Abstract
Hyaluronan (HA), an extracellular matrix glycosaminoglycan, is implicated in the pathogenesis of both type 1 diabetes (T1D) as well as type 2 diabetes (T2D) and has been postulated to be increased in these diseases due to hyperglycemia. We have examined the serum and tissue distribution of HA in human subjects with T1D and T2D and in mouse models of these diseases and evaluated the relationship between HA levels and glycemic control. We found that serum HA levels are increased in T2D but not T1D independently of hemoglobin-A1c, C-peptide, body mass index, or time since diabetes diagnosis. HA is likewise increased in skeletal muscle in T2D subjects relative to non-diabetic controls. Analogous increases in serum and muscle HA are seen in diabetic db/db mice (T2D), but not in diabetic DORmO mice (T1D). Diabetes induced by the β-cell toxin streptozotozin (STZ) lead to an increase in blood glucose but not to an increase in serum HA. These data indicate that HA levels are increased in multiple tissue compartments in T2D but not T1D independently of glycemic control. Given that T2D but not T1D is associated with systemic inflammation, these patterns are consistent with inflammatory factors and not hyperglycemia driving increased HA. Serum HA may have value as a biomarker of systemic inflammation in T2D.
Keywords: hyaluronan, diabetes, T1D, T2D, biomarker
Introduction
Hyaluronan (HA) is an extracellular matrix (ECM) polymer and glycosaminoglycan [1]. It is a long, unbranching polymer made of repeating disaccharides (glucuronic acid and N-acetyl-glucosamine). In healthy tissues, HA is produced by a diverse range of cells and tissues and is incorporated into tissues such as joints [2], basement membranes and the vitreous of the eye [3]. At these sites, it contributes to lubrication, acts as a structural component, and space filler [4]. HA bound within tissues interacts with a number of proteins that modify its function and longevity [1, 5–7].
In inflamed tissues, the amount of HA is greatly increased [3]. Cellular HA production in vitro is increased in response to a variety of pro-inflammatory cytokines [8, 9], microbial products [9, 10], hyperglycemia [11, 12], and other triggers [10, 13], and the presumption is that these factors also drive HA synthesis in vivo. HA undergoes extensive catabolism mediated by endogenous hyaluronidases, by bacterial hyaluronidases, by mechanical forces, and by oxidative stress [14]. Together, this increased synthesis and catabolism lead to the accumulation of HA fragments within inflamed tissues and tumors [15–19]. Increased HA may contribute to local inflammatory processes. HA fragments promote cellular activation, migration, and proliferation [1, 3, 20–24] via interactions with its receptors [25], including RHAMM [26], CD44 [27], and Toll-like receptors 2 and/or 4 [28].
Upon the resolution of inflammation HA fragments are cleared while in the setting of chronic inflammation, HA accumulates [29]. Abundant local HA characterizes chronic inflammation in many tissues including liver cirrhosis, asthma, and other diseases [30–33]. Reflecting this, serum HA is used as a biomarker of liver cirrhosis [34] and primary sclerosing cholangitis [35]. The amount and size of HA at sites of inflammation may thereby provide contextual information that reflects the stage of injury and its stage of resolution [17, 36].
HA has been implicated in the pathogenesis of autoimmune, type 1 diabetes (T1D). In healthy islets, HA is a normal component of the peri-islet and intra-islet regions adjacent to micro-vessels [37]. The development of autoimmune insulitis is associated with substantial changes in the islet ECM and deposition of intra-islet HA [38, 39]. In human T1D tissue samples from cadaveric organ donors obtained through the JDRF nPOD program, HA deposits were found to be present in islets from recent-onset T1D donors but not in non-diabetic controls [40]. Further, HA deposits were both temporally and anatomically associated with autoimmune insulitis in both T1D and in the DORmO mouse model of the disease. The DORmO mice are the offspring of DO11.10 and RIPmOVA transgenic mice. They carry a T-cell receptor transgene specific for OVA (emulating auto-reactive CD4+ T-cells) while simultaneously expressing OVA in conjunction with the insulin gene promoter on pancreatic b-cells (emulating the auto-antigen) [41]. In diabetic humans and mice the amount and distribution of HA closely tracked with the infiltration of CD3+ T-cells and the disappearance of insulin staining. HA was not increased within neighboring islets without active insulitis [41]. We and others have reported that interventions that clear HA or prevent its synthesis can prevent autoimmune diabetes in mice [41–44].
HA is also implicated in the pathogenesis of type 2 diabetes (T2D). HA deposition in T2D has been noted in skeletal muscle [45], adipose tissue [46, 47], and other tissues of obese and diabetic animals [48–52] as well as at sites of diabetes-associated pathology, such as vascular lesions [51, 53]. The T2D mouse model the db/db mice are leptin receptor deficient and uniformly exhibit severe obesity and hyperglycemia [54]. In humans and mice serum HA levels are likewise increased in T2D [51]. HA in peripheral tissues has been implicated in insulin resistance, as treatment with a recombinant hyaluronidase [45] or anti-CD44 antibodies [46] improve insulin sensitivity.
The finding that HA contributes in different ways to the pathogenesis of both T1D and T2D raises basic questions about the tissue distribution of HA, the role of hyperglycemia in this accumulation, and the specificity of serum HA as a biomarker for T2D versus T1D.
Here, we have asked how T1D and T2D status and glycemic control impact tissue deposition of HA. For these assessments, we have studied serum and skeletal muscle from human subjects with T1D, T2D or non-diabetic controls, as well as the analogous tissues collected from mouse models of these diseases.
Results
Serum and muscle HA concentrations are not increased in a mouse model of T1D
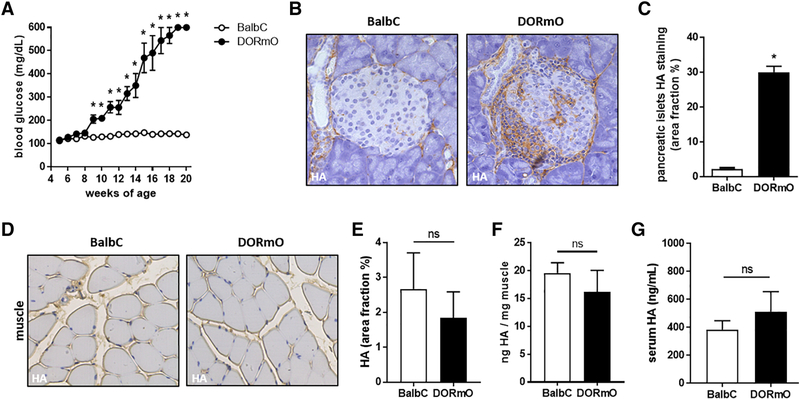
We first compared serum HA in the DORmO mouse model of T1D versus healthy, strain matched controls. At 15 weeks of age, DORmO mice had blood glucose levels over 250 mg/dL and were considered diabetic (Fig. 1A). At 10 weeks of age, we observed a significant increase in HA accumulation in the pancreatic islets of DORmO mice compared to BalbC controls (Fig. 1B,C).
