Abstract
Therapeutic adoptive transfer of natural regulatory T cells (nTreg, CD4+ CD25+ Foxp3+ T cells) or in vivo selective expansion of nTreg cells has been demonstrated to improve the cardiac function in various cardiovascular disease models. The differentiation of nTreg cells is mediated by catecholamines via β1-adrenergic receptor (β1-AR) activation. Autoantibody against β1-adrenoceptor (β1-AA) as a β1-AR agonist is closely associated with the occurrence and deterioration of cardiac dysfunction. However, whether β1-AA has any impact on nTreg cells has not been reported. The aim of the present study was intended to assess the potential impact of β1-AA on nTreg cell differentiation and explore the underlying mechanism. It was found that the expression of multiple proteins involved in nTreg cell differentiation, immunosuppressive function, and migration was up-regulated in mice after β1-AA administration, suggesting that β1-AA may promote nTreg cell activation. In vitro, β1-AA promoted nTreg cell differentiation by up-regulating mitochondrial fatty acid oxidation (FAO) in activated CD4+ T cells via AMP-activated protein kinase (AMPK) activation and mitochondrial membrane potential reduction. In addition, the AMPK agonist facilitated β1-AA-mediated FAO and nTreg cell differentiation. To further confirm the role of AMPK in β1-AA-mediated nTreg cell differentiation, β1-AA was acted on the CD4+ T cells isolated from AMPK-deficient (AMPK−/−) mice. The result showed that the effect of β1-AA on nTreg cell differentiation was attenuated markedly after AMPK knockout. In conclusion, AMPK-mediated metabolic regulation targeting for nTreg cell restoration may be a promising therapeutic target for β1-AA-positive patients with cardiac dysfunction.
Introduction
CD4+ T cells are known as the most important participant in adaptive immunity of the organism. Over-activation of CD4+ T cells and disproportion of their subpopulations play an important role in the pathogenesis of various cardiovascular diseases. Functionally, CD4+ T cells are classified as two major categories: effector T cells and regulatory T (Treg) cells1, among which natural Treg (nTreg, CD4+ CD25+ Foxp3+ T) cells play a critical role in inhibiting the immune response of effector T cells and maintaining immune tolerance2,3. Therapeutic adoptive transfer of nTreg cells or in vivo selective nTreg cell expansion has been demonstrated to attenuate post-infraction left ventricular remodeling, relief myocardial injury, and eventually improve the cardiac function in diverse cardiovascular disease models4,5. Studies have confirmed that the development and function of nTreg cells are regulated by catecholamines via the expression of α-, β1-, and β2-adrenergic receptors (β1/2-ARs)6–8. Compared with effector T cells, β1-AR expression in nTreg cells is more advantageous than β2-AR expression8, but the effect of β1-AR activation on nTreg cells remains unclear.
Autoantibody targeting the second extracellular loop of β1-adrenoceptor (β1-AA) is commonly detected in circulating blood of the patients with cardiac dysfunction caused by etiologies like dilated cardiomyopathy, ischemic heart disease, and arrhythmia9–11. β1-AA was found to exhibit the agonist-like effects on β1-AR, such as increasing the intracellular calcium level promoting the beating frequency of neonatal rat cardiomyocytes and inducing cAMP production12–14. The positive rate of β1-AA was reported to be as high as 80% in different cardiac dysfunction models15. Moreover, LVEF of the cardiac dysfunction patients improved obviously after removing β1-AA by immunoadsorption (IA) treatment16. However, it is not elucidated about the underlying mechanism related to β1-AA-induced cardiac dysfunction. Our previous and other studies found that in β1-AA-positive murine, not only the cardiac function was decreased but accompanied by an increase in the peripheral CD4+/CD8+ T cell ratio; in addition, part of the myocardium was infiltrated by large number of T cells17. In vitro, β1-AA isolated from the sera of cardiac dysfunction patients promoted proliferation of CD4+ T cells through the β1-AR/cAMP pathway14. Furthermore, accompanied by cardiac function improvement of the β1-AA-positive cardiac dysfunction after IA treatment, the number of circulating nTreg cells increased significantly18,19. It was shown that nTreg cell proportion in rat peripheral blood was inhibited by β1-AR blocker propranolol20. However, whether β1-AA as a agonist-like substance of β1-AR can exert a direct effect on nTreg cells has not been reported.
Therefore, the present study was intended to assess the potential impact of β1-AA on nTreg cell activation and differentiation, and the underlying mechanism was explored in an attempt to etiologically find a potential therapeutic target for β1-AA-positive cardiac dysfunction patients.
Results
Activation of circulating nTreg cells in mice was promoted by β1-AA
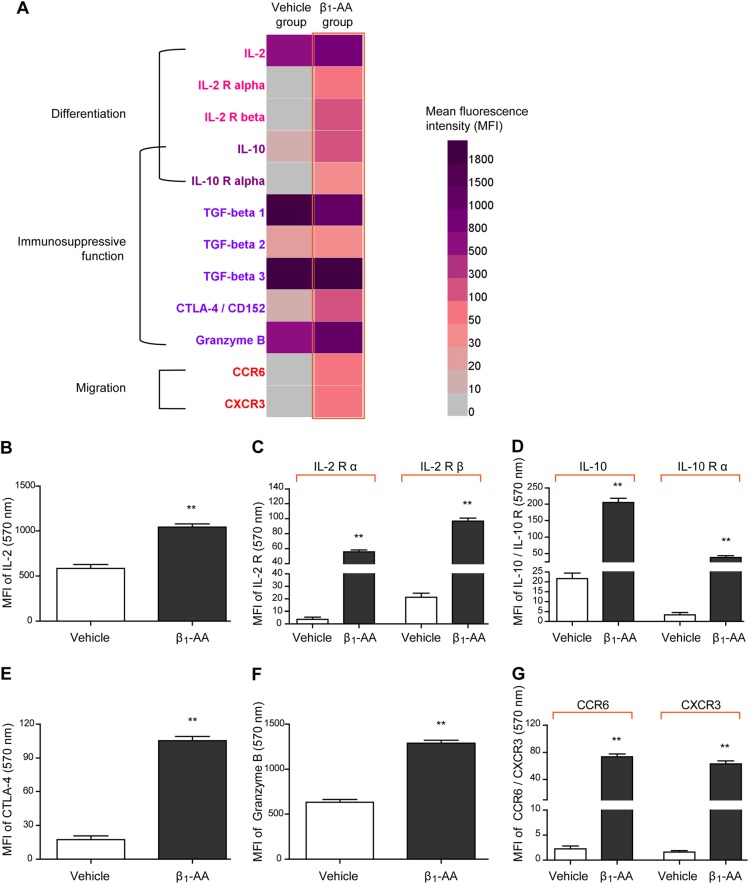
After 8 weeks β1-AR monoclonal antibody (β1-AR mAb) administration, optical density (OD) value of serum β1-AA was increased in mice, indicating that β1-AA-positive model was created successfully (Supplemental Fig. 1). Using the protein microarray chip technique, the expressions of nTreg cell-related proteins and cytokines were detected in β1-AA-positive mice at the eighth week after β1-AR mAb administration. The heat map of cluster analysis (Fig. 1a) showed that the expressions of interleukin-2 (IL-2)/IL-2 receptor (Fig. 1b, c), IL-10/IL-10 receptor (Fig. 1d), cytotoxic T-lymphocyte antigen 4 (CTLA-4) (Fig. 1e), granzyme B (Fig. 1f), chemokine receptor 3 (CXCR3) (Fig. 1g), and chemokine receptor CCR6 (Fig. 1g) in the sera of β1-AA-positive mice were enhanced significantly as compared with those in the vehicle group, of which IL-2 is known to be crucial for nTreg cell proliferation and differentiation21,22. IL-103, granzyme B, and CTLA-423 are known as important regulators in mediating the immunosuppressive activity of nTreg cells, while CCR6 and CXCR3 molecules are closely associated with nTreg cell recruitment24,25. Above all, β1-AA could promote nTreg cell activation in mice by up-regulating proteins related to nTreg cell differentiation, immunosuppressive function, and migration.
