Abstract
The aim of this study was to investigate the anticancer and antioxidant activities of low molecular weight subfractions isolated from secondary metabolites produced by the wood degrading fungus Cerrena unicolor. Human colon cancer cells (stage I) HT-29 and human normal colon epithelial cells CCD 841 CoTr were used in the research. The present study demonstrated that the low molecular weight subfractions exhibited inhibitory activity towards human colon cancer cells HT-29 at a concentration range of 25–200 μg/mL. All 6 subfractions inhibited proliferation of cells down to 47.5–9.2% at the highest concentrations in a dose-dependent manner. The most desired activity was exhibited by subfractions S, 3, 4, and 5, as the proliferation of HT-29 cells was inhibited to the greatest extent (16.5, 47.5, 42.7, and 26.1% of the control, respectively), while the effect on CCD 841 CoTr cells was the mildest (inhibition to 54.4, 71.4, 79.4, and 53.4%, compared to the control, respectively). The microscopic observation revealed that all extracts induced programmed cell death, i.e. apoptosis (up to 44.4% (subfraction 6) towards HT-29 and less than 20% (most fractions) towards CCD 841 CoTr), with no or a significantly low level of necrosis in both cell lines at the same time.
Introduction
Mushrooms offer a wide range of benefits associated with consumption thereof and, therefore, they are regarded as functional foods1. The health-enhancing properties of edible mushrooms have been analyzed for years, especially in the traditional Eastern medicine and in the folk medicine of the West. They are characterized by high contents of easily digestible protein, fatty acids with a favorable ratio of polyunsaturated to saturated acids, vitamins mainly from the B group and vitamins A, E, and C, as well as polysaccharides, including dietary fiber2. The bioactive substances produced by fungi can be divided into two main groups: low molecular compounds, e.g. terpenoids or phenolic compounds, and high molecular weight compounds, e.g. polysaccharides and enzymes3. Both groups of compounds exhibit biological activities such as antioxidant, anticancer, immunostimulating, antiatherosclerotic, neuroprotective, anti-inflammatory, antiallergic, antibacterial, antiviral, or hypoglycemic effects. The range of these activities results from the diversity of bioactive components present in fungi, such as proteins, e.g. immunomodulatory mushroom proteins (FIPS), lectins, glycoproteins, polysaccharides, phenolic compounds, indole compounds, terpenoids, or lipids (including ergosterol and its derivatives)4–8. The need for finding new compounds with effective antitumor properties and high selectivity towards cancer cells and low toxicity to normal cells prompts investigations of a wide variety of chemically and structurally related compounds. Therefore, the last few decades have been a period of the most intense research on substances isolated from fungi, both fruiting bodies and in vitro cultivated mushroom biomass. The largest group of compounds with anti-inflammatory properties is constituted by fungal terpenoids. They also exhibit antitumor, antiviral, antimalarial, anti-yeast, and antibacterial activity9. Monoterpenoids act cytotoxically against cervical carcinoma (HeLa) and liver cancer (HepG2) cells10. Diterpenoids and triterpenoids isolated from Ganoderma lucidum (Reishii) and Ganoderma orbiforme, such as ganoboninketals AG, numerous ganoderic acids, lucidadiol, luciden acids, and many others that exert a cytotoxic effect on tumor cells, have anti-malarial and anti-tuberculosis activity9,11. In addition, G. lucidum triterpenes inhibit angiogenesis, which is important in the development of tumor mass and metastasis12. Bisabolol produced by Inonotus rickii inhibits the development of myeloid leukemia as well as breast, colorectal, lung, and liver cancers13, and sesquiterpenoids isolated from Pleurotus cornucopiae are characterized by cytotoxicity to cervical cancer cells (HeLa) and liver cancer cells14. Phenols are another important group of low-molecular compounds of fungal origin. They have a wide range of bioactive properties; hence, they are used in the treatment of cancer, cardiovascular diseases as cardioprotective and vasodilator compounds, and agents in the treatment of brain disorders and degenerative diseases. Many of them have been found to have anti-inflammatory and anti-allergic effects15,16. The largest group among phenolic compounds of fungal origin comprises phenolic acids and polyphenols, for instance flavonoids, stilbene, lignans, tannins, and oxidized polyphenols. Phenolic acids are the basic phenolic compounds produced by fungi. Phenolic compounds from Pleurotus ostreatus, Lentinula edodes, or Hypsizygus tessellatus have antibacterial properties as well17. However, phenolic compounds occurring in Hericium erinaceus additionally induce the synthesis of a factor that activates the growth and development of nerves18. Another equally interesting group of fungal compounds is organic acids. These substances are compounds with antioxidant properties; therefore, they can be used in the treatment of many diseases related to oxidative stress19.
Colon cancer is one of the most common cancers worldwide and the second most common cancer in developed countries20. Surgery and chemotherapy are the routine treatments for colon cancer patients. However, the benefits of chemotherapy are still under discussion, because a large proportion of patients die due to tumor recurrence after such therapy21. Therefore, constant search for substances that would be effective in treating this type of cancer is crucial. An interesting fungal species, tested primarily as a very efficient source of extracellular laccase produced in non-induced growth conditions, is Cerrena unicolor. In addition to the laccase, it produces many other low molecular weight secondary metabolites with a broad spectrum of biological activity, which was confirmed in our previous work22–25. Studies carried out to date have been focused on the total low molecular subfraction obtained directly as a by-product of the production of biotechnologically important enzymes, e.g. laccase. From the point of view of possible biomedical applications, it was important to characterize and separate the preparation obtained from the culture fluid of C. unicolor, which was used for determination of its qualitative composition as well as biological properties (especially the anticancer effect).