Figure 1: Serum and muscle HA concentrations are not increased in a T1D mouse model.
A: Blood glucose of DORmO and BalbC mice from 4 to 20 weeks of age. B: Representative images of pancreatic islets from DORmO and BalbC mice stained for HA, in brown. C: Quantification of HA accumulation in pancreatic islets from IHC images. D: Representative images of skeletal muscle images from DORmO and BalbC mice stained for HA, in brown. E: Quantification of HA accumulation in skeletal muscle from IHC images. F: Quantification of HA content of skeletal muscle from DORmO and BalbC mice. G: Serum HA level from DORmO and BalbC mice. N = 5–10 mice per group. Data represent mean +/− SEM, *P < 0.05 vs the respective control by unpaired t test.
We further examined the skeletal muscle of 15 week old mice for HA content and found no significant difference between the DORmO and BalbC mice in histologic muscle sections stained for HA (Fig. 1D,E) or biochemically when determining the HA content of the muscle (Fig. 1F). The muscle HA content of the DORmO mice was 17.3 +/− 1.8 ng HA/mg muscle versus 19.6 +/− 0.9 ng HA/mg muscle for the BalbC controls (Fig. 1F).
We also collected serum from these animals and examined their HA content. We found that serum HA levels were not significantly elevated in 15 week old DORmO mice (514.0 +/− 71.1 ng/mL) compared to BalbC controls (384.5 +/− 31.6 ng/mL) (Fig. 1G).
These data indicate that serum and muscle HA are not increased in a mouse model of T1D but that HA is increased at the primary site of inflammation in this disease - the pancreatic islet.
Serum and muscle HA concentrations are increased in a mouse model of T2D
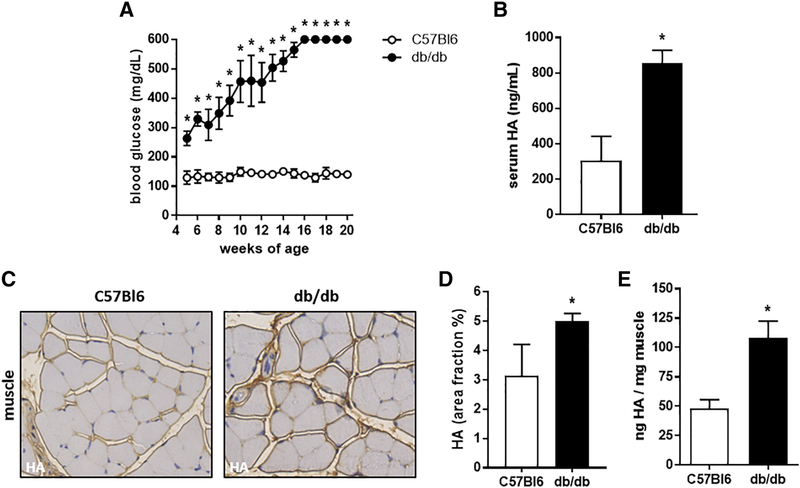
We next asked whether serum HA was elevated in the db/db mouse model of T2D compared to strain matched C57Bl6 controls. By 6 weeks of age db/db mice had blood glucose levels over 250 mg/dL and were considered diabetic (Fig. 2A). We then collected serum from these animals and examined the HA content. Serum HA levels were significantly higher in 16 week old db/db mice (854.3 +/− 37.1 ng/mL) compared to their C57Bl6 controls (304.8 +/− 68.6 ng/mL) (Fig. 2B).
Figure 2: Serum and muscle HA concentrations are significantly elevated in a T2D mouse model.
A: Blood glucose of db/db and C57Bl6 mice from 4 to 20 weeks of age. B: Serum HA level from db/db and C57Bl6 mice. C: Representative images of skeletal muscle images from db/db and C57Bl6 mice stained for HA, in brown. D: Quantification of HA accumulation in skeletal muscle from IHC images. E: Quantification of HA content of skeletal muscle from db/db and C57Bl6 mice. N = 5–10 mice per group. Data represent mean +/− SEM, *P < 0.05 vs the respective control by unpaired t test.
In skeletal muscle tissues from 16 week old db/db and control mouse muscle sections stained for HA we found that HA was increased in the db/db mice. This was seen by histologic assessments (Fig. 2C,D) as well as by biochemical measurements (Fig. 2E). The muscle HA content of the db/db mice was with 108.1 +/− 14.5 ng HA/mg muscle significantly higher than the muscle HA content of the C57Bl6 control mice with 48 +/− 7.7 ng HA/mg muscle (Fig. 2E).
Together these data indicate that serum and muscle HA are increased in a mouse model of T2D but not in a mouse model of T1D.
STZ treatment induces hyperglycemia in mice but HA serum levels are not significantly elevated
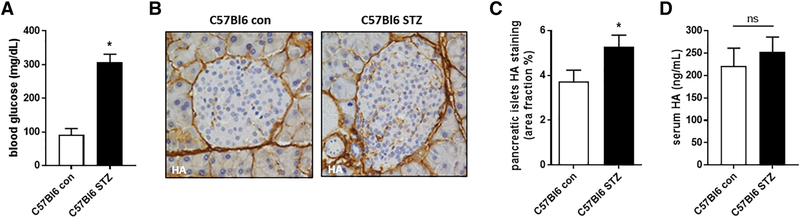
We also asked whether serum HA was elevated in C57Bl6 mice after treatment with STZ, a β-cell specific toxin. After administration of two consecutive high doses (200 mg/kg) of STZ, mice were diabetic within one week. Seven days after STZ administration we observed that blood glucose level were significantly increased (307.4 +/− 10.5 mg/dL) in the STZ treated group compared to the untreated controls (91.4 +/− 8.4 mg/dL) (Fig. 3A).
Figure 3: STZ treatment induces hyperglycemia in mice but HA serum levels are not significantly elevated.
A: Blood glucose of C57Bl6 mice with and without STZ treatment. B: Representative images of pancreatic islets from C57Bl6 mice with and without STZ treatment. C: Quantification of HA accumulation in pancreatic islets from IHC images. D: Serum HA measurements from STZ treated C57Bl6 mice and their untreated controls. N = 5–10 mice per group. Data represent mean +/− SEM, *P < 0.05 vs the respective control by unpaired t test.
We also saw a significant increase of pancreatic islet HA after STZ treatment compared to the non STZ treated mice (Fig. B,C). Serum HA level on the other hand remained unchanged after STZ treatment (220.7 +/− 18.1 ng/mL for controls vs 252.1 +/− 15.0 ng/mL for STZ treated mice) (Fig. 3D).
These data suggest that hyperglycemia does not immediately result in serum HA elevation.
