Genetic manipulations in hyperthermophiles have been studied for over 20 years. However, the extremely high temperatures under which these organisms grow have limited the development of genetic tools. In this study, an HHP-inducible promoter was used to control the expression of a toxin. Compared to sugar-inducible and cold-shock-inducible promoters, the HHP-inducible promoter rarely has negative effects on the overall physiology and central metabolism of microorganisms, especially piezophilic hyperthermophiles. Previous studies have used auxotrophic strains as hosts, which may interfere with studies of adaptation and metabolism. Using an inducible toxin-antitoxin (TA) system as a counterselectable marker enables the generation of a markerless gene disruption strain without the use of auxotrophic mutants and counterselection with 5-fluoroorotic acid. TA systems are widely distributed in bacteria and archaea and can be used to overcome the limitations of high growth temperatures and dramatically extend the selectivity of genetic tools in hyperthermophiles.
KEYWORDS: Pyrococcus, archaea, deep sea, high hydrostatic pressure, piezophilic hyperthermophile, toxin-antitoxin
ABSTRACT
The discovery of hyperthermophiles has dramatically changed our understanding of the habitats in which life can thrive. However, the extreme high temperatures in which these organisms live have severely restricted the development of genetic tools. The archaeon Pyrococcus yayanosii A1 is a strictly anaerobic and piezophilic hyperthermophile that is an ideal model for studies of extreme environmental adaptation. In the present study, we identified a high hydrostatic pressure (HHP)-inducible promoter (Phhp) that controls target gene expression under HHP. We developed an HHP-inducible toxin-antitoxin cassette (HHP-TAC) containing (i) a counterselectable marker in which a gene encoding a putative toxin (virulence-associated protein C [PF0776 {VapC}]) controlled by the HHP-inducible promoter was used in conjunction with the gene encoding antitoxin PF0775 (VapB), which was fused to a constitutive promoter (PhmtB), and (ii) a positive marker with the 3-hydroxy-3-methylglutaryl coenzyme A (HMG-CoA) reductase-encoding gene from P. furiosus controlled by the constitutive promoter Pgdh. The HHP-TAC was constructed to realize markerless gene disruption directly in P. yayanosii A1 in rich medium. The pop-out recombination step was performed using an HHP-inducible method. As proof, the PYCH_13690 gene, which encodes a 4-α-glucanotransferase, was successfully deleted from the strain P. yayanosii A1. The results showed that the capacity for starch hydrolysis in the Δ1369 mutant decreased dramatically compared to that in the wild-type strain. The inducible toxin-antitoxin system developed in this study greatly increases the genetic tools available for use in hyperthermophiles.
IMPORTANCE Genetic manipulations in hyperthermophiles have been studied for over 20 years. However, the extremely high temperatures under which these organisms grow have limited the development of genetic tools. In this study, an HHP-inducible promoter was used to control the expression of a toxin. Compared to sugar-inducible and cold-shock-inducible promoters, the HHP-inducible promoter rarely has negative effects on the overall physiology and central metabolism of microorganisms, especially piezophilic hyperthermophiles. Previous studies have used auxotrophic strains as hosts, which may interfere with studies of adaptation and metabolism. Using an inducible toxin-antitoxin (TA) system as a counterselectable marker enables the generation of a markerless gene disruption strain without the use of auxotrophic mutants and counterselection with 5-fluoroorotic acid. TA systems are widely distributed in bacteria and archaea and can be used to overcome the limitations of high growth temperatures and dramatically extend the selectivity of genetic tools in hyperthermophiles.
INTRODUCTION
Hyperthermophiles are a subset of extremophiles with optimum growth temperatures of over 80°C that are capable of surviving in extremely hot environments, thriving at temperatures of up to 122°C (1, 2). Many hyperthermophiles belong to archaea, the third domain of life. This unique group has attracted much attention, since its members occupy the deepest and shortest branches of a 16S rRNA gene sequence-based phylogenetic tree (3). Many studies have focused on elucidating the extreme environmental adaptation strategies of these organisms (4) and investigating the industrial applications of thermostable enzymes (5, 6).
Genetic manipulations of model organisms in the domain Archaea have been performed in methanogens, halophiles, Thermococcales, and Sulfolobales (7, 8). Although the study of genetic manipulations in hyperthermophiles has increased over the last 20 years, the diversity of available genetic tools, especially selectable markers, is still limited, primarily due to the extremely high temperatures in which these organisms live.
Currently available disruption systems for Thermococcus utilize amino acid/nucleotide auxotrophy for selection after single- or double-crossover recombination (9, 10). Thermococcus kodakarensis KOD1 was the first member of the order Thermococcales for which a gene disruption system was developed, using pyrF as a marker and uracil auxotrophic mutants as host strains in a synthetic medium consisting of 20 amino acids (10). Later, several amino acid auxotrophic markers, e.g., the trpE mutation for tryptophan auxotrophy and the hisD mutation for histidine auxotrophy, were developed (11). However, these markers could only be used in the amino acid medium. Subsequently, a simvastatin/3-hydroxy-3-methylglutaryl coenzyme A (HMG-CoA) reductase overexpression system that could be used in rich medium was developed (12). Unfortunately, a markerless gene targeting system that can be directly used for hyperthermophilic archaea without the use of an auxotrophic host strain and marker gene has not yet been reported in the literature. Auxotrophy can negatively affect the study of amino acid or nucleotide metabolism. Currently, many genetic manipulations must be performed in defined medium, which slows strain growth and thus requires a great deal of time. Therefore, there is a great need for novel systems in which gene manipulation can be carried out directly on wild-type strains in rich medium.
Toxin-antitoxin (TA) systems have been successfully used as selection markers to develop genetic tools in bacteria (13–15). Especially in type II TA systems, toxins and antitoxins are proteins that directly interact with each other. Toxins inhibit replication, transcription, or translation, resulting in growth inhibition (16), and are neutralized by the formation of a TA complex with corresponding antitoxins. Thus, toxin proteins are potentially useful as counterselectable markers in hyperthermophiles.
Pyrococcus yayanosii is a strictly anaerobic hyperthermophile that can grow at 80 to 108°C (optimum, 98°C) and at pressures of 0.1 to 120 MPa (optimum, 52 MPa) (17, 18). The complete genome sequence of P. yayanosii has been determined (19), and a gene replacement technique for P. yayanosii was established by deleting the pyrF gene, yielding an auxotrophic host that can be selected using simvastatin (20).