Methods
Strain, medium, growth processing, and separation of fungal samples
Cerrena unicolor (Bull. ex Fr.) Murr. was obtained from the culture collection of Regensburg University and deposited in the fungal collection of the Department of Biochemistry (Maria Curie-Sklodowska University, Poland) under strain number 139 (ITS sequence deposited in the GenBank under accession number DQ056858)26. The fermentor scale cultivation was performed according to the procedure described in earlier studies24. The post-culture fluid obtained from C. unicolor fungus culture was used in the study. The mycelium was separated from the culture fluid, which was subsequently subjected to preliminary separation on an ultrafiltration cartridge with 10 kDa MWCO (EMD Millipore™ Prep/Scale Spiral-Wound Ultrafiltration Modules: TFF-2), yielding two fractions with a molecular weight below 10 kDa and above 10 kDa. The lower mass fraction (ex-LMS) was further concentrated using Reverse Osmosis Membrane TFC-75F (Aquafilter Inc. USA) and subjected to further analysis. The fraction with a molecular weight below 10 kDa was fractionated on a chromatography column 5.0 × 30 cm (diameter × length) packed with Sephadex G-15. As a result of the separation, two low-molecular fractions were obtained - a fraction with a molecular mass above 1.5 kDa and a fraction corresponding to a mass below 1.5 kDa (subfraction S), which was lyophilized and used for further fractionation on a 2.5 × 120 cm Sephadex G-15 column. The resulting 6 subfractions (1, 2, 3, 4, 5, 6) were lyophilized and characterized. Fractions with the highest therapeutic potential were further fractionated on an Agilent Infinity 1260 chromatograph, using a 250 × 3 mm Knauer column packed with Eurospher II 100-3 C18A and equilibrated with 0.1% TFA in water. The 5-µl sample was eluted for 40 min at 0.1 ml/min with a 0.1% TFA solution in water and subjected to 30-min gradient separation: 0–50% water with 0.1% TFA and acetonitrile with 0.1% TFA at a 0.5 ml/min flow rate. Detection was conducted at 214 nm. The peaks with intensity above 200 µAu from 20 runs were collected, lyophilized, and analyzed with FTIR spectroscopy.
Cell cultures
The research was conducted on two cell lines: human normal colon epithelial cells CCD 841 CoTr (ATCC No. CRL-1807) and human colon cancer cells (stage I) HT-29 (ATCC No. HTB-38). The cells were cultured in RPMI 1640 medium (HT-29) or a mixture of DMEM and RPMI 1640 media (1:1)(CCD 841 CoTr), supplemented with 10% (v/v) FBS and antibiotics (100 U/mL penicillin, 100 μg/mL streptomycin), and kept at 37 °C (HT-29) or 34 °C (CCD 841 CoTr) in humidified atmosphere with 5% CO2.
Preparation of samples
Freeze-dried extracts were dissolved in RPMI medium to obtain a stock solution of 4 mg/mL. Subsequently, the extracts were dissolved in RPMI or RPMI:DMEM (1:1) with 2% (v/v) FBS to obtain desired concentrations.
Determination of proteins, carbohydrates, and phenolic compounds
Protein concentrations were determined using the Bradford reagent and bovine serum albumin as a standard27. The total content of the phenolic compounds was determined with diazosulfanilamide using the DASA test28, where absorbance was measured at 500 nm and vanillic acid was used as a standard. The total carbohydrate content was determined with the phenol-sulfuric acid assay using D-glucose as a standard29.
Antioxidant properties
DPPH free radical-scavenging test
The total antioxidant capacity of the sub-fractions was determined using the DPPH radical as a reagent, according to the procedure described by Paduch et al.30. This method is based on the ability of 1,1-diphenyl-2-picrylhydrazyl (DPPH) to decolorize in the presence of antioxidants. Subsequently, 100 μL of the test compound at concentrations ranging from 6.25 to 800 µg/mL were mixed with 100 μL of the DPPH solution (0.2 mg/mL in ethanol) and absorbance at 515 nm was determined after 2, 5, 10, 15, 20, and 30 min of incubation at room temperature. Trolox, i.e. the well-known standard with strong antioxidant activities, was used as a positive control. The results where expressed as trolox equivalents in mM per gram of dry analyte.
ABTS radical-scavenging test
The ABTS radical-scavenging activities of the fractions were determined according to the method proposed by van den Berg et al.31, Duo-Chuan32, and Re et al.33 with modification. The stock solution was prepared by dissolving 7.4 mM ABTS and 2.6 mM potassium persulfate in MQ water. After 16 h, the concentrated ABTS stock solution was diluted with phosphate buffered saline (PBS) pH 7.4 to absorbance recorded at 734 nm. Subsequently, 10 μL of the test compound at concentrations ranging from 6.25 to 800 μg/mL were mixed with 990 μL of the ABTS radical solution and absorbance was measured. The results where expressed as trolox equivalents in mM per gram of dry analyte.
Anticancer assay
MTT method
The method is based on the ability of mitochondrial succinate dehydrogenase to reduce yellow tetrazolium salt (MTT) to purple formazan crystals. Such activity is performed by live cells; therefore, absorbance is directly proportional to the quantity of live cells34,35.
After 96- h incubation of the cells in 96-well plates (100 μL of 1 × 105 cells/mL) with the studied extracts, 25 μL of 5 mg/mL MTT (Sigma) were added to each well. After 3 h, 100 μL of 10% SDS (Sigma) in 0.01 M HCl (POCH) were added and incubated for 24 h. The plates were read using a Microplate Reader and absorbance at 570 nm was measured (Molecular Devices Corporation; Menlo Park, CA, USA).
Apoptosis evaluation – Hoechst 33342/PI staining
Evaluation of apoptosis and necrosis in the cell cultures was performed by staining with DNA-intercalating fluorochromes – Hoechst 33342 (Sigma) and propidium iodide (PI) (Sigma)36. The morphological analysis was performed under a confocal microscope Axiovert 200 M (Zeiss, Jena, Germany) with a scanning head module LSM 5 PASCAL. Cells exhibiting blue fluorescence of nuclei (fragmented or with condensed chromatin) were interpreted as early apoptotic. Morphologically similar cells with pink fluorescence were defined as late apoptotic. Cells with pink fluorescence of whole nuclei were classified as necrotic.
The cultures were grown in Petri dishes (3 cm diameter); 2 mL of 1 × 105 cells/mL were poured to each dish. After 24 h incubation with the extracts, 5 μL of a Hoechst 33342 (0.4 mg/mL) and propidium iodide (0.5 mg/mL) mixture (2:3 ratio) were added. The samples were incubated for 5 min and then observed under a fluorescence microscope. At least 1000 cells in randomly selected areas were counted for each sample and the ratio of apoptosis and necrosis was calculated.
Wound assay
The cells were poured onto Petri dishes (3 cm diameter) and cultured until formation of a monolayer. Afterwards, wounds were made, the cells were washed twice with PBS, and fresh media with or without (control) the studied fractions were added. The cells were incubated for 24 h and stained according to the May-Grünwald-Giemsa method. One dish was stained immediately after wounding – the wound control. The images were taken under Olympus BX51 (Olympus Optical Co. Ltd, Japan) with an Olympus SC30 head module. The distance between the faces of migrating cells was measured using CellSans software and calculated in comparison to the control regarded as 100%.
