Abstract
Dihydromyricetin is the major flavonoid in vine tea, whose pharmacological action has attracted increasing attention in recent years. The triglyceride, albumin (ALB), alanine aminotransferase, aspartate aminotransferase, malondialdehyde, superoxide dismutase, glutathione (GSH), GSH peroxidase, tumor necrosis factor-α, interleukin (IL)-1β, IL-6 and IL-18 expression levels were measured using enzyme-linked immunosorbent assay kits. The protein levels of ALB and collagen I, PPARα, NF-κB, p53 and Bax were used to measure using western blotting. The results revealed that dihydromyricetin prevented the development of fatty liver, and inhibited oxidative stress, inflammation and apoptosis in a fatty liver rat model. In addition, treatment with dihydromyricetin inhibited the levels of ALB and collagen I, while it induced peroxisome proliferator-activated receptor α protein expression. Dihydromyricetin also suppressed the protein expression levels of nuclear factor (NF)-κB, p53 and B-cell lymphoma 2-associated X protein (Bax) in the rat model. Collectively, it is concluded that dihydromyricetin exerted a protective effect on fatty liver through NF-κB/p53/Bax signaling pathways in a rat model.
Keywords: dihydromyricetin, fatty liver, nuclear factor-κB, p53, B-cell lymphoma 2-associated X protein
Introduction
Dihydromyricetin, also known as ampelopsin, is a dihydroflavonol flavonoid that is found in ampelopsis, and its content in vine tea can reach up to 30% (1). It is also distributed in Myricaceae, Guttiferae, Euphorbiaceae, Burseraceae, Leguminosae, Sapotaceae and Clethraceae plants (2). Previous studies have verified that dihydromyricetin possesses multiple pharmacological effects, including antitumor, anti-inflammation, anti-oxidation, anti-alcohol, liver protection, anti-pathogen and blood lipid regulation activities (3). In addition, dihydromyricetin possesses bioactivities, such as an anti-hypertension effect, thrombosis suppression in vivo and hypoglycemic action (2).
The toxic effect of alcohol affects the vital organs of the body, particularly the liver. It has been reported that the overall and per capita alcohol consumption have been increasing (4). Therefore, the harmful effects of alcohol on the human body, and more specifically on the liver, have attracted increasing attention (5). Alcoholic liver disease (ALD) is a liver disease induced by long-term heavy drinking (5). Generally, fatty liver is the initial manifestation of ALD (6), which can then progress into alcoholic hepatitis and alcoholic fibrosis as alcohol consumption continues (6), finally leading to alcoholic cirrhosis and hepatocellular carcinoma. In the presence of alcohol stimulation, excessive lipid input results in liver lipoprotein synthesis disorder and insufficient fatty acid oxidation. As a result, the fat is deposited in the liver, thus giving rise to fatty liver (6).
Nuclear factor (NF)-κB is a common nuclear transcription factor in cells, with p50/p65 as the heterodimer form. NF-κB at an inactivated state can bind with the IkB inhibitor (7). It is involved in immune and inflammatory reactions, in the pathological and acute stage response of certain diseases, and in responses to cell proliferation and viral infection (7). In addition, NF-κB is widely involved in gene transcription and apoptosis (7); therefore, it has a relatively high expression in numerous diseases. Studies have suggested that NF-κB is a promising novel target of anti-inflammatory treatment. Therefore, the present study aimed to determine the effect of dihydromyricetin on fatty liver and its possible mechanism.
Materials and methods
Experimental animals
The study was conducted in accordance with the Guidelines for Animal Experimentation and approval obtained by the ethics committee of the Sixth People's Hospital of Qingdao (Qingdao, China). Male Sprague-Dawley rats (weight, 170±20 g; age, 4–5 weeks) were obtained from Beijing Vital River Laboratory Animal Technology Co., Ltd. (Beijing, China). The rats were maintained at 22–23°C, 55–60% humidity and a 12/12-h light/dark cycle, and provided with certified standard chow and tap water ad libitum. Rats were randomly assigned to three groups, including the normal (n=6), model (n=7) and dihydromyricetin groups (n=7). In the model and dihydromyricetin groups, rats were fed with a high-fat diet (36.5% fat, 44.6% carbohydrate and 18.9% protein) for 12 weeks. In the dihydromyricetin group, rats were intravenously given 2 mg/kg dihydromyricetin (Sigma-Aldrich; Merck KGaA, Darmstadt, Germany) every 3 days for 12 weeks as previously described (8).
