Vamorolone is a first-in-class dissociative drug that selectively targets the glucocorticoid receptor to safely treat chronic inflammation and the mineralocorticoid receptor to treat cardiomyopathy, providing efficacy with improved safety in mouse models of Duchenne muscular dystrophy.
Abstract
Cardiomyopathy is a leading cause of death for Duchenne muscular dystrophy. Here, we find that the mineralocorticoid receptor (MR) and glucocorticoid receptor (GR) can share common ligands but play distinct roles in dystrophic heart and skeletal muscle pathophysiology. Comparisons of their ligand structures indicate that the Δ9,11 modification of the first-in-class drug vamorolone enables it to avoid interaction with a conserved receptor residue (N770/N564), which would otherwise activate transcription factor properties of both receptors. Reporter assays show that vamorolone and eplerenone are MR antagonists, whereas prednisolone is an MR agonist. Macrophages, cardiomyocytes, and CRISPR knockout myoblasts show vamorolone is also a dissociative GR ligand that inhibits inflammation with improved safety over prednisone and GR-specific deflazacort. In mice, hyperaldosteronism activates MR-driven hypertension and kidney phenotypes. We find that genetic dystrophin loss provides a second hit for MR-mediated cardiomyopathy in Duchenne muscular dystrophy model mice, as aldosterone worsens fibrosis, mass and dysfunction phenotypes. Vamorolone successfully prevents MR-activated phenotypes, whereas prednisolone activates negative MR and GR effects. In conclusion, vamorolone targets dual nuclear receptors to treat inflammation and cardiomyopathy with improved safety.
Introduction
Duchenne muscular dystrophy (DMD) is a progressive X-linked disease characterized by muscle degeneration, chronic inflammation, loss of ambulation, and heart failure in the later stages. It is caused by deletion or loss-of-function mutations of the dystrophin gene (Monaco et al, 1986; Hoffman et al, 1987; Koenig et al, 1987). Elevated inflammatory NF-κB signaling is present in infants with DMD, with onset of muscle weakness in early childhood and diagnosis typically made around 5–7 yr of age (Chen et al, 2005). As patients grow older, cardiorespiratory disease develops, and cardiomyopathy is becoming a leading cause of morbidity and mortality (Nigro et al, 1990). Prednisone, an agonist of the glucocorticoid receptor (GR; gene NR3C1), is prescribed as a potent anti-inflammatory drug and considered standard of care for DMD (Griggs et al, 2013, 2016; Bello et al, 2015a). Chronic treatment with glucocorticoids leads to a broad range of side effects that detract from patient quality of life, including bone fragility, stunted growth, weight gain, behavior issues, cataracts, and adrenal suppression (Bello et al, 2015a). These negative side effects lead to delay in the age of treatment, poor compliance, and variation in clinical practice (Griggs et al, 2013; Bello et al, 2015a). Deflazacort was recently approved by the Food and Drug Administration (FDA) in efforts to provide a safer alternative to prednisone, after years of use in international and non-DMD populations. However, the approval of deflazacort has become controversial because of large price increases (highlighted by Senator Bernie Sanders in Sanders and Cummings [2017]), along with new reports suggesting it may have increased side effects compared with prednisone (Bello et al, 2015a).
Cardiomyopathy in early adulthood is a leading cause of death for DMD, responsible for 20–50% of mortalities in dystrophin-deficient patients (Eagle et al, 2002; Finsterer & Stollberger, 2003). The earlier, preclinical stages of DMD cardiomyopathy are characterized by normal heart systolic function with developing fibrosis (Hor et al, 2013). DMD myocardial fibrosis is progressive, ultimately leading to dilated cardiomyopathy and systolic dysfunction. Improved clinical management through anti-inflammatory glucocorticoid treatment and respiratory support has substantially improved survival and heart function in patients (Eagle et al, 2002; Silversides et al, 2003; Peterson et al, 2018). Recently, a different class of steroids demonstrated potential for treating DMD cardiomyopathy. These are antagonists of the mineralocorticoid receptor (MR; gene NR3C2). MR antagonists such as eplerenone and spironolactone have been used for the treatment of various forms of heart failure and hypertension for decades, and eplerenone is now shown to slow the progression of heart disease in DMD (Raman et al, 2015, 2017).
The GR and MR evolved from a shared ancestral receptor and show ligand cross-reactivity. The GR regulates inflammation and metabolism, whereas the MR regulates fluid and salt levels. The physiological GR hormone cortisol binds both receptors and circulates at higher levels than the MR hormone, aldosterone. To ensure MR signaling specificity, key aldosterone target tissues such as the kidney express the glucocorticoid-inactivating enzyme 11β-hydroxysteroid dehydrogenase 2 (HSD11B2) (Edwards et al, 1988). This enzyme is specifically expressed in epithelial tissues and metabolizes excess glucocorticoids (cortisol and prednisolone) into their inactive forms (cortisone and prednisone) (Funder et al, 1988). In non-epithelial tissues, where this steroid-inactivating enzyme is not expressed (HSD11B2-negative), glucocorticoids can be MR ligands (Edwards et al, 1988; Iqbal et al, 2014). A key non-epithelial MR target tissue in DMD is the heart (Young & Rickard, 2015; Cole & Young, 2017). It is now recognized that MR signaling can impact cardiomyopathy by acting locally, as cardiomyocyte and myeloid MRs promote fibrosis, inflammation, and dysfunction (Young & Rickard, 2015). Increases in MR activity can increase heart mass and promote the progression of pathophysiology in response to additional insults such as high salt or infarction (Brilla & Weber, 1992; Peters et al, 2009; Fraccarollo et al, 2011). Therefore, it would be beneficial to limit the MR cross-reactivity of agonists in HSD11B2-negative tissues such as the heart.
Vamorolone was recently developed as a dissociative GR ligand that provides anti-inflammatory efficacy with improved safety (Heier et al, 2013; Fiorillo et al, 2018; Hoffman et al, 2018). Vamorolone also binds to the MR and has a Δ9,11 structure which avoids enzymes that metabolize cortisol (such as HSD11B2). Here, we investigate the mechanisms of action for vamorolone using studies of MR ligand chemistry, MR reporter assays, and CRISPR knockout myoblasts. We also investigate the effects of MR signaling on dystrophin-deficient hearts using mdx mouse models. We report the sensitivity of dystrophin-deficient mdx hearts to MR activity, the efficacy of vamorolone as an MR antagonist, and the improved safety of vamorolone versus prednisolone. Our data provide new insights into steroid mechanisms of action, elucidate the molecular pathogenesis of dystrophic cardiomyopathy, and identify vamorolone as a first-in-class drug that targets dual receptors to treat both inflammation and heart failure pathways.
Results
Comparison of steroid ligand chemistries
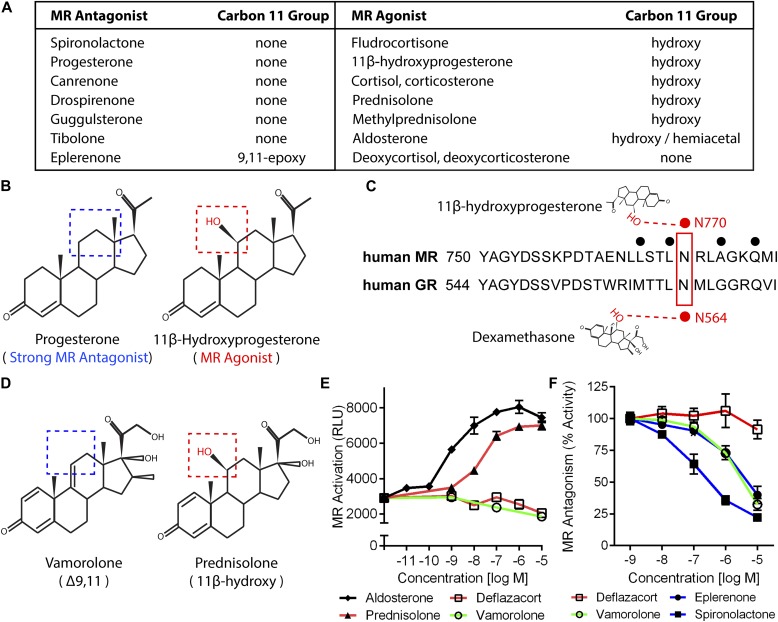
We began to investigate the consequences of MR-binding by the Δ9,11 compound vamorolone by performing in silico studies of the relationships between MR ligand structures, activities, and receptor interactions. By comparing structures of 14 physiological and pharmacological ligands, we found that an 11β-hydroxy group was only present on MR agonists (Fig 1A). Focusing on a pair of ligands with contrasting effects but similar structures, we found that 11β-hydroxy was the only structural distinction between a potent MR antagonist (progesterone) and MR agonist (11β-hydroxyprogesterone) (Fig 1B). We next queried available X-ray and structural data on ligands bound to their receptors to identify relevant moiety–residue interactions. The structural data showed that the 11β-hydroxy group of 11β-hydroxyprogesterone interacts with MR residue N770 (Fig 1C) through hydrogen bonding (Rafestin-Oblin et al, 2002). Because this residue is conserved between the MR and GR, we next queried whether a conserved interaction also existed between the GR and its ligands. Indeed, the 11β-hydroxy group of dexamethasone has been found to interact with this conserved residue on the GR (N564) through hydrogen bonding (Bledsoe et al, 2002; Hammer et al, 2003; Lind et al, 2000). Supporting its importance in modulating activity, disruption of this conserved interaction by MR or GR mutation (N770A or N564A, respectively) has been shown to maintain ligand binding but disrupt the transcription factor activity of that receptor (Hammer et al, 2003; Rafestin-Oblin et al, 2002). Together, this information indicated that 11β-hydroxysteroids can activate or enhance MR transcription factor functions through interaction with N770. Comparison of vamorolone and prednisolone structures (Fig 1D) provided a situation analogous to that of progesterone and 11β-hydroxyprogesterone, where the key structural difference is the 11β-hydroxy group (Hoffman et al, 2018). Based on these comparisons, vamorolone was anticipated to function as an antagonist of the MR, in direct contrast to prednisolone.
Figure 1. Vamorolone and MR antagonists lack 11-β hydroxyl groups linked to MR activation.
(A) Table of pharmacological and physiological MR ligands with their carbon 11 group identity provided. (B) Progesterone is a potent MR antagonist, whereas addition of an 11β-hydroxy (11β-Hydroxyprogesterone) results in an agonist compound. (C) The 11β-hydroxy group of hydroxyprogesterone interacts with MR residue N770 via hydrogen bonding. Dexamethasone also interacts with this conserved residue in the GR (N564) via hydrogen bonding. (D) Vamorolone is a Δ9,11 steroid where the 11β position features a carbon–carbon double bond, whereas prednisolone is an 11β-hydroxysteroid. (E) A stable MR reporter cell line was treated with drugs and quantified via chemiluminescence assay to determine their agonist properties. Prednisolone and aldosterone showed MR agonist activity. (F) Reporter cells were treated with drug in combination with a constant E80 dose of aldosterone to determine antagonist properties. Vamorolone acted as an MR antagonist, consistent with eplerenone and spironolactone. (Representative data from three experiments with each dose performed in triplicate; values are mean ± SEM).
Identification of vamorolone as an MR antagonist
We next compared the activities of vamorolone, glucocorticoids (prednisolone, deflazacort), aldosterone, and MR antagonists (eplerenone, spironolactone) on receptor activity using MR reporter cell lines (Yang et al, 2015). Testing for MR agonist activity, aldosterone showed a dose-dependent increase in MR reporter activity beginning at a concentration of 1 nM (Fig 1E). Prednisolone also caused a strong dose-dependent activation of the MR, beginning at 10 nM. In contrast, both deflazacort and vamorolone showed no MR agonist activity.
Next, we tested each drug's ability to act as an antagonist that competitively inhibits MR activity. MR reporter cells were pretreated with each drug for 1 h, and MR activity was induced by adding aldosterone (Fig 1F). Vamorolone, eplerenone, and spironolactone inhibited MR activity in a dose-dependent manner. The amount of MR inhibition shown by vamorolone closely resembled that of eplerenone in this in vitro system. Deflazacort showed no inhibition of MR activity. In addition to deflazacort, we also tested the active metabolite 21-desacetyl-deflazacort and found no agonist or antagonist activity (Fig S1). Together, these data indicate that clinical GR ligands differ in their effects on MR activity, and that the GR ligand vamorolone acts as an MR antagonist with potency similar to proven pharmacological antagonists.
