Abstract
Oxidized low-density lipoproteins (oxLDL) play a pivotal role in the etiopathogenesis of atherosclerosis through the activation of inflammatory signaling events eventually leading to endothelial dysfunction and senescence. In the present work, we investigated the effects of indicaxanthin, a bioavailable, redox-modulating phytochemical from Opuntia ficus indica fruits, with anti-inflammatory activity, against oxLDL-induced endothelial dysfunction. Human umbilical vein cord cells (HUVEC) were stimulated with human oxLDL, and the effects of indicaxanthin were evaluated in a range between 5 and 20 μM, consistent with its plasma level after a fruit meal (7 μM). Pretreatment with indicaxanthin significantly and concentration-dependently inhibited oxLDL-induced cytotoxicity; ICAM-1, VCAM-1, and ELAM-1 increase; and ABC-A1 decrease of both protein and mRNA levels. From a mechanistic perspective, we also provided evidence that the protective effects of indicaxanthin were redox-dependent and related to the pigment efficacy to inhibit NF-κB transcriptional activity. In conclusion, here we demonstrate indicaxanthin as a novel, dietary phytochemical, able to exert significant protective vascular effects in vitro, at nutritionally relevant concentrations.
1. Introduction
Atherosclerosis is a long, multifactorial, inflammatory process characterized by the accumulation of lipids in the wall of large- and medium-sized arteries [1, 2]. According to the oxidative theory of atherosclerosis, and its modifications over the last decade, hypercholesterolemia is one of the major risk factors of this condition as it triggers the accumulation of oxidized low-density lipoproteins (oxLDL) in the subintimal space [1, 2]. In areas at risk of atherosclerosis (e.g., at arterial bifurcations), the turbulent blood flow induces an endothelial activation, characterized by a local inflammatory response that generates an endocellular oxidative and nitrosative stress [3]. The increased levels of reactive oxygen and nitrogen species (RONS) activates selected redox-dependent transcription factors, such as NF-κB, that in turn induce an overexpression of endothelial adhesion molecules such as ICAM-1, VCAM-1, and ELAM-1 [1, 2]. This phenomenon increases the adhesion of leukocytes that eventually transmigrate in the subendothelial space where they are converted in macrophages. At the same time, the enhanced endothelial permeability favors the influx of LDL into the subendothelial intima where the inflammatory cells generate RONS inducing a progressive LDL oxidation. oxLDL, then, interact with endothelial cells (EC), disrupt the antioxidant defences of vascular endothelium, and boost leukocyte chemotaxis, reinforcing the whole inflammatory reaction that leads to a persistent EC dysfunction. As a consequence, a chronic systemic inflammatory response is established and eventually results in the proliferation of macrophagic foam cells and the formation of fatty streaks, the hallmark of the early atherosclerotic lesions [3].
In line with the key role of LDL in atherosclerosis, ATP-binding cassette A1 (ABC-A1), the cholesterol efflux system present in all tissues, endothelium included, has been demonstrated to offer antiatherogenic protection [4, 5]. Indeed, under physiological conditions, synthesized cholesterol can efflux from EC through ABC-A1 to maintain cholesterol homeostasis. Disturbed flow and oxLDL inhibit ABC-A1-mediated cholesterol efflux. As a result, cholesterol is removed through other pathways which may lead to lipid deposition in the subendothelial space [4–6].
Notwithstanding the solid basis of the oxidative theory of atherosclerosis, the vast majority of clinical trials and all meta-analyses surprisingly conclude that antioxidant vitamin supplementation has no beneficial effect on cardiovascular events. On the other hand, the association between “Mediterranean diet” and reduction of major chronic degenerative diseases, including the cardiovascular ones, appears robust and conclusive [1, 7, 8], suggesting the involvement of other redox-dependent molecular controls. In comparison to other fruits within the “Mediterranean diet” (e.g., oranges, lemons, and grapes), cactus pear (Opuntia ficus indica, L Mill) fruit is one that is less consumed and less studied, notwithstanding its abundance throughout selected areas of Italy, Spain, Greece, and North African countries.