In the present study, we developed a novel markerless gene disruption method based on a high hydrostatic pressure (HHP)-inducible toxin-antitoxin cassette (HHP-TAC) that can be used to directly manipulate the strain P. yayanosii A1. The toxin gene is under the control of a novel HHP-inducible promoter, while the antitoxin gene is under the control of a constitutive promoter (Pgdh) to neutralize background growth. The TA genes were amplified from Pyrococcus furiosus, a member of the same genus as P. yayanosii A1. The HHP-inducible promoter was identified from the transcriptomes of P. yayanosii A1 grown under different hydrostatic pressures and found to control a dramatically upregulated operon under HHP compared to atmospheric pressure.
To demonstrate the feasibility of our system, the PYCH_13690 gene, which encodes a 4-α-glucanotransferase, was successfully deleted from the strain P. yayanosii A1. Compared to the auxotrophic strategy, TA genetic manipulations can be realized in all hyperthermophiles using an appropriate TA system. Thus, a genetic strategy using a TA system under the control of an inducible promoter has potential for applications in all hyperthermophiles and precludes the need for an auxotrophic marker.
RESULTS
Searching for an HHP-inducible promoter.
Based on the transcriptomes of P. yayanosii strain A1 grown at different pressures, three of the most-transcribed neighboring genes were identified, namely, hhpA, hhpB, and hhpC (Table 1 and Fig. 1A). The quantitative PCR (qPCR) results confirmed that the transcription of these three genes was higher under HHP (52 MPa) than under atmospheric pressure (Fig. 1B). In addition, cotranscription analysis showed that these genes were cotranscribed, indicating that they likely constitute an operon (Fig. S1). Thus, our analysis indicated that this gene cluster is potentially regulated by a strong HHP-inducible promoter. Next, a 190-bp intergenic sequence and a 488-bp upstream sequence (Phhp2 and Phhp1, respectively) containing the putative HHP-inducible promoter were selected to verify the activity of the promoter (Fig. 1A).
TABLE 1.
Strains and plasmids used and constructed in this study
| Strain or plasmid | Origin or genetic features | Source or reference |
|---|---|---|
| P. yayanosii strains | ||
| A1 | Facultative piezophile, derivative strain of P. yayanosii CH1 | 20 |
| A1/pLMOS776TA | A1 with plasmid pLMOS776TA | This study |
| A1/pLMOS812TA | A1 with plasmid pLMOS812TA | This study |
| A1/pLMOS839TA | A1 with plasmid pLMOS839TA | This study |
| A1/pLMOS1206TA | A1 with plasmid pLMOS1206TA | This study |
| Δ1369 mutant | A1 with ΔPYCH_13690 | This study |
| Plasmids | ||
| pLMO12102 | Shuttle plasmid replicated in both P. abyssi GE5 and E. coli | X. Ma, unpublished data |
| pLMOS01 | pLMO12102::Pgdh-HMG-CoA from P. furiosus | This study |
| pLMOS011 | pLMO12102::Phhp1-HMG-CoA from P. furiosus | This study |
| pLMOS012 | pLMO12102::Phhp2-HMG-CoA from P. furiosus | This study |
| pLMOS776TA | pLMO12102::HMG-CoA-Pgdh-PhmbB-PF0775-PF0776-Phhp1 | This study |
| pLMOS812TA | pLMO12102::HMG-CoA-Pgdh-PhmbB-PF0813-PF0812-Phhp1 | This study |
| pLMOS839TA | pLMO12102::HMG-CoA-Pgdh-PhmbB-PF0883-PF0839-Phhp1 | This study |
| pLMOS1206TA | pLMO12102::HMG-CoA-Pgdh-PhmbB-PF1207-PF1206-Phhp1 | This study |
| pLMOS507Tpy | pLMO12102::Pgdh-HMG-CoA-Phhp1-PYCH05070 | This study |
| pLMOS1312Tpy | pLMO12102::Pgdh-HMG-CoA-Phhp1-PYCH13120 | This study |
| pLMOS1772Tpy | pLMO12102::Pgdh-HMG-CoA-Phhp1-PYCH17720 | This study |
| pLMOS944Tpf | pLMO12102::Pgdh-HMG-CoA-Phhp1-PF0944 | This study |
FIG 1.
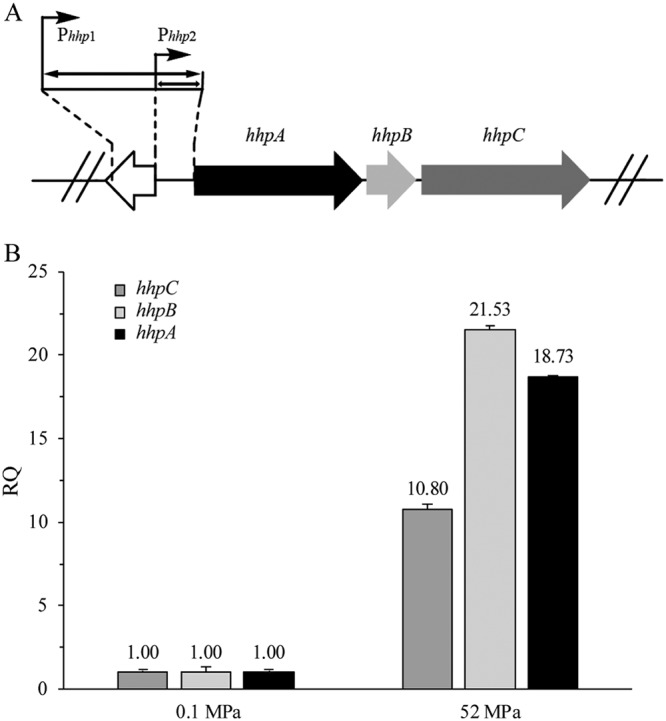
Searching for an HHP-inducible promoter. (A) Details of the high hydrostatic pressure (HHP)-responding operon in A1. The HHP gene cluster, which was identified from transcriptome analysis, consisted of hhpA (black), hhpB (light gray), and hhpC (dark gray). Phhp1 is a putative 488-bp promoter. Phhp2, the intergenic sequence, is a putative 190-bp promoter. (B) qPCR results for the HHP gene cluster at different pressures. The results are from three independent experiments, and the error bars represent the standard deviations. RQ, relative quantity.
Verifying the function of the HHP-inducible promoter.
