The role of semen in sexual transmission of human cytomegalovirus (HCMV) is currently unclear. This is surprising, as HCMV is frequently detected in this body fluid and infection is of high danger for neonates and pregnant women. In this study, we found that seminal plasma (SP) dose dependently inhibited HCMV infection. The infection inhibition was specific for HCMV, as other viruses, such as human immunodeficiency virus type 1 (HIV-1) and herpes simplex virus 2 (HSV-2), were not inhibited by SP. SP must contain a soluble, heat-resistant factor that limits attachment of HCMV particles to cells, probably by interaction with the trimeric glycoprotein complex gH/gL/gO. This novel virus-host interaction could possibly limit transmission of HCMV via semen during sexual intercourse.
KEYWORDS: HCMV, SEVI, attachment, gH/gL/gO, glycoprotein O, human cytomegalovirus, semen, seminal amyloids, sexual transmission
ABSTRACT
Human cytomegalovirus (HCMV) is the most frequent viral cause of congenital infections that can lead to severe birth defects. Although HCMV is frequently detected in semen and thus is potentially sexually transmitted, the role of semen in HCMV transmission is largely unclear. Here we describe that human seminal plasma (SP; the cell-free supernatant of semen) inhibits HCMV infection. The inhibition of HCMV infection was dose dependent and effective for different cell types, virus strains, and semen donors. This inhibitory effect was specific for HCMV, as herpes simplex virus 2 (HSV-2) and human immunodeficiency virus type 1 (HIV-1) infections were enhanced by SP. Mechanistically, SP inhibited infection by interfering with the attachment of virions to cells most likely via an interaction with the trimeric glycoprotein complex gH/gL/gO. Together, our findings suggest that semen contains a factor that potentially limits sexual transmission of HCMV.
IMPORTANCE The role of semen in sexual transmission of human cytomegalovirus (HCMV) is currently unclear. This is surprising, as HCMV is frequently detected in this body fluid and infection is of high danger for neonates and pregnant women. In this study, we found that seminal plasma (SP) dose dependently inhibited HCMV infection. The infection inhibition was specific for HCMV, as other viruses, such as human immunodeficiency virus type 1 (HIV-1) and herpes simplex virus 2 (HSV-2), were not inhibited by SP. SP must contain a soluble, heat-resistant factor that limits attachment of HCMV particles to cells, probably by interaction with the trimeric glycoprotein complex gH/gL/gO. This novel virus-host interaction could possibly limit transmission of HCMV via semen during sexual intercourse.
INTRODUCTION
Human betaherpesvirus 5, also known as human cytomegalovirus (HCMV), is an omnipresent pathogen with a seroprevalence from 30% to 90% in developed countries (1). It is mainly acquired during early childhood but also with the beginning of sexual activities (2, 3). HCMV infections have a considerable clinical relevance especially for immunocompromised patients and during pregnancy. Infection during pregnancy poses a high risk of in utero transmission to the fetus, especially for seronegative women, but also preexisting seropositivity does not protect from infection of the fetus (4). HCMV has a high birth prevalence (5, 6), from which approximately 10% of infected newborns are symptomatic (6, 7). Infection is transmitted via virus-containing body fluids (8–10). In agreement with this, HCMV and also other herpesviruses have been detected in urine, saliva, and semen (8, 11–14). Therefore, HCMV can potentially be transmitted during sexual intercourse via semen, in which the virus can be frequently detected and isolated (15–17). However, the role of semen as a vector for HCMV transmission and the risk of sexual transmission is currently unclear.
Numerous human pathogens, for example, HIV, are also present in semen (16) and are sexually transmitted. Several factors in semen may modulate viral infection and transmission. For example, seminal amyloids capture HIV virions and function as infection enhancers by promoting virus attachment to and fusion with target cells (18, 19). The best characterized of these amyloids is the semen-derived enhancer of virus infection (SEVI). Thus, seminal amyloids may play a major role in sexual transmission of HIV-1.
An enhancement of infection by SEVI has also been reported for herpes simplex virus 1 (HSV-1), HSV-2, and HCMV (20, 21), suggesting a similar mechanism of infection enhancement of these viruses. However, the role of amyloid fibrils in semen and the role of semen itself in HCMV infection have not been sufficiently explored. Here we report that human semen contains a heat-resistant factor that inhibits HCMV infection by interfering with virion attachment to cells through interaction with HCMV glycoprotein O (gO) or the trimeric glycoprotein complex gH/gL/gO. Our findings indicate the presence of a natural inhibitor of HCMV in semen, which might reduce or prevent HCMV transmission upon sexual intercourse.
RESULTS
Seminal plasma specifically inhibits HCMV infection.
The effect of seminal plasma (SP) on HCMV infection was investigated by infecting human foreskin fibroblasts (HFF) with cell-free virus of HCMV strain TB40/E in the presence of up to 5% SP. SP was generated by centrifugation of pooled, liquefied ejaculates derived from 10 individuals. To minimize cytotoxic effects of SP (18, 22, 23), the virus-SP mixtures were removed after 1 h of infection and replaced by fresh medium. Infection was determined 24 h postinfection (hpi) by detection of HCMV immediate early (IE) antigen-positive cells. SP inhibited HCMV infection in a dose-dependent manner, with a relative half-maximal inhibitory concentration (IC50) of 0.64% ± 0.09% (Fig. 1A and B) without affecting cell viability (Fig. 1C). The same SP pool enhanced infection of HFF with HSV-2 (Fig. 1E) and infection of TZM-bl cells with HIV-1 (Fig. 1D), consistent with previous reports (18, 24). Thus, SP possesses specific anti-HCMV properties.
FIG 1.
Effect of seminal plasma (SP) on HCMV, HIV-1, and HSV-2 infection. (A) HCMV (strain TB40/E, corresponding to an infection of about 20%) was incubated with the indicated concentrations of SP for 1 h at 37°C prior to infection of HFF with virus-SP mixtures for 1 h at 37°C. Infection was determined by indirect immunofluorescence staining for HCMV IE antigen 24 hpi. Blue, DAPI-positive cells; white, HCMV-positive cells. Scale bar is 200 μm. (B) Means ± SDs of relative infection from five individual infection experiments, each performed in triplicate. (C) Cytotoxicity of SP on HFF was controlled 24 h after incubation of cells with SP for 1 h at 37°C by using the MTT assay. (D and E) Means ± SDs of relative HIV-1 infection of TZM-bl cells (D) and HSV-2 infection of HFF (E) in the presence of SP from two individual experiments, each performed in triplicate. Controls (0% SP) were set to 100% and samples normalized accordingly. *, P < 0.01; **, P < 0.001; ***, P < 0.0001.
