Abstract
Tree bark represents an important source of medicinal compounds that may be useful for cancer therapy. In the current study, high-performance liquid chromatography with diode-array detection (HPLC-DAD) was used to determine the profile of the phenolic compounds of Catalpa speciosa, Taxus cuspidata, and Magnolia acuminata bark extracts. The antioxidant and anticancer bioactivities against different cancer cell lines were investigated. M. acuminata exerted significantly higher antioxidant activities in the diphenyl picrylhydrazine and β-carotene-linoleic acid assays than the other species. In C. speciosa, novel profiles of phenolic acids (ferulic acid was the predominant compound) and catechin were detected. In T. cuspidata, six phenolic acids were detected; the predominant compounds were hydroxycaffeic acid and protocatechuic acid. In M. acuminata, two phenolic acids and three catechins were detected; catechin was the predominant compound. The three species exerted clear anticancer activity against MCF-7, HeLa, Jurkat, T24, and HT-29 cells, with the strongest activity found in the extracts from M. acuminata. No antiproliferative activity against normal cells was found. Flow cytometry revealed greater accumulation of necrotic and early/late apoptotic cells in various treated cancer cells than in untreated control cells, and protocatechuic acid induced a similar accumulation of necrotic cells to that of the bark extracts. Caspase-3 and -7 activity was increased in cancer cells treated with different bark extracts; the highest activity was found in the M. acuminata treatment. Our results suggested that the treatment of cancer cells with bark extracts of M. acuminata, C. speciosa, and T. cuspidata, and protocatechuic acid induced apoptosis, suggesting an association between anticancer activities and individual phenolic compounds.
Keywords: Catalpa speciosa, Taxus cuspidata, Magnolia acuminata, phenols, antioxidants, anticancer
1. Introduction
Tree bark is used widely in folklore medicine to treat ailments and control the progress of many diseases and injuries, such as arthritis, gonorrhea, rheumatism, dysentery, malaria, inflammation, wounds, ulcers, and constipation [1,2,3]. Recent investigations found that tree bark may contain antioxidant and anticancer phenolic compounds [2,4,5,6]. However, the current knowledge of the pharmaceutical properties of tree bark is limited.
Catalpa belongs to Bignoniaceae and the genus contains several species, including Catalpa speciosa, which are distributed throughout North America and some parts of Europe [7]. Leaf extracts from other Catalpa species, including C. ovata, C. fargesii, C. bignonioides, and C. bungei are known to have antioxidant activity [8,9] and high phenolic composition. No pharmaceutical studies of C. speciosa have been conducted.
Magnolia species, which belong to Magnoliaceae, contain several species, including M. officinalis, M. obovata, and M. biondii, which have been used in folklore medicine for thousands of years in Asia [10]. Moreover, Magnoliae officinalis cortex is listed in the newest European Pharmacopoeia 9.0 as an official pharmaceutical raw plant material in Europe [11]. The dried parts of the magnolia are used to control diarrhea, abdominal swelling, or constipation, and cough [12]. The analyses of the bark and/or seeds of M. officinalis, M. obovata, and M. biondii revealed the availability of specific bioactive compounds, such as magnolol, honokiol, and obovatol, which have potent anticancer and antioxidant activities [4,6]. Other species, such as M. acuminata, have attracted little attention.
Taxus belongs to Taxaceae and contains approximately 13 species distributed throughout Asia, Europe, and North America [13]. The leaves and bark of some species. such as Taxus wallichiana, are used in the traditional Ayurveda and Unani medicines for the control of fever, cough, and cold, and exert anticancer activities [5]. The bark of Taxus baccata is a well-known pharmaceutical raw material that contains paclitaxel, a known anticancer compound [11]. T. cuspidata bark extract showed anticancer activities due to the presence of paclitaxel and other lignans in the roots [14,15]. However, no studies revealed the detailed phenolic and catechin profiles of this species.
The current study explores the phenolic, catechin, and flavonoid content of C. speciosa, T. cuspidata, and M. acuminata using HPLC-DAD method. The antioxidant, antiproliferative, apoptotic, and caspase-3/7 activities have been explored using several cancer cell lines.
2. Results
2.1. Targeted Profiling of Catechins and Phenols
2.1.1. C. speciosa
In C. speciosa methanolic bark extract, seven phenolic acids (caffeic acid, p-coumaric acid, ferulic acid, gallic acid, p-hydroxybenzoic acid, protocatechuic acid, and vanillic acid) were found out of the 22 screened (Table 1 and Supplementary files). Ferulic acid was the predominant compound (22.7 ± 0.18 mg 100 g−1 DW); other phenolic acids were detected in lower quantities. The content of p-hydroxybenzoic acid and vanillic acid was approximately 6 mg 100 g−1 DW, the content of caffeic acid, p-coumaric acid, and protocatechuic acid was approximately 3 mg 100 g−1 DW, and gallic acid was present at the lowest concentration (ca. 1.6 mg 100 g−1 DW). A low amount of catechin (ca. 1.2 mg 100 g−1 DW) was detected out of the five analyzed catechin derivatives (Table 2). No flavonoids were detected.
Table 1.
The phenolic acid compositions of Catalpa speciosa, Taxus cuspidata, and Magnolia acuminnata outer bark extracts.
| Species | Chemical Compound | Amount [mg 100g−1] D.W. |
|---|---|---|
| Catalpa speciosa | Caffeic acid | 3.04 ± 0.45 |
| p-Coumaric acid | 3.28 ± 0.44 | |
| Ferulic acid | 22.7 ± 0.18 | |
| Gallic acid | 1.57 ± 0.04 | |
| p-Hydroxybenzoic acid | 6.42 ± 0.03 | |
| Protocatechuic acid | 3.22 ± 0.02 | |
| Vanillic acid | 5.77 ± 0.22 | |
| Taxus cuspidata | Caffeic acid | 3.05 ± 0.01 |
| Chlorogenic acid | 8.30 ± 0.22 | |
| Gallic acid | 2.04 ± 0.07 | |
| p-Hydroxybenzoic acid | 2.42 ± 0.16 | |
| Hydroxycaffeic acid | 23.98 ± 1.3 | |
| Protocatechuic acid | 20.97 ± 0.56 | |
| Magnolia acuminnata | Ellagic acid | 0.43 ± 0.08 |
| Protocatechuic acid | 15.31 ± 1.19 |
Table 2.
The catechin derivatives compositions of Catalpa speciosa and Magnolia acuminnata outer bark extracts.
| Species | Chemical Compound | Amount [mg 100g−1] D.W. |
|---|---|---|
| Catalpa speciosa | Catechin | 1.19 ± 0.05 |
| Magnolia acuminnata | Catechin | 85.47 ± 1.30 |
| Epicatechin | 22.78 ± 0.53 | |
| Epigallocatechin gallate | 14.22 ± 0.95 |
2.1.2. T. cuspidata
In T. cuspidata bark extracts, caffeic acid, chlorogenic acid, gallic acid, p-hydroxybenzoic acid, hydroxycaffeic acid, and protocatechuic acid were identified (Table 1 and Supplementary files). The predominant compounds were protocatechuic acid (ca. 21 mg 100 g−1 DW) and hydroxycaffeic acid (ca. 24 mg 100 g−1 DW). In the extracts, no flavonoids or catechins were found.
