Abstract
Use of nanomaterials in the field of science and technology includes different fields in food industry, medicine, agriculture and cosmetics. Nanoparticle-based sensors have wide range of applications in food industry for identification and detection of chemical contaminants, pathogenic bacteria, toxins and fungal toxins from food materials with high specificity and sensitivity. Nanoparticle–microbe interactions play a significant role in disease treatment in the form of antimicrobial agents. The inhibitory mechanism of nanoparticles against different bacteria and fungi includes release of metal ions that interacts with cellular components through various pathways including reactive oxygen species (ROS) generation, pore formation in cell membranes, cell wall damage, DNA damage, and cell cycle arrest and ultimately inhibits the growth of cells. Nanoparticle-based therapies are growing to study the therapeutic treatments of plant diseases and to prevent the growth of phytopathogens leading to the growing utilization of engineered nanomaterials. Hence, with this background, the present review focuses thoroughly on detailed actions and responses of nanomaterials against different bacteria and fungi as well as food sensing and storage.
Keywords: Nanomaterials, Bacteria, Fungi, Mechanism of action
Introduction
Nanotechnology has received huge attention in recent years because it involves the synthesis of a material that is distinct from its bulk material with desired characteristics. The very exciting property of nanomaterials is the high ratio of surface area to volume that further upsurges the capability to penetrate cell membranes and help in biochemical activities. Nanomaterials which are produced normally for particular purposes, created inadvertently through sources such as environmental pollution or pollutants. Nanomaterials are being exposed to earth and environment because nanomaterials are designed for their utilization in agribusiness, personal care items, food, biotechnology and medicinal purpose. There is perception that nanomaterials may effectively add to betterment of diagnosis, analysis, treatment, and avoidance of diseases, including carcinoma or microbe-based infections. Engineered nanomaterials are widely used in distinct domains which including food industry, medicine, agriculture, cosmetics and many more fields of science and technology. The diverse applications of nanoparticles have been reported, which include medical applications, industrial along with involvement of nanoparticles in different materials of day to day life including cosmetics, clothes, etc. (Dubchak et al. 2010). Materials at nanoscale are observed to exhibit specific physicochemical characteristics, which can be attributed to their nano range size, large surface area to volume ratio, composition, hydrophilic or hydrophobic nature (Navya and Daima 2016). Therefore, nanotechnology-based techniques present novel opportunities for synthesizing nanoscale materials also known as engineered nanomaterials (ENMs) having extensive applications. Nanotechnology is now involved in shaping the global economy by commercialization of consumer products, such as medicines, cosmetics and also in industrial applications and remediation processes (Musee et al. 2011). After extensive studies from last two decades, the behavior of nanoparticles with living systems is still a matter of great debate. It might be due to its differential impact (both positive and negative). Sometimes nanoparticles play a beneficial role, however, in some cases, they cause severe damage to living system (Gwinn and vallyathan 2006; Lundqvist et al. 2008; Chatterjee et al. 2011; Tripathi et al. 2015, 2017a, b; Singh et al. 2016; Vishwakarma et al. 2017).
Metallic nanoparticles with antimicrobial properties have been used in medicine traditionally. For instance, silver was utilized in prevention of eye infections in infants (Lemire et al. 2013) and silver foils for treatment of surgical wounds (Silver et al. 2006). Medicinal applications of metals were significant until the finding of antibiotics and now, at the beginning of this century, with the increasing risk of multidrug resistance and lack of new antibiotics, use of metals having antimicrobial property is gaining pace in the form of different nanomaterials (Lemire et al. 2013). These nanomaterials are substantially significant and gradually being utilized for commercial applications in different fields of biotechnology and medicine (Lakshminarayanan et al. 2018).
Biosynthesis of nanoparticles
Synthesis of metallic nanoparticles can be performed using physical, chemical and biologically mediated pathways (Bansal et al. 2006b). In vitro biosynthesis method for synthesis of metallic nanoparticles utilizes the antioxidant activity of plant secondary metabolites. Both silver and gold nanoparticles in different sizes and shapes have been synthesized using the different plant extracts (Shankar et al. 2004). The mechanism of plant extract mediated synthesis includes initiation of nucleation by reduction of metal ions due to the antioxidant (reducing) compounds/phytochemicals present in the plant extracts. The nuclei further grow by deposition of metal atoms after reduction, thus resulting in formation of metallic nanoparticles (Rajeshkumar and Bharath 2017).
The metallic and non-metallic nanoparticles can also be synthesized in vivo by a number of bacterial and fungal species (Bansal et al. 2006a). The biosynthesis of these nanoparticles can either be intracellular and extracellular (Iravani 2014). Synthesis of gold and silver nanoparticles by Bacillus spp. with the average particle size ranging between 1 and 50 nm (Iravani 2014) has been reported. Similarly, the bacterium E. coli DH5α has been used in biosynthesis of silver (10–100 nm) (Ghorbani 2013) and gold (25 nm) (Du et al. 2007) nanoparticles. The other strains of E. coli have also been reported to biosynthesize gold and silver nanoparticles of different sizes (Iravani 2014). In similar studies, Pseudomonas aeruginosa, Klebsiella pneumonia and Lactobacillus spp. have also been reported for biosynthesis of metallic nanoparticles (Iravani 2014). Nanoparticle biosynthesis by different fungi and actinomycete has also been reported in different studies. In a study reported by Mukherjee et al. (2001), synthesis of silver nanoparticles using fungus Verticillium with the average particle size of 25 ± 12 nm. Both gold and silver nanoparticle biosynthesis using different fungi has been reported in either extracellular or intracellular conditions (Sastry et al. 2003). Some examples of synthesis of nanoparticles from microbes are highlighted in Table 1.
Table 1.
Biosynthesis of nanoparticles
Nanomaterial–microbe Interactions
Recent advancements in the strategy and knowledge of nanostructures have uncovered their biological activity through alterations in structure and functions prompted in bacteria. Special developments have been accomplished in figuring out the dimensions, form and surface chemistry of nanostructures and effects on biocidal activities. Microorganisms, bacteria, fungal spores, and the wide variety of zoo-/phytoplankton are examples of micro sized agents. Nanomaterials seemed to be interacting with all type of bacteria. These illustrations pointed that the microbial surface differs significantly in their attraction. For example, silica nanoparticles are associated with microbes in the following order: fungal spores larger than the microalgae and bacteria. The detailed mechanisms wherein these metallic nanostructures cause harm to bacteria nonetheless remain doubtful; however, unique attention has been paid at morphological alterations under in vitro studies in presence of nanostructures. The fungal infections have been considerably identified to the rising mortality and morbidity of immune compromised patients, with the need of serious treatment (Pfaller and Diekema 2007). Antifungal activities of metal nanoparticles were performed by very few studies. The fungicidal and fungistatic effects of the silver nanoparticles against certain pathogenic yeasts have been examined (Kim et al. 2008). For example, antifungal activities are higher for silver nanoparticles against pathogenic Candida spp. Silver nanoparticles showed antifungal activity to be comparable with that of ionic silver (Ag+) (Kim et al. 2008). The antifungal activities of spherical silver nanoparticles have been examined against various dermatophytes. Silver nanoparticles exhibited strong activity against ATCC strains and clinical isolates of Trichophyton mentagrophytes and Candida species that can be explained due to its effects on the mycelia of fungi (Kim et al. 2009).
Mechanism of action
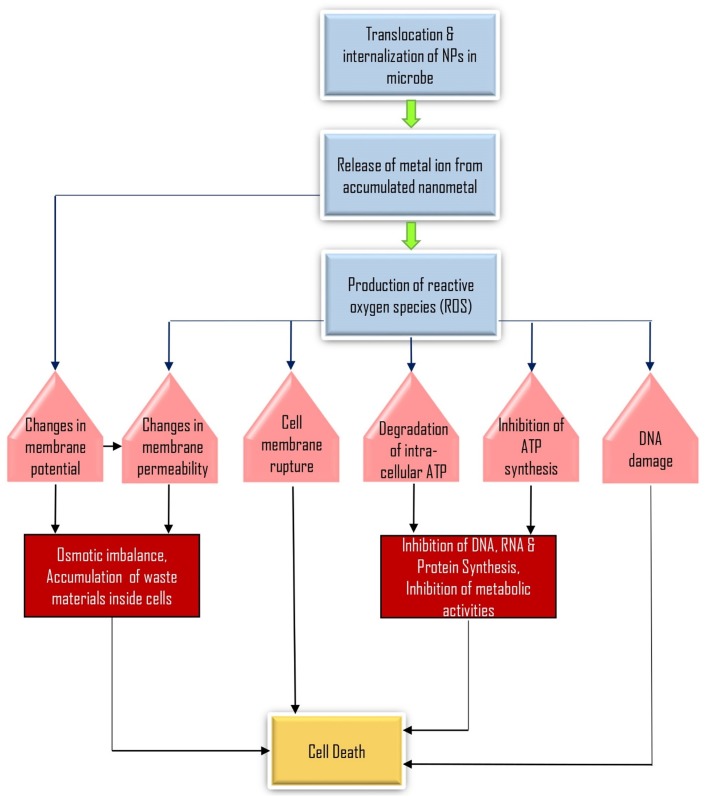
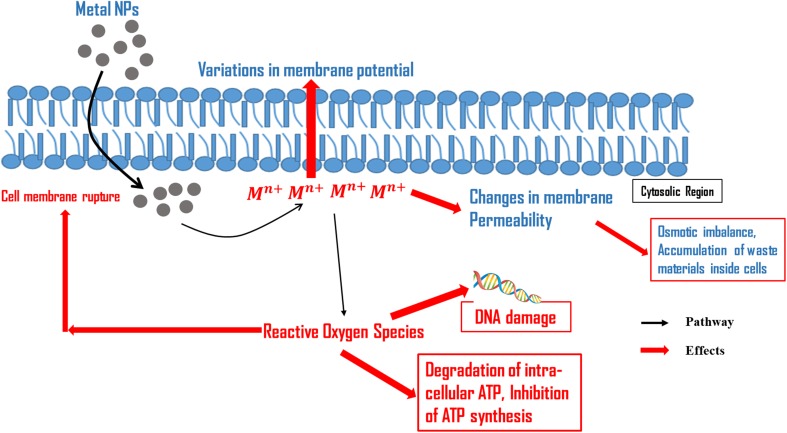
Despite the fact that the mechanisms responsible for biocidal activity shown by metallic nanoparticles are still not utterly explicated, three conjectural mechanisms are essentially the most generally authorized and mentioned in the literature. These are stated below:
Metal ions are taken up (translocation and internalization) inside the cells which results in degradation of intracellular ATP and interruption in DNA duplication (Lok et al. 2006),
Metal nanoparticles and ions generate the reactive oxygen species (ROS) causing damage to the cellular structures (Kim et al. 2007), and
These nanoparticles accumulate and dissolve in the bacterial membrane that leads to alterations in permeability of membrane (gradual liberation of lipopolysaccharides, membrane proteins and intracellular parameters) and dissipating the proton motive force (PMF) (McQuillan 2010).