In our attempts to test inhibition of infection at higher SP concentrations, we found that heat treatment of SP at 95°C for 10 min (hSP95°C) markedly reduced its cytotoxicity (Fig. 2A), while it conserved its anti-HCMV activity (Fig. 2B) (SP IC50, 0.42% ± 0.41%; hSP95°C IC50, 0.35% ± 0.24%). Consistent with a dose-dependent inhibitory effect, HCMV infection was completely blocked at 50% hSP95°C using the same infection procedure as before (Fig. 2C). To evaluate whether the HCMV inhibitory activity of SP is dependent on the viral inocula, HFF were infected in the presence of up to 50% hSP95°C with increasing virus doses of 1×, 10×, and 100× (leading to infections of 20, 50, and 100%). At the lowest virus dose, 50% hSP95°C entirely abrogated infection (Fig. 2D). However, with increasing virus doses, the antiviral activity of SP was less pronounced, demonstrating that HCMV inhibition is affected by the amount of input virus.
FIG 2.
Effect of higher SP concentrations on HCMV infection. (A) Cytotoxicity of SP and heat-treated seminal plasma (hSP95°C) on HFF was investigated by the MTT assay. (B and C) HCMV (strain TB40/E, corresponding to an infection of about 20%) was incubated with SP and hSP95°C for 1 h at 37°C prior to infection of HFF with virus-SP mixtures for 1 h at 37°C. Infection was determined by indirect immunofluorescence staining for HCMV IE antigen 24 hpi. Controls (0% SP and 0% hSP95°C) were set to 100% and samples normalized accordingly. (D) Different virus doses (1×, 10×, and 100×, corresponding to infections of 20%, 50%, and 100%, respectively) of TB40/E were used in infection experiments with the indicated concentrations of hSP95°C. Given are means ± SDs from two individual experiments, each performed in triplicate. *, P < 0.01; **, P < 0.001; ***, P < 0.0001.
A recent publication reported an infection-enhancing effect of seminal plasma at a concentration of 0.1% (20). Following their infection protocol, we did not detect infection-enhancing effects in indirect immunofluorescence or Western blot experiments at 0.1% SP (Fig. 3). Again, an inhibition of infection could be observed at 1% SP. Interestingly, the inhibitory effect of SP was even more pronounced when cells were incubated with virus-SP mixtures overnight compared to 1 h. Altogether, our data show that SP efficiently inhibits HCMV infection already at low concentrations.
FIG 3.
SP does not enhance HCMV infection at 0.1%. (A) HCMV strain TB40/E was incubated with the indicated concentrations of SP for 1 h at 37°C prior to infection of HFF with virus-SP mixtures for 24 h at 37°C. Infection was determined by indirect immunofluorescence staining for HCMV IE antigen 24 hpi. Blue, DAPI-positive cells; white, HCMV-positive cells. Scale bar is 200 μm. (B and D) HFF were infected with virus-SP mixtures for 1 h and 24 h. Infection was analyzed by indirect immunofluorescence (B) and Western blot analysis (D). (C) HFF were incubated with the indicated concentrations of SP for 1 and 24 h at 37°C. Cell viability of HFF was determined by the MTT assay. Given are means ± SDs from two individual experiments, each performed in triplicate. Controls (0% SP) were set to 100% and samples normalized accordingly. *, P < 0.01; **, P < 0.001; ***, P < 0.0001.
Inhibition of HCMV infection by seminal plasma is independent of seminal amyloids.
To further examine the anti-HCMV activity of SP, we investigated the role of seminal amyloids in infection (18). It has recently been reported that SEVI enhances HCMV as well as HSV-1 and -2 infections in vitro (20, 21). First, we verified the infection-enhancing capacity of SEVI on HCMV in infection experiments. We could show that HCMV infections rates increase about 50% in the presence of 10 μg/ml of SEVI (Fig. 4A and B). Next, we tested the hypothesis that SP counteracts the infection-enhancing effect of SEVI. Therefore, synthetic SEVI was added to SP and incubated with HCMV prior to infection of HFF. SP inhibited HCMV infection to similar extents independent of the presence of additional SEVI (Fig. 4B), indicating neutralization of the HCMV-enhancing properties of SEVI by SP. To verify that SEVI or seminal amyloids play no role in HCMV infection in this context, SP devoid of seminal amyloids was generated by an incubation overnight at 37°C (hSP37°C), which destroys the naturally occurring amyloid fibrils (25). As for untreated SP, HCMV infection was strongly reduced, by more than 60%, at a concentration of 5% hSP37°C (Fig. 4C) (SP IC50, 0.36% ± 0.52%; hSP37°C IC50, 0.34% ± 0.91%). Infection experiments with HIV-1 and HSV-2 (Fig. 4D and E) confirmed that the overnight incubation at 37°C was sufficient to inactivate the infection-enhancing properties of SP. These results suggest that the anti-HCMV activity in semen overpowers the infection-enhancing activity of seminal amyloids. It also shows that SP harbors no antiviral activity against HIV-1 and HSV-2 at concentrations that do not affect cell viability.
FIG 4.
Effect of seminal amyloids on virus infection. (A to C) HCMV (strain TB40/E, corresponding to an infection of about 20%) was incubated with the indicated concentrations of semen-derived enhancer of virus infection (SEVI), SP, combinations of SEVI and SP, and hSP37°C (heat-treated SP devoid of seminal amyloids) for 1 h at 37°C prior to infection of HFF with mixtures for 1 h at 37°C. Infection was determined by indirect immunofluorescence staining for HCMV IE antigen 24 hpi. (A) Blue, DAPI-positive cells; white, HCMV-positive cells. Scale bar is 200 μm. (B and C) Means ± SDs from two individual experiments, each performed in triplicate. (D and E) Means ± SDs of relative infection for HIV-1 infection of TZM-bl cells (D) and for HSV-2 infection of HFF (E) in the presence of the indicated concentrations of SP or hSP37°C from two individual experiments that were performed in triplicate. Infection of the controls (0% SP) was set to 100% and samples were normalized accordingly. *, P < 0.01; **, P < 0.001; ***, P < 0.0001.
The inhibition of HCMV infection by seminal plasma is effective for different cell types, virus strains, and semen donors.
HCMV can infect different cell types in the human host (26). Infection of these various cell types, e.g., fibroblasts and endothelial cells, is facilitated by different entry mechanisms (direct fusion at the plasma membrane or receptor-mediated endocytosis) and involves different glycoprotein complexes (27). To investigate whether the inhibitory property of SP on HCMV is a general feature and independent of entry mechanisms, the endothelial cell line HEC-LTT was included in infection experiments. In addition, another endotheliotropic HCMV strain (VHL/E) and three nonendotheliotropic strains (AD169, Towne, and Toledo) were used to exclude virus strain-specific effects (Fig. 5). Infection of both fibroblasts and endothelial cells was inhibited in the presence of SP for all tested endotheliotropic and nonendotheliotropic strains. Interestingly, higher SP concentrations were needed for similar infection inhibition in endothelial cells than for fibroblasts (Table 1).
FIG 5.
