Abstract
Mechanical ventilation may cause ventilator-induced lung injury (VILI). Canonical Wnt signaling has been reported to serve an important role in the pathogenesis of VILI. Bioinformatics analysis revealed that canonical and non-canonical Wnt signaling pathways were activated in VILI. However, the role of non-canonical Wnt signaling in the pathogenesis of VILI remains unclear. The present study aimed to analyze the potential role of non-canonical Wnt signaling in VILI pathogenesis. Lung injury was assessed via Evans blue albumin permeability and histological scoring, as well as by inflammatory cytokine expression and total protein concentration in bronchoalveolar lavage fluid. The relative protein expression of canonical and non-canonical Wnt signaling pathway components were examined via western blotting and immunohistochemistry. The results demonstrated that 6 h of mechanical ventilation at low tidal volume (LTV; 6 ml/kg) or moderate tidal volume (MTV; 12 ml/kg) induced lung injury in sensitive A/J mice. Ventilation with MTV increased the protein levels of Wnt-induced secreted protein 1 (WISP1), Rho-associated protein kinase 1 (ROCK1), phosphorylated (p)-Ras homolog gene family, member A and p-C-Jun N-terminal kinase (JNK). Inhibition of ROCK1 by Y27632 and JNK by SP600125 attenuated MTV-induced lung injury and decreased the expression of proteins involved in non-canonical Wnt signaling, including WISP1. In conclusion, non-canonical Wnt signaling participates in VILI by modulating WISP1 expression, which has been previously noted as critical for VILI development. Therefore, the non-canonical Wnt signaling pathway may provide a preventive and therapeutic target in VILI.
Keywords: WNT1-inducible secreted protein 1, non-canonical Wnt signaling pathway, ventilator-induced lung injury, moderate tidal volume, low tidal volume, c-Jun N-terminal kinase
Introduction
Mechanical ventilation may cause ventilator-induced lung injury (VILI) (1–3) that leads to acute respiratory distress syndrome (ARDS) (4). Mortality from ARDS has been reduced by controlling tidal volume and ventilation pressure (5). Increasing evidence has shown that specific target proteins have critical function in VILI; these proteins include α-1-antitrypsin, ephrin A2, sphingosine-1-phosphate lyase, mothers against decapentaplegic homolog (Smad)4, p120-catenin, and SN50 (a cell-permeable inhibitor of nuclear factor-κB) (6-8). For example, Smad4 expression is significantly increased by ventilation in C57BL/6 mice, and Smad4 knockdown dramatically inhibited extracellular matrix remodeling by reducing α-smooth muscle actin (SMA), collagen I and collagen III expression (9). Zhao et al (10) reported that the degradation of p120-catenin causes VILI in C57BL/6 mice, and inhibitors of protein kinase Cα block c-Src kinase activation and p120-catenin degradation, thus alleviating VILI. Chian et al (11) demonstrated that SN50 attenuates inflammation and VILI. However, the pathogenesis of VILI remains unclear.
Wnt-induced secreted protein 1 (WISP1), also known as CCN4, is a member of the CCN family (12). Our previous studies demonstrated that WISP1 contributes to VILI. WISP1 expression is increased in Evans blue albumin (EBA)-sensitive A/J mice after 4 h of ventilation, and anti-WISP1 antibodies reduce moderate tidal volume (MTV)-induced EBA increase in A/J mice (13). Furthermore, WISP1 is reported to act through toll-like receptor 4 (TLR4) signaling to influence VILI (13). In addition, it was found that WISP1 combined with αvβ3 integrin signaling increases the TLR-induced inflammatory response in sepsis-induced lung injury (14). Arg-Gly-Asp-Ser peptides alleviate acute lung injury through WISP1-integrin β6 pathway inhibition in septic mice (15). The administration of Poly(I:C) exacerbates MTV-induced lung injury in a WISP1- and integrin β3-dependent manner, which involves activation of the extracellular signal-related kinase (ERK) signaling pathway (16). The Wnt signaling pathway includes canonical and non-canonical pathways. The non-canonical Wnt signaling pathway includes the Wnt/planar cell polarity and the Wnt/Ca2+ pathways, which are stimulated by the Wnt ligands Wnt5a or Wnt11 and are transduced through the Frizzled family and receptor tyrosine kinase like orphan receptor (Ror)1 or Ror2 co-receptor complexes (17). Previous studies have shown that non-canonical Wnt signaling promotes epithelial cell differentiation, capillary development and autocrine motility factor differentiation during lung maturation and in adult lung fibroblasts, asthma and usual interstitial pneumonia (UIP) (18-20). For example, Vuga et al (21) compared the gene expression profiles of lung fibroblasts from UIP patients to those of normal lung fibroblasts, and revealed that Wnt5a is significantly upregulated in UIP fibroblasts. Further analysis revealed that WNT5a promotes proliferation, increases fibronectin expression and inhibits H2O2-induced apoptosis in lung fibroblasts. However, the role of non-canonical WNT signaling in the pathogenesis of VILI is unclear.
In the present study, GEO2R was used to analyze the differentially expressed genes (DEGs) in VILI. Gene Ontology (GO) function and Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway enrichment analysis of DEGs was conducted. Next, the interaction of the identified enrichment pathways was analyzed. The Wnt signaling pathway was associated with VILI. In addition, the non-canonical Wnt signaling pathway may serve an important role in VILI. Therefore, the functional role and mechanisms of the non-canonical Wnt signaling in the pathogenesis of VILI were investigated. Sensitive A/J mice were used to establish MTV-induced lung injury model to test the hypothesis that non-canonical Wnt signaling has critical function in VILI. The results indicated that modulation of non-canonical Wnt signaling may provide novel preventive and therapeutic strategies in VILI in the future.
Materials and methods
Bioinformatics analysis of microarray data
Three data sets (GSE11434, GSE58169 and GSE7742) associated with VILI in mice were collected from the GEO database (www.ncbi.nlm.nih.gov/geo) (22–24).
Identification of differentially expressed genes (DEGs)
GEO2R (www.ncbi.nlm.nih.gov/geo/info/geo2r.html), an interactive web tool for comparing two or more groups of samples in a GEO series, was used to identify DEGs across experimental conditions. P<0.05 and log fold change >1 were chosen as the cut-off criteria. A Venn diagram, heatmap, and cluster profiler analysis were performed with R 3.5.1 software (cran.r-project.org/bin/windows/base/) in the 'limma' package.
Functional enrichment analysis of DEGs
KEGG Orthology Based Annotation System (KOBAS) 3.0 (kobas.cbi.pku.edu.cn) is a web server for gene/protein functional annotation and for identifying the enrichment of gene functional sets. KEGG pathway analysis was performed using the KOBAS 3.0 online tool (25,26). P<0.05 was set as the cut-off criterion.
