Abstract
Hippocampal mossy fiber axons simultaneously activate CA3 pyramidal cells and stratum lucidum interneurons (SLINs), the latter providing feedforward inhibition to control CA3 pyramidal cell excitability. Filopodial extensions of giant boutons of mossy fibers provide excitatory synaptic input to the SLIN. These filopodia undergo extraordinary structural plasticity causally linked to execution of memory tasks, leading us to seek the mechanisms by which activity regulates these synapses. High-frequency stimulation of the mossy fibers induces long-term depression (LTD) of their calcium-permeable AMPA receptor synapses with SLINs; previous work localized the site of induction to be postsynaptic and the site of expression to be presynaptic. Yet, the underlying signaling events and the identity of the retrograde signal are incompletely understood. We used whole cell recordings of SLINs in hippocampal slices from wild-type and mutant mice to explore the mechanisms. Genetic and pharmacologic perturbations revealed a requirement for both the receptor tyrosine kinase TrkB and its agonist, brain-derived neurotrophic factor (BDNF), for induction of LTD. Inclusion of inhibitors of Trk receptor kinase and PLC in the patch pipette prevented LTD. Endocannabinoid receptor antagonists and genetic deletion of the CB1 receptor prevented LTD. We propose a model whereby release of BDNF from mossy fiber filopodia activates TrkB and PLCγ1 signaling postsynaptically within SLINs, triggering synthesis and release of an endocannabinoid that serves as a retrograde signal, culminating in reduced glutamate release. Insights into the signaling pathways by which activity modifies function of these synapses will facilitate an understanding of their contribution to the local circuit and behavioral consequences of hippocampal granule cell activity.
NEW & NOTEWORTHY We investigated signaling mechanisms underlying plasticity of the hippocampal mossy fiber filopodial synapse with interneurons in stratum lucidum. High-frequency stimulation of the mossy fibers induces long-term depression of this synapse. Our findings are consistent with a model in which brain-derived neurotrophic factor released from filopodia activates TrkB of a stratum lucidum interneuron; the ensuing activation of PLCγ1 induces synthesis of an endocannabinoid, which provides a retrograde signal leading to reduced release of glutamate presynaptically.
Keywords: BDNF, hippocampus, interneuron, synaptic plasticity, TrkB
INTRODUCTION
Feedforward inhibition, a circuit configuration in which principal cells and inhibitory interneurons are excited simultaneously by a common afferent, controls excitability of principal cells as well as the timing of action potentials (Isaacson and Scanziani, 2011). Within the hippocampus, the mossy fiber axons of the dentate granule cells provide monosynaptic excitatory synaptic input onto both CA3 pyramidal cells and interneurons within stratum lucidum (SLINs), the anatomic underpinnings of a feedforward inhibitory circuit. The structures of mossy fiber terminals at these targets is unique in that giant boutons form synapses on CA3 pyramidal cells, whereas filopodial extensions of these same giant boutons form synapses on the interneurons (Acsády et al. 2008). These filopodia exhibit extraordinary structural plasticity in animals performing hippocampus-dependent memory tasks, a plasticity causally linked to proper execution of the behaviors (Guo et al. 2018; Ruediger et al. 2011). Long-lasting increases in numbers of these filopodia have also been identified in animal models of temporal lobe epilepsy (Danzer et al. 2010).
These structural plasticities underscore the importance of understanding the mechanisms by which activity regulates the function of filopodial synapses with SLINs. Whereas high-frequency stimulation (HFS) of the mossy fibers induces long-term potentiation (LTP) of the mossy fiber synapses with CA3 pyramidal cells, this same stimulus induces long-term depression (LTD) of mossy fiber synapses with SLINs (Maccaferri et al. 1998). The LTD consists of two different forms distinguished by whether the α-amino-3-hydroxy-5-methylisoxazole propionic acid (AMPA) receptors (AMPAR) at the synapse with the SLIN are calcium permeable. Induction of each form requires calcium postsynaptically, yet the locale of expression differs. Induction of LTD at calcium-impermeable AMPAR synapses requires N-methyl-d-aspartate (NMDA) receptor activation, and expression resides postsynaptically, mediated by reduced density of synaptic AMPARs (Lei and McBain 2004). Induction of LTD at calcium permeable-AMPAR synapses (mossy fiber-CP-AMPAR) also requires calcium postsynaptically, yet its expression resides presynaptically, due to reduced release of glutamate from presynaptic terminals (Lei and McBain 2004).
The signaling events underlying induction of LTD at mossy fiber-CP-AMPAR synapses and the identity of the retrograde signal are incompletely understood. The presence of brain- derived neurotrophic factor (BDNF) in filopodial extensions of mossy fiber boutons (Danzer and McNamara 2004) together with the requirement of the neurotrophin BDNF and its receptor, TrkB, for LTP of the mossy fiber-CA3 synapse (Huang et al. 2008) led us to hypothesize that TrkB signaling is required for LTD of the mossy fiber-CP-AMPAR synapse. We tested this hypothesis with whole cell recordings of SLINs in acutely isolated hippocampal slices from wild-type (WT) and genetically modified mice.
MATERIALS AND METHODS
Animals were handled according to the National Institutes of Health Guide for the Care and Use of the Laboratory Animals, and protocols were approved by Duke University Animal Care and Welfare Committee.
Mice.
BDNF and TrkB conditional mutant mice were generated as described previously (He et al. 2004). Crossing either a BDNF or a TrkB floxed mouse to a transgenic mouse carrying Cre driven by a Synapsin1 promoter (Syn-Cre) generates progeny in which expression of the floxed gene is selectively eliminated in a subset of central nervous system (CNS) neurons. TrkBF616A mice enabled a chemical-genetic method for inhibition of TrkB kinase; a modification of the TrkB locus (TrkBF616A) in which alanine is substituted for phenylalanine at residue 616 within kinase subdomain V renders TrkB sensitive to inhibition by a small molecule, 1-(1,1-dimethylethyl)-3-(1-naphthalenylmethyl)-1H-pyrazolo[3,4-d]pyrimidin-4-amine (1NMPP1). Importantly, in the absence of 1NMPP1, no differences in TrkB kinase activity or overt behavior are detectable in TrkBF616A compared with WT mice (Chen et al. 2005). ZnT3 null mutant mice were generated by crossing male and female heterozygotes (Cole et al. 1999). The strains of mice were generously provided by the following investigators: floxed BDNF and TrkB by Dr. Luis Parada (Memorial Sloan Kettering), Synapsin I-Cre by Dr. Jamey Marth (University of California, San Diego), TrkBF616A by Dr. David Ginty (Harvard), ZnT3 by Dr. Richard Palmiter (University of Washington), and CB1 by Giovanni Marsicano and Beat Lutz (Johannes Gutenberg University). The genotype of each animal was verified twice using PCR of genomic DNA isolated from the tail before and after experiments.
Hippocampal slice preparation and electrophysiological recording.
Male and female mice ages postnatal day 21–29 were anesthetized with pentobarbital sodium and decapitated, and hippocampal slices were prepared for field potential and whole cell recordings. The brain was quickly removed and placed in ice-cold buffer containing (in mM) 110 sucrose, 60 NaCl, 3 KCl, 1.25 NaH2PO4, 28 NaHCO3, 0.5 CaCl2, 7.0 MgCl2, and 5 dextrose, saturated with 95% O2-5% CO2, pH 7.4. Following dissection of hippocampi, transverse slices (400 μm in thickness) were cut with a vibratome and incubated in oxygenated artificial cerebrospinal fluid (aCSF) containing (in mM) 124 NaCl, 1.75 KCl, 1.25 KH2PO4, 26 NaHCO3, 2.4 CaCl2, 1.3 MgCl2, and 11 dextrose for at least 1 h at 32–34°C before recording. The slices were then transferred to a recording chamber mounted on a Zeiss Axioskop2 FS Plus upright microscope.
A bipolar tungsten stimulating electrode was placed near the junction of the granule cell layer and hilus near the midpoint of the suprapyramidal blade of the dentate. Synaptic responses were filtered at 2 kHz and digitized at 5 kHz. Extracellular recordings were obtained with a glass micropipette filled with 2 M NaCl, 2–6 MΩ resistance, placed in stratum lucidum near the junction of CA3a and CA3b. Following placement of the extracellular recording electrode, SLINs were identified by their bipolar or spindle shape visualized by infrared differential interference contrast microscopy. Whole cell recordings of interneurons were established using a glass micropipette filled with the following solution (in mM): 100 CsCl, 0.6 EGTA, 5 MgCl2, 8 NaCl, 40 HEPES, 2 MgATP, 0.3 Na3GTP, 0.1 spermine tetrahydrochloride, and 1 QX-314, pH 7.3. Picrotoxin (100 uM) was included in the external solution to block GABAA receptor-mediated events. d,l-2-Amino-5-phosphonovaleric acid (d,l-APV; 100 μM) was included in the perfusion solution to eliminate contamination of associational-commissural afferents (Maccafferi et al. 1998). Series resistances ranged from 7 to 15 MΩ and were monitored throughout the experiment and not compensated. Experiments were discontinued if the series resistance increased by >20%. Data were collected from slices at room temperature using a Multiclamp 700A amplifier and pClamp 9.2 software (Axon Instruments).
