Abstract
We examined the association between the angiotensin I-converting enzyme (ACE) I/D gene polymorphism and isometric handgrip (IHG) training on cardiovascular and muscular responses among normotensive males. Thirty (II = 10, ID = 10, and DD = 10) normotensive untrained males underwent IHG training at 30% of their maximal voluntary contraction 3 days per week for 8 weeks. Cardiovascular and muscular variables were measured before IHG, after a session of IHG and after 8 weeks of IHG. No significant interaction effect was found between ACE I/D genotype and IHG training session on all dependent variables (all p > 0.05). There was a significant main effect of IHG training session on systolic blood pressure (SBP) (p = 0.002), mean arterial pressure (MAP) (p = 0.015) and handgrip strength (HGS) (p = 0.001) scores, while no difference in diastolic blood pressure (DBP), pulse pressure, or heart rate scores was found. A greater improvement in cardiovascular parameters following 8 weeks of IHG training was observed in participants with the D allele than the I allele (SBP reduction: ID+DD genotype group (-5.53 ± 6.2 mmHg) vs. II genotype group (-1.52 ± 5.3 mmHg)); MAP reduction: ID + DD genotype group (-2.80 ± 4.5 mmHg) vs. II genotype group (-1.45 ± 3.5 mmHg). Eight weeks of IHG training improved cardiovascular and muscular performances of normotensive men. Reduction in SBP and MAP scores in D allele carriers compared to I allele carriers indicates that the ACE I/D polymorphism may have an influence on IHG training adaptation in a normotensive population.
Keywords: ACE I/D gene polymorphism, IHG training, Normotensive, Blood pressure management
INTRODUCTION
Resistance exercise training, which has not been previously recommended for blood pressure (BP) management in hypertensive patients [1], has been shown to lower resting BP in normotensive and hypertensive individuals [2-5]. Reductions of 3 to 4 mmHg in resting systolic and diastolic BP were observed following four weeks of resistance exercise training [5]. Meanwhile, in another meta-analysis study conducted by Cornelissen and Smart [6], the largest reductions in resting BP were reported following the isometric resistance exercise training (systolic: -10.9 ± 2.86 mmHg), diastolic: -6.2 ± 3.34 mmHg) compared to after endurance (systolic: -3.5 ± 6.01 mmHg, diastolic: -3.7 ± 3.92 mmHg) and dynamic resistance exercise training (systolic: -1.8 ± 4.85 mmHg, diastolic: -2.5 ± 3.29 mmHg). It has been suggested that an isometric exercise training protocol consisting of four sets of 2-minute handgrip [7, 8] or leg contractions [9] at 30–50% of maximal voluntary contraction (MVC) [2, 10] with 1–4 minutes of passive rest between each contraction [2, 7] performed 3–5 times per week for 4–10 weeks [11, 12] is more effective at lowering resting BP than endurance and dynamic resistance exercise training.
Although the benefit of isometric exercise training for the management of hypertension has been well documented [13], it has remained unclear how factors [14] that can influence BP, such as sex and genetics, may influence the efficiency of this isometric exercise programme. Indeed, several studies using twins as subjects reported that BP is controlled by genetic factors [15, 16]. Given the fact that BP has a genetic basis, research efforts have been directed towards identifying the candidate genes involved in BP regulation [17, 18]. Among the proposed candidate genes, the angiotensin I-converting enzyme (ACE) gene has attracted much attention due to its role in the renin-angiotensin system (RAS), which is the body’s primary physiological system that regulates BP [19-21].
Within the ACE gene, the ACE I/D gene polymorphism showed a strong link with the level of ACE [22] in the RAS and accounted for 47% of the total phenotypic variance of ACE [23]. Rigat et al. [23] observed that ACE levels were higher among individuals with the DD genotype, followed by those with the ID and II genotypes of the ACE I/D gene polymorphism. Wong et al. (2012) reported that individuals with the II genotype had lower resting BP than those with the ID and DD genotypes. It has been suggested that individuals with two copies of the I allele might have lower resting BP compared to those with two copies of the D allele as the lower level of ACE decreased the production of ANG II, a potent vasodepressor and aldosterone stimulating peptide, besides activating bradykinin, a potent vasodilator that leads to a drop in BP [24]. Hence, given that the ACE I/D gene polymorphism has an important role in BP regulation, the BP response to exercise training may vary among individuals with different genotypes of ACE I/D gene polymorphism.
The influences of the ACE I/D gene polymorphism on BP in response to exercise training have been investigated previously, but the results have been inconsistent [25-31]. It has been reported that the ACE I/D gene polymorphism did not influence BP response to endurance [25, 27, 29] and dynamic resistance [31] exercise training. On the other hand, Hagberg et al. [26] reported a greater drop in resting BP among hypertensive men with the II and ID genotypes compared to those with the DD genotype after 9 months of endurance exercise training at 75–85% of maximal oxygen consumption. Zhang et al. [28] reported similar results for the impact of the ACE I/D gene polymorphism on BP response to 10 weeks of exercise therapy on a bicycle ergometer among 64 Japanese participants (16 males, 48 females) with mild to moderate essential hypertension. In contrast to these results, Kim [30] observed that adult women with the DD genotype had greater reduction in BP (diastolic) than those with the II and ID genotypes following 12 weeks of combined aerobic and resistance exercise training. The reasons for these inconsistent results are unclear, but differences in sample sizes, training protocols and ethnic background of the participants for eliciting substantial changes in resting BP may be involved. There is also a possibility that the effects of ACE I/D gene polymorphism on BP in response to exercise training may vary depending on the ethnic origin, which could explain the different findings in Korean and US populations as investigated by Kim [30] and Hagberg et al. [26], respectively. Ethnic variation has been demonstrated to exist in the distribution of ACE I/D gene polymorphism, with the highest frequency of the I allele being reported in the Black (Australian Aboriginal) population (0.97) [32], while the D allele was reported to be highest among the Caucasian population (0.77) [22]. Based on the previous reports that the distribution of ACE I/D gene polymorphism varies across ethnic groups and the disparity in findings between different ethnic groups, population-specific/ethnic-specific research is suggested to control this potential bias.
Based on the above-mentioned findings, there is a possibility that the ACE I/D gene polymorphism might also influence the BP response to isometric exercise training. More specifically, it raises the question of which if any ACE genotype group would be likely to benefit from this exercise programme. To our knowledge, no study has investigated the effect of isometric exercise training on BP response among individuals with different genotypes of the ACE I/D gene polymorphism. Thus, this investigation is important for determining whether ACE I/D gene polymorphism influences BP in response to exercise training. The results of this investigation would help to identify individuals who could reap potential clinical benefits from this type of exercise training programme.
MATERIALS AND METHODS
Study Design
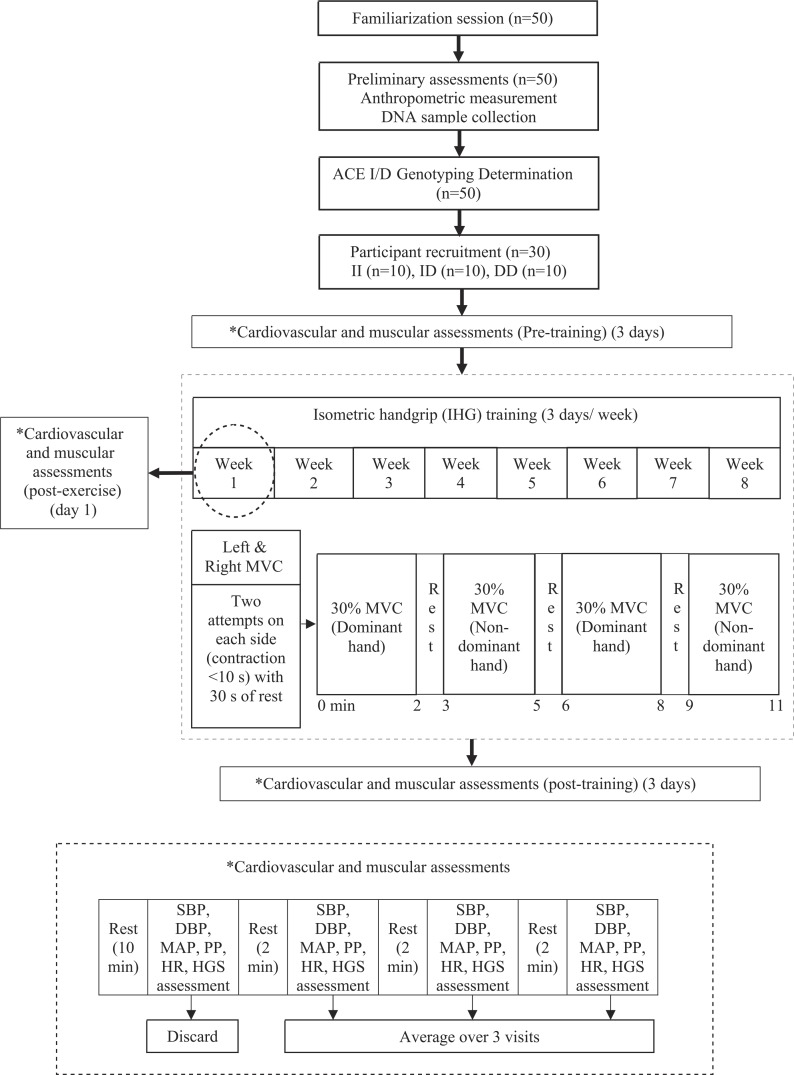
A single-blind, repeated measures study design was used in the present study. All participants underwent identical cardiovascular and muscular assessments before training (pre-training), after the initial acute exercise session (post-exercise), and after 8 weeks of training (post-training). They performed isometric handgrip (IHG) exercise 3 days per week for 8 weeks. All assessments and IHG training were performed under the supervision of the researchers and were conducted in a quiet, temperature-controlled room (20–25ºC). The study protocol was approved by the Human Research Ethics Committee in Universiti Sains Malaysia in accordance with the ethical standards of the Helsinki Declaration. A flowchart of the study design is presented in Figure 1.