VILI animal model
The animal study protocol was approved by University of Pittsburgh institutional animal care and use committee. Experiments were performed in accordance with the recommendations of the National Institutes of Health Guidelines for the Use of Laboratory Animals (27). Sensitive A/J mice (n=36; 8-9 weeks; 1:1 male:female; 20-25 g) were anesthetized by intraperitoneal pentobarbital injection (70 mg/kg). The trachea was exposed, cut and intubated. To analyze the effects of mechanical ventilation on lung injury, mice (n=6 each) were randomized to low tidal volume (LTV; 6 ml/kg, 140 breaths/min, 0 positive end-expiratory pressure), MTV (12 ml/kg, 100 breaths/min, 0 positive end-expiratory pressure), and spontaneous breathing groups for 6 h interventions, as described previously (28). To examine the Wnt pathway's effects on MTV-induced lung injury, MTV-exposed mice were randomized and intratracheally administered Y27632 [inhibitor of Rho-associated protein kinase 1 (ROCK1); 5 mg/kg], SP600125 [inhibitor of C-Jun N-terminal kinase (JNK); 6 mg/kg] prior to MTV, or were ventilated with MTV alone. Mice were subsequently sacrificed and one section of the right lung was stored in liquid nitrogen for western blotting and ELISAs; other lung sections were fixed in 10% formaldehyde for 24 h at room temperature for immunohistochemistry (IHC) analysis.
Measurement of alveolar-capillary permeability
The alveolar-capillary permeability was quantified by Evan's blue albumin (EBA) staining as previously described (13). In brief, EBA was prepared by adding bovine serum albumin (BSA; Sigma-Aldrich; Merck KGaA, Darmstadt, Germany) to 0.5% EB to a final concentration of 4% (0.6 mM). The solution was thoroughly dissolved by gently stirring with a magnet bar, and the EBA was then sterilely filtered through a 0.22 µm syringe filter and stored in aliquots at -80°C until use. Each aliquot was used only once for each animal to prevent cross-contamination. To evaluate the alveolar capillary barrier function, EBA (20 mg/kg) was administered via the internal jugular vein 1 h before sacrifice and tissue harvesting. At the termination of each experiment, all animals were euthanized, and blood samples were obtained via the right ventricle for plasma EBA measurement. The pulmonary vasculature was then flushed with PBS to remove blood components. The right lung was ligated at the level of the right mainstem bronchus, excised, and weighed and stored in liquid nitrogen for subsequent EB analysis. Once thawed, the lung tissue was homogenized in 2 ml PBS and then incubated with 2 ml formamide (Sigma-Aldrich; Merck KGaA) at 60°C for 18 h. Formamide extracts were centrifuged (Beckman TLX; Beckman Coulter, Inc., Brea, CA, USA) at 15,000×g for 30 min at 4°C, and the centrifuged supernatants were collected to quantify lung EBA content by dual-wavelength spectrophotometry (Beckman DU-640; Beckman Coulter) at 620 and 740 nm (29). EBA permeability index was calculated by dividing the corrected pulmonary tissue EBA absorbance at 620 nm (per g of lung tissue) by the corrected plasma EBA absorbance at 620 nm.
Histological examination
For histological examination, the lung of each animal was fixed in 4% paraformaldehyde overnight at 4°C. Then, 4 mm thick sections were paraffin embedded and stained with hematoxylin and eosin [(H&E) cat. no. P032IH; Auragene Bioscience] at room temperature for 10 min. As described previously, the histological score of the degree of lung injury was graded on a scale from 0 to 4 (0, absent and appeared normal; 1, light; 2, moderate; 3, strong; and 4, intense) for the following pathological features: Alveolar congestion, neutrophil infiltration, hemorrhage and thickness of alveolar wall membrane formation. Five fields of view were assessed (30).
Cytokine expression in the bronchoalveolar lavage fluid (BALF)
Mouse tumor necrosis factor (TNF)-α (cat. no. ab100747) and interleukin (IL)-6 (cat. no. ab100713) ELISA kits were purchased from Abcam (Cambridge, UK) and used to measure the concentrations of TNF-α and IL-6 in BALF. All reagents were equilibrated to room temperature and prepared prior to use according to the manufacturer's protocol. The standards and samples were added into wells and incubated for 2.5 h at room temperature. The wells were washed with wash buffer four times and biotinylated TNF-α and IL-6 were added into the wells and incubated 1 h at room temperature with gentle shaking. Following four washes, horseradish peroxidase (HRP)-streptavidin solution was added into the wells and incubated for 45 min at room temperature. The wash steps were repeated, and TMB substrate solution was added into the wells and incubated for 30 min in the dark at room temperature. Then, stop solution was added into the wells and a microplate reader was used to determine the optical density of each well at 450 nm.
Total protein concentration in BALF was detected by using bicinchoninic acid (BCA) protein assay kit (cat. no. P001B, Auragene Bioscience) according to the manufacturer's instructions.
Western blot analysis
Frozen lung tissues were thawed and lysed in radioimmunoprecipitation assay buffer (cat. no. P002A; Auragene Bioscience, Changsha, China) with protease inhibitors (cat. no. P019A; Auragene Bioscience) and phenylmethylsulfonyl fluoride (cat. no. P018A; Auragene Bioscience). Protein concentration was measured with a standard BCA protein assay. Proteins were separated by 10% SDS-PAGE and transferred to nitrocellulose membranes. Membranes were blocked in Tris-buffered saline with 0.5% Tween-20 (TBST) and 3% BSA for 1 h at room temperature and incubated overnight with WISP1 (cat. no. 18166-1-AP; 1:1,000; ProteinTech Group, Inc., Chicago, IL, USA), Ras homolog gene family, member A (RhoA; cat. no. ab54835; 1:1,000; Abcam), phosphorylated (p-)RhoA (cat. no. ab41435; 1:1,000; Abcam), JNK (cat. no. sc-7345; 1:500; Santa Cruz Biotechnology, Inc., Dallas, TX, USA), p-JNK (cat. no. sc-6254; 1:500; Santa Cruz Biotechnology, Inc.) or β-actin (cat. no. ab8226, 1:5,000; Abcam) antibodies. The membranes were washed with TBST five times and incubated with HRP-conjugated goat anti-mouse (cat. no. SA001, 1:15,000) and rabbit (cat. no. SA009; 1:15,000) IgG secondary antibodies (Auragene Bioscience) for 40 min at room temperature. Proteins were detected by chemiluminescence (cat. no. P020WB; Auragene Bioscience) and quantified using ImageJ version 6.0 software (National Institutes of Health, Bethesda, MA, USA).
Immunohistochemistry
For histological examination, a lung from each animal was fixed in 4% paraformaldehyde overnight at 4°C. Then, the samples were embedded in paraffin and cut into 3-µm-thick slices. Tissue slides were heated for 2 h at 65°C and subsequently incubated in xylene for 30 min at room temperature and in graded alcohol (100, 95, 85 and 75% alcohol for 5 min each). The slides were blocked in 3% H2O2 for 15 min at room temperature to inhibit endogenous peroxidase activity and placed in 1 M sodium citrate buffer (cat. no. P019IH; Auragene Bioscience) for antigen retrieval. The slides were incubated with the primary antibodies against WISP1 (cat. no. 18166-1-AP; 1:100; ProteinTech Group, Inc.), or p-Jnk1/2/3 (cat. no. YP0157; 1:200; ImmunoWay Biotechnology Company, Plano, TX, USA) overnight at 4°C. Then, the slides were rinsed with PBS five times and incubated with HRP-conjugated secondary antibodies (cat. no. SA009; 1:1,000; Auragene Bioscience) for 30 min. The slides were stained with DAB (cat. no. P013IH; Auragene Bioscience) solution for 1 min, counter-stained with hematoxylin (cat. no. C0107; Beyotime Institute of Biotechnology, Haimen, China) for 15 min at room temperature and dehydrated in a graded alcohol (85 and 95% for 5 min each) at room temperature prior to coverslipping.
