Abstract
Acute respiratory distress syndrome is a major cause of respiratory failure in critically ill patients. Despite extensive research into its pathophysiology, mortality remains high. No effective pharmacotherapy exists. Based largely on numerous preclinical animal studies, administration of mesenchymal stem or stromal cell (MSC) as a therapeutic for acute lung injury holds great promise, and phase I/II clinical trials are currently underway internationally. However, concern for the use of stem cells, specifically the risk of iatrogenic tumor formation, as well as the prohibitive cost of production, storage, and distribution of cells in bone marrow transplant facilities may limit access to this life saving therapy. Accumulating evidence now suggest that novel stem cell derived therapies including MSC conditioned medium and extracellular vesicles released from MSCs, might constitute compelling alternatives. The current review summarizes the preclinical studies testing MSC extracellular vesicles as treatment for acute lung injury and other inflammatory lung diseases. While certain logistical obstacles limit the clinical applications of MSC conditioned medium such as the volume required for treatment and lack of standardization of what constitutes the components of conditioned medium, the therapeutic application of MSC extracellular vesicles remains promising, primarily due to ability of extracellular vesicles to maintain the functional phenotype of the parent cell. However, utilization of MSC extracellular vesicles will require large-scale production and standardization concerning identification, characterization and quantification.
Keywords: Acute Lung Injury, Acute Respiratory Distress Syndrome, Exosomes, Extracellular Vesicles, Mesenchymal Stem Cells, Microvesicles
INTRODUCTION
Acute respiratory distress syndrome (ARDS), the clinical manifestation of acute lung injury (ALI), is a major cause of acute respiratory failure where mortality remains as high as 33–48% in critically ill patients.1,2 The most common triggering event for ARDS is severe sepsis from either a pulmonary or non-pulmonary source, accounting for 79% of the cases.1 However, various other inciting events including aspiration, toxic inhalation, lung contusion, acute pancreatitis, trauma, transfusion, burn injury, and cardiopulmonary bypass surgery can cause ARDS.3 Once triggered by infectious, chemical, or mechanical insult, the pathophysiology of ARDS results from complex interaction between the immune system and the alveolar-capillary barrier. Acute inflammatory response following the insult affects endothelial and epithelial type I/II cells in the alveoli. Damage to alveolar endothelium and epithelium directly increase the permeability of the alveolar-capillary barrier, and damage to type II cell impair pulmonary surfactant secretion and alveolar fluid clearance. The increased permeability of the alveolar-capillary barrier and impaired alveolar clearance result in pulmonary edema, which eventually cause impaired gas exchange and hypoxemia.4 Currently, there is no available pharmacotherapy based on the pathophysiology of ARDS. The therapeutic options are limited to primarily supportive measures such as lung-protective ventilation, fluid conservative strategy, and prone positioning.5–8
Recently, mesenchymal stem cells (MSC) have been regarded as a promising approach for ARDS because of their ability to attenuate the major pathologies underlying ALI. MSC are non-hematopoietic precursor cells from a variety of tissues in adult such as the bone marrow, adipose tissue, and placenta. MSC were first described as fibroblast-like cells of the bone marrow in 1968.9 The definition of MSC was established by the International Society of Cellular Therapy in 2006 as the following criteria: 1) adherence to plastic: MSC must be adherent to plastic under standard tissue culture conditions; 2) specific surface antigens: MSC must express certain cell surface markers such as CD105, CD90 and CD73, but must not express CD45, CD34, CD14 or CD11b, CD79a, CD19 or HLA-DR; 3) and multipotent differentiation: MSC must be able to differentiate into osteoblasts, adipocytes and chondroblasts under standard in vitro conditions.10 The therapeutic effects of MSC were initially thought to derive from their engraftment in the injury site and regeneration. However, the presence of MSC in the injured organ is only transient, and the replacement of the damaged tissues with transdifferentiated stem cells is very limited (<5%).11,12 Current view is that therapeutic properties of MSC are derived from soluble factors with paracrine or endocrine effects.13–15 The soluble factors include growth factors, anti-inflammatory cytokines, and antimicrobial peptides, which can stabilize the alveolar-capillary barrier, enhance alveolar fluid clearance, and decrease infection.16 Recent studies have also underscored the new potential role of extracellular vesicles (EV), small anuclear membrane bound particles released from MSC, as a paracrine or endocrine vehicle to deliver various soluble factors with a similar phenotype as the parent cell.17–21 Although the preclinical studies for the therapeutic use of MSC derived EVs in ARDS is still in its infancy, its potential as treatment vs. using intact live cells are significant such as: 1) ability to store EVs without the use preservatives such as DMSO; 2) no need for a bone marrow transplant facility to store living cells, potentially increasing the number of facilities which can deliver stem cell derived therapy; 3) and potential ability to give multiple doses without significantly effecting the patient’s hemodynamic or respiratory parameters. In the current review, we summarize the biological basis and the preclinical data available for the potential use of MSC-derived EV for ALI.