FIG. 1.
Flow chart of study design.
Participants
ifty healthy, normotensive, untrained males aged 30.3 ± 5.1 years, reportedly of Malay ancestry within the last three generations from Malaysia, were initially recruited to the study. After obtaining written consent from the participants, they were asked to complete a participant’s information detail form and the Physical Activity Readiness Questionnaire (PAR-Q) to record their health and physical activity status. The details obtained from the PAR-Q were used to determine whether the participant was sedentary (determined as 2 or fewer days a week of recreational exercise for < 30 minutes a day for the preceding 3 months [33]). These sedentary individuals were then screened for ACE I/D gene polymorphism (rs4646994). Thirty of these initial participants (10 each with the II, ID, and DD genotypes of the ACE I/D gene polymorphism) were then selected for IHG training.
The number of participants for this study was based on the results obtained by Hagberg et al. [26] and the sample size was calculated using the Power and Sample Size Calculation version 3.1.2 software [34] [Calculated sample size = 27 participants;Research sample size = 27 participants + (27*10% (expected drop out)) = 30 participants]. The statistical power of the study was set at 0.80 with a 95% confidence interval and an effect size of 0.25. The participants were briefed about the testing protocol and familiarized with the instruments and procedures involved in the isometric handgrip test. After they had signed and completed the consent form, they were interviewed to obtain personal information, including sex, age, ethnicity, and health status. Body height was measured using a portable stadiometer (Seca 213, Seca Corporation, Chino, California, USA). Body mass, body mass index, and body fat were measured using an Omron KARADA Scan Body Composition & Scale (HBF-362, Omron Corporation, Kyoto, Japan).
ACE I/D Genotyping
A deoxyribonucleic acid (DNA) sample from each participant was collected using a buccal swab with a sterile swab applicator (Classic Swabs by Copan Flock Technologies, Brescia, Italy). Genomic DNA was isolated from the swab samples using the GeneAll Exgene Cell SV kit following the manufacturer’s protocol (GeneAll Biotechnology Co. Ltd., Seoul, South Korea). Polymerase chain reaction (PCR) was carried out in a final volume of 25 μl consisting of 2.5 μl of 10X standard reaction buffer (25 mm Mg2+, 50 mm Tris-HCl, 50 mm KCl, 0.1 mm EDTA, 1 mm DTT, 0.5 mm PMSF, and 50% glycerol (GeneAll Biotechnology Co. Ltd., Seoul, South Korea), 2.0 μl of dNTP mix (200 μm from each dNTP (dATP, dCTP, dGTP, and dTTP)), 0.8 μm of each primer (forward primer: 5′-CTGGAGACCACTCCCATCCTTTCT-3′: reverse primer: 5′-CTGGAGACCACTCCCATCCTTTCT-3′), 0.5 units of Taq DNA polymerase, 2.5 μl of dimethylsulfoxide, 10.8 μl of sterilize distilled water, and 5 μl of genomic DNA (2–8 ng/μl). The target fragment bearing the ACE I/D gene polymorphism was amplified under the following conditions: 7 minutes at 95 °C followed by 25 cycles of 30 seconds at 95 °C, 30 seconds at 62 °C, and 1 minute at 72 °C, with a final step of 7 minutes at 72 °C. The amplified products were electrophoresed on a 1.5% agarose gel that was pre-stained with ethidium bromide at 70 volts for 1 hour. The presence of 490 base pair (bp) and 190 bp bands indicated the ACE insertion (I) and deletion (D) alleles, respectively. The PCR products for ACE I/D gene polymorphism were confirmed by sequencing (First BASE Laboratories Sdn Bhd, Selangor, Malaysia). The genotyping protocol used in the present study was adapted and modified from the established protocol by Mayne et al. [35].
Isometric Handgrip Training
Before every training session, the left and the right hand MVC values of the participants were assessed to reassess daily MVC of the participants with two attempts on each side (contraction duration < 10 seconds), separated by 30 seconds of passive rest between the attempts. If the variance from the two recordings for left or right hand MVC was < 5%, the highest value was taken as the participant’s MVC for that side. If the recordings for left or right hand MVC differed by > 5%, further attempts were made at 1-minute intervals until a stable maximal value was obtained. All participants were trained using unilateral (one hand), alternate IHG exercise 3 days per week for 8 weeks. During each session, participants performed four trials of 2-minute IHG exercise at 30% of their MVC. They performed the IHG exercise using alternate hands, starting with the dominant hand, with a 1-minute rest period between each trial while sitting with the working arm extended towards the front.
Cardiovascular and Muscular Assessments
Prior to measurement of the study variables, the participants were asked to refrain from performing vigorous exercise and consuming caffeinated beverages within 24 h before the assessments. A handgrip dynamometer (Takei A5401, Takei Scientific Instruments Co. Ltd., Tokyo, Japan) was used to measure the muscular variable, whereas the cardiovascular variables were assessed using a non-invasive automated brachial oscillometer (Omron HEM907XL, Omron Healthcare, Inc., Lake Forest, Illinois, USA). Overall, the cardiovascular and muscular assessments took approximately 30 minutes.
Cardiovascular systolic BP (SBP), diastolic BP (DBP), mean arterial pressure (MAP), pulse pressure (PP), and heart rate (HR) and muscular handgrip strength (HGS) variables were measured on 3 consecutive days at the same time (± 2 h) of the day, immediately prior to commencing training. During each visit, after 10 minutes of seated rest, all variables were measured on the dominant arm hand (self-reported by the participant) in the sitting position four successive times with 2-minute rest intervals. The first of the four measurements of all variables in each visit was discarded (due to the white coat effect), whereas the remaining three measurements were averaged over the three visits to represent the pre-training value [8].
One hour after the initial training session (post-exercise), the cardiovascular and muscular variables were assessed using the procedure described above to examine the acute effects of IHG exercise [36]. Considering the white coat effect following one hour of exercise, the first and the second measurements were discarded, whereas the last two measurements were averaged to represent the post-exercise value [36].
For 3 consecutive days after 8 weeks of training (post-training), the cardiovascular and muscular variables were again assessed using the procedure described above. The measurements of cardiovascular and muscular variables were averaged in the same way as described for the pre-training value to represent the post-training value [8].
Statistical Analysis
The descriptive data are presented as mean ± standard deviation (SD). Differences in pre- and post-exercise/post-training values were calculated as the final (post-exercise/post-training) minus the initial (pre-training) value. Positive and negative results indicated an increase and a decrease with IHG training, respectively. The mean values of all variables at pre-training, post-exercise, and post-training were compared for II, ID, and DD genotype groups via one-way analysis of variance (ANOVA) followed by Bonferroni’s post-hoc test when appropriate. A two-way repeated measures ANOVA test was used to examine whether there was an interaction effect between the ACE I/D genotype and IHG training on the cardiovascular and muscular variables (interaction: genotype x training;training effect (within subject);genotype effect (between subjects)). The main effects of training and genotype are presented as estimated marginal mean ± standard error of mean (SEM). All statistical evaluations were performed using IBM SPSS Statistics version 20.0 (Armonk, New York, USA), with the level of significance set at p < 0.050.
RESULTS
Characteristics of Participants
Table 1 shows the physical characteristics of the participants according to ACE I/D genotype. All ACE I/D genotype groups were similar in age, height, body weight, body mass index, and body fat.
TABLE 1.
Physical characteristics of participants according to ACE I/D genotype.
| Variables | II (n=10) | ID (n=10) | DD (n=10) | F value | p value |
|---|---|---|---|---|---|
| Age (years) | 27.8 ± 6.2 | 32.9 ± 3.1 | 30.0 ± 4.5 | 2.834 | 0.071 |
| Height (cm) | 169.0 ± 6.2 | 170.4 ± 9.3 | 169.0 ± 5.4 | 0.133 | 0.876 |
| Body Weight (kg) | 72.6 ± 10.0 | 74.7 ± 24.0 | 72.4 ± 7.4 | 0.066 | 0.961 |
| Body Mass Index (kg/m2) | 25.4 ± 3.1 | 27.1 ± 4.4 | 25.6 ± 3.2 | 0.672 | 0.519 |
| Body Fat (%) | 23.8 ± 3.5 | 26.0 ± 4.7 | 24.1 ± 3.7 | 0.937 | 0.404 |
Data shown as mean ± SD.
Cardiovascular and Muscular Responses in the Whole Samples
Table 2 shows the cardiovascular and muscular responses in the whole samples (regardless of their ACE I/D genotype) following a single session of IHG exercise (post-exercise) and 8 weeks of IHG training. SBP (t(29) = 2.456, p = 0.020) and MAP (t(29) = 2.506, p = 0.018), but not DBP, PP, and HR, were significantly lower following the initial session of IHG exercise than at pre-training. HGS did not increase significantly after the first session of IHG exercise. SBP (t(29) = 3.753, p = 0.001), MAP (t(29) = 3.008, p = 0.004), PP (t(29) = 2.401, p = 0.023), and HR (t(29) = 2.398, p = 0.023), but not DBP, were significantly lower following 8 weeks of IHG training than at pre-training. HGS (t(29) = -3.175, p = 0.004) significantly increased after IHG training.
TABLE 2.