Histological assessment
Following treatment with dihydromyricetin for 12 weeks, rats were sacrificed using decollation under 35 mg/kg pentobarbital, and liver tissue samples were collected. The tissues were fixed with 4% paraformaldehyde for 24 h, embedded in paraffin wax and then cut into 3-µM sections. Subsequently, the sections were stained with Oil Red O for 5 min and examined under a BX51 microscope (Olympus Corporation, Tokyo, Japan).
Enzyme-linked immunosorbent assay (ELISA)
Following treatment with dihydromyricetin, liver tissue samples were collected and homogenized by centrifugation at 18,000 × g for 10 min at 4°C. The supernatant was used to measure the triglyceride (F001), albumin (ALB; A028-1), collagen I (H142), alanine aminotransferase (ALT; C009-2), aspartate aminotransferase (AST; C010-2), malondialdehyde (MDA; A003-1), superoxide dismutase (SOD; A001-3), glutathione (GSH; A006-2), glutathione peroxidase (GSH-Px; A005), tumor necrosis factor-α (TNF-α; H052), interleukin (IL)-1β (H002), IL-6 (H007), IL-18 (H015), caspase-3 (G015), caspase-8 (G017) and caspase-9 (G018), and inducible nitric oxide synthase (iNOS, A014-1-1) levels with the corresponding ELISA kits (Nanjing Jiancheng Biology Engineering Institute, Nanjing, China).
Western blotting
Subsequent to treatment of rats with dihydromyricetin, total protein was extracted from frozen liver tissue using a radioimmunoprecipitation assay buffer (Beyotime Institute of Biotechnology, Haimen, China), and the protein concentration was determined using a bicinchoninic acid protein assay kit (Beyotime Institute of Biotechnology, Haimen, China). Next, 50-µg protein samples were separated using 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis and then transferred onto a polyvinylidene fluoride membrane (EMD Millipore, Billerica, MA, USA). The membrane was blocked with 5% non-fat milk in TBST for 1 h at 37°C, followed by overnight incubation at 4°C with the following primary antibodies: ALB (sc-271604; 1:2,000), collagen I (sc-8786; 1:1,000), peroxisome proliferator-activated receptor α (PPARα; sc-1982; 1:1,000), NF-κB (sc-71675; 1:1,000), p53 (sc-47698; 1:1,000), B-cell lymphoma 2-associated X protein (Bax; sc-6236; 1:1,000) and GAPDH (sc-51631; 1:5,000; all purchased from Santa Cruz Biotechnology, Inc., Dallas, TX, USA). Membranes were then incubated with horseradish peroxidase-conjugated goat anti-rabbit and anti-rat IgG secondary antibodies (sc-2004 and sc-2005; 1:5,000; Santa Cruz Biotechnology, Inc.) at 37°C for 1 h.
Statistical analysis
The data were expressed as the mean ± standard deviation using SPSS 17.0 (SPSS Inc., Chicago, IL, USA). All data were analyzed by one-way analysis of variance, followed by Tukey's post hoc test. P<0.05 was considered to indicate a statistically significant difference.
Results
Dihydromyricetin treatment prevents the development of fatty liver in a rat model
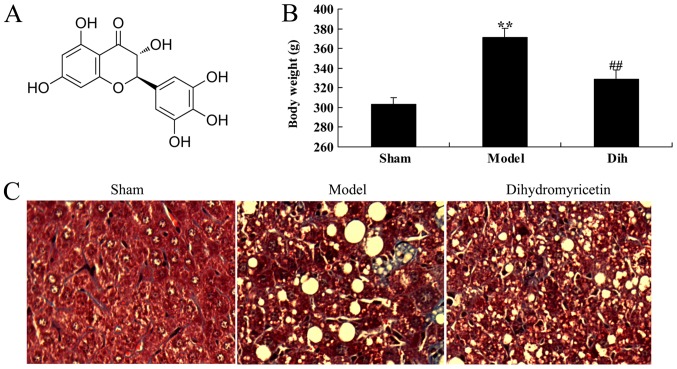
The chemical structure of dihydromyricetin is shown in Fig. 1A. First, the effects of dihydromyricetin on preventing fatty liver development were assessed in a rat model. As a result of a high-fat diet, the body weight was increased in the rat model of fatty liver as compared with that in the normal control group (Fig. 1B). Oil Red O staining of liver sections revealed steatosis in the liver tissues of rat in the fatty liver model group, which was absent in the normal control group tissues (Fig. 1C). However, treatment with dihydromyricetin markedly reduced the body weight and liver steatosis determined by Oil Red O staining in the fatty liver rat model, as compared with the untreated model group (Fig. 1B and C).