DEFINITION AND CHARACTERIZATION OF EXTRACELLULAR VESICLES
EVs are anuclear, membrane bound vesicles that are released by most cell types into the extracellular space. EVs may be secreted constitutively or following cell stimulation by soluble agonists, physical or chemical injury such as the oxidative stress and hypoxia, or shear stress.22 Although once considered cellular debris or as artifacts, EVs play an important role in intercellular communication by transporting various cargoes including proteins, RNAs, DNAs, and lipids between cells at both a paracrine and endocrine level.23–28 As EV transports complex biological information from donor cells to recipient cells, it can contribute to the maintenance of normal and pathophysiological conditions.25,29 EVs can be classified into exosomes, microvesicles, or apoptotic bodies according to size, biogenesis, and composition.30–32 Because there is no consensus on the nomenclature for the type of vesicles, especially for the microvesicles, other names such as shedding vesicles, ectosomes, exosome-like vesicles, nanoparticles, microparticles, and oncosomes have been employed in the literature. Microvesicles are generally heterogeneous in size ranging from 50 to 1000 nm in diameter depending on the state of the cell during release. Whereas, exosomes are relatively homogenous in size with a diameter ranging from 40 to 200 nm. Furthermore, the process of vesicle formation and release from cells also differs between exosomes and microvesicles. Microvesicles are released by direct shedding or budding from the plasma membrane. Whereas, exosomes are assembled through an invagination process of the endosomal membrane of multi-vesicular bodies (MVBs) within cells and, then, the fusion of the MVBs with the plasma membrane resulting in its release.32 Due to the difference in assembly and release, each of the EV subtypes has its own characteristic surface and intracellular markers.33 The release of exosomes is often detected within minutes after stimulation while microvesicles are released within seconds after stimulation. Although there can be some uncertainty or overlap for EV subtype markers, CD63 and CD61 are known as common stereotypic markers for exosomes, and TyA and C1q for microvesicles. However, despite such differences, both of the two EV subtypes are released upon physiological or pathological stimulus and have important effects on cell physiology and pathology by transferring their cargoes to target cells over short or long distances. Some EVs can break down shortly after release and excrete their contents to the extracellular space. The released agents bind their receptors in adjacent cells and provoke rapid responses.34–36 Most EVs that maintain their structure longer can travel to target cells, and thus EVs can be found in major body fluids, such as blood, lymph, and cerebrospinal fluid.33 There are various patterns of the interaction between EVs and the target cells; 1) Direct stimulation to target cell through surface-expressed ligands; 2) Direct fusion to target cell transferring membrane receptor and luminal material; 3) And uptake of entire EVs by target cell in the endocytosis process 22,33. The interactions enable EVs to directly stimulate or transfer various materials including proteins and genetic information to target cells.
MSCs also secrete EVs, and the MSC-derived EVs have been shown to be as effective as MSCs in accelerating recovery in various injury models37 by transferring their content to recipient cells, resulting in a therapeutic effect. In ALI models, various therapeutic agents including keratinocyte growth factor (KGF), angiopoietin-1 (Ang-1), cyclooxygenase 2, and mitochondria have been identified. Because there are no standardized methods for isolation, quantification and characterization of EVs, or differentiating MVs and exosomes, it is difficult to compare and analyze studies with MSC-derived EVs in terms of dose, route, and efficacy. However, in most studies involving MSC-derived EVs in ALI, EVs were isolated by ultracentrifugation at speeds of approximately 100,000 × g, and differential centrifugation or size columns were used to differentiate EVs into microvesicles and exosomes. In this review, we will use MSC-derived EVs as an umbrella term to include both microvesicles and exosomes.
While most studies have demonstrated the direct inhibition of inflammatory effector cells by MSCs, MSC can also have differential immunomodulatory effects, or even cause an inflammatory response, based largely by environmental cues. Depending on the level of inflammatory cytokines such as IFN-γ, IL-1ß, and TNFα, MSC can enhance the differentiation of naïve T cells into immunosuppressive Treg cells or pro-inflammatory Th17 cells, and can cause an increase or decrease of T cell proliferation.38 Furthermore, MSCs monitor their environment and recognize danger signals through various toll like receptors (TLR). The stimulation of different TLRs result in different phenotypes of MSCs, with TLR3-treated MSC having increased immunomodulatory potential, whereas TLR4 stimulation accelerating the pro-inflammatory response.39,40 Therefore, pre-treatment of MSC with inflammatory cytokines or TLR3 agonist can enhance the therapeutic effects in inflammatory disease models.41–44 The content and the therapeutic efficacy of MSC-derived EV can be differentiated according to the parental MSC phenotype. A recent study demonstrated that MSC pre-treated with IL-1β have greater immunomodulatory effects than naïve MSC by increasing microRNA-146a content in the MSC-derived EVs in a sepsis model.45 Another study using an ALI model showed that EV collected from pre-treated MSC with TLR3 agonist demonstrated increased antimicrobial effects than naïve MSC derived EV.46 Therefore, targeted pre-stimulation of MSC can be an efficient strategy to generate EVs with high therapeutic potential.