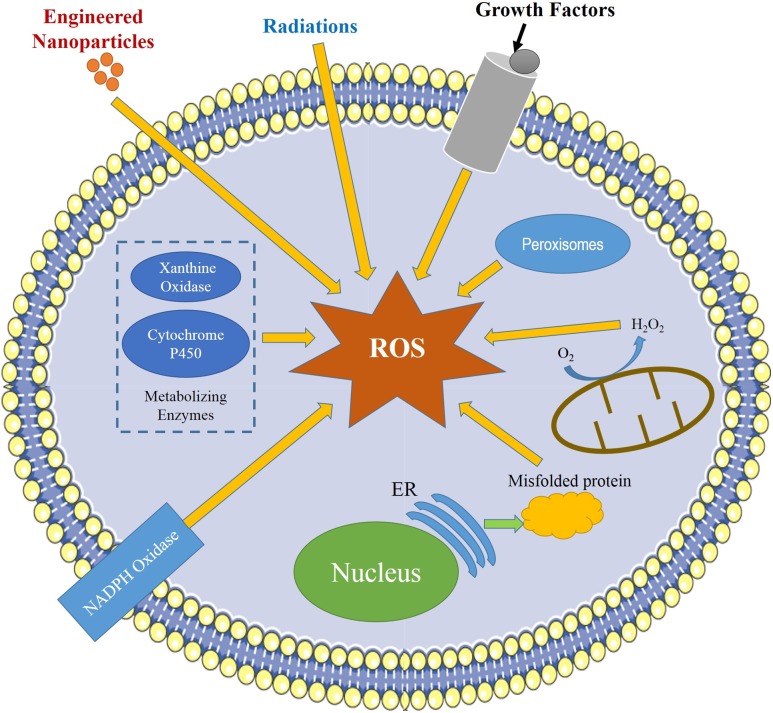
The oxidation reactions in the cell exhibit significant effects in the regulation of cell survival, cell death, differentiation, cell signaling and lead to generation of ROS under stress conditions (Touyz 2005; Mueller et al. 2005). The different components of reactive oxygen species include free radicals, such as superoxide (O2·−), singlet oxygen (1O2), hydroxyl (HO·), hydroperoxyl (HO2·), carbon dioxide radical (CO2·−), carbonate (CO3·−), peroxyl (RO2·), and alkoxyl (RO·) and nonradicals, such as ozone (O3), hydrogen peroxide (H2O2), nitric oxide (NO), hypochlorous acid (HOCl), hypochlorite (OCl−), hypobromous acid (HOBr), organic peroxides (ROOH), peroxynitrite (ONOO−), peroxynitrate (O2NOO–), peroxynitrous acid (ONOOH) and peroxomonocarbonate (HOOCO2−) (Pantopoulos and Schipper 2012; Wu et al. 2014; Halliwell 2006).
Due to instant reduction to H2O2, O2·− radical has a short lifespan, the reaction being catalyzed by superoxide dismutases (SOD) (Johnson and Giulivi 2005). Superoxide produced by nicotinamide adenine dinucleotide phosphate (NADPH) oxidase (NOX) and mitochondria (Fig. 1) inactivates different enzymes and initiates lipid peroxidation of cellular membranes (Brand 2010). The incomplete reduction of O2 results in superoxide generation and further its conversion into H2O2 by SOD. In different physiological conditions, intracellular stages are strictly modulated by different detoxifying enzymes, such as SOD, catalase (CAT), and glutathione peroxidase (GPX), or by different antioxidant compounds, including ascorbic acids, vitamin E, flavonoids, and glutathione (GSH) (Wu et al. 2014).
Fig. 1.
Different sources of generation of ROS in cells (slightly modified from Abdal-Dayem et al. 2017)
ROS production prompted by nanoparticles (silver, zinc oxide and copper oxide nanoparticles), both directly and indirectly plays an important role in genotoxicity. DNA degradation caused by oxidative stress is related to different biological mechanisms including mutagenesis. Stress due to the presence of oxidative species in higher concentration is an important mechanism that results in nano-toxicity and generation of oxidative stress leading to DNA damage (Fu et al. 2014). The DNA damage, owing to oxidative stress, involves base and sugar lesions, single- and double-strand breaks, DNA–protein crosslinks, and the generation of basic sites (Valko et al. 2006). Hydroxyl radicals are highly reactive radicals and can cause damage rapidly in the proximity, whereas the other relatively less-reactive forms of ROS may interact from a distance (Fu et al. 2014).
Bacteria are known to play important role in ecosystem. Being present in the bottom of food chain, they become key point for entry of nanomaterials to interact with organisms present at higher trophic level (Suresh et al. 2013). By acquiring knowledge regarding interaction, mechanism of bacteria with nanomaterials may be useful in redesigning environmentally benign materials. Presently, the nanomaterial toxicity mostly occurs by the concurrent dissemination of toxic metals and generation of reactive species of oxygen. Furthermore, carbon-based nanomaterials are shown to affect the mechanical stress on cell membranes. When nanostructures first interact with the cell wall, it results into disaggregated exopolysaccharide matrix, separated cells, followed by their elongation and re-arrangement into smaller groups. These changes allow the physical association of bacteria and nanostructures on available surfaces. The completely disrupted cell wall is the predominant step in second phase of interaction between nanostructures and bacteria. This results in the development of perforated and thickened cell wall. The enhancement in irregular and deformed structure can be related to the perforated cell wall accompanied with the excretion of intracellular matter and subsequent cell wall deformation (Nair et al. 2009). Most studied nanoparticle is silver nanoparticle, as its biocidal properties were observed widely. However, the mechanisms of how silver nanoparticles act on bacteria are not much clear. It was speculated that silver ions are able to interact with the bases of DNA instead of interacting with phosphate groups, thereby interfering with DNA replication (Xu et al. 2012). It is also anticipated that silver nanoparticles can liberate Ag+ ions and this mode of action performs a tremendous role in antimicrobial properties of silver nanoparticles that gets affected (Radzig et al. 2013). For instance, binding of silver ions to proteins with cysteine moieties on plasma membrane causes the damage physiologically and biochemically, resulting in compromised membrane stability. Consequently, silver penetrating into the cytoplasm inactivates potential enzyme functioning and ultimately leads to cell death (Ocsoy et al. 2013). Impacts of silver ions were observed to be powerful against wide range of microbes since ancient period. In addition, silver ions are being successfully utilized in controlling growth of bacteria for scientific purposes such as catheter, dentistry and remediation of wounds (Silver and Phung 1996; Jung et al. 2008). There are many complex mechanisms by which nanomaterials act on cells, but they are poorly understood especially when it comes to their composition, size, arrangement and shape (Van Aken 2015). For example, there was a rise in cytotoxic response of single-walled nanotubes (SWNTs) on E. coli, when its metallic and semiconductor forms were compared (Vecitis et al. 2010). On the basis of nanomaterial type and species of bacteria inhibitory, concentrations of nanomaterials are reported in the range of 2.5–500 mg/L (Hajipour et al. 2012). Similarly, in a study, different organisms such as plant, algae, bacteria, fishes and mammalian cells were tested for the toxic effects of different metal nanoparticles such as silver nanoparticles (Vishwakarma et al. 2017), copper oxide and zinc oxide nanoparticles (Bondarenko et al. 2013). Immense research has been carried out on copper nanoparticles. They have been studied to exhibit deleterious effects on E. coli (Deryabin et al. 2013; Jamshidi and Jahangiri-Rad 2014; Harikumar and Aravind 2016; Chatterjee et al. 2014), Bacillus subtilis (Yoon et al. 2007), Pseudomonas aeruginosa and Staphylococcus aureus (Azam et al. 2012).
Although sufficient development has been done in elaborating the mechanism of action of metallic nanoparticles, not much attention have been paid on iron (Fe), titanium (Ti), zinc (Zn) and nickel (Ni) nanoparticles. The use of silver and copper nanoparticles and its secreted products as biocidal measures has been a matter of huge debate during past years.
Responses of bacteria–nanomaterial interactions
Bacterial plasmolysis is a non-lytic slaying event referring to the substantial degradation of components of cytoplasm and alterations in morphology like reduction of cytoplasm as an outcome of loss in intracellular components and contraction of plasma membrane from the cell wall.
