Short abstract
Objective
Prevention of postoperative cognitive dysfunction (POCD) in patients of advanced age remains unclear. Studies have shown that the cholinergic anti-inflammatory pathway contributes to a decreased risk of POCD and that nicotine stimulates the cholinergic anti-inflammatory pathway. We investigated whether patients of advanced age with a preoperative smoking history have a decreased risk of POCD.
Methods
In total, 382 patients (190 smokers, 192 nonsmokers) aged ≥60 years who underwent major noncardiac surgery were enrolled. Cognitive function was assessed, and multivariate logistic regression analyses were performed to identify risk factors.
Results
On postoperative days 5 and 7, 111 (29.05%) and 90 (23.56%) patients exhibited POCD, respectively. A preoperative smoking history was significantly correlated with a decreased risk of POCD. A high serum tumor necrosis factor-α (TNF-α) level on the operative day was significantly associated with an increased risk of POCD. Early POCD was significantly associated with the sufentanil dosage, age, and education level. The hospital stay in patients with and without POCD was 10.54 ± 2.03 and 8.33 ± 1.58 days, respectively.
Conclusion
A preoperative smoking history was associated with a decreased risk of early POCD, and a high serum TNF-α level was significantly associated with an increased risk of POCD.
Keywords: Smoking, anesthesia, cognitive disorders, logistic regression, advanced age, noncardiac surgery
Introduction
Postoperative cognitive dysfunction (POCD) refers to short-term or long-term cognitive decline that can be detected through a battery of neuropsychological tests after surgery. POCD can be considered a mild cognitive disorder characterized by impaired memory, learning difficulties, and a reduced ability to concentrate.1 The occurrence of cognitive dysfunction is associated with multiple adverse effects, such as a prolonged hospital stay, increased healthcare costs, and a high mortality rate.2 However, the mechanism of POCD remains poorly understood and the prevention of POCD remains a serious problem, particularly in patients of advanced age.
Because increased cytokine secretion is associated with surgical trauma or neurodegenerative disease, the high incidence of delirium or cognitive dysfunction in patients of advanced age is associated with a central nervous system inflammatory response.3 In previous animal experiments and clinical trials, the experimental subjects exhibited an increase in the levels of inflammatory cytokines (tumor necrosis factor-α (TNF-α), interleukin (IL)-1β, and IL-6) in the circulatory system or central nervous system, particularly in the hippocampus and glial cells, when POCD occurred simultaneously.3 However, some studies have shown that augmentation of the cholinergic anti-inflammatory pathway contributes to a decreased risk of POCD via alleviation of the inflammatory response in the hippocampus.
According to Borovikova et al.,4 the nervous system rapidly and significantly suppresses the production of TNF-α and inflammatory cytokines through a process mediated by the vagus nerve, which may reduce the systemic inflammatory response; this mechanism is called the cholinergic anti-inflammatory pathway. The principle vagal neurotransmitter, acetylcholine (ACh), significantly attenuates the release of cytokines (TNF-α, IL-1β, IL-6, and IL-18).4 Vagal outflow diverges in the celiac ganglion, giving rise to the postganglionic splenic nerve, which terminates in the spleen. From there, it sends signals via the α7 nicotinic ACh receptor (α7nAChR) to reduce the synthesis of proinflammatory cytokines by inhibiting nuclear factor-κB (NF-κB) activity5–7 and stimulating the α7nAChR in macrophages to inhibit NF-κB activity. This process prevents postoperative monocyte migration into the hippocampus and consequent cognitive dysfunction. Some studies have shown that smoking is associated with the cholinergic anti-inflammatory pathway and proinflammatory cytokines. For example, Gracia8 reported that the nicotine in tobacco stimulates the cholinergic anti-inflammatory pathway. Nicotine, a major constituent of cigarettes, is anti-inflammatory and causes immune suppression through the nAChR.9 Therefore, the purpose of the present study was to determine the correlation between smoking and POCD in patients of advanced age undergoing noncardiac surgery to further clarify the role of the cholinergic anti-inflammatory pathway in the prevention of POCD.
Methods
This was a prospective cohort study. The present investigation was approved by the Ethics Committee of Shandong Provincial Hospital Affiliated to Shandong University (No. 2017-103). All patients provided written informed consent for the publication of any associated data and accompanying images. This study was completed in a manner consistent with the STROBE statement. A completed STROBE checklist is available on request. The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Patients
This study was performed from April 2017 to December 2017 at the Shandong Provincial Hospital Affiliated to Shandong University. Patients aged ≥60 years who underwent general anesthesia for noncardiac surgery were enrolled. General anesthesia was administered by injection and inhalation in all patients. Patients were excluded if they met the following criteria: underwent cardiac surgery or neurosurgery, had stopped smoking for more than 1 year, had a previous diagnosis of a mental or nervous system disease, had a preoperative Mini-Mental State Examination score confirming the existence of cognitive impairment (illiteracy, <17 points; primary school, <20 points; secondary school or above, <24 points), were currently taking antipsychotic or antidepressant medications, were unable to communicate or experienced difficultly in communicating with language, had severe hypotension (mean arterial pressure of ≤60 mmHg) or hypoxemia (blood oxygen saturation of ≤94%) for more than 10 minutes during surgery, were sent to the intensive care unit after surgery, and did not agree to participate in this study.