THERAPEUTIC USE OF MSC DERIVED EXTRACELLULAR VESICLES IN ALI MODELS
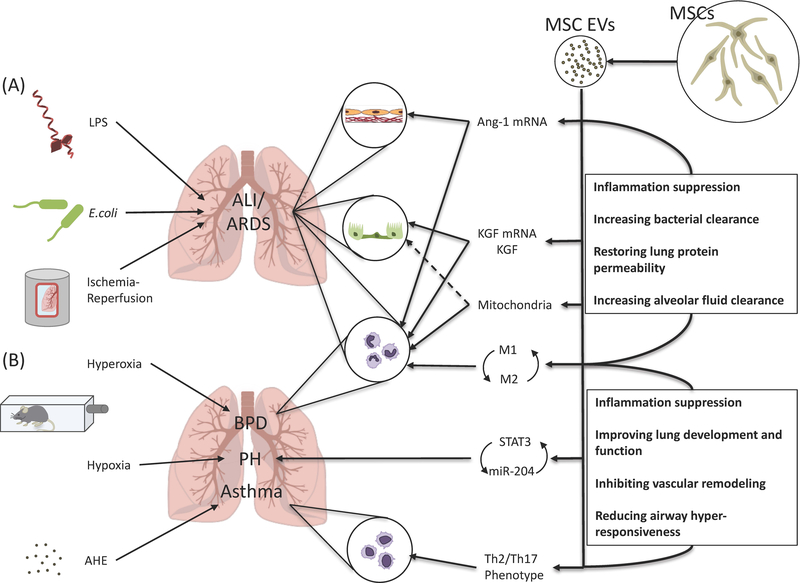
Cellular administration of MSCs as a therapeutic in preclinical models of ALI is very promising, and several phase 1/2 clinical trials with the use of MSCs in ARDS and/or sepsis are underway (such as NCT02097641 or NCT02421484). The biological rationale for the use of MSC in ALI include anti-inflammation on host tissue, reduction of the permeability of alveolar epithelium and endothelium, improvement of alveolar fluid clearance, enhancement of macrophage phagocytosis, and tissue repair through direct mitochondrial transfer with host cells.47,48 Similar to MSCs, the early pre-clinical studies using MSC-derived EV in several ALI models are promising and discussed below (Figure).
Figure. Therapeutic potential of extracellular vesicles derived from mesenchymal stem cells in acute lung injury.
(A) In ALI models with various etiologies including lipopolysaccharide, Escherichia coli bacteria, and ischemia-reperfusion injury, administration of MSC-derived EVs was associated with the transfer of Ang-11 and KGF mRNA and possibly mitochondria from the EVs to the alveolar epithelium and endothelium, contributing in preservation of alveolar-capillary permeability and improved alveolar fluid clearance. MSC-derived EVs also changed monocyte/macrophage towards an anti-inflammatory phenotype with increased phagocytic activity, which resulted in increased bacterial clearance. (B) In a model of hyperoxia-induced bronchopulmonary dysplasia, MSC-derived exosomes improved lung architecture and function through modulation of lung macrophage phenotype, suppressing the pro-inflammatory M1 and augmenting an anti-inflammatory M2-like state. In a model of hypoxia-induced pulmonary hypertension, MSC-derived exosomes also prevented vascular remodeling by suppressing the hypoxic induction of STAT3 and up-regulated miR-204 levels, interfering with the STAT3-miR-204-STAT3 feed-forward loop. In a model of aspergillus hyphal extract-induced asthma, MSC-derived EVs mitigated Th2/Th17-mediated airway hyper-responsiveness by shifting the Th2/Th17 inflammatory response towards a counter-regulatory Th1 response. MSC, mesenchymal stem cell; EV, extracellular vesicle; LPS, lipopolysaccharide; E. coli, Escherichia coli; ALI, acute lung injury; ARDS, acute respiratory distress syndrome; Ang-1, angiopoietin-1; KGF, keratinocyte growth factor; BPD, bronchopulmonary dysplasia; PH, pulmonary hypertension; STAT3, signal transducer and activator transcription 3; AHE, aspergillus hyphal extract.
