Abstract
Background
Although current guidelines now define heart failure with midrange ejection fraction (HFmrEF) as HF with a left ventricular EF of 40% to 49%, there are limited data on response to guideline‐directed medical therapy in patients with HFmrEF. The current study aimed to evaluate the association between β‐blocker, renin‐angiotensin system blocker (RASB), or aldosterone antagonist (AA) treatment with clinical outcome in patients with HFmrEF.
Methods and Results
We performed a patient‐level pooled analysis on 1144 patients with HFmrEF who were hospitalized for acute HF from the KorHF (Korean Heart Failure) and KorAHF (Korean Acute Heart Failure) registries. The study population was divided between use of β‐blocker, RASB, or AA to evaluate the guideline‐directed medical therapy in patients with HFmrEF. Sensitivity analyses, including propensity score matching and inverse‐probability‐weighted methods, were performed. The use of β‐blocker in the discharge group showed significantly lower rates of all‐cause mortality compared with those who did not use a β‐blocker (β‐blocker versus no β‐blocker, 30.7% versus 38.2%; hazard ratio, 0.758; 95% confidence interval, 0.615–0.934; P=0.009). Similarly, the RASB use in the discharge group was associated with the lower risk of mortality compared with no use of RASB (RASB versus no RASB, 31.9% versus 38.1%; hazard ratio, 0.76; 95% confidence interval, 0.618–0.946; P=0.013). However, there was no significant difference in all‐cause mortality between AA and no AA in the discharge group (AA versus no AA, 34.2% versus 34.0%; hazard ratio, 1.063; 95% confidence interval, 0.858–1.317; P=0.578). Multiple sensitivity analyses showed similar trends.
Conclusions
For treatment of acute HFmrEF after hospitalization, β‐blocker and RASB therapies on discharge were associated with reduced risk of all‐cause mortality.
Clinical Trial Registration
URL: http://www.clinicaltrials.gov. Unique identifier: NCT01389843.
Keywords: aldosterone antagonist, β‐blocker, heart failure with midrange ejection fraction, medical therapy, renin‐angiotensin system blocker
Subject Categories: Heart Failure, Mortality/Survival, Pharmacology
Clinical Perspective
What Is New?
Among patients with heart failure with midrange ejection fraction (HFmrEF) who were admitted for acute HF, patients treated with β‐blocker or renin‐angiotensin system blocker at discharge had lower risk of all‐cause mortality compared with those who did not use β‐blocker or renin‐angiotensin system blocker.
What Are the Clinical Implications?
The current guidelines suggest that HFmrEF might be managed in the same way as HF with preserved EF because there is limited evidence of the effect of guideline‐directed medical therapy in patients with HFmrEF.
Our results suggest that the use of β‐blocker and renin‐angiotensin system blocker in HFmrEF is associated with reduced risk of mortality, similar to heart failure with reduced ejection fraction.
Future randomized controlled trials are warranted to clarify whether guideline‐directed medical therapy would improve prognosis of patients with HFmrEF.
It is well known that neurohormonal antagonists (β‐blocker, renin‐angiotensin system blocker [RASB], and aldosterone antagonist [AA]) reduce morbidity and mortality in patients with heart failure with reduced ejection fraction (HFrEF).1, 2, 3, 4, 5, 6, 7, 8 β‐Blocker and RASB are recommended as class IA indications (unless contraindicated or not tolerated) in all symptomatic patients by the current guidelines.9, 10 An AA is also recommended for patients with HFrEF who remain symptomatic despite treatment with RASB and β‐blocker. However, guideline‐directed medical therapy (GDMT) has not been proved to reduce mortality and morbidity in patients with HF with preserved EF (HFpEF).
The new European Society of Cardiology guidelines suggest that patients with HF should be categorized as HFpEF (EF ≥50%), HFrEF (EF <40%), and HF with midrange EF (HFmrEF; EF 40%–49%).9 However, it is unclear if the prognosis of HFmrEF is similar to that of HFpEF, HFrEF, or a new “gray area” group.11, 12, 13, 14, 15, 16, 17, 18, 19 Furthermore, there are limited data about the effect of GDMT to reduce morbidity and mortality for patients with HFmrEF. Therefore, using 2 nationwide prospective multicenter registries from Republic of Korea, we investigated the association between GDMT, including β‐blocker, RASB, and AA, on discharge and clinical outcome in patients with HFmrEF.
Methods
The data, analytic methods, and study materials will not be made available to other researchers for purposes of reproducing the results or replicating the procedure.
Pooled Patient Population
The current study population was extracted from patient‐pooled cohorts from 2 nationwide, prospective, multicenter registries. First, the KorHF (Korean Heart Failure) registry included 3200 patients hospitalized for acute HF (AHF) from 24 hospitals in Korea, between June 2004 and April 2009.20 All consecutive patients with HF were enrolled, and HF was diagnosed on admission according to the Framingham criteria.21 Second, the KorAHF (Korean Acute Heart Failure) registry recruited 5625 hospitalized patients with AHF from 10 tertiary hospitals in Korea, between March 2011 and February 2014.22 Inclusion criteria of this registry were signs or symptoms of HF and at least 1 objective sign of lung congestion, left ventricular systolic dysfunction, or structural heart disease. There were no exclusion criteria in either registry, except withdrawal of consent. Patients with HF were categorized as having HFpEF (EF ≥50%), HFrEF (EF <40%), or HFmrEF (EF 40%–49%), according to the recent guidelines.9 Left ventricular EF (LVEF) was assessed by the biplane Simpson technique, M‐mode, or visual estimation.23 From the total pooled population of 8825 patients, 1144 with HFmrEF were selected for the current analysis (Figure 1). To identify the association between GDMT and clinical outcomes for patients with HFmrEF, the study population was stratified by use of each evidence‐based medical therapy at discharge (β‐blocker, RASB, or AA). RASB included angiotensin‐converting enzyme inhibitor or angiotensin receptor blocker. The primary outcome was all‐cause mortality during follow‐up. The study protocol was approved by the Institutional Review Board at each participating center, and all patients provided written informed consent before enrollment. This study protocol was conducted according to the principles of the Declaration of Helsinki.
Figure 1.
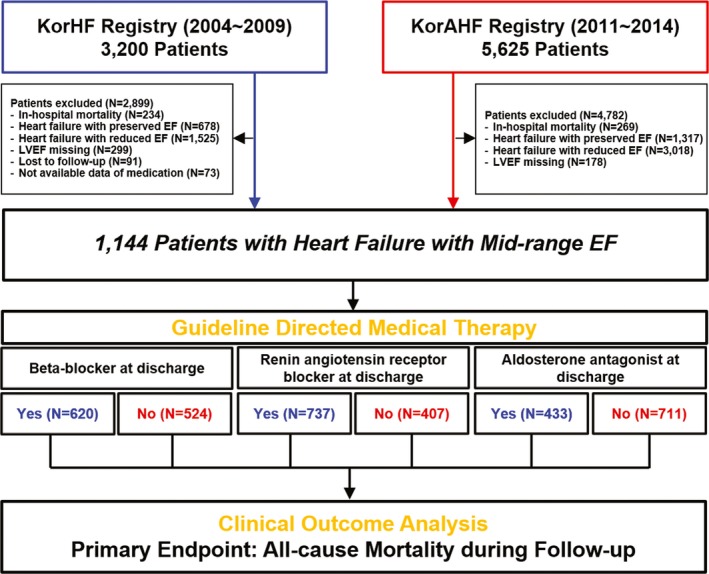
Study flow. KorAHF indicates Korean Acute Heart Failure; KorHF, Korean Heart Failure; LVEF, left ventricular ejection fraction.