For data collection, each patient’s cognitive function was assessed by an independent observer (resident in anesthesiology with >3 years of experience) 1 day preoperatively and 5 and 7 days postoperatively using the Mini-Mental State Examination, Stroop Color–Word Interference Test, and Concept Shifting Test. The following data were also collected: patient education level, age, weight, type of surgery, chronic diseases, history of alcohol drinking, allergy history, preoperative hematocrit and albumin level, history of general anesthesia for surgery, anesthesia duration, sufentanil dosage, intraoperative maximum lactate concentration, requirement for blood transfusion, duration of hospital stay, and preoperative smoking history (duration and amount).
Measurement of serum TNF-α concentration
Blood samples were obtained 1 day preoperatively and 5 and 7 days postoperatively. BD Vacutainer tubes (Becton, Dickinson and Company, Franklin Lakes, NJ, USA) were used for blood collection. Prior to the assay, the whole blood samples were centrifuged at 3,000 rpm for 20 minutes and stored at −20°C for no longer than 12 hours. The TNF-α concentration was measured using enzyme-linked immunosorbent assay kits (Jingmei Biological Engineering Co., Ltd., Jiangsu, China), according to the manufacturer’s protocol. The cytokine contents in the samples are expressed as picograms of antigen per milliliter of serum (pg/mL). The serum TNF-α concentration was divided into three levels for addition to the logistic regression.
Anesthesia and postoperative care
Our anesthesia protocol was as follows: penehyclidine (0.5 mg) was administered before intravenous induction; dexmedetomidine (1 µg/kg) was administered by pump for more than 10 minutes when venous access was successful; and etomidate (0.15–0.3 mg/kg), sufentanil (15–25 µg), cisatracurium (20 mg), and esmolol (10–40 mg) were used for induction. According to the changes in the patients’ vital signs, we used propofol, sevoflurane, and cisatracurium to sustain anesthesia before suturing the skin, and we maintained the blood oxygen saturation at 35 to 45 mmHg. We monitored the electrocardiogram, blood oxygen saturation, invasive blood pressure, and blood gas analysis during anesthesia, and we used ephedrine, atropine, norepinephrine, and nicardipine as cardiovascular drugs to manage the patients’ vital signs. When the surgery was complete, the patients were sent to the post-anesthesia care unit to regain consciousness; they were then returned to their ward. All patients received postoperative analgesia through a patient-controlled analgesia pump (8 mg of ondansetron, 100 mg of flurbiprofen, 10 mg of butorphanol, and 100 mL of normal saline). Hospital discharge was determined by the attending surgeon.
POCD assessment
The scores in the smoking and nonsmoking groups were calculated. These were estimated using “Z scores,” which were established by the International Study of Post-Operative Cognitive Dysfunction 1 (ISPOCD 1).10 Step 1: We calculated two differences in the scores for the nonsmokers: one was the difference between the score recorded 1 day preoperatively and the score recorded 5 days postoperatively, and the other was the difference between the score recorded 1 day preoperatively and the score recorded 7 days postoperatively. We then calculated the mean of these differences, which was defined as the learning effect. The learning effects were defined as an estimate of pseudo-scores that may be increased due to familiarity with the questions by repeated testing with the same questionnaire. Step 2: We calculated the difference between the preoperative and postoperative scores for the smokers, and the mean learning effect was subtracted to obtain the true difference. The defining Z score was equal to the true difference between the score of one patient in the smoking group divided by the standard deviation of the difference in the score of the same questionnaire in nonsmokers.10 The higher the Z score, the lower the postoperative test score compared with the score recorded before surgery. A patient was classified as displaying abnormal cognition if the Z score was ≥1.96.10 The study personnel who assessed cognitive dysfunction were blinded to the treatment details and smoking history.
Statistical analysis
Logistic regression requires adequate sample content. Generally, the sample size is 10 to 15 times the number of covariates.11 Sixteen covariates were included in this study, and the required sample size was 16 × 15 = 240. However, a larger sample size is more convincing. Hence, considering the limited research funding, we decided to collect data on 400 patients.
Smoking history was based on the smoking index (number of cigarettes per day multiplied by the years of smoking), and the index was divided into three levels (level 1: 0–500, level 2: 500–1000, and level 3: ≥1000). Measurement data are expressed as mean ± standard deviation. Continuous variables were analyzed using an independent-samples t-test, and categorical variables were analyzed using a chi-square test or single-sample binomial test. We checked the collinearity of continuous variables to be added to the logistic regression analysis. Initially, baseline and perioperative variables were evaluated for univariate associations with POCD. According to the above-mentioned standard of abnormal cognition, we converted the neuropsychological test scores into the binary outcome. We modeled the binary outcome of scores using a logistic regression analysis to determine which preoperative and demographic factors were associated with cognitive decline at 5 and 7 days postoperatively. All factors that were statistically significant (P ≤ 0.05) were included in the logistic regression model. The chi-square test was used to estimate significant differences in the incidence of POCD among the three smoking index levels. A P value of ≤0.05 was considered statistically significant. We used IBM SPSS Statistics for Windows, Version 19.0 (IBM Corp., Armonk, NY, USA) for all analyses.