Indicaxanthin (Figure 1), a betalain pigment from cactus pear fruit, has been the object of sound experimental work over the last years [9]. As with many phytochemicals, it is a redox-active compound and has been shown to act as antioxidant in a number of in vitro studies [9]. Interestingly, thanks to its charged portions, ionizable groups, and lipophilic moieties, it is amphiphilic [10] and has been demonstrated to interact with cell membranes [11]. This feature is critical to allow bioactive compounds to interact with cells and initiate signaling events. In this regard, indicaxanthin has been shown to modulate specific redox-dependent signaling pathways involved in macrophage activation and apoptosis and epithelial and endothelial dysfunction in vitro [9, 12, 13]. Remarkably, and in contrast with the majority of dietary phytochemicals, indicaxanthin is highly bioavailable [14]. The molecule has been shown to cross the unaltered intestinal epithelial cell in vitro being absorbed through paracellular junctions [11]. In line with that, indicaxanthin has been found in human plasma at a 7 μM peak concentration 3 h after the ingestion of four cactus pear fruits containing 28 mg of the pigment [14]. Moreover, its amphiphilicity allows it to cross the blood-brain barrier, located within the CNS, and modulate its bioelectric activity [15]. Finally, thanks to its bioavailability and redox-modulating properties, indicaxanthin exerts significant pharmacological effects in vivo. Indeed, oral administration of the phytochemical at nutritionally relevant doses (2 μmol/kg) generates, in rats, a plasma peak concentration of 0.2 μM able to exert strong anti-inflammatory effects in a model of acute inflammation [16].
Figure 1.
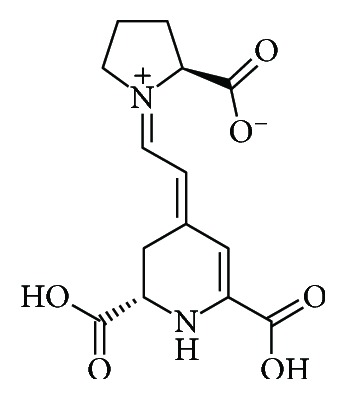
Chemical structure of indicaxanthin.
In the light of the strong interconnections between inflammation, atherosclerosis, and diet, and the anti-inflammatory and redox-modulating properties of indicaxanthin, the aim of this work was to evaluate the effects of the phytochemical, at nutritionally relevant concentrations, in an in vitro model of innate immunity activation represented by oxLDL-induced endothelial dysfunction. To this end, we investigated whether indicaxanthin prevented the oxLDL-mediated adhesion molecule overexpression and ABC-A1 downregulation, in human cultured EC. Mechanistic details of the redox-dependent NF-κB transcriptional activity have also been investigated.
2. Materials and Methods
2.1. Reagents
All reagents, unless otherwise stated, were from Sigma-Aldrich (Milan, Italy) and of the highest available purity grade.
2.2. Extraction and Purification of Indicaxanthin from Cactus Pear Fruits
Indicaxanthin was isolated from cactus pear (Opuntia ficus indica) fruits (yellow cultivar) as previously described [16].
2.3. Isolation and Oxidation of Human LDL
Blood samples from healthy subjects with informed consent were obtained after an overnight fasting. LDL were isolated from plasma as previously described [17] and diluted to 100 μg protein/ml in PBS. Protein concentration of the particle was evaluated by Bradford assay as reported elsewhere [17].
Aliquots of native LDL (nLDL) were then subjected to lipid oxidation by treatment with 5 μM CuSO4 at 37°C for 24 h, as previously described [14].
The oxidation state of LDL, in terms of conjugated dienes (CD) hydroperoxides and thiobarbituric acid-reactive substances (TBARS), was evaluated as previously reported [14].
oxLDL were then concentrated with centrifugal filter devices according to the manufacturer's instructions (Millipore, Milan, Italy) to a value of 100 μg protein/ml. All samples (nLDL and oxLDL) were filter-sterilized (0.2 μm Millipore filters, Milan, Italy), aliquoted, and stored at -80°C up to one month.
2.4. Cell Culture
HUVEC (human umbilical vein endothelial cells) were purchased from Lonza (Milan, Italy), grown in endothelial growth medium (Lonza) at 37°C in a humidified incubator under 5% CO2, subcultured by trypsinization, and used up to passage 4.
2.5. HUVEC Treatment
In order to perform the experiments, cells were seeded in 6-well plates at a density of 2 × 104 cells/well. Upon 80% confluence, HUVEC were incubated, after overnight starving, either in the absence (control cells) or in the presence of 100 μg oxLDL/ml for 16 h in a serum-free endothelial basal medium (Lonza, Milan, Italy). When necessary, HUVEC were pretreated for 1 h with indicaxanthin and then stimulated with oxLDL as described above.
2.6. Cell Viability Test
Cytotoxicity of oxLDL was assessed through MTT [3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl-2H-tetrazolium bromide] conversion assay according to the manufacturer's instructions (Invitrogen, Milan, Italy). Cell viability was expressed as percentage of the absorbance value measured in control HUVEC.
