Abstract
An integrated exposomic view of the relation between environment and cardiovascular health should consider the effects of both air and non-air related environmental stressors. Cardiovascular impacts of ambient air temperature, indoor and outdoor air pollution were recently reviewed. We aim, in this second part, to address the cardiovascular effects of noise, food pollutants, radiation, and some other emerging environmental factors.
Road traffic noise exposure is associated with increased risk of premature arteriosclerosis, coronary artery disease, and stroke. Numerous studies report an increased prevalence of hypertension in people exposed to noise, especially while sleeping. Sleep disturbances generated by nocturnal noise are followed by a neuroendocrine stress response. Some oxidative and inflammatory endothelial reactions are observed during experimental session of noise exposure. Moreover, throughout the alimentation, the cardiovascular system is exposed to persistent organic pollutants (POPs) as dioxins or pesticides, and plastic associated chemicals (PACs), such as bisphenol A. Epidemiological studies show positive associations of exposures to POPs and PACs with diabetes, arteriosclerosis and cardiovascular disease incidence. POPs and PACS share some abilities to interact with nuclear receptors activating different pathways leading to oxidative stress, insulin resistance and angiotensin potentiation. Regarding radiation, survivors of nuclear explosion have an excess risk of cardiovascular disease. Dose-effect relationships remain debated, but an increased cardiovascular risk at low dose of radiation exposure may be of concern. Some emerging environmental factors like electromagnetic fields, greenspace and light exposure may also require further attention.
Non-air related environmental stressors also play an important role in the burden of cardiovascular disease. Specific methodologies should be developed to assess the interactions between air and non-air related pollutants.
INTRODUCTION
Influences of environmental exposures on cardiovascular health are a growing concern. We recently reviewed the effects of indoor and outdoor air pollutants in Trends in Cardiovascular Medicine (1). To summarize, we documented a large body of epidemiological evidence supporting a strong link between air pollution exposure and cardiac clinical outcomes, such as myocardial infarction, stroke, heart failure, and cardiovascular mortality. We also presented some evidence from laboratory studies analyzing the role of endothelial oxidative stress, vascular inflammation, and sympathetic system activation into the air pollution related cardiovascular toxicity.
We aim, in this second part, to offer a more global view on the ecology of the cardiovascular system by also covering the effects of non-air related pollutants. The rationale of such a review is that humans are exposed to a cocktail of pollutants, which may affect the cardiovascular health, and that this exposomic view should integrate both the effects on air and non-air related pollutants. Indeed, in Europe, the environmental burden of disease has been estimated in order to rank the influence of nine different environmental risk factors (2). The highest overall public health impact was found for outdoor and indoor air pollution, followed by traffic noise, and radon exposure. Chemicals, like dioxins, had a significant but lower influence. Consequently, we dedicate this review to present the evidence on the association between cardiovascular health and environmental exposure to noise, organic and plastic pollutants from food and ionizing radiation. Because environmental sciences evolves constantly, we also present newly considered potential environmental factors, which may be shown in future studies to affect cardiovascular health.
We keep the same structure as our review of the air related pollutants health effects. We first describe the nature and sources of pollutants including, if available, the World Health Organisation (WHO) guidelines. Then, we present data from epidemiological and pathophysiological studies. The Pubmed database was searched using a combination of terms related to exposures (“noise”, “food pollutants”, “radiation”, “greenness”, “light”, “electromagnetic fields”) and cardiovascular outcomes (“myocardial infarction”, “stroke”, “heart failure”, “arrhythmia”, “hypertension”, “arterosclerosis”). In addition to general population studies, the epidemiological evidence regarding the effects of some pollutants, like food pollutants or radiation, also comes from prospective follow up of accidental contamination of population or survivors of atomic bombings. Data from occupational exposure were not included in this review. To strengthen the plausible link emerging from epidemiological studies, we also performed a Pubmed search on laboratory data with a special focus on oxidative stress and the sympathetic nervous system.
I. NOISE POLLUTION
1.1. Background
Noise-induced annoyance appears to be an important environmental factor influencing cardiovascular health (3). Community noise, also called environmental noise, is defined as noise emitted by all sources, except noise at the workplace. Effect of traffic noise on cardiovascular health has been widely investigated whereas effects of noise from public work, industries and indoor sources are less known. Besides noise from road and rail traffic, a growing environmental concern is the increasing noise pollution from aircraft sources because of airport-increased activities in populated areas. Noise is expressed in decibel using a logarithmic scale to measure sound pressure levels (SPL). After weighting for different sensitivities of the human ear at different sound frequencies, noise is expressed in energy-equivalent average A-weighted SPL (LAeq). LNight denotes the LAeq during the night-time hours. The LDN (day-night level) index corresponds to a LAeq over a 24 h period with a 10 dB penalty for nocturnal noise exposure. The LDEN index (day-evening-night level) is very similar in nature to the LDN, but with the 5 dB added penalty for the evening period. The WHO considered that 30% of the European population is exposed to a LNight exceeding 55dB (4).
1.2. Epidemiology
1.2.1. Hypertension:
A meta-analysis of 24 studies on the relationship between road traffic noise and the prevalence of hypertension reported an odds ratio (OR) of 1.07 (95% CI: 1.02–1.12) per 10 dB increase in the range of 48–78 dB (5). A meta-analysis of five studies on the relationship between aircraft noise and the prevalence of hypertension reported an OR of 1.13 (95% CI: 1.00–1.28) per 10 dB increase in the LDEN range of 48–68 dB (6). For aircraft noise, the Hypertension and Exposure to Noise Near Airports (HYENA) study demonstrated a relationship between night-time aircraft noise exposure and the risk of hypertension among 4681 middle-aged people who had lived for at least five years near one of the seven major European airports (7). A 10 dB increase in night-time aircraft noise exposure was associated with an OR of 1.14 for hypertension (95% CI: 1.01–1.29). However, this link between aircraft noise and hypertension was only significant in men. Many other epidemiological studies confirmed that nocturnal noise exposure had a greater effect on cardiovascular health than day-time noise exposure. Railway noise has been less studied than noise from other means of transportation. In the Swiss SALPADIA 2 study (8), railway noise, particularly during the night, was found to be significantly associated with increased systolic blood pressure (0.84 mmHg, 95% CI: 0.22–1.46 mmHg per 10 dB increase in LDEN).
1.2.2. Arteriosclerosis:
The Heinz–Nixdorf Recall study examined the associations between a 1-year average particulate air pollution and traffic noise with the thoracic aortic calcification (TAC) score in more than 4200 participants (9). A greater traffic noise at night was associated with a higher TAC score. The change in TAC score per 5 dB increase in night-time traffic noise was 3.9% (95% CI: 0.0 – 8.0%) in a model adjusted for PM2.5 concentration, suggesting a clinically relevant and independent association between traffic noise at night and pre-clinical atherosclerosis.