There are two different terminologies, viz. plasmolysis of bacteria and bacteriolysis. Quite often, bacteriolysis takes place when the rigid cell wall is substantially degraded because of unrestrained stimulation of autolytic wall enzymes (muramidases) (Ginsburg 1989). Bacteriolysis is considered to occur after death, and also depends on experimental conditions. However, the cause of bacteriolysis occurring after death of bacteria is not fully known yet. The biocidal components have been shown to be interacting with the cell membrane, when the released sodium (Na+) ions were exposed to copper nanoparticles. This resulted in disorganization of the membrane and production of cellular lysates (Diaz-Visurraga et al. 2010). Previously, it was noted that metallic nanoparticles impose pleiotropic activity on bacterial cells (Radzig et al. 2013). They are thought to bind with thiol moieties of bacterial proteins interrupting its activity and causing cell death and form the attachment with cellular membrane, thus disturbing its permeability by altering the cell electrical potential and disturbing the process of respiration (Radzig et al. 2013). ROS were also generated that inhibit the respiratory enzymes (Park et al. 2009). In addition, oxidized DNA precursors result in DNA lesions (Diaz-Visurraga et al. 2010).
Improved knowledge of cell-ghost formation, changes in swarming motility, sensibility against persistent and resistant strains leads to the encouragement of bacterial SOS (referring to a set of genes of the Ras subfamily of small GTPases encoding guanine nucleotide exchange factors that act on it). The response of SOS is enhanced by the metal nanoparticles which enable the scientists to take more informed judgments about the steps that have to be taken for new investigations in nanostructures with its applications as antibacterial agents. Based on the aforementioned responses, there were many studies conducted to analyze the expression of genes in response to nanoparticles. Gene expression patterns were found to be consistent, when exposed to metallic nanoparticles, by utilization of transcriptional analysis techniques, i.e., microarray or RT qPCR. For example, E. coli, when subjected to the treatment of silver nanoparticles, displayed differential expression in genes functioning, for regulating the oxidative balance, homeostasis of copper, silver and iron through whole genome microarray experiments and its properties to metabolize sulfur (Nagy et al. 2011; McQuillan and Shaw 2014). Furthermore, a number of researchers have worked on altered gene expression of bacteria in the presence of carbon-based nanomaterials. For instance, Kang et al. (2008) observed the reduced metabolism and viability, disruption of membrane integrity, leakage of cellular matter of E. coli when exposed to single walled nanotubes and found to be more severe than effect of multi-walled nanotubes.
In another microarray study on same model organism E. coli subjected to cerium oxide nanoparticles, the cerium oxide nanoparticles were observed to up-regulate number of oxidoreductases suggesting the alterations in cellular respiration, oxidation stress and iron deficiency (Pelletier et al. 2010). A similar study was conducted on Pseudomonas aeruginosa that was exposed to quantum dots and it was observed that genes responsible for oxidative stress and metal efflux transporters were up-regulated (Yang et al. 2012).
Nanoparticles could affect plant–microbe interaction by exposure of the plant growth-promoting bacterium, P. chlororaphis O6, to zinc oxide and copper oxide nanoparticles, thus leading to alterations in cellular levels of pyoverdine (siderophore) and expression of an inner membrane pyoverdine transporter gene (Dimkpa et al. 2012).
Responses of fungus–nanomaterial interactions
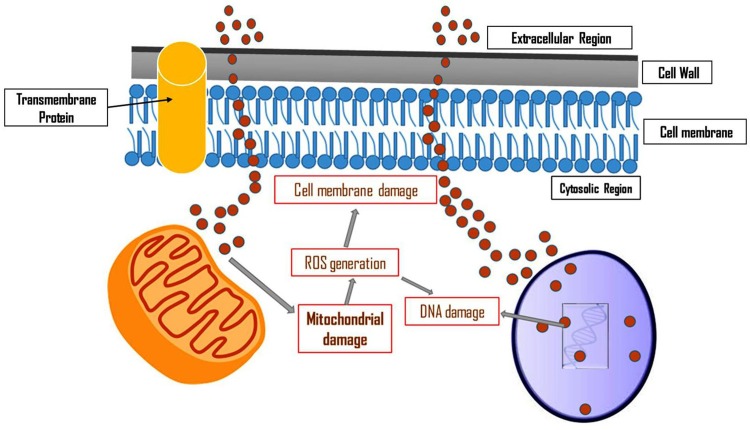
Different nanoparticles have capability to kill fungal cells (Kim et al. 2009). Formation of pores in the membrane leads to the release of biomolecules from the cells resulting in cell death. Kim et al. (2009) investigated that Candida albicans have antifungal effects on silver nanoparticles. In the experiment, the authors observed that membrane depolarization occurred in case of Candida albicans treated with silver nanoparticles. Formation of pores and pit formation in the cell wall were examined which led to release of intracellular glucose and trehalose into the suspension. By utilizing flow cytometry approach, cell cycle arrest at G2/M phase was also observed, thus preventing the growth of Candida albicans (Kim et al. 2009). Studies on antifungal activity of silver nanoparticles have been performed on different fungi including Trichophyton rubrum, Botrytis cinerea, Penicillium expansum, Phomopsis spp., Candida albicans, Candida tropicalis and Saccharomyces cerevisiae (Panácek et al. 2009; Lili et al. 2011; Nasrollahi et al. 2011; Mallmann et al. 2015). Studies showed that the inhibitory effect of silver nanoparticles on different fungi was reported and the cell membrane has been shown to be the target site for nanoparticles (Panácek et al. 2009; Lili et al. 2011; Nasrollahi et al. 2011; Mallmann et al. 2015). Prucek et al. (2011) studied the impact of Fe3O4 nanoparticles on Candida spp. and observed the inhibitory effect with site of action being the cell membrane. Cell wall and cell membrane of Cryptococcus neoformans have been found affected by silver nanoparticles (Ishida et al. 2013). In Table 2 and Figs. 2, 3 and 4, the mode of action of nanoparticles in case of different bacteria and fungi was presented.
Table 2.
Nanoparticle–microbe interaction and their responses
| S. no. | Nanoparticle | Organism | Target site | References |
|---|---|---|---|---|
| Effect of NP on bacteria | ||||
| 1. | Silver | Escherichia coli | Cell membrane | Sondi and Salopek-Sondi (2004) |
| 2. | Silver |
Escherichia coli
Pseudomonas. aeruginosa Vibrio cholera Scrub typhus |
Cell membrane | Morones et al. (2005) |
| 3. | Silver | Escherichia coli | Sulfur and phosphorous containing molecules | Pal et al. (2007) |
| 4. | Silver |
Escherichia coli
Staphylococcus aureus Listeria monocytogenes |
Cell membrane and sulfur and phosphorous | Fernández et al. (2008) |
| 5. | Silver | Recombinant bioluminescent Escherichia coli | Cell membrane | Hwang et al. (2008) |
| 6. | Fe3O4 silver |
Escherichia coli
Staphylococcus epidermidis Bacillus subtilis |
Cell membrane | Gong et al. (2007) |
| 7. | Styrene–acrylic acid/silver nanoparticles |
Escherichia coli
Staphylococcus aureus |
Cell membrane | Paula et al. (2009) |
| 8. | Silver-nHA/TiO2 |
Escherichia coli
Staphylococcus aureus Porphyromonas gingivalis Prevotella intermedia Streptococcus mutans |
Cell membrane | Liao and Li (2010) |
| 9. | Magnesium oxide |
Escherichia coli
Bacillus megaterium |
Cell membrane | Stoimenov et al. (2002) |
| 10. | Zinc oxide | Escherichia coli | Cell membrane | Zhang et al. (2007) |
| 11. | Copper |
Bacillus subtilis
Escherichia coli |
Cell membrane | Yoon et al. (2007) |
| 12. | Copper–silica |
Staphylococcus aureus
Escherichia coli Enterobacter cloacae |
Interaction with proteins | Kim et al. (2006) |
| 13. | Chitosan/copper ions | Escherichia coli | Cell membrane | Du et al. (2008) |
| Effect of NP on fungus | ||||
| 14. | Silver | Trichophyton rubrum | Cell membrane | Noorbakhsh et al. (2011) |
| 15. | Silver | Candida spp. | Cell membrane | Panácek et al. (2009) |
| 16. | Fe3O4 γ-Fe2O3 |
Candida albicans (I and II)
Candida tropicalis Candida parapsilosis |
Cell membrane | Prucek et al. (2011) |
| 17. | Zinc oxide | Botrytis cinerea, Penicillium expansum | Cell membrane | Lili et al. (2011) |
| 18. | Silver | Phomopsis spp. | Cell membrane | Mendes et al. (2014) |
| 19. | Silver |
Candida albicans
Candida tropicalis |
Cell membrane | Mallmann et al. (2015) |
| 20. | Silver |
Cryptococcus neoformans
Candida spp. |
Cell wall and cytoplasmic membrane | Ishida et al. (2013) |
| 21. | Silver |
Candida albicans
Candida parapsilosis Aspergillus niger |
Cell membrane and sulfur | Devi and Bhimba (2014) |
| 22. | Silver |
Candida albicans
Saccharomyces cerevisiae |
Cell membrane | Nasrollahi et al. (2011) |
Fig. 2.
Flow chart representing various mechanisms of nanoparticle actions
Fig. 3.
Mode of action of nanoparticles in bacterial cells
(modified from Kim et al. 2007)
Fig. 4.
Mode of action of nanoparticles in fungal cells
(modified from Kim et al. 2009)
Computer-based mathematical calculation and in silico modelling can provide more understandings of nanomaterial–microbe interactions. Experimental studies of the interactions of nanoparticles with microbes are the key and are extremely important for providing important information on the details of these interactions. Powerful bioinformatics tools are available that test statistical data and statement about a possible future event of the interactions of nanoparticles with parts of microbial membranes such as proteins, lipids or LPS. Methods include quantum mechanical, atom based by visualizing the force field, or rough grained strategies, and many more are now under development. Increasing our knowledge about something that causes disease or pathogen–nanomaterial interaction and interference could also be of importance for the development of future applications of targeted nanoparticles in medical approaches.