2.7. Flow Cytometry Analysis
At the end of the incubation time, HUVEC were washed with PBS, harvested with cell dissociation medium, and diluted in a washing buffer containing PBS, 0.1% bovine serum albumin, and 1 mM CaCl2. Aliquots (0.1 to 0.5 × 106 cells in 20 μl) were seeded in a 96-well plate and incubated at 4°C with 2 μg/well of a mouse anti-human ICAM-1 or VCAM-1 or ELAM-1 or ABC-A1 monoclonal antibody FITC-conjugated (Invitrogen, Milan, Italy). After 1 h at 4°C, cells were washed and analysed for fluorescence on a flow cytometer (Epics II, Beckman Coulter, US). At least 20000 events were analysed for each sample. Data are expressed as median fluorescence intensity (MFI) units, measured in the FL1 (green) channel with a 488 nm (excitation) and 530 nm (emission) filter set.
RONS concentration was evaluated by staining cells with 2′,7′–dichlorofluorescin diacetate dye (DCFDA) as reported elsewhere [18].
2.8. Quantitative Real-Time Reverse-Transcription Polymerase Chain Reaction
Total RNA was isolated by employing an RNeasy mini kit in accordance with the manufacturer's instructions (QIAGEN, Milan, Italy). RNA (5–10 μg) was then reverse-transcribed to cDNA using Superscript III Reverse Transcriptase following the manufacturer's protocol (Invitrogen, Milan, Italy) and stored at -20°C until tested. Real-time PCR for ICAM-1, VCAM-1, ELAM-1, or ABC-A1 and glucose-6-phosphate dehydrogenase (G6PDH) was carried out with an RT Real-Time™ SYBR Green PCR Master Mix and the RT2 PCR Primer Set in accordance with the manufacturer's instructions (SuperArray, Milan, Italy) using an ABI PRISM 7700 Sequence Detection System (Applied Biosystems, Warrington, UK). The following primers were used in the present study: human VCAM-1: (forward) 5′-CTTAAAATGCCTGGGAAGATGGT-3′, (reverse) 5′-GTCAATGAGACGGAGTCACCAAT-3′; human ICAM-1: (forward) 5′-GGCTGGAGCTGTTTGAGAAC-3′, (reverse) 5′-CTGACAAGTTGTGGGGGAGT-3′; human ELAM-1: (forward) 5′-GCCTGCAATGTGGTTGAGTG-3′, (reverse) 5′-ACGAACCCATTGGCTGGATT-3′; human ABC-A1: (forward) 5′-TGTCCAGTCCAGTAATGGTTCTGT-3′, (reverse) 5′-CGAGATATGGTCCGGATTGC-3′); and human GAPDH: (forward) 5′-CCACATCGCTCAGACACCAT-3′, (reverse) 5′-CCAGGCGCCCAATACG-3. Thermal cycling conditions included a prerun of 2 min at 50°C and 15 min at 95°C followed by 40 cycles at 95°C for 30s, 55°C for 30s, and 72°C for 30s. RT2-PCR data were quantified as cycle of threshold (Ct) values, and the relative expression of each gene was normalized to housekeeping gene GAPDH.
2.9. Reporter Gene Assay
NF-κB activity was examined by transfecting HUVEC with pNFκB-Luc luciferase construct (Stratagene, CA, USA) as reported elsewhere [19]. Transfected HUVEC were then cocultured with 100 μM oxLDL in the absence or in the presence of indicaxanthin as detailed above. After 6 h, cells were washed with PBS and lysed for 5 min at 4°C using a lysis buffer according to the manufacturer's instructions (Promega, WI, USA). Luciferase activity is expressed as relative luminescence units (RLU) × 103 following reading in a TD/2020 luminometer (Turner Biosystems, CA, USA).
2.10. Statistical Analysis
Data from all experiments are reported as mean ± SD. Data were analysed and presented using GraphPad Prism software (GraphPad). Comparisons between two values were made using Student's t-test, and P < 0.001 were designated with triple asterisks. Multiple comparisons were performed by one-way analysis of variance (ANOVA) followed by Tukey's correction. Significance was accepted when the null hypothesis was rejected at the P < 0.001 level.
3. Results
3.1. Oxidative State Assessment of oxLDL
The oxidative state of oxLDL was evaluated by assaying CD hydroperoxides and TBARS levels. When compared to nLDL, treatment with 5 μM CuSO4 for 24 h determined a significant increase (P < 0.001) of both CD and TBARS in the particle (Figure 2).
Figure 2.
