Abstract
The tailor-made character of deep eutectic solvents (DES) turns them very attractive to be used in several applications, including in health-related areas such as pharmaceutical, nutraceutical, and cosmetic industries. However, although DES has been touted as “green” solvents, several works proved that their potential toxicity should not be neglected. Using the premise of DES applicability in the cosmetic and pharmaceutical sectors, we chose two cell lines to work as a skin model (keratinocytes HaCaT and tumor melanocytes MNT-1), to assess DES cytotoxicity. The effect of three different hydrogen bond acceptors (HBA) ([Chol]Cl, [N1111]Cl and [N4444]Cl) and three different hydrogen bond donors (HBD) (hexanoic and butanoic acid, ethylene glycol, 1-propanol and urea) were evaluated through a common viability assay (MTT assay). Results were promising since [Chol]Cl and [N1111]Cl- based DES showed good biocompatibility for the tested cells. [N4444]Cl-based DES, however, showed cytotoxicity for both cell lines, with the HBA being the driver of the toxicity. Interestingly, some compounds increased cell viability in the HaCaT cell line, namely [Chol]Cl, ethylene glycol, hexanoic acid, urea, and all [Chol]Cl and [N1111]Cl-based DES and should be considered as targets for future studies. These results highlight their possible use in cosmetic or pharmaceutical formulations.
Introduction
The field of “designer solvents” such as ionic liquids (IL) and deep eutectic solvents (DES) has been growing in the past decades, under the scope of “Green Chemistry”, which promotes the design and application of chemical products and processes that could reduce or preferentially eliminate the use and generation of hazardous substances1. DES were firstly developed in 2003 by combining urea and cholinium chloride2. These are prepared through the mixing of two or three different starting materials (e.g., quaternary ammonium salts, amides, organic acids, polyalcohols) forming an eutectic mixture based on hydrogen bonding interactions between a hydrogen bond donor (HBD) and an acceptor (HBA). These present a melting point much lower than either of the individual components2–5. This significant decrease in the melting point compared to starting materials is the result of several factors, such as the interaction between the salt’s anionic species and the HBD, the lattice energies, the nature and asymmetry of the organic salts, and the charges delocalization through the hydrogen bonding4. These new solvents are simpler to prepare and do not need complex purification schemes3,4. Moreover, DES are recognized as having a cheap production, (due to the low cost of starting materials), and showing a good biocompatibility with different biomolecules6–8.
The possible aplications for DES are almost endless owing to their designer character, and presently are mainly focused on chemical, electrochemical and material applications9. More recently, health-related industries such as the pharmaceutical, nutraceutical and cosmetic are also exploring these compounds due to their compatibility with biomolecules like DNA and enzymes10, among others. DES are suitable for biotransformation processes11, as well as to process biomass12,13, perform extractions14 and stabilize natural pigments15 as reviewed by Mbous et al.16. DES based on natural compounds, such as primary metabolites, like organic acids, amino acids and sugars5,10 have been labelled Natural Deep Eutectic Solvents (NADES). Nowadays, their study is a promising area in the field of cellular metabolism and physiology. Some authors (Choi et al.10) consider that these solvents could be involved on the biosynthesis of non-water soluble molecules and can act as solvents in living organisms, as water and lipids. DES may be particularly interesting for cosmetic proposes17,18, since the extraction technology using DES can mimic the processes that plants use to solubilize their essential molecules (flavonoids, anthocyanins and polymers). Moreover, the DES physical and chemical properties render them highly efficient in solubilizing compounds that are normally poorly water- or lipid-soluble17. In addition, social awareness turns consumers increasingly interested in low-toxic and natural solvents17. Although DES were initially considered as “green solvents”, mainly due to the benign nature of their constituents, only a few studies are available that assessed their toxic potential, either regarding their ecotoxicity6,19–27, or their cytotoxicity22,23,28–30. These few studies show that the DES toxic profile should be better characterized before general classifications of their benign character can be assumed. Moreover, information about the toxicity of DES is critical for a proper risk assessment under regulatory frameworks worldwide (e.g. the REACH regulation in Europe, which ensure the safety of chemical products for people and environment31).
This work aims to assess the cytotoxicity of a set of DES towards two human skin cell lines, HaCaT (keratinocytes cells) and MNT-1 (melanoma cells), considering the increased relevance of these solvents in many sectors of industry. HaCaT32–35, in particular, was chosen as model for cosmetic applications, while MNT-136–38 was selected as a model to understand the potential of the DES under study on the treatment of skin disorders. The DES effects on the cell viability were assessed through the MTT assay. More specifically, the effects of three different HBA and three different structural groups as HBD in DES cytotoxicity were evaluated supporting the drawing of informative toxicity trends. In this way, we tested fifteen DES at a 1:1 molar ratio (HBA:HBD), and rationally selected the starting materials. Two ammonium chlorides with different alkyl chain lengths (the larger the most toxic was the underlying assumption39), and cholinium chloride, which has been argued as biocompatible40,41, were selected as HBA. Then, two alcohols and two acids differing in their alkyl chain length, and number of functional groups, as well as an amine (urea) were selected to represent the most commonly functional groups applied as HBD for DES formulation.
Materials and Methods
Cell culture
Human melanoma MNT-1 cells were generously provided by Doctor Manuela Gaspar (iMed.ULisboa, Portugal). Immortalized human keratinocyte HaCaT cells were obtained from Cell Lines Services (Eppelheim, Germany). MNT-1 and HaCaT cells were cultured in Dulbecco’s modified Eagle’s medium (DMEM) supplemented with 10% of fetal bovine serum (FBS) and 1% of L-glutamine, penicillin–streptomycin and fungizone (Life Technologies, Grand Island, NY, USA). Both cell cultures were incubated in a humidified atmosphere at 37 °C and 5% of carbon dioxide – CO2. Cell morphology was observed using an inverted microscope Nikon Eclipse 80i (Nikon, Tokyo, Japan).
DES preparation
The following chemical compounds were used for DES preparation. As HBA, cholinium chloride ([Chol]Cl − 98% of purity) was purchased from Acros Organic®; tetramethylammonium chloride ([N1111]Cl − 97% of purity) and tetrabutylammonium chloride [N4444]Cl − 97% of purity) were purchased from Sigma-Aldrich. As HBD, ethylene glycol (99.5% of purity) was purchased from Sigma-Aldrich; 1-propanol (99.5% of purity) was purchased from Merck; butanoic acid (99% of purity) was purchased from Riedel de Haën; hexanoic acid (98% of purity) was purchased from SAFC; and urea (99% of purity) from Panreac. All DES were prepared at a molar ratio of 1:1, HBA:HBD. Briefly, HBDs and HBAs were added gravimetrically to closed vials and heated in a heat block with constant agitation. After the formation of a transparent liquid, the mixture was cooled down to room temperature. For some of these eutectic mixtures a known volume of water was added. The water content of both starting materials and DES was determined by Karl Fischer titration as detailed elsewhere42 and considered in calculations regarding cytotoxicity benchmarks.