Nanomaterials in plant disease suppression
Plant diseases are induced with the aid of pathogenic bacteria, viruses, fungi and nematodes; the resulting contamination/ infestation leads to the economic loss by reduction in quality and yield of product and its shelf life. Patel et al. (2014) investigated that 25% of the food plants was effected by mycotoxins worldwide. Therefore, novel systems are required critically to manage crop diseases that would be a decisive component for any long-lasting process to sustain as well as enhance agriculture productivity. The possible utilization of nanoparticles to handle aforementioned necessities has been a subject of debate for several years. Earlier, Navarro et al. (2008) proposed that nanomaterials with large surface area are more efficient in retaining nutrients and may function as long-lasting and stable mineral store for crops. Thus, nanotechnology is progressively being included into the crop-industrial segment. It can present a novel, green and eco-friendly approach for managing diseases in plants very effectively. However, application of nano-biotechnology in pathological conditions of crop continues to be in the early stages.
For instance, nano-fungicides, nano-pesticides and nano-herbicides are being studied for their applications in agricultural practices (Dwivedi et al. 2016; Gul et al. 2014; Rai and Ingle 2012a). If such smart nano-supply systems are activated and monitored in remote areas, it will be of great assistance to agricultural growers in coming future to reduce the utilization of fungicides and pesticides (Rai and Ingle 2012b).
The effect of nanoparticles depends upon the species of plant and type of nanoparticle. Numbers of studies have been investigated to prove the positive cause of exposing metal oxide and metal nanoparticles on pathogen suppression and crop yield. For example, some nanoparticles such as silver, zinc oxide, magnesium, silicon, and TiO2 have been observed to be involved in direct suppression of crop ailments via their antimicrobial property (Prasad and Prasad 2014). Silver nanoparticles and ions were also assessed to examine the antifungal effect on Magnaporthe grisea and Bipolaris sorokiniana (Woo et al. 2009). In an in vitro and in vivo study by Woo et al. (2009), decrease in the progression of disease by phytopathogenic fungi was observed after treatment with both the silver nanoparticles and silver ions. Lamsal et al. (2011) investigated that the Colletotrichum spp. (anthracnose pathogen) inhibited by silver nanoparticles, when subjected to field trials. Authors also stated that there is enhancement in disease inhibition by applying nanoparticles (4–8 nm) prophylactically, giving an idea that alternate mechanisms induce significant resistance pathways.
Zinc oxide nanoparticles have also been observed to have efficient control on pathogen growth (Dimkpa et al. 2013). Zinc oxide nanoparticles offer strong benefits including lesser toxicity and adding to soil fertility over silver nanoparticles with respect to pathogen suppression (Dimkpa et al. 2013). It was also observed that zinc oxide nanoparticles in Mung bean diminished the growth of Fusarium graminearum in comparison with bulk oxide and control (Dimkpa et al. 2013). Similarly, a number of studies have proven that quantum dots could expand plant growth, probably via selective activity towards pathogens (Rispail et al. 2014). Currently, they confirmed the internalization of quantum dots within the cells (500 nM) by pathogen Fusarium oxysporum and observed a 20% cut down in growth of fungus and 15% decrease in hyphae development (Rispail et al. 2014).
Carbon-containing nanomaterials inclusive of C60/70 fullerenes, carbon nanoparticles, fullerols, single and multi-walled carbon nanotubes are also being given high attention with respect to increase in plant growth (Khot et al. 2012). Multi-walled carbon nanotubes and fullerenes have shown distinct impact on field-weathered chlordane and DDT along with metabolites buildup in a study carried out on soil system (De La Torre-Roche et al. 2013). Hamdi et al. (2014) confirmed that although non-functionalized multi-walled carbon nanotubes reduced chlordane matter in roots and shoots of lettuce by 78–88%, the repression was more reasonable with amino-functionalized tubes where the root showed 57% and shoots showed 23% reduction as compared to control. The amalgamation of biotechnology and nanotechnology in sensing field will lead to the development of methods/apparatus of great sensitivity that would enable a primitive reaction to environmental alterations and illnesses, as proposed in Fig. 4. The studies carried out till now are adequate to assure upcoming potential utilization of nanotechnology in management of several kinds of plant diseases.
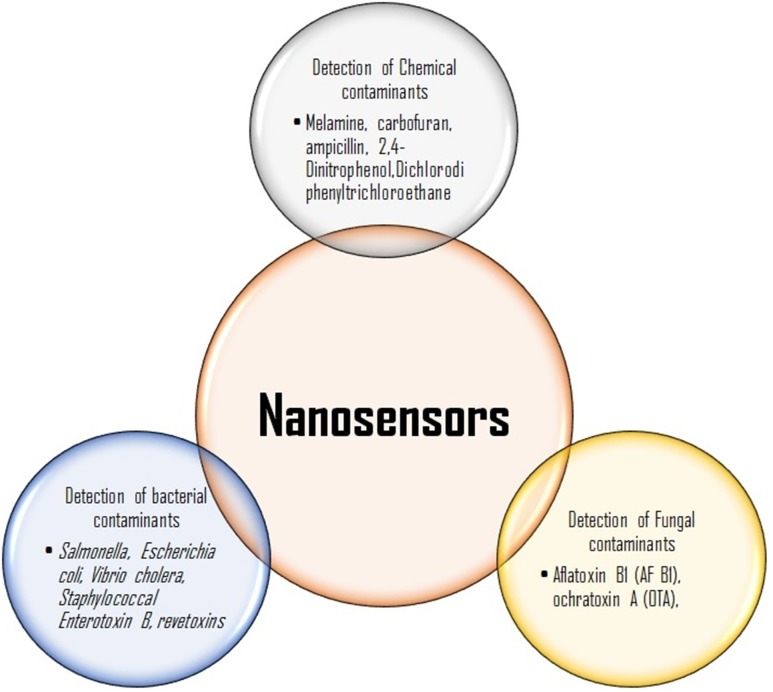
Nanosensors in contaminant detection from food materials
The most common pathogenic microbes Escherichia coli, Staphylococcus aureus, and Bacillus cereus are responsible for most of the food borne outbreaks (Park et al. 2001; Murphy et al. 2006; Gandhi and Chikindas 2007). There are many biological and immunological assays available such as sensitive ELISA-based tests for recognition of bacterial toxins, but they are not able to measure the activities of the toxin. It was investigated that different strains of toxic bacteria detected by sensitive, specific DNA probes and PCR-based tests but expression of toxin gene does not give a positive result (Priyanka et al. 2016; Wu et al. 2016; Pimbley and Patel 1998). Wu et al. (2016) have explained the rapid detection techniques for enterotoxins of Staphylococcus aureus. Among them, nanomaterials are shown to have great potential to be incorporated into various sensors to develop rapidly a sensitive and specific methodology for detecting pathogenic bacteria ensuring food safety and security, delivering an efficient alternative over the existing traditional, time-consuming and laborious methods as shown in Fig. 4. Joo et al. (2012) used an antibody-conjugated magnetic nanoparticle and were able to separate them from the samples by applying an external magnetic field to detect Salmonella spp. in milk. Several other studies have also reported the use of magnetic nanoparticles in detection of Salmonella spp. from milk (Poonlapdecha et al. 2018; Sung et al. 2013). The assay exhibited high sensitivity against low concentrations of Salmonella, hence absorption was found reversely proportional to the Salmonella concentration. Earlier, Chen et al. (2008) developed a piezoelectric biosensor using oligonucleotide fused GNPs for rapid detection the food borne pathogen Escherichia coli.
The lactose-stabilized nanoparticles in solution were observed to be red colored due to the surface plasmon absorption band at 524 nm and after binding of Cholera toxin deep purple color was observed due to lactose derivative formation. Viswanathan et al. (2006) demonstrated a sensitive method using an electrochemical immunosensor with liposomic and poly (3,4-ethylenedioxythiophene) coated carbon nanotubes for the recognition of cholera toxin. Sandwich type test on the electronic transducers was used for the identification of toxin. The amplification for the detection of toxin occurs by sandwich assay at ultra-trace levels. Liao and Li (2010) have developed a lateral flow strip for recognition of aflatoxin B1 (AF B1) in food. Wu et al. (2011) have developed a competitive fluorescence immuno-assay a rapid and sensitive method for the rapid detection by means of antigen-modified magnetic nanoparticles as immune sensing probes, and antibody functionalized rare earth doped NaYF4 up conversion nanoparticles as signal probes of AF B1 and Ochratoxin A (OTA) (another frequent mycotoxin produced by Aspergillus) in foodstuffs. The application of nanosensors in food industry is summarized in Fig. 5.
Fig. 5.
Applications of nanosensors in food industry
Nanomaterials in food storage
Gold nanomaterials are commonly used in food storage and packaging as well as food additives (Ahmed et al. 2016). Besides being common materials in food packaging and antimicrobial agents, gold nanomaterials have been used as biosensors to detect major contaminants in food products (Bajpai at al. 2018). Several reviews that focused on inorganic based nanomaterials and their applications in food analysis and other biological applications such as biosensors have been recently published (Bajpai et al. 2018). Most notably, for the detection of the presence of melamine in raw milk, where melamine induced the aggregation of the gold nanoparticles resulting in a color change from red to blue through gold nanoparticles (Noh et al. 2013; Li et al. 2010). Modifications in electrochemical DNA biosensors used by gold nanoparticles were also reported in studies (Hu et al. 2008).
