Abstract
RNA interference (RNAi) related pathways are essential for germline development and fertility in metazoa and can contribute to inter- and trans-generational inheritance. In the nematode Caenorhabditis elegans, environmental double-stranded RNA provided by feeding can lead to heritable changes in phenotype and gene expression. Notably, transmission efficiency differs between the male and female germline, yet the underlying mechanisms remain elusive. Here we use high-throughput sequencing of dissected gonads to quantify sex-specific endogenous piRNAs, miRNAs and siRNAs in the C. elegans germline and the somatic gonad. We identify genes with exceptionally high levels of secondary 22G RNAs that are associated with low mRNA expression, a signature compatible with silencing. We further demonstrate that contrary to the hermaphrodite germline, the male germline, but not male soma, is resistant to environmental RNAi triggers provided by feeding, in line with previous work. This sex-difference in silencing efficacy is associated with lower levels of gonadal RNAi amplification products. Moreover, this tissue- and sex-specific RNAi resistance is regulated by the germline, since mutant males with a feminized germline are RNAi sensitive. This study provides important sex- and tissue-specific expression data of miRNA, piRNA and siRNA as well as mechanistic insights into sex-differences of gene regulation in response to environmental cues.
Author summary
Environmental factors such as heat or pathogens influence living organisms, and in some cases these experiences can be passed on to the next generation. One such environmental factor is double stranded RNA that can be taken up by some animals and induce changes in gene expression via small RNA mediated silencing. Here, we use the roundworm Caenorhabditis elegans to show that the female and male germline react differently to the presence of such an environmental factor that may affect transmission to following generations. Sequencing of gonadal small RNA suggests that the underlying sex-differences affect downstream processing but not uptake. Furthermore, we describe the naturally occurring small RNA populations of female and male gonads and use genetic mutants to assign small RNAs to the two tissues of the gonad, namely the somatic gonad or the germline. These sex-and tissue-specific small RNA expression data provide an important resource for future research. In addition, this work reveals mechanistic insights into sex-differences in response to environmental cues.
Introduction
The environment can induce changes in phenotype and gene expression that persist across multiple generations [reviewed in 1]. Such intra- and trans-generational inheritance can pass both through the male and female germlines. Several studies have revealed sex-differences in transmission efficiency of heritable phenotypic changes, yet the underlying molecular mechanisms remain unknown [2–6].
In the nematode Caenorhabditis elegans, environmental cues such as starvation, viral RNA or environmental RNA can trigger heritable phenotypic changes that are transmitted via small RNA [7–9]. The mechanisms of trans-generational inheritance are best understood from studies of environmental RNA [5–7,10–12]. In C. elegans phenotypic changes induced by environmental RNAi require a double stranded RNA (dsRNA) entering the animal via a dedicated dsRNA transporter [13,14]. Thereafter, exogenous dsRNAs are processed by the conserved nuclease Dicer into ~22 nucleotide (nt) primary small interfering RNAs (siRNA) and incorporated into Argonaute proteins to form the RNA-induced silencing complex (RISC) [15,16]. This protein-RNA complex binds complementary mRNA sequences and initiates the production of secondary siRNA by RNA-dependent RNA polymerases (RdRP) (Fig 1A) [17–19]. Such secondary siRNAs are 22 nt with a 5’ guanine, thus named 22G RNAs [20]. Depending on the Argonaute, primary and secondary siRNA may trigger systemic gene silencing of complementary sequences through destabilization of mRNA or translational repression (Fig 1A) [21–23]. Once established, phenotypic changes induced by exogenous dsRNA can be transmitted over multiple generations [10,12,23,24].
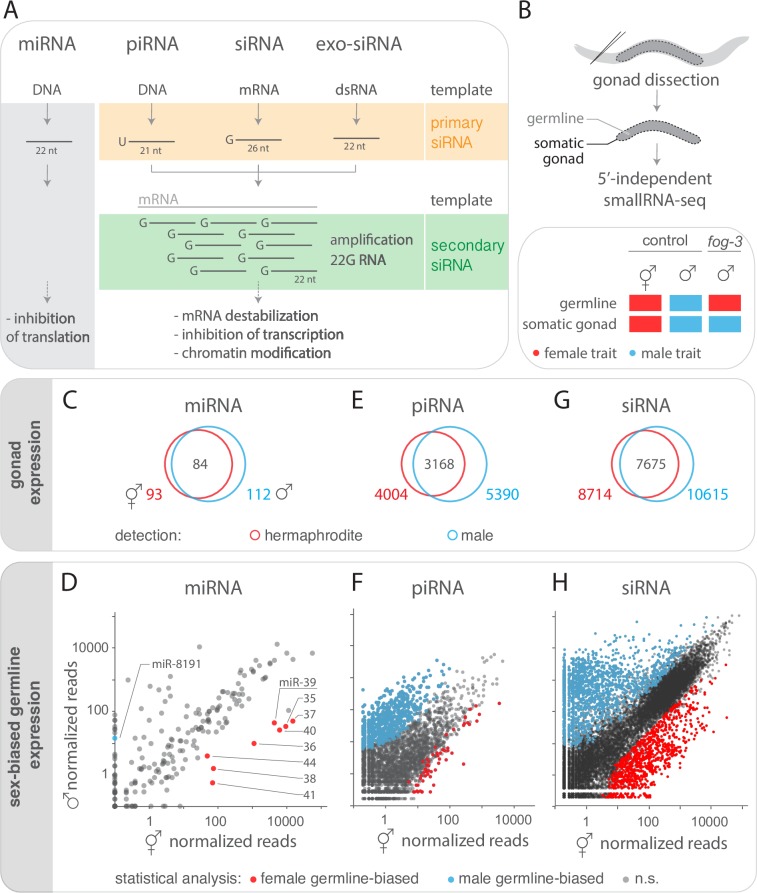
Fig 1. Tissue- and sex-specific small RNA profiling.
A) Small RNA types in C. elegans categorized by template (DNA or RNA), length and first nucleotide (nt). Distinct primary small RNA types (orange) induce the amplification of secondary 22G RNAs (green) by RNA-dependent RNA polymerase (RdRP) using mRNA as a template. B) Small RNAs were sequenced from dissected adult C. elegans gonads containing germline and somatic gonad tissue. Control hermaphrodites and males have sex-specific germline and somatic gonad tissue (red: female traits; blue: male traits). Mutant fog-3 males have a ‘female’ germline (red) and a male somatic gonad (blue). C, E, G) Gonad expression of miRNA (C), piRNA (E) and endogenous siRNA (G, >5 mean reads) in control hermaphrodites (red circle, n = 6 replicates) and males (blue circle n = 3). Total number of gonadal small RNAs in each sex is shown as well as the overlap. D, F, H) Sex-differences in expression of mean normalized miRNA (D), piRNA (F) and endogenous siRNA (H) reads in hermaphrodite (n = 6 replicates) and male gonads (n = 3). (D, H) miRNA and siRNA expression-differences with four-fold difference in abundance that were statistically different (Wilcoxon rank sum test with continuity correction; p adjusted<0.01) between hermaphrodites and males as well as between control males and fog-3 males (n = 2) are highlighted in red (female germline-biased) and blue (male germline-biased). (F) Since piRNA are germline-specific, expression-differences between hermaphrodites and males (n = 2) are highlighted in red (female germline-biased) and blue (male germline-biased) without comparison to fog-3. See S1 Fig for sex-differences in somatic gonad expression.
On top of their role in trans-generational inheritance, some endogenous small RNA types–such as miRNA, piRNA and siRNA–are essential for development and fertility in both sexes of C. elegans. Albeit distinguishable, those small RNA types share some biogenesis and silencing mechanisms (Fig 1A). One major class of small RNA, miRNAs, regulates translation of mRNA targets via partially complementary base-pairing. Many miRNAs are expressed in a tissue- and sex-specific specific manner reflecting functional sex-differences [25–28]. For instance, miR-35 family activity in the female germline is important for progeny viability and fecundity [29–31]. Furthermore, distinct Argonaute proteins associate with additional types of endogenous siRNAs in the male and female germline, suggesting functional sex-differences [20,32–36]. Importantly, germline-expressed PIWI-bound small RNA (piRNA) populations target mRNA and thus maintain genome stability in hermaphrodites and males [37–40]. Such piRNAs come in two flavours: type I piRNA are expressed from two genomic loci and have a conserved upstream motif, whereas type II piRNAs lack an apparent upstream motif and are dispersed throughout the genome [41]. In C. elegans, another group of endogenous small RNAs are expressed in the gonads of both sexes. Such siRNAs can be distinguished by length and are either (i) primary products of the RNAse III enzyme Dicer/DCR-1 (e.g., 26G RNA) or (ii) secondary products of RNA-dependent RNA polymerases (e.g., 22G RNA) (Fig 1A). Both types of endogenous siRNAs modulate gene expression and are essential for fertility, but very little is known about potential sex-differences in their gonadal expression [32,34,35].
The reproductive tissue of C. elegans consists of the germline surrounded by the somatic gonad, and development of both tissues is coordinated by multiple mechanisms [42]. Notably, miRNA activity in the somatic gonad is essential for gonad development and germline proliferation, and thus fertility [43,44]. However, a comprehensive tissue- and sex-specific expression study of gonadal siRNA is lacking.
Males and females also present germline sex-specific differences in response to environmental cues. First, RNAi in hermaphrodite gonads induces very strong knockdown phenotypes [2,45–47], in contrast to anecdotal evidence for mostly no detectable RNAi phenotypes in C. elegans sperm [6,45,48,49]. Second, siRNA induced trans-generational silencing is often more efficient through the female than the male germline [2–6]. The underlying mechanisms causing these sex-differences are not well understood.
Here we provide a comprehensive study of both the sex-determined small RNAome and environmentally-induced siRNA silencing. First, we quantify sex-biased expression of miRNA, piRNA and siRNA in isolated male and female gonads. We further ascribe small RNA sex-differences to the germline or somatic gonad by comparing gonads of each sex to mutant male gonads (fog-3) with a female germline. Quantitative analysis of gonadal expression of mRNA and 22G RNA identified genes with low mRNA expression and high 22G RNA levels, a signature compatible with silencing. Moreover, there was lower mRNA expression of WAGO than non-WAGO targets, further supporting the notion of endogenous silencing. Second, using environmental RNAi targeting a GFP-sensor we show that germline RNAi silencing efficacy is determined by germline sex, in line with previous work [6,45,48,49]. This tissue-specific sex-difference in silencing efficacy was associated with lower levels of RNAi amplification products in male than female gonads. These data provide a mechanistic basis for sex-differences in germline RNAi efficacy with implications for trans-generational inheritance.
