Abstract
Androgen deprivation therapy (ADT) has been the standard of care for the last 75 years in metastatic hormone sensitive prostate cancer (PCa). However, this approach is rarely curative. Recent clinical trials have demonstrated that ADT combined with other agents, notably docetaxel and abiraterone, lead to improved survival. The mechanisms surrounding this improved cancer outcomes are incompletely defined. The response of cancer cells to ADT includes apoptosis and cell death, but a significant fraction remains viable. Our laboratory has demonstrated both in vitro and in vivo that cellular senescence occurs in a subset of these cells. Cellular senescence is a phenotype characterized by cell cycle arrest, senescence-associated β-galactosidase (SA-β-gal), and a hypermetabolic state. Positive features of cellular senescence include growth arrest and immune stimulation, although persistence may release cytokines and growth factors that are detrimental. Senescent tumor cells generate a catabolic state with increased glycolysis, protein turnover and other metabolic changes that represent targets for drugs, like metformin, to be applied in a synthetic lethal approach. This review examines the response to ADT and the putative role of cellular senescence as a biomarker and therapeutic target in this context.
Keywords: Prostate cancer, Cellular senescence, Androgen deprivation therapy, Combination therapy, Synthetic lethal targeting, Metformin, Statins
1. Introduction
Prostate cancer (PCa) is the most common male malignancy in the western world and is a leading cause of cancer death [1]. For the last 75 years, advanced disease has been managed with androgen deprivation therapy (ADT), an approach that leads to disease regression, but rarely cure. Castration-resistant PCa generally results in death over 30–40 months depending on disease extent at diagnosis [2]. Multiple phase III clinical trials (ChemoHormonal Therapy Versus Androgen Ablation Randomized Trial for Extensive Disease in Prostate Cancer [CHAARTED], A Randomized, Double-blind, Comparative Study of Abiraterone Acetate Plus Low-Dose Prednisone Plus Androgen Deprivation Therapy [ADT] Versus ADT Alone in Newly Diagnosed Subjects with High-Risk, Metastatic Hormone-naïve Prostate Cancer [mHNPC] [LATITUDE], Randomized Phase III Trial Comparing an Association of Hormonal Treatment and Docetaxel Versus the Hormonal Treatment Alone in Metastatic Prostate Cancers [GETUG-AFU 15], and Systemic Therapy in Advancing or Metastatic Prostate Cancer: Evaluation of Drug Efficacy [STAMPEDE]) have recently demonstrated that in metastatic hormone-sensitive PCa, combining either docetaxel chemotherapy or abiraterone (an androgen signaling inhibitor) at the time of initiating ADT markedly improves overall survival in men [3], [4], [5], [6]. These clinical observations suggest that ADT induces susceptibilities in PCa cells that make them amenable to synergistic treatment and improve cell killing. This underexplored area has the potential to have a major impact on PCa survival by improving the cancer response to ADT.
One phenotype that arises with extended replication or after cell stress is cellular senescence, in which cells are arrested yet viable [7]. Cellular senescence can also be induced after exposure to oxidative stress, DNA damaging agents, and ADT [8]. In breast cancer, another hormonally dependent cancer, senescence results after tamoxifen exposure in estrogen receptor positive cells [9]. Beneficial aspects of cellular senescence include a therapeutic arrest and immune stimulation. However, negative features include reentry of a subset of these persistent cells into the cell cycle and a secretory phenotype that may induce the growth of surrounding tumor cells. Thus, removal of these cells may lead to improved cancer outcomes.
The late Dr. Donald Coffey was always a strong supporter of “thinking outside the box”. His work led to remarkable advances in their own right, but placed in the context of his mentees he had an even greater impact of the field. Dr. Coffey often marveled at nature and would often state “You are going to be surprised at the simplicity and beauty of the real answer”. This review, dedicated to him, focuses on a surprising and major shift in our approach to advanced hormone sensitive PCa, that of the role of ADT and combined therapy. It is an area that requires significant research effort to understand and refine recent clinical discoveries. We will review cancer cell responses to ADT, seek to understand the clinical responses and mechanisms underlying these new combinatorial therapies with ADT, and discuss putative targeting of senescent cells after ADT as a therapeutic niche to improve outcomes.
2. Cancer cell responses to ADT
One of the best studied responses to ADT is apoptosis or programmed cell death. Kyprianou and colleague [10], [11] performed some of the early work that androgen deprivation leads to apoptosis in pre-clinical PCa models. Androgen withdrawal in murine xenografts and human PCa tissues is associated with a decrease in the proliferative index, but surprisingly low levels of apoptosis encompassing only 2%–3% of cells [12], [13], [14]. Apoptosis typically occurs early after ADT within the first 72 h [15]. In addition to apoptosis, other cell death mechanisms induced with ADT include autophagy, necrosis, and necroptosis [16].
Autophagy is an evolutionarily conserved catabolic pathway that targets cellular organelles and cytoplasmic constituents to the lysosomes for degradation. Autophagy, although a cell death pathway, can also function as a survival mechanism exploited by cancer cells under various physiological stresses [17], [18]. Androgen deprivation and tissue hypoxia, conditions which occur in prostate tissue after surgical or medical castration, lead to an increased adenosine monophosphate (AMP)-activated protein kinase (AMPK) activity in a threshold dependent manner in murine xenografts [19]. Increased AMPK activity is associated with greater cell survival and the induction of autophagy. In vitro in hormone-sensitive PCa cells, autophagy has been reported to occur after bicalutamide, but this occurs at much higher toxic doses than those seen with the induction of cellular senescence [20].
Some tumor cells become quiescent after ADT and have the potential to reactivate. PCa stem-like cells are typically androgen receptor (AR) negative and may represent part of the normally quiescent cancer stem cell population that emerges and expands after ADT [21]. These cells are rare, but express a specific surface antigen profile (CD44+/α2β1hi/CD133+) when isolated from primary PCa tissues and show high levels of clonogenic ability [22].
