar]
- 99.Breidenstein EB, De La Fuente-Nunez C, Hancock RE. Pseudomonas aeruginosa: all roads lead to resistance. Trends Microbiol. 2011;19:419–426. doi: 10.1016/j.tim.2011.04.005. [DOI] [PubMed] [Google Scholar]
- 100.Gibson RL, Burns JL, Ramsey BW. Pathophysiology and management of pulmonary infections in cystic fibrosis. Am J Respir Crit Care Med. 2003;168:918–951. doi: 10.1164/rccm.200304-505SO. [DOI] [PubMed] [Google Scholar]
- 101.Doring G, Flume P, Heijerman H, Elborn JS. Treatment of lung infection in patients with cystic fibrosis: current and future strategies. J Cyst Fibr. 2012;11:461–479. doi: 10.1016/j.jcf.2012.10.004. [DOI] [PubMed] [Google Scholar]
- 102.Nilsson E, Larsson A, Olesen HV, Wejaker PE, Kollberg H. Good effect of IgY against Pseudomonas aeruginosa infections in cystic fibrosis patients. Pediatr Pulmonol. 2008;43:892–899. doi: 10.1002/ppul.20875. [DOI] [PubMed] [Google Scholar]
- 103.Quie PG. Lung defense against infection. J Pediatr. 1986;108:813–816. [DOI] [PubMed] [Google Scholar]
- 104.Brett MM, Ghoneim AT, Littlewood JM. Serum IgA antibodies against Pseudomonas aeruginosa in cystic fibrosis. Arch Dis Child. 1990;65:259–263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Thomsen K, Christophersen L, Jensen PO, Bjarnsholt T, Moser C, Hoiby N. Anti-Pseudomonas aeruginosa IgY antibodies promote bacterial opsonization and augment the phagocytic activity of polymorphonuclear neutrophils. Hum Vaccin Immunother. 2016;12:1690–1699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Honko AN, Mizel SB. Effects of flagellin on innate and adaptive immunity. Immunol Res. 2005;33:83–101. doi: 10.1385/IR:33:1:083. [DOI] [PubMed] [Google Scholar]
- 107.Prince A. Flagellar activation of epithelial signaling. Am J Respir Cell Mol Biol. 2006;34:548–551. doi: 10.1165/rcmb.2006-0022SF. [DOI] [PubMed] [Google Scholar]
- 108.Nilsson E, Amini A, Wretlind B, Larsson A. Pseudomonas aeruginosa infections are prevented in cystic fibrosis patients by avian antibodies binding Pseudomonas aeruginosa flagellin. J Chromatograp B, Anal Technol Biomed Life Sci. 2007;856:75–80. doi: 10.1016/j.jchromb.2007.05.029. [DOI] [PubMed] [Google Scholar]
- 109.Kollberg H, Carlander D, Olesen H, Wejaker PE, Johannesson M, Larsson A. Oral administration of specific yolk antibodies (IgY) may prevent Pseudomonas aeruginosa infections in patients with cystic fibrosis: a phase I feasibility study. Pediatr Pulmonol. 2003;35:433–440. doi: 10.1002/ppul.10290. [DOI] [PubMed] [Google Scholar]
- 110.Waters V, Smyth A. Cystic fibrosis microbiology: advances in antimicrobial therapy. J Cyst Fibr. 2015;14:551–560. doi: 10.1016/j.jcf.2015.02.005. [DOI] [PubMed] [Google Scholar]
- 111.Perlman S, Netland J. Coronaviruses post-SARS: update on replication and pathogenesis. Nat Rev Microbiol. 2009;7:439–450. doi: 10.1038/nrmicro2147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Rota PA, Oberste MS, Monroe SS, Nix WA, Campagnoli R, Icenogle JP, Penaranda S, Bankamp B, Maher K, Chen MH, et al. Characterization of a novel coronavirus associated with severe acute respiratory syndrome. Science (New York, NY). 2003;300:1394–1399. doi: 10.1126/science.1085952. [DOI] [PubMed] [Google Scholar]