Figure S1. An active metabolite of deflazacort shows no activity in MR reporter assays.
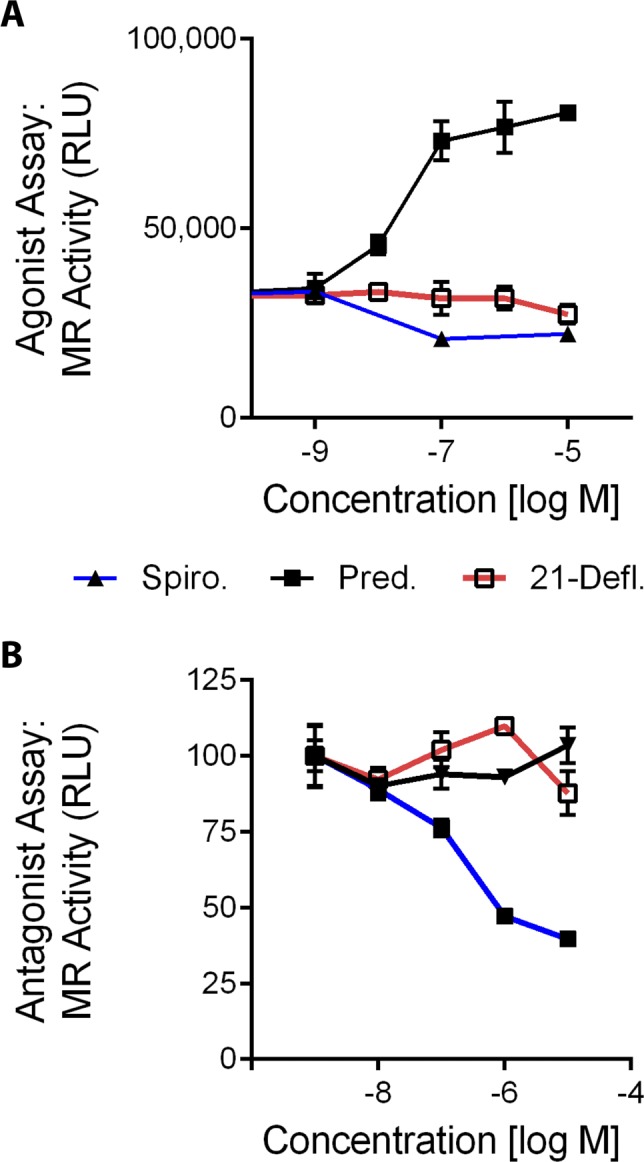
To determine if an active metabolite of deflazacort showed a lack of MR activity consistent with deflazacort, stable MR reporter cells were treated with 21-desacetyl deflazacort, prednisolone, or spironolactone. Used as controls here, prednisolone activated the MR as an agonist, and spironolactone acted as an antagonist to inhibit MR induction by aldosterone. Consistent with deflazacort, we found that the metabolite 21-desacetyl deflazacort avoided MR interaction. (A) Agonist activity was assayed by treating cells with each drug alone in increasing concentrations. (B) Antagonist activity was assayed by pretreating cells with each drug, then adding aldosterone (natural MR agonist) and assaying MR activity 6 h later. (Pred = Prednisolone, 21-Defl = 21-Desacetyl deflazacort, Spiro = Spironolactone).
Knockout shows anti-inflammatory activities of ligands are mediated via GR
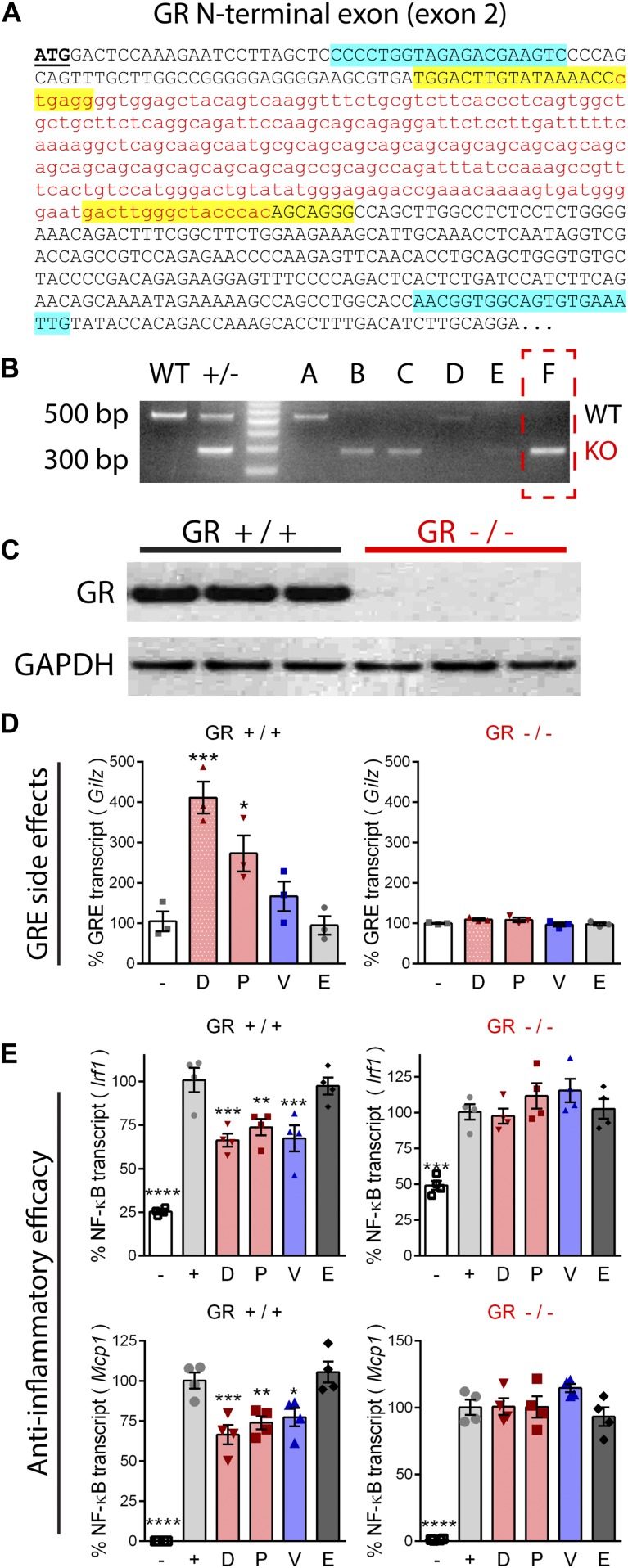
MR and GR ligands were tested for the ability to induce GR transactivation side effects, and to show efficacy at inhibiting inflammatory signaling in myogenic cell lines. CRISPR/Cas9 was used to create a GR knockout cell line in C2C12 myoblasts via an N-terminal exon deletion leading to a frameshift (loss of function). After transfection and single-cell clonal propagation, positive clones were identified by PCR and deletions defined by sequencing (Fig 2A–B). Successful elimination of GR protein expression was confirmed by Western blot (Fig 2C).
Figure 2. CRISPR knockout of the GR shows that anti-inflammatory pathways are GR-dependent.
A frame-shifting deletion mutation was introduced via CRISPR in C2C12 myoblasts to knockout GR expression. (A) The DNA sequence of the GR N-terminus is provided. Base pairs deleted in knockout cells are shown as red, lower case letters. Positions of CRISPR guide RNAs are highlighted in yellow, and genotyping primers in blue. (B) PCR of C2C12 myoblast clones (labeled A through F) identified WT (∼500 bp band) and GR deletion clones (∼300 bp band). (C) Western blot confirmed GR knockout in clone from PCR lane F. (D) Drug-treated wild-type and GR knockout myoblasts were assayed for activation of a GR transactivation target transcript (Gilz). (E) Wild-type and GR knockout myotubes were treated with drug, induced with TNF, and assayed for expression of inflammatory genes regulated by NF-κB (Irf1 and Mcp1). (n = 3–4, *P < 0.05, **P < 0.005, ***P < 0.001, ****P < 0.0001, ANOVA with post hoc versus vehicle [panel D] or + TNF [panel E] control; representative of three experiments; panels [D, E]: [−] = no drug or TNF, [+] = TNF plus vehicle, D = deflazacort, P = prednisolone, V = vamorolone, E = eplerenone).
GR-positive and knockout cells were treated with drugs at equimolar concentrations to determine their transactivation effects on genes directly regulated by a GR-bound promoter element (GRE). This property is associated with negative side effects of current glucocorticoids. For this, we assayed glucocorticoid-induced leucine zipper (Gilz; gene Tsc22d3). In wild-type cells, both prednisolone (P < 0.05) and deflazacort (P < 0.001) caused significant activation of Gilz expression (Fig 2D). Vamorolone and eplerenone both showed no significant effects on GR target gene expression. In contrast, and confirming loss of receptor function, GR knockout cells showed no activation of GRE-controlled Gilz gene expression by any of the drugs. These data are consistent with the activation of GRE-transactivation side effects by prednisone and deflazacort, but not by vamorolone.
To test for efficacy at inhibiting inflammatory NF-κB signaling, differentiated myotubes were pretreated with drug and then induced with TNF. Inflammatory gene expression was assayed by quantitative reverse transcriptase polymerase chain reaction (qRT-PCR) of Interferon regulatory factor 1 (Irf1) and Monocyte chemoattractant protein 1 (Mcp1; gene Ccl2). These encode an inflammatory transcription factor and a cytokine, respectively, expressed from gene promoters that are directly regulated by NF-κB (Ueda et al, 1994; Robinson et al, 2006). TNF caused a significant increase (P < 0.0005) of Irf1 and Mcp1 (Fig 2E). Vamorolone, prednisolone, and deflazacort all showed significant anti-inflammatory effects by reducing Irf1 (P < 0.005) and Mcp1 (P < 0.05) in GR-positive cells (Fig 2E). In contrast, the MR-specific drug eplerenone did not show anti-inflammatory effects on Irf1 or Mcp1. In GR knockout cells, no anti-inflammatory effects were seen by any of the drugs, confirming that the GR is specifically responsible for anti-inflammatory effects of vamorolone, prednisolone, and deflazacort. Together, these data indicate that vamorolone alone shows a combined profile of GR-dependent anti-inflammatory effects, selective avoidance of GR side effect pathways, and behavior as an MR antagonist.
Vamorolone has anti-inflammatory effects in muscle, immune, and heart cells
Our previous experiment demonstrated that GR ligands have anti-inflammatory effects in differentiated myotubes relevant to skeletal muscle pathophysiology in DMD. We next tested drugs for anti-inflammatory efficacy in immune and heart cells, both of which can impact DMD cardiomyopathy. For the first set of experiments, RAW 264.7 macrophages were induced with lipopolysaccharide (LPS) after pretreatment with a GR and/or MR ligand. Analysis by qRT-PCR showed LPS caused a significant increase (P < 0.0001) in Irf1 and Mcp1, consistent with data in myotubes (Fig S2). LPS also caused a significant increase (P < 0.0001) in Interleukin 1β (Il1b) and Interleukin 6 (Il6) (Fig 3A). These two cytokines are directly regulated by NF-κB and their chronic overexpression contributes to heart pathophysiology (Libermann & Baltimore, 1990; Hiscott et al, 1993; Wollert & Drexler, 2001; Bujak & Frangogiannis, 2009). Vamorolone, deflazacort, and prednisolone all showed a significant inhibition (P < 0.005) of Irf1, Mcp1, Il1b, and Il6 induction (Figs 3A and S2). The MR-specific drug eplerenone, in contrast, showed no effects on the expression of any of these inflammatory genes. To see if these transcriptional effects were consistent with the levels of cytokines secreted by macrophages, we assayed IL1B and IL6 protein levels in media from the same experiment using an AlphaLISA assay (Fig 3B). The results were consistent with qRT-PCR, showing a potent induction of both secreted IL1B and IL6 with LPS that was significantly attenuated by vamorolone, prednisolone, and deflazacort (P < 0.0001 for each), but not by eplerenone.
Figure S2. Vamorolone inhibits inflammatory signaling in macrophage cells.

RAW 264.7 macrophages were pretreated with 10 μM of GR and/or MR ligands and induced with LPS. Expression of NF-κB regulated inflammatory genes (Irf1 and Mcp1) was assayed by qRT-PCR. (n = 4, **P < 0.01, ***P < 0.005, ****P < 0.0001, ANOVA with post hoc versus [+] TNF control in gray; [−] = uninduced, [+] = TNF plus vehicle, D = deflazacort, P = prednisolone, V = vamorolone, E = eplerenone).