2.1.3. M. acuminata
In the M. acuminata bark extracts, protocatechuic acid (ca. 15 mg 100 g−1 DW) was the dominant phenolic acid (Table 1 and Supplementary files). A low composition of ellagic acid (less than 0.5 mg 100 g−1 DW) was also identified. However, catechins and catechin derivatives, epicatechin, and epigallocatechin gallate, were found in the extracts (Table 2). Catechin was the main compound (ca. 85.5 mg 100 g−1 DW), followed by epicatechin (ca. 23 mg 100 g−1 DW) (Table 2). No flavonoids were detected by using the HPLC-DAD method.
2.2. Antioxidant Activities
Bark extracts showed antioxidant activity as found in Table 3. M. acuminata exhibited the highest antioxidant activities in the diphenyl picryl hydrazyl (DPPH) (IC50, 3.1 µg mL−1) and β-carotene-linoleic acid (IC50, 3.6 µg mL−1) assays compared to other species. T. cuspidata exhibited higher antioxidant activities than C. speciosa. M. acuminata antioxidant power was comparable with those of the standard antioxidant (BHT).
Table 3.
Diphenyl picryl hydrazyl (DPPH) and β-carotene-linoleic acid of Catalpa speciosa, Taxus cuspidata, Magnolia acuminnata outer bark extracts.
| Plant/Standard | DPPH Free Radical Scavenging Activity (IC50, µg mL−1) |
β-Carotene-Linoleic Acid Assay (IC50, µg mL−1) |
|---|---|---|
| Catalpa speciosa | 4.4 ± 0.1a | 5.1 ± 0.1a |
| Taxus cuspidata | 4.2 ± 0.1b | 4.8 ± 0.1b |
| Magnolia acuminnata | 3.1 ± 0.1c | 3.6 ± 0.1c |
| BHT | 2.9 ± 0.1c | 3.2 ± 0.1c |
2.3. Antiproliferative Activities
The bark extracts exhibited antiproliferative activities against different cancer cell lines, as shown in Table 4. Anticancer activity against MCF-7, HeLa, Jurkat, T24, and HT-29 cells was noted. The strongest anticancer bioactivity was found in M. acuminata (IC50, 16.20–152.8 µg/mL). Further, no extract exerted antiproliferative activity against normal HEK-293 cells. Specific catechins and phenolics found in the bark extracts, including protocatechuic acid, catechin, ferulic acid, and hydroxycaffeic acid, exhibited notable antiproliferative activity against most cancer cells.
Table 4.
In vitro antiproliferative activity (IC50 (µg/mL)) of Catalpa speciosa, Taxus cuspidata, Magnolia acuminnata outer bark extracts on cancer cell lines.
| Plant/Standard | HeLa | MCF-7 | Jurkat | T24 | HT-29 | HEK-293 |
|---|---|---|---|---|---|---|
| Catalpa speciosa | 58.3 ± 1.7 | 41.19 ± 1.6 | 41.4 ± 1.1 | 249.5 ± 2.9 | 111.5 ± 2.9 | ˃400 |
| Taxus cuspidata | 54.5 ± 1.9 | 39.51 ± 0.9 | 37.3 ± 0.5 | 220.1 ± 2.9 | 102.2 ± 3.1 | ˃400 |
| Magnolia acuminnata | 28.4 ± 1.3 | 16.20 ± 1.1 | 25.1 ± 1.1 | 152.8 ± 2.9 | 89.2 ± 2.5 | ˃400 |
| Catechin | 36.48 ± 1.2 | 17.64 ± 1.8 | 38.16 ± 0.7 | 183.28 ± 4.3 | 96.16 ± 1.5 | ˃400 |
| Protocatechuic acid | 39.10 ± 2.3 | 18.97 ± 2.1 | 47.35 ± 2.1 | 176.35 ± 2.1 | 95.35 ± 3.3 | ˃400 |
| Ferulic acid | 51.73 ± 3.5 | 43.85 ± 1.3 | 39.11 ± 2.3 | 227.26 ± 5.1 | 126.26 ± 4.4 | ˃400 |
| Hydroxycaffeic acid | 65.31 ± 2.4 | 58.11 ± 0.9 | 63.09 ± 1.4 | 176.12 ± 3.6 | 113.15 ± 2.9 | ˃400 |
| Vinblastine sulfate | 2.7 ± 0.06 | ‒ | 0.1 ± 0.09 | 65.7 ± 2.1 | 21.0 ± 0.1 | 50.1± 2.3 |
| Vincristine | 8.5 ± 0.1 | 4.63 ± 1.8 | 0.4 ± 0.05 | 89.8 ± 2.5 | 47.3 ± 0.2 | 78.3 ± 1.6 |
| Taxol | ‒ | 0.09 ± 0.009 | ‒ | ‒ | ‒ | ‒ |
2.4. Apoptotic Cell Population
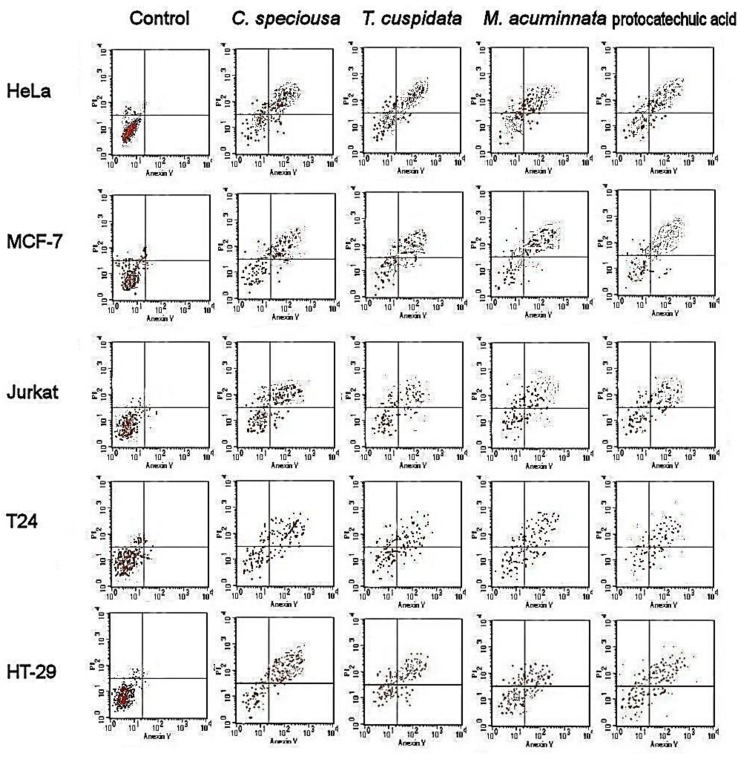
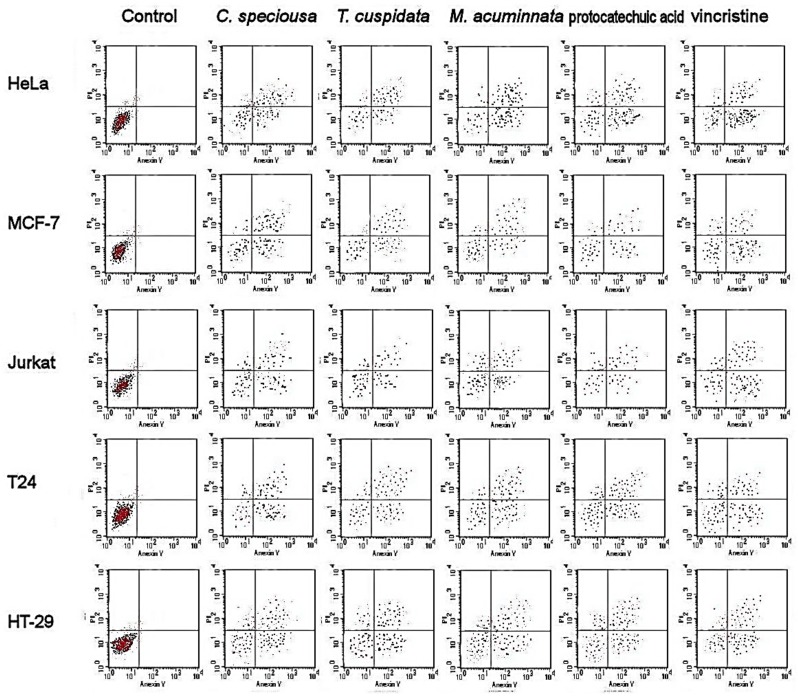
Flow cytometry revealed the extent of apoptosis in different cell lines subjected to the (50% Inhibition concentration) IC50 of bark extracts (Figure 1 and Figure 2). The results showed a greater accumulation of necrotic cells, and early and late apoptotic cells in treated cancer cells than in control cells in various cancer cell lines. Treatment with protocatechuic acid showed similar accumulation of necrotic cells as treatment with the bark extracts of the three species. The bioactivity of the extracts and protocatechuic acid was sustained after 48 h.
