Abstract
The aims of this study were to 1) examine pharyngoesophageal and cardiorespiratory responses to provoking pharyngeal stimuli, and 2) to determine potential contributory factors impacting heart rate (HR) changes to provide insight into cardiorespiratory events occurring in preterm infants. Forty-eight neonates (19 females and 29 males, born at 27.7 ± 0.5 wk; mean ± SE) pending discharge on full oral feeds were studied at 38.7 ± 0.2 wk postmenstrual age using concurrent pharyngoesophageal manometry, electrocardiography, respiratory inductance plethysmography, and nasal airflow thermistor. Pharyngoesophageal and cardiorespiratory responses (prevalence, latency, and duration) were quantified upon abrupt pharyngeal water stimuli (0.1, 0.3, and 0.5 ml in triplicate). Mixed linear models and generalized estimating equations were used for comparisons between HR changes. Contributory factors included stimulus characteristics and subject characteristics. Of 338 pharyngeal stimuli administered, HR increased in 23 (7%), decreased in 108 (32%), and remained stable in 207 (61%) neonates. HR decrease resulted in repetitive swallowing, increased respiratory-rhythm disturbance, and decreased esophageal propagation rates (all, P < 0.05). HR responses were related to stimulus volume, stimulus flow rate, and extreme prematurity (all, P < 0.05). In preterm infants, HR remains stable in a majority of pharyngeal provocations. HR decrease, due to pharyngeal stimulation, is related to aberrant pharyngoesophageal motility and respiratory dysregulation and is magnified by prematurity. We infer that the observed aberrant responses across digestive, respiratory, and cardiovascular systems are related to maladaptive maturation of the parasympathetic nervous system. These aberrant responses may provide diagnostic clues for risk stratification of infants with troublesome cardiorespiratory events and swallowing difficulty.
NEW & NOTEWORTHY Cardiorespiratory rhythms concurrent with pharyngeal, upper esophageal sphincter, and esophageal body responses were examined upon pharyngeal provocation in preterm-born infants who were studied at full-term maturation. Decreased heart rate (HR) was associated with extreme preterm birth and stimulus flow/volume. With HR decrease responses, aerodigestive reflex abnormalities were present, characterized by prolonged respiratory rhythm disturbance, repetitive multiple swallowing, and poor esophageal propagation. Promoting esophageal peristalsis may be a potential therapeutic target.
Keywords: cardiorespiratory rhythms, esophageal motility, life-threatening events, prematurity, swallowing
INTRODUCTION
Convalescing infants pending discharge from the neonatal intensive care unit (NICU) frequently experience cardiorespiratory events (apnea, bradycardia, and desaturation) and/or feeding difficulties (sucking and/or swallowing dysfunction) that further interfere with nutrition and oral intake, growth, and discharge plans. Per American Academy of Pediatrics policy, before discharge, preterm infants must exhibit: 1) oral feeding competency without cardiorespiratory compromise, and 2) physiological maturity with stable cardiorespiratory function for a sufficient duration, typically 5–7 days (2, 11). Life-threatening cardiorespiratory events frequently resolve by 36 wk postmenstrual age (PMA), but in high-risk infants may persist or reemerge after discharge until and beyond 44 wk PMA (4, 10). Additionally, the reasoning for the 5–7 days of waiting period is not completely understood, and in some situations, emergency hospital visits escalate socioeconomic consequences. The prevalence of life-threatening cardiorespiratory concerns is ~12.5% with varying economic burden, as they can be brief and self-resolving or prolonged and/or persistent requiring interventions (4, 6, 10, 22, 43, 53).
Lack of physiology-guided evaluations has led to nonspecific diagnostic criteria for problematic cardiorespiratory function. The term apparent life-threatening event (ALTE) (1), in which the observer fears an infant is dying, is defined as “an episode that is frightening to the observer and that is characterized by some combination of apnea (central or occasionally obstructive), color change (usually cyanotic or pallid, but occasionally erythematous or plethoric), marked change in muscle tone (usually marked limpness), choking, or gagging.” Recently, this term was replaced by brief resolved unexplained events (BRUE) (53) and defined as “an event occurring in an infant younger than 1 yr when the observer reports a sudden, brief, now resolved episode of ≥1 of the following 1) cyanosis or pallor; 2) absent, decreased, or irregular breathing; 3) marked change in tone (hypertonia or hypotonia); and 4) altered level of responsiveness.” These BRUE events are diagnosed when there is no explanation of the event despite appropriate history and can be classified as low-risk (likely not to occur again) or high-risk (likely for repeat occurrences and needs further investigation) (39). The high-risk scenario does not currently have well-defined guidelines. Regardless of the risk magnitude, these events pose a significant public health concern and contribute to parental anxiety, particularly among infants suffering from the consequences of extreme prematurity (18).
Identification of the causes for life-threating events still remains elusive, as well as the events that restore cardiorespiratory normalcy, whether self-regulated or requiring intervention. The influence of comorbidities on these mechanisms is also unclear. Evaluation of cardiorespiratory and pharyngoesophageal characteristics may provide insight into the physiology and pathophysiology of life-threatening events. We and others have previously shown that maturation and the neonate’s capability to adapt to bolus volume and flow can impact suck-swallow-breathing coordination (17, 25, 30, 35, 36, 40, 44, 47), thus contributing to feeding difficulties and swallowing dysfunction. A majority of these works have focused extensively on interactions between the airway (apnea) and digestive (swallowing) systems, but not the interactions of the autonomic system (heart rate, HR). In general, HR varies widely in awake and sleeping infants, ranging from 90 to 180 beats per min (bpm) (12) and may be related to cardiac, vagal, and sympathetic tone. In neonates and infants, a rate of less than 100 bpm requires monitoring and/or potential interventions (41, 55). Davies et al. (8) has shown that when heart rate change occurs during pharyngeal stimulation, it occurs after apnea and pharyngeal swallowing. However, esophageal characteristics were not evaluated. The importance of esophageal clearance can be implicated in facilitating autoresuscitation (16, 31). This study was undertaken to evaluate the frequency and magnitude of heart rate changes during adaptive cardiorespiratory and pharyngoesophageal reflexes in preterm infants studied at full-term status, when feeding skills should be developed and cardiorespiratory systems stable for discharge (2, 11).
Aims of the current study were to 1) examine and compare the pharyngoesophageal and cardiorespiratory responses to pharyngeal stimulation, and stratify the responses on the basis of heart rate changes, and 2) determine contributory factors that may alter heart rate changes in preterm born infants that were able to achieve successful oral feeds by discharge. We hypothesized that the response resulting in heart rate changes: 1) has distinct pharyngoesophageal and cardiorespiratory response characteristics, and 2) is modified by stimulus and subject characteristics.
MATERIALS AND METHODS
Subjects
Premature-born neonates (n = 48, 19 females and 29 males, born at 27.7 ± 0.5 wk weighing 1.2 ± 0.1 kg), who were transitioning to independent oral feeds and pending discharge from the NICU, underwent water perfusion manometry between 36 and 40 wk PMA for the evaluation of aerodigestive pathophysiology at The Infant Feeding Disorders Program at Nationwide Children’s Hospital, Columbus, OH. Informed parental consent and Institutional Research Review Board approval at Nationwide Children’s Hospital were obtained, and Health Insurance Portability and Accountability Act guidelines were followed. For subject safety, the patient’s vital signs were continuously monitored by the study physician and registered nurse.
