Abstract
The extracts prepared from various areal parts of the Adenium obesum (Forssk.) Roem. & Schult. (Family: Apocynaceae) including leaves, fruit and seeds ethanolic extracts and seed aqueous extract were evaluated against MCF-7 cells in order to investigate its potential of cytogenotoxicity and induction of apoptosis. The ethanolic seeds extract had comparatively higher cytotoxicity (IC50 ∼ 337 µg/ml). Further, apoptosis and DNA damaging potential of seeds ethanolic extract was analyzed by applying multiple sub-lethal concentrations and durations. Flow cytometry results revealed that maximum percentage of early apoptosis (37%) and late apoptosis (35%) were observed after 12 h exposure in concentrations 200 µg/ml and 300 µg/ml, respectively. Similarly, the higher effect of extract in terms of DNA damage by alkaline comet assay was registered after 12 h treatment at concentrations 200 and 300 µg/mL. The calculated total damage score (TDS) for these concentrations were 614 and 617, respectively. The above findings indicate that A. obesum ethanolic seeds extracts has cytogenotoxic properties that could be further explored for the potential source of chemotherapeutic lead against cancer.
Keywords: Comet assay, Apoptosis, Cytotoxicity, Cancer cells, Adenium obesum, Apocynaceae
1. Introduction
Female breast cancer is the most commonly diagnosed cancer and the leading cause of death (Bray et al., 2018). The malignancy (invasion and metastasis) of cancer makes its treatment difficult (Kumar et al., 2005). The treatment of cancer using the modern methods such as chemotherapy and radiotherapy are well-known for various side effects (Stopeck and Thompson, 2012), and their resistance to the conventional chemotherapy and radiotherapy too, the alternative treatment options of cancer continues. Therefore, the exploration of an effective drug candidate for the treatment of cancer including other diseases too from the wild plant resources through plant bioprospecting is of big interest worldwide today owing to the drug candidate derived from the plant natural resources are safer and having least side effects (da Rocha et al., 2001, Vermani and Garg, 2002, Gurib-Fakim, 2006, Baker et al., 2007, Shukla and Singh, 2007).
The majority of the chemotherapeutic anticancer candidates currently in the clicinal use such as paclitaxel (Razis and Fountzilas, 2001, Goodman and Walsh, 2001), etoposide (Hainsworth and Greco, 1995), camptothecin (Garcia-Carbonero and Supko, 2002) and vinca alkaloids (Jordan et al., 1991) originally derived from plants. These anticancer agents act through a range of mechanism which includes enzyme modulation and gene expression (Webb and Ebeler, 2004), antimutagenic activity (De Flora et al., 2001) and antioxidant and apoptosis (Miadokova et al., 2008).
Adenium obesum (Forssk) Roem. & Schult, family Apocynaceae (-commonly known as ‘desert rose’), previously been reported to have anti-influenza virus (Kiyohara et al., 2012), antimicrobial (Hossain et al., 2017), and cytotoxic activity (Almehdar et al., 2012). It is a rich source of cardiac glycosides, containing approximately 50 phytochemicals belong to the class of cardenolides, pregnanes, triterpenes, and flavonoids (Versiani et al., 2014). Traditionally, different parts of this plant is used for the treatment of various ailments in the middle-East region such as skin diseases, wounds, muscle pain, and joint pain. (Mouza and Hossain, 2015). Recently, a concise review on A. obesum have been presented on its medicinal attributes and biological activities (Paul et al., 2016). However, it’s cytotoxic and genotoxic potential and modes of action of chemicals constituents have not been clearly defined. Hence, it is aimed to unravel the anticancer potential of the leaves extracts of A. obesum against the MCF-7 breast cancer cells.
2. Materials and methods
2.1. Preparation of crude extracts
The leaves, fruits and seeds of A. obesum were collected from natural habitat near to Riyadh, Saudi Arabia, and oven-dried at 50 °C. The powdered plant material (1000 g) was used to prepare crude extract either in water (aqueous) or 95% ethanol. The rotary evaporator at low temperature and pressure was used to bring the extract into semi solid state. This crude extracts was stored at −20 °C until used in the experiments.
2.2. Cell viability and determination of IC50 value
The MTT colorimetric assay (Mosmann, 1983) with modifications (Farah et al, 2016) was used to analyse the cytotoxic activity of extracts against the MCF-7 human breast adenocarcinoma cells cultured in MEM supplemented with 15% FBS, maintained at 37 °C with supply of 5% CO2. A two-fold dilutions starting from 1000 μg/mL (total 6 concentrations) of all the extracts of A. obesum were prepared and MCF-7 cells were exposed for 24 h. Subsequently, seeds ethanolic extracts was applied in four narrow range of concentrations for IC50 determination. Further, to identify any changes in the morphology, MCF-7 cells were exposed to the three sub-lethal concentrations below IC50 value (100, 200, and 300 μg/mL). At the end of desired treatment (24 h), cells were observed at 100× magnification under phase contrast inverted microscope (Olympus IX51, Tokyo, Japan).