Figure 1.
Cellular apoptosis induced in cancer cells at 24 h following treatment with bark extracts and catechin. Lower left, viable cells; upper left, necrotic cells; lower right, early apoptotic cells; and upper right, late apoptotic cells.
Figure 2.
Cellular apoptosis induced in cancer cells at 48 h following treatment with bark extracts and catechin. Lower left, viable cells; upper left, necrotic cells; lower right, early apoptotic cells; and upper right, late apoptotic cells.
2.5. Detection of Caspase-3/7 Activity
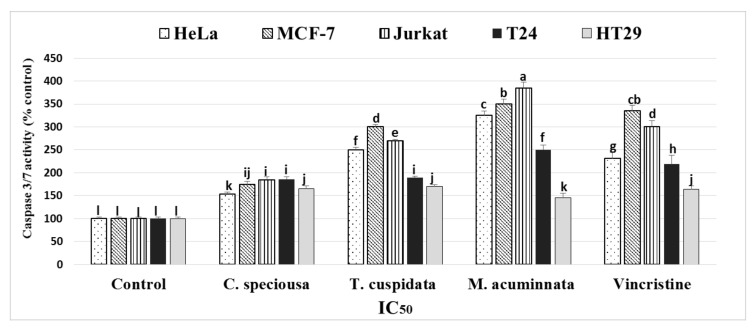
The effects of bark extracts on caspase-3/7 activity were investigated in HeLa, MCF-7, Jurkat, HT-29, and T24 cells (Figure 3). The results showed that greater caspase 3/7 activity occurred after M. acuminata, T. cuspidata, and C. speciosa treatment in all cancer cell lines than in the untreated control cells. M. acuminata resulted in a higher activity of caspase 3/7 than the other treatments.
Figure 3.
Enzyme activity of caspase 3/7 following treatment of different cancer cells with C. speciousa, T. cuspidate, and M. acuminnata bark extracts (IC50). The activity was expressed as a percentage (%) of untreated cells.
2.6. Western Blotting Analyses of Caspases-3 and Caspase-7
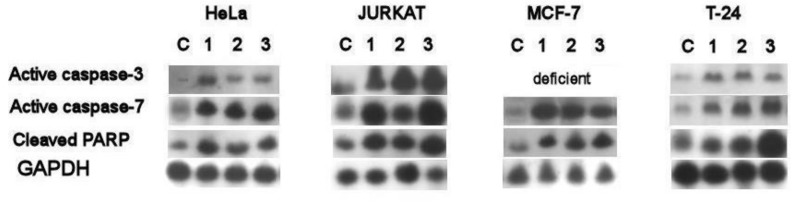
Western blotting of caspase-3 and caspase-7 activation by bark extracts was tested as shown in Figure 4. Bark extracts increased the activation of caspase-3 and caspase-7 in all cancer cells, except in MCF-7 which was deficient for caspase-3. Further, high proteolytic cleavage of the nuclear enzyme poly (adenosine diphosphate ribose) polymerase/PARP (caspase substrates) was found in all treatments compared to control. This increased cleavage is required for the activation of key apoptosis executioners (caspase-3 and caspase-7).
Figure 4.
Western blot analysis of active caspase-3, caspase-7 and cleaved PARP using bark extracts (IC50) of C. speciousa (1), T. cuspidata (2), and M. acuminnata (3).
3. Discussion
This is the first report to document detailed phenolic and catechin profiles of C. speciosa bark extract. The phenolic acid profile of C. speciosa bark was relatively unique (compared to other species) and contained ferulic acid (as the dominant compound), caffeic acid, p-coumaric acid, gallic acid, p-hydroxybenzoic acid, protocatechuic acid, and vanillic acid. Previous investigations into C. ovata, C. fargesii, and C. bungei leaves [8] have revealed only flavonoids such as luteolin and apigenin. C. bignonioides flowers and leaves had high phenolic contents [9]. The current investigation was the first to identify several phenolics in the T. cuspidata bark extract. Previously detected paclitaxel, a diterpenoid with anticancer activity, in the T. cuspidata bark extract [14]. In addition, several lignans were isolated from other parts of this species [15]. In different species (Taxus wallichiana Zucc.), the leaves and bark were found to contain paclitaxel and exhibit potent anticancer activities [5].
In M. acuminata bark, two phenolic acids (ellagic acid and protocatechuic acid) and three catechins (catechin, epicatechin, and epigallocatechin gallate) were found. Catechin was the predominant compound, followed by epicatechin; neither had been previously isolated from M. acuminata. Previous investigations described other phenols in M. officinalis stem bark; these phenols included magnolianone and its derivatives, which exerted antioxidant activities [4,16], and magnoloside, a phenylethanoid glycoside [6].
The antioxidant activity assays showed that M. acuminata bark extract had the highest antioxidant activity. This high antioxidant activity was mainly attributable to the high concentration of catechin (85.47 ± 1.30 mg 100 g−1) and catechin derivatives in the bark extracts. Catechin is strongly associated with the overall antioxidant activities in various species [17,18]. The barks of other species, such as Magnolia officinalis, were shown to have antioxidant activities [19], which were attributed to the glycosides and flavonoids in another study [20]. Studied on other species, such as Magnolia grandiflora, it revealed antioxidant effect in the flower extract [21] and comparable activity in the leaf extract [22]. T. cuspidata exerted stronger antioxidant activities than C. speciosa. A study on T. cuspidata bark revealed polyphenols that might have antioxidant effects, including lignans and catechins [23]; the authors found catechin and epicatechin, although we did not detect these types of catechins in the current study. In C. speciosa, we found a moderate antioxidant effect for the bark. Previous investigations revealed the antioxidant effect of the leaves of C. ovata, C. fargesii, and C. bungei [8], as well as in the inflorescence and leaves of C. bignonioides [9]. These antioxidant activities were attributed to the high total phenolic content [9] or the presence of flavonoids such as luteolin and apigenin [8]. In our study, this antioxidant bioactivity of C. speciosa bark extract was attributed to specific phenolic contents, including ferulic acid (22.7 ± 0.18 mg 100 g−1), which are known to be strong antioxidants [24].