Recording Techniques
A custom-designed silicone catheter (Dentsleeve International, Mui Scientific, Toronto, ON, Canada) with pressure channels spanning from the pharynx to the stomach (pharyngeal port, upper esophageal sphincter sleeve, proximal esophagus port, middle esophagus port, distal esophagus port, lower esophageal sphincter sleeve, gastric port) with a pharyngeal infusion port for stimulus attached to a water-perfused manometry system (Solar GI; Laborie Medical Technologies, Mississauga, ON, Canada), and passed nasally in the supine subject to evaluate pharyngeal and esophageal reflexes (17, 24, 25, 28). Catheter size (2.0 mm diameter, 3–5 cm length for sleeves, 11.0–18.75 cm length from stomach to pharynx), channel spacing (pharyngeal infusion port between 0.75–1.75 cm above upper esophageal sphincter sleeve, and 1.5–3 cm between esophageal ports), and perfusion rates (0.01 to 0.08 ml/min for each port) were selected on the basis of the infant’s weight (kg) at the time of evaluation (17, 20, 21, 24, 25, 28). Concurrent dual-band respiratory inductance plethysmography (Respitrace; Viasys, Conshohocken, PA), nasal thermistor (Integra Life Sciences, Plainsboro, NJ), and electrocardiography (Laborie Medical Technologies or BioRadio 150; Great Lakes NeuroTechnologies, Cleveland, OH) were used to evaluate cardiorespiratory rhythms (16, 17, 26, 28).
Experimental Protocol
The catheter was placed using the pull-through technique for proper positioning, and the infant was given adequate time for adaptation (17, 25, 26, 28, 30). Sterile water infusions through the pharyngeal port were administered using a graded stimulus dose (0.1, 0.3, and 0.5 ml) escalation protocol during aerodigestive quiescence (normal breathing and absence of pharyngoesophageal activity to record pharyngoesophageal and cardiorespiratory responses (17, 25, 26, 28, 30). Each stimulus volume was administered in triplicate, and patient safety was monitored before escalating to the subsequent higher dose.
Data Analysis
Pharyngoesophageal and cardiorespiratory rhythm characteristics were evaluated using MMS analysis software (v. 2.04; Laborie Medical Technologies) and VivoSense software (v 2.8; Vivonoetics, San Diego, CA). Responses were measured from stimulus onset to return to pharyngoesophageal and cardiorespiratory quiescence (17, 25–27, 30).
Pharyngoesophageal Motility
Pharyngeal provocation-induced reflexes were analyzed as previously published (17, 25–27, 30). Briefly, primary response type was characterized as pharyngeal reflexive swallow (PRS), pharyngo-upper esophageal sphincter contractile reflex (PUCR), or none (17, 25–27, 30). If PRS was observed (as either a primary or secondary response), the following pharyngeal characteristics were analyzed: 1) recruitment occurrence as the number of pharyngeal peaks (17, 25), 2) frequency (Hz) as number of pharyngeal peaks divided by duration of pharyngeal activity (17), 3) variability as standard deviation of pharyngeal peak to peak intervals (17), and 4) stability as a standard deviation of pharyngeal peak-to-peak interval durations divided by the average pharyngeal peak-to-peak interval duration (similar to coefficient of variation, lower values indicate greater stability) (17). Esophageal body characteristics analyzed response latency as esophageal body activity onset, response duration as esophageal body response activity onset to offset, and esophageal body inhibition as pharyngeal response onset to esophageal body response onset (17, 25–27, 30). Esophageal body propagation rate was defined as the ratio between the number of pharyngeal contractions to the number of esophageal body contractions.
Cardiorespiratory Rhythms
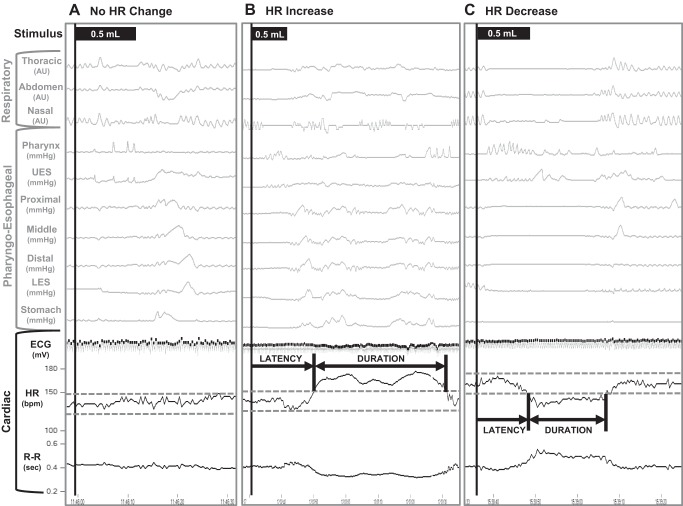
Basal heart rate was measured for a duration of 10 s immediately before stimulus onset. On the basis of prior experience testing sympathomimetic bronchodilator response in both infants and infants with bronchopulmonary dysplasia (13, 51), HR response for the current study was defined using a conservative definition of 10% change in HR from baseline. Increase in HR reflects increasing sympathetic tone (excitatory neuronal response), and a decrease reflects increasing parasympathetic tone (inhibitory vagal cardiac response). Cardiac changes (Fig. 1) included 1) HR response prevalence, %, as increase, decrease, or no change; 2) HR response latency (seconds), as the duration between infusion onset to 10% HR change from baseline; 3) response duration (in seconds), as the time interval between 10% HR change onset to 10% HR change offset, and 4) heart rate variability as the as the beat-to-beat (R-R wave intervals) changes in HR.
Fig. 1.
Evaluation of cardiac responses to pharyngeal stimuli. Depicted is a representative tracing of concurrent pharyngoesophageal motility and cardiorespiratory responses to pharyngeal stimuli (0.5 ml). Potential responses included no heart rate (HR) change (A), HR increase (B), or HR decrease (C). HR change was defined as 10% change from baseline (indicated by the gray dashed lines). HR response latency was defined as the time between stimulus onset (black vertical line) and 10% HR change. HR response duration was defined as the time between 10% change onset to 10% change offset. Note with the HR increase response, the R-R interval decreases, indicating increased sympathetic tone. Conversely, with the HR decrease response, the R-R interval increases, indicating increased parasympathetic tone.
Respiratory changes were analyzed as previously published (16, 17, 26–28). Briefly, respiratory adaptation measures included 1) respiratory rhythm disturbance prevalence (as a percentage) as a change in basal respiratory rhythm; 2) respiratory rhythm disturbance response latency as the duration between pharyngeal stimulus onset to onset of respiratory rhythm disturbance; 3) respiratory rhythm disturbance response duration (in seconds) as the onset of respiratory rhythm disturbance to offset; 4) prevalence (as a percentage) of deglutition apnea defined as cessation of breathing associated with initial pharyngeal response; and 5) deglutition apnea duration (in seconds) defined as the onset to offset of breathing cessation associated with initial pharyngeal response.