Results
The small RNAome of male and hermaphrodite gonadal tissues
To identify small RNAs expressed in hermaphrodite and male gonads, we quantified small RNA populations (Fig 1A) from dissected C. elegans gonads by high-throughput sequencing (Fig 1B). This approach allowed us to simultaneously analyse and compare tissue-specific expression of miRNA, piRNA and siRNAs of the C. elegans gonads.
miRNAs with sex-biased germline or somatic gonad expression
Of the 257 annotated miRNAs in the C. elegans genome (assembly WS235, miRBase release 21) we detected 93 in hermaphrodite gonads and 112 miRNAs in male gonads (Fig 1C; minimum 5 mean sense reads across replicates, S1 Table). To uncover quantitative differences in miRNA expression between sexes, we compared normalized read counts between hermaphrodite and male gonads. By using a cut-off of four-fold difference in abundance and a false-discovery rate of 1%, we identified 37 miRNAs with sex-biased gonad expression (S1 Table).
Since the gonads consist of two tissue types, the germline and the somatic gonad, the observed difference between hermaphrodite and male may stem from expression differences in either tissue. To determine the contribution of the germline, we made use of mutant males that have a feminized germline and a male somatic gonad (i.e., the loss of function allele fog-3(q849[E126K]) called fog-3 or ‘feminized male’ for clarity) [50,51]. The comparison of the control male and feminized male gonads allows one to uncover differences that stem only from the germline, since both types of individuals have identical male somatic gonads (Fig 1B). Of the 37 miRNAs with sex-biased gonad expression, nine were differentially expressed between the male and feminized male gonads (Fig 1D). Eight of them (including all from the miR-35-41 cluster and miR-44) were expressed more highly in the feminized germline (fog-3 gonads) whereas only one (miR-8191) was more highly expressed in the male germline (male gonads) (Fig 1D). This result shows for the first time sex-specific expression of miRNAs in the C. elegans germline.
The comparison of hermaphrodite and feminized males allows one also to identify the contribution of the somatic gonad to differences in sex expression between hermaphrodite and male gonads, since germlines are identical (both female). This comparison revealed that 16 of the 37 miRNAs with sex-biased gonad expression were differentially expressed between the hermaphrodite and feminized gonads. One miRNA was more highly expressed in hermaphrodites, and 15 miRNAs were more highly expressed in feminized males, indicating sex-differences that stem from the somatic gonad (see S1 Fig, S1 Table).
piRNAs with sex-biased expression
Next, we compared hermaphrodite and male gonads to identify type I and II piRNAs with sex-biased expression. Our cloning technique allowed us to capture (5’-monophosphate-) piRNAs, yet at lower frequency than other types of (5’-triphosphate-) siRNA. We detected sense reads of 4004 annotated piRNAs [38,52] in hermaphrodite gonads and 5390 in male gonads, including 3168 piRNAs present in both sexes (Fig 1E, S1 Table). To uncover putative quantitative differences in piRNA expression between sexes, we compared normalized read counts between hermaphrodite and male gonads. Using a cutoff of four-fold difference in abundance and a false-discovery rate of 1%, 66 piRNAs with hermaphrodite-biased expression and 919 piRNAs with male-biased expression were revealed (Fig 1F). piRNAs are germline specific, and previous studies have identified male- and hermaphrodite-specific piRNAs by sequencing of whole animals or purified gametes [38,39,53]. We identified 93% of the previously identified male- and 32% of the female-biased piRNAs in another study [53] despite differences between the two (i.e., cell type, genotype, cloning technique and statistics). Thus, our method captures differences in piRNA expression with more sex-specific piRNAs in males than in hermaphrodites, further suggesting sex-specific regulatory mechanisms.
Endogenous siRNAs with sex-biased germline expression
As a third group of endogenous small RNA, we focused on siRNAs expressed in gonads of both sexes. We analysed all siRNAs that map to protein coding genes with a minimum of five mean antisense reads, thus excluding mRNA degradation products with sense orientation. This revealed 8714 genes with siRNAs in hermaphrodite gonads and 10615 genes in male gonads (Fig 1G). 7675 of these siRNA targeted genes were found in both sexes (Fig 1G; S1 Table). Additional expression data of small RNA (antisense and sense) mapping to other annotated features in the genome such as pseudogenes, transposons and different types of small RNA for gonads of both sexes is available as S1 Table.
To detect potential quantitative expression differences between sexes, we compared normalized antisense read counts per gene between hermaphrodite and male gonads, selecting only genes with at least a four-fold difference in siRNA abundance and a false-discovery rate of 1%. 4508 genes exhibited a sex-biased expression of siRNAs: 1138 were higher expressed in hermaphrodites and 3370 were higher expressed in males. siRNAs of 68 genes were detected exclusively in hermaphrodite and 748 only in male gonads.
To determine whether these expression differences stem from the germline or the somatic gonad, we again made use of the fog-3 mutants. Of the 4508 genes with sex-biased siRNAs expression in the gonad, 3173 were also differentially expressed between the gonads of control males and feminized males. siRNAs mapping to 2245 genes were more highly expressed in the male gonads, and 928 were more highly expressed in the feminized male gonads (Fig 1H, S1 Table). Thus, most of the sex-differences between hermaphrodite and male gonads stem from expression differences in the germline proper.
To relate sex-differences in germline siRNA expression to biological processes, we carried out gene set enrichment analysis using WormExp v1.0 [54]. This tool queries C. elegans mRNA and siRNA expression data in functional groups called ‘gene sets’ based on individual experiments for statistically significant overlap with a given gene list. We predicted that genes with sex-biased germline siRNA expression (i.e., 928 hermaphrodite- and 2245 male-biased genes) may overlap with gene sets corresponding to germline mRNA or siRNA expression. For statistical analysis, we compared enrichment to all 18106 genes with siRNAs detected (S2 Table). From the selected 2293 gene sets queried (WormExp categories: mutants, tissue, other), there was significant overlap in both sexes with multiple gene sets related to germline mRNA expression (such as down-regulation in germline-less glp-1 mutant; here and in the following comparisons FDR<0.001) and siRNA regulation (such as alteration in csr-1, rde-1 and rrf-3 mutants). In addition, this analysis revealed sex-specific overlap with several prior siRNA experiments probing siRNA pathways. Genes with hermaphrodite-biased germline siRNA expression overlapped with siRNA targets detected in whole animals, for example those regulated by rde-8, ergo-1, eri-6/-7, mut-16. Likewise, genes with male-biased germline siRNA expression overlapped with spermatogenesis specific alg-3/-4 targets. Thus, as expected, gonadal siRNAs with tissue-and sex-specific expression are involved in germline and siRNA functions, confirming the specificity of our approach.
High 22G RNA levels are associated with low mRNA expression, compatible with silencing
Since 22G RNAs of several biogenesis pathways induce gene silencing, we determined the relative abundance of 22G RNAs in the gonads of both hermaphrodites and males. 22G RNAs represented 27.4% of reads in the hermaphrodite gonads and 27.9% of reads in the male gonads (S2A Fig). The 22G RNA levels per gene were not significantly different between the male (mean reads 234±1433) and hermaphrodite gonads (173±858; two-sided Wilcoxon rank sum test p<0.19, S2B Fig). Comparison of our expression data for 22G RNA and the level of gonadal mRNA measured in a previous study [55] revealed that the vast majority of mRNAs were targeted by 22G RNAs (83% in male gonads and 81% in hermaphrodite gonads; minimum 10 reads of each RNA type). Moreover, 22G RNA levels were not consistently different between hermaphrodites and males when comparing genes with similar mRNA levels (15 of the 17 bins were not significant, and the two that were significant (both p<0.041) were among the three with the lowest mRNA levels, S2C Fig).
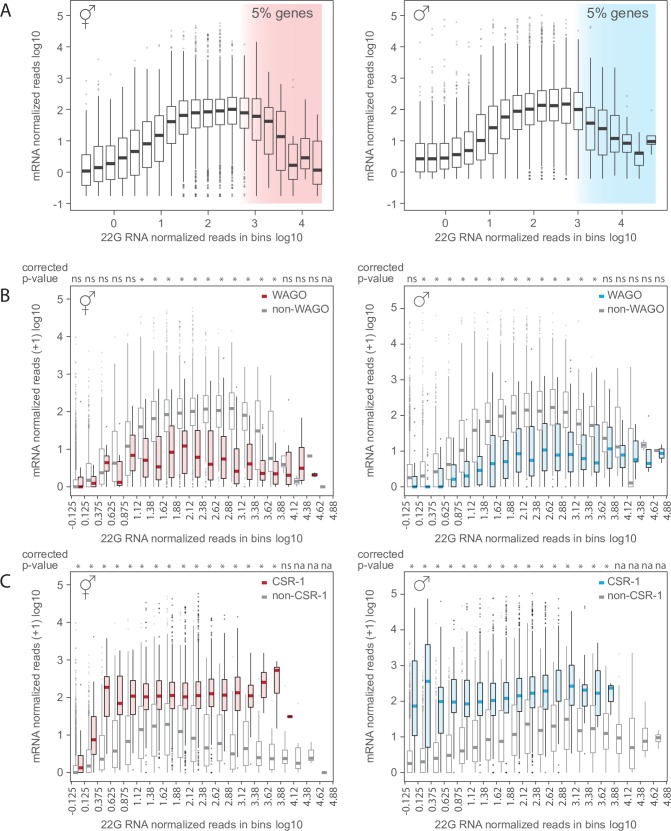
To gain insights into 22G RNA-mediated mRNA silencing, we tested if there was an association between the levels of 22G RNAs that we measured in the gonads of hermaphrodites and males and the corresponding mRNA template expression measured in a previous study [55]. We hypothesized that there should be a positive correlation between the levels of mRNA and 22G RNA if mRNA induces 22G RNA synthesis. Alternatively, if 22G RNA expression causes mRNA silencing, a negative correlation between the levels of mRNA and 22G RNA is expected. We first grouped genes in 20 bins according to their 22G RNA expression level. Across bins, there was a positive association between the levels of 22G RNA and gonadal mRNA for groups of genes with low and medium levels of 22G RNA (Fig 2A, S3 Table). In contrast, in both hermaphrodites and males, the level of mRNA decreased with extremely high 22G RNA levels (Fig 2A), a pattern suggestive of silencing. For statistical analyses we next grouped genes in 5% intervals with equal number of genes again according to 22G RNA expression (S3A Fig). The most significant negative association between the levels of 22G RNA and mRNA was found only for the 5% of the genes with highest level of 22G RNA expression (hermaphrodites p<1x10-12, males p<1x10-16; S3B Fig). These data indicate that mRNA expression is globally decreased for genes with very high levels of 22G RNAs, a signature compatible with siRNA mediated gene silencing. Because 22G RNA and mRNA engage in a feedback loop with unknown dynamics, other interpretations remain possible.
Fig 2. High 22G RNA levels are associated with low mRNA expression, compatible with silencing.
A) Mean normalized mRNA [55] and 22G RNA reads detected in hermaphrodite and male gonads (data in 20 bins according to 22G RNA level; number of genes per bin: 4–1101 in hermaphrodites, 5–1343 in males; mean: solid line, outliers: circles). mRNA expression is globally decreased for the top 5% of genes (shaded area) with high levels of 22G RNA. See S3A Fig and S3B Fig for statistical analysis. B) mRNA expression for WAGO-target genes and non-target genes (Bonferroni corrected p-values of the Wilcoxon rank sum test q<0.01 indicated by stars, ns = not significant, na = not applicable). Data are the same as for Fig 2A but plotted as WAGO targets [20] and non-WAGO targets in hermaphrodites and males. WAGO targets were available only for hermaphrodites and interpolated to male samples. C) mRNA expression for CSR-1-target genes and non-target genes (Bonferroni corrected p-values of the Wilcoxon rank sum test q<0.01 indicated by stars, ns = not significant, na = not applicable). Data are the same as for Fig 2A but plotted as CSR-1 targets and non-CSR-1 targets in hermaphrodite and male [35,56]. CSR-1 targets were available for each sex and analyzed separately.