Results
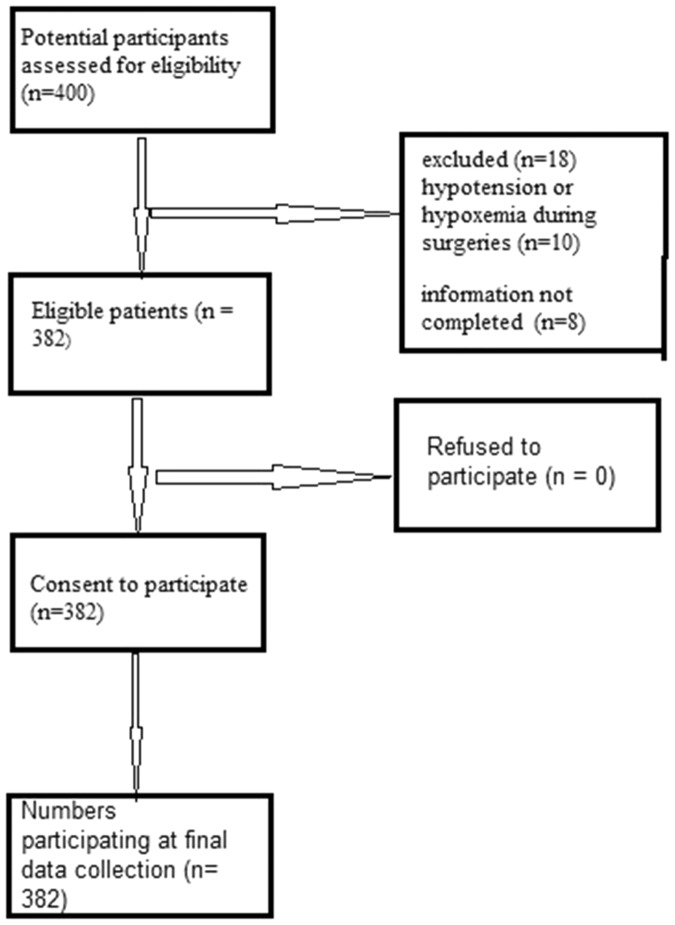
In total, 400 patients who underwent surgery by general anesthesia during the study period were recruited, and 382 patients (190 smokers, 192 nonsmokers) matched the selection criteria. All 382 patients provided written informed consent and were enrolled in this study (Figure 1). The demographic characteristics and perioperative characteristics of all enrolled patients are listed in Tables 1 and 2.
Figure 1.
Flow chart of the present study.
Table 1.
Demographic characteristics of the POCD and non-POCD groups.
| Variable | POCD group (n = 111) | Non-POCD group (n = 271) | P value |
|---|---|---|---|
| Smoking history | 50 (45.0) | 140 (51.6) | 0.009 |
| Male | 56 (50.4) | 133 (49.0) | 0.062 |
| Age, years | 66.42 ± 4.2 | 64.81 ± 3.9 | 0.001 |
| Weight, kg | 65.55 ± 9.7 | 67.76 ± 9.0 | 0.748 |
| Education level | |||
| Illiteracy | 25 (22.5) | 53 (19.5) | 0.039 |
| Primary school | 19 (17.1) | 45 (16.6) | 0.021 |
| Junior high school | 32 (28.8) | 61 (22.5) | 0.025 |
| High school | 30 (27.0) | 79 (29.1) | 0.306 |
| College | 5 (4.5) | 33 (12.1) | 0.310 |
| History of alcohol drinking | 77 (69.3) | 151 (55.7) | 0.057 |
| Chronic disease | 0.102 | ||
| Hypertension | 26 (23.4) | 84 (30.9) | |
| Coronary heart disease | 7 (6.3) | 23 (8.4) | |
| Diabetes | 9 (8.1) | 21 (7.7) | |
| History of general anesthesia | 20 (18.0) | 33 (12.1) | 0.001 |
| History of allergy | 23 (20.7) | 34 (12.5) | 0.001 |
Data are presented as n (%) or mean ± standard deviation.
POCD, postoperative cognitive dysfunction.
Table 2.
Perioperative characteristics of the POCD and non-POCD groups.
| POCD group (n = 111) | Non-POCD group (n = 271) | P value | |
|---|---|---|---|
| Preoperative Hct, % | 44.20 ± 3.8 | 40.83 ± 3.8 | 0.946 |
| Preoperative albumin, g/L | 45.49 ± 5.0 | 43.51 ± 3.6 | 0.512 |
| Anesthesia time, h | 3.64 ± 2.3 | 3.26 ± 1.0 | 0.001 |
| Sufentanil dosage, μg | 58.18 ± 14.9 | 46.18 ± 7.8 | 0.026 |
| IMLC, mmol/L | 1.04 ± 0.6 | 0.74 ± 0.2 | 1.000 |
| Blood transfusion | 20 (18.0) | 37 (13.6) | 0.001 |
Data are presented as mean ± standard deviation or n (%).