1). Endotoxin-induced ALI
Zhu et al. demonstrated the therapeutic efficacy and mechanism of human MSC-derived EV in a mice ALI model induced by intra-tracheal administration of E.coli endotoxin.21 In the study, MSC-derived EV reduced alveolar inflammation and edema by decreasing the influx of inflammatory cells and total protein levels in the endotoxin-damaged alveolus. In addition, the therapeutic effects of the EV were comparable regardless of route of administration, intra-tracheal or intravenous. Elimination of KGF activity found in the EVs using either siRNA or KGF antibody partially abrogated the therapeutic effects of MSC-derived EV, which suggested that the transfer of KGF mRNA to the target tissue was one of mechanisms of action. KGF, also known as FGF7, is an epithelial specific growth factor and a major paracrine factor released from MSCs with significant reparative properties. In ALI models, KGF from MSC has been shown to restore protein permeability and increase fluid clearance in the alveolus following injury.47,48
A recent study by Tang et al.49 also demonstrated MSC-derived EVs as a therapeutic agent in endotoxin-induced ALI in mice. Intra-tracheal administration of MSC EVs ameliorated lung inflammation and restored alveolar-capillary permeability after endotoxin induced injury. Furthermore, administration of the EVs suppressed TNFα and increased IL-10 secretion in a mouse macrophage cell line (RAW264.7) following endotoxin stimulation. Administration of EVs from Ang-1 SiRNA transfected MSCs partly abrogated the beneficial effects on alveolar inflammation and permeability in mice as well as immunomodulation in macrophages. Ang-1 is an angiogenic factor that stabilizes endothelial cells during injury, reduces endothelial permeability, and suppresses leukocyte-endothelium interactions. Ang-1 is also significantly secreted by MSCs.47,48
Recently, Morrison et al.50 demonstrated that MSC-derived EV protected against endotoxin-induced ALI by altering alveolar macrophage towards an anti-inflammatory phenotype with enhance phagocytic activity via EV-mediated mitochondrial transfer. Intra-tracheal administration of alveolar macrophages pre-treated with MSC-derived EV reduced inflammatory cells recruitment and the levels of TNFα and protein in the alveolus of mice with endotoxin-induced lung injury. Previously, using MSC as a therapeutic to prevent silica-induced lung inflammation and fibrosis, Phinney et al.51 also found that MSCs shed exosomes that modulated toll-like receptor signaling and cytokine secretion in macrophages, in part, by transfer of regulatory microRNAs; miR-451, known to suppress TNF and macrophage migration inhibitory factor, was highly abundant in MSC-derived exosomes, suggesting that the possible transfer of miR-451 to and increased expression in macrophages inhibited TNF secretion in response to silica. The authors also demonstrated that MSC-derived exosomes prevented the recruitment of Ly6Chi monocytes and reduced secretion of pro-fibrotic IL-10 and TGFβ by these cells. Lastly, the author found that MSCs managed intracellular oxidative stress by the transfer of depolarized mitochondria by MSCs. MSC-derived vesicles containing the mitochondria were engulfed and re-utilized by macrophages, resulting in enhanced bioenergetics. These two studies demonstrated that mitochondrial transfer by MSC-derived EVs was a significant mechanism for its therapeutic effect. It was already known that MSC was able to transfer mitochondria to the alveolar epithelium52 and macrophage53 through direct cell contact via tunneling nanotubes, but, now, MSC-derived EVs was also able to transfer mitochondria to recipient cells.
2). Bacteria-induced ALI
Monsel et al.46 instilled live E.coli bacteria into trachea, which caused bacterial pneumonia and lung injury, in order to demonstrate the therapeutic effect of MSC-derived EV on bacteria-induced ALI. The authors demonstrated that intravenous administration of MSC-derived MVs improved survival and reduced the bacterial load, inflammation, and protein permeability in the injured alveolus with live E.coli. The therapeutic effects were mediated in part by KGF. More specifically, MSC-derived EV increased the phagocytosis of bacteria and suppressed TNFα secretion in monocytes stimulated with bacteria. MSC-derived EV also restored intracellular ATP levels in injured alveolar epithelial type II cells. These results confirmed that the mechanisms of MSC-derived EV are as variable as those of MSCs including anti-inflammatory, anti-microbial, and with direct metabolomic effects. Furthermore, the EVs isolated from pre-stimulated MSC with TLR3 agonist further increased phagocytosis, decreased TNFα secretion, increased IL-10 secretion in monocytes compared to naïve MSC-derived EVs in mice with bacteria-induced ALI, suggesting that the contents and effects of the MSC-derived EVs can be modified by changing the phenotype of the parent cell. Lastly, Monsel et al. also found that the surface receptor CD44 was crucial for incorporation of MSC-derived EVs into injured monocytes and alveolar epithelial type 2 cells. CD44 is the principal surface receptor for hyaluronic acid, a major extracellular matrix component.54 It is expressed in almost every cell type including MSC, and associated with the regulation of cell proliferation, differentiation, survival, migration, and trafficking to tissues. CD44 is essential for the migration of MSC into the extracellular matrix.55 CD44 on the surface of MSC-derived EV appears to be important for in the incorporation into injured tissues, which is critical step for the therapeutic effects.