Synaptic events were evoked by a stimulus pulse (0.2-ms square-wave pulses delivered at 0.03 Hz with a DS3 Digitimer constant-current stimulator). In order for the SLIN to be identified as a calcium-permeable AMPAR (CP-AMPAR)-expressing interneuron, the following criteria were met: 1) injection of depolarizing current evoked high-frequency firing of action potentials with little or no adaptation; 2) the ratio of the amplitude of the second excitatory postsynaptic current (EPSC) to the first EPSC induced by paired pulses at a 60-ms interval did not exceed 1.5; 3) the rectification index as defined by Lei and McBain (2004) did not exceed 0.3; and 4) bath application of the group II metabotropic glutamate receptor (mGluR) agonist 2-(2,3-dicarboxycyclopropyl)glycine (DCG-IV; 1 µM) at the end of the experiment reduced the evoked EPSC by at least 70%, thereby confirming their mossy fiber origin. The effects of HFS on the mossy fiber synapse with CA3 pyramidal cells were examined in field potential recordings in a subset of experiments simultaneously with whole cell recordings of SLINs. In order for the CA3 pyramidal cell field potential to be considered an excitatory postsynaptic potential (fEPSP) evoked by mossy fibers, the following criteria were met: 1) the ratio for paired-pulse facilitation (PPF) at a 60-ms interval was 1.75 or greater; 2) frequency facilitation at 20 Hz was 2.0 or greater as determined by the ratio of the amplitude of the response to the third pulse compared with that to the first pulse (Toth et al. 2000); and 3) application of the DCG-IV (1 μM) at the end of the experiment reduced the amplitude of the evoked fEPSP by at least 70%.
LTD of the mossy fiber-SLIN synapse and LTP of the mossy fiber-CA3 synapse were evoked by HFS and assessed as follows. With respect to the mossy fiber-SLIN synapse, LTD was defined as the mean percent reduction of EPSC amplitude 10–20 min after HFS relative to that 10 min immediately preceding the HFS. Paired-pulse ratio (PPR) was calculated as the amplitude of the second synaptic response divided by the amplitude of the first synaptic response. The amplitude of fEPSPs was measured, and LTP was defined as the mean percent change in the amplitude 20 min after HFS relative to that 10 min immediately preceding the HFS. The numbers reported for whole cell recordings in the text and figure legends reflect the number of animals; in slices from approximately half of animals, recordings were obtained from more than one cell, in which case the results were averaged and reported as a single value for the animal. All data are presented as means ± SE. PPR data were analyzed by repeated-measures two-way ANOVA and a post hoc paired t-test. Unless specified otherwise, P values refer to results of paired t-tests comparing values collected 10–20 min following HFS with baseline (10 min immediately before HFS) values collected from the same cell. To assure objectivity, the individual performing all experiments with WT and mutant mice was blinded as to genotype.
Cell types.
A total of 323 cells were included in this work. Preliminary studies of biocytin-filled cells confirmed previous observations (Lei and McBain 2004; Tóth and McBain 1998) of CP-AMPARs on diverse morphologically defined subgroups of SLINs (data not shown).
RESULTS
Induction of LTD at mossy fiber-CP-AMPAR interneuron synapses within stratum lucidum requires postsynaptic calcium, yet its expression is due to reduced release of glutamate from mossy fiber afferents (Lei and McBain 2004). The mechanisms underlying its induction postsynaptically and the identity of the retrograde messenger are incompletely understood. The high level of expression of BDNF in mossy fiber terminals together with the requirement of the BDNF receptor TrkB for induction of presynaptic LTP of the mossy fiber-CA3 synapse (Huang et al. 2008) led us to hypothesize that TrkB activation is required for induction of LTD of the mossy fiber-CP-AMPAR synapse.
Mossy fiber EPSCs were evoked (holding potential Vhold = −65 mV, 0.03 Hz) in visually identified SLINs of acutely isolated mouse hippocampal slices by placement of a stimulating electrode in the dentate granule cell layer (Huang et al. 2008). Recordings of SLINs were restricted to cells exhibiting a rectification index of AMPAR-EPSCs of 0.3 or less, reflecting the presence of CP-AMPARs at the synapses (Lei and McBain 2004; Tóth and McBain 1998). To assure recording of synapses exhibiting LTD that is independent of NMDA receptors, d,l-APV (100 μM) was included in the solutions bathing the slices. To confirm the mossy fiber origin of EPSCs, we required an inhibition of at least 70% by addition of the group II mGluR agonist DCG-IV (1 μM) to the perfusing solution at the end of the experiment.
LTD of mossy fiber-CP-AMPAR synapse requires TrkB.
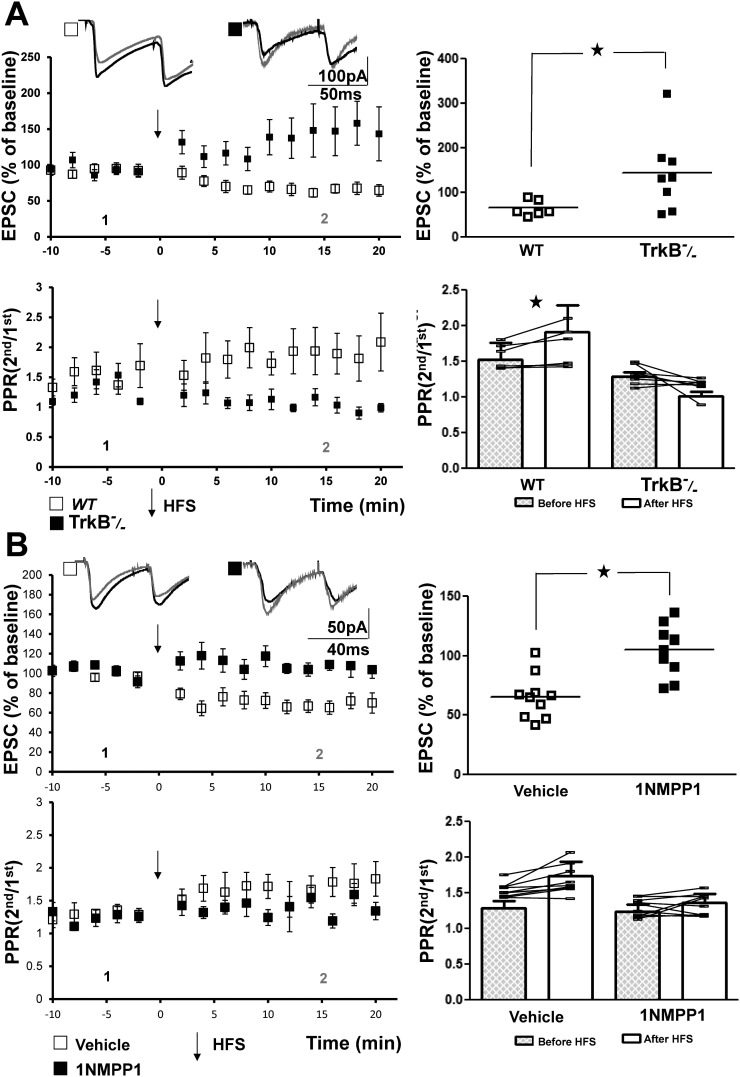
To address the requirement of TrkB signaling for LTD of the mossy fiber-CP-AMPAR synapse, hippocampal slices were isolated from conditional TrkB mutant mice in which exon one of the TrkB gene was floxed; crossing this line to Synapsin-Cre transgenic mice results in elimination of TrkB from the hippocampal dentate granule cells and other hippocampal neurons (He et al. 2004). HFS induced LTD in slices from WT animals (65 ± 7%, n = 6, P = 0.003). The LTD was associated with an increase of the PPR, consistent with a presynaptic locus of expression as shown by Lei and McBain (2004). In contrast, HFS-induced LTD of the mossy fiber-CP-AMPAR synapse was abolished in slices from TrkB conditional null mutant mice (Fig. 1A, top); indeed, HFS not only eliminated the decrease but also produced an increase of the EPSC in slices isolated from several TrkB conditional null mutant mice (5 of 8; Fig. 1A, top). Repeated-measures ANOVA analyses of PPR revealed significant differences between the WT and TrkB−/− mice (P = 0.005). In contrast to the significantly increased PPR in slices of WT mice (paired t-test, P = 0.03), the trend toward a decreased PPR in slices of TrkB−/− mice was not significant (P = 0.1; Fig. 1A). Together these findings demonstrate that TrkB is required for HFS-induced LTD of the mossy fiber-CP-AMPAR-SLIN synapse.
Fig. 1.