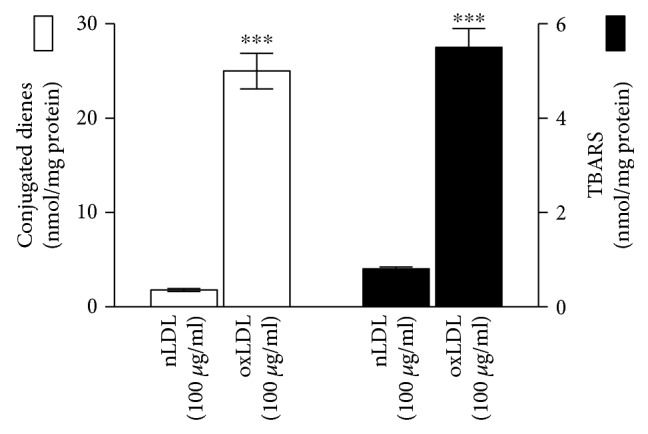
Amount of CD hydroperoxides and TBARS in the oxLDL employed in the study in comparison with untreated ones (nLDL). Values are the mean ± SD of three separate experiments carried out in duplicate; ∗∗∗P < 0.001.
3.2. Effects of Indicaxanthin on the Viability of oxLDL-Treated EC
Incubation of HUVEC with 100 μg oxLDL/ml for 16 h determined a significant decrease (P < 0.001) of cell viability as compared to control EC (Figure 3). On the other hand, pretreatment of HUVEC with indicaxanthin at 5 μM did not improve cell viability, while at 15 μM it caused a significant (P < 0.001) inhibition of cell death that was completely prevented with 20 μM indicaxanthin (Figure 3).
Figure 3.
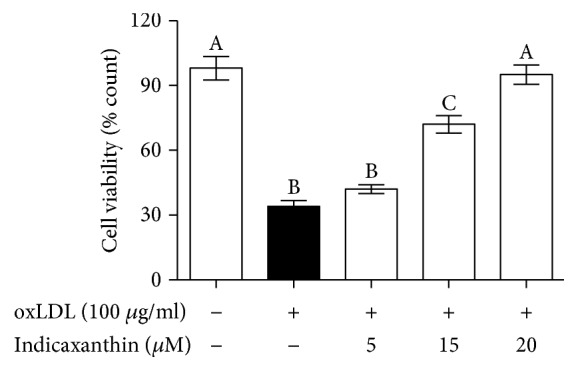
Protective effects of indicaxanthin against oxLDL-induced cytotoxicity in EC. Values are the mean ± SD of three separate experiments carried out in triplicate. Labeled means without a common letter differ with P < 0.001.
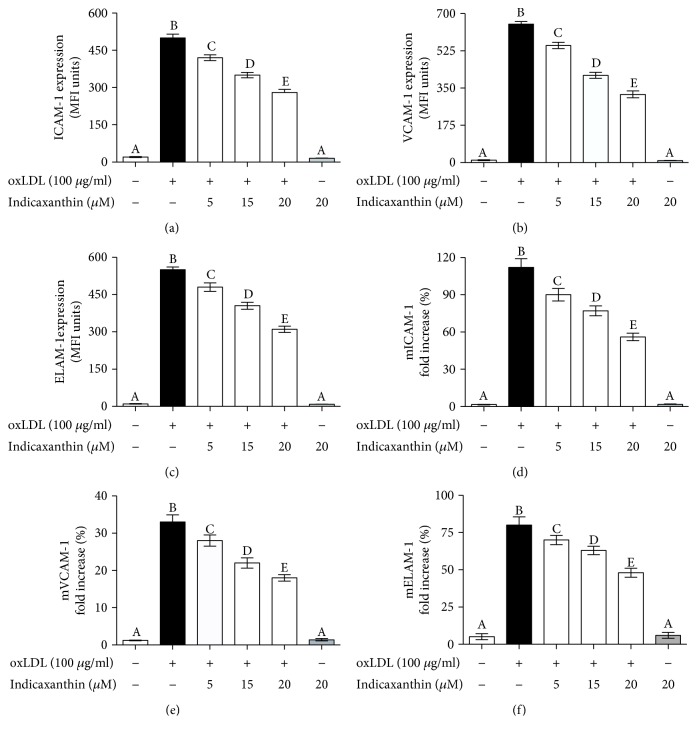
3.3. Effects of Indicaxanthin on oxLDL-Induced Protein and mRNA Upregulation of Adhesion Molecules in EC
Incubation of HUVEC with 100 μg oxLDL/ml for 16 h induced a significant (P < 0.001) increase of ICAM-1, VCAM-1, and ELAM-1 regarding both protein and mRNA levels (Figures 4(a)–4(f), respectively) in comparison with control cells. Noteworthy, pretreatment of EC with indicaxanthin in a concentration range between 5 and 20 μM caused a significant (P < 0.001) concentration-dependent decrease of all the above-cited adhesion molecules regarding both protein and mRNA levels (Figures 4(a)–4(f), respectively). Importantly, incubation of EC with 20 μM indicaxanthin in the absence of oxLDL did not modify the adhesion molecule, regarding both protein and mRNA levels, in comparison with control HUVEC.
Figure 4.