POCD, postoperative cognitive dysfunction; IMLC, intraoperative maximum lactate concentration; Hct, hematocrit.
According to the Z score, at 5 days postoperatively, 111 patients (29.05%; 95% confidence interval (CI), 25.11–35.57) exhibited POCD, including 61 nonsmokers and 50 smokers. At 7 days postoperatively, 90 patients (23.56%; 95% CI, 16.89–27.32) exhibited POCD, including 51 nonsmokers and 39 smokers. The neuropsychological test results are shown in Table 3.
Table 3.
Neuropsychological test results at 1 day preoperatively and 5 and 7 days postoperatively.
| 1 day preoperatively | 5 days postoperatively | 7 days postoperatively | |
|---|---|---|---|
| MMSE | 24.36 ± 3.03 | 24.33 ± 1.64 | 25.78 ± 1.45 |
| Concept Shifting Test, part C, time in seconds | 40.6 ± 15.7 | 41.0 ± 16.1 | 40.4 ± 17.3 |
| Concept Shifting Test, part C, number of errors | 0.6 ± 1.0 | 0.9 ± 1.6 | 0.7 ± 1.4 |
| Stroop test, part 3, number of errors | 2.6 ± 3.9 | 2.2 ± 3.1 | 1.7 ± 3.5 |
| Stroop test, part 3, time in seconds | 52.5 ± 15.2 | 52.7 ± 12.8 | 53.4 ± 13.2 |
Data are presented as mean ± standard deviation.
MMSE: Mini-Mental State Examination.
Variables that were statistically significant in the univariate analyses are shown in Table 4. After the multicollinearity test, no variables were excluded from the subsequent logistic regression analysis. Thus, the nine above-described variables were included in the multivariate analysis. Because of hospital discharge, we included the cognitive status at 5 and 7 days postoperatively in the multivariate analysis.
Table 4.
Predictors of cognitive dysfunction at 5 and 7 days postoperatively.
|
Univariate analysis |
Multivariate logistic regression analysis 5 days postoperatively |
Multivariate logistic regression analysis7 days postoperatively |
|||
|---|---|---|---|---|---|
| Predictors | P value | OR (95% CI) | P value | OR (95% CI) | P value |
| Smoking history | 0.009 | 0.833 (0.497‒0.911) | 0.006 | 0.684 (0.583‒0.952) | 0.002 |
| Dose of sufentanil | 0.026 | 2.901 (2.127‒16.381) | 0.007 | 1.073 (1.005‒1.632) | 0.036 |
| Age | 0.001 | 1.922 (1.057‒2.132) | 0.018 | 2.890 (1.583‒9.581) | 0.023 |
| Lower education level | 0.039 | 2.512 (1.603‒15.244) | 0.034 | 3.800 (1.200‒13.493) | 0.040 |
| Serum TNF-α concentration | 0.001 | 2.903 (1.572–4.484) | 0.042 | 1.437 (1.206–1.680) | 0.027 |
| Anesthesia duration | 0.001 | 1.783 (1.514‒7.366) | 0.047 | ||
| History of general anesthesia | 0.001 | 3.674 (2.496‒4.217) | 0.044 | ||
| History of allergy | 0.001 | ||||
| Blood transfusion | 0.001 | ||||
OR, odds ratio; CI, confidence interval; TNF-α, tumor necrosis factor-α.
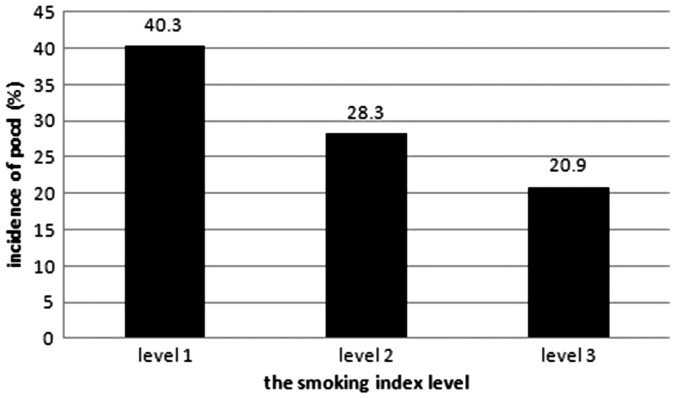
According to the logistic regression analysis, seven variables were identified as independent predictors of cognitive dysfunction 5 days postoperatively, and five variables were identified as independent predictors of cognitive dysfunction 7 days postoperatively (Table 4). Compared with nonsmokers, a preoperative smoking history (smoking index) was a protective factor for a lower risk of cognitive dysfunction at 5 days postoperatively (odds ratio (OR), 0.833; 95% CI, 0.497–0.911; P = 0.006; B = −2.312) and at 7 days postoperatively (OR, 0.684; 95% CI, 0.583–0.952; P = 0.002; B = −0.949) (Table 4). Patients with a high smoking index level had a significantly lower incidence of early POCD (Figure 2).