TrkB signaling is required for mossy fiber calcium permeable-AMPA receptor (mossy fiber-CP-AMPAR) long-term depression (LTD). A, top: representative traces (inset) show individual excitatory postsynaptic currents (EPSCs) at time points collected 10 min before (1, black) and between 10 and 20 min after (2, gray) application of high-frequency stimulation (HFS) in either wild-type (WT; □) or TrkB−/− (■) mice. Top left: HFS produced LTD of mossy fiber-CP-AMPAR stratum lucidum interneuron synapses in WT (65 ± 7%, n = 6, paired t-test, P = 0.003) but not in TrkB null mice (144 ± 30%, n = 8, paired t-test, P = 0.2). Top right: results of individual cells are plotted. Comparison of WT and TrkB−/− groups with Student’s t-test revealed a significant difference (*P = 0.01). Bottom: repeated-measures two-way ANOVA revealed a P value of 0.005 for paired-pulse ratio (PPR) data in the two groups. Post hoc paired t-tests revealed increased PPR in cells from WT (PPR was 1.5 ± 0.2 before and 1.9 ± 0.4 after HFS, n = 6, *P = 0.03) but not TrkB−/− mice (PPR was 1.3 ± 0.1 before and 1.0 ± 0.1 after HFS, n = 8, P = 0.1). B: chemical-genetic approach reveals requirement of TrkB kinase for mossy fiber-CP-AMPAR LTD in slices from TrkBF616A.mice. Inset presents representative traces of EPSCs collected 10 min before (1, black) and between 10 and 20 min after (2, gray) HFS of mossy fibers (□, vehicle group; ■, 2 μM 1NMPP1). Top left: treatment with 1NMPP1 impaired mossy fiber-CP-AMPAR LTD (105 ± 7%, n = 10, paired t-test, P = 0.3) compared with vehicle (68 ± 7%, n = 10, paired t-test, P = 0.001). Top right: results of individual animals are plotted. *P = 0.01, vehicle vs. 1NMPP1 groups (Student’s t-test). Bottom: repeated-measures two-way ANOVA revealed P = 0.089 for PPR data in the two groups.
We also used a chemical-genetic approach to assess the effect of TrkB signaling on LTD of the mossy fiber-CP-AMPAR-SLIN synapse. In contrast to the conditional null mutant in which TrkB protein has been eliminated from hippocampal neurons early in development, the chemical-genetic approach permitted addressing whether acutely inhibiting TrkB kinase in slices isolated from a juvenile mouse will also prevent LTD of the mossy fiber-CP-AMPAR-SLIN synapse. Substitution of alanine for phenylalanine in the kinase domain of TrkBF616A permits inhibition of TrkB kinase by addition of 1NMPP1 to the solution bathing the slices (Chen et al. 2005); importantly, in the absence of 1NMPP1, TrkB kinase functions similarly in WT and TrkBF616A mice (Chen et al. 2005). Inhibition of TrkB kinase activity inhibited HFS-induced LTD of the mossy fiber-CP-AMPAR-SLIN synapse. HFS of the mossy fibers reduced the amplitude of the mossy fiber-CP-AMPAR-SLIN EPSC in vehicle-treated slices from TrkBF616A mice (68 ± 7%, n = 10, P = 0.001, paired t-test; Fig. 1B). By contrast, in the presence of 1NMPP1 (2 μM), HFS failed to reduce the amplitude of the EPSC (105 ± 7%, n = 10, P = 0.3; Fig. 1B). Like the TrkB conditional mice, HFS resulted in an increase of the mossy fiber-CP-AMPAR-EPSC in a subset (4 of 10) of the 1NMPP1-treated slices from the TrkBF616A mice. Importantly, inclusion of 1NMPP1 (2 uM) in the bath of slices of WT animals did not prevent HFS-induced LTD of the mossy fiber-CP-AMPAR-SLIN synapse (70 ± 6%, n = 4, P < 0.05; data not shown). Taken together, evidence from both TrkB conditional null mutant mice and the TrkBF616A mice reveals a requirement for TrkB signaling for induction of LTD of the mossy fiber-CP-AMPAR-SLIN synapse.
Vesicular zinc is not required for LTD of mossy fiber-CP-AMPAR-SLIN synapse.
The requirement of TrkB signaling for LTD of the mossy fiber-CP-AMPAR-SLIN synapse raised the question as to the identity of the ligand that promotes this TrkB-dependent plasticity. In addition to the prototypic ligand of TrkB BDNF, we considered the possibility of zinc, because it is present in high concentration in synaptic vesicles of mossy fiber axon terminals (Haug 1967) and has been demonstrated to transactivate TrkB (Huang et al. 2008). Moreover, vesicular zinc together with BDNF contributes to induction of another TrkB-dependent plasticity, namely, LTP of the mossy fiber-CA3 synapse (Huang et al. 2008; Pan et al. 2011).
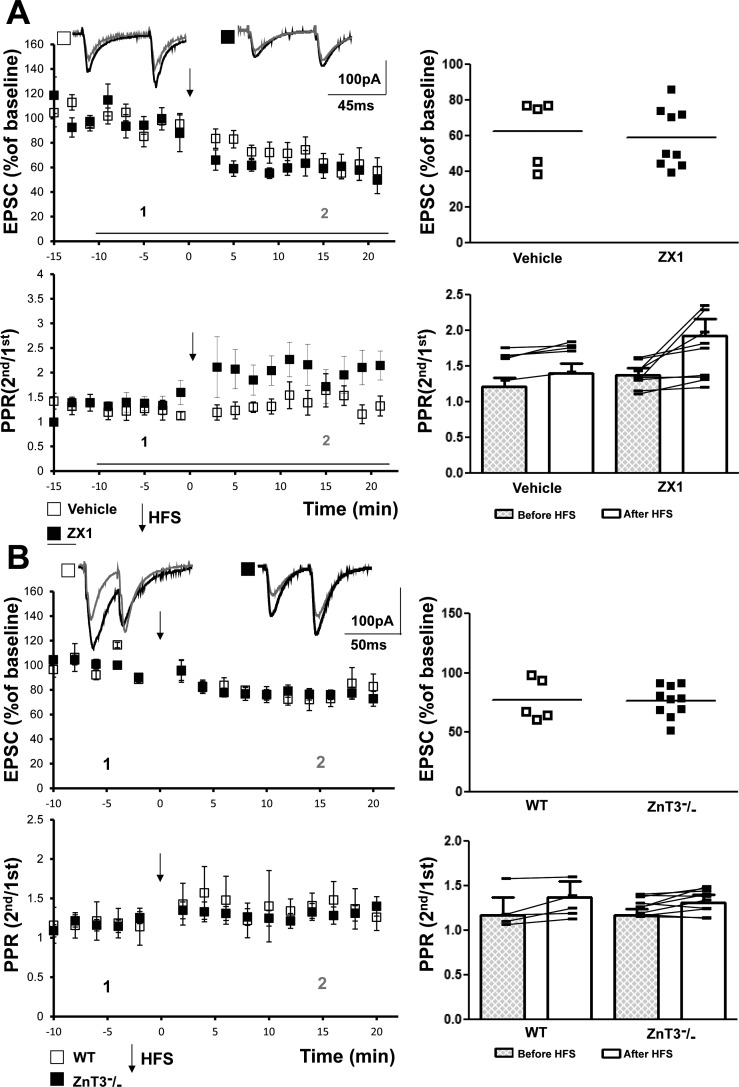
To assess the contribution of vesicular zinc to induction of mossy fiber-CP-AMPAR-SLIN LTD, we first examined the effects of ZX1, an extracellular zinc chelator with selectivity and kinetic properties suitable for study of the large transient of zinc in the synaptic cleft induced by mossy fiber stimulation (Pan et al. 2011). To our surprise, inclusion of ZX1 did not inhibit induction of LTD of the mossy fiber-CP-AMPAR-SLIN synapse or the associated increase of PPR (Fig. 2A). HFS of the mossy fibers induced a similar magnitude of LTD in the presence of vehicle (63 ± 9%, n = 5, P = 0.02) or ZX1 (59 ± 6%, n = 9, P = 0.0002; Fig. 2A).
Fig. 2.
Synaptically released zinc is not required for mossy fiber calcium permeable-AMPA receptor (mossy fiber-CP-AMPAR) long-term depression (LTD). A, top: representative traces (inset) show individual excitatory postsynaptic currents (EPSCs) at time points before (1, black) and after (2, gray) high-frequency stimulation (HFS) of mossy fibers. Top left: HFS induced similar amounts of LTD in the presence of vehicle (63 ± 9%, n = 5, P = 0.02, paired t-test) or ZX1 (59 ± 6%, n = 9, P = 0.0002, paired t-test). Top right: values from individual cells are plotted. Comparison of vehicle- and ZX1-treated groups with Student’s t-test revealed no significant differences (P = 0.4). Bottom: repeated-measures two-way ANOVA revealed a P value of 0.227 for paired-pulse ratio (PPR) data in the two groups. B, top: representative traces (inset) of mossy fiber-CP-AMPAR EPSC were collected before (1, black) and after (2, gray) HFS of mossy fibers. Top left: HFS induced LTD of mossy fiber-CP-AMPAR EPSC in slices from both WT (77 ± 8%, n = 5, paired t-test, P = 0.02) and ZnT3−/− mice (76 ± 4%, n = 10, paired t-test, P = 0.002). Top right: values from individual cells are plotted. Comparison of WT and ZnT3−/− mice with Student’s t-test revealed no significant difference (P = 0.5). Bottom: repeated-measures two-way ANOVA revealed a P value of 0.564 for PPR data in the two groups.