Statistical Analysis
Statistical analysis was performed using SAS (v 9.3; SAS Institute, Cary, NC). Demographic characteristics of subjects were compared using Kruskal-Wallis (nonparametric) test for continuous variables and χ2-tests for categorical variables. Linear mixed models and generalized estimation of equations were used to compare pharyngoesophageal cardiorespiratory responses for HR change (vs. no change). Potential contributing factors to HR change analyzed included stimulus (flow, volume) and subject (extreme prematurity defined as birth <28 wk gestation, bronchopulmonary dysplasia defined as oxygen requirement at 36 wk PMA, neuropathology, and gavage tube feeding) characteristics, were assessed using generalized estimating equations. Data are presented as median (IQR), means ± SE, or as a percentage, unless otherwise noted. A P value <0.05 was considered significant.
RESULTS
Demographic and Clinical Characteristics
Subjects were evaluated at 38.7 ± 0.2 wk PMA (2.8 ± 0.1 kg). Of the 48 preterm infants studied: 1) 27 (56%) were born extremely preterm (defined as birth at <28 wk gestation) averaging 25 ± 0.27 wk gestational age, and median (IQR) of 25 (24–26) wk gestational age; 2) 30 (63%) had bronchopulmonary dysplasia; and 3) 12 (25%) had neuropathology (intraventricular hemorrhage categorized as grade I: n = 5, grade II: n = 1, grade III: n = 1, grade IV: n = 1, and grade uncertain: n = 1, or periventricular leukomalacia). Feeding methods at evaluation were gavage feeding in 24 (50%) and oral feeding in 24 (50%). Respiratory support at evaluation was by nasal continuous positive airway pressure in 9 (19%), nasal cannula oxygen in 20 (42%), and none in 19 (39%). At discharge, all 48 (100%) infants were orally feeding and weighed 3.5 ± 0.1 kg, and 26 (54%) were discharged on nasal cannula oxygen. Length of hospitalization from birth was 99.2 ± 5.8 days.
Pharyngeal Stimuli-Induced Heart Rate Changes
An example of pharyngoesophageal motility and cardiorespiratory responses stratified by HR changes are shown (Fig. 1). Out of the 338 pharyngeal stimuli administered, poststimulus HR increased in 23 (7%), decreased in 108 (32%), and did not change in 207 (61%). The median HR during increase was 175 (150–181) bpm and during decrease was 103 (87–117) bpm. The frequency of HR decreases with stimuli resulting in HR < 100 bpm was 44 (13%), with HR <80 bpm was 18 (5%), and with HR <60 bpm was 5 (1%). However, the median oxygen saturation during these bradycardic events was 100% ranging from 63 to 100%. Out of the 338 pharyngeal stimuli, only 2 (0.6%) resulted in desaturation (event < 85%) with HR decrease (58 and 69 bpm) from two subjects, and was associated with prolonged deglutition apnea, repetitive swallowing, impaired peristaltic reflexes, coughing, movement, and arching. Neither event required bagging or chest compressions and were resolved with tactile stimulation and facilitated swallowing, all resulting in restoration to pharyngoesophageal and cardiorespiratory normalcy. Both subjects that had a desaturation event were born extremely preterm and had bronchopulmonary dysplasia, with one on nasal continuous positive airway pressure and the other on 0.3 liters/min nasal cannula oxygen. No other desaturation events were documented for any of the remaining 336 pharyngeal stimuli or subjects.
Primary motility response type (PRS:PUCR:none, %) did not differ between HR responses: (74:13:13) for HR increase, (89:8:3) for HR decrease, and (62:12:26) for no HR change, (P > 0.05). Specific pharyngoesophageal and cardiorespiratory characteristics during heart rate changes are shown in Table 1. Note the increased prevalence of multiple pharyngeal swallowing events, decreased esophageal body propagation rates, and increased prevalence and duration of respiratory perturbation with HR decrease responses. Pharyngeal frequency, pharyngeal peak-to-peak variability, pharyngeal stability, and esophageal body inhibition did not differ between HR changes (increase or decrease) vs. no HR change P > 0.05. Additionally, the prevalence of terminal swallow did not differ between no HR change (83%), HR increase (55%), and HR decrease (73%), P > 0.05. The occurrence of multiple vs. solitary swallows differed between HR decrease vs. HR increase (OR = 19.9, 95% CI = 1.39–283.7) and for HR decrease vs. no HR change (OR = 47.4, 95% CI = 6.5–347.2), but it did not significantly differ for HR increase vs. no change (OR = 2.4, 95% CI = 0.4–15.0). For the responses resulting in HR increases (n = 23), we noted these occurring 1) after respiratory response onset in 22 (96%), 2) after pharyngeal response onset in 22 (96%), and 3) after esophageal body response onset in 14 (61%). For the responses resulting in HR decrease (n = 108), we noted these occurring 1) after respiratory response onset in 104 (96%), 2) after pharyngeal response onset in 105 (97%), and 3) before esophageal body response onset in 63 (58%).
Table 1.
Pharyngeal provocation-induced pharyngoesophageal and cardiorespiratory responses categorized by HR response
| Characteristic | No HR Change | HR Increase | HR Decrease |
|---|---|---|---|
| Events, n | 207 | 23 | 108 |
| Stimulus duration, s | 10.0 ± 0.4 | 10.1 ± 0.8 | 9.9 ± 0.5 |
| Stimulus flow, ml/s | 0.05 ± 0.00 | 0.04 ± 0.01 | 0.04 ± 0.00 |
| Pharyngeal phase | |||
| PRS (solitary: multiple), % | 31: 69 | 16: 84 | x1: 99*† |
| Recruitment, no. of peaks/stimulus | 5.7 ± 0.4 | 5.9 ± 0.8 | 7.4 ± 0.4* |
| Response latency, s | 4.1 ± 0.4 | 4.6 ± 0.6 | 4.5 ± 0.4 |
| Response duration, s | 14.9 ± 1.2 | 17.4 ± 4.3 | 18.2 ± 2.0 |
| Esophageal phase | |||
| Response latency, s | 11.1 ± 0.7 | 13.2 ± 1.7 | 13.0 ± 0.8 |
| Response duration, s | 16.8 ± 2.3 | 23.7 ± 6.5 | 22.7 ± 2.8 |
| Propagation rate, % | 46.2 ± 3.1 | 56.6 ± 6.3 | 33.9 ± 3.5*† |
| Cardiac | |||
| Basal HR, beats/min | 152 ± 2 | 145 ± 2* | 151 ± 2† |
| Response HR, beats/min | 151 ± 2 | 170 ± 4*‡ | 102 ± 2*†‡ |
| HR response latency, s | N/A | 13.2 ± 1.4 | 9.4 ± 0.9† |
| HR response duration, s | N/A | 47.9 ± 3.7 | 8.0 ± 1.9† |
| HR response magnitude, % | 4 ± 0 | 22 ± 2* | 31 ± 2*† |
| Basal R-R interval, sec | 0.40 ± 0.01 | 0.42 ± 0.01* | 0.40 ± 0.01† |
| Response R-R interval, s | 0.40 ± 0.01 | 0.36 ± 0.02‡ | 0.61 ± 0.01*†‡ |
| Respiratory | |||
| Rhythm change prevalence, % | 73 | 87 | 100*† |
| Rhythm change latency, s | 4.2 ± 0.4 | 4.3 ± 0.8 | 4.0 ± 0.4 |
| Rhythm change duration, s | 18.9 ± 2.3 | 31.7 ± 6.4 | 29.9 ± 2.8* |
| Deglutition apnea prevalence, % | 98 | 100* | 98† |
| Deglutition apnea duration, s | 3.5 ± 0.6 | 7.8 ± 1.4* | 6.1 ± 0.7* |
Values are means ± SE or percentages. N/A, not applicable.