Data Collection and Follow‐Up Information
In both registries, patient demographics, baseline characteristics, medical history, clinical presentation, laboratory test results, treatments, and outcomes from the initial presentation through discharge were recorded via a web‐based case‐report form by each attending physician. After discharge, follow‐up data, including all‐cause mortality, death from HF aggravation, and rehospitalization for HF aggravation, were prospectively collected using medical records or telephone interviews. Mortality data for patients who were unavailable for follow‐up were obtained from the National Insurance Data or National Death Records. All clinical events were monitored and verified by a Clinical Event Committee composed of independent experts in HF who did not participate in patient enrollment for this study. The mean observational periods of the KorHF and KorAHF registries were 1.7 years (range, 0.1–4.9 years) and 2.1 years (range, 0.1–4.2 years), respectively. Follow‐up of patients in the KorAHF registry is planned until 2018.
Statistical Analysis
Continuous variables were compared using the Welch's t test, which is broadly applicable without the need for an equal variance assumption. The χ2 test was performed to compare the categorical variables. Cumulative incidences of all‐cause mortality were assessed by Kaplan‐Meier estimates, and significance levels were compared using the log‐rank test. To compare risk of all‐cause mortality between use and no use of β‐blocker, RASB, or AA at discharge, we used Cox proportional hazards models stratified by each GDMT. Multiple sensitivity analyses, including inverse‐probability‐weighted (IPW) and propensity score matching methods, were performed to reduce selection bias and to adjust the baseline difference. Enrolled subjects were matched 1:1 for β‐blocker versus no β‐blocker, RASB versus no RASB, and AA versus no AA comparisons using a caliper of width 0.2. Variables selected for use in the propensity score matching and IPW analysis included age, sex, body mass index, current smoker, hypertension, diabetes mellitus, chronic kidney disease, previous myocardial infarction, previous cerebrovascular accident, previous HF admission, de novo AHF, New York Heart Association classification, ischemic cardiomyopathy, dilated cardiomyopathy, valvular heart disease, atrial fibrillation, systolic blood pressure, LVEF, white blood cell count, creatinine level, hemoglobin level, use of intravenous diuretics, intravenous inotropes, mechanical ventilation, transfusion, intensive care unit admission, and use of other GDMT (Table S1). Balance of baseline difference after propensity score matching or IPW adjustment was assessed by calculating percentage standardized mean differences. Percentage standardized mean differences after propensity score matching or IPW adjustment were within ±10% across all matched covariates, demonstrating successful balance between comparative groups (Table S1). Stratified and IPW adjusted Cox proportional hazard models were used to compare outcomes of matched groups. All statistical analyses were performed using R Statistical Software, version 3.4.3 (R Foundation for Statistical Computing, Vienna, Austria), with P<0.05 considered statistically significant.
Results
Baseline Characteristics
Among the total population of the pooled cohort, 59.1% of patients presented with HFrEF, 26.0% of patients presented with HFpEF, and 14.9% of patients presented with HFmrEF. Among the 1144 patients with HFmrEF, β‐blockers were prescribed for 620 (54.2%), RASBs were prescribed for 737 (64.4%), and AAs were prescribed for 433 (37.8%) at discharge (Table 1). The mean age of the study population was 70.7 years, and 532 patients (46.5%) were men. The most common cause for HFmrEF was ischemic cardiomyopathy (47.6%). Table 2 presents baseline clinical characteristics of patients with HFmrEF, according to use or not of each GDMT at discharge.
Table 1.
Baseline Clinical Characteristics of the Total Study Population
| Characteristics | Value for All Patients With Heart Failure With Midrange Ejection Fraction (N=1144) |
|---|---|
| Demographics | |
| Age, y | 70.7±13.4 |
| Male sex | 532 (46.5) |
| Body mass index, kg/m2 | 23.2±3.7 |
| Cardiovascular risk factors | |
| Current smoker | 167 (14.7) |
| Hypertension | 692 (60.5) |
| Diabetes mellitus | 402 (35.1) |
| Chronic kidney disease | 167 (14.6) |
| Previous myocardial infarction | 190 (16.6) |
| Previous cerebrovascular accident | 163 (14.3) |
| Previous heart failure admission | 248 (21.7) |
| Presentation and cause of heart failure | |
| De novo acute heart failure | 457 (39.9) |
| NYHA class ≥3 | 901 (78.8) |
| Ischemic cardiomyopathy | 544 (47.6) |
| Dilated cardiomyopathy | 54 (4.7) |
| Valvular heart disease | 162 (14.2) |
| Arrhythmia | 333 (29.1) |
| Atrial fibrillation | 295 (25.8) |
| Hemodynamic parameters at admission | |
| Systolic blood pressure, mm Hg | 137.0±32.0 |
| Diastolic blood pressure, mm Hg | 79.9±19.1 |
| Pulse rate, beats/min | 92.0±27.0 |
| Left ventricular ejection fraction, % | 44.2±2.8 |
| Laboratory data | |
| WBCs, /mm3 | 8908.2±4282.1 |
| Creatinine, mg/dL | 1.6±1.7 |
| Hemoglobin, g/dL | 11.9±2.3 |
| NT‐proBNP, pg/mL | 9149.2±10 378.0 |
| In‐hospital management | |
| Intravenous diuretics | 827 (72.3) |
| Intravenous inotropes | 265 (23.2) |
| Digoxin | 276 (24.1) |
| Nitrates | 595 (52.0) |
| Mechanical ventilation | 125 (10.9) |
| Transfusion | 198 (17.3) |
| ICU admission | 543 (47.5) |
| Length of stay, d | 9.0 (6.0–14.0) |
| Medications at discharge | |
| β‐Blocker | 620 (54.2) |
| Renin‐angiotensin system blockera | 737 (64.4) |
| Aldosterone antagonist | 433 (37.8) |
| No use of the 3 drugs | 170 (14.9) |
| β‐Blocker only | 97 (8.5) |
| Renin‐angiotensin system blocker only | 167 (14.6) |
| Aldosterone antagonist only | 77 (6.7) |
| β‐Blocker+renin‐angiotensin system blocker | 277 (24.2) |
| β‐Blocker+aldosterone antagonist | 63 (5.5) |
| Renin‐angiotensin system blocker+aldosterone antagonist | 110 (9.6) |
| All the 3 drugs | 183 (16.0) |
| Loop diuretics | 898 (78.5) |
Values are mean±SD, median (quartile 1–quartile 3), or number (percentage). ICU indicates intensive care unit; NT‐proBNP, N‐terminal pro‐B‐type natriuretic peptide; NYHA, New York Heart Association; WBC, white blood cell.
Renin‐angiotensin system blocker included angiotensin‐converting enzyme inhibitor and angiotensin receptor blocker.
Table 2.