Statistical analysis
Data are presented as the mean ± standard error of the mean, and were analyzed by one-way analysis of variance followed by Tukey's post-hoc test. Statistical analyses were performed using GraphPad Prism 5.0 (GraphPad Software, Inc., La Jolla, CA, USA). P<0.05 was considered to indicate a statistically significant difference.
Results
Identification of differentially expressed genes
A total of 157, 757 and 1,086 DEGs were identified from GSE11434, GSE58169, and GSE7742, respectively. There were 10 common DEGs from the three datasets that were associated with lung injury (Fig. 1), including IL-1β, IL-1 receptor 2, stratifin, C-type lectin domain family 4 member D, amphiregulin, suppressor of cytokine signaling 3, S100 calcium binding protein A1, matrix metalloproteinase 8, solute carrier family 7 member 11 and early growth response 1.
Figure 1.
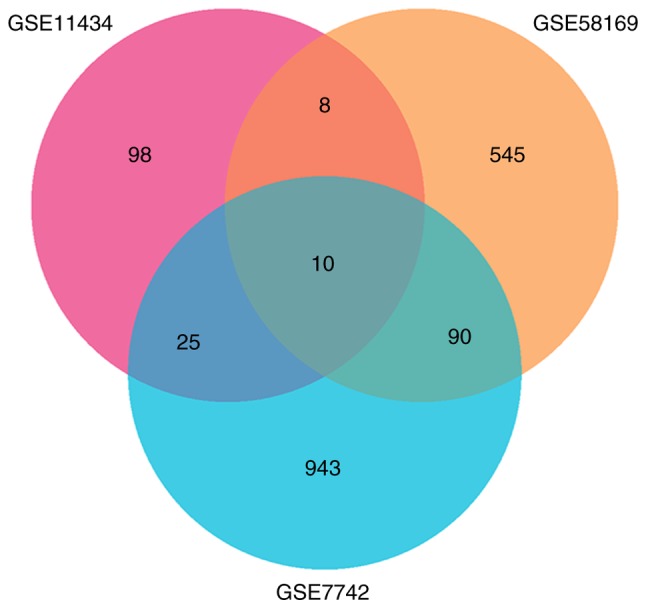
Identification of differentially expressed genes in ventilator-induced lung injury by Venn diagram. DEGs were identified from GSE11434, GSE58169, and GSE7742 datasets in VILI, with 10 common DEGs in the three datasets.
Functional enrichment analysis of DEGs
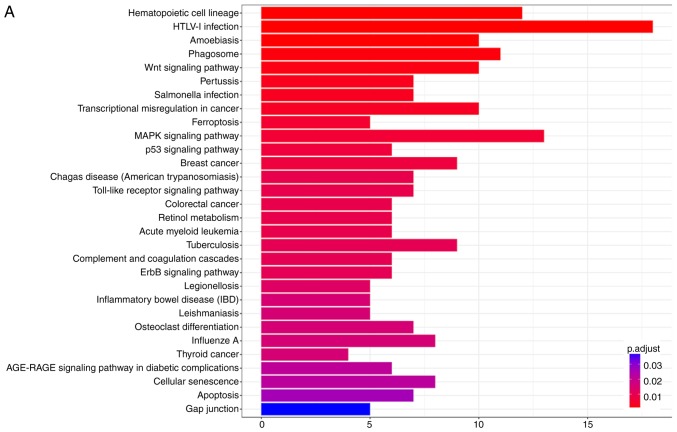
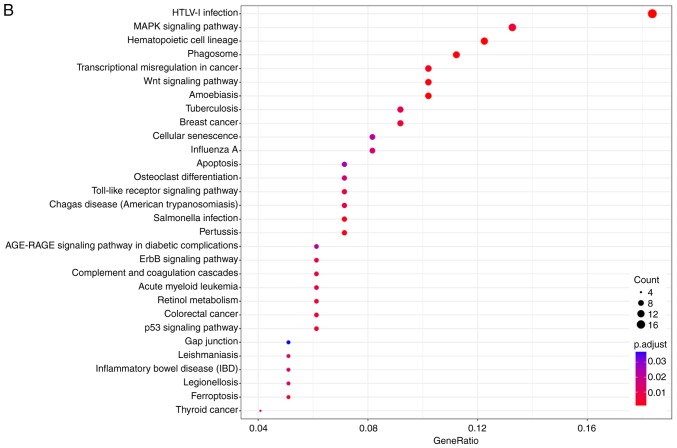
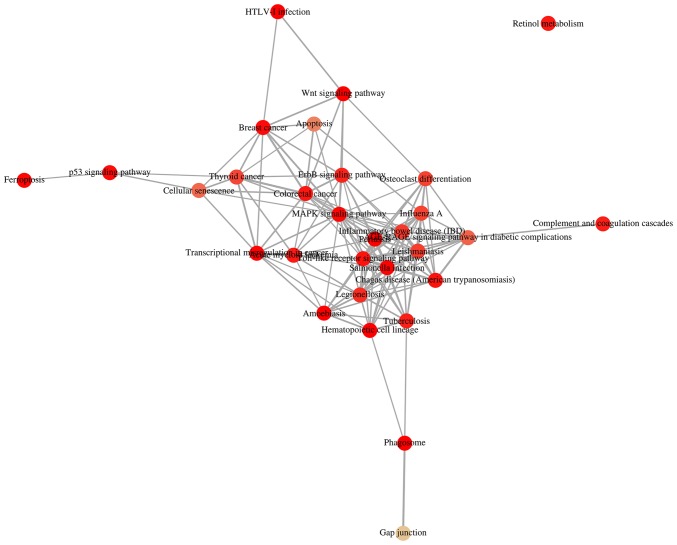
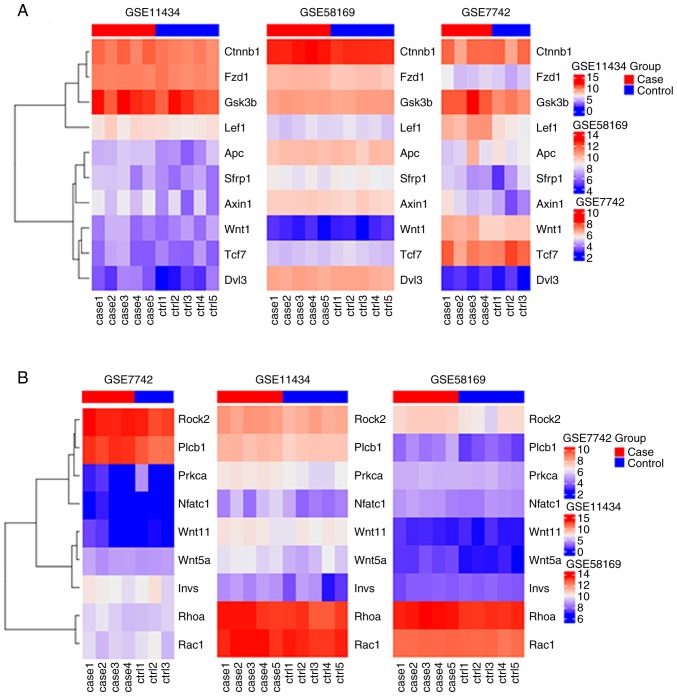
KOBAS 3.0 was used to analyze the pathway enrichment of identified DEGs. KEGG analysis revealed that the upregulated DEGs were predominantly enriched in 'HTLV-1 infection', 'Wnt signaling pathway' and 'MAPK signaling pathway', as well as pathways associated with VILI (31,32) (Fig. 2A and B). It was demonstrated that 'HTLV-1 infection' and 'MAPK signaling pathway' were associated with 'Wnt signaling pathway' (Fig. 3). It has been reported that Wnt/β-catenin signaling pathway is activated in early VILI, and inhibition of the Wnt/β-catenin signaling pathway suppresses VILI (31,33) Thus, the Wnt pathway was selected for further study. Heatmap analysis revealed that Wnt1 and GSK3β were significantly increased in VILI, compared with the control groups (Fig. 4A). In addition, the non-canonical signaling pathway factors Wnt5a, Wnt11 and ROCK2 were activated in VILI (Fig. 4B). The KEGG pathway enrichment demonstrated a relationship between the identified DEGs and VILI. These analyses support the reliability of the results in the present study.