ICAM-1, VCAM-1, and ELAM-1 protein (a–c) and mRNA (d–f) levels of HUVEC stimulated with oxLDL, either in the absence or in the presence of indicaxanthin at different concentrations for 16 h. Values are the mean ± SEM of three separate experiments carried out in triplicate. Labeled means without a common letter differ; P < 0.001.
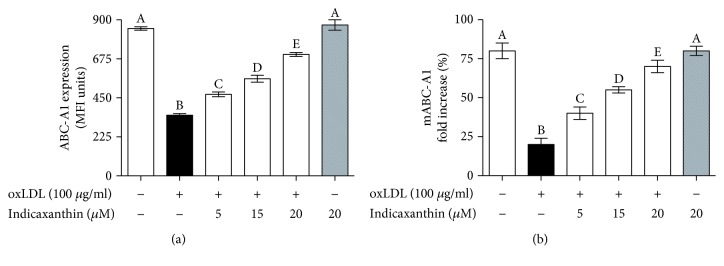
3.4. Effects of Indicaxanthin on oxLDL-Induced Downregulation of ABC-A1 in EC
Stimulation of EC with 100 μg oxLDL/ml induced a significant (P < 0.001) decrease of ABC-A1 regarding both protein and mRNA levels, in comparison with control HUVEC (Figures 5(a) and 5(b), respectively). Noteworthy, pretreatment of EC with indicaxanthin in a range between 5 and 20 μM caused a significant (P < 0.001) concentration-dependent increase of ABC-A1 expression, regarding both protein and mRNA levels (Figure 5). On the other hand, incubation of EC with 20 μM indicaxanthin in the absence of oxLDL did not modify ABC-A1 at both protein and mRNA levels, in comparison with control HUVEC.
Figure 5.
ABC-A1 protein (a) and mRNA (b) levels in HUVEC stimulated with oxLDL, either in the absence or in the presence of indicaxanthin at different concentrations for 16 h. Values are the mean ± SEM of three separate experiments carried out in triplicate. Labeled means without a common letter differ; P < 0.001.
3.5. Effects of Indicaxanthin on oxLDL-Induced RONS Production
In comparison with control HUVEC, stimulation of EC with 100 μg oxLDL/ml induced a significant increase of RONS levels (P < 0.001) (Figure 6). On the other hand, pretreatment of EC with indicaxanthin in a concentration range between 5 and 20 μM caused a significant (P < 0.001) concentration-dependent decrease of endocellular RONS production when compared to control EC (Figure 6). Conversely, incubation of EC with 20 μM indicaxanthin in the absence of oxLDL did not modify RONS levels, in comparison with control HUVEC.
Figure 6.
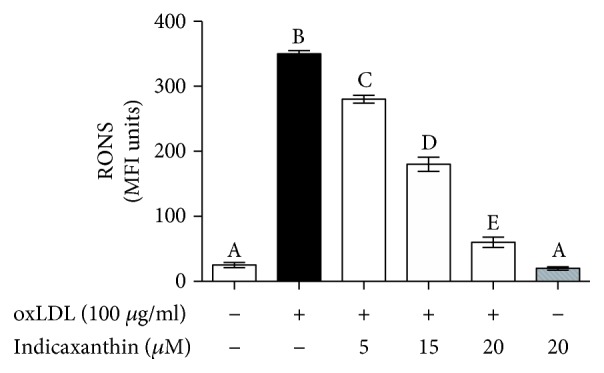
RONS levels in HUVEC stimulated with oxLDL, either in the absence or in the presence of indicaxanthin at different concentrations for 16 h. Values are the mean ± SEM of three separate experiments carried out in triplicate. Labeled means without a common letter differ; P < 0.001.
3.6. Effects of Indicaxanthin on oxLDL-Induced NF-κB Transcriptional Activity
Stimulation of EC with 100 μg oxLDL/ml induced a significant (P < 0.001) increase of NF-κB transcriptional activity, in comparison with control HUVEC (Figure 7). Noteworthy, pretreatment of EC with indicaxanthin in a concentration range between 5 and 20 μM caused a significant (P < 0.001) concentration-dependent decrease of the transcriptional activity (Figure 7). On the other hand, incubation of EC with 20 μM indicaxanthin in the absence of oxLDL did not modify the transcriptional activity of NF-κB, in comparison with control HUVEC.
Figure 7.