Serum HA concentrations are not elevated in T1D patients
We next sought to determine whether our findings in mouse models of diabetes would be mirrored in human subjects with T1D or T2D. For these studies, blood samples were drawn from a total of 60 patients (n = 20 T1D patients, n = 20 T2D patients and n = 20 non-diabetic controls). Groups were matched for age (all between 20–45 years), gender (each group had n = 10 male and n = 10 female patients), and HbA1c (mean 7.5 % (T1D) - 7.1 % (T2D). Mean C-peptide levels were 3.7 ng/mL for the T2D patients; for the T1D subjects C-peptide data were not available. The body mass index (BMI) from non-diabetic controls (BMI 27.9) was found to be between T1D (BMI 22.8) and T2D (BMI 32.7) patients. None of the patients had any comorbidity. These data are shown in Table 1.
Table 1: Characteristics of the subjects from whom serum was collected.
Blood was drawn from a total of 20 patients per group. The group consisted of n = 10 male and n = 10 female patients. The age at time of blood draw was between 20–45 years. For the T1D and T2D patients the days since diagnosis, the HbA1c and C-peptide serum level as well as BMI were reported. None of the patients had any comorbidity. Data are represented as mean +/− SEM.
| Healthy Controls (n=20) | T1D (n=20) | T2D (n=20) | |
|---|---|---|---|
| Sex (M/F) | 10/10 | 10/10 | 10/10 |
| Age (years) | 20–45 | 20–45 | 20–45 |
| Days since diagnosis | N/A | 60 ± 32 | 645 ± 547 |
| HbA1c (%) | N/A | 7.5 ± 1.5 | 7.1 ± 1.3 |
| C-peptide (ng/mL) | N/A | N/A | 3.7 ± 0.4 |
| BMI | 27.9 ± 1.1 | 22.8 ± 0.7 | 32.7 ± 1.3 |
| Comorbidity (i.e. renal failure, retinopathy) | None | None | None |
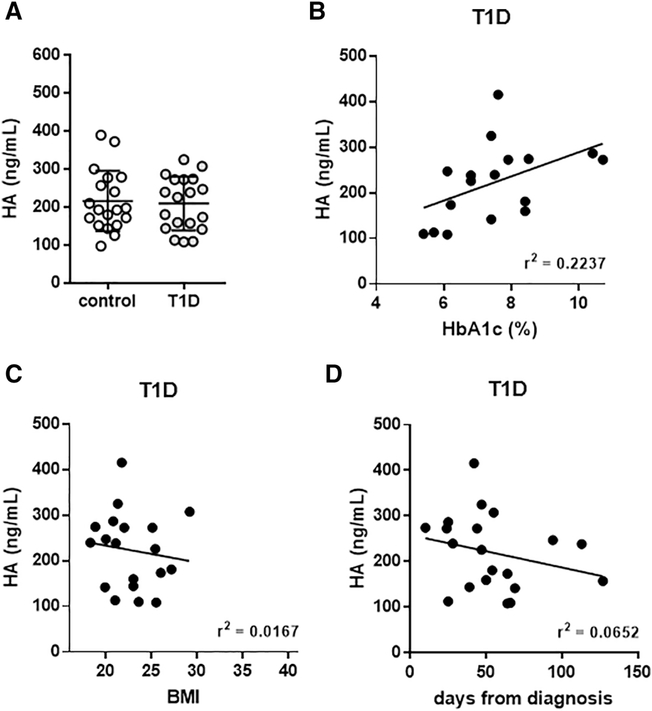
We observed that serum HA levels in T1D subjects (219.4 +/− 18.5 ng/mL) were no different from those in non-diabetic controls (216.6 +/− 17.3 ng/mL) (Fig. 4A)(Table 2). There was no correlation between serum HA levels and HbA1c in T1D patients (Fig. 4B). We did observed a slight decrease of HA with increasing BMI in T1D patients (Fig. 4C). There was no correlation between serum HA levels and duration of T1D (Fig. 4D).
Figure 4: Serum and muscle HA concentrations are not elevated in T1D patients.
A: Serum HA measurements in T1D and healthy control subjects. B: Relationship between serum HA and HbA1c. Mean serum HA values for each individual in the study are shown in conjunction with the HbA1c level measured for that given individual at the time of blood draw. C: BMI and (D) days from diagnosis are shown in correlation with HA. N = 1720 per group. Data in A represent mean +/− SEM, data in B-D are shown as regression plots. *P < 0.05 vs control by unpaired t test.
Table 2: Overview of HA accumulation at different diabetic sites in human and mouse.
The HA content in human and mouse T1D is elevated in pancreatic islets. In T2D HA in human and mouse is elevated in the serum and muscle. STZ treatment in mice increased the islet HA content, but did not significantly change the HA content in serum.
| HA content | T1D | T2D | STZ (toxin) |
|---|---|---|---|
| Serum | ↔ | ↑ | ↔ |
| Muscle | ↔ | ↑ | N/A |
| Islets | ↑ | N/A | ↑ |
Together these data mirror our findings in the serum of mice with autoimmune diabetes and these data strongly refute the hypothesis that serum HA reflects long-term hyperglycemia. In regards to HA at other sites, we previously reported that islet HA is increased in human subjects with T1D within 1 year of diagnosis with the disease but not in T1D subjects with long standing (> 1 year) disease [40]. Unfortunately, it was not possible to obtain muscle tissue from individuals with T1D.
HA levels are elevated in the serum and muscle of individuals with T2D
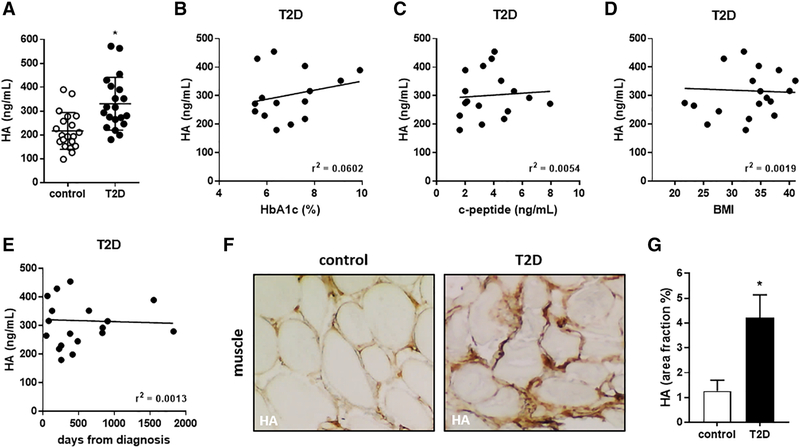
We observed that serum HA levels were significantly higher in T2D subjects (330.6 +/− 24.7 ng/mL) than in non-diabetic controls (216.6 +/− 17.3 ng/mL)(Fig. 5A). However, we found no correlation between HA levels and HbA1c in T2D patients (Fig. 5B). We also found no correlation between HA levels and C-peptide levels (Fig. 5C), BMI (Fig. 5D), or the duration of T2D (Fig. 5E) in these subjects.