Cardiovascular and muscular responses to a session of IHG exercise and 8 weeks of IHG training.
| Variables | Pre-training | Mid-training | Post-training | Change (Δ) with IHG exercise | Change (Δ) with IHG training | t valuea | p valuea | t valueb | p valueb |
|---|---|---|---|---|---|---|---|---|---|
| SBP (mmHg) | 121.3 ± 9.1 | 118.1 ± 9.4* | 117.1 ± 6.6* | -3.2 ± 7.2 | -4.2 ± 6.1 | 2.456 | 0.020 | 3.753 | 0.001 |
| DBP (mmHg) | 76.2 ± 7.2 | 73.8 ± 8.5 | 74.7 ± 7.9 | -2.4 ± 6.5 | -1.4 ± 4.5 | 2.024 | 0.052 | 1.737 | 0.093 |
| MAP (mmHg) | 91.2 ± 6.9 | 88.5 ± 8.0* | 88.9 ± 6.8* | -2.7 ± 5.8 | -2.4 ± 4.2 | 2.506 | 0.018 | 3.008 | 0.004 |
| PP (mmHg) | 45.1 ± 8.2 | 44.3 ± 8.0 | 42.4 ± 6.8* | -0.9 ± 7.1 | -2.8 ± 6.3 | 0.655 | 0.518 | 2.401 | 0.023 |
| HR (bpm) | 79.4 ± 7.7 | 78.5 ± 9.1 | 76.8 ± 8.9* | -0.9 ± 7.6 | -2.6 ± 5.9 | 0.608 | 0.548 | 2.398 | 0.023 |
| HGS (kg) | 43.5 ± 6.7 | 43.2 ± 6.9 | 46.0 ± 7.5* | -0.3 ± 2.3 | 2.6 ± 4.4 | 0.800 | 0.430 | -3.175 | 0.004 |
Data shown as mean ± SD
Significantly different compared to pre-training value (p < 0.050)
p value for change (Δ) with IHG exercise
p value for change (Δ) with IHG training.
ACE I/D Genotype and Cardiovascular and Muscular Responses
Cardiovascular and muscular variables among ACE genotype groups (II, ID, DD) at pre-training, post-exercise and post-training are shown in Table 3. Two-way repeated measures ANOVA revealed no significant interaction effect between ACE I/D gene polymorphism and IHG training session on all dependent variables. However, a significant main effect of IHG intervention (pre-training, post-exercise and post-training) on SBP (p = 0.002), MAP (p = 0.015) and HGS (p = 0.001) scores was found, while no significant differences were found in DBP, PP, and HR scores across the IHG training sessions.
TABLE 3.
Repeated-Measures 2-way ANOVA on Cardiovascular and muscular responses to a session of IHG exercise and 8 weeks of IHG training according to ACE I/D genotype.
| Repeated-Measures 2-way ANOVA | ||||||||
|---|---|---|---|---|---|---|---|---|
| Variables | Genotype | n | Pre-training | Mid-training | Post-training | Interaction (genotype x training session) | Training effect (within subject) | Genotype effect (between subjects) |
| SBP (mmHg) |
II ID DD |
10 10 10 |
115.3 ± 7.2 125.0 ± 9.3 123.6 ± 8.2 |
113.2 ± 7.5 123.1 ± 8.9 117.9 ± 9.8 |
113.8 ± 6.3 118.7 ± 4.9 118.8 ± 7.6 |
0.198 | 0.002* | 0.038* |
| DBP (mmHg) |
II ID DD |
10 10 10 |
70.1 ± 4.6 80.6 ± 5.9 77.8 ± 6.7 |
67.3 ± 7.0 78.5 ± 5.2 75.6 ± 9.1 |
68.6 ± 6.7 78.8 ± 6.7 76.7 ± 6.9 |
0.996 | 0.098 | 0.001* |
| MAP (mmHg) |
II ID DD |
10 10 10 |
85.2 ± 4.5 95.4 ± 5.8 93.1 ± 6.0 |
82.6 ± 6.4 93.3 ± 4.5 89.7 ± 8.8 |
83.7 ± 5.3 92.1 ± 5.3 90.7 ± 6.7 |
0.839 | 0.015* | 0.001* |
| PP (mmHg) |
II ID DD |
10 10 10 |
45.2 ± 7.3 44.4 ± 9.2 45.8 ± 8.7 |
45.9 ± 7.0 44.6 ± 10.3 42.4 ± 6.6 |
45.2 ± 8.2 39.9 ± 6.4 42.1 ± 5.1 |
0.278 | 0.067 | 0.692 |
| HR (bpm) |
II ID DD |
10 10 10 |
78.6 ± 6.5 79.8 ± 9.5 79.8 ± 7.7 |
77.0 ± 8.7 77.0 ± 10.2 81.7 ± 8.3 |
78.9 ± 8.1 76.1 ± 9.3 75.5 ± 9.8 |
0.140 | 0.164 | 0.923 |
| HGS (kg) |
II ID DD |
10 10 10 |
43.4 ± 4.9 46.3 ± 8.0 40.8 ± 6.3 |
43.3 ± 5.7 46.3 ± 7.9 39.9 ± 6.0 |
47.6 ± 4.6 47.1 ± 9.5 43.5 ± 7.8 |
0.199 | 0.001* | 0.223 |
Data shown as mean ± SD
Represent the main effect (Significant at p < 0.050).
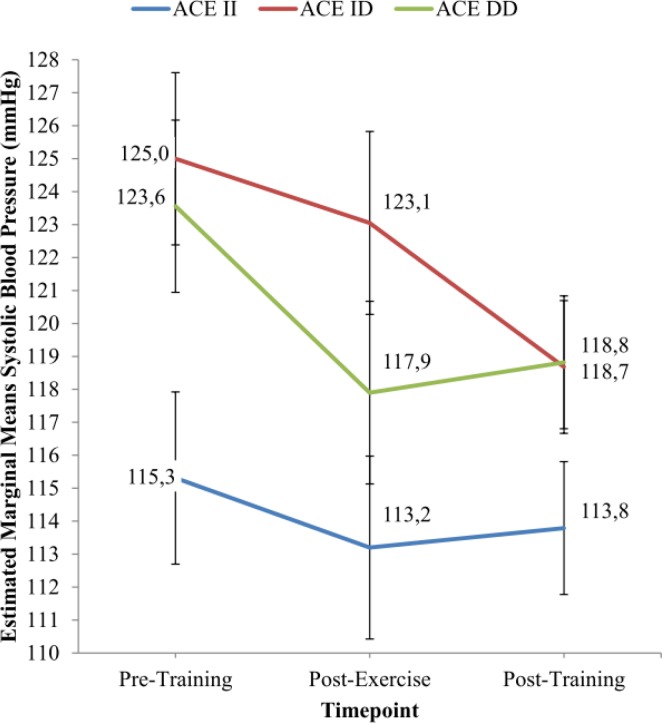
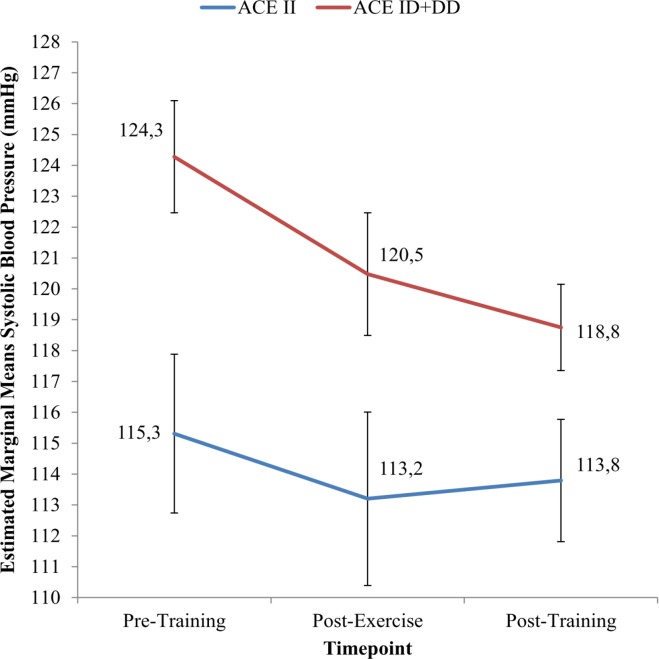
Post hoc tests using the Bonferroni correction revealed that IHG training elicited a significant reduction in SBP score from pre-training to after 8 weeks of IHG training (p = 0.02). However, a session of IHG exercise (post-exercise) elicited a slight reduction in SBP score from pre-training, which was not statistically significant (p = 0.064). There was also a significant main effect of ACE I/D gene polymorphism on SBP score (p = 0.038). Figure 2 shows that the SBP scores (estimated marginal means) after 8 weeks of IHG training for the II genotype group (113.8 ± 2.0 mmHg) are lower than those for the ID (118.7 ± 2.0 mmHg) and DD (118.8 ± 2.0 mmHg) genotype groups. However, as shown in Table 4, the SBP reduction was significantly greater in the ID genotype group (-6.32 ± 7.3 mmHg) than the II (-1.52 ± 5.3 mmHg) and DD (-4.74 ± 5.2 mmHg) genotype groups.
FIG. 2.
Estimated marginal means of Systolic Blood Pressure (SBP) in ACE II, ACE ID and ACE DD genotype groups at Pre-Training, Post-Exercise and Post-Training.
TABLE 4.