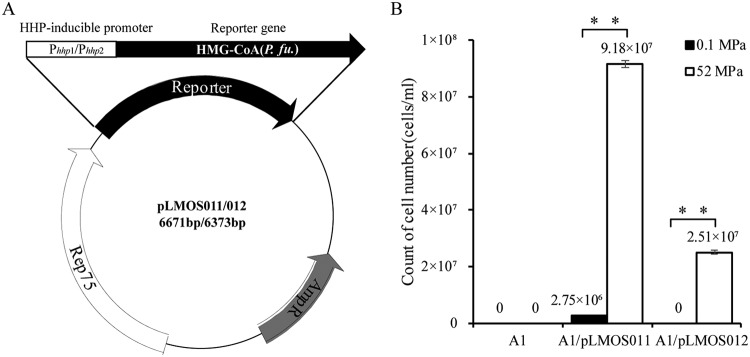
To verify the function of the promoter, recombinant plasmids were constructed using a shuttle vector, pLMO12102, that can replicate in both Pyrococcus abyssi GE5 and Escherichia coli (Xiaopan Ma, unpublished data). The recombinant plasmids pLMOS011 and pLMOS012, which were derived from the shuttle vector pLMO12102, contained the putative Phhp1 and Phhp2 promoter regions, respectively, fused to a heterologous 3-hydroxy-3-methylglutaryl coenzyme A (HMG-CoA) reductase gene from P. furiosus (Fig. 2A). The recombinant plasmids were introduced into P. yayanosii A1, and the transformants were cultured under different pressures with simvastatin. The results showed that the transformants grew well at 52 MPa, and the activity of promoter Phhp1 was stronger than that of Phhp2 (Fig. 2B), which indicated that the HHP-inducible promoter can be used to control gene transcription under different pressures.
FIG 2.
(A) Map of the recombinant plasmids. Phhp1 and Phhp2 are the HHP-inducible promoters. The HMG-CoA reductase gene from P. furiosus was used as a reporter gene. (B) Biomasses of the recombinant strains at different pressures. A1/pLMOS011 and A1/pLMOS012 indicate the A1 strain containing the plasmids pLMOS011 and pLMOS012, respectively. The strains were cultured in TRM supplemented with 10 μM simvastatin at different pressures. The results are from three independent experiments, and the error bars represent the standard deviations. **, P < 0.01.
Construction of an HHP-inducible toxin-antitoxin cassette (HHP-TAC).
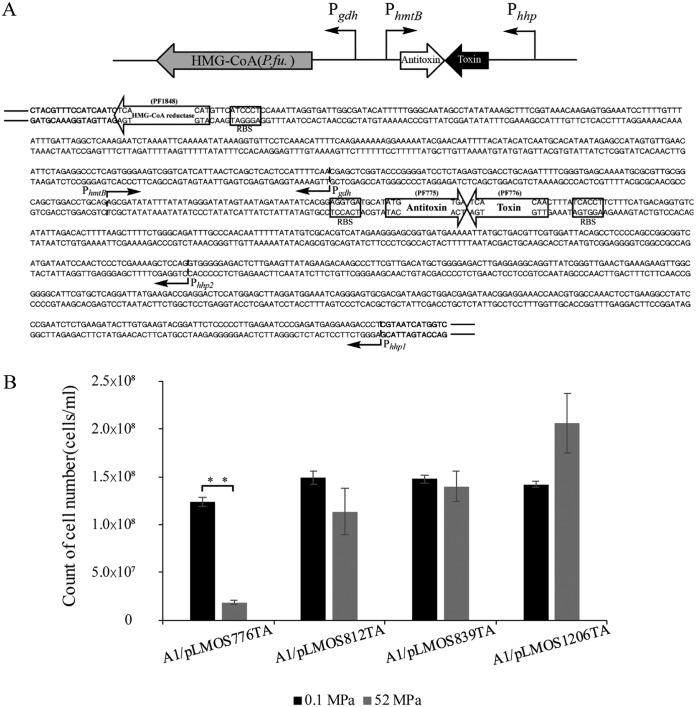
Type II TA systems in P. yayanosii (see Table S1 in the supplemental material) and P. furiosus (Table S2) were identified using TAfinder (21). The results showed that the expression of either of the identified toxin genes from P. yayanosii or highly similar exogenous toxin genes from P. furiosus was neutralized by the endogenous antitoxins in P. yayanosii (Fig. S2). Next, we selected four exogenous TA systems from P. furiosus (PF0776, GenBank accession no. AAL80900.1; PF0775, GenBank accession no. AAL80899.1; PF0812, GenBank accession no. AAL80936.1; PF0813, GenBank accession no. AAL80937.1; PF0839, GenBank accession no. AAL80963; PF0838, GenBank accession no. AAL80962.1; PF1206, GenBank accession no. AAL81330.1; PF1207, GenBank accession no. AAL81331.1) with low similarities (toxin identities, <36%) to their P. yayanosii counterparts. The toxins were controlled by the inducible promoter Phhp1, while the corresponding antitoxins were under the control of the constitutive promoter PhmtB, which was obtained from pTS535 (22), with the two elements oriented in opposite directions to avoid transcriptional interference. The fused fragments were cloned into the shuttle plasmid pLMO12102 with the HMG-CoA reductase gene under the control of a constitutive promoter (Pgdh), and the resulting plasmids were named pLMOS776TA, pLMOS812TA, pLMOS839TA, and pLMOS1206TA. The recombinant fragments were used to construct an HHP-TAC (Fig. 3A). The recombinant plasmids were introduced into strain A1, and the biomass of the recombinant strains was measured under different pressures. Under HHP, the recombinant strains A1/pLMOS812TA, A1/pLMOS839TA, and A1/pLMOS1206TA grew normally, whereas strain A1/pLMOS776TA grew poorly. These results showed that the growth of A1/pLMOS776TA was significantly inhibited by toxin PF0776 (Fig. 3B), indicating that the toxin-antitoxin cassette controlled by an HHP-inducible promoter could be used as a counterselection marker in the genetic manipulation of piezophiles.
FIG 3.
Construction and selection of an HHP-inducible toxin-antitoxin cassette. (A) Genetic organization of the HHP-inducible toxin-antitoxin cassette (HHP-TAC). Phhp1 and Phhp2 are the HHP-inducible promoters. PhmtB and Pgdh are constitutive promoters. The HMG-CoA reductase gene (PF1848 gene) from P. furiosus (P.fu.) was used as a positive selection marker. Antitoxin (PF775) and toxin (PF776) from P. furiosus were used as a counterselectable marker fused with the promoters. The sequence of the shuttle plasmid pLMO12102 is in boldface. RBS, ribosomal binding site. (B) Biomasses of the recombinant strains with different type II TA systems at different pressures. A1/pLMOS776TA, A1/pLMOS812TA, A1/pLMOS839TA, and A1/pLMOS1206TA indicate the A1 strain with the plasmids pLMOS776TA, pLMOS812TA, pLMOS839TA, and pLMOS1206TA, respectively. Black indicates the biomasses of the strains at atmospheric pressure. Dark gray indicates the biomasses of the strains at 52 MPa. The results are from three independent experiments, and the error bars represent the standard deviations. **, P < 0.01.