3). Ischemia/reperfusion-induced ALI
Gennai et al.56 used ex vivo perfused human lungs rejected for transplantation treated with MSC-derived EVs as a technique for rehabilitating marginal donor lungs.57 In the study, intravenous administration of MSC-derived EV improved alveolar fluid clearance rate in a dose dependent manner and decreased lung weight gain or pulmonary edema. MSC-derived EVs also increased nitric oxide level in the perfusate, which was associated with improvement of lung compliance and decrease in pulmonary artery pressure/resistance. Furthermore, MSC-derived EV reduced perfusate pH and lactate level, showing the metabolomic effects of MSC-derived EV. Finally, co-administration of CD44 neutralizing antibody with MSC-derived EVs abrogated the therapeutic effects on alveolar fluid clearance, lung weight, compliance, and pulmonary artery pressure/resistance, demonstrating the critical role of CD44 in the activity of the EVs.
4). Other lung disease models
There have been several investigations using MSC-derived EVs in the other lung diseases aside from ALI, which can help us understand the biological mechanisms of MSC-derived EVs in the pathophysiology of ALI. Cruz et al.58 demonstrated that MSC-derived EVs relieved allergic airway inflammation induced by repeated mucosal exposure of Aspergillus hyphal extract (AHE) in mice as a model of severe refractory asthma. Systemic administration of MSC-derived EVs isolated from both human and murine MSC suppressed the AHE-specific release of Th2- (IL-4 and IL-5) and Th17- (IL-17) mediated cytokines and boosted a counter-regulatory Th1 response (IFN-γ) in bronchoalveolar lavage fluid and in mixed lymphocyte culture. This study demonstrated that both syngeneic and xenogeneic administration of MSC-derived EV are effective in ameliorating airway hyper-reactivity and lung inflammation by modulating Th2/Th17-mediated allergic airway inflammation. Blocking the release of both soluble factors and/or EVs from MSC completely abrogated the therapeutic effects of human MSC, but only partly inhibited those of murine MSC, which suggests that some different mechanisms exists between murine and human MSCs.
Lee et al.20 revealed in a mouse model of hypoxic pulmonary hypertension that MSC-derived EV can partially reduce pulmonary hypertension specifically using the exosome fraction isolated by size-exclusion chromatography. The authors confirmed the morphological features (30 to 100 nm in diameter with biconcave shape) and the marker expressions (HSP90, Flotilin-1, CD63) of MSC derived exosomes. In their study, MSC-derived exosomes inhibited the expression of pro-inflammatory mediators such as monocyte chemoattractant protein-1 and hypoxia-induced mitogenic factor after hypoxia, suppressed vascular remodeling, and thus prevented pulmonary hypertension. Signal transducer and activator of transcription 3 (STAT3) was activated following hypoxia in the lung, which was suppressed by the MSC-derived exosomes. Furthermore, the MSC-derived exosomes increased the levels of miR-204, a key microRNA which is decreased in human pulmonary hypertension. STAT3 activation is associated with respiratory epithelial inflammation and pro-proliferative miR-17 induction. In addition, STAT3 and miR-204 are in the STAT3-miR-204-STAT3 feed-forward loop. Therefore, MSC-derived exosomes can alleviate pulmonary hypertension by suppressing STAT3 activation and interfering with the STAT3-miR-204-STAT3 feed-forward loop.
More recently, Willis et al.59 demonstrated the role of MSC-derived exosomes in a model of bronchopulmonary dysplasia. The authors also used MSC-derived exosomes isolated by flotation on an iodixanol cushion (30– 150 nm in diameter, expressing CD9, CD63, and flotilin-1). Treatment with MSC-derived exosomes in newborn mice significantly improved lung architecture injured by hyperoxia. Additionally, MSC-derived exosomes improved pulmonary function and pulmonary hypertension. In lung transcriptome analysis, hyperoxia up-regulated genes related to the adaptive immune response, inflammatory response, and leukocyte mediated immunity. MSC-derived exosomes treatment blunted the hyperoxic up-regulation of genes related to inflammation, adaptive immune responses, IFN-γ mediated-signaling, granulocyte production, and cytokine production. This study confirmed that MSC-derived exosomes regulated macrophage phenotype. In in vitro analysis with bone marrow derived macrophages, stimulation of classically activated (M1) macrophages with MSC-derived exosomes suppressed the levels of TNFα, IL-6, and CCL5. Addition of MSC-derived exosomes to alternatively activated (M2) macrophages enhanced Arg-1 expression, but suppressed CD206 induction. In experiments assessing lung macrophage in vivo, MSC-derived exosomes suppressed the hyperoxic induction of CD40 (M1 phenotype marker) and CD206 (M2 phenotype marker).
