Abstract
Osteoarthritis is a progressive and debilitating condition. An increasing number of total knee replacements are being performed under the age of 65. Improved understanding of the action of mesenchymal stem cells (MSC) has seen renewed interest in their role in cartilage repair. A 43-year-old man presented with grade IV medial compartment knee osteoarthritis. The patient underwent high tibial osteotomy (HTO) and arthroscopic abrasion arthroplasty in combination with adipose-derived MSC therapy. The patient reported improvement in pain and function as measured by validated outcome scores. Repeat MRI including T2 mapping techniques showed hyaline-like cartilage regeneration. This case highlights the potential benefit of surgical interventions including HTO in combination with MSC therapy in early-onset severe osteoarthritis. This technique may considerably delay or prevent the need for total knee replacement in young patients. Further controlled trials are needed to confirm the reproducibility of this outcome.
Keywords: orthopaedics, degenerative joint disease, osteoarthritis, sports and exercise medicine, osteoarthritic knww
Background
Osteoarthritis (OA) is a chronic and progressive degenerative condition and can result in considerable pain and functional limitation. Symptomatic OA is not just a disease of the elderly and has an observed radiological prevalence rate of 10% of men and 18% of women over the age of 45 years.1–4 Early degeneration is often attributable to secondary OA as a consequence of previous trauma. Of concern is an observed increase in the number of patients undergoing total knee replacement below the age of 65.5
In patients with symptomatic unicompartmental medial OA and associated genu varus malalignment, the surgical technique of high tibial osteotomy (HTO) may be considered to delay the need for total knee replacement (TKR). Previous research has shown a mean survival time to TKR of up to 10 years following HTO.6 Past research has indicated the benefits of arthroscopic techniques including arthroscopic abrasion arthroplasty and microfracture in combination with HTO to promote chondral repair.7 There remains questionable long-term benefits of these arthroscopic techniques however, as subsequent histopathology has shown type I collagen fibrocartilage as opposed to type II collagen hyaline-like cartilage formation.8–10 Furthermore, fibrocartilage has poor load bearing properties with an observed decrease in clinical outcome as early as 2 years.11
The use of cellular therapies including mesenchymal stem cells (MSCs) has been postulated as a technique to promote the conversion of fibrocartilage towards mature hyaline-like cartilage.12 Preclinical trials have shown significant structural and histological improvements in cartilage formation following intra-articular MSC injections following microfracture/microdrilling.13 14 Clinical trials using bone marrow or peripheral blood-derived MSCs in combination with HTO and arthroscopic chondral stimulation techniques, including microfracture or microdrilling, have observed successful hyaline-like cartilage regeneration with type II collagen shown on histopathology analysis.15–17 This case study describes the novel use of intra-articular injections of autologous adipose-derived MSCs (AdMSCs) in combination with a single-stage HTO and arthroscopic abrasion arthroplasty in the treatment of a grade IV medial compartment knee OA with an associated significant varus malalignment.
Case presentation
A 43-year-old man presents with progressive knee pain over the last 10 years. He notes a medical history of previous knee arthroscopy at age 17 with multiple subsequent arthroscopies. The last arthroscopy was performed 10 years ago. He is otherwise well.
On initial examination, the patient had a varus malalignment of his knee on stance. He had a moderate effusion and his knee range of motion was limited with a fixed flexion deformity of 10° and flexion through to 90° (measured by a handheld goniometer). He had a stable knee, and hip examination was normal.
Radiological examination included routine knee series X-ray (including a weight-bearing Rosenberg view), X-ray long leg mechanical axis and an MRI. Weight-bearing X-ray confirmed grade IV medial compartment OA based on Kellgren and Lawrence criteria (figure 1). Long leg mechanical axis alignment indicated a varus angulation of 6.8° (figure 2). MRI showed evidence of the previous near-complete medial meniscus resection with subsequent diffuse full-thickness cartilage loss over the medial femoral condyle and medial tibial plateau.
Figure 1.
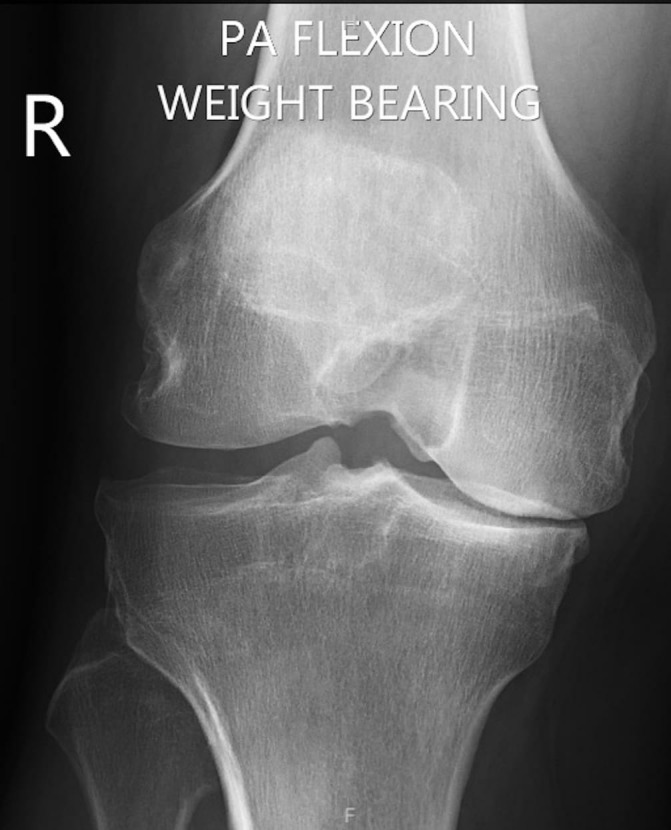
Weight-bearing X-ray in flexion (Rosenburg view) showing grade IV medial compartment osteoarthritis.
Figure 2.