Anticancer activities against the MCF-7, HeLa, Jurkat, T24, and HT-29 cells were found; the highest activities were exhibited by M. acuminata. The main components detected in M. acuminata were catechins, which were relatively abundant, including catechin, epicatechin, protocatechuic acid, and epigallocatechin gallate; the observed effects were related to these components of the bark. Protocatechuic acid (PCA, 3,4-dihydroxybenzoic acid) has been correlated to anticancer activities against different cancer cells, as found in this investigation, and is a relatively strong antioxidant [25,26]. Catechins are strongly associated with anticancer effects, such as reports of green tea catechins [27] and the synergistic effects with other treatments [28]. Epicatechin is also associated with the anticancer activity and the induction of apoptosis [29], especially epicatechin oligomers [30]. In addition, it is believed that epicatechin and epigallocatechin in green tea have anticancer and apoptosis-inducing activities [31]. Other species of Magnolia also have anticancer activities [32].
The anticancer bioactivity of C. speciosa is correlated with its major phenolic compartments, including ferulic acid, p-hydroxybenzoic acid, vanillic acid, and caffeic acid. Ferulic acid suppresses the metastasis in breast cancer cells by regulating the reversal of epithelial-mesenchymal transition [33] and inducing the cell cycle arrest in the cells of cervical cancer [34]. In our study, ferulic acid was the major phenol found in the bark extract (22.7 ± 0.18 mg 100 g−1) and is greatly associated with anticancer activity. We found that C. speciosa bark extracts have potential antioxidant and anticancer activities against MCF-7 breast cancer, which may be attributable to phenolic acid compounds such as protocatechuic acid and p-hydroxybenzoic acid. These compounds were found to have the power to control MCF-7 breast and PC-3 prostate cancers [35].
T. cuspidata showed antiproliferative activities against Jurkat, MCF-7, and HeLa cancer cells and these activities were directly associated with the major phenolic and catechin components, including hydroxycaffeic acid (23.98 ± 1.3 mg 100 g−1), chlorogenic acid (8.3 ± 0.22 mg 100 g−1), protocatechuic acid (20.97 ± 0.56 mg 100 g−1), and gallic acid (2.04 ± 0.07 mg 100 g−1). These phenolic and catechin compounds were shown to have antiproliferative effects against different cancer cells [35,36]. In addition to our results that the bark extracts had clear antiproliferative activities against specific cancer cell types, it was previously reported that twigs and needles of the same plant exert anticancer activities against human cancer [37].
The results of flow cytometry showed an accumulation of necrotic cells and early and late apoptotic cells in various cancer cells subjected to bark extracts compared with the untreated control cells. The apoptotic activities of M. acuminata are strongly connected with the catechin composition, as similar effects to those of catechin were found. This conclusion was strongly supported by the apoptosis induction described before for catechins such as epicatechin [29], epicatechin oligomers [30], epicatechin, and epigallocatechin [31]. The flower extract of M. grandiflora showed an apoptotic effect in lymphocytic leukemia cells [38]. There was a high accumulation of early and late apoptotic cells after following bark extracts and catechin treatment. In C. speciosa, ferulic acid was suggested to be the cornerstone of the apoptosis-inducing activities as it was associated with the death of osteosarcoma cells through the promotion of caspase-3 and apoptosis [39]. In another study, ferulic acid induced cell cycle arrest in cervical cancer cells [34]. In T. cuspidata, the apoptotic activities observed in this study agreed with a previous investigation into the needle and twig extracts of the same species [37]. The authors reported HeLa cells apoptosis in HeLa cells and low toxicity in normal cells and G(2)/M cell cycle arrest. The major phenolic acid in T. cuspidata is protocatechuic acid which showed apoptosis and slow metastasis in cancer cells [40].
Caspase-3 and -7 enzymes function as mediators of apoptosis through DNA fragmentation and apoptotic chromatin condensation, which lead to cell death [41,42]. We found increased activities of these enzymes in bark extract-treated cancer cells except in MCF-7 which was deficient for caspase-3 only [43,44]. The western plotting of these enzymes confirmed the antiproliferative and apoptotic activities of the three bark extracts as found in in the extracts of other plants [45]. Previous investigations into the use of ferulic acid to control cancer cells revealed that this compound promoted the apoptosis pathway through the activation of caspase-3 [39]. This was the case for C. speciosa, in which ferulic acid was the major phenol found in the bark extracts studied. C. speciosa might be a potential new natural source of ferulic acid. The highest activities of caspase-3/7 were found after M. acuminata treatment and this activity may be associated with the important catechins in this bark, such as epigallocatechin gallate and epicatechin, and the phenolic acid, protocatechuic acid. Epigallocatechin gallate was one of the major catechins found in M. acuminata bark in this study and was strongly related to the increased activities of caspase-3 in green tea catechins [46]. Protocatechuic acid was shown to increase caspase-3 activities [40]; however, another study reported contrasting results [25]. Epicatechin is strongly related to caspase-3 activity in several studies [47,48]. T. cuspidata showed some degree of caspase-3/7 activation, which may be attributable to several phenolic compounds found in the bark extract that stimulate the activity of caspase-3, such as chlorogenic acid [49] and gallic acid [50].
4. Materials and Methods
4.1. Plant Material
Catalpa speciosa (Bignoniaceae), Taxus cuspidata Siebold & Zucc.(Taxaceae), and Magnolia acuminata L. (Magnoliaceae) outer bark was obtained from identified plants at the Arboretum of University of Guelph, Ontario, Canada. The samples were identified by Hosam Elansary and a voucher was deposited at the University of Guelph and Alexandria University (Hosam000980-2018).
4.2. Sample Preparation and Cell Cultures
Fresh bark (0.25 g) was dried at 35 °C until a constant weight was obtained. The samples were ground and then dissolved in 3 mL methanol (99%) for 1 h in the dark at 25 °C. Bark solutions were centrifuged for 5 min at 10,000 rpm (7000 × g) and the supernatant was obtained (~2.7 mL). The samples were passed through a 0.45 μm polytetrafluroethylene (PTFE) nylon filter and then stored at −80 °C. Analytical grade chemicals (Sigma Aldrich, Germany) were used in the bioassays. The cancer cell lines, including cervical adenocarcinoma (HeLa), breast adenocarcinoma (MCF-7), T-cell lymphoblast like (Jurkat), urinary bladder carcinoma (T24), and colon adenocarcinoma (HT-29), were obtained from the American Type Culture Collection (ATCC).
4.3. Analyses of Phenolic Compounds
C. speciosa, T. cuspidata, and M. acuminata bark samples were dried by lyophilization (Labconco, Kansas City, MO, USA) and powdered. Bark samples were extracted (0.5 g) as described before [51]. Validated chromatographic analyses were performed using the HPLC [52,53]. A Purospher® RP-18e analytical column (4 × 250 mm, 5 mL; Merck, Berlin, Germany) was used in the HPLC-DAD (Merck-Hitachi, Tokyo, Japan) equipment. A gradient program was used with a flow rate of 1 mL/min, a detection wavelength of 254 nm, and an injection volume of 10 µL [51,54,55]. UV-DAD spectra and tr values were used for the quantification of the compounds alongside the phenolic, catechin, and flavonoid standards. These included benzoic acid and related derivatives: Ellagic, gallic, 3,4-dihydroxyphenylacetic, protocatechuic, gentisic, p-hydroxybenzoic, salicylic, vanillic, and syringic acids. In addition to cinnamic acid and the related derivatives, such as caffeic, coumaric, ferulic, o-coumaric, m-coumaric, p-hydrocaffeic, isoferulic, sinapic acids, and depsides (chlorogenic, rosmarinic, and neochlorogenic acids). The catechins included catechin, epicatechin, epigallocatechin gallate, epicatechin gallate, and epigallocatechin. The flavonoid standards included aglycones (kaempferol, myricetin, quercetin luteolin, and rhamnetin) and glycosides (apigetrin, cynaroside, robinin, hyperoside, isoquercetin, quercitrin, rutin, trifolin, and vitexin). The standards were obtained from Sigma-Aldrich (Berlin, Germany).