MTT assay
The cytotoxic effects of HBAs, HBDs and DES were assessed by the colorimetric MTT assay43. Briefly, MNT-1 and HaCaT cells were seeded in 96-well plates and allowed to adhere. After adhesion, cells were incubated for 72 h exposed to a range of six concentrations (50–500 µg.mL−1), of the tested compounds diluted in DMEM medium (these previously sterilized with a 0.22 µm syringe filter), at 37 °C in 5% of CO2. After 72 h of exposure, 50 µL of MTT solution (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) from Sigma-Aldrich) (1 mg.mL−1 in PBS, pH 7.2) was added to each well. After 4 h of incubation, the medium was replaced with 150 µL of dimethyl sulfoxide (DMSO) to dissolve the formazan crystals. The plate was shaken for approximately 2 h, protected from light. Cell viability was measured through the optical density of reduced MTT at 570 nm using a microplate reader (Synergy HT from BioTeK Instruments Inc., Winooski, VT, USA). The percentage of viable cells was calculated as the ratio between the absorbance of treated versus control cells. Likewise, IC50 was defined as the concentration of chemical that leads to a 50% decrease in cell viability, calculated through a non-linear regression, logistic function.
Statistical analysis
Data were expressed as the mean ± standard deviation (SD) of at least three independent experiments with three technical replicates each. Data from each test (with HBD, HBA and DES) were analysed by one-way ANOVA, followed by a Dunnett’s test to evaluate the significance of disparities between the treatment groups and the control. In the absence of normality or homogeneity of variances, as assessed with Shapiro-Wilk and Brown-Forsythe tests, respectively, data were analysed by non-parametric one-way ANOVA (Kruskall-Wallis) followed by Dunnett’s test (only for [N1111]Cl:1-propanol in HaCaT cell line, the Dunn’s tests was used instead, due to unequal samples size). A value of p < 0.05 was considered statistically significant.
Results
In this study, keratinocyte cells (HaCaT) were used as a model of non-tumoral skin cells, and melanoma cells (MNT-1 cells) were used as a model of skin tumor cells. Most of the compounds showed to be non-cytotoxic after 24 and 48 h of exposure at low concentrations in preliminary trials. Thus, the concentration range was increased (0–500 µg.mL−1), as well as the exposure time for 72 h, in order to discard the possibility of toxic effects under more dramatic conditions.
Cytotoxicity of HBA and HBD
From the HBAs under study, only [N4444]Cl was found to be toxic for both cell types after 72 h of single exposure (see Table 1 and Supplementary Figs S1, S3). Regarding the HBDs, only butanoic acid showed a toxic effect for both cell types. [N1111]Cl did not exert any effect in cell viability in MNT-1, but increased the viability of HaCaT at 50 μg.mL−1 and significantly decreased it at 500 μg.mL−1. On the opposite, 1-propanol also did not exert any effect in cell viability in HaCaT, but significantly increased the viability in MNT-1 cells following exposure to all tested concentrations. Hexanoic acid increased cell viability in HaCaT, but produced a slight, yet statistically significant, reduction considering the higher concentrations towards the MNT-1 cells. All the remaining compounds increased cell viability at both cell lines (see Table 1 and Supplementary Figs S1, S3). A least pronounced effect was caused by [Chol]Cl, which only produced a significant increase at the concentration 50, 200 and 300 μg.mL−1 for the HaCaT cell line, and at the concentration of 200 μg.mL−1 for MNT-1 cells. Ethylene glycol caused the higher increase in cell viability especially in HaCaT cells, with some treatments reaching 40% of increase in cell viability compared to the control (see Supplementary Figs S1, S3).
Table 1.
Summary of the effects of HBAs and HBDs, on the two studied cell lines, after 72 h of exposure.
| Chemical compounds | Cell line | Effect trend | IC50 ± SD (μg.mL−1) | LOEC (μg.mL−1) | |
|---|---|---|---|---|---|
| HBA | [Chol]Cl | HaCaT MNT-1 |
Increase (slightly) Increase (slightly) |
— — |
50 200 |
| [N1111]Cl | HaCaT MNT-1 |
No effect No effect |
— — |
50 — |
|
| [N4444]Cl | HaCaT MNT-1 |
Toxic effect Toxic effect |
220.8 ± 6.2 316.1 ± 25.7 |
200 50 |
|
| HBD | Butanoic acid | HaCaT MNT-1 |
Toxic effect Toxic effect |
309.2 ± 11.6 274.8 ± 5.0 |
— — |
| Hexanoic acid | HaCaT MNT-1 |
Increase Toxic (slightly) |
— — |
50 50 |
|
| Ethylene glycol | HaCaT MNT-1 |
Increase Increase |
— — |
50 50 |
|
| 1-Propanol | HaCaT MNT-1 |
No effect Increase |
— — |
— 50 |
|
| Urea | HaCaT MNT-1 |
Increase Increase |
— — |
100 50 |
The effect trend concerns the effect of the chemicals in cell viability. Inhibitory concentration values (IC50) and Lowest Observed Effect Concentration (LOEC) (Dunnett test following one-way ANOVA; p < 0.05; see Supplementary Table S1 for the ANOVA summary) are also presented.