Fig. 1. Expressions of nTreg cell-related proteins and cytokines in β1-AA positive mice.
a Heat map of cluster analysis for expressions of nTreg cell-related proteins in β1-AA-positive mice at the eighth week after β1-AR mAb administration. Dark purple and light gray represented high and low levels of expression for the indicated proteins, respectively. All cytokines and cytokine receptors were detected by protein microarray chip analysis. b–g The quantitative levels of serum IL-2 (b), IL-2 receptor (c), IL-10/IL-10 receptor (d), CTLA-4 (e), granzyme B (f), and CXCR3 and CCR6 (g) in the sera of the model mice. Data are presented as means ± SD (n = 3 per group). **P < 0.01 vs. vehicle group
Differentiation of nTreg cells from activated CD4+ T cells was facilitated by β1-AA
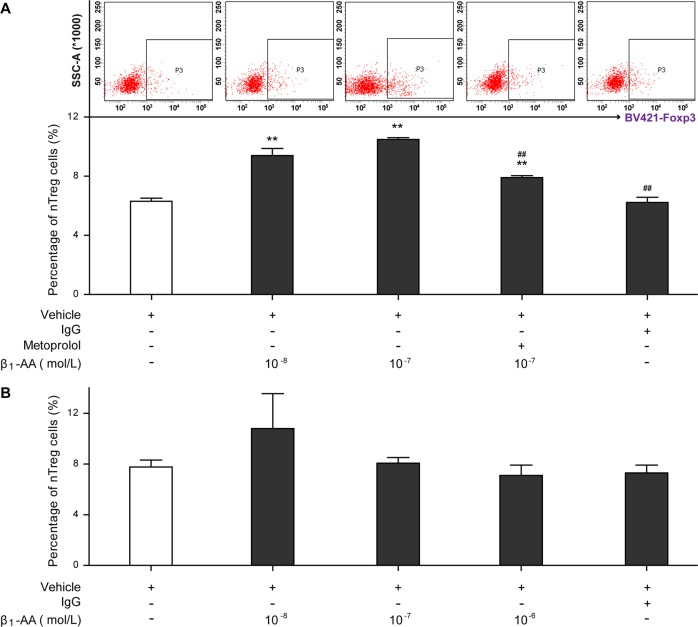
CD4+ T cells sorted from the splenocytes of healthy mice were with or without preactivation by anti-CD3/CD28 mAbs (1 μg/mL) for 72 h, and then stimulated with different concentrations of β1-AA (10−8, 10−7, or 10−6 mol/L). Flow cytometry analysis showed that the proportion of nTreg cells among the preactivated CD4+ T cells was increased significantly 24 h after β1-AA administration (Fig. 2a). In addition, this effect can be reversed drastically by the β1-AR-specific blocker metoprolol (10−7 mol/L) (Fig. 2a). However, β1-AA was unable to promote the differentiation of nTreg cells from quiescent CD4+ T cells (Fig. 2b, P > 0.05).
Fig. 2. The proportion of nTreg cells among CD4+ T cells after β1-AA stimulation.
a Percentage of nTreg cells in the preactivated CD4+ T cells at the 24th hour after β1-AA stimulation with or without metoprolol. b Percentage of nTreg cells in quiescent CD4+ T cells at the 24th hour after β1-AA stimulation. Data are presented as means ± SD (n = 5 per group). **P < 0.01 vs. vehicle group; ##P < 0.01 vs. 10−8 mol/L β1-AA group
A metabolic shift toward fatty acid metabolism was associated with the increased nTreg cell differentiation induced by β1-AA
The mechanism underlying β1-AA-mediated nTreg differentiation was explored in the further experiments. Knowing that mitochondrial fatty acid oxidation (FAO) is a decisive factor for CD4+ T cell differentiation, which promotes CD4+ T cell differentiation towards Treg cells as opposed to an effector phenotype26,27. To assess whether FAO was affected by β1-AA in CD4+ T cells, the uptake of palmitate was measured in the course of stimulation. Flow cytometry demonstrated that the absorption of palmitate was increased in activated CD4+ T cells with anti-CD3/CD28 mAbs after β1-AA stimulation (Fig. 3a). Therefore, we postulated that the metabolic alteration in activated CD4+ T cells induced by β1-AA participated in enhanced nTreg cell differentiation. Etomoxir, a selective inhibitor of carnitine palmitoyltransferase I28, was used to confirm our hypothesis. Indeed, it was found that etomoxir was able to reverse the effect of β1-AA in nTreg cell differentiation (Fig. 3b). Thus, the enhanced nTreg cell differentiation elicited by β1-AA is accompanied by a metabolic shift toward FAO in activated CD4+ T cells and is reversible by FAO inhibitor.
Fig. 3. Influence of β1-AA on FAO in activated CD4+ T cells.
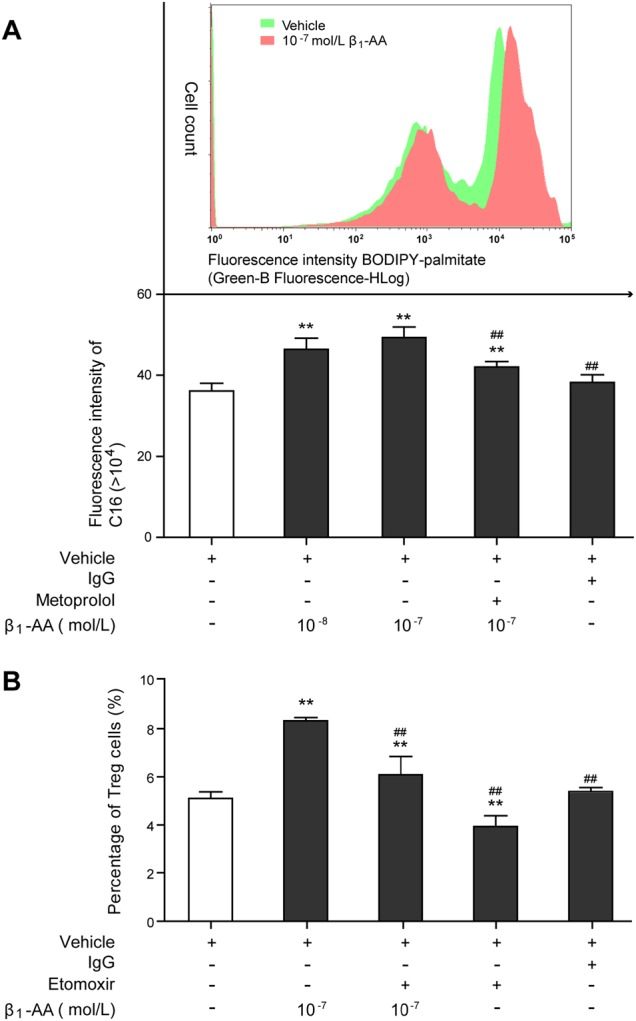
a The absorption level of palmitate in activated CD4+ T cells after β1-AA stimulation with or without metoprolol. b Percentage of nTreg cells in activated CD4+ T cells after β1-AA stimulation with or without Etomoxir. Data are presented as means ± SD (n = 5 per group). **P < 0.01 vs. vehicle group; ##P < 0.01 vs. 10−7 mol/L β1-AA group
