Abstract
Background:
Fetal tracheal occlusion (TO) is an emerging surgical therapy in congenital diaphragmatic hernia that improves the fetal lung growth. Different animal models of congenital diaphragmatic hernia and TO present advantages and disadvantages regarding ethical issues, cost, surgical difficulty, size, survival rates, and available genetic tools. We developed a minimally invasive murine transuterine TO model, which will be useful in defining how TO impacts lung molecular biology, cellular processes, and overall lung physiology.
Materials and methods:
Time-mated C57BL/6 mice underwent laparotomy at embryonic day 16.5 (E16.5) with transuterine TO performed on two fetuses in each uterine horn. At E18.5, dams were sacrificed and fetuses harvested. The lungs of the TO fetuses were compared with the nonmanipulated counterparts by morphometric and histologic analysis.
Results:
Successful TO was confirmed in 16 of 20 TO fetuses. Twelve of them survived to E18.5 (75%). Fetal weights were comparable, but lung weights were significantly greater in TO (28.41 ± 5.87 versus 23.38 ± 3.09, P = 0.043). Lung to body weight ratio was also greater (0.26 ± 0.003 versus 0.22 ± 0.002, P = 0.006). E18.5 TO lungs demonstrated dilated central and distal airspaces with increased cellularity. DNA/protein and DNA/lung weight ratios were elevated while protein/lung weight ratio was lower in TO compared to control.
Conclusions:
Mice fetal transuterine TO is feasible with comparable outcomes to other current animal models. The increase in the lung weight, lung to body weight ratio and the DNA/protein ratio indicate organized lung growth rather than edema or cell hypertrophy.
Keywords: CHAOS, Congenital diaphragmatic hernia, Fetal lung development, Fetal tracheal occlusion, Lung growth, Mice
Introduction
Fetal tracheal occlusion (TO) is an emerging prenatal therapy for severe congenital diaphragmatic hernia (CDH).1 TO prevents the egress of pulmonary fluid, thereby increasing tissue stretch and accelerating lung growth, and may ameliorate the pulmonary hypoplasia of CDH. In animals and humans developing CDH, their lungs experience increased growth after fetal TO during the canalicular and saccular phases of their development.2 The persistently high mortality of CDH over the past decades, despite advances in neonatal resuscitation and intensive care, increases the need for more investigation into the pathophysiology and prenatal treatment of the disease. To better understand how TO impacts human lung growth and development in CDH, mouse, rat, rabbit, and sheep models of TO have been developed.1,3–5 Each model presents advantages and disadvantages regarding ethical issues; cost; surgical difficulty; animal, fetus, and lung size; survival rates; and available genetic tools.4 However, each of these prior models requires hysterotomy for TO. Herein, we present an easy and minimally invasive murine TO model, which will be useful in defining how TO impacts in lung molecular biology, cellular processes, and overall lung physiology both in normal and CDH lungs.
Material and methods
Experimental design
Following approval of IACUC protocol #2016–0068 by the Cincinnati Children’s Research Foundation Institutional Animal Care and Use Committee, age-matched wild-type C57BL/6 mice were mated, and the date when the vaginal plug is seen was accepted as embryonic day 0 (E0). At E16.5, pregnant dams underwent laparotomy and transuterine TO was performed upon two fetuses in each uterine horn with 2.5× magnification. At E18.5, all dams were harvested by cesarean section and fetuses were weighed by using a scale measuring accurately up to 0.001 g (Mettler Toledo, Columbus, OH). Lungs were dissected from mouse embryos and were weighed to calculate total lung to body weight ratio (LBWR). Normal fetuses of the litter served as nonmanipulated controls.