Figure 2.
Relationship between the smoking index level and incidence of early POCD. POCD, postoperative cognitive dysfunction.
The TNF-α concentrations were significantly higher in the POCD than non-POCD group at 5 and 7 days postoperatively. Compared with 1 day preoperatively, the TNF-α concentrations in the POCD group increased significantly at 5 and 7 days postoperatively (P = 0.042 and P = 0.027, respectively) (Table 4). There was no significant difference between before and after surgery in the non-POCD group (Table 5).
Table 5.
Concentration of TNF-α in POCD and non-POCD groups.
| Group | Patients | 1 day preoperatively | 5 days postoperatively | P value | 7 days postoperatively | P value | |
|---|---|---|---|---|---|---|---|
| TNF-α, pg/mL | POCD | 111 | 32.23 ± 4.81b1 | 41.61 ± 8.02a,b2 | 0.042 | 37.67 ± 6.73a | 0.027 |
| Non-POCD | 271 | 29.67 ± 4.55 | 31.62 ± 6.21 | 0.194 | 28.04 ± 7.71 | 0.077 |
Data are presented as mean ± standard deviation.
POCD, postoperative cognitive decline; TNF-α, tumor necrosis factor-α.
Compared with 1 day preoperatively, aP ≤ 0.05; Compared with the non-POCD group, bP ≤ 0.05, b1P = 0.003, b2P < 0.001.
The hospital stay for patients without POCD was 8.33 ± 1.58 days, whereas that of patients with POCD (5 and 7 days postoperatively) was 10.54 ± 2.03 days. Patients with POCD exhibited a prolonged postoperative hospital stay compared with patients without POCD.
Discussion
In the present study, a preoperative smoking history was associated with a decreased risk of early POCD in patients of advanced age who underwent noncardiac surgery. A high serum TNF-α level was significantly associated with an increased risk of POCD. Patients with a high smoking index level had a significantly lower incidence of early POCD. Other independent risk factors were age, a history of general anesthesia, the sufentanil dosage, a lower education level, and the anesthesia duration. Moreover, POCD prolonged the hospital stay.
In 2008, Monk et al.12 conducted a study of 1064 patients and found that 36.6% of young people (18–39 years old), 30.4% of middle-aged people (40–59 years old), and 41.4% of advanced-age people (≥60 years old) exhibited POCD at discharge. In 1998, Moller et al.10 performed the ISPOCD in which they analyzed the occurrence of POCD in 1218 patients of advanced age who received general anesthesia during noncardiac surgery and showed that the incidence of POCD was 25.8% at 1 week postoperatively and 9.9% after 3 months; these values were obviously higher than those recorded in normal people who did not undergo surgery (the control group). The reasons for this variation include differences in the patient population (such as age, severity of illness, and type of procedure), the sensitivity of the screening instrument, and local medical practice. In our study, the incidence was less than that in some previously reported studies, perhaps because we administered dexmedetomidine to every patient and used strict exclusion criteria, which may have selected healthier patients in our study than in other studies.
Our understanding of the pathophysiology of POCD is limited, and researchers have not been able to determine whether it is caused by anesthesia.13 Numerous studies have been performed to identify the risk factors for developing POCD, and substantially different views have been reported.
Some studies indicate that the inflammatory and stress response to the surgical procedure play a significant role in the development of cognitive disorders. Mu et al.2 revealed that high serum cortisol levels were associated with an increased risk of postoperative delirium, and Glumac et al.14 demonstrated that the preoperative use of dexamethasone reduced the inflammatory response and decreased the risk of early POCD after cardiac surgery. These studies revealed that reducing inflammation may help to prevent POCD. Many researchers believe that augmentation of the cholinergic anti-inflammatory pathway contributes to a decreased risk of POCD via alleviation of the inflammatory response in the hippocampus. Vagal outflow diverges in the celiac ganglion, giving rise to the postganglionic splenic nerve; this nerve terminates in the spleen, where it sends signals via the α7nAChR to reduce synthesis of proinflammatory cytokines by inhibiting nuclear NF-κB activity5–7 and stimulating the α7nAChR in macrophages to inhibit NF-κB activity. This prevents postoperative monocyte migration into the hippocampus and consequent cognitive dysfunction.
