Abstract
Cordyceps militaris (C. militaris) was utilized to ferment cornmeal by solid state fermentation. The main objective of this study was to investigate effect of fermentation on the dynamic state of water and microstructure distribution of water within cornmeal with Low-field nuclear magnetic resonance, as well as the effect on composition and microstructure properties. The spin–spin relaxation time (T2) showed significant changes in solid-state fermented cornmeal. Principal component analysis further revealed that the variations within different fermentation stage could be discriminated by the T2 parameters. Bulk water (T22) was the main form of water present and lost in substrates. The weights of different indicators, as assessed by multiple regression analysis, demonstrated that there was a strong correlation between starch and T2 relaxation. Scanning electron microscopy demonstrated that fermentation can cause the appearance of micropores. The longer relaxation time of T22 during logarithmic period can be interpreted as a loosening of the structure at the starch hydrolysis, introducing more water into the structure. Thus, the differences in composition and structure of the substrate at different fermentation time produce different T2 values.
Electronic supplementary material
The online version of this article (10.1007/s13197-019-03569-0) contains supplementary material, which is available to authorized users.
Keywords: Solid-state fermentation, Cornmeal, LF-NMR, Cordyceps militaris
Introduction
Cordyceps militaris (C. militaris) is traditional medicinal and edible mushrooms in China, which is widely used in food processing. In order to meet the needs of commercial production, developments cultivation techniques have been conducted, such as liquid fermentation for mycelium and SSF for the production of fruiting bodies, which can be widely used as ingredients in functional foods. Xiao et al. (2015) applied solid-state fermentation by C. militaris to improve the functional and nutritional properties of chickpea flour. Both the chickpea and C. militaris have functional components, SSF of legumes and cereals is a new area of research. However, despite there the presence of many studies about the SSF of legumes and cereals (Bartkiene et al. 2015; Kang et al. 2017), the focus of most studies is on the documentation of health and safety effects. There are few reports on the online monitoring of SSF in industry. Furthermore, some of the traditional methods used are cumbersome and require long time. These factors all restrict the development of solid-state fermentation by C. militaris.
Because of the popularity of SSF in the food processing, such as the preparation of enzymes, production of metabolism, and bioconversion of agricultural by-products, it was recognized that a reliable, in-depth and effective method is required to describe and manage the processing (Jiang et al. 2012). However, because of the heterogeneity of fermented matrix, some measurements are difficult to conduct. Furthermore, processes of SSF in actual manufacture vary obviously from batch to batch, which can be ascribed to compositional variations that differ from the standardized batch (Cozzolino et al. 2010). Therefore, accurate and real-time process quality control technologies are needed in SSF process. Since the discovery of NMR, this technology has been widely applied in the field of food science (Kirtil and Oztop 2016). Low-field nuclear magnetic resonance (LF-NMR) technologies have been widely applied in quality testing and in the control of food, e.g., the measurement of the water holding capacity of fresh meat (Pearce et al. 2011); the measurement of the water content in glutinous rice (Li et al. 2015) and soybean antioxidant peptide power (Lin et al. 2016). Diverse food systems including starch are also widely studied using LF-NMR technology, and the LF-NMR can provide structure and compositional information of starch (Tang et al. 2001). The structure of starch has been widely studied with microscopic technologies, such as Scanning electron microscope (SEM) and Differential scanning calorimetry (DSC). The microscopic technologies do not show the microscopic distribution of water within the granule and dynamic changes of water. These characters are recognized to have important influence on fermentation. Compared with these analytical techniques, LF-NMR can provide novel insights into the matrix microstructure and dynamic changes of water. NMR is complementary to other analytical methods for the description of complex systems (Zhu 2017). In particular, LF-NMR and Magnetic Resonance Imaging (MRI) are highly sensitive to the changes in the moisture distribution and dynamics inside substrates, which makes them to be promising for the analyzing of SSF (Wu et al. 2016). However, the correlations between substrates and water distribution are currently not defined in heterogenous systems during SSF.
Recent developments of LF-NMR have led us to consider the application of LF-NMR to non-invasively monitor the SSF of cornmeal by C. militaris. In this study, the distribution and variation of water molecules in the fermented samples were investigated by LF-NMR, the comparison of these changes with composition and structure of substrate, and the possibility of using these water mobility changes to probe into the relationship with the substrate characteristic.
Materials and methods
Materials
Corn from Jilin Province was obtained from a local market (Chuncheng Market, Changchun, China) (approximately 63% total starch, 9% protein, 4.5% fat, and 14% water). Cordyceps militaris was preserved in the National Engineering Laboratory for Wheat and Corn Deep Processing (HMJAU 202, Herbarium Mycology of Jilin Agriculture University, Chang Chun, China). The Total Starch Assay Kit was obtained from Megazyme (K-TSTA-100A, Megazyme, Ireland). 3, 5-2 Nitro Salicylic Acid (609-99-4, Aladdin Chemistry Co. Ltd., Shanghai, China). Trichloroacetic acid solution (TCA) (76-03-9, Aladdin Chemistry Co. Ltd., Shanghai, China). HCL and petroleum ether were obtained from Dingguo biotechnology co. LTD (Beijing, China). All the chemicals used in this work were of analytical grade.
Methods
Preparation of seeding culture for cornmeal fermentation
The preparation of seeding culture was the same as described previously (Yang et al. 2018).