Figure 1.
Dihydromyricetin prevented the development of fatty liver in a rat model of fatty liver. (A) Structural formula of dihydromyricetin. (B) Body weight of rats at the end of the experimental period, and (C) Oil Red O staining of liver tissues in the different groups. Magnification, ×200. **P<0.01 vs. normal control group; ##P<0.01 vs. fatty liver model group. Dih, dihydromyricetin.
Dihydromyricetin treatment prevents liver function deterioration in a fatty liver rat model
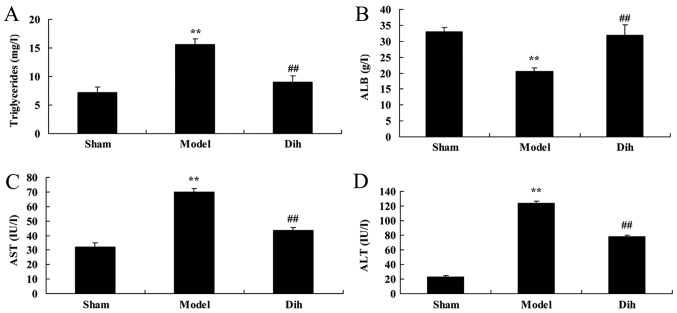
The levels of triglycerides, ALB, ALT and AST were decreased in the rat model of fatty liver, as compared with those in the normal control group (Fig. 2). By contrast, dihydromyricetin administration in the fatty liver rat model significantly increased the levels of triglycerides, ALB, ALT and AST as compared with those in the untreated model group, and indicating the inhibition of liver steatosis in rats (Fig. 2).
Figure 2.
Dihydromyricetin prevented liver function in a rat model of fatty liver. (A) Triglyceride, (B) ALB, (C) ALT and (D) AST levels were assessed by enzyme-linked immunosorbent assay kits. **P<0.01 vs. normal control group; ##P<0.01 vs. fatty liver model group. Dih, dihydromyricetin; ALB, albumin; ALT, alanine aminotransferase; AST, aspartate aminotransferase.
Dihydromyricetin treatment inhibits oxidative stress in a fatty liver rat model
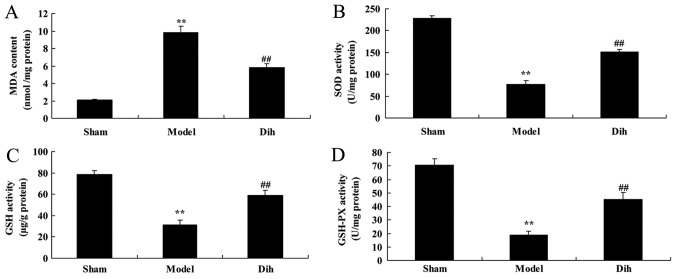
In rat of the fatty liver model group, the MDA level was increased, while the levels of SOD, GSH and GSH-Px were decreased, as compared with those in the normal control group (Fig. 3). However, treatment with dihydromyricetin significantly reduced the MDA level, and significantly increased SOD, GSH and GSH-Px levels, as compared with those of rat in the fatty liver model group without dihydromyricetin administration (Fig. 3).
Figure 3.
Dihydromyricetin inhibited oxidative stress in a rat model of fatty liver. (A) MDA, (B) SOD, (C) GSH and (D) GSH-Px levels were measured by enzyme-linked immunosorbent assay. **P<0.01 vs. normal control group; ##P<0.01 vs. fatty liver model group. Dih, dihydromyricetin; MDA, malondialdehyde; SOD, superoxide dismutase; GSH, glutathione; GSH-Px, glutathione peroxidase.