- 113.Drosten C, Gunther S, Preiser W, Van Der Werf S, Brodt H-R, Becker S, Rabenau H, Panning M, Kolesnikova L, Fouchier RAM, et al. Identification of a novel coronavirus in patients with severe acute respiratory syndrome. N Engl J Med. 2003;348:1967–1976. doi: 10.1056/NEJMoa030747. [DOI] [PubMed] [Google Scholar]
- 114.Lechartier B, Rybniker J, Zumla A, Cole ST. Tuberculosis drug discovery in the post-post-genomic era. EMBO Mol Med. 2014;6:158–168. doi: 10.1002/emmm.201201772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Bhatt K, Salgame P. Host innate immune response to Mycobacterium tuberculosis. J Clin Immunol. 2007;27:347–362. doi: 10.1007/s10875-007-9084-0. [DOI] [PubMed] [Google Scholar]
- 116.World Health Organization Global Tuberculosis Report, 2016. Geneva, Switzerland: WHO; 2017. Accessed http://www.who.int/tb/publications/global_report/en/. [Google Scholar]
- 117.Wang XZ, Chen SS, Xu YJ, Zheng HJ, Xiao TY, Li YQ, Chen X, Huang MX, Zhang HF, Fang XJ, et al. Identification and evaluation of the novel immunodominant antigen Rv2351c from Mycobacterium tuberculosis. Emerg Microbes Infect. 2017;6(6):6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Sudjarwo SA, Eraiko K, Sudjarwo GW. Koerniasari. The potency of chicken egg yolk immunoglobulin (IgY) specific as immunotherapy to Mycobacterium tuberculosis infection. J Adv Pharm Technol Res. 2017;8:91–96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Stott EJ, Taylor G. Respiratory syncytial virus. Brief Rev Arch Virol. 1985;84:1–52. [DOI] [PubMed] [Google Scholar]
- 120.Nair H, Nokes DJ, Gessner BD, Dherani M, Madhi SA, Singleton RJ, O’Brien KL, Roca A, Wright PF, Bruce N, et al. Global burden of acute lower respiratory infections due to respiratory syncytial virus in young children: a systematic review and meta-analysis. Lancet (London, England). 2010;375:1545–1555. doi: 10.1016/S0140-6736(10)60206-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Taylor G. Animal models of respiratory syncytial virus infection. Vaccine. 2017;35:469–480. doi: 10.1016/j.vaccine.2016.11.054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Valarcher JF, Taylor G. Bovine respiratory syncytial virus infection. Vet Res. 2007;38:153–180. doi: 10.1051/vetres:2006053. [DOI] [PubMed] [Google Scholar]
- 123.Ferella A, Bellido D, Chacana P, Wigdorovitz A, Santos MJD, Mozgovoj MV. Chicken egg yolk antibodies against bovine respiratory syncytial virus neutralize the virus in vitro. Procedia Vaccinol. 2012;6:33–38. doi: 10.1016/j.provac.2012.04.006. [DOI] [Google Scholar]
- 124.Lipman NS, Jackson LR, Trudel LJ, Weis-Garcia F. Monoclonal versus polyclonal antibodies: distinguishing characteristics, applications, and information resources. ILAR J. 2005;46:258–268. [DOI] [PubMed] [Google Scholar]
- 125.Lee W, Syed Atif A, Tan SC, Leow CH. Insights into the chicken IgY with emphasis on the generation and applications of chicken recombinant monoclonal antibodies. J Immunol Methods. 2017;447:71–85. doi: 10.1016/j.jim.2017.05.001. [DOI] [PubMed] [Google Scholar]
- 126.Spillner E, Braren I, Greunke K, Seismann H, Blank S, Du Plessis D. Avian IgY antibodies and their recombinant equivalents in research, diagnostics and therapy. Biol J Int Assoc Biol Standard. 2012;40:313–322. doi: 10.1016/j.biologicals.2012.05.003. [DOI] [PMC free article] [PubMed] [Google Scholar]