Preparation of solid-state fermented cornmeal
The corn was rinsed several times to remove soluble impurities, and then was dried in the oven (ALS-225, HD-Standard Oven Corporation, Shanghai, China) at 35 °C for 48 h. The whole kernels were milled to pass through a 40-US mesh screen. Then, the kernels were dried in an oven at 45 °C for approximately 24 h. The cornmeal was mixed with distilled water at a ratio of 1:1.2 (w/w; 30 g of dry cornmeal to 36 g of water), and was further sterilized at 121 °C for 15 min in an autoclave (YXQ-LS-SII, Boxun Corporation. Shanghai, China). Then, the cooled samples were inoculated with C. militaris seeding, inoculating at a ratio of 10:1, and a cooled sample was not inoculated with seeding to act as a control. During SSF, the temperature of the incubator (HWS-temperature-and-humidity constant box, Ningbo Jiangnan Instrument Factory, China) was maintained at 24 °C and was controlled by mechanical convection aeration based on a temperature sensor. The humidity of the incubator was kept at 55%, which was controlled by a steam humidifier based on humidity sensor. The samples were analysed based on five replicates at different fermentation time (0 to 10 days).
Moisture content (MC)
The ratio of weight loss before and after drying was defined as MC. Fermented samples were placed into an oven. The samples were dried at 105 °C until they were a constant weight (Monsoor et al. 2004). The experiments were conducted five times.
Biomass content
The biomass content was with reference to Wei et al. (2006). The establishment of a nucleic acid standard curve was based on the measurement of nucleic acid content from pure mycelium. Pure mycelium was obtained by filtering the fermented seeding. The fermented seedings were washed with deionized water several times and then filtered to obtain the pure mycelium, and then were dried in the oven to a constant weight at 60 °C. For the extraction of nucleic acid from pure mycelium, a known weight of mycelium was put in 25 ml 5% TCA, extracted for 25 min at 80 °C in a water bath, and then was centrifuged at 7000×g for 10 min after having been diluted 5 times (X-30R, Allegra, Denmark). The absorbance of the samples was determined at 260 nm, and 5% TCA was used as a blank control. The fermented samples were treated the same as those of the extraction of nucleic acid from pure mycelium. The unfermented sample was used as control. Linear equations: y = 9.553x + 0.10, R2 = 0.99. The experiments were conducted five times.
Total starch
The total starch content was determined by the AOAC Method 996 (1999), using the Total Starch Assay Kit (K-TSTA-100A, Megazyme, Ireland). The experiments were conducted five times.
Reducing sugar
The DNS method (3, 5-2 Nitro Salicylic Acid) was used to determine reducing sugar (Pothiraj and Eyini 2007). The experiments were conducted five times.
Protein
The protein content was measured by the micro-Kjeldahl method (K100, Hanon Instrument, Jinan, China) (Onweluzo and Nwabugwu 2009). The experiments were conducted five times.
Free amino acid
The amino acid analyser (L-8900, Hitachi, Tokyo, Japan) was used to determine the free amino acid content. The samples (0.1 g) were added to 14 ml hydrolysis tubes, and mixed with 10 ml HCl (6 mol/L). The samples were then digested under nitrogen atmosphere at 110 °C for 22 h. The mixtures were dried in a water bath, and then were diluted using sodium citrate buffer (pH 2.2). The samples were filtered with 0.22 µm organic filter membranes, and used for analysis. The experiments were conducted five times.
Acid-soluble protein
A procedure presented by Cuevas-Rodríguez et al. (2004) with some modifications, was utilized. The acid-soluble protein content was calculated as the acid-soluble nitrogen amount multiplied by 0.625. The acid-soluble nitrogen of samples was measured previously by, adding 25 ml 10% TCA, and then the samples were shaken at 200 rpm at a temperature of 25 °C for 1 h. The sediment was removed by centrifugation at 4000×g for 20 min. Then, distilled water was added to the supernatant to make up a constant volume, and 5 ml was used for the measurement of acid-soluble nitrogen by the micro-Kjeldahl. The experiments were conducted five times.
Crude fat
The content of crude fat was measured by the Soxhlet extractor method based on AOAC (2000).
Low-field nuclear magnetic resonance
A procedure proposed by Li et al. (2015) was used, with modification. The LF-NMR (NM120, Niumag Electronic Technology, Shanghai, China) was applied to determine the water distribution and migration during the solid-state fermentation by fungi. Approximately 1.5 g of fermented or unfermented samples with different fermentation times were put into 15 mm NMR tubes. Carr–Purcell–Meiboom–Gill (CPMG) sequences were applied to determine the T2 parameters. The test parameters were as follows: aτ-value = 100 μs, echoes = 3000, scan repetitions = 16, and a repetition time = 5 s. MultiExpInv Analysis software (Niumag Electronic Technology, Shanghai, China) was used for data analysis. The experiments were conducted five times.
Differential scanning calorimetry (DSC)
A TA Instruments Q2000 instrument (Pyris6, Perkin Elmer, America) was applied to obtain DSC data. The sample chamber was purged with nitrogen gas (40 ml/min). Fermented or unfermented samples (10 mg) were weighted in pan and heated from 40 °C to 250 °C at a rate of 10 °C/min. Then thermograms obtained were analysed.
Scanning electron microscope (SEM)
The microstructure of the samples was observed with the SEM (PW-100-011, LASER Company, China). Dried, ground samples were put on an aluminum stub using stick tape, and then were coated with gold. The SEM pictures were taken at an accelerating voltage of 20 kV and 2000 × magnification (Shim and Mulvaney 2010).
Multiple regression analysis (MRA)
Pearson’s correlation analysis was used to select the indicators for inclusion in the MRA. The T2 parameters were as the dependent variable and the indicators were as independent variables in the MRA. The weights of each indicator were determined as the quotient of its standardized regression coefficient divided by the sum of the standardized regression coefficient for all indicators. The weights of each indicator were applied to compare the relative importance of different independent variables to the dependent variable.