Dihydromyricetin treatment inhibits inflammation in a fatty liver rat model
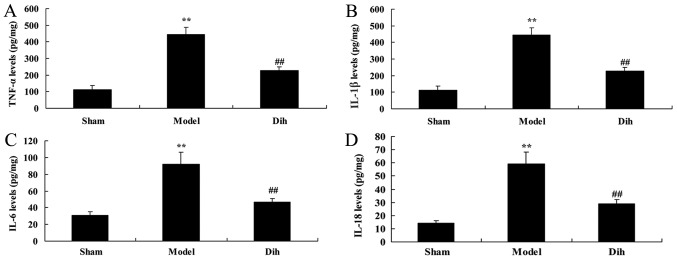
Next, the anti-inflammation effects of dihydromyricetin in rat model of fatty liver were assessed. As shown in Fig. 4, the levels of TNF-α, IL-1β, IL-6 and IL-18 were increased in the rat model of fatty liver, compared with the normal control group (Fig. 4). However, dihydromyricetin treatment in the rat model of fatty liver reduced the levels of TNF-α, IL-1β, IL-6 and IL-18 levels, in comparison with those in model rat without dihydromyricetin administration (Fig. 4).
Figure 4.
Dihydromyricetin inhibited inflammation in a rat model of fatty liver. (A) TNF-α, (B) IL-1β, (C) IL-6 and (D) IL-18 levels were measured by enzyme-linked immunosorbent assay. **P<0.01 vs. normal control group; ##P<0.01 vs. fatty liver model group. Dih, dihydromyricetin; TNF-α, tumor necrosis factor-α; IL, interleukin.
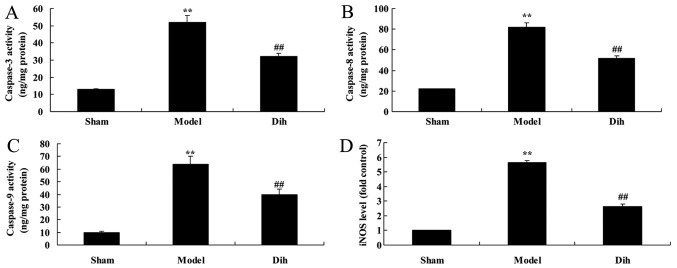
Dihydromyricetin inhibits hepatic cell apoptosis in a rat model of fatty liver
The anti-apoptotic effects of dihydromyricetin in the rat model of fatty liver were further evaluated. As shown in Fig. 5, the activities of caspase-3, caspase-8 and caspase-9, and inducible nitric oxide synthase (iNOS) levels were significantly induced in the rat model of fatty liver, compared with the normal control group. By contrast, treatment with dihydromyricetin in the rat model of fatty liver markedly reduced the activities of caspase-3, caspace-8 and caspase-9, as well as iNOS protein expression, as compared with the levels in untreated model rat (Fig. 5).
Figure 5.
Dihydromyricetin inhibited liver cell apoptosis in a rat model of fatty liver. (A) Caspase-3, (B) caspase-8 and (C) caspase-9 activities, and (D) iNOS levels were measured, by ELISA. **P<0.01 vs. normal control group; ##P<0.01 vs. fatty liver model group. Dih, dihydromyricetin; iNOS, inducible nitric oxide synthase.
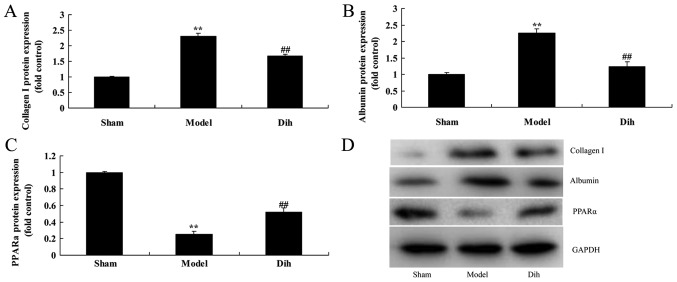
Dihydromyricetin decreases the protein expression levels of Albumin and collagen I, and induced PPARα protein expression in the rat model
Subsequently, the current study aimed to investigate the mechanism of dihydromyricetin in preventing fatty liver development in a rat model. The expression levels of Albumin and collagen I were markedly induced, and PPARα protein expression was suppressed in the rat model of fatty liver, compared with normal control group (Fig. 6). However, dihydromyricetin administration in the fatty liver rat model significantly suppressed the protein levels of ALB and collagen I, and induced PPARα protein expression in comparison with those in the untreated model group (Fig. 6).