CONCLUSIONS
Besides MSCs, EVs released from human MSCs have therapeutic properties in various pre-clinical models of ALI. The therapeutic potency of MSC-derived EVs can be equivalent to those of MSC, and the mechanism of action of MSC may be also be recapitulated in MSC-derived EVs. MSC-derived EVs can be an attractive alternative to MSCs when considering the risk of iatrogenic tumor formation or of pulmonary embolism with IV administration of MSC. However, many questions will need to be addressed prior to any serious translation to clinical trials: 1) Standardized techniques to characterize and quantify EVs; 2) Appropriate assessments for potency to compare between MSC and MSC-derived EVs; 3) Whether MSC derived exosomes, microvesicles or both are more effective; and 4) Cost-effectiveness between the productions for MSC and MSC-derived EVs. Regardless, cell-based therapy with EVs has become a legitimate alternative to using live cells and should be studied further.
Acknowledgement:
We thank Drs. Airan Liu and Qi Hao for editing of this review.
Funding Source: Grant HL-113022 from NHLBI (Dr. Jae-Woo Lee)
Footnotes
Conflicts of Interests: None.
REFERENCES
- 1.Rubenfeld GD, Caldwell E, Peabody E, et al. Incidence and outcomes of acute lung injury. N Eng J Med 2005;353:1685–93. [DOI] [PubMed] [Google Scholar]
- 2.Li G, Malinchoc M, Cartin-Ceba R, et al. Eight-year trend of acute respiratory distress syndrome: a population-based study in Olmsted County, Minnesota. Am J Resp Crit Care Med 2011;183:59–66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Bernard GR, Artigas A, Brigham KL, et al. The american-european consensus conference on ARDS. Definitions, mechanisms, relevant outcomes, and clinical trial coordination. Am J Resp Crit Care Med 1994;149:818–24. [DOI] [PubMed] [Google Scholar]
- 4.Ware LB, Matthay MA. The acute respiratory distress syndrome. N Eng J Med 2000;342:1334–49. [DOI] [PubMed] [Google Scholar]
- 5.Silversides JA, Ferguson ND. Clinical review: Acute respiratory distress syndrome - clinical ventilator management and adjunct therapy. Crit Care 2013;17:225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Brower RG, Matthay MA, Morris A, et al. Ventilation with lower tidal volumes as compared with traditional tidal volumes for acute lung injury and the acute respiratory distress syndrome. N Eng J Med 2000;342:1301–8. [DOI] [PubMed] [Google Scholar]
- 7.Wiedemann HP, Wheeler AP, Bernard GR, et al. Comparison of two fluid-management strategies in acute lung injury. N Eng J Med 2006;354:2564–75. [DOI] [PubMed] [Google Scholar]
- 8.Hu SL, He HL, Pan C, et al. The effect of prone positioning on mortality in patients with acute respiratory distress syndrome: a meta-analysis of randomized controlled trials. Crit Care 2014;18:R109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Friedenstein AJ, Petrakova KV, Kurolesova AI, et al. Heterotopic of bone marrow. Analysis of precursor cells for osteogenic and hematopoietic tissues. Transplantation 1968;6:230–47. [PubMed] [Google Scholar]
- 10.Dominici M, Le Blanc K, Mueller I, et al. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy 2006;8:315–7. [DOI] [PubMed] [Google Scholar]
- 11.Lange C, Tögel F, Ittrich H, et al. Administered mesenchymal stem cells enhance recovery from ischemia/reperfusion-induced acute renal failure in rats. Kidney Int 2005;68:1613–7. [DOI] [PubMed] [Google Scholar]
- 12.Tögel F, Hu Z, Weiss K, et al. Administered mesenchymal stem cells protect against ischemic acute renal failure through differentiation-independent mechanisms. Am J Physiol Renal Physiol 2005;289:F31–F42. [DOI] [PubMed] [Google Scholar]
- 13.Gnecchi M, Zhang Z, Ni A, et al. Paracrine mechanisms in adult stem cell signaling and therapy. Circ Res 2008;103:1204–19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Phinney DG, Prockop DJ. Concise review: mesenchymal stem/multipotent stromal cells: the state of transdifferentiation and modes of tissue repair--current views. Stem Cells 2007;25:2896–902. [DOI] [PubMed] [Google Scholar]
- 15.Tögel FE, Westenfelder C. Mesenchymal stem cells: a new therapeutic tool for AKI. Nat Rev Nephrol 2010;6:179–83. [DOI] [PubMed] [Google Scholar]
- 16.Lee JW, Fang X, Krasnodembskaya A, et al. Concise review: Mesenchymal stem cells for acute lung injury: role of paracrine soluble factors. Stem Cells 2011;29:913–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Biancone L, Bruno S, Deregibus MC, et al. Therapeutic potential of mesenchymal stem cell-derived microvesicles. Nephrol Dial Transplant 2012;27:3037–42. [DOI] [PubMed] [Google Scholar]
