Abstract
Maize originated as a tropical plant that required short days to transition from vegetative to reproductive development. ZmCCT10 [CO, CONSTANS, CO-LIKE and TIMING OF CAB1 (CCT) transcription factor family] is a regulator of photoperiod response and was identified as a major QTL controlling photoperiod sensitivity in maize. We modulated expression of ZmCCT10 in transgenic maize using two constitutive promoters with different expression levels. Transgenic plants over expressing ZmCCT10 with either promoter were delayed in their transition from vegetative to reproductive development but were not affected in their switch from juvenile-to-adult vegetative growth. Strikingly, transgenic plants containing the stronger expressing construct had a prolonged period of vegetative growth accompanied with dramatic modifications to plant architecture that impacted both vegetative and reproductive traits. These plants did not produce ears, but tassels were heavily branched. In more than half of the transgenic plants, tassels were converted into a branched leafy structure resembling phyllody, often composed of vegetative plantlets. Analysis of expression modules controlling the floral transition and meristem identity linked these networks to photoperiod dependent regulation, whereas phase change modules appeared to be photoperiod independent. Results from this study clarified the influence of the photoperiod pathway on vegetative and reproductive development and allowed for the fine-tuning of the maize flowering time model.
Introduction
Plants display an astonishing diversity in body plan architecture [1]. However much of the complexity of plant morphology can be explained by variations of the basic architectural unit–the phytomer. The phytomer is comprised of the leaf, attached to the node on the stem, the internode, the stem segment between two successive nodes, and the axillary bud positioned in the axil of the leaf [1]. The number of phytomers is determined by the activity of the shoot apical meristem (SAM), or the apical bud, which contains a pool of undifferentiated pluripotent stem cells capable of producing the above ground organs [2]. The axillary bud is also composed of meristematic cells which initiate the growth of side branches [3]. The SAM represses the outgrowth of the axillary bud to control the extent of branching which is known as apical dominance [4].
Maize is an annual grass with a determinate habit of growth. The main stalk of the mature plant is composed of a variable number of phytomers depending on genotype. The apex of the plant terminates when the SAM converts into the male inflorescence, called the tassel. The female inflorescence, the ear, is initiated from axillary buds. These buds form in leaf axils but usually only the top one or two buds on the plant develop into ears and bear seed; whereas the ears positioned lower on the shoot abort [5].
After germination, maize seedlings transition through distinct developmental phases. Vegetative development is divided into two phases—juvenile and adult [6]. Seedlings in the juvenile phase display characteristic traits such as the presence of adventitious roots, short internodes, and narrow leaves [7]. Juvenile leaves also have a number of distinctive epidermal traits including weakly invaginated cell walls, epicuticular waxes, the absence of trichomes (macrohairs) and the presence of bulliform cells [7–9]. The transition from the juvenile to adult phase is regulated by the balance of two micro-RNAs, miR156 and miR172, where miR156 promotes the juvenile phase and miR172 promotes the adult phase [10, 11].
High expression of miRNA156 maintains the juvenile phase by repression of SQUAMOSA PROMOTER BINDING PROTEIN (SBP/SPL) genes, which encode plant-specific transcription factors required for development of adult traits [11, 12]. In maize, over-expression of miR156 results in the striking grass-like phenotype of the Corngrass1 (Cg1) mutant [12]. This mutant retains juvenile traits such as short internodes, multiple tillers, slender culms, an increased number of vegetative nodes, adventitious roots, narrow leaves with epicuticular wax and the absence of trichomes [13]. Transcripts of at least seven SBP/SPL genes are down regulated in the Cg1 mutant including teosinte glume architecture1 (tga1), a SBP transcription factor involved in maize domestication [12]. As seedlings grow, expression of miR156 declines allowing expression of SBP genes to increase, leading to the transition to adult growth [6, 10, 11].
By contrast, miR172 functions antagonistically to miR156 by repressing juvenile traits and accelerating the transition to the adult phase by down regulation of APETALA2-like (AP2) transcription factor genes [10, 11]. The key regulator of the juvenile-to-adult transition in maize is the AP2-like GLOSSY15 (GL15) gene [7, 14]. Over-expression of miR172 degrades GL15 mRNA that results in an accelerated transition to the adult phase [15]. Genetically, GL15 functions downstream of Cg1 [7, 14], which is consistent with molecular data showing expression of miR172 transcripts are reduced in Cg1 mutants [12].
After the juvenile-to-adult phase change, plants acquire the competence for reproductive development. The switch from adult vegetative growth to reproductive growth is called the floral transition. During the floral transition, the SAM ceases leaf initiation and is transformed into an inflorescence meristem (IM). In maize, this is marked by the SAM becoming committed to tassel development [16]. The total number of leaves produced is often used as a quantitative measurement of the length of the vegetative stage of growth. The transition to reproductive development is regulated by numerous environmental and endogenous cues that stimulate accumulation of the flowering hormone florigen in leaves. Florigen is transmitted from leaves to the shoot apical meristem through the phloem where the transition to reproductive development occurs [17–20].
Florigen was identified in Arabidopsis as the 23 kD protein encoded by the FLOWERING locus T (FT) gene [17, 21]. Afterwards, homologs of the FT gene were detected in virtually all plants and the FT protein is postulated to be a universal florigen [18, 22, 23]. However, the discovery of one type of florigen does not exclude the possibility of others. In the SAM, FT interacts with a 14-3-3 receptor protein and the bZIP transcription factor FLOWERING LOCUS D (FD) forming the florigen activation complex (FAC) as demonstrated in rice [24, 25]. Formation of the FAC seems to be a universal feature in flowering plants but direct evidence of a FAC beyond rice is lacking [26]. The FAC activates transcription of the APETELA 1 (AP1)-like MADS box genes which marks the onset of reproductive development [24].
The FT-FD genetic module is conserved in maize. The DELAYED FLOWERING1 (DLF1) gene encodes an FD-like bZIP transcription factor which mediates floral signals in the shoot apex [27]. The expanded family of FT-like genes in maize were named Zea CENTRORADIALIS (ZCN) reflecting their functional diversification [28]. A florigenic function was shown for the ZCN8 gene [29, 30], which has recently been shown to contribute to adaptation to flowering at higher latitudes [31]. Its nearly identical paralog ZCN7 is thought to also possess florigenic activity [32]. The flowering regulator, Zn-finger transcription factor INDETERMINATE1 (ID1) [33] controls expression of ZCN8 and ZCN7 [29, 31]. Although it is not clear how ID1 regulates ZCN7 and ZCN8, recent data suggests it may be via epigenetic modification of their chromatin structure [32]. It is likely that the FAC is also formed in maize because the ZCN8 protein was shown to interact with the DLF1 protein [30]. Similar to Arabidopsis and rice, the onset of reproductive development in maize is demarcated by expression of the AP1-like MADS box genes ZMM4 and ZMM15 [34], which supports a conserved developmental genetic pathway between these species.
Maize was domesticated from the tropical grass teosinte (Zea mays ssp. parviglumis) that requires short days to flower [35]. Over time as maize cultivation moved to higher latitudes with longer summer days, selection for short day sensitivity was significantly weakened [36]. There is wide variation in the photoperiod sensitivity among maize inbred lines ranging from complete day length insensitivity, to moderately and highly sensitive groups [37]. Day neutral lines produce equal number of leaves under both short (SD) and long (LD) days. Gaspé Flint, which is the earliest flowering cultivar of maize, produces on average 10 leaves under both conditions. By contrast, tropical lines can be day-length sensitive, producing up to 30 leaves under LDs compared to 23 leaves under SDs [37]. Thus, tropical maize can be considered facultative SD plants because while SDs accelerate their flowering, they still flower under non-inductive LDs, although flowering is delayed.
Using a maize-teosinte mapping population and genome-wide association a major photoperiod response regulator the ZmCCT10 gene, was identified on chromosome 10 [38]. The ZmCCT10 gene encodes a CCT (CO, CO-LIKE and TIMING OF CAB1) domain protein. Further studies revealed the insertion of a CACTA-like transposon in the upstream promoter regions of the ZmCCT10 gene in day-neutral temperate lines which presumably disrupts ZmCCT10 expression and attenuates photoperiod sensitivity under LD conditions [39]. Recently, the ZmCCT9 gene, on chromosome 9, was also identified to control flowering similar to ZmCCT10 [40].
The maize ZmCCT10 is a homolog of the rice photoperiod response regulator Ghd7, which was identified as a quantitative trait locus (QTL) for Grains, plant height and heading date on chromosome 7 [38, 41]. Ghd7 is a negative regulator of flowering (heading date) [41, 42] and is expressed in leaves with a diurnal pattern, peaking in the early morning under LDs. In contrast, its expression is low under SDs [41]. The GHD7 protein represses transcription of the floral inducer Early Heading Date1 (Ehd1) which promotes flowering under SDs [43]. EHD1 is a B-type response regulator with DNA binding properties suggesting it functions as a transcription factor [43]. Ehd1 is required for expression of the rice florigen genes Heading Date3a (Hd3a) [23] and Rice flowering locus T1 (RTF1), major floral activators under LD conditions [44, 45]. The floral promoter Ehd1 and the floral repressor Ghd7 fine tune the expression of the Hd3a/RFT1 genes to recognize a critical day length for transition to reproductive development [46].
The Ghd7-Ehd1 genetic module seems to be conserved in other short day tropical grass crops as was demonstrated for sorghum [47, 48] and also for maize [39, 49]. In the long day winter crops wheat and barley, VERNALIZATION2 (VRN2), a homolog of Ghd7, represses FT-like genes prior to cold exposure [50, 51]. But the Ghd7- Ehd1 module is not present in Arabidopsis or other eudicots [41, 52].
Ghd7 over-expression or down-regulation in transgenic plants revealed its role as a central regulator of growth, development and stress response in rice [42]. Over-expression of Ghd7 affected plant architecture resulting in taller plants with thick stems, fewer tillers but increased panicle branching which led to more grain per plant [41, 42]. Ghd7 is one of the major targets for increasing grain yield in rice breeding programs [53].
Our knowledge of how the ZmCCT10 gene functions at the molecular level is limited [39]. However, being a repressor of the photoperiod pathway, manipulation of ZmCCT10 expression provides an opportunity to study the role of this pathway in floral transition and other developmental processes. To investigate these roles, we over-expressed ZmCCT10 driven by constitutive promoters of different strengths in the day neutral early-flowering maize line Gaspé Flint. The resulting transgenic plants displayed dramatic modification of plant morphology producing tall, late flowering phenotypes with about 50% of the events showing vegetative reversion of the tassel to a branched, leafy structure (phyllody).
Methods
Plant materials
Extremely early temperate cultivar Gaspé Flint, temperate inbred line B73, tropical CML436 and CML311maize lines were used due to their distinct photoperiod sensitivities and differences in flowering times. Diurnal experiment using these lines was described in [30]. Teosinte lines were obtained from North Central Regional Plant Introduction Station, Ames, Iowa, USA. Teosinte accession# PI 441934 is Zea mays sp huehuetenangensis originally from Guatemala. Teosinte accession # PI 422162 is Zea luxurians originally from Mexico.
T-DNA constructs and plant transformation
GATEWAYTECHNOLGY (Invitrogen, CA) was used for vector construction. The co-integrated vectors were constructed, and maize Gaspé Flint plants were transformed as described previously [30, 54]. Typically, 10 independent single copy events were generated for each construct. T1 seeds were generated by pollination with Gaspé Flint as a pollen donor.
CS184 (ft-2) and CS185(ft-3), the Arabidopsis later flowering mutants in the ectotype Lansberg erecta, were obtained from ABRC (Columbus, OH). Plants were grown under fluorescent lamps in growth chambers with 16 hr light period at 24°C and 8 hr dark period at 23°C and 50% relative humidity. Gateway technology (Invitrogen, CA) was used to construct transformation vectors. The binary vector, pBC.Yellow, was used for Arabidopsis transformation [55]. Using the flower dipping method, plants of Ler WT, CS-184 and CS-185 were inoculated by the Agrobacterium strain GV3101. Genes transformed into the binary vector was constitutively expressed under control of the promoter CaMV35S. Positive transgenic lines were identified by color selection from yellow fluorescent protein (YFP) and herbicide selection from the BAR gene. Seeds of at least three positive transgenic lines for every construct were saved for further study.
Phenotypic data collection
Vegetative growth stages (V stage) were defined according to the appearance of the leaf collar of the uppermost leaf [56]. The staging notes were taken twice a week. Using staging notes leaf appearance rate was calculated with linear regression models. Growth rate calculated as (HF-H1)/(DAPF-DAP1) where H—height, F—final height, 1—first recorded measurement, where DAP stands for days after planting. Leaf length was measured on fully expanded leaves with a visible ligule as the distance from the leaf collar/ligule to the tip of the blade. Leaf width was measured at the widest point of the blade. The ratio of the length to the width was calculated. Nodes were identified by the leaf number originating from that node. Internode lengths were measured when fully mature plants were harvested and were calculated as the distance between nodes. Only above-ground internodes were measured.
Toluidine blue O staining of epidermal peels
To determine when plants switched from juvenile to adult growth, leaf sections were collected from all leaves starting with leaf # 2 until adult traits were detected. Leaf segments from margin to margin were collected from the base and the tip of each leaf. Segments were fixed in a mixture of 1-part ethanol to 3 parts acetic acid [57]. The abaxial epidermis and mesophyll were removed using abrasive techniques. Once cleared, the adaxial epidermis was stained with 0.05% toluidine blue O/acetic acid, pH 4.5 (TBO solution) [7, 58] for 30 seconds. The peel was then rinsed with deionized water and immediately photographed using bright-field optics on a microscope.
Tissue collection for qRT-PCR
Plants were grown in the greenhouse under LD conditions with a 14-hour day length. To cover much of the developmental range of candidate gene expression, coleoptiles were sampled from NTG (non-transgenic plants) and UBIpro:ZmCCT10 transgenic plants but not from the BSVpro:ZmCCT10 plants due to limited seed availability. Afterwards, the first true leaf was sampled, and then leaves were sampled (with a punch) twice a week beginning from leaf # 4. Due to expression of the maize florigen gene ZCN8 at the tip of the leaf [30], leaves were consistently sampled 3–5 cm from the tip. To capture diurnal expression patterns, leaves were sampled in the morning when lights were turned on in the greenhouse and 12 hours later in the evening. When the BSVpro:ZmCCT10 plants reached 24 leaves, it was technically challenging to sample such tall plants and thus leaves 24–31 were sampled at the final dissection of the plants in the afternoon. Tissues collected for qRT-PCR were immediately flash frozen after being sampled. Additional NTG and UBIpro:ZmCCT10 transgenic plants were grown in a Conviron CMP6050 growth chamber under 14-hour days (28°C days and 26°C nights) to sample meristems. Meristems were dissected at every V stage starting at V1 until the meristem transitioned to a tassel (~V4 for NTG and ~V4 to V7 for UBIpro:ZmCCT10 transgenic plants depending on the CCT10 allele). Due to limited seed availability, BSVpro:ZmCCT10 plant meristems were sampled in the greenhouse under LD conditions with a 14-hour day length during the initial T1 experiment. Meristems were sampled at stages V6, V9, V11, V13, V17 and V19 from adult plants and from plantlets at stages V32 and V33. Images were taken of each meristem with a Leica MSV269 dissecting scope, then immediately flash frozen to be used for qRT-PCR.
