Abstract
Interest in nicotinic acetylcholine receptor (nAChR) ligands as potential therapeutic agents for cognitive disorders began more than 30 years ago when it was first demonstrated that the tobacco alkaloid nicotine could improve cognitive function in nicotine-deprived smokers as well as nonsmokers. Numerous animal and human studies now indicate that nicotine and a variety of nAChR ligands have the potential to improve multiple domains of cognition including attention, spatial learning, working memory, recognition memory, and executive function. The purpose of this review is to (1) discuss several pharmacologic strategies that have been developed to enhance nAChR activity (eg, agonist, partial agonist, and positive allosteric modulator) and improve cognitive function, (2) provide a brief overview of some of the more common rodent behavioral tasks with established translational validity that have been used to evaluate nAChR ligands for effects on cognitive function, and (3) briefly discuss some of the topics of debate regarding the development of optimal therapeutic strategies using nAChR ligands. Because of their densities in the mammalian brain and the amount of literature available, the review primarily focuses on ligands of the high-affinity α4β2* nAChR (“*” indicates the possible presence of additional subunits in the complex) and the low-affinity α7 nAChR. The behavioral task discussion focuses on representative methods that have been designed to model specific domains of cognition that are relevant to human neuropsychiatric disorders and often evaluated in human clinical trials.
Implications
The preclinical literature continues to grow in support of the development of nAChR ligands for a variety of illnesses that affect humans. However, to date, no new nAChR ligand has been approved for any condition other than nicotine dependence. As discussed in this review, the studies conducted to date provide the impetus for continuing efforts to develop new nAChR strategies (ie, beyond simple agonist and partial agonist approaches) as well as to refine current behavioral strategies and create new animal models to address translational gaps in the drug discovery process.
Introduction
Interest in the prototypic tobacco alkaloid nicotine as a potential therapeutic agent began nearly more than 30 years ago when it was discovered that tobacco smoking could result in enhancements in the performance of certain cognitive tasks. A limitation to these early observations was that at least some of the task improvements were only reversals of withdrawal-induced performance deficits in tobacco-dependent smokers. However, with additional studies and more extensive reviews of the literature, a general consensus developed that smoking or other means of nicotine administration could have a modest pro-cognitive effect in nonsmokers as well as nondeprived or minimally deprived smokers.1–3 There is now a large body of evidence to support the premise that nicotine can enhance information processing and cognitive function in experimental animals as well as in human nonsmokers.4 A small representative list of examples includes positive effects on sensorimotor gating and prepulse inhibition,5 sustained attention,6 working memory,7 and recognition memory8 in rats; working and/or short-term memory9,10 and distractibility11 in monkeys; and prepulse inhibition,12 visual information processing, attention, and short-term memory in humans.13–16 In addition to its pro-cognitive effects, nicotine has also been shown to have neuroprotective activity in multiple disease models (ie, both in vitro and in vivo), and this has bolstered the argument that nicotine might have both symptomatic and disease-modifying effects in neurodegenerative illnesses such as Alzheimer’s disease (AD) and Parkinson’s disease.17,18
Concerns about its relatively short half-life, cardiovascular effects, and abuse potential have (at least to some extent), however, limited enthusiasm for the use of nicotine as a therapeutic agent for neurologic and psychiatric conditions to date with the exception of nicotine dependence. It should be noted, however, that nicotine delivered via a transdermal patch for smoking cessation showed no increased risk of cardiovascular events compared with placebo in patients with cardiovascular disease.19 Moreover, nicotine delivered via a transdermal patch to patients with mild cognitive impairment improved cognition without signs of significant side effects.20 For smoking cessation, nicotine is now delivered safely as a replacement therapy via multiple formulations including patches, tablets, gums, lozenges, nasal sprays, and inhalers to relieve withdrawal symptoms and cravings without the harmful effects of tobacco smoke.21 Collectively, this information supports the argument that nicotine should continue to be evaluated as a potential therapeutic agent in conditions beyond nicotine dependence.
Central Nervous System Targets of Nicotinic Acetylcholine Receptor Ligands
In an attempt to address the limitations of nicotine noted earlier, and to enhance pro-cognitive actions and other desirable therapeutic properties, a wide range of ligands targeting specific subtypes of nicotinic acetylcholine receptors (nAChRs) have been developed in the last 20 years, and there is now evidence to support their use in multiple types of diseases that affect humans. (See Table 1 for a list of these potential indications and representative references cited for review.) As therapeutic targets, nAChRs belong to the large superfamily of pentameric, ligand-gated ion channel receptors that include serotonin 5-HT3, type-A γ-aminobutyric acid, and glycine receptors. By modulating the flux of cations such as Na+, K+, and Ca++ across cell membranes, nAChRs regulate neuronal excitability and neurotransmitter release to influence multiple physiologic processes including behavior.22 Neuronal nAChRs are formed through a combination of α and β subunits composing heteromeric receptors, or through homomeric α configurations. Although there have been nine α (α2−α10) and three β (β2−β4) subunits identified to date that can form many different combinations, for the purposes of this review, the heteromeric α4β2* and homomeric α7 nAChR subtypes are primarily discussed. These two nAChR receptors are the most predominant subtypes found in the mammalian brain and the most commonly targeted receptor subtypes in drug discovery programs to date, especially for disorders of cognitive function. Compounds selective for the α4β2* subtype were the first to be developed in drug discovery programs for cognition enhancement. This may (at least in part) be related to the high density of α4β2 nAChRs in the brain (relative to other nAChR subtypes), their high affinity for nicotine, and the large body of literature available on their role in reward, self-administration, and dependence.22 During the characterization of α4β2* nAChR agonists, it was discovered that α4 and β2 subunits assembled to form two stoichiometries with different pharmacological sensitivities toward acetylcholine (ACh) activation. The α4(2):β2(3) subunit possesses high sensitivity (EC50 = 1 μM) to ACh whereas the α4(3):β2(2) subunit possesses low sensitivity (EC50 = 100 μM) to ACh.23,24 Moreover, it was demonstrated that several α4β2* nAChR ligands (eg, nicotine, NS9283, LY2087101, and varenicline) showed distinct preferences for the high- and low-sensitivity α4β2 subunit populations. Although the therapeutic relevance has yet to be determined, the ability to target one α4β2 nAChR subtype over another will likely rejuvenate future α4β2 nAChR drug development efforts.
Table 1.
Therapeutic Potential of Nicotinic Acetylcholine Receptor Ligands
In addition to α4β2* nAChR ligands, considerable attention has also been given toward the development of homomeric α7 nAChR ligands for the treatment of cognitive disorders. Neuronal α7 nAChRs are abundant in brain regions (eg, prefrontal cortex and hippocampus) that are important for cognition, they influence the phosphorylation of ERK and CREB (signaling pathways linked to long-term potentiation and memory formation), they are often reduced in AD and schizophrenia, and they can promote neuroprotection in model systems.22
The majority of nAChR ligands developed to date are agonists or partial agonists targeted to orthosteric α4β2* and α7 nAChR binding sites, although more recently, several positive allosteric modulators (PAMs) at nAChRs have been developed.22 PAMs avoid direct receptor activation by binding at sites that are distinct from the orthosteric sites targeted by traditional α4β2* and α7 nAChR agonists and thus may offer some pharmaceutical advantages. For example, in contrast to traditional nAChR agonists, PAMs would be expected to reduce the likelihood of agonist-induced nAChR desensitization (and potential tolerance) following continuous administration to maintain cholinergic activity. PAMs require endogenous ligand (ie, ACh or choline) to elicit a nAChR response and are, therefore, thought to maintain a more natural (temporal phasic) stimulation pattern of the cholinergic receptor than agonists. However, unlike nAChR agonists (and a potential caveat), there is the requirement that PAMs have sufficient levels of endogenous cholinergic ligand present to activate the nAChR, which may not be the case in all types of cognitive disorders (eg, late stage AD). Despite the functional differences between the nAChR ligands (ie, full vs. partial agonist vs. PAM) and the question of which might be more beneficial as a therapeutic agent, results from early stage clinical trials indicate that, at therapeutically relevant doses, pharmacological tolerance and loss of clinical efficacy (cognitive improvement) do not occur following stabilized daily dosing with either full or partial nAChR agonists.16,20,22,31 Unfortunately, too few clinical investigations exist for nAChR PAMs to determine their “therapeutic-use” potential or advantages over nAChR agonists.22,39
In the nAChR field, most of the attention on PAMs to date has focused on the α7 nAChR, though a few reports have identified selective α4β2* nAChR PAMs,40 with compounds divided into two classes based on their functional properties: type I and type II PAMs. Type I PAMs are defined as molecules that predominately affect the apparent peak current, agonist sensitivity, and Hill coefficient, but not the receptor desensitization profile, and type II PAMs are defined as compounds that possess the aforementioned properties described for type I PAMs, as well as the ability to modify the desensitization profile of agonist responses. It has been argued that type II PAMs are less prone to induce tolerance, which may occur after the chronic administration of nAChR agonists, whereas, type I PAMs may have advantages over type II PAMs as they can minimize potential calcium-induced cytotoxicity. See the following reviews to support the information in the earlier paragraphs related to the physiology and pharmacology of nAChRs and their role as therapeutic targets.22,41
Animal Models and Animal Behavior Tasks: Translational Validity
With the long list of recent clinical trial failures of novel neurologic and psychiatric medications (including nAChR ligands) has come increasing scrutiny of both the animal models and the behavioral test procedures used in preclinical drug discovery research. The subject of “translational validity” in preclinical animal studies has become a hotly debated topic. Regarding the animal models used (eg, transgenic mice, brain lesion models, and pharmacologic impairment models), the degree of phenomenological similarity between the animal model and the human condition it is meant to simulate is known as “face validity”. Here, for example, an animal model of AD would be expected to exhibit deficits in attention, learning, memory, recall, etc. The level of homology in pathological mechanisms (ie, between the animal model and the human illness) that underlie disease symptoms is known as “construct validity”. An optimal AD model would thus be expected to demonstrate amyloid plaques, neurofibrillary tangles, synaptic loss, etc. in brain regions that are affected in human AD (eg, cortex and hippocampus). In drug discovery research, “predictive validity” is primarily defined by the degree to which the model can be used to predict efficacy of a new therapeutic agent in humans. In an animal model of AD, this might be demonstrated by its response to a positive control compound (eg, donepezil). However, in AD (as well as most neurologic and psychiatric conditions), demonstrating construct and predictive validity in animal models is very challenging because the neurobiology of AD remains poorly understood and because the currently marketed prescription medications (ie, the positive controls) are only marginally effective.