Importantly, field potential recordings of the mossy fiber-CA3 EPSP conducted simultaneously with the mossy fiber-CP-AMPAR-SLIN EPSC confirmed the previously observed effects of ZX1 on mossy fiber-CA3 LTP. That is, inclusion of ZX1 (100 μM) reduced the magnitude of LTP of the mossy fiber-CA3 synapse by 56% (vehicle: 157 ± 10%, n = 6; ZX1: 125 ± 12%, n = 5; P < 0.05, Student’s t-test; data not shown) in response to the same HFS in which no effect on mossy fiber-CP-AMPAR-SLIN LTD was detected.
We also addressed the effect of zinc on mossy fiber-CP-AMPAR-SLIN LTD in slices isolated from ZnT3 null mutant animals that lack vesicular zinc in CNS neurons (Cole et al. 1999). HFS induced similar amounts of LTD in slices from WT (77 ± 8%, n = 5, P = 0.02) and ZnT3−/− mice (76 ± 4%, n = 10, P = 0.002; Fig. 2B). Collectively, these pharmacological and genetic perturbations reveal that vesicular zinc is not required for induction of LTD of the mossy fiber-CP-AMPAR-SLIN synapse.
BDNF is required for LTD of mossy fiber-CP-AMPAR-SLIN synapse.
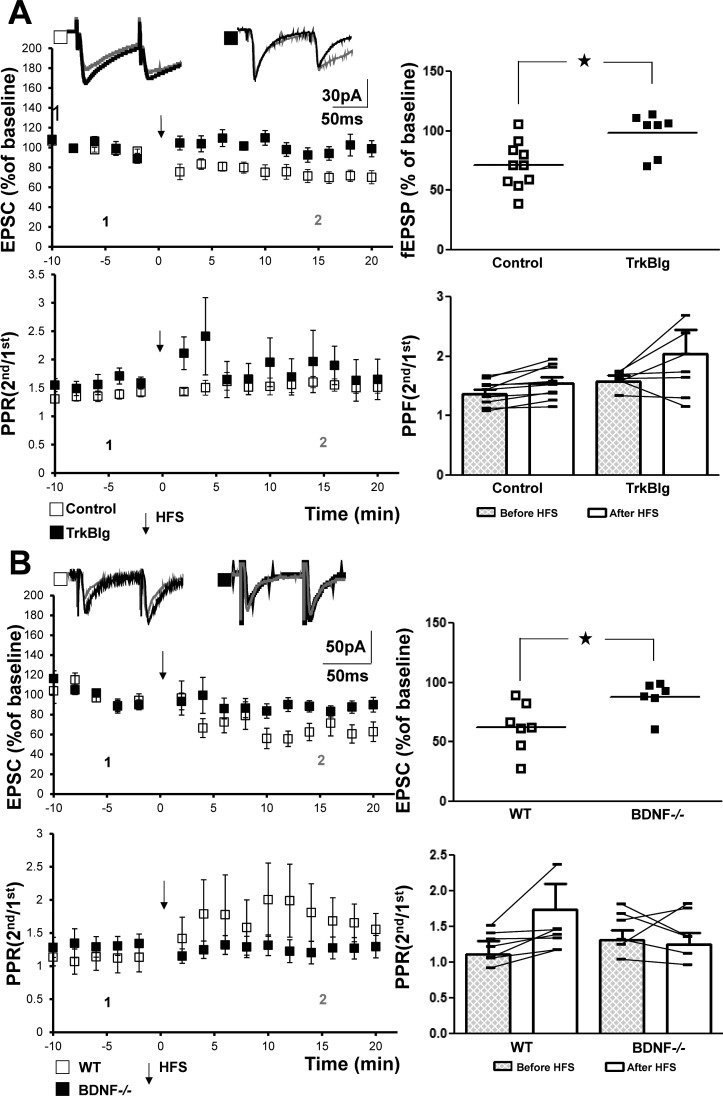
Next, we asked whether the prototypic ligand of TrkB, BDNF, is required for this plasticity. Notably, the presence of BDNF within the filopodial extensions of the mossy fiber boutons that innervate SLIN has been established (Danzer and McNamara 2004). Initially, we examined the effects of addition of TrkB-Ig to the solution bathing hippocampal slices acutely isolated from WT animals. TrkB-Ig is a recombinant protein in which the ligand-binding domain of human TrkB is fused to the Fc region of human IgG1 (Glass et al. 1996); its inclusion in the aCSF serves to scavenge extracellular BDNF. HFS induced LTD of the mossy fiber-CP- AMPAR-SLIN synapse in the presence of control IgG (72 ± 6%, n = 10, P = 0.005) but not in the presence of TrkB-Ig (98 ± 7%, n = 7, P = 0.4; Fig. 3A). Comparison of the magnitude of LTD in slices treated with IgG vs. TrkB-Ig revealed significant differences (P = 0.005, Student’s t-test). We also addressed the effect of BDNF on mossy fiber-CP-AMPAR-SLIN LTD in slices isolated from conditional BDNF mutant mice in which exon 5 of the BDNF gene was floxed; crossing this line to Synapsin-Cre transgenic mice results in elimination of BDNF from the hippocampal dentate granule cells and other hippocampal neurons (He et al. 2004).
Fig. 3.
Brain-derived neurotrophic factor (BDNF) is required for mossy fiber calcium permeable-AMPA receptor (mossy fiber-CP-AMPAR) long-term depression (LTD). A, top: representative traces (inset) of mossy fiber-CP-AMPAR excitatory postsynaptic current (EPSC) were collected before (1, black) and after (2, gray) high-frequency stimulation (HFS) of mossy fibers. Top left: HFS induced LTD of mossy fiber-CP-AMPAR synapses in the presence of control (72 ± 6%, n = 10, paired t-test, P = 0.005) but not in TrkB-Ig groups (98 ± 7%, n = 7, paired t-test, P = 0.4). Controls included 5 human IgG and 5 vehicle experiments; these were pooled for presentation because similar results were obtained with each. Top right: values from individual experiments are plotted. Comparison of control vs. TrkB-Ig with Student’s t-test revealed significant differences (*P = 0.005). Bottom, repeated-measures two-way ANOVA revealed a P value of 0.368 for paired-pulse ratio (PPR) data in the two groups. B, top: representative traces (inset) of mossy fiber-CP-AMPAR EPSC were collected before (1, black) and after HFS (2, gray). Top left: HFS induced LTD of mossy fiber-CP-AMPA synapse in slices from WT (63 ± 8%, n = 7, paired t-test, P = 0.002) and conditional BDNF mutant mice (88 ± 6%, n = 6, paired t-test, P = 0.04). Top right: values from individual experiments are plotted. Comparison of the LTD evident in WT vs. conditional BDNF mutant mice with Student’s t-test revealed a significant difference (P = 0.01). Bottom: repeated-measures two-way ANOVA revealed a P value of 0.06 for PPR data in the two groups.
Elimination of the BDNF gene attenuated induction of LTD of the mossy fiber-CP-AMPAR-SLIN synapse. HFS induced significant LTD in slices from both WT (63 ± 8%, n = 7, P = 0.002) and conditional BDNF−/− mice (88 ± 6%, n = 6, P = 0.04; Fig. 3B). Importantly, the magnitude of the LTD in slices from the WT was significantly greater than in slices from the BDNF−/− mice (Student’s t-test, P = 0.01). Collectively, these findings demonstrate a requirement for BDNF for HFS-induced LTD of the mossy fiber-CP-AMPAR-SLIN synapse.
Activation of TrkB and PLC within a SLIN is required for induction of LTD.
Our findings demonstrate that BDNF-mediated activation of TrkB is required for LTD of the mossy fiber-CP-AMPAR interneuron synapse. The increased PPR accompanying LTD in slices from WT animals supports previous work establishing a presynaptic locus of expression of LTD (Lei and McBain 2004). Left unanswered is the locale at which TrkB activation occurs and the signaling pathway downstream of TrkB that promotes LTD. That inclusion of the calcium chelator BAPTA within the SLIN prevented induction of LTD (Laezza et al. 1999) led us to favor two hypotheses: 1) that TrkB intrinsic to the SLIN itself mediated induction of LTD, or 2) that the signaling pathway by which TrkB activation induced LTD involved PLCγ1, because activation of this enzyme hydrolyzes phosphatidyl inositol 4,5-bisphosphate (PIP2), resulting in formation of inositol (1,4,5)-trisphosphate (IP3), with the ensuing activation of the IP3 receptor evoking release of calcium from endoplasmic reticulum.