RNA isolation and qRT-PCR
Total RNA was isolated using Qiagen RNeasy reagents (https://www.qiagen.com/us/shop/Lab-Basics/Buffers-and-Reagents/) with the nucleic acid bound to columns of a 384 well binding plate purchased from the Pall Corporation (http://www.pall.com/main/oem-materials-and-devices/product.page?lid=gri78l6g). DNA was removed from the RNA samples using Roche DNAse I Recombinant (https://www.lifescience.roche.com/shop/en/us/products/dnase-i-recombinant-rnase-free) and synthesis of cDNA was done using Applied Biosystems High Capacity cDNA Reverse Transcription kits (https://www.thermofisher.com/order/catalog/product/4368813?ICID=search-product). Quantitative PCR was done using hydrolysis probe and SYBR based reactions. Primers and probes were designed using Applied Biosystems Primer Express software (https://www.thermofisher.com/order/catalog/product/4363993?ICID=search-product) using nucleotide sequences published in Genbank. Hydrolysis probe-based PCR was performed using Bioline Sensi-fast mix (http://www.bioline.com/us/sensifast-probe-lo-rox-kit.html) while SYBR-based PCR was run using Applied Biosystems PowerUp SYBR Green Master Mix (https://www.thermofisher.com/order/catalog/product/A25741?ICID=search-product). All reactions were run on an Applied Biosystem Viia7 Real-Time PCR instrument using the manufacturer’s conditions. Relative gene expression was calculated by normalizing against maize eukaryotic initiation factor 4-gamma gene (GenBank accession # EU967723). A list of primers and probes are shown in S5 Table. To identify genes which expressions were different in transgenic apices compared to NTG plants, T-test was performed for every developmental stage. Expression level were considered statistically significant with p<0.05 (S6 Table).
Results
Over-expression of ZmCCT10 produces dramatic effects on multiple traits in T0 maize plants
To investigate how over expression of ZmCCT10 may impact flowering, we used a transgenic approach and chose to constitutively over-express the ZmCCT10 coding region. To explore how diverse allelic variation of the ZmCCT10 protein might differentially alter flowering, ZmCCT10 alleles from different maize inbred lines with distinct flowering characteristics were selected, including the day-length neutral early flowering Gaspé Flint, the temperate B73, and the SD-sensitive tropical CML436 and CML311 lines [30]. Two teosinte accessions, the SD-sensitive wild progenitor of maize, were also used (accessions PI 441934 and PI 422162). To identify conserved protein domains, maize ZmCCT10 and sorghum SbGHD7, the closest maize CCT10 homolog which functions as a floral repressor under long days, were compared [47]. Their amino acid alignment showed the proteins were conserved with 61% amino acid identity (S1 Fig).
To investigate how the level of ZmCCT10 expression could affect flowering, we used two constitutive promoters of different strengths to drive expression of six different alleles. The maize ubiquitin promoter (UBIpro) is widely used in cereal transgenic studies as a constitutive promoter with a high level of expression [59]. However, the Banana Streak Virus promoter (BSVpro) exceeds the level of expression driven by the UBIpro and directs expression in all tissue tested with exception of pollen [60]. Using both the UBIpro and BSVpro and the genomic and/or cDNA sequences of six ZmCCT10 alleles, we constructed a cohort of constructs that provided a wide range of allelic and expression combinations in which to study the effect of ZmCCT10 overexpression on plant phenotype (S1 Table).
All constructs were transformed into the day-neutral early flowering Gaspé Flint line (Fig 1A). Between 9–10 single copy transgenic events were generated for each construct. Because T0 transgenic plants were produced in batches, representative T0 data were collected for each experimental batch grown in the greenhouse at the same time (S1 Table).
Fig 1. Representative images of non-transgenic (NTG), UBIpro:ZmCCT10, and BSVpro:ZmCCT10 transgenic plants.
A) Image of a non-transgenic (NTG) Gaspé Flint line used for transformation. B) UBIpro:ZmCCT10 transgenic plant with a long ear shank (green arrow). C) BSVpro:ZmCCT10 transgenic plants at 119 days after planting and a Gaspé Flint parental line (red arrow). D) BSVpro:ZmCCT10 transgenic plants at 171 days after planting and a Gaspé Flint parental line (red arrow). Vegetative structures replace tassels in transgenic plants. E) A closeup view of extensive brace roots on a BSVpro:ZmCCT10 transgenic plant at the 15–20 internode region. F) A typical Gaspé Flint plant with brace roots only at the base of the plant.
As expected for over-expression of a flowering repressor, the transgenic plants exhibited a late flowering phenotype which was manifested as taller plants with more leaves compared to the non-transgenic (NTG) parental Gaspé Flint line (Fig 1B and 1C). Delayed flowering, recorded as time to pollen shed (shedding) and silk exertion (silking), was also observed (S1 Table). Typical time for Gaspe Flint to shed and silk is about 30 days.
In addition to the expected late flowering phenotype, the transgenic plants also displayed unexpected features. The UBIpro:ZmCCT10 T0 plants produced atypically long axillary branches subtending the ears (Fig 1B). Moreover, BSVpro:ZmCCT10 transgenic plants exhibited a severe modification to the entire plant architecture (Fig 1C and 1D). The BSVpro:ZmCCT10 transgenic plants were extremely tall, up to 300 cm, on average, and produced up to 38 leaves (Fig 1C and 1D and S2A Fig; S1 Table). The stalks of BSVpro:ZmCCT10 transgenic plants were strong yet flexible with increased mechanical strength and could withstand bending up to 80° (S3 Fig). Brace roots formed up to the 35th-37th nodes compared to the base internode of NTG plants (Fig 1E and 1F, S2B Fig). Secondary aerial brace roots developed as the plants aged (S2D Fig). In contrast, the non-transgenic Gaspé Flint line produced only a few brace roots at the base of the plant (Fig 1F). Unexpectedly, as plants matured, a vegetative, highly branched leafy structure emerged from the top of many of the BSVpro:ZmCCT10 plants in lieu of tassels (Fig 1C). More than 50% of the BSVpro:ZmCCT10 transgenic plants produced these vegetative apical structures—phyllody—(S2E Fig) and were composed of reversion plantlets. This phenotype was not observed in any of the NTG or UBIpro:ZmCCT10 plants.
Pleiotropic effects of ZmCCT10 are independent of allelic variations but are dosage dependent
We further examined ZmCCT10 overexpression effects in the T1 families. We selected 12 constructs composed of six cDNA ZmCCT10 alleles driven by either the UBIpro or BSVpro (S2 Table). The T1 families of 20 plants from two events were planted for the UBIpro:ZmCCT10 constructs. Due to the reduced fertility of the BSVpro:ZmCCT10 transgenic plants, the available T1 seed was limited and, in a few cases, only 3–4 plants were planted (S2 Table). Consistent with T0 observations, both UBIpro:ZmCCT10 and BSVpro:ZmCCT10 constructs modified plant morphology in the T1 plants, with the more extreme manifestations being observed in the BSVpro:ZmCCT10 plants. A weak allelic effect was detected in the UBIpro:ZmCCT10 constructs where UBIpro:ZmCCT10Gaspé and UBIpro:ZmCCT10B73 showed a smaller effect on plant height and leaf number. This allelic effect was not observed in the BSVpro:ZmCCT10 plants and we surmise that this lack of phenotypic effect may have been overridden by the very high level of transgene expression (S2 Table). Thus, we considered the level of transgene expression as the major factor correlated with phenotype severity. For this reason, we analyzed data by grouping the UBIpro:ZmCCT10 and BSVpro:ZmCCT10 results regardless of their allele background.
The T1 families showed a strong association between the level of transgene expression and trait measured. This occurred for plant height (S4A Fig), leaf number (S4B Fig), the uppermost node with brace roots (S4C Fig), and the primary ear position (S4D Fig). Higher levels of transgene expression resulted in enhancement of the trait modification. However, there was a threshold for transgene expression in the BSVpro:ZmCCT10 plants beyond which trait enhancement plateaued (S4 Fig).
Modification of vegetative traits in T1 maize transgenic plants
The T1 transgenic families exhibited modification of plant phenotypes consistent with the T0 generation. The average BSVpro:ZmCCT10 T1 plant height was 263 cm compared to 105 cm for the UBIpro:ZmCCT10 plants and ~ 70 cm for the NTG siblings (Table 1). The growth rate of the BSVpro:ZmCCT10 plants was only 2.8 cm/day compared to ~ 4.2 cm/day of the UBIpro:ZmCCT10 and NTG plants (Table 1). Despite their slower growth rate, the BSVpro:ZmCCT10 transgenic plants produced more internodes because they remained in a vegetative stage longer, which also resulted in the increased plant height.
Table 1. Vegetative traits collected for T1 families 1.
| Line | Total No. Plants | Relative Expression of ZmCCT10 qRT-PCR | Plant Height (cm) | Growth rate (cm/day) | Leaf No. | Leaf appearance rate (leaves/day) | Top Nodes with brace roots | Nodes with initiated primary ears |
|---|---|---|---|---|---|---|---|---|
| NTG siblings | 60 | 0 | 74.4 ± 14.2 | 4.4 ± 1.0 | 9.8 ± 0.8 | 0.28 | 4 to 6 | 6 to 7 |
| UBIpro:ZmCCT10 | 120 | 2.7 ± 1.3 | 104.9 ± 29.8* | 4.3 ± 0.8 | 12.6 ± 2.1* | 0.30 | 5 to 9 | 6 to 8 |
| NTG siblings | 45 | 0 | 67.9 ± 16.2 | 4.1 ± 1.1 | 9.5 ± 0.8 | 0.28 | 4 to 6 | 6 to 7 |
| BSVpro:ZmCCT10 | 69 | 11.9 ± 5.7 | 263.0 ± 51.8** | 2.8 ± 0.7 | 35.9 ± 5.8** | 0.31 | 16 to 37 | 18 to 33 |
Measurements represent means ± SD. Leaf appearance rate calculated with linear regression models. BSVpro:ZmCCT ear traits were collected only for one event (11 plants).
*Means are statistically different from NTG at p < 0.001.
**Means are statistically different from NTG at p < 0.00001.
Internode length depends on position within the plant shoot. As observed for NTG and UBIpro:ZmCCT10 plants, the internodes below the ear were progressively longer closer to the ear and then became shorter above the ear (Fig 2A). Thus, internode length displayed an acropetal, bottom-up gradient below the ear and then switched to a top-down basipetal gradient above the ear, which is characteristic for organ growth before and after floral transition [4]. The BSVpro:ZmCCT10 internodes were on average 1.5-fold shorter and 2-fold wider than NTG. No significant differences were found between UBIpro:ZmCCT10 and NTG plants (Fig 2A and S5 Fig). Diameters of the BSVpro:ZmCCT10 stalks measured at the 1st internode averaged 31 mm compared to 15 mm for NTG and 20 mm for UBIpro:ZmCCT10 plants (S3 Table). This observation may explain the slower relative growth of the BSVpro:ZmCCT10 plants since they are allocating more assimilates to stalk-width growth.
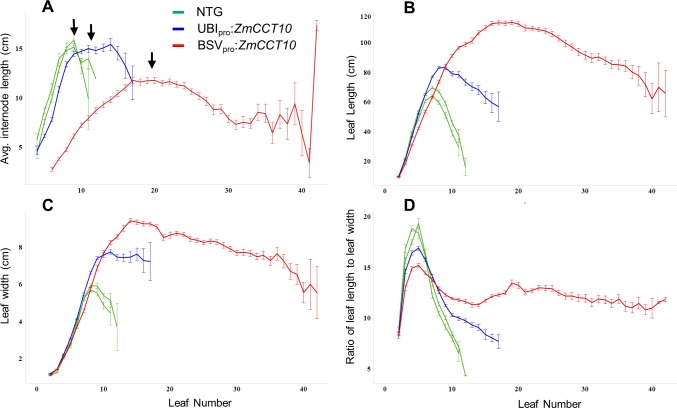
Fig 2. Internodes length and leaf morphology in non-transgenic (NTG), UBIpro:ZmCCT10, and BSVpro:ZmCCT10 transgenic plants.
A) Internode length by node position, B) leaf length by leaf position, C) leaf width by leaf position, and D) length/width ratios by leaf position. Node and leaf number are numbered from the base to the apex of the shoot. The ear nodes marked by black arrows. Error bars represent the ± the SE.
The UBIpro:ZmCCT10 plants produced an average of 13 leaves compared to 9–10 leaves for NTG plants, whereas the BSVpro:ZmCCT10 plants produced 36 leaves (Table 1). Leaf appearance rate was accelerated in transgenic plants: 0.30 and 0.31 leaves/day in UBIpro:ZmCCT10 and BSVpro:ZmCCT10 plants, respectively, versus 0.28 leaves/day in NTG plants (Table 1 and S6 Fig). This observation suggested that leaves in the SAM were initiated at a faster rate in transgenic plants relative to NTG plants.
Leaf size showed an acropetal growth pattern below the ear and a basipetal pattern above the ear (Fig 2). In NTG and UBIpro:ZmCCT10 plants, leaves below the ear displayed a sharp increase in length and width closer to the ear resulting in an increased length/width ratio. Leaves above the ear became shorter and narrower and the length/width ratio sharply declined (Fig 2B, 2C and 2D). Typically, the biggest leaf on a plant is the ear leaf. The BSVpro:ZmCCT10 plants displayed the characteristic acropetal pattern of leaf size but the basipetal pattern was disrupted after the 10th leaf. Leaf size remained relatively constant above node 10 reflecting the longer and wider leaf shape (Fig 2B, 2C and 2D). This indicated an increased leaf growth rate, longer duration of growth, or both in the BSVpro:ZmCCT10 plants compared to NTG or UBIpro:ZmCCT10 plants.
