Abstract
Fas Ligand (FasL) and Fas (APO-1/CD95) are members of the TNFR superfamily and may trigger apoptosis. Here, we aimed to elucidate the functional role of Fas signaling in an experimental model of chronic liver disease, the hepatocyte-specific NEMO knockout (NEMOΔhepa) mice. We generated NEMOΔhepa /Faslpr mice, while NEMOΔhepa, NEMOf/f as well as Faslpranimals were used as controls, and characterized their phenotype during liver disease progression. Liver damage was evaluated by serum transaminases, histological, immunofluorescence procedures, and biochemical and molecular biology techniques. Proteins were detected by western Blot, expression of mRNA by RT-PCR, and infiltration of inflammatory cells was determined by FACs analysis, respectively. Faslpr mutation in NEMOΔhepa mice resulted in overall decreased liver injury, enhanced hepatocyte survival, and reduced proliferation at 8 weeks of age compared with NEMOΔhepa mice. Moreover, NEMOΔhepa/Faslpr animals elicited significantly decreased parameters of liver fibrosis, such as Collagen IA1, MMP2, and TIMP1, and reduced proinflammatory macrophages and cytokine expression. At 52 weeks of age, NEMOΔhepa/Faslpr exhibited less malignant growth as evidenced by reduced HCC burden associated with a significantly decreased number of nodules and LW/BW ratio and decreased myeloid populations. Deletion of TNFR1 further reduced tumor load of 52-weeks-old NEMOΔhepa/Faslpr mice. The functionality of FasL/Fas might affect inflammation-driven tumorigenesis in an experimental model of chronic liver disease. These results help to develop alternative therapeutic approaches and extend the limitations of tumor therapy against HCC.
Introduction
The transmembrane proteins Fas-Ligand (FasL) and Fas (Fas/ CD95/APO-1) are members of the tumor necrosis factor (TNF) and TNF receptor gene superfamilies (TNFR gene superfamily), respectively, and are expressed in numerous cell types. FasL–Fas interaction plays a crucial role in immune regulation via the ability of FasL to transmit an apoptotic signal to Fas-expressing cells1. Particularly, the importance of the FasL/Fas axis in pathophysiology and homeostasis has been well-documented in the liver where these proteins are expressed in hepatocytes, cholangiocytes, activated stellate cells (HSC), and Kupffer cells (KC)2.
Downregulation or loss of Fas expression and function is frequently found in the progression of a number of human malignancies, including colon, breast, lung, and liver carcinoma3,4. Thus, the FasL–Fas pathway plays a crucial role in tumor initiation and progression. It might be a plausible therapeutic target not only for progression of liver disease, but also hepatocellular carcinoma (HCC).
HCC is the fifth most common solid cancer affecting one million people per year representing the third cause of mortality by cancer worldwide5,6. Escape from the immune surveillance may play an important role in liver tumorigenesis. Alteration of the FasL/Fas system is regarded as one of the mechanisms preventing the immune system from rejecting tumor cells7. However, little attention has been paid to the role of the Fas/FasL interaction in vivo.
Hepatocyte-specific NEMO knockout (NEMOΔhepa) mice are susceptible to spontaneous apoptosis, which leads to chronic hepatocyte injury and regenerative proliferation, constituting a risk factor for cancer development8. NEMOΔhepa livers are hypersensitive towards TRAIL stimulation9. Moreover, we have also shown that the death receptor TNFR1, but not TRAIL, is involved in determining progression of liver injury in NEMOΔhepa10.
In the present study, we examined the functional role of deficient FasL/Fas signaling on disease progression and end-stage tumorigenesis in the NEMOΔhepa model.
Materials and methods
Housing and Generation of Knockout mice
Animals were maintained in the animal facility of the University Hospital RWTH Aachen according to the German legal requirements. Hepatocyte-specific IKKγ/NEMO mice were generated by crossing loxP site-flanked (floxed [f]) NEMO gene (NEMOf/f) with Alfp-cre transgenic animals as described before9. These mice were further crossed either with Faslpr knockout mice (purchased from The Jackson Laboratory, Bar Harbor, Maine, USA) to yield NEMOΔhepa/Faslpr. Finally, we crossed NEMOΔhepa/Faslpr with TNFR1−/− mice to further generate NEMOΔhepa/Faslpr/TNFR1−/− and investigated the impact of TNFR1 in NEMOΔhepa/Faslpr mice. To use the proper controls, NEMOΔhepa mice were backcrossed from NEMOΔhepa/Faslpr, and Faslpr and Faslpr/TNFR1−/− were used as controls. Genotypes were confirmed via PCR specific for the respective alleles using DNA from tail biopsies. Progression of liver disease was investigated in male mice between 8–9 weeks and 52–54 weeks of age. Liver injury experiments were performed on mice between 8–9 weeks of age. Serum AST and ALT were measured by standard procedures in the Institute of Clinical Chemistry, University Hospital, RWTH Aachen.
TUNEL assay
TUNEL test was performed by standard procedures.
Quantitative real-time PCR
Total RNA was purified from liver tissue using Trizol reagent (Invitrogen, Karlsruhe, Germany). Total RNA (1 μg) was used to synthesize cDNA using SuperScript first-Stand Synthesis System (Invitrogen) and was resuspended in 100 μl of H2O. Quantitative real-time PCR was performed using SYBR Green Reagent (Invitrogen) in 7300 real-time PCR system (Applied Biosystem, Darmstadt, Germany). GAPDH expression was used to normalize gene expression, which is represented as times versus WT basal expression. Primer sequences can be provided upon request.
Histological, immunofluorescence, and immunohistochemical analysis
Livers from mice were harvested and after fixation with 4% PFA, were embedded in paraffin for further histological evaluation. H&E and Sirius Red staining were performed on liver sections. For immunofluorescence analysis, liver cryosections of 5 μm were stained with Ki-67, CD11b (BD Biosciences, Heidelberg, Germany), and F4/80 (BioRad, Hercules, USA). Slides were fixed in 4% PFA at room temperature. Secondary antibody conjugated with Cy3 (Jackson Immunoresearch, West Grove, PA) was used to obtain red fluorescence signal. Mounting solution containing DAPI (Vector Laboratories, Burlingame, CA) was used to counterstain the nuclei of hepatocytes.