Baseline Clinical Characteristics of Patients With HFmrEF Stratified by GDMT
| Characteristics | β‐Blocker at Discharge | Renin‐Angiotensin System Blocker at Discharge | Aldosterone Antagonist at Discharge | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Yes (N=620) | No (N=524) | P Value | Yes (N=737) | No (N=407) | P Value | Yes (N=433) | No (N=711) | P Value | |
| Age, y | 70.0±12.9 | 71.4±13.9 | 0.074 | 70.9±13.1 | 70.3±13.9 | 0.505 | 70.8±14.0 | 70.6±12.9 | 0.784 |
| Male sex | 286 (46.1) | 246 (46.9) | 0.828 | 346 (46.9) | 186 (45.7) | 0.732 | 178 (41.1) | 354 (49.8) | 0.005 |
| Body mass index, kg/m2 | 23.5±3.7 | 23.0±3.6 | 0.034 | 23.4±3.7 | 22.9±3.6 | 0.011 | 23.1±3.9 | 23.3±3.6 | 0.393 |
| Current smoker | 103 (16.6) | 64 (12.2) | 0.044 | 101 (13.7) | 66 (16.2) | 0.287 | 58 (13.4) | 109 (15.3) | 0.416 |
| Hypertension | 396 (63.9) | 296 (56.5) | 0.013 | 472 (64.0) | 220 (54.1) | 0.001 | 247 (57.0) | 445 (62.6) | 0.072 |
| Diabetes mellitus | 236 (38.1) | 166 (31.7) | 0.028 | 269 (36.5) | 133 (32.7) | 0.218 | 131 (30.3) | 271 (38.1) | 0.008 |
| Chronic kidney disease | 99 (16.0) | 68 (13.0) | 0.179 | 96 (13.0) | 71 (17.4) | 0.052 | 32 (7.4) | 135 (19.0) | <0.001 |
| Previous myocardial infarction | 109 (17.6) | 81 (15.5) | 0.378 | 130 (17.6) | 60 (14.7) | 0.239 | 60 (13.9) | 130 (18.3) | 0.062 |
| Previous cerebrovascular accident | 98 (15.8) | 65 (12.4) | 0.120 | 116 (15.7) | 47 (11.5) | 0.064 | 59 (13.6) | 104 (14.6) | 0.702 |
| Previous heart failure admission | 125 (20.2) | 123 (23.5) | 0.200 | 175 (23.7) | 73 (17.9) | 0.027 | 111 (25.6) | 137 (19.3) | 0.014 |
| De novo acute heart failure | 233 (37.6) | 224 (42.7) | 0.086 | 293 (39.8) | 164 (40.3) | 0.908 | 193 (44.6) | 264 (37.1) | 0.015 |
| NYHA class ≥3 | 500 (80.6) | 401 (76.5) | 0.104 | 589 (79.9) | 312 (76.7) | 0.224 | 357 (82.4) | 544 (76.5) | 0.021 |
| Ischemic cardiomyopathy | 339 (54.7) | 205 (39.1) | <0.001 | 358 (48.6) | 186 (45.7) | 0.384 | 168 (38.8) | 376 (52.9) | <0.001 |
| Dilated cardiomyopathy | 24 (3.9) | 30 (5.7) | 0.182 | 36 (4.9) | 18 (4.4) | 0.836 | 32 (7.4) | 22 (3.1) | 0.001 |
| Valvular heart disease | 64 (10.3) | 98 (18.7) | <0.001 | 93 (12.6) | 69 (17.0) | 0.054 | 67 (15.5) | 95 (13.4) | 0.365 |
| Arrhythmia | 155 (25.0) | 178 (34.0) | 0.001 | 214 (29.0) | 119 (29.2) | 0.997 | 143 (33.0) | 190 (26.7) | 0.027 |
| Atrial fibrillation | 141 (22.7) | 154 (29.4) | 0.013 | 189 (25.6) | 106 (26.0) | 0.938 | 129 (29.8) | 166 (23.3) | 0.019 |
| Systolic blood pressure, mm Hg | 138.0±32.1 | 135.7±31.9 | 0.239 | 140.3±31.2 | 131.0±32.5 | <0.001 | 135.3±31.0 | 138.0±32.6 | 0.164 |
| Diastolic blood pressure, mm Hg | 80.6±19.1 | 79.0±19.1 | 0.156 | 81.6±19.2 | 76.8±18.5 | <0.001 | 78.7±17.3 | 80.6±20.1 | 0.081 |
| Pulse rate, beats/min | 93.5±26.7 | 90.3±27.3 | 0.046 | 92.0±26.7 | 92.1±27.4 | 0.948 | 92.0±27.0 | 92.0±26.9 | 0.975 |
| Left ventricular ejection fraction, % | 44.2±2.9 | 44.2±2.9 | 0.855 | 44.1±2.8 | 44.3±2.9 | 0.373 | 44.3±2.9 | 44.1±2.8 | 0.470 |
| WBCs, /mm3 | 9317.6±4598.3 | 8428.7±3828.3 | <0.001 | 8762.8±4021.8 | 9173.3±4713.6 | 0.141 | 8456.2±3822.9 | 9181.1±4517.7 | 0.004 |
| Creatinine, mg/dL | 1.6±1.8 | 1.5±1.5 | 0.193 | 1.6±1.6 | 1.7±1.7 | 0.345 | 1.2±0.9 | 1.8±1.9 | <0.001 |
| Hemoglobin, g/dL | 12.0±2.4 | 11.9±2.3 | 0.874 | 11.9±2.4 | 11.9±2.2 | 0.922 | 12.1±2.4 | 11.9±2.3 | 0.151 |
| NT‐proBNP, pg/mL | 9778.2±11 025.9 | 8420.8±9539.4 | 0.091 | 8648.4±10 165.0 | 10 176.4±10 752.8 | 0.077 | 8324.7±9608.3 | 9621.7±10 777.2 | 0.112 |
| Intravenous diuretic | 449 (72.4) | 378 (72.1) | 0.968 | 538 (73.0) | 289 (71.0) | 0.515 | 341 (78.8) | 486 (68.4) | <0.001 |
| Intravenous inotrope | 129 (20.8) | 136 (26.0) | 0.047 | 120 (16.3) | 145 (35.6) | <0.001 | 88 (20.3) | 177 (24.9) | 0.088 |
| Digoxin | 140 (22.5) | 136 (26.0) | 0.208 | 187 (25.4) | 89 (21.9) | 0.210 | 135 (31.2) | 141 (19.8) | <0.001 |
| Nitrates | 355 (57.3) | 240 (45.8) | <0.001 | 394 (53.5) | 201 (49.4) | 0.208 | 209 (48.3) | 386 (54.3) | 0.055 |
| Mechanical ventilation | 68 (11.0) | 57 (10.9) | >0.999 | 67 (9.1) | 58 (14.3) | 0.010 | 46 (10.6) | 79 (11.1) | 0.874 |
| Transfusion | 102 (16.5) | 63 (18.3) | 0.451 | 107 (14.5) | 91 (22.4) | 0.001 | 67 (15.5) | 131 (18.4) | 0.230 |
| ICU admission | 312 (50.3) | 231 (44.1) | 0.041 | 331 (44.9) | 212 (52.1) | 0.023 | 188 (43.4) | 355 (49.9) | 0.038 |
| Length of stay, d | 9.0 (6.0–13.5) | 9.0 (6.0–15.0) | 0.110 | 9.0 (6.0–13.0) | 10.0 (7.0–17.0) | <0.001 | 9.0 (6.0–13.0) | 9.0 (6.0–15.0) | 0.114 |
| β‐Blocker | 620 (100) | 0 (0) | <0.001 | 460 (62.4) | 160 (39.3) | <0.001 | 246 (56.8) | 374 (52.6) | 0.185 |
| Renin‐angiotensin system blockera | 460 (74.2) | 277 (52.9) | <0.001 | 737 (100) | 0 (0) | <0.001 | 293 (67.7) | 444 (62.4) | 0.085 |
| Aldosterone antagonist | 246 (39.7) | 187 (35.7) | 0.185 | 293 (39.8) | 140 (34.4) | 0.085 | 433 (100) | 0 (0) | <0.001 |
| Loop diuretic | 489 (78.9) | 409 (78.1) | 0.792 | 581 (78.8) | 317 (77.9) | 0.766 | 384 (88.7) | 514 (72.3) | <0.001 |
Values are mean±SD, median (quartile 1–quartile 3), or number (percentage). GDMT indicates guideline‐directed medical therapy; HFmrEF, heart failure with midrange ejection fraction; ICU, intensive care unit; NT‐proBNP, N‐terminal pro‐B‐type natriuretic peptide; NYHA, New York Heart Association; WBC, white blood cell.