Statistical analysis
All the diagrams were plotted by Origin version 8.5 (Origin Lab Corp., USA). The significant differences in parameters were evaluated by Duncan’s multiple range tests. The data shown in Supplementary material Table S3 were subjected to one-way analysis of variance (ANOVA). Pearson correlation coefficients for the relationship between properties were calculated using SPSS version 22.0 (IBM Corp., Armonk, NY, USA). The significance correlation was defined as *(p < 0.05) and **(p < 0.01). Principal component analysis (PCA) was applied to show the variables among the samples during different fermentation time, using the normalization data obtained by LF-NMR.
Results and discussion
Analysis of the chemical compositions of the samples
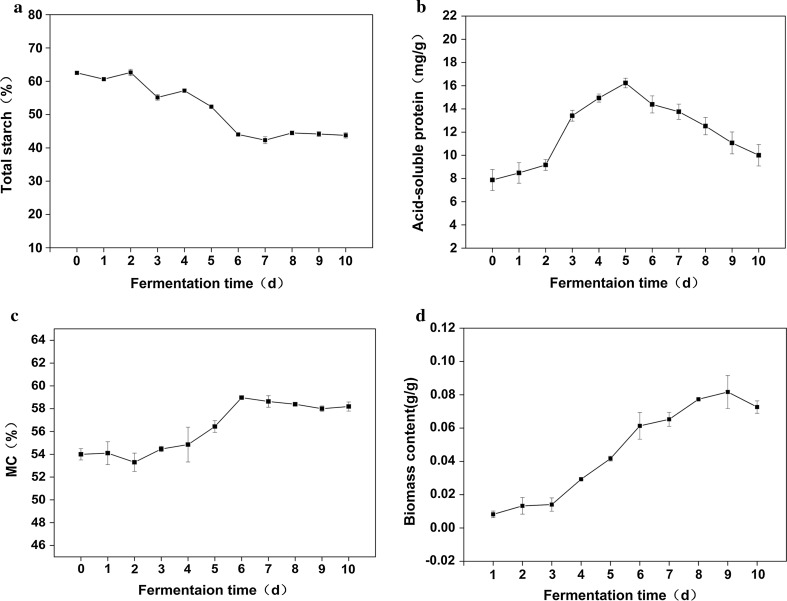
Solid-state fermentation is often accompanied with by changes in chemical compositions and metabolites (Ilowefah et al. 2015). Therefore, chemical compositions are often used to reflect the solid-state fermentation process. The solid-state fermentation of cornmeal is the process by which starch is hydrolyzed and converted into reducing sugar and other metabolites. As shown in Fig. 1a, the content of total starch remained relatively constant in the first 2 days of fermentation. After 2 days, the total starch content showed significantly decrease (p < 0.05), and it decreased to 42.326% at 7 days, and then kept relatively constant. In addition to starch hydrolysis, protein was hydrolyzed into small molecular weight protein and free amino acid. Figure 1b shows the change of acid-soluble protein content during SSF. Acid-soluble proteins showed a similar surging point at approximately 2 days of fermentation, implying the start of the logarithmic phase. MC is an important parameter of the status of the fermentation processing, and affects microbial growth (Madhuri et al. 2012). Therefore, MC is usually used to reflect and control solid-state fermentation. As shown in Fig. 1c, the MC increased statistically significantly from 54 to 59% during SSF (p < 0.05), because of the metabolism of fungi. The surging point appeared at approximately 2 days, the same as that of total starch and acid-soluble protein. The biomass content presented a similar tendency, except that its surging point came later, at approximately 3 days, compared with that of total starch, acid-soluble protein and moisture content (Fig. 1d). Because of the inhomogeneity of the substrate, some of the traditional measurements are time-consuming and have delay phenomenon. In addition, the traditional measurements only presented chemical information, and were unable to provide microstructure changes, which led to a lack of in-depth understanding of SSF.
Fig. 1.