Zymography
After the incubation with the studied extracts at 100 μg/mL, the supernatants above the cell cultures were collected. 20 μL of the supernatants were loaded on a 10% polyacrylamide gel with addition of 0.1% gelatin (POCH) and 0.001% SDS (Sigma). Electrophoresis was performed at 120 V (starting at 90 V, until the face reached the separating gel). Afterwards, the gels were washed twice (2 × 15 min) in renaturing buffer (2.5% Triton X-100 (Sigma) in 50 mM Tris-HCl (Sigma) followed by two 15-min washes in 50 mM Tris-HCl buffer. Subsequently, the gels were incubated in incubation buffer (5 mM CaCl2 in 50 mM Tris-HCl) at 37 °C for 24 h. After the incubation, the gels were stained with 0.15% Coomassie Brilliant Blue R-250 dissolved in methanol: acetic acid: glycerol: water (16:2:1:23, by vol.) for 30 min. and destained in methanol: acetic acid: glycerol: water (8:2:1:29, by vol.)37–39. Densitometric analysis was performed using Image Studio Lite software (LI-COR Biosciences).
FT-IR Spectroscopy Analysis of ex-LMS Samples
The analyses of ex-LMS subfractions were carried out using lyophilizates. FTIR spectroscopy was performed with a spectrometer (Thermo Scientific Nicolet 8700 A with FT Raman Nicolet NXR module) in the wavelength range 4000–400 cm−1.
Statistical analysis
All analyses were performed in at least 3 replications and the data were analyzed using GraphPad Prism ver. 5.01. The results are presented as mean ± SD. Statistical significance was evaluated with the one-way Anova test and post-hoc Dunnett’s test (all columns compared to the control).
Results
Our previous studies demonstrated that the extracellular fraction of secondary metabolites of less than 10 kDa had cytotoxic, antioxidant, and antimicrobial effects; therefore, further fractionation was performed in order to examine the individual subfractions22–25. In this study, we focused on selected properties of extracellular secondary metabolites with a molecular mass below 1.5 kDa. The separations of the starting fractions based on the molecular weight of their compounds yielded six separate formulations (S,1–6) differing in their qualitative composition and biological properties.
Characterization of the main biochemical properties of the extracellular low molecular subfractions
The analysis of the chemical composition of the obtained subfractions (1–6) showed the presence of protein (except for subfraction 3), sugars, and phenolic compounds (Table 1). The highest concentration of low molecular weight peptides was recorded for fraction 6 (31.8 μg/mL): it was about 10 times higher than for fraction 1–5. The highest concentration of total carbohydrates was recorded for fractions 4 and 5 (404.4 and 328.7 μg/mL, respectively). The concentration of phenolic compounds was similar in all the analyzed subfractions.
Table 1.
Chemical composition of the extracellular C. unicolor ≤ 1.5 kDa subfraction of low molecular weight metabolites (S) and its subfractions (1–6); yield of total carbohydrates, protein content, and concentration of phenolic compounds.
| Subfraction | S | 1 | 2 | 3 | 4 | 5 | 6 |
|---|---|---|---|---|---|---|---|
| Protein (μg/mL) | 10.9 ± 1.8 | 8.0 ± 0,4 | 1.9 ± 0.1 | 0 ± 0.0 | 2.3 ± 0.1 | 3.7 ± 0.2 | 31.8 ± 0.5 |
| Total carbohydrates (μg/mL) | 220.2 ± 2.8 | 253.8 ± 31.4 | 287.2 ± 44.1 | 215.5 ± 11.1 | 404.4 ± 30.5 | 328.7 ± 33.6 | 190.4 ± 44.2 |
| Total phenolic compounds (μg/mL) | 55.4 ± 1.3 | 64.7 ± 0.2 | 50.5 ± 1.5 | 57.9 ± 1.2 | 61.5 ± 2.1 | 57.8 ± 1.1 | 73.5 ± 1.1 |
Antioxidant properties of the extracellular low molecular subfractions
The antioxidant capacity of the subfractions prepared was evaluated in the study (Table 2). All subfractions showed antioxidant activity, but the highest values were recorded for subfraction 6: these were 569.8 mM/g for the ABTS method and 168.4 mM/g for the DPPH method. The weakest antioxidant properties were found for the fraction corresponding to a mass below 1.5 kDa (subfraction S) and subfraction 3.
Table 2.
Radical scavenging effects of the extracellular ≤ 1.5 kDa subfraction of low molecular weight metabolites (S) from C. unicolor and its subfractions (1–6) assessed with the ABTS and DPPH radical-scavenging method and expressed as trolox equivalents [mM/g].
| Subfraction | S | 1 | 2 | 3 | 4 | 5 | 6 |
|---|---|---|---|---|---|---|---|
| ABTS radical-scavenging | 176.2 ± 0.4 | 308.8 ± 1.1 | 243.1 ± 0.1 | 169.0 ± 0.1 | 239.3 ± 1.0 | 303.0 ± 0.6 | 569.8 ± 3.8 |
| DPPH radical-scavenging | 52.0 ± 0.7 | 117.7 ± 7.2 | 119.5 ± 5.3 | 0 ± 0.2 | 91.5 ± 5.1 | 119.6 ± 4.3 | 168.4 ± 3.2 |
Data are means ± SD of three measurements (n = 3).