We next asked whether 22G RNA of distinct small RNA pathways may have distinct effects on target gene regulation. To investigate the role of individual small RNA pathways, we used three datasets: published germline small RNA targets (WAGO/WAGO-1 [20], CSR-1 [35,56], ALG-3/-4 [34]), gonadal 22G RNA expression from hermaphrodites and males (this study), and gonadal mRNA expression from hermaphrodites and males [55] (see S3 Table). Using those three independent datasets, we grouped genes in 20 bins based on their 22G RNA expression, and next compared within bins the mRNA expression of Argonaute-target and non-target categories (some bins had only genes of a single category and were therefore not included in the analyses). We first analysed the WAGO/WAGO-1 dependent 22G RNAs that are involved in mRNA silencing [20]. mRNA levels were significantly lower for WAGO/WAGO-1 target genes than non-target genes in 11 of the 19 bins in hermaphrodites and 14 of the 20 bins in males (Fig 2B; S3C Fig), in line with the known function of WAGO/WAGO-1 in silencing. In addition, this analysis revealed that a wide range of 22G RNA levels mediate endogenous gene silencing in gonadal tissues (Fig 2B). Thus, genes with intermediate 22G RNA levels are also subject to silencing.
To investigate how other germline small RNA pathways may impact gonadal mRNA expression, we next analysed CSR-1 dependent 22G RNAs, which license expression of germline transcripts [36]. We found that, for both sexes, the level of mRNA expression was higher for CSR-1 targets [35,56] than for non-CSR-1 targets (significant difference in 16 bins (of 17) in hermaphrodite and 16 bins (of 16) in males, Fig 2C), a signature consistent with licensing, as shown previously [36]. Likewise, mRNA expression in males was higher for spermatogenesis specific ALG-3/-4 target genes [34] than non-target genes (14 of 14 bins; S3D Fig). In conclusion, germline-specific Argonautes CSR-1 and ALG-3/-4 do not appear to be involved in global mRNA silencing. Furthermore, these results highlights that small RNA sorting into distinct Argonaute complexes affects mRNA expression beyond 22G RNA levels.
Germline sex regulates environmental siRNA accumulation
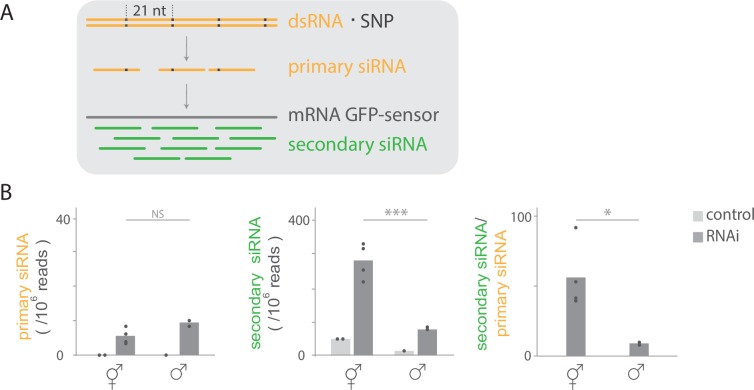
Since endogenous siRNA pathways share secondary siRNAs owing to similar biogenesis routes [17,19,32], we cannot experimentally determine the fraction of 22G RNAs induced by a specific primary siRNA type. To compare expression levels of both primary and secondary siRNA and their impact on protein expression, we developed a sensor-based assay. To this end, we used a germline expressed GFP-sensor [57] targeted by gfp(RNAi). Environmental gfp dsRNA provided by feeding allows one to manipulate primary siRNA levels and thus directly measure the impact of altered levels of primary siRNA on secondary siRNA levels. To distinguish exogenous primary siRNAs from worm-generated secondary siRNAs, we generated gfp dsRNA with single-nucleotide-polymorphisms (SNPs) every 21 nucleotides relative to the GFP-sensor transgene. Thus, primary siRNAs can be discriminated from secondary siRNAs (Fig 3A). To investigate potential sex-specific regulation of siRNA levels during exogenous RNAi, we quantified primary and secondary siRNAs by sequencing siRNAs from gonads of males and hermaphrodites expressing the GFP-sensor. Uptake and primary siRNA processing were active in both male (9 siRNA/ 106 reads) and hermaphrodite gonads (6 siRNA/ 106 reads). Moreover, primary siRNA levels were not statistically different between the sexes (t-test p<0.08, Fig 3B). By contrast, the secondary siRNA level was significantly lower in the male (82 siRNA/106 reads) than hermaphrodite gonads (280 siRNA/106 reads, t-test p<4.3x10-3, Fig 3B). Accordingly, the ratio of secondary siRNA/ primary siRNA was significantly lower in the male gonads compared to the hermaphrodite gonads (respectively, 9 and 56 secondary siRNA/primary siRNA, t-test p<0.03, Fig 3B). Taken together, we conclude that environmental dsRNA triggers primary and secondary siRNA production in the germline, with notably higher levels of secondary siRNA products in hermaphrodites than in males.
Fig 3. Sex-differences in gonadal siRNA levels during germline RNAi silencing.
A) Assay to quantify primary and secondary siRNA levels (from dissected gonads from hermaphrodites and males; see Fig 1B). Animals expressing the GFP-sensor in the germline were raised on gfp(RNAi) containing SNPs (black square) each 21 nucleotides. Primary siRNAs (orange) are identified by the presence of the SNPs, while secondary siRNAs (green) lack SNPs. B) Mean primary and secondary siRNA reads normalized to total reads in gonads from hermaphrodites (circles indicate replicates, n = 4) and males (n = 2) and mean secondary siRNA/ primary siRNA ratio (only shown for gfp(RNAi) treated animals). Animals were raised on gfp(RNAi) or control empty vector (hermaphrodite n = 2, male n = 1). Means were compared with t-tests; * = p<0.05, *** = p<0.001. Low levels of secondary siRNA are generated from the transgene in animals raised on control RNAi.
Our data do not allow one to formally ascribe primary siRNA production to either somatic gonad or germline tissue. However, several indirect lines of evidence support the presence of primary siRNAs specifically in the male germline. The GFP-sensor is expressed specifically in the germline and dsRNA feeding increases the level of 22G RNAs targeting the GFP-sensor (Fig 3B). Secondary siRNAs are not mobile [58] and therefore must be generated in the germline. Since the generation of secondary siRNAs requires primary siRNAs in the tissue of secondary siRNA production [59], we conclude that primary siRNAs are present in the male germline. Thus, uptake and transport of silencing RNA (dsRNA and/or primary siRNA) from the environment across male somatic tissues, notably the somatic gonad, is functional.
RNAi efficacy is germline sex dependent
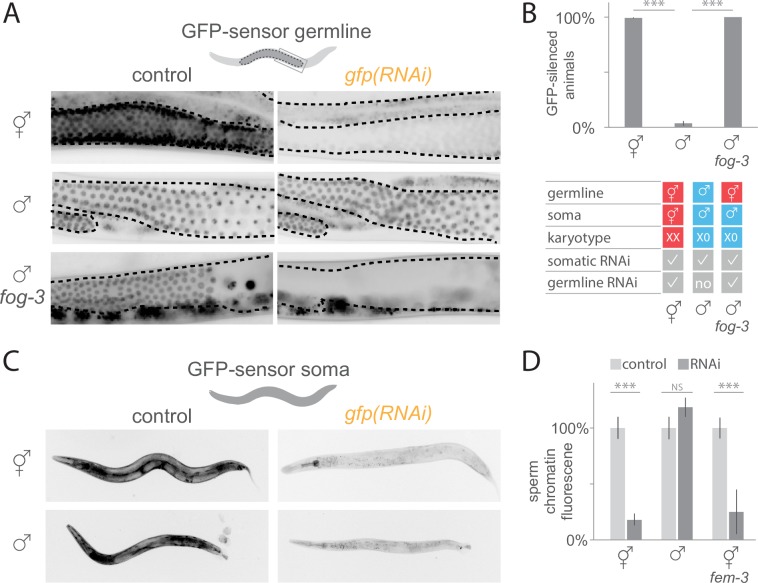
Anecdotal evidence suggests that germline RNAi silencing may differ between C. elegans hermaphrodites and males [6,45,48,49]. RNAi in hermaphrodite germlines is extremely potent as depletion phenotypes often appear within 24 hours of dsRNA exposure [47], even at reduced dsRNA dose [46]. In contrast, similar experiments generally cause suspiciously few phenotypic changes in the male germline, likely because of inefficient RNAi depletion [45,48,49]. Presumably stronger RNAi depletion occurs in male germlines directly injected with high doses of dsRNA [60], or by exposing the parental generation [61–64]. However, direct comparison of silencing efficacy of endogenous genes solely based on phenotypes in germlines of hermaphrodites and males is insufficient, notably due to sex differences in physiology and gene function. The GFP-sensor targeted by RNAi feeding allows one to measure both siRNA levels and the resulting silencing in hermaphrodites and males raised in the same environment. The difference in siRNA amplification products observed above (see Fig 3B) prompted us to ask whether germline sex affects RNAi silencing. To assess RNAi silencing in both sexes, we monitored the presence or absence of GFP-sensor fluorescence upon gfp(RNAi) (Fig 4A). These analyses revealed that germline silencing by dsRNA is sex specific. Of the 307 hermaphrodites analysed, 305 (99.4%) silenced the germline GFP-sensor in the entire germline (Fig 4B), in contrast to only 3 (3.7%) of the 81 males analysed (chi-square p<0.001; Fig 4B). This difference pertained to the entire germline of males since GFP-sensor expression was visible in proliferating germ cells and differentiated spermatocytes (Fig 4A). Contrary to the germline, the soma of males was RNAi sensitive, as evidenced by the complete silencing of a ubiquitous GFP-sensor [65] in all non-neuronal somatic cells (Fig 4C; 60/60 males). This is in line with successful RNAi-based screens targeting somatic tissues in male, notably the somatic gonad [66,67] and shows that somatic RNAi efficacy is sex independent. Thus, isogenic worms raised in the same environment show phenotypic differences in response to environmental cues. Since RNAi silencing is dependent on the dsRNA levels and because mutants defective in siRNA amplification are partly RNAi resistant [17,68–70], the observed sex-difference in secondary siRNA abundance in gonads (Fig 3B) may explain the difference between sexes in germline RNAi efficacy (Fig 4A and 4B).
Fig 4. Sex-differences in germline RNAi efficacy.