Variations of traditional parameters during SSF: a total starch, b acid-soluble protein, c moisture content, d biomass content
LF-NMR analysis of solid-state fermented cornmeal
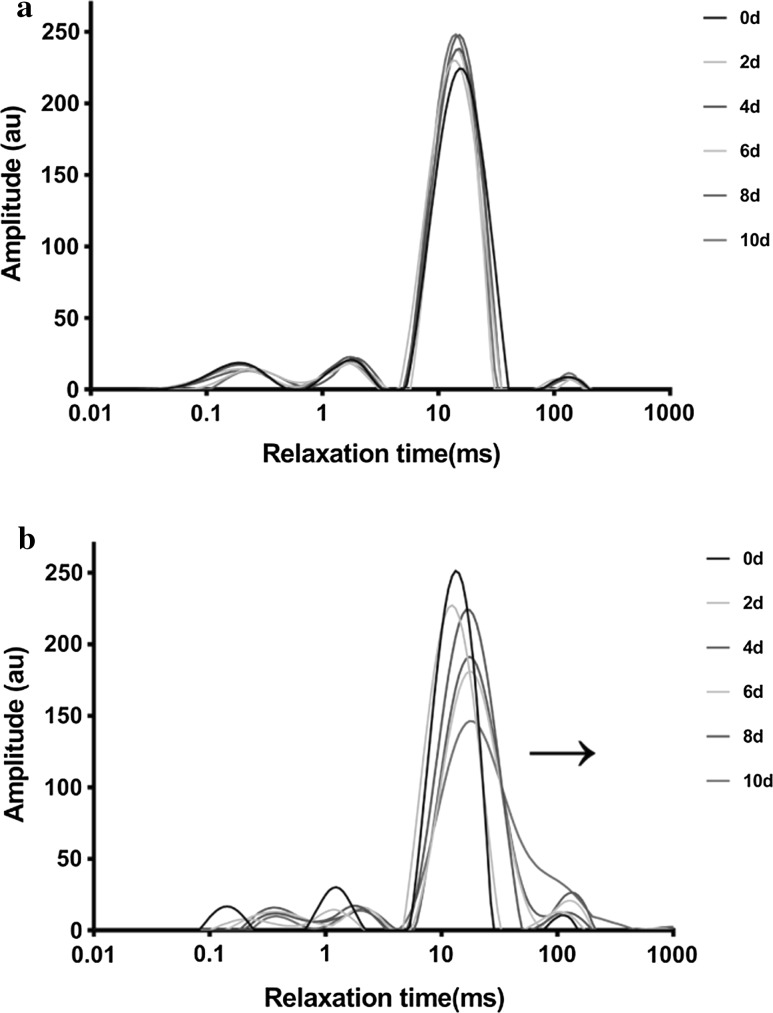
Figure 2 shows the relaxation time distribution curves of the unfermented samples and fermented samples at different times. The unfermented samples shows that the T2 parameters had no statistical significant difference within the 10 days of the fermentation time (p > 0.05). The paired-samples T test was implemented to determine whether there was a considerable significant difference between the unfermented samples and the fermented samples within 10 days. The test results are given in the Online Resource (Table S2). According to the results of the paired-samples T test, it was found that there was a considerable significant difference between unfermented samples and fermented samples (p < 0.05), indicating that solid-state fermentation significantly changes the water motivation and distribution of samples, which demonstrates that the T2 parameters changes could be ascribed to fermentation.
Fig. 2.
LF-NMR T2 relaxation time distribution curves of unfermented samples (a) and fermented samples (b)
In the presence of water, the gelatinization of starch occurs when the cornmeal is heated, and the starch forms a three-dimensional network structure, which can be characterized by meshes with different diameters in the structure (Utrillacoello et al. 2013). Additionally, cornmeal contains approximately 9.0% protein (Online Resource Table S1), which may participate in the formation of network structure. Thus, the fermented cornmeal is a three-dimensional network microstructure formed by gelatinized starch and denatured protein (Shim and Mulvaney 2010). There are many reports on the water proton relaxation of beef and tofu, demonstrating that relaxation behaviour is influenced by the matrix microstructure (Li et al. 2014; Yasir et al. 2007). Similar to many food materials (Mcdonnell et al. 2013), there were four kinds of water in the solid-state fermented cornmeal by C. militaris, including T2b, T21, T22 and T23. It is generally considered that a relaxation time of less than 1 ms corresponds to bound water, as previously reported (Li et al. 2012; Mendes et al. 2015). T2b, can be assigned to bound water, and do not have extensive contact with water, such as CH protons in amorphous and/or crystalline region of the starch. The T21 fraction, contains weakly bound water, and can be ascribed to intragranular confined water. This fraction included the exchangeable protons of starch and protein, including hydroxyl, carboxyl and sulfydryl protons. T22 was the major fraction, which was ascribed to mobile bulk water existing in the interstices of the microstructure of gel cornmeal. T23 was considered free water.
The T22 relaxation time showed a significant increase to a longer relaxation time after 2 days (Supplementary material Table S3), indicating this fraction water (T22) was in a more mobile state, which demonstrated the start of the logarithmic phase. The decreased M22 was due to the water evaporation and water consumption for substrate hydrolysis, which also demonstrates that T22 was the main available water for fungi during SSF. For solid-state fermentation for 8–10 days, the results show that the populations of T2b, T21 and T22 did not change significantly (Online Resource Table S3), and they were accompanied by a relatively constant content of chemical compositions. Based on this metabolic analysis, these phenomena indicated the beginning of the stationary growth. Research on the cellulose hydrolysis ascribed the limiting factor for hydrolysis to water availability (Selig et al. 2012). Our results also showed that the available water (M22) did not change significantly (p > 0.05). This result demonstrated that the decreasing rate of hydrolysis during the stationary phase can be caused by a decrease in water mobility and available water.
PCA analysis
To achieve a more straightforward description on SSF process, PCA of the LF-NMR was used to demonstrate the systematic relationship of the fermentation time with T2 variables. By extracting the principal components from the T2 dates, PCA can explore the trend of samples and scores of samples taken throughout solid state fermentation (Han et al. 2014). As seen in Fig. 3, PC1 explained 83.37% of the variation in the data, and PC2 explained 12.63% of the variation, and clearly discriminated according to the fermentation times. Significant differences were observed between the samples on the 2 days and 3 days. These results indicated that the exponential phase evaluated by LF-NMR began at approximately 3 days, which was in agreement with the results suggested by the chemical composition measurement. The samples fermented for 0–2 days, were located on the left side. The samples fermented for 3–6 days were placed on the lower and right sides of the plot, and the samples fermented for 7–10 days held their position in the higher and right sides. These results indicated that the different fermentation stages can be distinguished by LF-NMR. LF-NMR measurements were fast and required no sample pretreatment, while providing useful information of the fermentation progress.
Fig. 3.