1.2.3. Coronary heart disease and cardiovascular mortality:
A meta-analysis of 14 studies on the relationship between road traffic noise and the prevalence of CAD reported an OR of 1.08 (95% CI: 1.04–1.13) per 10 dB increase in the LDEN range of 53–78 dB (10). A meta-analysis of 10 studies on the relationship between aircraft noise and the prevalence of CAD reported an OR of 1.06 (95% CI: 1.04–1.08) per 10 dB increase in the LDEN range of 48–68 dB (11). A large cohort study in Switzerland reported a trend in increased mortality due to myocardial infarction with a hazard ratio of 1.3 (95% CI: 0.96–1.7) for individuals exposed to LDN≥60 dB (12). This association between residential exposure to aircraft noise and mortality from myocardial infarction was only significant for people who had lived in thesame highly exposed location for at least 15 years and was not found with mortality from other circulatory disease. In another study performed on populations surrounding Heathrow Airport (13), the relative risk of coronary heart disease was 1.21 (95% CI: 1.12–1.31) for individuals living in areas experiencing the highest levels of daytime aircraft noise compare to those experiencing the lowest levels (>63 dB vs. ≤51 dB).
1.2.4. Stroke:
Exposure to residential road traffic noise was also associated with a higher risk of stroke among people older than 64.5 years (RR=1.27 (95% CI: 1.13–1.43) per 10 dB increase of LDEN (14). When areas experiencing the highest levels of daytime aircraft noise around Heathrow Airport were compared to those experiencing the lowest levels (>63 dB vs ≤51 dB), the relative risk of hospital admissions for stroke in adjusted model for confounding variables was 1.24 (95% CI 1.08–1.43) (13).
1.2.5. Dose-response relationship:
For the prevention of noise-induced cardiovascular health effects, the most recent review of evidence performed by the WHO in 2018 for the European region, indicates that the risk of ischemic heart disease increases continuously for road traffic noise levels from a LDEN of about 50 dB (15). Above this threshold, the shape of the association between road traffic noise and the incidence of CAD seems to follow a linear relationship (11). However, personal exposure to noise pollution cannot simply be assessed by sound level. Other acoustic characteristics are important, such as the frequency spectrum, the cumulative exposure to sound, and some psychoacoustic parameters. Consequently, for the same decibel levels, different noise sources may produce different individual effects and generate different exposure–response curves. When comparing the different meta-analyses presented above regarding the effects of noise from road traffic and aircraft on ischaemic heart diseases, the excess of risk moves from 7 to 15% per 10 dB increase in equivalent noise levels, respectively (16). This suggests that for the same level of dB, aircraft noise may be more disturbing than traffic noise. Future studies are needed to confirm the exposure-response relationship, detect the exact threshold of cardiovascular effects appearance, and estimate the effect of multiple noise sources (e.g., aircraft, traffic).
1.2. Pathophysiology with focus on sympathetic nervous system and oxidative stress
Few studies have examined the noise-mediating mechanisms leading to vascular dysfunction and why nocturnal noise exposure had a greater effect on cardiovascular health than day-time noise exposure (17). The effects of night-time aircraft noise exposure on the endothelial function were measured in a group of healthy volunteers (n=75) by using nocturnal aircraft noise played back with loudspeakers at 60 dB. The endothelial function of the brachial artery decreases as noise exposure increases (18). Furthermore, the effect of noise was more pronounced in individuals with previous exposure to noise, suggesting a cumulative effect of repeated noise exposure. In a small subgroup of this controlled study (n=5), the antioxidant effect of Vitamin C was able to reverse the noise-induced endothelial dysfunction which suggests an oxidative vascular reaction following noise exposure (18). A similar study design applied to patients (n=60) with high risk of coronary artery disease also concluded that noise exposure also affects the vascular endothelial function (19).
These noise-related changes in arterial endothelial function were initially attributed to the release of stress hormones, which are able to act directly on the endothelial surface. Indeed, nocturnal noise is followed by stress responses characterized by a release of catecholamines and an imbalance of the autonomic nervous system in favor of an increased sympathetic tone. The sympathomimetic effects of noise exposure are likely triggered indirectly by the limbic system as part of an emotional reaction to a perceived discomfort and sleep disturbance. Another possibility is a more direct non-conscious physiological interaction between the auditory system and the central regulatory structure of autonomic nervous system (16). Indeed, activation of some sensitive or sensorial afferent projection on the centralnervous system may produce sustained changes in autonomic reflexes ensuring cardiovascular homeostasis as baroreflex or chemoreflex (20,21). Furthermore, this direct pathway might be more sensitive and may explain the occurrence of cardiovascular reaction with relatively low noise levels exposure. Beside sympathetic reactions, animal studies also suggest a release of cortisol and an increase in inflammatory markers (17).
1.3. Effect of mitigation maneuvers
Individual strategies, like hearing protection devices or other mitigation measures like facade noise insulation with double/triple glassing can reduce noise annoyance and sleep disturbance (22). However, studies examining the usefulness of such strategies to prevent noise related cardiovascular disease are still lacking. As previously done with smoking bans and more recently with diesel ban to control indoor and outdoor air pollution, some cities have tried to reduce nocturnal noise exposure by bans on night flights. However, contrary to air pollution, a positive impact of noise bans on cardiovascular health is not yet proved. However, WHO estimates that a 5 dB noise reduction would reduce the prevalence of hypertension by 1.4% and coronary artery disease by 1.8%. The annual economic benefit of such intervention was estimated to be 3.9 billion for the US (23).
II. POLLUTANTS FROM FOOD
2.1. Background
Because of the propagation and bioaccumulation of chemical pollutants in the food chain, food is a major source of toxic chemical contaminant exposure for humans (24). One major characteristic of this type of pollution concerns its biological persistence because of the resistance of these molecules to transformation or degradation. Persistent organic pollutants (POPs), such as polychlorinated biphenyls (PCB), dioxins, and pesticides, are important food hazards. Dichlorodiphenyltrichloroethane (DDT) is perhaps one of the most famous and controversial pesticides used extensively on agricultural crops. Dioxins occur as a complex mixture of different molecules presented in food and feed, and tetrachlorodibenzodioxin (TCDD) is the most toxic member of this family. More than 90% of human exposure to POPs arises from food, mainly meat, dairy products, fish, and shellfish (25). Global analysis of seafood found that concentrations and bioaccumulation of POPs differ among species and across ocean basins world (26). POPs are now being monitored in human milk according to a WHO recommendation and a recent worldwide analysis of this database showed that the highest levels of PCB are observed in East and West Europe (27). A second category of food pollutants is plastic-associated chemicals (PACs), such as Bisphenol A and phthalates. Bisphenol A is a chemical product used primarily as a monomer in the production of polycarbonate plastic and epoxy resins. Human exposure assessment study has shown that BPA is present at detectable levels in urine of over 90% of individuals of US population (28). Other inorganic food pollutants include mercury, lead, cadmium, nitrates and ammonia.
In 2004, the Stockholm Convention on POPs was ratified to decrease environmental and human exposure to POPs (29). Toxic equivalency factors (TEFs) are now international, and all dioxins are classified according to their toxic potency relative to the referent compound, namely TCDD. These international TEFs have been developed for application in risk assessment but numerous discrepancies exist internationally for accepted maximum intake levels of contaminants in food. A recent study performed in the Netherlands, a country affected by repeated food dioxin contamination crisis, shows that 8% of the population is exposed to intake levels above the “safe” weekly intake for dioxins (30).