As the case for animal models, the same terms used for translational validity are often applied to the behavioral procedures that are used, although it should be noted that the task requires an animal model, thus the terms often overlap and become more difficult to clearly define. For example, a working memory task that shows a delay-dependent decrement in accuracy by an animal would demonstrate both face validity and at least some components of construct validity (relative to human working memory tasks). In the context of drug discovery research, construct validity of the tasks that assess specific domains of cognition is the most common focus. Here, it is important to note that a clear definition of the specific cognitive domain (construct) must be formulated. The concept of etiological validity is closely related to construct validity and focuses on the anatomical substrates that have been determined to be essential to a specific cognitive domain as reflected in performance on a particular task. As an example, the dependence of a spatial learning task such as the Morris water maze on an intact hippocampus can be used as an evidence of the construct (etiological) validity of the task since damage to the hippocampus impairs spatial learning in humans. For a comprehensive review related to animal behavior task validity, see reference.42
In the text provided later, an overview of the domains of cognition commonly targeted in nAChR drug discovery programs is provided. The review focuses on several rodent behavioral tasks that have been developed and validated (using the criteria discussed earlier). These tasks also map well onto the domains of cognition that are often evaluated in clinical trials in humans for dementia and other neuropsychiatric disorders such as schizophrenia where cognitive deficits are a primary debilitating feature. Important developments in areas more closely related to fear memories and anxiety disorders are beyond the scope of this review. Nicotine has been extensively characterized (ie, acute, chronic, and withdrawal effects) in the fear conditioning model,43,44 see also reviews by Kaplan and Moore45 and Kutlu and Gould.46 In Table 2, an overview of representative ligands at α4β2* and α7 nAChRs that have been developed to date to treat cognitive disorders is provided. Although not all-inclusive, the table includes nAChR agonists, partial agonists, and PAMs that have been evaluated in the specific behavioral tasks that are discussed. Table 2 also provides information on additional properties of the compounds beyond memory enhancement (eg, neuroprotective effects and effects on sensorimotor gating) that might be useful for treating conditions such as AD and schizophrenia.
Table 2.
In vivo Pharmacological Effects of Nicotinic Acetylcholine Receptor Ligands
| Compound name | Receptor function | Cognitive domain enhanced | Additional properties | References |
|---|---|---|---|---|
| ABT-089 | α4β2 and α6β2 partial agonist | Attention, working memory, spatial reference memory | Neuroprotective (glutamate excitotoxicity) | 47–49 |
| ABT-594 | α4β2 Full agonist | Attention, working memory | Analgesic | 50–52 |
| AZD-3480 (TC-1734) |
α4β2 Partial agonist | Attention, object recognition, working memory, executive function | Neuroprotective (glutamate), antidepressant | 53–55 |
| Sazetidine-A | α4β2 Full agonist (silent desensitizer) | Attention | Analgesic, antidepressant | 56–58 |
| Varenicline | α4β2 Partial agonist and full α7 agonist | Attention, object recognition, working memory | Auditory and sensorimotor gating | 59–61 |
| NS9283 | (α4)3(β2)2 PAM | Attention, social recognition, spatial reference memory | Sensorimotor gating | 23 |
| ABBF | α7 Full agonist and 5-HT3 antagonist | Object and social recognition, working memory | 62 | |
| ABT-107 | α7 Full agonist | Attention, working memory, social recognition | Auditory gating neuroprotective (6-OHDA lesion model), | 63–65 |
| AQ051 | α7 Partial agonist | Object and social recognition, spatial reference memory | 66 | |
| BMS-933043 | α7 Partial agonist | Object recognition, working memory, executive function | Auditory gating | 67 |
| BMS-902483 | α7 Partial agonist and 5-HT3 antagonist | Object recognition, executive function | Auditory gating | 68 |
| EVP-6124 | α7 Partial agonist and 5-HT3 antagonist | Object recognition | 69 | |
| EVP-5141 | α7 Full agonist and 5-HT3 antagonist | Object and social recognition, working memory | 70 | |
| SEN-12333 | α7 Full agonist and histamine H3 antagonist | Object recognition | Neuroprotective (quisqualate lesion model), sensorimotor gating | 71 |
| RG 3487 | α7 Partial agonist and 5-HT3 antagonist | Attention, object recognition, spatial reference memory, executive function | Sensorimotor gating | 72 |
| TC-5619 | α7 Full agonist | Object recognition | Sensorimotor gating | 73 |
| Tropisetron | α7 Partial agonist and 5-HT3 antagonist | Attention, object recognition, working memory, spatial reference memory | Sensorimotor gating | 74–76 |
| AVL-3288 (Compound 6, CCMI or XY4083) |
α7 Type I PAM | Object recognition, working memory, executive function | Auditory gating | 77,78 |
| Compound 7z | α7 Type I PAM | Object recognition | 79 | |
| JNJ-39393406 | α7 Type I, type II PAM | Object recognition, executive function | Auditory gating | 39 |
| Lu AF58801 | α7 Type I PAM | Object recognition | 80 | |
| NS1738 | α7 Type I PAM | Object and social recognition, spatial reference memory | 81,82 | |
| PAM-2 | α7 Type II PAM | Object recognition, executive function | Social interaction model | 83 |
| PNU 120596 | α7 Type II PAM | Recognition memory, spatial learning, working memory, executive function | Auditory gating | 84,85 |
PAM = positive allosteric modulator.
Domains of Cognition Commonly Targeted in Preclinical nAChR Drug Discovery Programs
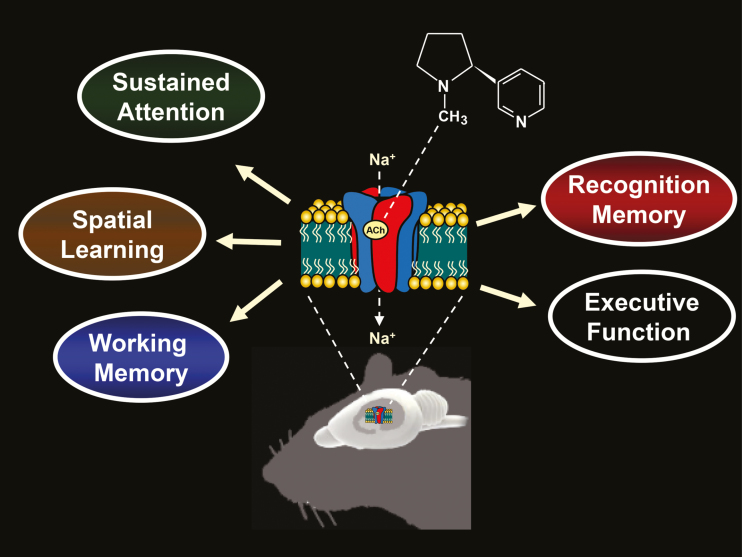
Please see Figure 1 for an illustration of several domains of cognition that are commonly targeted in rodent models in nAChR drug discovery programs.
Figure 1.
Illustration of the several domains of cognition often targeted in rodent models by nicotinic acetylcholine receptor ligands in drug discovery programs for neuropsychiatric disorders.
Attention
A simple definition of attention is the concentration of nervous system resources on specific (environmentally relevant) stimuli while ignoring other (nonrelevant or less relevant) stimuli. The ability to focus one’s attention is inextricably linked to perception, learning and memory, and executive function, and this ability is often impaired in a variety of neuropsychiatric disorders, including attention-deficit hyperactivity disorder (ADHD), depression, schizophrenia, and AD. Accordingly, attention is considered as a major therapeutic target in these disorders.86 In drug discovery research, the most commonly used attention-related tasks are the five-choice serial reaction time task (5-CSRTT) and the five-choice continuous performance task (5C-CPT) followed by the signal detection task (SDT). Each of these behavioral tasks has features that are similar to the continuous performance task (CPT) that has been used successfully to detect attention deficits in the aforementioned human neuropsychiatric disorders.86
The 5-CSRTT (Figure 2A), a task of sustained attention, assesses the subject’s ability to spatially divide its attention across multiple signal locations to select the correct target stimulus (light in a single aperture and nose-poke location) that, in turn, produces a food reward. In addition to measuring attention (choice accuracy), the 5-CSRTT can assess a number of other cognitive domains such as impulsivity (premature responses), cognitive inflexibility and/or compulsivity (perseverative and timeout responses), and processing speed (response latency). Like the 5-CSRTT, the 5C-CPT assesses the subject’s ability to spatially divide its attention across five signal locations and when illumination of a single aperture (correct target) occurs the subject must choose that hole to receive a reward. However, the 5C-CPT adds a nontarget component in which all five apertures are illuminated, thus requiring the subject to inhibit choice selection. The addition of nontarget trials (false alarms) with correct target trials increases task similarity to the human CPT version42 and (along with the other standard 5-CSRTT measurements) allows the assessment of response inhibition, which is an important component of executive functioning.