- 18.Bruno S, Grange C, Collino F, et al. Microvesicles derived from mesenchymal stem cells enhance survival in a lethal model of acute kidney injury. PLoS One 2012;7:e33115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Bruno S, Grange C, Deregibus MC, et al. Mesenchymal stem cell-derived microvesicles protect against acute tubular injury. J Am Soc Nephrol 2009;20:1053–67. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Lee C, Mitsialis SA, Aslam M, et al. Exosomes mediate the cytoprotective action of mesenchymal stromal cells on hypoxia-induced pulmonary hypertension. Circulation 2012;126:2601–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Zhu Y, Feng X, Abbott J, et al. Human mesenchymal stem cell microvesicles for treatment of Escherichia coli endotoxin-induced acute lung injury in mice. Stem Cells 2014;32:116–25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Camussi G, Deregibus MC, Bruno S, et al. Exosomes/microvesicles as a mechanism of cell-to-cell communication. Kidney Int 2010;78:838–48. [DOI] [PubMed] [Google Scholar]
- 23.Simpson RJ, Lim JW, Moritz RL, et al. Exosomes: proteomic insights and diagnostic potential. Expert Rev Proteomics 2009;6:267–83. [DOI] [PubMed] [Google Scholar]
- 24.Rak J, Guha A. Extracellular vesicles--vehicles that spread cancer genes. BioEssays 2012;34:489–97. [DOI] [PubMed] [Google Scholar]
- 25.Tauro BJ, Mathias RA, Greening DW, et al. Oncogenic H-ras reprograms Madin-Darby canine kidney (MDCK) cell-derived exosomal proteins following epithelial-mesenchymal transition. Mol Cell Proteomics 2013;12:2148–59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Colombo M, Raposo G, Théry C. Biogenesis, Secretion, and Intercellular Interactions of Exosomes and Other Extracellular Vesicles. Annu Rev Cell Dev Biol 2014;30:255–89. [DOI] [PubMed] [Google Scholar]
- 27.Christianson HC, Svensson KJ, Belting M. Exosome and microvesicle mediated phene transfer in mammalian cells. Semin Cancer Biol 2014;28:31–8. [DOI] [PubMed] [Google Scholar]
- 28.Rajendran L, Bali J, Barr M, et al. Emerging roles of extracellular vesicles in the nervous system. J Neurosci 2014;34:15482–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.De Toro J, Herschlik L, Waldner C, et al. Emerging roles of exosomes in normal and pathological conditions: new insights for diagnosis and therapeutic applications. Front Immunol 2015;6:203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Théry C, Ostrowski M, Segura E. Membrane vesicles as conveyors of immune responses. Nat Rev Immunol 2009;9:581–93. [DOI] [PubMed] [Google Scholar]
- 31.El Andaloussi S, Mäger I, Breakefield XO, et al. Extracellular vesicles: biology and emerging therapeutic opportunities. Nat Rev Drug Discov 2013;12:347–57. [DOI] [PubMed] [Google Scholar]
- 32.Yáñez-Mó M, Siljander PRM, Andreu Z, et al. Biological properties of extracellular vesicles and their physiological functions. J Extracell Vesicles 2015;4:27066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Cocucci E, Meldolesi J. Ectosomes and exosomes: shedding the confusion between extracellular vesicles. Trends Cell Biology 2015;25:364–72. [DOI] [PubMed] [Google Scholar]
- 34.Dubyak GR. P2X7 receptor regulation of non-classical secretion from immune effector cells. Cell Microbiol 2012;14:1697–706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Proia P, Schiera G, Mineo M, et al. Astrocytes shed extracellular vesicles that contain fibroblast growth factor-2 and vascular endothelial growth factor. Int J Mol Med 2008;21:63–7. [PubMed] [Google Scholar]
- 36.Lo Cicero A, Schiera G, Proia P, et al. Oligodendroglioma cells shed microvesicles which contain TRAIL as well as molecular chaperones and induce cell death in astrocytes. Int J Oncol 2011;39:1353–7. [DOI] [PubMed] [Google Scholar]
- 37.Monsel A, Zhu Y, Gudapati V, et al. Mesenchymal stem cell derived secretome and extracellular vesicles for acute lung injury and other inflammatory lung diseases. Expert Opin Biol Ther 2016;16:859–71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Uccelli A, de Rosbo NK. The immunomodulatory function of mesenchymal stem cells: mode of action and pathways. Ann N Y Acad Sci 2015;1351:114–26. [DOI] [PubMed] [Google Scholar]
- 39.Waterman RS, Tomchuck SL, Henkle SL, et al. A new mesenchymal stem cell (MSC) paradigm: polarization into a pro-inflammatory MSC1 or an immunosuppressive MSC2 phenotype. PLoS One 2010;5:e10088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Le Blanc K, Mougiakakos D. Multipotent mesenchymal stromal cells and the innate immune system. Nat Rev Immunol 2012;12:383–96. [DOI] [PubMed] [Google Scholar]
