Abstract
There has been no nationwide study of prognostic factors and outcomes in patients on peritoneal dialysis (PD) in Japan. We conducted a cohort study using data from the nationwide registry of the Japanese Society for Dialysis Therapy. We followed 8,954 prevalent PD patients for 2 years, 2014–2015. Cox proportional hazards regression analysis was used to determine factors that were independently associated with patient survival. Survival rates were compared between patients with and without diabetes after adjusting for potential confounders. During the 2-year study period, 893 (10.0%) of 8,954 patients died, 148 (1.6%) underwent kidney transplantation, and 2,637 (29.4%) were switched to hemodialysis; 5,276 (58.9%) patients were alive at the end of the study period. After multivariate adjustment, older age, longer duration of dialysis, presence of diabetes, cardiovascular comorbidity, use of 2.5% glucose dialysate, higher C-reactive protein and phosphate levels, and a lower serum albumin level were independently associated with increased hazard ratios for all-cause mortality. A combination of PD and hemodialysis was associated with a lower mortality rate. The new-onset cardiovascular event rate was significantly higher in the diabetes group than in the non-diabetes group (P < 0.0001). After adjusting for all variables, the hazard ratio was 1.509 (95% confidence interval 1.029–2.189, P = 0.036) in the diabetes group. Diabetes, older age, longer duration of dialysis, cardiovascular comorbidity, and inflammation were predictors of mortality in patients on PD.
Introduction
In Japan, the limited availability of kidney transplantation as a form of renal replacement therapy means that most patients with end-stage kidney disease (ESKD) require lifelong dialysis. Therefore, the amount of time spent on hemodialysis (HD) or peritoneal dialysis (PD) is longer in Japan than in the US or European countries. The number of Japanese patients on long-term dialysis has been steadily increasing and reached 314,180 at the end of 2013, with 4.2% of these patients having been on dialysis for over 25 years1. Many studies have shown that patients on HD in Japan have lower rates of hospitalization and mortality than their counterparts in other countries2–4. However, there is limited information on hospitalization and mortality rates in patients on PD. Furthermore, the number of patients with diabetes-related ESKD continues to increase5,6, which means that the number of patients with diabetes on PD is also increasing7.
In view of the lack of data on outcomes in patients on PD in Japan, we undertook a cohort study using a nationwide registry of such patients to clarify the outcomes, including mortality and cardiovascular events, in patients with and without diabetes on PD.
Methods
Database creation
Data were obtained from annual nationwide surveys of patients on dialysis conducted by the Japanese Society for Dialysis Therapy (JSDT) and stored in the JSDT Renal Data Registry (JRDR). The surveys were conducted by volunteers, as described previously7–10. The study protocol was approved by the Medicine Ethics Committee of the Japanese Society for Dialysis Therapy and by our university. All procedures fully adhered to the Declaration of Helsinki. The study was registered with the University Hospital Medical Information Network (UMIN000018641). The need for informed consent was waived in view of the anonymity of the study data.
The study population comprised patients who were receiving PD on 31 December 2013 and were followed for 2 years. The inclusion criterion was PD for at least 90 days at the time of enrollment in the cohort. We excluded patients who had received only HD, those aged <20 years, those whose records for date of birth, initiation of dialysis, or outcome were incomplete, and those who were considered outliers. Patients were considered outliers if their anthropometric and laboratory measurements were outside the following ranges: height 120–200 cm, body weight 20–150 kg, serum albumin 1.0–5.0 g/dL, C-reactive protein (CRP) <30 mg/dL, and hemoglobin 5.0–20.0 g/dL. Demographic and clinical data at enrollment were collected, including age, sex, duration of dialysis, primary cause of ESKD, body mass index (BMI), smoking status, use of antihypertensive agents, and history of complications of cardiovascular disease (CVD), including cerebral infarction, cerebral hemorrhage, myocardial infarction, and limb amputation. The patients with diabetes in the study were those with a diagnosis of diabetic nephropathy as the primary cause of ESKD and those on antidiabetic medications. Blood samples were drawn and assayed at each dialysis center or hospital, and the most recent values, including serum albumin, hemoglobin, calcium, phosphate, intact parathyroid hormone (i-PTH), high-density lipoprotein (HDL) cholesterol, non-HDL cholesterol, and CRP at the time of survey were collected. Weekly Kt/V urea from residual renal function (renal Kt/V) and weekly Kt/V urea from PD (PD Kt/V) values were recorded. Total Kt/V was calculated as the sum of renal Kt/V and PD Kt/V. The ratio of creatinine in the dialysate to that in plasma (D/P Cr) was obtained using the peritoneal equilibration test. The type of PD fluid used, namely, 2.5% glucose or icodextrin dialysate, was recorded, since 1.5% glucose dialysate is used frequently in Japan. The use of an automated PD or connecting system was also recorded. The type of treatment was also recorded because combination PD + HD therapy, that is, HD on 1 day per week (performed in the same facility as PD) and PD on the other days, has been approved in Japan since 2010.
Cause of death was classified as CVD, infection, malignancy, gastrointestinal disease, encapsulating peritoneal sclerosis (EPS), hepatic disease, other, or unknown. We defined cardiovascular death as that due to chronic heart failure, pulmonary edema, ischemic heart disease (including acute myocardial infarction, i.e., death within 30 days of onset), arrhythmia, valvular heart disease, endocarditis, other cardiac disease, subarachnoid hemorrhage, intracerebral hemorrhage, cerebral infarction, or other brain disease. New-onset cardiovascular events, including cerebral infarction, cerebral hemorrhage, myocardial infarction, or limb amputation, and episodes of peritonitis were also recorded. The definition of peritonitis used was that in the International Society for Peritoneal Dialysis guidelines11. The dates and causes of death were obtained from the JRDR at the end of 2014 and 2015.