Consistent with the T0 observations, brace roots were found as high as node 37 in the BSVpro:ZmCCT10 plants, which was above the node of primary ear formation (Table 1); whereas NTG and UBIpro:ZmCCT10 plants produced brace roots below the ear (NTG internode 4–6 and UBIpro:ZmCCT10 internode 5–9). Because brace roots typically form at juvenile nodes [7] we decided to examine phase change in T1 families. To determine juvenile and adult growth phases, we first observed the presence of macro-hairs (an adult trait). Macro-hairs were present in NTG, UBIpro:ZmCCT10 and BSVpro:ZmCCT10 plants starting at leaf #4 (S4 Table). To determine juvenile and adult traits at the cellular level, we examined epidermal peels stained with Toluidine Blue O (TBO) [7]. Based on observations of bulliform cells, epidermal hairs, cell wall invagination, and TBO staining, all epidermal peels were juvenile at leaves 2–3 and began transitioning at leaf 4 (S7 Fig). NTG leaves were fully adult by leaf 7 and above, whereas UBIpro:ZmCCT10 and BSVpro:ZmCCT10 leaves were fully adult by leaf 8. There were no significant differences between NTG and transgenic plants for the juvenile-to-adult transition based on cell morphology and TBO staining of epidermal peels. Therefore, we conclude that juvenile-to-adult phase change was not affected in these transgenic plants.
Modification of reproductive traits in T1 transgenic plants
Transgenic plants exhibited a modification of reproductive traits. Relative to NTG plants, all transgenic plants displayed a delayed flowering phenotype (Table 2). NTG plants shed pollen on average 35–36 days after sowing. The UBIpro:ZmCCT10 plants shed pollen ~6 days later. Technical difficulties associated with maintaining the exceedingly tall T1 BSVpro:ZmCCT10 plants resulted in their final harvest and dissection at 130 days after sowing. On the day of harvest, only 51% (35 plants) of the BSVpro:ZmCCT10 plants had shed pollen and they shed pollen 65 days later than NTG plants (Table 2), 43% (30 plants) produced the abnormal bushy phyllody, and 2 plants still had developing inflorescence meristem indicating an extreme delay in flowering.
Table 2. Reproductive traits collected for T1 families.
| Line | Total No. Plants | Shedding plants | Days to shed | Silking plants | Days to silk | Ear length (cm) | Nodes with primary ears | Shank length (cm) | No. Primary tassel branches | No. secondary tassel branches | No. plants with phyllody |
|---|---|---|---|---|---|---|---|---|---|---|---|
| NTG siblings | 60 | 100% | 36.2 ± 1.1 | 93% | 38.9 ± 2.7 | 9.2 ± 2.8 | 6 to 7 | 12.3 ± 6.0 | 9.0 ± 2.7 | 0 | 0 |
| UBIpro:ZmCCT10 | 120 | 100% | 42.2 ± 4.4* | 100% | 48.8 ± 6.2* | 12.7 ± 1.8* | 6 to 8 | 31.2 ± 15.8** | 14.1 ± 3.2* | 0.7 ± 1.1 | 0 |
| NTG siblings | 45 | 100% | 35.6 ± 1.6 | 96% | 38.4 ± 2.5 | 8.6 ± 2.5 | 6 to 7 | 12.7 ± 5.4 | 7.2 ± 2.4 | 0 | 0 |
| BSVpro:ZmCCT10 | 69 | 51% | 101.2 ± 12.9** | 16% | 94.3 ± 8.4** | 10.5 ± 2.9* | 18 to 33 | 13.3 ± 7.8 | 27.6 ± 7.2** | 10.9 ± 4.6** | 30 |
Measurements represent means ± SD. Only one BSVpro:ZmCCT event produced ears that silked (11 plants).
*Means are statistically different from NTG at p < 0.001.
**Means are statistically different from NTG at p< 0.00001.
Transgene expression had a significant effect on tassel branching. NTG plants produced tassels with an average of 7–9 primary branches and no secondary branches (Table 2 and S8 Fig). The UBIpro:ZmCCT10 plants produced tassels with an average of 14 primary branches and, in a few examples, secondary branches were observed. The BSVpro:ZmCCT10 plants developed highly branched tassels with an average of 28 primary and 11 secondary branches (Table 2 and S8 Fig).
For the NTG and UBIpro:ZmCCT10 plants, 93–100% of the plants produced ears that silked 2 to 6 days later than they shed pollen. The unusually long shank subtending the ears observed in T0 UBIpro:ZmCCT10 plants was reproduced in the T1 families. The average shank length of the UBIpro:ZmCCT10 plants was 31 cm which was a 3-fold increase compared to the ~12 cm long shanks produced by NTG (Table 2). There was one event (11 plants) of the BSVpro:ZmCCT10PI422162 construct that produced silking ears. These plants were used to measure ear and shank length for the BSVpro:ZmCCT10 plants (Table 2). The transgenic ears were ~2 cm longer than NTG ears (Table 2). No differences in shank length were found. After careful dissection of the mature T1 BSVpro:ZmCCT10 plants with no visible ears, we found initiated ears but most them had aborted at early stages of development and never exerted silks.
A high level of ZmCCT10 expression caused formation of apically-induced plantlets
A striking feature of the BSVpro:ZmCCT10 transgenic plants was the development of the phyllody phenotype upon plant maturity (Fig 1D). More than 50% of the T0 plants developed this phenotype (S2E Fig), which was reproduced in 43% of the T1 BSVpro:ZmCCT10 plants (Table 2). This feature was never observed in the NTG or UBIpro:ZmCCT10 plants suggesting a strong relationship between the phyllody phenotype and the higher level of ZmCCT10 transgene expression.
Dissection of the highly branched vegetative structure from the T0 plants revealed complex arrangements of vegetative structures resembling multiple plantlets, each with an individual shoot axis (Fig 3). The number of countable plantlets varied from 2 to 26 (S2C Fig). Plantlets were at different stages of development with larger, more mature plantlets positioned on the periphery of the tassel and younger, less developed plantlets closer to the center. Some apices still had a central growing point that could generate more plantlets (Fig 3B and 3C). Dissection of the individual plantlets revealed a complex mix of abnormal tassel-like and ear-like organs. In a few cases, ear-like organs were fertile and could be fertilized to produce kernels (Fig 3A, Row 4). Dissection of the T1 BSVpro:ZmCCT10 phyllody tops revealed the presence of apices consisting of only plantlets or a combination of plantlets and tassel branches (S9 Fig). The most mature looking plantlets (S 10A) when planted into soil developed large root systems (S10B, S10C and S10D Fig). However, the shoot growth of these transplanted plantlets was stunted with secondary plantlets developing in place of reproductive organs (S10C and S10D Fig).
Fig 3. Representative images of apically-induced plantlets (phyllody top) dissected from T0, BSVpro:ZmCCT10 transgenic plants.
Panel images are arranged in the following horizontal order—Row 1: Total view of apex of plant, Row 2: Individual apically-induced plantlets dissected from the apex above, Row 3: Internal structures of dissected apically-induced plantlets, Row 4: Internal structures of a single apically-induced plantlet showing propagation of phenotype (left) and additional dissection of the apex. Panel images are arranged in the following vertical order—A) BSVpro:ZmCCT10Gaspe plants. B) BSVpro:ZmCCT10B73 plants. C) BSVpro:ZmCCT10tropical plants. D) BSVpro:ZmCCT10teosinte plants. White arrows indicate the central growing point. Scale bars = 10 cm.
To capture morphological changes in the meristem during and after floral transition, we dissected and imaged shoot apices from NTG and T1 transgenic sib plants. NTG plants developed very fast, as is typical for the Gaspé Flint line. Transitioning of the vegetative SAM to an inflorescence meristem (IM) occurred between V1-V2 stages (Fig 4A and 4B) followed by branch meristem (BM) initiation (Fig 4C) and developing tassel (DT) (Fig 4D) at V2-V3. By the V4 stage the immature tassel (IT) was fully formed and committed to maturation (Fig 4E). The apices of the UBIpro:ZmCCT10 transgenic plants were sampled every V-stage until an immature tassel had formed (Fig 4F–4J). The floral transition occurred at the V3-V5 stages, two stages later than NTG plants (Fig 4G). UBIpro:ZmCCT10 apices showed initiation of multiple BMs (Fig 4H) that resulted in highly branched tassels (Fig 4I and 4J and S8B Fig). Due to the limited number of BSVpro:ZmCCT10 T1 plants, we sampled apices at fewer V-stages and were only able to observe the vegetative SAM up to the V19 stage (Fig 4K). At this late stage, the SAM was swollen at the base (Fig 4K), that was not observed in NTG or UBIpro:ZmCCT10 plants (Fig 4A and 4F). During removal and dissection of the very tall, latest flowering BSVpro:ZmCCT10 plants at 130 days after sowing, we found two plants with an IM, and several plants with developing tassels (DT), which were characteristically highly branched (Fig 4L and 4M). Interestingly, two BSVpro:ZmCCT10 plants were found that had a phyllody top, consisting of a developing tassel with plantlets initiating at lower positions (Fig 4N).
Fig 4. Representative images of apices dissected from non-transgenic and UBIpro:ZmCCT10, and BSVpro:ZmCCT10 transgenic plants during vegetative and reproductive development.

A to E) Apices of non-transgenic plants. F to J) Apices of UBIpro:ZmCCT10 plants. K to N) Apices of BSVpro:ZmCCT10 plants. A, F, K) Shoot apical meristem (SAM) at the vegetative stage. B, G) Inflorescence meristem (IM). C, H) Branch meristem (BM) initiation. D, I, L) Developing tassel (DT), the stage when all BM are initiated and branches continue to initiate spikelet pair (SPM) and spikelet meristems (SM). E, J, M) Immature tassel (IT), the stage of tassel growth when all spikelets are initiated and meristems are consumed. (N) The apex with combination of the tassel spike (arrowhead) and emerging plantlets (stars). Scale bars (A, B, C, F, G, H, K, N) = 500 mm, (D, E, I, J, L, M) = 1 mm.
To investigate the morphology of the apical meristem in the phyllody plantlets, we dissected the plantlets from several T1 BSVpro:ZmCCT10 plants. The younger plantlets had a vegetative appearing SAM while others had developed a typical BSVpro:ZmCCT10 highly branched immature tassel (S11A, S11B, S11E and S11F Fig). The shoot meristem of older plantlets had a mixed morphology, showing a combination of an immature main tassel rachis with numerous shoot-like structures basally positioned (S11C, S11D, S11M and S11O Fig), which we expected might form secondary plantlets. Other abnormalities were observed in the plantlet apical meristems, including ear-like organs surrounded by leaf-like primordia (S11J, S11K and S11L Fig) and a dissected spikelet containing both anthers and a cluster of pistillate florets (S11P, S11Q and S11R Fig).
Expression of flowering regulators in leaves affected by ZmCCT10 overexpression
To gain insight into the gene network regulated by ZmCCT10 in leaves, we selected candidate genes based on the regulatory flowering network models proposed for maize [61] and rice [62, 63]. Even though ZmCCT10 might regulate as many as 1117 genes in leaves [39], we selected 20 representative genes with proven or predicted functions in flowering pathways in leaves and assayed their expression by qRT-PCR (S5 Table).
The circadian clock plays a central role in regulation of photoperiodic flowering time in plants and so several clock genes were assayed. The maize genes GIGZ1A/1B and CONZ1 are homologs of Arabidopsis flowering genes GIGANTEA (GI) and CONSTANS (CO) [64]. As previously shown, the duplicated genes GIGZ1A and GIGZ1B have identical expression patterns [64]. For this reason, GIGZ1A/1B were tested using the same assay (S5 Table). GIGZ1A/1B were not expressed in the coleoptiles but from leaf # 1 and onwards GIGZ1A/1B exhibited a typical diurnal expression pattern with an evening peak (Fig 5A). Hence GIGZ1A/1B genes appeared to be not regulated by ZmCCT10 and likely functions upstream of ZmCCT10 in the photoperiod pathway, as is the case in rice [62]. CONZ1 gene expression was slightly reduced in the UBIpro:ZmCCT10 transgenic plants and was significantly repressed in the BSVpro:ZmCCT10 plants suggesting its regulation by ZmCCT10 (Fig 5B).
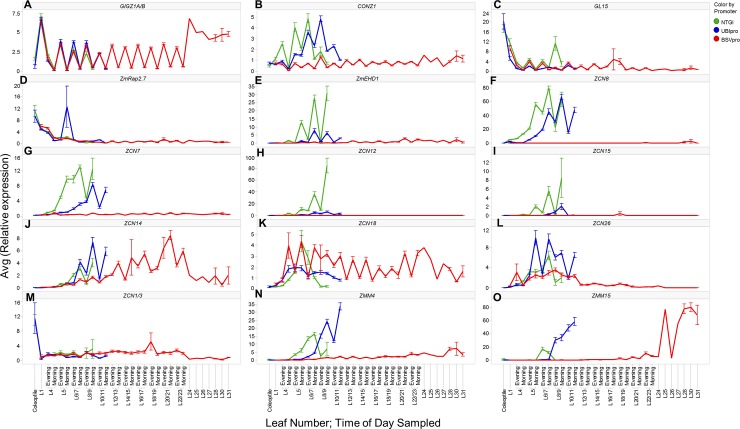
Fig 5. Expression patterns of flowering regulators in leaves of non-transgenic (NTG), UBIpro:ZmCCT10, and BSVpro:ZmCCT10 transgenic plants.
A) GIGZ1A/1B, a circadian clock gene. B) CONZ1, a circadian clock gene. C) GL15, the regulator of transition from a juvenile to adult leaf identity, a target of miR172. D) ZmRap2.7, a repressor of flowering, a target of miR172. E) ZmEHD1, an activator of flowering. F) ZCN8, the major florigen gene, an activator of flowering. G) ZCN7, another putative florigen gene, an activator of flowering. H) ZCN12, I) ZCN15, J) ZCN14, K) ZCN18, FT-like genes with florigenic activity in Arabidopsis. L) ZCN26, a FT-like gene with no florigenic activity in Arabidopsis. M) ZCN1/3, TFL1-like genes, repressors of flowering. N) ZMM4 and O) ZMM15 MADS genes, activators of flowering, markers for floral transition. The X-axis represents the tissue sampled: Coleoptile, leaves 1 to 22 (sampled either in the morning or evening), leaves 24 to 31 (sampled in the evening). The Y-axis represents the average relative gene expression normalized against eukaryotic initiation factor 4-gamma (GenBank EU967723). Error bars represent ± the SE.
The APETALA2-like transcription factor GL15, that regulates the transition from juvenile to adult leaf identity in maize and is a target of miR172 [15], was highly expressed in coleoptiles and in juvenile leaf # 1. Thereafter, its expression decreased in leaf # 4 (Fig 5C), which is consistent with our observation that the onset of the transition from juvenile-to-adult phases occurred in leaf # 4 (S7 Fig). GL15 was diurnally expressed with an evening peak and its expression pattern was nearly identical in NTG and transgenic plants suggesting no regulation by ZmCCT10 (Fig 5C).