In specific, silver nanoparticles are currently being used as an antimicrobial agent in food storage applications, anti-odorant, and health supplement (Sekhon 2010; Duncan 2011; Mahdi et al. 2012). In 2009, the Food and Drug Administration (FDA) allowed the use of silver as silver nitrate in bottled water at concentrations less than 17 g/kg (Duncan 2011). This is because the silver nanoparticles are effective in killing bacteria and preventing their growth, including Bacillus spp., Micrococcus luteus, and Escherichia coli (Sekhon 2010; Duncan 2011; Mahdi et al. 2012). In addition, it has been reported that using them in minced meat, reduced the growth of bacteria and increased the shelf life of the food up to 7 days (Mahdi et al. 2012). The applications of nanomaterials in food storage are summarized in Table 3 (Pradhan et al. 2015).
Table 3.
Applications of nanoparticles in food storage
| S. no. | Food material/packaging | Nanoparticle used | Application/role | References |
|---|---|---|---|---|
| 1. | Low density polyethylene | Silver, zinc oxide | Inhibition of yeast, and bacterial growth | Emamifar et al. (2010) |
| 2. | Polyurethane | Polyurethane | Inhibition of E. coli and S. aureus growth | Toker et al. (2013) |
| 3. | Low density polyethylene | Silver | Inhibition of aerobic bacterial growth | Valipoor Motalgh et al. (2013) |
| 4. | Polyethylene | Silver, TiO2 | Inhibition of Penicillium, Lactobacillus growth | Metak and Ajaal (2013) |
| 5. | Polyethylene | Silver, TiO2 | Inhibition of S. aureus, Coliforms, E. coli, Listeria growth | Mihindukulasuriya and Lim (2014) |
| 6. | Cellulose | Silver | Inhibition of total aerobic bacteria, lactic acid bacteria, Pseudomonas spp., Enterobacteriaceae | Fernandez et al. (2010) |
Conclusions and future prospects
Engineered nanomaterials have shown significant role in regulation of microbial growth and exhibit bactericidal, bacteriostatic, fungicidal and fungistatic effects through various biochemical pathways which can be further explored to control microbial growth. Use of biomolecular interactions with nanomaterial-based biosensors and nanosensors transduce the signal into detectable limits for rapid detection of food contaminants, and hence, it would be suggested to incorporate nanosensors to assure food safety and its quality. From this point of view, the development of nanosensors to detect microbes and contaminants would be an important application of food nanotechnology. in addition, the recently documented role of nanomaterials for detection of adulterant and toxins for its future role of interaction with microbes and applications can be considered for its development. Further, exhaustive research is necessary need to exploit new nanomaterials and its interaction with microbes for improving and assuring food safety and novel applications to agriculture industry.
Acknowledgements
The authors are grateful to Director MNNIT Allahabad, Central Instrumentation Facility under Department of Biotechnology, and Design and Innovation Centre, MNNIT Allahabad, Prayagraj for providing necessary research facilities. Shivesh Sharma and Durgesh K. Tripathi are thankful for the financial assistance provided by CSIR sponsored project (No. 38(1460)/18/EMR-II).
Compliance with ethical standards
Conflict of interest
Authors declared that they do not have any conflict of interest.
Contributor Information
Vivek Kumar Singh, Email: vivek.singh@smvdu.ac.in.
Durgesh Kumar Tripathi, Email: dktripathiau@gmail.com.
Shivesh Sharma, Email: shiveshs@mnnit.ac.in, Email: ssnvsharma@gmail.com.
References
- Abdal-Dayem A, Hossain MK, Lee SB, Kim K, Saha SK, Yang GM, Choi HY, Cho SG. The role of reactive oxygen species (ROS) in the biological activities of metallic nanoparticles. Int J Mol Sci. 2017;18(1):120. doi: 10.3390/ijms18010120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ahmed A, Khan AK, Anwar A, et al. Biofilm inhibitory effect of chlorhexidine conjugated gold nanoparticles against Klebsiella pneumoniae. Microb Pathog. 2016 doi: 10.1016/j.micpath.2016.06.016. [DOI] [PubMed] [Google Scholar]
- Azam A, Ahmed AS, Oves M, Khan MS, Memic A. Size-dependent antimicrobial properties of CuO nanoparticles against Gram-positive and-negative bacterial strains. Int J Nanomed. 2012;7:3527. doi: 10.2147/IJN.S29020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bajpai VK, Kamle M, Shukla S, et al. Prospects of using nanotechnology for food preservation, safety, and security. J Food Drug Anal. 2018 doi: 10.1016/j.jfda.2018.06.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bansal V, Ahmad A, Sastry M. Fungus-mediated biotransformation of amorphous silica in rice husk to nanocrystalline silica. J Am Chem Soc. 2006;128(43):14059–14066. doi: 10.1021/ja062113+. [DOI] [PubMed] [Google Scholar]
- Bansal V, Poddar P, Ahmad A, Sastry M. Room-temperature biosynthesis of ferroelectric barium titanate nanoparticles. J Am Chem Soc. 2006;128(36):11958–11963. doi: 10.1021/ja063011m. [DOI] [PubMed] [Google Scholar]
- Bondarenko O, Juganson K, Ivask A, et al. Toxicity of Ag, CuO and ZnO nanoparticles to selected environmentally relevant test organisms and mammalian cells in vitro: a critical review. Arch Toxicol. 2013;87(7):1181–1200. doi: 10.1007/s00204-013-1079-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brand MD. The sites and topology of mitochondrial superoxide production. Exp Gerontol. 2010;45(7–8):466–472. doi: 10.1016/j.exger.2010.01.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chatterjee S, Bandyopadhyay A, Sarkar K. Effect of iron oxide and gold nanoparticles on bacterial growth leading towards biological application. J Nanobiotechnol. 2011;9(1):34. doi: 10.1186/1477-3155-9-34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chatterjee AK, Chakraborty R, Basu T. Mechanism of antibacterial activity of copper nanoparticles. Nanotechnology. 2014;25(13):135101. doi: 10.1088/0957-4484/25/13/135101. [DOI] [PubMed] [Google Scholar]
- Chen SH, Wu VCH, Chuang YC, Lin CS. Using Oligonucleotide-functionalized Au nano particles to rapidly detect food borne pathogens on a piezoelectric biosensor. J Microbiol Meth. 2008;73:7–17. doi: 10.1016/j.mimet.2008.01.004. [DOI] [PubMed] [Google Scholar]
- De La Torre-Roche R, Hawthorne J, Deng Y, et al. Multiwalled carbon nanotubes and c60 fullerenes differentially impact the accumulation of weathered pesticides in four agricultural plants. Environ Sci Technol. 2013;47(21):12539–12547. doi: 10.1021/es4034809. [DOI] [PubMed] [Google Scholar]
- Deryabin DG, Aleshina ES, Vasilchenko AS, et al. Investigation of copper nanoparticles antibacterial mechanisms tested by luminescent Escherichia coli strains. Nanotechnol Russ. 2013;8(5–6):402–408. doi: 10.1134/S1995078013030063. [DOI] [Google Scholar]
- Devi JS, Bhimba BV. Antibacterial and antifungal activity of silver nanoparticles synthesized using Hypnea muciformis. Biosci Biotechnol Res Asia. 2014;11(1):235–238. doi: 10.13005/bbra/1260. [DOI] [Google Scholar]
- Diaz-Visurraga J, Cardenas G, Garcia A. Morphological changes induced in bacteria as evaluated by electron microscopy. In: Mendez-Vilas A, Diaz J, editors. Microscopy science, technology, applications. Spain: Formatex, Madrid; 2010. [Google Scholar]
- Dimkpa CO, Mclean JE, Britt DW, Anderson AJ. CuO and ZnO nanoparticles differently affect the secretion of fluorescent siderophores in the beneficial root colonizer, Pseudomonas chlororaphis O6. Nanotoxicology. 2012;6(6):635–642. doi: 10.3109/17435390.2011.598246. [DOI] [PubMed] [Google Scholar]
- Dimkpa CO, McLean JE, Britt DW, Anderson AJ. Antifungal activity of ZnO nanoparticles and their interactive effect with a biocontrol bacterium on growth antagonism of the plant pathogen Fusarium graminearum. Biometals. 2013;26(6):913–924. doi: 10.1007/s10534-013-9667-6. [DOI] [PubMed] [Google Scholar]
- Du L, Jiang H, Liu X, et al. Biosynthesis of gold nanoparticles assisted by Escherichia coli DH5α and its application on direct electrochemistry of hemoglobin. Electrochem Commun. 2007;9(5):1165–1170. doi: 10.1016/j.elecom.2007.01.007. [DOI] [Google Scholar]
- Du WL, Xu YL, Xu ZR, Fan CL. Preparation, characterization and antibacterial properties against Escherichia coli K88 of chitosan nanoparticle loaded copper ions. Nanotechnology. 2008;19(8):085707–085712. doi: 10.1088/0957-4484/19/8/085707. [DOI] [PubMed] [Google Scholar]