Figure 2.
Diagrams of representative rodent tasks often used in drug discovery programs that were developed to model specific domains of cognition. (A) Sustained attention: the five-choice serial reaction time task (5-CSRTT). In the 5-CSRTT, subjects are required to scan five apertures in an operant chamber for the appearance of a brief light stimulus and to make a nose-poke response in the correct spatial location (ie, the aperture where the light stimulus was presented) to receive a food reward. (B) Spatial learning and reference memory: the Morris water maze (MWM). The MWM task assesses the ability of rodents to learn and remember the spatial location of visual cues in a room to find and escape onto a hidden platform located in a pool of water. In probe trials (reference memory) conducted after hidden platform tests, the platform is removed from the pool and the time spent and distance traveled in the quadrant that previously contained the platform is measured. (C) Spatial working memory: the radial arm maze (RAM). In The RAM, subjects move down adjacent arms in a radial maze to retrieve food pellets without reentering empty (ie, unbaited) arms. By remembering which arms were previously entered (ie, by using spatial cues in the room on each trial) the rats use “spatial working memory” to successfully complete the task. (D) Recognition memory: spontaneous novel object recognition (NOR). During the sample trial (A/A), the subject is presented with two identical objects and allowed to explore the objects for a predetermined length of time. After a delay interval, a choice test trial (A/B) is performed in which the subject is presented with one of the previously observed “familiar” objects and a “novel”, unfamiliar object. Recognition memory is demonstrated in the NOR task when the subject explores the novel object more than a previously observed familiar one. (E) Executive function: -attentional set-shifting task (AST). In the food cup-based version of the AST, rodents are trained to retrieve food rewards buried in containers that contain fillers of different textures and (typically) different scents (olfactory cues). To obtain rewards the subjects have to be able to discriminate between the different textures and odors and then to switch between different textures or between odors (intradimensional set shift) or to shift from a specific texture to an odor (extradimensional set shifts) or vice versa. They are also required to perform reversals either within or between dimensions. SD = simple discrimination; CD = complex discrimination; ID = intradimensional set shift; ED = extradimensional set shift.
The SDT was designed to emphasize temporal components of attention by presenting a target stimulus repeatedly in a single spatial location and varying the timing of its presentation. The SDT thus differs from the 5-CSRTT and 5C-CPT in that the SDT requires the detection of a single centrally presented visual signal instead of detection of a target stimulus across multiple choice locations. Briefly, the SDT requires the subject to monitor a central panel for the presence (signal trial) or absence (nonsignal or blank trial) of a visual light stimulus that varies in time and intensity. Following the trial (signal or nonsignal) presentation and a short variable delay, two response levers are extended into the operant chamber and the subject must press the appropriately designated lever, based on trial type presented, to receive a reward. The SDT (similar to the human CPT) thus incorporates the use of a rule about the properties of a single stimulus. The method also allows the investigator to alter stimulus duration, intensity, and modality, which can place greater cognitive load on the subject to better assess sensory function and attentiveness. Distraction (eg, flashing house light) can be added to the protocol to further increase task demands. For a more comprehensive review of attention and rodent models designed to study attentional processes see Callahan and Terry.86
Results with nicotine and α4β2* nAChR agonists (eg, ABT-418, A-82695, AZD 3480, sazetidine, and varenicline) on improving sustained attention and inhibitory control have been mixed (Table 2).86 This drug class has routinely been shown to reverse the pharmacologic-induced impairments (ie, scopolamine and MK-801) of sustained attention, whereas their ability to consistently act as “stand-alone” pro-attentional drugs varies across laboratories.86 Fewer studies exist for α4β2* nAChR PAMs. Timmermann et al.23 assessed the low-sensitivity α4(3)β2(2) nAChR PAM NS 9283 on sustained attention using the 5-CSRTT. Preliminary results indicated that NS 9283 produced small improvements in accuracy and latency to response with decreases in trial omissions and premature responding. In a follow-up study, positive effects on trial omissions were replicated, whereas the effects on accuracy, response latency, and premature responding were not reproduced, indicating that additional investigations on attentional processes are warranted. Mixed results on sustained attention with α7 nAChR agonists and α7 nAChR PAMs have also been reported.86 The α7 nAChR partial agonist/5-HT3 antagonist RG 3487 was shown to exert beneficial effects on sustained attention comparable to that of nicotine in the rat SDT whereas, the α7 nAChR agonists (ie, AR-R17779 and PNU 282987) and PAMs (ie, CCMI and PNU 120596) did not improve attentional performance in the 5-CSRTT.84,86 Nicotinic α4β2* and α7 nAChR agonist administration has also resulted in mixed outcomes in clinical trials of ADHD despite being well tolerated. Agonists ABT-089, AZD-3480, and ABT-894 were reported to improve ADHD symptoms as measured by the Conner’s Adult ADHD Rating Scale-Investigator Version (CAARS-INV) whereas the α4β2 agonist AZD 1446 and the α7 nAChR agonist TC-5619 failed to significantly improve ADHD symptoms compared with placebo. Interestingly, the α7 nAChR partial agonist/5-HT3 antagonist tropisetron was shown to improve sustained visual attention (ie, rapid visual information processing) in patients with schizophrenia (for review see Terry et al.87). Because RG 3487 and tropisetron are also potent 5-HT3 receptor antagonists and blockade of this receptor site also increases the release of ACh, the dual action at both α7 nAChR and 5-HT3 receptors may account for the observed behavioral and clinical differences on attentional processing.
In summary, there is considerable preclinical (and some clinical) data to support the premise that ligands at both α4β2 and α7 nAChRs have the potential to enhance attention and to improve other behavioral domains related to attention (impulsivity). However, not all studies have reached this conclusion, warranting further investigation. There is a variety of potential reasons for the mixed results including the nature of the specific attention task used and its parameters (eg, stimulus types and durations), and routes of drug administration. Interestingly, Hahn et al.88 recently observed distinct rat strain-related differences in the attention-enhancing effects of nicotine in the 5-CSRTT, which would indicate that genetic predispositions may predict variability in the efficacy of nAChR compounds for enhancing attention.
Spatial Learning and Reference Memory
For more than 35 years, the Morris water maze has been used extensively by behavioral physiologists and pharmacologists for evaluating the effects of aging, experimental lesions, and drugs on learning and memory in rodents. The most commonly used version of the task (the fixed, hidden platform task; Figure 2B) is a test of spatial learning where the test subject relies on distal cues to navigate from start locations around the perimeter of an open swimming pool to locate a submerged platform to escape the water. Spatial learning ability is assessed across repeated (hidden platform) trials and reference memory (recall) is determined in probe trials by the preference of the subject for the platform area when the platform has been removed from the pool. The task requires many of the same processes known to be important components of human cognition including information acquisition and processing, consolidation, retention, and retrieval. Moreover, with a computer-automated video tracking system a number of nonmnemonic processes such as thigmotaxis, motor function (swim speeds), or motivational responses can be evaluated (for a more comprehensive review of water maze procedures used in drug discovery, see Terry89).
Nicotine, α4β2*, and α7 nAChR agonists have been shown to consistently improve Morris water maze spatial reference memory in aged (24-month-old) rodents, in rodents with chemical and electrolytic lesions, as well as in rodents with pharmacologic-induced impairments (Table 2).47,66,70,72 The α4(3)β2(2) nAChR PAM NS 9283 was also shown to reverse the impairing effects of subchronic phencyclidine administration on spatial reference memory in rats.59 The α7 nAChR type I PAM NS 1738 has been shown to reverse scopolamine-induced water maze deficits in young rats81 whereas the α7 nAChR type II PAM PNU 120596 failed to reverse the spatial reference memory deficits of aged, cognitively impaired F344 rats.90 Interestingly, increasing cholinergic tone via administration of a subthreshold dose of donepezil in combination with PNU 120596 improved spatial reference memory performance in aged, cognitively impaired animals.90
In summary, most of the data collected to date across a variety of animal models support the argument that both α4β2* and α7 nAChR agonists have the potential to enhance spatial learning and reference memory, whereas additional data are needed before this conclusion can be made for types I and II nAChR PAMs.