2.2. Epidemiology
2.2.1. Cardiovascular morbidity and mortality:
In a cross-sectional analysis from the US National Health and Nutrition Examination Survey (NHANES) 1999–2002, concentrations of PCBs and pesticides in serum were positively related to cardiovascular disease prevalence (31). NHANES (2003–2004) demonstrated that Bisphenol A levels in human urine were also positively associated with cardiovascular disease prevalence, with an of 1.6 (95% CI: 1.1–2.2) for a change of 1 standard deviation (SD) in Bisphenol A levels in urine (32). Further analysis of the NHANES 2005–2006 study confirmed these associations of PCBs and Bisphenol A with cardiovascular disease, but no clear distinction was made between different cardiovascular disorders. A large prospective population-based study is still required to provide high-level evidence of the independent roles of POPs and PACs on cardiovascular morbidity and mortality.
2.2.2. Arteriosclerosis:
Mono-methyl phthalate and a number of PCBs were related to the occurrence of carotid plaques, even after adjustment for lipids and known cardiovascular disease risk factors. Furthermore, levels of highly chlorinated PCBs were related to lipid infiltration into the intima space.
2.2.3. Diabetes:
Exposure to POPs, and especially DDT, has consistently been associated with diabetes in numerous prospective studies. Other studies show significant associations between PCB exposure and future diagnosis of diabetes. These data are largely supported by cohort data from the population surrounding Seveso (Italy), which suffered a POPs contamination in 1976 (33). In the previously mentioned cohort from Uppsala, Sweden, a number pesticides were found to be associated with diabetes prevalence (34). Exposure to PCB was a stronger risk factor for incidental diabetes than the body mass index (BMI), with a respective OR of 7.5 (95% CI: 1.4–38) for PCBs and 4.5 (95% CI: 1.2–22) for BMI. In the NHANES study of 2003–2004, the RR of diabetes was 1.3 (95% CI: 1.2–1.6) for a standard deviation change in Bisphenol A levels in urine (32).
2.2.4. Hypertension and other CV risk factors:
In the NHANES 1999–2002 study, the for hypertension was 2.3 (95% CI: 0.8–6.6) for the highest quartile of a number of PCBs in women (35). The same survey showed that the RR for newly diagnosed hypertension was 5 (95% CI: 1.2–21.5) for the highest quartiles of dioxin concentration, but this association was only present in women. In the Anniston cohort (Alabama, USA), which suffered a PCB contamination, a cross-sectional analysis suggested also an increased risk of hypertension (36). Recently, environmental exposure to beta-hexachlorocyclohexane, a lipophilic by-product of the production of the insecticide called lindane, has been associated with higher systolic blood pressure among people living close to an industrial area (37). The association between POCs and obesity, metabolic syndrome and dyslipidaemia is mainly based on cross-sectional data and requires further investigations to be confirmed.
2.2.5. Coronary artery disease and stroke:
In the cross-sectional analysis of the combined NHANES 2003–2004 and 2005–2006 data, the RR of myocardial infarction was 1.2 (95% CI: 1.1–1.4) for a change of 1 SD of the Bisphenol A levels in urine (38). A longitudinal study from the European Perspective Investigation of Cancer (EPIC) cohort also assessed the effects of BPA on the development of coronary artery disease and found an incident coronary artery disease diagnosis OR of 1.143 (IC 95%: 1.02–1.24) per standard deviation increase in urinary BPA concentration (39). In another study, which used coronary angiography results as end-points, the OR for multi-vessel coronary disease was 2.09 (CI 95%: CI 1.62–3.46) with higher levels of BPA in urine, but there was no significant association with less severe coronary artery disease (40). Considering the risks of coronary artery disease and strokes triggered by POP exposure, OR were respectively of 1.36 (95% CI: 1.18–1.56) and 1.15 (95% CI: 1.05–1.26) in a population study of people living close to a POP-contaminated waste site in New York (41)(42). Role of dioxin in the risk of developing CAD has been mostly investigated in occupational medicine and evidence from environmental research needs to be strengthened.
2.3. Pathophysiology with focus on oxidative stress
The accumulation of experimental evidence supports the findings that POPs and PACs increase the incidence of cardiovascular risk factors, such as diabetes, arterial hypertension, and vascular dysfunction. POPs have the ability to interact with sex hormone receptors with both agonist and antagonist properties. Other steroid nuclear receptors can also be affected by POPs. Dioxin interacts with the aryl hydrocarbon receptor (AhR), which promotes reactive oxygen species production through the cytochrome P450 enzyme CYP1A1 and increases 8-hydroxydeoxyguanosine, a product of DNA-based oxidation (43). When stimulated, this pathway leads to an activation of cyclooxygenase 2, which releases prostaglandins and eicosanoids, both acting as mediators of inflammation and atherothrombotic phenomenon (44). In TCDD-exposed mice aorta, an increased superoxide anion production and impairment of endothelium-dependent vasorelaxation were observed. In addition to dioxin-mediated endothelial dysfunction, the AhR pathway activation by TCDD increased blood pressure and induced cardiac hypertrophy (45). The treatment of macrophages by TCDD leads to AhR-dependent activation of inflammatory mediators and the formation of cholesterol-laden foam cells (46). The exposure of ApoE−/− mice to TCDD also caused a time-dependent progression of atherosclerosis through the stimulation of a G protein-coupled receptor, called CXCR2, by Interleukin 8 (IL-8) (46). The relation between POPs and diabetes has also been studied in animals. For instance, insulin secretion levels in mice was found to be significantly decreased by islets of Langerhans following TCDD exposure throughout a pathway involving the AhR (47). An increase in sensitivity towards angiotensin II was reported in animals and may be considered as a potential mechanism of POP-related arterial hypertension.
BPA is an endocrine disrupting chemical. As early as the 1930s, it was found to produce estrogenic activities. Mice exposed to Bisphenol A developed high blood pressure with increased level of angiotensin II (48). A vascular oxidative stress reaction was also observed with an eNOS-dependent superoxide production followed by an impairment of endothelium-dependent vasorelaxation (49).
2.4. Effect of mitigation maneuvers
Compared to conventional foods, organic food includes lower pesticide levels and may also present some higher antioxidant capacity. Some long-term cohort studies, as the Nutrinet- Santé study, demonstrated that organic food consumers present a decrease risk of hypertension, hypercholesterolemia, type 2 diabetes, and that organic food intake is associated with a reduced cardiovascular disease in men (50). Even if this study has performed numerous adjustments for age, education, occupation, smoking and physical activity level, a confounding effect of general life style between organic and conventional food consumers cannot be formerly ruled out. Strong evidence arising from controlled randomized study between organic or conventional diets, is still missing. Consequently, the ability of organic food consumption to prevent chronic disease arising from POCs and PACs exposure, such as diabetes or cardiovascular disease, requires further investigations.
Concerning people already exposed, in vitro study suggests that vitamin C and vitamin D intake may decrease the cardiovascular toxicity of PCBs (51). Detoxification by retrieving these persistent pollutants accumulated in fatty tissue is challenging and may require development of specific compounds, such as liposome and nano-carriers technologies (51).