Figure 3. Vamorolone inhibits inflammatory signaling in macrophage and heart cells.
(A) RAW 264.7 macrophages were pretreated with drug at 10 μM and inflammatory signaling was induced for 24 h using LPS. Expression of NF-κB–regulated inflammatory genes (Il1b and Il6) was assayed by qRT-PCR. (B) IL1B and IL6 protein levels were assayed in media from the same experiment via AlphaLISA assay. (C) Primary cardiomyocytes were pretreated with vehicle, vamorolone, or the GR-specific ligand deflazacort, and inflammatory signaling induced with TNF. NF-κB–regulated inflammatory gene expression (Il1b and Il6) was assayed by qRT-PCR. (D) IL6 protein levels were assayed by AlphaLISA. (E) HL-1cells were pretreated with 10 μM drug and induced with TNF for 24 h. Expression of Il6 was assayed by qRT-PCR. (n = 4, **P < 0.005, ****P < 0.0001, ANOVA with post hoc versus [+] TNF control in gray; [−] = no TNF control, [+] = TNF plus vehicle, D = deflazacort, P = prednisolone, V = vamorolone, E = eplerenone).
Next, we tested the ability of GR ligands to inhibit inflammatory signaling in heart cells. We first performed an experiment using primary cardiomyocytes obtained from postnatal day 2 wild-type mice and treated with either vamorolone or the GR-specific ligand deflazacort. Primary cardiomyocytes displayed spontaneous contractions in culture, characteristic of a heart phenotype, before and throughout treatment. TNF induction caused a significant increase (P < 0.0001) in Il1b and Il6 gene expression (Fig 3C). Administration of vamorolone and deflazacort significantly dampened induction of these genes (P < 0.005). Analysis of IL6 protein levels by AlphaLISA showed consistent results, as TNF caused an increase in IL6 and this was effectively inhibited (P < 0.0001) by both vamorolone and deflazacort (Fig 3D). Next, we repeated drug treatments using HL-1 cells. HL-1 cells are an immortalized cardiac muscle cell line that displays phenotypic characteristics consistent with adult atrial cardiomyocytes (Claycomb et al, 1998). Again, in HL-1 cells TNF increased Il6 expression (P < 0.0001) and this was successfully decreased (P < 0.005) by vamorolone and deflazacort (Fig 3E). Together, our in vitro data indicate that vamorolone, prednisolone, and deflazacort all possess a GR-dependent ability to inhibit inflammatory signaling in muscle, immune, and heart cells.
Vamorolone antagonizes the MR in vivo to protect dystrophic hearts
After investigating the mechanism of vamorolone in vitro, we next tested the ability of vamorolone to act as an MR antagonist in vivo. Randomized and blinded treatment groups of wild-type and mdx mice were implanted with subcutaneous osmotic pumps that secreted either vehicle or aldosterone, the physiological MR agonist, at a calculated dose of 0.25 mg/kg/d (n ≥ 8 per group). The mdx mice receiving aldosterone via osmotic pump were also treated with vehicle, vamorolone (20 mg/kg/d), eplerenone (100 mg/kg/d), or spironolactone (20 mg/kg/d), using daily oral administration via ingestion of sugar syrup formulations. After 6 wk, heart function was assayed by echocardiography, blood pressure was measured, organ weights were obtained, and terminal endpoint measures were performed.
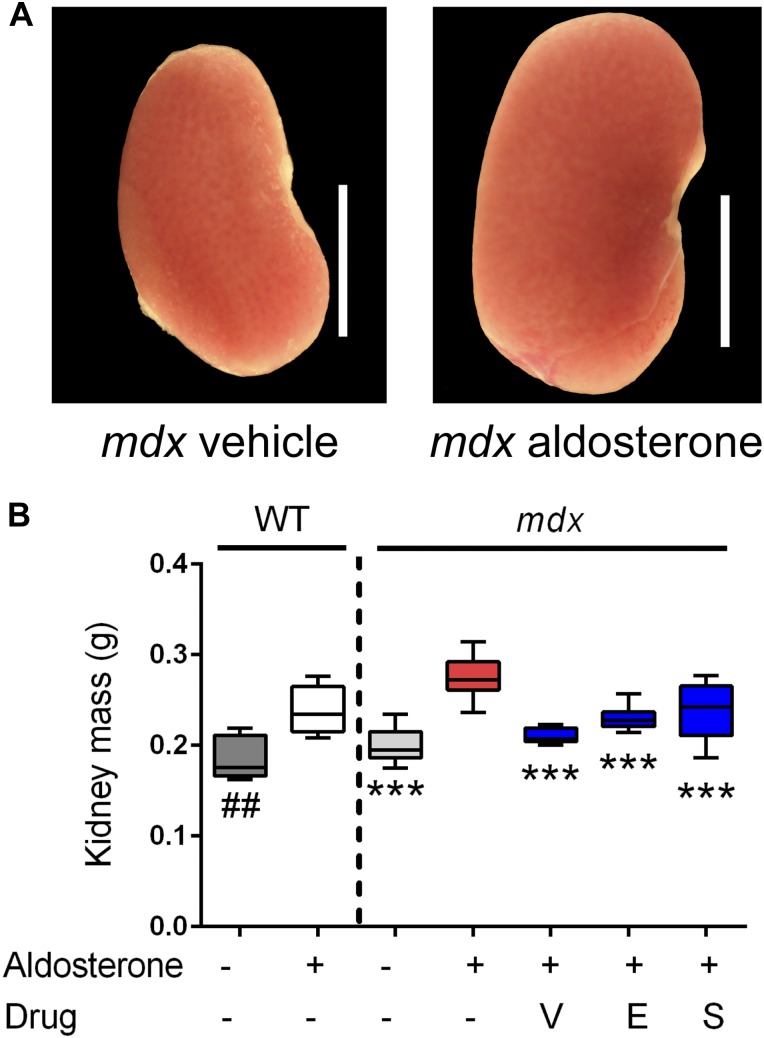
Kidney size was assayed as an aldosterone-specific effect driven by MR activation in epithelial (HSD11B2 positive) tissues. This also provided a readout of MR activity that was independent of direct effects from mdx pathophysiology. Aldosterone caused significant increases in kidney size for both wild-type (P < 0.005) and mdx (P < 0.0005) mice (Fig 4A and B). In the mdx treatment groups, vamorolone, eplerenone, and spironolactone all showed significant protective effects (P < 0.0005), maintaining normal kidney size. This indicates that vamorolone is an efficient MR antagonist in a tissue that is specifically responsive to aldosterone in vivo, consistent with our in vitro data.
Figure 4. Vamorolone is an effective MR antagonist in vivo.
Mice were treated with daily oral drug (vehicle, vamorolone, eplerenone, or spironolactone) and implanted with osmotic pumps that secreted either aldosterone or vehicle for 6 wk. Kidney size was assayed as an epithelial MR target tissue, where aldosterone-specificity of the MR is maintained through expression of the glucocorticoid-inactivating enzyme HSD11B2. (A) Representative kidney images are provided. (B) Aldosterone induced an increase in kidney mass in both wild-type and mdx. This was prevented in mdx by all three MR antagonists. (n ≥ 8 per group; ***P < 0.0005, ANOVA with post hoc test versus mdx + aldosterone in red; ##P < 0.005, t test comparing WT groups; V = Vamorolone, E = Eplerenone, S = Spironolactone).
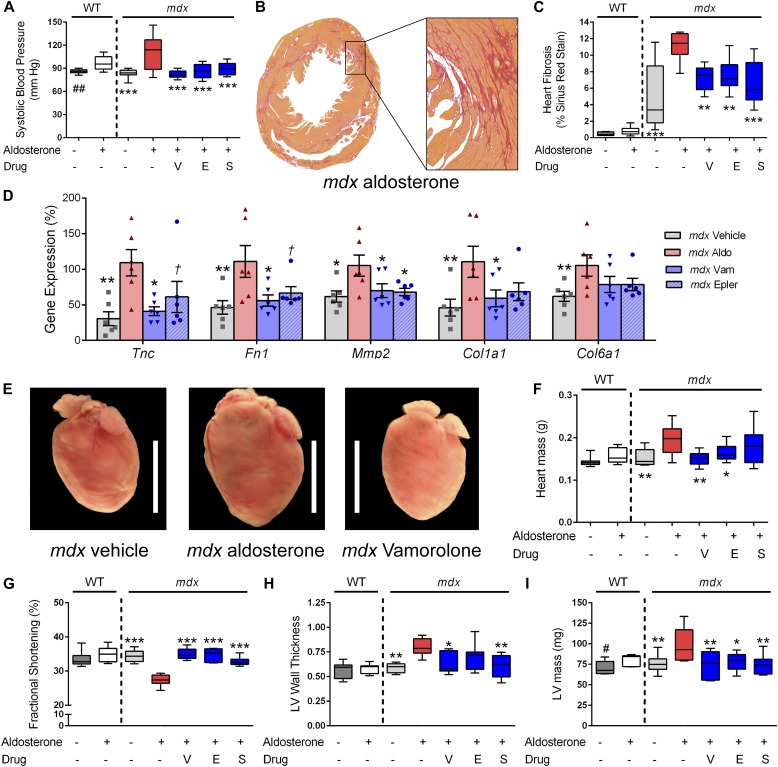
Hypertension and cardiomyopathy were assayed as non-epithelial phenotypes that are both related to cardiovascular morbidity. We found that aldosterone caused significant increases in blood pressure for both wild-type (P < 0.005) and mdx (P < 0.0005) mice (Fig 5A). In the mdx treatment groups, vamorolone caused a significant improvement in blood pressure, as did eplerenone and spironolactone (P < 0.0005).
Figure 5. Vamorolone improves MR-mediated hypertension and mdx cardiomyopathy phenotypes.
Aldosterone introduced via osmotic pumps caused an activation or progression of several phenotypes relevant to cardiomyopathy that were all improved or prevented by vamorolone, eplerenone, and spironolactone. (A) Blood pressure was measured by tail cuff, and increased by aldosterone in both wild-type and mdx mice. (B, C) Sirius Red staining of collagen deposition was used to assay heart fibrosis, where aldosterone increased heart fibrosis specifically in mdx mice in (B) representative images and upon (C) quantification. (D) qRT-PCR of mdx heart tissue was used to assay MR-activated gene expression changes relevant to fibrosis and cardiomyopathy (n = 6 per group). (E, F) Aldosterone increased mdx heart size in (E) representative images and (F) quantitative measurement of heart mass. (G–I) Heart function was assayed by echocardiography, with (G) fractional shortening (H) left ventricular wall thickness (LV wall thickness, in millimetre) and (I) left ventricular mass (LV mass) each showing aldosterone-mediated phenotypes that improved with antagonist treatment. (n ≥ 8; †P < 0.1, *P < 0.05, **P < 0.005, ***P < 0.0005, ANOVA with post hoc versus mdx + aldosterone in red; #P < 0.05, ##P < 0.005, t test comparing WT groups; V = Vamorolone, E = Eplerenone, S = Spironolactone).
In control groups that received no aldosterone, mdx hearts showed expected signs of mild cardiac pathology, including cardiac fibrosis, whereas wild-type hearts did not (Fig 5B and C). Aldosterone exposure in mdx mice exacerbated heart fibrosis, causing a significant increase of sirius red staining of collagen deposition in mdx hearts (P < 0.0005). Vamorolone, eplerenone, and spironolactone all successfully mitigated the development of fibrosis (P < 0.005) to no-aldosterone control levels. To examine MR-mediated gene expression changes associated with heart pathology, we used qRT-PCR to assay the expression of MR-regulated genes in heart tissue (Fig 5D). We found that aldosterone significantly increased (P < 0.05) expression of Tenascin C (Tnc), Fibronectin 1 (Fn1), Matrix metalloproteinase 2 (Mmp2), Collagen 1a1 (Col1a1), and Collagen 6a1 (Col6a1). Vamorolone and eplerenone both inhibited the aldosterone-mediated effects on each of these five genes.
Aldosterone exposure additionally caused a large, significant increase in mdx heart mass (∼30%, P < 0.005), whereas in WT mice, it caused a smaller increase that did not reach significance (∼10%, P = 0.15) (Fig 5E and F). Treatment of mdx mice with vamorolone or eplerenone effectively mitigated the increase in heart mass (P < 0.005 and P < 0.05, respectively). Together, these findings indicate that dystrophin-deficient hearts have increased sensitivity to damage from aldosterone and that vamorolone is effective at preventing this in vivo.