Working Memory
The term working memory has been defined in multiple ways, but in short, it refers to a brain system that enables the acquisition, temporary storage, and manipulation of information that is necessary to solve complex cognitive tasks such as language comprehension, learning, reasoning, and problem solving. Working memory is limited in capacity and may contain information about rules, recent stimulus events, expected goals, and planned actions that are maintained and manipulated in real time to affect ongoing behavior. Behavioral tasks in laboratory animals typically assess their ability to use this system to encode, temporarily maintain, and use sensory stimulus information to make a decision (sensory discrimination of some type) to receive a reward. These include the so-called delayed response tasks where the stimuli are either visuospatial or object oriented (eg, spatial delayed alternation or response, delayed match or nonmatch to position, and delayed match or nonmatch to sample).91 The tasks can be computer-automated in Skinner-type operant chambers with levers or nose-poke devices for response selection. One of the oldest and simplest but best validated methods of assessing working memory in rodents is the win-shift task in the eight-arm radial arm maze (RAM). In brief, the RAM typically uses an octagonal central chamber with eight attached arms (Figure 2C). Each arm is baited with a food reward and the animal is required to enter each arm to retrieve the reward. To complete the task most efficiently, the animal must not reenter a previously visited arm and adopt a win-shift foraging strategy. Spatial working memory performance of the animal is often measured by the number of baited arms entered before reentering a previously visited arm. Successful performance of the task is known to depend on the hippocampus and other brain regions such as the prefrontal cortex that are vital to human mnemonic processes. Performance of the RAM procedure is thought to reflect normal foraging strategies used by rodents in their natural environments, thus it is an ethologically relevant, positive reinforcing (ie, appetitively motivated) paradigm.92
Nicotine, α4β2*, and α7 nAChR agonists have been shown to significantly improve spatial working memory in rodents (eg, in the RAM) when administered alone and in pharmacologic-impairment models.7,53,93 To date, α4β2* nAChR PAMs have not been assessed in the RAM task whereas the results of α7 nAChR PAM (ie, CCMI and PNU 120596) evaluations have been mixed.77,94 When given alone, the type I PAM CCMI was shown to improve trial accuracy77 whereas the type II PAM PNU 120596 was without effects on working and reference memory errors.94 PNU 120596 did, however, counteract scopolamine-induced working and reference memory errors.94 Very little published clinical evidence exists describing the specific effects of nAChR ligands on spatial reference and working memory. The α4β2* nAChR agonist ispronicline was shown to improve working memory (immediate and delayed word recall) in elderly volunteers tested with the Cognitive Drug Research test battery,95 whereas AZD 1446 tested in adults with ADHD failed to show improvements in working memory or the spatial problem-solving test components of the CAARS-INV.96 Varenicline also failed to enhance spatial working memory in patients with schizophrenia.97 Similarly, α7 nAChR agonists (eg, ABT-126, GTS-21, and RG 3487) have produced inconsistent effects on spatial and working memory during clinical assessment.22,87,98 Clinical trials with α4β2* and α7 nAChR PAMs have either not been conducted or the effects on spatial and working memory were not published.
In summary, as in the case of spatial learning and reference memory, most of the preclinical data collected to date across a variety of animal models support the argument that both α4β2* and α7 nAChR agonists have the potential to enhance spatial working memory, whereas additional data are needed before this conclusion can be made for types I and II nAChR PAMs. The nature of the equivocal results in human clinical trials is yet to be determined.
Recognition Memory
Recognition memory refers to the ability to recognize previously encountered stimuli, (eg, objects, smells, people, conspecifics, and events). This form of memory is believed to consist of a recollective (episodic) and a familiarity component and it is one of the domains of cognition that often deteriorates with age in humans. Moreover, it is commonly impaired in AD99–101 and schizophrenia.102,103
Novel Object Recognition
The rodent spontaneous novel object recognition (NOR) task104 has become a particularly popular method for evaluating compounds (including nAChR ligands) for potential as pro-cognitive agents in preclinical drug discovery programs for a variety of reasons. From a practical standpoint, the task is quite efficient because of the short training times required, and it is not aversive to the test subject as it does not require food restriction, water immersion, foot shock, or other nociceptive stimulus. The NOR task (see diagram and general task procedure in Figure 2D) has ethological relevance because it exploits the natural preference of rodents for novelty and translational relevance because it uses brain regions (eg, perirhinal cortex and hippocampus) in the rodent that support recognition memory processes in humans.42,105,106 A variation of this procedure is the spatial object recognition task where subjects explore and prefer novel spatial configurations of objects over previously experienced spatial configurations. This procedure has been used on occasion to evaluate nicotine for effects on spatial recognition memory (see next).
Social Recognition
Social recognition tasks in rodents retain the recognition memory component of NOR, but they also incorporate features of social behavior, thus providing an additional translational component. Although much less complex than human social behavior, social recognition tasks in rodents do use behaviors that are relevant to humans. These include (1) sociability, ie, the preference for interacting with social (conspecifics) as compared with nonsocial objects or spending time alone in an empty chamber; (2) social preference and bias toward novelty, ie, the preference of one individual (usually an unfamiliar individual) over another; and (3) social recognition, ie, the ability to remember an individual and discriminate between individuals.107 The most typical version of the task is conducted similarly to NOR, with the objects replaced with novel and familiar conspecifics in the test phase.
Nicotine, α4β2*, and α7 nAChR agonists as well as α4β2* and α7 nAChR PAMs have been shown to exert robust pro-cognitive effects on object recognition and social recognition across a variety of experimental conditions. These include “stand-alone effects” and in pharmacologic-induced impairment models (eg, scopolamine and acute and subchronic administration of N-methyl-d-aspartate receptor antagonists). Moreover, the pro-cognitive effects elicited by these agents were attenuated by α4β2* or α7 nAChR antagonists, indicating the receptor selectivity of the pro-cognitive effect (Table 2).8,59,67,74,79,84 In addition, improvements in episodic memory (ie, word and picture recognition tests) have been observed with these compounds during early phase clinical trials in normal human volunteers, patients with mild-to-moderate AD, and patients with schizophrenia. Unfortunately, in later (larger-scale) clinical studies, the effects were not replicated.22,32,87,98,108
Although receptor subtype-selective nAChR ligands have not been systematically evaluated in the spatial object recognition task, nicotine has been observed to improve performance of the task when administered acutely.109 Acute administration of the nicotine metabolite cotinine has also been shown to attenuate impairments of a spatial object recognition task in a mouse model of Fragile X syndrome.110 In the case of nicotine, the effects can shift from enhancement of performance to impairments as administration changes from acute to chronic and during withdrawal, however.109 These effects were specific for spatial object recognition (as opposed to NOR), which led the authors to hypothesize that this could be because of differing underlying neural substrates involved performance of the two tasks. Melichercik et al.,111 however, demonstrated that nAChR activation of either the perirhinal cortex or the hippocampus via intracranial infusions of nicotine facilitated performance on both object recognition and object-location memory tasks. These findings were interpreted as further supporting the putative interactive relationship between the hippocampus and perirhinal cortex in object information processing and highlight the potential therapeutic value of nAChR activation in amnesic disorders.
Collectively, the preclinical data described previously support the argument that activation of both α4β2* and α7 nAChRs is a rational strategy for improving various forms of recognition memory including object recognition, social recognition, and object location memory.
Executive Function—Attentional Set Shifting Tasks
Executive function is an integrative component of normal cognition that is essential for the adaptive abilities and complex human behaviors. Optimal executive function requires attention, working memory, the planning, organization and initiation of tasks, cognitive flexibility (task or rule switching), as well as the inhibition of inappropriate responses.112,113 Impairments in executive function are among the most robust cognitive deficits known to be associated with schizophrenia and other neuropsychiatric disorders, especially where there is damage to the prefrontal cortex.114 Over the last several years, new behavioral tasks have been developed specifically for rodents to evaluate executive function. For example, intradimensional and extradimensional set shifting tasks115 are considered rodent analogs of procedures such as the Wisconsin Card Sorting Tasks, which are commonly performed in humans to assess executive function (see Figure 2E and the legend for a diagram and brief, general overview of this task). More recently, computer-automated (operant) tasks for rodents have been developed that retain many of the features of the Birrell and Brown (manual food cup-based tasks), but are somewhat less labor intensive and also allow multiple animals to be tested per day.40 In the manual, food cup-based subjects are typically required to discriminate between the different textures and odors to acquire a food reward. The cognitive flexibility component of the tasks is assessed when the subjects are required switch within dimensions (eg, between different textures) or between dimensions (eg, from texture to odor) and to perform reversals either within or between dimensions. In the computer-automated (operant) tasks, the dimensions (or rule strategies) commonly include spatial location (left vs. right lever) and visual stimuli (light presentation).
Similar to the previously reported pro-cognitive effects across multiple domains, nicotine, α4β2* (ie, 5IA-85380), α7 nAChR agonists (eg, A-582941, RG 3487, and SSR 180711), and α7 nAChR PAMS (eg, CCMI, PAM-2, and PNU 120596) have been shown to improve executive function in the attentional set-shift task whether given alone or after pharmacologic-induced impairment with either acute or subchronic administration of N-methyl-d-aspartate receptor antagonists (Table 2).67,72,78,83,116 A literature search indicated that α4β2* PAMs have not been assessed in the attentional set-shifting task to date. Clinically, improvements in executive function following α4β2* and α7 nAChR agonists during early phase clinical trials have been reported, although as observed for the other cognitive domains discussed earlier, results have not been replicated.22,32,87,98,108
In summary, even though the evaluation of novel nAChR ligands in animal tasks of executive function is in the relatively early stages, the data published to date suggest that α4β2* agonists, α7 nAChR agonists, and α7 nAChR PAMs have significant potential as therapeutic agents.
Some Topics of Debate Regarding the Development of Optimal Therapeutic Strategies Using nAChR Ligands
As noted earlier, although the preclinical literature continues to grow in support of the development of nAChR ligands for a variety of conditions that affect humans, no new nAChR ligand has been approved to date for any condition other than nicotine dependence. It has been argued that one major limitation of the rodent models used in drug discovery research to date (particularly mice) is their limited genetic diversity. This is a major consideration as the two most common causes of early withdrawal from clinical trials, lack of drug efficacy and unanticipated side effects, can be strongly influenced by the genetic heterogeneity of the clinical trial subjects. The recent development of the Collaborative Cross and Diversity Outbred mouse initiatives is providing new tools capable of approaching the genetic diversity found in human populations.