Figure 2.
KEGG pathway enrichment analysis of ventilator-induced lung injury. (A) KEGG pathway enrichment analysis of differentially expressed genes. (B) Gene ratio of DEGs in KEGG pathway enrichment analysis. DEGs, differentially expressed genes; KEGG, Kyoto Encyclopedia of Genes and Genomes.
Figure 3.
Relationship of top KEGG pathways enriched by ventilator-induced lung injury. Notably, DEGs were enriched in HTLV-1 infection, Wnt signaling, and MAPK signaling. KEGG, Kyoto Encyclopedia of Genes and Genomes; DEG, differentially expressed genes; HTLV-1, human T-lymphotropic virus; MAPK, mitogen-activated protein kinase.
Figure 4.
Wnt signaling pathway gene expression in VILI. (A) Canonical and (B) non-canonical Wnt signaling pathway genes in VILI. VILI, ventilator-induced lung injury.
The non-canonical Wnt signaling pathway is activated in VILI
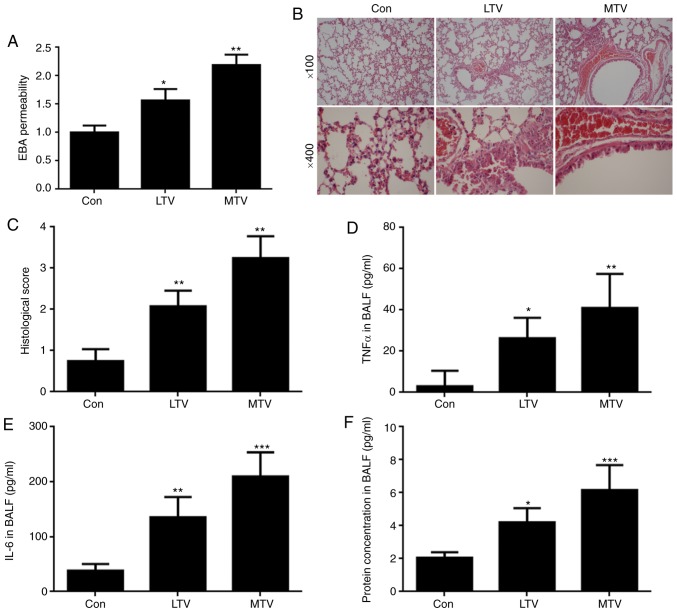
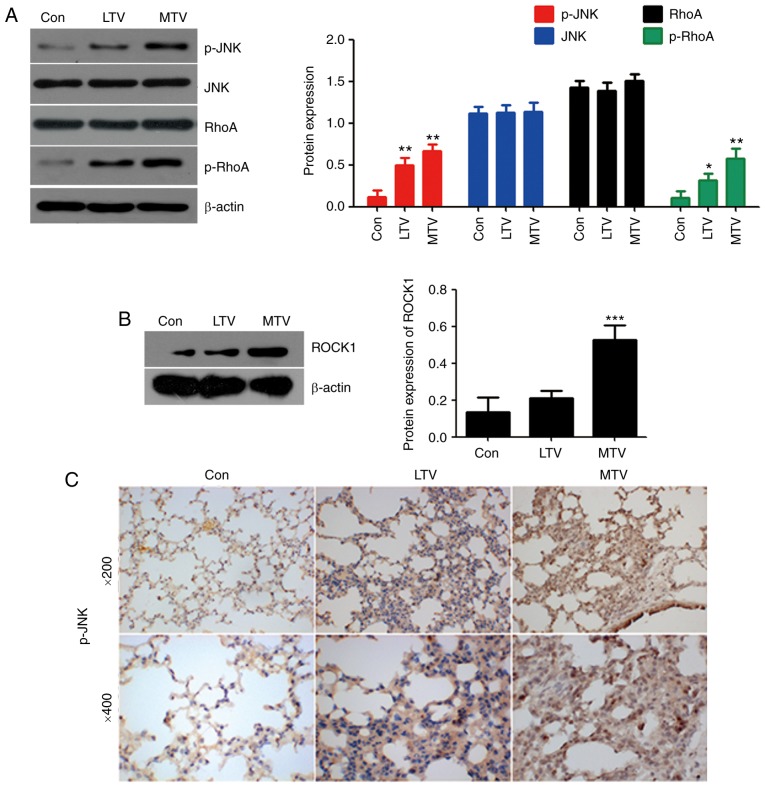
To investigate the role of non-canonical Wnt signaling in VILI, animal experiments in sensitive A/J mice. To assess VILI, alveolar-capillary permeability was measured, H&E staining was performed, and the expression of TNF-α and IL-6, as well as total protein, was measured. Permeability was increased in the lungs of A/J mice ventilated with LTV and MTV compared with the spontaneous breathing group (Fig. 5A). Histological examination illustrated that ventilation with LTV and MTV significantly increased inflammatory cell infiltration, pulmonary edema and alveolar septal thickening, findings which were confirmed by histological scoring (Fig. 5B and C). Similarly, increased TNF-α (Fig. 5D), IL-6 (Fig. 5E), and total protein (Fig. 5F) expression was detected in the LTV and MTV groups. Collectively, these results demonstrated that mechanical ventilation with LTV or MTV induced lung injury in A/J mice. To investigate the role of non-canonical Wnt signaling in VILI, the protein levels of important non-canonical Wnt signaling pathway components were examined via western blotting and IHC. RhoA and JNK are important proteins in non-canonical Wnt signaling (34). Mechanical ventilation did not alter the expression of RhoA or JNK. However, the expressions of p-RhoA, ROCK1 and p-JNK were elevated in the LTV and MTV groups, compared with the spontaneous breathing group (Fig. 6A and B). Furthermore, IHC revealed that p-JNK was dramatically increased in the LTV and MTV groups, compared with the spontaneous breathing group (Fig. 6C). Taken together, these data indicated that non-canonical Wnt signaling pathway participated in the pathogenesis of VILI in A/J mice.
Figure 5.
Lung injury following mechanical ventilation with LTV or MTV in A/J sensitive mice. (A) EBA permeability increased in mice ventilated with LTV (6 ml/kg) and MTV (12 ml/kg) for 6 h (n=4-6). (B) Ventilation with LTV and MTV significantly increased inflammatory cell infiltration, pulmonary edema, alveolar septal thickening and (C) the histological score (n=4-6). (D) TNF-α and (E) IL-6 expression, as well as (F) total protein concentration in BALF was increased in mice ventilated with LTV and MTV (n=4–6). *P<0.05, **P<0.01, ***P<0.001 vs. control. LTV, low tidal volume; MTV, moderate tidal volume; BALF, bronchoalveolar lavage fluid; EBA, Evan's blue albumin; TNF, tumor necrosis factor; IL-6, interleukin 6.