Flow cytometry analysis
Hepatocytes were stained with Annexin V-FITC (BD Biosciences). Immune cells from whole liver were isolated and stained with fluorochrome-conjugated antibodies (CD4-PE, CD8-FITC, CD45-APC-Cy7, CD11b-PE, Ly6G-FITC, and F4/80 Biotin) (BD Biosciences, Heidelberg, Germany). All samples were acquired by flow cytometry (FACS Canto II; BD Biosciences) and analyzed using the Flowjo software.
Immunoblot analysis
Isolated protein samples were probed with antibodies against RIPK1 (#5389) (Pro-Sci, Poway, CA, USA), Cleaved Caspase-3 (Asp175) (Cell Signaling Technology, Massachusetts, USA), PCNA (clone PC10) (Dianova GmbH, Hamburg, Germany), and GAPDH (MCA4739; 1:5000; AbD SeroTec, Düsseldorf, Germany). As secondary antibodies, anti-rabbit-HRP (#7074; Cell Signaling) and anti-mouse-HRP (#sc-2005; Santa Cruz) were used.
Statistical analysis
Data are expressed as the mean ± standard error of the mean (SEM). Statistical significance was determined using one-way analysis of variance (ANOVA) with Bonferroni post-hoc test.
Results
Generation and characterization of the NEMOΔhepa/Faslpr mice
To address the functional relevance of FasL/Fas signaling for chronic disease progression in the NEMOΔhepa model10,11, we used mutant Fas mice, the lymphoproliferative (lpr) mice (Faslpr)12. Upon generation (Supplementary Figure 1a), we observed that these animals develop lymphadenopathy by accumulating abnormal T cells and suffer from systemic lupus erythematosus-like autoimmune disease12. As expected, Faslpr and NEMOΔhepa/Faslpr displayed splenomegalia and presence of lymph nodes in the peritoneum (Supplementary Figure 1b–d).
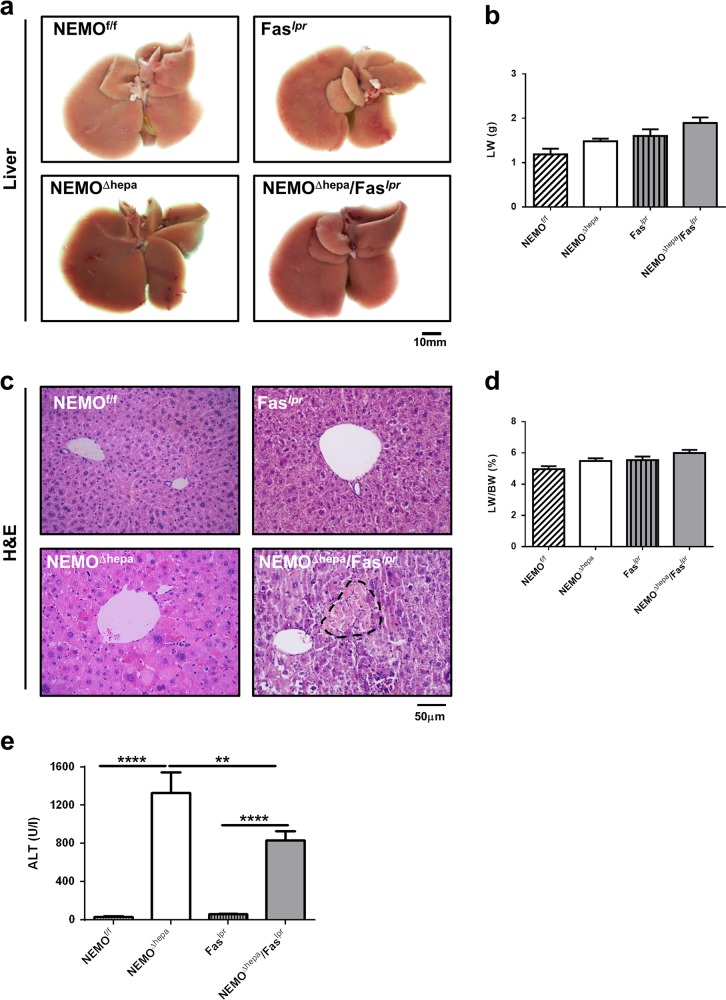
Macroscopic appearance of NEMOΔhepa/Faslpr livers was normal. Eight-week-old NEMOΔhepa livers are histologically characterized by lack of lobular disorganization, hepatocellular hyperplasia, hypertrophy, and severe diffuse hepatocellular anisokaryosis with marked increase in the apoptotic and mitotic rate. In turn, no neoplasia was present in the hepatic parenchyma of 8-week-old NEMOΔhepa/Faslpr livers markedly characterized by multifocal necrosis (Fig. 1a-d). A significant decrease in markers of liver injury at 8 weeks of age was observed in serum ALT (Fig. 1e) compared with NEMOΔhepa mice. No differences were found in AP and GLDH compared with NEMOΔhepa mice (Supplementary Figure 2a, b).
Fig. 1. Generation and characterization of 8-week-old NEMOΔhepa/Faslpr mice.