Renin‐angiotensin system blocker included angiotensin‐converting enzyme inhibitor and angiotensin receptor blocker.
Compared with the no β‐blocker group, patients in the β‐blocker group had significantly higher prevalence of hypertension and diabetes mellitus. Ischemic cause was more frequent in the β‐blocker group, but valvular heart disease and atrial fibrillation were more frequent in the no β‐blocker group. Patients with RASB at discharge also had significantly higher hypertension. Systolic and diastolic pressure at admission and history of HF admission were significantly higher in the RASB group than in the no RASB group. Both the β‐blocker and RASB groups were less likely to receive treatment with intravenous inotropes during admission compared with the no β‐blocker and no RASB groups. Compared with the no AA group, the AA group had a higher proportion of female patients and patients with a history of HF admission, New York Heart Association classification ≥3, and use of intravenous diuretics, but a lower proportion of patients with diabetes mellitus, chronic kidney disease, and ischemic cause. Also, the AA group had lower levels of creatinine than the no AA group. There was no significant difference in LVEF in all stratified groups.
Propensity score matching was performed with 375 matched pairs of patients for comparison between the β‐blocker and no β‐blocker groups, 302 matched pairs of patients for comparison between the RASB and no RASB groups, and 362 matched pairs of patients for comparison between the AA and no AA groups. The C statistics for the propensity score model were 0.69 for β‐blocker, 0.74 for RASB, and 0.70 for AA (Hosmer‐Lemeshow goodness of fit, P=0.94, P=0.81, and P=0.09, respectively).
Clinical Outcome
GDMT for HFpEF and HFrEF
Among patients with HFpEF, β‐blocker, RASB, and AA at discharge were not associated with reduced risk of all‐cause mortality compared with the no drug group (Figure S1). β‐Blocker and RASB at discharge were significantly associated with lower risk of all‐cause mortality compared with no β‐blocker and no RASB at discharge in patients with HFrEF (Figure S2). However, among patients with HFrEF, there was no significant difference in the rates of all‐cause mortality between the AA at discharge and no AA at discharge groups (Figure S2).
Overall population of HFmrEF
The median follow‐up duration was 27 months (interquartile range, 17–37 months). A total of 354 patients (30.9%) died during a 3‐year follow‐up period. Patients prescribed β‐blocker at discharge showed a significantly lower risk of all‐cause mortality compared with those without β‐blocker at discharge (β‐blocker versus no β‐blocker, 30.7% versus 38.2%; hazard ratio [HR], 0.758; 95% confidence interval [CI], 0.615–0.934; P=0.009) (Table 3). Similarly, prescription of RASB at discharge was associated with lower rates of all‐cause mortality compared with those without RASB at discharge (RASB versus no RASB, 31.9% versus 38.1%; HR, 0.76; 95% CI, 0.618–0.946; P=0.013) (Table 3). However, there was no significant difference in the rate of all‐cause mortality between the AA and no AA at discharge groups (AA versus no AA, 34.2% versus 34.0%; HR, 1.063; 95% CI, 0.858–1.317; P=0.578) (Table 3).
Table 3.
Comparison of All‐Cause Mortality During Follow‐Up, According to GDMT at Discharge
| Variable | Cumulative Incidence, No. %a | Univariate Analysis | IPW Adjusted Analysis | PS‐Matched Analysis | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| HR | 95% CI | P Value | HR | 95% CI | P Value | HR | 95% CI | P Value | ||
| β‐Blocker at discharge | (375 pairs) | |||||||||
| No (n=524) | 183 (38.2) | 1.000 | Reference | NA | 1.000 | Reference | NA | 1.000 | Reference | NA |
| Yes (n=620) | 171 (30.7) | 0.758 | 0.615–0.934 | 0.009 | 0.827 | 0.708–0.967 | 0.017 | 0.734 | 0.565–0.954 | 0.021 |
| Renin‐angiotensin system blocker at discharge | (302 pairs) | |||||||||
| No (n=407) | 143 (38.1) | 1.000 | Reference | NA | 1.000 | Reference | NA | 1.000 | Reference | NA |
| Yes (n=737) | 211 (31.9) | 0.765 | 0.618–0.946 | 0.013 | 0.814 | 0.698–0.950 | 0.009 | 0.755 | 0.570–0.999 | 0.048 |
| Aldosterone antagonist at discharge | (362 pairs) | |||||||||
| No (n=711) | 218 (34.0) | 1.000 | Reference | NA | 1.000 | Reference | NA | 1.000 | Reference | NA |
| Yes (n=433) | 136 (34.2) | 1.063 | 0.858–1.317 | 0.578 | 1.138 | 0.974–1.330 | 0.103 | 1.176 | 0.904–1.526 | 0.229 |
CI indicates confidence interval; GDMT, guideline‐directed medical therapy; HR, hazard ratio; IPW, inverse probability weighted; NA, not applicable; PS, propensity score.
Cumulative incidences of all‐cause mortality are presented as Kaplan‐Meier estimates.
Sensitivity analyses
After 1:1 propensity score matching of 375 pairs for use of β‐blocker or not, the β‐blocker at discharge group showed significantly lower risk of mortality than the no β‐blocker group among patients with HFmrEF (29.5% versus 37.8%; HR, 0.734; 95% CI, 0.565–0.954; P=0.021) (Table 3, Figure 2A). RASB at discharge was associated with significantly lower risk of all‐cause mortality compared with no RASB at discharge in the 302 pairs of propensity‐matched cohorts (32.5% versus 39.6%; HR, 0.755; 95% CI, 0.570–0.999; P=0.048) (Table 3, Figure 2B). However, all‐cause mortality rates did not differ between the AA and no AA groups in the 362 pairs of propensity‐matched populations (35.1% versus 33.3%; HR, 1.176; 95% CI, 0.904–1.526; P=0.229) (Table 3, Figure 2C). IPW analysis consistently showed similar outcomes between the stratified groups (Table 3).
