Abstract
Rectal neuroendocrine tumors (NETs) are often found as small lesions, which can be treated by endoscopic resection. However, high risk cases with lymph node (LN) metastasis are indication of radical surgery. Furthermore, rectal NETs are often associated with late recurrences and/or multiple cancer development. Therefore, proper surgical indication and patients’ management are required. We investigated the clinicopathological features of 79 rectal NET cases in order to elucidate risk factors for synchronous LN metastasis, recurrence, and multiple cancers. Recently, we reported that in pancreatic NET patients, a loss of heterozygosity (LOH) in PHLDA3 was associated with poorer prognosis, and that LOH of both PHLDA3 and MEN1 was frequently observed. Therefore, PHLDA3 and MEN1 LOH were also assessed in rectal NET patients for their association with clinicopathological features. Of the 79 patients, LN metastases were found in 12.7%, recurrences in 3.8%, and multiple cancers in 30.4% of the subjects. PHLDA3 and MEN1 LOH were found in 60.0% and 66.7% of the subjects, respectively. Lymphatic invasion and WHO classification 2010 were found to be independent risks for LN metastasis. There were three cases of recurrence, all of which occurred more than 3 years after resection and two of which exhibited LN metastasis. Older age and LOH in PHLDA3 were associated with the presence of multiple cancers. Long-term and systemic management of patients with rectal NETs is therefore recommended in accordance with these risk factors.
Introduction
Due to the increase in colonoscopy screening, the incidence of rectal neuroendocrine tumors (NETs) has been rising, as is the incidence of other NETs throughout the body1–3. In many cases, rectal NETs are found as small lesions measuring less than 10 mm, and if they are not accompanied by lymph node metastasis, they are generally amenable to treatment by endoscopic resection. Resectable rectal NETs are associated with better prognosis than NETs in other organs. On the other hand, cases with possible lymph node metastasis may require radical surgery with lymph node dissection, which can result in anal dysfunction or permanent stoma. Therefore, accurately predicting lymph node metastasis by endoscopic and pathological examination is important. It is reported that tumor size, depth of invasion, presence of lymphovascular invasion, presence of central depression and Ki-67 index are associated with lymph node metastasis, and these factors can therefore influence the therapeutic strategy chosen2–6. On the other hand, some rectal NETs, including small lesions without vascular invasion, may have metastatic potential3,7. Therefore, further investigation is required to clarify risk factors for lymph node metastasis. Recently, we have reported that PHLDA3 is a novel tumor suppressor and loss of PHLDA3 heterozygosity is associated with poorer prognosis in pancreatic NET patients8,9. MEN1 is another tumor suppressor gene frequently inactivated in pancreatic NET, and we previously observed that double loss of heterozygosity (LOH) of PHLDA3 and MEN1 frequently occurs in pancreatic NET8,9. Identifying such markers may contribute to decisions for rectal NET treatment10. Next, although resectable rectal NETs generally have a favorable clinical course, it is reported that they also are associated with an increased risk of secondary cancers1,11. Several cases of late recurrence have been reported12,13. Therefore, an accurate clinicopathological analysis of rectal NETs based on long-term follow-up data is needed to help establish a more standardized clinical approach to treat rectal NETs. In this study, we have examined the clinicopathological features of rectal NETs for their association with synchronous lymph node metastasis, recurrence, and the occurrence of multiple cancers. These features were also examined for their association with PHLDA3 and MEN1 LOH.