PCA plot obtained using distributed NMR T2 relaxation data of different fermentation time
MRA analysis
It is recognized that the NMR relaxation behaviour of different matrixes can be ascribed to different biopolymer composition (Shao and Li 2013). Meanwhile, the changes in the chemical compositions of substrates are complicated during SSF, and many chemical compositions are correlated with T2 parameters (Online Resource Table S4). The standardized regression coefficient of MRA is frequently used to compare the relative importance of different independent variables to the dependent variable (Bring 1994). Thus, the MRA was conducted to further compare the relative importance of the chemical compositions to T2 parameters. Based on the Pearson’s correlation analysis between the T2 parameters and chemical compositions, the indicators retained for the MRA included moisture content, total starch, reducing sugar, protein, free amino acid, acid-soluble protein and fat (Table 1). The weights of the different indicators were calculated by stepwise regression. If the indicator weight was assigned based on the MRA, the indicator that affected the T2 parameters the most obtained the greatest weight. Among the selective indicators of the regression models, total starch was assigned the highest weight (> 0.46), which revealed a strong correlation between total starch and T2 relaxation. This result further suggested that the changes in T2 parameters were caused by the hydrolysis of starch during SSF.
Table 1.
The weights of the chemical composition indicators that were assigned using multiple regression analysis
| Model | Unstandardized coefficients | Standardized coefficients | Weight | Model | Unstandardized coefficients | Standardized coefficients | Weight | ||
|---|---|---|---|---|---|---|---|---|---|
| T2b | Constant | 2.519 | M2b | Constant | 0.23 | ||||
| Total starch | 0.025 | − 1.009 | 0.639 | Total starch | − 0.002 | − 1.063 | 0.694 | ||
| Reducing sugar | − 0.018 | − 0.286 | 0.181 | Reducing sugar | − 0.001 | − 0.191 | 0.125 | ||
| Protein | − 0.082 | − 0.283 | 0.179 | Protein | − 0.004 | − 0.181 | 0.118 | ||
| Free amino acid | − 0.008 | − 0.097 | 0.063 | ||||||
| T21 | Constant | 20.104 | M21 | Constant | 0.117 | ||||
| Total starch | − 0.15 | − 1.109 | 0.574 | Total starch | − 0.001 | − 0.931 | 0.465 | ||
| Reducing sugar | − 0.125 | − 0.354 | 0.183 | Protein | 0.007 | 0.714 | 0.355 | ||
| Protein | − 0.49 | − 0.307 | 0.159 | Free amino acid | − 0.012 | − 0.358 | 0.179 | ||
| Free amino acid | − 0.874 | − 0.163 | 0.084 | ||||||
| T22 | Constant | 90.530 | M22 | Constant | 0.587 | ||||
| Total starch | − 0.581 | − 1.04 | 0.555 | Total starch | 0.005 | 1.084 | 0.828 | ||
| Reducing sugar | − 0.443 | − 0.304 | 0.162 | Reducing sugar | 0.003 | 0.225 | 0.172 | ||
| Protein | − 2.417 | − 0.367 | 0.196 | ||||||
| Free amino acid | − 3.592 | − 0.163 | 0.087 | ||||||
| T23 | Constant | 240.671 | M23 | Constant | 0.174 | ||||
| Total starch | − 1.123 | − 1.076 | 0.661 | Total starch | − 0.002 | − 1.184 | 0.777 | ||
| Reducing sugar | − 0.648 | − 0.238 | 0.146 | Reducing sugar | − 0.002 | − 0.34 | 0.223 | ||
| Protein | − 2.434 | − 0.198 | 0.122 | ||||||
| Free amino acid | − 4.809 | − 0.117 | 0.072 |
DSC analysis
After the gelatinized matrix is rearranged, many crystalline structures are formed. To destroy these crystal structures, additional energy is required to redissolve starch molecules. Therefore, the recrystallization matrix had heat absorption peaks that occurred in DSC; the area of the heat absorption peak is the heat enthalpy of recrystallization (ΔH). DSC technology has been used to examine starches following defined thermal treatments to provide samples exhibiting degrees of structure loss, which can reflect the structure stability of the matrix (Cooke and Gidley 1992). Thus, the DSC analysis was applied to determine the structure characteristics of samples with different fermentation times. As shown in Table 2, an absorption peak at 138.02 °C for the unfermented sample was observed, which was higher than that of the fermented samples. Lower temperature crystal melting peaks in the DSC of the fermented samples might be attributed to the disorganization of the starch crystallites. As expected, the ΔH of the fermented samples decreased significantly (p < 0.05) compared to that of the unfermented samples, which indicated the decrease crystallization. At the same time, the ΔH showed a similar trend to the T22 relaxation time, which explained the reason of increasing the T22 during SSF. In general, T2 relaxation behaviour of various matrixes could be ascribed to water fractions existing in different microstructures (Belton et al. 1988). Considering the aforementioned facts, we propose that the increase in T22 can be ascribed to loosening of the structure at starch hydrolysis, introducing more water into the structure. The decrease in ΔH indicated that the relative stabilized network could be destroyed, which increases the water motivation, causing an increase in T22.
Table 2.