Figure 5: Serum and muscle HA concentrations are elevated in T2D patients.
A: Serum HA measurements in T2D and healthy control subjects. B: Relationship between serum HA and HbA1c and (C) c-peptide. Mean serum HA values for each individual in the study are shown in conjunction with the HbA1c and c-peptide level measured for that given individual at the time of blood draw. D: BMI and (E) days from diagnosis are shown in correlation with HA. N = 17–20 per group. F: Representative images of muscle sections from healthy donors and T2D patients stained for HA, shown in brown. G: Quantification of HA muscle staining shown as % area fraction. N = 5 individuals per group. Data in A and G represent mean +/− SEM, data in B-E are shown as regression plots. *P < 0.05 vs control by unpaired t test.
We next sought to determine whether HA in muscle tissue was increased in T2D patients. To this end, we examined discarded surgical biopsy specimens collected from individuals with or without T2D following amputations of lower extremity tissues due to peripheral vascular disease. We found that the percentage area positive for HA was significantly increased in T2D patients relative to non-diabetic controls (Fig. 5G).
Together, these data indicate that both serum and muscle HA are elevated in human and mouse T2D (Table 2).
Discussion
We report three notable findings from these studies. First, HA is increased in the serum and muscles of humans with T2D as well as in the db/db mouse model of the disease. This corroborates previous reports linking increased tissue HA to T2D [45, 51, 55] and extends this finding to human skeletal muscle, the major tissue type involved in glucose uptake.
Second, we report that HA is not increased in the serum of individuals with T1D or in the DORmO mouse model of the disease. HA has been implicated in a number of autoimmune diseases like lupus [56], inflammatory bowel disease [57–60], autoimmune hepatitis [61] and others [62–64]. More recent studies show, that the local tissue environment is thought to contribute to immune regulation and the development of T1D [65–67], and that T1D is associated with substantial changes in the islet ECM and deposition of intra-islet HA [68]. Together with our previous report that autoimmune insulitis in T1D was associated with islet-specific deposition of HA [40, 41], the data presented here suggest that HA accumulates at the primary site of inflammation in T1D – the pancreatic islet – but not in distant sites, such as the serum or skeletal muscle. This pattern is consistent with T1D being a disease of local, isletspecific inflammation.
Third, we report that HA levels in muscle and tissue are not correlated with hyperglycemia, as measured by HbA1c. Serum HA levels in T2D are also not correlated with insulin levels, as measured by C-peptide, obesity, as measured by BMI, or with age, gender, or duration of disease.
It has been shown, that the total amount of serum HA in individuals with diseases like arthritis [69] and cancer [70, 71] is elevated [72]. We also know, that not only the total amount of HA is important but also its size. HA size has been called a natural biosensor for the state of tissue integrity [14]. For instance, it was discovered, that low molecular mass serum HA can be used to differentiate metastatic and non-metastatic breast cancer [73]. Also, the serum of healthy individuals usually contains low molecular mass between 100–300 kDa [74], and it is reported that even lower molecular mass HA, <50 kDa, is found in the serum of cancer patients [73]. Furthermore, it is known that HA breakdown products predominate during injury and inflammation [20]. These findings are in line with the elevated human serum HA data found in the inflammatory setting of T2D.
Together, these data suggest that serum and tissue HA may be elevated in diabetes in response to inflammation rather than hyperglycemia. In light of this, serum HA levels may have value as a potential biomarker for systemic inflammation in T2D independent of glycemic control.
We and others have previously reported that a great diversity of inflammatory factors, including cytokines and microbial products, drive HA synthesis [8, 9, 75]. The specific factors that promote HA synthesis in T2D and T1D are unknown but will be important to identify given reports linking HA and its receptors in the pathogenesis of these diseases [46, 47, 76].
In conclusion, these data support the use of circulating HA as a biomarker in T2D and suggest that its elevation may reflect inflammatory aspects of the disease that are distinct from glycemic control, insulin production, or BMI.
Methods
Human tissue donors and tissue procurement
Serum samples from 20, auto-antibody positive, T1D patients aged between 20 and 45 years, with disease duration of less than 1 year were collected. C-peptide data were not available from those T1D patients. Serum samples were also collected from 20 T2D patients aged between 20 and 45 years, and 20 age-matched autoantibody-negative healthy individuals. These samples were obtained with informed consent, under the auspices of a research protocol approved by the Benaroya Research Institute institutional review board. Clinical characteristics of donors are presented in Table 1.
Human muscle tissues were obtained from de-identified, discarded surgical specimens collected from T2D subjects or controls. These were provided through the Stanford Pathology Department in the form of formalin-fixed, paraffin-embedded histologic specimens. This study was carried out with the approval of the Institutional Review Board of Stanford University.
Mice
All animals were bred and maintained under specific pathogen-free conditions, with free access to food and water, in the vivarium at Stanford University. DO11.10 transgenic mice were purchased from The Jackson Laboratory (Bar Harbor, ME) and bred with BalbC mice expressing RIPmOVA (available at the Benaroya Research Institute) to generate the DORmO double-transgenic mice. Both C57Bl6 mice and Leptin receptor-deficient, db/db mice were purchased from The Jackson Laboratory (Bar Harbor, ME). All experiments and animal use procedures were approved by the Animal Care & Use Committee at Stanford University Medical School.
Weight and diabetes monitoring were performed on these mice. DORmO and db/db mice were weighed weekly as well as bled via the tail tip for the determination of their blood glucose level (BG) using a Contour blood glucose meter and blood glucose monitoring strips (Bayer Healthcare, Tarrytown, NY). When two consecutive blood glucose readings of 250 mg/dL were recorded, animals were considered diabetic.
C57Bl6 mice were treated two times with STZ at 200 mg/kg intraperitoneally. STZ was administered within 10 min of its dissolution. Mice in the untreated control group received citrate buffer (vehicle) alone. Blood glucose in those experimental groups was monitored daily. Mice were sacrificed following the development of hyperglycemia.
Serum, pancreas and skeletal muscle samples were collected. Serum samples were collected through cardiac puncture from deceased mice immediately after euthanization with CO2. Murine skeletal muscle was isolated from the quadriceps tissue and immediately transferred into methyl carnoys (MC) fixation. Tissue for paraffin embedding was processed using a Leica ASP300 Tissue Processor (Leica Microsystems Inc., Buffalo Grove, IL).
Measurement of serum levels of HA
Both human as well as murine serum samples were thawed and subsequently assayed for HA levels using a modified HA Enzyme-Linked Immunosorbent Assay (ELISA) [77]. Each sample was analyzed in triplicate with a mean value obtained for each individual.
