Abstract
Purpose
Inherited retinal diseases (IRDs) are clinically and genetically heterogeneous showing progressive retinal cell death which results in vision loss. IRDs include a wide spectrum of disorders, such as retinitis pigmentosa (RP), Leber congenital amaurosis (LCA), cone-rod dystrophy (CRD), and Stargardt disease (STGD1).
Methods
In this study, we performed targeted next-generation sequencing based on molecular inversion probes (MIPs) that allowed the sequence analysis of 108 IRD-associated genes in 50 Iranian IRD probands.
Results
The sequencing and variant filtering led to the identification of putative pathogenic variants in 36 out of 50 (72%) probands. Among 36 unique variants, we identified 20 novel variants in 15 genes. Four out of 36 probands carry compound heterozygous variants, and 32 probands carry homozygous variants.
Conclusions
Employing a cost-effective targeted next-generation sequencing procedure, we identified the genetic causes of different retinal disorders in the majority of Iranian families in this study.
Introduction
Inherited retinal dystrophies (IRDs) are a group of clinically and genetically heterogeneous disorders leading to vision loss because of degeneration of retinal cells [1-5]. Mutations in more than 250 genes have been reported to cause IRDs. Most identified mutations are inherited in an autosomal recessive fashion (50–60%) although some can be autosomal dominant (30–40%), X-linked (2–20%), or mitochondrial [6]. IRDs are divided into non-syndromic forms, such as retinitis pigmentosa (RP), cone-rod dystrophy (CRD), Leber congenital amaurosis (LCA), and Stargardt disease (STGD1), as well as syndromic forms [7]. These inherited retinal dystrophies have an estimated incidence of 1:2,000 individuals [4].
The earliest clinical symptoms of RP (OMIM 268000) are night blindness and peripheral vision loss (tunnel vision) due to rod cell degeneration. In later stages of the disease, central vision can also be lost once cone cells degenerate, and eventually, patients with RP can become (legally) blind [6,8]. In CRD (OMIM 120970), the main manifestations are loss of visual acuity, color vision, and photophobia followed by night blindness and tunnel vision [9]. The rarest and most severe retinal dystrophy form is LCA (OMIM 204000), in which patients have severe visual impairment in the first year of life [10]. LCA appears with severe and early visual loss, nystagmus, and sluggish pupils [11]. STGD1 (OMIM 248200) is one of the most common macular degenerative disorders with the features of macular atrophy, impaired central vision, and decreased visual acuity. Color vision, especially a red-green defect, is apparent in most affected individuals [12].
Because of the complexity of IRDs, it is often necessary to reconcile the clinical diagnosis, family diagnosis, and molecular diagnosis. This can be done in a multidisciplinary team which ideally includes trained ophthalmologists, genetic counselors, and molecular geneticists. Thus, identification of the pathogenic DNA variants can help clinicians and health professionals to improve and expedite the diagnosis of the disease and provide patients and their families with more accurate genetic counseling. Moreover, increasing knowledge of the molecular mechanisms involved in IRD pathogenesis may also lead to the development of new treatment options for affected individuals in the future [13].
Since the introduction of next-generation sequencing (NGS), several different targeted gene panel tests that sequence a large set of disease-associated genes have been developed for specific inherited eye disorders [14]. In this study, we applied a targeted gene panel–based NGS approach based on molecular inversion probes (MIPs) to obtain a molecular diagnosis for 50 patients who had been diagnosed with IRDs and for whom no genetic tests had been performed previously. Using this method, variants in 108 genes associated with non-syndromic IRDs were screened.
Methods
Subjects
In this study, we included 50 IRD probands with Iranian origin and the clinical diagnosis of RP (n=31), CRD (n=5), LCA (n=11), or STGD1 (n=3) who had not been genetically investigated previously. The clinical diagnosis was established by a trained ophthalmologist including the patient pedigree and history of the disease, electroretinography (MonPack3; Metrovision, Perenchies, France), evaluation of visual acuity using slit-lamp biomicroscopy, and fundoscopy (TRC-50EX retinal camera; Topcon, Tokyo, Japan). Written informed consent was obtained from all study subjects. This study was approved by the institutional review board of the ethics committee of Isfahan Medical University, Iran. The study adhered to the tenets of the Declaration of Helsinki and the ARVO statement on human subjects was approved by the institutional review board of the ethics committee of Isfahan Medical University, Iran.
Targeted sequencing and genetic analysis
Five cc of peripheral blood samples of affected and unaffected members were collected and mixed with EDTA anticoagulant (Merck KGaA, Darmstadt, Germany). Genomic DNA of all samples was extracted using the phenol-chloroform method [15].
Targeted NGS was performed to identify the genetic etiology of IRDs in 50 patients with different types of IRDs. The 108 gene panel MIP analysis designed at the Department of Human Genetics at Radboud University Medical Center (Nijmegen, The Netherlands) consisted of genes implicated in non-syndromic achromatopsia, cone dystrophy (CD) and CRD (autosomal dominant, autosomal recessive, and X-linked), congenital stationary night blindness (autosomal recessive and X-linked), macular degeneration (autosomal recessive and autosomal dominant), LCA (autosomal dominant and autosomal recessive), RP (autosomal recessive, autosomal dominant, and X-linked), and choroideremia. MIPs with 5-bp molecular tags were used to perform targeted NGS of 108 genes associated with IRD (Appendix 1). The 1,524 coding exons and at least 10 bp flanking each exon were targeted using 6,129 MIPs for an overall target size of 647,574 bp. On average, four to six MIPs cover one exon. When known single nucleotide polymorphisms (SNPs) resided in the genomic regions complementary to the annealing or ligation arm, SNP-specific MIPs were designed. The panel also included the frequent LCA-associated pathogenic intronic variant c.2992+1655A>G in CEP290 (Gene ID 80184, OMIM 610142) [16], as well as five deep-intronic variants in ABCA4 (Gene ID 496442, OMIM 248200) [17]. Pooled and phosphorylated probes were added to the capture reactions with 100 ng of genomic DNA from each individual to produce a sequence library for each individual. The libraries were amplified with 21 cycles of PCR (first denaturation step at 95 ºC for 4 min, followed by 21 cycles at 94 ºC for 30 s, 57 ºC for 30 s, 72 ºC for 1 min and then a final extension step at 72 ºC for 5 min), during which an 8-bp sample barcode was introduced. The barcoded libraries were then pooled and purified with AMPPureXP beads (Beckman-Coulter, Indianapolis, IN). Sequencing was performed on an Illumina NextSeq 500 system (San Diego, CA). Demultiplexed BAM files were aligned to a human reference sequence (UCSC Genome Browser hg19) via the Burrows-Wheeler Aligner (BWA) v.0.6.2 [18]. In-house automated data analysis pipeline and variant interpretation tools were used for variant calling. Only non-synonymous single nucleotide variants (nsSNVs), nonsense variants, putative splice site (±10 bps) variants, insertions, duplications, and deletions represented by more than 20 sequence reads were considered for further analysis. In addition, variants with a minor allele frequency (MAF) >0.5% in the Exome Aggregation Consortium Database (ExAC) Version 0.3.1 [19] were excluded from further investigation, with the exception of a few frequent variants in ABCA4 (i.e., c.2588G>C, c.5882G>A, and c.5603A>T [20-25]) that were selected separately.