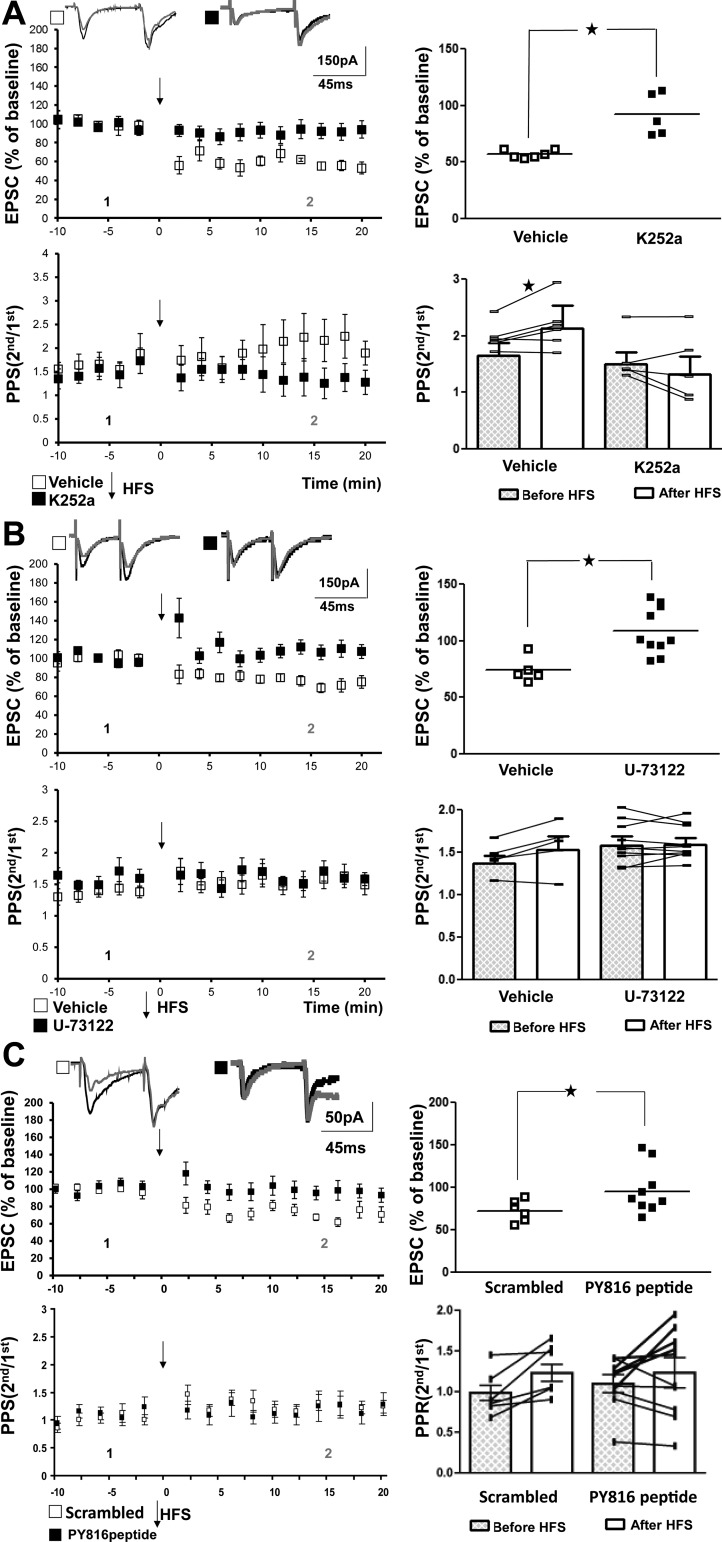
To address the first of these hypotheses, we asked whether inclusion of K252a, a Trk kinase inhibitor, within a CP-SLIN prevented induction of LTD. Inclusion of K252a (200 nM) in the patch pipette inhibited induction of LTD (vehicle: 57 ± 2%, n = 6, P = 0.0002; K252a: 92 ± 9%, n = 5, P = 0.2; Fig. 4A). Significant increases of PPR were found in cells from vehicle but not K252a experiments (Fig. 4A). These results provide additional evidence of a requirement for TrkB for LTD and suggest a postsynaptic locus of its activation.
Fig. 4.
Inclusion of inhibitors of Trk/PLC signaling in stratum lucidum interneurons inhibits mossy fiber calcium permeable-AMPA receptor (mossy fiber-CP-AMPAR) long-term depression (LTD). A, top left: inclusion of K252a (200 nM) in the recording pipette prevented high-frequency stimulation (HFS)-induced mossy fiber-CP-AMPAR LTD (vehicle: 57 ± 2%, n = 6, paired t-test, P = 0.00002; K252a: 92 ± 9%, paired t-test, P = 0.2). Top right: results of individual cells are plotted. Comparison of vehicle vs. K252a groups with Student’s t-test revealed a significant difference (*P = 0.007). Bottom: repeated-measures two-way ANOVA revealed a P value of 0.029 for paired-pulse ratio (PPR) data in the two groups. Post hoc paired t-tests revealed increased PPR in cells from vehicle (1.7 ± 0.2 before and 2.1 ± 0.4 after, paired t-test, P = 0.03) but not K2525a (1.5 ± 0.2 before and 1.3 ± 0.3 after, paired t-test, P = 0.2). B, top left: inclusion of U-73122 (5 μM) in recording pipette prevented HFS-induced mossy fiber-CP-AMPAR LTD (vehicle: 74 ± 5%, n = 5, paired t-test, P = 0.003; U-73122: 109 ± 7%, n = 10, paired t-test, P = 0.11). Top right: results of individual cells are plotted. Comparison of vehicle vs. U-73122 groups with Student’s t-test revealed a significant difference (*P = 0.0005). Bottom: repeated-measures two-way ANOVA revealed a P value of 0.058 for PPR data in the two groups. C, top left: inclusion of pY816 peptide (10 µM) in recording pipette prevented HFS-induced mossy fiber CP-AMPAR LTD (Scramble peptide: 73 ± 5, n = 6, paired t-test, P = 0.003; pY816 peptide: 96 ± 9, n = 10, paired t-test, P = 0.5). Top right: results of individual cells are plotted. Comparison of scrambled vs. pY816 peptide groups with Student’s t-test revealed a significant difference (*P = 0.04). Bottom: repeated-measures two-way ANOVA revealed a P value of 0.125 for PPR data in the two groups.
To address the second hypothesis, we asked whether inclusion of U-73122, a phospholipase inhibitor, within a CP-SLIN prevented induction of LTD. Inclusion of U-73122 (5 μM) in the patch pipette inhibited induction of LTD (vehicle: 74 ± 5%, n = 5, P = 0.003; U-73122: 109 ± 7%, n = 10, P = 0.11; Fig. 4B). Among the family of phospholipase enzymes, PLCγ1 in particular is bound and activated by TrkB (Middlemas et al. 1994). To address the contribution of PLCγ1 in particular, we examined the effects of a peptide (pY816) that binds the SH2 domain of PLCγ1 and thereby inhibits TrkB-mediated activation of PLCγ1 both in vitro and in vivo compared with a scrambled control peptide (Gu et al. 2015). Inclusion of pY816 peptide (10 µM) in the recording pipette prevented LTD (Scr peptide: 73 ± 5, n = 6, paired t-test P = 0.003; pY816 peptide: 96 ± 9, n = 10, paired t-test P = 0.5; Fig. 4C). Collectively, these data support the idea that TrkB-mediated activation of PLCγ1 signaling within a SLIN is required for induction of LTD of the mossy fiber-CP-AMPAR-SLIN synapse.
An endocannabinoid serves as retrograde messenger.