There are also several unresolved topics of debate regarding the development of optimal therapeutic strategies using nAChR ligands to improve cognition that should be discussed. Such topics include receptor subtype selectivity and the desired net pharmacologic effect of the ligand (eg, receptor stimulation, desensitization, and antagonism). In each of these scenarios, in vivo conditions can be very complex and difficult to interpret. As noted earlier regarding nAChR selectivity, the field has primarily concentrated on developing ligands selective for heteromeric α4β2 and homomeric α7 nAChRs to date because these receptor subtypes are thought to be the most prevalent in memory-related brain regions (eg, cortex and hippocampus) and because other subtypes (eg, α3β4 nAChRs) have been associated with undesired size effects (eg, abuse potential and cardiovascular side effects). In the text at the end of this section, the potential value of α4β2*-selective nAChR agonists to promote phasic neurotransmission is also discussed. However, the administration of a single pharmacologic agent with selectivity at α4β2 or α7 nAChRs may not be adequate or the most rational therapeutic due to the complex anatomical distributions of nAChRs as well as the nature and/or extent of the specific disease pathology. As just one example of the anatomical complexity, differing combinations of α7, α4β2, and α3β4 nAChRs found at specific locations on interneurons in the hippocampus affect both inhibitory and excitatory tones to collectively drive theta wave synchronization and long-term potentiation, the most widely proposed mechanism of memory storage, see Rogers et al.117. Differing physical properties of these receptor subtypes (eg, rates of activation and desensitization, and variable permeability to calcium vs. sodium) also make it unclear if pure selectivity at one of these specific nAChR subtypes is optimal. Moreover, as has been reviewed previously,87 given the complexity of both the pathophysiology and symptom profile of conditions such as AD and schizophrenia, it would appear rational to attempt to modulate several therapeutic targets simultaneously.
The classical view that receptor stimulation is the key action responsible for nAChR-induced cognitive enhancement can also be challenged by the observation that nAChRs are easily desensitized by nicotine and other nAChR agonists and that in some cases nAChR antagonists can exert pro-cognitive effects. As an example, although an extensive literature exists on the amnesic effects of the nonselective nAChR antagonist mecamylamine, pro-cognitive effects of this compound have been reported in working memory tasks in both rodents and monkeys as well as a recognition memory task in humans with ADHD. For details of these studies, additional examples of nAChR antagonist-related pro-cognitive effects, and a discussion of the provocative subject of “nAChR desensitization of as a strategy for pro-cognitive drug development,” see Refs. 22,118.
Another topic of debate related to the best cholinergic-based approaches to improving cognitive function is the type of neurotransmission or neuromodulation that should be targeted (eg, volume vs. phasic transmission). The diffuse projections of basal forebrain cholinergic neurons to the cortex and the locations of cholinergic receptors at multiple sites on neurons (presynaptic, postsynaptic, extrasynaptic) as well as on nonneuronal sites in the central nervous system have fueled the argument that they primary facilitate paracrine (also referred to as diffuse, nonjunctional, or volume) transmission. In this scenario, ACh slowly reaches the extrasynaptic space as opposed to remaining confined to the synaptic cleft (wired neurotransmission) and mediates different arousal states to influence cognition, especially attentional performance. This model would appear to fit well with the most commonly prescribed treatments for AD, the acetylcholinesterase inhibitors that, by blocking the hydrolysis of ACh, significantly elevate basal extracellular levels of ACh to improve cognition. However, the modest clinical efficacy of acetylcholinesterase inhibitors, and the observation that they often fail to significantly improve the performance of attention-based tasks in animals, argues that this may not be the most optimal approach to improving attention. Recent studies demonstrated that phasic release of ACh, at the scale of seconds, mediates precisely defined cognitive operations. Moreover, an increasing body of evidence suggests that nAChR agonists, especially α4β2*-selective nAChR agonists, generate phasic cholinergic signals to robustly enhance cognitive function (reviewed in McGonigle119). This type of observation would support the future development of selective nAChR agonists as opposed to additional efforts to enhance “volume transmission” in cholinergic pathways. Alternatively, adjunctive strategies where a selective nAChR agonist or PAM (to enhance phasic cholinergic signals) is combined with an acetylcholinesterase inhibitor (which promotes volume transmission) may be promising. Notably, in previous studies in our laboratory, we have observed robust cognitive enhancements in young and aged rats as well as aged monkeys when we combined the α7-nAChR-selective PAM, PNU 120596 with the acetylcholinesterase inhibitor donepezil.90
Conclusions and Future Directions
The high rate of clinical trial failures for novel compounds developed to treat neuropsychiatric disorders has fueled the argument that there is a significant translational gap between the preclinical animal studies and the human clinical trials.120,121 The clinical failures include novel nAChR ligands at both α4β2 (eg, ispronicline) and α7 nAChRs (encenicline) for AD and/or schizophrenia where the compounds did not show statistically significant efficacy as pro-cognitive agents or they were discontinued prematurely due to unanticipated side effects.121,122 Both study failures occurred despite a large body of encouraging preclinical data and early clinical results. There may be a variety of reasons for these failures, at both the preclinical and clinical trial design levels.22 However, the studies do provide the impetus for continuing efforts to develop new nAChR strategies beyond simple agonist and partial agonist approaches (to include PAMs) as well as to refine current behavioral strategies and create new animal models to address translational gaps in the drug discovery process. The preclinical literature continues to grow in support of the development of new nAChR ligands for a variety of conditions that affect humans. Moreover, there is some literature to support the repurposing of nAChR ligands that are approved and prescribed for other therapeutic purposes (eg, tropisetron) for disorders of cognition (eg, AD, Parkinson disease, and schizophrenia)74,87 and nicotine withdrawal-induced cognitive impairments.97
Funding
The corresponding author’s laboratories and/or salary are supported in part by the following funding sources: National Institutes of Health (grants MH097695, MH083317, and NS099455), Institut de Recherches Servier, and Prime Behavior Testing Laboratories, Evans, Georgia.
Declaration of Interests
None declared.
Supplementary Material
Acknowledgments
The authors wish to thank Ms. Ashley Davis for her administrative assistance in preparing this article.
References
- 1. Sherwood N. Effects of nicotine on human psychomotor performance. Hum Psychopharmacol Clin Exp. 1993;8(3):155–184. doi: 10.1002/hup.470080303 [DOI] [Google Scholar]
- 2. Heishman SJ, Taylor RC, Henningfield JE. Nicotine and smoking: a review of effects on human performance. Exp Clin Psychopharmacol. 1994;2(4):345–395. [Google Scholar]
- 3. Heishman SJ. What aspects of human performance are truly enhanced by nicotine?Addiction. 1998;93(3):317–320. [DOI] [PubMed] [Google Scholar]
- 4. Levin ED, Cauley M, Rezvani AH. Improvement of attentional function with antagonism of nicotinic receptors in female rats. Eur J Pharmacol. 2013;702(1–3):269–274. doi: 10.1016/j.ejphar.2013.01.056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Curzon P, Kim DJ, Decker MW. Effect of nicotine, lobeline, and mecamylamine on sensory gating in the rat. Pharmacol Biochem Behav. 1994;49(4):877–882. [DOI] [PubMed] [Google Scholar]
- 6. Mirza NR, Stolerman IP. Nicotine enhances sustained attention in the rat under specific task conditions. Psychopharmacology (Berl). 1998;138(3–4):266–274. [DOI] [PubMed] [Google Scholar]
- 7. Levin ED, Simon BB. Nicotinic acetylcholine involvement in cognitive function in animals. Psychopharmacology (Berl). 1998;138(3–4):217–230. [DOI] [PubMed] [Google Scholar]
- 8. Puma C, Deschaux O, Molimard R, Bizot JC. Nicotine improves memory in an object recognition task in rats. Eur Neuropsychopharmacol. 1999;9(4):323–327. [DOI] [PubMed] [Google Scholar]
- 9. Elrod K, Buccafusco JJ, Jackson WJ. Nicotine enhances delayed matching-to-sample performance by primates. Life Sci. 1988;43(3):277–287. [DOI] [PubMed] [Google Scholar]