In silico predictions
Several criteria were taken into account to evaluate the potential pathogenicity of variants: 1) The variants had previously been reported to be pathogenic, 2) the variants were absent in the ExAC database or showed a low MAF (<0.5%), 3) the variants caused a loss-of-function (LOF) of the encoded protein, such as nonsense, canonical splice site, and frameshift mutations, and 4) missense variants were predicted to be damaging by in silico prediction algorithms. The pathogenicity prediction tools used for missense variants were PolyPhen-2, Sorting Intolerant From Tolerant (SIFT), PhyloP score, the Grantham score, Mutation Taster, and Combined Annotation Dependent Depletion (CADD) score assessment. For the selection of non-canonical splice site variants, in silico predictions were performed by using five algorithms (SpliceSiteFinder-like, MaxEntScan, NNSPLICE, GeneSplicer, and Human Splicing Finder) via Alamut Visual software version 2.7 [26–30], and Biosoftware (Interactive Biosoftware, Rouen, France), by comparing the splicing scores for wild-type and variant nucleotides.
Segregation analysis
Candidate variants were validated and tested for cosegregation within the families using Sanger sequencing. For this purpose, specific primers were designed for PCR amplification of fragments containing the identified candidate causal mutations in each patient. PCR amplicons were sequenced using Sanger sequencing on an ABI 3730 DNA analyzer (Cologne, Germany) with BigDye chemistry v3.1.
Results
Study population
Fifty unrelated Iranian patients with non-syndromic IRD were included in this study, and their parents were first (34 families) or second cousins (16 families). All clinical information for the 50 families is summarized in Table 1. Among the 50 patients, 31 have RP, five have CRD, 11 have LCA, and three have STGD1.
Table 1. Clinical information overview for 50 Iranian families with inherited retinal dystrophies.
| Diagnosis | Symptoms | Number of patients | Sex |
|
Mean age of onset (±SD) | Mean age (±SD) | Family history of IRDs |
|
|---|---|---|---|---|---|---|---|---|
| Male | Female | Yes | No | |||||
| RP |
NB, Tunnel vision, near blindness |
31 |
15 |
16 |
11.6±4.2 years |
31.5±6.3 years |
21 |
10 |
| CRD |
Photophobia, defect in color vision, near blindness |
5 |
1 |
4 |
11.7± 4.2 years |
31.7±6.3 years |
4 |
1 |
| LCA |
Nystagmus and total blindness |
11 |
7 |
4 |
9.6±2.08 months |
31.3±6.7 years |
7 |
4 |
| STGD1 | Photophobia, defect in color vision, difficulty in adaption to the dark after sunlight exposure, central vision loss | 3 | 0 | 3 | 11.2±5.1 years | 30.6±2.5 years | 2 | 1 |
NB: Night blindness, RP: Retinitis pigmentosa, CRD: Cone-rod dystrophy, LCA: Leber congenital amaurosis, STGD1: Stargardt disease, IRD: Inherited retinal dystrophies, SD: Standard deviation
Variant prioritization to identify causal variants
One affected individual from each of the 50 families was subjected to targeted NGS of 108 known IRD-associated genes (Appendix 1). The averages of 1.2 million reads on target per sample were obtained using our capture panel technology, with an average coverage of 213 reads per MIP. Moreover, an average of 88% of targeted regions had 10X coverage or more, which was sufficient for accurate variant calling. The pipeline initially called an average of 532 single nucleotide variants and 64 insertions or deletions for each sample. In this study, putative pathogenic variants were identified in 36 index cases (Table 2), and we could not find the pathogenic variants for the remaining 14 families. All putative disease-associated variants were validated with conventional Sanger sequencing, and segregation analysis was performed for all 36 families with identified pathogenic variants. Homozygosity was observed for 32/36 patients (88%), and four patients were compound heterozygous.
Table 2. Pathogenic variants identified by targeted next-generation sequencing in 36 Iranian probands affected with IRDs.
| Index ID | Gene | Refseq | Phenotype | Exon/Intron | Nucleotide change | Protein change | Zygosity | Reference |
|---|---|---|---|---|---|---|---|---|
| 066,592 |
ABCA4 |
NM_000350 |
STGD1 |
E1 |
c.1A>G |
p.(Met1?) |
Homo |
[31] |
| 066,595 |
ABCA4 |
NM_000350 |
STGD1 |
I43 |
c.6005+1G>A |
p.? |
Compound het |
[32] |
| 066,595 |
ABCA4 |
NM_000350 |
STGD1 |
E6 |
c.634C>T |
p.(Arg212Cys) |
Compound het |
[33] |
| 066,599 |
ABCA4 |
NM_000350 |
STGD1 |
E30 |
c.4462T>C |
p.(Cys1488Arg) |
Compound het |
[34] |
| 066,599 |
ABCA4 |
NM_000350 |
STGD1 |
E8 |