P < 0.05 vs. pharyngeal infusions resulting in no heart rate (HR) change.
P < 0.05 vs. pharyngeal infusions resulting in HR increase. HR change was defined as 10% change from resting baseline.
P < 0.01 for comparison vs. basal HR or basal R-R interval, respectively.
Contributory Factors of Pharyngeal Stimulus-Induced Heart Rate Changes
Effects of stimulus volume and flow.
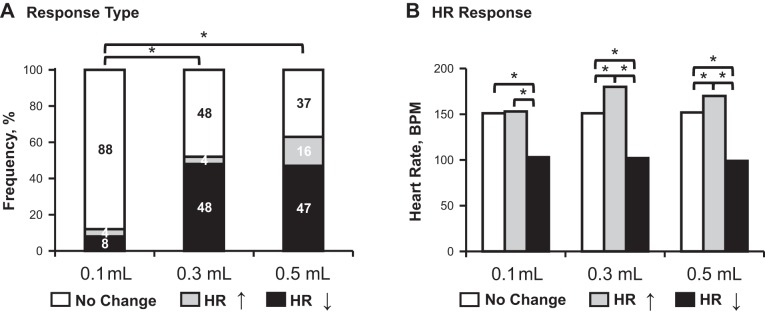
The effects of graded pharyngeal stimulus volume on cardiac responses are shown (Fig. 2). Note with higher stimulus volumes (0.3 and 0.5 ml), heart rate changes are more frequent, but they are not significantly different between 0.3 and 0.5 ml (Fig. 2A). Also note the response magnitude is significantly greater with heart rate decrease (Fig. 2B). Stimulus volume did not affect HR response latency or duration (P > 0.05). Infusion flows per volume were 0.08 ± 0.003 ml/s for 0.1 ml, 0.03 ± 0.004 ml/s for 0.3 ml, and 0.03 ± 0.004 for 0.5 ml, with P < 0.05 for 0.1 ml. With HR decrease responses, for every 0.01 ml/s increase in flow, the HR response latency decreases by 1.7 s (β ± SE, −1.74 ± 0.53 s, P < 0.01), and HR response magnitude increases by 1.04% over baseline (β ± SE, 1.04 ± 0.40%, P = 0.01).
Fig. 2.
Effects of pharyngeal stimulus volume on HR. *P < 0.05 for comparison. A: HR response frequency is given as a percentage. The HR increase or decrease is increased with the higher stimulus volumes (0.3 and 0.5 ml). If a HR response occurs, HR decrease is more frequent. B: actual HR (in beats per minute) also significantly differs with HR response, with HR decrease greater in magnitude.
Effects of subject characteristics on HR changes.
The effects of subject characteristics (extreme prematurity, bronchopulmonary dysplasia, neuropathology, or gavage feeding) on HR changes are shown (Table 2). Note extreme prematurity had a significant impact on the magnitude of HR change, but no effect on HR change frequency, response latency, or response duration. Additionally, the presence of neuropathology, bronchopulmonary dysplasia, and gavage tube feeding did not have an impact on HR responses.
Table 2.
Effects of subject characteristics on HR response to pharyngeal stimuli
| Characteristic | Extreme Prematurity |
Bronchopulmonary Dysplasia |
Neuropathology |
Gavage Fed |
||||
|---|---|---|---|---|---|---|---|---|
| Yes | No | Yes | No | Yes | No | Yes | No | |
| Infants, n | 27 | 21 | 30 | 18 | 12 | 36 | 24 | 24 |
| Stimuli, n | 187 | 151 | 202 | 136 | 86 | 252 | 169 | 160 |
| Postmenstrual age, wk | 38.5 ± 0.3 | 39.0 ± 0.2 | 38.4 ± 0.2 | 38.4 ± 0.2 | 38.2 ± 0.5 | 39.9 ± 0.1 | 38.2 ± 0.6 | 38.6 ± 0.2 |
| Basal HR, beats/min | 151 ± 2 | 150 ± 3 | 150 ± 2 | 151 ± 3 | 151 ± 4 | 151 ± 2 | 149 ± 3 | 152 ± 3 |
| HR response (increase: decrease: none), % | 3: 39: 58 | 12: 22: 66 | 4: 35: 61 | 11: 27: 62 | 1: 41: 58 | 8: 30: 62 | 4: 33: 63 | 9: 31: 60 |
| HR response, beats/min | 131 ± 4* | 146 ± 4 | 134 ± 4 | 144 ± 4 | 128 ± 6 | 141 ± 3 | 135 ± 4 | 140 ± 4 |
| HR response latency, s | 10.7 ± 1.0 | 12.6 ± 1.2 | 11.3 ± 1.0 | 11.6 ± 1.2 | 12.8 ± 1.4 | 10.9 ± 0.9 | 12.0 ± 1.1 | 10.8 ± 1.1 |
| HR response duration, s | 12.4 ± 3.3 | 21.7 ± 4.4 | 14.4 ± 3.5 | 18.3 ± 4.4 | 16.8 ± 3.2 | 12.3 ± 5.1 | 13.6 ± 3.8 | 18.3 ± 3.8 |
| HR increase magnitude, % | 23 ± 4 | 21 ± 2 | 21 ± 3 | 22 ± 2 | 23 ± 7 | 21 ± 2 | 24 ± 3 | 20 ± 2 |
| HR decrease magnitude, % | 35 ± 2 | 28 ± 2* | 32 ± 2 | 32 ± 2 | 35 ± 2 | 31 ± 2 | 33 ± 2 | 32 ± 2 |
Values are means ± SE or percentages.
P < 0.05 vs. no category.
DISCUSSION
Salient findings of the current study in preterm born infants are as follows: 1) HR remains stable in a majority of pharyngeal provocations; 2) HR responses primarily occur after respiratory and pharyngeal activity; 3) HR decrease is related to aberrant pharyngoesophageal motility and respiratory dysregulation, resulting in repetitive swallowing, decreased esophageal propagation rates, and increased prolonged respiratory rhythm disturbance, all influenced by stimulus flow/volume; and 4) extreme preterm birth is a significant risk factor for heart rate decrease.
Cardiorespiratory events are a major concern in NICU infants pending discharge, often escalating parental and provider anxiety, as well as the socioeconomic burden from extensive diagnostic testing to identify potential causes (53, 54). This study attempted to systematically clarify the neurophysiological, aerodigestive, and cardiac regulatory mechanisms involving apneas, bradycardias, and desaturations in relation to pharyngeal provocation, such as may happen during oral feeding or during full-column reflux events. During spontaneous events, the causes and mechanisms can be mere speculation, resulting in escalating investigations and empiric therapies (53). By attempting to cautiously provoke the pharynx using minute quantities of sterile water, we examined the receptor-afferent-cross systems regulation-efferent effects in a controlled setting, measured as pharyngeal, esophageal, cardiac, and respiratory rate change characteristics. This study provides the basis for commonly seen symptoms, i.e., apnea, bradycardia, and desaturation amidst swallowing difficulties, which are often the reasons for prolonged hospital stays or readmissions after discharge from the NICU.