2.3. Detection of apoptosis by flow cytometry
The MCF-7 cells were incubated with ethanolic seed extract (100 μg/mL, 200 μg/mL and 300 μg/mL) for three durations (i.e., 6 h, 12 h and 24 h), and the Annexin-V FITC apoptosis detection Kit (BD, Biosciences) was used for the sample preparation for the differentiation of early and late apoptotic, and necrotic cells using flow cytometry (BD FACS Calibur).
2.4. Determination of DNA damage
The MCF-7 cells were grown, and then treated with concentrations of 100, 200 and 300 μg/mL) of ethanolic seed extract the three durations (i.e. 6 h, 12 h and 24 h). The alkaline comet assay (Singh et al., 1988) was performed with additional modifications described previously in detail (Ateeq et al., 2005). The DNA damage was evaluated by the visual scoring of 300 comets of each concentration according to the standard criteria (Collins, 2004, Azqueta et al., 2009) by dividing the tail size into four classes (0, 1, 2 and 3). The total damage score (TDS) was calculated by multiplying the number of cells in each class by the damage class.
2.5. Statistical analysis
Three replicates in each experiments were maintained including controls. The values were presented as mean ± standard error of mean (SEM). The calculation and plotting of mean and standard deviation estimates in the graphs was performed using Microsoft Office Excel. Student’s t-test was used to analyze the data applying a significance level of p < 0.05.
3. Results
3.1. Cell viability and determination of IC50 value
The MTT assay was performed for preliminary screening of the ethanolic extract of leaves, fruit and seeds and aqueous extract of seeds in order to compare and select the more effective extract against MCF-7 cells for subsequent experiments. Fig. 1(A) represents summarized data of cell viability of all extracts for comparison. The ethanolic leaves extract shows random effects on MCF-7 cells. The cell growth inhibition was not dependent on the concentration of extract. The minimum cell viability was 62.2% at 250 µg/mL, while at the highest concentration cell viability was 79.5% and at the lowest concentration it reached 82.8%. There was an insignificant (p < 0.05) weak inverse linear correlation between the extract concentrations and cell viability. The ethanolic fruit extract showed biphasic effect of this extract on MCF-7 cells. The highest concentration (1000 µg/mL) of ethanolic fruit extract significantly inhibited cell growth and the cell viability was reduced to 46.4% compared to control. At lowest concentration (31 µg/mL) 84.7% of the cell viability was observed. The effect of fruit ethanolic extract was dependent on concentration at higher ranges only. There is a highly significant (p < 0.001) inverse linear correlation between the extract concentration and cell viability; while, the ethanolic seed extract showed a significant positive effect. More than 40% of the cells growth was inhibited at concentration 125 µg/mL, while at the maximum concentration (1000 µg/mL), only 12.1% of cells were found to be viable. At the lowest concentration of (31 µg/mL), 79.7% viable cells were observed. There is a highly significant (p < 0.001) inverse linear correlation between the extract concentrations and cell viability; while, the aqueous seed extract shows random effects on MCF-7 cells, and was not dependent on concentrations of the extract. However, minimum cell viability of 87.4% was observed at the highest concentration. The aqueous seed extract was found to be an ineffective to the MCF-7 cells. There is a weak, statistically insignificant (p < 0.05), inverse linear correlation between the aqueous extract concentrations and cell viability.
Fig. 1.
(A) Comparison of MTT cell viability assays of MCF-7 cell treated with four extracts of A. obesum (ethanolic extract of leaves, fruits and seeds and aqueous extract of seeds) after 24 h of exposure. (B) MTT cell viability assay of MCF-7 cells treated with ethanolic seeds extract with indicated concentrations for 24 h. The IC50 value was estimated as 337 µg/mL.
The ethanolic seed extract exerted significant cytotoxic effect on MCF-7 cells. Therefore, further analysed to determine IC50. Since two higher concentrations of 500 and 1000 µg/mL inhibited more than 50% cell proliferation, hence four concentrations between 100 and 400 µg/mL were used for determination of IC50 value. The results are summarized in Fig. 1 (B) that showed strong effect of ethanolic seeds extract on MCF-7 cells. At concentration 100 µg/mL of ethanolic seeds extract about 81.4% of cells are viable, whereas only 31.8% of cells were viable at concentration 400 µg/mL of this extract. The IC50 of ethanolic seeds extract was calculated from trendline equations using MS Excel®, it was estimated as 337 µg/mL; hence, the ethanolic seeds extract was used for subsequent experimentation for apoptosis detection and DNA damage evaluation in the present study. Three sub-lethal concentrations (100, 200 and 300 µg/mL) below IC50 were applied to MCF-7 cells for multiple durations (i.e. 6 h, 12 h, and 24 h).
The morphological changes in MCF-7 cells was presented in Fig. 2(A–D). Cellular changes were progressive from normal morphology of regular, spindle or polygonal adherent cells to irregular, detached cells, with increased intercellular space due to treatment with ethanolic seeds extract.
Fig. 2.
(A) Photomicrograph of untreated MCF-7 cells. Cells are polygonal or spindle-shaped and tightly packed. MCF-7 cells treated for 24 h with ethanolic seeds extracts. (B) 100 µg/mL, more cells are detached (become round or irregular instead of spindle shape), there is increase in intercellular spaces and there is increase of cell-free area. (C) 200 µg/mL, there are many irregularly shaped cells and increase in the intercellular space. (D) 300 µg/mL, almost all cells are shrinked, irregular round, losing their normal spindle shape morphology.