c.913C>T |
p.(Gln305*) |
Compound het |
[35] |
| 066,854 |
ABCA4 |
NM_000350 |
RP |
E46 |
c.6385A>G |
p.(Ser2129Gly) |
Homo |
[36] |
| 066,863 |
ABCA4 |
NM_000350 |
RP |
I20 |
c.3051–2A>G |
p.? |
Homo |
Novel |
| 066,876 |
ABCA4 |
NM_000350 |
RP |
I27 |
c.4128+1G>A |
p.? |
Homo |
Novel |
| 066,877 |
AIPL1 |
NM_014336 |
LCA |
E6 |
c.834G>A |
p.(Trp278*) |
Homo |
[37] |
| 066,872 |
BBS1 |
NM_024649 |
RP |
E5 |
c.479G>A |
p.(Arg160Gln) |
Homo |
[38] |
| 066,568 |
BBS2 |
NM_031885 |
RP |
E14 |
c.1705C>T |
p.(Gln569*) |
Compound het |
Novel |
| 066,568 |
BBS2 |
NM_031885 |
RP |
I1 |
c.117+1G>T |
p.? |
Compound het |
Novel |
| 066,574 |
BBS2 |
NM_031885 |
RP |
E2 |
c.224T>G |
p.(Val75Gly) |
Homo |
[39] |
| 066,891 |
CEP290 |
NM_025114 |
LCA |
E54 |
c.7304T>G |
p.(Val2435Gly) |
Homo |
Novel |
| 066,871 |
CERKL |
NM_001030311 |
RP |
E6 |
c.847C>T |
p.(Arg283*) |
Homo |
[40] |
| 066,889;066892 |
CERKL |
NM_001030311 |
CRD, RP |
E13 |
c.1453_1454insA |
p.(Pro485Hisfs*4) |
Homo |
Novel |
| 066,880 |
CRB1 |
NM_201253 |
RP |
E7 |
c.2234C>T |
p.(Thr745Met) |
Homo |
[41] |
| 066,886 |
CRB1 |
NM_201253 |
RP |
E2 |
c.548G>A |
p.(Cys183Tyr) |
Homo |
Novel |
| 066,607 |
GUCY2D |
NM_000180 |
LCA |
E12 |
c.2383C>T |
p.(Arg795Trp) |
Homo |
Novel |
| 066,858 |
GUCY2D |
NM_000180 |
LCA |
E4 |
c.1216G>C |
p.(Asp406His) |
Homo |
Novel |
| 066,855 |
LCA5 |
NM_001122769 |
RP |
E4 |
c.720+1G>A |
p.? |
Homo |
[42] |
| 066,852 |
LRAT |
NM_004744 |
RP |
E2 |
c.487C>G |
p.(His163Asp) |
Homo |
Novel |
| 066,853 |
LRAT |
NM_004744 |
RP |
E2 |
c.157_159dup |
p.(Val53dup) |
Homo |
Novel |
| 066,882 |
MERTK |
NM_006343 |
RP |
E2 |
c.390G>A |
p.(Trp130*) |
Homo |
[43] |
| 066,867 |
PDE6B |
NM_000283 |
RP |
E5 |
c.782_784del |
p.(Phe261del) |
Homo |
Novel |
| 066,884 |
PDE6C |
NM_006204 |
CRD |
E12 |
c.1612T>A |
p.(Phe538Ile) |
Homo |
Novel |
| 066,887 |
PDZD7 |
NM_001195263 |
RP |
E6 |
c.793A>G |
p.(Arg265Gly) |
Homo |
Novel |
| 066,865 |
PROM1 |
NM_006017 |
RP |
E25 |
c.2461C>T |
p.(Arg821*) |
Homo |
Novel |
| 066,859 |
RP1 |
NM_006269 |
RP |
E4 |
c.2749C>T |
p.(Gln917*) |
Homo |
Novel |
| 066,885 |
RP1 |
NM_006269 |
RP |
E4 |
c.1498_1499del |
p.(Met500Valfs*7) |
Compound het |
[44] |
| 066,885 |
RP1 |
NM_006269 |
RP |
E4 |
c.4105C>T |
p.(Gln1369*) |
Compound het |
[40] |
| 066,860 |
RPE65 |
NM_000329 |
RP |
E12 |
c.1299T>A |
p.(Tyr433*) |
Homo |
Novel |
| 066,864;066869 |
RPE65 |
NM_000329 |
RP |
E10 |
c.1102T>C |
p.(Tyr368His) |
Homo |
[45] |
| 066,879 |
RPE65 |
NM_000329 |
RP |
I4 |
c.354–1G>A |
p.? |
Homo |
Novel |
| 066,584;066866;066868 |
RPGRIP1 |
NM_020366 |
CRD, LCA |
E10 |
c.1306G>T |
p.(Ala436Ser) |
Homo |
Novel |
| 066,878 | SPATA7 | NM_018418 | RP | E6 | c.506del | p.(Thr169Lysfs*25) | Homo | Novel |
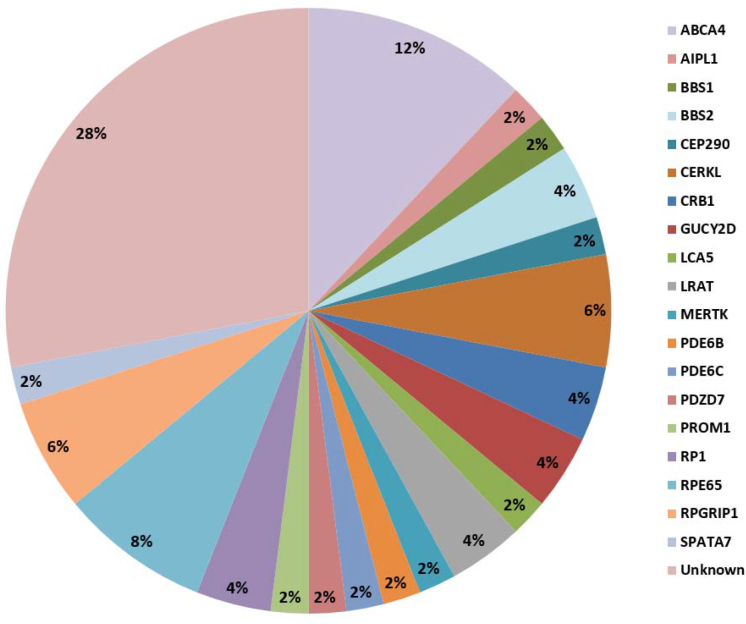
Overall, we identified 36 unique pathogenic variants based on the low allele frequency or absence of variants in ExAC, their high evolutionary conservation, the type of the amino acid substitutions, and the high predicted impacts of an amino acid substitution on the structure and function of a human protein in 19 genes. Of these, 20 were novel with the remaining 16 previously reported as IRD-causing variants [31-45]. Furthermore, 14 out of the 50 families remained genetically unexplained (Figure 1). Thus, the sensitivity of the gene panel for detection rate was 72%. The unique variants comprised 15 missense variants, nine nonsense variants, three deletions or insertions leading to a frameshift, six canonical splice site variants, two in-frame deletion and duplications, and one start loss. Pathogenicity was interpreted in accordance with different predictions (Table 3). According to the prediction tools in this study, all variants were annotated as pathogenic except one missense variant in ABCA4 (c.6385A>G; p.(Ser2129Gly)) that is predicted to be benign by PolyPhen-2 and tolerated by SIFT. However, this variant was predicted to be disease causing by MutationTaster and a high CADD score. As shown in Appendix 1, this variant is situated at the penultimate position of exon 46 and is part of the splice donor site consensus sequence. The canonical splice site nucleotides are GC, and therefore, the corresponding splice site was recognized only by SpliceSiteFinder-like. It predicted a strength of 91.8 (on a scale from 0 to 100) for the wild-type sequence and 80.3 for the mutant c.6385A>G, which is considered a large decrease in splice strength. Accordingly, we included the p.? in the predicted protein change. In addition, this homozygous variant has not been reported in ExAC.