A) Fluorescence microscopy images of germline (dotted line) GFP-sensor expression in hermaphrodites, males and fog-3 males (left). Silencing of the GFP-sensor upon gfp(RNAi) in hermaphrodite and fog-3 male germlines, while control male germlines are RNAi resistant (right). Gut autofluorescence is visible outside the germline. B) Percentage of worms (±SEM) silencing the GFP-sensor in the germline upon gfp(RNAi) (n = 307 hermaphrodites, 81 males, 106 fog-3 males). Chi-square test was applied; *** = p<0.001. Table with hermaphrodite/ female (red) and male (blue) traits in hermaphrodites, males and fog-3 males. C) Fluorescence microscopy images of ubiquitous GFP-sensor expression in the soma of hermaphrodites and males (left, for both n = 54). gfp(RNAi) silencing of somatic GFP in non-neuronal cells was observed in all hermaphrodites and males (right, respectively n = 57 and n = 60). D) Background subtracted sperm GFP-sensor chromatin signal intensity (mean ±SE) upon gfp(RNAi) in hermaphrodites (n = 10), males (n = 9) and fem-3 hermaphrodites producing only sperm (n = 15). Signal intensity was normalized to animals not exposed to dsRNA (n = 10 hermaphrodites, 11 males, 12 fem-3 hermaphrodites). Note that silencing efficiency may be underestimated because quantification was based on residual chromatin fluorescence. T-test was applied for significance test; *** = p<0.001.
To determine whether the observed differences in germline RNAi silencing between male and hermaphrodites was induced by differences in the germline or the soma, we again made use of the feminized males. All other traits of the feminized males such as soma, karyotype and feeding-behaviour are undistinguishable from wild-type males (Fig 4B). Upon feeding gfp(RNAi), all 106 feminized males silenced the GFP-sensor (Fig 4A and 4B), indicating that germline-intrinsic factors regulate RNAi silencing, rather than other male traits such as the soma, karyotype or behaviour.
To test whether differences in germline RNAi silencing between control and feminized mutant male gonads could stem from altered siRNA levels or altered downstream silencing activities, for instance target slicing by Argonautes, we compared siRNA levels in gonads. The gonads of feminized males contained higher levels of primary gfp siRNAs (74/106 reads, t-test p<9.5x10-4) and secondary gfp siRNAs (798/106 reads, t-test p<0.04) than control male gonads (S4 Fig). Since RNAi silencing is dose-dependent, the higher siRNA levels in feminized mutant compared to male (or hermaphrodite) gonads are a plausible explanation for the sex-difference in germline RNAi silencing and may indicate increased uptake/ processing or stability. Exogenous siRNA levels may also depend on turnover of germline tissue: while control animals regularly release gametes containing siRNA, feminized males do not ovulate and thus may accumulate higher siRNA levels. Intriguingly, the ratio of secondary siRNA/ primary siRNA was not significantly different between control and feminized male gonads, suggesting that the soma, which is male in both types of gonads, regulates accumulation of siRNA amplification products.
Male germlines are constantly undergoing spermatogenesis, therefore we asked if sperm producing tissue is generally RNAi resistant. We tested RNAi silencing efficiency in hermaphrodite germlines in the L4 larval stage prior to and during spermatogenesis. The GFP-sensor expression was variable in animals of the same developmental stage raised in the same environment. We observed 22% of animals without GFP-sensor expression, 48% with intermediate and variable expression (stronger fluorescence intensity in proximal nuclei compared to distal nuclei), 15% with intermediate and uniform expression and 15% with high GFP expression (S5A Fig). This suggests that developing male germ cells are partially susceptible to RNAi silencing.
To further examine whether the male germline RNAi resistance was inherent to sperm or influenced by the surrounding germline environment, we quantified RNAi silencing between the sperm of males and hermaphrodites. If the germline environment regulates RNAi, silencing should differ between the sperm of males and hermaphrodites. By contrast, if the sperm physiology confers RNAi resistance, both types of sperm should be RNAi resistant. We measured fluorescence intensities of a ubiquitous GFP-sensor [71] on chromatin using both types of sperm imaged in whole worms fed with gfp(RNAi) (S5B Fig). This experiment revealed RNAi silencing in hermaphrodite sperm, since gfp(RNAi) significantly decreased sperm GFP signal intensity by a mean of 82%±5% compared to animals not exposed to dsRNA (Fig 4D; t-test p<1.8x10-6). By contrast, the sperm of males was completely RNAi resistant, with the GFP-sensor signal intensity being not significantly different between males fed with gfp(RNAi) and males not being exposed to dsRNA (Fig 4D; t-test p<0.2). The finding that, in contrast to sperm in males, sperm from hermaphrodites is RNAi sensitive, indicates that the surrounding germline environment plays a crucial role in RNAi silencing of sperm. An alternative interpretation is that intrinsic differences between the sperm produced by males and hermaphrodites may directly affect RNAi silencing [72].
Because germ cell precursors for sperm and oocytes grow in the same syncytium in hermaphrodites, silencing agents such as siRNAs or RNAi proteins produced in oocytes may diffuse in the germline and thus influence silencing in sperm. To test this hypothesis, we investigated whether mutant hermaphrodites producing only sperm initiate RNAi independently of oogenic germ cells using the gain-of-function allele fem-3(q96) [73]. We found that such sperm were RNAi sensitive, since fluorescence GFP-sensor [71] signal intensity was on average 75%±20% lower in fem-3 worms fed gfp(RNAi) compared to animals not exposed to RNAi (Fig 4D; t-test p<3.0x10-7, S5B Fig). Thus, the sperm of hermaphrodites is RNAi sensitive even in the complete absence of oogenic germ cells in the syncytium.
Sex-biased gonadal mRNA expression of RNAi genes
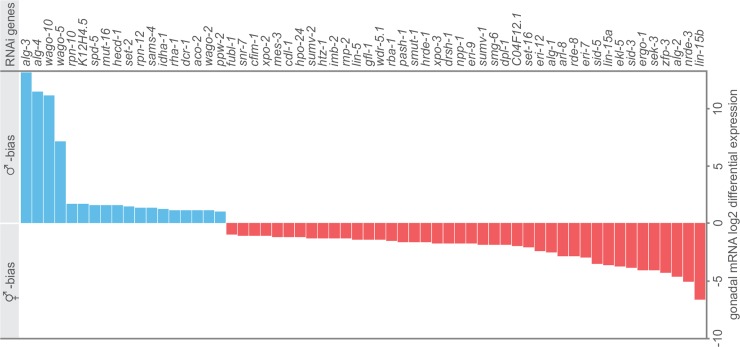
In an attempt to identify candidate genes that may regulate sex-differences in germline RNAi efficacy, we analysed mRNA expression of 178 RNAi genes in hermaphrodite and male gonads [55]. While several essential RNAi genes (e.g. rde-1, rde-2, rde-4) were not differentially expressed between sexes, 60 genes had sex-biased gonad expression using a cut-off of four-fold difference in abundance and a false-discovery rate of 1%. Forty-two RNAi genes were expressed more highly in the hermaphrodite gonads and 18 RNAi genes were expressed more highly in the male gonads (Fig 5). This candidate gene list serves as a starting point for future functional investigations of sex-differences in germline RNAi efficacy.
Fig 5. RNAi genes with sex-biased gonadal mRNA expression.
Gonadal mRNA expression data from [55]. Blue: male-biased genes, red: hermaphrodite-biased genes. We selected 178 candidate RNAi genes from WormMine based on their phenotype (classification contains “RNAi”) as well as all RNAi related genes from wormbase (such as argonautes, sid, rde, rsd and eri, see S4 Table).
Discussion
Here we provide the first comprehensive small RNA profile of isolated C. elegans male and hermaphrodite gonads and show qualitative as well as quantitative sex-differences in siRNA expression for both somatic gonad and germline tissue. We demonstrate tissue-specific sex-differences in response to environmental RNAi triggers: in contrast to the hermaphrodite germline, the male germline is resistant to silencing and accumulates lower levels of RNAi amplification products. Taken together, these results provide a mechanistic explanation for sex-differences in RNAi efficacy in response to the environment with implications for trans-generational inheritance.
Sex-differences in gonadal miRNA, piRNA and siRNA expression
miRNA regulation, notably via the conserved miR-35 family is crucial for embryonic viability, proliferation of the germline and is implicated in sex-determination [29,30,43,74]. In addition, miRNAs expressed in the C. elegans somatic gonad maintain germline proliferation and differentiation [43]. We identified female germline-biased expression of the miR35-41 cluster in isolated gonads, which is in line with studies conducted on whole animals [29,31,75].
The transgene reporter for miR-246 expression, which typically escapes germline expression, was previously observed in hermaphrodite gonadal sheath cells [25]. Here we establish sex-biased expression of endogenous miR-246 specifically in the hermaphrodite somatic gonad as opposed to the germline or the male gonad. Since miR-246 expression positively correlates with lifespan [76,77], it will be interesting to identify miR-246 targets in the somatic gonad or germline. Thus, small RNA sequencing of isolated gonads and comparison of expression differences between wild-type and sex-transformed mutant gonads captures known sex-differences in expression and in addition provides more detailed, tissue-specific information.
Our data provide the first tissue-specific analysis of miRNA expression in males. Of the 46 male-biased miRNAs with searchable names in miRBase detected in whole animals [26], 13 miRNAs showed male-biased gonad expression, underlining the importance of a tissue-specific approach. Thus, sex-biased miRNA expression occurs in both soma and gonad. Some miRNAs with male-biased gonad expression were previously not detected as sex-biased [26] and could have been masked by somatic expression in both sexes.
In contrast to miRNAs, the piRNA pathway acts exclusively in the germline. Since the germline makes up about half of the adult C. elegans cells [78], whole worm sequencing is a good proxy for germline-restricted piRNA expression. Our analysis of sex-biased piRNA expression thus complements previous studies on whole animals that reported distinct piRNA expression in hermaphrodites and males [26,39]. While the cloning method applied here causes relatively low (5’-monophasphate) piRNA detection level compared to a previous study [39], it also causes preferential cloning of 5’-triphosphate RNA species and thus high coverage of endogenous siRNA. The tissue-specific analysis of endogenous 22G RNA and mRNA identified transcripts that are potentially regulated by RNAi. Importantly, hermaphrodites and males share common as well as distinct siRNA targeted gene sets in gonadal tissue. Most gonadal sex-differences in siRNA expression stem from the germline, which represents the vast majority of the gonadal tissue. Further comparison of gonadal siRNA and corresponding mRNA uncovers uncoupling of siRNA and mRNA levels, indicative of RNAi silencing. It will be interesting to investigate this regulation in the future.
RNAi efficacy is regulated by germline sex
Our study provides novel insights into how the sex of an animal influences RNA-induced environmental changes in the phenotype. In particular, using environmental dsRNA triggers, we demonstrated that RNAi resistance affects exclusively the male germline. Sequencing primary and secondary siRNA with unique SNPs in isolated gonads from males and hermaphrodites allowed us to quantitatively compare steady-state levels. We detected similar levels of primary siRNAs in gonads of both sexes, suggesting functional transport of RNAi trigger from the environment to the soma and processing of primary siRNA in the male germline. In contrast, we discovered low levels of secondary siRNA amplification products in males compared to hermaphrodites. Thus, RNAi resistance in the male germline affects downstream processes that likely impact sex-differences in secondary siRNA processing or siRNA stability. It follows that RNAi is generally not suitable for functional studies of the C. elegans male germline if provided during one generation only. Since RNAi silencing is efficient in sperm of hermaphrodites, such animals are more suitable for reverse genetics targeting the germline.