Outcome analysis by demographic factors, dialysis dose, and nutritional factors
We divided the patients into six a priori categories based on duration of PD (<2, 2 to <4, 4 to <6, 6 to <8, 8 to <10, and ≥10 years) to examine the relationship between category of dialysis duration and risk of death. The patients were also categorized by dialysis-related factors, including residual renal function, i.e., anuric or non-anuric state, use of 2.5% glucose or icodextrin dialysate, and use of an automated PD or connecting system. We also divided the patients into five a priori categories based on total Kt/V, i.e., <1.1 and ≥2.0, with intervening increments of 0.3, to examine the dose-response association between the categories of Kt/V and risk of death. BMI, hemoglobin, albumin, non-HDL cholesterol, HDL cholesterol, calcium, phosphate, and CRP levels were included as nutrition-related and inflammation-related factors. Age, hemoglobin, CRP, BMI, and serum albumin, non-HDL cholesterol, HDL cholesterol, calcium, and phosphate levels were analyzed as continuous variables. We estimated the association between demographic factors at enrollment and survival using multivariate Cox proportional hazards regression and analyzed models that were both unadjusted and adjusted for relevant covariates measured at study enrollment, including age, sex, duration of PD, presence or absence of diabetes, comorbid CVD, use of antihypertensive agents, residual renal function, total Kt/V, type of dialysate, hemoglobin, CRP, BMI, albumin, non-HDL cholesterol, HDL cholesterol, calcium, and phosphate. Patients who were switched to HD or underwent kidney transplantation during follow-up were censored from the analysis of outcomes.
Outcome analysis by diabetes and non-diabetes groups
We divided the patients into two groups based on the presence or absence of diabetes. Survival analyses with Cox proportional hazards regression were used to examine whether the presence or absence of diabetes predicted survival for up to 2 years of follow-up. Kaplan-Meier survival analysis was used to compare all-cause, cardiovascular, and infection-related mortality rates. The log-rank statistic was used to test for differences between the two groups. New-onset cardiovascular events were also analyzed. Finally, we examined associations between the two groups and all-cause mortality. Models were analyzed when adjusted for the above-mentioned demographic, dialysis-related, nutrition-related, and inflammation-related factors measured at enrollment.
Statistical analysis
Data were summarized using proportions, with mean ± standard deviation or median (interquartile range) as appropriate. Categorical variables were analyzed using the chi-square test and continuous variables using the Student’s t-test. Categorical data were compared between groups using repeated-measures analysis of variance and Tukey’s honestly significant difference test or Kruskal-Wallis test as appropriate. Missing covariate data were imputed by the conventional method for multivariate regression as appropriate. Proportional hazards assumption was tested by graphical and formal testing. All analyses were performed using JMP® version 13.0 (SAS Institute, Cary, NC). A P-value < 0.05 was considered statistically significant.
Results
Patient demographics and clinical characteristics
In total, 315,631 patients on maintenance dialysis were registered at the end of 2013. After exclusions, 8,954 patients on PD were enrolled in the study (Fig. 1). Demographic and clinical characteristics of the 8,954 patients at enrollment are shown in Table 1. Mean patient age was 62.7 ± 13.1 years, 63.8% of the patients were male, and 15.1% had comorbid CVD, comprising coronary artery disease, stroke, or limb amputation. Mean duration of PD was 33 (15–60) months, mean BMI was 23.4 ± 3.8 kg/m2, and mean albumin and hemoglobin levels were 3.3 ± 0.5 g/dL and 10.8 ± 1.4 g/dL, respectively. The most common cause of ESKD was glomerulonephritis (39.2%), followed by diabetic nephropathy (30.3%) and hypertension (12.9%). Renal Kt/V and PD Kt/V values at enrollment were 0.3 (0–0.8) and 1.4 ± 0.6, respectively. An automated PD system was used in 43.2% of patients and a connecting device in 68.2%. During the 2-year study period, 893 patients (10.0%) died, 148 (1.6%) underwent kidney transplantation, 2,637 (29.4%) were switched to HD; 5,276 (58.9%) patients were still alive at the end of the study period.
Figure 1.
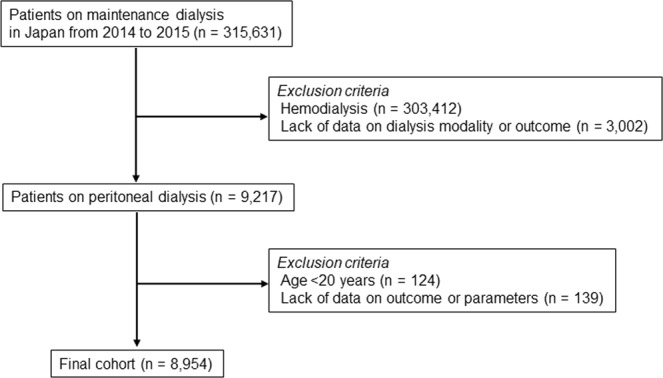
Flowchart showing enrolment of participants.
Table 1.
Demographic, clinical, and laboratory data at baseline in 8,954 patients on peritoneal dialysis.
| Variable | |
|---|---|
| Patients (n) | 8,954 |
| Age (years) | 62.7 ± 13.1 |
| Male sex (%) | 63.8 |
| Dialysis duration (months) | 33 [15–60] |
| Primary cause of ESKD, n (%) | |
| Chronic glomerulonephritis | 3,513 (39.2) |
| Diabetic nephropathy | 2,710 (30.3) |
| Hypertension | 1,154 (12.9) |
| Polycystic kidney disease | 223 (2.5) |
| Other | 476 (5.3) |
| Unknown | 878 (9.8) |
| Body mass index (kg/m2) | 23.4 ± 3.8 |
| Smoking (%) | 8.1 |
| Antihypertensive agents (%) | 79.2 |
| Comorbid CVD (%) | 15.1 |
| Hemoglobin (g/dL) | 10.8 ± 1.4 |
| Albumin (g/dL) | 3.3 ± 0.5 |
| Non-HDL cholesterol (mg/dL) | 128 ± 37 |
| HDL cholesterol (mg/dL) | 50 ± 18 |
| C-reactive protein (mg/dL) | 0.13 [0.07–0.40] |
| Calcium (mg/dL) | 8.7 ± 0.8 |
| Phosphate (mg/dL) | 5.2 ± 1.3 |
| Intact PTH (pg/mL) | 155 [81–262] |
| Renal Kt/V | 0.3 [0–0.8] |
| PD Kt/V | 1.4 ± 0.6 |
| Total Kt/V | 1.8 ± 0.8 |
| D/P Cr | 0.65 ± 0.13 |
| Use of 2.5% glucose dialysate (%) | 37.5 |
| Use of icodextrin (%) | 40.8 |
| Use of automated PD (%) | 43.2 |
| Connecting system (%) | |
| Device (+) | 68.2 |
| PD + HD combination (%) | 18.8 |
Data are presented as the number, percentage, mean ± standard deviation, or median [interquartile range]. Cr, creatinine; CVD, cardiovascular disease; D/P, dialysate/plasma; ESKD, end-stage kidney disease; HD, hemodialysis; HDL, high-density lipoprotein; PD, peritoneal dialysis; PTH, parathyroid hormone.