3.2. Apoptosis induction by A. obesum ethanolic seeds extracts
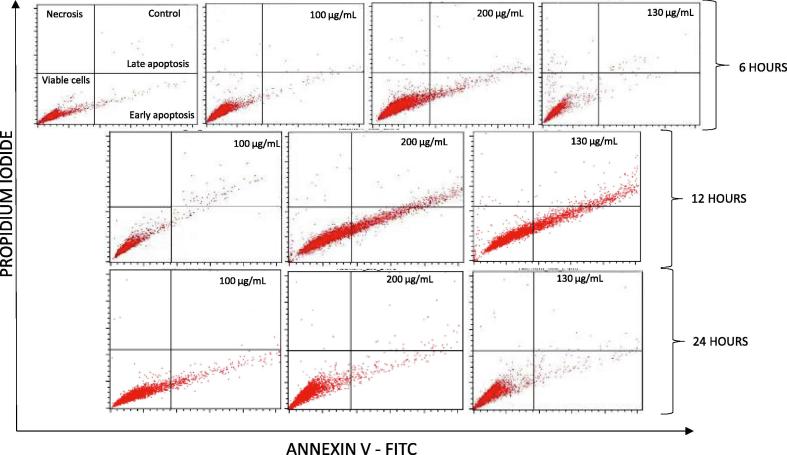
Fig. 3 shows the cytotoxic effect of A. obesum ethanolic seeds extract (100, 200 and 300 µg/mL) on MCF-7 cells treated for the duration of 6 h, 12 h and 24 h measured by flow cytometry. In the absence of extract (control), only 2% of cells were in early apoptosis (Annexin V +ve/PI −ve) and less than 1% cells were in late apoptosis (Annexin V +ve/PI +ve), while necrosis was not observed; however, when the treatment were given with the extract for 6 h, there was an increase in the early, late apoptosis and necrosis as well. The highest early apoptotic events in this duration group was at the concentration 200 followed by 300 and 100 µg/mL where 4.2%, 0.96% and 0.46% relative to viable cells of control, respectively. Similarly, the late apoptotic events were higher in 200 than 300 and 100 µg/mL with 4.3%, 3.9% and 0.46%. The highest necrotic events were 4.2% observed at 300 µg/mL.
Fig. 3.
Apoptosis assay by flow cytometry of MCF-7 cells treated A. obesum ethanolic seeds extract for indicated cencentrations and durations. Representative figures showing population of viable (annexin V− PI−), early apoptotic (annexin V+ PI−), late apoptotic (annexin V+ PI + ) and necrotic (annexin V− PI+) cells.
In 12 h treatment group, early apoptosis and late apoptosis relative to viable cells of control increased in both 200 and 300 µg/mL groups; they attained levels of 28.4% and 37.5% for early and 16.5% and 35% for late apoptosis, respectively. The 100 µg/mL concentration also induced 9.31% and 3.99% of early and late apoptosis, respectively. The effect of the extract was relatively decreased after 24 h compared to treatment of 12 h, but the trend was somewhat similar to 6 h treatment group. The maximum early apoptosis in this duration groups was 3.6%, which found in 100 µg/mL concentration groups, while it was 2% and 1.3% in 200 and 300 µg/mL concentrations respectively. Late apoptosis was 4.22%, 7% and 8.2% for 100, 200 and 300 µg/mL concentrations.
Fig. 4 illustrates summarized results of apoptosis analysis of MCF-7 cells induced by A. obesum ethanolic seeds extract at concentrations 100, 200 and 300 µg/mL for 6, 12 and 24 h respectively, it is clear that 12 h treatment groups exerted the maximum effect in induction of apoptosis (more than 70% cells were found to be in early and late apoptosis stages at the highest concentration of 300 µg/mL).
Fig. 4.
Showing the percentage of apoptotic cells relative to the viable cells of control in MCF-7 cells induced by A. obesum ethanolic seeds extract at concentrations 100, 200 and 300 µg/mL for 6, 12 and 24 h, respectively.
3.3. Genotoxicic effect of A. obesum ethanolic seeds extracts
Fig. 5(A–C) shows representive images of comets obtained after the treatment of MCF-7 cells with ethanolic seeds extract of A. obesum after 12 h with three concentrations (100, 200 and 300 µg/mL). Results of genotoxic effect of the extract after three durations and concentrations were presented in Table 1 and Fig. 5(D). The total damage score (TDS) of 6 h treatment group were 253, 204 and 217 for 100, 200 and 300 µg/mL treatment, respectively. No signifcant difference was observed between the control and 6 h treatment groups.
Fig. 5.
Representative images of comets showing the genotoxic effect of A. obesum ethanolic seeds extract on MCF-7 cells for 12 h treatment analysed by comet assay. (A) 100 µg/mL (B) 200 µg/mL (C) 300 µg/mL. Numbers at or near each comet are the visual scoring of DNA damage according to the comet class. (D) Total damage score of comet assay by A. obesum ethanolic seeds extract on MCF-7 cells for three concentrations and durations (*p < 0.01, **p < 0.05).