Inhibition of proliferation
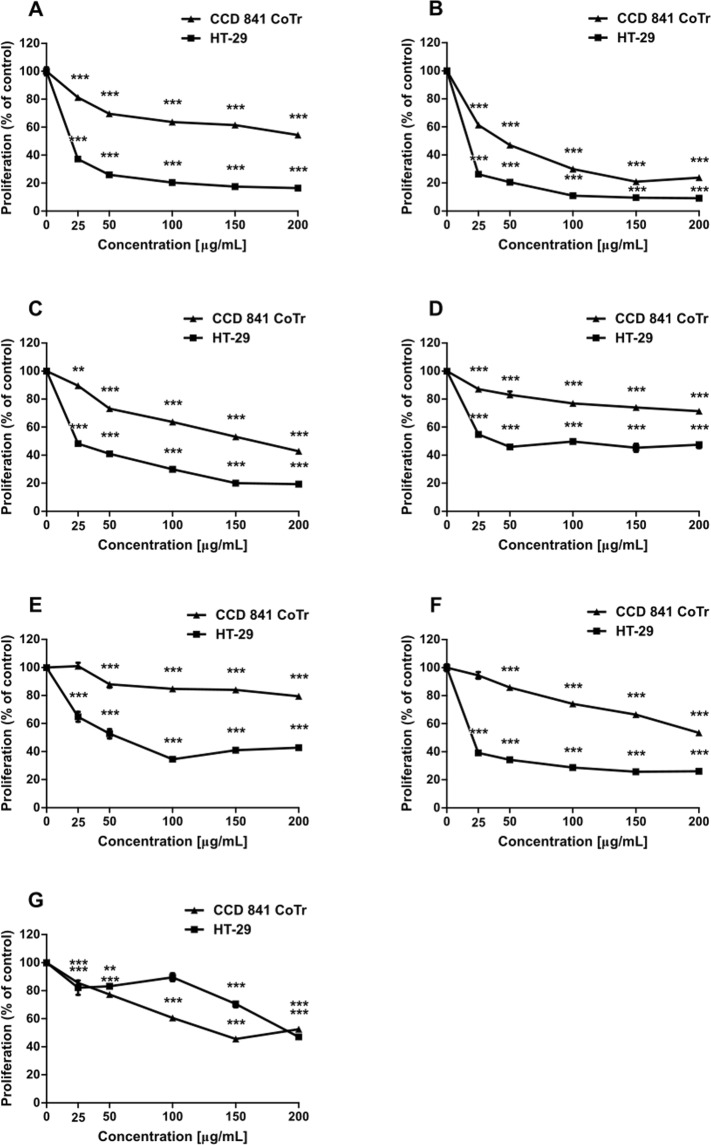
All subfractions inhibited cell proliferation in a dose-dependent manner. Furthermore, except for 6, the activity was stronger towards cancer cells, as the proliferation of the HT-29 cells was reduced to 16.5, 9.2, 19.4, 47.5, 42.7, and 26.2% of the control by fractions S, 1, 2, 3, 4, and 5 at 200 μg/mL, respectively (Fig. 1A–F). Subfraction 6 at concentrations of 25 μg/mL and 200 μg/mL had a stronger effect on the cancer cells (proliferation reduced to 82.2% and 47.1% of the control, respectively) than on the normal cells (proliferation inhibited to 85.6% and 52.6%, compared to the control, respectively) (Fig. 1G). In turn, at the concentrations of 50, 100, and 150 μg/mL, a greater effect was exerted on the CCD 841 CoTr cells. The most desired activity was exhibited by subfractions S, 3, 4, and 5, i.e. the proliferation of the HT-29 cells was inhibited to the greatest extent, while their effect on the CCD 841 CoTr cells was the mildest (Fig. 1A, D–F).
Figure 1.
Inhibition of proliferation of CCD 841 CoTr and HT-29 cells by fractions S (A), 1 (B), 2 (C), 3 (D), 4 (E), 5 (F), and 6 (G) measured with the MTT method. Values are expressed as a percentage of the control regarded as 100%; *p < 0.01, **p < 0.005, ***p < 0.001, one-way Anova, Dunnett’s test.
Induction of apoptosis
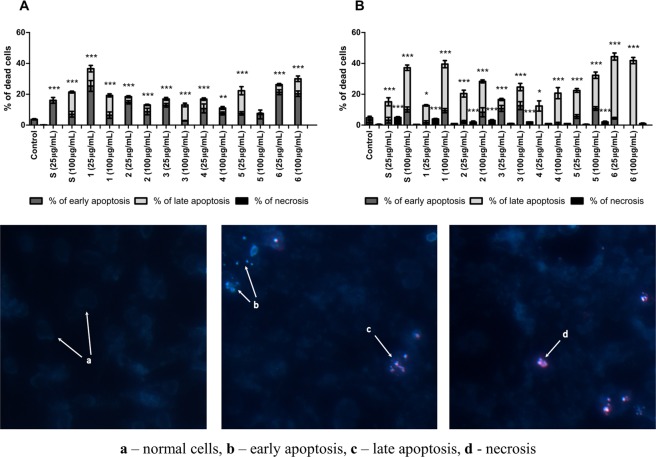
The microscopic observations revealed that all extracts induced programmed cell death (PCD), i.e. apoptosis, with no or a significantly low level of necrosis in both cell lines at the same time. There was a correlation between the apoptosis phase (early/late) and the cell line. Early apoptosis was dominant in the CCD 841 CoTr cell line, while the late phase dominated in HT-29. Among the extracts at 25 μg/mL, the highest level of programmed cell death in HT-29 was induced by subfraction 6 (44.4%) (Fig. 2B), while the rate of apoptosis in CCD 841 CoTr was significantly lower (26.2%) (Fig. 2A). In turn, at 100 μg/mL, the highest level of PCD was also induced by subfraction 6 in HT-29 (41.9%) and in CCD 841 CoTr (30.1%) (Fig. 2). All fractions at 100 μg/mL exhibited significantly greater pro-apoptotic activity in the HT-29 cell line, compared to the level of PCD in CCD 841 CoTr. All results were compared to the control in both cell lines. The control exhibited a significantly low level of apoptosis and necrosis (3.8% and 4.7% of apoptosis in CCD 841 CoTr and HT-29 cells, respectively, and 0.3% and 0.4% of necrosis in CCD 841 CoTr and HT-29 cells, respectively). The highest pro-apoptotic activity towards HT-29 cells was exhibited by subfractions S, 1, 5, and 6. All variants, except for 1 and 4 at 25 μg/mL, exerted a stronger pro-apoptotic effect on cancer cells than on normal cells.
Figure 2.
Ratio of apoptosis (early and late) and necrosis in CCD 841 CoTr (A) and HT-29 (B) cells induced by the studied fractions (upper panel) and representation of particular effects in cells: (a) normal cells, no effect, (b) early apoptosis, (c) late apoptosis, and (d) necrosis.