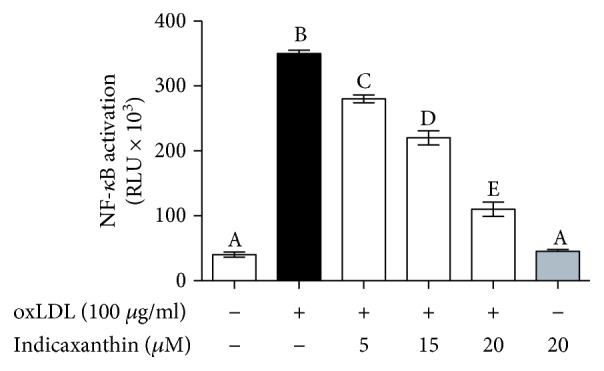
NF-κB transcriptional activity in HUVEC stimulated with oxLDL, either in the absence or in the presence of indicaxanthin at different concentrations for 16 h. Values are the mean ± SEM of three separate experiments carried out in triplicate. Labeled means without a common letter differ; P < 0.001.
4. Discussion
The present investigation falls within the intense research on the interplay between diet and immune function. The identity, the number, and the mechanisms of action of the protective agents we daily assume through our diet remain largely unknown and require further research. However, before defining appropriate clinical trials, it is crucial to evaluate, through suitable in vitro assays, the health-promoting effects of plant-derived compounds in order to identify the most promising ones [1–3].
In line with this perspective, we investigated here the protective effects exerted by indicaxanthin from cactus pear fruits, at nutritionally relevant concentrations, in an in vitro model of innate immunity activation represented by oxLDL-induced endothelial dysfunction. Our findings demonstrate that indicaxanthin protects EC from oxLDL-induced damage in vitro, as evaluated by an increase of the viability of EC pretreated with the phytochemical in the range between 15 and 20 μM. ICAM-1, VCAM-1, and ELAM-1 overexpression is a key event in the onset and the development of EC dysfunction. Relevantly, it not only increased indicaxanthin cell viability but also preserved cellular functions, counteracting the oxLDL-induced adhesion molecule overexpression, from 5 to 20 μM. From a mechanistic perspective, these effects were mediated by a modulation of gene expression that led to a downregulation of ICAM-1, VCAM-1, and ELAM-1 mRNA levels.
As stated above, many phytochemicals have shown immunomodulatory effects, in vitro. However, a major limitation for the employment of these compounds is their low bioavailability. On the contrary, present results appear of interest as indicaxanthin is effective at nutritionally relevant concentrations, achievable in humans after the ingestion of the fruit [14].
It is well established that endothelial adhesion molecule expression is under control of several redox-regulated transcription factors, including the master minder NF-κB [20]. Moreover, oxLDL have been demonstrated to activate various transcription factors, NF-κB included. In line with this, we demonstrated here an inhibition by indicaxanthin of the transcriptional activity of NF-κB. This result appears consistent with our previously reported findings on the NF-κB-dependent immune-modulatory and anti-inflammatory effects of indicaxanthin both in vivo and in vitro. Relevantly, the inhibition of the NF-κB transcriptional activity appears to be strictly consistent with the ability of indicaxanthin to inhibit oxLDL-induced endocellular oxidative and nitrosative stress. Along these lines, we therefore propone that the phytochemical counteracted the oxLDL-induced adhesion molecule overexpression by inhibiting NF-κB transcriptional activity, through a redox-dependent mechanism.
Along with ICAM-1, VCAM-1, and ELAM-1 overexpression, disruption of the reverse cholesterol transfer across the endothelium, via ABC-A1 downregulation, represents another key aspect of EC dysfunction and cholesterol deposition within the arterial wall. It is well known that oxLDL decreases ABC-A1 levels in EC via inhibiting liver receptor X (LXR) [4]. We found here that indicaxanthin counteracted the oxLDL-induced ABC-A1 downregulation. The observed inhibitory effects of indicaxanthin of the transcriptional activity of NF-κB can also be connected with its ability to counteract the oxLDL-induced ABC-A1 downregulation. Indeed, several lines of evidence show that ABC-A1 can be upregulated, rather than through the activation of the LXR pathway, via the inhibition of the redox-sensitive ERK/NF-κB pathway [21–25].
Finally, notwithstanding that indicaxanthin at 5 μM does not improve EC viability, it does, however, significantly reduce adhesion molecule overexpression, NF-κB transcriptional activity, and RONS production and increases ABCA-1 expression.
5. Conclusions
Evaluating the activities of healthy, plant-derived compounds to select the most promising ones through suitable in vitro assays is imperative before defining appropriate clinical trials. We demonstrated here that indicaxanthin, a bioavailable, redox-modulating phytochemical with anti-inflammatory activity, is able to exert remarkable protective effects in an in vitro model of vascular inflammation, at nutritionally relevant concentrations. In light of its ability to modulate specific endothelial genes involved in leukocyte adhesion and cholesterol transport, through a redox-dependent, NF-κB-mediated mechanism, indicaxanthin might be further investigated in vivo as a potential endothelial-protective agent of dietary origin.