Cardiovascular and muscular changes to a session of IHG exercise and 8 weeks of IHG training according to ACE I/D genotype.
| Variables | Geno-type | n | Pre-training | Mid-training | Post-training | Change (Δ) with IHG exercise | Change (Δ) with IHG training | t valuea | p valuea | t valueb | p valueb |
|---|---|---|---|---|---|---|---|---|---|---|---|
| SBP (mmHg) |
II ID DD |
10 10 10 |
115.3 ± 7.2 125.0 ± 9.3 123.6 ± 8.2 |
113.2 ± 7.5 123.1 ± 8.9 117.9 ± 9.8* |
113.8 ± 6.3 118.7 ± 4.9* 118.8 ± 7.6* |
-2.11 ± 6.8 -1.95 ± 8.4 -5.66 ± 6.5 |
-1.52 ± 5.3 -6.32 ± 7.3 -4.74 ± 5.2 |
-0.986 -0.738 -2.734 |
0.350 0.479 0.023 |
-0.909 -2.750 -2.905 |
0.387 0.022 0.017 |
| DBP (mmHg) |
II ID DD |
10 10 10 |
70.1 ± 4.6 80.6 ± 5.9 77.8 ± 6.7 |
67.3 ± 7.0 78.5 ± 5.2 75.6 ± 9.1 |
68.6 ± 6.7 78.8 ± 6.7 76.7 ± 6.9 |
-2.77 ± 5.6 -2.14 ± 3.9 -2.26 ± 9.4 |
-1.43 ± 3.7 -1.76 ± 5.7 -1.11 ± 4.4 |
-1.576 -1.747 -0.759 |
0.149 0.115 0.467 |
-1.231 -0.971 -0.801 |
0.250 0.357 0.444 |
| MAP (mmHg) |
II ID DD |
10 10 10 |
85.2 ± 4.5 95.4 ± 5.8 93.1 ± 6.0 |
82.6 ± 6.4 93.3 ± 4.5 89.7 ± 8.8 |
83.7 ± 5.3 92.1 ± 5.3 90.7 ± 6.7 |
-2.55 ± 5.0 -2.08 ± 4.9 -3.39 ± 7.7 |
-1.45 ± 3.5 -3.28 ± 5.3 -2.32 ± 3.7 |
-1.599 -1.347 -1.387 |
0.144 0.211 0.199 |
-1.309 -1.953 -1.987 |
0.223 0.083 0.078 |
| PP (mmHg) |
II ID DD |
10 10 10 |
45.2 ± 7.3 44.4 ± 9.2 45.8 ± 8.7 |
45.9 ± 7.0 44.6 ± 10.3 42.4 ± 6.6 |
45.2 ± 8.2 39.9 ± 6.4 42.1 ± 5.1 |
0.66 ± 6.9 0.19 ± 6.5 -3.40 ± 7.9 |
-0.09 ± 5.2 -4.56 ± 7.2 -3.63 ± 6.0 |
0.303 0.092 -1.364 |
0.769 0.928 0.206 |
-0.054 -2.009 -1.899 |
0.958 0.075 0.090 |
| HR (bpm) |
II ID DD |
10 10 10 |
78.6 ± 6.5 79.8 ± 9.5 79.8 ± 7.7 |
77.0 ± 8.7 77.0 ± 10.2 81.7 ± 8.3 |
78.9 ± 8.1 76.1 ± 9.3* 75.5 ± 9.8* |
-1.55 ± 6.8 -2.89 ± 9.1 1.90 ± 6.8 |
0.37 ± 8.1 -3.75 ± 3.5 -4.30 ± 4.2 |
-0.726 -1.009 0.884 |
0.486 0.339 0.400 |
0.144 -3.392 -3.246 |
0.889 0.008 0.010 |
| HGS (kg) |
II ID DD |
10 10 10 |
43.4 ± 4.9 46.3 ± 8.0 40.8 ± 6.3 |
43.3 ± 5.7 46.3 ± 7.9 39.9 ± 6.0 |
47.6 ± 4.6* 47.1 ± 9.5 43.5 ± 7.8 |
-0.11 ± 2.1 -0.06 ± 1.4 -0.84 ± 3.2 |
4.18 ± 4.1 0.75 ± 2.8 2.71 ± 5.6 |
-0.149 -0.136 0.844 |
0.885 0.895 0.400 |
3.212 0.851 0.420 |
0.011 0.417 0.158 |
Data shown as mean ± SD
Significantly different compared to pre-training value (p < 0.050)
p value for change (Δ) with IHG exercise
p value for change (Δ) with IHG training.
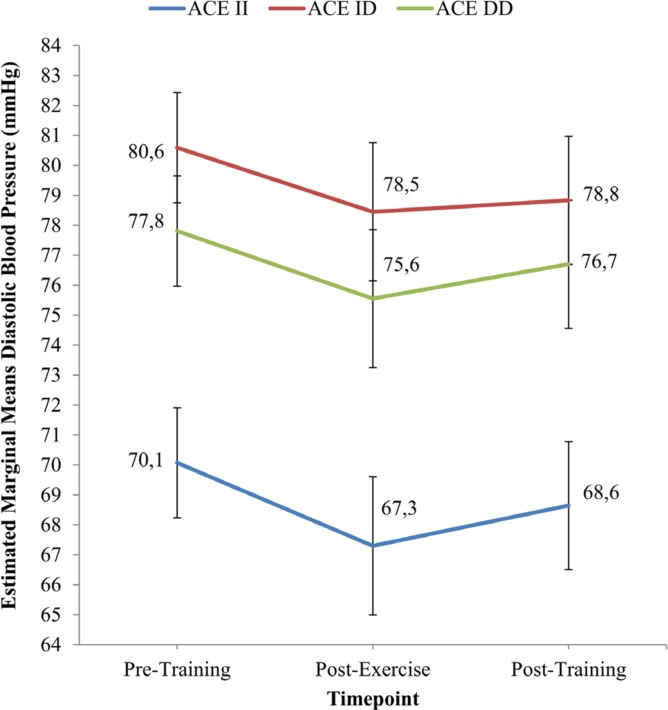
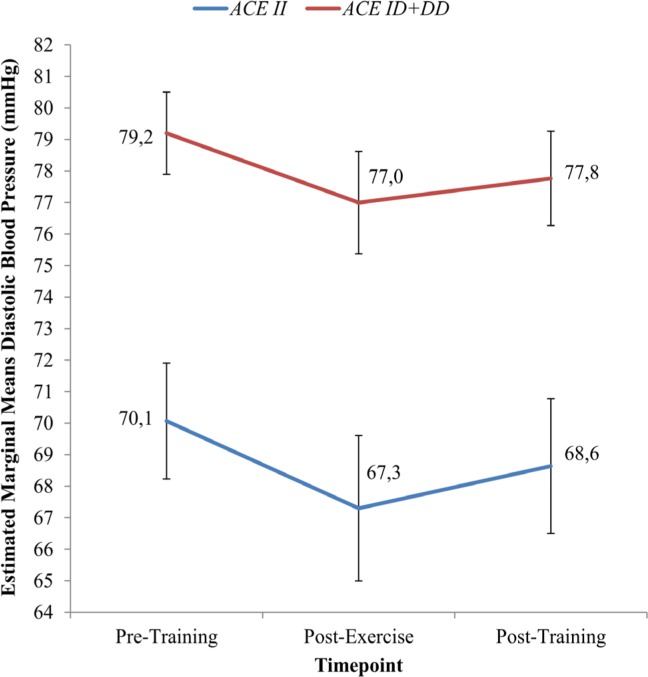
There was also a significant main effect of ACE I/D gene polymorphism on DBP score (p = 0.001). The DBP scores (estimated marginal means) after 8 week of IHG training for the II genotype group (68.6 ± 2.1 mmHg) are lower than those for the ID (78.8 ± 2.1 mmHg) and DD (76.7 ± 2.1 mmHg) genotype groups (Figure 3). However, Table 4 shows that the DBP reduction was greater in the ID genotype group (-1.76 ± 5.7 mmHg) than II (-1.43 ± 3.7 mmHg) and DD (-1.11 ± 4.4 mmHg) genotype groups, though it was not significantly difference from pre-training values.
FIG. 3.
Estimated marginal means of Diastolic Blood Pressure (DBP) in ACE II, ACE ID and ACE DD genotype groups at Pre-Training, Post-Exercise and Post-Training.