- Dubchak S, Ogar A, Mietelski JW, Turnau K. Influence of silver and titanium nanoparticles on arbuscular mycorrhiza colonization and accumulation of radiocaesium in Helianthus annuus. Span J Agric Res. 2010;8(1):103–108. doi: 10.5424/sjar/201008S1-1228. [DOI] [Google Scholar]
- Duncan TV. Applications of nanotechnology in food packaging and food safety: barrier materials, antimicrobials and sensors. J Colloid Interface Sci. 2011;363(1):1–24. doi: 10.1016/j.jcis.2011.07.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dwivedi S, Saquib Q, Al-Khedhairy AA, Musarrat J. Understanding the role of nanomaterials in agriculture. InMicrobial Inoculants in Sustainable Agricultural Productivity 2016. New Delhi: Springer; 2016. pp. 271–288. [Google Scholar]
- Emamifar A, Kadivar M, Shahedi M, et al. Evaluation of nanocomposite packaging containing Ag and ZnO on shelf life of fresh orange juice. Innov Food Sci Emerg Technol. 2010;11(4):742–748. doi: 10.1016/j.ifset.2010.06.003. [DOI] [Google Scholar]
- Fernández EJ, Barrasa JG, Laguna A, Luzuriaga JML, Monge M, Torres C. The preparation of highly active antimicrobial silver nanoparticles byan organometallic approach. Nanotechnology. 2008;19:185602–185608. doi: 10.1021/acs.jpcc.5b11724. [DOI] [PubMed] [Google Scholar]
- Fernández A, Picouet P, Lloret E. Cellulose-silver nanoparticle hybrid materials to control spoilage-related microflora in absorbent pads located in trays of fresh-cut melon. Int J Food Microbiol. 2010;142(1–2):222–228. doi: 10.1016/j.ijfoodmicro.2010.07.001. [DOI] [PubMed] [Google Scholar]
- Fu PP, Xia Q, Hwang HM, Ray PC, Yu H. Mechanisms of nanotoxicity: generation of reactive oxygen species. J Food Drug Anal. 2014;22(1):64–75. doi: 10.1016/j.jfda.2014.01.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gandhi M, Chikindas ML. Listeria: a foodborne pathogen that knows how to survive. Int J Food Microbiol. 2007;113:1–15. doi: 10.1016/j.ijfoodmicro.2006.07.008. [DOI] [PubMed] [Google Scholar]
- Ghorbani HR. Biosynthesis of silver nanoparticles by Escherichia coli. Asian J Chem. 2013 doi: 10.14233/ajchem.2013.12805. [DOI] [Google Scholar]
- Gong P, Li H, He X, et al. Preparation and antibacterial activity of Fe3O4@Ag nanoparticles. Nanotechnology. 2007;18:285604–285611. doi: 10.1088/0957-4484/18/28/285604. [DOI] [Google Scholar]
- Ginsburg I. Bacteriolysis is inhibited by hydrogen peroxide and by proteases. Agents Actions. 1989;28:238–242. doi: 10.1007/BF01967409. [DOI] [PubMed] [Google Scholar]
- Gul HT, Saeed S, Khan FZ, Manzoor SA. Potential of nanotechnology in agriculture and crop protection: A. Appl Sci Bus Econ. 2014;1(2):23–28. [Google Scholar]
- Gwinn MR, Vallyathan V. Nanoparticles: health effects: pros and cons. Environ Health Perspect. 2006 doi: 10.1289/ehp.8871. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hajipour MJ, Fromm KM, Ashkarra AA, et al. Antibacterial properties of nanoparticles. Trends Biotechnol. 2012;30(10):499–511. doi: 10.1016/j.tibtech.2012.06.004. [DOI] [PubMed] [Google Scholar]
- Halliwell B. Reactive species and antioxidants. Redox biology is a fundamental theme of aerobic life. Plant Physiol. 2006;141(2):312–322. doi: 10.1104/pp.106.077073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hamdi H, De La Torre-Roche R, Hawthorne J, White JC. Impact of non-functionalized and amino-functionalized multiwall carbon nanotubes on pesticide uptake by lettuce (Lactuca sativa L.) Nanotoxicology. 2014;9(2):172–180. doi: 10.3109/17435390.2014.907456. [DOI] [PubMed] [Google Scholar]
- Harikumar PS, Aravind A. Antibacterial activity of copper nanoparticles and copper nanocomposites against Escherichia Coli bacteria. Int J Sci. 2016;5(2):83–90. doi: 10.18483/ijSci.957. [DOI] [Google Scholar]
- Hu K, Lan D, Li X, Zhang S. Electrochemical DNA biosensor based on nanoporous gold electrode and multifunctional encoded DNA—Au Bio Bar Codes. Anal Chem. 2008;80(23):9124–9130. doi: 10.1021/ac8017197. [DOI] [PubMed] [Google Scholar]
- Hwang ET, Lee JH, Chae YJ, et al. Analysis of the toxic mode of action of silver nanoparticles using stress-specific bioluminescent bacteria. Small. 2008;4(6):746–750. doi: 10.1002/smll.200700954. [DOI] [PubMed] [Google Scholar]
- Iravani S. Bacteria in nanoparticle synthesis: current status and future prospects. Int Sch Res Notices. 2014 doi: 10.1155/2014/359316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ishida K, Cipriano TF, Rocha GM, et al. Silver nanoparticle production by the fungus Fusarium oxysporum: nanoparticle characterisation and analysis of antifungal activity against pathogenic yeasts. Rio de Janeiro: Mem Inst Oswaldo Cruz; 2013. pp. 1–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jamshidi A, Jahangiri-Rad M. Synthesis of copper nanoparticles and its antibacterial activity against Escherichia coli. Asian J Biol Sci. 2014;7:183–186. doi: 10.3923/ajbs.2014.183.186. [DOI] [Google Scholar]
- Johnson F, Giulivi C. Superoxide dismutases and their impact upon human health. Mol Asp Med. 2005;26(4–5):340–352. doi: 10.1016/j.mam.2005.07.006. [DOI] [PubMed] [Google Scholar]
- Joo J, Yim C, Kwon D, et al. A facile and sensitive detection of pathogenic bacteria using magnetic nano particles and optical nanocrystal probes. Analyst. 2012;137:3609–3612. doi: 10.1039/C2AN35369E. [DOI] [PubMed] [Google Scholar]
- Jung WK, Koo HC, Kim KW, et al. Antibacterial activity and mechanism of action of the silver ion in Staphylococcus aureus and Escherichia coli. Appl Environ Microbiol. 2008;74:2171–2178. doi: 10.1128/AEM.02001-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kang S, Herzberg M, Rodrigues DF, Elimelech M. Antibacterial effects of carbon nanotubes: size does matter! Langmuir. 2008;24(13):6409–6413. doi: 10.1021/la800951v. [DOI] [PubMed] [Google Scholar]
- Khot LR, Sankaran S, Maja JM, Ehsani R, Schuster EW. Applications of nanomaterials in agricultural production and crop protection: a review. Crop Prot. 2012;35:64–70. doi: 10.1016/j.cropro.2012.01.007. [DOI] [Google Scholar]
- Kim YH, Lee DK, Cha HG, et al. Preparation and characterization of the antibacterial Cu nanoparticle formed on the surface of SiO2 nanoparticles. J Phy ChemB. 2006;49:24923–24928. doi: 10.1021/jp0656779. [DOI] [PubMed] [Google Scholar]
- Kim JS, Kuk E, Yu KN, et al. Antimicrobial effects of silver nanoparticles. Nanomedicine. 2007;3:95–101. doi: 10.1016/j.nano.2006.12.001. [DOI] [PubMed] [Google Scholar]
- Kim KJ, Sung WS, Moon SK, et al. Antifungal effect of silver nanoparticles on dermatophytes. J Microbiol Biotechnol. 2008;18:1482–1484. doi: 10.1016/j.nano.2006.12.001. [DOI] [PubMed] [Google Scholar]
- Kim KJ, Sung WS, Suh BK, et al. Antifungal activity and mode of action of silver nano-particles on Candida albicans. Biometals. 2009;22:235–242. doi: 10.1007/s10534-008-9159-2. [DOI] [PubMed] [Google Scholar]
- Lakshminarayanan R, Ye E, Young DJ, Li Z, Loh XJ. Recent advances in the development of antimicrobial nanoparticles for combating resistant pathogens. Adv Healthc Mater. 2018 doi: 10.1002/adhm.201701400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lamsal K, Kim SW, Jung JH, et al. Application of silver nanoparticles for the control of Colletotrichum species in vitro and pepper anthracnose disease in field. Mycobiology. 2011;39(3):194–199. doi: 10.5941/MYCO.2011.39.3.194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lemire JA, Harrison JJ, Turner RJ. Antimicrobial activity of metals. Nat Rev Microbiol. 2013;11(6):71–84. doi: 10.1038/nrmicro3028. [DOI] [PubMed] [Google Scholar]
- Li L, Li B, Cheng D, Mao L. Visual detection of melamine in raw milk using gold nanoparticles as colorimetric probe. Food Chem. 2010;122(3):895–900. doi: 10.1016/j.foodchem.2010.03.032. [DOI] [Google Scholar]
- Liao JY, Li H. Lateral flow immunodipstick for visual detection of aflatoxin B1 in food using immuno-nanoparticles composed of a silver core and a gold shell. Microchim Acta. 2010;171:289. doi: 10.1007/s00604-010-0431-0. [DOI] [Google Scholar]
- Lili H, Yang L, Mustapha A, Lin M. Antifungal activity of zinc oxide nanoparticles against Botrytis cinerea and Penicillium expansum. Microbiol Res. 2011;166:207–215. doi: 10.1016/j.micres.2010.03.003. [DOI] [PubMed] [Google Scholar]