Markerless gene disruption using TA as a selection marker.
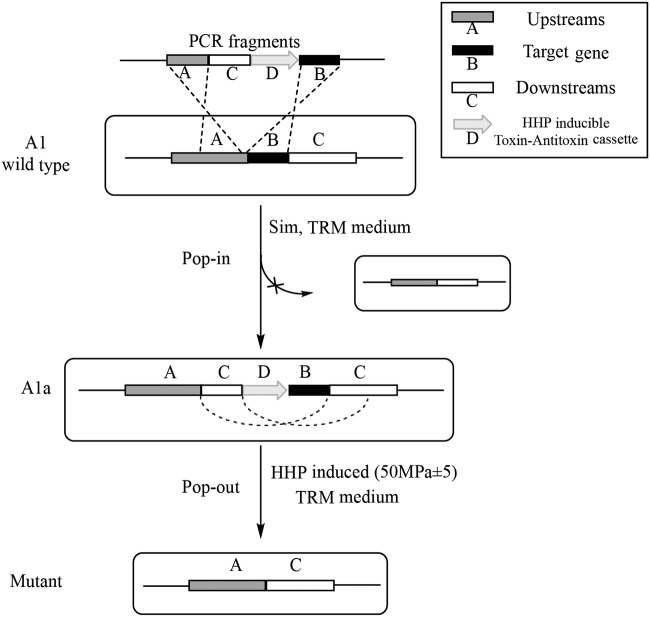
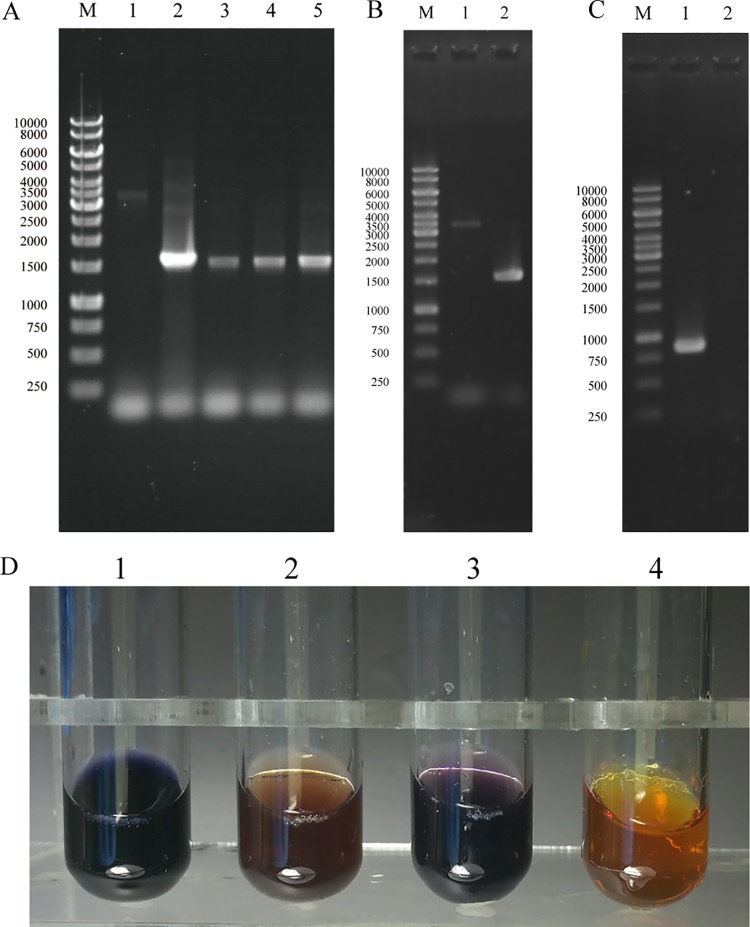
We performed markerless gene disruption of the PYCH_13690 gene, which encodes a 4-α-glucanotransferase, using the HHP-TAC. The constitutively overexpressed gene HMG-CoA was used as a selectable marker, and the HHP-inducible TA system was used as a counterselectable marker. To avoid reverse mutation, linearized fragments were PCR amplified from the suicide plasmid pUS776TA1369 (see Materials and Methods) using the primers M13-47 and M13-48. The linearized fragments were then inserted into the strain P. yayanosii A1 using the CaCl2 procedure described in Materials and Methods (Fig. 4). After the first recombination, single colonies were picked and cultured in liquid Thermococcales rich medium (TRM). Diagnostic PCR was used to identify the targeted fragment insertions in the genome (Fig. 5A). The confirmed intermediate strains were then cultured under HHP (approximately 50 MPa) to promote a second recombination (Fig. 4). The Δ1369 mutant was isolated and verified by PCR. As shown in Fig. 5B, two bands were visible: a 3,501-bp band corresponding to the wild-type allele and a 1,554-bp band corresponding to the mutant allele. Primers for the internal fragments were designed, and the results showed that no amplification of the target gene occurred in the deletion mutant (Fig. 5C). These results confirmed that the PYCH_13690 gene was successfully deleted by markerless gene disruption.
FIG 4.
Schematic diagram of targeted gene disruption in P. yayanosii A1. PCR fragments were amplified from the gene disruption plasmid pUS776TA1369 using the primers M13-47 and M13-48 for gene disruption. The pop-in recombination step was performed using a positive selectable marker in TRM supplemented with simvastatin. The pop-out recombination step was performed using a counterselectable marker in TRM under HHP.
FIG 5.
Analysis of the gene disruption strain (Δ1369). (A) PCR analysis of fragments from the upstream and downstream regions of the PYCH_13690 gene. Lane M, 1-kb marker; lane 1, A1; lanes 2 to 5, the intermediate strain. (B) PCR analysis of fragments from the upstream and downstream regions of the PYCH_13690 gene. Lane M, 1-kb marker; lane 1, A1; lane 2, Δ1369 mutant. (C) PCR analysis of internal fragments of the PYCH_13690 gene. Lane M, 1-kb marker; lane 1, A1; lane 2, Δ1369 mutant. (D) Measurement of residual starch via Lugol’s iodine method after 36 h at 95°C. Lane 1, TRM with 2‰ (wt/vol) soluble starch; lane 2, strain A1 cultured in TRM with 2‰ (wt/vol) soluble starch; lane 3, Δ1369 mutant cultured in TRM with 2‰ (wt/vol) soluble starch; lane 4, TRM.
Efficiency of gene disruption using the HHP-TAC.