Results
Patient and tumor characteristics
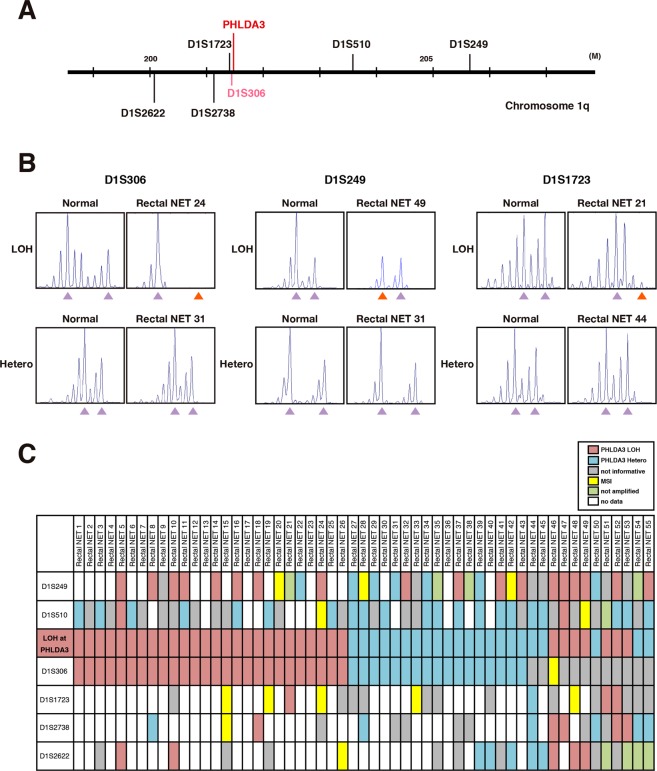
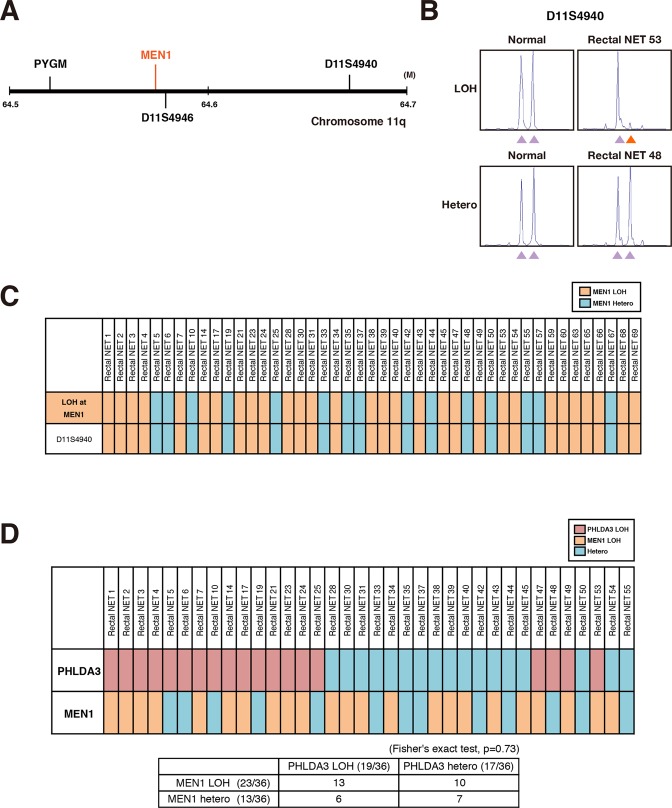
The clinicopathological features of 79 patients with rectal NET are shown in Table 1. Rectal NETs were found to occur predominantly in males (73.4%; n = 58); the mean age of the patients was 58.5 ± 12.8 years; and all patients were Asian. Of the 20 patients undergoing surgery, lymph node metastases were found in 10 and assessed as N1 (12.7%), and the tumor size averaged 9.6 ± 9.1 mm (consisting of <10 mm size in 51 patients [64.6%], 10–20 mm in 21 patients [26.6%], and >20 mm in 7 patients [8.9%]). All cases were NET G1 and G2 according to WHO classification 2010, and NET G3 were not included in this study. Lymphatic invasion was found in 13 cases (16.5%) and venous invasion in 22 cases (27.8%). We also analyzed LOH of PHLDA3 and MEN1, two tumor suppressor genes frequently inactivated in pancreatic NETs. LOH analysis of PHLDA3 was successfully performed in 55 cases, of which 60.0% (33/55) were positive, and LOH analyses of MEN1 was successful in 45 cases, of which 66.7% (30/45) were positive (Figs 1 and 2). The strikingly high incidence of LOH at these loci indicates that PHLDA3 and MEN1 are important in tumor suppression in rectal NETs. We next combined the LOH data for both loci. As shown in Fig. 2D, LOH at the PHLDA3 and MEN1 loci did not show a mutually exclusive pattern, which would be expected if PHLDA3 and MEN1 were on the same tumor suppressing pathway. Interestingly, a high frequency of double LOH, i.e. occurring at both the PHLDA3 and MEN1 loci was observed, although the frequency did not reach statistical significance (13 out of 36 samples were double-positive, p = 0.73 by Fisher’s exact test). These data suggest that the PHLDA3 and MEN1 tumor suppressing pathways are distinct, and that rectal NET development involves the functional loss of both pathways. As shown in Fig. S1, determination of LOH status was unsuccessful in 24 cases for PHLDA3 and 34 cases for MEN1. This was due to a failure in PCR amplification caused by low quality or insufficient DNA amounts (green columns), microsatellite instability (yellow columns), homozygosity (gray columns), or DNA sample was unavailable (pink columns).
Table 1.
Clinicopathological features of rectal NETs.
| Gender | Male (%) | 58 (73.4%) |
|---|---|---|
| Female (%) | 21 (26.6%) | |
| Age (years) | Mean ± SD | 58.5 ± 12.8 |
| AJCC stage | Stage I | 66 (83.5%) |
| Stage II | 3 (3.8%) | |
| Stage III | 9 (11.4%) | |
| Stage IV | 1 (1.3%) | |
| T stage | T1 | 73 (92.4%) |
| T2,3 | 6 (7.6%) | |
| N stage | N0 | 69 (87.3%) |
| N1 | 10 (12.7%) | |
| Tumor size (mm) | Mean ± SD | 9.6 ± 9.1 |
| <10 mm | 51 (64.6%) | |
| 10–20 mm | 21 (26.6%) | |
| ≥20 mm | 7 (8.9%) | |
| WHO classification 2010 | NET G1 | 72 (91.1%) |
| NET G2 | 7 (8.9%) | |
| Mitotic count (/10HPF) | <2 | 74 (93.7%) |
| 2–20 | 5 (6.3%) | |
| Ki-67 index (%) | ≤2 | 77 (97.5%) |
| 3–20 | 2 (2.5%) | |
| Lymphatic invasion | Positive | 13 (16.5%) |
| Negative | 66 (83.5%) | |
| Venous invasion | Positive | 22 (27.8%) |
| Negative | 57 (72.2%) | |
| PHLDA3 LOH | Positive | 33 (60.0%) |
| Negative | 22 (40.0%) | |
| MEN1 LOH | Positive | 30 (66.7%) |
| Negative | 15 (33.3%) | |
| Recurrence | Positive | 3 (3.8%) |
| Negative | 76 (96.2%) | |
| Disease-specific death | Positive | 2 (2.5%) |
| Negative | 77 (97.5%) | |
| Multiple cancers | Positive | 24 (30.4%) |
| Negative | 55 (69.6%) |
Abbreviations: NET, neuroendocrine tumor; HPF, high power field.