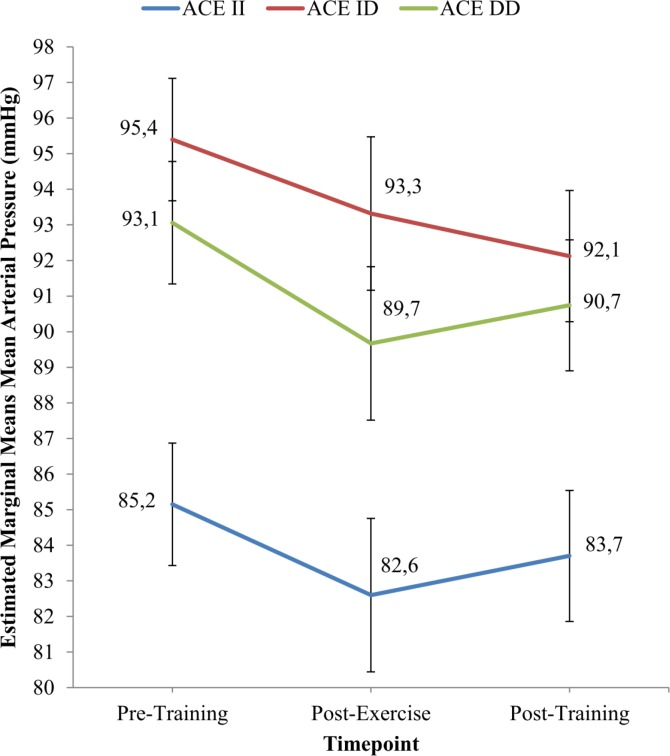
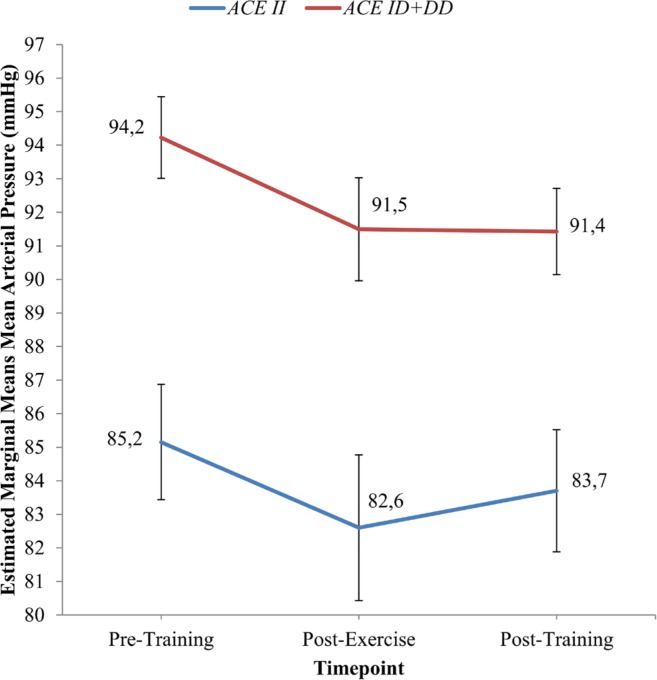
For MAP, IHG exercise elicited a significant reduction in MAP score from pre-training to 8 weeks of IHG training (p = 0.016). However, the MAP score was not significantly lower at post-exercise compared to pre-training (p = 0.066). There was a significant main effect of ACE I/D gene polymorphism on MAP score (p = 0.001). The MAP score (estimated marginal means) after 8 weeks of IHG training for the II genotype group (83.7 ± 1.8 mmHg) was lower than those for the ID (92.1 ± 1.8 mmHg) and DD genotype groups (90.7 ± 1.8 mmHg) (Figure 4). However, Table 4 shows that the MAP reduction was greater in the ID genotype group (-3.28 ± 5.3 mmHg) than II (-1.45 ± 3.5 mmHg) and DD (-2.32 ± 3.7 mmHg) genotype groups though it was not significantly different from pre-training values.
FIG. 4.
Estimated marginal means of Mean Arterial Pressure (MAP) in ACE II, ACE ID and ACE DD genotype groups at Pre-Training, Post-Exercise and Post-Training.
Post hoc tests using the Bonferroni correction also revealed that the increase in HGS as a result of IHG training was statistically significant following 8 weeks of IHG training (p = 0.001). Conversely, a session of IHG exercise did not lead to any increment in HGS score (p > 0.095). Additionally, the gains realized in HGS score were not significantly affected by ACE I/D gene polymorphism (p = 0.223) (Table 3).
To further analyse the association between the ACE I/D gene polymorphism and training adaptation, the dominant model was used in which the data for ID and DD genotype groups were combined and then compared to the data of the II genotype group. Table 5 shows the cardiovascular and muscular variables at pre-training, post-exercise and post-training according to the dominant model (II vs. ID + DD genotypes). A two-way repeated measure ANOVA revealed no significant interaction effect between dominant ACE genotype (II vs. ID+DD groups) and IHG training session on all dependent variables. However, a significant main effect of IHG intervention (pre-training, post-exercise and post-training) was found in SBP (p = 0.015), MAP (p = 0.029), and HGS (p = 0.002) scores, while no difference was found in DBP, PP, and HR scores across the IHG training session.
TABLE 5.
Repeated-Measures 2-way ANOVA on Cardiovascular and muscular responses to a session of IHG exercise and 8 weeks of IHG training according to dominant model (II vs. ID+DD).
| Repeated-Measures 2-way ANOVA | ||||||||
|---|---|---|---|---|---|---|---|---|
| Variables | Genotype | n | Pre-training | Mid-training | Post-training | Interaction (dominant genotype x training session) | Training effect (within subject) | Dominant Genotype effect (between subjects) |
| SBP (mmHg) |
II ID+DD |
10 20 |
115.3 ± 7.2 124.3 ± 8.6 |
113.2 ± 7.5 120.5 ± 9.5 |
113.8 ± 6.3 118.8 ± 6.2 |
0.285 | 0.015* | 0.013* |
| DBP (mmHg) |
II ID+DD |
10 20 |
70.1 ± 4.6 79.2 ± 6.3 |
67.3 ± 7.0 77.0 ± 7.4 |
68.6 ± 6.7 77.8 ± 6.7 |
0.959 | 0.100 | 0.002* |
| MAP (mmHg) |
II ID+DD |
10 20 |
85.2 ± 4.5 94.2 ± 5.8 |
82.6 ± 6.4 91.5 ± 7.1 |
83.7 ± 5.3 91.4 ± 6.0 |
0.774 | 0.029* | 0.002* |
| PP (mmHg) |
II ID+DD |
10 20 |
45.2 ± 7.3 45.1 ± 8.7 |
45.9 ± 7.0 43.5 ± 8.5 |
45.2 ± 8.2 41.0 ± 5.8 |
0.292 | 0.232 | 0.394 |
| HR (bpm) |
II ID+DD |
10 20 |
78.6 ± 6.5 79.8 ± 8.4 |
77.0 ± 8.7 79.3 ± 9.4 |
78.9 ± 8.1 75.8 ± 9.3 |
0.135 | 0.431 | 0.960 |
| HGS (kg) |
II ID+DD |
10 20 |
43.4 ± 4.9 43.6 ± 7.6 |
43.3 ± 5.7 43.1 ± 7.6 |
47.6 ± 4.6 45.3 ± 8.7 |
0.196 | 0.002* | 0.776 |
Data shown as mean ± SD
Represent the main effect (Significant at p < 0.050).
There was also a significant main effect of ACE I/D gene polymorphism on SBP (p = 0.013), DBP (p = 0.002) and MAP (p = 0.002) according to the dominant model. Figure 5 shows a main effect of IHG training and ACE genotype according to the dominant model, and no interaction between these two variables on SBP. The SBP score after 8 weeks of IHG training (post-training) for the II genotype group (113.8 ± 2.0 mmHg) was lower than that for the ID + DD genotype group (118.8 ± 1.4 mmHg). However, as shown in Table 6, the SBP reduction following the 8 weeks of IHG training was greater in the ID+DD genotype group (-5.53 ± 6.2 mmHg) than the II genotype group (-1.52 ± 5.3 mmHg).
FIG. 5.
Estimated marginal means of Systolic Blood Pressure (SBP) in ACE II and ACE ID + DD genotype groups at Pre-Training, Post-Exercise and Post-Training.
TABLE 6.
Cardiovascular and muscular changes to a session of IHG exercise and 8 weeks of IHG training according to according to dominant model (II vs. ID+DD).
| Variables | Geno- type | n | Pre-training | Mid-training | Post-training | Change (Δ) with IHG exercise | Change (Δ) with IHG training | t valuea | p valuea | t valueb | p valueb |
|---|---|---|---|---|---|---|---|---|---|---|---|
| SBP (mmHg) |
II ID+DD |
10 20 |
115.3 ± 7.2 125.0 ± 9.3 |
113.2 ± 7.5 123.1 ± 8.9* |
113.8 ± 6.3 118.7 ± 4.9* |
-2.11 ± 6.8 -3.81 ± 7.6 |
-1.52 ± 5.3 -5.53 ± 6.2 |
-0.986 -2.254 |
0.350 0.036 |
-0.909 -3.997 |
0.387 0.001 |
| DBP (mmHg) |
II ID+DD |
10 20 |
70.1 ± 4.6 79.2 ± 6.3 |
67.3 ± 7.0 77.0 ± 7.4 |
68.6 ± 6.7 77.8 ± 6.7 |
-2.77 ± 5.6 -2.20 ± 7.0 |
-1.43 ± 3.7 -1.44 ± 5.0 |
-1.576 -1.404 |
0.149 0.213 |
-1.231 -1.289 |
0.250 0.176 |
| MAP (mmHg) |
II ID+DD |
10 20 |
85.2 ± 4.5 94.2 ± 5.8 |
82.6 ± 6.4 91.5 ± 7.1 |
83.7 ± 5.3 91.4 ± 6.0* |
-2.55 ± 5.0 -2.74 ± 6.3 |
-1.45 ± 3.5 -2.80 ± 4.5 |
-1.599 -1.933 |
0.144 0.068 |
-1.309 -0.704 |
0.223 0.012 |
| PP (mmHg) |
II ID+DD |
10 20 |
45.2 ± 7.3 45.1 ± 8.7 |
45.9 ± 7.0 43.5 ± 8.5 |
45.2 ± 8.2 41.0 ± 5.8* |
0.66 ± 6.9 -1.61 ± 7.3 |
-0.09 ± 5.2 -4.10 ± 6.5 |
0.303 -0.987 |
0.769 0.336 |
-0.054 -2.828 |
0.958 0.011 |
| HR (bpm) |
II ID+DD |
10 20 |
78.6 ± 6.5 79.8 ± 8.4 |
77.0 ± 8.7 79.3 ± 9.4 |
78.9 ± 8.1 75.8 ± 9.3* |
-1.55 ± 6.8 -0.50 ± 8.2 |
0.37 ± 8.1 -4.03 ± 3.8 |
-0.726 -0.271 |
0.486 0.789 |
0.144 -4.780 |
0.889 0.001 |
| HGS (kg) |
II ID+DD |
10 20 |
43.4 ± 4.9 46.6 ± 7.6 |
43.3 ± 5.7 43.1 ± 7.6 |
47.6 ± 4.6* 45.3 ± 7.6 |
-0.11 ± 2.1 -0.45 ± 2.4 |
4.18 ± 4.1 1.73 ± 4.4 |
-0.149 -0.838 |
0.885 0.413 |
3.212 1.760 |
0.011 0.095 |
Data shown as mean ± SD
Significantly different compared to pre-training value (p < 0.050)
p value for change (Δ) with IHG exercise;
p value for change (Δ) with IHG training.