Histologic staining of skeletal muscle and pancreas tissues for HA
For both human and murine muscle and murine pancreas tissue, 5 μm thick sections were cut on a Leica RM 2255 Microtomes (Leica Microsystems Inc.). All staining steps were performed on a Leica Bond Max™ automated immune histochemistry (IHC) stainer (Leica Microsystems Inc.). For HA affinity histochemistry (AFC) the Bond Intense R Detection kit, a streptavidin-horse radish peroxidase (HRP) system, (Leica Microsystems, Inc.) was used with 4 μg/mL biotinylated-HABP in 0.1 % BSA-PBS as the primary. The Bond Polymer Detection Kit was used for all other immunohistochemistry. This detection kit contains a goat anti-rabbit conjugated to polymeric HRP and a rabbit anti-mouse post primary reagent for use with mouse primaries.
All images were visualized using a Leica DMIRB inverted fluorescence microscope equipped with a Pursuit 4-megapixel cooled color/monochrome charge-coupled device camera (Diagnostic Instruments, Sterling Heights, MI). Images were acquired using the Spot Pursuit camera and Spot Advance Software (SPOT Imaging Solutions; Diagnostic Instruments). Image analysis was performed using Image J (NIH), as described previously [40].
Biochemical Quantification of HA in skeletal muscle
Tissues were first lyophilized and weighed, then digested with proteinase K (250 μg/mL) in 100 mM ammonium acetate pH 7.0 overnight at 60°C. After digestion, the enzyme was inactivated by heating to 100°C for 20 minutes. Total amount of HA was determined by a modified competitive ELISA in which the samples to be assayed were first mixed with biotinylated HA-binding protein (b-HABP) and then added to HA-coated microtiter plates, the final signal being inversely proportional to the level of hyaluronan added to the biotinylated HABP [78].
Statistical analysis
Data are expressed as means +/− SEM of n independent measurements. Significance of the difference between the means of two or three groups of data was evaluated using the Mann-Whitney U test or one-way Anova, respectively. The statistical significance of differences in serum hyaluronan levels between T1D, T2D and healthy donors was calculated by the Kruskal-Wallis test. Correlation analysis was performed using the non-parametric Spearman correlation test. A p value less than <0.05 was considered statistically significant.
Highlights.
Hyaluronan (HA) is an extracellular matrix polymer that has been implicated in the pathogenesis of both type 1 diabetes (T1D) and type 2 diabetes (T2D). In T1D, HA accumulates at sites of autoimmune insulitis and has been implicated in local immune dysregulation. In T2D, HA is increased in multiple tissues and has been linked to insulin resistance. In vitro, hyperglycemia is one pro-inflammatory factor that drives HA synthesis by multiple cell types raising the possibility that increased tissue HA in these diseases is a consequence of poor glycemic control. We have asked how glycemic control and T1D and T2D status impact tissue deposition of HA. We report that HA is increased in the serum and muscles in T2D but not in T1D. We find no relationship between serum HA and glycemic control, insulin production, or BMI. Together these results suggest that inflammatory factors rather than hyperglycemia drive local HA accumulation in diabetes. These data support the use of circulating HA as a biomarker in T2D and suggest that its elevation occurs independently of glycemic control, insulin production, or BMI.
Acknowledgements
We would like to thank Christina Chan of the Matrix Biology Program, BRI, for her technical help. We also thank members of the BRI Diabetes Clinical Research Program who collected samples for these studies, including Carla J. Greenbaum, Marli McCulloch-Olson, Mary Ramey, Kevin St. Jacques, Kristy Meyer, Daxa Sabhaya, Katrina Dziubkiewicz, Heather Vendettuoli, and Jani Klein.
Funding
This work was supported in part by the Deutsche Forschungsgemeinschaft (DFG) NA 965/2–1 to NN and KA 3441/1–1 to GK; and National Institutes of Health grants R01 DK096087–01, R01 HL113294–01A1, and U01 AI101984 to PLB. This work was also supported by grants from the JDRF 3-PDF-2014–224-A-N to NN and 1-SRA-2018–518-S-B Innovation Award to PLB and by grants from the Harrington Institute, Stanford SPARK, the Stanford Child Health Research Institute all to PLB, a grant from the Larry L. Hillblom Foundation to VGS, and by a grant from the Stanford Diabetes Research Center to NN.
Abbreviations
- HA
hyaluronan
- ECM
extracellular matrix
- JDRF
Juvenile Diabetes Research Foundation
- nPOD
National Pancreatic Organ Donor
- STZ
streptozotozin
- T1D
Type 1 Diabetes
- T2D
Type 2 Diabetes
Footnotes
Declaration of interest
PLB and NN are co-founders of Hyalos Therapeutics, a company developing novel small molecules to inhibit HA synthesis.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- [1].Jiang D, Liang J, Noble PW, Hyaluronan as an immune regulator in human diseases, Physiol Rev 91(1) (2011) 221–64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [2].Knudson W, Ishizuka S, Terabe K, Askew EB, Knudson CB, The pericellular hyaluronan of articular chondrocytes, Matrix Biol (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- [3].Laurent TC, Laurent UB, Fraser JR, The structure and function of hyaluronan: An overview, Immunol Cell Biol 74(2) (1996) A1–7. [DOI] [PubMed] [Google Scholar]
- [4].Toole BP, Hyaluronan: from extracellular glue to pericellular cue, Nat Rev Cancer 4(7) (2004) 528–39. [DOI] [PubMed] [Google Scholar]
- [5].Day AJ, Prestwich GD, Hyaluronan-binding proteins: tying up the giant, J Biol Chem 277(7) (2002) 4585–8. [DOI] [PubMed] [Google Scholar]