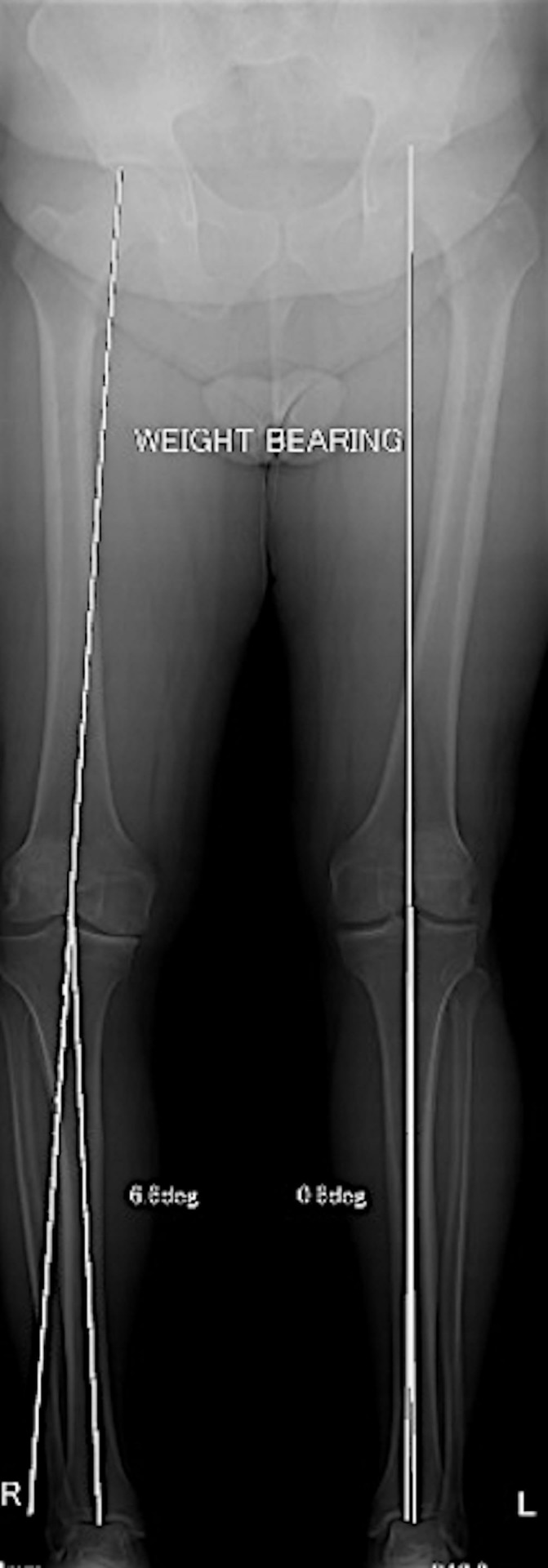
Long leg mechanical axis X-ray showing a varus knee alignment of 6.8°.
Unfortunately, despite a focused conservative management programme including simple analgesics, low impact exercise, attempted weight management and use of valgus heel wedges to offload the medial compartment, the patient had persistent and debilitating pain with significant impact on his work and quality of life.
After careful consideration, and with consultation between his treating physician and orthopaedic surgeon, the patient underwent a single-stage HTO with arthroscopic abrasion arthroplasty to areas of grade IV chondropathology of the medial compartment with planned postoperative AdMSC therapy.
The patient received formal written information regarding the relative risks of surgery and the use of MSC therapy. Prior to commencement of treatment, the patient completed formal written consent.
Investigations
See case presentation.
Treatment
Arthroscopic abrasion arthroplasty and HTO surgical procedure
The patient received a general anaesthetic and surgery was performed under tourniquet control.
Arthroscopic abrasion arthroplasty was performed as previously described by Johnson and colleagues.12 Using a spherical 4.5 mm arthroscopic bur, the area of eburnated bone was abraded down to the subchondral plate until capillary bleeding was observed. This was performed to both the medial femoral condyle and the medial tibial plateau (figures 3 and 4). Chondroplasty using a 4.5 mm arthroscopic shaver was performed to areas of unstable cartilage in both the medial and lateral compartment. A complex tear of the posterior horn and body of the medial meniscus remnant was debrided using the 4.5 mm arthroscopic shaver.
Figure 3.
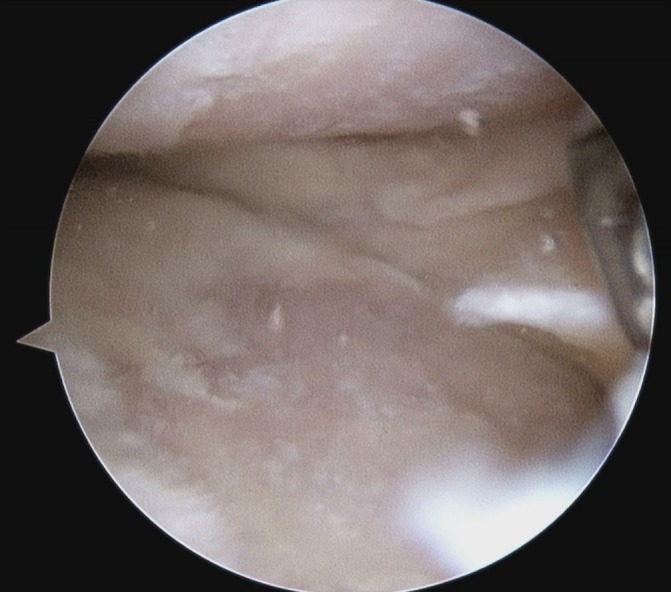
Arthroscopic picture showing areas of grade IV osteoarthritis of the medial compartment.
Figure 4.
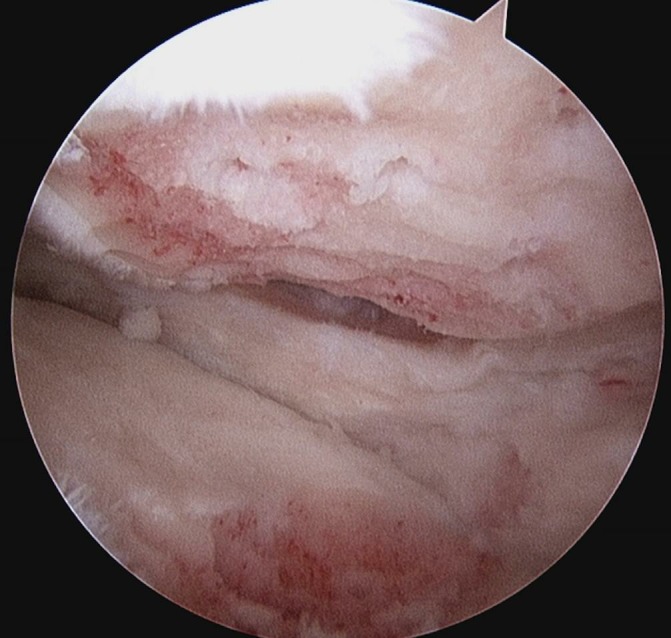
Arthroscopic picture showing the result of arthroscopic abrasion arthroplasty to areas of grade IV chondropathology with exposure of capillary bleeding.
After arthroscopic examination and abrasion arthroplasty, the patient underwent a medial opening HTO using a Flexit System plate (Neosteo, Nantes, France). Allograft bone crunch (Allovance Crunch, Australian Biotechnologies, Australia) was used to augment the opening HTO (figure 5).
Figure 5.