- 10. Buccafusco JJ, Jackson WJ. Beneficial effects of nicotine administered prior to a delayed matching-to-sample task in young and aged monkeys. Neurobiol Aging. 1991;12(3):233–238. [DOI] [PubMed] [Google Scholar]
- 11. Prendergast MA, Jackson WJ, Terry AV Jr, Decker MW, Arneric SP, Buccafusco JJ. Central nicotinic receptor agonists ABT-418, ABT-089, and (-)-nicotine reduce distractibility in adult monkeys. Psychopharmacology (Berl). 1998;136(1):50–58. [DOI] [PubMed] [Google Scholar]
- 12. Kumari V, Cotter PA, Checkley SA, Gray JA. Effect of acute subcutaneous nicotine on prepulse inhibition of the acoustic startle reflex in healthy male non-smokers. Psychopharmacology (Berl). 1997;132(4):389–395. [DOI] [PubMed] [Google Scholar]
- 13. White HK, Levin ED. Four-week nicotine skin patch treatment effects on cognitive performance in Alzheimer’s disease. Psychopharmacology (Berl). 1999;143(2):158–165. [DOI] [PubMed] [Google Scholar]
- 14. Newhouse PA, Kelton M. Nicotinic systems in central nervous systems disease: degenerative disorders and beyond. Pharm Acta Helv. 2000;74(2–3):91–101. [DOI] [PubMed] [Google Scholar]
- 15. Min SK, Moon IW, Ko RW, Shin HS. Effects of transdermal nicotine on attention and memory in healthy elderly non-smokers. Psychopharmacology (Berl). 2001;159(1):83–88. [DOI] [PubMed] [Google Scholar]
- 16. Sahakian B, Jones G, Levy R, Gray J, Warburton D. The effects of nicotine on attention, information processing, and short-term memory in patients with dementia of the Alzheimer type. Br J Psychiatry. 1989;154(6):797–800. [DOI] [PubMed] [Google Scholar]
- 17. Picciotto MR, Zoli M. Neuroprotection via nAChRs: the role of nAChRs in neurodegenerative disorders such as Alzheimer’s and Parkinson’s disease. Front Biosci. 2008;13(1):492–504. [DOI] [PubMed] [Google Scholar]
- 18. Quik M, O’Leary K, Tanner CM. Nicotine and Parkinson’s disease: implications for therapy. Mov Disord. 2008;23(12):1641–1652. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Benowitz NL. Pharmacology of nicotine: addiction, smoking-induced disease, and therapeutics. Annu Rev Pharmacol Toxicol. 2009;49(1):57–71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Newhouse P, Kellar K, Aisen P, et al. Nicotine treatment of mild cognitive impairment: a 6-month double-blind pilot clinical trial. Neurology. 2012;78(2):91–101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Shahab L, Brose LS, West R. Novel delivery systems for nicotine replacement therapy as an aid to smoking cessation and for harm reduction: rationale, and evidence for advantages over existing systems. CNS Drugs. 2013;27(12):1007–1019. [DOI] [PubMed] [Google Scholar]
- 22. Bertrand D, Terry AV Jr. The wonderland of neuronal nicotinic acetylcholine receptors. Biochem Pharmacol. 2018;151:214–225. [DOI] [PubMed] [Google Scholar]
- 23. Timmermann DB, Sandager-Nielsen K, Dyhring T, et al. Augmentation of cognitive function by NS9283, a stoichiometry-dependent positive allosteric modulator of α2- and α4-containing nicotinic acetylcholine receptors. Br J Pharmacol. 2012;167(1):164–182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Zwart R, Vijverberg HP. Four pharmacologically distinct subtypes of alpha4beta2 nicotinic acetylcholine receptor expressed in Xenopus laevis oocytes. Mol Pharmacol. 1998;54(6):1124–1131. [PubMed] [Google Scholar]
- 25. Changeux JP. Nicotine addiction and nicotinic receptors: lessons from genetically modified mice. Nat Rev Neurosci. 2010;11(6):389–401. [DOI] [PubMed] [Google Scholar]
- 26. D’Souza MS, Markou A. Neuronal mechanisms underlying development of nicotine dependence: implications for novel smoking-cessation treatments. Addict Sci Clin Pract. 2011;6(1):4–16. [PMC free article] [PubMed] [Google Scholar]
- 27. Chatterjee S, Bartlett SE. Neuronal nicotinic acetylcholine receptors as pharmacotherapeutic targets for the treatment of alcohol use disorders. CNS Neurol Disord Drug Targets. 2010;9(1):60–76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Philip NS, Carpenter LL, Tyrka AR, Price LH. Nicotinic acetylcholine receptors and depression: a review of the preclinical and clinical literature. Psychopharmacology (Berl). 2010;212(1):1–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Mineur YS, Picciotto MR. Nicotine receptors and depression: revisiting and revising the cholinergic hypothesis. Trends Pharmacol Sci. 2010;31(12):580–586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Rollema H, Russ C, Lee TC, Hurst RS, Bertrand D. Functional interactions of varenicline and nicotine with nAChR subtypes implicated in cardiovascular control. Nicotine Tob Res. 2014;16(6):733–742. [DOI] [PubMed] [Google Scholar]
- 31. Freedman R. α7-Nicotinic acetylcholine receptor agonists for cognitive enhancement in schizophrenia. Annu Rev Med. 2014;65(1):245–261. [DOI] [PubMed] [Google Scholar]
- 32. Wallace TL, Bertrand D. Neuronal α7 nicotinic receptors as a target for the treatment of schizophrenia. Int Rev Neurobiol. 2015;124:79–111. [DOI] [PubMed] [Google Scholar]
- 33. Wilens TE, Decker MW. Neuronal nicotinic receptor agonists for the treatment of attention-deficit/hyperactivity disorder: focus on cognition. Biochem Pharmacol. 2007;74(8):1212–1223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Potter AS, Schaubhut G, Shipman M. Targeting the nicotinic cholinergic system to treat attention-deficit/hyperactivity disorder: rationale and progress to date. CNS Drugs. 2014;28(12):1103–1113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Posadas I, López-Hernández B, Ceña V. Nicotinic receptors in neurodegeneration. Curr Neuropharmacol. 2013;11(3):298–314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Quik M, Mallela A, Ly J, Zhang D. Nicotine reduces established levodopa-induced dyskinesias in a monkey model of Parkinson’s disease. Mov Disord. 2013;28(10):1398–1406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Bencherif M, Lippiello PM, Lucas R, Marrero MB. Alpha7 nicotinic receptors as novel therapeutic targets for inflammation-based diseases. Cell Mol Life Sci. 2011;68(6):931–949. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Umana IC, Daniele CA, McGehee DS. Neuronal nicotinic receptors as analgesic targets: it’s a winding road. Biochem Pharmacol. 2013;86(8):1208–1214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Winterer G, Gallinat J, Brinkmeyer J, et al. Allosteric alpha-7 nicotinic receptor modulation and P50 sensory gating in schizophrenia: a proof-of-mechanism study. Neuropharmacology. 2013;64:197–204. [DOI] [PubMed] [Google Scholar]
- 40. Brady AM, Floresco SB. Operant procedures for assessing behavioral flexibility in rats. J Vis Exp. 2015;15(96):e52387. doi: 10.3791/52387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Albuquerque EX, Pereira EF, Alkondon M, Rogers SW. Mammalian nicotinic acetylcholine receptors: from structure to function. Physiol Rev. 2009;89(1):73–120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Young JW, Light GA, Marston HM, Sharp R, Geyer MA. The 5-choice continuous performance test: evidence for a translational test of vigilance for mice. PLoS One. 2009;4(1):e4227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Raybuck JD, Gould TJ. The role of nicotinic acetylcholine receptors in the medial prefrontal cortex and hippocampus in trace fear conditioning. Neurobiol Learn Mem. 2010;94(3):353–363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Tian S, Huang F, Li P, et al. Nicotine enhances contextual fear memory reconsolidation in rats. Neurosci Lett. 2011;487(3):368–371. [DOI] [PubMed] [Google Scholar]
- 45. Kaplan GB, Moore KA. The use of cognitive enhancers in animal models of fear extinction. Pharmacol Biochem Behav. 2011;99(2):217–228. [DOI] [PubMed] [Google Scholar]
- 46. Kutlu MG, Gould TJ. Nicotine modulation of fear memories and anxiety: implications for learning and anxiety disorders. Biochem Pharmacol. 2015;97(4):498–511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Decker MW, Bannon AW, Curzon P, et al. ABT-089 [2-methyl-3-(2-(S)-pyrrolidinylmethoxy)pyridine dihydrochloride]: II. A novel cholinergic channel modulator with effects on cognitive performance in rats and monkeys. J Pharmacol Exp Ther. 1997;283(1):247–258. [PubMed] [Google Scholar]
- 48. Rueter LE, Anderson DJ, Briggs CA, et al. ABT-089: pharmacological properties of a neuronal nicotinic acetylcholine receptor agonist for the potential treatment of cognitive disorders. CNS Drug Rev. 2004;10(2):167–182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Sullivan JP, Donnelly-Roberts D, Briggs CA, et al. ABT-089 [2-methyl-3-(2-(S)-pyrrolidinylmethoxy)pyridine]: I. A potent and selective cholinergic channel modulator with neuroprotective properties. J Pharmacol Exp Ther. 1997;283(1):235–246. [PubMed] [Google Scholar]
- 50. Buccafusco JJ, Terry AV Jr, Decker MW, Gopalakrishnan M. Profile of nicotinic acetylcholine receptor agonists ABT-594 and A-582941, with differential subtype selectivity, on delayed matching accuracy by young monkeys. Biochem Pharmacol. 2007;74(8):1202–1211. [DOI] [PubMed] [Google Scholar]