Inhibition of migration
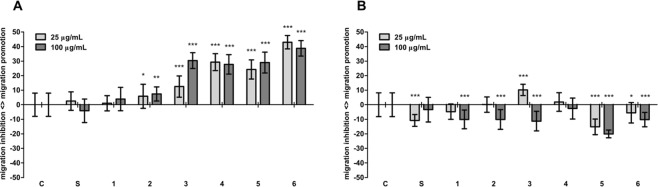
Inhibition of cancer cell migration may indicate anti-metastatic properties. Except for subfraction 3 and 4 at 25 μg/mL, all subfractions inhibited migration of cancer cells by 2.5 and 3.4% (crude subfraction S), 4.8 and 10.1% (subfraction 1), 0.1 and 10.1% (subfraction 2), 11.2% (subfraction 3), 2.6% (subfraction 4), 15.2 and 20.1% (subfraction 5), and 5.5 and 10.3% (subfraction 6), compared to the control, at 25 and 100 μg/mL, respectively (Figs. 3B and 4 bottom panel). In contrast, all subfractions promoted migration of normal cells (Figs. 3A and 4 upper panel). Although subfraction S at 100 μg/mL inhibited migration of normal cells, the effect was mild (4.1% of inhibition, compared to the control) and not statistically significant. The most potent activity was exhibited by subfractions 5 and 6. They inhibited migration of HT-29 cells and promoted migration of CCD 841 CoTr cells to the greatest extent. The effect on cancer cells did not exceed 20% of inhibition, but it must be noted that the HT-29 cells originated from stage I cancer.
Figure 3.
Effect of the fractions on the migration of CCD 841 CoTr (A) and HT-29 cells (B) (wound assay); *p < 0.01, **p < 0.005, ***p < 0.001, one-way Anova, Dunnett’s test.
Figure 4.
Effect of the fractions on the migration of CCD 841 CoTr (upper panel) and HT-29 (bottom panel) cells – wound assay. The width of the control wound is indicated with lines in each picture. The measurement bar indicates 200 μm.
Zymography
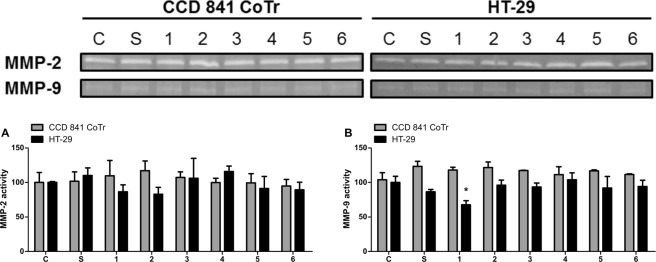
In a majority of cases, the zymographic analysis revealed no statistically significant influence of the subfractions on the activity of matrix metalloproteinases (MMPs) 2 and 9 secreted by the cells (Fig. 5). Despite the evident changes in the MMP activity in each variant, only subfraction 1 exerted a statistically significant effect towards the HT-29 cells, i.e. a decrease in the MMP-9 activity to 67.6% of the control (Fig. 5B).
Figure 5.
Activity of MMP-2 and MMP-9 secreted by CCD 841 CoTr and HT-29 cells incubated with the studied extracts at 100 μg/mL evaluated with zymography; the activity was measured densitometrically and presented in comparison to the control (regarded as 100%); *p < 0.01, **p < 0.005, ***p < 0.001, one-way Anova, Dunnett’s test.
After analysis of the zymography gels, another band with molecular mass <50 kDa was observed. The densitometric analysis revealed that all subfractions promoted the activity of the protease with this particular mass secreted by CCD 841 CoTr. The strongest effect was exerted by subfraction 6 (to 128.9%, compared to the control) (Supplementary, Figs. S1A and S2), and it was the only statistically significant result in the case of normal cells. These results correspond with the migration assay, where the strongest migration induction of CCD 841 CoTr was exhibited by subfraction 6 (Fig. 3A). The activity of this protease secreted by the HT-29 cells was inhibited by subfractions 2, 3, 4, 5, and 6, but only subfraction 4 had a statistically significant effect, i.e. a decrease in the activity to 40.9% of the control.
FT-IR Spectroscopy Analysis
Fraction 5 was selected for the FT-IR analysis due to its antiproliferative effect on the HT-29 cells and at the same time the weakest effect on the normal cell line (CCD 841 CoTr) out of the studied fractions. The comparison of the FT-IR spectra of the HPLC-purified subfraction 5 components against the Pharmaceuticals, Drugs and Antibiotics Spectra database St. JAPAN indicates the presence of compounds derived from hydroquinone, resorcinol, salicylaldehyde, and p-methoxyphenol.
Discussion
Colon cancer is the fourth most commonly diagnosed and the third most lethal cancer in the world. Despite intense efforts to develop treatments, effective measures are still not available. Therefore, extracts from natural products are still a very promising source of new anticancer drugs. Mushrooms are known as one of the most productive groups of useful natural products, including anticancer compounds. White rot fungi release a broad spectrum of low-molecular metabolites to the substrate, which not only support the decomposition of lignocellulosic material, but also have interesting health-promoting properties. In previous studies, we have shown that C. unicolor is an interesting fungus due to the unique properties of the laccase produced by this species as well as the interesting properties of extracellular secondary metabolites, which are a source of a waste product generated during the preparation of enzymes22–25,40.
Our previous analyses showed that the scavenging ability of the extracellular low molecular weight preparation was the same or even higher than that of the model antioxidant substances, i.e. trolox and ascorbic acid. The activity of this fraction determined using DPPH in the concentration range of 6.25–800 μg/mL ranged from 10% to 59% of free radical scavenging, but was at the level of 20% to 90% in an ABTS test22. In this study, we have also found that the six new low molecular weight preparations from C. unicolor have antioxidant properties. The antioxidative properties of some fungal species were also identified by other authors using the same determination methods. Kim et al. showed that edible fungi Pleurotus ostreatus, Agaricus bisporus, Pleurotus eryngii, and Lentinus edodes and medicinal mushrooms Agaricus blazei, Phellinus linteus, Ganoderma lucidum, and Inonotus obliquus had antioxidant activity and free radical scavenging ability, as demonstrated by the DPPH assay11,41. The white rot fungus belongs to the group of medical fungi characterized by slightly higher antioxidant activity, compared to edible fungi41. Other authors have described isolation of polyphenols with antioxidant properties from Inonotus xeranticus and Phellinus linteus42 as well as Ramaria flava10. The antioxidant profile of the studied preparations is closely related to the content of phenolic compounds and -OH groups of carbohydrates. Subfraction 6 showed the highest concentration of phenolic compounds among all the studied subfractions (73.5 μg/mL) and had the best antioxidant properties at the same time. To the best of our knowledge, these are the first studies showing that the byproduct obtained during isolation of laccase from the C. unicolor fungus is such an interesting source of antiproliferative substances in relation to colon cancer cells. Our results showed that apoptosis was gradually enhanced with an increase in the concentration of the subfractions. All fractions at 100 μg/mL exhibited significantly greater pro-apoptotic activity in the HT-29 cell line in relation to the level of PCD in CCD 841 CoTr. Similar results were obtained by Lim et al., who studied the effect of hispidin isolated from P. linteus on colon cancer cells, i.e., CMT-93 and HCT 116. They reported anticancer activities of the phenolic compound hispidin from the medicinal fungus P. linteus, which were reflected in induction of apoptosis in colon cancer cells. They found that hispidin inhibited cell viability in a dose-dependent manner43. Lee et al. reported anticancer activities of cordycepin isolated from Cordyceps militaris through induction of apoptosis in human HT-29 colon cancer cells44. Doskocil et al. examined mycelial extracts for cytotoxic properties against colon cancer cell line HT-29 and human Caco-2 colon adenoma cells. White rot fungi Daedalea quercina, Merulius tremellosus, Piptoporus betulinus, Pycnoporus cinnabarinus, Lentinula edodes, and Ganoderma lucidum showed cytotoxicity towards HT-29 tumor cells expressed as EC50 of 60, 186, 73, 31, and 302.115 μg/mL, respectively45. In their studies, these authors used isolated substances from mycelia or full ethanol extracts obtained from the mycelium of selected species, while our research was conducted on the hitherto untested culture fluid. In this work, we also demonstrated that, except for subfractions 3 and 4, the other subfractions inhibited the migration of cancer cells, favoring the migration of normal cells, which may indicate anti-metastatic properties. Therefore, the studied preparations may prevent invasion of adjacent tissues by cancer cells at early stages and prevent metastasis before the cells acquire the ability to move and release from the primary tumor to reach distant niches.