The DBP score (estimated marginal means) after 8 weeks of IHG training for the II genotype group (68.6 ± 2.1 mmHg) was lower than that for the ID + DD group (77.8 ± 1.5 mmHg) (Figure 6). However, Table 6 shows that the DBP reduction was slightly greater in the ID + DD genotype group (-1.44 ± 5.0 mmHg) than the II genotype group (-1.43 ± 3.7 mmHg), though it was not significantly different from pre-training values.
FIG. 6.
Estimated marginal means of Diastolic Blood Pressure (DBP) in ACE II and ACE ID + DD genotype groups at Pre-Training, Post-Exercise and Post-Training.
The MAP score (estimated marginal means) after 8 weeks of IHG training for the II genotype group (83.7 ± 1.8 mmHg) was lower than for the ID + DD genotype group (91.4 ± 1.3 mmHg) (Figure 7). However, Table 6 shows that the MAP reduction was significantly greater in the ID + DD genotype group (-2.80 ± 4.5 mmHg) than the II genotype group (-1.45 ± 3.5 mmHg).
FIG. 7.
Estimated marginal means of Mean Arterial Pressure (MAP) in ACE II and ACE ID + DD genotype groups at Pre-Training, Post-Exercise and Post-Training.
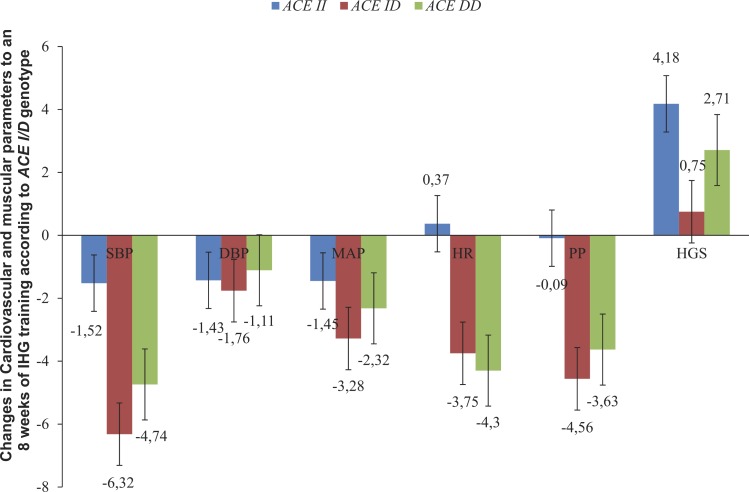
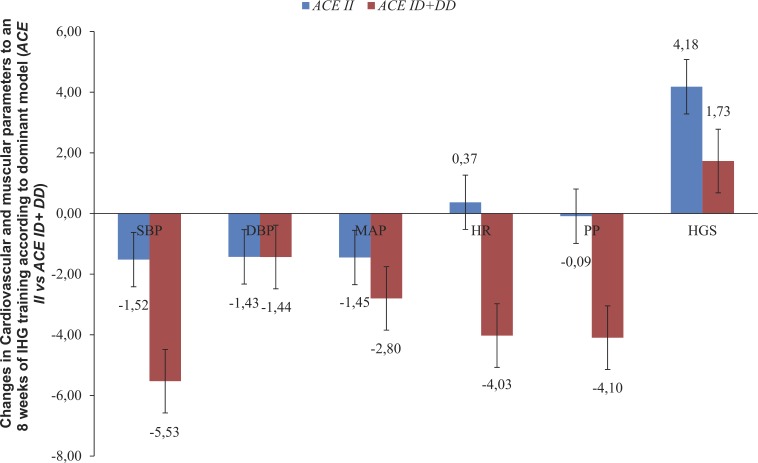
Figure 8 presents the cardiovascular and muscular changes in all variables following 8 weeks of IHG training among three ACE genotype groups (II, ID and DD). Meanwhile, Figure 9 displays the changes in the studied variables following 8 weeks of IHG training according to the dominant model (II vs. ID+DD).
FIG. 8.
Cardiovascular and muscular changes to an 8 weeks of IHG training (Post-Training value minus Pre-Training value) according to ACE I/D genotype.
SBP=Systolic Blood Pressure; DBP=Diastolic Blood Pressure; MAP=Mean Arterial Pressure; PP=Pulse Pressure; HGS=Handgrip Strengt.
FIG. 9.
Cardiovascular and muscular changes to 8 weeks of IHG training (Post-Training value minus Pre-Training value) according to dominant model (ACE II vs ACE ID+DD).
SBP=Systolic Blood Pressure; DBP=Diastolic Blood Pressure; MAP=Mean Arterial Pressure; PP=Pulse Pressure; HGS=Handgrip Strength.
DISCUSSION
The present study found significant differences in SBP, MAP and HGS scores between the pre-training and post-training, which showed that an 8-week IHG training programme elicits improvements in cardiovascular and muscular performances of normotensive men. In addition, greater improvements in cardiovascular parameters (SBP, DBP and MAP) were observed in participants with the D allele than those with the I allele. This finding was indicated by significant reductions in SBP, DBP and MAP scores (parameters for cardiovascular parameters) in the D allele group compared to the I allele group from the genotyping analysis (Table 4) and additional analysis by using the dominant model (Table 6).
The current study showed that cardiovascular responses to IHG training varied among normotensive individuals with different ACE I/D genotypes. Consistent with the results obtained by Kim [30], this study revealed that normotensive men with DD and ID genotypes tended to have decreased resting SBP and HR with IHG training more than those with II genotype (Table 6). Nevertheless, this finding was not comparable to the findings obtained from some previous studies [26, 28] which showed that individuals with the II genotype had lower resting BP than other genotypes after exercise. This inconsistency may be due to the differences in the mode of exercise performed, as for instance a study by Kim [30] which involved mixed aerobic and resistance exercise training compared with only aerobic training (endurance training) in a study by Hagberg et al. [26]. Several studies have demonstrated that the physiological adaptation for aerobic training was greater among individuals with the II genotype and those with the DD genotype responded better to resistance training [37-39]. Thus, the inconsistency between the above-mentioned studies may be due to differences in the intensity of exercise performed.
At present, the mechanism by which isometric exercise training elicits a reduction in BP has remained unclear [40]. Wiles et al. [41] suggested that the rise in BP during isometric exercise will stimulate the baroreceptors, which are sensory afferent nerve endings located in the carotid sinus and the aortic arch. When the BP is elevated, the baroreceptors are stretched and result in a reflex-mediated increase in parasympathetic nerve activity, as well as a decrease in sympathetic nerve activity [41]. Consequently, it causes a decline in the heart rate, while the diameter of blood vessels increases and further leads to a drop in the BP [41]. Moreover, it has been suggested that the reduction in BP after isometric exercise training is related to the repeated power of hydrogen (pH) changes due to muscle fatigue and lactate production that act as a metaboreceptor stimulus [42], augmentation in vasodilator substances, for instance, nitric oxide (NO) [43], and reduction in peripheral vascular adaptations [44].
Data from the HEalth, RIsk factors, exercise Training And Genetics (HERITAGE) Family Study suggest that reduction in BP after exercise may be influenced by genetic factors [45]. In this regard, the ACE gene was initially believed to influence the BP response to exercise [19-21] due to its role in the RAS. Rigat et al. [23] reported that individuals with the DD genotype had higher ACE activity compared to those with the II genotype. A higher level of ACE in the circulation and skeletal muscle renin-angiotensin system (RAS) would increase the production of angiotensin II (ANG II) [46, 47]. Nevertheless, ANG II has different effects on circulating and skeletal muscle RAS [46]. In circulating RAS, ANG II binds to several receptors that construct the blood vessels to increase BP [47]. However, ANG II in skeletal muscle RAS stimulates the production of angiotensin (ANG) (1-7) peptide, a potent vasodilator that causes a decrease in BP [46]. As this study employed IHG training that particularly involved the contraction of skeletal muscle, the reduction in SBP and HR in individuals with DD genotype observed in this study might be interpreted as due to high production of ANG (1-7) during exercise. Nonetheless, the present study did not measure the components of the skeletal muscle RAS. This therefore warrants future studies to confirm this possible mechanism.
The greater reduction in SBP and HR among DD genotype carriers could also be due to their higher baseline BP values. This present result was consistent with the finding obtained by Millar et al. [48], who demonstrated that normotensive individuals with higher baseline values of resting SBP and HR had a more pronounced reduction in these parameters after IHG training than those with lower baseline values. This finding was also consistent with Wiley et al. [2], who observed a larger reduction in resting BP in hypertensive patients following isometric exercise training compared with normotensive individuals. Collectively, these findings support the idea of Badrov et al. [12], who had previously suggested that individuals with higher baseline BP values might have greater capacity for reduction in BP following IHG training compared to those with lower baseline BP values. This speculation is supported by the fact that those with higher resting BP, such as hypertensive patients, present greater sympathetic activity at rest [49], which could lead to greater hemodynamic responsiveness to sympathetic activation [50].