Figure 1.
Frequency of LOH at the PHLDA3 gene locus in rectal NETs. (A) Chromosomal locations of PHLDA3 gene and microsatellite markers used in this study. D1S306 is located just next to the PHLDA3 gene (32 kb upstream). (B) Representative microsatellite analysis results of PHLDA3 gene. In normal tissues, two peaks derived from maternal and paternal alleles were detected, whereas in tumors, one allele was lost or changed (shown by orange arrows), indicating LOH at the locus. (C) Microsatellite analysis of the PHLDA3 gene locus region. Rectal NET samples were analyzed for LOH around the PHLDA3 gene locus. Because D1S306, D1S1723 and D1S2738 are located near to the PHLDA3 gene, the LOH status of the PHLDA3 gene was determined from the LOH status of these markers. For samples with no data for these markers, the LOH status of the locus was determined from the surrounding LOH status. In some cases, LOH status was unavailable because of failure of PCR due to insufficiency or low quality of DNA (shown in green columns), microsatellite instability (shown in yellow columns) or homozygosity (shown in gray columns).
Figure 2.
Frequency of LOH at the MEN1 gene locus in rectal NETs. (A) Chromosomal locations of the MEN1 gene and microsatellite marker used in this study. (B) Representative microsatellite analysis results of MEN1 gene. (C) Microsatellite analysis of LOH at the MEN1 gene locus. The LOH status of the MEN1 gene was determined from the LOH status of D11S4940, D11S4946 or PYGM. (D) Relationship between LOH status of the PHLDA3 and MEN1 loci. In total, 36 samples informative for both PHLDA3 and MEN1 loci were analyzed.
Clinicopathological features associated with lymph node metastasis, recurrence, and multiple cancers
Synchronous lymph node metastases were found in 10/79 cases (12.7%). The associations between various clinicopathological features and lymph node metastasis are shown in Table 2. Both WHO classification 2010 and lymphatic vessel invasion were found to be independent risk factors for lymph node metastasis. MEN1 and PHLDA3 LOH were not associated with lymph node metastasis. The clinicopathological features of patients with lymph node metastasis are shown in Supplementary Table S1. All cases had at least one of following risk factors; tumor size >10 mm, lymphatic invasion, venous invasion, or G2 in the WHO classification 2010.
Table 2.
Comparison of characteristics between cases with and without lymph node metastasis.
| With lymph node metastasis (n = 10) (%) | Without lymph node metastasis (n = 69) (%) | Univariate | Multivariate | |
|---|---|---|---|---|
| P value | P value | |||
| Gender | ||||
| Male | 7 (70.0%) | 51 (73.9%) | NS | |
| Female | 3 (30.0%) | 18 (26.1%) | ||
| Age (years) | 57.9 ± 11.1 | 58.6 ± 13.1 | NS | |
| WHO classification 2010 | ||||
| NET G1 | 5 (50.0%) | 67 (97.1%) | 0.01> | 0.01> |
| NET G2 | 5 (50.0%) | 2 (2.9%) | ||
| Mitotic count (/10HPF) | ||||
| <2 | 6 (60.0%) | 68 (98.6%) | 0.01> | |
| 2–20 | 4 (40.0%) | 1 (1.4%) | ||
| Ki-67 index (%) | ||||
| ≤2 | 9 (90.0%) | 68 (98.6%) | 0.239 | |
| 3–20 | 1 (10.0%) | 1 (1.4%) | ||
| Tumor size | ||||
| Mean, mm | 22.3 ± 17.6 | 7.8 ± 5.3 | 0.029 | 0.06 |
| Depth of tumor invasion | ||||
| submucosa | 7 (70.0%) | 68 (98.6%) | 0.01> | |
| Muscular layer or deeper | 3 (30.0%) | 1 (1.4%) | ||
| Lymphatic invasion | ||||
| Positive | 7 (70.0%) | 6 (8.7%) | 0.01> | 0.01> |
| Negative | 3 (30.0%) | 63 (91.3%) | ||
| Venous invasion | ||||
| Positive | 8 (80.0%) | 14 (20.3%) | 0.01> | 0.21 |
| Negative | 2 (20.0%) | 55 (79.7%) | ||
| MEN1 LOH (n = 45) | ||||
| Positive | 6 (75.0%) | 24 (64.9%) | NS | |
| Negative | 2 (25.0%) | 13 (35.1%) | ||
| PHLDA3 LOH (n = 55) | ||||
| Positive | 3 (33,3%) | 30 (65.2%) | NS | |
| Negative | 6 (66.7%) | 16 (34.8%) | ||
| Recurrence | ||||
| Positive | 2 (20.0%) | 1 (1.4%) | 0.041 | |
| Negative | 8 (80.0%) | 68 (98.6%) | ||
| Multiple cancers | ||||
| Positive | 2 (20.0%) | 22 (31.9%) | NS | |
| Negative | 8 (80.0%) | 47 (68.1%) | ||
Abbreviations: NET, neuroendocrine tumor; HPF, high power field; LOH, loss of heterozygosity; NS, not significant.