SP inhibits infection of different HCMV strains and cell types. HCMV strains TB40/E, VHL/E, AD169, Towne, and Toledo (corresponding to an infection of about 20%) were incubated with indicated concentrations of SP for 1 h at 37°C prior to infection of HFF and endothelial cells (HEC-LTT) with mixtures for 1 h at 37°C. Infection was determined by indirect immunofluorescence staining for HCMV IE antigen 24 hpi. (A) Blue, DAPI-positive cells; white, HCMV-positive cells. Scale bar is 200 μm. (B and C) Means ± SDs of relative infection from two individual experiments that were performed in triplicate. Controls (0% SP) were set to 100% and samples normalized accordingly. *, P < 0.01; **, P < 0.001; ***, P < 0.0001.
TABLE 1.
IC50s of seminal plasma in infections of fibroblasts and endothelial cells with different HCMV strains
| Cell type | HCMV strain | IC50 (%) |
|---|---|---|
| Fibroblasts | TB40/E | 0.15 ± 0.02 |
| VHL/E | 0.64 ± 0.43 | |
| AD169 | 0.41 ± 0.25 | |
| Towne | 0.51 ± 0.16 | |
| Toledo | 0.38 ± 0.09 | |
| Endothelial cells | TB40/E | 4.06 ± 0.73 |
| VHL/E | 4.74 ± 0.78 |
Furthermore, fresh ejaculates were tested to exclude that the freeze-thaw cycle to aliquot and store SP is responsible for its anti-HCMV activity. SP prepared from fresh ejaculates of six donors inhibited HCMV infection dose dependently (Fig. 6). Although SP from the different donors varied slightly in their inhibitory capacity, HCMV infection was reduced by more than 80% at 5% SP from all donors and the inhibitory activity seemed to be unrelated to the HCMV status of the donor (Table 2). Thus, SP from fresh ejaculates of all tested donors inhibited HCMV infection, corroborating our results obtained with frozen SP.
FIG 6.
Inhibition of HCMV is a general property of human semen. HCMV (strain TB40/E, corresponding to an infection of about 50%) was incubated for 1 h at 37°C with the indicated concentrations of SP from fresh ejaculates of six donors prior to infection of HFF with mixtures for 1 h at 37°C. Infection was determined by indirect immunofluorescence staining for HCMV IE antigen 24 hpi. Given are means ± SDs of relative infection from triplicate infection. Infection of the controls (0% SP) was set to 100% and samples were normalized accordingly. *, P < 0.01; **, P < 0.001; ***, P < 0.0001.
TABLE 2.
IC50s of seminal plasma from fresh ejaculates of individual donors in infections of fibroblasts with HCMV strain TB40/E
| Donor (HCMV status) | IC50 (%) |
|---|---|
| 1 (NDa ) | 0.79 ± 0.09 |
| 2 (ND) | 0.81 ± 0.25 |
| 3 (HCMV negative) | 1.36 ± 0.02 |
| 4 (ND) | 1.30 ± 0.02 |
| 5 (HCMV positive) | 0.70 ± 0.08 |
| 6 (HCMV negative) | 0.34 ± 0.18 |
ND, not determined.
Seminal plasma blocks HCMV attachment.
To clarify the mechanism of HCMV infection inhibition, we first tested whether the antiviral activity of SP is directed against the viral particle or the cell. HFF were incubated with up to 5% of SP or hSP95°C for 1 h, washed, and subsequently infected with HCMV. Cells incubated prior their infection with 5% SP and 5% hSP95°C exhibited similar HCMV infection as control cells (Fig. 7A). This mostly excludes a direct effect of SP on cells by altering their susceptibility to HCMV infection or permanent blocking of a cellular entry receptor. To test whether the inhibitory effect of SP is directed against virus particles, HCMV virions were incubated with either 5% SP, hSP95°C, or medium as a control for 1 h at 37°C. Afterwards, virus particles were pelleted by ultracentrifugation for 2 h at 100,000× g, resuspended in medium, and subsequently used for infection of HFF. Similar infections of cells were measured for virus particles treated with 5% SP (86.4% ± 3.6%), 5% hSP95°C (88.6% ± 3.3%), and medium (93.1% ± 1.6%) (Fig. 7B). This suggests that the inhibitory component of SP did not directly inactivate virus particles and did not pellet together with them. Thus, the antiviral factor remained in the supernatant of SP. To verify this, SP and hSP95°C were subjected directly to ultracentrifugation and tested for infection inhibition. As expected, supernatants of SP as well as of hSP95°C were still able to inhibit infection of HFF with HCMV after ultracentrifugation (Fig. 7C). Altogether, this excludes an irreversible damage of HCMV virus particles by SP and rather indicates a transient interaction with either the cell or the virus.
FIG 7.

The anti-HCMV factor in SP does not inactivate virus particles irreversibly. (A) HFF were pretreated with indicated concentrations of SP and hSP95°C (heat-treated SP) for 1 h at 37°C. Afterwards, media were removed and cells were washed and subsequently infected with TB40/E for 1 h at 37°C. (B) Cell-free supernatants of TB40/E were incubated in either medium or 5% SP and hSP95°C for 1 h at 37°C. Then viral particles were pelleted by ultracentrifugation for 2 h at 100,000 × g and 4°C. Virus pellets were resuspended in medium and used for infection experiments with HFF. (C) SP or hSP95°C in medium was processed via ultracentrifugation (100,000 × g, 2 h, and 4°C). Then supernatants were used for infection experiments with TB40/E. Cells were fixed 24 hpi and stained for HCMV IE antigen by indirect immunofluorescence. Given are means ± SDs from two individual experiments that were performed in triplicate. Infection of the controls was set to 100% and samples were normalized accordingly. *, P < 0.01; **, P < 0.001; ***, P < 0.0001.
HCMV infection is a multistep process that is initiated by the attachment of virus particles to the cell surface. To test if this step is affected by SP, we performed attachment assays at 4°C, which allows binding of particles to cells but prevents subsequent steps of virus entry (28). A strong reduction of attached virus particles per cell, about 70%, could be observed in the presence of 5% of SP and hSP95°C (Fig. 8). Heparin was used as a control, as it blocks attachment of herpesvirus particles to cell surfaces (28). Unlike with heparin, SP and hSP95°C had no effect on attachment when added either prior to or after the virus incubation at 4°C (Fig. 8B). This suggests an impaired attachment of HCMV particles in the presence of SP that appears to be different from the mode of action of heparin.
FIG 8.