Figure 6.
Dihydromyricetin decreased ALB and collagen I protein expression levels, and induced PPARα protein expression in a rat model of fatty liver. Quantified protein expression levels of (A) collagen I, (B) ALB and (C) PPARα. (D) Western blotting results. **P<0.01 vs. normal control group; ##P<0.01 vs. fatty liver model group. Dih, dihydromyricetin; ALB, albumin; PPARα, peroxisome proliferator-activated receptor α.
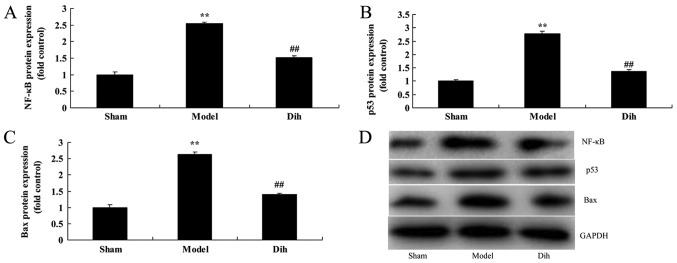
Dihydromyricetin suppresses the protein levels of NF-κB, p53 and Bax in the rat model
As shown in Fig. 7, the protein expression levels of NF-κB, p53 and Bax were significantly enhanced in the rat model of fatty liver, compared with the normal control group (Fig. 7). However, dihydromyricetin treatment in fatty liver model rat evidently suppressed the protein expression levels of NF-κB, p53 and Bax when compared with the model rat without dihydromyricetin administration (Fig. 7). These results showed that Dihydromyricetin suppresses NF-κB protein levels to reduce inflammation and decreased p53 and Bax protein expression to inhibit apoptosis in the rat model.
Figure 7.
Dihydromyricetin suppressed NF-κB, p53 and Bax protein expression levels in a rat model of fatty liver. Quantified protein levels of (A) NF-κB, (B) p53 and (C) Bax. (D) Western blotting results. **P<0.01 vs. normal control group; ##P<0.01 vs. fatty liver model group. Dih, dihydromyricetin; NF, nuclear factor; Bax, B-cell lymphoma 2-associated X protein.
Discussion
Alcohol consumption may also lead to digestive disease, chronic gastritis, alcoholic fatty liver, alcoholic hepatitis and even alcoholic cirrhosis (4). ALD is a disease resulting from excessive alcohol uptake, which finally leads to hepatic toxic damage (9). Research indicates that the major pathogenic factors of ALD are the metabolites of ethyl alcohol in hepatocytes and the induced metabolic disorder (9). The disease begins with fatty degeneration of hepatocytes, and may gradually develop into alcoholic hepatitis and fibrosis with the increase in alcohol consumption. Finally, alcoholic cirrhosis and even primary hepatocellular carcinoma may be developed (10). In the present study, it was observed that treatment with dihydromyricetin reduced body weight, triglycerides, ALT and AST levels, and increased ALB levels in a rat model of fatty liver.
Excessive highly active molecules, such as reactive oxygen species (ROS) and reactive nitrogen species, are produced in the presence of harmful stimulations (11). As a result, the oxidative degree exceeds the scavenging of oxides, and the balance between the oxidative and anti-oxidative systems is broken. Thus, tissue injury is induced (12), and such a process is referred to as oxidative stress. The major alcohol metabolic pathway exists in the liver (13). The body metabolism produces ROS (including superoxide anion, hydroxyl radical and hydrogen peroxide) through different pathways (13), and excessive ROS-induced oxidative stress response is the important cause of hepatocyte injury. Alcohol can trigger and aggravate oxidative stress, and reduce the free radical scavenging and anti-peroxidation capacity (14). As a result, peroxide production in vivo is increased, thereby promoting the genesis and development of alcoholic liver injury (12). Oxidative stress and pro-inflammatory factor production are important factors among alcohol-induced cell injury mechanisms (13).