III. IONIZING RADIATION
3.1. Background
Ionizing radiation carries sufficient energy to ionize atoms and molecules and break chemical bonds. These radiations take the form of electromagnetic waves (gamma or X-rays) or particles (neutrons, beta, or alpha). Radiation comes from different sources, including more than 60 natural sources such as radon, a naturally occurring gas emanating from rock and soil. Human exposure to radiation also comes from human-made sources. Medical use of radiation currentl accounts for 98% of the population’s dose contribution from all artificial sources (52). Massive releases of high nuclear decay rates of radioactive material into the environment after the Chernobyl (1986) and Fukushima (2011) nuclear power plant accidents were estimated at 5200 and 900 PentaBecquerel, respectively. Similar to POCs, iodine, caesium, and strontium radionuclides can also accumulate in the food chain. The effective dose received by humans is expressed in Sievert (Sv), a unit that takes into account the dose of absorbed radiation, usually expressed in Gray (Gy), but calibrated by the type of radiation and sensitivity of tissues and organs. The International Commission on Radiological Protection recommends, for public exposure, a maximum permissible dose of 1 millisievert per year, averaged over five years (53). High radiation doses induce a variety of non-cancer effects, such as damage to the structures of the heart valve, coronary, carotid, and other large arteries. An association between lower doses (< 0.5 Gy) and late circulatory diseases has recently been suggested (54).
3.2. Epidemiology
3.2.1. Cardiovascular morbity-mortality:
An excess risk of other non-cancer diseases, and especially circulatory diseases, was demonstrated among Japanese survivors with a clear correlation between the radiation dose received from the explosions. In the Life Span Study on Japanese bomb survivors, the ERR/Sv was 0.17 (90% CI: 0.08–0.26) for heart disease and 0.12 (90% CI: 0.02–0.22) for stroke (55). In a recent meta-analysis of studies on circulatory diseases from exposure to low-level ionizing radiation in the general population, the ERR/Sv for ischemic heart disease was 0.10 (95% CI: 0.04–0.15) and 0.21 (95% CI: 0.02–0.39) for stroke and 0.19 (95% CI: 0.00–0.38) for other circulatory diseases (56). Population-based excess absolute risk (EAR) estimates for the risk of exposure-induced death (REID) for all circulatory diseases ranged from 2.50%/Sv (CI: 0.77–4.22) in France to 8.51%/Sv (95% CI: 4.00–13.02) in Russia, reflecting the differentiations in the baseline prevalence of cardiovascular disease between these countries. Interestingly, the EAR of mortality from circulatory diseases are similar to those of radiation-induced cancer. This is in accordance with recent analyses of the Japanese atomic-bomb survivors, which have suggested that excess mortality from non-cancer diseases was comparable to that from cancer (57). The consequence of such findings is that the overall excess risk of mortality after exposure to low doses of radiation may be about twice the currently assumed level expected from the estimated risk of mortality due only to cancers.
3.2.2. Dose-effect relationship:
The debate on the existence of a threshold in the appearance of radiation-related cardiovascular effects is important in ascertaining whether these vascular effects are stochastic, like the association between radiation exposure and cancer, or non-stochastic, like the dose-dependent effect of radiation on skin. A threshold of 500 mSv would be compatible with epidemiological data on circulatory disease after radiation exposure. A review performed by the Public Health England endorsed the excess risks for ischemic heart disease and stroke, but concluded that a significantly elevated risk was detectable only for exposures above 500 mGy (58). However, the meta-analysis of studies with cumulative doses <500 mSv, or low dose rates of <10 mSv per day, estimated a significant dose-risk relationship with circulatory disease mortality (56). Consequently, other groups consider that a raised risk is apparent below this threshold and that the dose-response is compatible with a linear non-threshold relationship, as encountered with radiation-related cancer. Indeed, in the Life Span study, radiation effects on non-cancer mortality is described by a linear dose-response model with a risk increase of about 14% per Sv (55). This dose-effect relationship between radiation and cardiovascular disease remains debatable, and only further investigations using a precise evaluation of confounding lifestyles may change our understanding of the risks of relative low radiation exposure levels.
3.3. Pathophysiology with focus on endothelial function
An underlying causal relationship between low level radiation and circulatory disease is supported by animal research. A great number of studies have investigated radiation effects on endothelial cells in vitro, and a few studies have confirmed some of these observations in vivo (59). Myocardial perfusion in humans was studied after breast cancer radiotherapy revealed inhomogeneities and local defects in myocardial perfusion, suggesting an endothelial dysfunction in the myocardial microcirculation (65). An inflammatory phenotype is expressed by endothelial cells exposed to a dose of >0.5 Gy with a transient over-expression of adhesion molecules such as E-selectin, ICAM −1 and cytokines, including IL-6, IL-8 and TGF-ß (60,61). An increased transcription factor NFkB has also been reported after radiation exposure, and as demonstrated in other models, arteriosclerosis may play a pivotal role in radiation-induced vascular inflammation. Furthermore, irradiated endothelial cells release the von Willebrand factor, which may favor a prothrombotic reaction (62). Radiation-induced arteriosclerosis has been confirmed in the APOE−/− mouse (63). Local radiation of the carotid arteries of these mice with a single dose of 14 Gy caused an earlier onset and faster growth of arteriosclerotic plaques in the irradiated vessels. A histological study revealed also increased signs of plaque instability, such as intra-plaque haemorrhage or macrophage accumulation in the irradiated carotid arteries. Further investigation may determine whether a low dose total body irradiation might modulate atherosclerosis progression, considering pre-existing disease and other cardiovascular risk factors at time of exposure.
IV. EMERGING ENVIRONMENTAL FACTORS
4.1. Electromagnetic fields (EMFs):
During the last decades, exposure to electromagnetic fields in daily life has been on the increase. Low frequency EMFs have frequencies up to 300 Hz and exposure of population is mainly arising from electrical appliances, household wiring, and electric power lines. Numerous electrical systems, like Bluetooth, wi-fi, mobile phones, generates EMFs in a higher frequency range between 3KHz and 300 GHz. Due to tissue penetration, high frequency EMFs can physically impact cells and tissue function by triggering a thermal effect. Existence of non-thermal effects associated with low frequency EMFs remains not proved. In 2011, WHO classified radio electromagnetic fields into a category 2B as potentially carcinogenic. EMFs were first considered a concern in cardiology for patients with pacemakers and defibrillators. The heating of the implant, disturbances of the electronic circuit and of the sensing capabilities have been problematic with first-generation cell phones and older generations of implantable devices. More recently, some cardiovascular effects of electromagnetic fields have been suspected from studies demonstrating an altered heart rate variability with low frequency magnetic field exposure, suggesting some potential effects on the autonomic nervous system (64). However, interventional studies on healthy volunteers tried to replicate this finding with variables results. Some studies have shown an increase in activity in the sympathetic nervous s stem’s exposure to MF while others have shown an increase in activity of the parasympathetic nervous system or no effect (64). Thus, based on these conflictual studies, the possible deleterious cardiovascular health effects of EMFs and the characterization of potentially subtle physiological changes in the autonomic nervous system warrant further studies to be confirmed.
4.2. Green space:
In the opposite direction than the previously presented environmental stressors, greenness appears recently as a new factor, which may favorably influence the cardiovascular system. In one of the first reports, which was based on an analysis of the entire population of England, the rate of cardiovascular disease mortality in the least green areas was found to be twice that of greenest areas (65). Such a finding was not confirmed by another study performed on the population living in the 49 largest US cities, in which no association between greenness and mortality from heart disease was found (66). In a meta-analysis of 8 studies on the effect of greenness, no significant reduction of cardiovascular mortality risk was observed (RR 0.99; 95%CI 0.98–1.00)(67). An independent effect of green space remains difficult to prove because of the association between greenness environment and decreased levels of local air and noise pollutions. A recent study performed on the population of Switzerland has tried to overcome this limitation by discriminating the effects of greenness from other confounding variables. Based on satellite imaging data, this study shows that, even after adjustment for air pollution, traffic noise and other socio-economic confounders, greenness surrounding the living address was associated with a reduced cardiovascular mortality (RR 0.96; 95% CI: 0.95–0.98) (68). Further studies are required to confirm the independent benefit of greenness by including also as covariables the level of physical activity, which may also indirectly explain some parts of the benefit of green spaces.