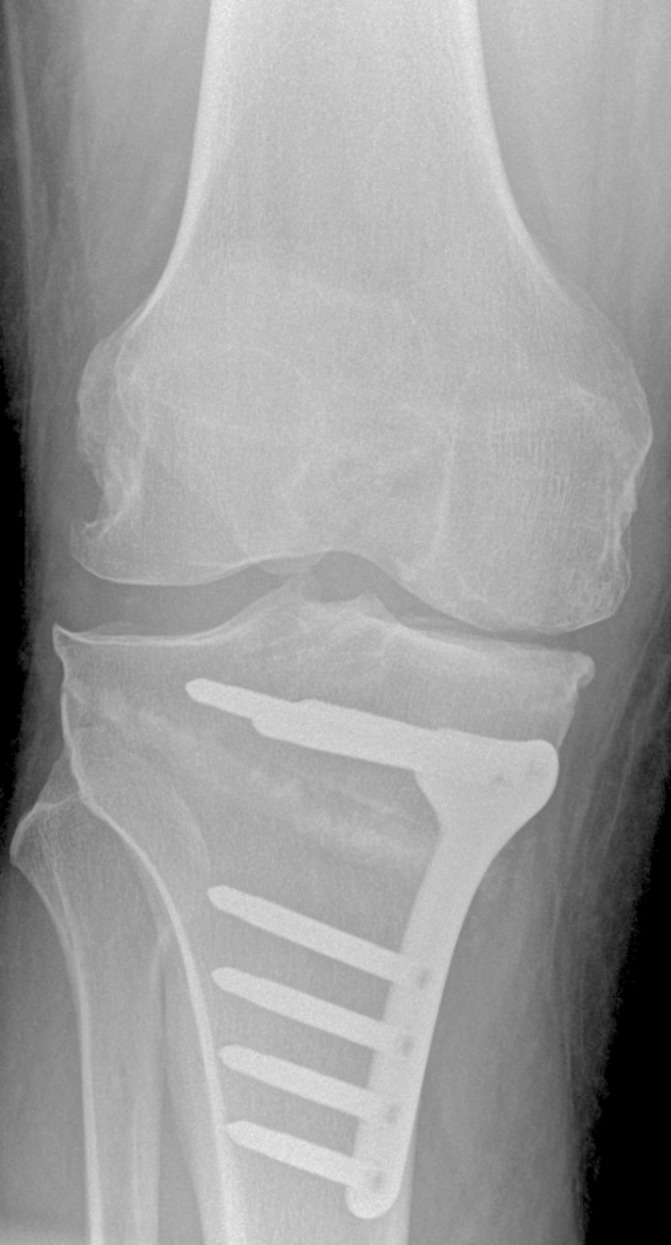
Postoperative X-ray of the high tibial osteotomy.
The patient was placed in a continuous passive motion (CPM) device in the immediate postoperative period with ROM set at 0–45°.
Autologous adipose-derived MSC preparation
Harvest procedure
Due to ease of harvest, relative abundance and observed chondrogenic potential, adipose tissue was chosen as a source of autologous MSCs.18 19
The patient underwent an abdominal lipoharvest procedure as previously described in past publications.20 After infiltration of 300 mL of tumescent fluid (comprising 30 mL of 2% lignocaine, 1 mL of 1:1000 epinephrine and 1 mL of 8.4% bicarbonate suspended in normal saline to a total volume of 1000 mL), 80 mL of lipoaspirate was collected using manual syringe suction and a 3 mm lipoaspirate cannula. Directly after collection, the lipoaspirate was transferred to a clean room stem cell laboratory (Magellan Stem Cells, Melbourne, Australia).
Isolation and expansion of MSCs
Isolation and expansion of autologous AdMSCs was achieved using techniques that have been previously published.20–22 All processing of cellular material was performed under strict sterile conditions within a clean room laboratory with equivalent of >ISO 5 air quality and class II biological safety cabinets.
Isolated and expanded AdMSCs were cryopreserved in clinical grade-qualified MSC cryoprotectant media using a validated control rate freezing method and stored in liquid nitrogen until required.23 24
Characterisation and sterility testing
Criteria established by the International Society of Cellular Therapy were used to characterise the isolated cell population.25 Fluorescent-activated cell sorting (FACS) analysis, performed independently at Monash University, assessed for the presence of MSC surface markers CD90, CD73 and CD105 and absence of haematopoietic surface markers CD14, CD19, CD34 and CD45 (table 1).
Table 1.
Flow cytometry fluorescent-activated cell sorting surface marker analysis showing results consistent with mesenchymal stem cells as per the International Society of Cellular Therapy guidelines
| Histograms—positive markers | Histograms—negative markers | ||||||
| CD90+ve | CD73+ve | CD105+ve | CD14+ve | CD19+ve | CD34+ve | CD45+ve | |
| Percentage | 97.54 | 99.35 | 99.60 | 0.92 | 0.22 | 0.17 | 0.34 |
Independent sterility testing for microbial growth/contamination was performed on a cell sample at completion of isolation and expansion.
AdMSC carrier media preparation
Prior to commencement of therapy, an autologous carrier media, which the AdMSCs are suspended in at time of injection, was prepared. Previous research had indicated that the combination of MSCs with a blood-derived carrier media may assist in increased expression of collagen type II and reduced chondrocyte apoptosis.26 Growth factors found in autologous blood-derived therapies (ie, platelet-rich plasma and autologous conditioned serum) including transforming growth factor beta1 (TGFβ1) and basic fibroblast growth factor (bFGF) have been shown to assist in migration of stromal cells to suite of injury.27 bFGF may also play a critical role in suppressing collagen type X formation and prevent possible hypertrophic endochondral ossification.28 For the purpose of this case series, autologous conditioned serum was chosen as the carrier media.
MSC treatment and injection method
Cryopreserved cells were thawed in a sterile water bath and cryoprotectant media removed using both centrifugation and washing in chilled phosphate buffered saline. The resultant cell pellet was resuspended in autologous conditioned serum to a total of 3 mL. Cell number and viability was confirmed using a Muse Cell Analyser (Merck, Millipore, USA).
The patient underwent an intra-articular injection of AdMSC therapy at day 3 post knee HTO and arthroscopic abrasion and another injection at 6 months.
The patient received a total of 48 million AdMSCs (viability 89%) on Day 3 and a total of 51 million AdMSCs (viability 87%) at 6 months.
At the time of injection, the patient’s knee was prepped using standard sterile procedural protocols. A total of 2 mL of 1% lignocaine was infiltrated to the subcutaneous tissue at the site of the injection. Under ultrasound guidance and using a sterile technique, the autologous AdMSCs were injected into the intra-articular knee space using a superolateral approach to the patella. On day 3, at commencement of MSC therapy, 15 mL of haemarthrosis effusion—the result of arthroscopic abrasion—was aspirated prior to the intra-articular injection of MSCs.