The results of the present study suggested that dihydromyricetin treatment inhibited oxidative stress, inflammation and liver cell apoptosis in a rat model of fatty liver. Previously, Chen et al (15) identified that dihydromyricetin protects against liver ischemia/reperfusion-induced apoptosis. In addition, Song et al (1) reported that dihydromyricetin attenuated angiotensin II-induced cardiac fibroblast proliferation associated with inhibition of oxidative stress.
NF-κB, an important transcription factor, is a nuclear protein binding to κ light chain enhancer of activated B cells (16). In addition, NF-κB is the upstream signaling molecule of multiple inflammatory mediators and can promote their production (17), and is thus key in the inflammatory reaction. NF-κB also regulates the transcription of various acute reactive proteins, cytokines and cell adhesion molecules. Thus, it can directly participate in acute and chronic liver inflammation (17). Liver injury in alcoholic fatty liver is closely associated with apoptosis of hepatocytes, which is in turn associated with the activation of NF-κB (16,18). Zhou et al (19) indicated that dihydromyricetin protects against lipopolysaccharide-induced cardiomyocyte injury via Toll-like receptor 4 and NF-κB pathways.
NF-κB is activated under the stimulation of pro-inflammatory factors in ALD, thus upregulating the expression of pro-inflammatory and chemotactic factors (7). NF-κB activation can enhance the transcription of pro-inflammatory cytokines (20), which in turn stimulate NF-κB activation (20). Such positive feedback reaction can certainly amplify the inflammatory signal and aggravate tissue injury. The production of abnormal large amounts of inflammatory factors serves a vital role in the pathogenesis of ALD (21). Therefore, NF-κB is an important central link, and control over the production of NF-κB can control the inflammatory damage of liver, thus serving as an effective therapeutic target (21). In the present study, dihydromyricetin suppressed NF-κB, p53 and Bax protein expression levels in a fatty liver rat model. Zhou et al (19) reported that dihydromyricetin induced apoptosis and cytoprotective autophagy in human melanoma cells through ROS-NF-κB signaling. Overall, the NF-κB signaling pathway regulates a great number of signaling pathways, and further investigation is required to analyze these signaling pathways.
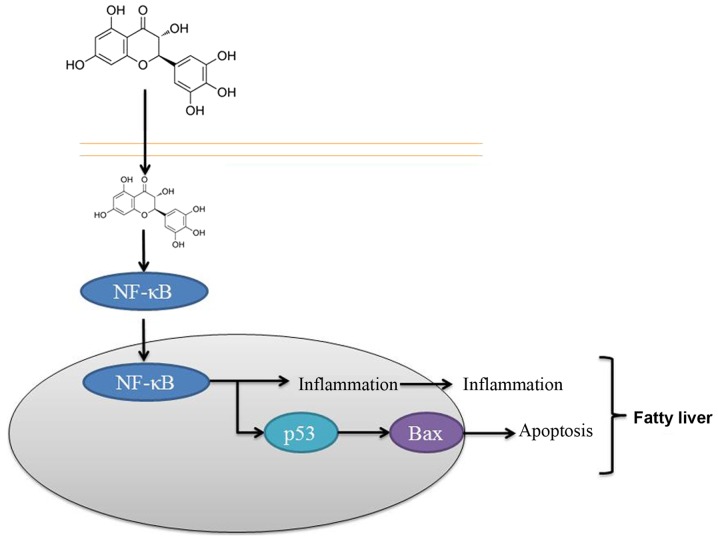
In conclusion, the results of the present study suggested that dihydromyricetin administration suppressed NF-κB, p53 and Bax protein expression levels in a fatty liver rat model. It is, thus, concluded that the protective effect of dihydromyricetin on the liver was through NF-κB/p53/Bax signaling pathways in the rat model (Fig. 8). These findings suggest that dihydromyricetin is associated with the pathogenesis of liver fibrosis and that dihydromyricetin treatment may thus be a strategy for preventing hepatic fibrosis of patients, although this requires further examination.
Figure 8.