4.3. Light pollution:
Some studies reported that exposure to light at night are accompanied by neuroendocrine reactions as impairment of melatonin release (69). Furthermore, evening light exposure before bedtime may affect circadian phase more than exposure during the afternoon. Chronic light exposure at night exposure may have negative effects on the psychological, cardiovascular and/or metabolic functions. Recent increasing exposure to blue light from light-emitting diodes screen has raised some concerns on possible circadian rhythm disruption and related increase in cardiovascular risk. However, exposure assessment is complex because numerous parameters (e.g., light intensity, wavelength, time, and duration of exposure) need to be considered to study the cardiovascular health effect of blue light exposure at night.
V. METHODOLOGICAL ISSUES FOR AN INTEGRATED APPROACH OF THE ECOLOGY OF THE CARDIOVASCULAR SYSTEM
Most of the presented studies report an independent effect of one pollutant on different cardiovascular events. Such an approach does not fit the multifactorial nature of air pollution and the ecological possibility of an interaction between pollutants. Indeed, the reality is that humans are not exposed to one pollutant at a time but to a simultaneous “cocktail” of chemicals, particles, noise, and electromagnetic waves (figure 1). Furthermore, some humans may be more vulnerable to the effect of environment because of pre-existing cardiovascular risk factors. A statistical approach considering such multi-factorial mixtures of environmental pollutants, and patient predisposition, requires insights from classical experimental design, complex models, and a very large population study. Few studies have performed a multi-pollutant approach on cardiovascular risk. In a study regarding the risk to develop thoracic aortic calcification, a bi-pollutant model was used to uncover the independent effect of air and noise pollution. After mutual adjustment, this study concluded that long-term exposure to fine PM and night-time traffic noise are both independently associated with subclinical atherosclerosis (9). However, numerous other interactions between air and non-air pollutants are possible and need to be explored (figure 1). As previously mentioned, greenness may interfere with air and noise pollution. Another possible interaction is that the autonomic mediated stress reaction observed with noise pollution may also interfere with change in circadian rhythm observed with light-emitting diode. Even more speculative, magnetite nanosphere arising from air pollution may also interfere with electromagnetic field in generating oxidative stress reaction (70). Interaction between air and food pollutions and numerous other hypothetical scenarios of interactions are possible and will need to be investigated.
Figure 1: Exposomic view of the effects of environmental stressors on the cardiovascular system.
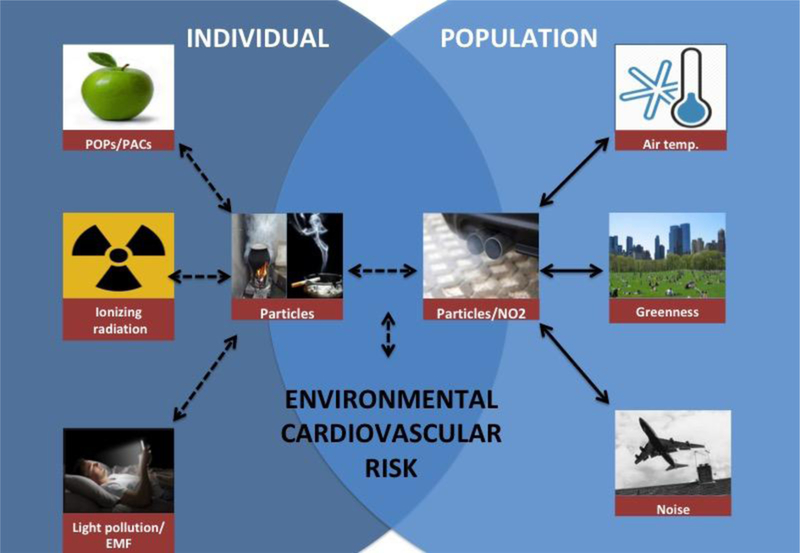
Solid arrows represent interactions between pollutants already studied whereas dashed arrows represent interactions that remain to be investigated.
Abbreviations: NO2: Nitrogen dioxide; Air temp.: Air temperature; POPs: Persistent Organic Pollutants, PACs: Plastic Associated Chemicals; EMF: Electromagnetic fields
Analyse of the interactions between air and non-air related pollutant is a difficult task because of the high correlations often existing between pollutants. Indeed, evaluating the independent effect of a pollutant from each other can be violated in the presence of collinearity. General linear model analysis has been largely used to address the environmental health effects of air pollution and noise. However, considering a statistical analysis with various air and non-air pollutants, cardiovascular risk factors, socio-economic subgroups leads to a high dimensional general linear model, and considering interactions between pollutants and cardiovascular risk factors further increases this dimensionality. The high dimensionality of such general linear model is prone to decrease inference, correct interpretation of the model parameters and possible prediction. At least, analysis of air temperature and pollutants effects requires a temporal approach of the lag effect. An optimal method to screen for the interaction between air and non-air related pollutants has not yet been clearly identified. Some techniques used for high dimensional regression problems, like the Least Absolute Shrinkage and Selection Operator (LASSO) method, may be translate in a next future to epidemiological studies of environmental risk factors (71). Only large-scale studies, integrating a maximum number of pollutant exposures into complex high dimension models, may be able to build a global scoring of the individual cardiovascular risk generated by exposures to environmental stressors (figure 2).
Figure 2: Fictive evaluation of individual cardiovascular risk arising from environmental stressors.
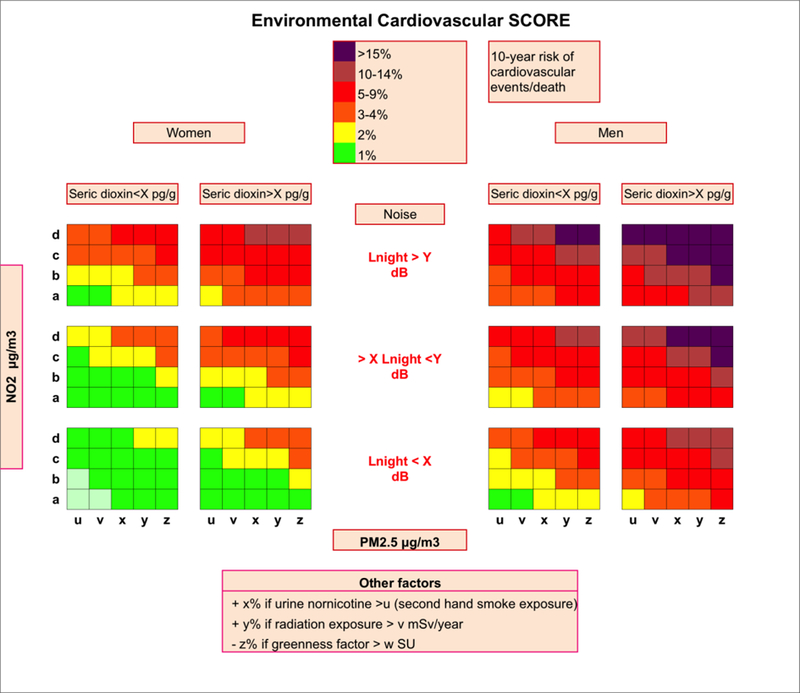
This figure is a facsimile of ESC SCORE Risk charts assessing the global risk of cardiovascular death related to conventional risk factor but transposed to environmental stressors.