Figure 2.
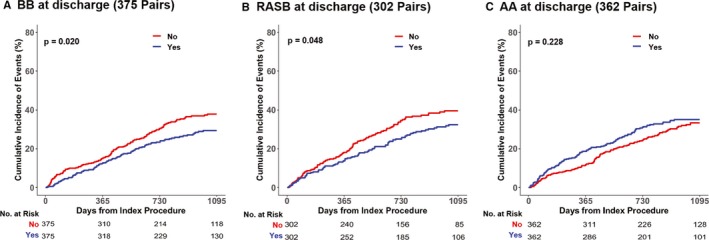
Comparison of all‐cause mortality at 3 years, according to use of guideline‐directed medical therapy among each propensity‐matched population. Kaplan‐Meier curves for all‐cause mortality of patients with acute heart failure with midrange ejection fraction, according to use of β‐blocker (blue line) or no β‐blocker (red line) in the 375 pairs from a propensity score–matched population (A), use of renin‐angiotensin system blocker (RASB; blue line) or no RASB (red line) in the 302 pairs from a propensity‐matched population (B), and use of aldosterone antagonist (AA; blue line) or no AA (red line) in the 362 pairs from a propensity‐matched population (C), are presented.
Use of β‐blocker and RASB in patients with HFmrEF
To evaluate the combination effect of β‐blocker and RASB, which were initially recommended for patients with HF by the current guidelines, we stratified 4 groups (no drug, β‐blocker only, RASB only, and β‐blocker plus RASB) and compared the clinical outcomes. On a multivariable Cox proportional hazard model of total population, the β‐blocker only, RASB only, and β‐blocker plus RASB groups associated with lower risk of all‐cause mortality compared with the no drug group (Table 4, Figure 3). The lowest HR and cumulative incidence of all‐cause mortality were observed in the β‐blocker plus RASB group on multivariable analysis. Variables selected for a multivariate model included age, sex, body mass index, hypertension, diabetes mellitus, chronic kidney disease, current smoker, New York Heart Association class ≥3, history of cerebrovascular events, history of HF admission, history of myocardial infarction, ischemic cause, dilated cardiomyopathy, atrial fibrillation, valvular heart disease, systolic blood pressure, hemoglobin, white blood cell count, LVEF, and use of intravenous inotropes during admission. Other predictors of all‐cause mortality among patients with HFmrEF are presented in Table 4.
Table 4.
Predictors of All‐Cause Mortality in Patients With HFmrEF
| Variable | Adjusted HR (95% CI)a | P Value |
|---|---|---|
| Use of β‐blocker and/or RASB (n=1144) | ||
| No drug (n=247) | 1.000 (Reference) | NA |
| β‐Blocker only (n=160) | 0.667 (0.464–0.960) | 0.029 |
| RASB only (n=277) | 0.673 (0.496–0.915) | 0.011 |
| β‐Blocker and RASB (n=460) | 0.636 (0.476–0.851) | 0.002 |
| Age (per 1 increase) | 1.042 (1.031–1.054) | <0.001 |
| Body mass index (per 1 increase) | 0.964 (0.932–0.997) | 0.031 |
| Hemoglobin (per 1 increase) | 0.858 (0.814–0.904) | <0.001 |
| WBCs (per 1000 increase) | 1.045 (1.022–1.070) | <0.001 |
| Male sex | 1.453 (1.156–1.828) | 0.001 |
| Chronic kidney disease | 1.430 (1.071–1.909) | 0.015 |
| Previous heart failure admission | 1.384 (1.057–1.811) | 0.018 |
| Use of intravenous inotrope at admission | 1.353 (1.051–1.741) | 0.019 |
Adjusted variables included age, sex, hypertension, diabetes mellitus, chronic kidney disease, current smoker, body mass index, history of myocardial infarction, cerebrovascular event, heart failure admission, ischemic cardiomyopathy, dilated cardiomyopathy, valvular heart disease, atrial fibrillation, left ventricular ejection fraction, systolic blood pressure, New York Heart Association class ≥3, white blood cell count, hemoglobin, use of β‐blocker and/or RASB, and aldosterone antagonist. CI indicates confidence interval; HFmrEF, heart failure with midrange ejection fraction; HR, hazard ratio; NA, not applicable; RASB, renin‐angiotensin system blocker; WBC, white blood cell.
Harrell's C‐index of the Cox regression model for all‐cause death was 0.723 (95% CI, 0.692–0.754).
Figure 3.
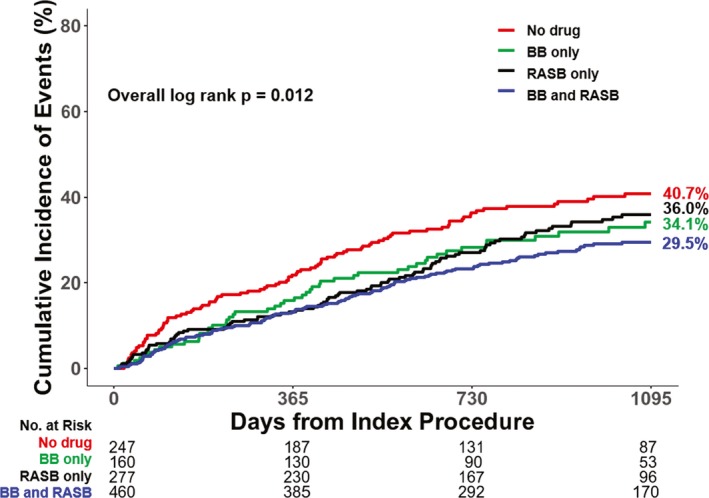
Comparison of all‐cause mortality at 3 years among patients with heart failure with midrange ejection fraction according to use of renin‐angiotensin system blocker (RASB) and β‐blocker. The cumulative incidence of all‐cause mortality was compared among 4 groups divided according to use of RASB and/or β‐blocker. Red line denotes no drug group, green line denotes β‐blocker only group, black line denotes RASB only group, and blue line denotes β‐blocker and RASB groups.
Use of digoxin and nitrates in patients with HFmrEF
We performed the additional analysis for evaluating the effects of digoxin and nitrates on the risk of mortality in patients with acute HFmrEF. Both digoxin and nitrate were not associated with reduced risk of mortality in patients with HFmrEF (Figure S3).
Subgroup Analysis
Figure 4 presents the forest plot for various subgroups to identify the consistency of outcomes of each GDMT. The difference of rate of all‐cause mortality between use of each GDMT or not was consistent across the various subgroups without significant interaction.
Figure 4.