What determines the sex-differences of gene regulation in response to environmental cues? It is conceivable that sex-differences in gonadal mRNA expression of RNAi regulators impact RNAi efficiency. In hermaphrodite gonads, RNAi genes having diverse functions–such as transport, processing or negative regulators–were expressed more highly than in male gonads. Notably, the endoribonuclease RDE-8, which is essential for amplification of siRNA products and silencing, was expressed predominantly in hermaphrodite gonads [70]. Thus, RNAi resistance in male germlines may possibly be linked to reduced RDE-8 activity.
Alternatively, competition between endogenous and exogenous RNAi pathways may cause male-specific germline RNAi resistance. Intriguingly, male-specific gonad expression affected most prominently four Argonautes. In addition to ALG-3/-4 that are implicated in spermatogenesis [34], we detected male-specific expression of WAGO-10 and WAGO-5. Although the molecular functions of WAGO-10 and WAGO-5 remain to be determined, their sex-specific expression suggests a role in male small RNA regulation.
Also, sex-differences in localization may affect RNAi efficacy. Notably CID-1, a poly(U)-polymerase modulating germline RNAi efficacy shows distinct subcellular localization in male and female germlines–perinuclear and chromosomal, respectively—that may affect function [79]. Intriguingly, in addition, multiple sex-differences are well described for the Argonaute CSR-1 that targets endogenous germline transcripts via secondary 22G RNAs [20,36]. First, males and hermaphrodites express distinct CSR-1 isoforms [36,78]. Second, csr-1 mutant hermaphrodites are mostly sterile, while csr-1 mutant males show only modestly reduced fertility over multiple generations [35,56]. Thus, sex-differences in CSR-1 function may contribute to the observed sex-difference in RNAi sensitivity, in line with CSR-1 function in germline RNAi [80]. Overall, multiple mechanisms may contribute to sex-specific gene regulation and untangling the individual contributions will provide exiting answers on male and female biology.
In conclusion, this study provides the tissue-specific small RNAome of C. elegans hermaphrodite and male gonads and identifies quantitative sex-differences in miRNA, piRNA and siRNA expression. Furthermore, we demonstrate that the male germline is resistant to RNAi triggers taken up from the environment and accumulates lower levels of RNAi amplification products. We thus provide mechanistic insights into sex-differences of gene regulation in response to environmental cues that may play a role in transgenerational inheritance.
Materials and methods
Nematode strains
C. elegans strains of the following genotype were cultured according to standard procedures [81]: him-8(e1489) IV [81], fog-3(q849[E126K]) I/ hT2[qIs48] (I;III) (kind gift from Scott Aoki and Judith Kimble) and fem-3(q96) IV [73]. The following GFP-sensors were used: single copy mjIs145 [mex-5p::GFP::his-58::21UR-1sense::tbb-2 3'UTR] II ([57]; Figs 1–3, 4A and 4B, S4 Fig and S5A Fig), multicopy adIs2122 [lgg-1p::GFP::lgg-1 3’UTR + rol-6(su1006)] [65]; Fig 4C) and multicopy zuIs178 [his-72p::his-72::GFP:his-72 3’UTR] ([71]; Fig 4D and S5B Fig). All strains were wild-type for mut-16(mg461) by PCR [82]. him-8(e1489) was used to generate otherwise wild-type males and hermaphrodites, for simplicity we refer to such animals as ‘control males’ and ‘control hermaphrodites’.
RNA interference
RNAi feeding plates where prepared from freshly streaked HT115 bacteria containing L4440 vector according to [83]. Those were used to feed synchronized L1 worms 40h at 20°C until L4 stage (S5A Fig) or minimum 48h at 20°C for adults unless otherwise stated. Initial gfp(RNAi) experiments were carried out with a construct targeting the full gfp coding sequence including three introns. The recoded gfp(RNAi) with SNP every 21 nucleotides targets only exons and was synthesized by Integrated DNA Technologies and cloned into the L4440 plasmid. Silencing efficiency of recoded gfp(RNAi) is slightly reduced compared to full length gfp(RNAi), which may be caused by SNPs or lacking introns.
Microscopy
RNAi silencing was scored in live adults mounted on slides with reaction wells (Paul Marienfeld) using 20x or 40x objective on a Zeiss Axio Scope.A1 microscope. Worms scored as RNAi sensitive on a binary scale if GFP-sensor was absent or greatly diminished as compared to animals treated with empty vector RNAi. Differential interference contrast (DIC) and fluorescence images were acquired on a Axiocam 506 mono CCD camera and processed with Fiji software [84]. Outlines of the germlines where drawn on the DIC image in Adobe Illustrator.
Gonad isolation and replicates
Bleach-synchronized L1 worms were grown on RNAi bacteria 48h at 20°C until L4/ young adult, then washed 5 times in 15mL M9 to remove bacteria and grown overnight at 20°C on OP50 bacteria. Prior dissection, adults were picked on empty plates and transferred in groups of 3–4 in a drop of 0.01% levamisole + sperm buffer [85] to wash and paralyze animals. Animals were cut with a 21G needle behind the pharynx to liberate the gonad in a new drop of buffer on a depression slide (cavity 15-18mm, depth 0.6–0.8 mm; Marienfeld). Non-gonadal somatic tissue was removed, including the intestine; the spermatheca was removed from hermaphrodite gonads. Using a mouth pipette with glass capillary gonads where transferred to a tube with RNAlater (50–100μl) and after TRIzol (Thermo Fisher) addition frozen at -80°C.
Gonads from hermphrodites (100), males or fog-3 males (each 200) were processed as independent replicates for sequencing as follows (# replicates; including # recoded gfp(RNAi) samples): hermaphrodites (6;4), males (3;2) and fog-3 males (2;2). Since endogenous siRNAs levels were not different from germlines treated with or without gfp(RNAi), both were used for sex-biased expression analysis.
RNA extraction
RNA was extracted with 1 ml of TRIzol according to manufactor’s protocol. Briefly, RNA was precipitated adding 1 volume of isopropanol and 20 μg glycogen (Roche). Samples forming a white precipitate at this point were cleared by addition of 500 μL isopropanol: water 50% (v/v). Samples were frozen at -80°C, thawed on ice and RNA was pelleted by centrifugation for 30 min at +4°C, 16000 g. The pellet was washed in ice cold 70% (v/v) ethanol and recovered by centrifugation for 20 min at +4°C, 16000 g and finally resuspended in 10 μL water. RNA concentration was determined by Qubit RNA BR Assay Kit (Thermo Fisher).
5’ independent library preparation and sequencing
For RNA dephosphorylation 400 ng of RNA were treated with 20 Units of 5’ polyphosphatase (Epicenter) in 20 μL reaction volume, purified using acid-phenol-chloroform, pH 4.5 (Thermo Fisher) and isopropanol precipitated using 20 μg of glycogen. Subsequently, RNA was suspended in 6 μL water and directly used for TruSeq Small RNA library kit (Illumina) following the manufacturer’s instructions with exception that 15 cycles of PCR amplification were used. The cDNA libraries were separated on 6% TBE PAGE gels (Life Technologies) and bands with 147–157 nucleotides were cut from the gel. The gel matrix was broken by centrifugation through gel breaker tubes (IST Engineering Inc.) and size-selected cDNA eluted with 400 μl of 0.3M Na-Chloride. cDNA was purified by centrifugation through Spin-X 0.22μm cellulose acetate filter columns (Costar) followed by isopropanol precipitation. Libraries were sequenced on a HiSeq 1500 Sequencer (Illumina).
Computational analysis of small RNA high-throughput sequencing data
Small RNA sequencing results were obtained from https://basespace.illumina.com/ as fastq files after demultiplexing. Sequencing data is available in the European Nucleotide Archive under accession number PRJEB12010 (S5 Table). Firstly, adapter sequences, reads shorten than 21 nt and reads longer than 34 nt were removed using cutadapt v1.9.1. Secondly, the remaining reads were aligned using bowtie 1.1.2 with at most 2 mismatches to Escherichia coli str. K-12 MG1655. Thirdly, the remaining reads were aligned with at most 2 mismatches to C. elegans WS235 tRNAs and rRNAs. Fourthly, the remaining reads were aligned with no mismatches to the worm gfp sequence. Fifthly, the remaining reads were aligned with no mismatches to the recoded gfp sequence. Sixthly, the remaining reads were aligned with no mismatches to C. elegans miRNAshairpins (miR-base release 21). Seventhly, the remaining reads were aligned with no mismatches to C. elegans genome cel235. Overall, we obtained ~2.3–12.8 million trimmed reads mapping to the genome (WS235) for each sex. endo-siRNAs was quantified per gene, antisense reads mapping to coding exons, which account for more than 96% of reads in this class.
Silencing of mRNA targeted by very high levels of 22G RNA
Fastq-mcf was used to trim adapters from single-end reads [86]. Bowtie 1.1.2 (parameters “—best—strata—tryhard -m 1”) was used to map single-ended reads to C. elegans WS250 genome [87]. Reads were classified as 22G/26G based on length and starting base. HTSeq 0.6.0 was used to generate counts for reads that map to WS250 features in the sense and antisense directions [88]. Reads mapping against transposons were retrieved using c_elegans.PRJNA13758.WS250.annotations.exons.genes.with_transposons.gff. Features counts from separate samples were normalized and differences in expression were determined using DESeq2 1.13.8 [89]. Because alternative biological processes generate sense and antisense mapping reads, reads mapping in the sense direction from each library were normalized together, likewise reads mapping in the antisense direction were separately normalized. The cutoff for sex-biased expression was >4 fold difference in abundance and adjusted p-value <0.01. For correlations with gonad transcriptome and differential expression of RNAi genes, we used data from [55] (Sequence Read Archive, accession number SRP096640). For miRNAs both star and non-star sequences from the same gene were summed.
Assay and quantification of primary and secondary gfp siRNA levels: Reads were trimmed with cutadapt v1.9.1 [90]. Bacteria, tRNA and rRNA reads contaminations were removed using bowtie 1.1.2 alignment to the Escherichia coli str. K-12 MG1655 and to C. elegans WS235 tRNAs and rRNAs [87]. Remaining reads were aligned with no mismatches to the worm gfp sequence. Then the remaining reads were aligned with no mismatches to the recoded gfp sequence.
Supporting information
A) Mean normalized miRNA reads (sense) in hermaphrodite (n = 6 replicates) and male gonads (n = 3). miRNA expression-differences with four-fold difference in abundance that were statistically different (Wilcoxon rank sum test with continuity correction; p adjusted<0.01) between gonads of hermaphrodites and males as well as between hermaphrodites and fog-3 males (n = 2) are highlighted in red (female somatic gonad-biased) and blue (male somatic gonad-biased). Related to Fig 1D. B) Mean normalized endogenous siRNA reads (antisense) in hermaphrodite and male gonads; same analyses and representation used as in S1A Fig. Related to Fig 1H.