Acknowledgments
This project was supported by a 2014–2020 PON Ricerca e Innovazione grant from the Italian Ministry of Education, University and Research, entitled “PROGEMA - Processi Green per l'Estrazione di Principi Attivi e la Depurazione di Matrici di Scarto e Non” (ARS01_00432) to P.C.
Data Availability
The data used to support the findings of this study are available from the corresponding author upon request.
Conflicts of Interest
The authors declare that there is no conflict of interest regarding the publication of this paper.
References
- 1.Salvayre R., Negre-Salvayre A., Camare C. Oxidative theory of atherosclerosis and antioxidants. Biochimie. 2016;125:281–296. doi: 10.1016/j.biochi.2015.12.014. [DOI] [PubMed] [Google Scholar]
- 2.Suciu C. F., Prete M., Ruscitti P., Favoino E., Giacomelli R., Perosa F. Oxidized low density lipoproteins: the bridge between atherosclerosis and autoimmunity. Possible implications in accelerated atherosclerosis and for immune intervention in autoimmune rheumatic disorders. Autoimmunity Reviews. 2018;17(4):366–375. doi: 10.1016/j.autrev.2017.11.028. [DOI] [PubMed] [Google Scholar]
- 3.Negre-Salvayre A., Auge N., Camare C., Bacchetti T., Ferretti G., Salvayre R. Dual signaling evoked by oxidized LDLs in vascular cells. Free Radical Biology & Medicine. 2017;106:118–133. doi: 10.1016/j.freeradbiomed.2017.02.006. [DOI] [PubMed] [Google Scholar]
- 4.Zhu Y., Liao H., Xie X., et al. Oxidized LDL downregulates ATP-binding cassette transporter-1 in human vascular endothelial cells via inhibiting liver X receptor (LXR) Cardiovascular Research. 2005;68(3):425–432. doi: 10.1016/j.cardiores.2005.07.003. [DOI] [PubMed] [Google Scholar]
- 5.Westerterp M., Tsuchiya K., Tattersall I. W., et al. Deficiency of ATP-binding cassette transporters A1 and G1 in endothelial cells accelerates atherosclerosis in mice. Arteriosclerosis, Thrombosis, and Vascular Biology. 2016;36(7):1328–1337. doi: 10.1161/ATVBAHA.115.306670. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Van Eck M. ATP-binding cassette transporter A1: key player in cardiovascular and metabolic disease at local and systemic level. Current Opinion in Lipidology. 2014;25(4):297–303. doi: 10.1097/MOL.0000000000000088. [DOI] [PubMed] [Google Scholar]
- 7.Mulvihill E. E., Burke A. C., Huff M. W. Citrus flavonoids as regulators of lipoprotein metabolism and atherosclerosis. Annual Review of Nutrition. 2016;36(1):275–299. doi: 10.1146/annurev-nutr-071715-050718. [DOI] [PubMed] [Google Scholar]
- 8.Cirillo P., Conte S., Cimmino G., et al. Nobiletin inhibits oxidized-LDL mediated expression of tissue factor in human endothelial cells through inhibition of NF-κB. Biochemical Pharmacology. 2017;128:26–33. doi: 10.1016/j.bcp.2016.12.016. [DOI] [PubMed] [Google Scholar]
- 9.Rahimi P., Abedimanesh S., Mesbah-Namin S. A., Ostadrahimi A. Betalains, the nature-inspired pigments, in health and diseases. Critical Reviews in Food Science and Nutrition. 2018;30:1–30. doi: 10.1080/10408398.2018.1479830. [DOI] [PubMed] [Google Scholar]
- 10.Turco Liveri M. L., Sciascia L., Allegra M., Tesoriere L., Livrea M. A. Partition of indicaxanthin in membrane biomimetic systems. A kinetic and modeling approach. Journal of Agricultural and Food Chemistry. 2009;57(22):10959–10963. doi: 10.1021/jf902266m. [DOI] [PubMed] [Google Scholar]
- 11.Tesoriere L., Gentile C., Angileri F., et al. Trans-epithelial transport of the betalain pigments indicaxanthin and betanin across Caco-2 cell monolayers and influence of food matrix. European Journal of Nutrition. 2013;52(3):1077–1087. doi: 10.1007/s00394-012-0414-5. [DOI] [PubMed] [Google Scholar]
- 12.Allegra M., D’Acquisto F., Tesoriere L., Attanzio A., Livrea M. A. Pro-oxidant activity of indicaxanthin from Opuntia ficus indica modulates arachidonate metabolism and prostaglandin synthesis through lipid peroxide production in LPS-stimulated RAW 264.7 macrophages. Redox Biology. 2014;2:892–900. doi: 10.1016/j.redox.2014.07.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Tesoriere L., Attanzio A., Allegra M., Gentile C., Livrea M. A. Indicaxanthin inhibits NADPH oxidase (NOX)-1 activation and NF-κB-dependent release of inflammatory mediators and prevents the increase of epithelial permeability in IL-1β-exposed Caco-2 cells. British Journal of Nutrition. 2014;111(3):415–423. doi: 10.1017/S0007114513002663. [DOI] [PubMed] [Google Scholar]