Protective effect of dihydromyricetin on the liver through NF-κB/p53/Bax signaling pathways in a rat model of fatty liver. NF, nuclear factor; Bax, B-cell lymphoma 2-associated X protein.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The analyzed data sets generated during the study are available from the corresponding author on reasonable request.
Authors' contributions
XY designed the experiments. LG and HZ performed the experiments. XY and LG analyzed the data. XY wrote the manuscript.
Ethics approval and consent to participate
The present study was conducted in accordance with the Guidelines for Animal Experimentation and approval obtained by the ethics committee of the Sixth People's Hospital of Qingdao.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Song Q, Liu L, Yu J, Zhang J, Xu M, Sun L, Luo H, Feng Z, Meng G. Dihydromyricetin attenuated Ang II induced cardiac fibroblasts proliferation related to inhibitory of oxidative stress. Eur J Pharmacol. 2017;807:159–167. doi: 10.1016/j.ejphar.2017.04.014. [DOI] [PubMed] [Google Scholar]
- 2.Huang H, Hu M, Zhao R, Li P, Li M. Dihydromyricetin suppresses the proliferation of hepatocellular carcinoma cells by inducing G2/M arrest through the Chk1/Chk2/Cdc25C pathway. Oncol Rep. 2013;30:2467–2475. doi: 10.3892/or.2013.2705. [DOI] [PubMed] [Google Scholar]
- 3.Rotman Y. Similarity between studies of dihydromyricetin and reservatrol for NAFLD. Pharmacol Res. 2015;100:335. doi: 10.1016/j.phrs.2015.07.034. [DOI] [PubMed] [Google Scholar]
- 4.Houghton D, Thoma C, Hallsworth K, Cassidy S, Hardy T, Burt AD, Tiniakos D, Hollingsworth KG, Taylor R, Day CP, et al. Exercise reduces liver lipids and visceral adiposity in patients with nonalcoholic steatohepatitis in a randomized controlled trial. Clin Gastroenterol Hepatol. 2017;15:96–102.e3. doi: 10.1016/j.cgh.2016.07.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Alexander KS, Zakai NA, Lidofsky SD, Callas PW, Judd SE, Tracy RP, Cushman M. Non-alcoholic fatty liver disease, liver biomarkers and stroke risk: The reasons for geographic and racial differences in stroke cohort. PLoS One. 2018;13:e0194153. doi: 10.1371/journal.pone.0194153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Kim W, Kim BG, Lee JS, Lee CK, Yeon JE, Chang MS, Kim JH, Kim H, Yi S, Lee J, et al. Randomised clinical trial: The efficacy and safety of oltipraz, a liver X receptor alpha-inhibitory dithiolethione in patients with non-alcoholic fatty liver disease. Aliment Pharmacol Ther. 2017;45:1073–1083. doi: 10.1111/apt.13981. [DOI] [PubMed] [Google Scholar]
- 7.Li L, Hai J, Li Z, Zhang Y, Peng H, Li K, Weng X. Resveratrol modulates autophagy and NF-κB activity in a murine model for treating non-alcoholic fatty liver disease. Food Chem Toxicol. 2014;63:166–173. doi: 10.1016/j.fct.2013.08.036. [DOI] [PubMed] [Google Scholar]
- 8.Bu W, Luo T. miR-1297 promotes cell proliferation of non-small cell lung cancer cells: Involving in PTEN/Akt/Skp2 signaling pathway. DNA Cell Biol. 2017;36:976–982. doi: 10.1089/dna.2017.3886. [DOI] [PubMed] [Google Scholar]
- 9.Chan DC, Watts GF, Gan S, Wong AT, Ooi EM, Barrett PH. Nonalcoholic fatty liver disease as the transducer of hepatic oversecretion of very-low-density lipoprotein-apolipoprotein B-100 in obesity. Arterioscler Thromb Vasc Biol. 2010;30:1043–1050. doi: 10.1161/ATVBAHA.109.202275. [DOI] [PubMed] [Google Scholar]