4.4. Antioxidant Activity
The DPPH and β-carotene-linoleic acid assays were used in the Faculty of Food and Agricultural Sciences, King Saud University to determine the antioxidant activities of bark extract [56]. For the DPPH assay, the samples were incubated for 30 min and the absorbance of the samples at 517 nm was measured. In the β-carotene-linoleic acid assay, the absorbance of the samples at 470 nm was measured. The concentration of the sample required to scavenge 50% of the DPPH/β-carotene-linoleic acid solutions, the IC50 (µg/mL) was determined by plotting the inhibition percentage against extract concentration. A standard antioxidant (butylated hydroxytoluene, BHT) was used as a positive control and the inhibition by the concentration of each sample was compared with that of the BHT and blank. The antioxidant activities were repeated twice in duplicates.
4.5. Antiproliferative Activity
The antiproliferative activity of bark extracts was examined in different cancer cells lines (HeLa, MCF-7, Jurkat, HT-29, and T24) and in normal cells (HEK-293) by using a modified 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) method [57]. The cells were grown in 75 cm2 flasks in MEM supplemented with 10% FBS, 17.8 mM NaHCO3, 0.1 mM non-essential amino acids, and 1 mM sodium pyruvate. They were seeded into 96-well plates at a density of 4 × 10−4 per well, left to stand in 270 µL of medium, and incubated in an atmosphere of 37 °C and 5% CO2. Sterilin-filtered leaf extracts were added to the culture media in microtiter plates. Five doses of bark extracts were used at final concentrations of 50, 100, 200, 300, and 400 µg/mL in culture medium. Untreated cells were considered used as negative controls and vinblastine sulfate and taxol treatment were used as the positive controls. After incubation of the culture medium for 2 days at 37 °C and 5% CO2, phosphate buffer saline (PBS) washes were performed to remove extract traces and the medium supplied was 12 mM MTT dissolved in PBS. Subsequently, 0.04 N HCl dissolved in isopropanol was mixed in each well, left to stand for 40 min, and the absorbance at 570 nm was determined by using a microplate reader (Thermo Fisher Scientific, Waltham, Massachusetts, USA). The percentage inhibition of antiproliferation activity was calculated in triplicate [58]:
% Inhibition = (Abs.570 nm control—Abs.570 nm sample)/Abs.570 nm control × 100. Subsequently, IC50 values were obtained by plotting the percentage of cell viability against the extract concentration and expressed in µg/mL.
4.6. Apoptotic Cell Population
Flow cytometry (FAC Scan, Becton Dickinson, Iowa, USA) was used to measure the apoptotic cell population [59]. Different types of cancer cells were cultured (37 °C, 5% CO2) in 6-well plates and treated for 24/48 h with the IC50 of bark extract, as determined from the MTT assay, and catechin and untreated samples were considered as the control. The cells were detached by using trypsin (0.25%) in Hank’s balanced salt solution (Thermo Fisher Scientific, Berlin, Germany). For staining the cells, the Annexin V apoptosis detection kit (Sigma, St. Louis, MO, USA) was used. Briefly, the cells were incubated in the dark at 37 °C for 15 min and washed with cold PBS, and the apoptotic populations were shown by the flow cytometer in quadrants: Lower left (viable cells), upper left (necrotic cells), lower right (early apoptotic cells), and upper right (late apoptotic cells).
4.7. Caspase-Glo 3/7 Assay
The effect of different bark extracts on caspase-3/7 activity using different cancer cell lines was detected by the Caspase-Glo 3/7 Assay kit (Promega, Berlin, Germany). The cancer cell lines were cultured in Roswell Park Memorial Institute (RPMI) growth medium (Sigma-Aldrich, St. Louis, MO, USA) in 96-well plates in the presence of the IC50 of the extracts, catechin, or DMSO (solvent control) for 24 h. Caspase-Glo 3/7 reagent (100 µL) was added to each well, mixed, and then incubated at room temperature for 1 h. The luminescence was of each well was detected by using an Infinite M2000 Pro™ (Tecan). The activity of caspase-3/7 was expressed as a percentage (%) of the untreated samples.
4.8. Western Blotting of Caspase-3 and Caspase-7
Cancer cells were treated with bark extracts (IC50) for 24 h then harvested, washed with PBS, lysed in protease-inhibitor cocktail buffer (Roche Diagnostic, Bern, Switzerland). The supernatant was collected by centrifugation (2500 ×g for 15 min), then the protein was extracted and the concentration was determined by the bicinchoninic acid protein assay kit (Sigma-Aldrich, Berlin, Germany). Sample proteins (60–80) µg were separated by SDS-PAGE (10%) then blotted on a Polyvinylidene difluoride (PVDF) membrane. Blocking of proteins was achieved by treating the membrane with 5% skimmed milk + 1XTBS + 0.1% Tween 20 for 60 min. The membrane was treated with caspase-3 (#9661), Caspase-7 primary antibodies (#9492), poly (adenosine diphosphate ribose) polymerase (PARP) (#9542), and GAPDH (internal control, #sc-32233) (1:1,000; Cell Signaling Technology, Danvers, MA, USA) at 4 °C overnight. The membrane was washed then incubated with anti-rabbit (cat. no. sc-2030)/ anti-mouse (cat. no. sc-2005) secondary antibody (1:2,000; Santa Cruz Biotechnology, Inc. Dallas, Texas, USA). The bands were detected by ECL reagent and GE Healthcare Bio-Sciences AB Image Quant LAS 4000 (GE Healthcare, Berlin, Germany). The results shown are representative of three independent experiments.
4.9. Statistical Analyses
The least significant difference (LSD) was computed by using SPSS software (version 22.0, IBM, New York, USA). The quantitative results of the chromatographic analyses are expressed in (mg 100 g−1 DW) as the mean ± SD of three series of experiments.
5. Conclusions
M. acuminata showed significantly higher antioxidant activities than the other species tested and standard antioxidants. In C. speciosa, seven phenolic acids (ferulic acid, caffeic acid, p-hydroxybenzoic acid, p-coumaric acid, gallic acid, protocatechuic acid, and vanillic acid) and catechin were detected by using HPLC-DAD analysis. In T. cuspidata, five phenolics were detected and the dominant compound was hydroxycaffeic acid. In M. acuminata, two phenolic acids (ellagic acid and protocatechuic acid) and three catechins (catechin, epicatechin, and epigallocatechin gallate) were detected; catechin was the predominant compound. The extracts exerted clear anticancer activities against MCF-7, HeLa, Jurkat, T24, and HT-29 cells. The strongest anticancer activity was exerted by the extract of M. acuminata. Further, no antiproliferative activities in normal cells were observed. Flow cytometry showed a greater accumulation of necrotic cells and early and late apoptotic cells in various cancer cell lines treated with the extracts compared with the untreated control cells. Protocatechuic acid resulted in similar accumulation of necrotic cells as the bark extracts of the three species. Increased caspase-3/7 activities were observed in cancer cells treated with different bark extracts and the highest activity was induced by M. acuminata treatment. In conclusion, our results indicate the induction of apoptosis after the treatment of cancer cells with bark extracts of M. acuminata, C. speciosa, T. cuspidata, and protocatechuic acid, and suggest the association between anticancer activities and the individual phenolic constituents.