The mean follow-up of the patients included in the analysis was 2136.0 ± 1472.9 days. During follow-up, recurrences occurred in 3 patients (3.1%), and 2 of these dying of the disease (Table 3). All cases were classified as G2 in the WHO classification 2010 and were shown to be positive for PHLDA3 LOH. Synchronous lymph node metastasis was found in 2 of the 3 recurrent cases. Of the 3 recurrent cases, 2 had liver metastases and 1 had a local recurrence. The time to recurrence from resection in these 3 recurrent cases was 1798, 1916, and 2595 days, respectively. And, all recurrences occurred over a 3-year period following NET resection.
Table 3.
Cases with recurrences and disease-specific death.
| Age | Gender | Resection | pT (tumor size) | pN | Lymphatic invasion | Venous invasion | WHO classification 2010 | MEN1 LOH | PHLDA3 LOH | Site of recurrence | Follow-up before recurrence | Total follow-up | Clinical outcome |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 61 | Male | Radical | 3 (55 mm) | 1 | (+) | (+) | Grade 2 | (+) | (+) | Liver | 1916 days | 5550 days | Died of disease |
| 66 | Female | Radical | 3 (55 mm) | 1 | (+) | (+) | Grade 2 | NA | (+) | Liver | 2595 days | 3162 days | Lost to follow-up |
| 70 | Male | Local | 1 (10 mm) | 0 | (−) | (−) | Grade 2 | (+) | (+) | Local | 1798 days | 3781 days | Died of disease |
Abbreviations: NA, data not available.
Table 4 and Supplementary Table S2 list the clinicopathological features of rectal NETs presenting with multiple cancers. Multiple cancers were detected in 24 of the 79 cases (30.4%). Of these, 5 cases had multiple lesions involving 2 organs (2 cases involving the stomach and colorectum, 2 involving the colorectum and lung, and 1 case involving the esophagus and stomach). Of the 29 tumors, 23 were histologically confirmed, 5 were recorded as a past history, and 1 case of pancreatic cancer was detected radiographically. Multiple colorectal cancers were found in 10 subjects, gastric cancer in 4, esophageal cancer in 3 (all squamous cell carcinoma), lung cancer in 3, prostate cancer in 2, and liver cancer in 2 subjects (1 hepatocellular carcinoma and 1 cholangiocellular carcinoma). One case involved lesions of the skin (malignant melanoma), kidney, pancreas, and breast. In addition, one gastric GIST was found. Fifteen tumors were found concurrent with rectal NETs, with 10 multiple tumors detected over 6 months before the resection of the rectal NETs. Four multiple tumors were detected over 6 months after removal of the rectal NET. The occurrence of multiple cancers was significantly associated with older age and the presence of PHLDA3 LOH, but not with any other clinicopathological feature. Importantly, 5 patients died of multiple cancers other than rectal NETs during the follow-up period.
Table 4.
Comparison of characteristics of patients with and without multiple cancers.
| Patients with multiple cancer | Patients without multiple cancer | P value | |
|---|---|---|---|
| Gender | |||
| Male | 18 (75.0%) | 40 (72.7%) | NS |
| Female | 6 (25.0%) | 15 (27.3%) | |
| Age (years) | 64.5 ± 8.7 | 55.9 ± 13.5 | 0.01> |
| 60 years old ≤ | 18 (75.0%) | 24 (43.6%) | 0.01 |
| 60 years old> | 6 (25.0%) | 31 (56.4%) | |
| pT stage | |||
| pT1 | 21 (87.5%) | 52 (94.5%) | NS |
| pT23 | 3 (12.5%) | 3 (5.5%) | |
| pN stage | |||
| pN0 | 22 (91.7%) | 47 (85.5%) | NS |
| pN1 | 2 (8.3%) | 8 (14.5%) | |
| Tumor size | |||
| Mean (mm) | 10.3 ± 11.4 | 9.3 ± 8.1 | NS |
| WHO classification 2010 | |||
| NET G1 | 22 (91.7%) | 50 (90.9%) | NS |
| NET G2 | 2 (8.3%) | 5 (9.1%) | |
| Lymphatic invasion | |||
| Positive | 4 (16.7%) | 9 (16.4%) | NS |
| Negative | 20 (83.3%) | 46 (83.6%) | |
| Venous invasion | |||
| Positive | 6 (25.0%) | 16 (29.1%) | NS |
| Negative | 18 (75.0%) | 39 (70.9%) | |
| MEN1 LOH (n = 45) | |||
| Positive | 7 (63.6%) | 23 (67.6%) | NS |
| Negative | 4 (36.4%) | 11 (32.4%) | |
| PHLDA3 LOH (n = 55) | |||
| Positive | 14 (82.4%) | 19 (50.0%) | 0.02 |
| Negative | 3 (17.6%) | 19 (50.0%) | |
| Recurrence | |||
| Positive | 1 (4.2%) | 2 (3.6%) | NS |
| Negative | 23 (95.8%) | 53 (96.4%) | |
Abbreviations: NET, neuroendocrine tumor; LOH, loss of heterozygosity; NS, not significant.
The association between LOH and various clinicopathological features is shown in Table 5. The observed frequency of PHLDA3 LOH in rectal NET (60.0%) was similar to the reported frequency in pancreatic NET (72.0%)8. Of the various clinicopathological features recorded, PHLDA3 LOH was associated only with the occurrence of multiple cancers. MEN1 LOH was found in 66.7% of the rectal NET, and was not associated with any clinicopathological features.
Table 5.