(TIF)
A) Percentage of antisense reads in hermaphrodite and male gonads classified as primary 26G RNA, secondary 22G RNA or other siRNA types. B) Normalized mean 22G RNA reads in hermaphrodite and male gonads per gene, median dotted line, mean full line. C) Mean 22G RNA reads targeting mRNAs of similar abundance in hermaphrodite (red) and male (blue) gonads. Boxes represent first and second quartile, 1.5 interquartile range: vertical line, mean: horizontal line, outliers: circles. Bonferroni corrected p-value of Wilcoxon rank sum test give the probability that samples from a continuous distribution have equal medians.
(TIF)
A) Mean normalized mRNA reads [55] and siRNAs in percentiles in hermaphrodite and male gonads. For 20 bins (each representing 5% of data i.e. 427 genes per bin in hermaphrodites and 504 in males) a linear model was fitted (black line). Related to Fig 2A. B) p-values indicate whether the linear model deviates from zero. The most significant negative correlation between the levels of 22G RNA and mRNA was found for the 5% of the genes with highest level of 22G RNA expression (shaded). C) mRNA expression for WAGO-1 target genes and non-target genes (Bonferroni corrected p-values of the Wilcoxon rank sum test q<0.01 indicated by stars, ns = not significant, n/a = not applicable). Data are the same as for Fig 2A but plotted as WAGO-1 targets [20] and non-WAGO-1 targets in hermaphrodites and males. Data were only available for hermaphrodites and interpolated to male samples. D) mRNA expression for ALG-3/-4 target genes and non-target genes (Bonferroni corrected p-values of the Wilcoxon rank sum test q<0.01 indicated by stars, ns = not significant, n/a = not applicable). Data are the same as for Fig 2A but plotted as ALG-3/-4 targets [34] and non-ALG-3/-4 targets in males.
(TIF)
Mean primary and secondary siRNA reads normalized to total reads in gonads from fog-3 males (n = 2), control hermaphrodites (n = 4) and control males (n = 2) raised on gfp(RNAi). Data for control hermaphrodites and control males are taken from Fig 3B. The right panel shows the ratio of secondary siRNA/ primary siRNA. Means were compared with t-tests w* = p<0.05, *** = p<0.001.
(TIF)
A) Fluorescence microscopy images of GFP-sensor expression of L4 hermaphrodite germlines (dotted line, distal end indicated by star); animals fed (gfp)RNAi or control RNAi. Percentage of animals with germline expression classified as: high, intermediate uniform, intermediate variable and no GFP-sensor expression (from bottom to top) for (gfp)RNAi (n = 27). 100% of animals raised on control RNAi showed high GFP-sensor expression (n = 24). B) Fluorescence microscopy images of sperm chromatin marked with a ubiquitous GFP-sensor in adult hermaphrodites, males and fem-3 hermaphrodites producing only sperm (top). Partial silencing of the GFP-sensor upon gfp(RNAi) at 24°C in control and fem-3 hermaphrodites, while control male germlines are RNAi resistant (bottom). Images were acquired with the same exposure time in control and RNAi condition, but are different between sexes due to different expression levels and physiology. For illustration purposes images are background subtracted, as explained in Fig 4D.
(TIF)
Analyses of sex-biased miRNA, piRNA, germline siRNA, sense and antisense reads.
(XLSX)
WormExp input and output.
(XLSX)
Expression levels of 22G RNA and mRNA and classification as WAGO, WAGO-1, CSR-1, ALG-3/-4.
(XLSX)
(XLSX)
Accession and genotype of each replicate.
(XLSX)
Acknowledgments
We are grateful to Scott Aoki and Judith Kimble for sharing strains prior to publication, Benita Wolf, Adria LeBoeuf, Tamara Mikeladze-Dvali, Nicolas Bologna, David Jordan and two anonymous reviewers for comments on the manuscript and the Gönczy and Miska lab members for advice. We thank Kay Harnish of the Gurdon Institute Sequencing Facility for managing the high-throughput sequencing, Coralie Busso and Nazife Bega for assistance and Wormbase. Some strains were provided by the CGC, which is funded by NIH Office of Research Infrastructure Programs (P40 OD010440).
Data Availability
Small RNA library sequencing data is available on the European Nucleotide Archive in the Study: PRJEB12010. A sample description can be found in additional file 3.
Funding Statement
This work was funded by grants from the Swiss National Science Foundation and an advanced European Research Council grant to LK, grants from Cancer Research UK (C13474/A18583, C6946/A14492) and the Wellcome Trust (104640/Z/14/Z, 092096/Z/10/Z) to EAM, and grants from the National Institutes of Health to SW (NIGMS NHRA 5F32GM100614) and to FP and KCG (NHGRI U01 HG004276, NICHD R01 HD046236), and by research funding from New York University Abu Dhabi to FP and KCG. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1.Miska EA, Ferguson-Smith AC. Transgenerational inheritance: Models and mechanisms of non-DNA sequence-based inheritance. Science. 2016. pp. 59–63. 10.1126/science.aaf4945 [DOI] [PubMed] [Google Scholar]
- 2.Grishok A, Tabara H, Mello CC. Genetic requirements for inheritance of RNAi in C. elegans. Science. 2000;287: 2494–2497. 10.1126/science.287.5462.2494 [DOI] [PubMed] [Google Scholar]
- 3.Luteijn MJ, van Bergeijk P, Kaaij LJT, Almeida MV, Roovers EF, Berezikov E, et al. Extremely stable Piwi-induced gene silencing in Caenorhabditis elegans. EMBO J. 2012;31: 3422–3430. 10.1038/emboj.2012.213 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Shirayama M, Seth M, Lee HC, Gu W, Ishidate T, Conte D, et al. PiRNAs initiate an epigenetic memory of nonself RNA in the C. elegans germline. Cell. 2012;150: 65–77. 10.1016/j.cell.2012.06.015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Schott D, Yanai I, Hunter CP. Natural RNA interference directs a heritable response to the environment. Sci Rep. 2014;4: 7387 10.1038/srep07387 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Marré J, Traver EC, Jose AM. Extracellular RNA is transported from one generation to the next in Caenorhabditis elegans. Proc Natl Acad Sci. 2016;113: 12496–12501. 10.1073/pnas.1608959113 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Fire A, Xu S, Montgomery MK, Kostas SA, Driver SE, Mello CC. Potent and specific genetic interference by double-stranded RNA in Caenorhabditis elegans. Nature. 1998;391: 806–811. 10.1038/35888 [DOI] [PubMed] [Google Scholar]
- 8.Rechavi O, Minevich G, Hobert O. Transgenerational Inheritance of an Acquired Small RNA-Based Antiviral Response in C. elegans. Cell. 2011;147: 1248–1256. 10.1016/j.cell.2011.10.042 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Jobson MA, Jordan JM, Sandrof MA, Hibshman JD, Lennox AL, Baugh LR. Transgenerational Effects of Early Life Starvation on Growth, Reproduction, and Stress Resistance in Caenorhabditis elegans. Genetics. 2015;201: 201–12. 10.1534/genetics.115.178699 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Vastenhouw NL, Brunschwig K, Okihara KL, Muller F, Tijsterman M, Plasterk RHA. Gene expression: long-term gene silencing by RNAi. Nature. 2006;442: 882 10.1038/442882a [DOI] [PubMed] [Google Scholar]
- 11.Alcazar RM, Lin R, Fire AZ. Transmission dynamics of heritable silencing induced by double-stranded RNA in Caenorhabditis elegans. Genetics. 2008;180: 1275–1288. 10.1534/genetics.108.089433 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Lev I, Seroussi U, Gingold H, Bril R, Anava S, Rechavi O. MET-2-Dependent H3K9 Methylation Suppresses Transgenerational Small RNA Inheritance. Curr Biol. 2017;27: 1138–1147. 10.1016/j.cub.2017.03.008 [DOI] [PubMed] [Google Scholar]
- 13.Winston WM, Molodowitch C, Hunter CP. Systemic RNAi in C. elegans requires the putative transmembrane protein SID-1. Science. 2002;295: 2456–9. 10.1126/science.1068836 [DOI] [PubMed] [Google Scholar]
- 14.Winston WM, Sutherlin M, Wright AJ, Feinberg EH, Hunter CP. Caenorhabditis elegans SID-2 is required for environmental RNA interference. Proc Natl Acad Sci. 2007;104: 10565–10570. 10.1073/pnas.0611282104 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Tabara H, Yigit E, Siomi H, Mello CC. The dsRNA binding protein RDE-4 interacts with RDE-1, DCR-1, and a DExH-box helicase to direct RNAi in C. elegans. Cell. 2002;109: 861–71. [DOI] [PubMed] [Google Scholar]
- 16.Parker GS, Eckert DM, Bass BL. RDE-4 preferentially binds long dsRNA and its dimerization is necessary for cleavage of dsRNA to siRNA. RNA. 2006;12: 807–18. 10.1261/rna.2338706 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Sijen T, Fleenor J, Simmer F, Thijssen KL, Parrish S, Timmons L, et al. On the Role of RNA Amplification in dsRNA-Triggered Gene Silencing. Cell. 2001;107: 465–476. 10.1016/S0092-8674(01)00576-1 [DOI] [PubMed] [Google Scholar]
- 18.Sijen T, Steiner FA, Thijssen KL, Plasterk RHA. Secondary siRNAs Result from Unprimed RNA Synthesis and Form a Distinct Class. Science. 2007;315: 244–247. 10.1126/science.1136699 [DOI] [PubMed] [Google Scholar]
- 19.Pak J, Fire A. Distinct Populations of Primary and Secondary Effectors During RNAi in C. elegans. Science. 2007;315: 241–244. 10.1126/science.1132839 [DOI] [PubMed] [Google Scholar]
- 20.Gu W, Shirayama M, Conte D, Vasale J, Batista PJ, Claycomb JM, et al. Distinct Argonaute-Mediated 22G-RNA Pathways Direct Genome Surveillance in the C. elegans Germline. Mol Cell. 2009;36: 231–244. 10.1016/j.molcel.2009.09.020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Grishok A, Sinskey JL, Sharp PA. Transcriptional silencing of a transgene by RNAi in the soma of C. elegans. Genes Dev. 2005;19: 683–696. 10.1101/gad.1247705 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Guang S, Bochner AF, Burkhart KB, Burton N, Pavelec DM, Kennedy S. Small regulatory RNAs inhibit RNA polymerase II during the elongation phase of transcription. Nature. 2010;465: 1097–101. 10.1038/nature09095 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Buckley BA, Burkhart KB, Gu SG, Spracklin G, Kershner A, Fritz H, et al. A nuclear Argonaute promotes multigenerational epigenetic inheritance and germline immortality. Nature. 2012;489: 447–51. 10.1038/nature11352 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Devanapally S, Ravikumar S, Jose AM. Double-stranded RNA made in C. elegans neurons can enter the germline and cause transgenerational gene silencing. Proc Natl Acad Sci U S A. 2015;112: 2133–8. 10.1073/pnas.1423333112 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Martinez NJ, Ow MC, Reece-Hoyes JS, Barrasa MI, Ambros VR, Walhout AJM. Genome-scale spatiotemporal analysis of Caenorhabditis elegans microRNA promoter activity. Genome Res. 2008;18: 2005–15. 10.1101/gr.083055.108 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Kato M, de Lencastre A, Pincus Z, Slack FJ. Dynamic expression of small non-coding RNAs, including novel microRNAs and piRNAs/21U-RNAs, during Caenorhabditis elegans development. Genome Biol. 2009;10: R54 10.1186/gb-2009-10-5-r54 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Alberti C, Manzenreither RA, Sowemimo I, Burkard TR, Wang J, Mahofsky K, et al. Cell-Type specific sequencing of microRNAs from complex animal tissues. Nat Methods. 2018;15: 283–289. 10.1038/nmeth.4610 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Diag A, Schilling M, Klironomos F, Ayoub S, Rajewsky N. Regulation of spatial and temporal gene expression in an animal germline. bioRxiv. Cold Spring Harbor Laboratory; 2018; 348425 10.1101/348425 [DOI] [Google Scholar]