SP inhibits attachment of HCMV virions to cell membranes. (A) Attachment of viral particles to HFF was assessed by indirect immunofluorescence staining with antibodies against the HCMV tegument protein pp150 after incubation of virus particles in the presence of SP for 1 h at 4°C. (Left) Bright field; (right) enhanced GFP (EGFP) channel (green, HCMV particles). Scale bar is 5 μm. (B) Black, quantification of attachment as in panel A with two additional conditions. Red, HFF were treated under the indicated conditions for 1 h and washed before TB40/E was added for attachment for 1 h. Green, TB40/E was added to HFF for 1 h, and cells were washed and incubated for 1 h under the indicated conditions. Given are relative numbers of attached HCMV particles per cell from 100 cells from two individual experiments. The controls (medium) were set to 100% and samples normalized accordingly. *, P < 0.01; **, P < 0.001; ***, P < 0.0001.
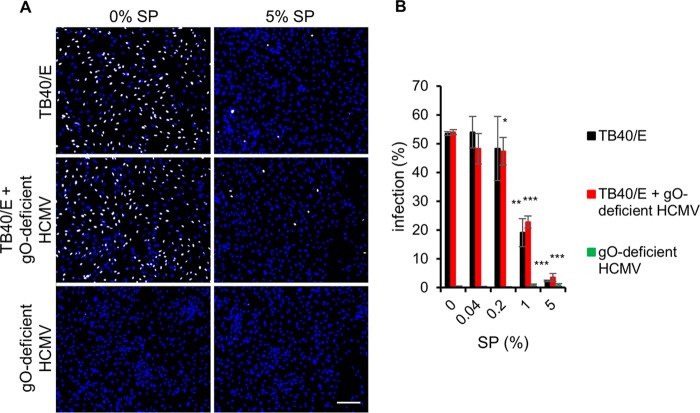
Finally, we sought to identify viral targets of the anti-HCMV compound of SP. One potential candidate was the betaherpesvirus-specific glycoprotein O (gO) and the trimeric glycoprotein complex gH/gL/gO of HCMV, because it is involved in HCMV attachment and entry (29, 30). To address this, a gO-deficient mutant of HCMV, lacking the trimeric complex, was used in infection assays. Concentrated cell-free virus was used, since deletion of gO results in an impaired infectivity of virions (29). Furthermore, infection assays were performed on endothelial cells due to their higher susceptibility to a gO-deficient virus. Supporting our hypothesis, SP could not inhibit infection of endothelial cells with the gO-deficient mutant. In contrast, enhanced infection was measured (Fig. 9A and B). Amyloid fibrils in SP were found to be responsible for the infection enhancement of the gO-deficient mutant, because their disruption in hSP37°C led to no enhanced infection (Fig. 9C), whereas the addition of synthetic SEVI to hSP37°C restored the enhancing effect (Fig. 9D). Importantly, inhibition of TB40/E infections was not affected by addition of gO-deficient virus, showing that the inhibitory capacity of SP was not altered (Fig. 10). Infection of HFF with mixtures of TB40/E and the gO-deficient virus can only result from TB40/E because of the requirement of the trimeric complex for fibroblast infection (29). Together, these results indicate that HCMV gO or the trimeric gH/gL/gO complex is targeted by the anti-HCMV compound in SP.
FIG 9.

Infection of cells with a gO-deficient HCMV mutant is enhanced by SP. Cell-free virus particles of the gO-deficient mutant TB40-BAC4-UL74stop were incubated with the indicated concentrations of SP, hSP37°C (heat-treated SP devoid of seminal amyloids), and SEVI for 1 h at 37°C prior to infection of HEC-LTT with mixtures for 1 h at 37°C. Infection was determined by indirect immunofluorescence staining for HCMV IE antigen 24 hpi. (A) Blue, DAPI-positive cells; white, HCMV-positive cells. Scale bar is 200 μm. (B to D) Means ± SDs of relative infection from at least two individual experiments that were performed in triplicates. Infection of the controls was set to 100% and the samples were normalized accordingly. *, P < 0.01; **, P < 0.001; ***, P < 0.0001.
FIG 10.
Ultracentrifugation of a gO-deficient HCMV does not saturate the anti-HCMV factor in SP. TB40/E, concentrated gO-deficient HCMV and a combination of both were incubated with the indicated concentrations of SP for 1 h at 37°C prior to infection of HFF with mixtures for 1 h at 37°C. Infection was determined by indirect immunofluorescence staining for HCMV IE antigen 24 hpi. (A) Blue, DAPI-positive cells; white, HCMV-positive cells. Scale bar is 200 μm. (B) Means ± SDs of infection from triplicates. *, P < 0.01; **, P < 0.001; ***, P < 0.0001.
DISCUSSION
We here show that human semen contains anti-HCMV activity. SP efficiently inhibited HCMV infection at concentrations of only 1% to 5% (vol/vol). Infection inhibition was observed with pooled SP derived from different donors as well as with SP from fresh ejaculates from single donors (HCMV positive and HCMV negative), showing that the inhibitory effect is a general property of human semen. The previously reported infection enhancement of SP could not be verified (20). The reason for the infection enhancement of HCMV by SP at a concentration of 0.1% as reported by Tang et al. is currently unclear, because 0.1% SP repeatedly did not enhance HCMV infection in our experiments. In contrast to Tang et al., we have tested several pooled SP preparations as well as fresh SP of single donors at various concentrations, and we never detected any enhancing activity on five different HCMV strains on fibroblasts and endothelial cells. Thus, our findings suggest that semen harbors a factor that inhibits infection and therefore likely limits the probability of HCMV transmission during sexual intercourse.
HCMV is frequently detected in human semen from various population groups, such as HIV-infected individuals (31, 32). HCMV is also detected in semen of healthy individuals lacking signs of HCMV infection (12, 33, 34). Infectious virus can be directly isolated from semen (15), altogether indicating that semen may be a potential vector. While median HCMV genome copies in semen samples differ greatly among various studies, the studies have in common that detection of HCMV genomes via PCR is more frequent than isolation of virus via tissue culture (35–37). This is in agreement with an inhibitory property of SP against HCMV, which could explain the low infectivity in semen. Furthermore, our data suggest that the viral load might be an important determinant for the infectivity of semen, because infection at higher virus doses could not be completely blocked by SP. However, there are only limited data available on HCMV genome copies in semen and infectivity measured by virus isolation in cell culture. An additional difficulty is the high cell culture toxicity of semen. Known factors associated with semen cytotoxicity are amine oxidases oxidizing spermine (23, 38). We found that heat treatment of SP for 10 min at 95°C minimizes the cell culture cytotoxicity. Heat treatment likely denatures and thereby inactivates, e.g., amine oxidases that contribute to the cytotoxicity of semen. This treatment, however, did not affect its inhibitory activity against HCMV, and we found that SP concentrations of 50% completely inhibited HCMV infection depending on the virus dose. Consistent with our data, Eggert-Kruse et al., who examined male and female partners simultaneously (33), suggest that transmission of HCMV by sexual contact is not a frequent event. However, higher HCMV loads, which are often found in HIV patients (32, 39, 40), could still result in transmission. In addition, our data suggest that virus particles are not irreversibly inactivated by human semen and dilution of the body fluid after ejaculation might allow infection of target cells. In the light of the high frequency of HCMV in semen and the severe sequelae of congenital HCMV infection, the role of semen in sexual transmission of HCMV deserves further investigation to clarify viral load and actual infectivity and evaluate the risk of HCMV transmission via semen.