The selection efficiency of gene disruption using the HHP-TAC was calculated in two independent experiments. When the second recombination was selected under 52 MPa, 32 of 33 positive colonies were proven to harbor the expected genotype, judging from diagnostic PCR. However, when the second recombination was selected under 45 MPa, only half (5 out of 10) of the positive colonies were correct (Fig. S3). This result showed that the counterselection efficiency of HHP-TAC was significantly higher under 52 MPa than under 45 MPa.
Phenotypic analyses of the gene disruption strains.
The PYCH_13690 gene, which encodes a 4-α-glucanotransferase (NCBI accession no. WP_013906095.1), belongs to glycoside hydrolase family 57 (GH57) and catalyzes the formation of cycloamylose from amylose, a key step in starch metabolism. Previous studies confirmed that an enzyme from Thermococcus litoralis, which shares 76% amino acid sequence similarity to PYCH_13690, catalyzes the transglycosylation of maltooligosaccharides (23). To measure residual starch levels after culturing, mutant strains were cultured in medium supplemented with soluble starch, and Lugol’s iodine was then added. In contrast with the brown color produced in the medium of the wild-type A1 culture, a blue-violet color was produced in the medium of the cultured Δ1369 mutant (Fig. 5D). These results indicate that the ability of the Δ1369 mutant to hydrolyze starch was greatly reduced compared to that of the wild-type strain.
Universality of the HHP-inducible promoter Phhp in other Thermococcales.
To confirm that the promoter Phhp works in other Thermococcales species, it was fused with HMG-CoA from Thermococcus eurythermalis, inserted into the shuttle plasmid pS341 (Xiaopan Ma, unpublished), and then introduced into T. eurythermalis. We observed that the recombinant strain could survive at 20 MPa when supplemented with 4 μM simvastatin, while no biomass was detected at atmospheric pressure (data not shown). Thus, these results showed that the HHP-inducible promoter Phhp can also be used in T. eurythermalis. However, Phhp was not regulated by changing pressure in T. kodakarensis.
DISCUSSION
In this study, we performed markerless gene disruption of the PYCH_13690 locus directly in P. yayanosii A1 by utilizing an HHP-TAC that uses an HHP-inducible TA system as a counterselectable marker. Although several studies using TA systems have been published (24–26), this report describes the development of a novel markerless gene disruption system in a hyperthermophilic archaeon in rich medium.
The auxotrophic strain P. yayanosii A1 containing a pyrF deletion, named A2, was previously constructed and used by our laboratory (20). However, uracil-based negative selection or counterselection suffers from a lack of efficiency in P. yayanosii, which has also been observed in other piezophilic hyperthermophiles (27). Although we optimized the construction of a new uracil auxotrophic strain (As1), the biomass of the auxotrophic strain reached no more than 80% that of strain A1, even in cultures supplemented with 10 μg/ml uracil (Fig. S4). Thus, a markerless gene disruption strategy for strains without an auxotrophic marker is needed.
For selectable markers, overexpression of the HMG-CoA reductase gene under the control of a strong constitutive promoter is an efficient choice to confer simvastatin resistance. Because of the risk of homologous recombination using the HMG-CoA reductase-encoding gene from P. yayanosii (20), the heterologous HMG-CoA reductase-encoding genes from P. furiosus and P. abyssi, placed under the control of the constitutive promoter Pgdh, were selected as positive markers. The results showed that the biomass of the recombinant strain with the HMG-CoA reductase from P. furiosus cultured in the presence of 10 μM simvastatin reached 5 × 107 cells/ml, which is almost equal to that of cells overexpressing homologous HMG-CoA reductase. However, the biomass of the recombinant strain with the HMG-CoA reductase from P. abyssi reduced to half when selected with 10 μM simvastatin.
Inducible systems allow for dose-dependent responses and are ideal for studying controlled expression. In Thermococcales, only two types of inducible promoters are known: a cold shock-inducible promoter (28, 29) and a sugar-inducible promoter (30). The cold shock-inducible system has potential industrial applications, but this induction strategy dramatically affects the physiology of organisms due to the requirement for a nearly 30°C drop in temperature. The sugar-inducible system provides a variety of choices for the use of multiple sugars but may interfere with the central metabolism of the organism of interest, which must be taken into consideration. Recently, an archaeal fluoride-responsive riboswitch composed of the Tk-FRR cassette was also constructed as an inducible expression system for hyperthermophiles (31). In our study, we identified an HHP-inducible promoter (Phhp) in P. yayanosii. The results confirmed that the genes were overexpressed under the control of Phhp under HHP. Furthermore, the strength of the HHP-inducible promoter Phhp1 was stronger than that of Phhp2, making HHP-TAC an ideal potential inducible system for metabolic studies.
The construction of markerless gene disruption systems in Thermococcales still relies on auxotrophic host strains (10, 27). Auxotrophy complicates the study of strain adaptation and metabolism, especially for studies of whole metabolic networks. In addition, the uracil marker results in a high background due to uracil contamination or thermal degradation of the utilized drugs (10, 27). In our study, the piezophilic strain P. yayanosii A1 served as an ideal model for studying high-pressure adaptation. High-pressure adaptation is believed to be achieved by a common adaptation strategy used with multiple other stresses (32). The use of auxotrophic strains as hosts may interfere with studies of the relationship between target genes and high-pressure adaptation. In addition, for Thermococcales, high temperature limits the usability of selectable markers. Thus, in Thermococcales, there is an urgent need to develop novel counterselectable markers and to construct a markerless gene disruption system without the need to develop auxotrophic host strains.
TA systems are widely distributed in bacteria and archaea and affect cell growth. Type II TA systems consist of toxin and antitoxin proteins that directly interact with each other (16). Type II TA systems are potential selectable markers that can greatly enrich the available choices of markers, especially in Thermococcales. Recently, type II TA systems have been used as counterselectable markers in bacteria (14, 15).
In the present study, we used Phhp1 to construct an HHP-TAC. In addition, to reduce the effects of background expression on recombination efficiency (15, 33), an antitoxin protein controlled by the constitutive promoter PhmtB was constructed to neutralize toxins (Fig. 3A). To identify workable TA systems, several candidate type II TA systems from P. yayanosii and P. furiosus were examined, as described in Results. Finally, the gene encoding toxin PF0776, a hypothetical protein with a virulence-associated protein C (VapC)-like PIN domain, was selected as a counterselectable marker associated with the gene encoding antitoxin PF0775 (VapB). The VapBC protein family induces the dormancy and resuscitation of strains by regulating translation (34).
The HHP-TAC developed in this study permits markerless gene disruption directly in P. yayanosii A1 in rich medium. It is a powerful genetic tool to study the function of nonessential genes. In particular, the HHP-inducible promoter Phhp can also be used to induce the overexpression of proteins derived from piezophilic microorganisms.