Heart function was assessed using echocardiography after 6 wk of aldosterone exposure (Fig 5G–I). As expected for this age, mdx mice did not show a clear decline in heart function as a result of their genotype, as no–aldosterone control wild type and mdx were similar in all measures. However, aldosterone caused significant changes in heart function and size which were specific to mdx mice. Fractional shortening showed a significant decrease with aldosterone (P < 0.0005) exposure (Fig 5G). This decline was successfully improved by treatment with vamorolone, eplerenone, or spironolactone (P < 0.0005). Consistent with the increase in heart mass observed in dissections, aldosterone increased left ventricular (LV) wall thickness, in addition to LV mass (Fig 5H and I). Treatment with vamorolone was successful at preventing increases in both wall thickness (P < 0.05) and LV mass (P < 0.005) toward no-aldosterone controls as well, consistent with eplerenone and spironolactone. In wild-type mice, the only effect observed for aldosterone was a smaller increase in LV mass (P < 0.05), which was similar to dissection data (though dissection differences were not significant). Together, these data indicate that MR activation causes a progression of dystrophic cardiomyopathy, whereas vamorolone prevents this progression through a mechanism of MR antagonism.
Vamorolone improves heart function with improved MR/GR safety versus prednisolone
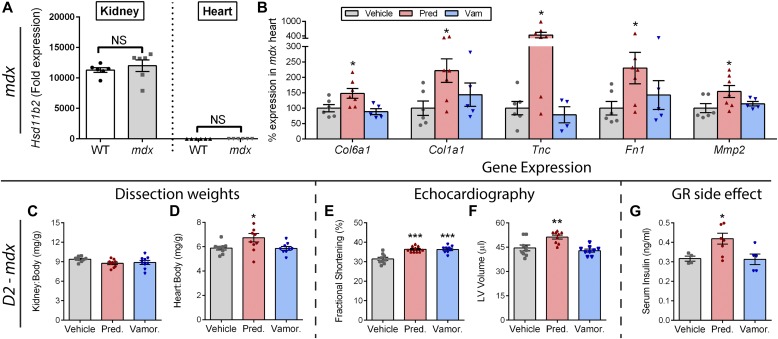
Prednisolone can act as an MR ligand in non-epithelial tissues (such as heart) that lack expression of the inactivating enzyme HSD11B2. To investigate if this occurs in mdx hearts, we first assayed expression of HSD11B2 in heart versus kidney tissue, and then determined if prednisolone activates MR-driven side effects in dystrophic hearts. For the first experiment, we used qRT-PCR to quantify expression of Hsd11b2 in the kidneys and in the hearts of both wild-type and mdx mice (Fig 6A). Consistent with the literature which indicates that HSD11B2 is not expressed in the heart, we did not detect a substantial level of Hsd11b2 expression in heart tissue from either genotype of mice. Specifically, we found a greater than 10,000-fold decrease in Hsd11b2 expression in heart tissue in comparison with kidneys (P < 0.00001). No significant effect of genotype was observed between mdx and wild-type mice for either tissue. These data suggest that kidney MR should be protected from activation by prednisolone, whereas heart MR should be sensitive to activation by prednisolone.
Figure 6. Prednisolone activates MR and GR side effect pathways in mdx and severe D2-mdx mice.
(A) Expression of the glucocorticoid-metabolizing gene Hsd11b2 was assayed by qRT-PCR in kidneys and hearts from wild-type and mdx mice. Lack of substantial expression in the heart indicated that the heart is sensitive to MR activation by prednisolone. (B) Standard mdx mice on a BL10 background were treated with vehicle, prednisolone (5 mg/kg), or vamorolone (45 mg/kg) for 4 months, beginning at 6 weeks of age. The heart was assayed for expression of MR-regulated genes via qRT-PCR and expressed as % vehicle levels (n ≥ 5). (C–G) Severe D2-mdx mice on an LTBP4 modifier background were treated for 8 weeks with daily oral vehicle, vamorolone (30 mg/kg), or prednisolone (5 mg/kg), beginning at 2 mo of age (n ≥ 9). (C, D) Kidney and heart mass at trial conclusion. Note, kidney expresses the glucocorticoid-inactivating enzyme (HSD11B2) that protects it from prednisolone, whereas the heart does not. Prednisolone increased D2-mdx heart size but not kidney size. (E, F) Echocardiography was performed near the trial endpoint. (E) Fractional shortening increased with both vamorolone and prednisolone. (F) Left ventricular volume (end diastolic) did not change with vamorolone but increased with prednisolone, consistent with premature dilatation. (G) Serum insulin levels increased with prednisolone (n ≥ 5), consistent with GR-mediated, metabolic side effects of glucocorticoids. (*P < 0.05, **P < 0.005, ***P < 0.0005; one panel B outlier removed after significant Grubbs test; (A) t test of mdx versus wild-type for each tissue, (B–G) ANOVA with post hoc versus vehicle in gray).
Following this, we obtained archival heart tissue to examine gene expression in the standard mdx model of DMD, where no aldosterone exposure was present and mice show a mild phenotype. Previously, we found that prednisolone (5 mg/kg) caused increases in heart mass and fibrosis, whereas vamorolone did not show either of these effects (Heier et al, 2013). Here, we used qRT-PCR to assay the expression of MR-regulated genes in heart tissue (Fig 6B). We found that prednisolone caused significant increases in the expression of Col6a1, Col1a1, Tnc, Fn1, and Mmp2 (P < 0.05), consistent with effects observed for aldosterone in prior experiments. Vamorolone avoided activation of each of these genes.
Next, we performed a new trial to determine the effects of daily oral prednisolone versus vamorolone on the heart in a dystrophin-deficient mouse model that exhibits a more severe heart phenotype. Specifically, a genetic modifier shown to be relevant to both human DMD and mdx mice is a polymorphism in the Latent transforming growth factor beta binding protein 4 (Ltbp4) gene (Heydemann et al, 2009; Flanigan et al, 2013; Bello et al, 2015b). In mice, the DBA/2J background harbors a deleterious Ltbp4 genotype. When crossed to the mdx dystrophin-deficient strain to produce the D2-mdx line of mice, this background significantly worsens mdx phenotypes, including an earlier onset of heart dysfunction and cardiomyopathy (Coley et al, 2016). Here, D2-mdx mice received daily oral vehicle, prednisolone (5 mg/kg), or vamorolone (30 mg/kg).
Prednisolone treatment showed no effect on kidney size (Fig 6C), but caused a significant increase (P < 0.05) in heart size (Fig 6D). This was consistent with expression of the glucocorticoid-inactivating enzyme (HSD11B2) in the kidneys, but not in the heart. No increase in kidney or heart mass was seen with vamorolone treatment. Both vamorolone and prednisolone improved cardiac function (P < 0.0005) in D2-mdx mice as measured by fractional shortening (Fig 6E). However, prednisolone also caused an increase in LV volume (P < 0.005) consistent with an increase in size and early dilatation (Fig 6F). Hyperinsulinemia is a GR-driven side effect induced in human patients treated with prednisone. It is considered a precursor and reliable biomarker for insulin resistance. As expected, prednisolone caused hyperinsulinemia (Fig 6G). Vamorolone did not induce hyperinsulinemia as a precursor of insulin resistance, which is consistent with recent human Phase 1 data where no changes in blood insulin and glucose levels were observed up through the highest dose (20 mg/kg/d) tested (Hoffman et al, 2018). Together, these data are consistent with activation of MR and GR side effects by prednisolone, whereas vamorolone shows improved safety.
Discussion
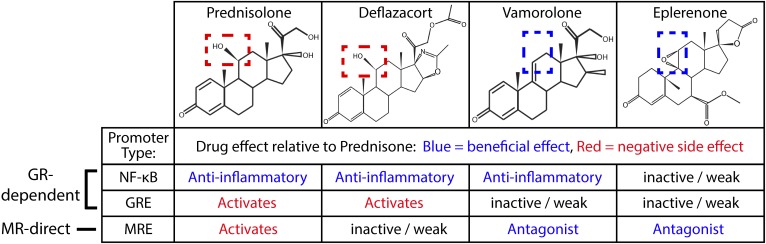
We find that vamorolone targets dual nuclear receptors in a manner that simultaneously treats cardiac disease and chronic inflammation pathways. Vamorolone is a first-in-class MR antagonist/dissociative GR ligand, with MR antagonist potency similar to eplerenone. This has the potential to benefit dystrophic cardiomyopathy, as we find that dystrophin-deficient hearts are specifically sensitive to damage mediated by activated MR. Simultaneously, vamorolone acts as a dissociative GR ligand to provide efficacy as a potent anti-inflammatory with improved safety over the current drugs prednisone and deflazacort. In contrast, eplerenone (MR antagonist) and deflazacort (GR agonist) show more receptor-specific effects, whereas prednisolone acts as a dual MR/GR agonist. A summary of ligand/receptor sub-activities is provided in (Table 1). These data inform both current DMD treatments and future drug development for a broader group of conditions impacted by steroid treatments.
Table 1.
Summary of drug sub-activity profiles on three promoter types for GR and MR ligands.
 |
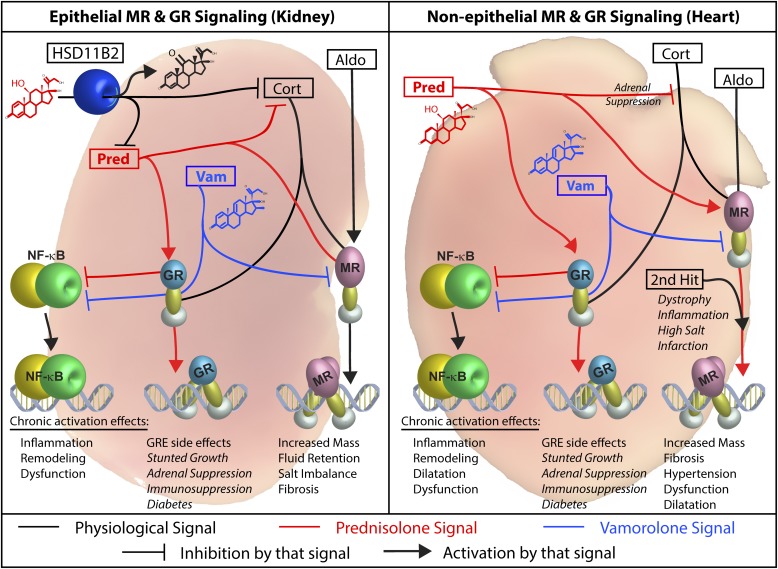
Steroids are multi-mechanistic and show particular cross-reactivity for the MR and GR. We find vamorolone and prednisolone have contrasting dual-receptor profiles; this has important consequences for heart pathophysiology and is summarized by the model in (Fig 7). The differing activity profiles of vamorolone and prednisolone are consistent with their contrasting structure (Hoffman et al, 2018) and corresponding ability to interact with a conserved residue that modulates receptor transactivation (N770/N564) (Rafestin-Oblin et al, 2002; Hammer et al, 2003). Loss of this transactivation function is likely responsible for the behavior of vamorolone as an MR antagonist, as it can competitively replace MR agonists (aldosterone and prednisolone) with a transactivation-inert ligand. Despite analogous reduction of the GR transactivation function, vamorolone is able to retain its anti-inflammatory efficacy function because it maintains inhibitory protein–protein interactions between the GR and NF-κB (Ratman et al, 2013). In contrast, the 11β-hydroxysteroid prednisolone activates all three of these functions. These findings are consistent with genomic-scale in vivo profiling of the muscle miRNAome, where prednisolone both activates and inhibits miRNA expression, whereas vamorolone exclusively inhibits miRNA expression (Fiorillo et al, 2018). By avoiding off-target transactivation while optimizing two important efficacy pathways, vamorolone can provide both a basic science tool to dissect steroid signaling and a safer therapeutic for inflammatory and heart conditions.
Figure 7. Working model of dual-receptor drug mechanisms in epithelial and non-epithelial tissues.