Although seminal amyloids like SEVI are known to harbor infection-enhancing properties (18, 19, 24), the inhibitory effect of SP on HCMV could not be overcome by addition of SEVI to SP. This suggests that endogenous seminal amyloids are ineffective in promoting infection of HCMV in SP. Furthermore, SP, depleted of infection-enhancing amyloid fibrils, failed to enhance HIV-1 and HSV-2 infection but retained its anti-HCMV activity. This indicates an HCMV-specific antiviral effect of semen. Mechanistically, SP inhibited an early step of HCMV infection. The anti-HCMV activity in SP could be narrowed down to a heat-resistant factor that reversibly blocks attachment of HCMV particles to cells. This seminal factor presumably interferes with the interaction of HCMV gO and/or the trimeric glycoprotein complex gH/gL/gO and its receptor, because infection of cells with a gO-deficient mutant was not inhibited by SP. Interestingly, an infection-enhancing effect of endogenous seminal amyloids of SP was observed in the absence of gO. This demonstrates that seminal amyloids are able to enhance HCMV infection in the physiological context but that they are neutralized due to the inhibitory activity of SP when gO is present. An antiviral activity of SP against betaherpesvirus-specific gO not only explains the specificity for HCMV but also explains why inhibition of HCMV infection is more efficient on fibroblasts than on endothelial cells. It is generally accepted that the trimeric gH/gL/gO complex mediates cell-type-specific entry into fibroblasts, while the pentameric gH/gL/UL128-131 complex is required for entry into endothelial cells (27, 41). However, the trimeric complex is involved in HCMV entry of all cell types, most likely by promoting fusion with the cell membrane (42). The use of different glycoprotein complexes for cell-type-specific entry could explain the higher IC50s of HCMV infection of endothelial cells than for fibroblasts. The inhibition of HCMV entry into endothelial cells by interfering with gO functions or the trimeric complex is not a new phenomenon and has been reported previously (29). The cellular receptor for the HCMV trimeric complex on fibroblasts is the platelet-derived growth factor receptor α (PDGFR-α) (29, 30). A soluble derivative of PDGFR-α inhibits both attachment to and fusion with cells, most likely via direct binding to gO (29). Although PDGFR-α is not expressed on endothelial cells, infection of endothelial cells is inhibited by soluble PDGFR-α. Therefore, gO seems to be a key player of HCMV entry, which is targeted by a natural component of semen.
Inhibition of infection by semen is apparently not restricted to HCMV, as an antiviral activity of semen has recently been reported against the flaviviruses Zika, dengue, and West Nile viruses (43). Müller et al. identified extracellular vesicles (EVs) of semen to be likely responsible for Zika virus inhibition. Whether EVs also account for the anti-HCMV effect of SP needs to be elucidated. In the light of a specific inhibition of HCMV and not of other herpesviruses (HSV-2), it is hardly probable that EVs are the anti-HCMV factor in SP. Apart from EVs, small, heat-resistant peptides, as well as lipids, are potential candidates that could be responsible for the specific inhibition of HCMV infection and will be tested in future studies.
In summary, our study highlights a previously unknown antiviral activity of semen interfering with HCMV attachment by gO or the trimeric complex and thereby inhibiting infection. The identification of the SP component with anti-HCMV activity therefore not only may reveal how semen inhibits HCMV infection but also could give new insights into HCMV entry processes and ways to prevent infection.
MATERIALS AND METHODS
Cell culture and virus strains.
Primary human foreskin fibroblasts (HFF) and the endothelial cell line HEC-LTT, derived from primary human vein endothelial cells, were used as described previously (44, 45). The human TZM-bl HIV reporter cell line was obtained through the NIH AIDS Research and Reference Reagent Program (ARRRP) and used as described elsewhere (46). Two low-passage-number, endotheliotropic HCMV strains (TB40/E [47] and VHL/E [48]), three nonendotheliotropic laboratory-adapted strains (AD169, Towne, and Toledo), and a gO-deficient mutant HCMV (TB40-BAC4-UL74stop) (49) (kindly provided by K. Laib Sampaio and C. Stegmann) were used for HCMV infections. HSV-2 infections were performed with a virus expressing the green fluorescent protein (GFP; kindly provided by P. Spear) (50). To generate cell-free virus stocks, supernatants of infected HFF were collected 48 h postinfection (hpi) for HSV-2 and 120 to 168 hpi for HCMV, cleared from cell debris by centrifugation at 1,000 rpm for 10 min, and stored in aliquots at −80°C before use. The R5-tropic HIV-1 NL4-3 92TH014.12 (51) was used for infection experiments with HIV-1. HIV-1 stocks were generated by transient transfection of HEK293T cells as described previously (52).
Immunofluorescence microscopy.
Infected cells were fixed with 4% paraformaldehyde in phosphate-buffered saline (PBS) for 10 min at 4°C. For detection of HCMV antigens, cells were permeabilized with 0.1% Triton X-100 for 5 min followed by incubation in blocking buffer (PBS, 1% bovine serum albumin [BSA], 5% fetal calf serum [FCS]) for 30 min at room temperature (RT). Primary monoclonal antibodies (MAbs; directed against immediate early [IE] 1 protein [Mab63-27; mouse IgG2a] or tegument protein pp150 [Mab36-14; mouse IgG2b]) were diluted in PBS–1%BSA and incubated for 2 h at RT. Nuclei were stained with 4′,6-diamidino-2-phenylindole (DAPI; Roche).
Infection assays.
HFF and HEC-LTT cells were used for HCMV infection assays, whereas only HFF were used for infections with HSV-2. Infection assays were performed in the presence of 100 μg/ml of gentamicin. A total of 1.7 × 104 cells/well were seeded in 96-well flat-bottom microtiter plates 1 day prior to infection. Semen was obtained from the Kinderwunsch-Zentrum Ulm, a fertility center in Ulm, Germany, or from healthy volunteers at Ulm University (ethics vote 89/16; 200/17). Ejaculates were allowed to liquefy for 30 min at RT and then either used directly or stored at −80°C as pooled semen samples. Seminal plasma (SP) was obtained by removal of spermatocytes and other cells through centrifugation for 10 min at 10,000 rpm. Heat treatment of SP was performed by incubation of SP overnight at 37°C to obtain hSP37°C or for 10 min at 95°C to generate hSP95°C. SEVI stock solution was generated as described previously (18) and diluted in PBS. Cell-free virus dilutions of HCMV (corresponding to infections of 20%, 50%, or 100%), the gO-deficient HCMV (concentrated by ultracentrifugation for 70 min at 80,000 × g and 4°C), and HSV-2 (corresponding to 104 PFU/ml) were incubated with different concentrations of either SEVI or SP in serum-free minimal essential medium (MEM) for 1 h at 37°C prior to infection of cells with these mixtures for 1 h at 37°C or, when indicated, for 24 h at 37°C. Subsequently, cells were washed and media were renewed. HCMV-infected cells were either fixed at 24 hpi and stained for HCMV IE antigen by indirect immunofluorescence or lysed at 24 hpi for Western blot analysis with detection of cellular actin (anti-β-actin MAb [AC15, Sigma]) and HCMV IE1/2 (anti-IE1/2 MAb [E13, Argene]). In HSV-2 infection assays, HFF were overlaid with 0.6 to 0.7% methylcellulose in serum-free MEM 1 hpi and fixed 12 hpi. Infection experiments with HIV-1 were conducted as described previously (52).