All-cause mortality according to demographic, dialysis-related, nutrition-related, and inflammation-related factors at enrollment
Hazard ratios (HRs) in the unadjusted analysis of variables evaluated as potential predictors of mortality in all patients are presented in Table 2. There was no significant effect of sex on patient survival (P = 0.114). Older age, longer duration of PD, presence of diabetes, and comorbid CVD were significant predictors of mortality. Lower dialysis dose, assessed by total Kt/V, was associated with higher risk of mortality. Use of 2.5% glucose or icodextrin dialysate and an anuric state at enrollment were also significant predictors of mortality. Poor nutritional status and more severe inflammatory status, indicated by lower hemoglobin, higher CRP, lower BMI, lower serum albumin, lower calcium, and lower phosphate, were also associated with higher mortality in patients on PD. PD + HD combination therapy was associated with a lower mortality risk. After multivariate adjustment, older age, longer duration of PD, presence of diabetes, comorbid CVD, use of 2.5% glucose dialysate, higher CRP, lower BMI, and lower albumin and phosphate levels continued to be independently associated with increased HR for all-cause mortality (Table 2). Use of antihypertensive agents, higher HDL levels, and PD + HD combination therapy continued to be associated with lower mortality risk.
Table 2.
Hazard ratios (with 95% CIs) for variables evaluated as potential predictors of all-cause mortality in the study population.
| Factors | n (%) | Unadjusted | Adjusted | ||||
|---|---|---|---|---|---|---|---|
| HR | 95% CI | P-value | HR | 95% CI | P-value | ||
| Sex | |||||||
| Male | 5,714 (63.8) | 1.000 | Reference | Reference | 1.000 | Reference | Reference |
| Female | 3,240 (36.2) | 0.898 | 0.787–1.025 | 0.114 | 0.772 | 0.438–1.378 | 0.376 |
| Age | |||||||
| 1-year increase | 8,954 (100) | 1.081 | 1.073–1.087 | <0.0001 | 1.113 | 1.078–1.149 | <0.0001 |
| PD duration (years) | |||||||
| <2 | 3,507 (39.2) | 1.000 | Reference | Reference | 1.000 | Reference | Reference |
| ≥2 <4 | 2,344 (26.2) | 1.170 | 0.978–1.399 | 0.086 | 1.374 | 0.966–1.953 | 0.076 |
| ≥4 <6 | 1,422 (15.9) | 1.432 | 1.165–1.762 | 0.001 | 1.627 | 1.173–2.256 | 0.004 |
| ≥6 <8 | 761 (8.5) | 1.676 | 1.233–2.278 | 0.001 | 1.720 | 1.041–2.841 | 0.045 |
| ≥8 <10 | 414 (4.6) | 1.545 | 1.093–2.183 | 0.013 | 2.211 | 1.377–3.549 | 0.002 |
| ≥10 | 506 (5.6) | 1.506 | 1.113–2.036 | 0.007 | 2.979 | 1.687–5.259 | 0.002 |
| Diabetes | |||||||
| No | 5,635 (62.9) | 1.000 | Reference | Reference | 1.000 | Reference | Reference |
| Yes | 3,319 (37.1) | 1.500 | 1.318–1.708 | <0.0001 | 1.648 | 1.097–2.474 | 0.016 |
| Comorbid CVD | |||||||
| No | 7,399 (84.9) | 1.000 | Reference | Reference | 1.000 | Reference | Reference |
| Yes | 1,313 (15.1) | 2.861 | 2.464–3.322 | <0.0001 | 1.367 | 1.055–1.771 | 0.017 |
| Use of antihypertensive medication | |||||||
| No | 1,264 (20.8) | 1.000 | Reference | Reference | 1.000 | Reference | Reference |
| Yes | 4,802 (79.2) | 0.509 | 0.422–0.615 | <0.0001 | 0.667 | 0.501–0.889 | 0.005 |
| Residual renal function | |||||||
| Non-anuric | 3,479 (77.2) | 1.000 | Reference | Reference | 1.000 | Reference | Reference |
| Anuric | 1,026 (22.8) | 1.677 | 1.330–2.114 | <0.0001 | 1.257 | 0.785–1.984 | 0.335 |
| Total Kt/V | |||||||
| <1.1 | 286 (8.7) | 2.313 | 1.481–3.614 | 0.001 | 2.431 | 0.925–6.386 | 0.071 |
| ≥1.1 <1.4 | 326 (9.9) | 1.392 | 0.806–2.405 | 0.235 | 1.162 | 0.419–3.222 | 0.772 |
| ≥1.4 <1.7 | 682 (20.7) | 1.000 | Reference | Reference | 1.000 | Reference | Reference |
| ≥1.7 <2.0 | 853 (25.9) | 1.159 | 0.784–1.713 | 0.457 | 0.792 | 0.350–1.791 | 0.576 |
| ≥2.0 | 1,144 (34.8) | 0.832 | 0.563–1.230 | 0.358 | 0.614 | 0.262–1.437 | 0.262 |
| Use of 2.5% dialysate | |||||||
| No | 3,422 (62.5) | 1.000 | Reference | Reference | 1.000 | Reference | Reference |
| Yes | 2,056 (37.5) | 1.502 | 1.267–1.779 | <0.0001 | 2.551 | 1.433–4.539 | 0.001 |
| Use of icodextrin dialysate | |||||||
| No | 3,241 (59.2) | 1.000 | Reference | Reference | 1.000 | Reference | Reference |
| Yes | 2,237 (40.8) | 1.307 | 1.102–1.548 | 0.002 | 0.968 | 0.641–1.445 | 0.876 |
| PD + HD combination therapy | |||||||
| No | 7,275 (81.2) | 1.000 | Reference | Reference | 1.000 | Reference | Reference |