3. Cellular senescence as a response to ADT in PCa
The phenotype of these residual cells after ADT is complex, but cellular senescence represents an intriguing response that has potential for therapeutic exploitation. Replicative senescence was first described as a phenotype in primary cells after extensive culture and replicative exhaustion in vitro, which was linked to telomere shortening [23]. More recently, DNA damage, increased oncogenic signaling, and oxidative stress have been found to result in induced or accelerated senescence [24]. The propensity of tumor cells to undergo senescence induction in response to multiple anti-cancer therapies has been demonstrated [25]. Notably, multiple solid tumor cell lines have shown dose-dependent cellular senescence induction in response to DNA damaging agents such as doxorubicin, diaziquone, cisplatin, and to a lesser degree ionizing radiation. Recently reported data suggest enzalutamide, an antiandrogen, potentiates DNA damage caused by radiation and significantly increases radiation-induced cellular senescence in LNCaP cells [26].
Senescent cells remain viable and metabolically active, but are permanently growth-arrested (Table 1) [24]. They are persistent, in contrast to cells undergoing programmed responses including apoptosis, autophagy, and/or mitotic catastrophe using conventional cytotoxic agents. Growth arrest is achieved and maintained in either G1 or G2/M phage, in part, by the increased expression of specific cyclin-dependent kinase inhibitors (CDKIs), including p16Ink4a, p21Waf1/Cip1 and p27Kip1 [27]. Interestingly, transformed neoplastic cells that lack cellular senescence-associated tumor suppressor genes present in non-transformed cells (e.g., p53 and Rb) retain the capacity to become senescent with exposure to doxorubicin, docetaxel, and other chemotherapy agents [28], [29].
Table 1.
Summary of clinical trial results using ADT in combination with other therapies for the treatment of metastatic hormone sensitive PCa.
| Clinical trial | ADT+ | Number of patients | Follow-up (month) | HR 95% CI | Reference |
|---|---|---|---|---|---|
| GETUG-AFU 15a | Docetaxel | 385 | 50 | 1.01 (0.75–1.36) | Gravis et al., 2013 [40] |
| CHAARTEDb | Docetaxel | 790 | 28.9 | 0.61 (0.47–0.80) | Sweeney et al., 2015 [3] |
| LATITUDEc | Abiraterone | 1199 | 30.4 | 0.62 (0.51–0.76) | Fizazi et al., 2017 [4] |
| STAMPEDE-Docd | Docetaxel | 2962 | 43 | 0.78 (0.66–0.93) | James et al., 2016 [5] |
| STAMPEDE-Abie | Abiraterone | 1917 | 40 | 0.63 (0.52–0.76) | James et al., 2017 [6] |
PCa, prostate cancer; HR, hazard ratio; CI, confidential interval.
a Randomized Phase III Trial Comparing an Association of Hormonal Treatment and Docetaxel Versus the Hormonal Treatment Alone in Metastatic Prostate Cancers (GETUC-AFU 15).
b ChemoHormonal Therapy Versus Androgen Ablation Randomized Trial for Extensive Disease in Prostate Cancer (CHAARTED).
c A Randomized, Double-blind, Comparative Study of Abiraterone Acetate Plus Low-Dose Prednisone Plus Androgen Deprivation Therapy (ADT) Versus ADT Alone in Newly Diagnosed Subjects with High-Risk, Metastatic Hormone-naïve Prostate Cancer (mHNPC) (LATITUDE).
d Systemic Therapy in Advancing or Metastatic Prostate Cancer: Evaluation of Drug Efficacy (STAMPEDE) with Docetaxel (Doc).
e Systemic Therapy in Advancing or Metastatic Prostate Cancer: Evaluation of Drug Efficacy (STAMPEDE) with Abi (Abiraterone).
Cultured in vitro, senescent cells develop a distinct and recognizable flattened and enlarged morphology with a prominent nucleus and increased cytoplasmic granularity. Most notably, these cells can be visualized using a staining technique based on senescence-associated β-galactosidase (SA-β-gal) activity [30]. This technique, which stains lysosomes in the perinuclear compartment blue at pH 6.0, is a widely accepted and utilized marker of cellular senescence, but has limitations in vivo, as it is not applicable to formalin-fixed archival tissues.
To resolve this issue, we validated one of the first antibodies to SA-β-gal protein (GLB1) in vitro, subsequently allowing the biology of cellular senescence in PCa in vivo to be interrogated [31]. In hormonally intact prostate tissues, quantitative imaging detects increased GLB1 expression in high-grade prostatic intraepithelial neoplasia (HGPIN) known to contain senescent cells compared to benign prostate tissues [31]. This work also demonstrated that in intermediate grade PCa increased GLB1 predicts prostate-specific antigen (PSA)-free survival. Furthermore, senescent cells are found less commonly in high grade (Gleason score 8–10) versus intermediate grade (Gleason score 6–7) cancers. These findings support a tumor suppression aspect of cellular senescence seen in skin and many other aging organs.
These studies in our laboratory, and subsequently others, have demonstrated that cellular senescence is induced in androgen sensitive cells after ADT [32], [33]. Increased expression of the senescence-related proteins GLB1, the CDKI p27Kip1, and chromatin-regulating heterochromatin protein 1γ (HP1γ) are detected in 50%–80% of androgen sensitive LNCaP cells after being cultured in androgen-free media [8]. In mice bearing LuCaP xenograft tumors in vivo, surgical castration similarly increases senescent markers [8]. In another study, immunohistochemistry of human prostate tumors removed after ADT induced with goserelin acetate (Zoladex) showed a similar induction of GLB1, HP1γ, and decreased Ki-67 [34]. ADT induces a cellular growth arrest consistent with cellular senescence, including hypophosphorylation of Rb, reduction of cyclin-dependent kinase activity, and a G1/S block [27]. More recently, we have found that in patients undergoing ADT prior to prostate removal for cancer, elevated cellular GLB1 levels measured by VECTRA automated immunoquantitation are noted in as many as 50% of cancer cells [35]. GLB1 levels increase in tumors with longer ADT duration.