Activation of AMPK positively regulated nTreg cell differentiation induced by β1-AA
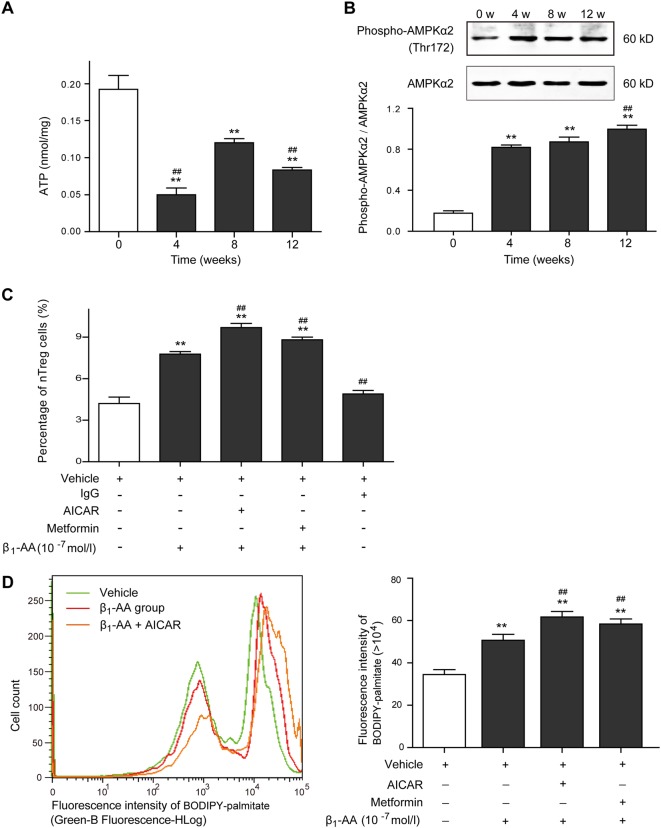
AMP-activated protein kinase (AMPK) activation is known to enhance mitochondrial FAO in response to decreased ATP level29, which is a crucial pathway attributable to nTreg cell differentiation27,30. AMPK is mainly activated by increased AMP level and phosphorylation of a threonine residue (Thr-172)31. To investigate the role of AMPK in β1-AA-induced nTreg cell differentiation, AMPK phosphorylation and ATP levels were estimated in the primary CD4+ T cells isolated from the splenic tissue of β1-AA-positive mice. Significant decrease in ATP levels has been observed in the primary CD4+ T cells since the fourth week of β1-AR mAb administration until the 12th week (Fig. 4a), accompanied by constantly increased Thr(172)-AMPKα phosphorylation (Fig. 4b). Unlike the expression changes in the inhibitory phosphorylation site serine 49132, which increased at the fourth week, and then decreased at the eighth week (Supplemental Fig. 2), Thr(172)-AMPKα phosphorylation have been constantly increased since the fourth week of β1-AR mAb administration until the 12th week. What is more, the direct AMPK activator, 5-aminoimidazole-4-carboxamide riboside (AICAR) facilitated β1-AA-mediated nTreg cell differentiation in vivo (Fig. 4c) and promoted palmitate absorption in activated CD4+ T cells with anti-CD3/CD28 mAbs (Fig. 4d). Metformin, known as a indirect activator of AMPK by lowering the energy supply30, exhibited similar effects on β1-AA-mediated nTreg cell differentiation and palmitate absorption (Fig. 4c–d).
Fig. 4. The role of AMPK in β1-AA-induced nTreg cell differentiation.
ATP levels (a) and Thr(172)-AMPKα phosphorylation (b) were estimated in the primary CD4+ T cells isolated from the splenic tissue of β1-AA-positive mice at different time points during β1-AR mAb administration (n = 4 per group). c Percentage of nTreg cells in activated CD4+ T cells after β1-AA stimulation with or without AICAR (n = 5 per group). d The absorption level of palmitate in activated CD4+ T cells after β1-AA stimulation with or without AICAR/metformin (n = 5 per group). Data are presented as means ± SD. a, b: **P < 0.01 vs. 0 week since β1-AR mAb administration; ##P < 0.01 vs. the eighth week. c, -d **P < 0.01 vs. vehicle group; ##P < 0.01 vs. β1-AA group
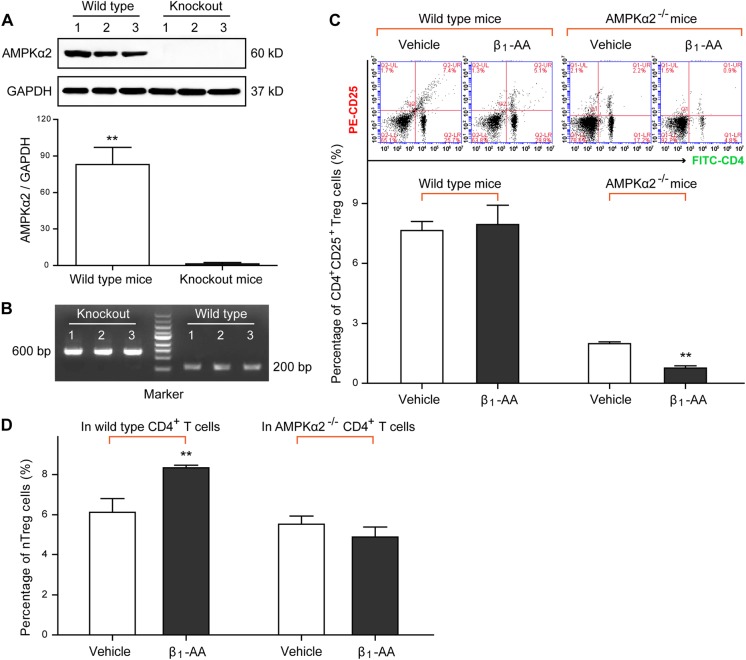
To further explore the role of AMPK in β1-AA-induced nTreg cell differentiation, β1-AA was utilized on the CD4+ T cells isolated from AMPK-deficient (AMPKα2−/−) mice (Fig. 5a, b). Percentage of the circulating CD4+ CD25+ Treg cells in AMPKα2−/− mice was lower than that in wild=type mice, and it was further reduced by 4-week β1-AA administration (Fig. 5c). In vitro, the result showed that nTreg cell proportion in the preactivated AMPK−/− CD4+ T cells was lower than that in CD4+ T cells isolated from wild-type mice after β1-AA stimulation (10−7 mol/L) for 24 h (Fig. 5d). The evidence suggesting that knockout of the AMPKα2 gene decreased the effect of β1-AA in promoting nTreg cell differentiation. To sum up, these data demonstrate that AMKP activation plays a moderate positive role in nTreg cell differentiation mediated by β1-AA.
Fig. 5. Influence of the knockout of the AMPKα2 gene in β1-AA-induced nTreg cell differentiation.