- 14.Tesoriere L., Allegra M., Butera D., Livrea M. A. Absorption, excretion, and distribution of dietary antioxidant betalains in LDLs: potential health effects of betalains in humans. The American Journal of Clinical Nutrition. 2004;80(4):941–945. doi: 10.1093/ajcn/80.4.941. [DOI] [PubMed] [Google Scholar]
- 15.Allegra M., Carletti F., Gambino G., et al. Indicaxanthin from Opuntia ficus-indica crosses the blood-brain barrier and modulates neuronal bioelectric activity in rat hippocampus at dietary-consistent amounts. Journal of Agricultural and Food Chemistry. 2015;63(33):7353–7360. doi: 10.1021/acs.jafc.5b02612. [DOI] [PubMed] [Google Scholar]
- 16.Allegra M., Ianaro A., Tersigni M., Panza E., Tesoriere L., Livrea M. A. Indicaxanthin from cactus pear fruit exerts anti-inflammatory effects in carrageenin-induced rat pleurisy. The Journal of Nutrition. 2014;144(2):185–192. doi: 10.3945/jn.113.183657. [DOI] [PubMed] [Google Scholar]
- 17.Bradford M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Analytical Biochemistry. 1976;72(1-2):248–254. doi: 10.1016/0003-2697(76)90527-3. [DOI] [PubMed] [Google Scholar]
- 18.Steffen Y., Jung T., Klotz L. O., Schewe T., Grune T., Sies H. Protein modification elicited by oxidized low-density lipoprotein (LDL) in endothelial cells: protection by (−)-epicatechin. Free Radical Biology & Medicine. 2007;42(7):955–970. doi: 10.1016/j.freeradbiomed.2006.12.024. [DOI] [PubMed] [Google Scholar]
- 19.Su Q., Sun Y., Ye Z., Yang H., Kong B., Li L. Pinocembrin protects endothelial cells from oxidized LDL-induced injury. Cytokine. 2018;111:475–480. doi: 10.1016/j.cyto.2018.05.033. [DOI] [PubMed] [Google Scholar]
- 20.Allegra M., D’Acquisto F., Tesoriere L., Livrea M. A., Perretti M. Cross-talk between minimally primed HL-60 cells and resting HUVEC reveals a crucial role for adhesion over extracellularly released oxidants. Biochemical Pharmacology. 2011;81(3):396–401. doi: 10.1016/j.bcp.2010.10.018. [DOI] [PubMed] [Google Scholar]
- 21.Wang Y., Wu J. F., Tang Y. Y., et al. Urotensin II increases foam cell formation by repressing ABCA1 expression through the ERK/NF-κB pathway in THP-1 macrophages. Biochemical and Biophysical Research Communications. 2014;452(4):998–1003. doi: 10.1016/j.bbrc.2014.09.030. [DOI] [PubMed] [Google Scholar]
- 22.Zhao G. J., Mo Z. C., Tang S. L., et al. Chlamydia pneumoniae negatively regulates ABCA1 expression via TLR2-nuclear factor-kappa B and miR-33 pathways in THP-1 macrophage-derived foam cells. Atherosclerosis. 2014;235(2):519–525. doi: 10.1016/j.atherosclerosis.2014.05.943. [DOI] [PubMed] [Google Scholar]
- 23.Jiang J., Mo Z. C., Yin K., et al. Epigallocatechin-3-gallate prevents TNF-α-induced NF-κB activation thereby upregulating ABCA1 via the Nrf2/Keap1 pathway in macrophage foam cells. International Journal of Molecular Medicine. 2012;29(5):946–956. doi: 10.3892/ijmm.2012.924. [DOI] [PubMed] [Google Scholar]
- 24.Gerbod-Giannone M. C., Li Y., Holleboom A., et al. TNFα induces ABCA1 through NF-κB in macrophages and in phagocytes ingesting apoptotic cells. Proceedings of the National Academy of Sciences of the United States of America. 2006;103(9):3112–3117. doi: 10.1073/pnas.0510345103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Chen M., Li W., Wang N., Zhu Y., Wang X. ROS and NF-κB but not LXR mediate IL-1β signaling for the downregulation of ATP-binding cassette transporter A1. American Journal of Physiology Cell Physiology. 2007;292(4):C1493–C1501. doi: 10.1152/ajpcell.00016.2006. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The data used to support the findings of this study are available from the corresponding author upon request.