Effect of fermentation time on thermal properties of samples
| Time (d) | To onset (°C) | Tp peak (°C) | Tc stop (°C) | ΔH (J/g) |
|---|---|---|---|---|
| 0 | 92.53 ± 0.7a | 138.02 ± 1.1c | 247.07 ± 1.2a | 266.1 ± 5.3e |
| 1 | 104.55 ± 1.1c | 133.69 ± 1.3b | 243.94 ± 2.1a | 244.9 ± 6.2d |
| 2 | 103.69 ± 0.7c | 132.51 ± 1.9b | 247.03 ± 1.1a | 231.5 ± 2.7d |
| 3 | 101.53 ± 0.9c | 131.33 ± 1.8b | 242.32 ± 1.4a | 226.5 ± 6.7c |
| 4 | 92.44 ± 1.2a | 131.28 ± 0.9b | 242.75 ± 0.4a | 216.8 ± 5.7b |
| 5 | 94.16 ± 1.3a | 128.7 ± 1.5a | 245.29 ± 1.9a | 208.3 ± 7.3b |
| 6 | 98.89 ± 0.8b | 127.69 ± 1.9a | 247.02 ± 0.8a | 190.3 ± 5.2a |
| 7 | 96.58 ± 1.5b | 129.4 ± 2.1a | 245.3 ± 2.5a | 212.3 ± 7.5b |
| 8 | 95.85 ± 0.6ab | 129.12 ± 0.9a | 247.01 ± 2.4a | 208.9 ± 8.3b |
| 9 | 93.34 ± 0.7a | 131.11 ± 0.8b | 247.01 ± 1.8a | 210.3 ± 3.7b |
| 10 | 92.52 ± 0.5a | 130.54 ± 0.9ab | 247.1 ± 1.9a | 207.4 ± 3.5b |
Data are expressed as means ± standard deviations of three experiments; Samples means with different superscript letters in the same column are significantly different at p < 0.05
SEM analysis
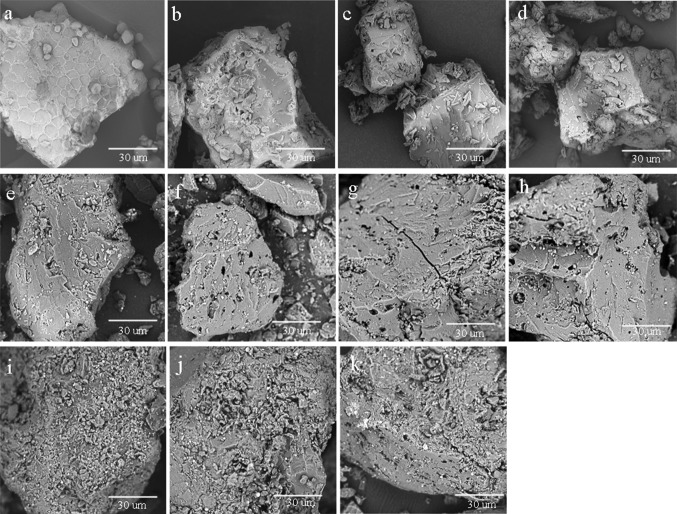
Figure 4 illustrates the microstructure of unfermented gelatinized and fermented samples. Large aggregates of flour particles with typical starch granular shapes were formed after gelatinization. The structure of those cornmeal systems was a tightly packed system of swollen granules after gelatinization. In the case of the fermented sample produced with 1–2 days of fermentation (Fig. 4), the process resulted in a product with numerous micropores. With the increase in the fermentation time, more micropores can be seen in Fig. 4. This result can be attributed to the action of α-amylase that caused the deepening of existing micropores as well as the creation of new ones. The results from the LF-NMR during fermentation are interpreted in terms of its effect on the cornmeal microstructure as shown in Fig. 4. SEM demonstrated that more micropores took place as a result of fermentation during the logarithmic phase, as observed on the substrate–water interactions in LF-NMR. Thus, the significant increase in T22 relaxation time during logarithmic phase can be ascribed to the hydrolysis mechanism of the enzyme from C. militaris, hydrolysing the starch on the surface of the granule. During the logarithmic period, this process introduces more cavities and micropores on the surface of starch (Fig. 4), increasing the chances of water to permeate into the starch structure as seen in the increase in the total water content during fermentation (Fig. 1c). Thus, the increased water promoted water motivation and, consequently, increased the relaxation time of T22. Li and Yue (2012) also found the similar results, who found thatα-amylase treatment of mung bean starch increased its water holding capacity. Besides, it can also be seen how fermentation increases the T22 relaxation time (Fig. 2). Based on the results of MRA, the hydrolysis of starch has, by far, the most remarkable effect on the T2 relaxation behavior during fermentation. The longer T22 relaxation time can be ascribed to loosening of the structure at the beginning of starch hydrolysis. Thus, the actions of the fermentation not only hydrolyze the starch but also introduce water into the matrix by the formation of numbers micropores.
Fig. 4.
Scanning electron micrographs of unfermented sample (a), fermented sample at 1 days (b), fermented sample at 2 days (c), fermented sample at 3 days (d), fermented sample at 4 days (e), fermented sample at 5 days (f), fermented sample at 6 days (g), fermented sample at 7 days (h), fermented sample at 8 days (i), fermented sample at 9 days (j), fermented sample at 10 days (k)
It was worth noted that T22 relaxation time showed decreasing tendency during the stationary period. The T22 relaxation time did not increase with the appearance of more micropores on the surface of granule (Fig. 4). During the stationary period, the rate of hydrolysis decreased, coinciding with the deceasing in water motivation (T22) and relatively constant available water (M22), although the amount of water maintained a relatively higher content compared with that of the beginning period. The decreasing in T22 relaxation time during the stationary period may be ascribed to hydrolysis mechanism of the enzyme. As shown in DSC results (Table 2), the fermented samples still maintained the overall structural characteristics. The hydrolysis the starch was on the surface of the granule, and the fermented substrate still maintained the overall structural characteristics. The remaining structure water peak can be interpreted as recalcitrant structures in the matrix. These changes occur mainly at the molecular level and are usually detectable by microscopic methods, but the LF-NMR can clearly provide information on the structure and starch–water interactions during SSF. The hydrolysis mechanism of enzyme is also good description of amylase action, which can provide information for enzyme production from C. militaris.