Table 1.
Genotoxic effect of A. obesum ethanolic seeds extracts on MCF-7 cells using comet assay.
| Group | Concentration | Cells per damage class |
Total count | TDS† | ||||
|---|---|---|---|---|---|---|---|---|
| 0 | 1 | 2 | 3 | 4 | ||||
| 6 h | Control | 131 | 118 | 27 | 23 | 1 | 300 | 245 |
| 100 µg/mL | 151 | 84 | 31 | 29 | 5 | 300 | 253 | |
| 200 µg/mL | 182 | 53 | 46 | 17 | 2 | 300 | 204 | |
| 300 µg/mL | 159 | 90 | 33 | 11 | 7 | 300 | 217 | |
| 12 h | Control | 186 | 15 | 13 | 7 | 0 | 300 | 221 |
| 100 µg/mL | 74 | 34 | 47 | 129 | 16 | 300 | 579** | |
| 200 µg/mL | 59 | 29 | 61 | 141 | 10 | 300 | 614** | |
| 300 µg/mL | 47 | 50 | 54 | 137 | 12 | 300 | 617** | |
| 24 h | Control | 175 | 25 | 17 | 13 | 5 | 300 | 235 |
| 100 µg/mL | 83 | 87 | 84 | 32 | 14 | 300 | 407* | |
| 200 µg/mL | 87 | 88 | 80 | 33 | 12 | 300 | 395* | |
| 300 µg/mL | 96 | 79 | 75 | 39 | 11 | 300 | 390* | |
TDS: total damage score.
p < 0.05.
p < 0.01.
The maximum effect of extract in terms of DNA damage was recorded after 12 h teatment at 200 and 300 µg/mL concentrations. The calculated TDS for these concentrations were 614 and 617, respectively and it was 579 for 100 µg/mL treatment. The increase in TDS in the treatment group of 12 h was highly significant (p < 0.01) and concentration dependent; however, in the treatment groups of 24 h the extent of DNA damage was decreased when compared to 12 h treatment groups, but still significant (p < 0.05), where the TDS were 407, 395 and 390 for 100, 200 and 300 µg/mL treatment concentrations respectively.
4. Discussion
The present study analysed the cytotoxic activity of a total of four extracts from different vegetative and reporductiove parts of A. obesum on MCF-7 cells. The ethanolic seed extract exhibited strongest cytotoxic activity, while ethanolic fruits extract shown moderate cytotoxicity and the ethanolic leaves extract and aqueous seeds extract have lowest effect. Results of MTT assay indicated that the ethanolic seed extract had the most remarkable cytotoxicity with an IC50 value of ∼337 µg/mL. This indicated that seed possesses very potent cytotoxic activity like other parts of the plant, such as ground stem (Hoffmann and Cole, 1977) and flowers (Bungihan and Matias, 2013). Although there is difference in the IC50 value when compared to the other studies, these differences are related to the method of preparation of extract, duration of treatment, type of cell line used and part of plant being tested. For example, Cepleanu et al. (1994) used dichloromethane (DCM) extract of the plant for 5 days, further they used different cell lines for example two human colon carcinoma cell lines, Col 15 and SW 480. On the other hand, Almehdar et al. (2012) found that IC50 of A. obesum methanolic extract on MCF-7 cells was 11.6 µg/mL. However, methanolic extract of the whole arial part was used and the duration of treatment was 48 h while duration in the present study was 24 h. Cytotoxic activity in several studies concerned on the induction of cell death in cancerous cell lines using MTT cytotoxicity assay has been documented (Venkatadri et al., 2017, Aghapour et al., 2018). The cytotoxicity of seeds extract might be attributed to the occurrence of the high content of cardiac glycosides present in A. obesum (Versiani et al., 2014, Ahmed et al., 2017). As anticancer agents, cardiac glycosides triggered different cell death mechanisms including the intrinsic or the extrinsic pathway of apoptosis (Schneider et al., 2018, Juncker et al., 2011). Furthermore, cardiac glycosides induced autophagic cell death was described in breast, ovarian, colorectal, and lung cancer cells (Farah et al., 2016, Hsu et al., 2016, Kang et al., 2016, Wang et al., 2012). More recently, the ability of cardiac glycosides to trigger immunogenic cell death were reported (Diederich et al., 2017).
In order to confirm that A. obesum induces apoptosis as the main mode of cell death in this study, annexin V apoptosis assay by flow cytometry was conducted which is one of the standard method for apoptosis analysis. Results of this assay showed high incidence of early and late apoptosis specially among 12 h treatment group at highest concentration of 300 µg/mL followed by the lower concerttion used in the experiment. The results of this test also showed low level of cell necrosis, which can be explained by the fact that the in vitro apoptosis finally leads to permeabilization of the plasma membrane, but this event was not found the in vivo since the apoptotic cells are digested by the macrophages or by the surrounding cells before their plasma membrane disrupted (Proskuryakov et al., 2003). It also suggest major role of cardiac glycosides in inducing apoptosis which is supposed to be one of the major constituents contained in the extract. The effect of extract was reduced after 24 h treatment; this may be due to metabolic inactivation of the active component(s), or due to the repair/recovery process of the cells.