Increasing age is a universally identified risk factor for POCD,10 suggesting that the naturally aged brain might be the basis of the occurrence of cognitive dysfunction. A history of general anesthesia and a long duration of anesthesia indicate a more complex procedure and a higher likelihood of postoperative complications, and these factors are also associated with the occurrence of POCD15,16 via the loss of neurons and glucocorticoid receptors in the hippocampal region.17
Chronic diseases have consistently been shown to be associated with cognitive dysfunction. According to Reinprecht et al.,18 high blood pressure, particularly high blood pressure in late middle age, is associated with a decline in cognitive function in people of advanced age. Long-term hypertension may cause cerebral vasospasm, cerebral ischemia, necrosis, and atrophy. The loss of cognitive function may be accelerated by the effects of traumatic stimulation by surgery and general anesthetics.19 Research by Feinkohl et al.20 revealed a higher risk of POCD in patients with than without diabetes, and people with diabetes who have poor blood glucose control have a higher risk of POCD. Our study did not reveal an association between a history of chronic disease and early POCD; this lack of association may have been due to the small proportion or small sample size of patients with chronic diseases.
In the present study, the sufentanil dosage was an independent risk factor for POCD. Previous studies of opioids, such as morphine and sufentanil, have shown that these drugs play an important role in the inflammatory response. The immunosuppressive effect of morphine has been confirmed, and many studies have revealed a similar effect of fentanyl.21 Sufentanil and fentanyl are both opioids, but sufentanil exerts a stronger analgesic effect. Leung et al.22 postulated that the administration of high opioid doses increases the risk of postoperative delirium in all patients. However, Silbert et al.23 hypothesized that a high dose of fentanyl is not correlated with the incidence of POCD from 3 to 12 months after coronary bypass surgery. In summary, the effect of high sufentanil doses on POCD requires further study.
A lower education level was associated with early cognitive decline, as previously shown in the ISPOCD1.10 As shown in the study by Scarmeas and Stern,24 a larger cognitive reserve capacity protects against the development of dementia in patients of advanced age and is associated with a better functional status after traumatic brain injury.25 Our study is similar to another investigation of a normal aging population that showed protective effects of a higher education level or better intellectual capacity on the development of cognitive decline.
Glumac et al.26 revealed that the stress response to surgical procedures is unlikely to represent an exclusive trigger for the occurrence of POCD and that interdependent mechanisms, such as inflammatory and stress responses, may be involved in the pathogenesis of POCD through their interaction effects. In the present study, we evaluated different operations performed by different surgeons. The surgeons had their own characteristic operative techniques, and the surgical trauma may not have been the same among the patients. Therefore, we used the anesthetic duration and requirement for blood transfusion to assess the effects of different surgical procedures.
This study mainly focused on the effect of nicotine on POCD. Nicotine exerts multiple effects on health. Nicotine simulates ACh signaling, which has a variety of functions in the entire organism. Most of these functions are unrelated or even antagonistic. In this study, we found that a preoperative smoking history might reduce the risk of POCD in patients of advanced age, potentially because of nicotine-mediated activation of the cholinergic anti-inflammatory pathway and inhibition of central nervous system inflammation or inflammatory factors (TNF-α, IL-1β, and IL-6).
The impulses of the vagus nerve induce ACh release near the macrophages in the reticuloendothelial tissue, and ACh specifically binds to the α7nAChR; the α7 subunit on immune cells inhibits the release of inflammatory cytokines through the cholinergic anti-inflammatory pathway.4 Pelissier-Rota et al.27 found that nicotine-mediated activation of the α7nAChR exerts a protective effect on epithelial morphology and maintains barrier integrity; therefore, it may be helpful for nerve cells. According to Borovikova et al.,4 the nervous system rapidly and significantly suppresses the production of inflammatory cytokines, such as TNF-α, IL-1β, IL-6, and IL-18, through a process mediated by the vagus nerve; this may reduce the systemic inflammatory response. A study by Chen et al.28 showed that nicotine, the main active ingredient in tobacco, can inhibit TNF-α secretion by activating the cholinergic anti-inflammatory pathway. Researchers have also found that a cigarette extract or nicotine inhibited the production of IL-1β, IL-2, interferon-γ, and TNF-α in humans and mice.29,30
Inflammatory cytokines and central nervous system inflammatory reactions play important roles in the occurrence of POCD. In previous animal experiments or clinical trials, the experimental subjects presented an increase in the levels of inflammatory cytokines (TNF-α, IL-1β, and IL-6) in the circulatory system or central nervous system when POCD occurred simultaneously.3 Wei et al.31 observed a neuroprotective effect of nicotine on adult rats, and its anti-POCD neuroprotective mechanism may involve activation of the brain-derived neurotrophic factor/tropomyosin-related receptor kinase B signaling pathway and inhibition of the NF-κB signal transduction pathway. In the current study, we found that the incidence of early POCD decreased as the smoking index level increased, which may be demonstrated by nervous inflammatory reactions and the cholinergic anti-inflammatory pathway mentioned above. Thus, we may conclude that a preoperative smoking history is associated with a decreased risk of POCD through activation of the cholinergic anti-inflammatory pathway, which inhibits inflammatory cytokine release, suppresses central nervous inflammatory reactions, and subsequently decreases the incidence of POCD.