The findings of the current study reaffirm previous reports of a reduction in resting BP after 8 weeks of IHG training among normotensive individuals [7, 8, 12]. Similar to previous studies [7, 8, 12], this study found that 8 weeks of IHG training significantly decreased resting SBP, MAP, and PP by 4.2 ± 6.1 mmHg, 2.4 ± 4.2 mmHg, and 2.8 ± 6.3 mmHg, respectively (Table 2). However, no significant difference was observed in resting DBP after the IHG training programme, which was similar to the observations of Badrov et al. [12] in normotensive women. However, the reason for the lack of change in DBP following 8 weeks of IHG training remains unclear. The underlying mechanism could be that DBP has a smaller range of values than SBP, which would limit the maximal change in DBP value [51]. Future studies with larger sample sizes may increase the chance of discovering a significant difference.
Concomitant with the reduction in resting BP, resting HR was also significantly lower after 8 weeks of IHG training in this study (Table 2). This decrease was similar to that reported previously by Singh et al. [52]. In fact, previous studies have suggested that the decrease in resting BP and HR may be due to a reduction in sympathetic nerve activity during IHG exercise [2, 53]. When BP is elevated during IHG exercise, the baroreceptors are stretched, resulting in a reflex-mediated increase in parasympathetic nerve activity, and a decrease in sympathetic nerve activity [2]. Consequently, it caused the decline in the heart rate, while the diameter of blood vessels increased, leading to a drop in the BP [2].
Besides resting BP and HR, the present study also showed that 8 weeks of IHG training significantly improved muscle strength (Table 2). The increase in muscle strength observed in the present study is consistent with results of previous studies demonstrating that IHG exercise at 30% of MVC improved muscle strength [9, 54]. Considering the significant changes in resting BP and HR as well as muscle strength following IHG training, the training protocol adopted in the present study may be prescribed as part of lifestyle modification for maintaining a desirable BP level.
Comparable with the findings of a cardiovascular response to IHG training, a significant reduction in cardiovascular variables immediately following the first session of IHG exercise was observed (acute effects) (Table 2). Resting SBP and MAP were significantly reduced by 3.2 ± 7.2 mm Hg and 2.7 ± 5.8 mmHg, respectively, in response to acute IHG exercise. These findings indicated that IHG exercise may provide substantial benefits for hypertensive patients by lowering their resting BP and HR for a period of time after acute isometric exercise exposure. Nevertheless, for individuals with uncontrolled hypertension, the IHG exercise can cause a temporary increase in BP due to the increase in muscle tension. Hence, to prevent any potential risk, the IHG exercise should be performed at low intensity (30% MVC) and needs to be closely monitored by the clinician.
Although the present study has yielded some useful findings, there is a limitation that should be taken into consideration. The sample size in each genotype group may have been too small, and further, larger studies are required to confirm these results. The present study did not include a non-exercising control group to reduce the variability of the results, as the study was primarily designed to investigate the effects of ACE I/D gene polymorphism on IHG training adaptation by comparison among genotype groups. Despite this limitation, the current results remain valid and applicable, as the participants in each group were relatively homogeneous in terms of sex, physical characteristics, health status, and ethnic background. The training intervention was standardized, and the same trained investigator conducted all the training sessions.
CONCLUSIONS
In conclusion, the present study showed that individuals with the D allele exhibited a significantly greater improvement in cardiovascular parameters after IHG training compared to individuals with the I allele. Future studies on hypertensive patients are warranted to observe whether the ACE I/D gene polymorphism drives similar cardiovascular and muscular changes in hypertensive patients following 8 weeks of IHG training. Moreover, as hypertension is a multifactorial outcome, future studies are needed to address the role of ACE I/D gene polymorphism and other genetic variants in the BP response following IHG training.
Acknowledgements
The authors thank the volunteers who made this study possible. This study was funded by the Student Research Fund of the Advanced Medical and Dental Institute (AMDI), Universiti Sains Malaysia.
Conflict of Interest
The authors declare that they have no conflict of interest.
REFERENCES
- 1.Palatini P, Mos L, Munari L, Valle F, Del Torre M, Rossi A, Varotto L, Macor F, Martina S, Pessina AC. Blood pressure changes during heavy-resistance exercise. J Hypertens Suppl. 1989;7(6):S72–73. doi: 10.1097/00004872-198900076-00032. [DOI] [PubMed] [Google Scholar]
- 2.Wiley RL, Dunn CL, Cox RH, Hueppchen NA, Scott MS. Isometric exercise training lowers resting blood pressure. Med Sci Sports Exerc. 1992;24(7):749–754. [PubMed] [Google Scholar]
- 3.Wood RH, Reyes R, Welsch MA, Favaloro-Sabatier J, Sabatier M, Matthew Lee C, Johnson LG, Hooper PF. Concurrent cardiovascular and resistance training in healthy older adults. Med Sci Sports Exerc. 2001;33(10):1751–1758. doi: 10.1097/00005768-200110000-00021. [DOI] [PubMed] [Google Scholar]
- 4.Vincent KR, Vincent HK, Braith RW, Bhatnagar V, Lowenthal DT. Strength training and hemodynamic responses to exercise. Am J Geriatr Cardiol. 2003;12(2):97–106. doi: 10.1111/j.1076-7460.2003.01588.x. [DOI] [PubMed] [Google Scholar]
- 5.Collier SR, Kanaley JA, Carhart R, Jr, Frechette V, Tobin MM, Hall AK, Luckenbaugh AN, Fernhall B. Effect of 4 weeks of aerobic or resistance exercise training on arterial stiffness, blood flow and blood pressure in pre- and stage-1 hypertensives. J Hum Hypertens. 2008;22(10):678–686. doi: 10.1038/jhh.2008.36. [DOI] [PubMed] [Google Scholar]
- 6.Cornelissen VA, Smart NA. Exercise training for blood pressure: a systematic review and meta-analysis. J Am Heart Assoc. 2013;2(1):e004473. doi: 10.1161/JAHA.112.004473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Mcgowan CL, Visocchi A, Faulkner M, Verduyn R, Rakobowchuk M, Levy AS, Mccartney N, Macdonald MJ. Isometric handgrip training improves local flow-mediated dilation in medicated hypertensives. Eur J Appl Physiol. 2007;99(3):227–234. doi: 10.1007/s00421-006-0337-z. [DOI] [PubMed] [Google Scholar]
- 8.Millar PJ, Bray SR, Macdonald MJ, Mccartney N. The hypotensive effects of isometric handgrip training using an inexpensive spring handgrip training device. J Cardiopulm Rehabil Prev. 2008;28(3):203–207. doi: 10.1097/01.HCR.0000320073.66223.a7. [DOI] [PubMed] [Google Scholar]
- 9.Howden R, Lightfoot JT, Brown SJ, Swaine IL. The effects of isometric exercise training on resting blood pressure and orthostatic tolerance in humans. Exp Physiol. 2002;87(4):507–515. doi: 10.1111/j.1469-445x.2002.tb00064.x. [DOI] [PubMed] [Google Scholar]
- 10.Ray CA, Carrasco DI. Isometric handgrip training reduces arterial pressure at rest without changes in sympathetic nerve activity. Am J Physiol Heart Circ Physiol. 2000;279(1):H245–249. doi: 10.1152/ajpheart.2000.279.1.H245. [DOI] [PubMed] [Google Scholar]
- 11.Devereux G, Wiles J, Swaine I. Reductions in resting blood pressure after 4 weeks of isometric exercise training. Euro J Appl Physiol. 2010;109(4):601–606. doi: 10.1007/s00421-010-1394-x. [DOI] [PubMed] [Google Scholar]
- 12.Badrov MB, Bartol CL, Dibartolomeo MA, Millar PJ, Mcnevin NH, Mcgowan CL. Effects of isometric handgrip training dose on resting blood pressure and resistance vessel endothelial function in normotensive women. Euro J Appl Physiol. 2013;113(8):2091–2100. doi: 10.1007/s00421-013-2644-5. [DOI] [PubMed] [Google Scholar]
- 13.Carlson DJ, Dieberg G, Hess NC, Millar PJ, Smart NA. Isometric exercise training for blood pressure management: a systematic review and meta-analysis. Mayo Clin Proc. 2014;89(3):327–334. doi: 10.1016/j.mayocp.2013.10.030. [DOI] [PubMed] [Google Scholar]
- 14.Pescatello LS, Franklin BA, Fagard R, Farquhar WB, Kelley GA, Ray CA. American College of Sports Medicine position stand. Exercise and hypertension. Med Sci Sports Exerc. 2004;36(3):533–553. doi: 10.1249/01.mss.0000115224.88514.3a. [DOI] [PubMed] [Google Scholar]
- 15.Van Den Bree MB, Schieken RM, Moskowitz WB, Eaves LJ. Genetic regulation of hemodynamic variables during dynamic exercise. The MCV twin study. Circulation. 1996;94(8):1864–1869. doi: 10.1161/01.cir.94.8.1864. [DOI] [PubMed] [Google Scholar]