Metalloproteinases of the extracellular matrix (MMPs) and their tissue inhibitors play an important role in the process of colon carcinogenesis. One of the metalloproteinases with significant importance in the development of colon cancer is MMP-946. Colon cancer cells have been shown to be able to synthesize metalloproteinases, including MMP-9, a proteolytic enzyme capable of digesting type IV collagen - the main component of the basal membrane. The degradation of type IV collagen by MMP-9 is found in both invasion and metastasis47. Similarly, Wu et al. showed that the expression of MMP-2 and MMP-9 was significantly higher in colon cancer tissues48. The zymographic analyses carried out in this work showed some effect of the subfractions on the activity of matrix metalloproteinase (MMP) 9 secreted by the cells. The studied subfractions promoted the migration of the normal cells, while inhibiting the process in the HT-29 cells. Furthermore, subfraction 1 exhibited strong inhibition of the activity of MMP-9 secreted by the HT-29 cells. The results suggest that inhibition or promotion of migration of both CCD 841 CoTr and HT-29 cells may be dependent on the activity of MMPs, as well as other factors engaged in cancer progression. Furthermore, another band with molecular weight ~50 kDa was observed. The activity of this protease corresponded to the migration assay. A protease with such molecular mass may be another MMP (i.e. MMP-14) regulated by MMP-249,50 or another form of MMP-9. Active MMP-9 may appear in the form of 82 kDa, but also ~65 or ~50 kDa truncated forms. It seems that low-mass forms of MMP-9 are resistant to TIMP-1 inhibition, thus the decrease in the activity of low-mass MMP by the studied subfractions is a desired result51,52.
The FT-IR analysis of subfraction 5, which acted pro-apoptotically on the tumor cells and affected the normal cells to a smallest extent, performed in the last stage of the study showed the presence of compounds that were relatively similar in their spectra to hydroquinone, resorcinol, salicylaldehyde, and p-methoxyphenol, e.g. phenolic substances with antioxidant activity. This result confirms previous analyses of the presence of phenolic compounds and their antioxidant properties.
To sum up the presented results, it can be concluded that the preparations isolated from the LMS fraction produced by C. unicolor can induce apoptosis in human HT-29 colon cancer cells and prevent metastasis. These facts suggest that these substances, obtained and characterized for the first time in this work, should be further evaluated as potential therapeutic and/or preventive agents in human colon cancer.
Supplementary information
Gelatin zymography analysis of enzymatic activity of matrix metalloproteinases MMP-2, MMP-9 and ~50kDa MMP
Author Contributions
A.M., D.S., M.J. contributed to the conception and design of the study, contributed to the experimental part of the study, and wrote the first draft of the manuscript; M.P. and A.Z. wrote sections of the manuscript, performed the statistical analysis, and contributed to the experimental part of the study. Ł.M., R.B. contributed to the manuscript preparation; I.C., M.G. contributed to the experimental part of the study. R.P. meritorical consultations and wrote sections of the manuscript. All authors contributed to the revision of the manuscript, read, and approved the submitted version.
Competing Interests
The authors declare no competing interests.
Footnotes
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Anna Matuszewska, Email: anna.matuszewska@poczta.umcs.lublin.pl.
Dawid Stefaniuk, Email: dawid.stefaniuk@umcs.pl.
Electronic supplementary material
Supplementary information accompanies this paper at 10.1038/s41598-018-37947-z.