Postoperative rehabilitation
Following surgical HTO and arthroscopic abrasion arthroplasty, the patient was non-weight bearing for a period of 8 weeks. During this time, he used a CPM device for 2 hours per day. He was encouraged to adjust the degree of flexion as tolerated. Furthermore, the patient was instructed on non-weight-bearing muscle activation exercises.
At 8 weeks, the patient was allowed to weight bear as tolerated and commenced active range of motion exercises on a stationary bike at minimal load. The patient was advised to do this for 30–60 min at least twice per day. Load was increased as tolerated.
A formal strengthening programme was commenced under physiotherapist supervision with avoidance of high impact exercise.
Analysis method and outcome measures
Pain and functional outcome was assessed using validated questionnaires and were completed at baseline, 2, 6, 12 and 24 months postoperatively.
These measures included:
The Knee Injury and Osteoarthritis Outcome Score (KOOS). KOOS consists of five subscales—pain, symptoms, function in daily living, function in sport and recreation and knee-related quality of life. Each subscale receives a score with 100 indicating no symptoms and 0 indicating maximum symptoms.29
The Western Ontario and McMaster Universities Arthritis Index (WOMAC Index 3.0). WOMAC is a validated quality of life score in patients with symptomatic osteoarthritis. In this case study, the global WOMAC score is presented as in inverse percentage to be directly comparable to KOOS subscales. Scores ranging from 100 indicate no symptoms and 0 indicates maximum symptoms.30
The Numeric Pain Rating Scale (NPRS). The patient was asked to rate their knee pain intensity over the previous week on an 11-point scale of 0–10. The NPRS has been validated for use in people with musculoskeletal pain.31
Questionnaires were completed remotely and online using the software programme Clinical Intelligence (Clinical Intelligence, Melbourne, Australia).
Longitudinal structural outcome was assessed using MRI performed prior to surgery and at 1 month, 6 months, 12 months and 24 months postoperatively. A modified International Cartilage Repair Society (ICRS) score was used to describe the cartilage defect32:
Grade 0: normal cartilage
Grade 1: focal blistering and intracartilaginous low-signal intensity area with an intact surface and bottom
Grade 2: irregularities on the surface or bottom and loss of thickness of less than 50%
Grade 3: deep ulceration with loss of thickness of more than 50%
Grade 4: full-thickness cartilage wear with exposure of subchondral bone.
The MRI technique of T2 relaxation time was used as a validated non-invasive method to assess cartilage quality at 12 months and again at 24 months.33 34
Outcome and follow-up
Pain and functional outcome
The patient experienced a routine postoperative period with minimal discomfort during the period of non-weight bearing and use of the CPM. At 8 weeks, the patient was able to commence full weight bearing and a progressive lower limb strengthening programme. Pain scores remained consistently improved throughout follow-up with NPRS at 2 years recorded as 4—an improvement from 7 at baseline and hence an observed 43% reduction in perceived pain (figure 6).
Figure 6.
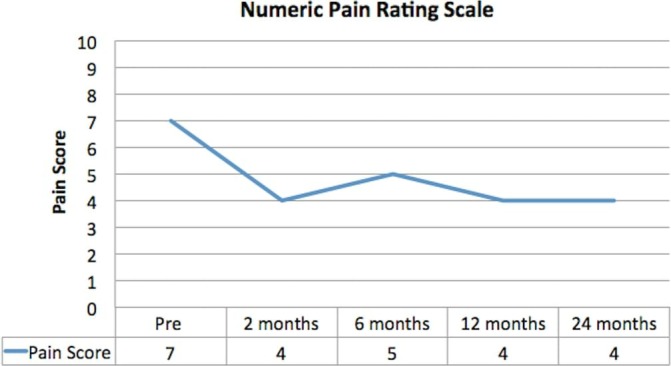
Numeric Pain Rating Scale. Pain improved throughout follow-up and remained improved until completion of follow-up at 24 months.
The global WOMAC score consistently improved with scores increasing from 45 at baseline to 78 at 24 months—a total improvement of 73% (figure 7).
Figure 7.
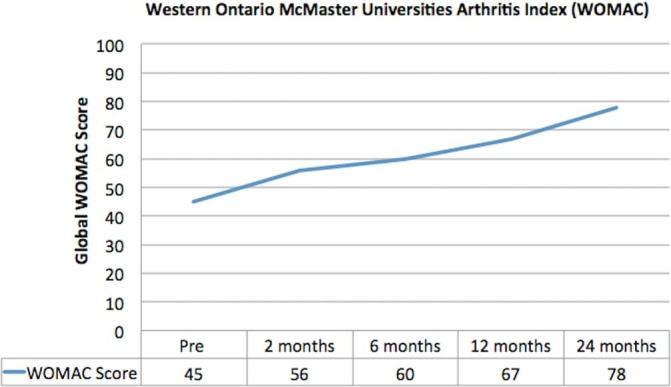
Western Ontario and McMaster Universities Arthritis Index (presented as an inverse percentage). Pain, stiffness and function improved following mesenchymal stem cell therapy.
All separate parameters of the KOOS showed improvement across the follow-up period (figure 8). At 24 months, the symptom score had improved by 100%. Quality of life as measured by KOOS improved by greater than 260%.
Figure 8.
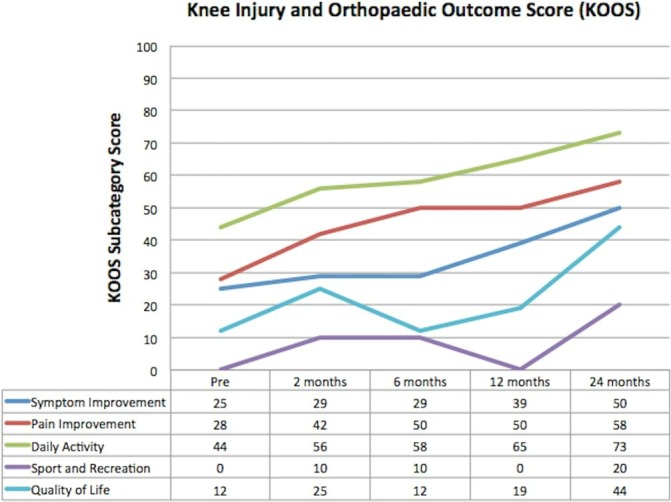
Knee Injury and Osteoarthritis Outcome Score (KOOS). All subscales of KOOS showed progressive improvement over the period of follow-up.