a Macroscopic appearance of livers 8-week-old NEMOf/f, NEMOΔhepa, Faslpr, and NEMOΔhepa/Faslpr. The liver (LW) (b) was calculated and represented. c Representative H&E staining of liver sections of 8-week-old animals. d The liver versus the body weight (LW/BW) ratio was calculated and represented. Arrows indicate infiltration and dotted area points to necrosis. e Serum ALT levels of 8-week-old NEMOf/f, NEMOΔhepa, Faslpr, and NEMOΔhepa/Faslpr mice were determined. Results are expressed as mean ± SEM (n = 50 mice, p < 0.001–0.01)
Impact of FasL/Fas disruption on cell death and compensatory proliferation in NEMOΔhepa livers
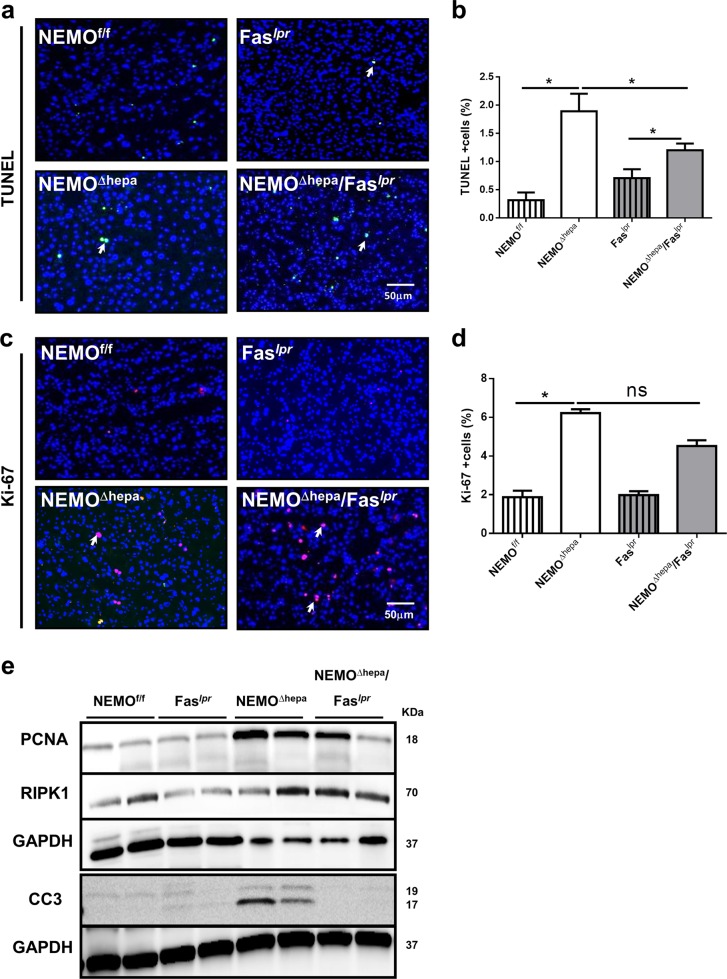
The decrease in transaminase levels observed in mutant NEMOΔhepa/Faslpr prompted us to investigate the impact of Fas deletion on cell death in NEMOΔhepa mice. Significant differences were evident in TUNEL-positive cells between NEMOΔhepa and NEMOΔhepa/Faslpr livers (Fig. 2a, b). Interestingly, the mRNA transcripts of Bcl2, a pro-survival protein, was significantly upregulated in NEMOΔhepa/Faslpr compared with NEMOΔhepa livers (Supplementary Figure 2c). Since enhanced cell death triggers compensatory proliferation in NEMOΔhepa livers8, we further tested whether disruption of FasL/Fas signalling would have an impact on cell proliferation in NEMOΔhepa/Faslpr mice. By using cell cycle markers for total cell cycle activity (Ki-67), we found a clear trend towards reduced cell proliferation and significantly lower cell cycle activity (CcnD1) in NEMOΔhepa/Faslpr livers compared with NEMOΔhepa (Fig. 2c, d; Supplementary Figure 2d). Interestingly, most Ki-67-positive cells were hepatocytes in NEMOΔhepa livers, whereas immune cells were proliferating in the hepatic parenchyma of NEMOΔhepa/Faslpr animals (Fig. 2c, d). Additionally, PCNA protein levels were augmented in NEMOΔhepa compared with NEMOΔhepa/Faslpr livers (Fig. 2e). Moreover, cleavage of Caspase-3—but not protein levels of RIPK1—was absent in NEMOΔhepa/Faslpr livers (Fig. 2e). Altogether, these results suggest that Fas signaling might promote apoptotic cell death in NEMOΔhepa-deficient livers.
Fig. 2. Loss of the FasL/Fas signaling exacerbates cell death and compensatory proliferation in NEMOΔhepa mice.
a Representative TUNEL staining of 8-week-old NEMOf/f, NEMOΔhepa, Faslpr, and NEMOΔhepa/Faslpr livers. b TUNEL-positive cells were quantified and graphed. c Proliferation was determination by Ki-67-positive cells immunofluorescence. d Ki-67-positive cells were quantified and graphed. e Expression of PCNA, cleaved Caspase-3 (CC3), and RIPK1 was analysed by immunoblotting of whole liver extracts. GAPDH served as a loading control. Results are expressed as mean ± SEM (n = 8 livers, p < 0.001–0.05)
Loss of Fas attenuates fibrogenesis in NEMOΔhepa livers
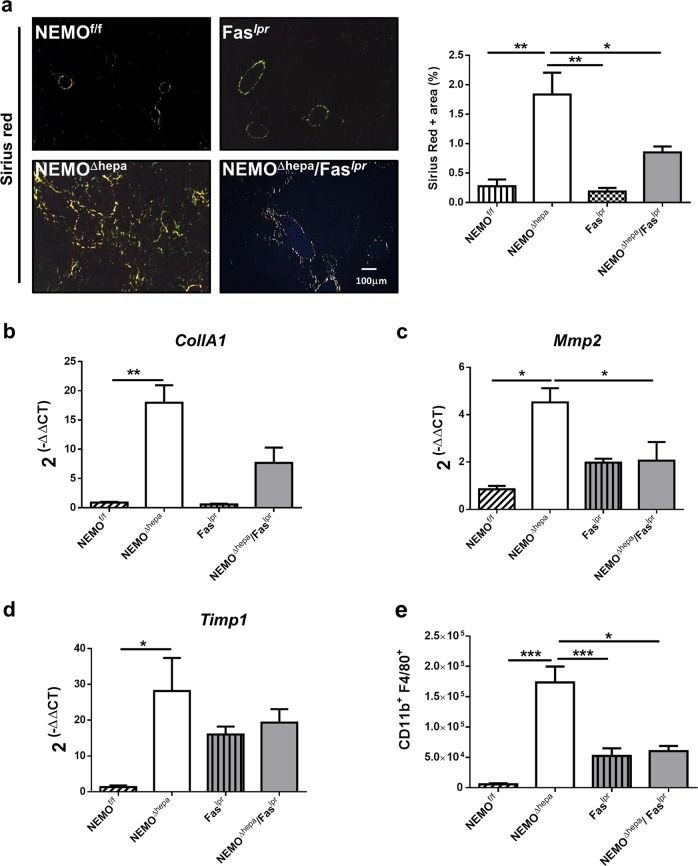
Fas might attenuate the effect of gut-derived products on liver injury. Therefore, we next aimed to investigate if this finding might also affect liver disease progression and evaluated its relevance for liver fibrogenesis in 8-week-old animals. Interestingly, deletion of Fas attenuated liver fibrosis in NEMOΔhepa. We first measured hepatic fibre formation by Sirius red staining and quantification (Fig. 3a, b), and determined additional well-characterized pro-fibrotic markers, such as Collagen IA1, MMP2, and TIMP1 mRNA expression (Fig. 3c, d).