MATERIALS AND METHODS
Strains, media, and growth conditions.
Escherichia coli strains were cultured in Luria-Bertani (LB) medium at 37°C. P. yayanosii A1, a derivative strain of P. yayanosii CH1, was cultured under anaerobic conditions in TRM at 95°C (17–20). When necessary, TRM was supplemented with simvastatin at a final concentration of 10 μM for positive selection, and growth in TRM under HHP (approximately 50 MPa) was used for negative selection. The liquid medium was supplemented with sulfur and Na2S⋅9H2O. The solid medium in a roll tube was supplemented with a polysulfide solution (12) as an electron acceptor and maintained under anaerobic conditions.
General DNA manipulations.
Plasmids were constructed and replicated in E. coli strain DH5α. Plasmids were extracted using the plasmid DNA minikit I (Omega). DNA fragments were PCR amplified using Pfu DNA polymerase (Tiangen, Beijing, People’s Republic of China) for amplicons less than 1,000 bp and with PrimeSTAR Max DNA polymerase (TaKaRa, Dalian, People’s Republic of China) for amplicons longer than 1,000 bp. The fragments were purified from agarose gels using a gel extraction kit (Omega). Linearized DNA fragments were fused via fusion PCR and ClonExpress II one-step cloning kits (Vazyme, Nanjing, People’s Republic of China).
Construction of shuttle and disruption vectors.
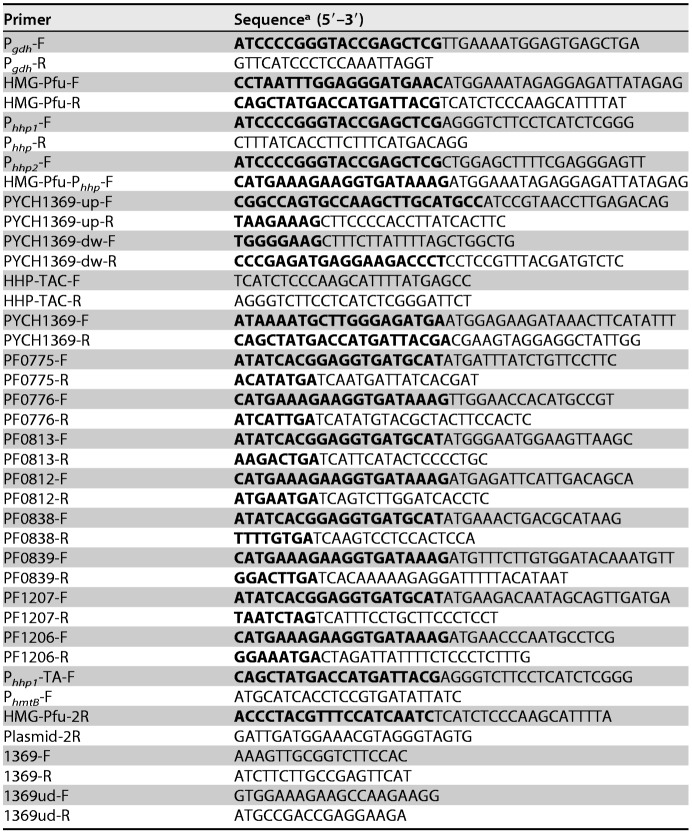
The primers used in this study are listed in Table 2. The shuttle vectors used in this study were derived from the shuttle plasmid pLMO12102. To avoid homologous recombination in the host strain, an HMG-CoA reductase gene from P. furiosus was amplified using the primers HMG-Pfu-F/R and fused with the constitutive promoter Pgdh from P. furiosus via fusion PCR to generate a positive selection marker. The promoter Pgdh was amplified using the primers Pgdh-F/R. The positive selection marker was inserted into the EcoRI site of pLMO12102 using a ClonExpress II one-step cloning kit (Vazyme) and was subsequently named pLMOS01. The putative HHP-inducible promoters Phhp1 and Phhp2 were fused with the HMG-CoA reductase-encoding gene via fusion PCR. The fragments were amplified using the primers Phhp1-F/Phhp-R, Phhp2-F/Phhp-R, and HMG-Pfu-Phhp-F/HMG-Pfu-R, respectively. Next, the fused fragments were inserted into the plasmid pLMO12102, resulting in pLMOS011/pLMOS012. To construct the recombinant shuttle plasmid, pLMO12102 was fused with promoter Phhp1, a toxin gene, an antitoxin gene, the promoters PhmtB and Pgdh, and the HMG-CoA reductase gene, in a stepwise manner, using ClonExpress MultiS one-step cloning kits (Vazyme). Four shuttle plasmids (pLMOS776TA, pLMOS812TA, pLMOS839TA, and pLMOS1206TA) were each obtained by incorporation of a different toxin gene and antitoxin gene pair. The fragments were amplified using the primers Phhp1-TA-F/Phhp-R; PF0776-F/R, PF0812-F/R, PF0839-F/R, or PF1206-F/R (for one of the four different toxins); PF0775-F/R, PF0813-F/R, PF0838-F/R, or PF1207-F/R (for one of the four different antitoxins); PhmtB-F/Pgdh-R; and HMG-Pfu-F/HMG-Pfu-2R, respectively. The plasmid pLMO12102 was linearized by PCR and amplified using the primers plasmid-F/plasmid-2R.
TABLE 2.
Primers used in this study
aOverlap regions are indicated in boldface.
To construct the suicide plasmid pUS776TA1369, the plasmid pUC18 was fused with the upstream fragment 1369-up, the downstream fragment 1369-down, the HHP-TAC, and the target gene PYCH_13690. The upstream fragment 1369-up (890 bp), the downstream fragment 1369-down (656 bp), and the target gene fragment (820 bp) were all amplified using template DNA from strain A1 using the primers PYCH1369-up-F/R, PYCH1369-dw-F/R, and PYCH1369-F/R, respectively. The plasmid pUC18 was linearized by PCR and amplified using the primers plasmid-F (5′-TCGTAATCATGGTCATAGCTGTTTCCTGTG-3′) and plasmid-R (5′-GGCATGCAAGCTTGGCACTGGCCG-3′). The HHP-TAC was PCR amplified using pLMOS776TA as the template DNA with the primers HHP-TAC-F/R.
Transformation of P. yayanosii.