Despite permanent growth arrest, senescent cells are metabolically active and have increased energy demand, protein turnover, and glycolysis [36]. The increased energy demand is marked by activation of AMPK, a cellular energy sensor [37]. Increased glucose transport and amplified glycolytic enzyme and pyruvate kinase expression promote the utilization of glucose through non-oxidative glycolysis, similar to the Warburg phenomenon described in tumor cells [36]. Additionally, higher amino acid transport and increased protein synthesis generate cytokines that characterize the proinflammatory secretory-associated senescent phenotype (SASP). These metabolic changes present an opportunity for improved therapeutic approaches when combined with ADT.
Interestingly, AR signaling may play an important role in the cellular senescence response. Blocking the AR, or paradoxically applying supraphysiologic levels of androgens, such as those used in bipolar androgen therapy, may induce cellular senescence [38]. This may represent part of the biphasic growth response seen in AR expressing PCa cells, and induction of senescence in this context may function through the Src-P13K-Akt signaling pathway. Pharmacologic induction of senescence has also been demonstrated in murine xenografts and human PCa tissues using the naturally occurring AR antagonist atraric acid [39].
4. Clinical trials suggest unique PCa susceptibilities after ADT
The application of ADT has always been approached as the sole systemic therapy for metastatic hormone sensitive PCa, traditionally using agents that target the substrate (testosterone) or block the AR. In a major paradigm shift, several recent trials using either docetaxel or androgen signaling inhibitors in combination with ADT for metastatic hormone sensitive PCa have demonstrated remarkable improvements in survival. This suggests ADT induces susceptibilities in cancer cells that may be exploited. The phase III CHAARTED trial (ChemoHormonal Therapy Versus Androgen Ablation Randomized Trial for Extensive Disease in Prostate Cancer), demonstrated upfront chemotherapy with concurrent docetaxel and ADT improves survival by 13.6 months versus ADT alone in hormone naïve patients (Table 1) [3]. The docetaxel group also experienced a significant delay in biochemical, symptomatic, or radiographic progression compared to controls (20.1 vs. 11.7 months; hazard ratio [HR] = 0.61; 95% confidence interval [CI] = 0.51 to 0.72; p < 0.001). These results were confirmed in the STAMPEDE (Systemic Therapy in Advancing or Metastatic Prostate Cancer: Evaluation of Drug Efficacy) [5] and GETUG-AFU 15 trials (Randomized Phase III Trial Comparing an Association of Hormonal Treatment and Docetaxel Versus the Hormonal Treatment Alone in Metastatic Prostate Cancers) [40]. Docetaxel functions to inhibit the transport of the AR, potentiating ADT action [41]. It is also a microtubule inhibitor having direct toxicity to cells. We anticipate this combination results in fewer persistent senescent cells than ADT alone post-treatment, but further mechanistic study is required regarding the synergistic activity of this combination.
The phase III LATITUDE trial (A Randomized, Double-blind, Comparative Study of Abiraterone Acetate Plus Low-Dose Prednisone Plus Androgen Deprivation Therapy [ADT] Versus ADT Alone in Newly Diagnosed Subjects with High-Risk, Metastatic Hormone-naïve Prostate Cancer [mHNPC]) demonstrated upfront therapy with concurrent abiraterone and luteinizing hormone-releasing hormone agonists improves overall survival versus ADT alone (median not reached versus 34.7 months, HR = 0.62; 95% CI: 0.51–0.76; p < 0.001) in hormone naïve patients [4]. The abiraterone group also experienced a significant delay in radiographic progression-free survival compared to control (33.0 months vs. 14.8 months; HR = 0.47; 95% CI: 0.39–0.55; p < 0.001). The abiraterone and ADT arm of the STAMPEDE trial also showed improved outcomes [6]. Abiraterone irreversibly inhibits the Cytochrome P450 17α-hydroxylase/17,20-lyase (CYP17) enzyme expressed in adrenal tissues, reducing androgen synthesis from all sources [42]. Although the combination of CYP17 inhibition and ADT demonstrates more effective androgen depletion than either agent alone [43], resistance to abiraterone develops, likely through increase CYP17 expression [44], enzymatic alterations [45], and/or gain of function 3β-hydroxysteroid dehydrogenase type 1 mutation [46]. These clinical observations suggest that the initiation of ADT induces susceptibilities in PCa cells that make them amenable to synergistic treatment and improved cell killing. Further study is required to delineate the synergistic activity of abiraterone and ADT, along with clearly defining the mechanisms of resistance in castration-resistant PCa.
5. Exploiting cellular senescence as synthetic therapy
Cellular senescence may have detrimental features. Oncogene-induced cellular senescence imposes selective pressures that promote the outgrowth of senescence-resistant aggressive tumor cell subpopulations [47]. Although cellular senescence is cytostatic and offers a potential survival advantage in some models, long-term exposure of surrounding cells to the SASP, and resulting proinflammatory cytokines and growth factors, may have deleterious effects on surrounding cells [7]. In a recent Nature paper, it was reported that lymphoma cells released from chemotherapy-induced senescence results in a population of cells exhibiting a stem cell phenotype that exhibits highly aggressive growth potential upon escape from cell-cycle blockade [48]. This population is enriched in relapsing hematologic tumors. It has been proposed that ADT-induced cellular senescence might play a role in the chemoresistance that arises with intermittent ADT [32].