The AP2-like ZmRap2.7 gene is a repressor of flowering and is a target of miR172 [65]. The ZmRap2.7 expression pattern was similar to GL15 which was higher in the coleoptiles and in juvenile leaf # 1 with a diurnal evening peak (Fig 5D). No significant differences were observed in NTG and transgenic plants placing ZmRap2.7 outside of ZmCCT10 regulation.
ZmEhd1 is a homolog of the rice Ehd1 activator of flowering under short days [43]. According to the rice photoperiod model, Ehd1 is directly repressed by Ghd7 [62, 63]. In agreement with the rice model, expression of ZmEhd1 was reduced in the UBIpro:ZmCCT10 transgenic plants and completely repressed in the BSVpro:ZmCCT10 plants positioning ZmEhd1 downstream of ZmCCT10 (Fig 5E).
There are 15 FT-like genes in maize (the ZCN genes) that encode florigen-like proteins but only seven of them are expressed in leaves at some stages of development [28]. The ZCN8 and ZCN7 genes were shown to have florigen activity [29, 30, 66]. We examined the function of five leaf-expressed ZCN genes in Arabidopsis and found that four genes, ZCN12/14/15/18, displayed florigenic activity in Arabidopsis while ZCN26 did not (S12 and S13 Figs). This finding suggested the potential for six ZCN proteins (including ZCN7/8) to function as florigen triggers in maize. Expression of all seven leaf-expressed ZCN genes was examined in the ZmCCT10 transgenic plants. Expression levels of ZCN8, ZCN7, ZCN12 and ZCN15 were reduced in the UBIpro:ZmCCT10 transgenic plants and completely repressed in the BSVpro:ZmCCT10 transgenic plants (Fig 5F, 5G, 5H and 5I). Thus ZCN8, ZCN7, ZCN12 and ZCN15 appeared to be negatively regulated by ZmCCT10. Three of the ZCN genes, ZCN14, ZCN18 and ZCN26, exhibited complex expression patterns (Fig 5J, 5K and 5L). ZCN14 expression was slightly up-regulated in the UBIpro:ZmCCT10 transgenic plants and its expression was continuously elevated in the BSVpro:ZmCCT10 transgenic plants reaching its maximum in leaves 20–21 around the time when floral transition took place in at least some BSVpro:ZmCCT10 plants (Fig 5J). Thus, ZCN14 is not downstream of ZmCCT10 and may potentially be influenced by long day-dependent mechanisms. ZCN18 expression was slightly reduced in the UBIpro:ZmCCT10 plants but was up-regulated in the BSVpro:ZmCCT10 plants where it showed diurnal expression with a morning peak (Fig 5K). Thus, expression of ZCN18 appears to be induced by high levels of ZmCCT10 expression. ZCN26 showed the opposite expression pattern of ZCN18. It was up-regulated in the UBIpro:ZmCCT10 transgenic plants and was slightly repressed in the BSVpro:ZmCCT10 plants in leaves #10–11 and completely repressed in older leaves (Fig 5L). Because ZNC26 does not have florigenic activity and displays this unusual pattern, we speculate that its function may be unrelated to flowering time.
We also examined the TFL1-like ZCN1/3 genes which are antagonists of FT-like genes and have been shown to delay flowering and modify plant architecture when over-expressed in maize plants [54]. Out of six TFL1-like genes (ZCN1-6), only ZCN1 and ZCN3, are expressed in leaves at early growth stages [54]. The duplicated ZCN1 and ZCN3 genes showed higher expression in the coleoptiles and much lower expression in leaves at all stages of development in both NTG and transgenic plants (Fig 5M) and thus, are likely not regulated by ZmCCT10.
The MADS box genes, ZMM4 and ZMM15 are markers of the floral transition in maize. They are activated during this transition, first in the reproductive inflorescences and then in the leaves [34]. ZMM4 expression increased in leaves two stages later after the floral transition in NTG and UBIpro:ZmCCT10 plants (Fig 5N). In the BSVpro:ZmCCT10 plants, a slight expression increase was observed in leaves # 28–30 (Fig 5N). ZMM15 expression increased in leaves # 6–7 of NTG plants and 2 leaf stages later in the UBIpro:ZmCCT10 transgenic plants (Fig 5O). In the BSVpro:ZmCCT10 transgenic plants, ZMM15 expression was first detected in leaf #22 and increased in leaves # 24–31 reaching a higher level than in NTG or UBIpro:ZmCCT10 plants (Fig 5O). This finding suggests that both ZMM4 and ZMM15 are regulated by ZmCCT10 but ZMM15 may be activated independently of the photoperiod pathway.
Expression of meristem identity genes in the shoot apices affected by ZmCCT10 overexpression
To gain insight into the gene network(s) regulated by ZmCCT10 in the shoot apex, genes were selected with known and predicted functions in the floral transition and inflorescence development (S5 Table). For qRT-PCR analysis, the apices were grouped into five categories according to their developmental stage by morphology, including vegetative SAM, reproductive inflorescence meristem (IM), branch meristem (BM) initiation, developing tassel (DT), and immature tassel (IT) (Fig 4). Due to the limited number of BSVpro:ZmCCT10 plants available for dissection, fewer apices from this genotype were sampled and those at the BM initiation stage were missed. Overall, we surveyed 30 genes with various meristem functions (S5 Table). Expression of 20 genes were different in transgenic apices at more than one developmental stage (S6 Table). As a general trend, the magnitude of transcriptional changes was higher in the BSVpro:ZmCCT10 apices compared to UBIpro:ZmCCT10 apices (Fig 6 and S14 Fig).
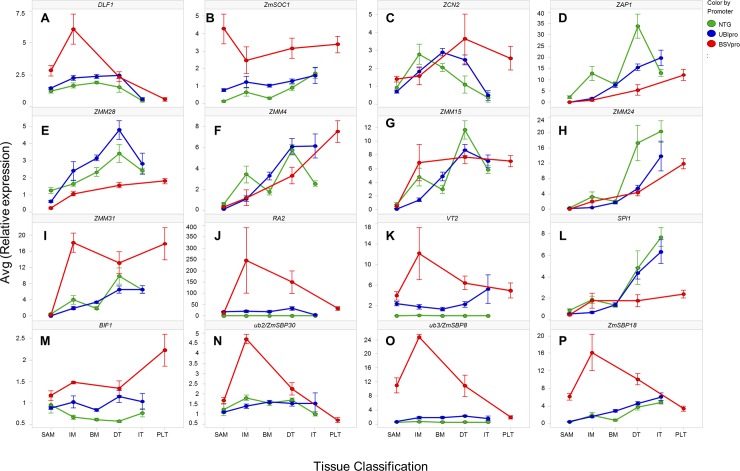
Fig 6. Expression patterns of inflorescence developmental genes in the shoot apices of non-transgenic (NTG), UBIpro:ZmCCT10, and BSVpro:ZmCCT10 transgenic plants.
A) DLF1 and B) ZmSOC1, floral transition genes. C) ZCN2, maize homolog of Arabidopsis TFL1, a repressor of the floral transition. AP1-like MADS genes D) ZAP1, E) ZMM28, F) ZMM4, and G) ZMM15. SEP-like MADS genes H) ZMM24 and I) ZMM31. J) RA2, SPM determinacy gene. K) VT2 and L) SPI1, auxin biosynthesis genes. M) BIF1, auxin signaling gene. N) UB2/SBP30. O) UB3/ZmSBP8, and P) ZmSBP18 genes, targets of miR156. The X-axis represents developmental stages of apices as defined in Fig 5. SAM: shoot apical meristem, IF: inflorescence meristem, BM: branch meristem, DT: developing tassel, IT: immature tassel, PLT: plantlets. The Y-axis represents the average relative gene expression normalized against eukaryotic initiation factor 4-gamma (GenBank EU967723). Error bars represent ± the SE.
The flowering activators DLF1 [27] and ZmSOC1 [67] known also as ZmMADS1[68] showed increased expression in transgenic apices (Fig 6A and 6B) suggesting their function independent of ZmCCT10. Expression of the flowering repressor ZCN2, a maize ortholog of Arabidopsis TFL1, showed similar expression trends in NTG and transgenic plants (Fig 6C). Expression of ZFL1 that controls inflorescence phyllotaxy in maize [69, 70] was slightly lower in transgenic apices compared to NTG plants (S14A Fig, S6 Table) suggesting a partial regulation of ZFL1 by the photoperiod pathway.
The inflorescence meristem identity AP1-like and SEP-like MADS genes were selected based on their key functions in inflorescence meristem specification in maize [34, 71] and rice [71–73]. As expected, the AP1-like MADS box genes, ZAP1, ZMM28, ZMM4 and ZMM15 were expressed at very low levels at vegetative stages in all genotypes. Their expression increased in the IM in NTG plants with ZAP1 having the highest expression level (Fig 6D). In BSVpro:ZmCCT10 apices, the expression levels of ZAP1 and ZMM28 were significantly reduced (Fig 6D and 6E) which indicates they function downstream of ZmCCT10. However, the expression of ZMM4 and ZMM15 was not significantly different in transgenic and NTG apices (Fig 6F and 6G).
The SEP-like MADS box genes ZMM24 and ZMM31 displayed contrasting expression patterns (Fig 6H and 6I). Expression of ZMM24 was repressed in both UBIpro:ZmCCT10 and BSVpro:ZmCCT10. But ZMM31 expression was moderately increased in UBIpro:ZmCCT10 apices at certain stages and was strongly increased in BSVpro:ZmCCT10 apices. ZMM31 expression was also high in plantlet apices (Fig 6I). This observation suggests that expression of the AP1-like and SEP-like genes is not tightly co-regulated by ZmCCT10 even though AP1 and SEP MADS proteins seems to function together in specifying inflorescence meristem identity [73].
To address the morphological changes in the tassel, such as the increased number of branches, the RAMOSA (RA) genes that control branching were assayed [74, 75]. In NTG plants, RA1, RA2, RA3 and REL2 showed normal developmental expression patterns, consistent with original observations [75, 76]. Expression was not detected in the SAM, was detected in the IM, was increased in the BM and DT, and declined in the IT stages in NTG (Figs 6J, S14B, S14C and S14D). Although minor expression differences were observed for RA1, RA3 and REL2 in both UBIpro:ZmCCT10 and BSVpro:ZmCCT10 transgenic apices (S14B, S14C and S14D Fig), RA2 expression was strongly increased in the transgenic plants, with BSVpro:ZmCCT10 apices showing a 250-fold increase (Fig 6J). The RA2 gene is required for transcriptional activation of RA1 [77], thus one would expect RA1 expression to also be up-regulated in transgenic plants, but this was not observed.
Given the critical function of auxin in plant development and shoot architecture [78, 79] we assayed the auxin biosynthetic genes VT2 [80] and SPI1 [81]. Surprisingly expression of these genes showed opposing patterns (Fig 6K and 6L). Expression of VT2 was significantly increased in UBIpro:ZmCCT10 and BSVpro:ZmCCT10 apices (Fig 6K) but expression of SPI1 was either not different than NTG or was reduced in UBIpro:ZmCCT10 and BSVpro:ZmCCT10 apices (Fig 6L). Elevated expression of VT2 was also observed in the roots of dissected apical plantlets (S10 Fig), but SPI1 expression was not detected in roots. VT2 encodes a tryptophan aminotransferase [80] and SPI1 encodes a YUCCA-like flavin monooxygenase [81]. Both genes belong to different tryptophan dependent auxin biosynthesis pathways [81]. This finding suggests that those pathways may be regulated independently of each other and that regulation of the SPI1 pathway might be influenced by photoperiod. To further investigate effects on auxin pathway genes, expression of the auxin efflux transporter ZmPIN1a (Zea mays pinformed1a) [82] and the auxin signaling genes BIF1, BIF4 and BA1 [83] were assayed. Expression of ZmPIN1a was not affected in transgenic plants (S14L Fig). Expression of BIF1 was increased for both transgenic constructs, whereas expression of BIF4 and BA1 were not statistically different (Figs 6M, S14M and S14N).
The novel phenotypic conversion of the tassel into a highly branched structure bearing plantlets in transgenic plants resembled the dominant Corngrass1 (Cg1) mutant which results from over-expression of miR156 and down-regulation of targeted SBP genes [12]. For this reason, we assayed six SBP genes regulated by miRNA156 cleavage including TSH4/SBP6 (TASSELSHEATH4) [84], TGA1 (TEOSINTE GLUME ARCHITECTURE1) [12], UB2 (UNBRANCHED2) and UB3 (UNBRANCHED3) [85], and ZmSBP14 and ZmSBP18 [86]. None of six tested SBP genes showed decreased expression in transgenic plants compared to NTG plants (Fig 6N, 6O and 6P and S14H, S14I and S14J Fig). In fact, increased transcript accumulation was observed for ub2/ZmSBP30, ub3/ZmSBP8 and ZmSBP18 (Fig 6N, 6O and 6P). Thus, it is likely that the apically induced plantlets are not caused by down-regulation of SBP gene expression.
To study the contribution of genes regulated by miR172, which has an antagonistic function to miR156, we selected the spikelet meristem identity genes AP2-like transcription factor BD1 (BRANCHED SILKLESS1 [87], TS6/IDS1 (TASSELSEED6/INDETERMINATE SPIKELET1, and SID1 SISTER OF INDETERMINATE SPIKELET1) [88, 89]. The overall expression patterns of these genes were not statistically different between NTG and transgenic apices (S14E, S14F and S14G Fig), although BD1 transcript accumulation was slightly decreased in UBIpro:ZmCCT10 apices (S14F Fig).
We took advatage of the prolonged vegetative phase of transgenic plants to compare expression of the miR156 and miR172 targeted genes during aging of the SAM. The SAM of the UBIpro:ZmCCT10 plants transtioned to an IM on average 20 days after sowing whereas the SAM of the BSVpro:ZmCCT10 plants took up to 60 days. Transcript accumulation of the miR156-targeted genes, ZmSBP18, TSH4/SBP6, TGA1, UB2/ZmSBP30, UB3/ZmSBP8 and ZmSBP14, showed an age dependent increase in expression (S15A, S15B, S15C, S15D, S15E and S15F Fig). This finding suggests that the expression of miR156 decreased over the extended vegetative period of growth resulting in accumulation of targeted SBP transcripts. Interestingly TGA1 mRNA accumulation peaked around the floral transition in the apices of UBIpro:ZmCCT10 and BSVpro:ZmCCT10 transgenic plants (S15C Fig) suggesting a putative involvement in the transition from vegetative to reproductive development.
Transcript accumulation for the miR172-targeted AP2-like genes showed an inverse pattern to the SBP genes, with expression being higher in the younger stage SAM and declining in older SAMs (S15G, S15H and S15I Fig). This pattern suggests that expression of miR172 is low in young apices and increased during extended vegetative growth. This finding is consistent with observations that miR156-miR172 regulation of the juvenile-to-adult transition in leaves is not altered in transgenic plants and these miRNA species are not regulated by ZmCCT10.