Abbreviations: CV: cardiovascular, PM: Particle matter; NO2: Nitrogen dioxide; dB: decibel, LNight: noise energy equivalent at night
GENERAL CONCLUSION ON THE ECOLOGY OF CARDIOVASCULAR SYSTEM
The cardiovascular system is vulnerable to both air and non-air related pollutant throughout common physiopathological pathways involving endothelial oxidative stress and autonomic system imbalance. Deterioration of environment has an impact on cardiovascular health and, as presented in the first part of this review, exposures to extreme temperatures and decrease air quality are likely the main contributors to environmental mediated cardiovascular mortality and morbidity. Noise from road traffic also has shown some important effects on coronary artery disease, whereas effect of aircraft noise seems strongly associated with hypertension. Chemical pollutions from food have been associated with premature occurrence of diabetes and hypertension, but effects on coronary disease or stroke differ across studies. Low dose exposure to ionizing radiation also seems associated with cardiovascular disease. Interestingly, most of these environmental triggers display no clear exposure threshold before observing adverse cardiovascular effects. This concept that low levels of exposure may already generate a significant cardiovascular health effect strengthens the need of studies examining the synergistic effects of multiple pollutant exposures.
Acknowledgments
T. Bourdrel is a member of the Strasbourg Respire Association. Research reported in this publication was supported by the John Harvard Distinguished Science Fellows program within the FAS Division of Science of Harvard University and by the Office of the Director, National Institutes of Health under Award Number DP5OD021412. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Disclosure of conflicts of interest:
The authors declare no competing financial interests.
REFERENCES
- 1.Argacha JF, Bourdrel T, van de Borne P. Ecology of the cardiovascular system: A focus on air-related environmental factors. Trends Cardiovasc Med 2018. February;28(2):112–26. [DOI] [PubMed] [Google Scholar]
- 2.Hänninen O, Knol AB, Jantunen M, Lim T-A, Conrad A, Rappolder M, et al. Environmental burden of disease in Europe: assessing nine risk factors in six countries. Environ Health Perspect National Institute of Environmental Health Science; 2014. May;122(5):439–46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Münzel T, Gori T, Babisch W, Basner M. Cardiovascular effects of environmental noise exposure. Eur Heart J 2014. April;35(13):829–36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Fritschi L, Brown AL, Kim R, Schwela D, Kephalopoulos S. Burden of disease from environmental noise: Quantification of healthy life years lost in Europe. World Health Organization World Health Organization; 2011.
- 5.van Kempen E, Babisch W. The quantitative relationship between road traffic noise and hypertension: a meta-analysis. J Hypertens 2012. June;30(6):1075–86. [DOI] [PubMed] [Google Scholar]
- 6.Babisch W, Kamp I van. Exposure-response relationship of the association between aircraft noise and the risk of hypertension. Noise Health Medknow Publications; 2009;11(44):161–8. [DOI] [PubMed] [Google Scholar]
- 7.Jarup L, Babisch W, Houthuijs D, Pershagen G, Katsouyanni K, Cadum E, et al. Hypertension and exposure to noise near airports: the HYENA study. Environ Health Perspect National Institute of Environmental Health Science; 2008. March;116(3):329– 33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Dratva J, Phuleria HC, Foraster M, Gaspoz J- M, Keidel D, Künzli N, et al. Transportation noise and blood pressure in a population-based sample of adults. Environ Health Perspect 2012. January;120(1):50–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kälsch H, Hennig F, Moebus S, Möhlenkamp S, Dragano N, Jakobs H, et al. Are air pollution and traffic noise independently associated with atherosclerosis: the Heinz Nixdorf Recall Study. Eur Heart J 2014. April;35(13):853–60. [DOI] [PubMed] [Google Scholar]
- 10.Babisch W Updated exposure-response relationship between road traffic noise and coronary heart diseases: A meta-analysis. Noise Heal 2014;16(68):1–9. [DOI] [PubMed] [Google Scholar]
- 11.Vienneau D, Schindler C, Perez L, Probst-Hensch N, Röösli M. The relationship between transportation noise exposure and ischemic heart disease: a meta-analysis. Environ Res 2015. April;138:372–80. [DOI] [PubMed] [Google Scholar]
- 12.Huss A, Spoerri A, Egger M, Röösli M, Swiss National Cohort Study Group. Aircraft noise, air pollution, and mortality from myocardial infarction. Epidemiology 2010. November;21(6):829–36. [DOI] [PubMed] [Google Scholar]
- 13.Hansell AL, Blangiardo M, Fortunato L, Floud S, de Hoogh K, Fecht D, et al. Aircraft noise and cardiovascular disease near Heathrow airport in London: small area study. BMJ 2013;347:f5432. [DOI] [PubMed] [Google Scholar]
- 14.Sørensen M, Hvidberg M, Andersen ZJ, Nordsborg RB, Lillelund KG, Jakobsen J, et al. Road traffic noise and stroke: a prospective cohort study. Eur Heart J 2011. March;32(6):737–44. [DOI] [PubMed] [Google Scholar]
- 15.van Kempen E, Casas M, Pershagen G, Foraster M. WHO Environmental Noise Guidelines for the European Region: A Systematic Review on Environmental Noise and Cardiovascular and Metabolic Effects: A Summary. Int J Environ Res Public Health 2018. February 22;15(2):379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Basner M, Babisch W, Davis A, Brink M, Clark C, Janssen S, et al. Auditory and non-auditory effects of noise on health. Lancet (London, England) Elsevier; 2014. April 12;383(9925):1325–32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Münzel T, Sørensen M, Gori T, Schmidt FP, Rao X, Brook FR, et al. Environmental stressors and cardio-metabolic disease: part II-mechanistic insights. Eur Heart J Oxford University Press; 2017. February 21;38(8):557–64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Schmidt FP, Basner M, Kroger G, Weck S, Schnorbus B, Muttray A, et al. Effect of nighttime aircraft noise exposure on endothelial function and stress hormone release in healthy adults. Eur Heart J 2013;34(45):3508–14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Schmidt F, Kolle K, Kreuder K, Schnorbus B, Wild P, Hechtner M, et al. Nighttime aircraft noise impairs endothelial function and increases blood pressure in patients with or at high risk for coronary artery disease. Clin Res Cardiol Springer Berlin Heidelberg; 2015. January 22;104(1):23–30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Perez CM, Hazari MS, Farraj AK. Role of Autonomic Reflex Arcs in Cardiovascular Responses to Air Pollution Exposure. Cardiovasc Toxicol 2015. January 15;15(1):69–78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Argacha JF, Xhaët O, Gujic M, De Boeck G, Dreyfuss C, Lamotte M, et al. Facial cooling and peripheral chemoreflex mechanisms in humans. Acta Physiol 2008;194(2). [DOI] [PubMed] [Google Scholar]
- 22.Amundsen AH, Klæboe R, Aasvang GM. Long-term effects of noise reduction measures on noise annoyance and sleep disturbance: The Norwegian facade insulation study. J Acoust Soc Am 2013. June;133(6):3921–8. [DOI] [PubMed] [Google Scholar]
- 23.Swinburn TK, Hammer MS, Neitzel RL. Valuing Quiet: An Economic Assessment of U.S. Environmental Noise as a Cardiovascular Health Hazard. Am J Prev Med 2015. September;49(3):345–53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Lind L, Lind PM. Can persistent organic pollutants and plastic-associated chemicals cause cardiovascular disease? J Intern Med 2012. June;271(6):537–53. [DOI] [PubMed] [Google Scholar]
- 25.WHO fact sheets. Dioxins and their effects on human health [Internet] 2016. [cited 2018 Apr 30]. Available from: http://www.who.int/en/news-room/fact-sheets/detail/dioxins-and-their-effects-on-human-health
- 26.Bonito LT, Hamdoun A, Sandin SA. Evaluation of the global impacts of mitigation on persistent, bioaccumulative and toxic pollutants in marine fish. PeerJ PeerJ Inc.; 2016. January 28;4:e1573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.van den Berg M, Kypke K, Kotz A, Tritscher A, Lee SY, Magulova K, et al. WHO/UNEP global surveys of PCDDs, PCDFs, PCBs and DDTs in human milk and benefit–risk evaluation of breastfeeding. Vol. 91, Archives of Toxicology 2017. p. 83– 96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Calafat AM, Kuklenyik Z, Reidy JA, Caudill SP, Ekong J, Needham LL. Urinary concentrations of bisphenol A and 4-nonylphenol in a human reference population. Environ Health Perspect 2005. April;113(4):391–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.United Nations Environment Programme. Stockholm Convention on Persistent Organic Pollutants 2004.