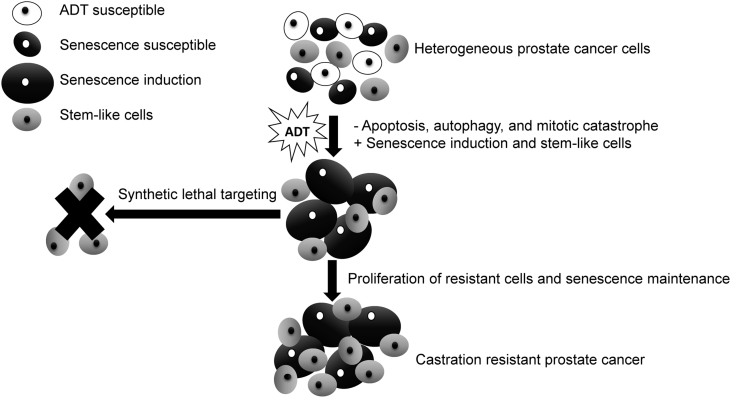
The cellular senescence phenotype presents unique opportunities that have the potential to be exploited for therapeutic cure (Fig. 1) [49]. Dörr and colleagues [36] have induced senescence in lymphoma cells and used compounds that target the inhibition of glucose transporters, glycolytic enzymes, and adenosine triphosphate (ATP) depletion to generate synthetic lethality in cancer cells (Table 2). These combinations lead to improved survival and elimination of cancer cells through caspase-12- and caspase-3-mediated endoplasmic-reticulum-related apoptosis [36]. Additionally, higher amino acid transport and increased protein synthesis generate cytokines that characterize the proinflammatory SASP. These findings highlight the hypercatabolic nature of senescent cells after induction with ADT and other agents that is therapeutically exploitable by synthetic lethal metabolic targeting. These approaches have been largely unexplored to date in PCa, but as outlined below, interesting supportive data exist.
Figure 1.
Synthetic lethal targeting of ADT induced cellular senescence for improved prostate cancer cell killing. ADT, androgen deprivation therapy.
Table 2.
Presence of the cellular senescence phenotype offers therapeutic opportunities.
| Characteristic | Synthetic lethal targeting of senescent cells |
|---|---|
| Cell morphology | |
| SA-β-gal positive | |
| Enlarged cell with prominent nuclei and cytoplasmic granularity | |
| Metabolic alterations | |
| Hypermetabolic | Glucose transport inhibitors phloretin, cytochalasin B, 2-deoxy-d-glucose [36] |
| Glycolysis | AMPK inhibitor compound C [36]/AMPK activator and mTOR inhibitor metformin [50], [51] |
| High protein turnover | Lysosomal V-ATPase inhibitors bafilomycin A1 and concanamycin A [36] |
| Secretory phenotype | |
| Pro-inflammatory cytokines | Tumor microenvironment cancer-based immunotherapy [52] |
| Growth factors | Growth factor and growth factor receptor inhibitors (e.g. VEGF, IGF-1 inhibitors) [53] |
AMPK, adenosine monophosphate-activated protein kinase; ATPase, adenosine triphosphatase; IGF-1, insulin-like growth factor 1; mTOR, mammalian target of rapamycin; SA-β-gal, senescence-associated β-galactosidas; VEGF, vascular endothelial growth factor.
The therapeutic induction of cellular senescence from chemotherapeutics, such as cyclophosphamide, results in augmented protein translation with the resultant accumulation of misfolded proteins, which activates the conserved proteotoxic stress response (PSR) [36]. The substantial proteotoxic stress in cellular senescence evokes energy-consuming countermeasures for cell survival. The PSR is orchestrated by the transcription factor heat shock factor 1 (Hsf1), which regulates the gene expression of several families of molecular chaperones (heat shock protein (Hsp) 27, Hsp70, Hsp90 and others) that attenuate proteotoxic stress and promote cell survival by preventing protein aggregation, refolding non-native proteins, or delivering them to the proteasome [54]. Anabolic tumor cells are more dependent on the Hsf1-mediated PSR than normal cells for cell survival [55]. The PSR may represent an additional novel targetable vulnerability of senescent transformed cells that can be exploited therapeutically, a hypothesis that has yet to be tested.
Metformin is an oral anti-diabetic agent in the biguanide class that has generated interest as a cancer therapy. Metformin is an intriguing drug that may not only enhance chemotherapy response for established tumors, but also demonstrates anticancer activity as a single agent [56]. Metformin inhibits the mammalian target of rapamycin (mTOR), a central regulator of cell growth and survival, in part by activating AMPK [51]. Additionally, metformin inhibits Hsf1 by activating AMPK, which in turns phosphorylates Hsf1 on Ser 121 to inhibit its activity [57]. This disables the protective effects of Hsf1 against proteotoxic stress. Synergistic activity after neoadjuvant chemotherapy was suggested in an analysis showing improved clinical response in diabetic patients with breast cancer receiving metformin and neoadjuvant chemotherapy [58]. These patients received doxorubicin, which is well known for its senescence-inducing properties in prostate and breast cancer [28], [29].
Given the metabolic susceptibilities that ADT induces, metformin leads to increased cell kill when combined with ADT. Metformin has been combined with the antiandrogen bicalutamide in vitro and in animal models [59]. This combination significantly reduced clonogenicity (p < 0.005) and tumor growth with greater effects in AR-positive cells. In unpublished data, we have found LNCaP, LaPC4, and CWR22 PCa lines all demonstrate synthetic lethal responses as calculated by Calcusyn (Biosoft, Cambridge, UK) to low dose bicalutamide (1–5 umol/L) to induce senescence initially, followed by metformin at low dose (0.1–1 mmol/L) [60]. Increased apoptosis peaks 2 days after metformin application.
In a large observational study of 87 344 veterans, we demonstrated that metformin combined with ADT improved overall survival (HR = 0.82, 95% CI: 0.78–0.86), reduced the risk of skeletal related events (HR = 0.84, 95% CI: 0.74–0.96), and improved cancer specific survival (HR = 0.70, 95% CI: 0.64–0.77) [61]. Skeletal related events, defined as pathologic fracture, spinal cord compression, and/or necessity for bone radiation or surgery due to pain or impending fracture, was used as one surrogate for disease progression. Although retrospective observational studies do not prove causality, these data merit further clinical trial investigation. Notably, metformin is an inexpensive, widely used drug associated with minimal side effects even when used in the non-diabetic population. Therefore, few barriers exist to prevent its clinical translation in PCa.