Cytotoxicity of DES
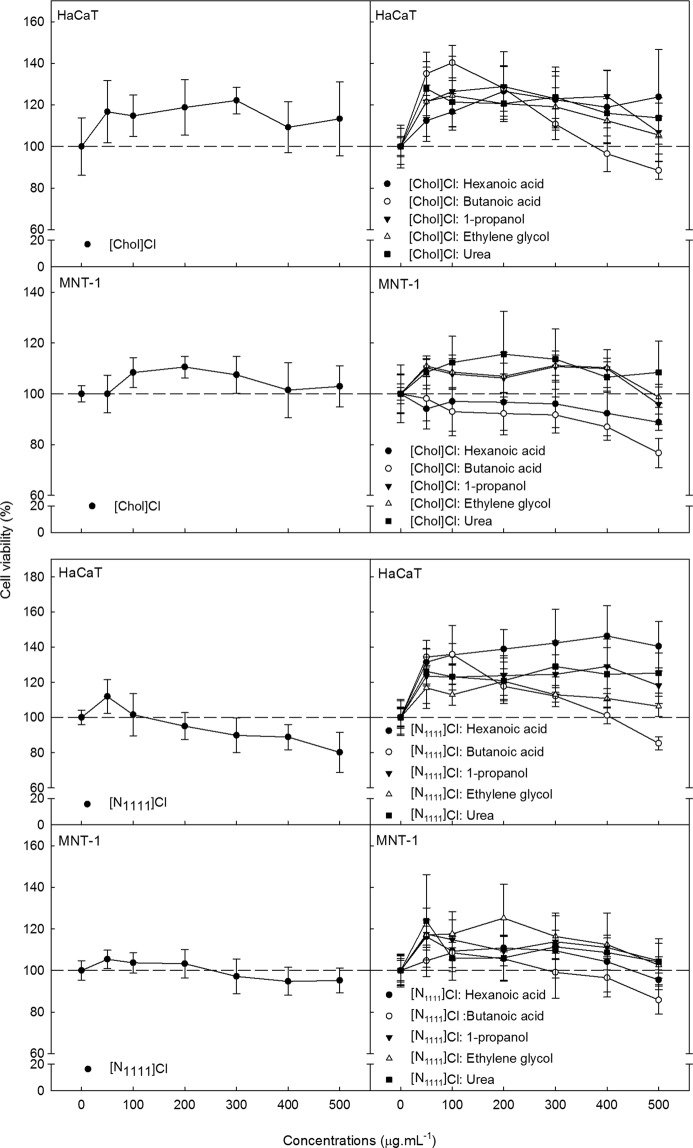
Cytotoxicity of DES is summarised in Table 2 and depicted in Supplementary Figs S2, S4. All [Chol]Cl-based DES promoted an increase in cell viability of HaCaT cells. The highest increase was found following exposure to [Chol]Cl:butanoic acid, reaching almost 40% at 50 and 100 µg.mL−1; however at 500 µg.mL−1, a decrease in cell viability was observed. This DES produced interesting results, since it was the only one promoting the cell viability in HaCaT, but it showed a toxic effect in MNT-1, at higher concentrations. However, and although significant, the decrease in MNT-1 cell viability was measured only up to 20% at the highest concentration tested (500 µg.mL−1), which prevented the calculation of median inhibitory concentrations. In MNT-1 cells, the viability increase observed following exposure to [Chol]Cl:ethylene glycol and [Chol]Cl:1-propanol (significant at 50, 100, 300 and 400 µg.mL−1) was smaller compared to the viability increase observed in HaCaT. Also for MNT-1 cells, [Chol]Cl:hexanoic acid and [Chol]Cl:urea produced no significant effects regarding cell viability.
Table 2.
Summary of the effect of the tested DES, for the two studied cell lines, after 72 h of exposure. The effect trend concerns the effect of the chemicals in cell viability.
| HBA: HBD 1:1 | Cell line | Effect trend | IC50 ± SD (μg.mL−1) | LOEC (μg.mL−1) | |
|---|---|---|---|---|---|
| DES | [Chol]Cl: Butanoic acid | HaCaT MNT-1 |
Increase Toxic (slightly) |
— — |
50 400 |
| [Chol]Cl: Hexanoic acid | HaCaT MNT-1 |
Increase No effect |
— — |
100 — |
|
| [Chol]Cl: Ethylene glycol | HaCaT MNT-1 |
Increase Increase |
— — |
50 50 |
|
| [Chol]Cl: 1-Propanol | HaCaT MNT-1 |
Increase Increase |
— — |
50 50 |
|
| [Chol]Cl: Urea | HaCaT MNT-1 |
Increase No effect |
— — |
50 — |
|
| [N1111]Cl: Butanoic acid | HaCaT MNT-1 |
Increase Toxic (slightly) |
— — |
50 500 |
|
| [N1111Cl: Hexanoic acid | HaCaT MNT-1 |
Increase Increase |
— — |
50 50 |
|
| [N1111]Cl: Ethylene glycol | HaCaT MNT-1 |
Increase Increase |
— — |
50 50 |
|
| [N1111]Cl: 1-Propanol | HaCaT MNT-1 |
Increase Increase |
— — |
50 50 |
|
| [N1111]Cl: Urea | HaCaT MNT-1 |
Increase Increase (slightly) |
— — |
50 50 |
|
| [N4444]Cl: Butanoic acid | HaCaT MNT-1 |
Toxic effect Toxic effect |
108.7 ± 4.2 472.6 ± 21.3 |
200 50 |
|
| [N4444]Cl: Hexanoic acid | HaCaT MNT-1 |
Toxic effect Toxic effect |
112.0 ± 4.3 476.1 ± 23.2 |
200 100 |
|
| [N4444]Cl: Ethylene glycol | HaCaT MNT-1 |
Toxic effect Toxic effect |
34.1 ± 5.0 300.9 ± 12.7 |
200 200 |
|
| [N4444]Cl: 1-Propanol | HaCaT MNT-1 |
Toxic effect Toxic effect |
77.9 ± 6.3 250.5 ± 7.6 |
200 50 |
|
| [N4444]Cl: Urea | HaCaT MNT-1 |
Toxic effect Toxic effect |
81.8 ± 3.1 496.7 ± 58.4 |
50 200 |
[N1111]Cl-based DES showed almost no toxicity for HaCaT and for MNT-1 cells. There was a slight cytotoxic effect observed following exposure to [N1111]Cl:butanoic acid towards both cell lines, but only at the highest concentration tested (500 µg.mL−1), while at the remaining concentrations an increase in cell viability was observed for HaCaT. Still regarding HaCaT, all [N1111]Cl-based DES produced an increase in cell viability, which is higher in [N1111]Cl:hexanoic acid by reaching the 40%. [N1111]Cl:hexanoic acid, [N1111]Cl:1-propanol and [N1111]Cl:ethylene glycol also increased MNT-1 viability in some treatments. Similarly to [Chol]Cl:urea, [N1111]Cl:urea induced no effects in MNT-1 cell viability except for the 50 µg.mL−1, where a significant increase of 20% was noticed.