- 51. Donnelly-Roberts DL, Puttfarcken PS, Kuntzweiler TA, et al. ABT-594 [®-5-(2-azetidinylmethoxy)-2-chloropyridine]: a novel, orally effective analgesic acting via neuronal nicotinic acetylcholine receptors: I. In vitro characterization. J Pharmacol Exp Ther. 1998;285(2):777–786. [PubMed] [Google Scholar]
- 52. Mohler EG, Franklin SR, Rueter LE, Fox GB, Decker MW, Browman KE. ABT-594 improves performance in the 5-choice serial reaction time task under conditions of increased difficulty, sub-chronic dosing, and in poorly-performing subjects. Pharmacol Biochem Behav. 2010;95(2):146–157. [DOI] [PubMed] [Google Scholar]
- 53. Gatto GJ, Bohme GA, Caldwell WS, et al. TC-1734: an orally active neuronal nicotinic acetylcholine receptor modulator with antidepressant, neuroprotective and long-lasting cognitive effects. CNS Drug Rev. 2004;10(2):147–166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Lippiello P, Letchworth SR, Gatto GJ, Traina VM, Bencherif M. Ispronicline: a novel alpha4beta2 nicotinic acetylcholine receptor-selective agonist with cognition-enhancing and neuroprotective properties. J Mol Neurosci. 2006; 30(1–2):19–20. PMID: 17192610. [DOI] [PubMed] [Google Scholar]
- 55. Obinu MC, Reibaud M, Miquet JM, Pasquet M, Rooney T. Brain-selective stimulation of nicotinic receptors by TC-1734 enhances ACh transmission from frontoparietal cortex and memory in rodents. Prog Neuropsychopharmacol Biol Psychiatry. 2002;26(5):913–918. [DOI] [PubMed] [Google Scholar]
- 56. Cucchiaro G, Xiao Y, Gonzalez-Sulser A, Kellar KJ. Analgesic effects of Sazetidine-A, a new nicotinic cholinergic drug. Anesthesiology. 2008;109(3):512–519. [DOI] [PubMed] [Google Scholar]
- 57. Rezvani AH, Cauley M, Xiao Y, Kellar KJ, Levin ED. Effects of chronic sazetidine-A, a selective α4β2 neuronal nicotinic acetylcholine receptors desensitizing agent on pharmacologically-induced impaired attention in rats. Psychopharmacology (Berl). 2013;226(1):35–43. [DOI] [PubMed] [Google Scholar]
- 58. Turner JR, Castellano LM, Blendy JA. Nicotinic partial agonists varenicline and sazetidine-A have differential effects on affective behavior. J Pharmacol Exp Ther. 2010;334(2):665–672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59. Rollema H, Hajós M, Seymour PA, et al. Preclinical pharmacology of the alpha4beta2 nAChR partial agonist varenicline related to effects on reward, mood and cognition. Biochem Pharmacol. 2009;78(7):813–824. [DOI] [PubMed] [Google Scholar]
- 60. Gould RW, Garg PK, Garg S, Nader MA. Effects of nicotinic acetylcholine receptor agonists on cognition in rhesus monkeys with a chronic cocaine self-administration history. Neuropharmacology. 2013;64:479–488. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61. Lange-Asschenfeldt C, Schäble S, Suvorava T, et al. Effects of varenicline on alpha4-containing nicotinic acetylcholine receptor expression and cognitive performance in mice. Neuropharmacology. 2016;107:100–110. [DOI] [PubMed] [Google Scholar]
- 62. Boess FG, De Vry J, Erb C, et al. The novel alpha7 nicotinic acetylcholine receptor agonist N-[(3R)-1-azabicyclo[2.2.2]oct-3-yl]-7-[2-(methoxy)phenyl]-1-benzofuran-2-carboxamide improves working and recognition memory in rodents. J Pharmacol Exp Ther. 2007;321(2):716–725. [DOI] [PubMed] [Google Scholar]
- 63. Bitner RS, Bunnelle WH, Decker MW, et al. In vivo pharmacological characterization of a novel selective alpha7 neuronal nicotinic acetylcholine receptor agonist ABT-107: preclinical considerations in Alzheimer’s disease. J Pharmacol Exp Ther. 2010;334(3):875–886. [DOI] [PubMed] [Google Scholar]
- 64. Bordia T, McGregor M, Papke RL, Decker MW, McIntosh JM, Quik M. The α7 nicotinic receptor agonist ABT-107 protects against nigrostriatal damage in rats with unilateral 6-hydroxydopamine lesions. Exp Neurol. 2015;263:277–284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65. Radek RJ, Robb HM, Stevens KE, Gopalakrishnan M, Bitner RS. Effects of the novel α7 nicotinic acetylcholine receptor agonist ABT-107 on sensory gating in DBA/2 mice: pharmacodynamic characterization. J Pharmacol Exp Ther. 2012;343(3):736–745. [DOI] [PubMed] [Google Scholar]
- 66. Feuerbach D, Pezous N, Weiss M, et al. AQW051, a novel, potent and selective α7 nicotinic ACh receptor partial agonist: pharmacological characterization and phase I evaluation. Br J Pharmacol. 2015;172(5):1292–1304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67. Bristow LJ, Easton AE, Li YW, et al. The novel, nicotinic alpha7 receptor partial agonist, BMS-933043, improves cognition and sensory processing in preclinical models of schizophrenia. PLoS One. 2016;11(7):e0159996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68. Pieschl RL, Miller R, Jones KM, et al. Effects of BMS-902483, an α7 nicotinic acetylcholine receptor partial agonist, on cognition and sensory gating in relation to receptor occupancy in rodents. Eur J Pharmacol. 2017;807:1–11. [DOI] [PubMed] [Google Scholar]
- 69. Prickaerts J, van Goethem NP, Chesworth R, et al. EVP-6124, a novel and selective α7 nicotinic acetylcholine receptor partial agonist, improves memory performance by potentiating the acetylcholine response of α7 nicotinic acetylcholine receptors. Neuropharmacology. 2012;62(2):1099–1110. [DOI] [PubMed] [Google Scholar]
- 70. Boess FG, de Vry J, Erb C, et al. Pharmacological and behavioral profile of N-[(3R)-1-azabicyclo[2.2.2]oct-3-yl]-6-chinolincarboxamide (EVP-5141), a novel α7 nicotinic acetylcholine receptor agonist/serotonin 5-HT3 receptor antagonist. Psychopharmacology (Berl). 2013;227(1):1–17. [DOI] [PubMed] [Google Scholar]
- 71. Roncarati R, Scali C, Comery TA, et al. Procognitive and neuroprotective activity of a novel alpha7 nicotinic acetylcholine receptor agonist for treatment of neurodegenerative and cognitive disorders. J Pharmacol Exp Ther. 2009;329(2):459–468. [DOI] [PubMed] [Google Scholar]
- 72. Wallace TL, Callahan PM, Tehim A, et al. RG3487, a novel nicotinic α7 receptor partial agonist, improves cognition and sensorimotor gating in rodents. J Pharmacol Exp Ther. 2011;336(1):242–253. [DOI] [PubMed] [Google Scholar]
- 73. Hauser TA, Kucinski A, Jordan KG, et al. TC-5619: an alpha7 neuronal nicotinic receptor-selective agonist that demonstrates efficacy in animal models of the positive and negative symptoms and cognitive dysfunction of schizophrenia. Biochem Pharmacol. 2009;78(7):803–812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74. Callahan PM, Bertrand D, Bertrand S, Plagenhoef MR, Terry AV Jr. Tropisetron sensitizes α7 containing nicotinic receptors to low levels of acetylcholine in vitro and improves memory-related task performance in young and aged animals. Neuropharmacology. 2017;117:422–433. [DOI] [PubMed] [Google Scholar]
- 75. Kohnomi S, Suemaru K, Goda M, et al. Ameliorating effects of tropisetron on dopaminergic disruption of prepulse inhibition via the alpha(7) nicotinic acetylcholine receptor in Wistar rats. Brain Res. 2010;1353(24):152–158. [DOI] [PubMed] [Google Scholar]
- 76. Pitsikas N, Borsini F. Different effects of tropisetron and ondansetron in learning and memory paradigms. Pharmacol Biochem Behav. 1997;56(4):571–576. [DOI] [PubMed] [Google Scholar]
- 77. Ng HJ, Whittemore ER, Tran MB, et al. Nootropic alpha7 nicotinic receptor allosteric modulator derived from GABAA receptor modulators. Proc Natl Acad Sci U S A. 2007;104(19):8059–8064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78. Nikiforuk A, Potasiewicz A, Kos T, Popik P. The combination of memantine and galantamine improves cognition in rats: the synergistic role of the α7 nicotinic acetylcholine and NMDA receptors. Behav Brain Res. 2016;313(15):214–218. [DOI] [PubMed] [Google Scholar]
- 79. Hogenkamp DJ, Ford-Hutchinson TA, Li WY, et al. Design, synthesis, and activity of a series of arylpyrid-3-ylmethanones as type I positive allosteric modulators of α7 nicotinic acetylcholine receptors. J Med Chem. 2013;56(21):8352–8365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80. Eskildsen J, Redrobe JP, Sams AG, et al. Discovery and optimization of Lu AF58801, a novel, selective and brain penetrant positive allosteric modulator of alpha-7 nicotinic acetylcholine receptors: attenuation of subchronic phencyclidine (PCP)-induced cognitive deficits in rats following oral administration. Bioorg Med Chem Lett. 2014;24(1):288–293. [DOI] [PubMed] [Google Scholar]
- 81. Timmermann DB, Grønlien JH, Kohlhaas KL, et al. An allosteric modulator of the alpha7 nicotinic acetylcholine receptor possessing cognition-enhancing properties in vivo. J Pharmacol Exp Ther. 2007;323(1):294–307. [DOI] [PubMed] [Google Scholar]
- 82. Marcus MM, Björkholm C, Malmerfelt A, et al. Alpha7 nicotinic acetylcholine receptor agonists and PAMs as adjunctive treatment in schizophrenia. An experimental study. Eur Neuropsychopharmacol. 2016;26(9):1401–1411. [DOI] [PubMed] [Google Scholar]
- 83. Potasiewicz A, Hołuj M, Kos T, Popik P, Arias HR, Nikiforuk A. 3-Furan-2-yl-N-p-tolyl-acrylamide, a positive allosteric modulator of the α7 nicotinic receptor, reverses schizophrenia-like cognitive and social deficits in rats. Neuropharmacology. 2017;113(Pt A):188–197. [DOI] [PubMed] [Google Scholar]