Fig. 3. Loss of Fas attenuates liver fibrogenesis in NEMOΔhepa livers.
a Sirius red staining of paraffin-embedded liver tissue derived from 8-week-old livers of NEMOf/f, NEMOΔhepa, Faslpr, and NEMOΔhepa/Faslpr mice. Representative photomicrographs taken under polarized light are shown. SR-positive area was quantifed using ImageJ© and represented. MRNA relative expression of b Collagen-1A1, c Mmp2, and d Timp1 was quantified by RT-PCR. e Percentage of F4/80hiCD11hi-positive cells of each mouse strain was assessed using FACS analysis. Results are expressed as mean ± SEM (n = 8, p < 0.001–0.01)
The infiltration of distinct immune cell populations directed by chemotactic cytokines is a central pathogenic feature following chronic liver injury13. Thus, we characterized the hepatic inflammatory cell populations using qRT-PCR and FACS analysis. CD11b+ F4/80+-proinflammatory macrophages were significantly decreased in NEMOΔhepa/Faslpr mice compared to NEMOΔhepa livers (Fig. 3e). Altogether, these findings suggest that Fas deletion—very likely by changing the sensitivity versus LPS—has an anti-inflammatory effect on NEMOΔhepa-derived hepatitis.
To test whether decreased liver injury after Faslpr deletion in IKKγ/Nemo mice had an impact on pro-inflammatory cytokines, we performed qRT-PCR for TNF, a main driver of NEMOΔhepa-induced liver injury. Interestingly, TNF mRNA levels were significantly reduced in NEMOΔhepa/Faslpr compared with NEMOΔhepa livers (Supplementary Figure 3a). However, no differences were found in TGFβ levels between mouse strains (Supplementary Figure 3b). Therefore, we next sought to investigate immune cell infiltration in 8-week-old NEMOΔhepa/Faslpr livers. In addition to FACS analysis, we observed significantly decreased CD11b and F4/80-positive cells in NEMOΔhepa/Faslpr compared with NEMOΔhepa hepatic tissue (Supplementary Figure 4a-e). These results demonstrate that attenuated liver inflammation after Fas deletion in NEMOΔhepa livers reduces fibrogenesis, pro-inflammatory macrophages, and expression of pro-inflammatory cytokines e.g., TNF.
Fas signaling is involved in hepatocarcinogenesis (HCC) in NEMOΔhepa mice
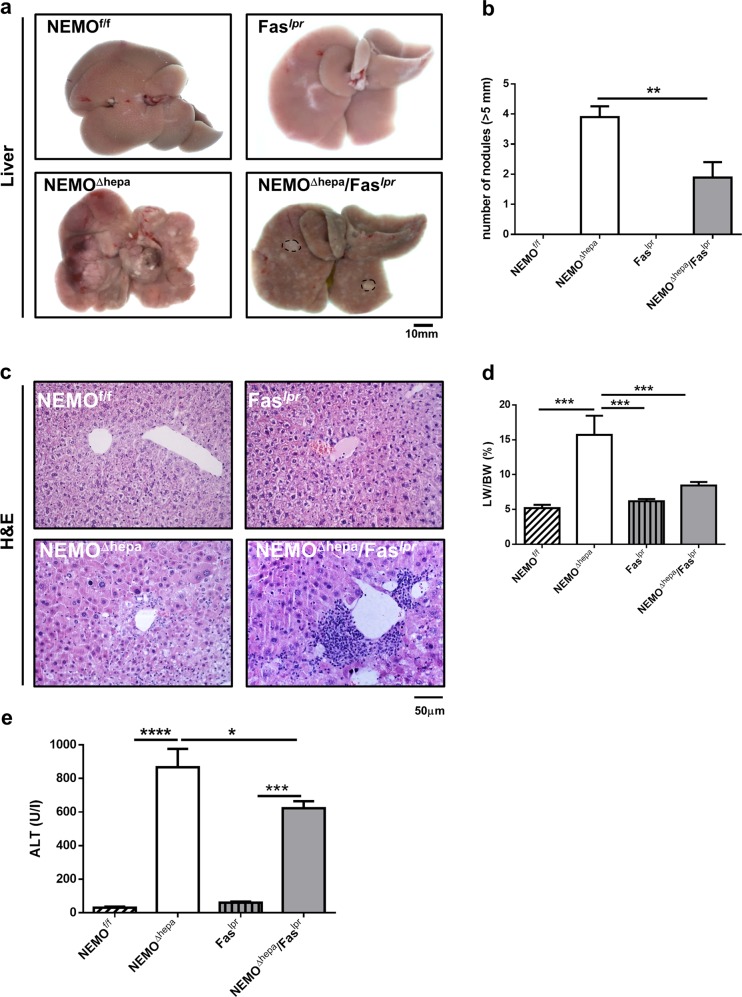
Fifty-two-week-old NEMOΔhepa mice develop not only liver fibrosis, but also HCC8. Next, we tested the relevance of Fas signaling for liver carcinogenesis. Macroscopically, livers from NEMOΔhepa mice revealed both regenerative nodules and well-defined, large vascularised tumors resulting in a significantly higher liver weight (LW) and liver weight/body weight ratio (LW/BW) compared to NEMOΔhepa/Faslpr (Fig. 4).
Fig. 4. Hepatocarcinogenesis is decreased in 1-year-old NEMOΔhepa/Faslpr mice.
NEMOf/f, NEMOΔhepa, Faslpr, and NEMOΔhepa/Faslpr mice at the age of 12 months were sacrificed and livers extracted and analyzed. a Macroscopic appearance of livers. Arrows show visible nodules. b The number of nodules (≥5 mm) diameter was quantified. c Representative H&E staining of liver sections. d Liver weight versus body weight (LW/BW) ratio is shown. e Serum ALT was determined in the same mice. Results are expressed as mean ± SEM (n = 50, *p < 0.05; **p < 0.01)
H&E stainings of 1-year-old NEMOΔhepa mice showed well-differentiated trabecular and solid HCC with nodules of proliferative hepatocytes with disturbed liver architecture and hepatocellular lipid storage, whereas NEMOΔhepa/Faslpr showed no neoplasia but atypia and hepatic hyperplasia accompanied by strong perivascular infiltration. Of note, lymphocyte accumulation was also found in Faslpr livers (Table 1). Concomitantly, NEMOΔhepa/Faslpr exhibited decreased number of nodules and significantly lower LW/BW ratio compared with NEMOΔhepa livers (Fig. 4a–d).