Association between PHLDA3, MEN1 LOH and clinicopathological features.
| PHLDA3 LOH (+) (%) | PHLDA3 LOH (−) (%) | P value | MEN1 LOH (+) (%) | MEN1 LOH (−) (%) | P value | |
|---|---|---|---|---|---|---|
| Gender | ||||||
| Male | 26 (78.8%) | 16 (72.7%) | NS | 24 (80.0%) | 9 (60.0%) | 0.15 |
| Female | 7 (21.2%) | 6 (27.3%) | 6 (20.0%) | 6 (40.0%) | ||
| Mean age, years | 60.0 ± 12.2 | 56.1 ± 14.1 | NS | 58.3 ± 10.1 | 51.3 ± 15.8 | NS |
| pT Stage | ||||||
| pT1 | 29 (87.9%) | 22 (100%) | NS | 28 (93.3%) | 13 (86.7%) | NS |
| pT23 | 4 (12.1%) | 0 (0%) | 2 (6.7%) | 2 (13.3%) | ||
| pN Stage | ||||||
| pN0 | 30 (90.9%) | 16 (72.7%) | NS | 24 (80.0%) | 13 (86.7%) | NS |
| pN1 | 3 (9.1%) | 6 (27.3%) | 6 (20.0%) | 2 (13.3%) | ||
| WHO classification 2010 | ||||||
| NET G1 | 30 (90.9%) | 20 (90.9%) | NS | 26 (86.7%) | 14 (93.3%) | NS |
| NET G2 | 3 (9.1%) | 2 (9.1%) | 4 (13.3%) | 1 (6.7%) | ||
| Tumor size | ||||||
| Mean (mm) | 9.5 ± 9.9 | 9.1 ± 4.7 | NS | 9.4 ± 9.7 | 10.1 ± 7.0 | NS |
| Recurrence | ||||||
| Positive | 2 (6.1%) | 0 (0%) | NS | 1 (3.3%) | 0 (0%) | NS |
| Negative | 31 (93.9%) | 22 (100%) | 29 (96.7%) | 15 (100%) | ||
| Multiple cancers | ||||||
| Positive | 14 (42.4%) | 3 (13.6%) | 0.02 | 7 (23.3%) | 4 (26.7%) | NS |
| Negative | 19 (57.6%) | 19 (86.4%) | 23 (76.7%) | 11 (73.3%) | ||
| Lymphatic invasion | ||||||
| Positive | 5 (15.2%) | 5 (22.7%) | NS | 6 (20.0%) | 2 (13.3%) | NS |
| Negative | 28 (84.8%) | 17 (77.3%) | 24 (80.0%) | 13 (86.7%) | ||
| Venous invasion | ||||||
| Positive | 9 (27.3%) | 7 (31.8%) | NS | 10 (33.3%) | 5 (33.3%) | NS |
| Negative | 24 (72.7%) | 15 (68.2%) | 20 (66.7%) | 10 (66.7%) | ||
Abbreviations: NET, neuroendocrine tumor; HPF, high power field; LOH, loss of heterozygosity; NS, not significant.
Discussion
Consistent with earlier reports, resectable rectal NETs showed a favorable clinical course in our study, with a recurrence rate of only 3.1% and a disease-specific survival of 96.9% during the mean follow-up period over 5 years. There were no recurrences in the first three years after resection, one after three years and two after five years. Of the 3 recurring cases, 2 had synchronous lymph node metastases. Therefore, long-term follow up may be especially required in patients showing synchronous lymph node metastasis. In addition, 30.4% of the patients were found to have multiple cancers other than NET, the likelihood of which was associated with older age and the presence of PHLDA3 LOH. Previous epidemiological studies have also reported an association with multiple tumors1,11. In a long-term follow-up at our institution, we also confirmed a high incidence of multiple tumors in various organs among these patients. To our knowledge, the data presented here is the first to describe the clinicopathological features of patients suffering from rectal NETs associated with multiple cancers. Many of the cases of multiple cancers were found prior to or at the same time as the diagnosis of rectal NET. In particular, our results suggest that comprehensive screening for multiple cancers is especially recommended for patients over 60 years of age. Biological markers that would predict the recurrence of NETs or the presence of multiple cancers would contribute to more efficient patient management, and PHLDA3 LOH may constitute one such marker to predict a higher probability of multiple cancers10. In this regard, higher gene methylation levels in normal tissue have been reported to be associated with metachronous gastric cancer14. While the role of PHLDA3 in carcinogenesis is less clear, higher level of epigenetic alterations in normal tissues may also be found in rectal NETs patients. It is likely that LOH of PHLDA3 in rectal NETs results in the production of some tumor promoting factors, but further study will be required to confirm this. In addition, we note that the most of multiple tumors we observed were in the colon, rectum, stomach, and these organs are differentiated from endoderm. We therefore speculate that the PHLDA3 LOH must have occurred in endoderm cell at a very early stage of the development. It is well-known that Li-Fraumeni syndrome patients, who have germline p53 mutation, also develop multiple cancers. PHLDA3 is a target gene and important down-stream mediator of p53, therefore dysfunction of PHLDA3 at an early stage of development may also contribute to multiple cancer formation.