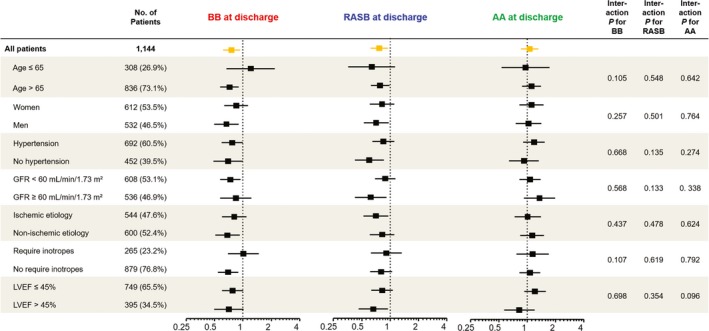
Subgroup analysis among the total population, according to use of guideline‐directed medical therapy. Comparative unadjusted hazard ratios of all‐cause mortality for subgroups in the overall population, according to use of β‐blocker (red text), renin‐angiotensin system blocker (RASB; blue text), and aldosterone antagonist (AA; green text). Box denotes hazard ratio, and line denotes 95% confidence interval. GFR indicates glomerular filtration rate; LVEF, left ventricular ejection fraction.
Discussion
The current study evaluated the effect of GDMT, including β‐blocker, RASB, and AA at discharge, on clinical outcomes in hospitalized patients with HFmrEF using patient‐pooled cohorts from 2 nationwide, prospective, multicenter registries in Korea. Major findings of this study are as follows. First, prescription of β‐blocker or RASB at discharge was associated with reduced risk of all‐cause mortality in patients with HFmrEF. These results were maintained after propensity score matching and IPW analyses. Second, rates of all‐cause mortality were not significantly different between prescription of AA at discharge or not in patients with AHF with HFmrEF. Third, when dividing patients into 4 groups according to use of β‐blocker and/or RASB, the β‐blocker only, RASB only, and β‐blocker plus RASB groups had a significantly lower all‐cause mortality rate compared with the no drug group. In addition, the lowest HR and cumulative incidence of all‐cause mortality were observed in the β‐blocker plus RASB group on multivariable analysis.
Characteristics and Prognosis of HFmrEF
HF is a clinical syndrome with various causes, characterized by dyspnea, fatigue, and signs of volume overload. It is well known that patients with HFpEF have different underlying causes, demographics, comorbidities, and responses to GDMT compared with those with HFrEF.24, 25 However, the prognosis and causes of HFmrEF, recently classified as a new group, show conflicting results on the basis of previous studies. Although most studies to evaluate the outcomes of HFmrEF have shown that the HFmrEF group has an intermediate clinical profile between HFrEF and HFpEF, several studies have reported that the prognosis of HFmrEF is similar to that of HFrEF,11, 12, 13 and other studies have shown that the prognosis of HFmrEF is similar to that of HFpEF.14, 15, 16 In addition, several studies have found that the prognosis of HFmrEF may vary according to cause or clinical presentation. Our group recently reported that prognosis of HFmrEF may differ according to de novo HF or acute decompensated HF using the KorAHF registry.17 Furthermore, Koh et al showed that HFmrEF is overall an intermediate phenotype between HFpEF and HFrEF, with the important exception of ischemic cause, which was both more common and associated with higher mortality in HFmrEF and HFrEF compared with HFpEF.18 Consistent with previous studies, 47.6% of the current study population had HFmrEF attributable to ischemic cause, similar to that of HFrEF. This indirectly supports the hypothesis that GDMT, such as β‐blocker and RASB, which are effective for reducing morbidity and mortality in acute coronary syndrome, will also be effective for patients with HFmrEF.
GDMT for HFmrEF
The current guidelines suggest that HFmrEF might be managed in the same way as HFpEF because there is limited evidence of the effect of GDMT in patients with HFmrEF.9 However, recent studies on the mortality benefit of GDMT, such as β‐blocker or RASB in patients with HFmrEF, have been reported. Patient‐level meta‐analysis of 11 double‐blind, randomized, placebo‐controlled trials, stratified by baseline LVEF and heart rhythm, demonstrated that β‐blocker significantly improves the prognosis for patients with HFmrEF as well as HFrEF.26 Furthermore, post hoc analysis of the CHARM (Candesartan in Heart Failure: Assessment of Reduction in Mortality and Morbidity) trial also suggests that candesartan improves outcomes in HFmrEF to a similar degree as in HFrEF.27 In concordance with previous studies, we identified the mortality benefits of β‐blocker and RASB in hospitalized patients with HFmrEF using patient‐pooled data from 2 nationwide, prospective, multicenter registries. In particular, our data showed the lowest HR of all‐cause mortality in the β‐blocker plus RASB group after classifying patients into 4 groups according to use of β‐blocker and/or RASB. This result would support the use of β‐blocker and RASB in HFmrEF to reduce the risk of mortality, similar to HFrEF. Future large randomized controlled trials for the effects of β‐blocker and RASB in patients with HFmrEF will be useful to confirm our results. In addition, future guidelines for patients with HFmrEF could be changed to treatment strategies similar to those for HFrEF, not HFpEF, on the basis of the results of several trials, including the present study.
AAs, including spironolactone and eplerenone, are associated with reducing morbidity and mortality in patients with systolic HF (EF <35%) in addition to recommended therapy.7, 8 However, the TOPCAT (Treatment of Preserved Cardiac Function Heart Failure With an Aldosterone Antagonist) trial failed to prove the benefit of spironolactone therapy in patients with HFpEF (EF ≥45%).28 Although post hoc analysis from the TOPCAT trial, stratified by EF, showed that the potential efficacy of spironolactone tended to be maximized at the lower end of the LVEF spectrum29; to date, there are no data comparing the outcomes of use of AAs in patients with HFmrEF. The present study demonstrated that the risk of mortality did not differ between use of AA or not in patients with HFmrEF. However, observational studies consistently show a lack of benefit with AA, but randomized controlled trials consistently show a benefit for patients with HFrEF. Perhaps, this result might be a failure of method rather than a lack of benefit from AA. Because AAs were recommended to be prescribed to those who remained symptomatic taking the medication of RASB and β‐blocker, those with AAs might be more severe patients with worse prognosis, although we performed propensity matching analysis. In addition, the failure to reach the statistical significance of the effect of AA use on clinical outcome might be because of type II error. In this regard, well‐designed randomized controlled trials focusing on the effect of AA in patients with HFmrEF will be needed.
Limitations
The current study had several limitations. First, the nonrandomized nature of registry data could introduce selection bias, and use of GDMT was based on physician's discretion. Although we performed various risk adjustments for potential confounding factors, including propensity score matching and IPW analysis, we cannot correct for unmeasured variables in the present study. Second, LVEF was measured by various methods, including biplane Simpson technique, M‐mode, and visual estimation, rather than by a single method. However, this variability may only minimally affect the present study, because LVEF was only used to stratify the groups. Third, there are no data on doses or on postdischarge initiation or discontinuation of GDMT. Fourth, the analysis was performed on the pooled data set from 2 distinct registries that captured hospitalizations from 2004 to 2009 and from 2011 to 2014. Furthermore, baseline characteristics and clinical outcomes slightly differ between the 2 registries (Table S2). However, there was no change of medical treatment strategy for patients with HF between 2004 and 2009 and between 2011 and 2014. Finally, because the current study only included patients who were admitted for AHF, we were not able to extrapolate the effects of GDMT on patients with chronic stable HF.
Conclusions
For the treatment of acute HFmrEF after hospitalization, β‐blocker and RASB at discharge were associated with reduced risk of all‐cause mortality. However, treatment with AA at discharge was not associated with reduced risk of all‐cause mortality for the management of HFmrEF.