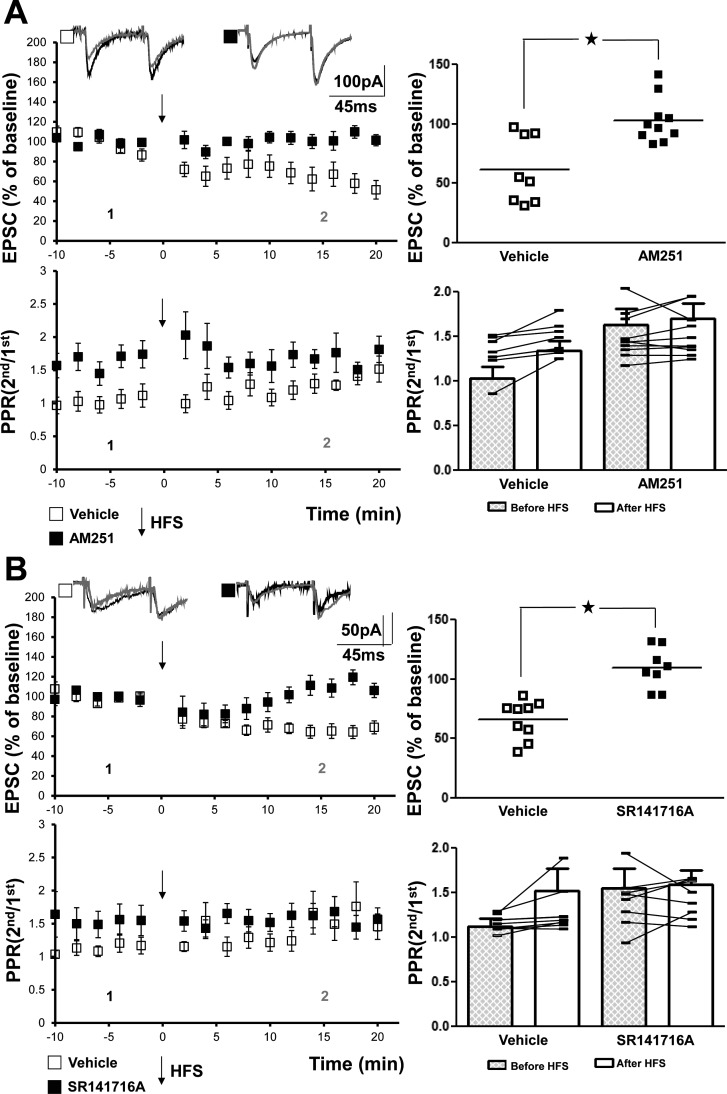
The locus of expression of LTD of the mossy fiber-CP-AMPAR synapse is thought to reside presynaptically in the filopodial extension of mossy fiber terminals, a conclusion supported by multiple measures including increases of PPR accompanying the LTD (Lei and McBain 2004). That inclusion of BAPTA postsynaptically within the CP-SLIN prevented LTD (Laezza et al. 1999; Lei and McBain 2002) together with the above findings with K252a, U-73122, and pY816 implies the existence of a retrograde signal that somehow instructs the presynaptic terminal to reduce glutamate release. Our pharmacological evidence implicating a requirement for TrkB/PLCγ1 signaling within a SLIN for induction of LTD led us to hypothesize that the retrograde signal may be an endocannabinoid (Zhao et al. 2015). We therefore asked whether an antagonist of CB1 receptors prevents induction of mossy fiber-CP-AMPAR-SLIN LTD. Inclusion of the CB1 receptor antagonist AM 251 (5 µM) in the bath abolished mossy fiber-CP-AMPAR-SLIN LTD (vehicle: 61 ± 10%, n = 8, P = 0.003; AM 251: 102 ± 6%, n = 10, P = 0.2; Fig. 5A); field potential recordings of CA3 pyramidal cell responses performed in a subset of these experiments revealed no detectable inhibition of LTP of mossy fiber-CA3 EPSP (vehicle: 151 ± 11%, n = 6, P = 0.01; AM 251: 165 ± 17%, n = 8, P = 0.009) or the associated reduction of PPR examined in simultaneous field potential recordings (not shown). Similar results were obtained with a structurally distinct CB1 receptor antagonist, SR141716A. Like AM 251, inclusion of SR141716A (5 µM) abolished mossy fiber-CP-AMPAR LTD (vehicle: 66 ± 5%, n = 9, P = 0.0001; SR141716A: 109 ± 6%, n = 8, P = 0.08; Fig. 5B). Field potential recordings of CA3 pyramidal cell responses performed in a subset of experiments examining mossy fiber LTD revealed no detectable effect of SR141716A on LTP of the mossy fiber-CA3 EPSP (vehicle: 142 ± 7%, n = 5, P = 0.04; SR141716A: 152 ± 14%, n = 6, P = 0.04; not shown). In sum, each of two CB1 receptor antagonists inhibited induction of mossy fiber-CP-AMPAR-SLIN LTD, yet had no effect on induction of mossy fiber-CA3 LTP.
Fig. 5.
Bath application of CB1 receptor antagonists prevents mossy fiber calcium permeable-AMPA receptor (mossy fiber-CP-AMPAR) long-term depression (LTD). A, top left: inclusion of AM251 (5 μM) prevented high-frequency stimulation (HFS)-induced mossy fiber-CP-AMPAR LTD (vehicle: 61 ± 10%, n = 8, paired t-test, P = 0.003; AM251: 102 ± 6%, n = 10, paired t-test, P = 0.2). Top right: results of individual cells are plotted. Comparison of vehicle vs. AM251 groups with Student’s t-test revealed a significant difference (*P = 0.002). Bottom: repeated-measures two-way ANOVA revealed a P value of 0.124 for paired-pulse ratio (PPR) data in the two groups. B, top left: inclusion of SR141716A (5 μM) prevented HFS-induced mossy fiber-CP-AMPAR LTD (vehicle: 66 ± 5%, n = 9, paired t-test, P = 0.0001; SR141716A: 109 ± 6%, n = 8, paired t-test, P = 0.08). Top right: results of individual cells are plotted. Comparison of vehicle vs. SR141716A groups with Student’s t-test revealed a significant difference (*P = 0.00005). Bottom: repeated-measures two-way ANOVA revealed a P value of 0.205 for PPR data in the two groups.
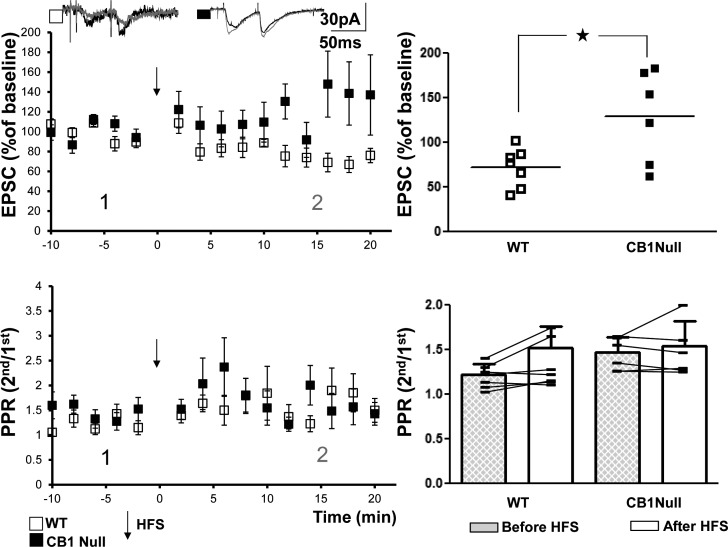
The presence of off target effects of CB1 receptor antagonists (Bukoski et al. 2002; Jin et al. 2004) led us to seek genetic evidence to confirm a CB1 receptor requirement for mossy fiber-CP-AMPAR-SLIN LTD. HFS induced LTD in slices from WT animals (73 ± 8%, n = 7, P = 0.006). In contrast, HFS-induced LTD of the mossy fiber-CP-AMPAR-SLIN synapse was abolished in slices from CB1 receptor null mutant mice (129 ± 21%, n = 6, P = 0.1; Fig. 6A, top). Field potential recordings of CA3 pyramidal cell responses performed in a subset of experiments examining mossy fiber LTD revealed induction of LTP in slices from both WT (125 ± 3%, n = 5, P = 0.001) and CB1 receptor null mutant mice (138 ± 16%, n = 5, P = 0.03). In sum, these data reveal the requirement of CB1 receptors for LTD of the mossy fiber-CP- AMPAR-SLIN synapse but not for LTP of the mossy fiber-CA3 synapse.
Fig. 6.
High-frequency stimulation (HFS)-mediated induction of mossy fiber calcium permeable-AMPA receptor (mossy fiber-CP-AMPAR) long-term depression (LTD) is impaired in slices from CB1 receptor knockout (CB1R−/−) mice. Top left: HFS induced LTD in slices from wild-type (WT) but not CB1R−/− mice (WT: 73 ± 8%, n = 7, paired t-test, P = 0.006; CB1R−/−: 129 ± 21%, n = 6, paired t-test, P = 0.1). Top right: results of individual cells are plotted. Comparison of vehicle vs. CB1R−/− mice with Student’s t-test revealed a significant difference (*P = 0.02). Bottom: repeated-measures two-way ANOVA revealed a P value of 0.309 for paired-pulse ratio (PPR) data in the two groups.
DISCUSSION
We used whole cell recordings of SLINs receiving mossy fiber-CP-AMPAR synapses in hippocampal slices acutely isolated from WT and mutant mice to explore the mechanisms mediating HFS-induced LTD. Five principal findings emerged. 1) Either a conditional deletion of TrkB or acute inhibition of TrkB kinase with a chemical-genetic method prevented LTD. 2) In contrast to presynaptic LTP of the mossy fiber-CA3 synapse, neither genetic elimination of vesicular zinc nor application of an extracellular zinc chelator prevented LTD. 3) Both conditional deletion of BDNF and acute scavenging of extracellular BDNF prevented LTD. 4) Inclusion of inhibitors of Trk receptor kinase and phospholipase as well as a peptide inhibitor of the binding of TrkB to PLCγ1 in the patch pipette solution prevented LTD. 5) Either deletion of the CB1 receptor or bath application of CB1 receptor antagonists prevented induction of LTD. These findings elucidate the signaling mechanisms by which HFS induces LTD of the subset of mossy fiber synapses with SLINs that utilize CP-AMPARs.