- [6].Itano N, Atsumi F, Sawai T, Yamada Y, Miyaishi O, Senga T, Hamaguchi M, Kimata K, Abnormal accumulation of hyaluronan matrix diminishes contact inhibition of cell growth and promotes cell migration, Proc Natl Acad Sci U S A 99(6) (2002) 3609–14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [7].Bell TJ, Brand OJ, Morgan DJ, Salek-Ardakani S, Jagger C, Fujimori T, Cholewa L, Tilakaratna V, Ostling J, Thomas M, Day AJ, Snelgrove RJ, Hussell T, Defective lung function following influenza virus is due to prolonged, reversible hyaluronan synthesis, Matrix Biol (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- [8].Bollyky PL, Evanko SP, Wu RP, Potter-Perigo S, Long SA, Kinsella B, Reijonen H, Guebtner K, Teng B, Chan CK, Braun KR, Gebe JA, Nepom GT, Wight TN, Th1 cytokines promote T-cell binding to antigen-presenting cells via enhanced hyaluronan production and accumulation at the immune synapse, Cell Mol Immunol 7(3) (2010) 211–20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [9].Evanko SP, Potter-Perigo S, Bollyky PL, Nepom GT, Wight TN, Hyaluronan and versican in the control of human T-lymphocyte adhesion and migration, Matrix Biol 31(2) (2012) 90–100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [10].Lauer ME, Mukhopadhyay D, Fulop C, de la Motte CA, Majors AK, Hascall VC, Primary murine airway smooth muscle cells exposed to poly(I,C) or tunicamycin synthesize a leukocyte adhesive hyaluronan matrix, J Biol Chem 284(8) (2009) 5299–312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [11].Shakya S, Wang Y, Mack JA, Maytin EV, Hyperglycemia-Induced Changes in Hyaluronan Contribute to Impaired Skin Wound Healing in Diabetes: Review and Perspective, Int J Cell Biol 2015 (2015) 701738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [12].Wang A, Hascall VC, Hyperglycemia, intracellular hyaluronan synthesis, cyclin D3 and autophagy, Autophagy 5(6) (2009) 864–5. [DOI] [PubMed] [Google Scholar]
- [13].Homann S, Grandoch M, Kiene LS, Podsvyadek Y, Feldmann K, Rabausch B, Nagy N, Lehr S, Kretschmer I, Oberhuber A, Bollyky P, Fischer JW, Hyaluronan synthase 3 promotes plaque inflammation and atheroprogression, Matrix Biol 66 (2018) 67–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [14].Stern R, Jedrzejas MJ, Hyaluronidases: their genomics, structures, and mechanisms of action, Chem Rev 106(3) (2006) 818–39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [15].Jiang D, Liang J, Noble PW, Hyaluronan in tissue injury and repair, Annu Rev Cell Dev Biol 23 (2007) 435–61. [DOI] [PubMed] [Google Scholar]
- [16].Noble PW, Hyaluronan and its catabolic products in tissue injury and repair, Matrix Biol 21(1) (2002) 25–9. [DOI] [PubMed] [Google Scholar]
- [17].Ruppert SM, Hawn TR, Arrigoni A, Wight TN, Bollyky PL, Tissue integrity signals communicated by high-molecular weight hyaluronan and the resolution of inflammation, Immunol Res 58(2–3) (2014) 186–92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [18].Tammi MI, Oikari S, Pasonen-Seppanen S, Rilla K, Auvinen P, Tammi RH, Activated hyaluronan metabolism in the tumor matrix - Causes and consequences, Matrix Biol (2018). [DOI] [PubMed] [Google Scholar]
- [19].Heldin P, Lin CY, Kolliopoulos C, Chen YH, Skandalis SS, Regulation of hyaluronan biosynthesis and clinical impact of excessive hyaluronan production, Matrix Biol (2018). [DOI] [PubMed] [Google Scholar]
- [20].Bollyky PL, Falk BA, Wu RP, Buckner JH, Wight TN, Nepom GT, Intact extracellular matrix and the maintenance of immune tolerance: high molecular weight hyaluronan promotes persistence of induced CD4+CD25+ regulatory T cells, J Leukoc Biol 86(3) (2009) 567–72. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [21].Bollyky PL, Wu RP, Falk BA, Lord JD, Long SA, Preisinger A, Teng B, Holt GE, Standifer NE, Braun KR, Xie CF, Samuels PL, Vernon RB, Gebe JA, Wight TN, Nepom GT, ECM components guide IL-10 producing regulatory T-cell (TR1) induction from effector memory T-cell precursors, Proc Natl Acad Sci U S A 108(19) (2011) 7938–43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [22].de la Motte C, Nigro J, Vasanji A, Rho H, Kessler S, Bandyopadhyay S, Danese S, Fiocchi C, Stern R, Platelet-derived hyaluronidase 2 cleaves hyaluronan into fragments that trigger monocyte-mediated production of proinflammatory cytokines, Am J Pathol 174(6) (2009) 2254–64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [23].McKee CM, Penno MB, Cowman M, Burdick MD, Strieter RM, Bao C, Noble PW, Hyaluronan (HA) fragments induce chemokine gene expression in alveolar macrophages. The role of HA size and CD44, J Clin Invest 98(10) (1996) 2403–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [24].Scheibner KA, Lutz MA, Boodoo S, Fenton MJ, Powell JD, Horton MR, Hyaluronan fragments act as an endogenous danger signal by engaging TLR2, J Immunol 177(2) (2006) 1272–81. [DOI] [PubMed] [Google Scholar]
- [25].Jackson DG, Hyaluronan in the lymphatics: The key role of the hyaluronan receptor LYVE-1 in leucocyte trafficking, Matrix Biol (2018). [DOI] [PubMed] [Google Scholar]
- [26].Savani RC, Wang C, Yang B, Zhang S, Kinsella MG, Wight TN, Stern R, Nance DM, Turley EA, Migration of bovine aortic smooth muscle cells after wounding injury. The role of hyaluronan and RHAMM, J Clin Invest 95(3) (1995) 1158–68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [27].Johnson P, Ruffell B, CD44 and its role in inflammation and inflammatory diseases, Inflamm Allergy Drug Targets 8(3) (2009) 208–20. [DOI] [PubMed] [Google Scholar]
- [28].Termeer C, Benedix F, Sleeman J, Fieber C, Voith U, Ahrens T, Miyake K, Freudenberg M, Galanos C, Simon JC, Oligosaccharides of Hyaluronan activate dendritic cells via toll-like receptor 4, J Exp Med 195(1) (2002) 99–111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [29].Nagy N, Freudenberger T, Melchior-Becker A, Rock K, Ter Braak M, Jastrow H, Kinzig M, Lucke S, Suvorava T, Kojda G, Weber AA, Sorgel F, Levkau B, Ergun S, Fischer JW, Inhibition of hyaluronan synthesis accelerates murine atherosclerosis: novel insights into the role of hyaluronan synthesis, Circulation 122(22) (2010) 2313–22. [DOI] [PubMed] [Google Scholar]
- [30].Ayars AG, Altman LC, Potter-Perigo S, Radford K, Wight TN, Nair P, Sputum hyaluronan and versican in severe eosinophilic asthma, Int Arch Allergy Immunol 161(1) (2013) 65–73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [31].Back SA, Tuohy TM, Chen H, Wallingford N, Craig A, Struve J, Luo NL, Banine F, Liu Y, Chang A, Trapp BD, Bebo BF Jr., Rao MS, Sherman LS, Hyaluronan accumulates in demyelinated lesions and inhibits oligodendrocyte progenitor maturation, Nat Med 11(9) (2005) 966–72. [DOI] [PubMed] [Google Scholar]