Under the flow cytometric evaluation, it was noted that treatment of MCF-7 cells increased early and late apoptotic cells. This was supported by translocation of PS to outer surface of PM (Fadok et al., 1998) under microscopy, the DNA damage (Nagata, 2000) induced in comet assay also clearly suggest that the seed extract has therapeutic value against cancer cells. The anti-cancer potential of Carissa spinarum (Sehar et al., 2011, Rao et al., 2005), Wrightia tomentosa (Chakravarti et al., 2012) and several other members of Apocynaceae have also previously been documented (Acebedo et al., 2014).
The fundamental reason to conduct genotoxicity studies is to evaluate a compound to be a potential genotoxin. Genotoxic anticancer agents induce DNA damage, which, if severe enough, will lead to apoptosis. The genotoxic chemotherapeutic drugs affect both normal and cancerous cells. Rapidly dividing cells, such as cancer cells, are particulary sensitive to these agents (Havelka et al., 2007); however, there are three primary effects that genotoxic substances can have on organisms by affecting their genetic informatio i.e. the genotoxins can be carcinogens, mutagens or teratogens (Shah, 2012). In this study, genotoxicity was investigated through single cell gel electrophresis. The results showed the ability of A. obesum extract to induce DNA damge at single cell level. Studies on A. obesum genotoxicity has been reported before on MCF-7 cells with silver nano particles obtained from A. obesum leaves extract. The study demonstrated the ability of nanoparticles to increase the level of ROS (reactive oxygen species) that leads to the DNA damage (Farah et al., 2016). The comet assay data showed the maximum DNA damage was related to the duration of exposure rather than to the concentration, particulary 12 h treatment groups which have the highest effect (TDS is more than double of the control group), compared with the effect of 24 h treatment groups where the TDS declines but still higher than the control level. These results resemble the data obtained from flow cytometry apoptosis analysis. Therefore, the posibility of DNA damage as secondary to cytotoxic effect was suggested (Vock et al., 1999). Other explantion to these damage may be attributed to cardiac glycosides might be present in the extract (Newman et al., 2006). Agents that induce DNA damage and activate programmed cell death in cancer cells could be valuable anti-cancer therapeutics (Kumar et al., 2008).
The genotoxic effect of some members of apocynaceae family have been reported. For example, C. roseus is the source of well-known Vinca alkaloids anticancer drugs, vinblastine and vincristine (van Der Heijden et al., 2004), both drugs have genotoxic effects (Tiburi et al., 2002). Several studies have demonstrated presence of bioactive compounds from the family apocynaceae, but detailed evaluation of extracts from A. obesum has not been undertaken so far. Hence, the cytotoxic potential of A. obesum against MCF-7 cells is likely candidate for the development as chemotherapeutic drugs. The results obtained through our studies demonstrated that the alcoholic seed extract of the A. obesum is genotoxic and cytotoxic at the concentrations used in this study after 12 h treatment. Further studies to investigate the anticancer effects of isolated compounds of this plant species should provide a better understanding of the mechanisms described herein.
Acknowledgments
The authors would like to extend their sincere appreciation to the Deanship of Scientific Research at King Saud University for its funding of the research through the research group project #RG-1438-015.
Footnotes
Peer review under responsibility of King Saud University.
References
- Acebedo A.R., Amor E.C., Jacinto S.D. Apoptosis-inducing activity of HPLC fraction from Voacanga globosa (Blanco) Merr. on the human colon carcinoma cell. Asian Pac. J. Cancer Prev. 2014;15(2):617–622. doi: 10.7314/apjcp.2014.15.2.617. [DOI] [PubMed] [Google Scholar]
- Aghapour F., Moghadamnia A.A., Nicolini A., Kani S.M., Barari L., Morakabati P., Kazemi S. Quercetin conjugated with silica nanoparticles inhibits tumor growth in MCF-7 breast cancer cell lines. Biochem. Biophys. Res. Comm. 2018;500(4):860–865. doi: 10.1016/j.bbrc.2018.04.174. [DOI] [PubMed] [Google Scholar]