Attachment assay.
Cell-free TB40/E was incubated with 5% SP, 5% hSP95°C, and 10 μg/ml of heparin (Santa Cruz) in serum-free MEM for 1 h at 37°C. Attachment was conducted at 4°C, using precooled media and cells. Confluent monolayers of HFF in 8-well μ-slides (ibidi) were washed once with PBS before cooled mixtures were added to the cells for 1 h at 4°C. Alternatively, either cells were pretreated with 5% SP, 5% hSP95°C, and 10 μg/ml of heparin for 1 h at 4°C and washed and virus was added thereafter or SP was added after virus attachment for 1 h. Unbound HCMV particles were removed by washing three times with 4°C PBS. Then cells were fixed with 80% acetone for 10 min at RT, followed by indirect immunofluorescence staining for HCMV protein pp150. The number of attached viral particles per cell was determined for 100 cells per condition from two individual experiments from images randomly taken using an Axio-Observer.Z1 fluorescence microscope (Zeiss), Axiovision software, and the 63× objective.
Quantification of infection and statistical analysis.
HCMV infection was quantified after staining of HCMV IE-positive cells from 2 × 2 mosaic images, acquired with the 10× objective of the Axio-Observer.Z1 fluorescence microscope (Zeiss), by using the Image-based Tool for Counting Nuclei from ImageJ (https://imagej.nih.gov/ij/) with the following settings: width, 30 pixels; minimum distance, 18 pixels; and threshold, 0.1. HSV-2 infection was quantified by counting the total numbers of GFP-positive plaques per well. HIV-1 infectivity was quantified by Tat-induced β-galactosidase expression as described before (52). GraphPad Prism 5 was used to analyze data and to calculate the relative half-maximal inhibitory concentrations (IC50). One-way analysis of variance (ANOVA; nonparametric), followed by Bonferroni’s multiple-comparison test was performed for the statistical analysis.
Cytotoxicity.
Potential cytotoxic effects of the various conditions and different concentrations of SP were measured by determining cell viability using the MTT (tetrazolium salt) assay (Sigma).
ACKNOWLEDGMENTS
We thank all donors for providing semen samples. We further thank Kerstin Laib Sampaio, Cora Stegmann, Christian Sinzger, and Diana Lieber for cells, viruses, and support.
S.L., M.H., and R.G. are part of the International Graduate School in Molecular Medicine Ulm. J.A.M. is indebted to the Baden-Wurttemberg Stiftung for the financial support of this research project by the Eliteprogramm für Postdocs. J.M. acknowledges funding by Volkswagen Stiftung and Deutsche Forschungsgemeinschaft (DFG; German Research Foundation), project number 316249678-SFB 1279.
REFERENCES
- 1.Staras SA, Dollard SC, Radford KW, Flanders WD, Pass RF, Cannon MJ. 2006. Seroprevalence of cytomegalovirus infection in the United States, 1988–1994. Clin Infect Dis 43:1143–1151. doi: 10.1086/508173. [DOI] [PubMed] [Google Scholar]
- 2.Fowler KB, Pass RF. 2006. Risk factors for congenital cytomegalovirus infection in the offspring of young women: exposure to young children and recent onset of sexual activity. Pediatrics 118:e286–e292. doi: 10.1542/peds.2005-1142. [DOI] [PubMed] [Google Scholar]
- 3.Lanzieri TM, Kruszon-Moran D, Gambhir M, Bialek SR. 2016. Influence of parity and sexual history on cytomegalovirus seroprevalence among women aged 20–49 years in the USA. Int J Gynecol Obstet 135:82–85. doi: 10.1016/j.ijgo.2016.03.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Rawlinson WD, Boppana SB, Fowler KB, Kimberlin DW, Lazzarotto T, Alain S, Daly K, Doutr S, Gibson L, Giles ML, Greenlee J, Hamilton ST, Harrison GJ, Hui L, Jones CA, Palasanthiran P, Schleiss MR, Shand AW, van Zuylen WJ. 2017. Congenital cytomegalovirus infection in pregnancy and the neonate: consensus recommendations for prevention, diagnosis, and therapy. Lancet Infect Dis 17:e177–e188. doi: 10.1016/S1473-3099(17)30143-3. [DOI] [PubMed] [Google Scholar]
- 5.Cannon MJ, Davis KF. 2005. Washing our hands of the congenital cytomegalovirus disease epidemic. BMC Public Health 5:70. doi: 10.1186/1471-2458-5-70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Dollard SC, Grosse SD, Ross DS. 2007. New estimates of the prevalence of neurological and sensory sequelae and mortality associated with congenital cytomegalovirus infection. Rev Med Virol 17:355–363. doi: 10.1002/rmv.544. [DOI] [PubMed] [Google Scholar]
- 7.van Zuylen WJ, Hamilton ST, Naing Z, Hall B, Shand A, Rawlinson WD. 2014. Congenital cytomegalovirus infection: clinical presentation, epidemiology, diagnosis and prevention. Obstet Med 7:140–146. doi: 10.1177/1753495X14552719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Cannon MJ, Hyde TB, Schmid DS. 2011. Review of cytomegalovirus shedding in bodily fluids and relevance to congenital cytomegalovirus infection. Rev Med Virol 21:240–255. doi: 10.1002/rmv.695. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Sia IG, Patel R. 2000. New strategies for prevention and therapy of cytomegalovirus infection and disease in solid-organ transplant recipients. Clin Microbiol Rev 13:83–121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Pass RF, Anderson B. 2014. Mother-to-child transmission of cytomegalovirus and prevention of congenital infection. J Pediatric Infect Dis Soc 3:S2–S6. doi: 10.1093/jpids/piu069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Handsfield HH, Chandler SH, Caine VA, Meyers JD, Corey L, Medeiros E, McDougall JK. 1985. Cytomegalovirus infection in sex partners: evidence for sexual transmission. J Infect Dis 151:344–348. [DOI] [PubMed] [Google Scholar]