Statins are another widely used and inexpensive drug that may provide synergistic lethality with ADT given the metabolic profile of senescent cells. Statins are a class of oral anti-hypercholesterolemia agents that inhibit 3-hydroxy-3-methylglutaryl-CoA (HMG CoA) reductase. Although they are associated with myopathy and elevated transaminases, the side effect profile is favorable [62]. The use of statins reduces PSA in vivo [63], through downregulation of AR expression and activity due to enhanced proteolysis of the AR protein [64]. In LNCaP cells, the combination of simvastatin and ADT demonstrates increased growth inhibition in AR positive lines [65]. Statins also compete with the dihydrotestosterone precursor for Solute Carrier Organic Anion Transporter Family Member 2B1 mediated transport, thus decreasing androgen availability in PCa cells [66]. Men with hormone sensitive PCa undergoing ADT and taking statin drugs experienced a delayed time to disease progression compared to men not taking statins (median 27.5 months; 95% CI: 21.1–37.7 vs. 17.4 months; 95% CI: 14.9–21.1). Simvastatin has also been shown to decrease the secretory phenotype of senescent human fibroblasts, including interleukin-6, by inhibiting protein prenylation [67]. Whether statins directly induce cell death in senescent cells is unknown as of yet.
Other characteristics of senescent cells include a hypermetabolic phenotype comprised of enhanced glycolysis and protein turnover, providing a critical cellular senescence associated metabolic liability that may be targeted therapeutically. Senescent cells are selectively susceptible to inhibition of glucose transporters (e.g. cytochalasin B), or to the pharmacological competitor (2-deoxy-d-glucose) [36]. Senescent cells also rely on an intact lysosomal protein degradation machinery to buffer proteotoxic stress. Exposure of senescent cells to bafilomycin A1 or concanamycin A, specific inhibitors of lysosomal V-ATPases, or to a cocktail of lysosomal protease inhibitors all generate increased death in therapy induced senescent cells [36]. With use of these inhibitor in cell culture, senescent cell death occurred at significantly higher rates. They provide an avenue for therapeutic exploitation that should be examined further.
6. Conclusion and future directions
ADT has been used as the primary approach to advanced hormone sensitive PCa for over 75 years. Recent clinical trials have demonstrated combining ADT with other agents improves survival. ADT and docetaxel (or abiraterone) should be considered standard of care for patients with metastatic hormone sensitive PCa given recent trial results. Research is needed to define and understand these observations to further improve our progress in this area. Cellular senescence is a distinctive phenotype characterized by metabolic alterations and growth arrest. ADT induced cellular senescence in PCa may be deleterious; however, it may offer a unique opportunity for synthetic lethal targeting of residual PCa cells. Several recent retrospective hypothesis-generating studies suggest combining ADT with agents that target metabolism, including metformin, may improve patient survival. Future work should focus on further delineating the response of tumors to ADT and cellular senescence biology in this context.
Conflicts of interest
The study was supported by DOD Prostate Cancer Research Program PC150221, R. Stephenson Family Fund.
Author contributions
Study concept and design: Kyle A. Richards, Bing Yang, Vincent Cryns, David F. Jarrard.
Drafting of manuscript: Tyler Etheridge, Shivashankar Damodaran, Adam Schultz, Kyle A. Richards, Joseph Gawdzik, Bing Yang, Vincent Cryns, David F. Jarrard.
Footnotes
Peer review under responsibility of Second Military Medical University.
References
- 1.Siegel R.L., Miller K.D., Jemal A. Cancer statistics, 2017. CA Cancer J Clin. 2017;67:7–30. doi: 10.3322/caac.21387. [DOI] [PubMed] [Google Scholar]
- 2.Moul J.W. Hormone naïve prostate cancer: predicting and maximizing response intervals. Asian J Androl. 2015;17:929–935. doi: 10.4103/1008-682X.152821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Sweeney C.J., Chen Y.H., Carducci M., Liu G., Jarrard D.F., Eisenberger M. Chemohormonal therapy in metastatic hormone-sensitive prostate cancer. N Engl J Med. 2015;373:737–746. doi: 10.1056/NEJMoa1503747. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Fizazi K., Tran N., Fein L., Matsubara N., Rodriguez-Antolin A., Alekseev B.Y. Abiraterone plus prednisone in metastatic, castration-sensitive prostate cancer. N Engl J Med. 2017;377:352–360. doi: 10.1056/NEJMoa1704174. [DOI] [PubMed] [Google Scholar]