Linearized fragments were PCR amplified from the suicide plasmid pUS776TA1369 using the primers M13-47 (5′-AGGGTTTTCCCAGTCACG-3′) and M13-48 (5′-GAGCGGATAACAATTTCACAC-3′). The linearized fragments were then introduced into strain A1 using the CaCl2 method, as described previously (20). After transformation, the strains were spread on solid TRM supplemented with a polysulfide solution in the presence of 10 μM simvastatin and cultured at 95°C for approximately 1.5 days until colonies were observed. Several single colonies were picked and cultured in 5 ml of liquid TRM with 10 μM simvastatin. PCR amplification was performed using primers PYCH1369-up-F and PYCH1369-dw-R to determine whether the first recombination was successful. For the HHP-induced markerless gene disruption method, the first recombination was performed as described above. Next, the recombinant strain that was confirmed by PCR amplification was inoculated into liquid TRM at 1%. The culture was transferred into an injector with a rubber plug for anaerobic growth. The strain was cultured in a high-pressure/high-temperature incubation system that was maintained at 52 MPa and 95°C. After 24 h, the culture was diluted to approximately 104 to 105 cells/ml, 1 ml of which was subsequently transferred to a roll tube with solid TRM supplemented with a polysulfide solution and rotated to achieve uniform distribution in the tube. The superfluous liquid was then discarded to ensure single colony isolation. The tubes were incubated at 95°C and atmospheric pressure. After 1.5 days, single colonies were selected and transferred to 5 ml of liquid TRM for further culturing. As an option, single colonies were inoculated in liquid TRM with 10 μM simvastatin to exclude the first recombination strain. Total DNA was extracted from each single colony using the phenol-chloroform method and detected by PCR amplification using primers PYCH1369-up-F/PYCH1369-dw-R, PYCH1369-F/R, 1369-F/R, and 1369ud-F/R. The PCR fragments amplified with the primer sets 1369-F/R and 1369ud-F/R annealed outside homologous regions to exclude amplification from plasmid contamination.
Measurement of amylase activity.
The strains were cultured in liquid TRM supplemented with 2‰ (wt/vol) soluble starch at 95°C for 36 h. Next, 10 μl of Lugol’s iodine was added to 1 ml of cell suspension culture to measure starch hydrolysis.
Supplementary Material
ACKNOWLEDGMENTS
This study was supported by the National Key R&D Program of China (2018YFC0310704), the National Natural Science Foundation of China (41676121 and 41530967), and the State Key Laboratory of Ocean Engineering (GKZD010071).
Q.S., Z.L., and J.X. designed the experiments. Q.S., Z.L., R.C., and X.M. performed the experiments. Q.S., X.X., and J.X. analyzed the data. Q.S. and J.X. drafted the manuscript. All authors discussed and reviewed the manuscript.
Footnotes
Supplemental material for this article may be found at https://doi.org/10.1128/AEM.02662-18.
REFERENCES
- 1.Stetter KO. 2013. A brief history of the discovery of hyperthermophilic life. Biochem Soc Trans 41:416–420. doi: 10.1042/BST20120284. [DOI] [PubMed] [Google Scholar]
- 2.Takai K, Nakamura K, Toki T, Tsunogai U, Miyazaki M, Miyazaki J, Hirayama H, Nakagawa S, Nunoura T, Horikoshi K. 2008. Cell proliferation at 122°C and isotopically heavy CH4 production by a hyperthermophilic methanogen under high-pressure cultivation. Proc Natl Acad Sci U S A 105:10949–10954. doi: 10.1073/pnas.0712334105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Stetter KO. 2006. Hyperthermophiles in the history of life. Philos Trans R Soc Lond B Biol Sci 361:1837–1843. doi: 10.1098/rstb.2006.1907. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Stetter KO. 1999. Extremophiles and their adaptation to hot environments. FEBS Lett 452:22–25. doi: 10.1016/S0014-5793(99)00663-8. [DOI] [PubMed] [Google Scholar]
- 5.Adams MWW, Kelly RM. 1998. Finding and using hyperthermophilic enzymes. Trends Biotechnol 16:329–332. doi: 10.1016/S0167-7799(98)01193-7. [DOI] [PubMed] [Google Scholar]
- 6.Vieille C, Zeikus GJ. 2001. Hyperthermophilic enzymes: sources, uses, and molecular mechanisms for thermostability. Microbiol Mol Biol Rev 65:1–43. doi: 10.1128/MMBR.65.1.1-43.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Farkas JA, Picking JW, Santangelo TJ. 2013. Genetic techniques for the archaea. Annu Rev Genet 47:539–561. doi: 10.1146/annurev-genet-111212-133225. [DOI] [PubMed] [Google Scholar]
- 8.Leigh JA, Albers SV, Atomi H, Allers T. 2011. Model organisms for genetics in the domain Archaea: methanogens, halophiles, Thermococcales and Sulfolobales. FEMS Microbiol Rev 35:577–608. doi: 10.1111/j.1574-6976.2011.00265.x. [DOI] [PubMed] [Google Scholar]
- 9.Hileman TH, Santangelo TJ. 2012. Genetics techniques for Thermococcus kodakarensis. Front Microbiol 3:195. doi: 10.3389/fmicb.2012.00195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Sato T, Fukui T, Atomi H, Imanaka T. 2003. Targeted gene disruption by homologous recombination in the hyperthermophilic archaeon Thermococcus kodakaraensis KOD1. J Bacteriol 185:210–220. doi: 10.1128/JB.185.1.210-220.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Sato T, Fukui T, Atomi H, Imanaka T. 2005. Improved and versatile transformation system allowing multiple genetic manipulations of the hyperthermophilic archaeon Thermococcus kodakaraensis. Appl Environ Microbiol 71:3889–3899. doi: 10.1128/AEM.71.7.3889-3899.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Matsumi R, Manabe K, Fukui T, Atomi H, Imanaka T. 2007. Disruption of a sugar transporter gene cluster in a hyperthermophilic archaeon using a host-marker system based on antibiotic resistance. J Bacteriol 189:2683–2691. doi: 10.1128/JB.01692-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kimelman A, Levy A, Sberro H, Kidron S, Leavitt A, Amitai G, Yoder-Himes DR, Wurtzel O, Zhu Y, Rubin EM, Sorek R. 2012. A vast collection of microbial genes that are toxic to bacteria. Genome Res 22:802–809. doi: 10.1101/gr.133850.111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Wang H, Bian X, Xia L, Ding X, Muller R, Zhang Y, Fu J, Stewart AF. 2014. Improved seamless mutagenesis by recombineering using ccdB for counterselection. Nucleic Acids Res 42:e37. doi: 10.1093/nar/gkt1339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Wu J, Deng A, Sun Q, Bai H, Sun Z, Shang X, Zhang Y, Liu Q, Liang Y, Liu S, Che Y, Wen T. 2018. Bacterial genome editing via a designed toxin-antitoxin cassette. ACS Synth Biol 7:822–831. doi: 10.1021/acssynbio.6b00287. [DOI] [PubMed] [Google Scholar]