Sources of Funding
This work was supported by the Research of Korea Centers for Disease Control and Prevention (2010‐E63003‐00, 2011‐E63002‐00, 2012‐E63005‐00, 2013‐E63003‐00, 2013‐18 E63003‐01, 2013‐E63003‐02, and 2016‐ER6303‐00) for the KorAHF (Korean Acute Heart Failure) registry and the Korean Society of Heart Failure for the KorHF (Korean Heart Failure) registry.
Disclosures
None.
Supporting information
Table S1. Standardized Differences of Variables Used in PS Matching and IPW Adjustment to Adjust for Baseline Differences
Table S2. Comparison of KorHF and KorAHF Populations
Figure S1. Comparison of all‐cause mortality at 3 years according to use of guideline directed medical therapy among patients with heart failure with preserved ejection fraction.
Figure S2. Comparison of all‐cause mortality at 3 years according to use of guideline directed medical therapy among patients with heart failure with reduced ejection fraction.
Figure S3. Comparison of all‐cause mortality at 3 years according to use of digoxin and nitrate among patients with heart failure with mid‐range ejection fraction.
(J Am Heart Assoc. 2018;7:e009806 DOI: 10.1161/JAHA.118.009806.)
References
- 1. The SOLVD Investigators. Effect of enalapril on survival in patients with reduced left ventricular ejection fractions and congestive heart failure. N Engl J Med. 1991;325:293–302. [DOI] [PubMed] [Google Scholar]
- 2. Garg R, Yusuf S; Collaborative Group on ACE Inhibitor Trials . Overview of randomized trials of angiotensin‐converting enzyme inhibitors on mortality and morbidity in patients with heart failure. JAMA. 1995;273:1450–1456. [PubMed] [Google Scholar]
- 3. Pfeffer MA, McMurray JJ, Velazquez EJ, Rouleau JL, Kober L, Maggioni AP, Solomon SD, Swedberg K, Van de Werf F, White H, Leimberger JD, Henis M, Edwards S, Zelenkofske S, Sellers MA, Califf RM; Valsartan in Acute Myocardial Infarction Trial Investigators . Valsartan, captopril, or both in myocardial infarction complicated by heart failure, left ventricular dysfunction, or both. N Engl J Med. 2003;349:1893–1906. [DOI] [PubMed] [Google Scholar]
- 4. Effect of metoprolol CR/XL in chronic heart failure: metoprolol CR/XL randomised intervention trial in congestive heart failure (MERIT‐HF). Lancet. 1999;353:2001–2007. [PubMed] [Google Scholar]
- 5. The Cardiac Insufficiency Bisoprolol Study II (CIBIS‐II): a randomised trial. Lancet. 1999;353:9–13. [PubMed] [Google Scholar]
- 6. Packer M, Coats AJ, Fowler MB, Katus HA, Krum H, Mohacsi P, Rouleau JL, Tendera M, Castaigne A, Roecker EB, Schultz MK, DeMets DL; Carvedilol Prospective Randomized Cumulative Survival Study Group . Effect of carvedilol on survival in severe chronic heart failure. N Engl J Med. 2001;344:1651–1658. [DOI] [PubMed] [Google Scholar]
- 7. Pitt B, Zannad F, Remme WJ, Cody R, Castaigne A, Perez A, Palensky J, Wittes J; Randomized Aldactone Evaluation Study Investigators . The effect of spironolactone on morbidity and mortality in patients with severe heart failure. N Engl J Med. 1999;341:709–717. [DOI] [PubMed] [Google Scholar]
- 8. Zannad F, McMurray JJ, Krum H, van Veldhuisen DJ, Swedberg K, Shi H, Vincent J, Pocock SJ, Pitt B; EMPHASIS‐HF Study Group . Eplerenone in patients with systolic heart failure and mild symptoms. N Engl J Med. 2011;364:11–21.21073363 [Google Scholar]
- 9. Ponikowski P, Voors AA, Anker SD, Bueno H, Cleland JGF, Coats AJS, Falk V, Gonzalez‐Juanatey JR, Harjola VP, Jankowska EA, Jessup M, Linde C, Nihoyannopoulos P, Parissis JT, Pieske B, Riley JP, Rosano GMC, Ruilope LM, Ruschitzka F, Rutten FH, van der Meer P; ESC Scientific Document Group . 2016 ESC guidelines for the diagnosis and treatment of acute and chronic heart failure: the Task Force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC): developed with the special contribution of the Heart Failure Association (HFA) of the ESC. Eur Heart J. 2016;37:2129–2200. [DOI] [PubMed] [Google Scholar]
- 10. Yancy CW, Jessup M, Bozkurt B, Butler J, Casey DE Jr, Colvin MM, Drazner MH, Filippatos GS, Fonarow GC, Givertz MM, Hollenberg SM, Lindenfeld J, Masoudi FA, McBride PE, Peterson PN, Stevenson LW, Westlake C. 2017 ACC/AHA/HFSA focused update of the 2013 ACCF/AHA guideline for the management of heart failure: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Failure Society of America. Circulation. 2017;136:e137–e161. [DOI] [PubMed] [Google Scholar]
- 11. Vedin O, Lam CSP, Koh AS, Benson L, Teng THK, Tay WT, Braun OO, Savarese G, Dahlstrom U, Lund LH. Significance of ischemic heart disease in patients with heart failure and preserved, midrange, and reduced ejection fraction: a nationwide cohort study. Circ Heart Fail. 2017;10:e003875. [DOI] [PubMed] [Google Scholar]
- 12. Rickenbacher P, Kaufmann BA, Maeder MT, Bernheim A, Goetschalckx K, Pfister O, Pfisterer M, Brunner‐La Rocca HP; TIME‐CHF Investigators . Heart failure with mid‐range ejection fraction: a distinct clinical entity? Insights from the Trial of Intensified versus standard Medical therapy in Elderly patients with Congestive Heart Failure (TIME‐CHF). Eur J Heart Fail. 2017;19:1586–1596. [DOI] [PubMed] [Google Scholar]
- 13. Bhambhani V, Kizer JR, Lima JAC, van der Harst P, Bahrami H, Nayor M, de Filippi CR, Enserro D, Blaha MJ, Cushman M, Wang TJ, Gansevoort RT, Fox CS, Gaggin HK, Kop WJ, Liu K, Vasan RS, Psaty BM, Lee DS, Brouwers FP, Hillege HL, Bartz TM, Benjamin EJ, Chan C, Allison M, Gardin JM, Januzzi JL Jr, Levy D, Herrington DM, van Gilst WH, Bertoni AG, Larson MG, de Boer RA, Gottdiener JS, Shah SJ, Ho JE. Predictors and outcomes of heart failure with mid‐range ejection fraction. Eur J Heart Fail. 2018;20:651–659. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Guisado‐Espartero ME, Salamanca‐Bautista P, Aramburu‐Bodas O, Conde‐Martel A, Arias‐Jimenez JL, Llacer‐Iborra P, Davila‐Ramos MF, Cabanes‐Hernandez Y, Manzano L, Montero‐Perez‐Barquero M; RICA Investigators Group . Heart failure with mid‐range ejection fraction in patients admitted to internal medicine departments: findings from the RICA Registry. Int J Cardiol. 2018;255:124–128. [DOI] [PubMed] [Google Scholar]