Structural outcome
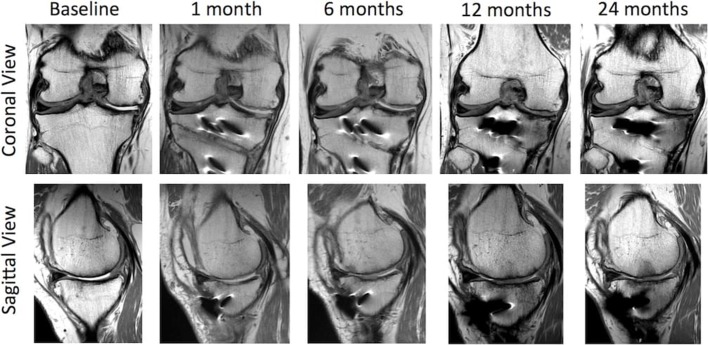
Following HTO, postoperative long leg mechanical axis measurements showed correction of the 6.8° varus angulation to 2.9°of varus angulation (figures 2 and 9). MRI performed 1 month postoperatively convincingly showed cartilage generation at the site of abrasion arthroplasty over both the medial femoral condyle and medial tibial plateau (figure 10). MRI at 12 months showed further maturation of cartilage regrowth with the modified ICRS score improving from grade IV at baseline to grades I–II at 12 months. This was maintained until completion of follow-up at 2 years. At 12 months, the average T2-weighted relaxation time values of the medial femoral condyle and medial tibial plateau were 43 ms and 39 ms, respectively (figure 11). This was comparable to areas of native cartilage which had not undergone arthroscopic abrasion. Further MRI T2 mapping at 2 years indicated progressive maturation of the area of regenerated cartilage with average T2 values of the medial femoral condyle and medial tibial plateau recorded as 40 ms and 33 ms, respectively. No regeneration of the medial meniscus (which had undergone a previous near total resection) was observed.
Figure 9.
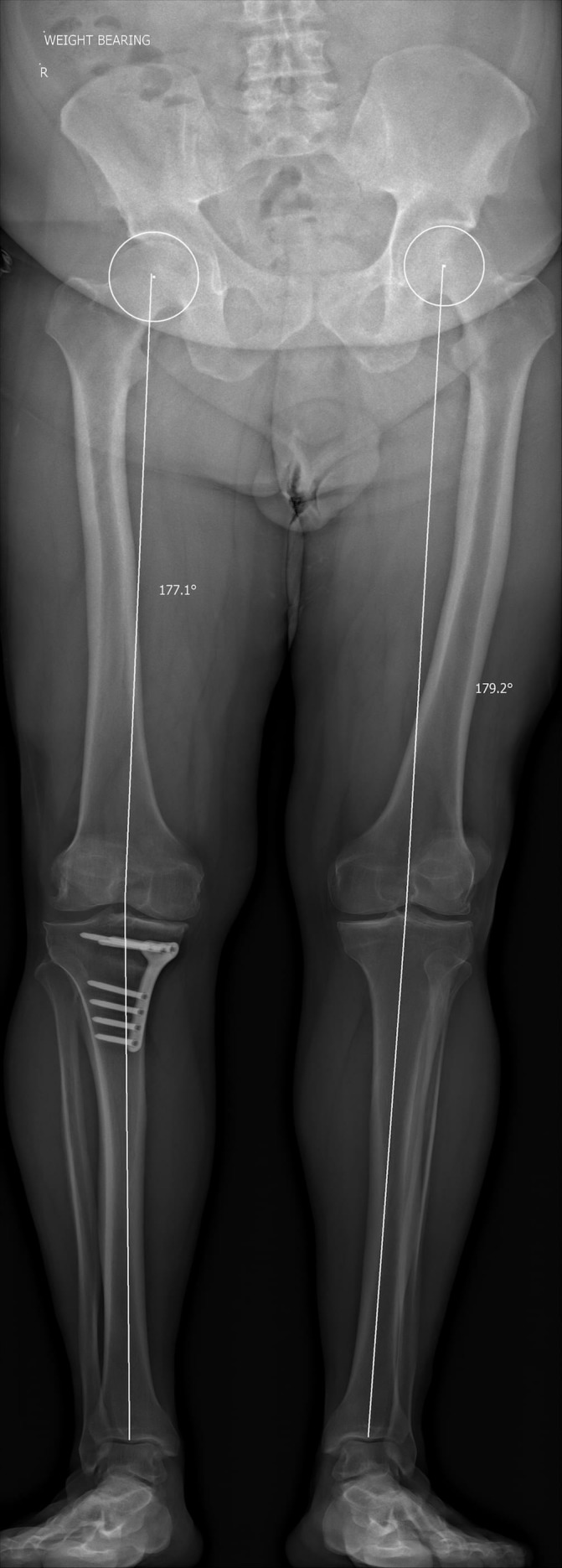
Postoperative long leg mechanical axis X-ray showing a varus knee alignment of 2.9°.
Figure 10.
Sequential proton density (PD) coronal and sagittal MRI images from baseline to 24 months showing articular cartilage regeneration at the site of chondropathology.
Figure 11.
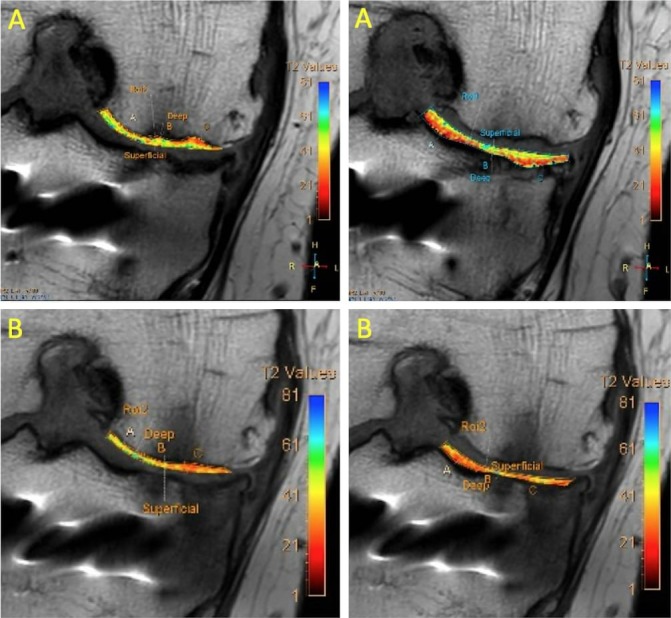
(A) MRI T2 mapping of medial compartment at 12 months. (B) MRI T2 mapping of the medial compartment showing improved values at 24 months indicating further maturation of the regenerative cartilage.
Complications and adverse events
No significant adverse event was observed throughout the period of observation. Mild bruising at the site of abdominal liposuction was observed and this resolved within 4 weeks of the harvest procedure. The patient had routine postoperative care following HTO and arthroscopic abrasion with a planned overnight stay in hospital. Pain and swelling of the knee following both the day 3 and 6 month intra-articular AdMSC injections were observed though these episodes were self-limiting, lasting 1 week, and required only simple analgesia. No complications were observed.