- 30.Baars AJ, Bakker MI, Baumann RA, Boon PE, Freijer JI, Hoogenboom LAP, et al. Dioxins, dioxin-like PCBs and non-dioxin-like PCBs in foodstuffs: occurrence and dietary intake in The Netherlands. Toxicol Lett 2004. June 15;151(1):51–61. [DOI] [PubMed] [Google Scholar]
- 31.Ha M-H, Lee D-H, Jacobs DR. Association between serum concentrations of persistent organic pollutants and self-reported cardiovascular disease prevalence: results from the National Health and Nutrition Examination Survey, 1999–2002. Environ Health Perspect 2007. August;115(8):1204–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Lang IA, Galloway TS, Scarlett A, Henley WE, Depledge M, Wallace RB, et al. Association of urinary bisphenol A concentration with medical disorders and laboratory abnormalities in adults. JAMA 2008. September 17;300(11):1303–10. [DOI] [PubMed] [Google Scholar]
- 33.Consonni D, Pesatori AC, Zocchetti C, indaco R, D’Oro LC, Rubagotti M, et al. Mortality in a population exposed to dioxin after the Seveso, Italy, accident in 1976: 25 Years of follow-up. Am J Epidemiol 2008. April 1;167(7):847–58. [DOI] [PubMed] [Google Scholar]
- 34.Lee D-H, Lind PM, Jacobs DR, Salihovic S, van Bavel B, Lind L. Polychlorinated biphenyls and organochlorine pesticides in plasma predict development of type 2 diabetes in the elderly: the prospective investigation of the vasculature in Uppsala Seniors (PIVUS) study. Diabetes Care 2011. August;34(8):1778–84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Ha M-H, Lee D-H, Son H-K, Park S-K, Jacobs DR. Association between serum concentrations of persistent organic pollutants and prevalence of newly diagnosed hypertension: results from the National Health and Nutrition Examination Survey 1999–2002. J Hum Hypertens 2009. April;23(4):274–86. [DOI] [PubMed] [Google Scholar]
- 36.Goncharov A, Bloom M, Pavuk M, Birman I, Carpenter DO. Blood pressure and hypertension in relation to levels of serum polychlorinated biphenyls in residents of Anniston, Alabama. J Hypertens 2010. October;28(10):2053–60. [DOI] [PubMed] [Google Scholar]
- 37.Bruno RM, Di Lascio N, Faita F, Sicari R, Fantini F, Blasetti F, Porta D, Narduzzi SFF. Environmental exposure to beta-hexachlorocyclohexane is associated with higher systolic blood pressure among people living close to an industrial area. Eur Heart J 2015;36(Abstract Supplement):710. [Google Scholar]
- 38.Melzer D, Rice NE, Lewis C, Henley WE, Galloway TS. Association of urinary bisphenol a concentration with heart disease: evidence from NHANES 2003/06. PLoS One 2010;5(1):e8673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Melzer D, Osborne NJ, Henley WE, Cipelli R, Young A, Money C, et al. Urinary Bisphenol A Concentration and Risk of Future Coronary Artery Disease in Apparently Healthy Men and WomenClinical Perspective. Circulation 2012;125(12). [DOI] [PubMed] [Google Scholar]
- 40.Melzer D, Gates P, Osborne NJ, Osborn NJ, Henley WE, Cipelli R, et al. Urinary bisphenol a concentration and angiography-defined coronary artery stenosis. PLoS One 2012;7(8):e43378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Shcherbatykh I, Huang X, Lessner L, Carpenter DO. Hazardous waste sites and stroke in New York State. Environ Health 2005;4:18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Sergeev AV, Carpenter DO. Hospitalization rates for coronary heart disease in relation to residence near areas contaminated with persistent organic pollutants and other pollutants. Environ Health Perspect National Institute of Environmental Health Science; 2005. June;113(6):756–61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Shertzer HG, Nebert DW, Puga A, Ary M, Sonntag D, Dixon K, et al. Dioxin Causes a Sustained Oxidative Stress Response in the Mouse. Biochem Biophys Res Commun Academic Press; 1998;253(1):44–8. [DOI] [PubMed] [Google Scholar]
- 44.Puga A, Hoffer A, Zhou S, Bohm JM, Leikauf GD, Shertzer HG. Sustained Increase in Intracellular Free Calcium and Activation of Cyclooxygenase-2 Expression in Mouse Hepatoma Cells Treated with Dioxin. Biochem Pharmacol 1997;54(12):1287–96. [DOI] [PubMed] [Google Scholar]
- 45.Kopf PG, Huwe JK, Walker MK. Hypertension, cardiac hypertrophy, and impaired vascular relaxation induced by 2,3,7,8-tetrachlorodibenzo-p-dioxin are associated with increased superoxide. Cardiovasc Toxicol 2008. December;8(4):181–93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Wu D, Nishimura N, Kuo V, Fiehn O, Shahbaz S, Van Winkle L, et al. Activation of aryl hydrocarbon receptor induces vascular inflammation and promotes atherosclerosis in apolipoprotein E−/− mice. Arterioscler Thromb Vasc Biol NIH Public Access; 2011. June;31(6):1260–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Kurita H, Yoshioka W, Nishimura N, Kubota N, Kadowaki T, Tohyama C. Aryl hydrocarbon receptor-mediated effects of 2,3,7,8-tetrachlorodibenzo-p-dioxin on glucose-stimulated insulin secretion in mice. J Appl Toxicol 2009. November;29(8):689–94. [DOI] [PubMed] [Google Scholar]
- 48.Saura M, Marquez S, Reventun P, Olea-Herrero N, Arenas MI, Moreno-Gómez-Toledano R, et al. Oral administration of bisphenol A induces high blood pressure through angiotensin II/CaMKII-dependent uncoupling of eNOS. FASEB J 2014. November;28(11):4719–28. [DOI] [PubMed] [Google Scholar]
- 49.Gao X, Wang H-S. Impact of bisphenol a on the cardiovascular system - epidemiological and experimental evidence and molecular mechanisms. Int J Environ Res Public Health Multidisciplinary Digital Publishing Institute (MDPI); 2014. August;11(8):8399–413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Baudry J, Méjean C, Péneau S, Galan P, Hercberg S, Lairon D, et al. Health and dietary traits of organic food consumers: results from the NutriNet-Santé study. Br J Nutr 2015. December 28;114(12):2064–73. [DOI] [PubMed] [Google Scholar]