Conclusion
The T2 parameters showed significant changes in the solid-state fermented cornmeal. PCA analysis of T2 variations showed significant differences between different fermentation stages. The mobile bulk water (T22) was the main available water for fungi, and increased significantly during logarithmic growth. The weights of the different indicators by MRA demonstrated that there was a strong correlation between starch and T2 relaxation behaviour. The fermentation not only hydrolyze the starch but also introduce water into the matrix by the formation of micropores, which explained the reason for the increase in T22 relaxation time. The decrease in the relaxation time of T22 can be interpreted as responsible for the decrease in hydrolysis rate during stationary period. Therefore, LF-NMR combined with statistical analysis is an effective method for further clarification and understanding of the fungi–starch matrix–water fermentation system and its interactions.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Acknowledgements
This work was supported by the National Key R&D Program of China. (Grant Number: 2016YFD0400702), and Jilin Province “Double Ten Project” Major Scientific and Technological Projects - Key Technology Research and Product Development of Health Staple Food of Coarse Grains. (Grant Number: 20150201010NY).
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- AACC I (1999) Approved Methods ofAnalysis, 11th Ed. Method 76-13.01. Total starch assay procedure (megazyme amyloglucosidase/alpha-amylase method). Approval November 3. AACC International, St. Paul, MN, USA
- AOAC . Official methods of analysis. 17. Washington, DC: Association of Official Analytical Chemists; 2000. [Google Scholar]
- Bartkiene E, Krungleviciute V, Juodeikiene G, Vidmantiene D, Maknickiene Z. Solid state fermentation with lactic acid bacteria to improve the nutritional quality of lupin and soya bean. J Sci Food Agric. 2015;95:1336–1342. doi: 10.1002/jsfa.6827. [DOI] [PubMed] [Google Scholar]
- Belton PS, Hills BP, Raimbaud ER. The effects of morphology and exchange on proton N.M.R. relaxation in agarose gels. Mol Phys. 1988;63:825–842. doi: 10.1080/00268978800100591. [DOI] [Google Scholar]
- Bring J. How to standardize regression coefficients. Am Stat. 1994;48:209–213. [Google Scholar]
- Cooke D, Gidley MJ. Loss of crystalline and molecular order during starch gelatinisation: origin of the enthalpic transition. Carbohydr Res. 1992;227:103–112. doi: 10.1016/0008-6215(92)85063-6. [DOI] [Google Scholar]
- Cozzolino D, Parker M, Dambergs RG, Herderich M, Gishen M. Chemometrics and visible-near infrared spectroscopic monitoring of red wine fermentation in a pilot scale. Biotechnol Bioeng. 2010;95:1101–1107. doi: 10.1002/bit.21067. [DOI] [PubMed] [Google Scholar]
- Cuevas-Rodríguez EO, MiIán-Carrillo J, Mora-Escobedo R, Cárdenas-Valenzuela OG, Reyes-Moreno C. Quality protein maize (Zea mays L.) tempeh flour through solid state fermentation process. LWT-Food Sci Technol. 2004;37:59–67. doi: 10.1016/S0023-6438(03)00134-8. [DOI] [Google Scholar]
- Han MY, Wang P, Xu XL, Zhou GH. Low-field NMR study of heat-induced gelation of pork myofibrillar proteins and its relationship with microstructural characteristics. Food Res Int. 2014;62:1175–1182. doi: 10.1016/j.foodres.2014.05.062. [DOI] [Google Scholar]
- Ilowefah M, Bakar J, Ghazali HM, Mediani A, Muhammad K. Physicochemical and functional properties of yeast fermented brown rice flour. J Food Sci Technol. 2015;52:5534–5545. doi: 10.1007/s13197-014-1661-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jiang H, Liu G, Xiao X, Mei C, Ding Y, Yu S. Monitoring of solid-state fermentation of wheat straw in a pilot scale using FT-NIR spectroscopy and support vector data description. Microchem J. 2012;102:68–74. doi: 10.1016/j.microc.2011.12.003. [DOI] [Google Scholar]
- Kang M, Zhai FH, Li XX, Cao JL, Han JR. Total phenolic contents and antioxidant properties of buckwheat fermented by three strains of Agaricus. J Cereal Sci. 2017;73:138–142. doi: 10.1016/j.jcs.2016.12.012. [DOI] [Google Scholar]
- Kirtil E, Oztop MH. 1H nuclear magnetic resonance relaxometry and magnetic resonance imaging and applications in food science and processing. Food Eng Rev. 2016;8:1–22. doi: 10.1007/s12393-015-9118-y. [DOI] [Google Scholar]
- Li XH, Yue XJ. An enzymatic method for inhibiting retrogradation of mung bean starch and its application in ice cream. Food Science. 2012;33:339–344. [Google Scholar]
- Li X, Ma LZ, Tao Y, Kong BH, Li PJ. Low field-NMR in measuring water mobility and distribution in beef granules during drying process. Adv Mater Res. 2012;550:3406–3410. [Google Scholar]
- Li Y, Jia W, Zhang CH, Li X, Wang JZ, Zhang DQ, Mu GF. Fluctuated low temperature combined with high-humidity thawing to reduce physicochemical quality deterioration of beef. Food Bioprocess Technol. 2014;7:3370–3380. doi: 10.1007/s11947-014-1337-3. [DOI] [Google Scholar]