Discussion
Duration of the vegetative phase shapes maize plant architecture
Genetic evidence suggests that ZmCCT10 is a regulator of the photoperiod pathway in maize repressing flowering under long days in sensitive lines [39, 49]. ZmCCT10 is a nuclear localized protein with the potential to repress transcription, as was demonstrated for the homologous rice GHD7 protein [42]. For these reasons, over-expression of the ZmCCT10 protein in transgenic day-neutral (DN) maize was expected to act as a dominant inhibitor of the photoperiod pathway. This transgenic model allowed us to decipher which components of the photoperiod pathway are still responsive to ZmCCT10 modulation in day neutral maize, to identify genes in the autonomous pathway that connect to the photoperiod pathway, and to study the impact of modulating these pathways on maize growth and development.
Over-expression of ZmCCT10 with promoters of different strength, UBIpro:ZmCCT10 and BSVpro:ZmCCT10, revealed that the phenotypic effects of ZmCCT10 are quantitatively related to transcript abundance. Both UBIpro:ZmCCT10 and BSVpro:ZmCCT10 transgenic plants showed multiple changes to plant architecture, but the BSVpro:ZmCCT10 transgenic plants developed more extreme phenotypes, in general. Quantitative transgene action may be explained by the diurnal turnover of the ZmCCT10 protein. This idea is supported by the observation that the homologous GHD7 protein in rice is degraded at night, but high protein levels accumulate each day correlated with high levels of GHD7 transcription [42]. If this is the case for the ZmCCT10 protein, a higher transcriptional level may maintain a higher protein level.
Manipulation of expression of the ZmCCT10 repressor with promoters of different strength revealed a strong relationship between duration of vegetative growth and plant architecture. As expected during the prolonged vegetative phase, plants produced more phytomers, recorded as the number of nodes with an attached leaf. This is clearly demonstrated by comparison of NTG Gaspé Flint plants with the shortest vegetative phase (1–2 days) producing a maximum of 10 leaves at maturity, compared to the BSVpro:ZmCCT10 transgenic plants with the longest vegetative stage (up to 60 days) producing up to 60 leaves. Since NTG Gaspé Flint plants transitioned to reproductive development at the V1 stage, when they are still in the juvenile phase, this suggests that phase change might not be required for initiation of reproductive development in maize.
Morphological parameters of the individual phytomer are also modified in transgenic plants. Internode length is shorter and stalk diameter is wider compared to NTG plants. Leaf shape along the shoot is also modified (Fig 2) with leaves above the ear continuing growth resulting in long, wide leaves on the upper shoot of transgenic plants (Fig 1). Transgenic BSVpro:ZmCCT10 plants also developed adventitious brace roots at every internode up to the top of the plants (Fig 1). Another atypical phenotype produced only by UBIpro:ZmCCT10 transgenic plants was an unusually long axillary branch (shank) subtending the ears (Fig 1D, Table 2). Extreme elongation of ear shanks was reported for the jasmonic acid-deficient double opr7, opr8 mutants [90] hinting to a possible perturbation of jasmonic acid biosynthesis in UBIpro:ZmCCT10 transgenic plants. All these modifications dramatically changed the overall architecture of the BSVpro:ZmCCT10 transgenic plants indicating that prolonged vegetative growth could disturb multiple developmental processes.
Delaying the transition to reproductive development also induced changes in reproductive traits. We observed increased tassel branching in both UBIpro:ZmCCT10 and BSVpro:ZmCCT10 transgenic plants (S8 Fig) relative to controls. In BSVpro:ZmCCT10 transgenic plants, all ears aborted, suggesting axillary inflorescence development was suppressed by increased apical dominance. Alternatively, constant strong ZmCCT10 transcription may repress transition of the axillary branch meristem from vegetative to reproductive growth. It is important to emphasize that all maize plants with delayed floral transition displayed similar modifications to plant architecture. Production of more and bigger leaves, thicker stalks, brace roots at higher nodes, and highly branched tassels, was seen in the late flowering mutants id1 [33] and dlf1 [27]; in transgenic lines with less florigenic ZCN8 gene activity [91], in TFL1-like over-expression lines [54, 91], and in tropically adapted maize lines grown under long days [37, 92]. Moreover, transformation of the tassel into a phyllody phenotype observed in the BSVpro:ZmCCT10 transgenic lines (Fig 1), was similar to the “ball of shoots” phenotype observed in the null id1-m1 mutant [33] and in some tropical lines grown under long day conditions [92]. Regardless of the genetic or environmental cues, prolonged vegetative growth leads to similar phenotypic modifications to plant architecture suggesting a common physiological mechanism. The nature of this mechanism remains unclear.
Suppression of the photoperiod pathway does not affect transition from juvenile to adult phase but does delay the transition from vegetative to reproductive development
We found no effect of transgene expression on juvenile-to-adult phase change in transgenic events using such leaf morphological features as bulliform cells, epidermal hairs, cell wall invagination, and TBO staining (S7 Fig). In agreement with phenotypic observations, expression of the GL15 gene, a regulator of juvenile-to-adult phase leaf traits and a target of miR172 [15], is not affected in transgenic plants (Fig 5C). mRNA levels of miR172 targeted gene, ZmRap2.7, a repressor of flowering [65] also did not change in transgenic plants (Fig 5D). This finding suggests that the “adult” miR172-mediated aging pathway is not regulated by ZmCCT10 and is not connected to the photoperiod pathway.
Higher ZmCCT10 expression did result in a significant increase in leaf number and later time to flower which is evidence of repression of floral transition. We investigated the morphology of shoot apices and found that ZmCCT10 expression delayed the transition of the vegetative SAM to an IM (Fig 4). The floral transition occurred two stages later in the UBIpro:ZmCCT10 transgenic plants (V3-V4) compared to NTG controls (V1) and up to 20 stages later in the BSVpro:ZmCCT10 transgenic plants. Suppression of the photoperiod pathway delayed transition from vegetative to reproductive development but had no effect on phase change.
Disruption of meristem identity gene expression may cause the perturbed inflorescence morphology in ZmCCT10 transgenic plants
We surveyed the expression of 30 meristem identity genes and found that for most of them, their expression was altered in BSVpro:ZmCCT10 apices. The most common expression change was increased expression in the IM samples (Fig 6I, 6J, 6K, 6N and 6P). Among them, the two most striking were the 250-fold increase in RA2 expression (Fig 6J) and the 12-fold increase in expression of the auxin biosynthesis gene VT2 (Fig 6K), suggesting that auxin levels might be elevated in transgenic apices that contribute to alteration of meristem development. The mechanisms by which over accumulation of RA2 or VT2 contributes to the highly branched, vegetative phyllody phenotype is not clear. In fact, RA2 normally functions to suppress the initiation of long branches in the tassel, since loss of function ra2 mutants have highly branched tassels. Perhaps, increased branching in the ZmCCT10 transgenics is mediated by a different branching pathway, like the UB2/UB3 pathway, and RA2 expression is activated in response to suppress the extra branching.
FT-like ZCN genes are differently regulated by ZmCCT10
Maize has seven FT-like genes (ZCN8,7,12,14,15,18,26) [28] with ZCN8 appearing to encode the primary florigenic signal [29–31]. However, other FT-like genes, like the close homolog of ZCN8, ZCN7, may also possess florigenic activity [32]. Complementation of the ft mutant in Arabidopsis indicates, of the seven FT-like ZCN genes, six possess florigenic activity, (excluding ZCN26) (S13 Fig), suggesting they may have a flowering function in maize. Analyses of their expression patterns in ZmCCT10 transgenic plants provides further evidence for their potential involvement in flowering time. ZCN8/7/12/15 expression was repressed by ZmCCT10 which places them downstream of this regulator in the photoperiod pathway. Expression of ZCN26 appears to not be regulated by ZmCCT10, consistent with previous results suggesting it has no flowering function [30].
Expression of ZCN14/18 is independent of ZmCCT10, positioning them outside of the photoperiod pathway. ZCN18 is likely not involved in flowering time control regardless of its potential florigenic activity in Arabidopsis (S13 Fig) because its expression is independent of the floral transition and the ZCN18 protein does not interact with DLF1 in a Y2H assay [30]. To the contrary, ZCN14 may function as part of the autonomous pathway that enables flowering even under non-inductive conditions in photoperiod sensitive backgrounds. We base this hypothesis on two lines of evidence. First, ZCN14 is expressed in leaves, consistent with a role as a floral activator, but only under LDs in tropical germplasm [30], indicating it is LD responsive. Second, the ZCN14 protein interacts with DLF1 in a Y2H assay[30], and is phylogenetically quite closely related to the rice, barley, and wheat florigenic proteins [28]. Therefore, we suggest a ZCN14-DLF1 FAC may form under LD conditons, allowing photoperiod sensitive genotypes to flower, even under non-inducitve conditions. It is important to emphasize that DLF1 expression is not suppressed in transgenic apices (Fig 6A), indicating the DLF1 protein is not rate limiting in forming a FAC.
A conceptual network model for regulation of flowering time in maize
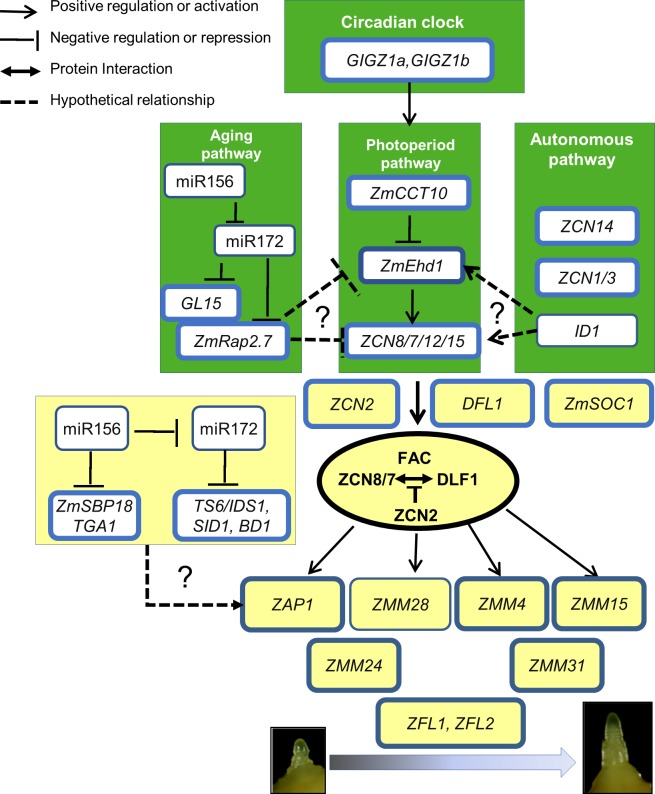
The genetic network model for flowering time in maize [61] is less elaborated compared to rice [93] due to a limited number of flowering time mutants and a lack of flowering time QTLs with a large effect [94]. An updated network model was recently advanced based on results from transcriptome profiling of temperate maize and obligate SD teosinte [95]. Another approach is transgenic manipulation of flowering time which provides additional information to populate the maize network. Over-expression of ZmCCT10 helped define components of the photoperiod dependent and independent pathways and allowed us to further refine the flowering network (Fig 7) published previously [61].
Fig 7. A conceptual gene network model for regulation of flowering time in maize.
The model is divided into genes expressed in leaves (green background) and those expressed in the shoot apical meristem (yellow background). Genes tested in this study are framed by a blue outline. In the leaves, three pathways are depicted: aging, photoperiod and autonomous. The photoperiod pathway is represented by the module ZmCCT10 (rice Ghd7)—ZmEhd1–ZCN8/7/12/15 (FT-like genes) which is conserved in other short-day monocots, rice and sorghum. ZmCCT10 is likely a repressor of the flowering activator ZmEhd1 gene which is a homolog of rice Ehd1 and sorghum SbEhd1. ZmCCT10 is not expressed under inductive SDs releasing ZmEhd1 from repression and activating the FT-like ZCN8/7/12/15 genes. ZmCCT10 is expressed under non-inductive LDs leading to repression of the FT-like ZCN8/7/12/15genes which occurs directly or indirectly via repression of ZmEhd1. However, the FT-like ZCN14 gene is not repressed by ZmCCT10 and may be placed in the autonomous pathway. In the aging pathway, ZmRap2.7 represses flowering via suppression of ZmEhd1 or ZCN8/7 genes. In the autonomous pathway, ID1 is required for activation of ZCN8/7 and possibly ZmEhd1 or other upstream regulators of ZCN8/7. Once transported to the shoot apical meristem (SAM), the florigenic ZCN8/7proteins interact with DLF1 forming the florigen activation complex (FAC, yellow oval). The florigen-antagonistic protein ZCN2 may compete with ZCN8/7 in the FAC leading to delayed flowering. Expression of DFL1 and ZCN2 is independent of the photoperiod pathway. Formation of the FAC in the SAM induces transcription of the AP1-like MADS box meristem identity genes ZAP1, ZMM4 and ZMM15 marking the onset of inflorescence specification. The ZFL1/2 genes are positioned downstream of the AP1-like genes.
In our model, the photoperiod pathway is represented by the conserved module ZmCCT10 (homolog of Ghd7)—ZmEhd1—ZCN8/7 (FT-like florigen). The circadian clock is the primary regulator of the photoperiod pathway but over expression of ZmCCT10 does not affect expression of the circadian genes GIGZ1A/B (homolog of GIGANTIA), indicating the lack of feedback from ZmCCT10 to the circadian clock. The conz1 gene, the maize homolog of CONSTANS, is down regulated in ZmCCT10 transgenic plants but its connection to other genes in this pathway is not clear, so it was not included in this model.
ZCN8 appears to be a conversion point of the photoperiod, autonomous and aging pathways [31, 40, 95, 96] and its regulation is complex. In rice, Ehd1 is one of many upstream activators of the florigenic genes Hd3a/RFT1 [93, 97]. In maize, ZmEhd1 may play a similar activation function of ZCN8 and other florigenic genes but this gene order must be further validated. Recently, the MADS-box transcription factor ZmMADS1 (alias ZmSOC1, in the model) was shown to be a direct, positive regulator of ZCN8 reinforcing the idea of multiple inputs for ZCN8 regulation.
In ZmCCT10 transgenic plants, expression of the florigenic genes ZCN8/7/12/15 are repressed, positioning them downstream of ZmCCT10. In contrast, expression of ZCN14 increases over time in ZmCCT10 transgenic plants, suggesting it function outside the photoperiod pathway, perhaps in the autonomous pathway. Within the autonomous pathway, ID1 is a major flowering time regulator and the pathway itself may be regulated by sugar signaling through a recently proposed "sugar clock" [95].