Discussion
Although consanguineous marriages are common in the Iranian population [46], which makes the Iranian gene pool a valuable asset for genetic studies, little is known about inherited retinal degeneration in the Iranian community. Overall, 37.4% of Iranian marriages are consanguineous. Among them, 19.3% are first cousins and 18.1% second cousins [47]. Thus far, three small studies on genetic causes of Iranian patients with IRD have been reported [41,48,49].
Compared to the multiple targeted gene panel tests for different subtypes of IRDs that have been developed in the past few years with a detection rate of approximately 50–70% [50], we found similar percentages of pathogenic variants using our panel testing. Haer-Wigman et al. showed that using an in silico gene panel (based on whole exome sequencing) that was performed for 266 Dutch patients with visual impairment, in 52% of the cases, the genetic cause was identified [51]. However, in some studies the detection rate using panel testing was low. For instance, Fu et al. recruited 31 families with autosomal recessive RP and identified the pathogenic variant through a gene panel comprising 163 retinal disease genes. The authors achieved a detection rate of approximately 40% [52].
Variant c.6385A>G, apart from a predicted amino acid exchange, p.(Ser2129Gly), lowers the strength of a splice donor site of exon 46. A splice assay can shed light on its effect. Moreover, most of the variants detected in the patients are homozygous except four families that we identified as carrying compound heterozygous variants in RP1 (Gene ID 19888, OMIM 180100), ABCA4, and BBS2 (Gene ID 67378, OMIM 615981). The most frequently mutated gene was RPE65 (Gene ID 6121, OMIM 180069), mutated in 4/50 (8%) of the families (Figure 1). Sundaresan et al. reported that although the mutations in some genes such as RPE65 are a common cause of LCA in North America, they are rare in southern India [53]. To determine whether mutations in RPE65 are a common cause of RP in the Iranian population, we need to screen a larger cohort of RP probands in the future.
Our genetic findings, in particular the recurrence for variants in CERKL (Gene ID 375298, OMIM 608381), RPE65, and RPGRIP1 (Gene ID 57096, OMIM 605446) will aid genetic testing for IRD in the Iranian population. Furthermore, different variants in one gene can cause the same phenotype as, for example, different homozygous missense variants in GUCY2D (Gene ID 3000, OMIM 600179) in two unrelated patients cause LCA. Similarly, various pathogenic variants in LRAT (Gene ID 9227, OMIM 604863) and CRB1 (Gene ID 23418, OMIM 604210 ) in unrelated patients cause RP. However, the same variant in one gene can cause different phenotypes, such as a homozygous frameshift variant in the CERKL gene that led to CRD and RP in two unrelated families. Another example of this category is a homozygous missense variant in RPGRIP1 that caused LCA in two unrelated patients and CRD in one patient. In addition, different variants in one gene can cause different phenotypes as exemplified by ABCA4 that causes STGD1 and autosomal recessive (arRP).
Mutations in genes associated with Bardet-Biedl syndrome (BBS), such as BBS1 (Gene ID 582, OMIM 209900) and BBS2, can also be found in non-syndromic arRP [54-56]. The variants within these two genes identified in this study are associated with non-syndromic RP as the probands did not show extraocular features.
We identified homozygous CRB1 variants in two RP probands. One of these missense mutations, p.(Thr745Met), was previously detected in three Iranian RP cases by Ghofrani et al. [41]. Mutations in CRB1 have been shown to be responsible for 10% of LCA cases in northwestern Europe and 2.5% of arRP cases in the French population [10,57].
Although finding known and new mutations in known IRD-associated genes through cost-effective gene panel testing is important for patients who cannot afford the price of whole exome sequencing (WES) and whole genome sequencing (WGS), WES and WGS provide the opportunity to discover novel genes, as well as copy number variations (CNVs) and noncoding variants in IRD-associated genes [58-60]. The sequencing costs for this gene panel testing (MIP analysis), excluding the MIP synthesis costs and Sanger sequencing validation, were about €40 per sample, which is 10–20 times lower than the commercial price for WES.
Despite the high yield in this study, for 28% of the patients with IRDs, we still do not know the causal gene defects. The MIP design was based on the non-syndromic IRD-associated genes known in October 2013. In the meantime, another 60 non-syndromic IRD genes have been discovered that together may well explain another 10–15% of IRD cases. In conclusion, we demonstrated that this panel testing, employing MIPs, is a cost-effective targeted NGS-based method with a high detection rate for pathogenic variants in patients with IRDs.
Figure 1.
Relative involvement of 19 inherited retinal disease-associated genes among the 50 probands.
Table 3. In-silico analysis of the non-truncating pathogenic variants identified in this study.
| Gene | Amino acid change | Number of Probands | Mutationtaster | Polyphen-2 | Grantham score | PhyloP score | SIFT | CADD score | ExAC MAF | Known/ Novel variant |
|---|---|---|---|---|---|---|---|---|---|---|
| ABCA4 |
p.(Arg212Cys) |
1 |
Disease causing |
Probably damaging |
180 |
5.2 |
Deleterious |
26.4 |
0.000117 |
Known |
| ABCA4 |
p.(Cys1488Arg) |
1 |
Disease causing |
Possibly damaging |
180 |
8.9 |
Deleterious |
21 |
1.42E-05 |
Known |
| ABCA4 |
p.(Ser2129Gly) |
1 |
Disease causing |
benign |
56 |
7.9 |
Tolerated |
19.96 |
N/A |
Known |
| BBS1 |
p.(Arg160Gln) |
1 |
Disease causing |
Probably damaging |
43 |
7.9 |
Deleterious |
33 |
4.12E-05 |
Known |
| BBS2 |
p.(Val75Gly) |
1 |
Disease causing |
Probably damaging |
109 |
9.1 |
Deleterious |
27.2 |
N/A |
Known |
| CEP290 |
p.(Val2435Gly) |
1 |
Disease causing |
Probably damaging |
109 |
8.4 |
Deleterious |
24.8 |
N/A |
Novel |
| CRB1 |
p.(Thr745Met) |
1 |
Disease causing |
Probably damaging |
81 |
5.5 |
Deleterious |
15 |
8.26E-05 |
Known |
| CRB1 |
p.(Cys183Tyr) |
1 |
Disease causing |
Probably damaging |
194 |
7.4 |
Deleterious |
15.9 |
N/A |
Novel |
| GUCY2D |
p.(Arg795Trp) |
1 |
Disease causing |
Probably damaging |
101 |
1.37 |
Deleterious |
21.7 |
8.26E-06 |
Novel |
| GUCY2D |
p.(Asp406His) |
1 |
Disease causing |
Probably damaging |
81 |
9.1 |
Deleterious |
20.9 |
N/A |
Novel |
| LRAT |
p.(His163Asp) |
1 |
Disease causing |
Probably damaging |
81 |
7.6 |
Deleterious |
20 |
N/A |
Novel |
| PDE6C |
p.(Phe538Ile) |
1 |
Disease causing |
Probably damaging |
21 |
7.4 |
Deleterious |
25.1 |
N/A |
Novel |
| PDZD7 |
p.(Arg265Gly) |
1 |
Disease causing |
Possibly damaging |
125 |
2.4 |
Deleterious |
15.1 |
8.24E-06 |
Novel |
| RPE65 |
p.(Tyr368His) |
2 |
Disease causing |
Probably damaging |
83 |
8.8 |
Deleterious |
24.4 |
6.6E-05 |
Known |
| RPGRIP1 | p.(Ala436Ser) | 3 | Disease causing | Probably damaging | 99 | 5.4 | Deleterious | 15 | N/A | Novel |
MAF: Minor allele frequency, N/A: Not available
Acknowledgments
We are grateful to the patients and their families for their participation in our study. We also thank Mina Bozorgzadeh Esfahani and Maryam Ghalyani for her help in this study to gather the families. We also thank the Medical Genetics Laboratory of Genome, Isfahan, Iran. This work was supported by the Algemene Nederlandse Vereniging ter Voorkoming van Blindheid and Landelijke Stichting voor Blinden en Slechtzienden that contributed through UitZicht 2014-13, together with the Rotterdamse Stichting Blindenbelangen, Stichting Blindenhulp and the Stichting tot Verbetering van het Lot der Blinden (to FPMC). The funding organizations had no role in the design or conduct of this research, and provided unrestricted grants.