- 41.Duijvestein M, Wildenberg ME, Welling M, et al. Pretreatment with interferon-γ enhances the therapeutic activity of mesenchymal stromal cells in animal models of colitis. Stem Cells 2011;29:1549–58. [DOI] [PubMed] [Google Scholar]
- 42.Zhao X, Liu D, Gong W, et al. The toll-like receptor 3 ligand, poly(I:C), improves immunosuppressive function and therapeutic effect of mesenchymal stem cells on sepsis via inhibiting MiR-143. Stem Cells 2014;32:521–33. [DOI] [PubMed] [Google Scholar]
- 43.Chen H, Min X-H, Wang Q-Y, et al. Pre-activation of mesenchymal stem cells with TNF-α, IL-1β and nitric oxide enhances its paracrine effects on radiation-induced intestinal injury. Sci Rep 2015;5:8718. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 44.Fan H, Zhao G, Liu L, et al. Pre-treatment with IL-1β enhances the efficacy of MSC transplantation in DSS-induced colitis. Cell Mol Immunol 2012;9:473–81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Song Y, Dou H, Li X, et al. Exosomal miR-146a contributes to the enhanced therapeutic efficacy of interleukin-1β-primed mesenchymal stem cells against sepsis. Stem Cells 2017;35:1208–21. [DOI] [PubMed] [Google Scholar]
- 46.Monsel A, Zhu Y, Gennai S, et al. Therapeutic effects of human mesenchymal stem cell–derived microvesicles in severe pneumonia in mice. Am J Resp Crit Care Med 2015;192:324–36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Walter J, Ware LB, Matthay MA. Mesenchymal stem cells: mechanisms of potential therapeutic benefit in ARDS and sepsis. Lancet Resp Med 2014;2:1016–26. [DOI] [PubMed] [Google Scholar]
- 48.Zhu Y, Hao Q, Monsel A, Feng X, Lee J. Adult stem cells for acute lung injury: remaining questions and concerns. Respirology 2013;18:744–56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Tang X, Shi L, Monsel A, et al. Mesenchymal stem cell microvesicles attenuate acute lung injury in mice partly mediated by Ang-1 mRNA. Stem Cells 2017;35:1849–59. [DOI] [PubMed] [Google Scholar]
- 50.Morrison TJ, Jackson MV, Cunningham EK, et al. Mesenchymal stromal cells modulate macrophages in clinically relevant lung injury models by extracellular vesicle mitochondrial transfer. Am J Resp Crit Care Med 2017;196:1275–86. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Phinney DG, Di Giuseppe M, Njah J, et al. Mesenchymal stem cells use extracellular vesicles to outsource mitophagy and shuttle microRNAs. Nat Commun 2015;6:8472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Islam MN, Das SR, Emin MT, et al. Mitochondrial transfer from bone-marrow-derived stromal cells to pulmonary alveoli protects against acute lung injury. Nat Med 2012;18:759–65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Jackson MV, Morrison TJ, Doherty DF, et al. Mitochondrial transfer via tunneling nanotubes is an important mechanism by which mesenchymal stem cells enhance macrophage phagocytosis in the in vitro and in vivo models of ARDS. Stem Cells 2016;34:2210–23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Noble PW, Liang J, Jiang D. Hyaluronan as an immune regulator in human diseases. Physiol Rev 2011;91:221–64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Zhu H, Mitsuhashi N, Klein A, et al. The role of the hyaluronan receptor CD44 in mesenchymal stem cell migration in the extracelular matrix. Stem Cells 2006;24:928–35. [DOI] [PubMed] [Google Scholar]
- 56.Gennai S, Monsel A, Hao Q, et al. Microvesicles derived from human mesenchymal stem cells restore alveolar fluid clearance in human lungs rejected for transplantation. Am J Transplant 2015;15:2404–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.McAuley DF, Curley GF, Hamid UI, et al. Clinical grade allogeneic human mesenchymal stem cells restore alveolar fluid clearance in human lungs rejected for transplantation. Am J Physiol Lung Cell Mol Physiol 2014;306:L809–15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Cruz F, Borg ZD, Goodwin M, et al. Systemic administration of human bone marrow-derived mesenchymal stromal cell extracellular vesicles ameliorates Aspergillus hyphal extract-induced allergic airway inflammation in immunocompetent mice. Stem Cells Transl Med 2015;4:1302–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Willis DGR, Fernandez-Gonzalez DA, Anastas DJ, et al. Mesenchymal stromal cell exosomes ameliorate experimental bronchopulmonary dysplasia and restore lung function through macrophage immunomodulation. Am J Resp Crit Care Med 2018;197:104–16. [DOI] [PMC free article] [PubMed] [Google Scholar]