Discussion
This case study indicates that AdMSC therapy in combination with surgical marrow stimulation techniques (arthroscopic abrasion arthroplasty) and HTO may be both a well-tolerated, safe and effective treatment for advanced medial compartment osteoarthritis. This may be a suitable option for young patients with advanced osteoarthritis who are not suitable for consideration of other surgical interventions including total knee replacement.
Current techniques for the treatment of medial compartment osteoarthritis include HTO. HTO may delay progression to total knee replacement by 10 years with some studies showing observed cartilage regeneration with this technique in isolation.6 35 36 Cartilage regeneration, however, is typically only scattered and only offers partial coverage.35 36 The use of arthroscopic abrasion in combination with HTO has been associated with improved cartilage formation in advanced grade IV OA. Unfortunately, previous studies have indicated that cartilage formation observed in HTO and after arthroscopic abrasion contains predominantly type I collagen and is hence fibrocartilaginous.7
Previous preclinical and clinical studies have shown the ability of MSCs when combined with surgical marrow stimulation techniques (microdrilling/microfracture) to result in hyaline-like cartilage formation rather than fibrocartilage. Saw and colleagues have published a limited case series on the use of open wedge HTO in combination with arthroscopic microdrilling and peripheral blood MSCs.6 Immunohistochemical staining of biopsies at the site of cartilage formation showed an abundance of type II collagen closely resembling native articular cartilage. Using the ICRS Visual Assessment Scale II (ICRS II), two independent and blinded histopathologists graded the regenerated articular cartilage at close to 95% of the scores achieved by normal articular cartilage.
The methods used in this case study differ from that described in Saw’s previous case series as we have chosen to use adipose-derived MSCs and chose the marrow stimulation technique of arthroscopic abrasion rather than microdrilling. AdMSCs were chosen due to the ease and safety of harvest (a limited liposuction under local anaesthetic control) and their relative abundance. Previous comparative studies have shown comparable chondrogenic and osteogenic properties of AdMSCs versus bone marrow MSCs.18 19 While peripheral blood MSCs have been shown to exhibit chondrogenic and osteogenic properties, the method of collection as described by Saw using bone marrow stimulation through a granulocyte colony stimulating factor (filgastrim) followed by apheresis to isolate and collect peripheral blood MSCs is time consuming and requires hospital admission. By comparison, a lipoharvest can be performed as an outpatient day procedure.
Marrow stimulation techniques including microfracture/microdrilling are thought to allow migration of endogenous bone marrow MSCs to the site of chondropathology. Bone marrow surprisingly has a relative paucity of MSCs of between 0.001% and 0.02% of nucleated cells within bone marrow aspirate and this suggests that the mechanism of action of bone marrow stimulation may be different.19 37 In advanced osteoarthritis with eburnated bone and an abnormally thickened and sclerotic subchondral plate, the altered and relative avascular environment is further to likely limit repair potential. We theorise that the approach of arthroscopic abrasion arthroplasty and removal of the sclerotic subchondral plate to bleeding subchondral bone brings the area back to normal anatomy. Exposing bleeding subchondral bone causes the release of growth factors including TGFβ1 and bFGF which assists in the migration of stromal cells to the site of injury.27 This enhances the ability of intra-articular injected MSCs to directly hone to the area of pathology without needing to use more arduous and difficult surgical techniques such as MSC impregnated scaffolds.
Following HTO and arthroscopic abrasion arthroplasty in combination with AdMSC therapy, the subject reported pain and functional improvements as measured by validated questionnaires. Perhaps the most striking improvement was seen in the KOOS Quality of Life (QoL) score, which improved by a value of 260% from baseline to completion of follow-up at 24 months.
Sequential MRIs showed progressive chondral regeneration with earliest chondral formation observed at 1 month. Subsequent MRIs showed progressive maturation of cartilage with improvement in ICRS score from 4 to 1. MRI T2 mapping techniques showed values in both the deep and superficial layers comparable to values obtained from areas of native cartilage. Past research has confirmed a significant correlation between comparable T2 mapping values of repair tissue and native cartilage and long-term positive outcome.38 Repeat T2 mapping at 24 months did show further improvement in values indicating reduced water content and increased collagen anisotropy which supports the past observation that the process of chondrogenesis is still maturing at 24 months.39 This is not uncommon to what is found with matrix autologous chondrocyte implants that exhibit greater type II collagen and hyaline-like cartilage properties at 24 months versus 12 months of follow-up.39 While it is accepted that an arthroscopic biopsy would have provided definitive proof of cartilage histopathology, this invasive investigation was not felt to be justifiable due to the significant improvement in the patient’s symptoms.
Whether the AdMSCs migrated to the site of abrasion and differentiated along the path of a chondrocyte is unknown. Past studies have not consistently shown integration of MSCs within articular cartilage repair suggesting that the action of MSCs may be more through cell to cell contact and paracrine mechanisms.40–43 The use of cell marking techniques to study cell migration/integration would be useful in further studies.
Importantly, no serious adverse events were recorded during the follow-up period of 24 months. A self-limiting flare up in pain following each AdMSC intra-articular injection was observed though this was self-limiting and required only the use of simple analgesics. Complications not uncommon to current surgical management techniques such as autologous chondrocyte implantation—including tissue overgrowth and osseous formation—were not observed in this case study.
It is recognised that conclusions from this level IV case study are limited by the nature of it being a singular case and outcome follow-up to only 24 months. Long-term follow-up of HTO has shown an effective delay to total knee replacement by a mean of 10 years. While past studies have effectively shown fibrocartilage formation when using bone marrow stimulation techniques in combination with HTO, pain and functional results have been comparable to HTO in isolation. Furthermore, it is understood that fibrocartilage has poor weight-bearing qualities and may show early degeneration. The use of AdMSCs in combination with arthroscopic abrasion resulting in regenerative tissue with hyaline-like cartilage morphology may theoretically improve the long-term results of HTO.
In this limited case study, the use of autologous AdMSCs intra-articular injections in combination with HTO and arthroscopic abrasion arthroplasty resulted in observed pain, functional and structural improvements over a 24-month period of follow-up. The procedure was well tolerated with no serious adverse events recorded. The results suggest that this approach may be a suitable option for the treatment of advanced unicompartmental medial knee osteoarthritis in patients, where due to age or other factors, the patient is not suitable for a total knee replacement.
Learning points.
Osteoarthritis is a leading cause of pain and disability across the world.
There is an observed increase in the number of patients undergoing total knee replacement under the age of 65.
The novel use of high tibial osteotomy, in combination with arthroscopic abrasion arthroplasty and adipose-derived mesenchymal stem cell therapy, may considerably delay the need for total knee replacement.