Left Panel: Epithelial MR target tissues (e.g., kidney, colon, skin) co-express the HSD11B2 enzyme to prevent over-activation of the MR by glucocorticoids. This protein metabolizes 11β-hydroxysteroids (cortisol and prednisolone) into their inactive ketone states (cortisone and prednisone) within that tissue. Aldosterone is not metabolized by HSD11B2, thus enabling aldosterone-specific signaling in vivo. We find that aldosterone challenge increases kidney size, whereas prednisolone treatment does not, consistent with this model. Right Panel: Non-epithelial MR target tissues express the MR but do not express the HSD11B2 enzyme (heart, brain, skin, fat, immune cells). These tissues may have increased risk of damage from MR activation by glucocorticoids. The heart is particularly important here, as (1) DMD patients naturally develop cardiomyopathy, and (2) a “second hit” provided by the DMD disease could worsen MR-mediated heart pathologies (such a “second hit” is seen in other aldosterone models). We find that both aldosterone and prednisolone increase mdx heart size and fibrosis, consistent with increased MR activation. Both Panels: Vamorolone is a potent MR antagonist that protects both epithelial and non-epithelial tissue types. (Black = physiological ligands, Blue = Vamorolone, Red = Prednisolone; Aldo = Aldosterone, Cort = Cortisol, HSD11B2 = 11β-hydroxysteroid dehydrogenase, Pred = Prednisolone, Vam = Vamorolone).
Vamorolone is effective against both hypertension and cardiomyopathy induced by hyperaldosteronism in vivo. Previous mdx cardiac challenges used dobutamine or isoproterenol, both of which stress the heart by stimulating sympathetic nerves to increase the rate and force of cardiac contraction (Danialou et al, 2001; Yasuda et al, 2005; Townsend et al, 2007; Spurney et al, 2011). This worsens cardiomyocyte injury, cardiac pump failure, and acute heart failure. Aldosterone also increases cardiac workload to stress the heart, by regulating blood volume and blood pressure. In addition, aldosterone activates MR signaling locally within specific tissues to increase myocardial fibrosis. Tissue-specific knockout in cardiomyocytes, macrophages, and endothelial cells show that local MR signaling within these tissues promotes heart inflammation, fibrosis, dilatation, and/or dysfunction (Rickard et al, 2009, 2014; Fraccarollo et al, 2011; Lother et al, 2011; Bienvenu et al, 2012). In the clinic, spironolactone improves severe heart failure at low doses shown to lack diuretic or blood pressure effects (Investigators, 1996; Pitt et al, 1999). Eplerenone, introduced as a safer and more MR-specific (de Gasparo et al, 1987) heart failure drug in 2003, also improves cardiac outcomes in severe and mild forms of heart failure (Pitt et al, 2003; Rossignol et al, 2011; Zannad et al, 2011). Recent DMD studies show that eplerenone improves or slows decline in LV strain, ejection fraction, and myocardial damage, and that these effects are heightened with earlier treatment (Raman et al, 2015, 2017). These findings are consistent with mouse models, where spironolactone used in combination with lisinopril improves cardiac strain and damage in a severe utrophin double mutant (utr+/−; mdx) model (Rafael-Fortney et al, 2011; Lowe et al, 2016). Excitingly, MR antagonists continue to evolve as they are relevant to a broader array of dystrophic, hypertensive, and non-hypertensive forms of heart failure.
Eplerenone and deflazacort show increased specificity over the other compounds for the MR and GR, respectively. Eplerenone is part of a series of spironolactone derivatives developed in the 1980's as MR antagonists, with the goal of eliminating spironolactone's off-target side effects on the androgen and progesterone receptors (Smith, 1962; de Gasparo et al, 1989). Modification of the 9,11α-position of spironolactone's structure maintains MR-binding affinity but can improve receptor specificity (de Gasparo et al, 1987). Deflazacort, on the other hand, is an 11β-hydroxysteroid developed as a more targeted anti-inflammatory GR agonist with differentiated tissue-specific bioavailability (Hahn et al, 1981; Diederich et al, 2002). Early reports suggest that it has reduced side effects on bone, adipose tissue, and weight (Mesa et al, 1991; Bonifati et al, 2000; Ferraris et al, 2000; Grosso et al, 2008). We and others find that deflazacort shows increased specificity for the GR, with reduced or absent effects on the MR (Grossmann et al, 2004). This specificity suggests that deflazacort may provide improved safety with regard to MR-specific side effects versus prednisone, which is a dual GR/MR agonist. However, deflazacort still shows potent GR transactivation, and recent DMD trials report increased cataracts, cushingoid features, and growth delay with deflazacort versus prednisolone (Bello et al, 2015a). Further study should be carried out to dissect MR- from GR-specific side effects, to determine tissue-specific impacts, and to directly compare deflazacort versus prednisone in the DMD clinic.
Future studies using tissue-specific knockout mice and receptor-specific ligands will be valuable to further dissect the roles of the mineralocorticoids, glucocorticoids, their receptors, and their regulatory enzymes. For example, we find that intracellular, inflammatory NF-κB signaling is inhibited by vamorolone, prednisolone, and deflazacort in a GR-specific manner. However, the renin-angiotensin-aldosterone system also impacts inflammation via effects on reactive oxygen species, blood pressure, blood volume, and inflammatory cell infiltration/adhesion. Accordingly, work by other groups indicate that MR antagonists can also show a level of anti-inflammatory and membrane-stabilizing effects driven independently from the GR and these may provide separate pathways of efficacy for drugs such as eplerenone (Chadwick et al, 2017). In addition, a recent report suggests that myeloid cells can synthesize aldosterone in a manner that could have local impacts on the dystrophic microenvironment of skeletal muscle (Chadwick et al, 2016). In contrast to skeletal muscle in that study, here we do not detect Hsd11b2 expression in heart tissue; this finding is consistent with our findings that prednisolone can worsen heart phenotypes of mdx mice. It will be intriguing to eventually compare a full dissection of steroidal pathways in muscle, heart, immune, and fibroblast tissue types. Doing so will provide valuable information on how to best apply current clinical drugs, on future drug development, and on the biology of muscle versus heart involvement in DMD.
Prednisolone activation of MR signaling can explain several reports that it has detrimental effects on mdx cardiac phenotypes at various stages of disease. At a young age, we find that prednisolone causes premature fibrosis and increased heart mass in mdx mice when vehicle controls show no signs of cardiomyopathy (Heier et al, 2013). In older mdx mice that naturally present with cardiomyopathy, we find that chronic prednisolone treatment increases heart fibrosis, increases blood pressure, and decreases heart function (Guerron et al, 2010; Heier et al, 2013; Uaesoontrachoon et al, 2014). We should note that there are differences in both heart function and blood pressure between the stages of mdx disease; in younger mdx here we saw no natural decrease in heart function and no natural blood pressure phenotype, whereas older mdx develop a cardiomyopathy with decreased heart function and a decrease in blood pressure (Spurney et al, 2009; Uaesoontrachoon et al, 2014). Other groups also report findings that prednisolone worsens cardiomyopathy in mdx mice. A previous article comparing prednisolone to angiotensin-converting enzyme inhibitors finds that administration of prednisolone on its own increases heart fibrosis, LV dilatation, and diastolic dysfunction (Bauer et al, 2009). Recently, two cardiac combination therapies, each containing spironolactone along with another drug, were tested on a background of either no treatment or prednisolone, representing the current standard of care (Janssen et al, 2014). In that study, prednisolone also increases myocardial damage, fibrosis, and strain, whereas this effect was lessened in groups that received spironolactone. Together, these reports are consistent with a model where drugs can impact mdx heart phenotypes by affecting MR activity.
We find that dystrophin deficiency is a second hit that specifically sensitizes mdx hearts to MR activity. Aldosterone exposure in mdx mice causes an increase in heart fibrosis, LV wall thickness, LV mass, and heart mass, with a concurrent decrease in heart function. The corresponding efficacy of MR-specific eplerenone and two other MR antagonists shows that these effects are mediated by activated MR. In contrast, aldosterone exposure by itself shows a lack of progressive heart pathology in wild-type mice, consistent with reports where mice are not additionally exposed to high salt or another insult (Brilla & Weber, 1992; Peters et al, 2009). In the literature, introduction of a “second hit” drives a progression of pathology to cardiac fibrosis and heart failure. This “second hit” is provided in various models by a high salt diet, inflammation, oxidative stress, or myocardial infarction (Brilla & Weber, 1992; Sun et al, 2002; Di Zhang et al, 2008; Fraccarollo et al, 2011; He et al, 2011; Ruhs et al, 2012). We find that the loss of dystrophin in mdx mice also provides such a second hit, which could be due to chronic inflammation or increases in cardiomyocyte injury due to the dystrophic phenotype.
Our work here can have both immediate and future clinical impacts. Prednisone, deflazacort, and eplerenone are already being used therapeutically in DMD. Our data strongly support the efficacy of eplerenone in preventing the progression of cardiac disease in DMD, particularly in a background of pharmacological glucocorticoids. Deflazacort shows increased specificity for the GR; however, it maintains GR side effects and lacks the cardiac benefits of MR antagonists. The dual-receptor profile of vamorolone is particularly exciting for its potential to simultaneously treat inflammatory muscle and cardiac pathology with improved safety in DMD. In preclinical animal trials, vamorolone has now been shown to have in vivo anti-inflammatory efficacy as measured by histology, live imaging, cytokine analysis, and/or flow cytometry in models of DMD, inflammatory bowel disease, asthma, limb girdle muscular dystrophy, and multiple sclerosis (Damsker et al, 2013, 2016; Heier et al, 2013; Dillingham et al, 2015; Sreetama et al, 2018). Vamorolone has completed phase I clinical trials in healthy adults, where it showed the loss of most side effects of corticosteroids even through high doses (20 mg/kg/d) (Hoffman et al, 2018). Vamorolone also recently completed phase 2a trials in DMD patients, where it showed anti-inflammatory efficacy and a dissociation of effects on safety versus efficacy biomarkers in patient serum (Conklin et al, 2018). Data from human phase I trials and preclinical animal model trials indicate that vamorolone, in comparison to prednisone, avoids or has substantially reduced side effects on bone, metabolism, adrenal suppression, and immunosuppression. Based on these data, vamorolone has moved forward and is currently being evaluated in phase 2b trials in subjects with DMD. Through shared mechanisms, this has the potential to further impact other diseases characterized by chronic inflammation or cardiovascular pathology.
Materials and Methods
MR activity assays
Activity of the MR was assayed via chemiluminescence assay using a stable cell line (PathHunter MR protein interaction assay; DiscoveRx) that expresses two fusion reporter proteins which detect the interaction of activated MR with an MR co-activator protein. To detect MR activation by agonist ligands, cells were treated with increasing concentrations of drug in DMSO for 6 h. To detect inactivation of the MR by antagonist ligands, cells were pretreated with drug for 1 h, then MR activity was induced with aldosterone at an EC80 concentration (2.1 nM) for 5 h.
CRISPR-mediated GR knockout experiments
CRISPR/Cas9 was used to introduce a deletion mutation into the first coding exon of the GR. A Neon transfection system (Thermo Fisher Scientific) was used to transfect the Cas9 vector into C2C12 myoblasts with gRNA sequences TGGACTTGTATAAAACCCTGAGG and GACTTGGGCTACCCACAGCAGGG. Single cells were isolated on 96-well plates and expanded into homogenous clonal populations. PCR analysis was performed to detect clones with a GR deletion, using forward primer CCCCTGGTAGAGACGAAGTC and reverse primer CAATTTCACACTGCCACCGTT, which produces a 578-bp amplicon for wild-type GR and an approximately 300-bp amplicon for the deletion clones. For subsequent experiments, we selected a knockout clone with a sequencing-confirmed 271-bp deletion which causes a frameshift and loss of protein expression as detected by Western blot with GR (m-20) antibody (Santa Cruz).
To test the effect of drugs on GR-activated targets, we plated C2C12 and knockout myoblasts in DMEM containing charcoal-stripped FBS overnight, then treated with MR and GR ligands at 100 nM for 24 h. To test the effect of drugs on inflammatory targets regulated by NF-κB, we first differentiated C2C12 cells into myotubes by replacing media with differentiation media (DMEM with 5% horse serum) for 4 d. Myotubes were pretreated with drug for 30 min, followed by the induction of inflammatory transcription by treating with recombinant mouse TNFα (R & D Systems) for 24 h. All cells were lysed in TRIzol for RNA and expression of target genes (Ccl2, Gilz, and Irf1) quantified by TaqMan Assay (Thermo Fisher Scientific).