- Li T, Tu C, Rui X, Wang K, Xiao Y, Dong MS. Study of water dynamics in the soaking, steaming, and solid-state fermentation of glutinous rice by LF-NMR: a novel monitoring approach. J Agric Food Chem. 2015;63:3261–3270. doi: 10.1021/acs.jafc.5b00769. [DOI] [PubMed] [Google Scholar]
- Lin S, Yang S, Li X, Chen F, Zhang M. Dynamics of water mobility and distribution in soybean antioxidant peptide powders monitored by LF-NMR. Food Chem. 2016;199:280–286. doi: 10.1016/j.foodchem.2015.12.024. [DOI] [PubMed] [Google Scholar]
- Madhuri A, Nagaraju B, Harikrishna N, Reddy G. Production of alkaline protease by Bacillus altitudinis GVC11 using Castor Husk in solid-state fermentation. Appl Biochem Biotechnol. 2012;167:1199–1207. doi: 10.1007/s12010-012-9570-6. [DOI] [PubMed] [Google Scholar]
- Mcdonnell CK, Allen P, Duggan E, Arimi JM, Casey E, Duane G, Lyng JG. The effect of salt and fibre direction on water dynamics, distribution and mobility in pork muscle: a low field NMR study. Meat Sci. 2013;95:51–58. doi: 10.1016/j.meatsci.2013.04.012. [DOI] [PubMed] [Google Scholar]
- Mendes GDO, Silva NM, Anastácio TC, Vassilev NB, Ribeiro JI, Silva IR, Costa MD. Optimization of Aspergillus nigerrock phosphate solubilization in solid-state fermentation and use of the resulting product as a P fertilizer. Microb Biotechnol. 2015;8:930. doi: 10.1111/1751-7915.12289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Monsoor MA, Proctor A, Siebenmorgen TJ. Surface lipid and free acids (FFA) content of head and broken rice produced by milling after different drying treatments. Cereal Chem. 2004;81:705–709. doi: 10.1094/CCHEM.2004.81.6.705. [DOI] [Google Scholar]
- Onweluzo JC, Nwabugwu CC. Fermentation of millet (Pennisetum americanum) and pigeon pea (Cajanus cajan) seeds for flour production: effects on composition and selected functional properties. Pak J Nutr. 2009;8:1136–1141. [Google Scholar]
- Pearce KL, Rosenvold K, Andersen HJ, Hopkins DL. Water distribution and mobility in meat during the conversion of muscle to meat and ageing and the impacts on fresh meat quality attributes—a review. Meat Sci. 2011;89:111–124. doi: 10.1016/j.meatsci.2011.04.007. [DOI] [PubMed] [Google Scholar]
- Pothiraj C, Eyini M. Enzyme activities and substrate degradation by fungal isolates on cassava waste during solid state fermentation. Mycobiology. 2007;35:196–204. doi: 10.4489/MYCO.2007.35.4.196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Selig MJ, Hsieh CW, Thygesen LG, Himmel ME, Felby C, Decker SR. Considering water availability and the effect of solute concentration on high solids saccharification of lignocellulosic biomass. Biotechnol Prog. 2012;28:1478. doi: 10.1002/btpr.1617. [DOI] [PubMed] [Google Scholar]
- Shao XL, Li YF. Application of low-field NMR to analyze water characteristics and predict unfrozen water in blanched sweet corn. Food Bioprocess Technol. 2013;6:1593–1599. doi: 10.1007/s11947-011-0727-z. [DOI] [Google Scholar]
- Shim J, Mulvaney SJ. Effect of heating temperature, pH, concentration and starch/whey protein ratio on the viscoelastic properties of corn starch/whey protein mixed gels. J Sci Food Agric. 2010;81:706–717. doi: 10.1002/jsfa.869. [DOI] [Google Scholar]
- Tang HR, Brun A, Hills B. A proton NMR relaxation study of the gelatinisation and acid hydrolysis of native potato starch. Carbohydr Polym. 2001;46:7–18. doi: 10.1016/S0144-8617(00)00265-4. [DOI] [Google Scholar]
- Utrillacoello RG, Bellopérez LA, Vernoncarter EJ, Rodriguez E, Alvarezramirez J. Microstructure of retrograded starch: quantification from lacunarity analysis of SEM micrographs. J Food Eng. 2013;116:775–781. doi: 10.1016/j.jfoodeng.2013.01.026. [DOI] [Google Scholar]
- Wei PL, Cen PL, Sheng CQ. Comparison of three biomass estimation methods in solid state fermentation. J Food Sci Biotechnol. 2006;25:60–62. [Google Scholar]
- Wu J, Li Y, Gao X. Monitoring a typical fermentation process of natto by low-field nuclear magnetic resonance (LF-NMR) and magnetic resonance imaging (MRI) techniques. Anal Methods. 2016 [Google Scholar]
- Xiao Y, Xing GL, Rui X, Li W, Chen XH, Jiang M, Dong MS. Effect of solid-state fermentation with Cordyceps militaris SN-18 on physicochemical and functional properties of chickpea (Cicer arietinum L.) flour. LWT-Food Sci Technol. 2015;63:1317–1324. doi: 10.1016/j.lwt.2015.04.046. [DOI] [Google Scholar]
- Yang S, Zheng MZ, Cao Y, Dong YJ, Sanabil LJ. Optimization of liquid fermentation conditions for biotransformation zein by Cordyceps militaris 202 and characterization of physicochemical and functional properties of fermentative hydrolysates. Braz J Microbiol. 2018;49:621–631. doi: 10.1016/j.bjm.2017.12.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yasir SBM, Sutton KH, Newberry MP, Andrews NR, Gerrard JA. The impact of Maillard cross-linking on soy proteins and tofu texture. Food Chem. 2007;104:1502–1508. doi: 10.1016/j.foodchem.2007.02.042. [DOI] [Google Scholar]
- Zhu F. NMR spectroscopy of starch systems. Food Hydrocolloids. 2017;63:611–624. doi: 10.1016/j.foodhyd.2016.10.015. [DOI] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.