Table 1.
Histopathological characteristics of the different mouse groups
| Nemo | Nemo/Faslpr | Nemo/Faslpr/TNFR1−/− | |
|---|---|---|---|
| Neoplasia | HCCa1 | Noa2 | Noa3 |
| Anisokaryosis | 4.17 ± 0.98 | 4.38 ± 0.51 | 2.88 ± 1.25c, d |
| Altered foci | 2.17 ± 1.17 | 2.00 ± 1.07 | 0.62 + 0.91d |
| Mitosis/HPF (40 × ) | 1.75 ± 2.47 | 1.43 ± 0.79 | 0.83 ± 1.33 |
| Cellular hypertrophy | 2.5 ± 1.22 | 2.25 ± 0.46 | 1.13 ± 0.99d |
| Dysplasia | 2.5 ± 1.27 | 2.30 ± 0.46 | 1.00 ± 0.93d |
| Oval cell proliferation | 2.17 ± 0.98 | 1.5 ± 0.53 | 0.87 ± 0.64c, d |
| Lymphoid aggregates/lymphocytic inflammation | 0.50 ± 0.00 | 1.00 ± 0.00 | 0.86 ± 0.89 |
| Apoptosis | 1.50 ± 0.70 | 1.28 ± 0.48 | 0.67 ± 0.51 |
| Additional lesions | Mild lipidosis diffuse vaculopathy | Multifocal necrosis | Biliary atresia early lymphomas |
a1Well-differentiated trabecular or solid HCCs
a2Early hepatocellular adenomas
a3Single well-differentiated trabecular and glandular HCC
cNemo versus Nemo/FasLPR/TNFR1−/−
dNemo/FasLPR versus Nemo/FasLPR/TNFR1−/−
Additionally, a tendency towards reduced ALT was observed in 52-weeks-old NEMOΔhepa/Faslpr compared with NEMOΔhepa mice (Fig. 4e). Together, these results indicate that loss of Fas protects against tumorigenesis in the NEMOΔhepa experimental model of chronic liver injury.
TNFR1 deficiency reduces tumor load in NEMOΔhepa/Faslpr livers
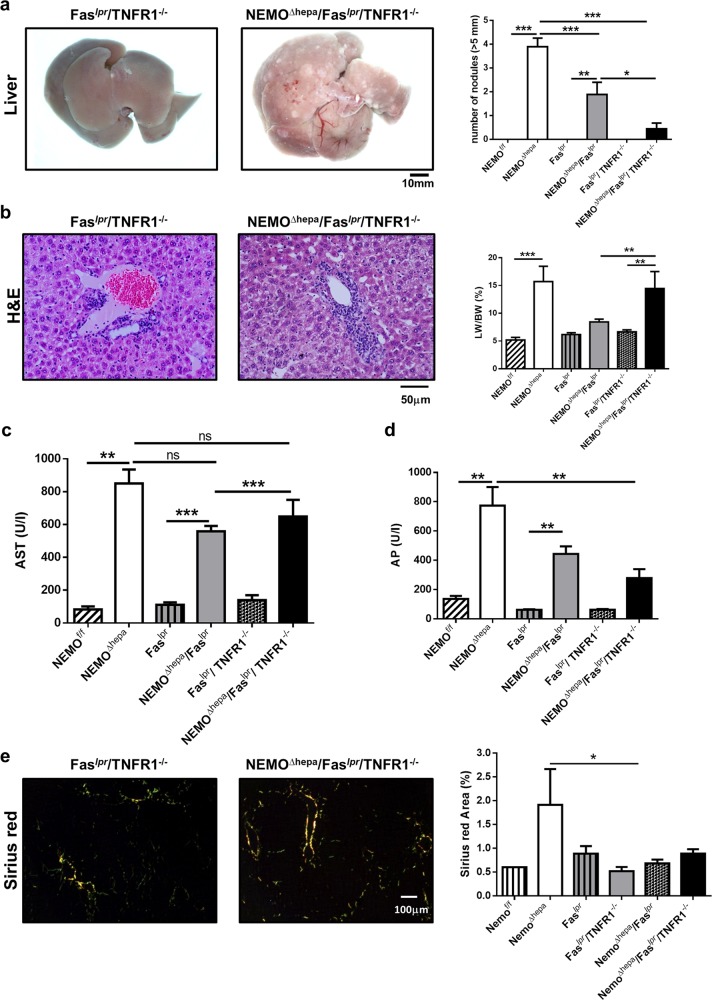
Since deletion of TNFR1 is highly beneficial not only to NEMOΔhepa 10 but also to NEMOΔhepa/p21−/− knockout mice14, we generated NEMOΔhepa/Faslpr/TNFR1−/− triple knockout (TKO) mice and investigated the progression of chronic liver disease in these animals. Eight-week-old TKO mice displayed increased spleen size and a significantly larger number of peritoneal lymph nodes (Supplementary Figure 5a–c). Macroscopically, 8-week-old NEMOΔhepa/Faslpr/TNFR1−/− livers revealed reduced number of nodules, accompanied by significantly ameliorated ALT, AP, and GLDH compared with NEMOΔhepa and NEMOΔhepa/Faslpr mice (Supplementary Figure 6a–d).
Histopathological examination indicated that 1-year-old NEMOΔhepa/Faslpr/TNFR1−/− knockout mice presented no signs of neoplasia, early lymphomas, and reduced number of nodules (Table 1). However, no differences in LW/BW ratio (Fig. 5a, b) or important changes in serum AST levels albeit significantly decreased AP levels in 52-week-old NEMOΔhepa/Faslpr/TNFR1−/− compared with NEMOΔhepa, and NEMOΔhepa/Faslpr mice were observed (Fig. 5c, d). Interestingly, NEMOΔhepa/Faslpr/TNFR1−/− knockout mice displayed similar inflammation as NEMOΔhepa/Faslpr livers as observed by Sirius red staining and quantification (Fig. 5e, Supplementary Figure 7). Altogether, these data show that TNF deficiency reduces tumorigenesis in NEMOΔhepa/Faslpr livers.