Figure 6.
MTV ventilation increased non-canonical Wnt signaling protein expression in the lungs of A/J sensitive mice. (A) p-RhoA, p-JNK and (B) ROCK1 protein expression was measured by western blotting. β-actin was the loading control (n=4-6). *P<0.05, **P<0.01, ***P<0.001 vs. control. (C) Immunohistochemical staining for p-JNK in the lungs following mechanically ventilating with LTV and MTV for 6 h in A/J mice. LTV, low tidal volume; MTV, moderate tidal volume; p-, phosphorylated; JNK, c-Jun N terminal kinase; ROCK1, Rho-associated protein kinase 1; RhoA, Ras homolog gene family, member A.
Inhibition of non-canonical Wnt signaling attenuated MTV-induced lung injury
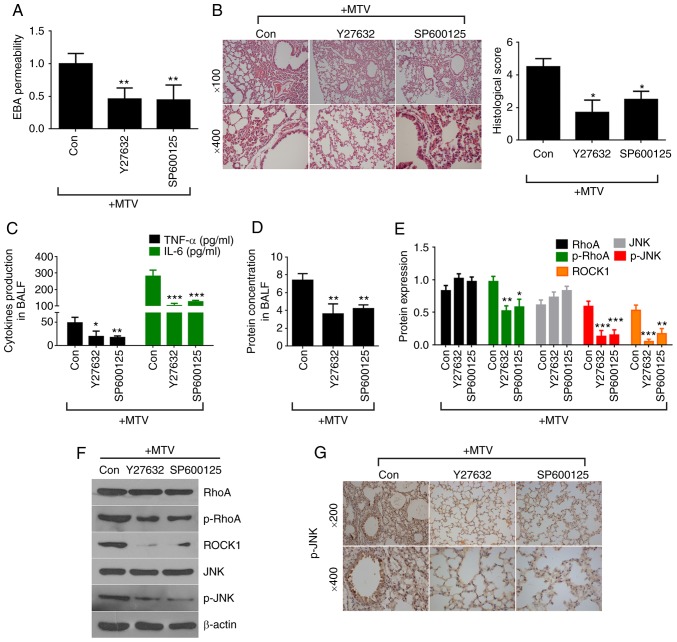
To further investigate the functional role of non-canonical Wnt signaling in the pathogenesis of VILI, inhibitors of target proteins in the pathway were used. Sensitive A/J mice were given Y27632 (ROCK1 inhibitor) or SP600125 (JNK inhibitor) intratracheally before undergoing ventilation with MTV for 6 h. Y27632 and SP600125 significantly decreased MTV-induced EBA permeability (Fig. 7A). Histological analysis and scoring illustrated that Y27632 and SP600125 attenuated MTV-induced inflammatory cell infiltration, pulmonary edema and alveolar septal thickening (Fig. 7B and C). Total protein concentration, as well as TNF-α and IL-6 expression in BALF were notably decreased following treatment with Y27632 or SP600125 (Fig. 7D and E). In addition, the protein expression of JNK, p-JNK, RhoA, p-RhoA, and ROCK1 was measured by western blotting and IHC. Western blot analysis showed that inhibition of ROCK1 (Y27632) and JNK (SP600125) dramatically decreased p-JNK, p-RhoA, and ROCK1 levels compared to the MTV group (Fig. 7F and G). IHC confirmed that p-JNK (Fig. 7H) expression was attenuated in VILI following administration of Y27632 or SP600125. Collectively, these data indicated that inhibition of non-canonical and canonical Wnt signaling pathways may effectively protect lung tissues against VILI in A/J mice.
Figure 7.
Inhibition of non-canonical Wnt signaling attenuates MTV-induced lung injury. (A) EBA permeability was decreased in A/J mice administered Y27632 or SP600125 prior to MTV ventilation compared to mice ventilated with MTV alone (n=3). (B) Administration of Y27632 or SP600125 reduced MTV-induced inflammatory cell infiltration, pulmonary edema, alveolar septal thickening and histological scores (n=4-6). (C) TNF-α and IL-6 expression, as well as (D) total protein concentration decreased in BALF in A/J mice following the administration of Y27632 or SP600125 prior to MTV ventilation (n=4-6). (E) Protein quantification in Y27632 or SP600125 treatment reduced the ventilation-induced increase in p-RhoA, p-JNK and ROCK1. *P<0.05, **P<0.01, ***P<0.001 vs. control. (F) Western blotting for p-RhoA, p-JNK and ROCK1 protein expression in A/J mice after administering Y27632 or SP600125 prior to MTV ventilation. (G) Immunohistochemical staining for p-JNK expression was decreased in A/J mice following the administration of Y27632 or SP600125 prior to MTV ventilation. BALF, bronchoalveolar lavage fluid; EBA, Evan's blue albumin; MTV, moderate tidal volume; p-, phosphorylated; JNK, c-Jun N terminal kinase; ROCK1, Rho-associated protein kinase 1; RhoA, Ras homolog gene family, member A; TNF, tumor necrosis factor; IL-6, interleukin 6; Y27632, ROCK1 inhibitor; SP600125, JNK inhibitor.
Non-canonical Wnt signaling regulates WISP1 expression in VILI
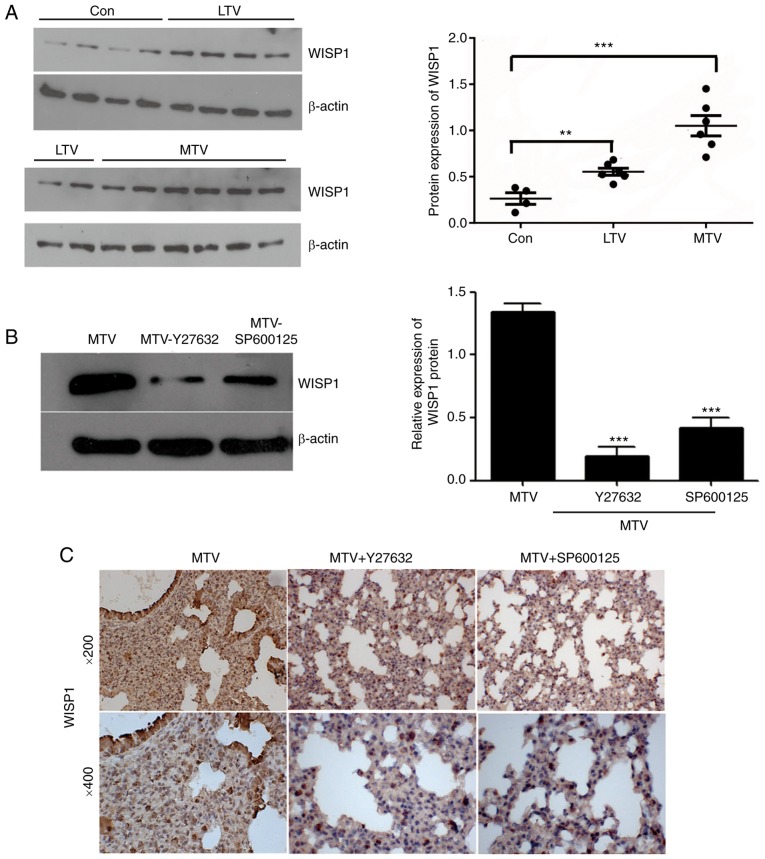
Western blotting was performed to analyze the regulation of WISP1 by the non-canonical Wnt pathway. The results revealed that WISP1 expression was increased in mice ventilated with LTV and MTV, compared with the control group (Fig. 8A). The inhibition of ROCK1 and JNK markedly decreased MTV-induced WISP1 expression (Fig. 8B and C). These results revealed that non-canonical Wnt signaling promoted VILI by upregulating WISP1 expression in A/J mice.