- Ahmed S.K., Versiani M.A., Ikram A., Sattar S.A., Faizi S. Cytotoxic cardiac glycosides from the fruit (pods) of Adenium obesum (Forssk.) Roem. & Schult. Nat. Prod. Res. 2017;31(10):1205–1208. doi: 10.1080/14786419.2016.1226826. [DOI] [PubMed] [Google Scholar]
- Almehdar H., Abdallah H.M., Osman A.M., Abdel-Sattar E.A. In vitro cytotoxic screening of selected Saudi medicinal plants. J. Nat. Med. 2012;66(2):406–412. doi: 10.1007/s11418-011-0589-8. [DOI] [PubMed] [Google Scholar]
- Ateeq B., Farah M.A., Ahmad W. Detection of DNA damage by alkaline single cell gel electrophoresis in 2,4-dichlorophenoxyacetic-acid- and butachlor-exposed erythrocytes of Clarias batrachus. Ecotoxicol. Environ. Saf. 2005;62(3):348–354. doi: 10.1016/j.ecoenv.2004.12.011. [DOI] [PubMed] [Google Scholar]
- Azqueta A., Lorenzo Y., Collins A.R. In vitro comet assay for DNA repair: a warning concerning application to cultured cells. Mutagenesis. 2009;24:379–381. doi: 10.1093/mutage/gep009. [DOI] [PubMed] [Google Scholar]
- Baker D.D., Chu M., Oza U., Rajgarhia V. The value of natural products to future pharmaceutical discovery. Nat. Prod. Rep. 2007;24(6):1225–4124. doi: 10.1039/b602241n. [DOI] [PubMed] [Google Scholar]
- Bray F., Ferlay J., Soerjomataram I., Siegel R.L., Torre L.A., Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA: Cancer J. Clin. 2018;68(6):394–424. doi: 10.3322/caac.21492. [DOI] [PubMed] [Google Scholar]
- Bungihan M.E., Matias C.A. Determination of the antioxidant, phytochemical and antibacterial profiles of flowers from selected ornamental plants in Nueva Vizcaya, Philippines. J. Agric. Sci. Technol. 2013;3:833–841. [Google Scholar]
- Cepleanu F., Hamburger M.O., Sordat B., Msonthi J.D., Gupta M.P., Saadou M., Hostettmann K. Screening of medical plants of tropics for molluscicidal, larvicidal, fungicidal and cytotoxic activities and brine shrimp toxicity. Pharma. Biol. 1994;32(3):294–307. [Google Scholar]
- Chakravarti B., Maurya R., Siddiqui J.A., Bid H.K., Rajendran S.M., Yadav P.P., Konwar R. In vitro anti-breast cancer activity of ethanolic extract of Wrightia tomentosa: role of pro-apoptotic effects of oleanolic acid and urosolic acid. J. Ethnopharmacol. 2012;142(1):72–79. doi: 10.1016/j.jep.2012.04.015. [DOI] [PubMed] [Google Scholar]
- Collins A.R. The comet assay for DNA damage and repair principles, applications, and limitations. Mol. Biotechnol. 2004;26:249–260. doi: 10.1385/MB:26:3:249. [DOI] [PubMed] [Google Scholar]
- da Rocha A., Lopes R., Schwartsmann G. Natural products in anticancer therapy. Curr. Opin. Pharmacol. 2001;1:364–369. doi: 10.1016/s1471-4892(01)00063-7. [DOI] [PubMed] [Google Scholar]
- De Flora S., Izzotti A., D’Agostini F., Balansky R.M., Noonam D., Albini A. Multiple points of intervention in the prevention of cancer and other mutation-induced diseases. Mut. Res. 2001;480–481:9–22. doi: 10.1016/s0027-5107(01)00165-8. [DOI] [PubMed] [Google Scholar]
- Diederich M., Muller F., Cerella C. Cardiac glycosides: From molecular targets to immunogenic cell death. Biochem. Pharmacol. 2017;125:1–11. doi: 10.1016/j.bcp.2016.08.017. [DOI] [PubMed] [Google Scholar]
- Fadok V.A., Bratton D.L., Frasch S.C., Warner M.L., Henson P.M. The role of phosphatidylserine in recognition of apoptotic cells by phagocytes. Cell Death Differ. 1998;5(7):551–562. doi: 10.1038/sj.cdd.4400404. [DOI] [PubMed] [Google Scholar]
- Farah M.A., Ali M.A., Chen S.M., Li Y., Al-Hemaid F.M., Abou-Tarboush F.M., Al-Anazi K.M., Lee J. Silver nanoparticles synthesized from Adenium obesum leaf extract induced DNA damage, apoptosis and autophagy via generation of reactive oxygen species. Colloids. Surf. B. Biointerf. 2016;141:158–169. doi: 10.1016/j.colsurfb.2016.01.027. [DOI] [PubMed] [Google Scholar]
- Garcia-Carbonero R., Supko J.G. Current perspectives on the clinical experience, pharmacology, and continued development of the camptothecins. Clin. Cancer Res. 2002;8(3):641–661. [PubMed] [Google Scholar]
- Goodman J., Walsh V. Cambridge University Press; 2001. The Story of Taxol: Nature and Politics in the Pursuit of an Anti-cancer Drug; p. 17. [Google Scholar]