- Lok CN, Ho CM, Chen R, et al. Proteomic analysis of the mode of antibacterial action of silver nanoparticles. J Proteome Res. 2006;5:916–924. doi: 10.1021/pr0504079. [DOI] [PubMed] [Google Scholar]
- Lundqvist M, Stigler J, Elia G, Lynch I, Cedervall T, Dawson KA. Nanoparticle size and surface properties determine the protein corona with possible implications for biological impacts. Proc Natl Acad Sci. 2008;105(38):14265–14270. doi: 10.1073/pnas.0805135105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mahdi SS, Vadood R, Nourdahr R. Study on the antimicrobial effect of nanosilver tray packaging of minced beef at refrigerator temperature. Glob Vet. 2012;9(3):284–289. doi: 10.5829/idosi.gv.2012.9.3.1827. [DOI] [Google Scholar]
- Mallmann EJJ, Cunha FA, Castro BNMF, et al. Antifungal activity of silver nanoparticles obtained by green synthesis. Rev Inst Med Trop Sao Paulo. 2015;57(2):165–167. doi: 10.1590/S0036-46652015000200011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McQuillan J (2010) Bacterial-nanoparticle interactions. Thesis for the degree of Doctor of Philosophy in Biological Sciences. University of Exeter
- McQuillan JS, Shaw AM. Differential gene regulation in the Ag nanoparticle and Ag+-induced silver stress response in Escherichia coli: a full transcriptomic profile. Nanotoxicology. 2014;8(sup1):177–184. doi: 10.3109/17435390.2013.870243. [DOI] [PubMed] [Google Scholar]
- Mendes JE, Abrunhosa L, Teixeira JA, et al. Antifungal activity of silver colloidal nanoparticles against phytopathogenic fungus (Phomopsis sp.) in soybean seeds. Int J Biol Vet Agri Food Eng. 2014;8(9):928–933. doi: 10.5281/zenodo.1095981. [DOI] [Google Scholar]
- Metak AM, Ajaal TT. Investigation on polymer based nano-silver as food packaging materials. Int J Biol Food Vet Agric Eng. 2013;7:772–777. [Google Scholar]
- Mihindukulasuriya SDF, Lim LT. Nanotechnology development in food packaging: a review. Trends Food Sci Technol. 2014;40(2):149–167. doi: 10.1016/j.tifs.2014.09.009. [DOI] [Google Scholar]
- Morones JR, Elechiguerra JL, Camacho A, et al. The bactericidal effect of silver nanoparticles. Nanotechnology. 2005;16:2346–2353. doi: 10.1088/0957-4484/16/10/059. [DOI] [PubMed] [Google Scholar]
- Mueller CF, Laude K, McNally JS, Harrison DG. Redox mechanisms in blood vessels. Arterioscler Thromb Vasc Biol. 2005;25(2):274–278. doi: 10.1161/01.ATV.0000149143.04821.eb. [DOI] [PubMed] [Google Scholar]
- Mukherjee P, Ahmad A, Mandal D, et al. Fungus-mediated synthesis of silver nanoparticles and their immobilization in the mycelial matrix: a novel biological approach to nanoparticle synthesis. Nano Lett. 2001;1(10):515–519. doi: 10.1021/nl0155274. [DOI] [Google Scholar]
- Murphy C, Carroll C, Jordan KN. Environmental survival mechanisms of the foodborne pathogen Campylobacter jejuni. J Appl Microbiol. 2006;100:623–632. doi: 10.1111/j.1365-2672.2006.02903.x. [DOI] [PubMed] [Google Scholar]
- Musee N, Thwala M, Nota N. The antibacterial effects of engineered nanomaterials: implications for wastewater treatment plants. J Environ Monit. 2011;13(5):1164–1183. doi: 10.1039/c1em10023h. [DOI] [PubMed] [Google Scholar]
- Nagy A, Harrison A, Sabbani S, et al. Silver nanoparticles embedded in zeolite membranes: release of silver ions and mechanism of antibacterial action. Int J Nanomed. 2011;6:1833. doi: 10.2147/IJN.S24019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nair S, Sasidharan A, Rani VVD, et al. Role of size scale of ZnO nanoparticles and microparticles on toxicity toward bacteria and osteoblast cancer cells. J Mater Sci Mater Med. 2009;20:S235–S241. doi: 10.1007/s10856-008-3548-5. [DOI] [PubMed] [Google Scholar]
- Nasrollahi A, Pourshamsian KH, Mansourkiaee P. Antifungal activity of silver nanoparticles on some of fungi. Int J Nano Dimens. 2011;1(3):233–239. doi: 10.7508/ijnd.2010.03.007. [DOI] [Google Scholar]
- Navarro E, Baun A, Behra R, et al. Environmental behavior and ecotoxicity of engineered nanoparticles to algae, plants, and fungi. Ecotoxicology. 2008;17(5):372–386. doi: 10.1007/s10646-008-0214-0. [DOI] [PubMed] [Google Scholar]
- Navya PN, Daima HK. Rational engineering of physicochemical properties of nanomaterials for biomedical applications with nanotoxicological perspectives. Nano Converg. 2016;3:1. doi: 10.1186/s40580-016-0064-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Noh HJ, Kim HS, Cho S, Park Y. Melamine nanosensing with chondroitin sulfate-reduced gold nanoparticles. J Nanosci Nanotechnol. 2013;12(13):8229–8238. doi: 10.1166/jnn.2013.7926. [DOI] [PubMed] [Google Scholar]
- Noorbakhsh F, Rezaie S, Shahverdi AR. International conference on bioscience, biochemistry and bioinformatics. Singapore: IACSIT Press; 2011. Antifungal effects of silver nanoparticle alone and with combination of antifungal drug on dermatophyte pathogen Trichophyton rubrum; pp. 364–367. [Google Scholar]
- Ocsoy I, Paret ML, Ocsoy MA, et al. Nanotechnology in plant disease management: DNA-directed silver nanoparticles on graphene oxide as an antibacterial against Xanthomonas perforans. ACS Nano. 2013;7(10):8972–8980. doi: 10.1021/nn4034794. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pal S, Tak YK, Song JM. Does the antibacterial activity of silver nanoparticles depend on the shape of the nanoparticle? A study of the Gram-negative bacterium Escherichia coli. Appl Environ Microbiol. 2007;73(6):1712–1720. doi: 10.1128/AEM.02218-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Panácek A, Kolár M, Vecerová R, et al. Antifungal activity of silver nanoparticles against Candida spp. Biomaterials. 2009;30:6333–6340. doi: 10.1016/j.biomaterials.2009.07.065. [DOI] [PubMed] [Google Scholar]
- Pantopoulos K, Schipper HM. Principles of free radical biomedicine. Hauppauge: Nova Science Publications; 2012. [Google Scholar]
- Park SS, Worobo RW, Durst RA. Escherichia coli O157: H7 as an emerging foodborne pathogen: a literature review. Crit Rev Biotechnol. 2001;21:27–48. doi: 10.1080/10408699991279259. [DOI] [PubMed] [Google Scholar]
- Park J, Kim JY, Kim J, et al. Silver-ion mediated reactive oxygen species generation affecting bactericidal activity. Water Res. 2009;43:1027–1032. doi: 10.1016/j.watres.2008.12.002. [DOI] [PubMed] [Google Scholar]
- Patel N, Desa P, Pael N, Jha A, Gautam HK. Agronatechlogy for plant fungal disease management: a review. Int J Cur Micobl Appl Sci. 2014;3(10):71–84. [Google Scholar]
- Paula MMDS, Franco CV, Baldin MC, et al. Synthesis, characterization and antibacterial activity studies of poly-{styrene-acrylic acid} with silver nanoparticles. Mater Sci Eng C. 2009;29(2):647–650. doi: 10.1016/j.msec.2008.11.017. [DOI] [Google Scholar]
- Pelletier DA, Suresh AK, Holton GA, et al. Effects of engineered cerium oxide nanoparticles on bacterial growth and viability. Appl Environ Microbiol. 2010;76(24):7981–7989. doi: 10.1128/AEM.00650-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pfaller MA, Diekema DJ. Epidemiology of invasive candidiasis: a persistent public health problem. Clin Microbiol Rev. 2007;20:133–163. doi: 10.1128/CMR.00029-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pimbley DW, Patel PD. A review of analytical methods for the detection of bacterialtoxins. J Appl Microbiol. 1998;84:98S–109S. doi: 10.1046/j.1365-2672.1998.0840s198S.x. [DOI] [PubMed] [Google Scholar]
- Poonlapdecha W, Seetang-Nun Y, Wonglumsom W, et al. Antibody-conjugated ferromagnetic nanoparticles with lateral flow test strip assay for rapid detection of Campylobacter jejuni in poultry samples. Int J Food Microbiol. 2018;286:6–14. doi: 10.1016/j.ijfoodmicro.2018.07.009. [DOI] [PubMed] [Google Scholar]
- Pradhan N, Singh S, Ojha N, Shrivastava A, et al. Facets of nanotechnology as seen in food processing, packaging, and preservation industry. Biomed Res Int. 2015 doi: 10.1155/2015/365672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Prasad VK, Prasad KS. Nanotechnology in sustainable agriculture: present concerns and future aspects. Afr J Biotechnol. 2014;13(6):706–713. doi: 10.5897/AJBX2013.13554. [DOI] [Google Scholar]
- Priyanka B, Patil RK, Dwarakanath S. A review on detection methods used for foodborne pathogens. Indian J Med Res. 2016;144(3):327. doi: 10.4103/0971-5916.198677. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Prucek R, Tucek J, Kilianová M, et al. The targeted antibacterial and antifungal properties of magnetic nanocomposite of iron oxide and silver nanoparticles. Biomaterials. 2011;32:4704–4713. doi: 10.1016/j.biomaterials.2011.03.039. [DOI] [PubMed] [Google Scholar]