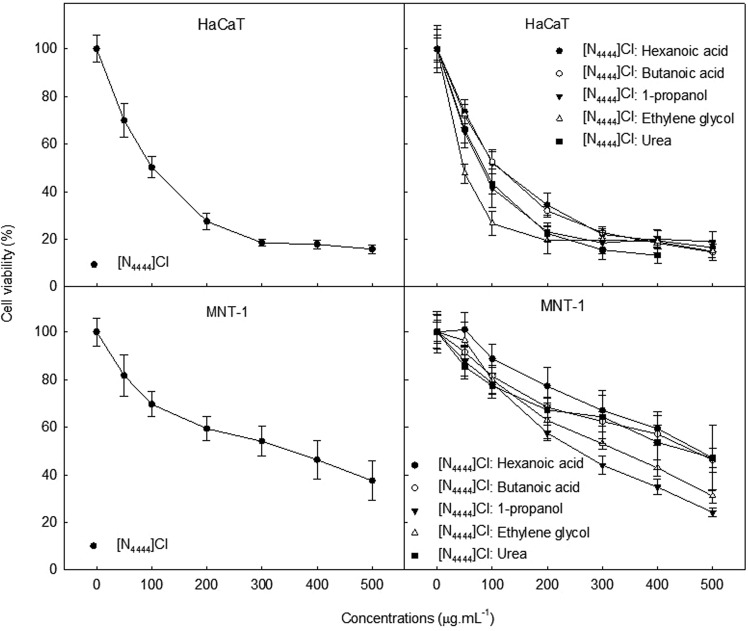
Considering [N4444]Cl-based DES, these bearing larger alkyl chains in comparison with the [N1111]Cl-based DES, all showed toxicity for both cell lines, regardless the HBD used. Such as [N4444]Cl, they were more toxic to HaCaT than to MNT-1 (compare IC50 values in Table 2). The most toxic DES with [N4444]Cl as HBA was [N4444]Cl:ethylene glycol, with an IC50 = 34.1 µg.mL−1 estimated for HaCaT cells. It is worth further noticing that the toxicity profile of [N4444]Cl-based DES is very similar to that observed for the starting material [N4444]Cl (see Fig. 1).
Figure 1.
Cell viability of [N4444]Cl (starting material) and [N4444]Cl-based DES for HaCaT and MNT-1 cells, after 72 h of exposure. Results are expressed as mean ± SD of three independent experiments.
Discussion
The study of DES toxicity is still in its infancy. Regarding their (eco)toxicity, the best studied systems are [Chol]Cl and phosphonium-based DES, which were assessed through a variety of organisms such as bacteria (Bacillus subtilis, Staphylococcus aureus, Pseudomonas aeruginosa6,19, Escherichia coli6,19,20, Aliivibrio fischeri21, Listeria monocytogenes, Salmonella enteriditis24); fungi (Phanerochaete chrysosporium, Candida cylindracea, Aspergillus niger, Lentinus tigrinus25, Sacharomyces cerevisae26); wheat seeds (Triticum aestivum)22; invertebrates, (Hydra sinensis20,27 and Artemia salina6,19); the plant Allium sativum20; the fish Cyprinus carpio25; and mice23.
The cytotoxicity of DES was also briefly addressed using different cell lines, vis. L929 fibroblast-like cells28, PC3 human prostate cancer, A375 human malignant melanoma23,29, OKF6 human oral keratinocyte, H413 carcinoma-derived human oral keratinocyte23, MCF-7 human breast cancer22,23,29,30, HelaS3 human cervical cancer29,30, CaOV3 human ovarian cancer, B16F10 mouse skin cancer30, AGS human gastric cancer, WRL-68 human hepatocyte29, and also CCO fish cells towards a non-human perspective22. While Hayyan et al.23 found that the studied [Chol]Cl-based DES exert relatively high cytotoxicity towards all cell lines, argued as higher than that by their individual components (i.e. glycerine, ethylene glycol, triethylene glycol and urea), Radošević et al.22 found that some DES (i.e.[Chol]Cl:oxalic acid) exerted moderate toxicity, while others (i.e.[Chol]Cl:glucose and [Chol]Cl:glycose) showed very low cytotoxicity (>2000 mg.L−1). Hayyan et al.30 and Paiva et al.28 found that NADES prepared with organic acids as HBD (e.g. malonic acid, citric acid and tartaric acid) were more cytotoxic to HelaS3, CaOV3, MCF-7, B16F10, and L929 cells. Through the main results, Hayyan et al.23 admitted that the HBD played a significant role in cytotoxicity. Finally, Mbous et al.29 used two [Chol]Cl-based NADES and a different DES (N,N-diethyl ethanolammonium chloride:triethylene glycol), and found that although all affect the viability of the tested cell lines, the DES was more toxic than the tested NADES.
In the present study, the cell models used reflected the potential applications of DES in the cosmetics and skin care industry. HaCaT32–35 and MNT-136–38 cell lines are widely used as human skin cell models. Surprisingly, given the previous records found in literature (see above), the tested DES did not show cytotoxicity with the exception of all [N4444]Cl-based DES, and many were able to stimulate cell viability instead, especially in non-tumoral cells. This shows that they could be considered as promising candidates for use in the cosmetic and/or pharmaceutical sectors.
Not much is known regarding the mechanisms of toxic action of DES and their starting materials, although the interaction with the biological membranes, as well as membrane damage possibly linked with oxidative stress imbalance, that cannot be hold by the antioxidant defence, have been argued relevant in this context23,29. Biological membranes are composed by a matrix of lipids and proteins, which regulates their permeability. One of the most important constituents of the lipid bilayer are phospholipids44, whose distribution across the membrane creates a membrane potential that regulates permeability and the diffusion of ionic and molecular species. Phospholipids consist on a ratio of functional groups on the cell surface (carboxyl, phosphate and amino groups), which depends on the cell type. The ratio between these functional groups determines the entry and the entry rate of extracellular materials44. According to Hayyan et al.23 and Mbous et al.29, the cytotoxic mechanism of DES involves an increase in the membrane permeability. Then, once inside the cell, the DES contributes to the increase in the concentration of reactive oxygen species (ROS), challenging the oxidative status of the cell. This could be the case of butanoic acid, [N4444]Cl and [N4444]Cl-based DES. Indeed, all of these compounds harm both non-tumoral (HaCaT) and tumor cells (MNT-1), probably through a similar mechanism. This was already observed by Mbous et al.29 with [Chol]Cl:fructose, [Chol]Cl:glucose and N,N-diethylethanolammonium chloride:triethylene glycol, in cancer cell lines (HelaS3, PC3, AGS, A375 and MCF-7) and the non-tumoral cell line WRL-68.