References
- 1.Elmastas M, Isildak O, Turkekul I, Temur N. Determination of antioxidant activity and antioxidant compounds in wild edible mushrooms. J. Food Compos. Anal. 2007;20:337–345. doi: 10.1016/j.jfca.2006.07.003. [DOI] [Google Scholar]
- 2.Grangeia C, Heleno SA, Barros L, Martins A, Ferreira IC. Effects of trophism on nutritional and nutraceutical potential of wild edible mushrooms. Food Res. Int. 2011;44:1029–1035. doi: 10.1016/j.foodres.2011.03.006. [DOI] [Google Scholar]
- 3.Cheung PCK. Mini-review on edible mushrooms as source of dietary fiber: Preparation and health benefits. Food Sci. Hum. Wellness. 2013;2:162–166. doi: 10.1016/j.fshw.2013.08.001. [DOI] [Google Scholar]
- 4.Palacios I, et al. Antioxidant properties of phenolic compounds occurring in edible mushrooms. Food Chem. 2011;128:674–678. doi: 10.1016/j.foodchem.2011.03.085. [DOI] [Google Scholar]
- 5.Muszynska B, et al. Edible mushrooms in prophylaxis and treatment of human diseases. Med. Int. Rev. 2013;101:170–183. [Google Scholar]
- 6.Patel S, Goyal A. Recent developments in mushrooms as anti-cancer therapeutics: a review. 3 Biotech. 2012;2:1–15. doi: 10.1007/s13205-011-0036-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Roupas P, Keogh J, Noakes M, Margetts C, Taylor P. The role of edible mushrooms in health: Evaluation of the evidence. J. Funct. Foods. 2012;4:687–709. doi: 10.1016/j.jff.2012.05.003. [DOI] [Google Scholar]
- 8.Ćilerdžić JL, et al. Neuroprotective Potential and Chemical Profile of Alternatively Cultivated Ganoderma lucidum Basidiocarps. Chem. Biodivers. 2018;15:e1800036. doi: 10.1002/cbdv.201800036. [DOI] [PubMed] [Google Scholar]
- 9.Duru ME, Çayan GT. Biologically active terpenoids from mushroom origin: a review. Rec. Nat. Prod. 2015;9:456. [Google Scholar]
- 10.Liu K, Wang J, Zhao L, Wang Q. Anticancer, antioxidant and antibiotic activities of mushroom Ramaria flava. Food Chem. Toxicol. 2013;58:375–380. doi: 10.1016/j.fct.2013.05.001. [DOI] [PubMed] [Google Scholar]
- 11.Ćilerdžić J, Vukojević J, Stajić M, Stanojković T, Glamočlija J. Biological activity of Ganoderma lucidum basidiocarps cultivated on alternative and commercial substrate. J. Ethnopharmacol. 2014;155:312–319. doi: 10.1016/j.jep.2014.05.036. [DOI] [PubMed] [Google Scholar]
- 12.Sliva D. Ganoderma lucidum (Reishi) in Cancer Treatment. Integr. Cancer Ther. 2003;2:358–364. doi: 10.1177/1534735403259066. [DOI] [PubMed] [Google Scholar]
- 13.Chen H-P, et al. Four new sesquiterpenoids from fruiting bodies of the fungus Inonotus rickii. J. Asian Nat. Prod. Res. 2014;16:581–586. doi: 10.1080/10286020.2014.925451. [DOI] [PubMed] [Google Scholar]
- 14.Wang S, et al. Isolation, Identification, and Bioactivity of Monoterpenoids and Sesquiterpenoids from the Mycelia of Edible Mushroom Pleurotus cornucopiae. J. Agric. Food Chem. 2013;61:5122–5129. doi: 10.1021/jf401612t. [DOI] [PubMed] [Google Scholar]
- 15.Ferreira CFRI, A Vaz J, Vasconcelos MH, Martins A. Compounds from wild mushrooms with antitumor potential. Anti-Cancer Agents Med. Chem. Former. Curr. Med. Chem.-Anti-Cancer Agents. 2010;10:424–436. doi: 10.2174/1871520611009050424. [DOI] [PubMed] [Google Scholar]
- 16.Valverde, M. E., Hernández-Pérez, T. & Paredes-López, O. Edible mushrooms: Improving human health and promoting quality life. International Journal of Microbiology, 10.1155/2015/376387 (2015). [DOI] [PMC free article] [PubMed]
- 17.Chowdhury MMH, Kubra K, Ahmed SR. Screening of antimicrobial, antioxidant properties and bioactive compounds of some edible mushrooms cultivated in Bangladesh. Ann. Clin. Microbiol. Antimicrob. 2015;14:8. doi: 10.1186/s12941-015-0067-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kawagishi H, Zhuang C, Yunoki R. Compounds for dementia from Hericium erinaceum. Drugs Future. 2008;33:149. doi: 10.1358/dof.2008.033.02.1173290. [DOI] [Google Scholar]
- 19.Barros L, Pereira C, Ferreira ICFR. Optimized analysis of organic acids in edible mushrooms from Portugal by ultra fast liquid chromatography and photodiode array detection. Food Anal. Methods. 2013;6:309–316. doi: 10.1007/s12161-012-9443-1. [DOI] [Google Scholar]
- 20.Ferlay J, et al. Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. Int. J. Cancer. 2015;136:E359–E386. doi: 10.1002/ijc.29210. [DOI] [PubMed] [Google Scholar]
- 21.Gill S, et al. Pooled analysis of fluorouracil-based adjuvant therapy for stage II and III colon cancer: who benefits and by how much? J. Clin. Oncol. 2004;22:1797–1806. doi: 10.1200/JCO.2004.09.059. [DOI] [PubMed] [Google Scholar]
- 22.Jaszek, M. et al. New bioactive fungal molecules with high antioxidant and antimicrobial capacity isolated from Cerrena unicolor idiophasic cultures. BioMed Res. Int. 2013 (2013). [DOI] [PMC free article] [PubMed]
- 23.Mizerska-Dudka M, et al. Fungus Cerrena unicolor as an effective source of new antiviral, immunomodulatory, and anticancer compounds. Int. J. Biol. Macromol. 2015;79:459–468. doi: 10.1016/j.ijbiomac.2015.05.015. [DOI] [PubMed] [Google Scholar]
- 24.Statkiewicz, M. et al. Antimelanomic effects of high- and low-molecular weight bioactive subfractions isolated from the mossy maze mushroom, Cerrena unicolor (Agaricomycetes). Int. J. Med. Mushrooms19 (2017). [DOI] [PubMed]
- 25.Matuszewska A, Jaszek M, Stefaniuk D, Ciszewski T, Matuszewski Ł. Anticancer, antioxidant, and antibacterial activities of low molecular weight bioactive subfractions isolated from cultures of wood degrading fungus Cerrena unicolor. PLOS ONE. 2018;13:e0197044. doi: 10.1371/journal.pone.0197044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Janusz G, et al. Cloning and characterization of a laccase gene from biotechnologically important basidiomycete Cerrena unicolor. J. Fac. Agric. Kyushu Univ. 2012;57:41–49. [Google Scholar]
- 27.Bradford MM. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976;72:248–254. doi: 10.1016/0003-2697(76)90527-3. [DOI] [PubMed] [Google Scholar]
- 28.Malarczyk, E. Transformation of phenolic acids by Nocardia. Acta Microbiol. Pol. 1 (1989).