It is nortworthy that in pancreatic endocrine tissue, PHLDA3 acts as a tumor suppressor, and methylation and/or LOH of PHLDA3 activates Akt-regulated biological processes. PHLDA3 status has also been reported to be associated with clinical outcome in pancreatic NET patients. Although we observed no association between PHLDA3 LOH and lymph node metastasis, all three cases of recurring NETs were found to have PHLDA3 LOH. Thus, further study is required to evaluate the clinical utility of PHLDA3 LOH in predicting tumor behavior. In addition, rectal NETs are not usually associated with the MEN1 syndrome, and MEN1 LOH has been thought to be rare15. However, previous reports analyzed only 1 or 2 patients, so reliable epidemiological data was not available16–18. Therefore, we investigated LOH of MEN1 as well as PHLDA3 to estimate the clinical utility of each as a biomarker. We report here the novel finding that LOH occurs at the PHLDA3 and MEN1 loci in 60.0% and 66.7% of rectal NETs cases, respectively. Using the LOH analysis method established in previous studies, we identified a slightly higher frequency of MEN1 LOH compared to PHLDA3 LOH in rectal NETs8. Thus, this is the first report to assess the frequency of LOH at the MEN1 locus and show its association with PHLDA3 LOH. Consistent with our previous report on pancreatic NETs, patients with rectal NETs frequently exhibited double LOH, which differed from matually exclusive pattern between K-RAS and BRAF mutations in colorectal cancer6. Further study will be required to confirm generality of our results from single institution. In addition, although most rectal NETs are thought to be sporadic without genetic disorder, some rectal NET with neurofibromatosis or Peutz-Jeghers syndrome has also been reported. Therefore, the molecular biological analysis of genes related to these diseases may also provide useful findings19–21.
One case showed local recurrence after trans-anal excision, thus it is important to clearly understand the parameters that might indicate radical surgery in the management of rectal NETs. Tumor size is a simple and objective measure to guide therapeutic decision proposed by the ENETS and NCCN guidelines22,23. Although there are some reported exceptions, lesions measuring < 10 mm in diameter, and without lympho-vascular invasion have been reported to generally have a low risk of metastatic disease and therefore can be resected endoscopically or by other local transanal resections3. Tumors > 20 mm, T3 or T4 stage, and having G3 grading by the ENETS guidelines or those with loco-regional lymph node involvement should indicate the need for additional surgical resection of the rectum with mesorectal lymph node dissection. On the other hand, for tumors measuring between 10 and 20 mm, the recommendations provided by the ENETS and NCCN guidelines are ambiguous, and their metastatic risk is considered to be between 10% and 15%. Our retrospective study showed that 8 of the 10 patients with lymph node metastasis had tumors measuring ≤ 20 mm in size. Five of these patients with lymph node metastasis fulfilled the criteria, including, tumor size ≤ 20 mm, NET G1 and submucosal invasion in these guidelines. These 5 cases could be observed after endoscopic resection or trans anal excision. However, 4 of these cases were shown to have had lymphatic or venous invasion in pathological examinations, which lead us undertake radical surgery. Therefore, a combined assessment of histological factors, WHO grading, lymphatic vessel invasion, and venous invasion should be used to guide the decision to undertake additional surgical resection in tumors sizes between 10 and 20 mm. Although vascular invasion is reported to be a less objective measure, the concordance of vascular invasion in pT1 colorectal cancers has reported to be 0.52–0.5624,25. Therefore, an assessment of vascular invasion in rectal NETs, most of which are small and within submucosa, may provide increased objectivity in determining a course of treatment. However, further pathological study is required to confirm this point.
In conclusion, we believe that special attention should be given to recurrences that are found more than 3 years after resection, especially in case showing synchronous lymph node metastasis. The risk for multiple cancers should be also considered especially in patients over 60 years of age and those exhibiting PHLDA3 LOH. Lymphatic invasion and the WHO classification 2010 were found to be good predictors of synchronous lymph node metastasis, and the combined use of these factors may assist in determining surgery after local resection. Rectal NET patients appear to require systemic screening before resection, as well as long-term and systemic management after resection. Biological and biomarker research focused on recurrence or multiple cancers would be beneficial for the long term management of rectal NET patients.
Materials and Methods
Informed consent
All experiments were performed after obtaining written comprehensive informed consents from all patients. This study was approved by the National Cancer Ethical Review Board (No. 2013-032), and was performed in accordance with relevant guidelines and regulations.
Patients
Using pathological data base, we enrolled 79 patients retrospectively diagnosed as rectal NET G1 and G2 according to the World Health Organization (WHO) classification 2010 between January 1, 1999 and March 31, 2014 at National Cancer Center East Hospital15. All cases were intended for curative resection. Endoscopic resections, transanal excision, and radical surgery were underwent in 54 (68.4%) cases, 5 (6.3%) cases, and 20 (25.3%) cases, respectively. Additional surgical resection of the rectum with mesorectal lymph node was considered after endoscopic resection if pathological risk factor such as a positive margin, tumor size ≥10 mm, invasion into the muscularis propria, the presence of lympatic and venous invasion or NETG2 were identified. Of the 20 patients undergoing radical resection, 5 underwent additional resection after initial endoscopic resection. All patients underwent colonoscopy and an abdominopelvic computed tomography (CT) scan to evaluate the presence of metastatic disease before resection. Regarding the postoperative course of the patient, we conducted an outpatient visit once every four months for the first two years, and once every six months for the following three years. At the time of follow-up, we conducted an examination (chest, abdomen, pelvic CT examination and blood sampling test) according to the protocol of the Japan Colon Cancer Research Group. In addition, we performed a lower gastrointestinal endoscopy in the 2nd and 5th postoperative periods. After 5 years postoperative surgery, patients’ visit was taken once a year. Clinical information were reviewed and recorded retrospectively including age, sex, method of resection used, recurrence, synchronous and metachronous multiple cancers other than neuroendocrine tumors. Multiple cancers detected 6 months before and after the diagnosis of rectal NETs were defined as synchronous lesions, with all other lesions defined as metachronous lesions11. Cancer staging was performed using the 7th edition of the American Joint Committee on Cancer (AJCC) Staging Manual26.