- 5.James N.D., Sydes M.R., Clarke N.W., Mason M.D., Dearnaley D.P., Spears M.R. Addition of docetaxel, zoledronic acid, or both to first-line long-term hormone therapy in prostate cancer (STAMPEDE): survival results from an adaptive, multiarm, multistage, platform randomised controlled trial. Lancet. 2016;387:1163–1177. doi: 10.1016/S0140-6736(15)01037-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.James N.D., de Bono J.S., Spears M.R., Clarke N.W., Mason M.D., Dearnaley D.P. Abiraterone for prostate cancer not previously treated with hormone therapy. N Engl J Med. 2017;377:338–351. doi: 10.1056/NEJMoa1702900. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Ewald J.A., Desotelle J.A., Wilding G., Jarrard D.F. Therapy-induced senescence in cancer. J Natl Cancer Inst. 2010;102:1536–1546. doi: 10.1093/jnci/djq364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ewald J.A., Desotelle J.A., Church D.R., Yang B., Huang W., Laurila T.A. Androgen deprivation induces senescence characteristics in prostate cancer cells in vitro and in vivo. Prostate. 2013;73:337–345. doi: 10.1002/pros.22571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Tuttle R., Miller K.R., Maiorano J.N., Termuhlen P.M., Gao Y., Berberich S.J. Novel senescence associated gene, YPEL3, is repressed by estrogen in ER+ mammary tumor cells and required for tamoxifen-induced cellular senescence. Int J Cancer. 2012;130:2291–2299. doi: 10.1002/ijc.26239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kyprianou N., Isaacs J.T. Activation of programmed cell death in the rat ventral prostate after castration. Endocrinology. 1988;122:552–562. doi: 10.1210/endo-122-2-552. [DOI] [PubMed] [Google Scholar]
- 11.Kyprianou N., English H.F., Isaacs J.T. Programmed cell death during regression of PC-82 human prostate cancer following androgen ablation. Cancer Res. 1990;50:3748–3753. [PubMed] [Google Scholar]
- 12.Agus D.B., Cordon-Cardo C., Fox W., Drobnjak M., Koff A., Golde D.W. Prostate cancer cell cycle regulators: response to androgen withdrawal and development of androgen independence. J Natl Cancer Inst. 1999;91:1869–1876. doi: 10.1093/jnci/91.21.1869. [DOI] [PubMed] [Google Scholar]
- 13.Ohlson N., Wikström P., Stattin P., Bergh A. Cell proliferation and apoptosis in prostate tumors and adjacent non-malignant prostate tissue in patients at different time-points after castration treatment. Prostate. 2005;62:307–315. doi: 10.1002/pros.20139. [DOI] [PubMed] [Google Scholar]
- 14.Westin P., Stattin P., Damber J.E., Bergh A. Castration therapy rapidly induces apoptosis in a minority and decreases cell proliferation in a majority of human prostatic tumors. Am J Pathol. 1995;146:1368–1375. [PMC free article] [PubMed] [Google Scholar]
- 15.Smitherman A.B., Gregory C.W., Mohler J.L. Apoptosis levels increase after castration in the CWR22 human prostate cancer xenograft. Prostate. 2003;57:24–31. doi: 10.1002/pros.10271. [DOI] [PubMed] [Google Scholar]
- 16.Zhang K.X., Firus J., Prieur B., Jia W., Rennie P.S. To die or to survive, a fatal question for the destiny of prostate cancer cells after androgen deprivation therapy. Cancers (Basel) 2011;3:1498–1512. doi: 10.3390/cancers3021498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Mathew R., Karantza-Wadsworth V., White E. Role of autophagy in cancer. Nat Rev Cancer. 2007;7:961–967. doi: 10.1038/nrc2254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Gerland L.M., Peyrol S., Lallemand C., Branche R., Magaud J.P., Ffrench M. Association of increased autophagic inclusions labeled for beta-galactosidase with fibroblastic aging. Exp Gerontol. 2003;38:887–895. doi: 10.1016/s0531-5565(03)00132-3. [DOI] [PubMed] [Google Scholar]
- 19.Chhipa R.R., Wu Y., Ip C. AMPK-mediated autophagy is a survival mechanism in androgen-dependent prostate cancer cells subjected to androgen deprivation and hypoxia. Cell Signal. 2011;23:1466–1472. doi: 10.1016/j.cellsig.2011.04.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Boutin B., Tajeddine N., Vandersmissen P., Zanou N., Van Schoor M., Mondin L. Androgen deprivation and androgen receptor competition by bicalutamide induce autophagy of hormone-resistant prostate cancer cells and confer resistance to apoptosis. Prostate. 2013;73:1090–1102. doi: 10.1002/pros.22658. [DOI] [PubMed] [Google Scholar]
- 21.Ojo D., Lin X., Wong N., Gu Y., Tang D. Prostate cancer stem-like cells contribute to the development of castration-resistant prostate cancer. Cancers (Basel) 2015;7:2290–2308. doi: 10.3390/cancers7040890. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Collins A.T., Berry P.A., Hyde C., Stower M.J., Maitland N.J. Prospective identification of tumorigenic prostate cancer stem cells. Cancer Res. 2005;65:10946–10951. doi: 10.1158/0008-5472.CAN-05-2018. [DOI] [PubMed] [Google Scholar]
- 23.Hayflick L. The limited in vitro lifespan of human diploid cell strains. Exp Cell Res. 1965;25:585–621. doi: 10.1016/0014-4827(61)90192-6. [DOI] [PubMed] [Google Scholar]
- 24.Campisi J. Cancer, aging and cellular senescence. In Vivo. 2000;14:183–188. [PubMed] [Google Scholar]
- 25.Gewirtz D.A., Holt S.E., Elmore L.W. Accelerated senescence: an emerging role in tumor cell response to chemotherapy and radiation. Biochem Pharmacol. 2008;76:947–957. doi: 10.1016/j.bcp.2008.06.024. [DOI] [PubMed] [Google Scholar]
- 26.Ghashghaei M., Paliouras M., Heravi M., Bekerat H., Trifiro M., Niazi T.M. Enhanced radiosensitization of enzalutamide via schedule dependent administration to androgen-sensitive prostate cancer cells. Prostate. 2018;78:64–75. doi: 10.1002/pros.23445. [DOI] [PubMed] [Google Scholar]
- 27.Bringold F., Serrano M. Tumor suppressors and oncogenes in cellular senescence. Exp Gerontol. 2000;35:317–329. doi: 10.1016/s0531-5565(00)00083-8. [DOI] [PubMed] [Google Scholar]