Acknowledgments
We acknowledge the funding of this work from the Deanship of Scientific Research at King Saud University through research group no. (RG - 1440-12).
Supplementary Materials
The following are available online at http://www.mdpi.com/1420-3049/24/3/412/s1, Figure S1: The representative HPLC-UV chromatogram (λ = 254 nm) of Magnolia acuminnata bark extract; 1 - protocatechuic acid (tR = 6.3 min), 2 – catechin (tR = 8.8 min), 3 - elgaic acid (tR = 12.2 min), 4 - epigallocatechin gallate (tR = 14.8 min), 5 – epicatechin (tR = 19.2 min), Figure S2: The representative HPLC-UV chromatogram (λ = 254 nm) of Taxus cuspidate bark extract; 1 - gallic acid (tR = 3.2 min), 2 - protocatechuic acid (tR = 6.3 min), 3 - chlorogenic acid (tR=9.2 min), 4 - p-hydroxybensoic acid (tR = 11.8 min), 5 - caffeic acid (tR = 15.2 min), 6 - hydroxycaffeic acid (tR = 15.8 min), Figure S3: The representative HPLC-UV chromatogram (λ = 254 nm) of Catalpa speciosa bark extract; 1 - gallic acid (tR = 3.2 min), 2 - protocatechuic acid (tR = 6.3 min), 3 – catechin (tR = 8.8 min), 4- p-hydroxybensoic acid (tR = 11.8 min), 5 - vanillic acid (tR = 12 min), 6 -caffeic acid (tR = 15.2 min), 7 - p-coumaric acid(tR = 26 min), 8 - ferulic acid(tR = 34.4 min).
Author Contributions
H.E., A.S., P.K., F.A., E.M., M.M., T K., and H.E. participated in conducting experiments, writing and approving the final manuscript.
Funding
Funding was approved by the Deanship of Scientific Research at King Saud University through research group no. (RG - 1440-12).
Conflicts of Interest
The authors declare no conflict of interest.
Footnotes
Sample Availability: Not available.
References
- 1.Grace O.M., Prendergast H.D.V., Jager A.K., van Staden J. Bark medicines used in traditional healthcare in KwaZulu-Natal, South Africa: An inventory. S. Afr. J. Bot. 2003;69:301–363. doi: 10.1016/S0254-6299(15)30318-5. [DOI] [Google Scholar]
- 2.Bello I., Shehu M.W., Musa M., Asmawi M.Z., Mahmud R. Kigelia africana (Lam.) Benth. (Sausage tree): Phytochemistry and pharmacological review of a quintessential African traditional medicinal plant. J. Ethnopharmacol. 2016;189:253–276. doi: 10.1016/j.jep.2016.05.049. [DOI] [PubMed] [Google Scholar]
- 3.Vermeulen W.J., Geldenhuys C.J., Esler K.J. Response of Ocotea bullata, Curtisia dentata and Rapanea melanophloeos to medicinal bark stripping in the southern Cape, South Africa: Implications for sustainable use. South. For. 2012;74:183–193. doi: 10.2989/20702620.2012.717384. [DOI] [Google Scholar]
- 4.Chuang D.Y., Chan M.H., Zong Y., Sheng W., He Y., Jiang J.H., Simonyi A., Gu Z., Fritsche K.L., Cui J., et al. Magnolia polyphenols attenuate oxidative and inflammatory responses in neurons and microglial cells. J. Neuroinflamm. 2013;10:15. doi: 10.1186/1742-2094-10-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Juyal D., Thawani V., Thaledi S., Joshi M. Ethnomedical properties of Taxus wallichiana zucc. (Himalayan yew) J. Tradit. Complement. Med. 2014;4:159–161. doi: 10.4103/2225-4110.136544. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Yu S.X., Yan R.Y., Liang R.X., Wang W., Yang B. Bioactive polar compounds from stem bark of Magnolia officinalis. Fitoterapia. 2012;83:356–361. doi: 10.1016/j.fitote.2011.11.020. [DOI] [PubMed] [Google Scholar]
- 7.Hemmings E.T., Core E.L. Archeological Evidence for Range Extension of the Catawba Tree (Catalpa speciosa Warder) in West Virginia. Castanea. 1976;41:9–11. [Google Scholar]
- 8.Xu H., Hu G., Dong J., Wei Q., Shao H., Lei M. Antioxidative Activities and Active Compounds of Extracts from Catalpa Plant Leaves. Sci. World J. 2014;2014:857982. doi: 10.1155/2014/857982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Dvorska M., Zemlicka M., Muselik J., Karafiatova J., Suchy V. Antioxidant activity of Catalpa bignonioides. Fitoterapia. 2007;78:437–439. doi: 10.1016/j.fitote.2007.03.025. [DOI] [PubMed] [Google Scholar]
- 10.Lee Y.J., Lee Y.M., Lee C.K., Jung J.K., Han S.B., Hong J.T. Therapeutic applications of compounds in the Magnolia family. Pharmacol. Ther. 2011;130:157–176. doi: 10.1016/j.pharmthera.2011.01.010. [DOI] [PubMed] [Google Scholar]
- 11.European Directorate for the Quality of Medicines and Healthcare . European Pharmacopoeia 9.0. Council of Europe; Strasbourg, France: 2017. [Google Scholar]
- 12.Park J., Lee J., Jung E.S., Park Y., Kim K., Park B., Jung K.S., Park E., Kim J., Park D. In vitro antibacterial and anti-inflammatory effects of honokiol and magnolol against Propionibacterium sp. Eur. J. Pharmacol. 2004;496:189–195. doi: 10.1016/j.ejphar.2004.05.047. [DOI] [PubMed] [Google Scholar]
- 13.Farjon A. World Checklists and Bibliographies, 3. 2nd ed. Royal Botanic Gardens, Kew; Kew, UK: 2001. World Checklist and Bibliography of Conifers; p. 300. [Google Scholar]
- 14.Kawamura F., Kikuchi Y., Ohira T., Yatagai M. Accelerated solvent extraction of paclitaxel and related compounds from the bark of Taxus cuspidata. J. Nat. Prod. 1999;62:244–247. doi: 10.1021/np980310j. [DOI] [PubMed] [Google Scholar]
- 15.Kawamura F., Kikuchi Y., Ohira T., Yatagai M. Phenolic constituents of Taxus cuspidata I: Lignans from the roots. J. Wood Sci. 2000;46:167–171. doi: 10.1007/BF00777366. [DOI] [Google Scholar]
- 16.Shen C.C., Ni C.L., Shen Y.C., Huang Y.L., Kuo C.H., Wu T.S., Chen C.C. Phenolic constituents from the stem bark of Magnolia officinalis. J. Nat. Prod. 2009;72:168–171. doi: 10.1021/np800494e. [DOI] [PubMed] [Google Scholar]
- 17.Grzesik M., Naparlo K., Bartosz G., Sadowska-Bartosz I. Antioxidant properties of catechins: Comparison with other antioxidants. Food Chem. 2018;241:480–492. doi: 10.1016/j.foodchem.2017.08.117. [DOI] [PubMed] [Google Scholar]
- 18.Kerio L.C., Wachira F.N., Wanyoko J.K., Rotich M.K. Total polyphenols, catechin profiles and antioxidant activity of tea products from purple leaf coloured tea cultivars. Food Chem. 2013;136:1405–1413. doi: 10.1016/j.foodchem.2012.09.066. [DOI] [PubMed] [Google Scholar]