- 12.Kaspersen MD, Höllsberg P. 2013. Seminal shedding of human herpesviruses. Virol J 10:226. doi: 10.1186/1743-422X-10-226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Yan XC, Wang JH, Wang B, Huang LL, Zhou LQ, Zhu B, Liang Y. 2015. Study of human cytomegalovirus replication in body fluids, placental infection, and miscarriage during the first trimester of pregnancy. J Med Virol 87:1046–1053. doi: 10.1002/jmv.24158. [DOI] [PubMed] [Google Scholar]
- 14.Yamamoto Y, Morooka M, Hashimoto S, Ihra M, Yoshikawa T. 2014. Analysis of the shedding of three β-herpesviruses in urine and saliva of children with renal disease. J Med Virol 86:505–511. doi: 10.1002/jmv.23782. [DOI] [PubMed] [Google Scholar]
- 15.Buimovici‐Klein E, Lange M, Ong KR, Grieco MH, Cooper LZ. 1988. Virus isolation and immune studies in a cohort of homosexual men. J Med Virol 25:371–385. [DOI] [PubMed] [Google Scholar]
- 16.Krieger JN, Coombs RW, Collier AC, Ross SO, Speck C, Corey L. 1995. Seminal shedding of human immunodeficiency virus type 1 and human cytomegalovirus: evidence for different immunologic controls. J Infect Dis 171:1018–1022. [DOI] [PubMed] [Google Scholar]
- 17.Aynaud O, Poveda J-D, Huynh B, Guillemotonia A, Barrasso R. 2002. Frequency of herpes simplex virus, cytomegalovirus and human papillomavirus DNA in semen. Int J STD AIDS 13:547–550. doi: 10.1258/095646202760159666. [DOI] [PubMed] [Google Scholar]
- 18.Münch J, Rücker E, Ständker L, Adermann K, Goffinet C, Schindler M, Wildum S, Chinnadurai R, Rajan D, Specht A, Giménez-Gallego G, Sánchez PC, Fowler DM, Koulov A, Kelly JW, Mothes W, Grivel JC, Margolis L, Keppler OT, Forssmann WG, Kirchhoff F. 2007. Semen-derived amyloid fibrils drastically enhance HIV infection. Cell 131:1059–1071. doi: 10.1016/j.cell.2007.10.014. [DOI] [PubMed] [Google Scholar]
- 19.Castellano LM, Shorter J. 2012. The surprising role of amyloid fibrils in HIV infection. Biology (Basel) 1:58–80. doi: 10.3390/biology1010058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Tang Q, Roan NR, Yamamura Y. 2013. Seminal plasma and semen amyloids enhance cytomegalovirus infection in cell culture. J Virol 87:12583–12591. doi: 10.1128/JVI.02083-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Torres L, Ortiz T, Tang Q. 2015. Enhancement of herpes simplex virus (HSV) infection by seminal plasma and semen amyloids implicates a new target for the prevention of HSV infection. Viruses 7:2057–2073. doi: 10.3390/v7042057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kiessling AA. 2005. Isolation of human immunodeficiency virus type 1 from semen and vaginal fluids. Methods Mol Biol 304:71–86. doi: 10.1385/1-59259-907-9:071. [DOI] [PubMed] [Google Scholar]
- 23.Fiore JR, La Grasta L, Di Stefano M, Buccoliero G, Pastore G, Angarano G. 1997. The use of serum-free medium delays, but does not prevent, the cytotoxic effects of seminal plasma in lymphocyte cultures: implications for studies on HIV infection. New Microbiol 20:339–344. [PubMed] [Google Scholar]
- 24.Zirafi O, Kim K-A, Roan NR, Kluge SF, Muller JA, Jiang S, Mayer B, Greene WC, Kirchhoff F, Munch J. 2014. Semen enhances HIV infectivity and impairs the antiviral efficacy of microbicides. Sci Transl Med 6:262ra157. doi: 10.1126/scitranslmed.3009634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Roan NR, Liu H, Usmani SM, Neidleman J, Muller JA, Avila-Herrera A, Gawanbacht A, Zirafi O, Chu S, Dong M, Kumar ST, Smith JF, Pollard KS, Fandrich M, Kirchhoff F, Munch J, Witkowska HE, Greene WC. 2014. Liquefaction of semen generates and later degrades a conserved semenogelin peptide that enhances HIV infection. J Virol 88:7221–7234. doi: 10.1128/JVI.00269-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Sinzger C, Digel M, Jahn G. 2008. Cytomegalovirus cell tropism. Curr Top Microbiol Immunol 325:63–83. [DOI] [PubMed] [Google Scholar]
- 27.Adler B, Scrivano L, Ruzcics Z, Rupp B, Sinzger C, Koszinowski U. 2006. Role of human cytomegalovirus UL131A in cell type-specific virus entry and release. J Gen Virol 87:2451–2460. doi: 10.1099/vir.0.81921-0. [DOI] [PubMed] [Google Scholar]
- 28.Compton T, Nowlin DM, Cooper NR. 1993. Initiation of human cytomegalovirus infection requires initial interaction with cell surface heparan sulfate. Virology 193:834–841. doi: 10.1006/viro.1993.1192. [DOI] [PubMed] [Google Scholar]
- 29.Stegmann C, Hochdorfer D, Lieber D, Subramanian N, Stöhr D, Laib Sampaio K, Sinzger C. 2017. A derivative of platelet-derived growth factor receptor alpha binds to the trimer of human cytomegalovirus and inhibits entry into fibroblasts and endothelial cells. PLoS Pathog 13:e1006273. doi: 10.1371/journal.ppat.1006273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Kabanova A, Marcandalli J, Zhou T, Bianchi S, Baxa U, Tsybovsky Y, Lilleri D, Silacci-Fregni C, Foglierini M, Fernandez-Rodriguez BM, Druz A, Zhang B, Geiger R, Pagani M, Sallusto F, Kwong PD, Corti D, Lanzavecchia A, Perez L. 2016. Platelet-derived growth factor-α receptor is the cellular receptor for human cytomegalovirus gHgLgO trimer. Nat Microbiol 1:16082. doi: 10.1038/nmicrobiol.2016.82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Lupton J, Vernamonti J, McCabe C, Noble J, Yin HZ, Eyre RC, Kiessling AA. 2014. Cytomegalovirus and human immunodeficiency virus in semen of homosexual men. Fertil Steril 101:350–358. doi: 10.1016/j.fertnstert.2013.10.040. [DOI] [PubMed] [Google Scholar]
- 32.Gianella S, Smith DM, Vargas MV, Little SJ, Richman DD, Daar ES, Dube MP, Zhang F, Ginocchio CC, Haubrich RH, Morris SR. 2013. Shedding of HIV and human herpesviruses in the semen of effectively treated HIV-1-infected men who have sex with men. Clin Infect Dis 57:441–447. doi: 10.1093/cid/cit252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Eggert-Kruse W, Reuland M, Johannsen W, Strowitzki T, Schlehofer JR. 2009. Cytomegalovirus (CMV) infection-related to male and/or female infertility factors? Fertil Steril 91:67–82. doi: 10.1016/j.fertnstert.2007.11.014. [DOI] [PubMed] [Google Scholar]