- Radzig MA, Nadtochenko VA, Koksharova VA, et al. Antibacterial effects of silver nanoparticles on gram-negative bacteria: Influence on the growth and biofilms formation, mechanisms of action. Colloids Surf B Biointerfaces. 2013;102:300–306. doi: 10.1016/j.colsurfb.2012.07.039. [DOI] [PubMed] [Google Scholar]
- Rai M, Ingle A. Role of nanotechnology in agriculture with special reference to management of insect pests. Appl Microbiol Biotechnol. 2012;94(2):287–293. doi: 10.1007/s00253-012-3969-4. [DOI] [PubMed] [Google Scholar]
- Rai M, Ingle A. Role of nanotechnology in agriculture with special reference to management of insect pests. Appl Microbiol Biotechnol. 2012;94:287–293. doi: 10.1007/s00253-012-3969-4. [DOI] [PubMed] [Google Scholar]
- Rajeshkumar S, Bharath LV. Mechanism of plant-mediated synthesis of silver nanoparticles–a review on biomolecules involved, characterisation and antibacterial activity. Chem-Biol Interact. 2017;273:219–227. doi: 10.1016/j.cbi.2017.06.019. [DOI] [PubMed] [Google Scholar]
- Rispail N, De Matteis L, Santos R, et al. Quantum dot and superparamagnetic nanoparticle interaction with pathogenic fungi: internalization and toxicity profile. ACS Appl Mater Interfaces. 2014;6(12):9100–9110. doi: 10.1021/am501029g. [DOI] [PubMed] [Google Scholar]
- Sastry M, Ahmad A, Khan MI, et al. Biosynthesis of metal nanoparticles using fungi and actinomycete. Curr Sci. 2003;85(2):162–170. [Google Scholar]
- Sekhon BS. Food nanotechnology—an overview. Nanotechnol Sci Appl. 2010;4(3):1–15. [PMC free article] [PubMed] [Google Scholar]
- Shankar SS, Rai A, Ahmad A, et al. Rapid synthesis of Au, Ag, and bimetallic Au core–Ag shell nanoparticles using Neem (Azadirachta indica) leaf broth. J Colloid Interface Sci. 2004;275(2):496–502. doi: 10.1016/j.jcis.2004.03.003. [DOI] [PubMed] [Google Scholar]
- Silver S, Phung LT. Bacterial heavy metal resistance: new surprises. Annu Rev Microbiol. 1996;50:753–789. doi: 10.1146/annurev.micro.50.1.753. [DOI] [PubMed] [Google Scholar]
- Silver S, Phung le T, Silver G. Silver as biocides in burn and wound dressings and bacterial resistance to silver compounds. J Ind Microbiol Biotechnol. 2006;33:627–634. doi: 10.1007/s10295-006-0139-7. [DOI] [PubMed] [Google Scholar]
- Singh S, Tripathi DK, Dubey NK, Chauhan DK. Effects of nano-materials on seed germination and seedling growth: striking the slight balance between the concepts and controversies. Mater Focus. 2016;5(3):195–201. doi: 10.1166/mat.2016.1329. [DOI] [Google Scholar]
- Sondi I, Salopek-Sondi B. Silver nanoparticles as antimicrobial agent: a case study on Escherichia coli as a model for Gram-negative bacteria. J Colloid Interface Sci. 2004;275(1):177–182. doi: 10.1016/j.jcis.2004.02.012. [DOI] [PubMed] [Google Scholar]
- Stoimenov P, Klinger R, Marchin GL, Klabunde KJ. Metal oxide nanoparticles as bactericidal agents. Langmuir. 2002;18(17):6679–6686. doi: 10.1021/la0202374. [DOI] [Google Scholar]
- Sung YJ, Suk HJ, Sung HY, Li T, et al. Novel antibody/gold nanoparticle/magnetic nanoparticle nanocomposites for immunomagnetic separation and rapid colorimetric detection of Staphylococcus aureus in milk. Biosens Bioelectron. 2013;43:432–439. doi: 10.1016/j.bios.2012.12.052. [DOI] [PubMed] [Google Scholar]
- Suresh AK, Pelletier DA, Doktycz MJ. Relating nanomaterial properties and microbial toxicity. Nanoscale. 2013;5(2):463–474. doi: 10.1039/C2NR32447D. [DOI] [PubMed] [Google Scholar]
- Toker RD, Kayaman-Apohan N, Kahraman MV. UV-curable nano-silver containing polyurethane based organic–inorganic hybrid coatings. Prog Org Coat. 2013;76(9):1243–1250. doi: 10.1016/j.porgcoat.2013.03.023. [DOI] [Google Scholar]
- Touyz RM. Molecular and cellular mechanisms in vascular injury in hypertension: role of angiotensin II–editorial review. Curr Opin Nephrol Hypertens. 2005;14(2):125–131. doi: 10.1097/00041552-200503000-00007. [DOI] [PubMed] [Google Scholar]
- Tripathi DK, Singh VP, Prasad SM, Chauhan DK, Dubey NK. Silicon nanoparticles (SiNp) alleviate chromium (VI) phytotoxicity in Pisum sativum (L.) seedlings. Plant Physiol Biochem. 2015;96:189–198. doi: 10.1016/j.plaphy.2015.07.026. [DOI] [PubMed] [Google Scholar]
- Tripathi DK, Tripathi A, Gaur S, et al. Uptake, accumulation and toxicity of silver nanoparticle in autotrophic plants, and heterotrophic microbes: a concentric review. Front Microbiol. 2017;8:7. doi: 10.3389/fmicb.2017.00007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tripathi DK, Singh S, Singh VP, Prasad SM, Dubey NK, Chauhan DK. Silicon nanoparticles more effectively alleviated UV-B stress than silicon in wheat (Triticum aestivum) seedlings. Plant Physiol Biochem. 2017;110:70–81. doi: 10.1016/j.plaphy.2016.06.026. [DOI] [PubMed] [Google Scholar]
- Valipoor Motlagh N, Hamed Mosavian MT, Mortazavi SA. Effect of polyethylene packaging modified with silver particles on the microbial, sensory and appearance of dried barberry. Packag Technol Sci. 2013;26(1):39–49. doi: 10.1002/pts.1966. [DOI] [Google Scholar]
- Valko M, Rhodes C, Moncol J, Izakovic MM, Mazur M. Free radicals, metals and antioxidants in oxidative stress-induced cancer. Chem Biol Interact. 2006;160(1):1–40. doi: 10.1016/j.cbi.2005.12.009. [DOI] [PubMed] [Google Scholar]
- Van Aken B. Gene expression changes in plants and microorganisms exposed to nanomaterials. Curr Opin Biotechnol. 2015;33:206–219. doi: 10.1016/j.copbio.2015.03.005. [DOI] [PubMed] [Google Scholar]
- Vecitis CD, Zodrow KR, Kang S, Elimelech M. Electronic-structure-dependent bacterial cytotoxicity of single-walled carbon nanotubes. ACS Nano. 2010;4(9):5471–5479. doi: 10.1021/nn101558x. [DOI] [PubMed] [Google Scholar]
- Vishwakarma K, Shweta, Upadhyay N, Singh J, Liu S, Singh VP, Prasad SM, Chauhan DK, Tripathi DK, Sharma S. Differential Phytotoxic Impact of Plant Mediated Silver Nanoparticles (AgNPs) and Silver Nitrate (AgNO3) on Brassica sp. Front Plant Sci. 2017;8:1501. doi: 10.3389/fpls.2017.01501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Viswanathan S, Wu LC, Huang MR, Ho JA. Electrochemical immunosensor for cholera toxin using liposomes and poly (3,4-ethylenedioxythiophene)-coated carbon nanotubes. Anal Chem. 2006;78:1115–1121. doi: 10.1021/ac051435d. [DOI] [PubMed] [Google Scholar]
- Woo KS, Kim KS, Lamsal K, et al. An in vitro study of the antifungal effect of silver nanoparticles on oak wilt pathogen Raffaelea sp. J Microbiol Biotechnol. 2009;19:760–764. doi: 10.4014/jmb.0812.649. [DOI] [PubMed] [Google Scholar]
- Wu S, Duan N, Zhu C, et al. Magnetic nanobead-based immunoassay for the simultaneous detection of aflatoxin B1 and ochratoxin A using upconversion nanoparticles as multicolor labels. Biosens Bioelectron. 2011;30(1):35–42. doi: 10.1016/j.bios.2011.08.023. [DOI] [PubMed] [Google Scholar]
- Wu H, Yin JJ, Wamer WG, Zeng M, Lo YM. Reactive oxygen species-related activities of nano-iron metal and nano-iron oxides. J Food Drug Anal. 2014;22(1):86–94. doi: 10.1016/j.jfda.2014.01.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu S, Duan N, Gu H, Hao L, et al. A review of the methods for detection of Staphylococcus aureus enterotoxins. Toxins. 2016;8(7):176. doi: 10.3390/toxins8070176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu H, et al. Role of reactive oxygen species in the antibacterial mechanism of silver nanoparticles on Escherichia coli O157:H7. Biometals. 2012;25:45–53. doi: 10.1007/s10534-011-9482-x. [DOI] [PubMed] [Google Scholar]
- Yang Y, Mathieu JM, Chattopadhyay S, et al. Defense mechanisms of Pseudomonas aeruginosa PAO1 against quantum dots and their released heavy metals. ACS Nano. 2012;6(7):6091–6098. doi: 10.1021/nn3011619. [DOI] [PubMed] [Google Scholar]
- Yoon KY, Byeon JH, Park JH, Hwang J. Susceptibility constants of Escherichia coli and Bacillus subtilis to silver and copper nanoparticles. Sci Total Environ. 2007;373(2–3):572–575. doi: 10.1016/j.scitotenv.2006.11.007. [DOI] [PubMed] [Google Scholar]
- Zhang L, Jiang Y, Ding Y, Povey M, York D. Investigation into the antibacterial behavior of suspensions of ZnO nanoparticles (ZnO nanofluids) J Nanoparticle Rev. 2007;9(3):479–489. doi: 10.1007/s11051-006-9150-1. [DOI] [Google Scholar]