- 16.Yamaguchi Y, Park JH, Inouye M. 2011. Toxin-antitoxin systems in bacteria and archaea. Annu Rev Genet 45:61–79. doi: 10.1146/annurev-genet-110410-132412. [DOI] [PubMed] [Google Scholar]
- 17.Zeng X, Birrien JL, Fouquet Y, Cherkashov G, Jebbar M, Querellou J, Oger P, Cambon-Bonavita MA, Xiao X, Prieur D. 2009. Pyrococcus CH1, an obligate piezophilic hyperthermophile: extending the upper pressure-temperature limits for life. ISME J 3:873–876. doi: 10.1038/ismej.2009.21. [DOI] [PubMed] [Google Scholar]
- 18.Birrien JL, Zeng X, Jebbar M, Cambon-Bonavita MA, Querellou J, Oger P, Bienvenu N, Xiao X, Prieur D. 2011. Pyrococcus yayanosii sp. nov., an obligate piezophilic hyperthermophilic archaeon isolated from a deep-sea hydrothermal vent. Int J Syst Evol Microbiol 61:2827–2831. doi: 10.1099/ijs.0.024653-0. [DOI] [PubMed] [Google Scholar]
- 19.Jun X, Lupeng L, Minjuan X, Oger P, Fengping W, Jebbar M, Xiang X. 2011. Complete genome sequence of the obligate piezophilic hyperthermophilic archaeon Pyrococcus yayanosii CH1. J Bacteriol 193:4297–4298. doi: 10.1128/JB.05345-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Li X, Fu L, Li Z, Ma X, Xiao X, Xu J. 2015. Genetic tools for the piezophilic hyperthermophilic archaeon Pyrococcus yayanosii. Extremophiles 19:59–67. doi: 10.1007/s00792-014-0705-2. [DOI] [PubMed] [Google Scholar]
- 21.Shao Y, Harrison EM, Bi D, Tai C, He X, Ou HY, Rajakumar K, Deng Z. 2011. TADB: a web-based resource for type 2 toxin-antitoxin loci in bacteria and archaea. Nucleic Acids Res 39:D606. doi: 10.1093/nar/gkq908. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Santangelo TJ, Cubonova L, Reeve JN. 2010. Thermococcus kodakarensis genetics: TK1827-encoded beta-glycosidase, new positive-selection protocol, and targeted and repetitive deletion technology. Appl Environ Microbiol 76:1044–1052. doi: 10.1128/AEM.02497-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Jeon BS, Taguchi H, Sakai H, Ohshima T, Wakagi T, Matsuzawa H. 1997. 4-α-Glucanotransferase from the hyperthermophilic archaeon Thermococcus litoralis: enzyme purification and characterization, and gene cloning, sequencing, and expression in Escherichia coli. Eur J Biochem 248:171–178. doi: 10.1111/j.1432-1033.1997.00171.x. [DOI] [PubMed] [Google Scholar]
- 24.Takagi H, Kakuta Y, Okada T, Yao M, Tanaka I, Kimura M. 2005. Crystal structure of archaeal toxin-antitoxin RelE-RelB complex with implications for toxin activity and antitoxin effects. Nat Struct Mol Biol 12:327–331. doi: 10.1038/nsmb911. [DOI] [PubMed] [Google Scholar]
- 25.Shinohara M, Guo JX, Mori M, Nakashima T, Takagi H, Nishimoto E, Yamashita S, Tsumoto K, Kakuta Y, Kimura M. 2010. The structural mechanism of the inhibition of archaeal RelE toxin by its cognate RelB antitoxin. Biochem Biophys Res Commun 400:346–351. doi: 10.1016/j.bbrc.2010.08.061. [DOI] [PubMed] [Google Scholar]
- 26.Li Z, Song Q, Wang Y, Xiao X, Xu J. 2018. Identification of a functional toxin-antitoxin system located in the genomic island PYG1 of piezophilic hyperthermophilic archaeon Pyrococcus yayanosii. Extremophiles 22:347–357. doi: 10.1007/s00792-018-1002-2. [DOI] [PubMed] [Google Scholar]
- 27.Thiel A, Michoud G, Moalic Y, Flament D, Jebbar M. 2014. Genetic manipulations of the hyperthermophilic piezophilic archaeon Thermococcus barophilus. Appl Environ Microbiol 80:2299–2306. doi: 10.1128/AEM.00084-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Weinberg MV, Schut GJ, Brehm S, Datta S, Adams MW. 2005. Cold shock of a hyperthermophilic archaeon: Pyrococcus furiosus exhibits multiple responses to a suboptimal growth temperature with a key role for membrane-bound glycoproteins. J Bacteriol 187:336–348. doi: 10.1128/JB.187.1.336-348.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Basen M, Sun J, Adams MWW. 2012. Engineering a hyperthermophilic archaeon for temperature-dependent product formation. mBio 3(2):e00053-12. doi: 10.1128/mBio.00053-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Kanai T, Akerboom J, Takedomi S, van de Werken HJ, Blombach F, van der Oost J, Murakami T, Atomi H, Imanaka T. 2007. A global transcriptional regulator in Thermococcus kodakaraensis controls the expression levels of both glycolytic and gluconeogenic enzyme-encoding genes. J Biol Chem 282:33659–33670. doi: 10.1074/jbc.M703424200. [DOI] [PubMed] [Google Scholar]
- 31.Speed MC, Burkhart BW, Picking JW, Santangelo TJ. 2018. An archaeal fluoride-responsive riboswitch provides an inducible expression system for hyperthermophiles. Appl Environ Microbiol 84:e02306-17. doi: 10.1128/AEM.02306-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Zhang Y, Li X, Bartlett DH, Xiao X. 2015. Current developments in marine microbiology: high-pressure biotechnology and the genetic engineering of piezophiles. Curr Opin Biotechnol 33:157–164. doi: 10.1016/j.copbio.2015.02.013. [DOI] [PubMed] [Google Scholar]
- 33.Zhang XZ, Yan X, Cui ZL, Hong Q, Li SP. 2006. mazF, a novel counter-selectable marker for unmarked chromosomal manipulation in Bacillus subtilis. Nucleic Acids Res 34:e71. doi: 10.1093/nar/gkl358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Gerdes K, Maisonneuve E. 2012. Bacterial persistence and toxin-antitoxin loci. Annu Rev Microbiol 66:103–123. doi: 10.1146/annurev-micro-092611-150159. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.