Appendix 1. Splice site prediction scores for the ABCA4 c.6385A and c.6385G variants according to five algorithms in Alamut Visual.
The splice donor site predictions (in blue; 5’) show a decrease from 91.8 to 80.3 for SpliceSiteFinder-like. There are no scores for the other programs as the ‘GC’ canonical splice site sequence only is recognized by SpliceSiteFinder-like. A strong cryptic splice donor site at c.6386+27 in intron 47 possibly could be activated in the ABCA4 gene carrying c.6385A>G. To access the data, click or select the words “Appendix 1.”
References
- 1.Verbakel SK, van Huet RAC, Boon CJF, den Hollander AI, Collin RWJ, Klaver CCW, Hoyng CB, Roepman R, Klevering BJ. Non-syndromic retinitis pigmentosa. Prog Retin Eye Res. 2018;66:157–86. doi: 10.1016/j.preteyeres.2018.03.005. [DOI] [PubMed] [Google Scholar]
- 2.Dias MF, Joo K, Kemp JA, Fialho SL, da Silva Cunha A, Jr, Woo SJ, Kwon YJ. Molecular genetics and emerging therapies for retinitis pigmentosa: Basic research and clinical perspectives. Prog Retin Eye Res. 2018;63:107–31. doi: 10.1016/j.preteyeres.2017.10.004. [DOI] [PubMed] [Google Scholar]
- 3.Farrar GJ, Carrigan M, Dockery A, Millington-Ward S, Palfi A, Chadderton N, Humphries M, Kiang AS, Kenna PF, Humphries P. Toward an elucidation of the molecular genetics of inherited retinal degenerations. Hum Mol Genet. 2017;26(R1):R2–11. doi: 10.1093/hmg/ddx185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Broadgate S, Yu J, Downes SM, Halford S. Unravelling the genetics of inherited retinal dystrophies: Past, present and future. Prog Retin Eye Res. 2017;59:53–96. doi: 10.1016/j.preteyeres.2017.03.003. [DOI] [PubMed] [Google Scholar]
- 5.Roosing S, Thiadens AA, Hoyng CB, Klaver CC, den Hollander AI, Cremers FP. Causes and consequences of inherited cone disorders. Prog Retin Eye Res. 2014;42:1–26. doi: 10.1016/j.preteyeres.2014.05.001. [DOI] [PubMed] [Google Scholar]
- 6.Berson EL. Retinitis pigmentosa: unfolding its mystery. Proc Natl Acad Sci USA. 1996;93:4526–8. doi: 10.1073/pnas.93.10.4526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Hohman TC. Hereditary Retinal Dystrophy. Handb Exp Pharmacol. 2017;242:337–67. doi: 10.1007/164_2016_91. [DOI] [PubMed] [Google Scholar]
- 8.Fahim A. Retinitis pigmentosa: recent advances and future directions in diagnosis and management. Curr Opin Pediatr. 2018;30:725–33. doi: 10.1097/MOP.0000000000000690. [DOI] [PubMed] [Google Scholar]
- 9.Hamel CP. Cone rod dystrophies. Orphanet J Rare Dis. 2007;2:7. doi: 10.1186/1750-1172-2-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.den Hollander AI, Roepman R, Koenekoop RK, Cremers FP. Leber congenital amaurosis: genes, proteins and disease mechanisms. Prog Retin Eye Res. 2008;27:391–419. doi: 10.1016/j.preteyeres.2008.05.003. [DOI] [PubMed] [Google Scholar]
- 11.Koenekoop RK. An overview of Leber congenital amaurosis: a model to understand human retinal development. Surv Ophthalmol. 2004;49:379–98. doi: 10.1016/j.survophthal.2004.04.003. [DOI] [PubMed] [Google Scholar]
- 12.Franceschetti A. A special form of tapetoretinal degeneration: fundus flavimaculatus. Trans Am Acad Ophthalmol Otolaryngol. 1965;69:1048–53. [PubMed] [Google Scholar]
- 13.Jones KD, Wheaton DK, Bowne SJ, Sullivan LS, Birch DG, Chen R, Daiger SP. Next-generation sequencing to solve complex inherited retinal dystrophy: A case series of multiple genes contributing to disease in extended families. Mol Vis. 2017;23:470–81. [PMC free article] [PubMed] [Google Scholar]
- 14.Lee K, Garg S. Navigating the current landscape of clinical genetic testing for inherited retinal dystrophies. Genet Med. 2015;17:245–52. doi: 10.1038/gim.2015.15. [DOI] [PubMed] [Google Scholar]
- 15.Ghaheri M, Kahrizi D, Yari K, Babaie A, Suthar RS, Kazemi E. A comparative evaluation of four DNA extraction protocols from whole blood sample. Cell Mol Biol. 2016;62:120–4. [PubMed] [Google Scholar]
- 16.den Hollander AI, Koenekoop RK, Yzer S, Lopez I, Arends ML, Voesenek KE, Zonneveld MN, Strom TM, Meitinger T, Brunner HG, Hoyng CB, van den Born LI, Rohrschneider K, Cremers FP. Mutations in the CEP290 (NPHP6) gene are a frequent cause of Leber congenital amaurosis. Am J Hum Genet. 2006;79:556–61. doi: 10.1086/507318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Bax NM, Sangermano R, Roosing S, Thiadens AA, Hoefsloot LH, van den Born LI, Phan M, Klevering BJ, Westeneng-van Haaften C, Braun TA, Zonneveld-Vrieling MN, de Wijs I, Mutlu M, Stone EM, den Hollander AI, Klaver CC, Hoyng CB, Cremers FP. Heterozygous deep-intronic variants and deletions in ABCA4 in persons with retinal dystrophies and one exonic ABCA4 variant. Hum Mutat. 2015;36:43–7. doi: 10.1002/humu.22717. [DOI] [PubMed] [Google Scholar]