a AMPK expression in AMPKα2−/− mice at the protein level (n = 3 per group). b Identification of AMPKα2−/− mice by gel electrophoresis on a 1.5% agarose gel. The result showed that two straps of 600 and 200 bp appeared, which represented the AMPKα2−/− and wild-type C57BL/6 mice. c Frequency of the CD4+ CD25+ Treg cells in mice peripheral blood at the fourth week after β1-AR mAb administration (n = 6 per group). d Percentage of nTreg cells in activated AMPK−/− CD4+ T cells or wild-type CD4+ T cells after β1-AA stimulation (10−7 mol/L) for 24 h (n = 5 per group). Data are presented as means ± SD. a **P < 0.01 vs. knockout mice; c, d: **P < 0.01 vs. vehicle group
Enhancement of fatty acid metabolism mediated by MMP reduction promoted β1-AA-induced nTreg cell differentiation
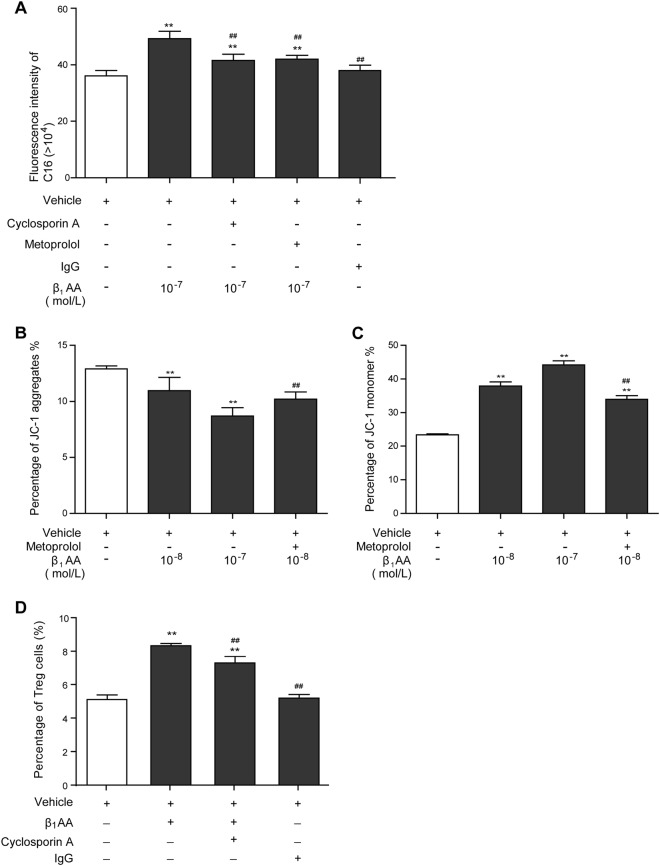
The mitochondrial FAO is closely associated with mitochondrial membrane potential (MMP) level. When MMP is reduced, intracellular absorption and utilization of palmitate are enhanced, and FAO is up-regulated33,34. To determine whether β1-AA-induced nTreg cell differentiation resulted from MMP alteration in CD4+ T cells, cyclosporin A, an MMP stabilizer, was used in the course of β1-AA stimulation. It was found that the enhanced palmitate absorption elicited by β1-AA in activated CD4+ T cells with anti-CD3/CD28 mAbs was reversed drastically by cyclosporin A and metoprolol (Fig. 6a). As shown by JC-1 staining, a concurrent reduction in the MMP of preactivated CD4+ T cells was found in the β1-AA group (Fig. 6b, c). Subsequently, cyclosporin A inhibited the elevation of nTreg cell differentiation mediated by β1-AA (Fig. 6d). Therefore, enhanced fatty acid metabolism mediated by MMP reduction in preactivated CD4+ T cells is one of the mechanisms underlying nTreg cell differentiation induced by β1-AA.
Fig. 6. Effect of mitochondrial membrane potential (MMP) on β1-AA-induced nTreg cell differentiation.
a The absorption level of palmitate in activated CD4+ T cells after β1-AA stimulation with or without cyclosporin A. b, c Percentages of JC-1 aggregate and JC-1 monomer in activated CD4+ T cells after β1-AA stimulation. d Percentage of nTreg cells in activated CD4+ T cells after β1-AA stimulation with or without cyclosporin A. Data are presented as means ± SD (n = 5 per group). **P < 0.01 vs. vehicle group; ##P < 0.01 vs. β1-AA group
Discussion
β1-AA was first identified in the sera of patients with dilated cardiomyopathy by Wallukat et al. in 198735. Subsequently, ample evidence has confirmed the pathogenic effect of β1-AA in cardiac dysfunction9,11,15. However, both our study and others found that β1-AA was positive in about 10% of healthy individuals of different age groups36–38, suggesting that β1-AA may participate in maintaining physiological homeostasis, although the correlative mechanism is unclear. nTreg cells play a very important role in maintaining the balance of the immune system by inhibiting effector T cells2,3, and their differentiation and function are regulated by the sympathetic nervous system7,8. Nevertheless, whether β1-AA as a agonist-like substance of β1-AR, could exert a direct effect on nTreg cells has not been reported. Therefore, the present study sought to assess the potential impact of β1-AA on nTreg cell differentiation and explore the underlying mechanism. It was found that β1-AA promoted nTreg cell differentiation by up-regulating fatty acid metabolism in activated CD4+ T cells via the AMPK pathway and MMP reduction.
To study the effect of β1-AA on Treg cells, a passive immunization mouse model was established successfully with the highly active and purified β1-AR mAb (Supplemental Fig. 3). Naïve CD4+ T cells differentiate into different subsets (Th1, Th2, Th17, or Treg cells) to establish immune tolerance and defense against pathogens. To quantify CD4+ T cell-related cytokine levels, the levels of Th1 cytokine (IFN-γ), Th2 cytokine (IL-4), Th17 cytokine (IL-17), and Treg cytokine (IL-10) in the sera of β1-AA-positive mice was assessed by a bead-based multianalyte flow assay kit. Quantitative analysis demonstrated that levels of Treg cytokine (IL-10) (Supplemental Fig. 4A), Th17 cytokine (IL-17) (Supplemental Fig. 4B), Th1 cytokine (IFN-γ) (Supplemental Fig. 4C), and Th2 cytokine (IL-4) (Supplemental Fig. 4D) increased in mice 8 weeks after β1-AA administration, indicating that β1-AA promoted a systemic activation of CD4+ T cell in vivo. What is more, the expression of multiple proteins related to nTreg cell differentiation, immunosuppressive function, and migration increased in mice peripheral blood, suggesting that β1-AA was able to promote nTreg cell activation. However, ultrasound analysis showed that β1-AA-induced cardiac dysfunction in mice, as illustrated by decreases in LVEF (Supplemental Fig. 5A), fractional shortening (Supplemental Fig. 5B), and cardiac output (Supplemental Fig. 5C), accompanied with a decreased proportion of circulating CD4+ CD25+ Treg cells (Supplemental Fig. 6). These results are consistent with the finding of many other studies that Treg cell frequency in cardiac dysfunction patients was decreased significantly39–41, and the number of Treg cells was positively correlated with LVEF, and negatively correlated with the NT-proBNP level42. Nevertheless, increased Treg cell infiltration was observed in the myocardium of mice with cardiac dysfunction43. For this reason, we explored whether β1-AA had a direct effect on nTreg cell differentiation in our subsequent experiment in vitro.
It was found that β1-AA induced a metabolic shift towards FAO in activated CD4+ T cells, thus promoting nTreg cell differentiation. In addition, the effect of β1-AA in promoting nTreg cell differentiation could be reversed drastically by the β1-AR-specific blocker metoprolol. Other studies also demonstrated that atecholamines such as epinephrine and norepinephrine increased the proportion of Treg cells, and the β1-AR blocker propranolol attenuated such elevation of Treg cells44. Regulation of metabolism decides the fate of CD4+ T cell differentiation, and FAO is crucial in inducing Treg cell differentiation vs. Teff cell lineages26,27,45. In other words, Treg cell differentiation depends on FAO, and we found that the FAO inhibitor etomoxir reversed β1-AA-mediated nTreg cell differentiation. MMP which effects fatty acid uptake33,34 and AMPK activation29,30,46 are two pivotal regulator for FAO. Furthermore, the underlying mechanism involved in β1-AA-mediated nTreg cell differentiation was explored.
Indeed, β1-AA reduced MMP of activated CD4+ T cells, and the MMP stabilizer cyclosporin A drastically reversed β1-AA-induced fatty acid absorption enhancement and nTreg cell differentiation. Our previous study29,30,47 demonstrated that β1-AA-induced cardiomyocyte apoptosis by reducing MMP. Similarly, the present study demonstrated that the level of CD4+ T cell apoptosis was increased significantly after β1-AA stimulation shown by Annexin V-FITC and propidium iodide double staining (Supplemental Figure 7). These findings suggest that β1-AA promoted nTreg cell differentiation through up-regulating FAO and reducing MMP, and this effect is closely associated with β1-AA-induced CD4+ T cell apoptosis. In addition, MMP reflects the integrity of mitochondrial function and is a key indicator of mitochondrial function48. The mitochondrial function alteration may participate in nTreg cell differentiation elicited by β1-AA.