Our findings confirm and extend earlier analyses of the mechanisms underlying NMDA receptor-independent LTD of the mossy fiber-SLIN synapse. Multiple criteria, including coefficient of variation and PPR, revealed that expression of LTD of mossy fiber-CP-AMPAR synapses was due to reduced release of glutamate from mossy fiber terminals (Lei and McBain 2004). Although expressed presynaptically, its induction required calcium signaling within the postsynaptic cell, because it was prevented by inclusion of the calcium chelator BAPTA within the SLIN (Laezza et al. 1999; Lei and McBain 2002). Its induction was also inhibited by bath application of (R,S)-α-methylserine-O-phosphate (MSOP), a compound selectively inhibiting the group III mGluR7 under the conditions of the experiment (Pelkey et al. 2005). Interestingly, induction of chemical LTD of the mossy fiber-SLIN synapse by combining bath application of AP4 with low-frequency stimulation of the mossy fibers induced internalization of mGluR7 from the mossy fiber terminals contacting the SLINs (Pelkey et al. 2005). Left unanswered was the source of calcium within the SLIN as well as the chain of events by which the SLIN signals the presynaptic terminal to induce activation and internalization of mGluR7.
Our findings are consistent with a model whereby HFS of the mossy fibers induces release of BDNF from a filopodial terminal, leading to activation of TrkB of a SLIN, thereby triggering activation of PLCγ1. We suggest that the TrkB-mediated activation of PLCγ1 promotes catalysis of phosphatidylinositol 4,5-bisphosphate (PIP2), producing diacylglycerol (DAG) and IP3. IP3 binds its receptor in the endoplasmic reticulum, triggering release of calcium and activation of calcium-regulated signaling pathways. We further suggest that catalysis of DAG by diacyl-arachidonoyl lipase (DAGL) produces 2-arachidonyl glycerol (2-AG), which is released and binds to and activates CB1 receptors (Mechoulam et al. 1995; Sugiura et al. 1995), the activation of which leads to sustained inhibition of glutamate release from mossy fiber terminals. The presence of DAGL in interneurons of CA3 (Péterfi et al. 2012) is consistent with this model. Although the rise of calcium due to activation of CP-AMPARs alone was not sufficient to induce LTD here, it likely promotes activation of PLCγ1, because the coincidence of postsynaptic calcium rise and Gq receptor activation markedly enhanced PLCβ-dependent endocannabinoid release (Hashimotodani et al. 2007). Inhibition of LTD by CB1 receptor antagonists and in CB1 receptor knockout mice clearly implicates CB1 receptors yet does not reveal the cellular locale of these receptors. One possibility is that the endocannabinoid activated CB1 receptors on astrocytes, triggering release of glutamate (Navarrete and Araque 2010) and subsequent activation of mGlu7, resulting in its internalization from the filopodial terminal membrane and reduced release of glutamate expressed as LTD (Pelkey et al. 2005). Although this proposed sequence of events provides a parsimonious explanation for our findings, we recognize that additional experiments are required to validate the model. A similar signaling pathway using TrkB-mediated activation of PLC-γ1 may contribute to theta burst-induced LTD of an inhibitory neuron synapse onto cortical layer 2/3 pyramidal neurons (Zhao et al. 2015).
Unexpectedly, HFS of mossy fibers not only inhibited LTD but also induced LTP in a subset of TrkB−/− mice (Fig. 1A). By contrast, acute inhibition of TrkB kinase activity using the chemical genetic method or K252a inhibited the LTD but did not produce LTP (median of the group response approximated 100% in each; Figs. 1B and 4A). One explanation for these inconsistencies may be the duration of reduced TrkB signaling before stimulation of mossy fibers, being lifelong in the TrkB−/− mice but only tens of minutes with the chemical-genetic inhibition of TrkB kinase or with K252a. Alternatively, these differences may be due to the absence of TrkB protein itself in the TrkB−/− mice compared with simply inhibiting TrkB kinase activity with 1NMPP1 or K252a. Importantly, the TrkB protein can undergo transactivation by zinc (and likely other factors) and convey signals even when TrkB kinase is inhibited (Huang and McNamara 2010).
Some interesting similarities and differences emerge with respect to the mechanisms underlying plasticity of the giant bouton synapses on CA3 pyramidal cells compared with the synapses formed by filopodial extensions of these boutons with SLINs. Although the sign of the plasticity is opposite (LTP vs. LTD), expression of the plasticity resides presynaptically for each. Inclusion of the calcium chelator BAPTA postsynaptically prevents mossy fiber SLIN LTD (Lei and McBain 2002) but not mossy fiber CA3 LTP (Pan et al. 2011), supporting a locus of induction postsynaptically for LTD but presynaptically for LTP. These differences notwithstanding, BDNF, TrkB, and PLC contribute to induction of each. Whereas vesicular zinc is required for induction of mossy fiber CA3 LTP (Pan et al. 2011), present results reveal no detectable role in mossy fiber SLIN LTD. Finally, the present results reveal a critical role for endocannabinoid signaling for LTD yet no contribution to mossy fiber CA3 LTP. These differences add to previous evidence demonstrating the compartmentalization of molecular signaling events in the filopodia compared with the giant boutons from which they arise (Maccaferri et al. 1998; Pelkey et al. 2005).
The present findings reveal a heretofore unrecognized role of BDNF in LTD of excitatory synapses on interneurons of the mammalian CNS. Neurotrophin signaling, specifically proBDNF, has been previously implicated in LTD of excitatory synapses on principal neurons. BDNF is translated as proBDNF, which binds p75NTR. Application of recombinant proBDNF promotes low-frequency stimulation-induced LTD of the excitatory synapse of Schaffer collateral axons with hippocampal CA1 pyramidal cells through p75NTR (Woo et al. 2005). Consistent with this, transgenic mice expressing a cleavage-resistant form of proBDNF exhibited exaggerated LTD compared with WT controls following low-frequency stimulation of Schaffer collateral axons (Yang et al. 2014). Interestingly, proBDNF has been found to be cleaved intracellularly into two proteins, the pro domain and mature BDNF, each of which is packaged in dense core vesicles and presumably released, raising the intriguing question as to the receptor for the pro domain and its biological effects (Dieni et al. 2012). In any case, the present findings differ in several ways from proBDNF-induced LTD. Here the synapse is formed by an excitatory afferent on an interneuron, not a principal cell. The pattern of afferent stimulation required for induction of LTD consisted of high, not low, frequency. Finally, the receptor mediating the LTD was not p75, but rather TrkB, as evident by both conditional deletion of TrkB and inhibition of TrkB kinase.
Connectivity of mossy fibers with SLINs has been implicated in the precision of both recent and remote memories (Guo et al. 2018; Ruediger et al. 2011). Degradation of this precision is evident in pathological memory decline with aging as well as in intrusive flashbacks unrelated to the initiating event in posttraumatic stress disorder. Control of excitability of CA3 pyramidal cells is thought to be critical to the precision of recall, with control mediated in part by filopodial connections with interneurons that mediate feedforward inhibition of CA3 pyramidal cells. Memory tasks in mice induce increased numbers of filopodial connections with SLINs, an event shown to be critical to precision of the memory (Guo et al. 2018; Ruediger et al. 2011). Our findings reveal mechanisms underlying the plasticity of filopodial synapses with a subset of these interneurons. These insights will facilitate elucidating how these synapses contribute to the function of these circuits and their role in the precision of memory in both health and disease.
GRANTS
This work was supported by National Institute of Neurological Disease and Stroke Grant NS097717 (to J. O. McNamara).
DISCLOSURES
No conflicts of interest, financial or otherwise, are declared by the authors.
AUTHOR CONTRIBUTIONS
J.O.M. conceived and designed research; E. Pan collected and analyzed the electrophysiology data; Z. Zhao colllected and analyzed the morphological data; J.O.M. interpreted results of experiments; J.O.M. edited and revised manuscript; J.O.M. approved final version of manuscript.
ACKNOWLEDGMENTS
We thank Wei-Hua Qian for support of mouse husbandry and genotyping and Elizabeth Mathews, PhD, for valuable discussion.