[N4444]Cl in particular, produced interesting results in the present study. Both [N4444]Cl and all [N4444]Cl-based DES proved to be toxic for both skin cell lines tested. For components like [N4444]Cl, strangers to the cell or that are required in smaller amounts, the intercellular diffusion is restricted and they are retained in the cell membrane, having a more pronounced deleterious effect29. Actually, the accumulation of ammonium cations (above a specific threshold concentration) on cellular membranes can disrupt the lipid bilayer and induce cell death45. The toxic profile of [N4444]Cl-based DES was almost the same as that observed for [N4444]Cl (see Fig. 1). This translated in the yielding of a toxic DES regardless the HBD combined with [N4444]Cl, supporting the assumption that this HBA is the driver of the toxicity within any of the DES. The role as a toxicity driver was not as clear for the other HBA studied ([N1111]Cl and [Chol]Cl) (see Fig. 2). De Morais (2015)21 assessed the ecotoxicity of [CholCl-based DES towards the marine bacteria A. fischeri and found that the HBD (acids) had a preponderant effect in the toxicity. In the present study, although butanoic acid was found toxic to both cell lines, when used as an HBD, the resulting DES were not always cytotoxic and often increased cell viability. According to literature, butanoic acid and similar compounds can induce apoptosis in different types of cancer cells46–49, but it can also serve as an anti-inflammatory agent and as a source of energy in some non-tumoral cells50. These converse mechanisms of toxicity may concur to explain the inconsistent toxicity trends observed, which importantly question the role of HBD as toxicity drivers. Contrasting to our results but in agreement with those by De Morais et al.21, which showed the importance of HBD, Hayyan et al.23 also evidenced that HBD such as ethylene glycol, triethylene glycol, glycerine and urea can play a significant role in DES toxicity. Overall, it seems clear that DES toxicity is hardly generalizable and dependent on the actual HBA:HBD combination and ratio.
Figure 2.
Cell viability of [Chol]Cl, and [N1111]Cl (as starting materials), and [Chol]Cl-based DES and [N1111]Cl-based DES for HaCaT and MNT-1 cells, after 72 h of exposure. Results are expressed as mean ± SD of three independent experiments.
Contributing to the melting pot above collecting on inconsistent responses to DES exposure, hexanoic acid and 1-propanol produced different results when assessed with tumoral or non-tumoral cells. The 1-propanol increases the viability in the tumor cell line MNT-1, while hexanoic acid increased viability in non-tumoral cells, and induced toxicity in cancer cells. This suggests that their mechanism of action is different in tumor and non-tumoral cells. A similar compound, the 6-hydroxyhexanoic acid, showed that human fibroblasts (HGF-1) growth inhibition only occurs at a high exposure concentration of 20 mM51. The toxicity of hexanoic acid (i.e. caproic acid), as well as other related fatty acids (e.g. capric and caprylic acids) in cancer cells lines (colorectal, skin and mammary human cell lines) was already examined by Narayanan et al.52, who found cell viability significantly inhibited after 48 h of exposure by 75% to 90% compared to the control. These fatty acids are involved in the down-regulation of genes important to cell division and in the up-regulations of genes necessary to apoptosis in skin and colon cancer cells. Moreover, the relative activity of caspase-8 of cancer cells (human colorectal carcinoma cells - HCT-116) treated with these fatty acids was significantly higher than that found in control cells52. Briefly, caspase-8 is located at the top of the hierarchy of the caspase cascade leading to the apoptotic death of the cells53.
Also relevant was the increase viability in HaCaT caused by [Chol]Cl:urea and [N1111]Cl:urea, which did not happened in MNT-1 cells. [Chol]Cl serves as cellular raw material for the synthesis of phospholipids membranes such as phatidycholine and sphingomyelin54. Thus, intracellular choline availability is crucial for the metabolism both in normal and cancer cells29. Therefore, we were not expecting to find cytotoxic signals as [Chol]Cl or derived DES were tested. Although the use of [Chol]Cl is forbidden in cosmetic formulations in UE55, [Chol]Cl-based DES are widely used in many different applications14,15,56–58 and therefore it is important to study possible irritation symptoms due to dermal contact. Indeed, [Chol]Cl was not toxic for either cell lines and was able to increase cell viability, a pattern also generally depicted for [Chol]Cl-based DES, in particular for cells exposed to [Chol]Cl:butanoic acid. Urea is also a natural compound and it is used in many commercial skin care lotions and creams, due to its moisturizing properties59. Urea and urea-based DES could increase cell viability in HaCaT and MNT-1. These were expected results, but still they contrast with those by Hayyan et al.23 who found toxic effects induced by [Chol]Cl:urea and respective starting materials towards the skin cancer cells A375. In our study, only [N4444]Cl:ethylene glycol exhibited toxicity records (IC50 = 34.1 µg.mL−1) similar to those shown by Hayyan et al.23. Ethylene glycol belongs to a family of chemicals that exert their pharmacological and/or toxicological effect through biotransformation. In the case of ethylene glycol, biotransformation involves the conversion of a substance to its active metabolite causing the biological response60. Ethylene glycol has little intrinsic toxicological activity61, but its metabolite glycolic acid exhibits nonlinear kinetics and has a dose-dependent transition that can lead to the development of toxicity in animals under specific circumstances60. Our results may hence have been driven by glycolic acid rather than ethylene glycol.
Regarding the putative role of the HBD functional group, inconsistent trends could be highlighted from the dataset. For example, butanoic but not hexanoic acid was cytotoxic, and when conjugated with [N1111]Cl, both acids contributed to an increase in cell viability. Conversely, the use of acids as HBD such as oxalic acid22, malonic acid30, tartaric acid, and citric acid28, generally present increased toxicity. Even extrapolating the results obtained for acids as HBD towards other organisms21, the mechanisms are still not easy to understand. If the pH imposed by the presence of acids could play an important role, some of the results obtained in this work prove that this specific condition should not be used as an heuristic rule to explain the main mechanism of (cyto)toxicity, as the results of this work show. Moreover, none of the tested alcohols produced cytotoxic effects, but ethylene glycol increased cell viability when tested alone, as well as when tested as HBD with [Chol]Cl and [N1111]Cl, while 1-propanol elicited no effects in HaCaT, but increased MNT-1 viability when dosed singly and when used as HBD with [Chol]Cl and [N1111]Cl in both cell lines.
Conclusions
DES have emerged in the last years as designer solvents with interesting properties and behavior, under the scope of “Green Chemistry”. Despite the increased attention given to DES, due to their promising applications in the cosmetic and pharmaceutical fields (just to mentioned a few), their characterization in terms of (cyto)toxicity is still very incomplete. In this context, most of the DES studied in this work were harmless for the cell lines HaCaT and MNT-1, even at high concentrations (500 μg.mL−1), thus both [Chol]Cl- and [N1111]Cl-based DES constitute promising benign compounds judging on their cytotoxic effects towards these two specific cell lines. Also, compounds like [Chol]Cl, ethylene glycol, hexanoic acid, urea, and all [Chol]Cl and [N1111]Cl-based DES allowed an increase in cell viability on the HaCaT cell line. Therefore, these compounds should be targets of future studies regarding their potential in skin regeneration. On the opposite, all the [N4444]Cl-based DES showed cytotoxicity. Moreover, the toxic profiles of these [N4444]Cl-based DES were similar to those exhibited by [N4444]Cl, suggesting that this HBA renders DES hazardous regardless the HBD used. Regarding the HBD, no consistent trends were obtained in the cytotoxic responses, with opposed effects being noticed between compounds bearing the same functional group. The increased cell viability caused by the majority of the tested compounds in non-tumoral cells (HaCaT) is an interesting aspect deserving further investigation. Still, it is important to understand the mechanisms behind this increase in cell viability, since it can represent cell proliferation promoting skin regeneration, but it can also indicate the possible activation of mutagenesis mechanisms.