In day neutral, and tropical lines under inductive SDs, florigen is supplied by the ZCN8/7 genes, which moves to the SAM [29, 30] where it interacts with DLF1 to form the FAC. In the shoot apical meristem, DLF1 is expressed independently of the photoperiod pathway, allowing for a constant supply of this bZIP transcription factor to the FAC [24]. This complex activates expression of the meristem identity AP1-like MADS box genes ZAP1, ZMM28, ZMM4 and ZMM15.
The aging pathway in leaves and apical meristems are not repressed in ZmCCT10 transgenic plants which places them outside of the photoperiod pathway. However, during prolonged vegetative growth, we observed an age-dependent activation of SBP genes in the SAM. This finding suggests that some SBP proteins might have the ability to bypass the need for FAC function and activate AP1-like MADS box genes independent of the florigen-dependent pathway for flowering. A similar situation was shown to function in Arabidopsis where expression of the SPL3, SPL13 and SPL9 (squamosa promoter binding protein-like) genes could trigger expression of AP1, FUL1 and LEAFY and, thus, bypass the need for FT-FD function to induce the floral transition [98, 99]. If a similar bypass mechanism functions in maize, it has yet to be determined.
Conclusion
Maize was domesticated from the tropical progenitor-grass teosinte, which requires short-day photoperiods to flower. Over time, as cultivation moved to higher latitudes, maize’s requirement for short-day photoperiod induction was reduced. As a result, maize has been adapted to grow in a wide range of photoperiods, with tropical maize flowering faster in short days, and temperate maize being relatively day neutral. Genetic studies revealed that ZmCCT10, which functions as a repressor of flowering, controls this short-day requirement. We investigated the role this gene plays in flowering and other developmental processes by generating transgenic plants overexpressing ZmCCT10. Our phenotypic analysis of transgenic events containing either a constitutive promoter with high strength, or a constitutive promoter with very-high strength, showed that ZmCCT10 over-expression produced the expected late flowering phenotype, with the very-high level expressing events showing a dramatically prolonged vegetative period of growth and severe morphological developmental defects. Transcript expression analyses indicated that many genes that promote flowering are repressed and thus are downstream of ZmCCT10. We also showed that specific genes in modules affecting other developmental transitions are linked to photoperiod dependent or independent regulation. Our analysis allowed us to update the maize flowering time network model.
Supporting information
Amino acids conserved in all genotypes are shown over yellow background. Amino acids found in only one genotype are shown over green background. Amino acids conserved in five or six genotypes out of seven are shown over the blue background. The conserved CCT domain is framed. Asterisks marked putative DNA/RNA binding motif C-X2-C-X4-CC-X-H-X2-H. Putative nuclear localization signals (NLS) are underlined.
(TIF)
NTG parent: no overexpression of ZmCCT10 allele, Gaspe: overexpression of Gaspé Flint ZmCCT10 allele, B73: overexpression of B73 ZmCCT10 allele, CML436: overexpression of the CML436 ZmCCT10 allele, CML311: overexpression of the CML311 ZmCCT10 allele, PI422162: overexpression of the teosinte PI422162 ZmCCT10 allele, PI441934: overexpression of the teosinte PI441934 ZmCCT10 allele. A) Internode length by leaf position. Internode distance between nodes 4 and 5 is referred to as node 5. Measurements represent means ± SD. B) The range of the highest nodes with the brace roots depicted by the box-plot. C) The number of apically–induced plantlets in T0 plants depicted by the box-plot. D) Example of secondary aerial brace roots formed at 153 days after planting. E) The percentage of T0 plants with normal and modified apex morphology (phyllody)”top by ZmCCT10 alleles. Apex morphology is classified as shoot apical meristem (SAM) in the vegetative state, the tassel, both tassel and plantlets, as well as plantlets only.
(TIF)
(TIF)
A) Plant height at harvest. B) Final leaf number. C) The highest nodes with brace root initiation. D) The highest nodes with primary ears (aborted ears in the BSVpro:ZmCCT10 transgenic plants).
(TIF)
Scale Bar = 1 m.
(TIF)
The plant leaf number was recorded twice a week. Linear regression lines show leaf appearance rate. b-value indicates average number of leaves appearing in one day. r2 indicates how well the data fit the trend line.
(TIF)
A) Leaf 2 from NTG, B) UBIpro:ZmCCT10 and C) BSVpro:ZmCCT10 plants representing the juvenile phase. Juvenile epidermal cells are elongated, stain violet, and possess wavy cell walls. D) Leaf 4 from NTG E) UBIpro:ZmCCT10 and F) BSVpro:ZmCCT10 plants in the transitioning stage showing a mixture of juvenile and adult traits. Macrohairs are visible, but files of bulliform cells are not formed yet. G) Leaf 7 from NTG, and leaf 8 H) UBIpro:ZmCCT10 and I) BSVpro:ZmCCT10 plants representing the adult phase. J) Graphic representation of leaf identity vs. leaf number. The epidermis is highly differentiated into aqua-staining cells with invaginated cell walls, files of purple bulliform cells with macrohairs. b, files of bulliform cells; m, macrohair. Scale bar = 500 mm.
(TIF)
A) Non-transgenic tassels, B) UBIpro:ZmCCT10 tassels, and C) BSVpro:ZmCCT10 tassels. Scale bar = 5 cm
(TIF)
A,B,C) Examples of the apices that produced only plantlets. D,E,F,G) Examples of the apices that produced plantlets and tassels. Scale bar = 5 cm.
(TIF)
A) Images of 9 plantlets and the main growing stalk dissected from one T1 BSVpro:ZmCCT10 plant. The more developed plantlets #1, #2 and #3 were planted into pots on June 30, 2015 and grown in a greenhouse until August 19, 2015. Plantlet #2 died. B) View of plantlet #1 showing well-developed roots (close-up in insert). C) Dissection of plantlet #1 showing continuous production of secondary plantlets. D) View of plantlet #3 showing well-developed roots, developed ear (close-up in insert on the right side) and the main growing stalk producing secondary plantlets (close-up in insert on the left side). Scale Bars = 30 cm.
(TIF)
Plantlets dissected from the same plant are grouped and numbered starting from the most mature plantlets (#1). A), B), H) Visibly normal immature tassels. C), D) M), N), O) Apices with the developed main tassel spike (arrowheads) and emerging secondary plantlets (stars) at the base of the tassel. E), F) Apices with the vegetative SAM. G), I) Severely impaired tassels with leaf-like structures. J) Apices with massive outgrowth of ear-like structures at the base of the main tassel spike and emerging secondary plantlets (stars). K) A close-up view of ear-like structures. L) Dissection of the secondary plantlet from the apex [(J) marked by arrow] show the ear-like structure with tertiary emerging plantlets (stars). P) The staminate spikelet dissected from the tassel-like structure with 2 normal (instead of 3) stamens at the right-side floret, close-up Q) and the ear-like structure at the left side floret, closeup. R) The main tassel spikes are marked by arrowheads and emerging plantlets are marked by stars. Scale bars (A, B, K, L) = 1 mm, (G, H, I, O) = 2 mm, (J, M, N) = 5 mm, (C, D, E, F, P, Q, R) = 500 mm.
(TIF)
Scale bar = 2 cm.
(TIF)
Over-expression of ZCN8 and ZCN12 not only complements ft mutants but also causes extremely early flowering and determinate inflorescences. ZCN12 T1 over-expression lines are sterile. Over-expression of ZCN14, ZCN15, and ZCN18 complements ft mutants and causes early flowering and indeterminate inflorescences. Over-expression of ZCN26 does not complement ft mutants and leads to a slightly late flowering. Number of rosette leaves are means ± SD.
(TIF)
X-axis represents developmental stages of apices as defined in Fig 5. SAM: shoot apical meristem, IF: inflorescence meristem, BM: branch meristem, DT: developing tassel, IT: immature tassel, PLT: plantlets. Y-axis represents average relative gene expression normalized against eukaryotic initiation factor 4-gamma (GenBank EU967723). Numeric t-test, p>0.05 are shown in S4 Table where expression in the transgenic shoot apices was compared to NTG apices. Error bars = ± SE.
(TIF)
A) to F) Quantification of SBP mRNA, regulated by miR156 cleavage. G) to I) Quantification of AP2-like mRNA, regulated by miR172 cleavage. X-axis represents days after sowing (DAS). Y-axis represents relative gene expression normalized against eukaryotic initiation factor 4-gamma (GenBank EU967723). Each data point represents a qRT-PCR value for an individual apex.
(TIF)
(XLS)
(XLS)
(XLS)
(XLS)
(XLS)
(XLS)
Acknowledgments
We are thankful to Ron Christensen, Jacque Hockenson and Tim Moriarty for assistance with growing plants in the green house; Kevin Simcox for supplying the seeds of teosinte, Shawn Thatcher for help with the statistical analysis and Gene Ananiev for figure preparation.
Data Availability
All relevant data are within the manuscript and its Supporting Information files.
Funding Statement
DuPont Pioneer provided support in the form of salaries for authors E.S., S.E., X.M., J.O., J.E.H., O.N.D. but did not have any additional role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript. The specific roles of these authors are articulated in the ‘author contributions’ section.
References
- 1.Barthélémy D, Caraglio Y. Plant Architecture: A Dynamic, Multilevel and Comprehensive Approach to Plant Form, Structure and Ontogeny. Annals of botany. 2007;99(3):375–407. 10.1093/aob/mcl260 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Ha CM, Jun JH, Fletcher JC. Shoot Apical Meristem Form and Function Current Topics in Developmental Biology: Elsevier; 2010. p. 103–40. 10.1016/S0070-2153(10)91004-1 [DOI] [PubMed] [Google Scholar]
- 3.Teichmann T, Muhr M. Shaping plant architecture. Frontiers in plant science. 2015;6:233 10.3389/fpls.2015.00233 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.McSteen P, Leyser O. Shoot branching. Annual review of plant biology. 2005;56:353–74. Epub 2005/05/03. 10.1146/annurev.arplant.56.032604.144122 . [DOI] [PubMed] [Google Scholar]
- 5.Cheng PC, Greyson RI, Walden DB. Organ Initiation and the Development of Unisexual Flowers in the Tassel and Ear of Zea mays. American Journal of Botany 1983;70(3):450–62. [Google Scholar]
- 6.Poethig RS. Vegetative phase change and shoot maturation in plants. Curr Top Dev Biol. 2013;105:125–52. 10.1016/B978-0-12-396968-2.00005-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Moose SP, Sisco PH. Glossy 15 Controls the Epidermal Juvenile-to-Adult Phase Transition in Maize. The Plant cell. 1994;6:1343–55. 10.1105/tpc.6.10.1343 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Evans MMS, Passas HJ, Poethig RS. Heterochronic effects of glossy15 mutations on epidermal cell identity in maize. Development (Cambridge, England). 1994;120:1971–81. [DOI] [PubMed] [Google Scholar]
- 9.Zheng XM, Feng L, Wang J, Qiao W, Zhang L, Cheng Y, et al. Nonfunctional alleles of long-day suppressor genes independently regulate flowering time. Journal of integrative plant biology. 2015. Epub 2015/07/30. 10.1111/jipb.12383 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Chuck G, Candela H, Hake S. Big impacts by small RNAs in plant development. Current opinion in plant biology. 2009;12(1):81–6. 10.1016/j.pbi.2008.09.008 . [DOI] [PubMed] [Google Scholar]
- 11.Teotia S, Tang G. To bloom or not to bloom: role of microRNAs in plant flowering. Molecular plant. 2015;8(3):359–77. Epub 2015/03/05. 10.1016/j.molp.2014.12.018 . [DOI] [PubMed] [Google Scholar]
- 12.Chuck G, Cigan AM, Saeteurn K, Hake S. The heterochronic maize mutant Corngrass1 results from overexpression of a tandem microRNA. Nat Genet. 2007;39(4):544–9. 10.1038/ng2001 . [DOI] [PubMed] [Google Scholar]
- 13.Abedon BG, Tracy WF. Corngrass 1 of Maize (Zea mays L.) Delays Development of Adult Plant Resistance to Common Rust (Puccinia sorghi Schw.) and European Corn Borer (Ostrinia nubilalis Hubner). Journal of Heredity. 1966;87(3):219–23. [Google Scholar]