Measuring the Effects of the Parasympathetic System via Vagal Reflexes
Stimulus (cause)-induced effects of parasympathetic system immaturity (aerodigestive and cardiorespiratory responses) were measured; the characteristics of regulatory and adaptive motility functions are impaired in those that had bradycardic (decreasing) rhythms. An adaptive stress response occurs when homeostasis is maintained as when no symptoms occur, while maladaptation disrupts homeostasis manifesting as symptoms/events. The vagus modulates pharyngorespiratory interactions (23, 26) and can be associated with cardiac rhythm changes. It has also been shown that recovery and restoration to normalcy from apneic or hypoxic events may involve vagal influence on pharyngoesophageal motility and clearance (32, 37). In the current study, HR increase or no HR change may be considered adaptive responses, as swallowing characteristics were not altered in response to a measured stimulus. In contrast, HR decrease was associated with significantly altered swallowing characteristics and may be considered maladaptive and require attention. When bradycardia is occurring, in the monitored NICU setting, the provider is alerted to respond on the basis of the neonatal resuscitation protocol (6), and often times, the consequences of these bradycardic events are aborted. We are concerned about those infants who are in the unmonitored, high-risk domestic settings, and are at risk of sudden death.
We and others (35, 36, 40, 44, 47) have previously shown that maturational deficits in adapting to a bolus volume and flow can affect suck-swallow-breathing coordination, thus contributing to feeding difficulties and swallowing dysfunction. Subject maturation and stimulus volume significantly alter adaptive respiratory, pharyngeal, and esophageal responses to pharyngeal stimuli in human neonates (17, 25, 30). In this study, we found that differences in cardiac rate change response was significant in those <28 wk of birth gestation, despite similar PMA at evaluation. We noted that extreme premature birth had a significant impact on the magnitude of HR change but did not influence the cardiac response frequency, response latency, or response duration. This variation may be due to modifications from postnatal maturation and growth. Additionally, the presence of neuropathology, bronchopulmonary dysplasia, and gavage tube feeding did not have an impact on HR responses, and this may be due to adaptation responses. Furthermore, extremely preterm infants typically have greater frequency of nasooropharyngeal sensory-motor interventions (nasogastric tube, endotracheal tube, suctioning, nasal CPAP, or other supplemental oxygen delivery systems), albeit some of these can be nociceptive to result in HR changes (3). The increased prevalence of comorbidities may also limit the positive orosensory and oromotor experiences ex utero. On the other hand, infants born at ≥28 wk gestation have more intrauterine opportunities to swallow amniotic fluid, and earlier introduction of oral feedings associated with lesser duration of nasooropharyngeal sensory-motor interventions. Further cautious studies are needed to examine the sensory-motor effects and characteristics of early oral nutritive stimulus opportunities in these prototypes of NICU infants who are vulnerable to long-term morbidities, such as bronchopulmonary dysplasia, dysphagia, and life-threatening events.
Clinico-Physiological Reasoning for Study Findings
Before stimulation (baseline), we noted normal resting pharyngoesophageal and cardiorespiratory rhythm quiescence. With pharyngeal provocation, we noted four neurophysiological events: 1) pharyngeal rhythm changes, 2) esophageal peristaltic changes, 3) respiratory rhythm changes, and 4) cardiac rhythm changes. A sequential summary (adapted from response latencies and durations from Table 1) of pharyngoesophageal and cardiorespiratory rhythm responses grouped by HR increase or HR decrease can be observed in Fig. 3. The sequential pattern of reflexes suggests that neuromotor activation results in respiratory adaptation, pharyngeal swallowing, and esophageal peristalsis, which are the first aerodigestive responses to pharyngeal stimulation, and the HR increase occurs after the fact. The HR increase is likely a marker of cardiac stress response activated upon pharyngeal stimulation. This is likely a normal stress response if it occurs, as there were no functional derangements. On the other hand, over 60% of the stimuli were not associated with any HR changes, and there were no functional derangements. This can also be considered a normal response. Conversely, the HR decreased when the sequential activation of reflexes was associated with delayed esophageal peristalsis (Fig. 3). We hypothesize that weak, infrequent, and delayed esophageal peristalsis and or persistent stimulus within the aerodigestive tract may be associated with the bradycardic response. These latter situations are the ones that are concerning for potential aerodigestive pathologies and cardiorespiratory compromise.
Fig. 3.
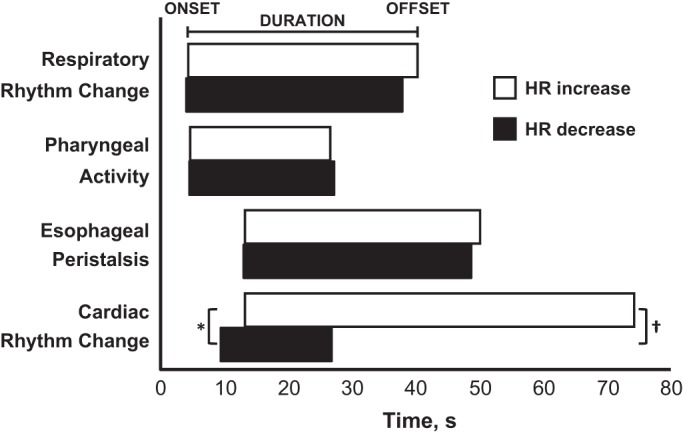
Diagrammatic representation of sequential timing of pharyngoesophageal and cardiorespiratory responses to pharyngeal stimuli. The figure was adapted from response latency and response duration values in Table 2. Along the x-axis is elapsed time with 0 s representing pharyngeal stimulus onset. *P < 0.05 for HR response onset. †P < 0.05 for HR response duration. Note that although respiratory, pharyngeal, and esophageal response timings were similar, the cardiac response is distinct between HR increase and HR decrease for both response onset and duration. Also note for HR decrease, cardiac response latency occurs after respiratory rhythm change and pharyngeal swallowing, but before esophageal peristalsis.
The phenomena observed in this study can be explained by the convergence of viscero-somatic-autonomic neural circuitry activities at the level of the brain stem, as explained before, for which the vagus nerve functions at multiple levels (42, 50). It is also likely that delays with vagus nerve maturation may be a factor involved with control and regulation of cross-system interactions, particularly with HR, and may involve receptors for vagal nerve afferents, brain stem nuclei, carotid sinuses, cardiac pacemaker, or the cardiac muscle (7, 15, 48). Alternatively, sympathetic nervous system, vagal efferents, endocrine influences, and neurotransmitters may be responsible, in addition to postreceptor signaling mechanisms (7, 19, 34, 49). In the current study, the frequency of HR decrease responses was more prevalent with increase in pharyngeal stimulus magnitude, suggesting progressive increase in cardiac vagal tone. The degree of HR (bpm) change with the HR decrease response was profound with an order of 50 bpm change from baseline, contrasting with no response or an HR increase response of up to ~20 bpm change from baseline HR. HR decrease responses were associated with greater stimulus volume, suggesting difficulties with pharyngeal, upper esophageal sphincter (UES), and esophageal body motility. This was evident as multiple PRS, poorer peristaltic propagation rates, altered and prolonged respiratory rhythm, along with prolonged deglutition apnea. In contrast, HR increase or no change in HR was associated with minimal adaptive shortcomings. Alterations in maturation of neurosensory and neuromotor pharyngoesophageal motility, and/or rapidly adaptive and slowly adaptive cardiorespiratory responses may be responsible for the troublesome symptoms, which further delay the acquisition of safe feeding, safe swallowing, and airway protection functions.