Fig. 5. Deletion of TNFR1 amplifies the protective effect of Fas signalling blockade in NEMO mice.
Faslpr/TNFR1−/− and NEMOΔhepa/Faslpr/TNFR1−/− mice at the age of 12 months were sacrificed and livers extracted and analyzed. a Macroscopic appearance of livers. Arrows show visible nodules. The number of nodules (≥5 mm) diameter was quantified. b Representative H&E staining of liver sections. Liver weight versus body weight (LW/BW) ratio is shown. Serum AST (c) and AP (d) were determined in the same mice and compared to 1-year-old NEMOf/f, NEMOΔhepa, Faslpr, and NEMOΔhepa/Faslpr. e Sirius red staining of paraffin-embedded liver tissue was performed and photomicrographs were taking. SR-positive area was quantifed using ImageJ© and represented including 1-year-old NEMOf/f, NEMOΔhepa, Faslpr, and NEMOΔhepa/Faslpr. Results are expressed as mean ± SEM (n = 25, *p < 0.05; **p < 0.01)
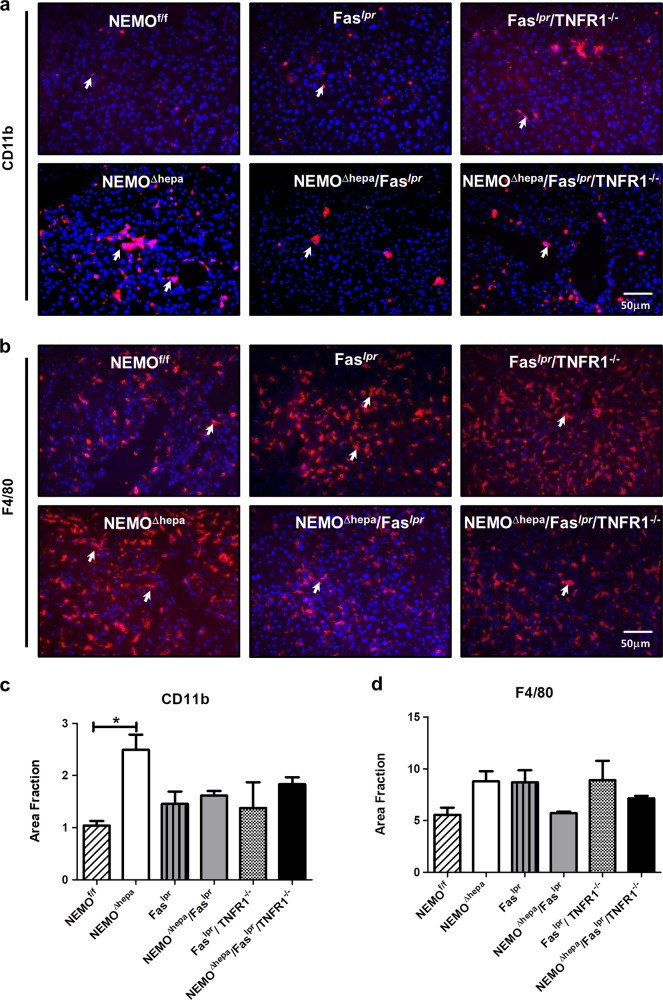
In order to better characterize the TNFR1-mediated effect in NEMOΔhepa/Faslpr, the inflammatory profile of 52-week-old livers was exhaustively investigated. The number of CD11b-positive cells was statistically significantly higher in 52-week-old NEMOΔhepa livers compared to WT animals (Fig. 6c). Elevated F4/80 cells were also found in NEMOΔhepa livers. In contrast, a trend towards reduction of CD11b and F4/80 cells in 52-week-old NEMOΔhepa/Faslpr livers was observed (Fig. 6a–d). However, no differences in cell infiltration were evident between NEMOΔhepa/Faslpr and NEMOΔhepa/Faslpr/TNFR1−/− animals, suggesting that TNFR1 might be more involved in modulating tumorigenesis.
Fig. 6. Impact of Fas and TNFR1 deletion on liver infiltrating inflammatory cells in NEMOΔhepa mice.
a Representative CD11b immunostaining of 52-week-old NEMOf/f, NEMOΔhepa, Faslpr NEMOΔhepa/Faslpr, Faslpr/TNFR1−/−, and NEMOΔhepa/Faslpr/TNFR1−/− livers. b The positive Area Fraction for CD11b was quantified in ImageJ© and graphed. c Representative F4/80 immunostaining of 52-week-old NEMOf/f, NEMOΔhepa, Faslpr NEMOΔhepa/Faslpr, Faslpr/TNFR1−/−, and NEMOΔhepa/Faslpr/TNFR1−/− livers. d The positive Area Fraction for F4/80 was quantified in ImageJ© and graphed
Discussion
Apart from TNFR1 and TRAILR/D5, FasL/Fas is the third death receptor of the TNF receptor superfamily that can activate caspase-8-mediated apoptosis15. Previously, we first showed that loss of TRAILR does not improve experimental hepatitis as others have recently confirmed10,16. Next, we observed that TNFR1 deletion is beneficial for the progression of chronic liver injury in NEMO∆hepa mice10,14. Moreover, we specifically showed that TNFR1 deletion in hepatocytes is protective, whereas TNFR1 inactivation in bone-marrow-derived cells might be deleterious in this experimental model of chronic liver disease. In contrast, a recent study suggested no role for TNFR1 in hepatocytes in the same model albeit showing a clear trend towards reduced transaminases and tumorigenesis16.
Since its first description in 1989, the FasR and FasL system has become the best-characterized extracellular system triggering apoptosis17. Accumulating evidence highlighted the importance of FasL/Fas expression in the pathogenesis of many gastrointestinal diseases including Wilson’s disease, cholestatic liver disease, alcoholic hepatitis, non-alcoholic steatohepatitis (NASH), and hepatocellular carcinoma (HCC)18,19. In the present study, we hypothesized that Fas mediates TNF-induced cell death in NEMO-deficient hepatocytes thus triggering the progression of chronic liver disease and end-stage HCC.