- 14.Moose SP, Sisco PH. Glossy15, an APETALA2-like gene from maize that regulates leaf epidermal cell identity. Genes & Development. 1996;10(23):3018–27. 10.1101/gad.10.23.3018 [DOI] [PubMed] [Google Scholar]
- 15.Lauter N, Kampani A, Carlson S, Goebel M, Moose SP. microRNA172 down-regulates glossy15 to promote vegetative phase change in maize. Proceedings of the National Academy of Sciences of the United States of America. 2005;102(26):9412–7. 10.1073/pnas.0503927102 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.McSteen P, Laudencia-Chingcuanco D, Colasanti J. A floret by any other name: control of meristem identity in maize. Trends in plant science. 2000;5(2):61–6. Epub 2000/02/09. . [DOI] [PubMed] [Google Scholar]
- 17.Jaeger KE, Wigge PA. FT protein acts as a long-range signal in Arabidopsis. Curr Biol. 2007;17(12):1050–4. 10.1016/j.cub.2007.05.008 . [DOI] [PubMed] [Google Scholar]
- 18.Turck F, Fornara F, Coupland G. Regulation and identity of florigen: FLOWERING LOCUS T moves center stage. Annual review of plant biology. 2008;59:573–94. 10.1146/annurev.arplant.59.032607.092755 . [DOI] [PubMed] [Google Scholar]
- 19.Turnbull C. Long-distance regulation of flowering time. Journal of experimental botany. 2011;62(13):4399–413. 10.1093/jxb/err191 . [DOI] [PubMed] [Google Scholar]
- 20.Zeevaart JA. Leaf-produced floral signals. Current opinion in plant biology. 2008;11(5):541–7. 10.1016/j.pbi.2008.06.009 . [DOI] [PubMed] [Google Scholar]
- 21.Corbesier L, Vincent C, Jang S, Fornara F, Fan Q, Searle I, et al. FT Protein Movement Contributes to Long-Distance Signaling in Floral Induction of Arabidopsis. Science (New York, NY). 2007;316(5827):1030–3. [DOI] [PubMed] [Google Scholar]
- 22.Pin PA, Nilsson O. The multifaceted roles of FLOWERING LOCUS T in plant development. Plant, cell & environment. 2012;35(10):1742–55. Epub 2012/06/16. 10.1111/j.1365-3040.2012.02558.x . [DOI] [PubMed] [Google Scholar]
- 23.Tamaki S, Matsuo S, Wong HL, Yokoi S, Shimamoto K. Hd3a Protein Is a Mobile Flowering Signal in Rice. Science (New York, NY). 2007;316(5827):1033–6. [DOI] [PubMed] [Google Scholar]
- 24.Taoka K, Ohki I, Tsuji H, Furuita K, Hayashi K, Yanase T, et al. 14-3-3 proteins act as intracellular receptors for rice Hd3a florigen. Nature. 2011;476(7360):332–5. Epub 2011/08/02. 10.1038/nature10272 . [DOI] [PubMed] [Google Scholar]
- 25.Taoka K, Ohki I, Tsuji H, Kojima C, Shimamoto K. Structure and function of florigen and the receptor complex. Trends in plant science. 2013;18(5):287–94. Epub 2013/03/13. 10.1016/j.tplants.2013.02.002 . [DOI] [PubMed] [Google Scholar]
- 26.Li C, Lin H, Dubcovsky J. Factorial combinations of protein interactions generate a multiplicity of florigen activation complexes in wheat and barley. Plant J. 2015;84(1):70–82. Epub 2015/08/08. 10.1111/tpj.12960 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Muszynski MG, Dam T, Li B, Shirbroun DM, Hou Z, Bruggemann E, et al. delayed flowering1 Encodes a basic leucine zipper protein that mediates floral inductive signals at the shoot apex in maize. Plant physiology. 2006;142(4):1523–36. 10.1104/pp.106.088815 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Danilevskaya ON, Meng X, Hou Z, Ananiev EV, Simmons CR. A genomic and expression compendium of the expanded PEBP gene family from maize. Plant physiology. 2008;146(1):250–64. Epub 2007/11/13. 10.1104/pp.107.109538 ; PubMed Central PMCID: PMCPmc2230559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Lazakis CM, Coneva V, Colasanti J. ZCN8 encodes a potential orthologue of Arabidopsis FT florigen that integrates both endogenous and photoperiod flowering signals in maize. Journal of experimental botany. 2011;62(14):4833–42. Epub 2011/07/07. 10.1093/jxb/err129 ; PubMed Central PMCID: PMCPmc3192997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Meng X, Muszynski MG, Danilevskaya ON. The FT-like ZCN8 Gene Functions as a Floral Activator and Is Involved in Photoperiod Sensitivity in Maize. The Plant cell. 2011;23(3):942–60. Epub 2011/03/29. 10.1105/tpc.110.081406 ; PubMed Central PMCID: PMCPmc3082274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Guo L, Wang X, Zhao M, Huang C, Li C, Li D, et al. Stepwise cis-Regulatory Changes in ZCN8 Contribute to Maize Flowering-Time Adaptation. Curr Biol. 2018;28(18):3005–15 e4. 10.1016/j.cub.2018.07.029 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Mascheretti I, Turner K, Brivio RS, Hand A, Colasanti J, Rossi V. Florigen-Encoding Genes of Day-Neutral and Photoperiod-Sensitive Maize Are Regulated by Different Chromatin Modifications at the Floral Transition. Plant physiology. 2015;168(4):1351–63. Epub 2015/06/19. 10.1104/pp.15.00535 ; PubMed Central PMCID: PMCPmc4528754. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Colasanti J, Yuan Z, Sundaresan V. The indeterminate gene encodes a zinc finger protein and regulates a leaf-generated signal required for the transition to flowering in maize. Cell research. 1998; 93(4):593–603. [DOI] [PubMed] [Google Scholar]
- 34.Danilevskaya ON, Meng X, Selinger DA, Deschamps S, Hermon P, Vansant G, et al. Involvement of the MADS-box gene ZMM4 in floral induction and inflorescence development in maize. Plant physiology. 2008;147(4):2054–69. Epub 2008/06/10. 10.1104/pp.107.115261 ; PubMed Central PMCID: PMCPmc2492622. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Doebley J. The genetics of maize evolution. Annu Rev Genet. 2004;38:37–59. 10.1146/annurev.genet.38.072902.092425 . [DOI] [PubMed] [Google Scholar]
- 36.Troyer AF. Temperate corn: Background, behavior, and breeding. Boca Raton, FL,: CRC Press; 2001. [Google Scholar]
- 37.Russell WK, Stuber CW. Effects of Photoperiod and Temperatures on the Duration of Vegetative Growth in Maize. Crop Science. 1982;23(5):847–50. [Google Scholar]
- 38.Hung HY, Shannon LM, Tian F, Bradbury PJ, Chen C, Flint-Garcia SA, et al. ZmCCT and the genetic basis of day-length adaptation underlying the postdomestication spread of maize. Proceedings of the National Academy of Sciences. 2012;109(28):E1913–E21. 10.1073/pnas.1203189109 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Yang Q, Li Z, Li W, Ku L, Wang C, Ye J, et al. CACTA-like transposable element in ZmCCT attenuated photoperiod sensitivity and accelerated the postdomestication spread of maize. Proceedings of the National Academy of Sciences of the United States of America. 2013;110(42):16969–74. Epub 2013/10/04. 10.1073/pnas.1310949110 ; PubMed Central PMCID: PMCPmc3801022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Huang C, Sun H, Xu D, Chen Q, Liang Y, Wang X, et al. ZmCCT9 enhances maize adaptation to higher latitudes. Proceedings of the National Academy of Sciences of the United States of America. 2018;115(2):E334–E41. 10.1073/pnas.1718058115 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Xue W, Xing Y, Weng X, Zhao Y, Tang W, Wang L, et al. Natural variation in Ghd7 is an important regulator of heading date and yield potential in rice. Nat Genet. 2008;40(6):761–7. Epub 2008/05/06. 10.1038/ng.143 . [DOI] [PubMed] [Google Scholar]
- 42.Weng X, Wang L, Wang J, Hu Y, Du H, Xu C, et al. Grain number, plant height, and heading date7 is a central regulator of growth, development, and stress response. Plant physiology. 2014;164(2):735–47. Epub 2014/01/07. 10.1104/pp.113.231308 ; PubMed Central PMCID: PMCPmc3912102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Doi K, Izawa T, Fuse T, Yamanouchi U, Kubo T, Shimatani Z, et al. Ehd1, a B-type response regulator in rice, confers short-day promotion of flowering and controls FT-like gene expression independently of Hd1. Genes Dev. 2004;18(8):926–36. 10.1101/gad.1189604 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Komiya R, Ikegami A, Tamaki S, Yokoi S, Shimamoto K. Hd3a and RFT1 are essential for flowering in rice. Development (Cambridge, England). 2008;135(4):767–74. 10.1242/dev.008631 . [DOI] [PubMed] [Google Scholar]
- 45.Komiya R, Yokoi S, Shimamoto K. A gene network for long-day flowering activates RFT1 encoding a mobile flowering signal in rice. Development (Cambridge, England). 2009;136(20):3443–50. Epub 2009/09/19. 10.1242/dev.040170 . [DOI] [PubMed] [Google Scholar]
- 46.Itoh H, Nonoue Y, Yano M, Izawa T. A pair of floral regulators sets critical day length for Hd3a florigen expression in rice. Nat Genet. 2010;42(7):635–8. Epub 2010/06/15. 10.1038/ng.606 . [DOI] [PubMed] [Google Scholar]
- 47.Murphy RL, Morishige DT, Brady JA, Rooney WL, Yang S, Klein PE, et al. Ghd7 (Ma6) Represses Sorghum Flowering in Long Days: Alleles Enhance Biomass Accumulation and Grain Production. The Plant Genome. 2014;7(2):0. 10.3835/plantgenome2013.11.0040 [DOI] [Google Scholar]
- 48.Yang S, Murphy RL, Morishige DT, Klein PE, Rooney WL, Mullet JE. Sorghum phytochrome B inhibits flowering in long days by activating expression of SbPRR37 and SbGHD7, repressors of SbEHD1, SbCN8 and SbCN12. PLoS One. 2014;9(8):e105352 Epub 2014/08/15. 10.1371/journal.pone.0105352 ; PubMed Central PMCID: PMCPmc4133345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Hung HY, Shannon LM, Tian F, Bradbury PJ, Chen C, Flint-Garcia SA, et al. ZmCCT and the genetic basis of day-length adaptation underlying the postdomestication spread of maize. Proceedings of the National Academy of Sciences of the United States of America. 2012;109(28):E1913–21. Epub 2012/06/20. 10.1073/pnas.1203189109 ; PubMed Central PMCID: PMCPmc3396540. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Woods D, McKeown M, Dong Y, Preston JC, Amasino RM. Evolution of VRN2/GhD7-like genes in vernalization-mediated repression of grass flowering. Plant physiology. 2016. 10.1104/pp.15.01279 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Yan L, Loukoianov A, Belchl A, Tranquilli G, Ramakrishna W, SanMiguel P, et al. The wheat VRN2 gene is a flowering repressor down-regulated by vernalization. Science (New York, NY). 2004;303:1640–4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Matsubara K, Hori K, Ogiso-Tanaka E, Yano M. Cloning of quantitative trait genes from rice reveals conservation and divergence of photoperiod flowering pathways in Arabidopsis and rice. Frontiers in plant science. 2014;5:193 Epub 2014/05/27. 10.3389/fpls.2014.00193 ; PubMed Central PMCID: PMCPmc4026727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Yan W, Liu H, Zhou X, Li Q, Zhang J, Lu L, et al. Natural variation in Ghd7.1 plays an important role in grain yield and adaptation in rice. Cell research. 2013;23(7):969–71. Epub 2013/03/20. 10.1038/cr.2013.43 ; PubMed Central PMCID: PMCPmc3698629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Danilevskaya ON, Meng X, Ananiev EV. Concerted modification of flowering time and inflorescence architecture by ectopic expression of TFL1-like genes in maize. Plant physiology. 2010;153(1):238–51. 10.1104/pp.110.154211 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Shi J, Habben JE, Archibald RL, Drummond BJ, Chamberlin MA, Williams RW, et al. Overexpression of ARGOS Genes Modifies Plant Sensitivity to Ethylene, Leading to Improved Drought Tolerance in Both Arabidopsis and Maize. Plant physiology. 2015;169(1):266–82. Epub 2015/07/30. 10.1104/pp.15.00780 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Abendroth LJ, Elmore ML, Boyer MJ, Marlay SK. Corn growth and development. Ames, Iowa Iowa State University; 2011. 10.1242/dev.058578 [DOI] [Google Scholar]
- 57.Evans MMS, Poethig RS. Gibberellins Promote Vegetative Phase Change and Reproductive Maturity in Maize. Plant physiology. 1995;108(475–487). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Dudley M, Poethig RS. The Heterochronic Teopod1 and Teopod2 Mutations of Maize Are Expressed Non-Cell-Autonomously. Genetics. 1993;133:389–99. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.McEIroy D, Brettell R. Foreign gene expression in transgenic cereals. Plant Biotechnolgy. 1994;12:62–8. [Google Scholar]
- 60.Diehn S, Lu AL, Simmons CRD. Viral promoter, truncations thereof, and methods of use. United States Patent. 2012;Patent No: US 8,338,662 B(Dec. 25, 2015).