- 28.Chang B.D., Broude E.V., Dokmanovic M., Zhu H., Ruth A., Xuan Y. A senescence-like phenotype distinguishes tumor cells that undergo terminal proliferation arrest after exposure to anticancer agents. Cancer Res. 1999;59:3761–3767. [PubMed] [Google Scholar]
- 29.Schwarze S.R., Fu V.X., Desotelle J.A., Kenowski M.L., Jarrard D.F. The identification of senescence-specific genes during the induction of senescence in prostate cancer cells. Neoplasia. 2005;7:816–823. doi: 10.1593/neo.05250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Dimri G.P., Lee X., Basile G., Acosta M., Scott G., Roskelley C. A biomarker that identifies senescent human cells in culture and in aging skin in vivo. Proc Natl Acad Sci USA. 1995;92:9363–9367. doi: 10.1073/pnas.92.20.9363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Wagner J., Damaschke N., Yang B., Truong M., Guenther C., McCormick J. Overexpression of the novel senescence marker β-galactosidase (GLB1) in prostate cancer predicts reduced PSA recurrence. PLoS One. 2015;10:e0124366. doi: 10.1371/journal.pone.0124366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Burton D.G., Giribaldi M.G., Munoz A., Halvorsen K., Patel A., Jorda M. Androgen deprivation-induced senescence promotes outgrowth of androgen-refractory prostate cancer cells. PLoS One. 2013;8:e68003. doi: 10.1371/journal.pone.0068003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Pernicová Z., Slabáková E., Kharaishvili G., Bouchal J., Král M., Kunická Z. Androgen depletion induces senescence in prostate cancer cells through down-regulation of Skp2. Neoplasia. 2011;13:526–536. doi: 10.1593/neo.11182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Bartkova J., Rezaei N., Liontos M., Karakaidos P., Kletsas D., Issaeva N. Oncogene-induced senescence is part of the tumorigenesis barrier imposed by DNA damage checkpoints. Nature. 2006;444:633–637. doi: 10.1038/nature05268. [DOI] [PubMed] [Google Scholar]
- 35.Blute M.L., Damaschke N., Wagner J., Yang B., Gleave M., Fazli L. Persistence of senescent prostate cancer cells following prolonged neoadjuvant androgen deprivation therapy. PLoS One. 2017;12:e0172048. doi: 10.1371/journal.pone.0172048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Dörr J.R., Yu Y., Milanovic M., Beuster G., Zasada C., Däbritz J.H. Synthetic lethal metabolic targeting of cellular senescence in cancer therapy. Nature. 2013;501:421–425. doi: 10.1038/nature12437. [DOI] [PubMed] [Google Scholar]
- 37.Jones R.G., Plas D.R., Kubek S., Buzzai M., Mu J., Xu Y. AMP-activated protein kinase induces a p53-dependent metabolic checkpoint. Mol Cell. 2005;18:283–293. doi: 10.1016/j.molcel.2005.03.027. [DOI] [PubMed] [Google Scholar]
- 38.Roediger J., Hessenkemper W., Bartsch S., Manvelyan M., Huettner S.S., Liehr T. Supraphysiological androgen levels induce cellular senescence in human prostate cancer cells through the Src-Akt pathway. Mol Cancer. 2014;13:214. doi: 10.1186/1476-4598-13-214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Hessenkemper W., Roediger J., Bartsch S., Houtsmuller A.B., van Royen M.E., Petersen I. A natural androgen receptor antagonist induces cellular senescence in prostate cancer cells. Mol Endocrinol. 2014;28:1831–1840. doi: 10.1210/me.2014-1170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Gravis G., Fizazi K., Joly F., Oudard S., Priou F., Esterni B. Androgen-deprivation therapy alone or with docetaxel in non-castrate metastatic prostate cancer (GETUG-AFU 15): a randomised, open-label, phase 3 trial. Lancet Oncol. 2013;14:149–158. doi: 10.1016/S1470-2045(12)70560-0. [DOI] [PubMed] [Google Scholar]
- 41.Zhu M.L., Horbinski C.M., Garzotto M., Qian D.Z., Beer T.M., Kyprianou N. Tubulin-targeting chemotherapy impairs androgen receptor activity in prostate cancer. Cancer Res. 2010;70:7992–8002. doi: 10.1158/0008-5472.CAN-10-0585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Thakur A., Roy A., Ghosh A., Chhabra M., Banerjee S. Abiraterone acetate in the treatment of prostate cancer. Biomed Pharmacother. 2018;101:211–218. doi: 10.1016/j.biopha.2018.02.067. [DOI] [PubMed] [Google Scholar]
- 43.Attard G., Reid A.H., Yap T.A., Raynaud F., Dowsett M., Settatree S. Phase I clinical trial of a selective inhibitor of CYP17, abiraterone acetate, confirms that castration-resistant prostate cancer commonly remains hormone driven. J Clin Oncol. 2008;26:4563–4571. doi: 10.1200/JCO.2007.15.9749. [DOI] [PubMed] [Google Scholar]
- 44.Mostaghel E.A., Marck B.T., Plymate S.R., Vessella R.L., Balk S., Matsumoto A.M. Resistance to CYP17A1 inhibition with abiraterone in castration-resistant prostate cancer: induction of steroidogenesis and androgen receptor splice variants. Clin Cancer Res. 2011;17:5913–5925. doi: 10.1158/1078-0432.CCR-11-0728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Cai C., Chen S., Ng P., Bubley G.J., Nelson P.S., Mostaghel E.A. Intratumoral de novo steroid synthesis activates androgen receptor in castration-resistant prostate cancer and is upregulated by treatment with CYP17A1 inhibitors. Cancer Res. 2011;71:6503–6513. doi: 10.1158/0008-5472.CAN-11-0532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Chang K.H., Li R., Kuri B., Lotan Y., Roehrborn C.G., Liu J. A gain-of-function mutation in DHT synthesis in castration-resistant prostate cancer. Cell. 2013;154:1074–1084. doi: 10.1016/j.cell.2013.07.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Halazonetis T.D., Gorgoulis V.G., Bartek J. An oncogene-induced DNA damage model for cancer development. Science. 2008;319:1352–1355. doi: 10.1126/science.1140735. [DOI] [PubMed] [Google Scholar]