- 18.Li H, Durbin R. Fast and accurate short read alignment with Burrows-Wheeler transform. Bioinformatics. 2009;25:1754–60. doi: 10.1093/bioinformatics/btp324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Lek M, Karczewski KJ, Minikel EV, Samocha KE, Banks E, Fennell T, O’Donnell-Luria AH, Ware JS, Hill AJ, Cummings BB, Tukiainen T, Birnbaum DP, Kosmicki JA, Duncan LE, Estrada K, Zhao F, Zou J, Pierce-Hoffman E, Berghout J, Cooper DN, Deflaux N, DePristo M, Do R, Flannick J, Fromer M, Gauthier L, Goldstein J, Gupta N, Howrigan D, Kiezun A, Kurki MI, Moonshine AL, Natarajan P, Orozco L, Peloso GM, Poplin R, Rivas MA, Ruano-Rubio V, Rose SA, Ruderfer DM, Shakir K, Stenson PD, Stevens C, Thomas BP, Tiao G, Tusie-Luna MT, Weisburd B, Won HH, Yu D, Altshuler DM, Ardissino D, Boehnke M, Danesh J, Donnelly S, Elosua R, Florez JC, Gabriel SB, Getz G, Glatt SJ, Hultman CM, Kathiresan S, Laakso M, McCarroll S, McCarthy MI, McGovern D, McPherson R, Neale BM, Palotie A, Purcell SM, Saleheen D, Scharf JM, Sklar P, Sullivan PF, Tuomilehto J, Tsuang MT, Watkins HC, Wilson JG, Daly MJ, MacArthur DG, Exome Aggregation C. Analysis of protein-coding genetic variation in 60,706 humans. Nature. 2016;536:285–91. doi: 10.1038/nature19057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Cremers FP, van de Pol DJ, van Driel M, den Hollander AI, van Haren FJ, Knoers NV, Tijmes N, Bergen AA, Rohrschneider K, Blankenagel A. Autosomal recessive retinitis pigmentosa and cone-rod dystrophy caused by splice site mutations in the Stargardt’s disease gene ABCR. Hum Mol Genet. 1998;7:355–62. doi: 10.1093/hmg/7.3.355. [DOI] [PubMed] [Google Scholar]
- 21.Sangermano R, Bax NM, Bauwens M, Van den Born LI, De Baere E, Garanto A, Collin RW, Goercharn-Ramlal AS, den Engelsman-van Dijk AH, Rohrschneider K. Photoreceptor progenitor mRNA analysis reveals exon skipping resulting from the ABCA4 c. 5461–10T→ C mutation in Stargardt disease. Ophthalmology. 2016;123:1375–85. doi: 10.1016/j.ophtha.2016.01.053. [DOI] [PubMed] [Google Scholar]
- 22.Zernant J, Xie YA, Ayuso C, Riveiro-Alvarez R, Lopez-Martinez MA, Simonelli F, Testa F, Gorin MB, Strom SP, Bertelsen M, Rosenberg T, Boone PM, Yuan B, Ayyagari R, Nagy PL, Tsang SH, Gouras P, Collison FT, Lupski JR, Fishman GA, Allikmets R. Analysis of the ABCA4 genomic locus in Stargardt disease. Hum Mol Genet. 2014;23:6797–806. doi: 10.1093/hmg/ddu396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Zernant J, Lee W, Collison FT, Fishman GA, Sergeev YV, Schuerch K, Sparrow JR, Tsang SH, Allikmets R. Frequent hypomorphic alleles account for a significant fraction of ABCA4 disease and distinguish it from age-related macular degeneration. J Med Genet. 2017;54:404–12. doi: 10.1136/jmedgenet-2017-104540. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Cornelis SS, Bax NM, Zernant J, Allikmets R, Fritsche LG, den Dunnen JT, Ajmal M, Hoyng CB, Cremers FP. In Silico Functional Meta‐Analysis of 5,962 ABCA4 Variants in 3,928 Retinal Dystrophy Cases. Hum Mutat. 2017;38:400–8. doi: 10.1002/humu.23165. [DOI] [PubMed] [Google Scholar]
- 25.Schulz HL, Grassmann F, Kellner U, Spital G, Ruther K, Jagle H, Hufendiek K, Rating P, Huchzermeyer C, Baier MJ, Weber BH, Stohr H. Mutation Spectrum of the ABCA4 Gene in 335 Stargardt Disease Patients From a Multicenter German Cohort-Impact of Selected Deep Intronic Variants and Common SNPs. Invest Ophthalmol Vis Sci. 2017;58:394–403. doi: 10.1167/iovs.16-19936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Cartegni L, Wang J, Zhu Z, Zhang MQ, Krainer AR. ESEfinder: A web resource to identify exonic splicing enhancers. Nucleic Acids Res. 2003;31:3568–71. doi: 10.1093/nar/gkg616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Desmet FO, Hamroun D, Lalande M, Collod-Beroud G, Claustres M, Beroud C. Human Splicing Finder: an online bioinformatics tool to predict splicing signals. Nucleic Acids Res. 2009;37:e67. doi: 10.1093/nar/gkp215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Pertea M, Lin X, Salzberg SL. GeneSplicer: a new computational method for splice site prediction. Nucleic Acids Res. 2001;29:1185–90. doi: 10.1093/nar/29.5.1185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Reese MG, Eeckman FH, Kulp D, Haussler D. Improved splice site detection in Genie. J Comput Biol. 1997;4:311–23. doi: 10.1089/cmb.1997.4.311. [DOI] [PubMed] [Google Scholar]
- 30.Yeo G, Burge CB. Maximum entropy modeling of short sequence motifs with applications to RNA splicing signals. J Comput Biol. 2004;11:377–94. doi: 10.1089/1066527041410418. [DOI] [PubMed] [Google Scholar]
- 31.Maia-Lopes S, Silva ED, Silva MF, Reis A, Faria P, Castelo-Branco M. Evidence of widespread retinal dysfunction in patients with stargardt disease and morphologically unaffected carrier relatives. Invest Ophthalmol Vis Sci. 2008;49:1191–9. doi: 10.1167/iovs.07-1051. [DOI] [PubMed] [Google Scholar]
- 32.Duno M, Schwartz M, Larsen PL, Rosenberg T. Phenotypic and genetic spectrum of Danish patients with ABCA4-related retinopathy. Ophthalmic Genet. 2012;33:225–31. doi: 10.3109/13816810.2011.643441. [DOI] [PubMed] [Google Scholar]
- 33.Ozgul RK, Durukan H, Turan A, Oner C, Ogus A, Farber DB. Molecular analysis of the ABCA4 gene in Turkish patients with Stargardt disease and retinitis pigmentosa. Hum Mutat. 2004;23:523. doi: 10.1002/humu.9236. [DOI] [PubMed] [Google Scholar]