In vitro macrophage and cardiomyocyte experiments
RAW 264.7 cells were cultured in DMEM with 10% FBS. Cells were pretreated with 10 μM drug. Inflammatory signaling was induced using LPS (Thermo Fisher Scientific) at a dilution of 1:1,000. After 24 h, cells were lysed for RNA using TRIzol and expression of target genes (Ccl2, Il1b, Il6, and Irf1) quantified by TaqMan Assay (Thermo Fisher Scientific). Levels of secreted proteins were assayed by luminescence-based IL1B and IL6 AlphaLISA assays (PerkinElmer) on media from the same cells assayed by qRT-PCR. Primary cardiomyocytes were isolated from ventricular tissue of hearts in postnatal day 2 C57/BL6 mice by dissecting the ventricles and enzymatically disassociating the cells, with subsequent culture in Cardiomyocyte Maintenance Medium (BrainBits). Cardiomyocytes were observed to display spontaneous contractions in culture, consistent with a cardiac phenotype. Cardiomyocytes were pretreated with 10 μM drug for 30 min and induced with TNF for 24 h. Expression of Il1β and Il6 was quantified by TaqMan qRT-PCR. IL6 protein was quantified by AlphaLISA (PerkinElmer). HL-1 atrial cardiomyocyte cells (Millipore Sigma) were cultured as previously described (Claycomb et al, 1998) with daily replacement of supplemented Claycomb media (10% FBS, 1× Penicillin/Streptomycin, 0.1 mM norepinephrine, 2 mM L-Glutamine). For treatment, cells were serum starved for 24 h, pretreated with drug for 30 min, and induced with TNF for 24 h.
Effects of steroid receptor ligands in mice
All animal work was conducted according to relevant institutional, national, and international guidelines, with adherence to standards of the National Institutes of Health (NIH) Guide for the Care and Use of Laboratory Animals. All mdx experiments were conducted according to the protocols approved by the Institutional Animal Care and Use Committee of Children's National Medical Center. All D2-mdx experiments were conducted according to protocols approved by the University Committee on Laboratory Animals of Dalhousie University. Animals were maintained in a controlled mouse facility with a 12 h light: 12 h dark photoperiod, fed ad libitum, and monitored daily for health. Wild-type (strain 000476; C57BL/10ScSnJ) and mdx (strain 001801; C57BL/10ScSn-Dmdmdx/J) mice were obtained from The Jackson Laboratory. All mice were initially assayed for weight, blood pressure, and echocardiography parameters (n ≥ 8 per group). Mice were randomly divided into groups evenly matched for baseline data, with subsequent phenotyping and histology experiments blinded to genotype and drug treatment. For the MR antagonism trial, groups were then pretreated with drug for one week, receiving daily oral drug in cherry syrup (vehicle, vamorolone at 20 mg/kg, eplerenone at 100 mg/kg, or spironolactone at 20 mg/kg). At three months of age, all mice were then implanted with osmotic pumps containing either vehicle (10% ethanol) or aldosterone secreted at a dose of 0.25 mg/kg/d. Drug dosing was continued daily for the duration of the trial. Two mice were removed from the trial, one mouse exhibited aortic stenosis at baseline and was removed at trial onset, the other mouse showed complications from pump implantation surgery and was euthanized. 6 wk after aldosterone pump implantation, mice were assayed for blood pressure and echocardiography. The following week, mice were sacrificed and tissues collected. Histopathology with Sirius Red staining for fibrosis was performed on cross-sections of heart at the level of the papillary muscles. Sirius Red staining was imaged using a VS120 scanning microscope (Olympus) with quantification performed using ImageJ (version 1.48) software.
Expression of Hsd11b2 was assayed in 5-mo-old mdx heart and kidney tissues. Frozen tissues were ground using a liquid nitrogen–cooled mortar and pestle, then RNA was extracted using TRIzol. Hsd11b2 expression was quantified by TaqMan qRT-PCR assay (Thermo Fisher Scientific).
To examine prednisolone versus vamorolone effects on gene expression linked to MR activation and fibrosis, we assayed archival heart samples. This sample set was taken from a trial in which mdx mice were subjected to treadmill running to unmask mild phenotypes, and were treated with either vehicle, prednisolone (5 mg/kg), or vamorolone (45 mg/kg) for four months beginning at 6 wk of age (Heier et al, 2013). In that trial, both drug groups showed improvements in strength measurements and in measurements of inflammation; however, prednisolone caused several side effects that were avoided by vamorolone.
For studies of prednisolone versus vamorolone effects in a more severe model of DMD, the DBA/2J-mdx model (or D2-mdx) was used. Groups (n ≥ 9 per group) of severe D2-mdx mice received daily oral vehicle (cherry syrup), vamorolone (30 mg/kg), or prednisolone (5 mg/kg) for a period of 8 wk, starting at 2 mo of age. One mouse from the D2-mdx vehicle group was removed from the study and euthanized due to complications from submandibular blood collection during the seventh week of treatment. Echocardiography was performed in the final week of treatment. At treatment endpoint, heart mass was measured and serum collected for measurement of insulin levels by ELISA using an Ultra-Sensitive Mouse Insulin ELISA Kit (Crystal Chem Inc., USA).
Blood pressure and echocardiography
Mouse blood pressure was measured by tail cuff (Hatteras Instruments). Echocardiography was performed under isoflurane anesthesia using a Vevo 770 micro-ultrasound imaging system in mdx and a Vevo 1100 system in D2-mdx (VisualSonics). Mice were assayed in M-mode of the parasternal short axis for ejection fraction, fractional shortening, and left ventricular wall thickness at the level of the papillary muscles. Image analysis and calculations were performed using Vevo software.
Statistical analysis
The n values for each experiment are provided in Figure legends. In vitro studies were analyzed by ANOVA with post hoc multiple comparisons of each treatment with vehicle. Bar graphs represent mean ± SEM. Aldosterone exposure studies were analyzed by ANOVA with post hoc multiple comparisons of each group with aldosterone-exposed mdx controls (aldosterone plus vehicle). Gene expression qPCR data were tested for outlier values using a Grubbs test as described in Figure legends. For box and whisker plots, boxes extend from the 25th to 75th percentile, with the whiskers marking the highest and lowest values for each group. The two wild-type groups (+/− aldosterone) were compared with each other using a two-tailed t test. D2-mdx studies were analyzed by ANOVA with post hoc multiple comparisons of each treatment to vehicle. Values of P < 0.05 were regarded as statistically significant.
Supplementary Material
Acknowledgements
The authors would like to thank Marina Moraca, Christina Bell, William Ross, Emma Gillis, and Todd Dow of AGADA Biosciences for their work on treatments and dissections in the D2-mdx trial. This work was supported by grants from the Foundation to Eradicate Duchenne, the Clark Charitable Foundation, Action Duchenne, and the NIH. CR Heier and this project were funded by the NIH (K99HL130035, R00HL130035, and L40AR068727). A Fiorillo is supported by the Department of Defense (W81XWH-17-1-047). CR Heier, AA Fiorillo, and DA Mazala were supported by the NIH (T32AR056993). Microscopic analysis for this study was conducted at the Children's Research Institute (CRI) Light Microscopy and Image Analysis Core, which is supported by CRI and the Intellectual and Developmental Disabilities Research Center Award (U54HD090257) through the National Institutes of Health and the National Institute of Child Health and Human Development (NICHD).
Author Contributions
CR Heier: conceptualization, methodology, investigation, formal analysis, supervision, funding acquisition, and writing—original draft, review and editing.
Q Yu: methodology, investigation, formal analysis, and writing—original draft.
AA Fiorillo: conceptualization, methodology, formal analysis, and writing—original draft, review and editing.
CB Tully: formal analysis, methodology, investigation, and writing—original draft, review and editing.
A Tucker: methodology and investigation.
DA Mazala: Formal analysis, methodology, investigation, and writing—original draft, review and editing.
K Uaesoontrachoon: investigation, formal analysis, and supervision.
S Srinivassane: investigation, formal analysis, and supervision.
JM Damsker: conceptualization and supervision.
EP Hoffman: conceptualization, supervision, funding acquisition, formal analysis, and writing—original draft, review and editing.
K Nagaraju: conceptualization, supervision, formal analysis, and funding acquisition.
CF Spurney: conceptualization, methodology, supervision, funding acquisition, formal analysis, and writing—original draft, review and editing.
Conflict of Interest Statement
ReveraGen BioPharma owns the method of use intellectual property relating to the use of Δ9,11 compounds to treat disease. EP Hoffman and K Nagaraju are co-founders of ReveraGen with shares in the company. JM Damsker is an employee of the company. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- Bauer R, Straub V, Blain A, Bushby K, MacGowan GA (2009) Contrasting effects of steroids and angiotensin-converting-enzyme inhibitors in a mouse model of dystrophin-deficient cardiomyopathy. Eur J Heart Fail 11: 463–471. 10.1093/eurjhf/hfp028 [DOI] [PubMed] [Google Scholar]
- Bello L, Gordish-Dressman H, Morgenroth LP, Henricson EK, Duong T, Hoffman EP, Cnaan A, McDonald CM (2015a) Prednisone/prednisolone and deflazacort regimens in the CINRG Duchenne Natural History Study. Neurology 85: 1048–1055. 10.1212/wnl.0000000000001950 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bello L, Kesari A, Gordish-Dressman H, Cnaan A, Morgenroth LP, Punetha J, Duong T, Henricson EK, Pegoraro E, McDonald CM, et al. (2015b) Genetic modifiers of ambulation in the Cooperative International Neuromuscular Research Group Duchenne Natural History Study. Ann Neurol 77: 684–696. 10.1002/ana.24370 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bienvenu LA, Morgan J, Rickard AJ, Tesch GH, Cranston GA, Fletcher EK, Delbridge LM, Young MJ (2012) Macrophage mineralocorticoid receptor signaling plays a key role in aldosterone-independent cardiac fibrosis. Endocrinology 153: 3416–3425. 10.1210/en.2011-2098 [DOI] [PubMed] [Google Scholar]
- Bledsoe RK, Montana VG, Stanley TB, Delves CJ, Apolito CJ, McKee DD, Consler TG, Parks DJ, Stewart EL, Willson TM, et al. (2002) Crystal structure of the glucocorticoid receptor ligand binding domain reveals a novel mode of receptor dimerization and coactivator recognition. Cell 110: 93–105. 10.1016/s0092-8674(02)00817-6 [DOI] [PubMed] [Google Scholar]
- Bonifati MD, Ruzza G, Bonometto P, Berardinelli A, Gorni K, Orcesi S, Lanzi G, Angelini C (2000) A multicenter, double-blind, randomized trial of deflazacort versus prednisone in Duchenne muscular dystrophy. Muscle Nerve 23: 1344–1347. [DOI] [PubMed] [Google Scholar]
- Brilla CG, Weber KT (1992) Mineralocorticoid excess, dietary sodium, and myocardial fibrosis. J Lab Clin Med 120: 893–901 [PubMed] [Google Scholar]