Acknowledgments
The authors would like to acknowledge the following people for their contribution to the case study: Dr Paul Marks and Dr Chris Holden (Radiologists at Imaging Associates) for radiological analysis.
Footnotes
Patient consent for publication: Obtained.
Contributors: JF and CN were involved in the conception and design of the study. JF, CN, JW, KS and AT were involved in the drafting of the report. JF, CN, JW, KS and AT have read and approved the final manuscript.
Funding: The authors have not declared a specific grant for this research from any funding agency in the public, commercial or not-for-profit sectors.
Competing interests: JF is a partner at Magellan Stem Cells and Melbourne Stem Cell Centre and is a member of Magellan Stem Cells Clinical and Scientific Advisory Board. KS is the Chief Scientific Officer of Magellan Stem Cells and a partner at Magellan Stem Cells. AT is a partner at Magellan Stem Cells and Melbourne Stem Cell Centre.
References
- 1. Gupta S, Hawker GA, Laporte A, et al. The economic burden of disabling hip and knee osteoarthritis (OA) from the perspective of individuals living with this condition. Rheumatology 2005;44:1531–7. 10.1093/rheumatology/kei049 [DOI] [PubMed] [Google Scholar]
- 2. Issa SN, Sharma L. Epidemiology of osteoarthritis: an update. Curr Rheumatol Rep 2006;8:7–15. 10.1007/s11926-006-0019-1 [DOI] [PubMed] [Google Scholar]
- 3. Peat G, McCarney R, Croft P. Knee pain and osteoarthritis in older adults: a review of community burden and current use of primary health care. Ann Rheum Dis 2001;60:91–7. 10.1136/ard.60.2.91 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Zhuo Q, Yang W, Chen J, et al. Metabolic syndrome meets osteoarthritis. Nat Rev Rheumatol 2012;8:729–37. 10.1038/nrrheum.2012.135 [DOI] [PubMed] [Google Scholar]
- 5. Knutson K, Robertsson O. Swedish Knee Arthroplasty Registry (www.knee.se): the inside story. Acta Orthop 2010;81:5–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Spahn G, Hofmann GO, von Engelhardt LV, et al. The impact of a high tibial valgus osteotomy and unicondylar medial arthroplasty on the treatment for knee osteoarthritis: a meta-analysis. Knee Surg Sports Traumatol Arthrosc 2013;21:96–112. 10.1007/s00167-011-1751-2 [DOI] [PubMed] [Google Scholar]
- 7. Akizuki S, Yasukawa Y, Takizawa T. Does arthroscopic abrasion arthroplasty promote cartilage regeneration in osteoarthritic knees with eburnation? A prospective study of high tibial osteotomy with abrasion arthroplasty versus high tibial osteotomy alone. Arthroscopy 1997;13:9–17. 10.1016/S0749-8063(97)90204-8 [DOI] [PubMed] [Google Scholar]
- 8. Jakobsen RB, Engebretsen L, Slauterbeck JR. An analysis of the quality of cartilage repair studies. J Bone Joint Surg Am 2005;87:2232–9. 10.2106/JBJS.D.02904 [DOI] [PubMed] [Google Scholar]
- 9. Johnson LL. Arthroscopic abrasion arthroplasty historical and pathologic perspective: present status. Arthroscopy 1986;2:54–69. 10.1016/S0749-8063(86)80012-3 [DOI] [PubMed] [Google Scholar]
- 10. Magnussen RA, Dunn WR, Carey JL, et al. Treatment of focal articular cartilage defects in the knee: a systematic review. Clin Orthop Relat Res 2008;466:952–62. 10.1007/s11999-007-0097-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. LaPrade RF, Bursch LS, Olson EJ, et al. Histologic and immunohistochemical characteristics of failed articular cartilage resurfacing procedures for osteochondritis of the knee: a case series. Am J Sports Med 2008;36:360–8. 10.1177/0363546507308359 [DOI] [PubMed] [Google Scholar]
- 12. Johnson LL. Arthroscopic abrasion arthroplasty: a review. Clin Orthop Relat Res 2001;391:S306–S317. 10.1097/00003086-200110001-00028 [DOI] [PubMed] [Google Scholar]
- 13. McIlwraith CW, Frisbie DD, Rodkey WG, et al. Evaluation of intra-articular mesenchymal stem cells to augment healing of microfractured chondral defects. Arthroscopy 2011;27:1552–61. 10.1016/j.arthro.2011.06.002 [DOI] [PubMed] [Google Scholar]
- 14. Saw KY, Hussin P, Loke SC. Articular cartilage regeneration with autologous marrow aspirate and hyaluronic acid: an experimental study in a goat model. Arthroscopy 2009:25. [DOI] [PubMed] [Google Scholar]
- 15. Saw KY, Anz A, Jee CS, Cs-y J, et al. High tibial osteotomy in combination with chondrogenesis after stem cell therapy: a histologic report of 8 cases. Arthroscopy 2015;31:1909–20. 10.1016/j.arthro.2015.03.038 [DOI] [PubMed] [Google Scholar]
- 16. Wakitani S, Imoto K, Yamamoto T, et al. Human autologous culture expanded bone marrow mesenchymal cell transplantation for repair of cartilage defects in osteoarthritic knees. Osteoarthritis Cartilage 2002;10:199–206. 10.1053/joca.2001.0504 [DOI] [PubMed] [Google Scholar]
- 17. Wong KL, Lee KB, Tai BC, et al. Injectable cultured bone marrow-derived mesenchymal stem cells in varus knees with cartilage defects undergoing high tibial osteotomy: a prospective, randomized controlled clinical trial with 2 years' follow-up. Arthroscopy 2013;29:2020–8. 10.1016/j.arthro.2013.09.074 [DOI] [PubMed] [Google Scholar]
- 18. Im GI, Shin YW, Lee KB. Do adipose tissue-derived mesenchymal stem cells have the same osteogenic and chondrogenic potential as bone marrow-derived cells? Osteoarthritis Cartilage 2005;13:845–53. 10.1016/j.joca.2005.05.005 [DOI] [PubMed] [Google Scholar]
- 19. Peng L, Jia Z, Yin X, et al. Comparative analysis of mesenchymal stem cells from bone marrow, cartilage, and adipose tissue. Stem Cells Dev 2008;17:761–74. 10.1089/scd.2007.0217 [DOI] [PubMed] [Google Scholar]