TO surgical method
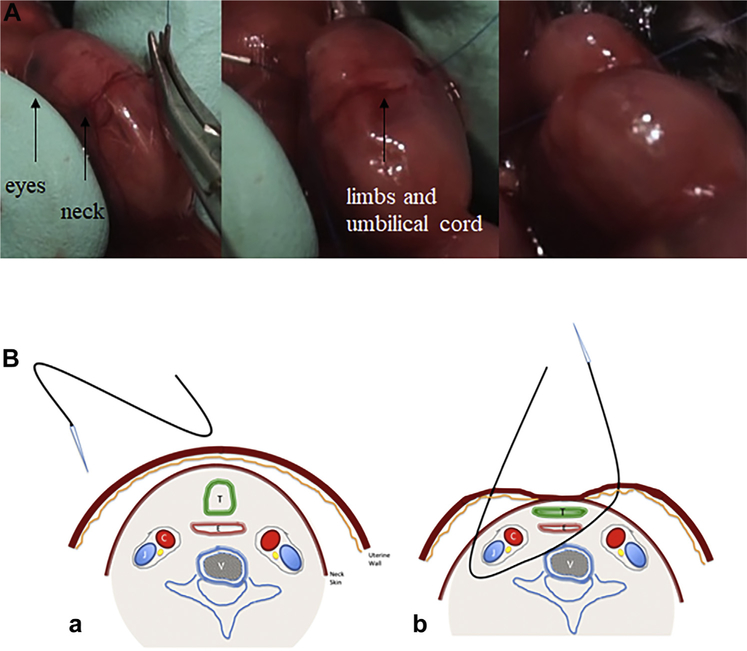
After performing a median laparotomy to the dams under general anesthesia, the uterine horn was positioned gently in a transverse fashion with the pups facing upward between two fingers of the surgeon. Using gentle pressure, pups’ heads were extended allowing visualization of the neck. A 6.0 polypropylene suture (13 mm 1/2c Taperpoint; Ethicon, Sommerville, NJ) with an atraumatic needle was used for TO. The needle was inserted through the side of the uterus opposite to the placenta, through 1/3 anterior of the neck in a transverse fashion (Fig. 1A). The tip of the needle was gently advanced till it passes the midline of the pup where it was directed through the anterior of the neck and exits the neck between the trachea and opposite carotid sheath and uterus, respectively (Fig. 1B). Finally, it was knotted gently without tearing the membranes and uterine wall and avoiding the umbilical cord (Video 1). The dam’s abdominal wall and skin were then closed. Buprenorphine 0.1 mg/kg was administered intraperitoneally for analgesia, and the pregnant mouse was subsequently allowed to be recovered.
Fig. 1 –
(A) The transuterine bypassing of the suture through the neck. (B) Schematic representation of the transuterine suture through the neck, respecting one side vascular (carotid and jugular) elements and surrounding trachea. (a) The entrance of the needle. (b) The structures included by the suture. C = carotid artery; J = jugular vein; T = trachea; E = esophagus; V = vertebra.
Histology
Right and left lungs were snap frozen in optimum cutting temperature (Sakura, NL) and snap frozen on dry ice. Ten micrometer sections were cut using cryostat and mounted on polylysine slides. Slides were baked at 60°C overnight, stained with hematoxylin and eosin, and mounted. Tile-scanned 4× and individual 20× images were obtained using a Nikon 90i wide-field microscope and Nikon Elements software.
Tissue processing for protein and DNA
Dissected fetal lungs were snap-frozen and then homogenized in 300 mL of RIPA buffer (Sigma-Aldrich, St Louis, MO) and centrifuged. Trizol (Life Technologies, Carlsbad, CA) was used to extract DNA, RNA, and protein as per manufacturer instructions. Protein was quantified using Bradford assay, and DNA and RNA concentrations were determined by NanoDrop (both Thermo Fisher, Waltham, MA) using previously published methods.6
Statistical analysis
Data were normalized to fetal body weights, lung weights, and lung protein content and expressed as means ± standard deviations. Statistical analysis was performed using Student’s t-test, with P-values of less than 0.05 considered significant. Data presented in bar chart form represent mean ± standard error of the mean.
Results
Gross results
There were 20 fetuses in the TO group and 17 fetuses in the control group. Four of the mice in the TO group were excluded due to unsuccessful TO, which were unable to be trachea occluded with the transuterine suture. The survival rate was comparable in both groups, 12 fetuses (75%) in the TO group and 15 fetuses (88%) in the control group survived the surgery (P = 0.334, odds ratio 2.5 95% confidence interval 0.39–16.05).
Morphometric results
The mean body weight, lung weight, and LBWR were all higher in TO fetuses than the control group (Table 1). When normalized to the mean LBWR of the control group, TO fetuses had significantly higher LBWR (Fig. 2A) (P = 0.006).
Table 1 –
Morphometrical results of groups.
| Parameters | TO | Control | P |
|---|---|---|---|
| Fetus weight (mg) | 1100.52 ± 229.38 | 1087.15 ± 172.32 | 0.896 |
| Lung weight (mg) | 28.41 ± 5.87 | 23.38 ± 3.09 | 0.043 |
| LBWR | 0.0259 ± 0.0021 | 0.0217 ± 0.0028 | 0.006* |
Values expressed as means ± standard deviations.
95% confidence interval 0.0222–0.0249.