Histological diagnosis
All specimens were pathologically reviewed and assessed retrospectively by two investigators (K.I and M.K). Maximum tumor size was determined based on H.E. slides and neuroendocrine differentiation was assessed by positive immunohistochemical staining for chromogranin A (diluted 1:200, clone DAK-A3, Dako, Glostrup, Denmark), synaptophysin (ready to use, DAK-A3, Dako, Glostrup, Denmark), and CD56 in all cases (diluted 1:50, clone 123C3, Dako, Glostrup, Denmark). Tumor specimens were considered positive for neuroendocrine markers if more than 5% of tumor cells were stained. All tumors are positive for at least two of three neuroendocrine markers. Elastica and D2–40 staining was used in all cases to assess lymph-vascular invasion (diluted 1:200, clone D2–40, Acris, Herford, Germany). The Ki-67 and mitotic index was evaluated, and grading was performed according to the WHO classification 201015. Lymphatic invasion and venous invasion were assessed as reported previously27. Lymph node metastasis was confirmed histologically using surgical specimens, and distant metastases were evaluated either radiologically or histologically.
LOH analysis
LOH analysis of MEN1 and PHLDA3 loci were performed as reported previously8. Five 10 μm-thick slides from formalin-fixed paraffin-embedded tumors were used for LOH analysis. Six primer pairs labeled with FAM with amplified microsatellite loci were used to accurately detect LOH at the PHLDA3 locus. For the MEN1 locus, 3 primer pairs were used. Amplified PCR products were analyzed with a 3100 automated sequencer and GeneScan and Genotyper software (Applied Biosystems, Foster City, CA, USA). The genotype was determined to be heterozygous if two bands of different sizes were obtained from normal tissues. A ratio of the two peaks in tumor DNA of less than 0.7 in comparison with the corresponding ratio of the two peaks in non-tumor DNA was considered as allelic loss. Primers used in this study are shown in Supplementary Table S3.
Statistical analysis
Continuous variables, such as age and tumor size, were expressed as mean ± SD. Their association with synchronous lymph node metastases, multiple cancer, MEN1 LOH, and PHLDA3 LOH were assessed using Student’s t-test. Other non-continuous clinicopathological characteristics were assessed with χ2 and Fisher’s exact tests. Multiple logistic regression analysis was performed on independent risk factors for synchronous lymph node metastasis. All analyses were performed using statistical software Minitab (Kozo Keikaku Engineering Inc., Tokyo, Japan).
Supplementary information
Acknowledgements
This work is supported by the National Cancer Center Research and Development Fund (29-A-5) (Tokyo, Japan), partly supported by a Grant-in-Aid for Scientific Research (B) (#17H03587) (R.O. and M.K.) from the Ministry of Education, Culture, Sports, Science and Technology of Japan, Development of Innovative Research on Cancer Therapeutics (P-DIRECT)/Ministry of Education, Culture, Sports, Science and Technology of Japan (R.O. and M.K.), grant from the National Cancer Center Research and Development Fund (to R.O., 29-E-2), and Foundation for Promotion of Cancer Research in Japan (to R.O.).
Author Contributions
M.K. and Y.C.: conceived the project, performed experiments, wrote the manuscript, and finalized the figures. K.I., Y.T., D.T., S.K., K.A. and M.I.: provided samples, discussed and reviewed manuscript from clinical aspect. R.O. and AO.: conceived the project, provided financial support and approved the final manuscript.
Competing Interests
The authors declare no competing interests.
Footnotes
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Motohiro Kojima and Yu Chen contributed equally.
Electronic supplementary material
Supplementary information accompanies this paper at 10.1038/s41598-018-37707-z.