- 10.Kalinowski P, Paluszkiewicz R, Ziarkiewicz-Wroblewska B, Wróblewski T, Remiszewski P, Grodzicki M, Krawczyk M. Liver function in patients with nonalcoholic fatty liver disease randomized to Roux-en-Y gastric bypass versus sleeve gastrectomy: A secondary analysis of a randomized clinical trial. Ann Surg. 2017;266:738–745. doi: 10.1097/SLA.0000000000002397. [DOI] [PubMed] [Google Scholar]
- 11.Gonzalez-Manan D, D'Espessailles A, Dossi CG, San Martin M, Mancilla RA, Tapia GS. Rosa mosqueta oil prevents oxidative stress and inflammation through the upregulation of PPAR-α and NRF2 in C57BL/6J rat fed a high-fat diet. J Nutr. 2017;147:579–588. doi: 10.3945/jn.116.243261. [DOI] [PubMed] [Google Scholar]
- 12.Mukai T, Egawa M, Takeuchi T, Yamashita H, Kusudo T. Silencing of FABP1 ameliorates hepatic steatosis, inflammation, and oxidative stress in rat with nonalcoholic fatty liver disease. FEBS Open Bio. 2017;7:1009–1016. doi: 10.1002/2211-5463.12240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Sharifi N, Amani R, Hajiani E, Cheraghian B. Does vitamin D improve liver enzymes, oxidative stress, and inflammatory biomarkers in adults with non-alcoholic fatty liver disease? A randomized clinical trial. Endocrine. 2014;47:70–80. doi: 10.1007/s12020-014-0336-5. [DOI] [PubMed] [Google Scholar]
- 14.Zhou Y, Ding YL, Zhang JL, Zhang P, Wang JQ, Li ZH. Alpinetin improved high fat diet-induced non-alcoholic fatty liver disease (NAFLD) through improving oxidative stress, inflammatory response and lipid metabolism. Biomed Pharmacother. 2018;97:1397–1408. doi: 10.1016/j.biopha.2017.10.035. [DOI] [PubMed] [Google Scholar]
- 15.Chen Y, Lv L, Pi H, Qin W, Chen J, Guo D, Lin J, Chi X, Jiang Z, Yang H, Jiang Y. Dihydromyricetin protects against liver ischemia/reperfusion induced apoptosis via activation of FOXO3a-mediated autophagy. Oncotarget. 2016;7:76508–76522. doi: 10.18632/oncotarget.12894. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Willy JA, Young SK, Stevens JL, Masuoka HC, Wek RC. CHOP links endoplasmic reticulum stress to NF-κB activation in the pathogenesis of nonalcoholic steatohepatitis. Mol Biol Cell. 2015;26:2190–2204. doi: 10.1091/mbc.E15-01-0036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Shankar E, Zhang A, Franco D, Gupta S. Betulinic acid-mediated apoptosis in human prostate cancer cells involves p53 and nuclear Factor-kappa B (NF-κB) Pathways. Molecules. 2017;22(pii):E264. doi: 10.3390/molecules22020264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Zhang H, Tan X, Yang D, Lu J, Liu B, Baiyun R, Zhang Z. Dietary luteolin attenuates chronic liver injury induced by mercuric chloride via the Nrf2/NF-κB/P53 signaling pathway in rats. Oncotarget. 2017;8:40982–40993. doi: 10.18632/oncotarget.17334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Zhou DZ, Sun HY, Yue JQ, Peng Y, Chen YM, Zhong ZJ. Dihydromyricetin induces apoptosis and cytoprotective autophagy through ROS-NF-κB signalling in human melanoma cells. Free Radic Res. 2017;51:517–528. doi: 10.1080/10715762.2017.1328552. [DOI] [PubMed] [Google Scholar]
- 20.Kim JY, Song EH, Lee HJ, Oh YK, Choi KH, Yu DY, Park SI, Seong JK, Kim WH. HBx-induced hepatic steatosis and apoptosis are regulated by TNFR1- and NF-kappaB-dependent pathways. J Mol Biol. 2010;397:917–931. doi: 10.1016/j.jmb.2010.02.016. [DOI] [PubMed] [Google Scholar]
- 21.Maraslioglu M, Weber R, Korff S, Blattner C, Nauck C, Henrich D, Jobin C, Marzi I, Lehnert M. Activation of NF-κB after chronic ethanol intake and haemorrhagic shock/resuscitation in rat. Br J Pharmacol. 2013;170:506–518. doi: 10.1111/bph.12224. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The analyzed data sets generated during the study are available from the corresponding author on reasonable request.