REFERENCES
- Acsády L, Kamondi A, Sik A, Freund T, Buzsaki G. GABAergic cells are the major postsynaptic targets of mossy fibers in the rat hippocampus. J Neurosci 18: 3386–3403, 1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bukoski RD, Bátkai S, Járai Z, Wang Y, Offertaler L, Jackson WF, Kunos G. CB1 receptor antagonist SR141716A inhibits Ca2+-induced relaxation in CB1 receptor-deficient mice. Hypertension 39: 251–257, 2002. doi: 10.1161/hy0202.102702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen X, Ye H, Kuruvilla R, Ramanan N, Scangos KW, Zhang C, Johnson NM, England PM, Shokat KM, Ginty DD. A chemical-genetic approach to studying neurotrophin signaling. Neuron 46: 13–21, 2005. doi: 10.1016/j.neuron.2005.03.009. [DOI] [PubMed] [Google Scholar]
- Cole TB, Wenzel HJ, Kafer KE, Schwartzkroin PA, Palmiter RD. Elimination of zinc from synaptic vesicles in the intact mouse brain by disruption of the ZnT3 gene. Proc Natl Acad Sci USA 96: 1716–1721, 1999. doi: 10.1073/pnas.96.4.1716. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Danzer SC, He X, Loepke AW, McNamara JO. Structural plasticity of dentate granule cell mossy fibers during the development of limbic epilepsy. Hippocampus 20: 113–124, 2010. doi: 10.1002/hipo.20589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Danzer SC, McNamara JO. Localization of brain-derived neurotrophic factor to distinct terminals of mossy fiber axons implies regulation of both excitation and feedforward inhibition of CA3 pyramidal cells. J Neurosci 24: 11346–11355, 2004. doi: 10.1523/JNEUROSCI.3846-04.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dieni S, Matsumoto T, Dekkers M, Rauskolb S, Ionescu MS, Deogracias R, Gundelfinger ED, Kojima M, Nestel S, Frotscher M, Barde YA. BDNF and its pro-peptide are stored in presynaptic dense core vesicles in brain neurons. J Cell Biol 196: 775–788, 2012. doi: 10.1083/jcb.201201038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Glass DJ, Bowen DC, Stitt TN, Radziejewski C, Bruno J, Ryan TE, Gies DR, Shah S, Mattsson K, Burden SJ, DiStefano PS, Valenzuela DM, DeChiara TM, Yancopoulos GD. Agrin acts via a MuSK receptor complex. Cell 85: 513–523, 1996. doi: 10.1016/S0092-8674(00)81252-0. [DOI] [PubMed] [Google Scholar]
- Gu B, Huang YZ, He XP, Joshi RB, Jang W, McNamara JO. A peptide uncoupling BDNF receptor TrkB from phospholipase Cγ1 prevents epilepsy induced by status epilepticus. Neuron 88: 484–491, 2015. doi: 10.1016/j.neuron.2015.09.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guo N, Soden ME, Herber C, Kim MT, Besnard A, Lin P, Ma X, Cepko CL, Zweifel LS, Sahay A. Dentate granule cell recruitment of feedforward inhibition governs engram maintenance and remote memory generalization. Nat Med 24: 438–449, 2018. doi: 10.1038/nm.4491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hashimotodani Y, Ohno-Shosaku T, Kano M. Ca2+-assisted receptor-driven endocannabinoid release: mechanisms that associate presynaptic and postsynaptic activities. Curr Opin Neurobiol 17: 360–365, 2007. doi: 10.1016/j.conb.2007.03.012. [DOI] [PubMed] [Google Scholar]
- Haug FM. Electron microscopical localization of the zinc in hippocampal mossy fibre synapses by a modified sulfide silver procedure. Histochemie 8: 355–368, 1967. [DOI] [PubMed] [Google Scholar]
- He XP, Kotloski R, Nef S, Luikart BW, Parada LF, McNamara JO. Conditional deletion of TrkB but not BDNF prevents epileptogenesis in the kindling model. Neuron 43: 31–42, 2004. doi: 10.1016/j.neuron.2004.06.019. [DOI] [PubMed] [Google Scholar]
- Huang YZ, McNamara JO. Mutual regulation of Src family kinases and the neurotrophin receptor TrkB. J Biol Chem 285: 8207–8217, 2010. doi: 10.1074/jbc.M109.091041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang YZ, Pan E, Xiong ZQ, McNamara JO. Zinc-mediated transactivation of TrkB potentiates the hippocampal mossy fiber-CA3 pyramid synapse. Neuron 57: 546–558, 2008. doi: 10.1016/j.neuron.2007.11.026. [DOI] [PubMed] [Google Scholar]
- Isaacson JS, Scanziani M. How inhibition shapes cortical activity. Neuron 72: 231–243, 2011. doi: 10.1016/j.neuron.2011.09.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jin K, Xie L, Kim SH, Parmentier-Batteur S, Sun Y, Mao XO, Childs J, Greenberg DA. Defective adult neurogenesis in CB1 cannabinoid receptor knockout mice. Mol Pharmacol 66: 204–208, 2004. doi: 10.1124/mol.66.2.204. [DOI] [PubMed] [Google Scholar]
- Laezza F, Doherty JJ, Dingledine R. Long-term depression in hippocampal interneurons: joint requirement for pre- and postsynaptic events. Science 285: 1411–1414, 1999. doi: 10.1126/science.285.5432.1411. [DOI] [PubMed] [Google Scholar]
- Lei S, McBain CJ. Distinct NMDA receptors provide differential modes of transmission at mossy fiber-interneuron synapses. Neuron 33: 921–933, 2002. doi: 10.1016/S0896-6273(02)00608-6. [DOI] [PubMed] [Google Scholar]
- Lei S, McBain CJ. Two Loci of expression for long-term depression at hippocampal mossy fiber-interneuron synapses. J Neurosci 24: 2112–2121, 2004. doi: 10.1523/JNEUROSCI.4645-03.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maccafferi G, Tóth K, McBain CJ. Target-specific expression of presynaptic mossy fiber plasticity. Science 279: 1368–1370, 1998. doi: 10.1126/science.279.5355.1368. [DOI] [PubMed] [Google Scholar]
- Mechoulam R, Ben-Shabat S, Hanus L, Ligumsky M, Kaminski NE, Schatz AR, Gopher A, Almog S, Martin BR, Compton DR, Pertwee RG, Griffin G, Bayewitch M, Barg J, Vogel Z. Identification of an endogenous 2-monoglyceride, present in canine gut, that binds to cannabinoid receptors. Biochem Pharmacol 50: 83–90, 1995. doi: 10.1016/0006-2952(95)00109-D. [DOI] [PubMed] [Google Scholar]
- Middlemas DS, Meisenhelder J, Hunter T. Identification of TrkB autophosphorylation sites and evidence that phospholipase C-γ1 is a substrate of the TrkB receptor. J Biol Chem 269: 5458–5466, 1994. [PubMed] [Google Scholar]
- Navarrete M, Araque A. Endocannabinoids potentiate synaptic transmission through stimulation of astrocytes. Neuron 68: 113–126, 2010. doi: 10.1016/j.neuron.2010.08.043. [DOI] [PubMed] [Google Scholar]
- Pan E, Zhang XA, Huang Z, Krezel A, Zhao M, Tinberg CE, Lippard SJ, McNamara JO. Vesicular zinc promotes presynaptic and inhibits postsynaptic long-term potentiation of mossy fiber-CA3 synapse. Neuron 71: 1116–1126, 2011. doi: 10.1016/j.neuron.2011.07.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pelkey KA, Lavezzari G, Racca C, Roche KW, McBain CJ. mGluR7 is a metaplastic switch controlling bidirectional plasticity of feedforward inhibition. Neuron 46: 89–102, 2005. doi: 10.1016/j.neuron.2005.02.011. [DOI] [PubMed] [Google Scholar]
- Péterfi Z, Urbán GM, Papp OI, Németh B, Monyer H, Szabó G, Erdélyi F, Mackie K, Freund TF, Hájos N, Katona I. Endocannabinoid-mediated long-term depression of afferent excitatory synapses in hippocampal pyramidal cells and GABAergic interneurons. J Neurosci 32: 14448–14463, 2012. doi: 10.1523/JNEUROSCI.1676-12.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ruediger S, Vittori C, Bednarek E, Genoud C, Strata P, Sacchetti B, Caroni P. Learning-related feedforward inhibitory connectivity growth required for memory precision. Nature 473: 514–518, 2011. doi: 10.1038/nature09946. [DOI] [PubMed] [Google Scholar]
- Sugiura T, Kondo S, Sukagawa A, Nakane S, Shinoda A, Itoh K, Yamashita A, Waku K. 2-Arachidonoylglycerol: a possible endogenous cannabinoid receptor ligand in brain. Biochem Biophys Res Commun 215: 89–97, 1995. doi: 10.1006/bbrc.1995.2437. [DOI] [PubMed] [Google Scholar]
- Tóth K, McBain CJ. Afferent-specific innervation of two distinct AMPA receptor subtypes on single hippocampal interneurons. Nat Neurosci 1: 572–578, 1998. doi: 10.1038/2807. [DOI] [PubMed] [Google Scholar]
- Toth K, Suares G, Lawrence JJ, Philips-Tansey E, McBain CJ. Differential mechanisms of transmission at three types of mossy fiber synapse. J Neurosci 20: 8279–8289, 2000. doi: 10.1523/JNEUROSCI.20-22-08279.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Woo NH, Teng HK, Siao CJ, Chiaruttini C, Pang PT, Milner TA, Hempstead BL, Lu B. Activation of p75NTR by proBDNF facilitates hippocampal long-term depression. Nat Neurosci 8: 1069–1077, 2005. doi: 10.1038/nn1510. [DOI] [PubMed] [Google Scholar]
- Yang J, Harte-Hargrove LC, Siao CJ, Marinic T, Clarke R, Ma Q, Jing D, Lafrancois JJ, Bath KG, Mark W, Ballon D, Lee FS, Scharfman HE, Hempstead BL. proBDNF negatively regulates neuronal remodeling, synaptic transmission, and synaptic plasticity in hippocampus. Cell Reports 7: 796–806, 2014. doi: 10.1016/j.celrep.2014.03.040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao L, Yeh ML, Levine ES. Role for endogenous BDNF in endocannabinoid-mediated long-term depression at neocortical inhibitory synapses. eNeuro 2: 1–11, 2015. doi: 10.1523/ENEURO.0029-14.2015. [DOI] [PMC free article] [PubMed] [Google Scholar]