- 20. Freitag J, Li D, Wickham J, et al. Effect of autologous adipose-derived mesenchymal stem cell therapy in the treatment of a post-traumatic chondral defect of the knee. BMJ Case Rep 2017;2017:bcr-2017-220852 10.1136/bcr-2017-220852 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Freitag J, Ford J, Bates D, et al. Adipose derived mesenchymal stem cell therapy in the treatment of isolated knee chondral lesions: design of a randomised controlled pilot study comparing arthroscopic microfracture versus arthroscopic microfracture combined with postoperative mesenchymal stem cell injections. BMJ Open 2015;5:e009332 10.1136/bmjopen-2015-009332 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Zuk PA, Zhu M, Mizuno H, et al. Multilineage cells from human adipose tissue: implications for cell-based therapies. Tissue Eng 2001;7:211–28. 10.1089/107632701300062859 [DOI] [PubMed] [Google Scholar]
- 23. Goh BC, Thirumala S, Kilroy G, et al. Cryopreservation characteristics of adipose-derived stem cells: maintenance of differentiation potential and viability. J Tissue Eng Regen Med 2007;1:322–4. 10.1002/term.35 [DOI] [PubMed] [Google Scholar]
- 24. Martinello T, Bronzini I, Maccatrozzo L, et al. Canine adipose-derived-mesenchymal stem cells do not lose stem features after a long-term cryopreservation. Res Vet Sci 2011;91:18–24. 10.1016/j.rvsc.2010.07.024 [DOI] [PubMed] [Google Scholar]
- 25. Dominici M, Le Blanc K, Mueller I, et al. Minimal criteria for defining multipotent mesenchymal stromal cells. The international society for cellular therapy position statement. Cytotherapy 2006;8:315–7. 10.1080/14653240600855905 [DOI] [PubMed] [Google Scholar]
- 26. Mifune Y, Matsumoto T, Takayama K, et al. The effect of platelet-rich plasma on the regenerative therapy of muscle derived stem cells for articular cartilage repair. Osteoarthritis Cartilage 2013;21:175–85. 10.1016/j.joca.2012.09.018 [DOI] [PubMed] [Google Scholar]
- 27. Zhu Y, Yuan M, Meng HY, et al. Basic science and clinical application of platelet-rich plasma for cartilage defects and osteoarthritis: a review. Osteoarthritis Cartilage 2013;21:1627–37. 10.1016/j.joca.2013.07.017 [DOI] [PubMed] [Google Scholar]
- 28. Weiss S, Hennig T, Bock R, et al. Impact of growth factors and PTHrP on early and late chondrogenic differentiation of human mesenchymal stem cells. J Cell Physiol 2010;223:84–93. 10.1002/jcp.22013 [DOI] [PubMed] [Google Scholar]
- 29. Roos EM, Roos HP, Lohmander LS, et al. Knee Injury and Osteoarthritis Outcome Score (KOOS)--development of a self-administered outcome measure. J Orthop Sports Phys Ther 1998;28:88–96. 10.2519/jospt.1998.28.2.88 [DOI] [PubMed] [Google Scholar]
- 30. Bellamy N, Buchanan WW, Goldsmith CH, et al. Validation study of WOMAC: a health status instrument for measuring clinically important patient relevant outcomes to antirheumatic drug therapy in patients with osteoarthritis of the hip or knee. J Rheumatol 1988;15:1833–40. [PubMed] [Google Scholar]
- 31. Dworkin RH, Turk DC, Farrar JT, et al. Core outcome measures for chronic pain clinical trials: IMMPACT recommendations. Pain 2005;113(1-2):9–19. 10.1016/j.pain.2004.09.012 [DOI] [PubMed] [Google Scholar]
- 32. Brittberg M, Winalski CS. Evaluation of cartilage injuries and repair. J Bone Joint Surg Am 2003;85:58–69. 10.2106/00004623-200300002-00008 [DOI] [PubMed] [Google Scholar]
- 33. Crema MD, Roemer FW, Marra MD, et al. Articular cartilage in the knee: current MR imaging techniques and applications in clinical practice and research. Radiographics 2011;31:37–61. 10.1148/rg.311105084 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Mamisch TC, Trattnig S, Quirbach S, et al. Quantitative T2 mapping of knee cartilage: differentiation of healthy control cartilage and cartilage repair tissue in the knee with unloading--initial results. Radiology 2010;254:818–26. 10.1148/radiol.09090335 [DOI] [PubMed] [Google Scholar]
- 35. Jung WH, Takeuchi R, Chun CW, et al. Second-look arthroscopic assessment of cartilage regeneration after medial opening-wedge high tibial osteotomy. Arthroscopy 2014;30:72–9. 10.1016/j.arthro.2013.10.008 [DOI] [PubMed] [Google Scholar]
- 36. Wakabayashi S, Akizuki S, Takizawa T, et al. A comparison of the healing potential of fibrillated cartilage versus eburnated bone in osteoarthritic knees after high tibial osteotomy: an arthroscopic study with 1-year follow-up. Arthroscopy 2002;18:272–8. 10.1053/jars.2002.30488 [DOI] [PubMed] [Google Scholar]
- 37. Alvarez-Viejo M, Menendez-Menendez Y, Blanco-Gelaz MA, et al. Quantifying mesenchymal stem cells in the mononuclear cell fraction of bone marrow samples obtained for cell therapy. Transplant Proc 2013;45:434–9. 10.1016/j.transproceed.2012.05.091 [DOI] [PubMed] [Google Scholar]
- 38. Jungmann PM, Brucker PU, Baum T, et al. Bilateral cartilage T2 mapping 9 years after Mega-OATS implantation at the knee: a quantitative 3T MRI study. Osteoarthritis Cartilage 2015;23:2119–28. 10.1016/j.joca.2015.06.013 [DOI] [PubMed] [Google Scholar]
- 39. Gobbi A, Karnatzikos G, Scotti C, et al. One-step cartilage repair with bone marrow aspirate concentrated cells and collagen matrix in full-thickness knee cartilage lesions: results at 2-year follow-up. Cartilage 2011;2:286–99. 10.1177/1947603510392023 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Caplan AI. Why are MSCs therapeutic? New data: new insight. J Pathol 2009;217:318–24. 10.1002/path.2469 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Jing XH, Yang L, Duan XJ, et al. In vivo MR imaging tracking of magnetic iron oxide nanoparticle labeled, engineered, autologous bone marrow mesenchymal stem cells following intra-articular injection. Joint Bone Spine 2008;75:432–8. 10.1016/j.jbspin.2007.09.013 [DOI] [PubMed] [Google Scholar]
- 42. Mokbel AN, El Tookhy OS, Shamaa AA, et al. Homing and reparative effect of intra-articular injection of autologus mesenchymal stem cells in osteoarthritic animal model. BMC Musculoskelet Disord 2011;12:259 10.1186/1471-2474-12-259 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Wu L, Leijten JC, Georgi N, et al. Trophic effects of mesenchymal stem cells increase chondrocyte proliferation and matrix formation. Tissue Eng Part A 2011;17:1425–36. 10.1089/ten.tea.2010.0517 [DOI] [PubMed] [Google Scholar]