- Gurib-Fakim A. Medicinal plants: traditions of yesterday and drugs of tomorrow. Mol. Asp. Med. 2006;27(1):1–93. doi: 10.1016/j.mam.2005.07.008. [DOI] [PubMed] [Google Scholar]
- Hainsworth J.D., Greco F.A. Etoposide: twenty years later. Ann. Oncol. 1995;6(4):325–341. doi: 10.1093/oxfordjournals.annonc.a059180. [DOI] [PubMed] [Google Scholar]
- Havelka A.M., Berndtsson M., Olofsson M.H., Shoshan M.C., Linder S. Mechanisms of action of DNA-damaging anticancer drugs in treatment of carcinomas: Is acute apoptosis an “off-target” effect? Mini Rev. Med. Chem. 2007;7(10):1035–1039. doi: 10.2174/138955707782110196. [DOI] [PubMed] [Google Scholar]
- Hoffmann J.J., Cole J.R. Phytochemical investigation of Adenium obesum Forskal (Apocynaceae): isolation and identification of cytotoxic agents. J. Pharm. Sci. 1977;66(9):1336–1338. doi: 10.1002/jps.2600660935. [DOI] [PubMed] [Google Scholar]
- Hossain M.A., Akhtar M.S., Said S., Al-Abri T.A. Two new flavonoids from Adenium obesum grown in Oman. J. KSU Sci. 2017;29(1):62–69. [Google Scholar]
- Hsu I.L., Chou C.Y., Wu Y.Y., Wu J.E., Liang C.H., Tsai Y.T., Hong T.M. Targeting FXYD2 by cardiac glycosides potently blocks tumor growth in ovarian clear cell carcinoma. Oncotarget. 2016;7(39):62925. doi: 10.18632/oncotarget.7497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jordan M.A., Thrower D., Wilson L. Mechanism of inhibition of cell proliferation by Vinca alkaloids. Cancer Res. 1991;51(8):2212–2222. [PubMed] [Google Scholar]
- Juncker T., Cerella C., Teiten M.H., Morceau F., Schumacher M., Ghelfi J., Gaascht F., Schnekenburger M., Henry E., Dicato M., Diederich M. UNBS1450, a steroid cardiac glycoside inducing apoptotic cell death in human leukemia cells. Biochem. Pharmacol. 2011;81(1):13–23. doi: 10.1016/j.bcp.2010.08.025. [DOI] [PubMed] [Google Scholar]
- Kang M.A., Kim M.S., Kim W., Um J.H., Shin Y.J., Song J.Y., Jeong J.H. Lanatoside C suppressed colorectal cancer cell growth by inducing mitochondrial dysfunction and increased radiation sensitivity by impairing DNA damage repair. Oncotarget. 2016;7(5):6074. doi: 10.18632/oncotarget.6832. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kiyohara H., Ichino C., Kawamura Y., Nagai T., Sato N., Yamada H., Salama M.M., Abdel-Sattar E. In vitro anti-influenza virus activity of a cardiotonic glycoside from Adenium obesum (Forssk) Phytomedicine. 2012;19(2):111–114. doi: 10.1016/j.phymed.2011.07.004. [DOI] [PubMed] [Google Scholar]
- Kumar A., Malik F., Bhushan S., Sethi V.K., Shahi A.K., Kaur J., Taneja S.C., Qazi G.N., Singh J. An essential oil and its major constituent isointermedeol induce apoptosis by increased expression of mitochondrial cytochrome c and apical death receptors in human leukaemia HL-60 cells. Chem. Biol. Interact. 2008;171(3):332–347. doi: 10.1016/j.cbi.2007.10.003. [DOI] [PubMed] [Google Scholar]
- Kumar V., Abbas A.K., Fausto N. seventh ed. Elsevier Saunders; Philadelphia: 2005. “Neoplasia” in Robbins and Cotran Pathologic Basis of Disease; pp. 1119–1155. [Google Scholar]
- Miadokova E., Nadova S., Vickova V., Duhova V., Kopaskova M., Cipak L., Rauko P., Mucaji P., Grancai D. Antigenotoxic effect of extract from Cynara cardunculus. Phytother. Res. 2008;22:77–81. doi: 10.1002/ptr.2268. [DOI] [PubMed] [Google Scholar]
- Mosmann T. Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J. Immunol. Methods. 1983;65:55–63. doi: 10.1016/0022-1759(83)90303-4. [DOI] [PubMed] [Google Scholar]
- Mouza K.G., Hossain M.A. Determination of total phenolics, flavonoids and antioxidant activity of root crude extracts of Adenium obesum traditionally used for the treatment of bone dislocations and rheumatism. Asian Pac. J. Trop. Dis. 2015;5(Suppl. 1):S155–S158. [Google Scholar]
- Nagata S. Apoptotic DNA fragmentation. Exp. Cell Res. 2000;256(1):12–18. doi: 10.1006/excr.2000.4834. [DOI] [PubMed] [Google Scholar]
- Newman R.A., Yang P., Hittelman W.N., Lu T., Ho D.H., Ni D., Chan D., Vijjeswarapu M., Cartwright C., Dixon S., Felix E., Addington C. Oleandrin-mediated oxidative stress in human melanoma cells. J. Exp. Ther. Oncol. 2006;5(3):167–181. [PubMed] [Google Scholar]
- Paul D., Biswas K., Sinha S.N. Biological activities of Adenium obesum (Forssk.) Roem. & Schult.: a concise review. Malaya J. Biosci. 2016;2(4):214–221. [Google Scholar]