Figure 8.
WISP1 expression in the lungs of A/J mice following the administration of Y27632 or SP600125 prior to MTV ventilation. (A) WISP1 expression was increased in mice ventilated with LTV and MTV. **P<0.01, ***P<0.001 vs. Con group. (B) Administration of Y27632 or SP600125 decreased WISP1 protein level. ***P<0.001 vs. MTV group. (C) WISP1 level expression detected by immunohistochemistry was decreased in the lungs of mice given Y27632 or SP600125. Con, control; LTV, low tidal volume; MTV, moderate tidal volume; Y27632, ROCK1 inhibitor; SP600125, JNK inhibitor; WISP1, Wnt-induced secreted protein 1.
Discussion
The present study demonstrated that non-canonical WNT pathway proteins were overexpressed in VILI mice. To the best of our knowledge, this is the first report to examine the non-canonical WNT pathway in an experimental model of VILI in A/J mice. The main findings were as follows: i) p-RhoA and JNK expression was increased in lungs following mechanical ventilation with LTV or MTV; ii) inhibition of the non-canonical WNT pathway by inhibiting ROCK1 and JNK markedly decreased MTV-induced lung injury as assessed by EBA permeability, histological scoring and proinflammatory cytokine levels; and iii) non-canonical Wnt signaling promoted VILI through the upregulation of WISP1 expression. These data suggested that non-canonical Wnt signaling served significant roles in the regulation of WISP1 and the pathogenesis of VILI in A/J mice.
Acute lung injury (ALI) has been investigated (35,36), but the molecular mechanisms involved in injury induced by mechanical ventilation are not well understood. In the present study, it was found that the upregulated DEGs in VILI were predominantly enriched in HTLV-1 infection, Wnt signaling and MAPK signaling pathways. The correlation of these signaling pathways was analyzed; HTLV-1 infection and the MAPK signaling pathway were associated with Wnt signaling. It has been previously reported that canonical WNT signaling is activated in ALI induced by lipopolysaccharide by driving the Th17 response in mice (37). The knockdown of TGF-β1 and FGF-2 via the inhibition of Wnt/β-catenin signaling may serve as a potential therapeutic strategy for bleomycin-induced pulmonary fibrosis (38). Additionally, Wnt signaling has critical involvement in VILI (31). For example, MTV exacerbates Poly(I:C)-induced lung injury in a WISP1- and integrin β3-dependent manner, involving, at least partially, activation of the ERK pathway (16). Furthermore, activation of the Wnt/β-catenin signaling pathway by mechanical ventilation is associated with ventilator-induced pulmonary fibrosis in healthy lungs (31). miR-127 acts downstream of TLR4 to promote proinflammatory M1 macrophage development in vitro and in vivo, which is achieved, at least partially, through a JNK-dependent mechanism (39).
The current in vitro study revealed that VILI activated non-canonical Wnt signaling in sensitive A/J mice. Initially, three GEO datasets were selected and DEGs were identified. KEGG analysis determined that the Wnt signaling pathway was a common node affecting other pathways. Wnt5a and Wnt11, which are important non-canonical Wnt ligands, are activated in VILI (40-42). Increasing evidence has shown that the non-canonical Wnt pathway functions in cell injury. For example, primary cilia regulate non-canonical Wnt signaling responses in the injured kidney (43). Nakamura et al (44) reported that secreted Frizzled-related protein 5 (SFRP5), an inhibitor of non-canonical Wnt signaling, protects cardiac cells following ischemia/reperfusion injury (44). Non-canonical Wnt signaling is also associated with fibrosis (45,46). The cardiac microenvironment uses non-canonical Wnt signaling to activate monocytes following myocardial infarction (45). Frizzled-7 mediates TGF-β-induced pulmonary fibrosis by mediating non-canonical Wnt signaling (46). In the present study, it was demonstrated that VILI induced by MTV increased p-RhoA and p-JNK expression in A/J mice. Treatment with Y27632 and SP600125, inhibitors of ROCK and JNK, respectively, attenuated MTV-induced lung injury by decreasing WISP1 protein expression.
This is the first demonstration that the non-canonical Wnt signaling pathway induced WISP1 expression and contributed to MTV-induced lung injury in A/J mice. However, the present study had several limitations. First, the data does not fully confirm that the non-canonical Wnt signaling pathway was involved in disrupting lung repair or in the early development of fibrosis in VILI. Second, WNT5A-deficient animals were not used to irrefutably demonstrate that this pathway contributed to the pathogenesis of VILI. Third, the mechanism by which the non-canonical Wnt signaling pathway regulated WISP1 remains unclear and requires further study.
In conclusion, the present study confirmed that mechanical ventilation induced lung injury, which was accompanied by increasing WISP1 expression. Non-canonical Wnt signaling was involved in mechanical VILI, and suppression of this signaling significantly alleviated VILI. WISP1 expression was regulated by the non-canonical Wnt signaling pathway in A/J mice. The modulation of both WISP1 and Wnt signaling may provide novel therapeutic strategies in VILI.
Acknowledgments
We thank Ms. Christine Heiner, Scientific Writer for the Departments of Anesthesiology and Surgery at the University of Pittsburgh, for her assistance with scientific editing that greatly improved the manuscript. We would also like to show our gratitude to Ms. Karla Woosloose, laboratory manager, for assisting with experiments.
Abbreviations
- WISP1
Wnt-induced secreted protein 1
- VILI
ventilator-induced lung injury
- MTV
moderate tidal volume
- LTV
low tidal volume
- p-JNK
phosphorylated c-Jun N-terminal kinase
- ROCK1
Rho-associated protein kinase 1
Funding
This study was supported by grants from the National Institute of Health (grant nos. R01-GM-50441 and R01-GM-108639; to TB and LMZ) and the Natural Science Foundation of Hunan Province of China (grant no. 2017JJ2177; to YFX).
Availability of data and materials
Three data sets (GSE11434, GSE58169 and GSE7742) related to ventilator-induced lung injury in mice were collected from the GEO database (www.ncbi.nlm.nih.gov/geo).
Authors' contributions
YFX and LMZ designed the experiments and study. JC, JFY and WO performed the experiments. BP and TB wrote the manuscript, analyzed and interpreted the data. All authors read and approved the final manuscript.
Ethics approval and consent to participate
All procedures performed in studies involving animals were in accordance with the ethical standards of the Institution Review of Hunan Cancer Hospital.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that there are no competing interests.