- 15. Farmakis D, Simitsis P, Bistola V, Triposkiadis F, Ikonomidis I, Katsanos S, Bakosis G, Hatziagelaki E, Lekakis J, Mebazaa A, Parissis J. Acute heart failure with mid‐range left ventricular ejection fraction: clinical profile, in‐hospital management, and short‐term outcome. Clin Res Cardiol. 2017;106:359–368. [DOI] [PubMed] [Google Scholar]
- 16. Lam CSP, Gamble GD, Ling LH, Sim D, Leong KTG, Yeo PSD, Ong HY, Jaufeerally F, Ng TP, Cameron VA, Poppe K, Lund M, Devlin G, Troughton R, Richards AM, Doughty RN. Mortality associated with heart failure with preserved vs. reduced ejection fraction in a prospective international multi‐ethnic cohort study. Eur Heart J. 2018;39:1770–1780. [DOI] [PubMed] [Google Scholar]
- 17. Choi KH, Lee GY, Choi JO, Jeon ES, Lee HY, Cho HJ, Lee SE, Kim MS, Kim JJ, Hwang KK, Chae SC, Baek SH, Kang SM, Choi DJ, Yoo BS, Kim KH, Park HY, Cho MC, Oh BH. Outcomes of de novo and acute decompensated heart failure patients according to ejection fraction. Heart. 2018;104:525–532. [DOI] [PubMed] [Google Scholar]
- 18. Koh AS, Tay WT, Teng THK, Vedin O, Benson L, Dahlstrom U, Savarese G, Lam CSP, Lund LH. A comprehensive population‐based characterization of heart failure with mid‐range ejection fraction. Eur J Heart Fail. 2017;19:1624–1634. [DOI] [PubMed] [Google Scholar]
- 19. Rastogi A, Novak E, Platts AE, Mann DL. Epidemiology, pathophysiology and clinical outcomes for heart failure patients with a mid‐range ejection fraction. Eur J Heart Fail. 2017;19:1597–1605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Choi DJ, Han S, Jeon ES, Cho MC, Kim JJ, Yoo BS, Shin MS, Seong IW, Ahn Y, Kang SM, Kim YJ, Kim HS, Chae SC, Oh BH, Lee MM, Ryu KH; KorHF Registry . Characteristics, outcomes and predictors of long‐term mortality for patients hospitalized for acute heart failure: a report from the Korean Heart Failure Registry. Korean Circ J. 2011;41:363–371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Ho KK, Anderson KM, Kannel WB, Grossman W, Levy D. Survival after the onset of congestive heart failure in Framingham Heart Study subjects. Circulation. 1993;88:107–115. [DOI] [PubMed] [Google Scholar]
- 22. Lee SE, Lee HY, Cho HJ, Choe WS, Kim H, Choi JO, Jeon ES, Kim MS, Kim JJ, Hwang KK, Chae SC, Baek SH, Kang SM, Choi DJ, Yoo BS, Kim KH, Park HY, Cho MC, Oh BH. Clinical characteristics and outcome of acute heart failure in Korea: results from the Korean Acute Heart Failure Registry (KorAHF). Korean Circ J. 2017;47:341–353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Schiller NB, Shah PM, Crawford M, DeMaria A, Devereux R, Feigenbaum H, Gutgesell H, Reichek N, Sahn D, Schnittger I, Silverman NH, Tajik AJ; American Society of Echocardiography Committee on Standards, Subcommittee on Quantitation of Two‐Dimensional Echocardiograms . Recommendations for quantitation of the left ventricle by two‐dimensional echocardiography. J Am Soc Echocardiogr. 1989;2:358–367. [DOI] [PubMed] [Google Scholar]
- 24. Butler J, Fonarow GC, Zile MR, Lam CS, Roessig L, Schelbert EB, Shah SJ, Ahmed A, Bonow RO, Cleland JG, Cody RJ, Chioncel O, Collins SP, Dunnmon P, Filippatos G, Lefkowitz MP, Marti CN, McMurray JJ, Misselwitz F, Nodari S, O'Connor C, Pfeffer MA, Pieske B, Pitt B, Rosano G, Sabbah HN, Senni M, Solomon SD, Stockbridge N, Teerlink JR, Georgiopoulou VV, Gheorghiade M. Developing therapies for heart failure with preserved ejection fraction: current state and future directions. JACC Heart Fail. 2014;2:97–112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Maciver DH, Townsend M. A novel mechanism of heart failure with normal ejection fraction. Heart. 2008;94:446–449. [DOI] [PubMed] [Google Scholar]
- 26. Cleland JGF, Bunting KV, Flather MD, Altman DG, Holmes J, Coats AJS, Manzano L, McMurray JJV, Ruschitzka F, van Veldhuisen DJ, von Lueder TG, Bohm M, Andersson B, Kjekshus J, Packer M, Rigby AS, Rosano G, Wedel H, Hjalmarson A, Wikstrand J, Kotecha D; Beta‐Blockers in Heart Failure Collaborative Group . Beta‐blockers for heart failure with reduced, mid‐range, and preserved ejection fraction: an individual patient‐level analysis of double‐blind randomized trials. Eur Heart J. 2018;39:26–35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Lund LH, Claggett B, Liu J, Lam CS, Jhund PS, Rosano GM, Swedberg K, Yusuf S, Granger CB, Pfeffer MA, McMurray JJV, Solomon SD. Heart failure with mid‐range ejection fraction in CHARM: characteristics, outcomes and effect of candesartan across the entire ejection fraction spectrum. Eur J Heart Fail. 2018;20:1230–1239. [DOI] [PubMed] [Google Scholar]
- 28. Pitt B, Pfeffer MA, Assmann SF, Boineau R, Anand IS, Claggett B, Clausell N, Desai AS, Diaz R, Fleg JL, Gordeev I, Harty B, Heitner JF, Kenwood CT, Lewis EF, O'Meara E, Probstfield JL, Shaburishvili T, Shah SJ, Solomon SD, Sweitzer NK, Yang S, McKinlay SM; TOPCAT Investigators . Spironolactone for heart failure with preserved ejection fraction. N Engl J Med. 2014;370:1383–1392. [DOI] [PubMed] [Google Scholar]
- 29. Solomon SD, Claggett B, Lewis EF, Desai A, Anand I, Sweitzer NK, O'Meara E, Shah SJ, McKinlay S, Fleg JL, Sopko G, Pitt B, Pfeffer MA; TOPCAT Investigators . Influence of ejection fraction on outcomes and efficacy of spironolactone in patients with heart failure with preserved ejection fraction. Eur Heart J. 2016;37:455–462. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Table S1. Standardized Differences of Variables Used in PS Matching and IPW Adjustment to Adjust for Baseline Differences
Table S2. Comparison of KorHF and KorAHF Populations
Figure S1. Comparison of all‐cause mortality at 3 years according to use of guideline directed medical therapy among patients with heart failure with preserved ejection fraction.
Figure S2. Comparison of all‐cause mortality at 3 years according to use of guideline directed medical therapy among patients with heart failure with reduced ejection fraction.
Figure S3. Comparison of all‐cause mortality at 3 years according to use of digoxin and nitrate among patients with heart failure with mid‐range ejection fraction.