| Yes | 1,679 (18.8) | 0.556 | 0.438–0.703 | <0.0001 | 0.595 | 0.413–0.858 | 0.005 |
| Hemoglobin | |||||||
| 1-g/dL increase | 6,130 (68.5) | 0.773 | 0.726–0.823 | <0.0001 | 0.925 | 0.844–1.014 | 0.098 |
| C-reactive protein | |||||||
| 1-mg/dL increase | 5,344 (59.7) | 1.571 | 1.467–1.682 | <0.0001 | 1.217 | 1.073–1.381 | 0.002 |
| Body mass index | |||||||
| 1-kg/m2 increase | 5,330 (59.5) | 0.910 | 0.885–0.935 | <0.0001 | 0.906 | 0.847–0.969 | 0.004 |
| Serum albumin | |||||||
| 1-g/dL increase | 6,092 (68.0) | 0.217 | 0.190–0.247 | <0.0001 | 0.363 | 0.234–0.562 | <0.0001 |
| Non-HDL cholesterol | |||||||
| 1-mg/dL increase | 3,658 (40.9) | 0.996 | 0.993–0.999 | 0.042 | 0.995 | 0.989–1.001 | 0.097 |
| HDL cholesterol | |||||||
| 1-mg/dL increase | 4,264 (47.6) | 0.982 | 0.976–0.989 | <0.0001 | 0.981 | 0.968–0.995 | 0.008 |
| Calcium (mg/dL) | |||||||
| 1-mg/dL increase | 6,175 (69.0) | 0.748 | 0.685–0.819 | <0.0001 | 1.145 | 0.877–1.493 | 0.317 |
| Phosphate (mg/dL) | |||||||
| 1-mg/dL increase | 7,340 (82.0) | 0.871 | 0.818–0.925 | <0.0001 | 1.231 | 1.046–1.450 | 0.012 |
CI, confidence interval; CVD, cardiovascular disease; HD, hemodialysis; HDL, high-density lipoprotein; HR, hazard ratio; PD, peritoneal dialysis.
In total, 1,240 episodes of PD-related peritonitis occurred in 859 patients during the study period. The mean peritonitis rate was 0.18 per patient-year. Multivariate analysis was performed to identify predictors of peritonitis (Supplementary Table 1). A previous history of peritonitis, use of a connecting device, comorbid CVD, and higher BMI were independent predictors of peritonitis.
Demographics and clinical characteristics in the diabetes and non-diabetes groups
Patients were divided into two groups based on the presence or absence of diabetes. Table 3 shows the demographics and clinical characteristics of each group. Patients with diabetes were older and more likely to be male, with shorter duration of PD, higher BMI, higher rate of CVD comorbidity, lower HDL cholesterol, and higher CRP levels. There was no significant difference in hemoglobin level, total Kt/V value, or frequency of use of an automated PD system. More patients in the diabetes group used 2.5% glucose and/or icodextrin dialysate than those in the non-diabetes group.
Table 3.
Demographic, clinical, and laboratory data in diabetes and non-diabetes patients on peritoneal dialysis.
| Diabetes group | Non-diabetes group | P-value | |
|---|---|---|---|
| Patients (n) | 3,319 | 5,635 | — |
| Age (years) | 63.9 ± 11.8 | 61.9 ± 13.6 | <0.0001 |
| Male sex (%) | 70.4 | 59.9 | <0.0001 |
| PD duration (m) | 26 [13–48] | 37 [17–69] | <0.0001 |
| Type 1 diabetes (%) | 8.3 | — | — |
| Body mass index (kg/m2) | 24.3 ± 3.8 | 22.7 ± 3.6 | <0.0001 |
| Smoking (%) | 9.9 | 6.9 | <0.0001 |
| Antihypertensive agents (%) | 81.7 | 77.5 | <0.0001 |
| Comorbid CVD (%) | 21.4 | 11.3 | <0.0001 |
| Albumin (g/dL) | 3.2 ± 0.5 | 3.3 ± 0.5 | <0.0001 |
| Non-HDL cholesterol (mg/dL) | 126 ± 38 | 130 ± 36 | 0.004 |
| HDL cholesterol (mg/dL) | 46 ± 16 | 52 ± 17 | <0.0001 |
| HbA1c (%) | 6.2 ± 1.1 | — | — |
| C-reactive protein (mg/dL) | 0.17 [0.09–0.49] | 0.11 [0.06–0.37] | 0.001 |
| Calcium (mg/dL) | 8.6 ± 0.8 | 8.8 ± 0.8 | <0.0001 |
| Phosphate (mg/dL) | 5.2 ± 1.3 | 5.3 ± 1.3 | 0.028 |
| Intact PTH (pg/mL) | 150 [77–249] | 158 [82–267] | 0.002 |
| Hemoglobin (g/dL) | 10.8 ± 1.4 | 10.8 ± 1.4 | 0.313 |
| Renal Kt/V | 0.4 [0–0.9] | 0.2 [0–0.7] | <0.0001 |
| PD Kt/V | 1.3 ± 0.6 | 1.4 ± 0.6 | <0.0001 |
| Total Kt/V | 1.8 ± 0.8 | 1.8 ± 0.8 | 0.848 |
| D/P Cr | 0.67 ± 0.13 | 0.64 ± 0.13 | <0.0001 |
| PET category (%) | <0.0001 | ||
| Low | 8.1 | 11.6 | |
| Low average | 36.7 | 39.1 | |
| High average | 42.1 | 40.5 | |
| High | 13.1 | 8.8 | |
| Use of 2.5% glucose dialysate (%) | 40.1 | 36.0 | 0.002 |
| Use of icodextrin (%) | 45.0 | 38.3 | <0.0001 |
| Use of APD system (%) | 43.3 | 43.1 | 0.924 |
| Connecting system (%) | |||
| Device (+) | 72.5 | 65.5 | <0.001 |
| PD + HD combination (%) | 18.5 | 19.0 | 0.537 |
Data are shown as the number, percentage, mean ± standard deviation, or median [interquartile range]. APD, automated peritoneal dialysis; Cr, creatinine; CVD, cardiovascular disease; D/P, dialysate/plasma; HD, hemodialysis; HDL, high-density lipoprotein; PD, peritoneal dialysis; PET, peritoneal equilibration test; PTH, parathyroid hormone.