As previously described20, Fas mice (Faslpr), mutant for Fas, develop splenomegaly, lymphadenopathy, and glomerulonephritis, including mononuclear cell infiltration, and display accumulation of CD4−CD8− T lymphocytes in peripheral organs such as the liver. These mice develop autoimmunity and are an excellent model of systemic lupus erythematosus. Interestingly, NEMOΔhepa/Faslpr displayed larger spleens than Faslpr mice but no differences in number of lymph nodes. On the other hand, Faslpr/TNFR1−/− and NEMOΔhepa/Faslpr/TNFR1−/− animals exhibited a larger number of peritoneal nodes.
Defective Fas signalling in NEMOΔhepa livers resulted in decreased serum transaminases and multifocal necrosis. In contrast, NEMOΔhepa with normal Fas signalling displayed high mitotic index, oval cell proliferation, mild lipidosis, and diffuse vasculopathy. FasL and Fas are expressed in NEMO-deficient livers, suggesting that they might be involved in mediating apoptosis in NEMOΔhepa hepatocytes16. Thus, we first explored the role of Fas in cell death of NEMOΔhepa mice. Decreased TUNEL-positive cells and absence of Caspase-3 activation were associated with lower compensatory proliferation in NEMOΔhepa/Faslpr compared with NEMOΔhepa livers. Moreover, Faslpr do not develop liver hyperplasia, but small amount of Fas protein may still be produced by the lpr mutant Fas allele and mice engineered to completely lack Fas protein did exhibit liver hyperplasia21. Altogether, neither systemic nor specific ablation of Fas in hepatocytes completely prevent cell death but increased hepatocyte survival in NEMOΔhepa animals.
Next, we measured TNF levels since this proinflammatory cytokine is responsible for cell death in NEMOΔhepa liver8. Interestingly, TNF levels were significantly downregulated in NEMOΔhepa/Faslpr. Moreover, NEMOΔhepa/Faslpr livers exhibited decreased liver fibrosis and significantly reduced presence of CD11b+ F4/80+ cells. Since inflammation is a critical factor for progression of liver injury, these observations suggest that an attenuated inflammatory response, and not reduced Fas-induced apoptosis was the cause for the protective effect in NEMOΔhepa mice22.
Previously, our group showed that NEMOΔhepa mice are resistant to Jo2 stimulation, a model of Fas-induced damage9. However, direct binding of Jo2 to hepatocytes in vivo is not completely clear23, and the effect of Jo2 is not restricted only to the liver; it could also affect other tissues of which cells often express a higher level of Fas than hepatocytes. Additionally, Ehlken16, using specific deletion of Fas in liver parenchymal cells, found that Fas is not required for the development of chronic liver damage in NEMOΔhepa mice.
In the present study, we employed NEMOΔhepa/Faslpr mice, which carry a mutation in their Fas gene caused by insertion of the Etn retrotransposon into intron 2 of this gene24. Hence, these result in the expression of a completely defective Fas antigen. Thus, our experiments overcome the underestimated effect of Fas-mediated signaling in other tissues that can directly affect the development of chronic liver disease. Concomitant to our results, Hatano et al.25 also showed that NF-κB inhibition sensitizes hepatocytes to Fas-mediated apoptosis. Altogether, our study takes into account overall Fas signaling rather than hepatocyte-specific Fas-mediated apoptosis to explain the phenotype of NEMOΔhepa mice.
The FasL/Fas system is a major mechanism of certain types of cancer cells for avoiding detection and destruction by the immune system through FasL expression7. Interestingly, NEMOΔhepa/Faslpr displayed reduced tumorigenesis compared to NEMOΔhepa mice. Moreover, in the chronic phase reduced inflammation-driven carcinogenesis and lack of functional T cells play an essential role in leading to reduced disease progression in NEMOΔhepa/Faslpr livers, which is associated with reduced fibrosis and liver tumorigenesis.
Besides a direct cytotoxic effect on hepatocytes, the TNF/TNFR1 system might be also required for Fas-mediated cell death, as demonstrated by the increased resistance of TNFR1/2 double knockout mice to Fas-induced fulminant liver injury26. Thus, we further assessed the role of TNFR signaling in Fas signaling. As previously described as beneficial in ethanol-mediated liver injury27, deletion of TNFR1 in NEMOΔhepa/Faslpr animals reduced the tumor load of these mice, indicating that TNFR1 deficiency might modulate tumor load in HCC development.
Therefore, therapeutic approaches aiming to inhibit Fas-mediated liver injury might be relevant in inhibiting inflammation-driven hepatic disease progression including growth of HCC.
Supplementary information
Acknowledgements
This work was supported by the IZKF, the START-Program of the Faculty of Medicine #691405, RWTH Aachen), the DFG TR285-10/1, the German Krebshilfe Grant Nr. 361209 and the MINECO Retos SAF2016-78711, EXOHEP-CM S2017/BMD-3727, AMMF 2018/117 and ERAB EA 18/14 and COST Action CA17112. F.J.C. is a Ramón y Cajal Researcher (RYC-2014-15242) and a Gilead Liver Research Scholar 2018. We are grateful to the Department of Human and Veterinary Anatomy and Embryology at the Complutense University School of Medicine for their continuous support.
Conflict of interest
The authors declare that they have no conflict of interest.
Footnotes
Edited by G. Raschellà
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
These authors contributed equally: Francisco Javier Cubero, Marius Maximilian Woitok and Miguel E. Zoubek
These author jointly supervised: Maximilian Hatting, Christian Trautwein
Contributor Information
Francisco Javier Cubero, Phone: +34-91394-1385, Email: fcubero@ucm.es.
Christian Trautwein, Phone: +49-241-80-80866, Email: ctrautwein@ukaachen.de.
Electronic supplementary material
Supplementary Information accompanies this paper at (10.1038/s41419-019-1391-x).