References
- 1.Hashemian SM, Mohajerani SA, Jamaati HR. Ventilator-induced lung injury. N Engl J Med. 2014;370:979–980. doi: 10.1056/NEJMc1400293. [DOI] [PubMed] [Google Scholar]
- 2.Rahaman U. Mathematics of ventilator-induced lung injury. Indian J Crit Care Med. 2017;21:521–524. doi: 10.4103/ijccm.IJCCM_411_16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Beitler JR, Malhotra A, Thompson BT. Ventilator-induced lung injury. Clin Chest Med. 2016;37:633–646. doi: 10.1016/j.ccm.2016.07.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Plötz FB. Ventilator-induced lung injury. Intensive Care Med. 2001;27:452. doi: 10.1007/s001340000831. [DOI] [PubMed] [Google Scholar]
- 5.Amato MB, Barbas CS, Medeiros DM, Magaldi RB, Schettino GP, Lorenzi-Filho G, Kairalla RA, Deheinzelin D, Munoz C, Oliveira R, et al. Effect of a protective-ventilation strategy on mortality in the acute respiratory distress syndrome. N Engl J Med. 1998;338:347–354. doi: 10.1056/NEJM199802053380602. [DOI] [PubMed] [Google Scholar]
- 6.Zhu H, He J, Liu J, Zhang X, Yang F, Liu P, Wang S. Alpha 1-antitrypsin ameliorates ventilator-induced lung injury in rats by inhibiting inflammatory responses and apoptosis. Exp Biol Med (Maywood) 2018;243:87–95. doi: 10.1177/1535370217740852. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Park BH, Shin MH, Douglas IS, Chung KS, Song JH, Kim SY, Kim EY, Jung JY, Kang YA, Chang J, et al. Erythropoietin-producing hepatoma receptor tyrosine kinase A2 modulation associates with protective effect of prone position in ventilator-induced lung injury. Am J Respir Cell Mol Biol. 2018;58:519–529. doi: 10.1165/rcmb.2017-0143OC. [DOI] [PubMed] [Google Scholar]
- 8.Suryadevara V, Fu P, Ebenezer DL, Berdyshev E, Bronova IA, Huang LS, Harijith A, Natarajan V. Sphingolipids in ventilator induced lung injury: Role of sphingosine-1-phosphate. lyase Int J Mol Sci. 2018;19:E114. doi: 10.3390/ijms19010114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Huang X, Zhou W, Ding S. Downregulated Smad4 affects extracellular matrix remodeling in ventilator-induced lung injury. Ann Clin Lab Sci. 2016;46:451–456. [PubMed] [Google Scholar]
- 10.Zhao T, Zhao H, Li G, Zheng S, Liu M, Gu C, Wang Y. Role of the PKCα-c-Src tyrosine kinase pathway in the mediation of p120-catenin degradation in ventilator-induced lung injury. Respirology. 2016;21:1404–1410. doi: 10.1111/resp.12858. [DOI] [PubMed] [Google Scholar]
- 11.Chian CF, Chiang CH, Chuang CH, Liu SL, Tsai CL. SN50, a cell-permeable-inhibitor of nuclear factor-κB, attenuates ventilator-induced lung injury in an isolated and perfused rat lung model. Shock. 2016;46:194–201. doi: 10.1097/SHK.0000000000000563. [DOI] [PubMed] [Google Scholar]
- 12.Jun JI, Lau LF. Taking aim at the extracellular matrix: CCN proteins as emerging therapeutic targets. Nat Rev Drug Discov. 2011;10:945–963. doi: 10.1038/nrd3599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Li HH, Li Q, Liu P, Liu Y, Li J, Wasserloos K, Chao W, You M, Oury TD, Chhinder S, et al. WNT1-inducible signaling pathway protein 1 contributes to ventilator-induced lung injury. Am J Respir Cell Mol Biol. 2012;47:528–535. doi: 10.1165/rcmb.2012-0127OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Chen Z, Ding X, Jin S, Pitt B, Zhang L, Billiar T, Li Q. WISP1-αvβ3 integrin signaling positively regulates TLR-triggered inflammation response in sepsis induced lung injury. Sci Rep. 2016;6:28841. doi: 10.1038/srep28841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Ding X, Wang X, Zhao X, Jin S, Tong Y, Ren H, Chen Z, Li Q. RGD peptides protects against acute lung injury in septic mice through Wisp1-integrin β6 pathway inhibition. Shock. 2015;43:352–360. doi: 10.1097/SHK.0000000000000313. [DOI] [PubMed] [Google Scholar]
- 16.Jin S, Chen Z, Ding X, Zhao X, Jiang X, Tong Y, Billiar TR, Li Q. Mechanical ventilation augments poly(I:C)induced lung injury via a WISP1-integrin β3 dependent pathway in mice. Mol Med. 2016;22:54–63. doi: 10.2119/molmed.2015.00233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Borcherding N, Kusner D, Kolb R, Xie Q, Li W, Yuan F, Velez G, Askeland R, Weigel RJ, Zhang W. Paracrine WNT5A signaling inhibits expansion of tumor-initiating cells. Cancer Res. 2015;75:1972–1982. doi: 10.1158/0008-5472.CAN-14-2761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Ishitani T, Kishida S, Hyodo-Miura J, Ueno N, Yasuda J, Waterman M, Shibuya H, Moon RT, Ninomiya-Tsuji J, Matsumoto K. The TAK1-NLK mitogen-activated protein kinase cascade functions in the Wnt-5a/Ca(2+) pathway to antagonize Wnt/beta-catenin signaling. Mol Cell Biol. 2003;23:131–139. doi: 10.1128/MCB.23.1.131-139.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Li C, Xiao J, Hormi K, Borok Z, Minoo P. Wnt5a participates in distal lung morphogenesis. Dev Biol. 2002;248:68–81. doi: 10.1006/dbio.2002.0729. [DOI] [PubMed] [Google Scholar]
- 20.Li C, Hu L, Xiao J, Chen H, Li JT, Bellusci S, Delanghe S, Minoo P. Wnt5a regulates Shh and Fgf10 signaling during lung development. Dev Biol. 2005;287:86–97. doi: 10.1016/j.ydbio.2005.08.035. [DOI] [PubMed] [Google Scholar]
- 21.Vuga LJ, Ben-Yehudah A, Kovkarova-Naumovski E, Oriss T, Gibson KF, Feghali-Bostwick C, Kaminski N. WNT5A is a regulator of fibroblast proliferation and resistance to apoptosis. Am J Respir Cell Mol Biol. 2009;41:583–589. doi: 10.1165/rcmb.2008-0201OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Spassov S, Pfeifer D, Strosing K, Ryter S, Hummel M, Faller S, Hoetzel A. Genetic targets of hydrogen sulfide in ventilator-induced lung injury-a microarray study. PLoS One. 2014;9:e102401. doi: 10.1371/journal.pone.0102401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Dolinay T, Wu W, Kaminski N, Ifedigbo E, Kaynar AM, Szilasi M, Watkins SC, Ryter SW, Hoetzel A, Choi AM. Mitogen-activated protein kinases regulate susceptibility to ventilator-induced lung injury. PLoS One. 2008;3:e1601. doi: 10.1371/journal.pone.0001601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Wray C, Mao Y, Pan J, Chandrasena A, Piasta F, Frank JA. Claudin-4 augments alveolar epithelial barrier function and is induced in acute lung injury. Am J Physiol Lung Cell Mol Physiol. 2009;297:L219–L227. doi: 10.1152/ajplung.00043.