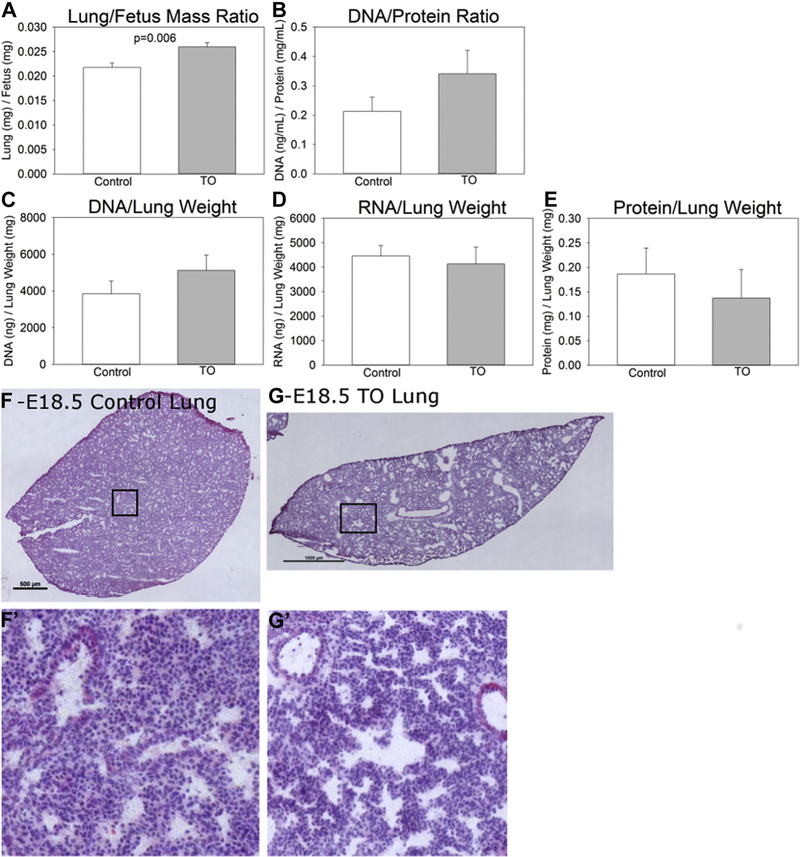
Fig. 2 ‒
Model characteristics. (A) Normalized fetal lung to body weight ratio. (B) Lung DNA to protein ratio. (C) Lung DNA, (D) RNA, and (E) protein content normalized to lung weight. Hematoxylin and eosin images of C57BL/6 E18.5 lungs (F) without (non-dilated airways) and (G) with (dilated airways) fetal transuterine tracheal occlusion.
Learning the model
The impetus to develop this technique derived from poor fetal survival using a previously published technique that involves hysterotomy and delivery of the fetal head.5 Our first three surgical attempts were unsuccessful with one mouse dying under anesthesia and two mice requiring euthanasia due to premature labor. The subsequent six surgeries were successful with >85% fetus survival noted at the time of collection. The average operation time was reduced from 90 to 45 min per dam over the course of this study suggesting that reduced surgical times and reduced uterine manipulation are key factors in the success of this model.
Lung DNA, RNA, and protein content
Because LBWR were increased in TO mice, we sought to determine whether this increase in lung size and an apparent increase in the relative number of nuclei were due to hyperplasia. DNA/protein ratios were increased in TO lungs (Fig. 2B), lung DNA tended to be increased (Fig. 2C), while lung RNA was no different (Fig. 2D), and protein tended to be decreased (Fig. 2E) in the TO group compared to controls.
Histology
TO lungs qualitatively showed an increase in airway diameter compared to control lungs; however, this was not specifically measured. The lungs of E18.5 control fetuses show histologic findings of lungs in the late canalicular/early saccular stage of lung development with developing airspaces and thickened interstitium between epithelial surfaces. E18.5 TO lungs show dilated central and distal airspaces with subjectively increased numbers of nuclei (Fig. 2F and G). This increased cellularity is consistent with the noted increase in lung DNA.
Discussion
It has long been known that naturally occurring airway occlusion secondary to atresia or stenosis of the larynx, trachea, or bronchus results in large fluid-filled lungs.7,8 Research done on animals have demonstrated that TO accelerates lung growth in normal lungs and in models of lung hypoplasia caused by oligohydramnios, bilateral nephrectomy, and CDH.7,9,10 We have described a novel and minimally invasive mouse surgical model of fetal TO. Our results confirm that fetal transuterine TO is feasible in the mice. The increases in the lung weight, LBWR, the DNA/protein ratio, and total DNA/body weight ratio are consistent with two prior reports in rabbit models that TO induces lung hyperplasia indicating that the phenotype of this new model is in agreement with previously reported ones.11,12
The learning curve in this study was relatively short, and fetal mortality dramatically decreased as we improved our technique. The principal limitations of the sheep and rabbit models of CDH and TO are related to the animal purchase and maintenance expenses, the small number of fetuses per dam, the long gestation, and the paucity of genetic tools available in these larger animals. Our technique offers several advantages over larger animal CDH/TO models. First, transuterine TO does not require a hysterotomy, which probably accounts for the high rates of fetal survival that we observed—levels that are comparable to other animal models of TO. Second, the reduced animal and housing costs and shorter gestational period of mice facilitate more highly powered studies. Third, shorter surgery time prevents complications due to hypothermia and other anesthetic duration. Finally, the transgenic tools available in the mouse in combination with the nitrofen model of CDH now permit a host of important studies to be performed such as lineage tracing of selected cell populations, general and tissue-specific gene manipulation. Our model offers an advantage over previously published mouse models in that it does not require a hysterotomy and could potentially be reversed by suture removal allowing delivery and possibly the survival of the TO fetus.13 Therefore, this model will not only enable us to study TO in the nitrofen and knockout models of CDH but also may let us work on transient TO that will mimic the human counterpart.