- 34.Fujinami K, Sergouniotis PI, Davidson AE, Mackay DS, Tsunoda K, Tsubota K, Robson AG, Holder GE, Moore AT, Michaelides M, Webster AR. The clinical effect of homozygous ABCA4 alleles in 18 patients. Ophthalmology. 2013;120:2324–31. doi: 10.1016/j.ophtha.2013.04.016. [DOI] [PubMed] [Google Scholar]
- 35.Sciezynska A, Ozieblo D, Ambroziak AM, Korwin M, Szulborski K, Krawczynski M, Stawinski P, Szaflik J, Szaflik JP, Ploski R, Oldak M. Next-generation sequencing of ABCA4: High frequency of complex alleles and novel mutations in patients with retinal dystrophies from Central Europe. Exp Eye Res. 2016;145:93–9. doi: 10.1016/j.exer.2015.11.011. [DOI] [PubMed] [Google Scholar]
- 36.Fujinami K, Zernant J, Chana RK, Wright GA, Tsunoda K, Ozawa Y, Tsubota K, Webster AR, Moore AT, Allikmets R, Michaelides M. ABCA4 gene screening by next-generation sequencing in a British cohort. Invest Ophthalmol Vis Sci. 2013;54:6662–74. doi: 10.1167/iovs.13-12570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Pennesi ME, Stover NB, Stone EM, Chiang PW, Weleber RG. Residual Electroretinograms in Young Leber Congenital Amaurosis Patients with Mutations of AIPL1. Invest Ophthalmol Vis Sci. 2011;52:8166–73. doi: 10.1167/iovs.11-8298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Kantaputra PN, Kayserili H, Guven Y, Kantaputra W, Balci MC, Tanpaiboon P, Tananuvat N, Uttarilli A, Dalal A. Clinical manifestations of 17 patients affected with mucopolysaccharidosis type VI and eight novel ARSB mutations. Am J Med Genet A. 2014;164A:1443–53. doi: 10.1002/ajmg.a.36489. [DOI] [PubMed] [Google Scholar]
- 39.Zaghloul NA, Liu Y, Gerdes JM, Gascue C, Oh EC, Leitch CC, Bromberg Y, Binkley J, Leibel RL, Sidow A, Badano JL, Katsanis N. Functional analyses of variants reveal a significant role for dominant negative and common alleles in oligogenic Bardet-Biedl syndrome. Proc Natl Acad Sci USA. 2010;107:10602–7. doi: 10.1073/pnas.1000219107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Fokkema IF, Taschner PE, Schaafsma GC, Celli J, Laros JF, den Dunnen JT. LOVD v.2.0: the next generation in gene variant databases. Hum Mutat. 2011;32:557–63. doi: 10.1002/humu.21438. [DOI] [PubMed] [Google Scholar]
- 41.Ghofrani M, Yahyaei M, Brunner HG, Cremers FP, Movasat M, Imran Khan M, Keramatipour M. Homozygosity Mapping and Targeted Sanger Sequencing Identifies Three Novel CRB1 (Crumbs homologue 1) Mutations in Iranian Retinal Degeneration Families. Iran Biomed J. 2017;21:294–302. doi: 10.18869/acadpub.ibj.21.5.294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Corton M, Avila-Fernandez A, Vallespin E, Lopez-Molina MI, Almoguera B, Martin-Garrido E, Tatu SD, Khan MI, Blanco-Kelly F, Riveiro-Alvarez R, Brion M, Garcia-Sandoval B, Cremers FPM, Carracedo A, Ayuso C. Involvement of LCA5 in Leber congenital amaurosis and retinitis pigmentosa in the Spanish population. Ophthalmology. 2014;121:399–407. doi: 10.1016/j.ophtha.2013.08.028. [DOI] [PubMed] [Google Scholar]
- 43.Wang F, Wang H, Tuan HF, Nguyen DH, Sun V, Keser V, Bowne SJ, Sullivan LS, Luo H, Zhao L, Wang X, Zaneveld JE, Salvo JS, Siddiqui S, Mao L, Wheaton DK, Birch DG, Branham KE, Heckenlively JR, Wen C, Flagg K, Ferreyra H, Pei J, Khan A, Ren H, Wang K, Lopez I, Qamar R, Zenteno JC, Ayala-Ramirez R, Buentello-Volante B, Fu Q, Simpson DA, Li Y, Sui R, Silvestri G, Daiger SP, Koenekoop RK, Zhang K, Chen R. Next generation sequencing-based molecular diagnosis of retinitis pigmentosa: identification of a novel genotype-phenotype correlation and clinical refinements. Hum Genet. 2014;133:331–45. doi: 10.1007/s00439-013-1381-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Payne A, Vithana E, Khaliq S, Hameed A, Deller J, Abu-Safieh L, Kermani S, Leroy BP, Mehdi SQ, Moore AT, Bird AC, Bhattacharya SS. RP1 protein truncating mutations predominate at the RP1 adRP locus. Invest Ophthalmol Vis Sci. 2000;41:4069–73. [PubMed] [Google Scholar]
- 45.Yzer S, van den Born LI, Schuil J, Kroes HY, van Genderen MM, Boonstra FN, van den Helm B, Brunner HG, Koenekoop RK, Cremers FPA. Tyr368His RPE65 founder mutation is associated with variable expression and progression of early onset retinal dystrophy in 10 families of a genetically isolated population. J Med Genet. 2003;40:709–13. doi: 10.1136/jmg.40.9.709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Hamamy H. Consanguineous marriages: Preconception consultation in primary health care settings. J Community Genet. 2012;3:185–92. doi: 10.1007/s12687-011-0072-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Hosseini-Chavoshi M, Abbasi-Shavazi MJ, Bittles AH. Consanguineous marriage, reproductive behaviour and postnatal mortality in contemporary Iran. Hum Hered. 2014;77:16–25. doi: 10.1159/000358403. [DOI] [PubMed] [Google Scholar]