References
- 1.Fecho K, Cohen PL. Fas ligand (gld)- and Fas (lpr)-deficient mice do not show alterations in the extravasation or apoptosis of inflammatory neutrophils. J. Leukoc. Biol. 1998;64:373–383. doi: 10.1002/jlb.64.3.373. [DOI] [PubMed] [Google Scholar]
- 2.Faubion WA, Gores GJ. Death receptors in liver biology and pathobiology. Hepatology. 1999;29:1–4. doi: 10.1002/hep.510290101. [DOI] [PubMed] [Google Scholar]
- 3.Ito Y, et al. Fas antigen expression in hepatocellular carcinoma tissues. Oncol. Rep. 1998;5:41–44. [PubMed] [Google Scholar]
- 4.Abrams SI. Positive and negative consequences of Fas/Fas ligand interactions in the antitumor response. Front. Biosci. 2005;10:809–821. doi: 10.2741/1575. [DOI] [PubMed] [Google Scholar]
- 5.Nicholes K, et al. A mouse model of hepatocellular carcinoma: ectopic expression of fibroblast growth factor 19 in skeletal muscle of transgenic mice. Am. J. Pathol. 2002;160:2295–2307. doi: 10.1016/S0002-9440(10)61177-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Mikhail S. & He A. R. Liver cancer stem cells. Int. J. Hepatol. (2011). 10.4061/2011/486954 [DOI] [PMC free article] [PubMed]
- 7.Nagao M, et al. The alteration of Fas receptor and ligand system in hepatocellular carcinomas: how do hepatoma cells escape from the host immune surveillance in vivo? Hepatology. 1999;30:413–421. doi: 10.1002/hep.510300237. [DOI] [PubMed] [Google Scholar]
- 8.Luedde T, et al. Deletion of NEMO/IKKgamma in liver parenchymal cells causes steatohepatitis and hepatocellular carcinoma. Cancer Cell. 2007;11:119–132. doi: 10.1016/j.ccr.2006.12.016. [DOI] [PubMed] [Google Scholar]
- 9.Beraza N, et al. Hepatocyte-specific NEMO deletion promotes NK/NKT cell- and TRAIL-dependent liver damage. J. Exp. Med. 2009;206:1727–1737. doi: 10.1084/jem.20082152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Cubero FJ, et al. TNFR1 determines progression of chronic liver injury in the IKKgamma/Nemo genetic model. Cell Death Differ. 2013;20:1580–1592. doi: 10.1038/cdd.2013.112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Cubero FJ, et al. Haematopoietic cell-derived Jnk1 is crucial for chronic inflammation and carcinogenesis in an experimental model of liver injury. J. Hepatol. 2015;62:140–149. doi: 10.1016/j.jhep.2014.08.029. [DOI] [PubMed] [Google Scholar]
- 12.Adachi M, et al. Enhanced and accelerated lymphoproliferation in Fas-null mice. Proc. Natl Acad. Sci. USA. 1996;93:2131–2136. doi: 10.1073/pnas.93.5.2131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Karlmark KR, Wasmuth HE, Trautwein C, Tacke F. Chemokine-directed immune cell infiltration in acute and chronic liver disease. Expert Rev. Gastroenterol. Hepatol. 2008;2:233–242. doi: 10.1586/17474124.2.2.233. [DOI] [PubMed] [Google Scholar]
- 14.Ehedego H, et al. p21 ablation in liver enhances DNA damage, cholestasis, and carcinogenesis. Cancer Res. 2015;75:1144–1155. doi: 10.1158/0008-5472.CAN-14-1356. [DOI] [PubMed] [Google Scholar]
- 15.Ashkenazi A, Dixit VM. Death receptors: signaling and modulation. Science. 1998;281:1305–1308. doi: 10.1126/science.281.5381.1305. [DOI] [PubMed] [Google Scholar]
- 16.Ehlken H, et al. Death receptor-independent FADD signalling triggers hepatitis and hepatocellular carcinoma in mice with liver parenchymal cell-specific NEMO knockout. Cell Death Differ. 2014;21:1721–1732. doi: 10.1038/cdd.2014.83. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Rudiger HA, Clavien PA. Tumor necrosis factor alpha, but not Fas, mediates hepatocellular apoptosis in the murine ischemic liver. Gastroenterology. 2002;122:202–210. doi: 10.1053/gast.2002.30304. [DOI] [PubMed] [Google Scholar]
- 18.Hammam O, et al. The role of fas/fas ligand system in the pathogenesis of liver cirrhosis and hepatocellular carcinoma. Hepat. Mon. 2012;12:e6132. doi: 10.5812/hepatmon.6132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Guicciardi ME, Gores GJ. Apoptosis: a mechanism of acute and chronic liver injury. Gut. 2005;54:1024–1033. doi: 10.1136/gut.2004.053850. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Hutcheson J, Perlman H. Loss of Bim results in abnormal accumulation of mature CD4-CD8-CD44-CD25- thymocytes. Immunobiology. 2007;212:629–636. doi: 10.1016/j.imbio.2007.05.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Adachi M, et al. Targeted mutation in the Fas gene causes hyperplasia in peripheral lymphoid organs and liver. Nat. Genet. 1995;11:294–300. doi: 10.1038/ng1195-294. [DOI] [PubMed] [Google Scholar]
- 22.Gujral JS, Liu J, Farhood A, Jaeschke H. Reduced oncotic necrosis in Fas receptor-deficient C57BL/6J-lpr mice after bile duct ligation. Hepatology. 2004;40:998–1007. doi: 10.1002/hep.1840400431. [DOI] [PubMed] [Google Scholar]
- 23.Jodo S, et al. Anti-CD95-induced lethality requires radioresistant Fcgamma RII + cells. A novel mechanism for fulminant hepatic failure. J. Biol. Chem. 2003;278:7553–7557. doi: 10.1074/jbc.M211229200. [DOI] [PubMed] [Google Scholar]
- 24.Li XK, et al. Fulminant hepatitis by Fas-ligand expression in MRL-lpr/lpr mice grafted with Fas-positive livers and wild-type mice with Fas-mutant livers. Transplantation. 2001;71:503–508. doi: 10.1097/00007890-200102270-00004. [DOI] [PubMed] [Google Scholar]
- 25.Hatano E, et al. The mitochondrial permeability transition augments Fas-induced apoptosis in mouse hepatocytes. J. Biol. Chem. 2000;275:11814–11823. doi: 10.1074/jbc.275.16.11814. [DOI] [PubMed] [Google Scholar]
- 26.Costelli P, et al. Mice lacking TNFalpha receptors 1 and 2 are resistant to death and fulminant liver injury induced by agonistic anti-Fas antibody. Cell Death Differ. 2003;10:997–1004. doi: 10.1038/sj.cdd.4401281. [DOI] [PubMed] [Google Scholar]
- 27.Nanji AA, French SW. Animal models of alcoholic liver disease--focus on the intragastric feeding model. Alcohol Res. Health. 2003;27:325–330. [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.