Mice have also the advantage to provide transgenic models for CDH and offer vast possibilities to analyze molecular pathways. Now, the option to have a feasible method to perform a minimally invasive fetal TO in these tiny animals opens new frontiers in the study of prenatal therapies for CDH.
Supplementary Material
Acknowledgments
Funding: This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Footnotes
Financial disclosure: There is no financial disclosure to be declared.
Ethical approval: All experiments have complied with the National Institutes of Health Guide for the care and use of Laboratory animals (NIH Publications No. 80023, revised 1978).
Disclosure
The authors reported no proprietary or commercial interest in any product mentioned or concept discussed in this article.
Supplementary data
Supplementary data related to this article can be found at https://doi.org/10.1016/j.jss.2018.04.028.
REFERENCES
- 1.Beck V, Davey MG, Mayer S, et al. A longer tracheal occlusion period results in increased lung growth in the nitrofen rat model. Prenat Diagn. 2012;32:39–44. [DOI] [PubMed] [Google Scholar]
- 2.Muensterer OJ, Nicola T, Farmer S, Harmon CM, Ambalavanan N. Temporary fetal tracheal occlusion using a gel plug in a rabbit model of congenital diaphragmatic hernia. J Pediatr Surg. 2012;47:1063–1066. [DOI] [PubMed] [Google Scholar]
- 3.Kitano Y, Davies P, von Allmen D, Adzick NS, Flake AW. Fetal tracheal occlusion in the rat model of nitrofen-induced congenital diaphragmatic hernia. J Appl Physiol (1985). 1999;87:769–775. [DOI] [PubMed] [Google Scholar]
- 4.Sananès N, Ruano R, Weingertner A-S, et al. Experimental fetal endoscopic tracheal occlusion in rhesus and cynomolgus monkeys: nonhuman primate models. J Matern Fetal Neonatal Med 2015;28:1822–1827. [DOI] [PubMed] [Google Scholar]
- 5.Maltais F, Seaborn T, Guay S, Piedboeuf B. In vivo tracheal occlusion in fetal mice induces rapid lung development without affecting surfactant protein C expression. Am J Physiol Lung Cell Mol Physiol. 2003;284:L622–L632. [DOI] [PubMed] [Google Scholar]
- 6.Chomczynski P A reagent for the single-step simultaneous isolation of RNA, DNA and proteins from cell and tissue samples. Biotechniques. 1993;15:532–534, [PubMed] [Google Scholar]
- 7.Kitano Y, Yang EY, von Allmen D, Quinn TM, Adzick NS, Flake AW. Tracheal occlusion in the fetal rat: a new experimental model for the study of accelerated lung growth. J Pediatr Surg. 1998;33:1741–1744. [DOI] [PubMed] [Google Scholar]
- 8.Wu CS, Chen CM, Chou HC. Pulmonary hypoplasia induced by oligohydramnios: findings from animal models and a population-based study. Pediatr Neonatol. 2017;58:3–7. [DOI] [PubMed] [Google Scholar]
- 9.Kitano Y, Flake AW, Quinn TM, et al. Lung growth induced by tracheal occlusion in the sheep is augmented by airway pressurization. J Pediatr Surg. 2000;35:216–222. [DOI] [PubMed] [Google Scholar]
- 10.Difiore BJW, Fauza D, Slavin R, et al. Experimental fetal tracheal ligation reversess the structural and physiological effects of pulmonary hypoplasia in congenital diaphragmatic hernia. J Pediatr Surg. 1994;29:248–256; [DOI] [PubMed] [Google Scholar]
- 11.Engels AC, Brady PD, Kammoun M, et al. Pulmonary transcriptome analysis in the surgically induced rabbit model of diaphragmatic hernia treated with fetal tracheal occlusion. Dis Model Mech. 2016;9:221–228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Varisco BM, Sbragia L. Excessive reversal of epidermal growth factor receptor and ephrin signaling following tracheal occlusion in rabbit model of congenital diaphragmatic hernia. Mol Med 2016;22:1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Beurskens N, Klaassens M, Rottier R, De Klein A, Tibboel D. Linking animal models to human congenital diaphragmatic hernia. Birth Defects Res A Clin Mol Teratol 2007;79:565–572. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.