- 34.Garolla A, Pizzol D, Bertoldo A, Menegazzo M, Barzon L, Foresta C. 2013. Sperm viral infection and male infertility: focus on HBV, HCV, HIV, HPV, HSV, HCMV, and AAV. J Reprod Immunol 100:20–29. doi: 10.1016/j.jri.2013.03.004. [DOI] [PubMed] [Google Scholar]
- 35.Bresson JL, Clavequin MC, Mazeron MC, Mengelle C, Scieux C, Segondy M, Houhou N, Thepot F, Obry E, Mayer G, Berjon JJ, Sauvalle A, Boucher D, Grizard G, Servoz-Gavin M, Hennebick S, Guerin JF, Cottinet D, Chalet M, Foliguet B, Jouannet P, Kunstmann JM, Dulioust E, Letur H, Alnot M, Melin MC, Mace B, Rives N, Cranz C, Mansat A, Bujan L, Barthelemy C. 2003. Risk of cytomegalovirus transmission by cryopreserved semen: a study of 635 semen samples from 231 donors. Hum Reprod 18:1881–1886. [DOI] [PubMed] [Google Scholar]
- 36.Bezold G, Politch JA, Kiviat NB, Kuypers JM, Wolff H, Anderson DJ. 2007. Prevalence of sexually transmissible pathogens in semen from asymptomatic male infertility patients with and without leukocytospermia. Fertil Steril 87:1087–1097. doi: 10.1016/j.fertnstert.2006.08.109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Diamond C, Speck C, Huang ML, Corey L, Coombs RW, Krieger JN. 2000. Comparison of assays to detect cytomegalovirus shedding in the semen of HIV-infected men. J Virol Methods 90:185–191. doi: 10.1016/S0166-0934(00)00232-9. [DOI] [PubMed] [Google Scholar]
- 38.Allen RD, Roberts TK. 1987. Role of spermine in the cytotoxic effects of seminal plasma. Am J Reprod Immunol Microbiol 13:4–8. [DOI] [PubMed] [Google Scholar]
- 39.Gianella S, Morris SR, Vargas MV, Young JA, Callahan B, Richman DD, Little SJ, Smith DM. 2013. Role of seminal shedding of herpesviruses in HIV type 1 transmission. J Infect Dis 207:257–261. doi: 10.1093/infdis/jis683. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Gianella S, Scheffler K, Mehta SR, Little SJ, Freitas L, Morris SR, Smith DM. 2015. Seminal shedding of CMV and HIV transmission among men who have sex with men. Int J Environ Res Public Health 12:7585–7592. doi: 10.3390/ijerph120707585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Loughney JW, Rustandi RR, Wang D, Troutman MC, Dick LW, Li G, Liu Z, Li F, Freed DC, Price CE, Hoang VM, Culp TD, De Phillips PA, Fu TM, Ha S. 2015. Soluble human cytomegalovirus gH/gL/pUL128-131 pentameric complex, but not gH/gL, inhibits viral entry to epithelial cells and presents dominant native neutralizing epitopes. J Biol Chem 290:15985–15995. doi: 10.1074/jbc.M115.652230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Zhou M, Lanchy J-M, Ryckman BJ. 2015. Human cytomegalovirus gH/gL/gO promotes the fusion step of entry into all cell types, whereas gH/gL/UL128-131 broadens virus tropism through a distinct mechanism. J Virol 89:8999–9009. doi: 10.1128/JVI.01325-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Müller JA, Harms M, Krüger F, Groß R, Joas S, Hayn M, Dietz AN, Lippold S, von Einem J, Schubert A, Michel M, Mayer B, Cortese M, Jang KS, Sandi-Monroy N, Deniz M, Ebner F, Vapalahti O, Otto M, Bartenschlager R, Herbeuval J-P, Schmidt-Chanasit J, Roan NR, Münch J. 2018. Semen inhibits Zika virus infection of cells and tissues from the anogenital region. Nat Commun 9:2207. doi: 10.1038/s41467-018-04442-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Dietz AN, Villinger C, Becker S, Frick M, von Einem J. 2018. A tyrosine-based trafficking motif of the tegument protein pUL71 is crucial for human cytomegalovirus secondary envelopment. J Virol 92:e00907-17. doi: 10.1128/JVI.00907-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Lieber D, Hochdorfer D, Stoehr D, Schubert A, Lotfi R, May T, Wirth D, Sinzger C. 2015. A permanently growing human endothelial cell line supports productive infection with human cytomegalovirus under conditional cell growth arrest. Biotechniques 59:127–136. doi: 10.2144/000114326. [DOI] [PubMed] [Google Scholar]
- 46.Wei X, Decker JM, Liu H, Zhang Z, Arani RB, Kilby JM, Saag MS, Wu X, Shaw GM, Kappes JC. 2002. Emergence of resistant human immunodeficiency virus type 1 in patients receiving fusion inhibitor (T-20) monotherapy. Antimicrob Agents Chemother 46:1896–1905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Sinzger C, Schmidt K, Knapp J, Kahl M, Beck R, Waldman J, Hebart H, Einsele H, Jahn G. 1999. Modification of human cytomegalovirus tropism through propagation in vitro is associated with changes in the viral genome. J Gen Virol 80:2867–2877. doi: 10.1099/0022-1317-80-11-2867. [DOI] [PubMed] [Google Scholar]
- 48.Waldman WJ, Roberts WH, Davis DH, Williams MV, Sedmak DD, Stephens RE. 1991. Preservation of natural endothelial cytopathogenicity of cytomegalovirus by propagation in endothelial cells. Arch Virol 117:143–164. [DOI] [PubMed] [Google Scholar]
- 49.Stegmann C, Abdellatif MEA, Laib Sampaio K, Walther P, Sinzger C. 2016. Importance of highly conserved peptide sites of HCMV gO for the formation of the gH/gL/gO complex. J Virol 91:e01339-16. doi: 10.1128/JVI.01339-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Taylor JM, Lin E, Susmarski N, Yoon M, Zago A, Ware CF, Pfeffer K, Miyoshi J, Takai Y, Spear PG. 2007. Alternative entry receptors for herpes simplex virus and their roles in disease. Cell Host Microbe 2:19–28. doi: 10.1016/j.chom.2007.06.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Papkalla A, Munch J, Otto C, Kirchhoff F. 2002. Nef enhances human immunodeficiency virus type 1 infectivity and replication independently of viral coreceptor tropism. J Virol 76:8455–8459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Müller JA, Münch J. 2016. Reporter assay for semen-mediated enhancement of HIV-1 infection. Bio-Protocol 6(14):e1871. doi: 10.21769/BioProtoc.1871. [DOI] [Google Scholar]