Comparison of outcomes between the diabetes and non-diabetes groups
Outcome variables are shown in Table 4. In total, 1,020 cardiovascular events (401 fatal, 619 non-fatal) were recorded during the study period. The cardiovascular event rate was significantly higher in the diabetes group than in the non-diabetes group (15.9% vs. 8.7%; P < 0.0001). The leading cause of death in the diabetes group was CVD (42.1%), followed by infection (27.2%). The leading cause of death in the non-diabetes group was also CVD (35.7%), followed by infection (21.7%). EPS was the cause of death in 1 patient in the diabetes group and in 5 patients in the non-diabetes group; the difference was statistically significant (P = 0.0013). Kaplan-Meier survival analysis (Fig. 2) showed a 2-year survival rate of 82.2% in the diabetes group and 87.4% in the non-diabetes group (P < 0.0001). Kaplan-Meier survival analyses for cardiovascular and infection-related deaths revealed significant differences in the patient survival rates between the diabetes and non-diabetes groups (both P < 0.0001; Supplementary Fig. 2a,b).
Table 4.
New-onset cardiovascular event rates and causes of death in diabetes and non-diabetes patients.
| All | Diabetes group | Non-diabetes group | P-valuea | |
|---|---|---|---|---|
| New-onset CV events, n (%) | 1,020 (11.4) | 527 (15.9) | 493 (8.7) | <0.0001 |
| Death, n (%) | 893 (14.5) | 397 (17.8) | 496 (12.7) | <0.0001 |
| Cause of death, n (%) | 0.001 | |||
| Cardiovascular disease | 344 (38.5) | 167 (42.0) | 177 (35.7) | |
| Infection | 215 (24.1) | 108 (27.2) | 107 (21.6) | |
| Malignancy | 50 (5.6) | 13 (3.3) | 37 (7.5) | |
| Gastrointestinal disease | 29 (3.2) | 7 (1.8) | 22 (4.4) | |
| EPS | 6 (0.7) | 1 (0.3) | 5 (1.0) | |
| Hepatic disease | 12 (1.3) | 6 (1.5) | 6 (1.2) | |
| Other | 130 (14.6) | 48 (12.1) | 82 (16.5) | |
| Unknown | 107 (12.0) | 47 (11.8) | 60 (12.1) |
aDiabetes vs. non-diabetes. CV, cardiovascular; EPS, encapsulating peritoneal sclerosis.
Figure 2.
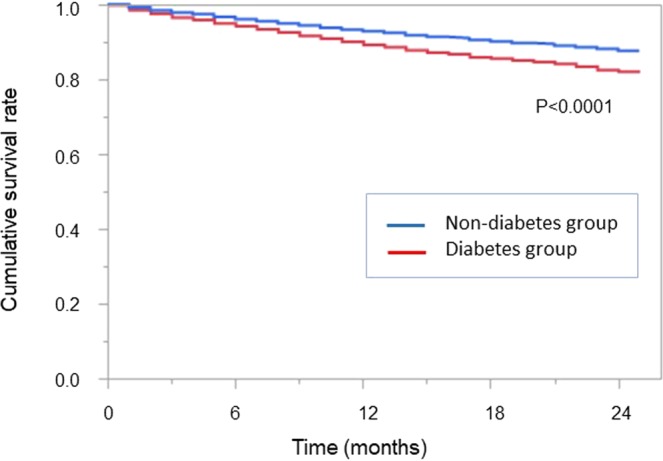
Comparison of cumulative survival rates between diabetes and non-diabetes patients on peritoneal dialysis.
HRs for all-cause mortality between the diabetes and non-diabetes groups
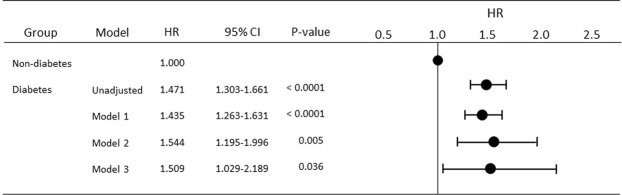
As shown in Fig. 3, unadjusted HR for all-cause mortality in the diabetes group when compared with that in the non-diabetes (reference) group was 1.471 (95% CI 1.303–1.661; P < 0.0001). After adjusting for sex, age, duration of PD, smoking status, and comorbid CVD, HR for all-cause mortality in the diabetes group when compared with the non-diabetes (reference) group was 1.435 (95% CI 1.263–1.631; P < 0.0001). After adjusting for modification of dialysis, including total Kt/V value, anuric state, use of 2.5% glucose dialysate or icodextrin, and PD + HD combination therapy, as well as for baseline factors, HR for the diabetes group compared with the non-diabetes (reference) group was 1.544 (95% CI 1.195–1.996; P = 0.005). Finally, after adjusting for nutrition-related and inflammation-related factors in addition to the demographic factors and dialysis-related factors, the HR for the diabetes group when compared with the non-diabetes (reference) group was 1.509 (95% CI 1.029–2.189; P = 0.036).
Figure 3.