- [32].Liang J, Jiang D, Jung Y, Xie T, Ingram J, Church T, Degan S, Leonard M, Kraft M, Noble PW, Role of hyaluronan and hyaluronan-binding proteins in human asthma, J Allergy Clin Immunol 128(2) (2011) 403–411 e3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [33].Yoshioka Y, Kozawa E, Urakawa H, Arai E, Futamura N, Zhuo L, Kimata K, Ishiguro N, Nishida Y, Suppression of hyaluronan synthesis alleviates inflammatory responses in murine arthritis and in human rheumatoid synovial fibroblasts, Arthritis Rheum 65(5) (2013) 1160–70. [DOI] [PubMed] [Google Scholar]
- [34].Orasan OH, Ciulei G, Cozma A, Sava M, Dumitrascu DL, Hyaluronic acid as a biomarker of fibrosis in chronic liver diseases of different etiologies, Clujul Med 89(1) (2016) 24–31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [35].de Vries EMG, Farkkila M, Milkiewicz P, Hov JR, Eksteen B, Thorburn D, Chazouilleres O, Pares A, Nygard S, Gilja OH, Wunsch E, Invernizzi P, Carbone M, Bernuzzi F, Boberg KM, Rosjo H, Rosenberg W, Beuers UH, Ponsioen CY, Karlsen TH, Vesterhus M, Enhanced liver fibrosis test predicts transplant-free survival in primary sclerosing cholangitis, a multi-centre study, Liver Int 37(10) (2017) 1554–1561. [DOI] [PubMed] [Google Scholar]
- [36].Bollyky PL, Bogdani M, Bollyky JB, Hull RL, Wight TN, The role of hyaluronan and the extracellular matrix in islet inflammation and immune regulation, Curr Diab Rep 12(5) (2012) 471–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [37].Hull RL, Johnson PY, Braun KR, Day AJ, Wight TN, Hyaluronan and hyaluronan binding proteins are normal components of mouse pancreatic islets and are differentially expressed by islet endocrine cell types, J Histochem Cytochem 60(10) (2012) 749–60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [38].Irving-Rodgers HF, Ziolkowski AF, Parish CR, Sado Y, Ninomiya Y, Simeonovic CJ, Rodgers RJ, Molecular composition of the peri-islet basement membrane in NOD mice: a barrier against destructive insulitis, Diabetologia 51(9) (2008) 1680–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [39].Korpos E, Kadri N, Kappelhoff R, Wegner J, Overall CM, Weber E, Holmberg D, Cardell S, Sorokin L, The peri-islet basement membrane, a barrier to infiltrating leukocytes in type 1 diabetes in mouse and human, Diabetes 62(2) (2013) 531–42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [40].Bogdani M, Johnson PY, Potter-Perigo S, Nagy N, Day AJ, Bollyky PL, Wight TN, Hyaluronan and hyaluronan-binding proteins accumulate in both human type 1 diabetic islets and lymphoid tissues and associate with inflammatory cells in insulitis, Diabetes 63(8) (2014) 2727–43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [41].Nagy N, Kaber G, Johnson PY, Gebe JA, Preisinger A, Falk BA, Sunkari VG, Gooden MD, Vernon RB, Bogdani M, Kuipers HF, Day AJ, Campbell DJ, Wight TN, Bollyky PL, Inhibition of hyaluronan synthesis restores immune tolerance during autoimmune insulitis, J Clin Invest 125(10) (2015) 3928–40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [42].Weiss L, Slavin S, Reich S, Cohen P, Shuster S, Stern R, Kaganovsky E, Okon E, Rubinstein AM, Naor D, Induction of resistance to diabetes in non-obese diabetic mice by targeting CD44 with a specific monoclonal antibody, Proc Natl Acad Sci U S A 97(1) (2000) 285–90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [43].Nagy N, de la Zerda A, Kaber G, Johnson PY, Hu KH, Kratochvil MJ, Yadava K, Zhao W, Cui Y, Navarro G, Annes JP, Wight TN, Heilshorn SC, Bollyky PL, Butte MJ, Hyaluronan content governs tissue stiffness in pancreatic islet inflammation, J Biol Chem 293(2) (2018) 567–578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [44].Kuipers HF, Nagy N, Ruppert SM, Sunkari VG, Marshall PL, Gebe JA, Ishak HD, Keswani SG, Bollyky J, Frymoyer AR, Wight TN, Steinman L, Bollyky PL, The pharmacokinetics and dosing of oral 4-methylumbelliferone for inhibition of hyaluronan synthesis in mice, Clin Exp Immunol 185(3) (2016) 372–81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [45].Kang L, Lantier L, Kennedy A, Bonner JS, Mayes WH, Bracy DP, Bookbinder LH, Hasty AH, Thompson CB, Wasserman DH, Hyaluronan accumulates with high-fat feeding and contributes to insulin resistance, Diabetes 62(6) (2013) 1888–96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [46].Kodama K, Horikoshi M, Toda K, Yamada S, Hara K, Irie J, Sirota M, Morgan AA, Chen R, Ohtsu H, Maeda S, Kadowaki T, Butte AJ, Expression-based genome-wide association study links the receptor CD44 in adipose tissue with type 2 diabetes, Proc Natl Acad Sci U S A 109(18) (2012) 7049–54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [47].Liu LF, Kodama K, Wei K, Tolentino LL, Choi O, Engleman EG, Butte AJ, McLaughlin T, The receptor CD44 is associated with systemic insulin resistance and proinflammatory macrophages in human adipose tissue, Diabetologia 58(7) (2015) 1579–86. [DOI] [PubMed] [Google Scholar]
- [48].Bowling FL, Rashid ST, Boulton AJ, Preventing and treating foot complications associated with diabetes mellitus, Nat Rev Endocrinol 11(10) (2015) 606–16. [DOI] [PubMed] [Google Scholar]
- [49].Dalferes ER Jr., Radhakrishnamurthy B, Crouch MS, Berenson GS, A study of connective tissue macromolecules in skin of mice with goldthioglucose-induced obesity, Proc Soc Exp Biol Med 148(3) (1975) 918–24. [DOI] [PubMed] [Google Scholar]
- [50].Dwyer TM, Banks SA, Alonso-Galicia M, Cockrell K, Carroll JF, Bigler SA, Hall JE, Distribution of renal medullary hyaluronan in lean and obese rabbits, Kidney Int 58(2) (2000) 721–9. [DOI] [PubMed] [Google Scholar]
- [51].Mine S, Okada Y, Kawahara C, Tabata T, Tanaka Y, Serum hyaluronan concentration as a marker of angiopathy in patients with diabetes mellitus, Endocr J 53(6) (2006) 761–6. [DOI] [PubMed] [Google Scholar]
- [52].Zhu Y, Crewe C, Scherer PE, Hyaluronan in adipose tissue: Beyond dermal filler and therapeutic carrier, Sci Transl Med 8(323) (2016) 323ps4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [53].Morita M, Yano S, Ishibashi Y, Nakata N, Kurioka S, Sugimoto T, Close relationship between serum hyaluronan levels and vascular function in patients with type 2 diabetes, Biomarkers 19(6) (2014) 493–7. [DOI] [PubMed] [Google Scholar]
- [54].Orland MJ, Permutt MA, Quantitative analysis of pancreatic proinsulin mRNA in genetically diabetic (db/db) mice, Diabetes 36(3) (1987) 341–7. [DOI] [PubMed] [Google Scholar]