AMPK is a key regulatory molecule in response to energy deprivation of the organism29. It provides energy quickly by promoting FAO and inhibiting the activity of acetyl coenzyme A carboxylase49,50. AMPK-dependent metabolic regulation plays an important role in Treg cell differentiation30,46. We found that the ATP level in the primary CD4+ T cells isolated from the splenic tissue of β1-AA positive mice was significantly lower than that in the vehicle group, which was accompanied by enhanced Thr(172)-AMPKα phosphorylation. It was reported that AICAR, a pharmacological analog of AMPK30,46, promoted Treg cell differentiation without affecting effector T cells51. Besides, norepinephrine induced AMPK activation via the cAMP/β-AApathway52. The present study showed that both the direct AMPK activator AICAR and the indirect AMPK activator metformin facilitated β1-AA-mediated FAO enhancement and nTreg cell differentiation in vitro. Moreover, AICAR promoted IL-2 level in the supernatant of activated CD4+ T cells after β1-AA stimulation, which is crucial for nTreg cell differentiation (Supplemental Figure 8). To further confirm the role of AMPK in β1-AA-mediated nTreg cell differentiation, β1-AA was acted on the CD4+ T cells isolated from the AMPKα2−/− mice. It was found that knockout of the AMPKα2 gene reduced the effect of β1-AA in promoting nTreg cell differentiation markedly, confirming that AMPK-induced FAO is a key mechanism underlying β1-AA-mediated nTreg cell differentiation.
According to the published paper52,53, the β2-AR/cAMP/PKA pathway played a positive moderate role in the immunosuppressive activity of Treg cells. Indeed, cAMP levels increased in the supernatants of nTreg cells after 30-min β1-AA stimulation, which is also the downstream signal molecule of β1-AR (Supplemental Figure 9A). However, immunofluorescence staining showed that the fluorescein-labeled β1-AA was incapable of binding to β2-ARs on Treg cells compared to the anti-β2-AR mAb (Supplemental Figure 9B). Moreover, by contrast to the activation effect of β2-AR pathway, proliferation assay of the CD4+ CD25− T effector cells revealed that compromised suppressive activity of nTreg cells were resulted from 48h β1-AA administration (Supplemental Figure 10). The inhibitory effect of nTreg cells on Teff cells is mainly mediated by IL-10 production54. In view of the fact that nTreg cell dysfunction may induced by β1-AA, IL-10 level was measured in the supernatant of nTreg cells after β1-AA stimulation. It was found that IL-10 secretion from nTreg cells was suppressed by β1-AA at concentrations of 10−6 and 10−7 mol/L compared with vehicle groups (Supplemental Figure 11). However, 10−8 mol/L β1-AA had a remarkable opposite effect, and 10−9 mol/L β1-AA did not appear to be a factor (Supplemental Figure 11). The evidences above indicated that β1-AA had bidirectional impact on the immunosuppressive function of nTreg cells. Yet, the decline in cardiac function induced by β1-AA (Supplemental Fig. 5) seems to outweigh the potentially beneficial effects on nTreg cell restoration. Nevertheless, like many other physiological processes, the influence of β1-AA on organism is a double-edged sword with therapeutic potential that is associated with the concentration of β1-AA.
Limitation and clinical perspective
Till now, the influence of β1-AR gene knockout on Treg maturation and function has not been reported. In order to investigate the role of β1-AR in β1-AA-induced nTreg differentiation, our lab had already acquired three pairs of homozygous β1-AR gene knockout mice (C57BL/6J background) from Nanjing BioMedical Research Institute of Nanjing University recently. However, yet the number of available transgenic mice is not sufficient to build our model.
Cardiac dysfunction associated with myocardial injury triggers β1-AA generation in different cardiac dysfunction models, and the positive rate of β1-AA is nearly 80%14. However, there is no specific and effective therapeutic strategy for β1-AA-positive patients. β1-AR blockers cannot entirely reverse the injurious effect of β1-AA on cardiomyocytes55,56. The present study showed that the impacts of β1-AA on nTreg cell differentiation cannot be fully counteracted by β1-AR-specific blocker metoprolol (Fig. 2a, Fig. 3a, and Fig. 6a–c), indicating that there are other mechanisms involved except for a receptor pathway. It is therefore an urgent task to find a more effective therapeutic target specific for β1-AA-positive cardiac dysfunction patients. The present study demonstrated that AMPK-mediated metabolic regulation targeting for nTreg cell restoration might be a promising therapeutic target for β1-AA-positive cardiac dysfunction patients.
Materials and methods
Synthesis and identification of β1-AR mAb
The sequence (amino-acid residues 197–222) of the second extracellular loop of the β1-AR: H-W-W-R-A-E-S-D-E-A-R-R-C-Y-N-D-P-K-C-C-D-F-VT-N-R-C was synthesized by solid-phase method using an automated peptide synthesizer. Subsequently, 0.5 mg synthetic polypeptide was coupled with the carrier protein keyhole limpet hemocyanin and bovine serum albumin (BSA) to acquire immunogenicity. The coupled polypeptide was applied to BALB/c mice to create active immunization and induce the production of β1-AR-ECII-specific antibodies. Finally, these specific antibodies were fused with the hybridoma cell line to synthesize mAbs specific to β1-AR-ECII. The synthesis of β1-AR-ECII peptide was conducted by Qiang Yao Bio Scientific Commercial Development Co., Ltd (Shanghai, China), and the hybridoma cells secreting β1-AR mAb were constructed by AbMax Biotechnology Co., Ltd (Beijing, China).
To induce the generation of ascites containing β1-AR mAb, log-phase hybridoma cells were injected intraperitoneally to female BALB/c mice aged 10 weeks at a dose of 106 cells per mL, 0.5 mL per mouse biweekly. The ascites was collected and then purified by using Protein G Affinity Chromatography Column (GE Healthcare Life Sciences, USA). The specificity and activity of the purified β1-AR mAbs were determined by enzyme-linked immunosorbent assay (ELISA) and the neonatal mouse cardiomyocyte beating experiment, respectively.
ELISA
The specificity of the purified β1-AR mAb and the OD value of β1-AA in mice serum were detected by ELISA. Briefly, the β1-AR-ECII peptide was dissolved in 100 mM 10 µg/mL Na2CO3 solution (pH = 11.0) at 4 °C overnight. The embedded 96-well plate was incubated with 1% BSA at 37 °C for 1 h, and then cultured with the primary antibody. Biotin-labeled anti-mouse immunoglobulin G (IgG) was diluted with the sealing solution at a ratio of 1:3000 and cultured at 37 °C for 1 h. Horseradish enzyme-labeled streptavidin was diluted at a ratio of 1:2000 and cultured at 37 °C for 1 h. The substrate ABTS (2,2′-azino-di-(ethyl-benzthiazoline) sulfonic acid) was dissolved in the substrate buffer with a final concentration of 1.1 mmol/L and cultured at 37 °C for 30 min. The optical density (OD) value of each well was measured at 405 nm. The titer of β1-AR mAb was determined by the positive/negative (P/N) ratio using the following equation: P/N = (sample OD–blank control OD)/(positive control OD–blank control OD). The positivity or negativity of β1-AA was determined by P/N ≥ 2.1 or P/N ≤ 1.5, respectively.
Establishment of the β1-AA-positive mouse model
Male C57BL/6 mice aged 8–10 weeks (weighing 19–28 g) were purchased from the Vital River Laboratory Animal Technology Co., Ltd (Beijing, China). Homozygous AMPKα2-deficient (AMKPα2−/−) mice in the C57BL/6 background (weighing 18–26 g) were kindly provided by Dr. Benoit Viollet (Institute National de la Santé et de la Recherche Médicale U567, Paris). The genotype of the AMKPα2−/− mice was evaluated by PCR using tail DNA. All the experimental mice were housed under the 26 ± 1.5 °C, 40–60% humidity and specific pathogen-free conditions (fewer than five animals in a cage). All animal experiments were performed according to the regulation for animal management issued by the Ministry of Health of the People’s Republic of China (Document No. 55, 2001), and approved by the ethics committee of the Capital Medical University (Beijing, China; Ethical number: AEEI-2016-013).