- Bujak M, Frangogiannis NG (2009) The role of IL-1 in the pathogenesis of heart disease. Arch Immunol Ther Exp (Warsz) 57: 165–176. 10.1007/s00005-009-0024-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chadwick JA, Bhattacharya S, Lowe J, Weisleder N, Rafael-Fortney JA (2017) Renin-angiotensin-aldosterone system inhibitors improve membrane stability and change gene-expression profiles in dystrophic skeletal muscles. Am J Physiol Cell Physiol 312: C155–C168. 10.1152/ajpcell.00269.2016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chadwick JA, Swager SA, Lowe J, Welc SS, Tidball JG, Gomez-Sanchez CE, Gomez-Sanchez EP, Rafael-Fortney JA (2016) Myeloid cells are capable of synthesizing aldosterone to exacerbate damage in muscular dystrophy. Hum Mol Genet 25: 5167–5177. 10.1093/hmg/ddw331 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen YW, Nagaraju K, Bakay M, McIntyre O, Rawat R, Shi R, Hoffman EP (2005) Early onset of inflammation and later involvement of TGFbeta in Duchenne muscular dystrophy. Neurology 65: 826–834. 10.1212/01.wnl.0000173836.09176.c4 [DOI] [PubMed] [Google Scholar]
- Claycomb WC, Lanson NA Jr., Stallworth BS, Egeland DB, Delcarpio JB, Bahinski A, Izzo NJ Jr (1998) HL-1 cells: A cardiac muscle cell line that contracts and retains phenotypic characteristics of the adult cardiomyocyte. Proc Natl Acad Sci U S A 95: 2979–2984. 10.1073/pnas.95.6.2979 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cole TJ, Young MJ (2017) 30 years of the mineralocorticoid receptor: Mineralocorticoid receptor null mice: Informing cell-type-specific roles. J Endocrinol 234: T83–T92. 10.1530/joe-17-0155 [DOI] [PubMed] [Google Scholar]
- Coley WD, Bogdanik L, Vila MC, Yu Q, Van Der Meulen JH, Rayavarapu S, Novak JS, Nearing M, Quinn JL, Saunders A, et al. (2016) Effect of genetic background on the dystrophic phenotype in mdx mice. Hum Mol Genet 25: 130–145. 10.1093/hmg/ddv460 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Conklin LS, Damsker JM, Hoffman EP, Jusko WJ, Mavroudis PD, Schwartz BD, Mengle-Gaw LJ, Smith EC, Mah JK, Guglieri M, et al. (2018) Phase IIa trial in Duchenne muscular dystrophy shows vamorolone is a first-in-class dissociative steroidal anti-inflammatory drug. Pharmacol Res 136: 140–150. 10.1016/j.phrs.2018.09.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Damsker JM, Conklin LS, Sadri S, Dillingham BC, Panchapakesan K, Heier CR, McCall JM, Sandler AD (2016) VBP15, a novel dissociative steroid compound, reduces NFkappaB-induced expression of inflammatory cytokines in vitro and symptoms of murine trinitrobenzene sulfonic acid-induced colitis. Inflamm Res 65: 737–743. 10.1007/s00011-016-0956-8 [DOI] [PubMed] [Google Scholar]
- Damsker JM, Dillingham BC, Rose MC, Balsley MA, Heier CR, Watson AM, Stemmy EJ, Jurjus RA, Huynh T, Tatem K, et al. (2013) VBP15, a glucocorticoid analogue, is effective at reducing allergic lung inflammation in mice. PLoS One 8: e63871 10.1371/journal.pone.0063871 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Danialou G, Comtois AS, Dudley R, Karpati G, Vincent G, Des Rosiers C, Petrof BJ (2001) Dystrophin-deficient cardiomyocytes are abnormally vulnerable to mechanical stress-induced contractile failure and injury. FASEB J 15: 1655–1657. 10.1096/fj.01-0030fje [DOI] [PubMed] [Google Scholar]
- de Gasparo M, Joss U, Ramjoue HP, Whitebread SE, Haenni H, Schenkel L, Kraehenbuehl C, Biollaz M, Grob J, Schmidlin J, et al. (1987) Three new epoxy-spirolactone derivatives: Characterization in vivo and in vitro. J Pharmacol Exp Ther 240: 650–656 [PubMed] [Google Scholar]
- de Gasparo M, Whitebread SE, Preiswerk G, Jeunemaitre X, Corvol P, Menard J (1989) Antialdosterones: Incidence and prevention of sexual side effects. J Steroid Biochem 32: 223–227. 10.1016/0022-4731(89)90169-6 [DOI] [PubMed] [Google Scholar]
- Di Zhang A, Nguyen Dinh Cat A, Soukaseum C, Escoubet B, Cherfa A, Messaoudi S, Delcayre C, Samuel JL, Jaisser F (2008) Cross-talk between mineralocorticoid and angiotensin II signaling for cardiac remodeling. Hypertension 52: 1060–1067. 10.1161/hypertensionaha.108.117531 [DOI] [PubMed] [Google Scholar]
- Diederich S, Eigendorff E, Burkhardt P, Quinkler M, Bumke-Vogt C, Rochel M, Seidelmann D, Esperling P, Oelkers W, Bahr V (2002) 11beta-hydroxysteroid dehydrogenase types 1 and 2: An important pharmacokinetic determinant for the activity of synthetic mineralo- and glucocorticoids. J Clin Endocrinol Metab 87: 5695–5701. 10.1210/jc.2002-020970 [DOI] [PubMed] [Google Scholar]
- Dillingham BC, Knoblach SM, Many GM, Harmon BT, Mullen AM, Heier CR, Bello L, McCall JM, Hoffman EP, Connor EM, et al. (2015) VBP15, a novel anti-inflammatory, is effective at reducing the severity of murine experimental autoimmune encephalomyelitis. Cell Mol Neurobiol 35: 377–387. 10.1007/s10571-014-0133-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eagle M, Baudouin SV, Chandler C, Giddings DR, Bullock R, Bushby K (2002) Survival in Duchenne muscular dystrophy: Improvements in life expectancy since 1967 and the impact of home nocturnal ventilation. Neuromuscul Disord 12: 926–929. 10.1016/s0960-8966(02)00140-2 [DOI] [PubMed] [Google Scholar]
- Edwards CR, Stewart PM, Burt D, Brett L, McIntyre MA, Sutanto WS, de Kloet ER, Monder C (1988) Localisation of 11 beta-hydroxysteroid dehydrogenase--tissue specific protector of the mineralocorticoid receptor. Lancet 2: 986–989. 10.1016/s0140-6736(88)90742-8 [DOI] [PubMed] [Google Scholar]
- Ferraris JR, Pasqualini T, Legal S, Sorroche P, Galich AM, Pennisi P, Domene H, Jasper H (2000) Effect of deflazacort versus methylprednisone on growth, body composition, lipid profile, and bone mass after renal transplantation. The Deflazacort Study Group. Pediatr Nephrol 14: 682–688. 10.1007/s004670000337 [DOI] [PubMed] [Google Scholar]
- Finsterer J, Stollberger C (2003) The heart in human dystrophinopathies. Cardiology 99: 1–19. 10.1159/000068446 [DOI] [PubMed] [Google Scholar]
- Fiorillo AA, Tully CB, Damsker JM, Nagaraju K, Hoffman EP, Heier CR (2018) Muscle miRNAome shows suppression of chronic inflammatory miRNAs with both prednisone and vamorolone. Physiol Genomics 50: 735–745. 10.1152/physiolgenomics.00134.2017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Flanigan KM, Ceco E, Lamar KM, Kaminoh Y, Dunn DM, Mendell JR, King WM, Pestronk A, Florence JM, Mathews KD, et al. (2013) LTBP4 genotype predicts age of ambulatory loss in Duchenne muscular dystrophy. Ann Neurol 73: 481–488. 10.1002/ana.23819 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fraccarollo D, Berger S, Galuppo P, Kneitz S, Hein L, Schutz G, Frantz S, Ertl G, Bauersachs J (2011) Deletion of cardiomyocyte mineralocorticoid receptor ameliorates adverse remodeling after myocardial infarction. Circulation 123: 400–408. 10.1161/circulationaha.110.983023 [DOI] [PubMed] [Google Scholar]
- Funder JW, Pearce PT, Smith R, Smith AI (1988) Mineralocorticoid action: Target tissue specificity is enzyme, not receptor, mediated. Science 242: 583–585. 10.1126/science.2845584 [DOI] [PubMed] [Google Scholar]
- Griggs RC, Herr BE, Reha A, Elfring G, Atkinson L, Cwik V, McColl E, Tawil R, Pandya S, McDermott MP, et al. (2013) Corticosteroids in Duchenne muscular dystrophy: Major variations in practice. Muscle Nerve 48: 27–31. 10.1002/mus.23831 [DOI] [PubMed] [Google Scholar]
- Griggs RC, Miller JP, Greenberg CR, Fehlings DL, Pestronk A, Mendell JR, Moxley RT 3rd, King W, Kissel JT, Cwik V, et al. (2016) Efficacy and safety of deflazacort vs prednisone and placebo for Duchenne muscular dystrophy. Neurology 87: 2123–2131. 10.1212/wnl.0000000000003217 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grossmann C, Scholz T, Rochel M, Bumke-Vogt C, Oelkers W, Pfeiffer AF, Diederich S, Bahr V (2004) Transactivation via the human glucocorticoid and mineralocorticoid receptor by therapeutically used steroids in CV-1 cells: A comparison of their glucocorticoid and mineralocorticoid properties. Eur J Endocrinol 151: 397–406. 10.1530/eje.0.1510397 [DOI] [PubMed] [Google Scholar]
- Grosso S, Farnetani M, Mostardini R, Cordelli D, Berardi R, Balestri P (2008) A comparative study of hydrocortisone versus deflazacort in drug-resistant epilepsy of childhood. Epilepsy Res 81: 80–85. 10.1016/j.eplepsyres.2008.04.016 [DOI] [PubMed] [Google Scholar]
- Guerron AD, Rawat R, Sali A, Spurney CF, Pistilli E, Cha HJ, Pandey GS, Gernapudi R, Francia D, Farajian V, et al. (2010) Functional and molecular effects of arginine butyrate and prednisone on muscle and heart in the mdx mouse model of Duchenne Muscular Dystrophy. PLoS One 5: e11220 10.1371/annotation/a82245dd-7379-4067-bd84-b0fc5ffb5869 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hahn BH, Pletscher LS, Muniain M (1981) Immunosuppressive effects of deflazacort: A new glucocorticoid with bone-sparing and carbohydrate-sparing properties: Comparison with prednisone. J Rheumatol 8: 783–790 [PubMed] [Google Scholar]
- Hammer S, Spika I, Sippl W, Jessen G, Kleuser B, Holtje HD, Schafer-Korting M (2003) Glucocorticoid receptor interactions with glucocorticoids: Evaluation by molecular modeling and functional analysis of glucocorticoid receptor mutants. Steroids 68: 329–339. 10.1016/s0039-128x(03)00030-8 [DOI] [PubMed] [Google Scholar]
- He BJ, Joiner ML, Singh MV, Luczak ED, Swaminathan PD, Koval OM, Kutschke W, Allamargot C, Yang J, Guan X, et al. (2011) Oxidation of CaMKII determines the cardiotoxic effects of aldosterone. Nat Med 17: 1610–1618. 10.1038/nm.2506 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heier CR, Damsker JM, Yu Q, Dillingham BC, Huynh T, Van der Meulen JH, Sali A, Miller BK, Phadke A, Scheffer L, et al. (2013) VBP15, a novel anti-inflammatory and membrane-stabilizer, improves muscular dystrophy without side effects. EMBO Mol Med 5: 1569–1585. 10.1002/emmm.201302621 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heydemann A, Ceco E, Lim JE, Hadhazy M, Ryder P, Moran JL, Beier DR, Palmer AA, McNally EM (2009) Latent TGF-beta-binding protein 4 modifies muscular dystrophy in mice. J Clin Invest 119: 3703–3712. 10.1172/jci39845 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hiscott J, Marois J, Garoufalis J, D'Addario M, Roulston A, Kwan I, Pepin N, Lacoste J, Nguyen H, Bensi G, et al. (1993) Characterization of a functional NF-kappa B site in the human interleukin 1 beta promoter: Evidence for a positive autoregulatory loop. Mol Cell Biol 13: 6231–6240. 10.1128/mcb.13.10.6231 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hoffman EP, Brown RH Jr., Kunkel LM (1987) Dystrophin: The protein product of the Duchenne muscular dystrophy locus. Cell 51: 919–928. 10.1016/0092-8674(87)90579-4 [DOI] [PubMed] [Google Scholar]
- Hoffman EP, Riddle V, Siegler MA, Dickerson D, Backonja M, Kramer WG, Nagaraju K, Gordish-Dressman H, Damsker JM, McCall JM (2018) Phase 1 trial of vamorolone, a first-in-class steroid, shows improvements in side effects via biomarkers bridged to clinical outcomes. Steroids 134: 43–52. 10.1016/j.steroids.2018.02.010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hor KN, Taylor MD, Al-Khalidi HR, Cripe LH, Raman SV, Jefferies JL, O'Donnell R, Benson DW, Mazur W (2013) Prevalence and distribution of late gadolinium enhancement in a large population of patients with Duchenne muscular dystrophy: Effect of age and left ventricular systolic function. J Cardiovasc Magn Reson 15: 107 10.1186/1532-429x-15-107 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Investigators TR. (1996) Effectiveness of spironolactone added to an angiotensin-converting enzyme inhibitor and a loop diuretic for severe chronic congestive heart failure (the Randomized Aldactone Evaluation Study [RALES]). Am J Cardiol 78: 902–907. 10.1016/S0002-9149(96)00465-1 [DOI] [PubMed] [Google Scholar]