- [55].Zhu Y, Kruglikov IL, Akgul Y, Scherer PE, Hyaluronan in adipogenesis, adipose tissue physiology and systemic metabolism, Matrix Biol (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- [56].Yung S, Chan TM, The Role of Hyaluronan and CD44 in the Pathogenesis of Lupus Nephritis, Autoimmune Dis 2012 (2012) 207190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [57].Ahrenstedt O, Knutson L, Hallgren R, Gerdin B, Increased luminal release of hyaluronan in uninvolved jejunum in active Crohn’s disease but not in inactive disease or in relatives, Digestion 52(1) (1992) 6–12. [DOI] [PubMed] [Google Scholar]
- [58].Soroosh A, Albeiroti S, West GA, Willard B, Fiocchi C, de la Motte CA, Crohn’s Disease Fibroblasts Overproduce the Novel Protein KIAA1199 to Create Proinflammatory Hyaluronan Fragments, Cell Mol Gastroenterol Hepatol 2(3) (2016) 358–368 e4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [59].Yamaguchi Y, Noda H, Okaniwa N, Adachi K, Shinmura T, Nakagawa S, Ebi M, Ogasawara N, Funaki Y, Zhuo L, Kimata K, Sasaki M, Kasugai K, Serum-Derived Hyaluronan Associated Protein Is a Novel Biomarker for Inflammatory Bowel Diseases, Digestion 95(2) (2017) 146–155. [DOI] [PubMed] [Google Scholar]
- [60].Petrey AC, de la Motte CA, Hyaluronan in inflammatory bowel disease: Cross-linking inflammation and coagulation, Matrix Biol (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- [61].Nishikawa H, Enomoto H, Iwata Y, Hasegawa K, Nakano C, Takata R, Nishimura T, Yoh K, Aizawa N, Sakai Y, Ikeda N, Takashima T, Iijima H, Nishiguchi S, Clinical significance of serum Wisteria floribunda agglutinin positive Mac-2-binding protein level and high-sensitivity C reactive protein concentration in autoimmune hepatitis, Hepatol Res 46(7) (2016) 613–21. [DOI] [PubMed] [Google Scholar]
- [62].Tishler M, Yaron I, Shirazi I, Yaron M, Salivary and serum hyaluronic acid concentrations in patients with Sjogren’s syndrome, Ann Rheum Dis 57(8) (1998) 506–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [63].Gianoukakis AG, Jennings TA, King CS, Sheehan CE, Hoa N, Heldin P, Smith TJ, Hyaluronan accumulation in thyroid tissue: evidence for contributions from epithelial cells and fibroblasts, Endocrinology 148(1) (2007) 54–62. [DOI] [PubMed] [Google Scholar]
- [64].Yoshizaki A, Iwata Y, Komura K, Ogawa F, Hara T, Muroi E, Takenaka M, Shimizu K, Hasegawa M, Fujimoto M, Tedder TF, Sato S, CD19 regulates skin and lung fibrosis via Toll-like receptor signaling in a model of bleomycin-induced scleroderma, Am J Pathol 172(6) (2008) 1650–63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [65].Glisic S, Ehlenbach S, Jailwala P, Waukau J, Jana S, Ghosh S, Inducible regulatory T cells (iTregs) from recent-onset type 1 diabetes subjects show increased in vitro suppression and higher ITCH levels compared with controls, Cell Tissue Res 339(3) (2010) 585–95. [DOI] [PubMed] [Google Scholar]
- [66].Putnam AL, Brusko TM, Lee MR, Liu W, Szot GL, Ghosh T, Atkinson MA, Bluestone JA, Expansion of human regulatory T-cells from patients with type 1 diabetes, Diabetes 58(3) (2009) 652–62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [67].Zhou X, Bailey-Bucktrout SL, Jeker LT, Penaranda C, Martinez-Llordella M, Ashby M, Nakayama M, Rosenthal W, Bluestone JA, Instability of the transcription factor Foxp3 leads to the generation of pathogenic memory T cells in vivo, Nat Immunol 10(9) (2009) 1000–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [68].Nagy N, Kuipers HF, Marshall PL, Wang E, Kaber G, Bollyky PL, Hyaluronan in immune dysregulation and autoimmune diseases, Matrix Biol (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- [69].Grigoreas GH, Anagnostides ST, Vynios DH, A solid-phase assay for the quantitative analysis of hyaluronic acid at the nanogram level, Anal Biochem 320(2) (2003) 179–84. [DOI] [PubMed] [Google Scholar]
- [70].Dahl IM, Laurent TC, Concentration of hyaluronan in the serum of untreated cancer patients with special reference to patients with mesothelioma, Cancer 62(2) (1988) 326–30. [DOI] [PubMed] [Google Scholar]
- [71].Oikari S, Kettunen T, Tiainen S, Hayrinen J, Masarwah A, Sudah M, Sutela A, Vanninen R, Tammi M, Auvinen P, UDP-sugar accumulation drives hyaluronan synthesis in breast cancer, Matrix Biol 67 (2018) 63–74. [DOI] [PubMed] [Google Scholar]
- [72].Tengblad A, Laurent UB, Lilja K, Cahill RN, Engstrom-Laurent A, Fraser JR, Hansson HE, Laurent TC, Concentration and relative molecular mass of hyaluronate in lymph and blood, Biochem J 236(2) (1986) 521–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [73].Wu M, Cao M, He Y, Liu Y, Yang C, Du Y, Wang W, Gao F, A novel role of low molecular weight hyaluronan in breast cancer metastasis, FASEB J 29(4) (2015) 1290–8. [DOI] [PubMed] [Google Scholar]
- [74].Sasaki Y, Uzuki M, Nohmi K, Kitagawa H, Kamataki A, Komagamine M, Murakami K, Sawai T, Quantitative measurement of serum hyaluronic acid molecular weight in rheumatoid arthritis patients and the role of hyaluronidase, Int J Rheum Dis 14(4) (2011) 313–9. [DOI] [PubMed] [Google Scholar]
- [75].Vigetti D, Genasetti A, Karousou E, Viola M, Moretto P, Clerici M, Deleonibus S, De Luca G, Hascall VC, Passi A, Proinflammatory cytokines induce hyaluronan synthesis and monocyte adhesion in human endothelial cells through hyaluronan synthase 2 (HAS2) and the nuclear factor-kappaB (NF-kappaB) pathway, J Biol Chem 285(32) (2010) 24639–45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [76].Kodama K, Toda K, Morinaga S, Yamada S, Butte AJ, Anti-CD44 antibody treatment lowers hyperglycemia and improves insulin resistance, adipose inflammation, and hepatic steatosis in diet-induced obese mice, Diabetes 64(3) (2015) 867–75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [77].Underhill CB, Hyaluronan is inversely correlated with the expression of CD44 in the dermal condensation of the embryonic hair follicle, J Invest Dermatol 101(6) (1993) 820–6. [DOI] [PubMed] [Google Scholar]
- [78].Wilkinson TS, Potter-Perigo S, Tsoi C, Altman LC, Wight TN, Pro- and anti-inflammatory factors cooperate to control hyaluronan synthesis in lung fibroblasts, Am J Respir Cell Mol Biol 31(1) (2004) 92–9. [DOI] [PubMed] [Google Scholar]