Potential Underlying Mechanisms for ALTE/BRUE
The current study findings serve as a framework for the further understanding of bradycardic rhythms by providing clinicopathological reasoning for commonly seen symptoms experienced by infants presenting with life-threatening events or BRUE, including apneas, bradycardias, and desaturations. These physiological metrics may be used to ascertain risk for BRUE occurrence, and/or to test the effects of therapies in modifying symptoms. It has been shown that if a bradycardia event occurs during pharyngeal stimuli, bradycardia was not an independent chemoreflex response, as it was preceded by apnea (8). However, in those studies, the pharyngeal, UES, and esophageal phase of swallowing was not evaluated during heart rate changes. Our study offers approaches to develop risk categories among infants with apparent life-threatening events or BRUEs (53). In those events in which HR decrease response was noted, we carefully examined them for causal mechanisms. Specifically, we noted heightened cardiac sensitivity with vagal parasympathomimetic effect in causing the following: HR to decrease (shorter response latency), prolonged inhibition of respiratory rhythm (deglutition apnea duration), efferent pharyngeal contractile activity (in the form of multiple pharyngeal contractions), rhythmic pharyngeal contractility (activated by the brain stem swallowing rhythms), and multiple pharyngeal reflexive swallows (as peristalsis, although less frequent full propagation rates). These cross-system adaptive responses reflect physiological responses; but exaggeration of such physiological responses or delays with adaptive recovery may result in perceived apparent life-threatening events or BRUEs. Further studies are needed to examine the opportunities for safe oral feeding, safe swallowing, and appropriate airway protection algorithms and strategies in infants at risk.
Limitations and Future Directions
Limitations of the current study exist as follows: 1) A graded dose escalation protocol was used as in previous studies (17, 20, 21, 25, 28, 30). Larger initial volumes may cause cardiorespiratory instability. Therefore, this dose escalation approach ensures subject safety and comfort before escalating to a higher stimulus volume to allow us to determine sensory thresholds, response latency, and response frequency. Although adaptation is possible, we administer three doses of the same volume before escalating to a higher dose and have shown volume-response relationships using this approach (17, 20, 21, 25, 28, 30). Further studies are needed to test adaptation using randomized stimulus volumes. 2) HR response was defined as a 10% change from baseline based on prior experience (13, 51). This threshold was chosen for the current study as a conservative definition and may be modified in future studies. Note as shown in Tables 1 and 2, the response magnitudes for HR increase and HR decrease were ~20–30%, respectively. This HR response threshold may be modified in the future to determine bradycardic pathology. 3) This was a limited heterogeneous study population from a single center. Further studies are needed to determine effects of recurrent LTE, neurological damage severity, and modes of respiratory support.
We and others have studied the group of laryngeal chemoreflex (LCR) in infants, which may serve to protect against aspiration and or increase airway vigilance expressed by startle, rapid swallowing, apnea, laryngeal constriction, hypertension, and/or bradycardia events (5, 8, 9, 14, 17, 23, 25, 26, 29, 33, 44, 52). Chemical composition of stimuli and maturation affect the LCR in that magnitude is increased at younger age (9, 44, 45). However, in these studies, the downstream protective vagal reflexes or potential therapeutic strategies were not examined. In infants with life-threating events, we have previously shown spontaneous respiratory events are primarily associated with esophageal dysmotility (16). As shown in the current study, HR decreases primarily occurred after respiratory and pharyngeal responses, but before esophageal responses, and was associated with esophageal dysmotility. Therefore, esophageal motility may be a potential therapeutic target for infants experiencing cardiorespiratory events, and adaptive pharyngoesophageal manometry may be a potentially useful diagnostic tool. Further cautious studies are needed to examine interregulation and adaptation between cardiorespiratory and pharyngoesophageal biorhythms, which are critical to feeding success. Reciprocal relationships between pharyngeal and respiratory biorhythms exist at younger maturational age and aerodigestive safety metrics advance with PMA and growth (17, 38, 39, 46). Knowledge about these mechanisms can provide anticipatory guidance to anxious parents and providers, as well as plans for safe feeding and discharge/follow up.
Conclusions
Pharyngeal-provoking stimulus is a driving factor resulting in changes in heart rate, pharyngeal rhythm, esophageal peristalsis, and respiratory rhythm. Under physiological conditions, the source of this stimulus can be regional secretions, oral bolus, retrograde esophageal bolus movement, or airway secretions. Parasympathetic control mechanisms are a common factor that links all of these effects and suggests that maturation of the convergence of viscero-somatic-autonomic neural circuitry at the level of brain stem and vagus nerve may play a role in clinically significant cardiopulmonary events. Physiological underdevelopment may be responsible for delays in acquisition of safe feeding, safe swallowing, and airway protection functions.
GRANTS
This study is supported in part by National Institutes of Health Grants RO1-DK-068158 (to S. R. Jadcherla) and PO1-DK-068051 (to R. Shaker, I. M. Lang, and S. R. Jadcherla).
DISCLOSURES
No conflicts of interest, financial or otherwise, are declared by the authors.
AUTHOR CONTRIBUTIONS
K.A.H. and S.R.J. conceived and designed research; K.A.H. and S.R.J. performed experiments; K.A.H., S.N., and S.R.J. analyzed data; K.A.H., S.N., I.M.L., R.S., and S.R.J. interpreted results of experiments; K.A.H. prepared figures; K.A.H., S.N., and S.R.J. drafted manuscript; K.A.H., S.N., I.M.L., R.S., and S.R.J. edited and revised manuscript; K.A.H., S.N., I.M.L., R.S., and S.R.J. approved final version of manuscript.
ACKNOWLEDGMENTS
We are grateful to Erika K. Osborn and Brittney Casey for providing clinical nursing support, demographic data extraction, and verification.