- Iqbal J, Andrew R, Cruden NL, Kenyon CJ, Hughes KA, Newby DE, Hadoke PW, Walker BR (2014) Displacement of cortisol from human heart by acute administration of a mineralocorticoid receptor antagonist. J Clin Endocrinol Metab 99: 915–922. 10.1210/jc.2013-2049 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Janssen PM, Murray JD, Schill KE, Rastogi N, Schultz EJ, Tran T, Raman SV, Rafael-Fortney JA (2014) Prednisolone attenuates improvement of cardiac and skeletal contractile function and histopathology by lisinopril and spironolactone in the mdx mouse model of Duchenne muscular dystrophy. PLoS One 9: e88360 10.1371/journal.pone.0088360 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koenig M, Hoffman EP, Bertelson CJ, Monaco AP, Feener C, Kunkel LM (1987) Complete cloning of the Duchenne muscular dystrophy (DMD) cDNA and preliminary genomic organization of the DMD gene in normal and affected individuals. Cell 50: 509–517. 10.1016/0092-8674(87)90504-6 [DOI] [PubMed] [Google Scholar]
- Libermann TA, Baltimore D (1990) Activation of interleukin-6 gene expression through the NF-kappa B transcription factor. Mol Cell Biol 10: 2327–2334. 10.1128/mcb.10.5.2327 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lind U, Greenidge P, Gillner M, Koehler KF, Wright A, Carlstedt-Duke J (2000) Functional probing of the human glucocorticoid receptor steroid-interacting surface by site-directed mutagenesis. Gln-642 plays an important role in steroid recognition and binding. J Biol Chem 275: 19041–19049. 10.1074/jbc.m000228200 [DOI] [PubMed] [Google Scholar]
- Lother A, Berger S, Gilsbach R, Rosner S, Ecke A, Barreto F, Bauersachs J, Schutz G, Hein L (2011) Ablation of mineralocorticoid receptors in myocytes but not in fibroblasts preserves cardiac function. Hypertension 57: 746–754. 10.1161/hypertensionaha.110.163287 [DOI] [PubMed] [Google Scholar]
- Lowe J, Floyd KT, Rastogi N, Schultz EJ, Chadwick JA, Swager SA, Zins JG, Kadakia FK, Smart S, Gomez-Sanchez EP, et al. (2016) Similar efficacy from specific and non-specific mineralocorticoid receptor antagonist treatment of muscular dystrophy mice. J Neuromuscul Dis 3: 395–404. 10.3233/jnd-160173 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mesa LE, Dubrovsky AL, Corderi J, Marco P, Flores D (1991) Steroids in Duchenne muscular dystrophy--deflazacort trial. Neuromuscul Disord 1: 261–266. 10.1016/0960-8966(91)90099-e [DOI] [PubMed] [Google Scholar]
- Monaco AP, Neve RL, Colletti-Feener C, Bertelson CJ, Kurnit DM, Kunkel LM (1986) Isolation of candidate cDNAs for portions of the Duchenne muscular dystrophy gene. Nature 323: 646–650. 10.1038/323646a0 [DOI] [PubMed] [Google Scholar]
- Nigro G, Comi LI, Politano L, Bain RJ (1990) The incidence and evolution of cardiomyopathy in Duchenne muscular dystrophy. Int J Cardiol 26: 271–277. 10.1016/0167-5273(90)90082-g [DOI] [PubMed] [Google Scholar]
- Peters J, Schluter T, Riegel T, Peters BS, Beineke A, Maschke U, Hosten N, Mullins JJ, Rettig R (2009) Lack of cardiac fibrosis in a new model of high prorenin hyperaldosteronism. Am J Physiol Heart Circ Physiol 297: H1845–H1852. 10.1152/ajpheart.01135.2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peterson JM, Wang DJ, Shettigar V, Roof SR, Canan BD, Bakkar N, Shintaku J, Gu JM, Little SC, Ratnam NM, et al. (2018) NF-kappaB inhibition rescues cardiac function by remodeling calcium genes in a Duchenne muscular dystrophy model. Nat Commun 9: 3431 10.1038/s41467-018-05910-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pitt B, Remme W, Zannad F, Neaton J, Martinez F, Roniker B, Bittman R, Hurley S, Kleiman J, Gatlin M (2003) Eplerenone, a selective aldosterone blocker, in patients with left ventricular dysfunction after myocardial infarction. N Engl J Med 348: 1309–1321. 10.1056/nejmoa030207 [DOI] [PubMed] [Google Scholar]
- Pitt B, Zannad F, Remme WJ, Cody R, Castaigne A, Perez A, Palensky J, Wittes J (1999) The effect of spironolactone on morbidity and mortality in patients with severe heart failure. Randomized Aldactone Evaluation Study Investigators. N Engl J Med 341: 709–717. 10.1056/nejm199909023411001 [DOI] [PubMed] [Google Scholar]
- Rafael-Fortney JA, Chimanji NS, Schill KE, Martin CD, Murray JD, Ganguly R, Stangland JE, Tran T, Xu Y, Canan BD, et al. (2011) Early treatment with lisinopril and spironolactone preserves cardiac and skeletal muscle in Duchenne muscular dystrophy mice. Circulation 124: 582–588. 10.1161/circulationaha.111.031716 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rafestin-Oblin ME, Fagart J, Souque A, Seguin C, Bens M, Vandewalle A (2002) 11beta-hydroxyprogesterone acts as a mineralocorticoid agonist in stimulating Na+ absorption in mammalian principal cortical collecting duct cells. Mol Pharmacol 62: 1306–1313. 10.1124/mol.62.6.1306 [DOI] [PubMed] [Google Scholar]
- Raman SV, Hor KN, Mazur W, Halnon NJ, Kissel JT, He X, Tran T, Smart S, McCarthy B, Taylor MD, et al. (2015) Eplerenone for early cardiomyopathy in Duchenne muscular dystrophy: A randomised, double-blind, placebo-controlled trial. Lancet Neurol 14: 153–161. 10.1016/s1474-4422(14)70318-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Raman SV, Hor KN, Mazur W, He X, Kissel JT, Smart S, McCarthy B, Roble SL, Cripe LH (2017) Eplerenone for early cardiomyopathy in Duchenne muscular dystrophy: Results of a two-year open-label extension trial. Orphanet J Rare Dis 12: 39 10.1186/s13023-017-0590-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ratman D, Vanden Berghe W, Dejager L, Libert C, Tavernier J, Beck IM, De Bosscher K (2013) How glucocorticoid receptors modulate the activity of other transcription factors: A scope beyond tethering. Mol Cell Endocrinol 380: 41–54. 10.1016/j.mce.2012.12.014 [DOI] [PubMed] [Google Scholar]
- Rickard AJ, Morgan J, Chrissobolis S, Miller AA, Sobey CG, Young MJ (2014) Endothelial cell mineralocorticoid receptors regulate deoxycorticosterone/salt-mediated cardiac remodeling and vascular reactivity but not blood pressure. Hypertension 63: 1033–1040. 10.1161/hypertensionaha.113.01803 [DOI] [PubMed] [Google Scholar]
- Rickard AJ, Morgan J, Tesch G, Funder JW, Fuller PJ, Young MJ (2009) Deletion of mineralocorticoid receptors from macrophages protects against deoxycorticosterone/salt-induced cardiac fibrosis and increased blood pressure. Hypertension 54: 537–543. 10.1161/hypertensionaha.109.131110 [DOI] [PubMed] [Google Scholar]
- Robinson CM, Hale PT, Carlin JM (2006) NF-kappa B activation contributes to indoleamine dioxygenase transcriptional synergy induced by IFN-gamma and tumor necrosis factor-alpha. Cytokine 35: 53–61. 10.1016/j.cyto.2006.07.007 [DOI] [PubMed] [Google Scholar]
- Rossignol P, Menard J, Fay R, Gustafsson F, Pitt B, Zannad F (2011) Eplerenone survival benefits in heart failure patients post-myocardial infarction are independent from its diuretic and potassium-sparing effects. Insights from an EPHESUS (Eplerenone Post-Acute Myocardial Infarction Heart Failure Efficacy and Survival Study) substudy. J Am Coll Cardiol 58: 1958–1966. 10.1016/j.jacc.2011.04.049 [DOI] [PubMed] [Google Scholar]
- Ruhs S, Stratz N, Schlor K, Meinel S, Mildenberger S, Rabe S, Gekle M, Grossmann C (2012) Modulation of transcriptional mineralocorticoid receptor activity by nitrosative stress. Free Radic Biol Med 53: 1088–1100. 10.1016/j.freeradbiomed.2012.06.028 [DOI] [PubMed] [Google Scholar]
- Sanders B, Cummings EE (2017) Letter to Jeffrey S. Aronin, Chairman and Chief Executive Officer, Marathon Pharmaceuticals, LLC. Congress of the United States (https://www.sanders.senate.gov/download/february-2017-letter-to-marathon?id=06256FA4-D5AA-4E3F-AD40-589E7D664E84&download=1&inline=file) [Google Scholar]
- Silversides CK, Webb GD, Harris VA, Biggar DW (2003) Effects of deflazacort on left ventricular function in patients with Duchenne muscular dystrophy. Am J Cardiol 91: 769–772. 10.1016/s0002-9149(02)03429-x [DOI] [PubMed] [Google Scholar]
- Smith WG. (1962) Spironolactone and gynaecomastia. Lancet 280: 886 10.1016/s0140-6736(62)90668-2 [DOI] [Google Scholar]
- Spurney CF, Gordish-Dressman H, Guerron AD, Sali A, Pandey GS, Rawat R, Van Der Meulen JH, Cha HJ, Pistilli EE, Partridge TA, et al. (2009) Preclinical drug trials in the mdx mouse: Assessment of reliable and sensitive outcome measures. Muscle Nerve 39: 591–602. 10.1002/mus.21211 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spurney CF, Guerron AD, Yu Q, Sali A, van der Meulen JH, Hoffman EP, Nagaraju K (2011) Membrane sealant Poloxamer P188 protects against isoproterenol induced cardiomyopathy in dystrophin deficient mice. BMC Cardiovasc Disord 11: 20 10.1186/1471-2261-11-20 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sreetama SC, Chandra G, Van der Meulen JH, Ahmad MM, Suzuki P, Bhuvanendran S, Nagaraju K, Hoffman EP, Jaiswal JK (2018) Membrane stabilization by modified steroid offers a potential therapy for muscular dystrophy due to dysferlin deficit. Mol Ther 26: 2231–2242. 10.1016/j.ymthe.2018.07.021 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sun Y, Zhang J, Lu L, Chen SS, Quinn MT, Weber KT (2002) Aldosterone-induced inflammation in the rat heart: Role of oxidative stress. Am J Pathol 161: 1773–1781. 10.1016/s0002-9440(10)64454-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Townsend D, Blankinship MJ, Allen JM, Gregorevic P, Chamberlain JS, Metzger JM (2007) Systemic administration of micro-dystrophin restores cardiac geometry and prevents dobutamine-induced cardiac pump failure. Mol Ther 15: 1086–1092. 10.1038/sj.mt.6300144 [DOI] [PubMed] [Google Scholar]
- Uaesoontrachoon K, Quinn JL, Tatem KS, Van Der Meulen JH, Yu Q, Phadke A, Miller BK, Gordish-Dressman H, Ongini E, Miglietta D, et al. (2014) Long-term treatment with naproxcinod significantly improves skeletal and cardiac disease phenotype in the mdx mouse model of dystrophy. Hum Mol Genet 23: 3239–3249. 10.1093/hmg/ddu033 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ueda A, Okuda K, Ohno S, Shirai A, Igarashi T, Matsunaga K, Fukushima J, Kawamoto S, Ishigatsubo Y, Okubo T (1994) NF-kappa B and Sp1 regulate transcription of the human monocyte chemoattractant protein-1 gene. J Immunol 153: 2052–2063 [PubMed] [Google Scholar]
- Wollert KC, Drexler H (2001) The role of interleukin-6 in the failing heart. Heart Fail Rev 6: 95–103. 10.1023/A:1011401825680 [DOI] [PubMed] [Google Scholar]
- Yang C, Balsells J, Chu HD, Cox JM, Crespo A, Ma X, Contino L, Brown P, Gao S, Zamlynny B, et al. (2015) Discovery of benzimidazole oxazolidinediones as novel and selective nonsteroidal mineralocorticoid receptor antagonists. ACS Med Chem Lett 6: 461–465. 10.1021/acsmedchemlett.5b00010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yasuda S, Townsend D, Michele DE, Favre EG, Day SM, Metzger JM (2005) Dystrophic heart failure blocked by membrane sealant poloxamer. Nature 436: 1025–1029. 10.1038/nature03844 [DOI] [PubMed] [Google Scholar]
- Young MJ, Rickard AJ (2015) Mineralocorticoid receptors in the heart: Lessons from cell-selective transgenic animals. J Endocrinol 224: R1–R13. 10.1530/joe-14-0471 [DOI] [PubMed] [Google Scholar]
- Zannad F, McMurray JJ, Krum H, van Veldhuisen DJ, Swedberg K, Shi H, Vincent J, Pocock SJ, Pitt B (2011) Eplerenone in patients with systolic heart failure and mild symptoms. N Engl J Med 364: 11–21. 10.1056/nejmoa1009492 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.