- 19.Oh S., Gadde U.D., Bravo D., Lillehoj E.P., Lillehoj H.S. Growth-Promoting and Antioxidant Effects of Magnolia Bark Extract in Chickens Uninfected or Co-Infected with Clostridium perfringens and Eimeria maxima as an Experimental Model of Necrotic Enteritis. Curr. Dev. Nutr. 2018;2:nzy009. doi: 10.1093/cdn/nzy009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Zhang M., Cheng S., Liang Y., Mu Y., Yan H., Liu Q., Geng Y., Wang X., Zhao H. Rapid purification of antioxidants from Magnolia officinalis by semi-prep-HPLC with a two-step separation strategy guided by on-line HPLC-radical scavenging detection. J. Chromatogr. B. 2018;1100–1101:140–147. doi: 10.1016/j.jchromb.2018.09.030. [DOI] [PubMed] [Google Scholar]
- 21.Huang H.C., Hsieh W.Y., Niu Y.L., Chang T.M. Inhibition of melanogenesis and antioxidant properties of Magnolia grandiflora L. flower extract. BMC Complement. Altern. Med. 2012;12:72. doi: 10.1186/1472-6882-12-72. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Zhang D., Dong W., Zhang D., Jin L., Zhang D., Zhang J., Tao S. Screening of Natural Antioxidants and Application In the Course of Perry Brewing. Energy Procedia. 2012;17:1811–1816. doi: 10.1016/j.egypro.2012.02.316. [DOI] [Google Scholar]
- 23.Veselova M.V., Fedoreev S.A., Vasilevskaya N.A., Denisenko V.A., Gerasimenko A.V. From leaf to flower: Revisiting Goethe’s concepts on the “metamorphosis” of plants. Pharm. Chem. J. 2007;41:88–93. doi: 10.1007/s11094-007-0019-0. [DOI] [Google Scholar]
- 24.Kikuzaki H., Hisamoto M., Hirose K., Akiyama K., Taniguchi H. Antioxidant properties of ferulic acid and its related compounds. J. Agric. Food Chem. 2002;50:2161–2168. doi: 10.1021/jf011348w. [DOI] [PubMed] [Google Scholar]
- 25.Semaming Y., Pannengpetch P., Chattipakorn S.C., Chattipakorn N. Pharmacological Properties of Protocatechuic Acid and Its Potential Roles as Complementary Medicine. Evid.-Based Complement. Altern. 2015;2015:593902. doi: 10.1155/2015/593902. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Xi X.J., Hu S.Q., Zhou Z.X., Liu X.R., Tang J.B., Shen Y.Q. Dendrimers with the protocatechuic acid building block for anticancer drug delivery. J. Mater. Chem. B. 2016;4:5236–5245. doi: 10.1039/C6TB01597B. [DOI] [PubMed] [Google Scholar]
- 27.Yu Y., Deng Y., Lu B.M., Liu Y.X., Li J., Bao J.K. Green tea catechins: A fresh flavor to anticancer therapy. Apoptosis. 2014;19:1–18. doi: 10.1007/s10495-013-0908-5. [DOI] [PubMed] [Google Scholar]
- 28.Manikandan R., Beulaja M., Arulvasu C., Sellamuthu S., Dinesh D., Prabhu D., Babu G., Vaseeharan B., Prabhu N.M. Synergistic anticancer activity of curcumin and catechin: An in vitro study using human cancer cell lines. Microsc. Res. Tech. 2012;75:112–116. doi: 10.1002/jemt.21032. [DOI] [PubMed] [Google Scholar]
- 29.Abdulkhaleq L.A., Assi M.A., Noor M.H.M., Abdullah R., Saad M.Z., Taufiq-Yap Y.H. Therapeutic uses of epicatechin in diabetes and cancer. Vet. World. 2017;10:869–872. doi: 10.14202/vetworld.2017.869-872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Takanashi K., Suda M., Matsumoto K., Toda C.I.K., Toda K., Kawaguchi K., Senga S., Kobayashi N., Ichikawa M., Katoh M., et al. Epicatechin oligomers longer than trimers have anti-cancer activities, but not the catechin counterparts. Sci. Rep. 2017;7:7791. doi: 10.1038/s41598-017-08059-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Azam S., Hadi N., Khan N.U., Hadi S.M. Prooxidant property of green tea polyphenols epicatechin and epigallocatechin-3-gallate: Implications for anticancer properties. Toxicol. In Vitro. 2004;18:555–561. doi: 10.1016/j.tiv.2003.12.012. [DOI] [PubMed] [Google Scholar]
- 32.Arora S., Singh S., Piazza G.A., Contreras C.M., Panyam J., Singh A.P. Honokiol: A Novel Natural Agent for Cancer Prevention and Therapy. Curr. Mol. Med. 2012;12:1244–1252. doi: 10.2174/156652412803833508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Zhang X., Lin D., Jiang R., Li H., Wan J., Li H. Ferulic acid exerts antitumor activity and inhibits metastasis in breast cancer cells by regulating epithelial to mesenchymal transition. Oncol. Rep. 2016;36:271–278. doi: 10.3892/or.2016.4804. [DOI] [PubMed] [Google Scholar]
- 34.Gao J., Yu H., Guo W., Kong Y., Gu L., Li Q., Yang S., Zhang Y., Wang Y. The anticancer effects of ferulic acid is associated with induction of cell cycle arrest and autophagy in cervical cancer cells. Cancer Cell Int. 2018;18:102. doi: 10.1186/s12935-018-0595-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Spilioti E., Jaakkola M., Tolonen T., Lipponen M., Virtanen V., Chinou I., Kassi E., Karabournioti S., Moutsatsou P. Phenolic acid composition, antiatherogenic and anticancer potential of honeys derived from various regions in Greece. PLoS ONE. 2014;9:e94860. doi: 10.1371/journal.pone.0094860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Elansary H.O., Szopa A., Kubica P., Ekiert H., Ali H.M., Elshikh M.S., Abdel-Salam E.M., El-Esawi M., El-Ansary D.O. Bioactivities of Traditional Medicinal Plants in Alexandria. Evid.-Based Complement. Altern. Med. 2018;2018:1463579. doi: 10.1155/2018/1463579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Shang W., Qiao J., Gu C., Yin W., Du J., Wang W., Zhu M., Han M., Lu W. Anticancer activity of an extract from needles and twigs of Taxus cuspidata and its synergistic effect as a cocktail with 5-fluorouracil. BMC Complement. Altern. Med. 2011;11:123. doi: 10.1186/1472-6882-11-123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Marin G.H., Mansilla E. Apoptosis induced by Magnolia Grandi fl ora extract in chlorambucil-resistant B-chronic lymphocytic leukemia cells. J. Cancer Res. Ther. 2010;6:463–465. doi: 10.4103/0973-1482.77107. [DOI] [PubMed] [Google Scholar]