Supplementary information
Cytotoxicity profiling of deep eutectic solvents to human skin cells
Acknowledgements
Thanks are due for the financial support to CESAM (UID/AMB/50017 - POCI-01-0145-FEDER-007638) to FCT/MCTES through national funds (PIDDAC), and the co-funding by the FEDER, within the PT2020 Partnership Agreement and Compete 2020. This work was also developed within the scope of the project CICECO-Aveiro Institute of Materials, POCI-01-0145-FEDER-007679 (FCT Ref. UID/CTM/50011/2013), financed by national funds through the FCT/MEC and when appropriate co-financed by FEDER under the PT2020 Partnership Agreement. This study was funded by FCT through the project PTDC/ATP-EAM/5331/2014 and PTDC/BTM-MAT/31794/2017 (POCI-01-0145-FEDER-031794), and through the individual research grants awarded to I.P.E. Macário (SFRH/BD/123850/2016), A.C. Menezes (BI/CESAM/012/AMB/50017) and A.M.M. Gonçalves (SFRH/BPD/97210/2013). J.L. Pereira and H. Oliveira are funded by national funds (OE), through FCT – Fundação para a Ciência e a Tecnologia, I.P., in the scope of the framework contract foreseen in the numbers 4, 5 and 6 of the article 23, of the Decree-Law 57/2016, of August 29, changed by Law 57/2017, of July 19. A. M. M. Gonçalves also acknowledges University of Coimbra for the contract IT057-18-7253 and the research unit MARE (UID/MAR/04292/2013). S.P.M. Ventura acknowledges FCT for the contract IF/00402/2015 under the Investigador FCT 2015 program.
Author Contributions
I.P.E.M., H.O., J.L.P., S.P.M.V., A.M.M.G., J.A.P.C. and F.J.M.G. contribute to conceive the idea; I.P.E.M. and C.M. perform the experiments; I.P.E.M., H.O., J.L.P., S.P.M.V. wrote the paper. F.J.M.G. and J.A.P.C. were responsible for the oversight of the project. All authors critically read and discussed the manuscript.
Competing Interests
The authors declare no competing interests.
Footnotes
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary information accompanies this paper at 10.1038/s41598-019-39910-y.
References
- 1.Anastas, P. T. & Warner, J. C. Green Chemistry: Theory and Practice. (Oxford University Press, 1998).
- 2.Abbott AP, Capper G, Davies DL, Rasheed RK, Tambyrajah V. Novel solvent properties of choline chloride/urea mixtures. Chem. Commun. 2003;99:70–71. doi: 10.1039/b210714g. [DOI] [PubMed] [Google Scholar]
- 3.Ruß C, König B. Low melting mixtures in organic synthesis – an alternative to ionic liquids? Green Chem. 2012;14:2969–2982. doi: 10.1039/c2gc36005e. [DOI] [Google Scholar]
- 4.Zhang QH, Vigier KD, Royer S, Jerome F. Deep eutectic solvents: syntheses, properties and applications. Chem. Soc. Rev. 2012;41:7108–7146. doi: 10.1039/c2cs35178a. [DOI] [PubMed] [Google Scholar]
- 5.Dai Y, van Spronsen J, Witkamp GJ, Verpoorte R, Choi YH. Natural deep eutectic solvents as new potential media for green technology. Anal. Chim. Acta. 2013;766:61–68. doi: 10.1016/j.aca.2012.12.019. [DOI] [PubMed] [Google Scholar]
- 6.Hayyan M, et al. Are deep eutectic solvents benign or toxic? Chemosphere. 2013;90:2193–2195. doi: 10.1016/j.chemosphere.2012.11.004. [DOI] [PubMed] [Google Scholar]
- 7.Jhong HR, Wong DSH, Wan CC, Wang YY, Wei TC. A novel deep eutectic solvent-based ionic liquid used as electrolyte for dye-sensitized solar cells. Electrochem. commun. 2009;11:209–211. doi: 10.1016/j.elecom.2008.11.001. [DOI] [Google Scholar]
- 8.Singh BS, Lobo HR, Shankarling GS. Choline chloride based eutectic solvents: Magical catalytic system for carbon – carbon bond formation in the rapid synthesis of β -hydroxy functionalized derivatives. Catal. Commun. 2012;24:70–74. doi: 10.1016/j.catcom.2012.03.021. [DOI] [Google Scholar]
- 9.Smith EL, Abbott AP, Ryder KS. Deep Eutectic Solvents (DESs) and Their Applications. Chem. Rev. 2014;114:11060–11082. doi: 10.1021/cr300162p. [DOI] [PubMed] [Google Scholar]
- 10.Choi YH, et al. Are Natural Deep Eutectic Solvents the Missing Link in Understanding Cellular Metabolism and Physiology? Plant Physiol. 2011;156:1701–1705. doi: 10.1104/pp.111.178426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Gutiérrez MC, Ferrer ML, Yuste L, Rojo F, Monte F. Angew. Chemie - Int. Ed. 2010. Del. Bacteria incorporation in deep-eutectic solvents through freezedrying; pp. 2158–2162. [DOI] [PubMed] [Google Scholar]
- 12.Xia S, Baker G, Li H, Ravula S, Zhao H. Aqueous ionic liquids and deep eutectic solvents for cellulosic biomass pretreatment and saccharification. RSC Adv. 2014;4:10586–10596. doi: 10.1039/c3ra46149a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.De Faria ELP, et al. Deep eutectic solvents as efficient media for the extraction and recovery of cynaropicrin from cynara cardunculus L. Leaves. Int. J. Mol. Sci. 2017;18:2276. doi: 10.3390/ijms18112276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Ruesgas-Ramón M, Figueroa-Espinoza MC, Durand E. Application of Deep Eutectic Solvents (DES) for Phenolic Compounds Extraction: Overview, Challenges, and Opportunities. J. Agric. Food Chem. 2017;65:3591–3601. doi: 10.1021/acs.jafc.7b01054. [DOI] [PubMed] [Google Scholar]
- 15.Dai Y, Verpoorte R, Choi YH. Natural deep eutectic solvents providing enhanced stability of natural colorants from safflower (Carthamus tinctorius) Food Chem. 2014;159:116–121. doi: 10.1016/j.foodchem.2014.02.155. [DOI] [PubMed] [Google Scholar]
- 16.Mbous YP, et al. Applications of deep eutectic solvents in biotechnology and bioengineering—Promises and challenges. Biotechnol. Adv. 2017;35:105–134. doi: 10.1016/j.biotechadv.2016.11.006. [DOI] [PubMed] [Google Scholar]
- 17.Laguerre, M. & Lavaud, A. The rise of deep eutectics from nature to cosmetics. Naturex 45–47 (2016).