- 51.Gupta P, Thompson BL, Wahlang B, Jordan CT, Zach Hilt J, Hennig B, et al. The environmental pollutant, polychlorinated biphenyls, and cardiovascular disease: a potential target for antioxidant nanotherapeutics. Drug Deliv Transl Res 2017. October 3; [DOI] [PMC free article] [PubMed]
- 52.UNSCEAR report. Sources and effects of ionizing radiation [Internet]. Vol. I 2008. [cited 2018 Apr 30]. Available from: http://www.unscear.org/unscear/en/publications/2008_1.html [Google Scholar]
- 53.The International Commission on Radiological Protection. Annals of the ICRP, Publication 103, The 2007 recommendations of the International Commission on Radiological Protection. Ann ICRP 2007;37(2–4):1–332. [DOI] [PubMed] [Google Scholar]
- 54.Wakeford R, Little MP. Epidemiological evidence for radiation-induced circulatorydisease – the Japanese atomic bomb survivors. EU cientific eminar 2008. “Emerging evidence for radiation induced circulator diseases”. Proceedings of a scientific seminar held in Luxembourg on 2009;158:21–31. [Google Scholar]
- 55.Preston DL, Shimizu Y, Pierce DA, Suyama A, Mabuchi K. Studies of mortality of atomic bomb survivors. Report 13: Solid cancer and noncancer disease mortality:1950–1997. Radiat Res 2003. October;160(4):381–407. [DOI] [PubMed] [Google Scholar]
- 56.Little MP, Azizova TV, Bazyka D, Bouffler SD, Cardis E, Chekin S, et al. Systematic review and meta-analysis of circulatory disease from exposure to low-level ionizing radiation and estimates of potential population mortality risks. Environ Health Perspect National Institute of Environmental Health Science; 2012. November;120(11):1503–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Ozasa K, Shimizu Y, Suyama A, Kasagi F, Soda M, Grant EJ, et al. Studies of the mortality of atomic bomb survivors, Report 14, 1950–2003: an overview of cancer and noncancer diseases. Radiat Res 2012. March;177(3):229–43. [DOI] [PubMed] [Google Scholar]
- 58.McMillan TJ, Bennett MR, Bridges BA, Hendry J, Jones B, Kanthou C, et al. “Circulator Disease Risk”, report of the independent Advisor Group on Ionising Radiation 2010.
- 59.Schultz-Hector S, Trott K-R. Radiation-induced cardiovascular diseases: is the epidemiologic evidence compatible with the radiobiologic data? Int J Radiat Oncol Biol Phys 2007. January 1;67(1):10–8. [DOI] [PubMed] [Google Scholar]
- 60.Van Der Meeren A, Squiban C, Gourmelon P, Lafont H, Gaugler MH. Differential regulation by IL-4 and IL-10 of radiation-induced IL-6 and IL-8 production and ICAM-1 expression by human endothelial cells. Cytokine 1999. November;11(11):831–8. [DOI] [PubMed] [Google Scholar]
- 61.Milliat F, François A, Isoir M, Deutsch E, Tamarat R, Tarlet G, et al. Influence of endothelial cells on vascular smooth muscle cells phenotype after irradiation: implication in radiation-induced vascular damages. Am J Pathol 2006. October;169(4):1484–95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Boerma M, Kruse JJCM, van Loenen M, Klein HR, Bart CI, Zurcher C, et al. Increased deposition of von illebrand factor in the rat heart after local ioni ing irradiation. trahlentherapie und Onkol Organ der Dtsch R ntgengesellschaft et al. 200 February;180(2):109–16. [DOI] [PubMed] [Google Scholar]
- 63.Stewart FA, Heeneman S, Te Poele J, Kruse J, Russell NS, Gijbels M, et al. Ionizing radiation accelerates the development of atherosclerotic lesions in ApoE−/− mice and predisposes to an inflammatory plaque phenotype prone to hemorrhage. Am J Pathol 2006. February;168(2):649–58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.McNamee DA, Legros AG, Krewski DR, Wisenberg G, Prato FS, Thomas AW. A literature review: the cardiovascular effects of exposure to extremely low frequency electromagnetic fields. Int Arch Occup Environ Health 2009. August 17;82(8):919–33. [DOI] [PubMed] [Google Scholar]
- 65.Mitchell R, Popham F. Effect of exposure to natural environment on health inequalities: an observational population study. Lancet (London, England) Elsevier; 2008. November 8;372(9650):1655–60. [DOI] [PubMed] [Google Scholar]
- 66.Richardson EA, Mitchell R, Hartig T, de Vries S, Astell-Burt T, Frumkin H. Green cities and health: a question of scale? J Epidemiol Community Health BMJ Publishing Group Ltd; 2012. February 1;66(2):160–5. [DOI] [PubMed] [Google Scholar]
- 67.Gascon M, Triguero-Mas M, Martínez D, Dadvand P, Rojas-Rueda D, Plasència A, et al. Residential green spaces and mortality: A systematic review. Environ Int 2016. January;86:60–7. [DOI] [PubMed] [Google Scholar]
- 68.Vienneau D, de Hoogh K, Faeh D, Kaufmann M, Wunderli JM, Röösli M. More than clean air and tranquillity: Residential green is independently associated with decreasing mortality. Environ Int 2017. November;108:176–84. [DOI] [PubMed] [Google Scholar]
- 69.Cho Y, Ryu S-H, Lee BR, Kim KH, Lee E, Choi J. Effects of artificial light at night on human health: A literature review of observational and experimental studies applied to exposure assessment. Chronobiol Int 2015. October 21;32(9):1294–310. [DOI] [PubMed] [Google Scholar]
- 70.Maher BA, Ahmed IAM, Karloukovski V, MacLaren DA, Foulds PG, Allsop D, et al. Magnetite pollution nanoparticles in the human brain. Proc Natl Acad Sci National Academy of Sciences; 2016. September 6;113(39):10797–801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Barrera-Gómez J, Agier L, Portengen L, Chadeau-Hyam M, Giorgis-Allemand L, Siroux V, et al. A systematic comparison of statistical methods to detect interactions in exposome-health associations. Environ Heal A Glob Access Sci Source 2017. December 14;16(1):74. [DOI] [PMC free article] [PubMed] [Google Scholar]