Comparison of hazard ratios for all-cause mortality between diabetes and non-diabetes patients on peritoneal dialysis using standard Cox proportional hazards regression. Model 1 is adjusted for age, sex, duration of PD, and presence or absence of comorbid cardiovascular disease. Model 2 is adjusted for basic demographic and dialysis-related factors, including total Kt/V, anuric or non-anuric state, type of dialysate, and PD + HD combination therapy. Model 3 is adjusted for demographic, dialysis-related, nutrition-related, and inflammation-related factors, including body mass index and C-reactive protein, hemoglobin, serum albumin, non-HDL cholesterol, HDL cholesterol, calcium, and phosphate levels. CI, confidence interval; HD, hemodialysis; HDL, high-density lipoprotein; HR, hazard ratio; PD, peritoneal dialysis.
Discussion
This study is the first to identify predictors of 2-year mortality in patients on PD in Japan. Duration of PD and the serum albumin level were the most common factors associated with patient survival. Interestingly, PD + HD combination therapy was associated with lower mortality risk. The patients with diabetes who were receiving PD had lower survival rates than those without diabetes. The main strength of this study is that it is a nationwide cohort analysis of outcomes in patients on PD in Japan. Moreover, this is the first Japanese study to address prognostic risk factors in these patients.
Data from the US Renal Data System for 2010 revealed that the overall 1-year, 2-year, and 3-year survival rates of patients on PD were 83%, 67.2%, and 53.8%, respectively, whereas those in a diabetes subgroup were 80.3%, 61.7%, and 46.6%12. A further study from Canada reported 1-year, 2-year, and 3-year survival rates of 91%, 76%, and 66%, respectively, for patients with diabetes and respective rates of 94%, 89%, and 84% for patients without diabetes13. Data from single centers in Asia and Europe also showed that survival rates were significantly lower in patients with diabetes on PD than in their counterparts without diabetes14. In the present study, the survival rate was significantly lower in the diabetes group than in the non-diabetes group. However, the 1-year and 2-year survival rates in all patients in our study were 91.4% and 85.5%, respectively, and higher than in previous reports; furthermore, the respective 1-year and 2-year survival rates were 91.3% and 82.2% in the diabetes group and 93.8% and 87.4% in the non-diabetes group. These findings suggest that patients with diabetes on PD have lower survival rates than their counterparts without diabetes. BMI and renal Kt/V values at enrollment were significantly higher in the diabetes group than in the non-diabetes group; however, the older age, higher rate of CVD comorbidity, higher CRP level, and lower HDL cholesterol level at enrollment might be attributed to lower survival rate in the diabetes group.
The diabetes group in our cohort consisted of patients with ESKD caused by diabetic nephropathy and those with diabetes as a comorbid condition. Previous studies of patients on HD have compared those with and without diabetic nephropathy as the primary kidney disease and showed poorer survival in those with diabetic nephropathy15,16. Furthermore, The Netherlands Cooperative Study on the Adequacy of Dialysis (NECOSAD) reported that mortality in patients with diabetes as the primary kidney disease was similar to that in patients with diabetes as a comorbid condition17. Therefore, diabetes has a strong impact on survival even if it is not the primary cause of ESKD; in our cohort, cardiovascular mortality and new-onset CVD rates were higher in the diabetes group. Patients on PD are exposed to a large amount of glucose absorbed from the dialysate; therefore, continuous exposure to dialysate might worsen glycemic control and cause peritoneal damage in patients with diabetes18. Peritoneal absorption of glucose degradation products might enhance formation of advanced glycosylation end products (AGEs), a nonenzymatic reaction between reducing sugars and proteins19,20. In the PD population, increased peritoneal membrane transport and protein permeability is more common in patients with diabetes than in those without diabetes, and these characteristics has been shown to be associated with a number of risk factors for mortality and technique failure, including age, racial origin, BMI, burden of comorbid illness, hypoalbuminemia, and elevated inflammatory markers21–23. Patients with diabetes on PD may have higher levels of peritoneal AGEs associated with endothelial dysfunction and atherosclerotic CVD24,25. However, it was reported that overall survival of patients on PD was significantly higher than that of those on HD with diabetes on incident dialysis, and that the better patient survival on PD was influenced by the degree of glycemic control, that is, an HbA1c level below 8.0%26. The present study revealed that 1.5% glucose dialysate and icodextrin dialysate are frequency used in Japan. However, given that the PD dialysate prescription patterns vary from country to country, further studies are needed to clarify whether use of dialysate with a low glucose concentration might affect patient survival.
PD + HD combination therapy has been approved in Japan since 2010 and was used by 18.8% of the patients in our study cohort. Combined therapy is indicated for patients in whom adequate solute clearance (total Kt/V urea >1.7) cannot be maintained with a standard PD prescription and for patients with symptoms of uremia despite a total Kt/V urea >1.7 or a state of persistent fluid overload. It was reported that 50% of patients on combined therapy had a PD duration of more than 8 years27. In the present cohort, the duration of PD was significantly longer in the patients receiving combination therapy than in those receiving PD alone (72 months vs. 38 months, P < 0.0001). On the day of HD and on the following day, patients on combination therapy have a PD holiday, that is, they are released from bag exchange for PD28,29, which might lead to a decrease in the D/P Cr ratio obtained from the peritoneal equilibration test, suggesting improved functioning of the peritoneal membrane. PD + HD combination therapy is widely used in Japanese patients on PD in view of its known effectiveness for solute clearance and fluid management30,31. In one report, there was a decrease in body weight, blood pressure, and serum creatinine, as well as an increase in hemoglobin level, after switching from PD alone to combined therapy, suggesting significant improvement in dialysis and decreased fluid overload32. In the present study, all-cause mortality was significantly lower in patients who were receiving combination therapy than in those on PD alone. However, patients selected for PD + HD combination therapy might be different from those in the general PD population, which would influence all-cause mortality and other clinical outcomes. Furthermore, we could not compare the characteristics, including duration of PD and HD, of patients on PD + HD combination therapy in this study. Therefore, further prospective cohort studies are needed to determine whether this combination therapy has a survival benefit, since the patients receiving PD + HD combination therapy and those receiving only PD therapy may constitute different cohorts.