- 61.Dong Z, Danilevskaya O, Abadie T, Messina C, Coles N, Cooper M. A gene regulatory network model for floral transition of the shoot apex in maize and its dynamic modeling. PLoS One. 2012;7(8):e43450 10.1371/journal.pone.0043450 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Itoh H, Izawa T. The coincidence of critical day length recognition for florigen gene expression and floral transition under long-day conditions in rice. Molecular plant. 2013;6(3):635–49. Epub 2013/02/19. 10.1093/mp/sst022 . [DOI] [PubMed] [Google Scholar]
- 63.Tsuji H, Taoka K-H., Shimamoto K. Regulation of flowering in rice: two florigen genes, a complex gene network, and natural variation. Current opinion in plant biology. 2010;14:1–8. 10.1016/j.pbi.2010.11.003 [DOI] [PubMed] [Google Scholar]
- 64.Miller TA, Muslin EH, Dorweiler JE. A maize CONSTANS-like gene, conz1, exhibits distinct diurnal expression patterns in varied photoperiods. Planta. 2008;227(6):1377–88. 10.1007/s00425-008-0709-1 . [DOI] [PubMed] [Google Scholar]
- 65.Salvi S, Sponza G, Morgante M, Tomes D, Niu X, Fengler KA, et al. Conserved noncoding genomic sequences associated with a flowering-time quantitative trait locus in maize. Proceedings of the National Academy of Sciences of the United States of America. 2007;104(27):11376–81. 10.1073/pnas.0704145104 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Mascheretti I, Turner K, Brivio RS, Hand A, Colasanti J, Rossi V. Florigen-Encoding Genes of Day-Neutral and Photoperiod-Sensitive Maize Are Regulated by Different Chromatin Modifications at the Floral Transition. Plant Physiology. 2015;168(4):1351–63. 10.1104/pp.15.00535 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Zhao S, Luo Y, Zhang Z, Xu M, Wang W, Zhao Y, et al. ZmSOC1, a MADS-box transcription factor from Zea mays, promotes flowering in Arabidopsis. International journal of molecular sciences. 2014;15(11):19987–20003. Epub 2014/11/06. 10.3390/ijms151119987 ; PubMed Central PMCID: PMCPmc4264151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Alter P, Bircheneder S, Zhou LZ, Schluter U, Gahrtz M, Sonnewald U, et al. Flowering Time-Regulated Genes in Maize Include the Transcription Factor ZmMADS1. Plant physiology. 2016;172(1):389–404. Epub 2016/07/28. 10.1104/pp.16.00285 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Bomblies K, Doebley JF. Pleiotropic effects of the duplicate maize FLORICAULA/LEAFY genes zfl1 and zfl2 on traits under selection during maize domestication. Genetics. 2006;172(1):519–31. 10.1534/genetics.105.048595 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Bomblies K, Wang RL, Ambrose BA, Schmidt RJ, Meeley RB, Doebley J. Duplicate FLORICAULA/LEAFY homologs zfl1 and zfl2 control inflorescence architecture and flower patterning in maize. Development (Cambridge, England). 2003;130(11):2385–95. Epub 2003/04/19. . [DOI] [PubMed] [Google Scholar]
- 71.Mena M, Mandel MA, Lerner DR, Yanofsky MF, Schmidt RJ. A characterization of the MADS-box gene family in maize. Plant J. 1995;8(6):845–54. Epub 1995/12/01. . [DOI] [PubMed] [Google Scholar]
- 72.Kobayashi K, Maekawa M, Miyao A, Hirochika H, Kyozuka J. PANICLE PHYTOMER2 (PAP2), encoding a SEPALLATA subfamily MADS-box protein, positively controls spikelet meristem identity in rice. Plant & cell physiology. 2010;51(1):47–57. 10.1093/pcp/pcp166 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Kobayashi K, Yasuno N, Sato Y, Yoda M, Yamazaki R, Kimizu M, et al. Inflorescence meristem identity in rice is specified by overlapping functions of three AP1/FUL-like MADS box genes and PAP2, a SEPALLATA MADS box gene. The Plant cell. 2012;24(5):1848–59. 10.1105/tpc.112.097105 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Gallavotti A, Long JA, Stanfield S, Yang X, Jackson D, Vollbrecht E, et al. The control of axillary meristem fate in the maize ramosa pathway. Development (Cambridge, England). 2010;137(17):2849–56. Epub 2010/08/12. 10.1242/dev.051748 ; PubMed Central PMCID: PMCPmc2938917. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Vollbrecht E, Springer PS, Goh L, Buckler ESt, Martienssen R. Architecture of floral branch systems in maize and related grasses. Nature. 2005;436(7054):1119–26. Epub 2005/07/26. 10.1038/nature03892 . [DOI] [PubMed] [Google Scholar]
- 76.Satoh-Nagasawa N, Nagasawa N, Malcomber S, Sakai H, Jackson D. A trehalose metabolic enzyme controls inflorescence architecture in maize. Nature. 2006;441(7090):227–30. Epub 2006/05/12. 10.1038/nature04725 . [DOI] [PubMed] [Google Scholar]
- 77.Bortiri E, Chuck G, Vollbrecht E, Rocheford T, Martienssen R, Hake S. ramosa2 encodes a LATERAL ORGAN BOUNDARY domain protein that determines the fate of stem cells in branch meristems of maize. The Plant cell. 2006;18(3):574–85. Epub 2006/01/10. 10.1105/tpc.105.039032 ; PubMed Central PMCID: PMCPmc1383634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Gallavotti A. The role of auxin in shaping shoot architecture. Journal of experimental botany. 2013;64(9):2593–608. Epub 2013/05/28. 10.1093/jxb/ert141 . [DOI] [PubMed] [Google Scholar]
- 79.McSteen P. Auxin and monocot development. Cold Spring Harbor perspectives in biology. 2010;2(3):a001479 Epub 2010/03/20. 10.1101/cshperspect.a001479 ; PubMed Central PMCID: PMCPmc2829952. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Phillips KA, Skirpan AL, Liu X, Christensen A, Slewinski TL, Hudson C, et al. vanishing tassel2 encodes a grass-specific tryptophan aminotransferase required for vegetative and reproductive development in maize. The Plant cell. 2011;23(2):550–66. Epub 2011/02/22. 10.1105/tpc.110.075267 ; PubMed Central PMCID: PMCPmc3077783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Gallavotti A, Barazesh S, Malcomber S, Hall D, Jackson D, Schmidt RJ, et al. sparse inflorescence1 encodes a monocot-specific YUCCA-like gene required for vegetative and reproductive development in maize. Proceedings of the National Academy of Sciences of the United States of America. 2008;105(39):15196–201. Epub 2008/09/19. 10.1073/pnas.0805596105 ; PubMed Central PMCID: PMCPmc2567514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Gallavotti A, Yang Y, Schmidt RJ, Jackson D. The Relationship between auxin transport and maize branching. Plant physiology. 2008;147(4):1913–23. Epub 2008/06/14. 10.1104/pp.108.121541 ; PubMed Central PMCID: PMCPmc2492655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Galli M, Liu Q, Moss BL, Malcomber S, Li W, Gaines C, et al. Auxin signaling modules regulate maize inflorescence architecture. Proceedings of the National Academy of Sciences of the United States of America. 2015;112(43):13372–7. Epub 2015/10/16. 10.1073/pnas.1516473112 ; PubMed Central PMCID: PMCPmc4629326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Chuck G, Whipple C, Jackson D, Hake S. The maize SBP-box transcription factor encoded by tasselsheath4 regulates bract development and the establishment of meristem boundaries. Development (Cambridge, England). 2010;137(8):1243–50. 10.1242/dev.048348 . [DOI] [PubMed] [Google Scholar]
- 85.Chuck GS, Brown PJ, Meeley R, Hake S. Maize SBP-box transcription factors unbranched2 and unbranched3 affect yield traits by regulating the rate of lateral primordia initiation. Proceedings of the National Academy of Sciences of the United States of America. 2014;111(52):18775–80. Epub 2014/12/17. 10.1073/pnas.1407401112 ; PubMed Central PMCID: PMCPmc4284592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Hultquist JF, Dorweiler JE. Feminized tassels of maize mop1 and ts1 mutants exhibit altered levels of miR156 and specific SBP-box genes. Planta. 2008;229(1):99–113. 10.1007/s00425-008-0813-2 . [DOI] [PubMed] [Google Scholar]
- 87.Chuck G, Muszynski M, Kellogg E, Hake S, Schmidt RJ. The control of spikelet meristem identity by the branched silkless1 gene in maize. Science (New York, NY). 2002;298(5596):1238–41. Epub 2002/11/09. 10.1126/science.1076920 . [DOI] [PubMed] [Google Scholar]
- 88.Chuck G, Meeley R, Hake S. Floral meristem initiation and meristem cell fate are regulated by the maize AP2 genes ids1 and sid1. Development (Cambridge, England). 2008;135(18):3013–9. 10.1242/dev.024273 . [DOI] [PubMed] [Google Scholar]
- 89.Chuck G, Meeley R, Irish E, Sakai H, Hake S. The maize tasselseed4 microRNA controls sex determination and meristem cell fate by targeting Tasselseed6/indeterminate spikelet1. Nat Genet. 2007;39(12):1517–21. 10.1038/ng.2007.20 . [DOI] [PubMed] [Google Scholar]
- 90.Yan Y, Christensen S, Isakeit T, Engelberth J, Meeley R, Hayward A, et al. Disruption of OPR7 and OPR8 reveals the versatile functions of jasmonic acid in maize development and defense. The Plant cell. 2012;24(4):1420–36. 10.1105/tpc.111.094151 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Danilevskaya ON, Meng X, McGonigle B, Muszynski MG. Beyond flowering time: pleiotropic function of the maize flowering hormone florigen. Plant signaling & behavior. 2011;6(9):1267–70. Epub 2011/08/19. 10.4161/psb.6.9.16423 ; PubMed Central PMCID: PMCPmc3258048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Coles ND, McMullen MD, Balint-Kurti PJ, Pratt RC, Holland JB. Genetic control of photoperiod sensitivity in maize revealed by joint multiple population analysis. Genetics. 2010;184(3):799–812. Epub 2009/12/17. 10.1534/genetics.109.110304 ; PubMed Central PMCID: PMCPmc2845347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Lee Y-S, An G. Regulation of flowering time in rice. Journal of Plant Biology. 2015;58(6):353–60. 10.1007/s12374-015-0425-x [DOI] [Google Scholar]
- 94.Buckler ES, Holland JB, Bradbury PJ, Acharya CB, Brown PJ, Browne C, et al. The Genetic Architecture of Maize Flowering Time. Science (New York, NY). 2009;325(5941):714–8. 10.1126/science.1174276 [DOI] [PubMed] [Google Scholar]
- 95.Minow MAA, Avila LM, Turner K, Ponzoni E, Mascheretti I, Dussault FM, et al. Distinct gene networks modulate floral induction of autonomous maize and photoperiod-dependent teosinte. Journal of experimental botany. 2018;69(12):2937–52. Epub 2018/04/25. 10.1093/jxb/ery110 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Liang Y, Liu Q, Wang X, Huang C, Xu G, Hey S, et al. ZmMADS69 functions as a flowering activator through the ZmRap2.7-ZCN8 regulatory module and contributes to maize flowering time adaptation. The New phytologist. 2018. 10.1111/nph.15512 . [DOI] [PubMed] [Google Scholar]
- 97.Song YH, Shim JS, Kinmonth-Schultz HA, Imaizumi T. Photoperiodic flowering: time measurement mechanisms in leaves. Annual review of plant biology. 2015;66:441–64. Epub 2014/12/24. 10.1146/annurev-arplant-043014-115555 ; PubMed Central PMCID: PMCPmc4414745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Wang JW, Czech B, Weigel D. miR156-regulated SPL transcription factors define an endogenous flowering pathway in Arabidopsis thaliana. Cell. 2009;138(4):738–49. 10.1016/j.cell.2009.06.014 . [DOI] [PubMed] [Google Scholar]
- 99.Yamaguchi A, Wu MF, Yang L, Wu G, Poethig RS, Wagner D. The microRNA-regulated SBP-Box transcription factor SPL3 is a direct upstream activator of LEAFY, FRUITFULL, and APETALA1. Developmental cell. 2009;17(2):268–78. 10.1016/j.devcel.2009.06.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Amino acids conserved in all genotypes are shown over yellow background. Amino acids found in only one genotype are shown over green background. Amino acids conserved in five or six genotypes out of seven are shown over the blue background. The conserved CCT domain is framed. Asterisks marked putative DNA/RNA binding motif C-X2-C-X4-CC-X-H-X2-H. Putative nuclear localization signals (NLS) are underlined.
(TIF)
NTG parent: no overexpression of ZmCCT10 allele, Gaspe: overexpression of Gaspé Flint ZmCCT10 allele, B73: overexpression of B73 ZmCCT10 allele, CML436: overexpression of the CML436 ZmCCT10 allele, CML311: overexpression of the CML311 ZmCCT10 allele, PI422162: overexpression of the teosinte PI422162 ZmCCT10 allele, PI441934: overexpression of the teosinte PI441934 ZmCCT10 allele. A) Internode length by leaf position. Internode distance between nodes 4 and 5 is referred to as node 5. Measurements represent means ± SD. B) The range of the highest nodes with the brace roots depicted by the box-plot. C) The number of apically–induced plantlets in T0 plants depicted by the box-plot. D) Example of secondary aerial brace roots formed at 153 days after planting. E) The percentage of T0 plants with normal and modified apex morphology (phyllody)”top by ZmCCT10 alleles. Apex morphology is classified as shoot apical meristem (SAM) in the vegetative state, the tassel, both tassel and plantlets, as well as plantlets only.
(TIF)
(TIF)
A) Plant height at harvest. B) Final leaf number. C) The highest nodes with brace root initiation. D) The highest nodes with primary ears (aborted ears in the BSVpro:ZmCCT10 transgenic plants).
(TIF)
Scale Bar = 1 m.
(TIF)
The plant leaf number was recorded twice a week. Linear regression lines show leaf appearance rate. b-value indicates average number of leaves appearing in one day. r2 indicates how well the data fit the trend line.
(TIF)
A) Leaf 2 from NTG, B) UBIpro:ZmCCT10 and C) BSVpro:ZmCCT10 plants representing the juvenile phase. Juvenile epidermal cells are elongated, stain violet, and possess wavy cell walls. D) Leaf 4 from NTG E) UBIpro:ZmCCT10 and F) BSVpro:ZmCCT10 plants in the transitioning stage showing a mixture of juvenile and adult traits. Macrohairs are visible, but files of bulliform cells are not formed yet. G) Leaf 7 from NTG, and leaf 8 H) UBIpro:ZmCCT10 and I) BSVpro:ZmCCT10 plants representing the adult phase. J) Graphic representation of leaf identity vs. leaf number. The epidermis is highly differentiated into aqua-staining cells with invaginated cell walls, files of purple bulliform cells with macrohairs. b, files of bulliform cells; m, macrohair. Scale bar = 500 mm.
(TIF)
A) Non-transgenic tassels, B) UBIpro:ZmCCT10 tassels, and C) BSVpro:ZmCCT10 tassels. Scale bar = 5 cm
(TIF)
A,B,C) Examples of the apices that produced only plantlets. D,E,F,G) Examples of the apices that produced plantlets and tassels. Scale bar = 5 cm.
(TIF)
A) Images of 9 plantlets and the main growing stalk dissected from one T1 BSVpro:ZmCCT10 plant. The more developed plantlets #1, #2 and #3 were planted into pots on June 30, 2015 and grown in a greenhouse until August 19, 2015. Plantlet #2 died. B) View of plantlet #1 showing well-developed roots (close-up in insert). C) Dissection of plantlet #1 showing continuous production of secondary plantlets. D) View of plantlet #3 showing well-developed roots, developed ear (close-up in insert on the right side) and the main growing stalk producing secondary plantlets (close-up in insert on the left side). Scale Bars = 30 cm.
(TIF)
Plantlets dissected from the same plant are grouped and numbered starting from the most mature plantlets (#1). A), B), H) Visibly normal immature tassels. C), D) M), N), O) Apices with the developed main tassel spike (arrowheads) and emerging secondary plantlets (stars) at the base of the tassel. E), F) Apices with the vegetative SAM. G), I) Severely impaired tassels with leaf-like structures. J) Apices with massive outgrowth of ear-like structures at the base of the main tassel spike and emerging secondary plantlets (stars). K) A close-up view of ear-like structures. L) Dissection of the secondary plantlet from the apex [(J) marked by arrow] show the ear-like structure with tertiary emerging plantlets (stars). P) The staminate spikelet dissected from the tassel-like structure with 2 normal (instead of 3) stamens at the right-side floret, close-up Q) and the ear-like structure at the left side floret, closeup. R) The main tassel spikes are marked by arrowheads and emerging plantlets are marked by stars. Scale bars (A, B, K, L) = 1 mm, (G, H, I, O) = 2 mm, (J, M, N) = 5 mm, (C, D, E, F, P, Q, R) = 500 mm.
(TIF)
Scale bar = 2 cm.
(TIF)
Over-expression of ZCN8 and ZCN12 not only complements ft mutants but also causes extremely early flowering and determinate inflorescences. ZCN12 T1 over-expression lines are sterile. Over-expression of ZCN14, ZCN15, and ZCN18 complements ft mutants and causes early flowering and indeterminate inflorescences. Over-expression of ZCN26 does not complement ft mutants and leads to a slightly late flowering. Number of rosette leaves are means ± SD.
(TIF)
X-axis represents developmental stages of apices as defined in Fig 5. SAM: shoot apical meristem, IF: inflorescence meristem, BM: branch meristem, DT: developing tassel, IT: immature tassel, PLT: plantlets. Y-axis represents average relative gene expression normalized against eukaryotic initiation factor 4-gamma (GenBank EU967723). Numeric t-test, p>0.05 are shown in S4 Table where expression in the transgenic shoot apices was compared to NTG apices. Error bars = ± SE.
(TIF)
A) to F) Quantification of SBP mRNA, regulated by miR156 cleavage. G) to I) Quantification of AP2-like mRNA, regulated by miR172 cleavage. X-axis represents days after sowing (DAS). Y-axis represents relative gene expression normalized against eukaryotic initiation factor 4-gamma (GenBank EU967723). Each data point represents a qRT-PCR value for an individual apex.
(TIF)
(XLS)
(XLS)
(XLS)
(XLS)
(XLS)
(XLS)
Data Availability Statement
All relevant data are within the manuscript and its Supporting Information files.