REFERENCES
- 1.American Academy of Pediatrics National Institutes of Health Consensus Development Conference on Infantile Apnea and Home Monitoring, Sept 29 to Oct 1, 1986. Pediatrics 79: 292–299, 1987. [PubMed] [Google Scholar]
- 2.American Academy of Pediatrics Committee on Fetus and Newborn Hospital discharge of the high-risk neonate. Pediatrics 122: 1119–1126, 2008. doi: 10.1542/peds.2008-2174. [DOI] [PubMed] [Google Scholar]
- 3.Anand KJ. Pain assessment in preterm neonates. Pediatrics 119: 605–607, 2007. doi: 10.1542/peds.2006-2723. [DOI] [PubMed] [Google Scholar]
- 4.Bakewell-Sachs S, Medoff-Cooper B, Escobar GJ, Silber JH, Lorch SA. Infant functional status: the timing of physiologic maturation of premature infants. Pediatrics 123: e878–e886, 2009. doi: 10.1542/peds.2008-2568. [DOI] [PubMed] [Google Scholar]
- 5.Ballester A, Gould F, Bond L, Stricklen B, Ohlemacher J, Gross A, DeLozier K, Buddington R, Buddington K, Danos N, German R. Maturation of the coordination between respiration and deglutition with and without recurrent laryngeal nerve lesion in an animal model. Dysphagia 33: 627–635, 2018. doi: 10.1007/s00455-018-9881-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Black L, Hulsey T, Lee K, Parks DC, Ebeling MD. Incremental hospital costs associated with comorbidities of prematurity. Manag Care 24: 54–60, 2015. [PubMed] [Google Scholar]
- 7.Broussard DL, Altschuler SM. Brainstem viscerotopic organization of afferents and efferents involved in the control of swallowing. Am J Med 108 (Suppl 4a): 79S–86S, 2000. doi: 10.1016/S0002-9343(99)00343-5. [DOI] [PubMed] [Google Scholar]
- 8.Davies AM, Koenig JS, Thach BT. Characteristics of upper airway chemoreflex prolonged apnea in human infants. Am Rev Respir Dis 139: 668–673, 1989. doi: 10.1164/ajrccm/139.3.668. [DOI] [PubMed] [Google Scholar]
- 9.Davies AM, Koenig JS, Thach BT. Upper airway chemoreflex responses to saline and water in preterm infants. J Appl Physiol (1985) 64: 1412–1420, 1988. doi: 10.1152/jappl.1988.64.4.1412. [DOI] [PubMed] [Google Scholar]
- 10.Eichenwald EC, Aina A, Stark AR. Apnea frequently persists beyond term gestation in infants delivered at 24 to 28 weeks. Pediatrics 100: 354–359, 1997. doi: 10.1542/peds.100.3.354. [DOI] [PubMed] [Google Scholar]
- 11.Eichenwald EC; Committee on Fetus and Newborn, American Academy of Pediatrics . Apnea of prematurity. Pediatrics 137: e1–e7, 2016. doi: 10.1542/peds.2015-3757. [DOI] [PubMed] [Google Scholar]
- 12.Fleming S, Thompson M, Stevens R, Heneghan C, Plüddemann A, Maconochie I, Tarassenko L, Mant D. Normal ranges of heart rate and respiratory rate in children from birth to 18 years of age: a systematic review of observational studies. Lancet 377: 1011–1018, 2011. doi: 10.1016/S0140-6736(10)62226-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Goldstein AB, Castile RG, Davis SD, Filbrun DA, Flucke RL, McCoy KS, Tepper RS. Bronchodilator responsiveness in normal infants and young children. Am J Respir Crit Care Med 164: 447–454, 2001. doi: 10.1164/ajrccm.164.3.2005080. [DOI] [PubMed] [Google Scholar]
- 14.Gould FD, Lammers AR, Ohlemacher J, Ballester A, Fraley L, Gross A, German RZ. The physiologic impact of unilateral recurrent laryngeal nerve (RLN) lesion on infant oropharyngeal and esophageal performance. Dysphagia 30: 714–722, 2015. doi: 10.1007/s00455-015-9648-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Goyal RK, Padmanabhan R, Sang Q. Neural circuits in swallowing and abdominal vagal afferent-mediated lower esophageal sphincter relaxation. Am J Med 111 (Suppl 8A): 95S–105S, 2001. doi: 10.1016/S0002-9343(01)00863-4. [DOI] [PubMed] [Google Scholar]
- 16.Hasenstab KA, Jadcherla SR. Respiratory events in infants presenting with apparent life threatening events: is there an explanation from esophageal motility? J Pediatr 165: 250–255.e1, 2014. doi: 10.1016/j.jpeds.2014.02.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Hasenstab KA, Sitaram S, Lang IM, Shaker R, Jadcherla SR. Maturation modulates pharyngeal-stimulus provoked pharyngeal and respiratory rhythms in human infants. Dysphagia 33: 63–75, 2018. doi: 10.1007/s00455-017-9833-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Jadcherla SR. Advances with neonatal aerodigestive science in the pursuit of safe swallowing in infants: invited review. Dysphagia 32: 15–26, 2017. doi: 10.1007/s00455-016-9773-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Jadcherla SR. Inflammation inhibits muscarinic signaling in in vivo canine colonic circular smooth muscle cells. Pediatr Res 52: 756–762, 2002. doi: 10.1203/00006450-200211000-00024. [DOI] [PubMed] [Google Scholar]
- 20.Jadcherla SR, Duong HQ, Hoffmann RG, Shaker R. Esophageal body and upper esophageal sphincter motor responses to esophageal provocation during maturation in preterm newborns. J Pediatr 143: 31–38, 2003. doi: 10.1016/S0022-3476(03)00242-7. [DOI] [PubMed] [Google Scholar]
- 21.Jadcherla SR, Duong HQ, Hofmann C, Hoffmann R, Shaker R. Characteristics of upper oesophageal sphincter and oesophageal body during maturation in healthy human neonates compared with adults. Neurogastroenterol Motil 17: 663–670, 2005. doi: 10.1111/j.1365-2982.2005.00706.x. [DOI] [PubMed] [Google Scholar]
- 22.Jadcherla SR, Durr B, Chan C, Slaughter J, Castile R, and Klebanoff M. Characteristics of infants admitted with an apparent life threatening event (ALTE) in the safe sleep era. E-Ped Acad Soc 2922.231, 2013. [Google Scholar]
- 23.Jadcherla SR, Gupta A, Coley BD, Fernandez S, Shaker R. Esophago-glottal closure reflex in human infants: a novel reflex elicited with concurrent manometry and ultrasonography. Am J Gastroenterol 102: 2286–2293, 2007. doi: 10.1111/j.1572-0241.2007.01401.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Jadcherla SR, Gupta A, Stoner E, Coley BD, Wiet GJ, Shaker R. Correlation of glottal closure using concurrent ultrasonography and nasolaryngoscopy in children: a novel approach to evaluate glottal status. Dysphagia 21: 75–81, 2006. doi: 10.1007/s00455-005-9002-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Jadcherla SR, Gupta A, Stoner E, Fernandez S, Shaker R. Pharyngeal swallowing: defining pharyngeal and upper esophageal sphincter relationships in human neonates. J Pediatr 151: 597–603, 2007. doi: 10.1016/j.jpeds.2007.04.042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Jadcherla SR, Gupta A, Wang M, Coley BD, Fernandez S, Shaker R. Definition and implications of novel pharyngo-glottal reflex in human infants using concurrent manometry ultrasonography. Am J Gastroenterol 104: 2572–2582, 2009. doi: 10.1038/ajg.2009.411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Jadcherla SR, Hasenstab KA, Shaker R, Castile RG. Mechanisms of cough provocation and cough resolution in neonates with bronchopulmonary dysplasia. Pediatr Res 78: 462–469, 2015. doi: 10.1038/pr.2015.131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Jadcherla SR, Hasenstab KA, Sitaram S, Clouse BJ, Slaughter JL, Shaker R. Effect of nasal noninvasive respiratory support methods on pharyngeal provocation-induced aerodigestive reflexes in infants. Am J Physiol Gastrointest Liver Physiol 310: G1006–G1014, 2016. doi: 10.1152/ajpgi.00307.2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Jadcherla SR, Parks VN, Peng J, Dzodzomenyo S, Fernandez S, Shaker R, Splaingard M. Esophageal sensation in premature human neonates: temporal relationships and implications of aerodigestive reflexes and electrocortical arousals. Am J Physiol Gastrointest Liver Physiol 302: G134–G144, 2012. doi: 10.1152/ajpgi.00067.