- 48.Milanovic M., Fan D.N.Y., Belenki D., Däbritz J.H.M., Zhao Z., Yu Y. Senescence-associated reprogramming promotes cancer stemness. Nature. 2018;553:96–100. doi: 10.1038/nature25167. [DOI] [PubMed] [Google Scholar]
- 49.Cairney C.J., Bilsland A.E., Evans T.R., Roffey J., Bennett D.C., Narita M. Cancer cell senescence: a new frontier in drug development. Drug Discov Today. 2012;17:269–276. doi: 10.1016/j.drudis.2012.01.019. [DOI] [PubMed] [Google Scholar]
- 50.Rocha G.Z., Dias M.M., Ropelle E.R., Osório-Costa F., Rossato F.A., Vercesi A.E. Metformin amplifies chemotherapy-induced AMPK activation and antitumoral growth. Clin Cancer Res. 2011;17:3993–4005. doi: 10.1158/1078-0432.CCR-10-2243. [DOI] [PubMed] [Google Scholar]
- 51.Dowling R.J., Zakikhani M., Fantus I.G., Pollak M., Sonenberg N. Metformin inhibits mammalian target of rapamycin-dependent translation initiation in breast cancer cells. Cancer Res. 2007;67:10804–10812. doi: 10.1158/0008-5472.CAN-07-2310. [DOI] [PubMed] [Google Scholar]
- 52.Nakamura K., Smyth M.J. Targeting cancer-related inflammation in the era of immunotherapy. Immunol Cell Biol. 2017;95:325–332. doi: 10.1038/icb.2016.126. [DOI] [PubMed] [Google Scholar]
- 53.Macfarlane R.J., Chi K.N. Novel targeted therapies for prostate cancer. Urol Clin North Am. 2010;37:105–119. doi: 10.1016/j.ucl.2009.11.011. Table of Contents. [DOI] [PubMed] [Google Scholar]
- 54.Westerheide S.D., Morimoto R.I. Heat shock response modulators as therapeutic tools for diseases of protein conformation. J Biol Chem. 2005;280:33097–33100. doi: 10.1074/jbc.R500010200. [DOI] [PubMed] [Google Scholar]
- 55.Dai C., Whitesell L., Rogers A.B., Lindquist S. Heat shock factor 1 is a powerful multifaceted modifier of carcinogenesis. Cell. 2007;130:1005–1018. doi: 10.1016/j.cell.2007.07.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Foretz M., Guigas B., Bertrand L., Pollak M., Viollet B. Metformin: from mechanisms of action to therapies. Cell Metab. 2014;20:953–966. doi: 10.1016/j.cmet.2014.09.018. [DOI] [PubMed] [Google Scholar]
- 57.Dai S., Tang Z., Cao J., Zhou W., Li H., Sampson S. Suppression of the HSF1-mediated proteotoxic stress response by the metabolic stress sensor AMPK. EMBO J. 2015;34:275–293. doi: 10.15252/embj.201489062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Jiralerspong S., Palla S.L., Giordano S.H., Meric-Bernstam F., Liedtke C., Barnett C.M. Metformin and pathologic complete responses to neoadjuvant chemotherapy in diabetic patients with breast cancer. J Clin Oncol. 2009;27:3297–3302. doi: 10.1200/JCO.2009.19.6410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Colquhoun A.J., Venier N.A., Vandersluis A.D., Besla R., Sugar L.M., Kiss A. Metformin enhances the antiproliferative and apoptotic effect of bicalutamide in prostate cancer. Prostate Cancer Prostatic Dis. 2012;15:346–352. doi: 10.1038/pcan.2012.16. [DOI] [PubMed] [Google Scholar]
- 60.McCormick J.R., Blute M.L., Yang B., Damaschke N., Jarrard D.F. Synthetic lethal metabolic targeting of cellular senescence in prostate cancer with the repurposed drug metformin. J Urol. 2016;195:e673–e674. [Google Scholar]
- 61.Richards K.A., Liou J.I., Cryns V.L., Downs T.M., Abel E.J., Jarrard D.F. Metformin use is associated with improved survival in patients with advanced prostate cancer on androgen deprivation therapy. J Urol. 2018;200:1256–1263. doi: 10.1016/j.juro.2018.06.031. [DOI] [PubMed] [Google Scholar]
- 62.Armitage J. The safety of statins in clinical practice. Lancet. 2007;370:1781–1790. doi: 10.1016/S0140-6736(07)60716-8. [DOI] [PubMed] [Google Scholar]
- 63.Hamilton R.J., Goldberg K.C., Platz E.A., Freedland S.J. The influence of statin medications on prostate-specific antigen levels. J Natl Cancer Inst. 2008;100:1511–1518. doi: 10.1093/jnci/djn362. [DOI] [PubMed] [Google Scholar]
- 64.Yang L., Egger M., Plattner R., Klocker H., Eder I.E. Lovastatin causes diminished PSA secretion by inhibiting AR expression and function in LNCaP prostate cancer cells. Urology. 2011;77:1508. doi: 10.1016/j.urology.2010.12.074. e1501–7. [DOI] [PubMed] [Google Scholar]
- 65.Syvälä H., Pennanen P., Bläuer M., Tammela T.L., Murtola T.J. Additive inhibitory effects of simvastatin and enzalutamide on androgen-sensitive LNCaP and VCaP prostate cancer cells. Biochem Biophys Res Commun. 2016;481:46–50. doi: 10.1016/j.bbrc.2016.11.021. [DOI] [PubMed] [Google Scholar]
- 66.Harshman L.C., Wang X., Nakabayashi M., Xie W., Valenca L., Werner L. Statin use at the time of initiation of androgen deprivation therapy and time to progression in patients with hormone-sensitive prostate cancer. JAMA Oncol. 2015;1:495–504. doi: 10.1001/jamaoncol.2015.0829. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Liu S., Uppal H., Demaria M., Desprez P.Y., Campisi J., Kapahi P. Simvastatin suppresses breast cancer cell proliferation induced by senescent cells. Sci Rep. 2015;5:17895. doi: 10.1038/srep17895. [DOI] [PMC free article] [PubMed] [Google Scholar]