This study has several limitations. First, we only performed neuropsychological tests on the enrolled patients 1 day preoperatively and 5 and 7 days postoperatively, and we did not observe the effect of smoking on the long-term prognosis of the patients. Second, due to the limitations of the study conditions, we failed to monitor and control the depth of anesthesia and to determine the cerebral oxygen saturation of the patients enrolled in this study; thus, we did not completely control for confounding factors. Third, smoking is an independent risk factor for intracranial arterial stenosis, and the prevalence of intracranial arterial stenosis increases with the number of years of smoking. Long-term smokers do not exhibit a reduction of this effect on intracranial artery stenosis even if they stop smoking.32 In smokers of advanced age, POCD may also be related to cerebrovascular disease caused by smoking, which we were not able to exclude in this study. Fourth, different surgeons have their own characteristic operative techniques, and the resulting surgical trauma may not have been the same among the patients. Thus, instead of using different types of surgeries, we used the anesthesia duration and requirement for blood transfusion to assess the effect of different surgical procedures. Finally, in this observational study, we were unable to confirm that a preoperative smoking history reduces the risk of POCD by activating the cholinergic anti-inflammatory pathway of the spleen; thus, further animal experiments are necessary. Additional studies are needed to illuminate the mechanisms by which a preoperative smoking history may affect POCD.
Conclusions
On the basis of the results of our study, a preoperative smoking history is associated with a decreased risk of early POCD by stimulating the cholinergic anti-inflammatory pathway. A high serum TNF-α level was significantly associated with an increased risk of POCD. Age, a history of general anesthesia, the sufentanil dosage, a lower education level, and the anesthesia duration are independent risk factors for POCD. Finally, POCD prolongs the hospital stay.
Acknowledgements
We wish to thank Dr Wei Li, Department of Anesthesiology, Shandong Provincial Hospital, affiliated to Shandong University, Jinan, China, for his help in revising the paper and raising parts of the funding.
Authors’ contributions
Planning: Runjia Wang and Mengyuan Zhang.
Study conduct: Runjia Wang and Gongming Wang.
Data analysis: Runjia Wang and Yang Liu.
Writing of the paper: Runjia Wang and Gongming Wang.
Revision of the paper: All authors.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This study was supported by “the research of preventive effect and dosage of dexmedetomidine in postoperative disorders in advanced patients undergoing arthroplasty” (Grant No. 26020112721741).
References
- 1.Miller RD. Miller’s Anesthesia. Philadelphia: ELSEVIER, 2015. [Google Scholar]
- 2.Mu DL, Wang DX, Li LH, et al. High serum cortisol level is associated with increased risk of delirium after coronary artery bypass graft surgery: a prospective cohort study. Crit Care 2010; 14: R238. doi: 10.1186/cc9393 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Tang Y, Ouyang W. Inflammation-relevant mechanisms for postoperative cognitive dysfunction and the preventive strategy. Zhong Nan Da Xue Xue Bao Yi Xue Ban 2017; 42: 1321–1326. [DOI] [PubMed] [Google Scholar]
- 4.Borovikova LV, Ivanova S, Zhang M, et al. Vagus nerve stimulation attenuates the systemic inflammatory response to endotoxin. Nature 2000; 405: 458–462. [DOI] [PubMed] [Google Scholar]
- 5.Rosas-Ballina M, Olofsson PS, Ochani M, et al. Acetylcholine-synthesizing T cells relay neural signals in a vagus nerve circuit. Science 2011; 334: 98–101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Pavlov VA, Tracey KJ. The vagus nerve and the inflammatory reflex-linking immunity and metabolism. Nat Rev Endocrinol 2012; 8: 743–754. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Olofsson PS, Rosas-Ballina M, Levine YA, et al. Rethinking inflammation: neural circuits in the regulation of immunity. Immunol Rev 2012; 248: 188–204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Gracia MC. Exposure to nicotine is probably a major cause of inflammatory diseases among non-smokers. Med Hypotheses 2005; 65: 253–258. [DOI] [PubMed] [Google Scholar]
- 9.Yang J, Shi S, Shi L, et al. Nicotine, an α7 nAChR agonist, reduces lipopolysaccharide-induced inflammatory responses and protects fetuses in pregnant rats. Am J Obstet Gynecol 2014; 211: 538.e1–7. [DOI] [PubMed] [Google Scholar]
- 10.Moller JT, Cluitmans P, Rasmussen LS, et al. Long-term postoperative cognitive dysfunction in the elderly ISPOCD1 study. ISPOCD investigators. International Study of Post-Operative Cognitive Dysfunction. Lancet 1998; 351: 857–861. [DOI] [PubMed] [Google Scholar]