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Jadcherla SR, Shubert TR, Gulati IK, Jensen PS, Wei L, Shaker R. Upper and lower esophageal sphincter kinetics are modified during maturation: effect of pharyngeal stimulus in premature infants. Pediatr Res 77: 99–106, 2015. doi: 10.1038/pr.2014.147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Khurana A, Thach BT. Effects of upper airway stimulation on swallowing, gasping, and autoresuscitation in hypoxic mice. J Appl Physiol (1985) 80: 472–477, 1996. doi: 10.1152/jappl.1996.80.2.472. [DOI] [PubMed] [Google Scholar]
- 32.Kiatchoosakun P, Dreshaj IA, Abu-Shaweesh JM, Haxhiu MA, Martin RJ. Effects of hypoxia on respiratory neural output and lower esophageal sphincter pressure in piglets. Pediatr Res 52: 50–55, 2002. doi: 10.1203/00006450-200207000-00011. [DOI] [PubMed] [Google Scholar]
- 33.Lang IM, Medda BK, Jadcherla SR, Shaker R. Characterization and mechanisms of the pharyngeal swallow activated by stimulation of the esophagus. Am J Physiol Gastrointest Liver Physiol 311: G827–G837, 2016. doi: 10.1152/ajpgi.00291.2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Lang IM, Medda BK, Shaker R, Jadcherla S. The effect of body position on esophageal reflexes in cats: a possible mechanism of SIDS? Pediatr Res 83: 731–738, 2018. doi: 10.1038/pr.2017.302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Lau C, Alagugurusamy R, Schanler RJ, Smith EO, Shulman RJ. Characterization of the developmental stages of sucking in preterm infants during bottle feeding. Acta Paediatr 89: 846–852, 2000. doi: 10.1111/j.1651-2227.2000.tb00393.x. [DOI] [PubMed] [Google Scholar]
- 36.Lau C, Sheena HR, Shulman RJ, Schanler RJ. Oral feeding in low birth weight infants. J Pediatr 130: 561–569, 1997. doi: 10.1016/S0022-3476(97)70240-3. [DOI] [PubMed] [Google Scholar]
- 37.Lumb KJ, Schneider JM, Ibrahim T, Rigaux A, Hasan SU. Afferent neural feedback overrides the modulating effects of arousal, hypercapnia and hypoxaemia on neonatal cardiorespiratory control. J Physiol 596: 6009–6019, 2018. doi: 10.1113/JP275682. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.MacFarlane PM, Ribeiro AP, Martin RJ. Carotid chemoreceptor development and neonatal apnea. Respir Physiol Neurobiol 185: 170–176, 2013. doi: 10.1016/j.resp.2012.07.017. [DOI] [PubMed] [Google Scholar]
- 39.Martin RJ, Abu-Shaweesh JM. Control of breathing and neonatal apnea. Biol Neonate 87: 288–295, 2005. doi: 10.1159/000084876. [DOI] [PubMed] [Google Scholar]
- 40.Mathew OP. Breathing patterns of preterm infants during bottle feeding: role of milk flow. J Pediatr 119: 960–965, 1991. doi: 10.1016/S0022-3476(05)83056-2. [DOI] [PubMed] [Google Scholar]
- 41.McInerny T, Adam H, Campbell D, DeWitt T, Foy J, Kamat D. American Academy of Pediatrics Textbook of Pediatric Care. Elk Grove Village, IL: American Academy of Pediatrics, 2017. [Google Scholar]
- 42.Miranda A, Peles S, Shaker R, Rudolph C, Sengupta JN. Neonatal nociceptive somatic stimulation differentially modifies the activity of spinal neurons in rats and results in altered somatic and visceral sensation. J Physiol 572: 775–787, 2006. doi: 10.1113/jphysiol.2006.108258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Montenegro BL, Amberson M, Veit L, Freiberger C, Dukhovny D, Rhein LM. Economics of home monitoring for apnea in late preterm infants. Respir Care 62: 42–48, 2017. doi: 10.4187/respcare.04954. [DOI] [PubMed] [Google Scholar]
- 44.Pickens DL, Schefft G, Thach BT. Prolonged apnea associated with upper airway protective reflexes in apnea of prematurity. Am Rev Respir Dis 137: 113–118, 1988. doi: 10.1164/ajrccm/137.1.113. [DOI] [PubMed] [Google Scholar]
- 45.Pickens DL, Schefft GL, Thach BT. Pharyngeal fluid clearance and aspiration preventive mechanisms in sleeping infants. J Appl Physiol (1985) 66: 1164–1171, 1989. doi: 10.1152/jappl.1989.66.3.1164. [DOI] [PubMed] [Google Scholar]
- 46.Poets CF. Apnea of prematurity: What can observational studies tell us about pathophysiology? Sleep Med 11: 701–707, 2010. doi: 10.1016/j.sleep.2009.11.016. [DOI] [PubMed] [Google Scholar]
- 47.Sakalidis VS, Kent JC, Garbin CP, Hepworth AR, Hartmann PE, Geddes DT. Longitudinal changes in suck-swallow-breathe, oxygen saturation, and heart rate patterns in term breastfeeding infants. J Hum Lact 29: 236–245, 2013. doi: 10.1177/0890334412474864. [DOI] [PubMed] [Google Scholar]
- 48.Sengupta JN. Electrophysiological recording from neurons controlling sensory and motor functions of the esophagus. Am J Med 111 (Suppl 8A): 169S–173S, 2001. doi: 10.1016/S0002-9343(01)00866-X. [DOI] [PubMed] [Google Scholar]
- 49.Sengupta JN. An overview of esophageal sensory receptors. Am J Med 108, Suppl 4a: 87S–89S, 2000. doi: 10.1016/S0002-9343(99)00344-7. [DOI] [PubMed] [Google Scholar]
- 50.Sengupta JN. Visceral pain: the neurophysiological mechanism. Handb Exp Pharmacol 194: 31–74, 2009. doi: 10.1007/978-3-540-79090-7_2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Shepherd EG, Clouse BJ, Hasenstab KA, Sitaram S, Malleske DT, Nelin LD, Jadcherla SR. Infant pulmonary function testing and phenotypes in severe bronchopulmonary dysplasia. Pediatrics 141: e20173350, 2018. doi: 10.1542/peds.2017-3350. [DOI] [PubMed] [Google Scholar]
- 52.Thach BT. Maturation and transformation of reflexes that protect the laryngeal airway from liquid aspiration from fetal to adult life. Am J Med 111, Suppl 8A: 69S–77S, 2001. doi: 10.1016/S0002-9343(01)00860-9. [DOI] [PubMed] [Google Scholar]
- 53.Tieder JS, Bonkowsky JL, Etzel RA, Franklin WH, Gremse DA, Herman B, Katz ES, Krilov LR, Merritt JL 2nd, Norlin C, Percelay J, Sapien RE, Shiffman RN, Smith MB; Subcommittee on Apparent Life Threatening Events . Brief resolved unexplained events (formerly apparent life-threatening events) and evaluation of lower-risk infants. Pediatrics 138: e20161488, 2016. [DOI] [PubMed] [Google Scholar]
- 54.Tieder JS, Cowan CA, Garrison MM, Christakis DA. Variation in inpatient resource utilization and management of apparent life-threatening events. J Pediatr 152: 629–635, 2008. doi: 10.1016/j.jpeds.2007.11.024. [DOI] [PubMed] [Google Scholar]
- 55.Wyckoff MH, Aziz K, Escobedo MB, Kapadia VS, Kattwinkel J, Perlman JM, Simon WM, Weiner GM, Zaichkin JG. Part 13: Neonatal resuscitation: 2015 American Heart Association Guidelines update for cardiopulmonary resuscitation and emergency cardiovascular care. Circulation 132 (Suppl 2): S543–S560, 2015. doi: 10.1161/CIR.0000000000000267. [DOI] [PubMed] [Google Scholar]