- 29.Alvarez-Saavedra E, Horvitz HR. Many Families of C. elegans MicroRNAs Are Not Essential for Development or Viability. Curr Biol. 2010;20: 367–373. 10.1016/j.cub.2009.12.051 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.McJunkin K, Ambros V. The embryonic mir-35 family of microRNAs promotes multiple aspects of fecundity in Caenorhabditis elegans. G3. 2014;4: 1747–54. 10.1534/g3.114.011973 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.McEwen TJ, Yao Q, Yun S, Lee C-Y, Bennett KL. Small RNA in situ hybridization in Caenorhabditis elegans, combined with RNA-seq, identifies germline-enriched microRNAs. Dev Biol. 2016;418: 248–257. 10.1016/j.ydbio.2016.08.003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Han T, Manoharan AP, Harkins TT, Bouffard P, Fitzpatrick C, Chu DS, et al. 26G endo-siRNAs regulate spermatogenic and zygotic gene expression in Caenorhabditis elegans. Proc Natl Acad Sci U S A. 2009;106: 18674–9. 10.1073/pnas.0906378106 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Vasale JJ, Gu W, Thivierge C, Batista PJ, Claycomb JM, Youngman EM, et al. Sequential rounds of RNA-dependent RNA transcription drive endogenous small-RNA biogenesis in the ERGO-1/Argonaute pathway. Proc Natl Acad Sci U S A. 2010;107: 3582–7. 10.1073/pnas.0911908107 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Conine CC, Batista PJ, Gu W, Claycomb JM, Chaves DA, Shirayama M, et al. Argonautes ALG-3 and ALG-4 are required for spermatogenesis-specific 26G-RNAs and thermotolerant sperm in Caenorhabditis elegans. Proc Natl Acad Sci U S A. 2010;107: 3588–93. 10.1073/pnas.0911685107 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Conine CC, Moresco JJ, Gu W, Shirayama M, Conte D, Yates JR, et al. Argonautes Promote Male Fertility and Provide a Paternal Memory of Germline Gene Expression in C. elegans. Cell. 2013;155: 1532–1544. 10.1016/j.cell.2013.11.032 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Gerson-Gurwitz A, Wang S, Sathe S, Green R, Yeo GW, Oegema K, et al. A Small RNA-Catalytic Argonaute Pathway Tunes Germline Transcript Levels to Ensure Embryonic Divisions. Cell. 2016;165: 396–409. 10.1016/j.cell.2016.02.040 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Wang G, Reinke V. A C. elegans Piwi, PRG-1, Regulates 21U-RNAs during Spermatogenesis. Curr Biol. 2008;18: 861–867. 10.1016/j.cub.2008.05.009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Batista PJ, Ruby JG, Claycomb JM, Chiang R, Fahlgren N, Kasschau KD, et al. PRG-1 and 21U-RNAs Interact to Form the piRNA Complex Required for Fertility in C. elegans. Mol Cell. 2008;31: 67–78. 10.1016/j.molcel.2008.06.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Shi Z, Montgomery TA, Qi Y, Ruvkun G. High-throughput sequencing reveals extraordinary fluidity of miRNA, piRNA, and siRNA pathways in nematodes. Genome Res. 2013;23: 497–508. 10.1101/gr.149112.112 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Das PP, Bagijn MP, Goldstein LD, Woolford JR, Lehrbach NJ, Sapetschnig A, et al. Piwi and piRNAs Act Upstream of an Endogenous siRNA Pathway to Suppress Tc3 Transposon Mobility in the Caenorhabditis elegans Germline. Mol Cell. 2008;31: 79–90. 10.1016/j.molcel.2008.06.003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Gu W, Lee HC, Chaves D, Youngman EM, Pazour GJ, Conte D, et al. CapSeq and CIP-TAP identify pol ii start sites and reveal capped small RNAs as C. elegans piRNA precursors. Cell. 2012;151: 1488–1500. 10.1016/j.cell.2012.11.023 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Killian DJ, Hubbard EJA. Caenorhabditis elegans germline patterning requires coordinated development of the somatic gonadal sheath and the germ line. Dev Biol. 2005;279: 322–335. 10.1016/j.ydbio.2004.12.021 [DOI] [PubMed] [Google Scholar]
- 43.Bukhari SIA, Vasquez-Rifo A, Gagné D, Paquet ER, Zetka M, Robert C, et al. The microRNA pathway controls germ cell proliferation and differentiation in C. elegans. Cell Res. 2012;22: 1034–1045. 10.1038/cr.2012.31 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Rios C, Warren D, Olson B, Abbott AL. Functional analysis of microRNA pathway genes in the somatic gonad and germ cells during ovulation in C. elegans. Dev Biol. 2017;426: 115–125. 10.1016/j.ydbio.2017.04.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Fraser AG, Kamath RS, Zipperlen P, Martinez-Campos M, Sohrmann M, Ahringer J. Functional genomic analysis of C. elegans chromosome I by systematic RNA interference. Nature. 2000;408: 325–330. 10.1038/35042517 [DOI] [PubMed] [Google Scholar]
- 46.Gönczy P, Echeverri C, Oegema K, Coulson A, Jones SJ, Copley RR, et al. Functional genomic analysis of cell division in C. elegans using RNAi of genes on chromosome III. Nature. 2000;408: 331–6. 10.1038/35042526 [DOI] [PubMed] [Google Scholar]
- 47.Green RA, Kao H-L, Audhya A, Arur S, Mayers JR, Fridolfsson HN, et al. A High-Resolution C. elegans Essential Gene Network Based on Phenotypic Profiling of a Complex Tissue. Cell. 2011;145: 470–482. 10.1016/j.cell.2011.03.037 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Reinke V, Gil IS, Ward S, Kazmer K. Genome-wide germline-enriched and sex-biased expression profiles in Caenorhabditis elegans. Development. 2003;131: 311–323. 10.1242/dev.00914 [DOI] [PubMed] [Google Scholar]
- 49.Ma X, Zhu Y, Li C, Xue P, Zhao Y, Chen S, et al. Characterisation of Caenorhabditis elegans sperm transcriptome and proteome. BMC Genomics. 2014;15: 168 10.1186/1471-2164-15-168 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Ellis RE, Kimble J. The fog-3 gene and Regulation of Cell Fate in the Germ line of Caenorhabditis elegans. Genetics. 1995;139: 561–577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Noble DC, Aoki ST, Ortiz MA, Kim KW, Verheyden JM, Kimble J. Genomic Analyses of Sperm Fate Regulator Targets Reveal a Common Set of Oogenic mRNAs in Caenorhabditis elegans. Genetics. 2016;202: 221–34. 10.1534/genetics.115.182592 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Ruby JG, Jan C, Player C, Axtell MJ, Lee W, Nusbaum C, et al. Large-Scale Sequencing Reveals 21U-RNAs and Additional MicroRNAs and Endogenous siRNAs in C. elegans. Cell. 2006;127: 1193–1207. 10.1016/j.cell.2006.10.040 [DOI] [PubMed] [Google Scholar]
- 53.Billi AC, Freeberg MA, Day AM, Chun SY, Khivansara V, Kim JK. A conserved upstream motif orchestrates autonomous, germline-enriched expression of Caenorhabditis elegans piRNAs. Copenhaver GP, editor. PLoS Genet. 2013;9: e1003392 10.1371/journal.pgen.1003392 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Yang W, Dierking K, Schulenburg H. WormExp: a web-based application for a Caenorhabditis elegans -specific gene expression enrichment analysis. Bioinformatics. 2016;32: 943–945. 10.1093/bioinformatics/btv667 [DOI] [PubMed] [Google Scholar]
- 55.West SM, Mecenas D, Gutwein M, Aristizábal-Corrales D, Piano F, Gunsalus KC. Developmental dynamics of gene expression and alternative polyadenylation in the Caenorhabditis elegans germline. Genome Biol. 2017;19: 8 10.1186/s13059-017-1369-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Claycomb JM, Batista PJ, Pang KM, Gu W, Vasale JJ, van Wolfswinkel JC, et al. The Argonaute CSR-1 and Its 22G-RNA Cofactors Are Required for Holocentric Chromosome Segregation. Cell. 2009;139: 123–134. 10.1016/j.cell.2009.09.014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Bagijn MP, Goldstein LD, Sapetschnig A, Weick E-M, Bouasker S, Lehrbach NJ, et al. Function, targets, and evolution of Caenorhabditis elegans piRNAs. Science. 2012;337: 574–578. 10.1126/science.1220952 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Jose AM, Garcia GA, Hunter CP. Two classes of silencing RNAs move between Caenorhabditis elegans tissues. Nat Struct Mol Biol. 2011;18: 1184–1188. 10.1038/nsmb.2134 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Raman P, Zaghab SM, Traver EC, Jose AM. The double-stranded RNA binding protein RDE-4 can act cell autonomously during feeding RNAi in C. elegans. Nucleic Acids Res. 2017;45: 8463–8473. 10.1093/nar/gkx484 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Chen P-J, Singal A, Kimble J, Ellis RE. A Novel Member of the Tob Family of Proteins Controls Sexual Fate in Caenorhabditis elegans Germ Cells. Dev Biol. Academic Press; 2000;217: 77–90. 10.1006/dbio.1999.9521 [DOI] [PubMed] [Google Scholar]
- 61.Luitjens C, Gallegos M, Kraemer B, Kimble J, Wickens M. CPEB proteins control two key steps in spermatogenesis in C. elegans. Genes Dev. 2000;14: 2596–609. 10.1101/GAD.831700 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Sumiyoshi E, Sugimoto A, Yamamoto M. Protein phosphatase 4 is required for centrosome maturation in mitosis and sperm meiosis in C. elegans. J Cell Sci. 2002;115: 1403–10. [DOI] [PubMed] [Google Scholar]