References
- 1.Modlin IM, Lye KD, Kidd M. A 5-decade analysis of 13,715 carcinoid tumors. Cancer. 2003;97:934–959. doi: 10.1002/cncr.11105. [DOI] [PubMed] [Google Scholar]
- 2.Konishi T, et al. Prognosis and risk factors of metastasis in colorectal carcinoids: results of a nationwide registry over 15 years. Gut. 2007;56:863–868. doi: 10.1136/gut.2006.109157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Kojima M et al Neuroendocrine tumor of the large intestine: clinicopathological features and predictive factors of lymph node metastasis. Front Oncol. 6, 173. 10.3389/fonc.2016.00173. eCollection (2016). [DOI] [PMC free article] [PubMed]
- 4.Federspiel BH, Burke AP, Sobin LH, Shekitka KM. Rectal and colonic carcinoids. A clinicopathologic study of 84 cases. Cancer. 1990;65:135–140. doi: 10.1002/1097-0142(19900101)65:1<135::AID-CNCR2820650127>3.0.CO;2-A. [DOI] [PubMed] [Google Scholar]
- 5.Kobayashi K, et al. Indications of endoscopic polypectomy for rectal carcinoid tumors and clinical usefulness of endoscopic ultrasonography. Dis Colon Rectum. 2005;48:285–291. doi: 10.1007/s10350-004-0765-y. [DOI] [PubMed] [Google Scholar]
- 6.Hotta K, Shimoda T, Nakanishi Y, Saito D. Usefulness of Ki-67 for predicting the metastatic potential of rectal carcinoids. Pathol Int. 2006;56:591–596. doi: 10.1111/j.1440-1827.2006.02013.x. [DOI] [PubMed] [Google Scholar]
- 7.Tsuboi K, et al. Liver metastases of a minute rectal carcinoid less than 5 mm in diameter: a case report. Hepatogastroenterology. 2004;51:1330–1332. [PubMed] [Google Scholar]
- 8.Ohki R, et al. PHLDA3 is a novel tumor suppressor of pancreatic neuroendocrine tumors. Proc Natl Acad Sci USA. 2014;111:E2404–2413. doi: 10.1073/pnas.1319962111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kawase T, et al. PH domain-only protein PHLDA3 is a p53-regulated repressor of Akt. Cell. 2009;136:535–550. doi: 10.1016/j.cell.2008.12.002. [DOI] [PubMed] [Google Scholar]
- 10.Modlin IM, Moss SF, Chung DC, Jensen RT, Snyderwine E. Priorities for improving the management of gastroenteropancreatic neuroendocrine tumors. J Natl Cancer Inst. 2008;100:1282–1289. doi: 10.1093/jnci/djn275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Tichansky DS, et al. Risk of second cancers in patients with colorectal carcinoids. Dis Colon Rectum. 2002;45:91–97. doi: 10.1007/s10350-004-6119-y. [DOI] [PubMed] [Google Scholar]
- 12.Yamaguchi T, Fujita S. A case of lateral pelvic lymph node recurrence of rectal carcinoid. Jpn J Clin Oncol. 2008;38:390. doi: 10.1093/jjco/hyn033. [DOI] [PubMed] [Google Scholar]
- 13.Promislow S, et al. Recurrence of a Thymic carcinoid tumour 15 years after resection with multiple myopericardial cardiac metastases: the role of multimodality imaging. Can J Cardiol. 2016;32:1577. doi: 10.1016/j.cjca.2016.06.008. [DOI] [PubMed] [Google Scholar]
- 14.Asada K, et al. Demonstration of the usefulness of epigenetic cancer risk prediction by a multicentre prospective cohort study. Gut. 2015;64:388–396. doi: 10.1136/gutjnl-2014-307094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Bosman T. C. F., Hruban R., Theise N. WHO Classification of Tumours of the Digestive System. 4th edn. IARC, Lyon (2010).
- 16.Toliat MR, Berger W, Ropers HH, Neuhaus P, Wiedenmann B. Mutation in the MEN I gene in sporadic neuroendocrine tumor of gastroenteropancreatic system. Lancet. 1997;350:1223. doi: 10.1016/S0140-6736(05)63453-8. [DOI] [PubMed] [Google Scholar]
- 17.Jakobovitz O, et al. Carcinoid tumor frequently display genetic abnormalities involving chromosome 11. J Clin Endocrinol Metab. 1996;81:3164–3167. doi: 10.1210/jcem.81.9.8784062. [DOI] [PubMed] [Google Scholar]
- 18.D’adda T, et al. Different patterns of 11q allelic losses in digestive endocrine tumors. Hum Pathol. 2002;33:322–329. doi: 10.1053/hupa.2002.32219. [DOI] [PubMed] [Google Scholar]
- 19.James AW, Chang L, Genshaft S, Dry SM. Coincident liposarcoma, carcinoid and gastrointestinal stromal tumor complicating type 1 neurofibromatosis: Case report and literature review. J Orthop. 2015;12(Suppl 1):S111–6. doi: 10.1016/j.jor.2014.08.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Wada K, et al. Rectal carcinoid tumor associated with the Peutz-Jeghers syndrome. J Gastroenterol. 1998;33:743–6. doi: 10.1007/s005350050166. [DOI] [PubMed] [Google Scholar]
- 21.Carty SE, et al. The variable penetrance and spectrum of manifestations of multiple endocrine neoplasia type 1. Surgery. 1998;124:1106–13. doi: 10.1067/msy.1998.93107. [DOI] [PubMed] [Google Scholar]
- 22.Ramage JK, et al. Vienna Consensus Conference participants. ENETS Consensus Guidelines Update for Colorectal Neuroendocrine Neoplasms. Neuroendocrinology. 2016;103:139–43. doi: 10.1159/000443166. [DOI] [PubMed] [Google Scholar]
- 23.National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines), Neuroendocrine Tumors, Version 3. http://www.nccn.org/professionals/physician_gls/PDF/neuroendocrine.pdf. (2017).
- 24.Harris EI, et al. Lymphovascular invasion in colorectal cancer: an interobserver variability study. Am J Surg Pathol. 2008;32:1816–1821. doi: 10.1097/PAS.0b013e3181816083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kojima M, et al. Blood and lymphatic vessel invasion in pT1 colorectal cancer: an international concordance study. J Clin Pathol. 2015;68:628–632. doi: 10.1136/jclinpath-2014-202805. [DOI] [PubMed] [Google Scholar]
- 26.Edge S. et al AJCC Cancer Staging Manual, 7th edn. Springer, New York (2010).
- 27.Kojima M, et al. Pathological diagnostic criterion of blood and lymphatic vessel invasion in colorectal cancer: a framework for developing an objective pathological diagnostic system using the Delphi method, from the Pathology Working Group of the Japanese Society for Cancer of the Colon and Rectum. J Clin Pathol. 2013;66:551–558. doi: 10.1136/jclinpath-2012-201076. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.