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Xie C, Mao X, Huang J, Ding Y, Wu J, Dong S, Kong L, Gao G, Li CY, Wei L. KOBAS 2.0: A web server for annotation and identification of enriched pathways and diseases. Nucleic Acids Res. 2011;39(Web Server issue):W316–W322. doi: 10.1093/nar/gkr483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Kanehisa M, Sato Y, Kawashima M, Furumichi M, Tanabe M. KEGG as a reference resource for gene and protein annotation. Nucleic Acids Res. 2016;44:D457–D462. doi: 10.1093/nar/gkv1070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Kilkenny C, Browne WJ, Cuthill IC, Emerson M, Altman DG. Improving bioscience research reporting: The ARRIVE guidelines for reporting animal research. PLoS Biol. 2010;8:e1000412. doi: 10.1371/journal.pbio.1000412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Chen T, Chen C, Zhang Z, Zou Y, Peng M, Wang Y. Toll-like receptor 4 knockout ameliorates neuroinflammation due to lung-brain interaction in mechanically ventilated mice. Brain Behav Immun. 2016;56:42–55. doi: 10.1016/j.bbi.2016.04.004. [DOI] [PubMed] [Google Scholar]
- 29.Li H, Su X, Yan X, Wasserloos K, Chao W, Kaynar AM, Liu ZQ, Leikauf GD, Pitt BR, Zhang LM. Toll-like receptor 4-myeloid differentiation factor 88 signaling contributes to ventilator-induced lung injury in mice. Anesthesiology. 2010;113:619–629. doi: 10.1097/ALN.0b013e3181e89ab2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Achouiti A, van der Meer AJ, Florquin S, Yang H, Tracey KJ, van't Veer C, de Vos AF, van der Poll T. High-mobility group box 1 and the receptor for advanced glycation end products contribute to lung injury during Staphylococcus aureus pneumonia. Crit Care. 2013;17:R296. doi: 10.1186/cc13162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Villar J, Cabrera NE, Valladares F, Casula M, Flores C, Blanch L, Quilez ME, Santana-Rodríguez N, Kacmarek RM, Slutsky AS. Activation of the Wnt/β-catenin signaling pathway by mechanical ventilation is associated with ventilator-induced pulmonary fibrosis in healthy lungs. PLoS One. 2011;6:e23914. doi: 10.1371/journal.pone.0023914. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Tao G, Pan L, Jing R, Lin F, Dai H, Ge W. Study on Rac1/MAPK/ERK pathway mediated mechanism and role in rats with ventilator induced lung injury. Zhonghua Wei Zhong Bing Ji Jiu Yi Xue. 2017;29:249–254. doi: 10.3760/cma.j.issn.2095-4352.2017.03.011. In Chinese. [DOI] [PubMed] [Google Scholar]
- 33.Villar J, Cabrera NE, Casula M, Valladares F, Flores C, López-Aguilar J, Blanch L, Zhang H, Kacmarek RM, Slutsky AS. WNT/β-catenin signaling is modulated by mechanical ventilation in an experimental model of acute lung injury. Intensive Care Med. 2011;37:1201–1209. doi: 10.1007/s00134-011-2234-0. [DOI] [PubMed] [Google Scholar]
- 34.Xiao J, Zhou H, Wu N, Wu L. The non-canonical Wnt pathway negatively regulates dendritic cell differentiation by inhibiting the expansion of Flt3+ lymphocyte-primed multipotent precursors. Cell Mol Immunol. 2015;13:593–604. doi: 10.1038/cmi.2015.39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Ouyang W, Zhou H, Liu C, Wang S, Han Y, Xia J, Xu F. 25-Hydroxycholesterol protects against acute lung injury via targeting MD-2. J Cell Mol Med. 2018;22:5494–5503. doi: 10.1111/jcmm.13820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Xu F, Diao R, Liu J, Kang Y, Wang X, Shi L. Curcumin attenuates staphylococcus aureus-induced acute lung injury. Clin Respir J. 2015;9:87–97. doi: 10.1111/crj.12113. [DOI] [PubMed] [Google Scholar]
- 37.Cheng L, Zhao Y, Qi D, Li W, Wang D. Wnt/β-catenin pathway promotes acute lung injury induced by LPS through driving the Th17 response in mice. Biochem Biophys Res Commun. 2018;495:1890–1895. doi: 10.1016/j.bbrc.2017.12.058. [DOI] [PubMed] [Google Scholar]
- 38.Chen X, Shi C, Meng X, Zhang K, Li X, Wang C, Xiang Z, Hu K, Han X. Inhibition of Wnt/β-catenin signaling suppresses bleomycin-induced pulmonary fibrosis by attenuating the expression of TGF-β1 and FGF-2. Exp Mol Pathol. 2016;101:22–30. doi: 10.1016/j.yexmp.2016.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Ying H, Kang Y, Zhang H, Zhao D, Xia J, Lu Z, Wang H, Xu F, Shi L. MiR-127 modulates macrophage polarization and promotes lung inflammation and injury by activating the JNK pathway. J Immunol. 2015;194:1239–1251. doi: 10.4049/jimmunol.1402088. [DOI] [PubMed] [Google Scholar]
- 40.Kim J, Chang W, Jung Y, Song K, Lee I. Wnt5a activates THP-1 monocytic cells via a β-catenin-independent pathway involving JNK and NF-κB activation. Cytokine. 2012;60:242–248. doi: 10.1016/j.cyto.2012.06.013. [DOI] [PubMed] [Google Scholar]
- 41.Koval A, Purvanov V, Egger-Adam D, Katanaev VL. Yellow submarine of the Wnt/Frizzled signaling: Submerging from the G protein harbor to the targets. Biochem Pharmacol. 2011;82:1311–1319. doi: 10.1016/j.bcp.2011.06.005. [DOI] [PubMed] [Google Scholar]
- 42.Hsieh JC, Kodjabachian L, Rebbert ML, Rattner A, Smallwood PM, Samos CH, Nusse R, Dawid IB, Nathans J. A new secreted protein that binds to Wnt proteins and inhibits their activities. Nature. 1999;398:431–436. doi: 10.1038/18899. [DOI] [PubMed] [Google Scholar]
- 43.Saito S, Tampe B, Müller GA, Zeisberg M. Primary cilia modulate balance of canonical and non-canonical Wnt signaling responses in the injured kidney. Fibrogenesis Tissue Repair. 2015;8:6. doi: 10.1186/s13069-015-0024-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Nakamura K, Sano S, Fuster JJ, Kikuchi R, Shimizu I, Ohshima K, Katanasaka Y, Ouchi N, Walsh K. Secreted Frizzled-related protein 5 diminishes cardiac inflammation and protects the heart from ischemia/reperfusion injury. J Biol Chem. 2016;291:2566–2575. doi: 10.1074/jbc.M115.693937. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Meyer IS, Jungmann A, Dieterich C, Zhang M, Lasitschka F, Werkmeister S, Haas J, Müller OJ, Boutros M, Nahrendorf M, et al. The cardiac microenvironment uses non-canonical WNT signaling to activate monocytes after myocardial infarction. EMBO Mol Med. 2017;9:1279–1293. doi: 10.15252/emmm.201707565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Guan S, Zhou J. Frizzled-7 mediates TGF-β-induced pulmonary fibrosis by transmitting non-canonical Wnt signaling. Exp Cell Res. 2017;359:226–234. doi: 10.1016/j.yexcr.2017.07.025. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Three data sets (GSE11434, GSE58169 and GSE7742) related to ventilator-induced lung injury in mice were collected from the GEO database (www.ncbi.nlm.nih.gov/geo).