Twenty-four C57BL/6 mice which were β1-AA negative confirmed by ELISA were equally randomized to three groups: vehicle group, β1-AR mAb group, and negative IgG group. Mice in β1-AR mAb group received intraperitoneal injection of β1-AR mAb at a dose of 5 μg/g biweekly. Mice in the vehicle group received the same dose of normal saline, and the mice in the negative IgG group received the same dose of negative IgG.
Protein microarray chip analysis
The expression of Treg cell-related proteins and cytokines in the β1-AA-positive mice was detected using a biotin-labeled mouse protein chip reagent kit. Briefly, each chip well was added with 100 µL sealing solution, cultured on the rocking bed for 30 min at room temperature. After sucking out the sealing solution, each well was added with 100 µL serum, cultured by oscillation at 4 °C overnight, and centrifuged at 13,000 rpm for 8 min. Each well was added with 70 µL biotin-labeled antibody and cultured at room temperature for 1 h after two washes of the plate. Then, 70 µL fluorant Cy3-streptomycin avidin was added to each well and cultured by oscillation at room temperature for 2 h. Finally, the fluorescent signal was detected by the Cy3 or green channel (532 nm).
Flow cytometric sorting for CD4+ T cells
Specific fluorescent antibody-labeled CD4+ T cells were separated from mouse spleen mononuclear cells by flow cytometry. C57BL/6 mice aged 10 weeks were euthanized by cervical dislocation to isolate splenocytes. Then, a single-cell suspension was prepared by using mechanical trituration method where the tissue was ground through a 300-mesh sieve. Subsequently, mouse spleen mononuclear cells were re-suspended using 50% and 70% Percoll separating media (GE Healthcare Life Sciences, USA). Density-gradient centrifugation was undertaken at 2500 rpm for 25 min following red blood cell lysis. The steps mentioned above were performed at a fast pace, at 4 °C or on ice. For cell surface staining, the antibodies (FITC-anti-CD4) were incubated with the single-cell suspension for at least 30 min at 4 °C. FITC-anti-CD4 antibody was purchased from BD Bioscience (USA). Cells were sorted with the flow cytometer FACS Aria II (Becton, Dickinson and Company).
Flow cytometric analysis for the proportion of nTreg cells
CD4+ T cells sorted from the splenocytes of healthy mice were with or without preactivation by anti-CD3/CD28 mAbs (1 μg/mL) (eBioscience, USA) for 72 h. Then, the cells were stimulated with different concentrations of β1-AA (10−8, 10−7, or 10−6 mol/L), with or without metoprolol (the β1-AR-specific blocker, 10−8 mol/L), for 24 h. Subsequently, CD4+ T cells were surface stained with anti-CD4/CD25 mAbs (FITC/phycoerythrin (PE)) for 30 min at 4 °C. After staining, cells were fixed and permeabilized using an intracellular fixation and permeabilization buffer set (eBioscience, USA), followed by intracellular staining with anti-FoxP3 mAb (BV421) (BD Bioscience, USA). The stained cells were centrifuged at 1500 rpm for 5 min, and re-suspended in 250 μL flow cytometric buffer solution (1% FBS in phosphate-buffered saline (PBS)). Flow cytometry was performed on the FACS Aria II flow cytometer (Becton, Dickinson and Company).
Effect of β1-AA on CD4+ T cell absorption of palmitic acid and subsequent FAO
BODIP is a fat-soluble fluorescent probe. Coupling of BODIP and palmitate can be used to observe the cellular FAO level (Life Technologies, USA). A 5 mmol stock solution was prepared by dissolving the BODIPY–palmitate into dimethyl sulfoxide, and then diluted in PBS buffer to a working concentration of 0.5 µmol. All intervention factors and BODIPY–palmitate were added to the activated CD4+ T cells and cultured at 37 °C for 48 h. Finally, the fluorescent intensity of each tube was detected with the FACS Aria II flow cytometer (Becton, Dickinson and Company).
Measurement of the ATP content
The ATP content in CD4+ T cells of β1-AA-positive mice was determined using a kit purchased from Beyotime Institute of Biotechnology (China) according to the manufacturer’s protocol.
Western blotting
The expression of AMPK in CD4+ T cells was determined by Western blot analysis. CD4+ T cells were separated from the splenic tissues of the β1-AA-positive mice and immediately lysed. The supernatant protein was extracted by centrifugation. The supernatant protein was analyzed by sodium dodecyl sulfate-polyacrylamide gel electrophoresis at a 50 μg sample volume. After electrophoresis, the PVDF membranes were transferred and blocked with 5% non-fat milk powder in TBST buffer for 1 h, and then incubated with anti-AMPKα2 mAb (1:1000; Abcam, Cambridge, UK), anti-phospho-AMPKα2 (Thr-172) mAb (1:1000; Abcam, Cambridge, UK) or anti-phospho-AMPKα2 (S491) mAb (1:1000; Abcam, Cambridge, UK) or anti-GAPDH mAb (Cell Signaling Tech., Danvers, MA, USA) at 4 °C overnight. The membranes were incubated with the corresponding secondary antibodies. Finally, the grayscale values of the straps were analyzed by Image J software after development.
Genotype identification of IL-10−/− mice
The genotype of AMPKα2−/− mice was identified by PCR of tail genomic DNA, using the following specific primers: 5′-GCT TAG CAC GTT ACC CTG GAT GG-3′ (forward, common), 5′-GCA TTG AAC CAC AGT CCT TCC TC-3′ (reverse 1, mutation), and 5′-GTT ATC AGC CCA ACT AAT TAC AC-3′ (reverse 2, wild type). Each 25-μl PCR mixture contained 20 pmol of each primer. The reaction conditions of PCR were as follows: 95 °C, 5 min; 94 °C, 30 s; 64 °C, 35 s, then 35 cycles of 72 °C, 45 s, followed by a final 3 min at 72 °C.
JC-1 staining
When the cellular MMP remained stable, JC-1 aggregated in the mitochondrial matrix, producing red fluorescence; when MMP was reduced, JC-1 was present as a monomer in the cellular matrix, producing green fluorescence. Therefore, changes in MMP can be detected by observing the percentage of the red and green fluorescence. Briefly, 48 h after stimulation of activated CD4+ T cells (106 cells per well) with β1-AA, cells were re-suspended in 0.5 mL RPMI medium 1640 (Hyclone, USA). After the addition of 0.5 mL JC-1 dye, cells were cultured in a 37 °C incubator for 20 min. Then, cells were re-suspended by the addition of 300 μL JC-1 staining buffer after being washed with JC-1 staining buffer in a centrifuge at 600 × g and 4 °C for 3 min twice. Finally, the red and green fluorescent intensity of each tube were detected and analyzed using the FACS Aria II flow cytometer.
Statistical analysis
Data are presented as mean ± SD. Statistical analysis was performed with the SPSS Statistics software (version 16.0, SPSS Inc., Chicago, IL, USA). The differences between groups were analyzed using independent sample t tests, one-way or two-way analysis of variance (between different mice strains). Histograms were produced by GraphPad Prism 6 (GraphPad Software Inc., USA). A P value <0.05 was considered statistically significant. Differences in the heat map of cluster analysis were statistically significant when the fold change between two groups was >1.5.
Electronic supplementary material
Supplemental methods and supplemental figures
Acknowledgements
We thank Professor Benoit Viollet (Institute National de la Santé et de la Recherche Médicale U567, Paris) for the generous gift of AMPKα2−/− mice. This study was supported by funds from the Natural Science Foundation of China (NSFC) to H.L. (Grant No. 81770393), the NSFC grant to W.L. (Grant No. 31871177), and the Scientific Research Base Construction-Scientific and Technological Innovation Platform Metabolic Disorders Related Cardiovascular Diseases Research Platform Construction to Huirong Liu (Grant No. PXM2015_014226_000062).