- 11.Jiqian Fang. Health statistics. Beijing: People’s Medical Publishing House, 2012: 399–400. [Google Scholar]
- 12.Monk TG, Weldon BC, Garvan CW, et al. Predictors of cognitive dysfunction after major noncardiac surgery. Anesthesiology 2008; 108: 18–30. [DOI] [PubMed] [Google Scholar]
- 13.Roberta L, Hines KEM. Stoelting’s anesthesia and co-existing disease. Philadelphia: ELSEVIER, 2012. [Google Scholar]
- 14.Glumac S, Kardum G, Sodic L, et al. Effects of dexamethasone on early cognitive decline after cardiac surgery: A randomised controlled trial. Eur J Anaesthesiol 2017; 34: 776–784. [DOI] [PubMed] [Google Scholar]
- 15.Krähenbühl ES, Immer FF, Stalder M, et al. Temporary neurological dysfunction after surgery of the thoracic aorta: a predictor of poor outcome and impaired quality of life. Eur J Cardiothorac Surg 2008; 33: 1025–1029. [DOI] [PubMed] [Google Scholar]
- 16.Spahr-Schopfer I, Vutskits L, Toni N, et al. Differential neurotoxic effects of propofol on dissociated cortical cells and organotypic hippocampal cultures. Anesthesiology 2000; 92: 1408–1417. [DOI] [PubMed] [Google Scholar]
- 17.Hokkanen SRK, Hunter S, Polvikoski TM, et al. Hippocampal sclerosis, hippocampal neuron loss patterns and Tdp-43 in the aged population. Brain Pathol 2018; 28: 548–559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Reinprecht F, Elmstahl S, Janzon L, et al. Hypertension and changes of cognitive function in 81-year-old men: a 13-year follow-up of the population study “Men born in 1914”, Sweden. J Hypertens 2003; 21: 57–66. [DOI] [PubMed] [Google Scholar]
- 19.Forette F, Seux ML, Staessen JA, et al. Prevention of dementia in randomised double-blind placebo-controlled Systolic Hypertension in Europe (Syst-Eur) trial. Lancet 1998; 352: 1347–1351. [DOI] [PubMed] [Google Scholar]
- 20.Feinkohl I, Winterer G, Pischon T. Diabetes is associated with risk of postoperative cognitive dysfunction: A meta-analysis. Diabetes Metab Res Rev 2017; 33. doi:10.1002/dmrr.2884 [DOI] [PubMed] [Google Scholar]
- 21.Odunayo A, Dodam JR, Kerl ME, et al. Immunomodulatory effects of opioids. J Vet Emerg Crit Care (San Antonio) 2010; 20: 376–385. [DOI] [PubMed] [Google Scholar]
- 22.Leung JM, Sands LP, Lim E, et al. Does preoperative risk for delirium moderate the effects of postoperative pain and opiate use on postoperative delirium? Am J Geriatr Psychiatry 2013; 21: 946–956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Silbert BS, Scott DA, Evered LA, et al. A comparison of the effect of high- and low-dose fentanyl on the incidence of postoperative cognitive dysfunction after coronary artery bypass surgery in the elderly. Anesthesiology 2006; 104: 1137–1145. [DOI] [PubMed] [Google Scholar]
- 24.Scarmeas N, Stern Y. Cognitive reserve: implications for diagnosis and prevention of Alzheimer's disease. Curr Neurol Neurosci Rep 2004; 4: 374–380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kesler SR, Adams HF, Blasey CM, et al. Premorbid intellectual functioning, education, and brain size in traumatic brain injury: an investigation of the cognitive reserve hypothesis. Appl Neuropsychol 2003; 10: 153–162. [DOI] [PubMed] [Google Scholar]
- 26.Glumac S, Kardum G, Karanović N. A prospective cohort evaluation of the cortisol response to cardiac surgery with occurrence of early postoperative cognitive decline. Med Sci Monit 2018; 24: 977–986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Pelissier-Rota MA, Pelosi L, Meresse P, et al. Nicotine-induced cellular stresses and autophagy in human cancer colon cells: A supportive effect on cell homeostasis via up-regulation of Cox-2 and PGE(2) production. Int J Biochem Cell Biol 2015; 65: 239–256. [DOI] [PubMed] [Google Scholar]
- 28.Chen Y, Guo Q, Pan X, et al. Smoking and impaired bone healing: will activation of cholinergic anti-inflammatory pathway be the bridge? Int Orthop 2011; 35: 1267–1270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Ouyang Y, Virasch N, Hao P, et al. Suppression of human IL-1beta, IL-2, IFN-gamma, and TNF-alpha production by cigarette smoke extracts. J Allergy Clin Immunol 2000; 106: 280–287. [DOI] [PubMed] [Google Scholar]
- 30.Li-Sha G, Jing-Lin Z, Li L, et al. Nicotine inhibits the production of proinflammatory cytokines of mice infected with coxsackievirus B3. Life Sci 2016; 148: 9–16. [DOI] [PubMed] [Google Scholar]
- 31.Wei P, Zheng Q, Liu H, et al. Nicotine-Induced Neuroprotection against Cognitive Dysfunction after Partial Hepatectomy Involves Activation of BDNF/TrkB Signaling Pathway and Inhibition of NF-kappaB Signaling Pathway in Aged Rats. Nicotine Tob Res 2018; 20: 515–522. [DOI] [PubMed] [Google Scholar]
- 32.Ran Liu, Yang Hua, Lingyun Jia, et al. Correlation between smoking and occurrence of intracranial artery stenosis by ultrasonography: an analysis of multi-center research results. Chinese Journal of Cerebrovascular Diseases 2017; 06: 297–301 + 312. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.