- 84. Nikiforuk A, Kos T, Potasiewicz A, Popik P. Positive allosteric modulation of alpha 7 nicotinic acetylcholine receptors enhances recognition memory and cognitive flexibility in rats. Eur Neuropsychopharmacol. 2015;25(8):1300–1313. [DOI] [PubMed] [Google Scholar]
- 85. Stevens KE, Zheng L, Floyd KL, Stitzel JA. Maximizing the effect of an α7 nicotinic receptor PAM in a mouse model of schizophrenia-like sensory inhibition deficits. Brain Res. 2015;1611(22):8–17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86. Callahan PM, Terry AV Jr. Attention. In: Kantak KM, Wettstein JG, eds. Cognitive Enhancement, Handbook of Experimental Pharmacology 228, Switzerland: Springer International Publishing; 2015: 161–189, Chap 5. [Google Scholar]
- 87. Terry AV Jr, Callahan PM, Hernandez CM. Nicotinic ligands as multifunctional agents for the treatment of neuropsychiatric disorders. Biochem Pharmacol. 2015;97(4):388–398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88. Hahn B, Riegger KE, Elmer GI. Strain dependency of the effects of nicotine and mecamylamine in a rat model of attention. Psychopharmacology (Berl). 2016;233(8):1427–1434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89. Terry AV., Jr Spatial navigation (water maze) tasks. In: Buccafusco JJ, ed. Methods of Behavior Analysis in Neuroscience. 2nd ed. Boca Raton, FL: CRC Press/Taylor & Francis; 2009, Chap 13. [PubMed] [Google Scholar]
- 90. Callahan PM, Hutchings EJ, Kille NJ, Chapman JM, Terry AV Jr. Positive allosteric modulator of α7 nicotinic-acetylcholine receptors, PNU-120596 augments the effects of donepezil on learning and memory in aged rodents and non-human primates. Neuropharmacology. 2013;67:201–212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91. Dudchenko PA, Talpos J, Young J, Baxter MG. Animal models of working memory: a review of tasks that might be used in screening drug treatments for the memory impairments found in schizophrenia. Neurosci Biobehav Rev. 2013;37(9, Pt B):2111–2124. [DOI] [PubMed] [Google Scholar]
- 92. Olton DS. The radial arm maze as a tool in behavioral pharmacology. Physiol Behav. 1987;40(6):793–797. [DOI] [PubMed] [Google Scholar]
- 93. Levin ED, Bettegowda C, Blosser J, et al. AR-R17779, an alpha7 nicotinic agonist, improves learning and memory in rats. Behav Pharmacol. 1999; 10(6–7):675–680. [DOI] [PubMed] [Google Scholar]
- 94. Pandya AA, Yakel JL. Effects of neuronal nicotinic acetylcholine receptor allosteric modulators in animal behavior studies. Biochem Pharmacol. 2013;86(8):1054–1062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95. Jucaite A, Öhd J, Potter AS, et al. A randomized, double-blind, placebo-controlled crossover study of α4β 2* nicotinic acetylcholine receptor agonist AZD1446 (TC-6683) in adults with attention-deficit/hyperactivity disorder. Psychopharmacology (Berl). 2014;231(6):1251–1265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96. Hong LE, Thaker GK, McMahon RP, et al. Effects of moderate-dose treatment with varenicline on neurobiological and cognitive biomarkers in smokers and nonsmokers with schizophrenia or schizoaffective disorder. Arch Gen Psychiatry. 2011;68(12):1195–1206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97. Valentine G, Sofuoglu M. Cognitive effects of nicotine: recent progress. Curr Neuropharmacol. 2018;16(4):403–414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98. Gault LM, Lenz RA, Ritchie CW, et al. ABT-126 monotherapy in mild-to-moderate Alzheimer’s dementia: randomized double-blind, placebo and active controlled adaptive trial and open-label extension. Alzheimers Res Ther. 2016;8(1):44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99. Flicker C, Ferris SH, Crook T, Bartus RT. A visual recognition memory test for the assessment of cognitive function in aging and dementia. Exp Aging Res. 1987;13(3):127–132. [DOI] [PubMed] [Google Scholar]
- 100. Purdy KS, McMullen PA, Freedman M. Changes to the object recognition system in patients with dementia of the Alzheimer’s type. Brain Cogn. 2002;49(2):213–216. [PubMed] [Google Scholar]
- 101. Schiavetto A, Köhler S, Grady CL, Winocur G, Moscovitch M. Neural correlates of memory for object identity and object location: effects of aging. Neuropsychologia. 2002;40(8):1428–1442. [DOI] [PubMed] [Google Scholar]
- 102. Pelletier M, Achim AM, Montoya A, Lal S, Lepage M. Cognitive and clinical moderators of recognition memory in schizophrenia: a meta-analysis. Schizophr Res. 2005;74(2–3):233–252. [DOI] [PubMed] [Google Scholar]
- 103. Ranganath C, Minzenberg MJ, Ragland JD. The cognitive neuroscience of memory function and dysfunction in schizophrenia. Biol Psychiatry. 2008;64(1):18–25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104. Ennaceur A, Delacour J. A new one-trial test for neurobiological studies of memory in rats. 1: behavioral data. Behav Brain Res. 1988;31(1):47–59. [DOI] [PubMed] [Google Scholar]
- 105. Lyon L, Saksida LM, Bussey TJ. Spontaneous object recognition and its relevance to schizophrenia: a review of findings from pharmacological, genetic, lesion and developmental rodent models. Psychopharmacology (Berl). 2012;220(4):647–672. [DOI] [PubMed] [Google Scholar]
- 106. Grayson B, Leger M, Piercy C, Adamson L, Harte M, Neill JC. Assessment of disease-related cognitive impairments using the novel object recognition (NOR) task in rodents. Behav Brain Res. 2015;285(15):176–193. [DOI] [PubMed] [Google Scholar]
- 107. Millan MJ, Bales KL. Towards improved animal models for evaluating social cognition and its disruption in schizophrenia: the CNTRICS initiative. Neurosci Biobehav Rev. 2013;37(9, Pt B):2166–2180. [DOI] [PubMed] [Google Scholar]
- 108. Dunbar GC, Kuchibhatla R. Cognitive enhancement in man with ispronicline, a nicotinic partial agonist. J Mol Neurosci. 2006;30(1–2):169–172. [DOI] [PubMed] [Google Scholar]
- 109. Kenney JW, Adoff MD, Wilkinson DS, Gould TJ. The effects of acute, chronic, and withdrawal from chronic nicotine on novel and spatial object recognition in male C57BL/6J mice. Psychopharmacology (Berl). 2011;217(3):353–365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110. Pardo M, Beurel E, Jope RS. Cotinine administration improves impaired cognition in the mouse model of Fragile X syndrome. Eur J Neurosci. 2017;45(4):490–498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111. Melichercik AM, Elliott KS, Bianchi C, Ernst SM, Winters BD. Nicotinic receptor activation in perirhinal cortex and hippocampus enhances object memory in rats. Neuropharmacology. 2012;62(5–6):2096–2105. [DOI] [PubMed] [Google Scholar]
- 112. Jurado MB, Rosselli M. The elusive nature of executive functions: a review of our current understanding. Neuropsychol Rev. 2007;17(3):213–233. [DOI] [PubMed] [Google Scholar]
- 113. Ardila A. On the evolutionary origins of executive functions. Brain Cogn. 2008;68(1):92–99. [DOI] [PubMed] [Google Scholar]
- 114. Liu KC, Chan RC, Chan KK, et al. Executive function in first-episode schizophrenia: a three-year longitudinal study of an ecologically valid test. Schizophr Res. 2011;126(1–3):87–92. doi: 10.1016/j.schres.2010.11.023. [DOI] [PubMed] [Google Scholar]
- 115. Birrell JM, Brown VJ. Medial frontal cortex mediates perceptual attentional set shifting in the rat. J Neurosci. 2000;20(11):4320–4324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116. Wood C, Kohli S, Malcolm E, Allison C, Shoaib M. Subtype-selective nicotinic acetylcholine receptor agonists can improve cognitive flexibility in an attentional set shifting task. Neuropharmacology. 2016;105:106–113. [DOI] [PubMed] [Google Scholar]
- 117.Rogers S, Gould T, Baker T. Mouse models and the genetics of nicotinic dependence. Chapter 4 in National Cancer Institutue Tobacco Control Monograph No. 20. In Phenotypes and Endophenotypes: Foundations for Genetic Studies of Nicotine Use and Dependence (pp: 133–186). Bethesda, MD: U.S. Department of Health and Human Services. 2009 NIH Publication No. 09-6366. [Google Scholar]
- 118. Buccafusco JJ, Beach JW, Terry AV Jr. Desensitization of nicotinic acetylcholine receptors as a strategy for drug development. J Pharmacol Exp Ther. 2009;328(2):364–370. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119. Sarter M, Parikh V, Howe WM. Phasic acetylcholine release and the volume transmission hypothesis: time to move on. Nat Rev Neurosci. 2009;10(5):383–390. doi: 10.1038/nrn2635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120. McGonigle P. Animal models of CNS disorders. Biochem Pharmacol. 2014;87(1):140–149. doi: 10.1016/j.bcp.2013.06.016. [DOI] [PubMed] [Google Scholar]
- 121. Lewis AS, van Schalkwyk GI, Bloch MH. Alpha-7 nicotinic agonists for cognitive deficits in neuropsychiatric disorders: a translational meta-analysis of rodent and human studies. Prog Neuropsychopharmacol Biol Psychiatry. 2017;75:45–53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122. Anonymous, Encenicline|Alzforum 2016. Retrieved October 18, 2017, from http://www.alzforum.org/therapeutics/encenicline
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.