- 18.Jeong KM, et al. Multi-functioning deep eutectic solvents as extraction and storage media for bioactive natural products that are readily applicable to cosmetic products. J. Clean. Prod. 2017;151:87–95. doi: 10.1016/j.jclepro.2017.03.038. [DOI] [Google Scholar]
- 19.Hayyan M, et al. Assessment of cytotoxicity and toxicity for phosphonium-based deep eutectic solvents. Chemosphere. 2013;93:455–459. doi: 10.1016/j.chemosphere.2013.05.013. [DOI] [PubMed] [Google Scholar]
- 20.Wen Q, Chen JX, Tang YL, Wang J, Yang Z. Assessing the toxicity and biodegradability of deep eutectic solvents. Chemosphere. 2015;132:63–69. doi: 10.1016/j.chemosphere.2015.02.061. [DOI] [PubMed] [Google Scholar]
- 21.De Morais P, Gonçalves F, Coutinho JAP, Ventura SPM. Ecotoxicity of Cholinium-Based Deep Eutectic Solvents. ACS Sustain. Chem. Eng. 2015;3:3398–3404. doi: 10.1021/acssuschemeng.5b01124. [DOI] [Google Scholar]
- 22.Radošević K, et al. Evaluation of toxicity and biodegradability of choline chloride based deep eutectic solvents. Ecotoxicol. Environ. Saf. 2015;112:46–53. doi: 10.1016/j.ecoenv.2014.09.034. [DOI] [PubMed] [Google Scholar]
- 23.Hayyan M, Looi CY, Hayyan A, Wong WF, Hashim MA. In Vitro and in Vivo toxicity profiling of ammonium-based deep eutectic solvents. PLoS One. 2015;10:1–18. doi: 10.1371/journal.pone.0117934. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Zhao BY, et al. Biocompatible Deep Eutectic Solvents Based on Choline Chloride: Characterization and Application to the Extraction of Rutin from Sophora japonica. ACS Sustain. Chem. Eng. 2015;3:2746–2755. doi: 10.1021/acssuschemeng.5b00619. [DOI] [Google Scholar]
- 25.Juneidi I, Hayyan M, Mohd Ali O. Toxicity profile of choline chloride-based deep eutectic solvents for fungi and Cyprinus carpio fish. Environ. Sci. Pollut. Res. 2016;23:7648–7659. doi: 10.1007/s11356-015-6003-4. [DOI] [PubMed] [Google Scholar]
- 26.Cardellini F, et al. Room temperature deep eutectic solvents of (1S)-(+)-10-camphorsulfonic acid and sulfobetaines: hydrogen bond-based mixtures with low ionicity and structure-dependent toxicity. RSC Adv. 2015;40:31772–31786. doi: 10.1039/C5RA03932K. [DOI] [Google Scholar]
- 27.Huang ZL, Wu BP, Wen Q, Yang TX, Yang Z. Deep eutectic solvents can be viable enzyme activators and stabilizers. J. Chem. Technol. Biotechnol. 2014;89:1975–1981. doi: 10.1002/jctb.4285. [DOI] [Google Scholar]
- 28.Paiva A, et al. Natural Deep Eutectic Solvents − Solvents for the 21st Century. ACS Sustain. Chem. Eng. 2014;2:1063–1071. doi: 10.1021/sc500096j. [DOI] [Google Scholar]
- 29.Mbous YP, Hayyan M, Wong WF, Looi CY, Hashim MA. Unraveling the cytotoxicity and metabolic pathways of binary natural deep eutectic solvent systems. Sci. Rep. 2017;7:41257. doi: 10.1038/srep41257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Hayyan M, et al. Natural deep eutectic solvents: cytotoxic profile. Springerplus. 2016;5:913. doi: 10.1186/s40064-016-2575-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.EU. Corrigendum to Regulation (EC) No 1907/2006 of the European Parliament and of the Council of 18 December 2006 concerning the Registration, Evaluation, Authorisation and Restriction of Chemicals (REACH) (2007).
- 32.Seo M, Kang T, Lee C, Lee A, Noh M. HaCaT Keratinocytes and Primary Epidermal Keratinocytes Have Different Transcriptional Profiles of Cornified Envelope-Associated Genes to T Helper Cell Cytokines. Biomol Ther (Seoul). 2012;20:171–6. doi: 10.4062/biomolther.2012.20.2.171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.McNeilly AD, Woods Ja, Ibbotson SH, Wolf CR, Smith G. Characterization of a human keratinocyte HaCaT cell line model to study the regulation of CYP2S1. Drug Metab. Dispos. 2012;40:283–9. doi: 10.1124/dmd.111.042085. [DOI] [PubMed] [Google Scholar]
- 34.Abdelkader DH, et al. Effect of poly(ethylene glycol) on insulin stability and cutaneous cell proliferation in vitro following cytoplasmic delivery of insulin-loaded nanoparticulate carriers – A potential topical wound management approach. Eur. J. Pharm. Sci. 2018;114:372–384. doi: 10.1016/j.ejps.2017.12.018. [DOI] [PubMed] [Google Scholar]
- 35.Genç H, et al. Biocompatibility of designed MicNo-ZnO particles: Cytotoxicity, genotoxicity and phototoxicity in human skin keratinocyte cells. Toxicol. Vitr. 2018;47:238–248. doi: 10.1016/j.tiv.2017.12.004. [DOI] [PubMed] [Google Scholar]
- 36.Menezes AC, Carvalheiro M. Ferreira de Oliveira, J. M. P., Ascenso, A. & Oliveira, H. Cytotoxic effect of the serotonergic drug 1-(1-Naphthyl)piperazine against melanoma cells. Toxicol. Vitr. 2018;47:72–78. doi: 10.1016/j.tiv.2017.11.011. [DOI] [PubMed] [Google Scholar]
- 37.Vighi E, et al. New cGMP analogues restrain proliferation and migration of melanoma cells. Oncotarget. 2018;9:5301–5320. doi: 10.18632/oncotarget.23685. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Silva Z, Veríssimo T, Videira PA, Novo C. Protein disulfide isomerases: Impact of thapsigargin treatment on their expression in melanoma cell lines. Int. J. Biol. Macromol. 2015;79:44–48. doi: 10.1016/j.ijbiomac.2015.04.029. [DOI] [PubMed] [Google Scholar]
- 39.Stolte S, et al. Effects of different head groups and functionalised side chains on the aquatic toxicity of ionic liquids. Green Chem. 2007;9:1170–1179. doi: 10.1039/b711119c. [DOI] [Google Scholar]
- 40.Florindo C, Oliveira FS, Rebelo LPN, Fernandes AM, Marrucho IM. Insights into the synthesis and properties of deep eutectic solvents based on choline chloride and carboxylic acids. ACS Sustain. Chem. Eng. 2014;2:2416–2425. doi: 10.1021/sc500439w. [DOI] [Google Scholar]
- 41.FEEDAP. Scientific Opinion on safety and efficacy of choline chloride as a feed additive for all animal species (2011).