This study has several limitations. First, as in any annual survey or observational cohort study, facility bias could not be excluded. Therefore, the mortality rate and incidence of peritonitis may have varied between centers because of differences in practice and in patient populations. Second, the follow-up duration of 2 years was relatively short, and prevalent patients rather than incident patients were included. Therefore, there are several factors that could vary according to duration of PD, including residual renal function, total Kt/V, and type of dialysate used. Furthermore, early technique failure (within the first 12 months of commencement of PD) is common, and peritonitis is one of the causes of early technique failure. Therefore, we could not detect patients with early technique failure or the exact rate of development of peritonitis. It is not known whether the rates of peritonitis were affected by differences in patient education on how to prevent these complications. Similarly, the effects of physical activity levels, employment status, and socioeconomic factors are unknown. Therefore, prospective studies on whether the outcomes for patients on PD are affected by differences in these factors are required in the future. Finally, glycemic control in patients with diabetes was not assessed because of the small number of patients with data on glycemic indices. Further studies would be needed to assess the important issue of whether the outcomes in diabetes patients on PD would be affected by glycemic control.
In conclusion, older age, longer duration of PD, use of 2.5% glucose dialysate, lower serum albumin level, more severe inflammatory status, comorbid CVD, and higher phosphate level were significant predictors of mortality in Japanese patients on PD. Higher BMI and HDL cholesterol levels were associated with lower mortality risk in these patients. This study identified that all-cause mortality rate is higher in diabetes patients on PD than in their counterparts without diabetes. Further studies are needed to clarify whether PD + HD combination therapy has a survival benefit. The findings of this study underscore the need for further research on the factors that influence patient outcomes to identify alternative interventions that would improve the outlook for patients receiving PD.
Supplementary information
Acknowledgements
We thank the members of the JSDT Renal Data Registry committee for all their efforts as well as the staff members of the dialysis facilities who participated in the survey. No financial support was received for this study or publication of this report.
Author Contributions
Research idea and study design: M.A.; data acquisition: M.A., J.H., A.W. and S.N.; data analysis/interpretation: M.A.; statistical analysis, M.A.; supervision or mentorship: T.H., I.M., N.H., K.N. and H.N. All authors contributed important intellectual content during drafting or revision of the manuscript and accept accountability for the overall work by ensuring that questions pertaining to the accuracy or integrity of any portion of the work are appropriately investigated and resolved. M.A. takes responsibility that this study has been reported honestly, accurately, and transparently; that no important aspects of the study have been omitted; and that any discrepancies from the study as planned and registered have been explained.
Competing Interests
The authors declare no competing interests.
Footnotes
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary information accompanies this paper at 10.1038/s41598-019-40692-6.
References
- 1.Masakane I, et al. An Overview of Regular Dialysis Treatment in Japan (As of 31 December 2013) Ther Apher Dial. 2015;19:540–574. doi: 10.1111/1744-9987.12378. [DOI] [PubMed] [Google Scholar]
- 2.Goodkin DA, et al. Association of comorbid conditions and mortality in hemodialysis patients in Europe, Japan, and the United States: the Dialysis Outcomes and Practice Patterns Study (DOPPS) J Am Soc Nephrol. 2003;14:3270–3277. doi: 10.1097/01.ASN.0000100127.54107.57. [DOI] [PubMed] [Google Scholar]
- 3.Held PJ, et al. Five-year survival for end-stage renal disease patients in the United States, Europe, and Japan, 1982–1987. Am J Kidney Dis. 1990;15:451–457. doi: 10.1016/S0272-6386(12)70363-3. [DOI] [PubMed] [Google Scholar]
- 4.Valderrabano F. Renal replacement therapy. What are the differences between Japan and Europe? Nephrol Dial Transplant. 1996;11:2151–2153. doi: 10.1093/oxfordjournals.ndt.a027129. [DOI] [PubMed] [Google Scholar]
- 5.Abe M, Kalantar-Zadeh K. Haemodialysis-induced hypoglycaemia and glycaemic disarrays. Nat Rev Nephrol. 2015;11:302–313. doi: 10.1038/nrneph.2015.38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Abe M, et al. Is there a “burnt-out diabetes” phenomenon in patients on hemodialysis? Diabetes Res Clin Pract. 2017;130:211–220. doi: 10.1016/j.diabres.2017.06.012. [DOI] [PubMed] [Google Scholar]
- 7.Masakane I, et al. Peritoneal Dialysis Registry With 2013 Survey Report. Ther Apher Dial. 2016;20:557–568. doi: 10.1111/1744-9987.12520. [DOI] [PubMed] [Google Scholar]
- 8.Hoshino J, et al. Committee of Renal Data Registry of the Japanese Society for Dialysis Therapy: Glycated albumin versus hemoglobin A1c and mortality in diabetic hemodialysis patients: a cohort study. Nephrol Dial Transplant. 2018;33:1150–1158. doi: 10.1093/ndt/gfy014. [DOI] [PubMed] [Google Scholar]
- 9.Abe M, Hamano T, Wada A, Nakai S, Masakane I. High-Performance Membrane Dialyzers and Mortality in Hemodialysis Patients: A 2-Year Cohort Study from the Annual Survey of the Japanese Renal Data Registry. Am J Nephrol. 2017;46:82–92. doi: 10.1159/000478032. [DOI] [PubMed] [Google Scholar]
- 10.Masakane I. An overview of regular dialysis treatment in Japan (As of 31 December 2014) [in Japanese]. http://docs.jsdt.or.jp/overview/index.html, (13 July 2018, date last accessed). [DOI] [PubMed]
- 11.Li PK, et al. International Society for Peritoneal Dialysis: International Society for Peritoneal Dialysis: Peritoneal dialysis–related infections recommendations: 2010 update. Perit Dial Int. 2010;30:393–423. doi: 10.3747/pdi.2010.00049. [DOI] [PubMed] [Google Scholar]
- 12.United states Renal Data System. 2010 USRDS Annual Data Report: Atlas of Chronic Kidney Disease and End-stage Renal Disease in the United States. Bethesda, MD: National Institute of Diabetes and Digestive and Kidney Diseases (2010).