- 63.Zhu G, Salazar G, Zlatic SA, Fiza B, Doucette MM, Heilman CJ, et al. SPE-39 family proteins interact with the HOPS complex and function in lysosomal delivery. Mol Biol Cell. 2009;20: 1223–40. 10.1091/mbc.E08-07-0728 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.von Tobel L, Mikeladze-Dvali T, Delattre M, Balestra FR, Blanchoud S, Finger S, et al. SAS-1 Is a C2 Domain Protein Critical for Centriole Integrity in C. elegans. Dutcher SK, editor. PLoS Genet. 2014;10: e1004777 10.1371/journal.pgen.1004777 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Kang C, You Y -j., Avery L. Dual roles of autophagy in the survival of Caenorhabditis elegans during starvation. Genes Dev. 2007;21: 2161–2171. 10.1101/gad.1573107 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Kalis AK, Kroetz MB, Larson KM, Zarkower D. Functional genomic identification of genes required for male gonadal differentiation in Caenorhabditis elegans. Genetics. 2010;185: 523–535. 10.1534/genetics.110.116038 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Nelson MD, Zhou E, Kiontke K, Fradin H, Maldonado G, Martin D, et al. A bow-tie genetic architecture for morphogenesis suggested by a genome-wide RNAi screen in Caenorhabditis elegans. Chisholm AD, editor. PLoS Genet. 2011;7: e1002010 10.1371/journal.pgen.1002010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Pak J, Maniar JM, Mello CC, Fire A. Protection from Feed-Forward Amplification in an Amplified RNAi Mechanism. Cell. 2012;151: 885–899. 10.1016/j.cell.2012.10.022 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Zhang C, Montgomery TA, Fischer SEJ, Garcia SMDA, Riedel CG, Fahlgren N, et al. The Caenorhabditis elegans RDE-10/RDE-11 Complex Regulates RNAi by Promoting Secondary siRNA Amplification. Curr Biol. 2012;22: 881–890. 10.1016/j.cub.2012.04.011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Tsai H-Y, Chen C-CG, Conte D, Moresco JJ, Chaves DA, Mitani S, et al. A Ribonuclease Coordinates siRNA Amplification and mRNA Cleavage during RNAi. Cell. 2015;160: 407–419. 10.1016/j.cell.2015.01.010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Ooi SL, Priess JR, Henikoff S. Histone H3.3 Variant Dynamics in the Germline of Caenorhabditis elegans. PLoS Genet. 2006;2: e97 10.1371/journal.pgen.0020097 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.LaMunyon CW, Ward S. Larger sperm outcompete smaller sperm in the nematode Caenorhabditis elegans. Proc R Soc B Biol Sci. 1998;265: 1997–2002. 10.1098/rspb.1998.0531 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Barton MK, Schedl TB, Kimble J. Gain-of-Function Mutations of fem-3, a Sex-Determination Gene in Caenorhabditis elegans. Genetics. 1987;115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.McJunkin K, Ambros V. A microRNA family exerts maternal control on sex determination in C. elegans. Genes Dev. 2017;31: 422–437. 10.1101/gad.290155.116 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Lim LP, Lau NC, Weinstein EG, Abdelhakim A, Yekta S, Rhoades MW, et al. The microRNAs of Caenorhabditis elegans. Genes Dev. 2003;17: 991–1008. 10.1101/gad.1074403 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.de Lencastre A, Pincus Z, Zhou K, Kato M, Lee SS, Slack FJ. MicroRNAs Both Promote and Antagonize Longevity in C. elegans. Curr Biol. 2010;20: 2159–2168. 10.1016/j.cub.2010.11.015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Pincus Z, Smith-Vikos T, Slack FJ. MicroRNA Predictors of Longevity in Caenorhabditis elegans. Kim SK, editor. PLoS Genet. 2011;7: e1002306 10.1371/journal.pgen.1002306 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Ortiz MA, Noble D, Sorokin EP, Kimble J. A new dataset of spermatogenic vs. oogenic transcriptomes in the nematode Caenorhabditis elegans. G3. 2014;4: 1765–72. 10.1534/g3.114.012351 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.van Wolfswinkel JC, Claycomb JM, Batista PJ, Mello CC, Berezikov E, Ketting RF. CDE-1 Affects Chromosome Segregation through Uridylation of CSR-1-Bound siRNAs. Cell. 2009;139: 135–148. 10.1016/j.cell.2009.09.012 [DOI] [PubMed] [Google Scholar]
- 80.Yigit E, Batista PJ, Bei Y, Pang KM, Chen C-CG, Tolia NH, et al. Analysis of the C. elegans Argonaute Family Reveals that Distinct Argonautes Act Sequentially during RNAi. Cell. 2006;127: 747–757. 10.1016/j.cell.2006.09.033 [DOI] [PubMed] [Google Scholar]
- 81.Hodgkin J, Horvitz HR, Brenner S. Nondisjunction Mutants of the Nematode Caenorhabditis elegans. Genetics. 1979;91: 67–94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Zhang C, Montgomery TA, Gabel HW, Fischer SEJ, Phillips CM, Fahlgren N, et al. mut-16 and other mutator class genes modulate 22G and 26G siRNA pathways in Caenorhabditis elegans. Proc Natl Acad Sci U S A. 2011;108: 1201–8. 10.1073/pnas.1018695108 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Kamath RS, Martinez-Campos M, Zipperlen P, Fraser AG, Ahringer J. Effectiveness of specific RNA-mediated interference through ingested double-stranded RNA in Caenorhabditis elegans. Genome Biol. 2001;2: RESEARCH0002. 10.1186/gb-2000-2-1-research0002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.O’Connell J, Arganda-Carreras I, Frise E, Kaynig V, Longair M, Pietzsch T, et al. Fiji: an open-source platform for biological-image analysis. Nat Methods. 2012;9: 676–682. 10.1038/nmeth.2019 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Peters N, Perez DE, Song MH, Liu Y, Muller-Reichert T, Caron C, et al. Control of mitotic and meiotic centriole duplication by the Plk4-related kinase ZYG-1. J Cell Sci. 2010;123: 795–805. 10.1242/jcs.050682 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Aronesty E. Comparison of Sequencing Utility Programs. Open Bioinforma J. 2013;7: 1–8. 10.2174/1875036201307010001 [DOI] [Google Scholar]
- 87.Langmead B, Trapnell C, Pop M, Salzberg SL. Ultrafast and memory-efficient alignment of short DNA sequences to the human genome. Genome Biol. 2009;10: R25 10.1186/gb-2009-10-3-r25 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Anders S, Pyl PT, Huber W. HTSeq—a Python framework to work with high-throughput sequencing data. Bioinformatics. 2015;31: 166–169. 10.1093/bioinformatics/btu638 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Love MI, Huber W, Anders S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 2014;15: 550 10.1186/s13059-014-0550-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Martin M. Cutadapt removes adapter sequences from high-throughput sequencing reads. EMBnet.journal. 2011;17: 10 10.14806/ej.17.1.200 [DOI] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
A) Mean normalized miRNA reads (sense) in hermaphrodite (n = 6 replicates) and male gonads (n = 3). miRNA expression-differences with four-fold difference in abundance that were statistically different (Wilcoxon rank sum test with continuity correction; p adjusted<0.01) between gonads of hermaphrodites and males as well as between hermaphrodites and fog-3 males (n = 2) are highlighted in red (female somatic gonad-biased) and blue (male somatic gonad-biased). Related to Fig 1D. B) Mean normalized endogenous siRNA reads (antisense) in hermaphrodite and male gonads; same analyses and representation used as in S1A Fig. Related to Fig 1H.
(TIF)
A) Percentage of antisense reads in hermaphrodite and male gonads classified as primary 26G RNA, secondary 22G RNA or other siRNA types. B) Normalized mean 22G RNA reads in hermaphrodite and male gonads per gene, median dotted line, mean full line. C) Mean 22G RNA reads targeting mRNAs of similar abundance in hermaphrodite (red) and male (blue) gonads. Boxes represent first and second quartile, 1.5 interquartile range: vertical line, mean: horizontal line, outliers: circles. Bonferroni corrected p-value of Wilcoxon rank sum test give the probability that samples from a continuous distribution have equal medians.
(TIF)
A) Mean normalized mRNA reads [55] and siRNAs in percentiles in hermaphrodite and male gonads. For 20 bins (each representing 5% of data i.e. 427 genes per bin in hermaphrodites and 504 in males) a linear model was fitted (black line). Related to Fig 2A. B) p-values indicate whether the linear model deviates from zero. The most significant negative correlation between the levels of 22G RNA and mRNA was found for the 5% of the genes with highest level of 22G RNA expression (shaded). C) mRNA expression for WAGO-1 target genes and non-target genes (Bonferroni corrected p-values of the Wilcoxon rank sum test q<0.01 indicated by stars, ns = not significant, n/a = not applicable). Data are the same as for Fig 2A but plotted as WAGO-1 targets [20] and non-WAGO-1 targets in hermaphrodites and males. Data were only available for hermaphrodites and interpolated to male samples. D) mRNA expression for ALG-3/-4 target genes and non-target genes (Bonferroni corrected p-values of the Wilcoxon rank sum test q<0.01 indicated by stars, ns = not significant, n/a = not applicable). Data are the same as for Fig 2A but plotted as ALG-3/-4 targets [34] and non-ALG-3/-4 targets in males.
(TIF)
Mean primary and secondary siRNA reads normalized to total reads in gonads from fog-3 males (n = 2), control hermaphrodites (n = 4) and control males (n = 2) raised on gfp(RNAi). Data for control hermaphrodites and control males are taken from Fig 3B. The right panel shows the ratio of secondary siRNA/ primary siRNA. Means were compared with t-tests w* = p<0.05, *** = p<0.001.
(TIF)
A) Fluorescence microscopy images of GFP-sensor expression of L4 hermaphrodite germlines (dotted line, distal end indicated by star); animals fed (gfp)RNAi or control RNAi. Percentage of animals with germline expression classified as: high, intermediate uniform, intermediate variable and no GFP-sensor expression (from bottom to top) for (gfp)RNAi (n = 27). 100% of animals raised on control RNAi showed high GFP-sensor expression (n = 24). B) Fluorescence microscopy images of sperm chromatin marked with a ubiquitous GFP-sensor in adult hermaphrodites, males and fem-3 hermaphrodites producing only sperm (top). Partial silencing of the GFP-sensor upon gfp(RNAi) at 24°C in control and fem-3 hermaphrodites, while control male germlines are RNAi resistant (bottom). Images were acquired with the same exposure time in control and RNAi condition, but are different between sexes due to different expression levels and physiology. For illustration purposes images are background subtracted, as explained in Fig 4D.
(TIF)
Analyses of sex-biased miRNA, piRNA, germline siRNA, sense and antisense reads.
(XLSX)
WormExp input and output.
(XLSX)
Expression levels of 22G RNA and mRNA and classification as WAGO, WAGO-1, CSR-1, ALG-3/-4.
(XLSX)
(XLSX)
Accession and genotype of each replicate.
(XLSX)
Data Availability Statement
Small RNA library sequencing data is available on the European Nucleotide Archive in the Study: PRJEB12010. A sample description can be found in additional file 3.