- 39.Zhang X.D., Wu Q., Yang S.H. Ferulic acid promoting apoptosis in human osteosarcoma cell lines. Pak. J. Med. Sci. 2017;33:127–131. doi: 10.12669/pjms.331.12066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Yin M.C., Lin C.C., Wu H.C., Tsao S.M., Hsu C.K. Apoptotic effects of protocatechuic acid in human breast, lung, liver, cervix, and prostate cancer cells: Potential mechanisms of action. J. Agric. Food Chem. 2009;57:6468–6473. doi: 10.1021/jf9004466. [DOI] [PubMed] [Google Scholar]
- 41.Alvarado-Sansininea J.J., Sanchez-Sanchez L., Lopez-Munoz H., Escobar M.L., Flores-Guzman F., Tavera-Hernandez R., Jimenez-Estrada M. Quercetagetin and Patuletin: Antiproliferative, Necrotic and Apoptotic Activity in Tumor Cell Lines. Molecules. 2018;23:2579. doi: 10.3390/molecules23102579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Bell R.A.V., Megeney L.A. Evolution of caspase-mediated cell death and differentiation: Twins separated at birth. Cell Death Differ. 2017;24:1359–1368. doi: 10.1038/cdd.2017.37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Turner C., Devitt A., Parker K., MacFarlane M., Giuliano M., Cohen G.M., Gregory C.D. Macrophage-mediated clearance of cells undergoing caspase-3-independent death. Cell Death Differ. 2003;10:302–312. doi: 10.1038/sj.cdd.4401170. [DOI] [PubMed] [Google Scholar]
- 44.Kagawa S., Gu J., Honda T., McDonnell T.J., Swisher S.G., Roth J.A., Fang B. Deficiency of Caspase-3 in MCF7 Cells Blocks Bax-mediated Nuclear Fragmentation but not Cell Death. Clin. Cancer Res. 2001;7:1474–1480. [PubMed] [Google Scholar]
- 45.Sung M.H., Kwon O.K., Oh S.R., Lee J., Park S.H., Han S.B., Ahn K.S. Azorella compacta methanolic extract induces apoptosis via activation of mitogen-activated protein kinase. Mol. Med. Rep. 2015;12:6821–6828. doi: 10.3892/mmr.2015.4317. [DOI] [PubMed] [Google Scholar]
- 46.Wu B.T., Hung P.F., Chen H.C., Huang R.N., Chang H.H., Kao Y.H. The apoptotic effect of green tea (-)-epigallocatechin gallate on 3T3-L1 preadipocytes depends on the Cdk2 pathway. J. Agric. Food Chem. 2005;53:5695–5701. doi: 10.1021/jf050045p. [DOI] [PubMed] [Google Scholar]
- 47.Spencer J.P., Schroeter H., Kuhnle G., Srai S.K., Tyrrell R.M., Hahn U., Rice-Evans C. Epicatechin and its in vivo metabolite, 3’-O-methyl epicatechin, protect human fibroblasts from oxidative-stress-induced cell death involving caspase-3 activation. Biochem. J. 2001;354:493–500. doi: 10.1042/bj3540493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Elbaz H.A., Lee I., Antwih D.A., Liu J., Huttemann M., Zielske S.P. Epicatechin stimulates mitochondrial activity and selectively sensitizes cancer cells to radiation. PLoS ONE. 2014;9:e88322. doi: 10.1371/journal.pone.0088322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Yang J.S., Liu C.W., Ma Y.S., Weng S.W., Tang N.Y., Wu S.H., Ji B.C., Ma C.Y., Ko Y.C., Funayama S., et al. Chlorogenic acid induces apoptotic cell death in U937 leukemia cells through caspase- and mitochondria-dependent pathways. In Vivo. 2012;26:971–978. [PubMed] [Google Scholar]
- 50.Ji B.C., Hsu W.H., Yang J.S., Hsia T.C., Lu C.C., Chiang J.H., Yang J.L., Lin C.H., Lin J.J., Suen L.J., et al. Gallic acid induces apoptosis via caspase-3 and mitochondrion-dependent pathways in vitro and suppresses lung xenograft tumor growth in vivo. J. Agric. Food Chem. 2009;57:7596–7604. doi: 10.1021/jf901308p. [DOI] [PubMed] [Google Scholar]
- 51.Szopa A., Kokotkiewicz A., Kubica P., Banaszczak P., Wojtanowska-Krosniak A., Krosniak M., Marzec-Wroblewska U., Badura A., Zagrodzki P., Bucinski A., et al. Comparative analysis of different groups of phenolic compounds in fruit and leaf extracts of Aronia sp.: A-melanocarpa, A-arbutifolia, and A. xprunifolia and their antioxidant activities. Eur. Food Res. Technol. 2017;243:1645–1657. doi: 10.1007/s00217-017-2872-8. [DOI] [Google Scholar]
- 52.Ellnain-Wojtaszek M., Zgorka G. High-performance liquid chromatography and thin-layer chromatography of phenolic acids from Ginkgo biloba L-leaves collected within vegetative period. J. Liq. Chromatogr. Relat. Technol. 1999;22:1457–1471. doi: 10.1081/JLC-100101744. [DOI] [Google Scholar]
- 53.Sulkowska-Ziaja K., Maslanka A., Szewczyk A., Muszynska B. Physiologically Active Compounds in Four Species of Phellinus. Nat. Prod. Commun. 2017;12:363–366. [PubMed] [Google Scholar]
- 54.Szopa A., Kokotkiewicz A., Bednarz M., Luczkiewicz M., Ekiert H. Studies on the accumulation of phenolic acids and flavonoids in different in vitro culture systems of Schisandra chinensis (Turcz.) Baill. using a DAD-HPLC method. Phytochem. Lett. 2017;20:462–469. doi: 10.1016/j.phytol.2016.10.016. [DOI] [Google Scholar]
- 55.Szopa A., Ekiert H., Szewczyk A., Fugas E. Production of bioactive phenolic acids and furanocoumarins in in vitro cultures of Ruta graveolens L. and Ruta graveolens ssp. divaricata (Tenore) Gams. under different light conditions. Plant Cell Tissue Org. 2012;110:329–336. doi: 10.1007/s11240-012-0154-5. [DOI] [Google Scholar]
- 56.Elansary H.O., Salem M.Z.M., Ashmawy N.A., Yessoufou K., El-Settawy A.A.A. In vitro antibacterial, antifungal and antioxidant activities of Eucalyptus spp. leaf extracts related to phenolic composition. Nat. Prod. Res. 2017;31:2927–2930. doi: 10.1080/14786419.2017.1303698. [DOI] [PubMed] [Google Scholar]
- 57.Mosmann T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods. 1983;65:55–63. doi: 10.1016/0022-1759(83)90303-4. [DOI] [PubMed] [Google Scholar]
- 58.Parry J., Su L., Moore J., Cheng Z., Luther M., Rao J.N., Wang J.Y., Yu L.L. Chemical compositions, antioxidant capacities, and antiproliferative activities of selected fruit seed flours. J. Agric. Food Chem. 2006;54:3773–3778. doi: 10.1021/jf060325k. [DOI] [PubMed] [Google Scholar]
- 59.Komina A., Palkina N., Aksenenko M., Tsyrenzhapova S., Ruksha T. Antiproliferative and Pro-Apoptotic Effects of MiR-4286 Inhibition in Melanoma Cells. PLoS ONE. 2016;11:e0168229. doi: 10.1371/journal.pone.0168229. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.