- Proskuryakov S.Y., Konoplyannikov A.G., Gabai V.L. Necrosis: a specific form of programmed cell death? Exp. Cell Res. 2003;283(1):1–16. doi: 10.1016/s0014-4827(02)00027-7. [DOI] [PubMed] [Google Scholar]
- Rao J.R., Sampath Kumar U., Venkat Reddy S., Tiwari A.K., Madhusudana Rao J. Antioxidants and a new germacrane sesquiterpene from Carissa spinarum. Nat. Prod. Res. 2005;19(8):763–769. doi: 10.1080/14786410512331330648. [DOI] [PubMed] [Google Scholar]
- Razis E.D., Fountzilas G. Paclitaxel: epirubicin in metastatic breast cancer-a review. Ann. Oncol. 2001;12(5):593–598. doi: 10.1023/a:1011108807105. [DOI] [PubMed] [Google Scholar]
- Schneider N.Z., Persich L., Rocha S.C., Ramos A.P., Cortes V.F., Silva I.T., Barbosa L.A. Cytotoxic and cytostatic effects of digitoxigenin monodigitoxoside (DGX) in human lung cancer cells and its link to Na, K-ATPase. Biomed. Pharmacol. 2018;97:684–696. doi: 10.1016/j.biopha.2017.10.128. [DOI] [PubMed] [Google Scholar]
- Sehar I., Pal H.C., Shukla S., Bhushan S., Hamid A., Gupta B.D., Saxena A.K. Cytotoxic evaluation and induction of mitochondria-mediated apoptosis in human leukaemia HL-60 cells by Carissa spinarum stem isolate. J. Pharm. Pharmacol. 2011;63(8):1078–1090. doi: 10.1111/j.2042-7158.2011.01310.x. [DOI] [PubMed] [Google Scholar]
- Shah S.U. Importance of genotoxicity & S2A guidelines for genotoxicity testing for pharmaceuticals. IOSR J. Pharm. Biol. Sci. 2012;1(2):43–54. [Google Scholar]
- Shukla Y., Singh M. Cancer preventive properties of ginger: a brief review. Food Chem. Toxicol. 2007;45:683–690. doi: 10.1016/j.fct.2006.11.002. [DOI] [PubMed] [Google Scholar]
- Singh N.P., McCoy M.T., Tice R.R., Schneider E.L. A simple technique for quantization of low levels of DNA damage in individual cells. Exp. Cell Res. 1988;175:184–191. doi: 10.1016/0014-4827(88)90265-0. [DOI] [PubMed] [Google Scholar]
- Stopeck, A.T., Thompson, P.A., 2012. Breast cancer treatment and anagement. (WebMD Health Professional Network) Retrieved september 29, 2012, from medscape: <http://emedicine.medscape.com/article/1947145-treatment>.
- Tiburi M., Reguly M.L., Schwartsmann G., Cunha K.S., Lehmann M., Rodrigues H. Comparative genotoxic effect of vincristine, vinblastine, and vinorelbine in somatic cells of Drosophila melanogaster. Mutat. Res. 2002;519(1–2):141–149. doi: 10.1016/s1383-5718(02)00136-5. [DOI] [PubMed] [Google Scholar]
- van Der Heijden R., Jacobs D.I., Snoeijer W., Hallard D., Verpoorte R. The Catharanthus alkaloids: pharmacognosy and biotechnology. Curr. Med. Chem. 2004;11(5):607–628. doi: 10.2174/0929867043455846. [DOI] [PubMed] [Google Scholar]
- Venkatadri R., Muni T., Iyer A.V., Yakisich J.S., Azad N. Role of apoptosis-related miRNAs in resveratrol-induced breast cancer cell death. Cell Death Dis. 2017;7(2):e2104. doi: 10.1038/cddis.2016.6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vermani K., Garg S. Herbal medicines for sexually transmitted diseases and AIDS. J. Ethnopharmacol. 2002;80:49–66. doi: 10.1016/s0378-8741(02)00009-0. [DOI] [PubMed] [Google Scholar]
- Versiani M.A., Ahmed S.K., Ikram A., Ali S.T., Yasmeen K., Faizi S. Chemical constituents and biological activities of Adenium obesum (Forsk.) Roem.etSchult. Chem. Biodivers. 2014;11(2):171–180. doi: 10.1002/cbdv.201200254. [DOI] [PubMed] [Google Scholar]
- Vock E.H., Lutz W.K., Ilinskaya O., Vamvakas S. Discrimination between genotoxicity and cytotoxicity for the induction of DNA double-strand breaks in cells treated with aldehydes and diepoxides. Mutat. Res. 1999;441(1):85–93. doi: 10.1016/s1383-5718(99)00038-8. [DOI] [PubMed] [Google Scholar]
- Wang Y., Qiu Q., Shen J.J., Li D.D., Jiang X.J., Si S.Y., Shao R.G., Wang Z. Cardiac glycosides induce autophagy in human non-small cell lung cancer cells through regulation of dual signaling pathways. Int. J. Biochem. Cell Biol. 2012;44(11):1813–1824. doi: 10.1016/j.biocel.2012.06.028. [DOI] [PubMed] [Google Scholar]
- Webb M.R., Ebeler S.E. Comparative analysis of topoisomerase IB inhibition and DNA intercalation by flavonoids and similar compounds: structural determinates of activity. Biochem. J. 2004;384:527–541. doi: 10.1042/BJ20040474. [DOI] [PMC free article] [PubMed] [Google Scholar]