- 13.Fang W, et al. Patient and technique survival of diabetics on peritoneal dialysis: one-center’s experience and review of the literature. Clin Nephrol. 2008;69:193–200. doi: 10.5414/CNP69193. [DOI] [PubMed] [Google Scholar]
- 14.Chung SH, Heimbürger O, Lindholm B, Lee HB. Peritoneal dialysis patient survival: a comparison between a Swedish and a Korean centre. Nephrol Dial Transplant. 2005;20:1207–1213. doi: 10.1093/ndt/gfh772. [DOI] [PubMed] [Google Scholar]
- 15.Vonesh EF, Snyder JJ, Foley RN, Collins AJ. The differential impact of risk factors on mortality in hemodialysis and peritoneal dialysis. Kidney Int. 2004;66:2389–2401. doi: 10.1111/j.1523-1755.2004.66028.x. [DOI] [PubMed] [Google Scholar]
- 16.Liem YS, Wong JB, Hunink MG, de Charro FT, Winkelmayer WC. Comparison of hemodialysis and peritoneal dialysis survival in The Netherlands. Kidney Int. 2007;71:153–158. doi: 10.1038/sj.ki.5002014. [DOI] [PubMed] [Google Scholar]
- 17.Schroijen MA, et al. NECOSAD Study Group: Survival in dialysis patients is not different between patients with diabetes as primary renal disease and patients with diabetes as a co-morbid condition. BMC Nephrol. 2011;12:69. doi: 10.1186/1471-2369-12-69. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Mehrotra R, et al. Chronic peritoneal dialysis in the United States: declining utilization despite improving outcomes. J Am Soc Nephrol. 2007;18:2781–2788. doi: 10.1681/ASN.2006101130. [DOI] [PubMed] [Google Scholar]
- 19.Holmes CJ, Shockley TR. Strategies to reduce glucose exposure in peritoneal dialysis patients. Perit Dial Int. 2000;20(Suppl 2):S37–S41. [PubMed] [Google Scholar]
- 20.Marshall J, Jennings P, Scott A, Fluck RJ, McIntyre CW. Glycemic control in diabetic CAPD patients assessed by continuous glucose monitoring system (CGMS) Kidney Int. 2003;64:1480–1486. doi: 10.1046/j.1523-1755.2003.00209.x. [DOI] [PubMed] [Google Scholar]
- 21.Davies SJ, Phillips L, Naish PF, Russell GI. Quantifying comorbidity in peritoneal dialysis patients and its relationship to other predictors of survival. Nephrol Dial Transplant. 2002;17:1085–1092. doi: 10.1093/ndt/17.6.1085. [DOI] [PubMed] [Google Scholar]
- 22.Rumpsfeld M, McDonald SP, Purdie DM, Collins J, Johnson DW. Predictors of baseline peritoneal transport status in Australian and New Zealand peritoneal dialysis patients. Am J Kidney Dis. 2004;43:492–501. doi: 10.1053/j.ajkd.2003.11.010. [DOI] [PubMed] [Google Scholar]
- 23.Rumpsfeld M, McDonald SP, Johnson DW. Higher peritoneal transport status is associated with higher mortality and technique failure in the Australian and New Zealand peritoneal dialysis patient populations. J Am Soc Nephrol. 2006;17:271–278. doi: 10.1681/ASN.2005050566. [DOI] [PubMed] [Google Scholar]
- 24.Kocak H, et al. Tuncer M: Advanced oxidative protein products are independently associated with endothelial function in peritoneal dialysis patients. Nephrology (Carlton) 2009;14:273–280. doi: 10.1111/j.1440-1797.2008.01062.x. [DOI] [PubMed] [Google Scholar]
- 25.Raj DS, Choudhury D, Welbourne TC, Levi M. Advanced glycation end products: a Nephrologist’s perspective. Am J Kidney Dis. 2000;35:365–380. doi: 10.1016/S0272-6386(00)70189-2. [DOI] [PubMed] [Google Scholar]
- 26.Lee MJ, et al. Glycemic control modifies difference in mortality risk between hemodialysis and peritoneal dialysis in incident dialysis patients with diabetes: Results from a nationwide prospective cohort in Korea. Medicine (Baltimore) 2016;95:e3118. doi: 10.1097/MD.0000000000003118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Hasegawa T, et al. Peritoneal Dialysis Registry With 2012 Survey Report. Ther Apher Dial. 2015;19:529–539. doi: 10.1111/1744-9987.12382. [DOI] [PubMed] [Google Scholar]
- 28.Fukui H, et al. PD + HD Combination Therapy Study Group: Review of combination of peritoneal dialysis and hemodialysis as a modality of treatment for end-stage renal disease. Ther Apher Dial. 2004;8:56–61. doi: 10.1111/j.1526-0968.2004.00107.x. [DOI] [PubMed] [Google Scholar]
- 29.Maruyama Y, Yokoyama K. Clinical efficacy of combined therapy with peritoneal dialysis and hemodialysis. Renal Replacement Therapy. 2016;2:11. doi: 10.1186/s41100-016-0023-5. [DOI] [Google Scholar]
- 30.Matsuo N, et al. Clinical impact of a combined therapy of peritoneal dialysis and hemodialysis. Clin Nephrol. 2010;74:209–216. doi: 10.5414/CNP74209. [DOI] [PubMed] [Google Scholar]
- 31.Maruyama Y, et al. EARTH (Evaluation of the Adequacy of Renal replacement THerapy) study group: Combined therapy with peritoneal dialysis and hemodialysis: a multicenter retrospective observational cohort study in Japan. Blood Purif. 2014;38:149–153. doi: 10.1159/000368389. [DOI] [PubMed] [Google Scholar]
- 32.Maruyama Y. Does Combined Therapy with Peritoneal Dialysis and Hemodialysis Improve Prognosis? Contrib Nephrol. 2018;196:64–70. doi: 10.1159/000485701. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.