Conflict of interest
The authors declare that they have no conflict of interest.
Footnotes
Edited by H.-U. Simon
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Supplementary Information accompanies this paper at (10.1038/s41419-018-1209-2).
References
- 1.Procaccini C, et al. The proteomic landscape of human ex vivo regulatory and conventional T cells reveals specific metabolic requirements. Immunity. 2016;44:406. doi: 10.1016/j.immuni.2016.01.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Cuadrado E, et al. Proteomic analyses of human regulatory T cells reveal adaptations in signaling pathways that protect cellular identity. Immunity. 2018;48:1046. doi: 10.1016/j.immuni.2018.04.008. [DOI] [PubMed] [Google Scholar]
- 3.Gabrysova L, et al. c-Maf controls immune responses by regulating disease-specific gene networks and repressing IL-2 in CD4(+) T cells. Nat. Immunol. 2018;19:497. doi: 10.1038/s41590-018-0083-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Dinh TN, et al. Cytokine therapy with interleukin-2/anti-interleukin-2 monoclonal antibody complexes expands CD4+ CD25+ Foxp3+ regulatory T cells and attenuates development and progression of atherosclerosis. Circulation. 2012;126:1256. doi: 10.1161/CIRCULATIONAHA.112.099044. [DOI] [PubMed] [Google Scholar]
- 5.Weirather J, et al. Foxp3+ CD4+ T cells improve healing after myocardial infarction by modulating monocyte/macrophage differentiation. Circ. Res. 2014;115:55. doi: 10.1161/CIRCRESAHA.115.303895. [DOI] [PubMed] [Google Scholar]
- 6.Sanders VM. The beta2-adrenergic receptor on T and B lymphocytes: do we understand it yet? Brain Behav. Immun. 2012;26:195. doi: 10.1016/j.bbi.2011.08.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Bellinger DL, Lorton D. Autonomic regulation of cellular immune function. Auton. Neurosci. 2014;182:15. doi: 10.1016/j.autneu.2014.01.006. [DOI] [PubMed] [Google Scholar]
- 8.Freier E, et al. Decrease of CD4(+) FOXP3(+) T regulatory cells in the peripheral blood of human subjects undergoing a mental stressor. Psychoneuroendocrinology. 2010;35:663. doi: 10.1016/j.psyneuen.2009.10.005. [DOI] [PubMed] [Google Scholar]
- 9.Luft FC. Activating autoantibodies and cardiovascular disease. Physiology (Bethesda). 2013;28:254. doi: 10.1152/physiol.00014.2013. [DOI] [PubMed] [Google Scholar]
- 10.Stavrakis S, et al. Opposing cardiac effects of autoantibody activation of beta-adrenergic and M2 muscarinic receptors in cardiac-related diseases. Int. J. Cardiol. 2011;148:331. doi: 10.1016/j.ijcard.2009.11.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Limas CJ, Limas C. Beta-adrenoceptor antibodies and genetics in dilated cardiomyopathy—an overview and review. Eur. Heart J. 1991;12(Suppl. D):175. doi: 10.1093/eurheartj/12.suppl_D.175. [DOI] [PubMed] [Google Scholar]
- 12.Christ T, Schindelhauer S, Wettwer E, Wallukat G, Ravens U. Interaction between autoantibodies against the beta1-adrenoceptor and isoprenaline in enhancing L-type Ca2+ current in rat ventricular myocytes. J. Mol. Cell. Cardiol. 2006;41:716. doi: 10.1016/j.yjmcc.2006.06.011. [DOI] [PubMed] [Google Scholar]
- 13.Wallukat G. The β-adrenergic receptors. Herz. 2002;27:683. doi: 10.1007/s00059-002-2434-z. [DOI] [PubMed] [Google Scholar]
- 14.Liu HR, Zhao RR, Jiao XY, Wang YY, Fu M. Relationship of myocardial remodeling to the genesis of serum autoantibodies to cardiac beta(1)-adrenoceptors and muscarinic type 2 acetylcholine receptors in rats. J. Am. Coll. Cardiol. 2002;39:1866. doi: 10.1016/S0735-1097(02)01865-X. [DOI] [PubMed] [Google Scholar]
- 15.Baba A, et al. Complete elimination of cardiodepressant IgG3 autoantibodies by immunoadsorption in patients with severe heart failure. Circ. J. 2010;74:1372. doi: 10.1253/circj.CJ-09-0748. [DOI] [PubMed] [Google Scholar]
- 16.Zuo L, et al. Long-term active immunization with a synthetic peptide corresponding to the second extracellular loop of beta1-adrenoceptor induces both morphological and functional cardiomyopathic changes in rats. Int. J. Cardiol. 2011;149:89. doi: 10.1016/j.ijcard.2009.12.023. [DOI] [PubMed] [Google Scholar]
- 17.Matsui S, et al. Transfer of rabbit autoimmune cardiomyopathy into severe combined immunodeficiency mice. J. Cardiovasc. Pharmacol. 2003;42(Suppl. 1):S99. doi: 10.1097/00005344-200312001-00021. [DOI] [PubMed] [Google Scholar]
- 18.Du Y, et al. β1-adrenoceptor autoantibodies from DCM patients enhance the proliferation of T lymphocytes through the β1-AR/cAMP/PKA and p38 MAPK pathways. PLoS ONE. 2012;7:e52911. doi: 10.1371/journal.pone.0052911. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Bulut D, Creutzenberg G, Mugge A. The number of regulatory T cells correlates with hemodynamic improvement in patients with inflammatory dilated cardiomyopathy after immunoadsorption therapy. Scand. J. Immunol. 2013;77:54. doi: 10.1111/sji.12000. [DOI] [PubMed] [Google Scholar]
- 20.Dimitrijevic M, et al. Beta-adrenoceptor blockade ameliorates the clinical course of experimental allergic encephalomyelitis and diminishes its aggravation in adrenalectomized rats. Eur. J. Pharmacol. 2007;577:170. doi: 10.1016/j.ejphar.2007.08.021. [DOI] [PubMed] [Google Scholar]
- 21.Feinerman O, et al. Single-cell quantification of IL-2 response by effector and regulatory T cells reveals critical plasticity in immune response. Mol. Syst. Biol. 2010;6:437. doi: 10.1038/msb.2010.90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Webster KE, et al. In vivo expansion of T reg cells with IL-2-mAb complexes: induction of resistance to EAE and long-term acceptance of islet allografts without immunosuppression. J. Exp. Med. 2009;206:751. doi: 10.1084/jem.20082824. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Paterson AM, et al. Deletion of CTLA-4 on regulatory T cells during adulthood leads to resistance to autoimmunity. J. Exp. Med. 2015;212:1603. doi: 10.1084/jem.20141030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Alikhan MA, Huynh M, Kitching AR, Ooi JD. Regulatory T cells in renal disease. Clin. Transl. Immunol. 2018;7:e1004. doi: 10.1002/cti2.1004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Oo YH, et al. Distinct roles for CCR4 and CXCR3 in the recruitment and positioning of regulatory T cells in the inflamed human liver. J. Immunol. 2010;184:2886. doi: 10.4049/jimmunol.0901216. [DOI] [PubMed] [Google Scholar]
- 26.Michalek RD, et al. Cutting edge: distinct glycolytic and lipid oxidative metabolic programs are essential for effector and regulatory CD4+ T cell subsets. J. Immunol. 2011;186:3299. doi: 10.4049/jimmunol.1003613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Lochner M, Berod L, Sparwasser T. Fatty acid metabolism in the regulation of T cell function. Trends Immunol. 2015;36:81. doi: 10.1016/j.it.2014.12.005. [DOI] [PubMed] [Google Scholar]