- 48.Eisenberger T, Neuhaus C, Khan AO, Decker C, Preising MN, Friedburg C, Bieg A, Gliem M, Charbel Issa P, Holz FG, Baig SM, Hellenbroich Y, Galvez A, Platzer K, Wollnik B, Laddach N, Ghaffari SR, Rafati M, Botzenhart E, Tinschert S, Borger D, Bohring A, Schreml J, Kortge-Jung S, Schell-Apacik C, Bakur K, Al-Aama JY, Neuhann T, Herkenrath P, Nurnberg G, Nurnberg P, Davis JS, Gal A, Bergmann C, Lorenz B, Bolz HJ. Increasing the yield in targeted next-generation sequencing by implicating CNV analysis, non-coding exons and the overall variant load: the example of retinal dystrophies. PLoS One. 2013;8:e78496. doi: 10.1371/journal.pone.0078496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Beheshtian M, Saee Rad S, Babanejad M, Mohseni M, Hashemi H, Eshghabadi A, Hajizadeh F, Akbari MR, Kahrizi K, Riazi Esfahani M, Najmabadi H. Impact of whole exome sequencing among Iranian patients with autosomal recessive retinitis pigmentosa. Arch Iran Med. 2015;18:776–85. [PubMed] [Google Scholar]
- 50.Combs R, McAllister M, Payne K, Lowndes J, Devery S, Webster AR, Downes SM, Moore AT, Ramsden S, Black G, Hall G. Understanding the impact of genetic testing for inherited retinal dystrophy. Eur J Hum Genet. 2013;21:1209–13. doi: 10.1038/ejhg.2013.19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Haer-Wigman L, van Zelst-Stams WA, Pfundt R, van den Born LI, Klaver CC, Verheij JB, Hoyng CB, Breuning MH, Boon CJ, Kievit AJ, Verhoeven VJ, Pott JW, Sallevelt SC, van Hagen JM, Plomp AS, Kroes HY, Lelieveld SH, Hehir-Kwa JY, Castelein S, Nelen M, Scheffer H, Lugtenberg D, Cremers FP, Hoefsloot L, Yntema HG. Diagnostic exome sequencing in 266 Dutch patients with visual impairment. Eur J Hum Genet. 2017;25:591–9. doi: 10.1038/ejhg.2017.9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Fu Q, Wang F, Wang H, Xu F, Zaneveld JE, Ren H, Keser V, Lopez I, Tuan HF, Salvo JS, Wang X, Zhao L, Wang K, Li Y, Koenekoop RK, Chen R, Sui R. Next-generation sequencing-based molecular diagnosis of a Chinese patient cohort with autosomal recessive retinitis pigmentosa. Invest Ophthalmol Vis Sci. 2013;54:4158–66. doi: 10.1167/iovs.13-11672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Sundaresan P, Vijayalakshmi P, Thompson S, Ko AC, Fingert JH, Stone EM. Mutations that are a common cause of Leber congenital amaurosis in northern America are rare in southern India. Mol Vis. 2009;15:1781–7. [PMC free article] [PubMed] [Google Scholar]
- 54.Estrada-Cuzcano A, Koenekoop RK, Senechal A, De Baere EB, de Ravel T, Banfi S, Kohl S, Ayuso C, Sharon D, Hoyng CB, Hamel CP, Leroy BP, Ziviello C, Lopez I, Bazinet A, Wissinger B, Sliesoraityte I, Avila-Fernandez A, Littink KW, Vingolo EM, Signorini S, Banin E, Mizrahi-Meissonnier L, Zrenner E, Kellner U, Collin RW, den Hollander AI, Cremers FP, Klevering BJ. BBS1 mutations in a wide spectrum of phenotypes ranging from nonsyndromic retinitis pigmentosa to Bardet-Biedl syndrome. Arch Ophthalmol. 2012;130:1425–32. doi: 10.1001/archophthalmol.2012.2434. [DOI] [PubMed] [Google Scholar]
- 55.Mykytyn K, Nishimura DY, Searby CC, Shastri M, Yen HJ, Beck JS, Braun T, Streb LM, Cornier AS, Cox GF, Fulton AB, Carmi R, Luleci G, Chandrasekharappa SC, Collins FS, Jacobson SG, Heckenlively JR, Weleber RG, Stone EM, Sheffield VC. Identification of the gene (BBS1) most commonly involved in Bardet-Biedl syndrome, a complex human obesity syndrome. Nat Genet. 2002;31:435–8. doi: 10.1038/ng935. [DOI] [PubMed] [Google Scholar]
- 56.Katsanis N, Ansley SJ, Badano JL, Eichers ER, Lewis RA, Hoskins BE, Scambler PJ, Davidson WS, Beales PL, Lupski JR. Triallelic inheritance in Bardet-Biedl syndrome, a Mendelian recessive disorder. Science. 2001;293:2256–9. doi: 10.1126/science.1063525. [DOI] [PubMed] [Google Scholar]
- 57.Bujakowska K, Audo I, Mohand-Said S, Lancelot ME, Antonio A, Germain A, Leveillard T, Letexier M, Saraiva JP, Lonjou C, Carpentier W, Sahel JA, Bhattacharya SS, Zeitz C. CRB1 mutations in inherited retinal dystrophies. Hum Mutat. 2012;33:306–15. doi: 10.1002/humu.21653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Weisschuh N, Mayer AK, Strom TM, Kohl S, Glockle N, Schubach M, Andreasson S, Bernd A, Birch DG, Hamel CP, Heckenlively JR, Jacobson SG, Kamme C, Kellner U, Kunstmann E, Maffei P, Reiff CM, Rohrschneider K, Rosenberg T, Rudolph G, Vamos R, Varsanyi B, Weleber RG, Wissinger B. Mutation Detection in Patients with Retinal Dystrophies Using Targeted Next Generation Sequencing. PLoS One. 2016;11:e0145951. doi: 10.1371/journal.pone.0145951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Van Schil K, Naessens S, Van de Sompele S, Carron M, Aslanidis A, Van Cauwenbergh C, Kathrin Mayer A, Van Heetvelde M, Bauwens M, Verdin H, Coppieters F, Greenberg ME, Yang MG, Karlstetter M, Langmann T, De Preter K, Kohl S, Cherry TJ, Leroy BP, Group CNVS, De Baere E. Mapping the genomic landscape of inherited retinal disease genes prioritizes genes prone to coding and noncoding copy-number variations. Genet Med. 2018;20:202–13. doi: 10.1038/gim.2017.97. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Mayer AK, Van Cauwenbergh C, Rother C, Baumann B, Reuter P, De Baere E, Wissinger B, Kohl S, Group AS. CNGB3 mutation spectrum including copy number variations in 552 achromatopsia patients. Hum Mutat. 2017;38:1579–91. doi: 10.1002/humu.23311. [DOI] [PubMed] [Google Scholar]