- 29.Dubois M, Gilles KA, Hamilton JK, Rebers PT, Smith F. Colorimetric method for determination of sugars and related substances. Anal. Chem. 1956;28:350–356. doi: 10.1021/ac60111a017. [DOI] [Google Scholar]
- 30.Paduch R, et al. Lamium Album Extracts Express Free Radical Scavenging and Cytotoxic Activities. Pol. J. Environ. Stud. 2008;17:569–580. [Google Scholar]
- 31.van den Berg R, Haenen GRMM, van den Berg H, Bast A. Applicability of an improved Trolox equivalent antioxidant capacity (TEAC) assay for evaluation of antioxidant capacity measurements of mixtures. Food Chem. 1999;66:511–517. doi: 10.1016/S0308-8146(99)00089-8. [DOI] [Google Scholar]
- 32.Duo-Chuan L. Review of Fungal Chitinases. Mycopathologia. 2006;161:345–360. doi: 10.1007/s11046-006-0024-y. [DOI] [PubMed] [Google Scholar]
- 33.Re R, et al. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. Med. 1999;26:1231–1237. doi: 10.1016/S0891-5849(98)00315-3. [DOI] [PubMed] [Google Scholar]
- 34.Mosmann T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods. 1983;65:55–63. doi: 10.1016/0022-1759(83)90303-4. [DOI] [PubMed] [Google Scholar]
- 35.van de Loosdrecht AA, Beelen RHJ, Ossenkoppele GJ, Broekhoven MG, Langenhuijsen MMAC. A tetrazolium-based colorimetric MTT assay to quantitate human monocyte mediated cytotoxicity against leukemic cells from cell lines and patients with acute myeloid leukemia. J. Immunol. Methods. 1994;174:311–320. doi: 10.1016/0022-1759(94)90034-5. [DOI] [PubMed] [Google Scholar]
- 36.Jakubowicz-Gil J, Langner E, Bądziul D, Wertel I, Rzeski W. Quercetin and sorafenib as a novel and effective couple in programmed cell death induction in human gliomas. Neurotox. Res. 2014;26:64–77. doi: 10.1007/s12640-013-9452-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Bai L, Charvin G, Siggia ED, Cross FR. Nucleosome-depleted regions in cell-cycle-regulated promoters ensure reliable gene expression in every cell cycle. Dev. Cell. 2010;18:544–555. doi: 10.1016/j.devcel.2010.02.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Ren, Z., Chen, J. & Khalil, R. A. Zymography as a research tool in the study of matrix metalloproteinase inhibitors. In Zymography 79–102, 10.1007/978-1-4939-7111-4_8 (Humana Press, New York, NY, 2017). [DOI] [PMC free article] [PubMed]
- 39.Paszczyński A, Miedziak I, Lobarzewski J, Kochmańska J, Trojanowski J. A simple method of affinity chromatography for the purification of glucoamylase obtained from Aspergillus niger C. FEBS Lett. 1982;149:63–66. doi: 10.1016/0014-5793(82)81072-7. [DOI] [PubMed] [Google Scholar]
- 40.Matuszewska A, et al. Laccase purified from Cerrena unicolor exerts antitumor activity against leukemic cells. Oncol. Lett. 2016;11:2009–2018. doi: 10.3892/ol.2016.4220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Kim M-Y, et al. Phenolic compound concentration and antioxidant activities of edible and medicinal mushrooms from Korea. J. Agric. Food Chem. 2008;56:7265–7270. doi: 10.1021/jf8008553. [DOI] [PubMed] [Google Scholar]
- 42.Jung J-Y, et al. Antioxidant polyphenols from the mycelial culture of the medicinal fungi Inonotus xeranticus and Phellinus linteus. J. Appl. Microbiol. 2008;104:1824–1832. doi: 10.1111/j.1365-2672.2008.03737.x. [DOI] [PubMed] [Google Scholar]
- 43.Lim J-H, Lee Y-M, Park SR, DA HYE K, Lim BO. Anticancer activity of hispidin via reactive oxygen species-mediated apoptosis in colon cancer cells. Anticancer Res. 2014;34:4087–4093. [PubMed] [Google Scholar]
- 44.Lee SY, Debnath T, Kim S-K, Lim BO. Anti-cancer effect and apoptosis induction of cordycepin through DR3 pathway in the human colonic cancer cell HT-29. Food Chem. Toxicol. 2013;60:439–447. doi: 10.1016/j.fct.2013.07.068. [DOI] [PubMed] [Google Scholar]
- 45.Doskocil I, et al. In vitro immunomodulatory activity, cytotoxicity and chemistry of some central European polypores. Pharm. Biol. 2016;54:2369–2376. doi: 10.3109/13880209.2016.1156708. [DOI] [PubMed] [Google Scholar]
- 46.Groblewska M, Mroczko B, Szmitkowski M. The role of selected matrix metalloproteinases and their inhibitors in colorectal cancer development. Postepy Hig. Med. Doswiadczalnej Online. 2010;64:22–30. [PubMed] [Google Scholar]
- 47.Murray D, Morrin M, Mcdonnell S. Increased invasion and expression of MMP-9 in human colorectal cell lines by a CD44-dependent mechanism. Anticancer Res. 2004;24:489–494. [PubMed] [Google Scholar]
- 48.Wu W, He JT, Ruan JD, Wang RB, Zhang YD. Expression of MMP-2, MMP-9 and collagen type IV and their relationship in colorectal carcinomas. Xi Bao Yu Fen Zi Mian Yi Xue Za Zhi Chin. J. Cell. Mol. Immunol. 2008;24:908–909. [PubMed] [Google Scholar]
- 49.Stanton H, et al. The activation of ProMMP-2 (gelatinase A) by HT1080 fibrosarcoma cells is promoted by culture on a fibronectin substrate and is concomitant with an increase in processing of MT1-MMP (MMP-14) to a 45 kDa form. J. Cell Sci. 1998;111:2789–2798. doi: 10.1242/jcs.111.18.2789. [DOI] [PubMed] [Google Scholar]
- 50.Guo, C. & Piacentini, L. Type I collagen-induced MMP-2 activation coincides with up-regulation of MT1-MMP and TIMP-2 in cardiac fibroblasts. J. Biol. Chem. (2003). [DOI] [PubMed]
- 51.Bellini T, et al. Matrix metalloproteinase-9 activity detected in body fluids is the result of two different enzyme forms. J. Biochem. (Tokyo) 2012;151:493–499. doi: 10.1093/jb/mvs014. [DOI] [PubMed] [Google Scholar]
- 52.Vandooren J, Steen PEVden, Opdenakker G. Biochemistry and molecular biology of gelatinase B or matrix metalloproteinase-9 (MMP-9): The next decade. Crit. Rev. Biochem. Mol. Biol. 2013;48:222–272. doi: 10.3109/10409238.2013.770819. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Gelatin zymography analysis of enzymatic activity of matrix metalloproteinases MMP-2, MMP-9 and ~50kDa MMP