- 28.Senni, N. et al. β-catenin-activated hepatocellular carcinomas are addicted to fatty acids. GUT68, 322 (2019). [DOI] [PubMed]
- 29.Hwang JH, et al. Enhanced production of adenosine triphosphate by pharmacological activation of adenosine monophosphate-activated protein kinase ameliorates acetaminophen-induced liver injury. Mol. Cells. 2015;38:843. doi: 10.14348/molcells.2015.0072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Gualdoni GA, et al. The AMP analog AICAR modulates the Treg/Th17 axis through enhancement of fatty acid oxidation. FASEB J. 2016;30:3800. doi: 10.1096/fj.201600522R. [DOI] [PubMed] [Google Scholar]
- 31.Deng M, et al. Deubiquitination and activation of AMPK by USP10. Mol. Cell. 2016;61:614. doi: 10.1016/j.molcel.2016.01.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Coughlan KA, et al. PKD1 inhibits AMPKalpha2 through phosphorylation of serine 491 and impairs insulin signaling in skeletal muscle cells. J. Biol. Chem. 2016;291:5664. doi: 10.1074/jbc.M115.696849. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Canto C, et al. Neuregulins increase mitochondrial oxidative capacity and insulin sensitivity in skeletal muscle cells. Diabetes. 2007;56:2185. doi: 10.2337/db06-1726. [DOI] [PubMed] [Google Scholar]
- 34.Bin F, et al. Silymarin protects against renal injury through normalization of lipid metabolism and mitochondrial biogenesis in high fat-fed mice. Free Radic. Biol. Med. 2017;110:240. doi: 10.1016/j.freeradbiomed.2017.06.009. [DOI] [PubMed] [Google Scholar]
- 35.Wallukat G, Wollenberger A. Effects of the serum gamma globulin fraction of patients with allergic asthma and dilated cardiomyopathy on chronotropic beta adrenoceptor function in cultured neonatal rat heart myocytes. Biomed. Biochim. Acta. 1987;46:S634. [PubMed] [Google Scholar]
- 36.Nagatomo Y, et al. A pilot study on the role of autoantibody targeting the beta1-adrenergic receptor in the response to beta-blocker therapy for congestive heart failure. J. Card. Fail. 2009;15:224. doi: 10.1016/j.cardfail.2008.10.027. [DOI] [PubMed] [Google Scholar]
- 37.Jahns R, et al. Autoantibodies activating human beta1-adrenergic receptors are associated with reduced cardiac function in chronic heart failure. Circulation. 1999;99:649. doi: 10.1161/01.CIR.99.5.649. [DOI] [PubMed] [Google Scholar]
- 38.Liu HR, Zhao RR, Zhi JM, Wu BW, Fu ML. Screening of serum autoantibodies to cardiac beta1-adrenoceptors and M2-muscarinic acetylcholine receptors in 408 healthy subjects of varying ages. Autoimmunity. 1999;29:43. doi: 10.3109/08916939908995971. [DOI] [PubMed] [Google Scholar]
- 39.Li N, et al. The Th17/Treg imbalance exists in patients with heart failure with normal ejection fraction and heart failure with reduced ejection fraction. Clin. Chim. Acta. 2010;411:1963. doi: 10.1016/j.cca.2010.08.013. [DOI] [PubMed] [Google Scholar]
- 40.Okamoto N, et al. Prognostic value of circulating regulatory T cells for worsening heart failure in heart failure patients with reduced ejection fraction. Int. Heart J. 2014;55:271. doi: 10.1536/ihj.13-343. [DOI] [PubMed] [Google Scholar]
- 41.Meng X, et al. Regulatory T cells in cardiovascular diseases. Nat. Rev. Cardio. 2016;13:167. doi: 10.1038/nrcardio.2015.169. [DOI] [PubMed] [Google Scholar]
- 42.Tang TT, et al. Defective circulating CD4+ CD25+ Foxp3+ CD127(low) regulatory T-cells in patients with chronic heart failure. Cell. Physiol. Biochem. 2010;25:451. doi: 10.1159/000303050. [DOI] [PubMed] [Google Scholar]
- 43.Bansal SS, et al. Activated T lymphocytes are essential drivers of pathological remodeling in ischemic heart failure. Circ. Heart Fail. 2017;10:e3688. doi: 10.1161/CIRCHEARTFAILURE.116.003688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Zhou L, et al. Propranolol attenuates surgical stress-induced elevation of the regulatory T cell response in patients undergoing radical mastectomy. J. Immunol. 2016;196:3460. doi: 10.4049/jimmunol.1501677. [DOI] [PubMed] [Google Scholar]
- 45.Berod L, et al. De novo fatty acid synthesis controls the fate between regulatory T and T helper 17 cells. Nat. Med. 2014;20:1327. doi: 10.1038/nm.3704. [DOI] [PubMed] [Google Scholar]
- 46.Lochner M, Berod L, Sparwasser T. Fatty acid metabolism in the regulation of T cell function. Trends Immunol. 2015;36:81. doi: 10.1016/j.it.2014.12.005. [DOI] [PubMed] [Google Scholar]
- 47.Wang L, et al. Decreased autophagy induced by beta1-adrenoceptor autoantibodies contributes to cardiomyocyte apoptosis. Cell Death Dis. 2018;9:406. doi: 10.1038/s41419-018-0445-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Perry SW, Norman JP, Barbieri J, Brown EB, Gelbard HA. Mitochondrial membrane potential probes and the proton gradient: a practical usage guide. Biotechniques. 2011;50:98. doi: 10.2144/000113610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.O’Neill HM, Holloway GP, Steinberg GR. AMPK regulation of fatty acid metabolism and mitochondrial biogenesis: implications for obesity. Mol. Cell. Endocrinol. 2013;366:135. doi: 10.1016/j.mce.2012.06.019. [DOI] [PubMed] [Google Scholar]
- 50.Herms A, et al. AMPK activation promotes lipid droplet dispersion on detyrosinated microtubules to increase mitochondrial fatty acid oxidation. Nat. Commun. 2015;6:7176. doi: 10.1038/ncomms8176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Brockhoff M, et al. Targeting deregulated AMPK/mTORC1 pathways improves muscle function in myotonic dystrophy type I. J. Clin. Invest. 2017;127:549. doi: 10.1172/JCI89616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Hutchinson DS, Chernogubova E, Dallner OS, Cannon B, Bengtsson T. Beta-adrenoceptors, but not alpha-adrenoceptors, stimulate AMP-activated protein kinase in brown adipocytes independently of uncoupling protein-1. Diabetologia. 2005;48:2386. doi: 10.1007/s00125-005-1936-7. [DOI] [PubMed] [Google Scholar]
- 53.Guereschi MG, et al. Beta2-adrenergic receptor signaling in CD4+ Foxp3+ regulatory T cells enhances their suppressive function in a PKA-dependent manner. Eur. J. Immunol. 2013;43:1001. doi: 10.1002/eji.201243005. [DOI] [PubMed] [Google Scholar]
- 54.Rubtsov YP, et al. Regulatory T cell-derived interleukin-10 limits inflammation at environmental interfaces. Immunity. 2008;28:546. doi: 10.1016/j.immuni.2008.02.017. [DOI] [PubMed] [Google Scholar]
- 55.Nikolaev VO, et al. A novel fluorescence method for the rapid detection of functional beta1-adrenergic receptor autoantibodies in heart failure. J. Am. Coll. Cardiol. 2007;50:423. doi: 10.1016/j.jacc.2007.03.051. [DOI] [PubMed] [Google Scholar]
- 56.Freedman NJ, Lefkowitz RJ. Anti-beta(1)-adrenergic receptor antibodies and heart failure: causation, not just correlation. J. Clin. Invest. 2004;113:1379. doi: 10.1172/JCI21748. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplemental methods and supplemental figures