- 42.Passos H, Tavares DJP, Ferreira AM, Freire MG, Coutinho JAP. Are Aqueous Biphasic Systems Composed of Deep Eutectic Solvents Ternary or Quaternary Systems? ACS Sustain. Chem. Eng. 2016;4:2881–2886. doi: 10.1021/acssuschemeng.6b00485. [DOI] [Google Scholar]
- 43.Twentyman PR, Luscombe M. A study of some variables in a tetrazolium dye (MTT) based assay for cell growth and chemosensitivity. Br. J. Cancer. 1987;56:279–285. doi: 10.1038/bjc.1987.190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Nelson, D. L. & Cox, M. M. Lehninger - Principles of Biochemistry. (Freeman, W. H. & Company, 2012).
- 45.Gal N, Malferarri D, Galletti P, Tagliavini E, Jelinek R. Biochimica et Biophysica Acta Membrane interactions of ionic liquids: Possible determinants for biological activity and toxicity. Biochim. Biophys. Acta. 2012;1818:2967–2974. doi: 10.1016/j.bbamem.2012.07.025. [DOI] [PubMed] [Google Scholar]
- 46.Leng SL, Leeding KS, Gibson PR, Bach LA. Insulin-like growth factor-II renders LIM 2405 human colon cancer cells resistant to butyrate-induced apoptosis: a potential mechanism for colon cancer cell survival in vivo. Carcinogenesis. 2001;22:1625–31. doi: 10.1093/carcin/22.10.1625. [DOI] [PubMed] [Google Scholar]
- 47.Rozental R, et al. Sodium butyrate induces apoptosis in MSN neuroblastoma cells in a calcium independent pathway. Neurochem. Res. 2004;29:2125–2134. doi: 10.1007/s11064-004-6886-9. [DOI] [PubMed] [Google Scholar]
- 48.Rabizadeh E, et al. Doxorubicin and a butyric acid derivative effectively reduce levels of BCL-2 protein in the cells of chronic lymphocytic leukemia patient. Eur. J. Haematol. 2001;66:263–271. doi: 10.1034/j.1600-0609.2001.066004263.x. [DOI] [PubMed] [Google Scholar]
- 49.Singh NP, Lai HC. Synergistic cytotoxicity of artemisinin and sodium butyrate on human cancer cells. Anticancer Res. 2005;25:4325–4331. [PubMed] [Google Scholar]
- 50.Treptow-Van Lishaut S, Rechkemmer G, Rowland I, Dolara P, Pool-Zobel BL. The carbohydrate crystalean and colonic microflora modulate expression of glutathione S-transferase subunits in colon of rats. Eur. J. Nutr. 1999;38:76–83. doi: 10.1007/s003940050047. [DOI] [PubMed] [Google Scholar]
- 51.Orchel A, Jelonek K, Kasperczyk J, Dzierzewicz Z. Growth of human fibroblasts in the presence of 6-hydroxyhexanoic acid. Acta Pol. Pharm. - Drug Res. 2010;67:710–712. [PubMed] [Google Scholar]
- 52.Narayanan A, Baskaran SA, Amalaradjou MAR, Venkitanarayanan K. Anticarcinogenic properties of medium chain fatty acids on human colorectal, skin and breast cancer cells in vitro. Int. J. Mol. Sci. 2015;16:5014–5027. doi: 10.3390/ijms16035014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Kruidering M, Evan G. Caspase-8 in Apoptosis: The Beginning of ‘The End’? IUBMB Life. 2000;50:85–90. doi: 10.1080/713803693. [DOI] [PubMed] [Google Scholar]
- 54.Lodish, H. et al. Molecular cell biology. (2000).
- 55.EU. Regulmento (CE) N°1223/2009 Do Parlamento Europeu e do Conselho. Jornal Oficial da União Europeia 151 (2009).
- 56.Zhao B-Y, et al. Biocompatible Deep Eutectic Solvents Based on Choline Chloride: Characterization and Application to the Extraction of Rutin from Sophora japonica. ACS Sustain. Chem. Eng. 2015;3:2746–2755. doi: 10.1021/acssuschemeng.5b00619. [DOI] [Google Scholar]
- 57.Alañón, M. E., Ivanović, M., Gómez-Caravaca, A. M., Arráez-Román, D. & Segura-Carretero, A. Choline chloride derivative-based deep eutectic liquids as novel green alternative solvents for extraction of phenolic compounds from olive leaf. Arab. J. Chem. in press (2018).
- 58.Vieira V, et al. Enhanced extraction of phenolic compounds using choline chloride based deep eutectic solvents from Juglans regia L. Ind. Crops Prod. 2018;115:261–271. doi: 10.1016/j.indcrop.2018.02.029. [DOI] [Google Scholar]
- 59.Björklund S, Engblom J, Thuresson K, Sparr E. Glycerol and urea can be used to increase skin permeability in reduced hydration conditions. Eur. J. Pharm. Sci. 2013;50:638–645. doi: 10.1016/j.ejps.2013.04.022. [DOI] [PubMed] [Google Scholar]
- 60.Slikker W, et al. Dose-dependent transitions in mechanisms of toxicity: Case studies. Toxicol. Appl. Pharmacol. 2004;201:226–294. doi: 10.1016/j.taap.2004.06.027. [DOI] [PubMed] [Google Scholar]
- 61.Carney EW. An integrated perspective on the developmental toxicity of ethylene glycol. Reprod. Toxicol. 1994;8:99–113. doi: 10.1016/0890-6238(94)90017-5. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Cytotoxicity profiling of deep eutectic solvents to human skin cells