- 16.Hottenga JJ, Boomsma DI, Kupper N, Posthuma D, Snieder H, Willemsen G, De Geus EJ. Heritability and stability of resting blood pressure. Twin Res Hum Genet. 2005;8(5):499–508. doi: 10.1375/183242705774310123. [DOI] [PubMed] [Google Scholar]
- 17.Sober S, Org E, Kepp K, Juhanson P, Eyheramendy S, Gieger C, Lichtner P, Klopp N, Veldre G, Viigimaa M, Doring A, Putku M, Kelgo P, Shaw-Hawkins S, Howard P, Onipinla A, Dobson RJ, Newhouse SJ, Brown M, Dominiczak A, Connell J, Samani N, Farrall M, Caulfield MJ, Munroe PB, Illig T, Wichmann HE, Meitinger T, Laan M. Targeting 160 candidate genes for blood pressure regulation with a genome-wide genotyping array. PloS One. 2009;4(6):e6034. doi: 10.1371/journal.pone.0006034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Arora P, Newton-Cheh C. Blood pressure and human genetic variation in the general population. Curr Opin Cardiol. 2010;25(3):229–237. doi: 10.1097/HCO.0b013e3283383e2c. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Hagberg JM, Park JJ, Brown MD. The role of exercise training in the treatment of hypertension: an update. Sports Med. 2000;30(3):193–206. doi: 10.2165/00007256-200030030-00004. [DOI] [PubMed] [Google Scholar]
- 20.Marteau JB, Zaiou M, Siest G, Visvikis-Siest S. Genetic determinants of blood pressure regulation. J Hypertens. 2005;23(12):2127–2143. doi: 10.1097/01.hjh.0000186024.12364.2e. [DOI] [PubMed] [Google Scholar]
- 21.Dhanachandra Singh K, Jajodia A, Kaur H, Kukreti R, Karthikeyan M. Gender Specific Association of RAS Gene Polymorphism with Essential Hypertension: A Case-Control Study. Biomed Res Int. 2014:10. doi: 10.1155/2014/538053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Tiret L, Rigat B, Visvikis S, Breda C, Corvol P, Cambien F, Soubrier F. Evidence, from combined segregation and linkage analysis, that a variant of the angiotensin I-converting enzyme (ACE) gene controls plasma ACE levels. Am J Hum Genet. 1992;51(1):197–205. [PMC free article] [PubMed] [Google Scholar]
- 23.Rigat B, Hubert C, Alhenc-Gelas F, Cambien F, Corvol P, Soubrier F. An insertion/deletion polymorphism in the angiotensin I-converting enzyme gene accounting for half the variance of serum enzyme levels. J Clin Invest. 1990;86(4):1343–1346. doi: 10.1172/JCI114844. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Coates D. The angiotensin converting enzyme (ACE) Int J Biochem Cell Biol. 2003;35(6):769–773. doi: 10.1016/s1357-2725(02)00309-6. [DOI] [PubMed] [Google Scholar]
- 25.Montgomery HE, Clarkson P, Dollery CM, Prasad K, Losi MA, Hemingway H, Statters D, Jubb M, Girvain M, Varnava A, World M, Deanfield J, Talmud P, Mcewan JR, Mckenna WJ, Humphries S. Association of angiotensin-converting enzyme gene I/D polymorphism with change in left ventricular mass in response to physical training. Circulation. 1997;96(3):741–747. doi: 10.1161/01.cir.96.3.741. [DOI] [PubMed] [Google Scholar]
- 26.Hagberg JM, Ferrell RE, Dengel DR, Wilund KR. Exercise training-induced blood pressure and plasma lipid improvements in hypertensives may be genotype dependent. Hypertension. 1999;34(1):18–23. doi: 10.1161/01.hyp.34.1.18. [DOI] [PubMed] [Google Scholar]
- 27.Rankinen T, Gagnon J, Perusse L, Chagnon YC, Rice T, Leon AS, Skinner JS, Wilmore JH, Rao DC, Bouchard C. AGT M235T and ACE ID polymorphisms and exercise blood pressure in the HERITAGE Family Study. Am J Physiol Heart Circ Physiol. 2000;279(1):H368–374. doi: 10.1152/ajpheart.2000.279.1.H368. [DOI] [PubMed] [Google Scholar]
- 28.Zhang B, Sakai T, Miura S, Kiyonaga A, Tanaka H, Shindo M, Saku K. Association of angiotensin-converting-enzyme gene polymorphism with the depressor response to mild exercise therapy in patients with mild to moderate essential hypertension. Clin Genet. 2002;62(4):328–333. doi: 10.1034/j.1399-0004.2002.620414.x. [DOI] [PubMed] [Google Scholar]
- 29.Dengel DR, Brown MD, Ferrell RE, Reynolds THT, Supiano MA. Exercise-induced changes in insulin action are associated with ACE gene polymorphisms in older adults. Physiol Genomics. 2002;11(2):73–80. doi: 10.1152/physiolgenomics.00048.2002. [DOI] [PubMed] [Google Scholar]
- 30.Kim K. Association of angiotensin-converting enzyme insertion/deletion polymorphism with obesity, cardiovascular risk factors and exercise-mediated changes in Korean women. Euro J Appl Physiol. 2009;105(6):879–887. doi: 10.1007/s00421-008-0973-6. [DOI] [PubMed] [Google Scholar]
- 31.Mota MR, Oliveira RJ, Terra DF, Pardono E, Dutra MT, De Almeida JA, Silva FM. Acute and chronic effects of resistance exercise on blood pressure in elderly women and the possible influence of ACE I/D polymorphism. Int J Gen Med. 2013;6:581–587. doi: 10.2147/IJGM.S40628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Lester S, Heatley S, Bardy P, Bahnisch J, Bannister K, Faull R, Clarkson A. The DD genotype of the angiotensin-converting enzyme gene occurs in very low frequency in Australian Aboriginal. Nephrol Dial Transplant. 1999;14(4):887–890. doi: 10.1093/ndt/14.4.887. [DOI] [PubMed] [Google Scholar]
- 33.Pate RR, O’neill JR, Lobelo F. The evolving definition of “sedentary”. Exerc Sport Sci Rev. 2008;36(4):173–178. doi: 10.1097/JES.0b013e3181877d1a. [DOI] [PubMed] [Google Scholar]
- 34.Dupont WD, Plummer WD. Power and sample size calculations: a review and computer program. Control Clin Trials. 1990;11(2):116–128. doi: 10.1016/0197-2456(90)90005-m. [DOI] [PubMed] [Google Scholar]
- 35.Mayne I. Examination of the ACE and ACTN3 Genes in UTC Varsity Athletes and Sedentary Students. The University of Tennessee at Chattanooga; 2006. [Google Scholar]
- 36.Hecksteden A, Grutters T, Meyer T. Association between postexercise hypotension and long-term training-induced blood pressure reduction: a pilot study. Clin J Sport Med. 2013;23(1):58–63. doi: 10.1097/JSM.0b013e31825b6974. [DOI] [PubMed] [Google Scholar]
- 37.Colakoglu M, Cam FS, Kayitken B, Cetinoz F, Colakoglu S, Turkmen M, Sayin M. ACE genotype may have an effect on single versus multiple set preferences in strength training. Eur J Appl Physiol. 2005;95(1):20–26. doi: 10.1007/s00421-005-1335-2. [DOI] [PubMed] [Google Scholar]
- 38.Defoor J, Vanhees L, Martens K, Matthijs G, Van Vlerken A, Zielinska D, Schepers D, Vlietinck R, Fagard R. The CAREGENE study: ACE gene I/D polymorphism and effect of physical training on aerobic power in coronary artery disease. Heart. 2006;92(4):527–528. doi: 10.1136/hrt.2004.054312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Cam S, Colakoglu M, Colakoglu S, Sekuri C, Berdeli A. ACE I/D gene polymorphism and aerobic endurance development in response to training in a non-elite female cohort. J Sports Med Phys Fitness. 2007;47(2):234–238. [PubMed] [Google Scholar]
- 40.Millar PJ, Mcgowan CL, Cornelissen VA, Araujo CG, Swaine IL. Evidence for the role of isometric exercise training in reducing blood pressure: potential mechanisms and future directions. Sports Med. 2014;44(3):345–356. doi: 10.1007/s40279-013-0118-x. [DOI] [PubMed] [Google Scholar]
- 41.Wiles JD, Coleman DA, Swaine IL. The effects of performing isometric training at two exercise intensities in healthy young males. Euro J Appl Physiol. 2010;108(3):419–428. doi: 10.1007/s00421-009-1025-6. [DOI] [PubMed] [Google Scholar]
- 42.Devereux GR. The Effects of Isometric Exercise Training on Resting Blood Pressure With Specific Reference to Selected Cardiovascular, Neuromuscular, and Metabolic Variables. Degree of Doctor of Philosophy. Canterbury Christ Church University; 2010. [Google Scholar]
- 43.Lopez RM, Castillo C, Castillo EF. Isometric contraction increases endothelial nitric oxide synthase activity via a calmodulin antagonist-sensitive pathway in rat aorta. Vasc Pharmacol. 2009;50(1-2):14–19. doi: 10.1016/j.vph.2008.08.001. [DOI] [PubMed] [Google Scholar]
- 44.Gregory M. The effects of isometric handgrip training on carotid arterial compliance and resting blood pressure in postmenopausal women. Degree of Master of Human Kinetics. University of Windsor; 2012. [Google Scholar]
- 45.Wilmore JH, Stanforth PR, Gagnon J, Rice T, Mandel S, Leon AS, Rao DC, Skinner JS, Bouchard C. Heart rate and blood pressure changes with endurance training:t he HERITAGE Family Study. Med Sci Sports Exerc. 2001;33(1):107–116. doi: 10.1097/00005768-200101000-00017. [DOI] [PubMed] [Google Scholar]
- 46.Jones A, Woods DR. Skeletal muscle RAS and exercise performance. Int J Biochem Cell Biol. 2003;35(6):855–866. doi: 10.1016/s1357-2725(02)00342-4. [DOI] [PubMed] [Google Scholar]
- 47.Sayed-Tabatabaei FA, Oostra BA, Isaacs A, Van Duijn CM, Witteman JC. ACE polymorphisms. Circ Res. 2006;98(9):1123–1133. doi: 10.1161/01.RES.0000223145.74217.e7. [DOI] [PubMed] [Google Scholar]
- 48.Millar PJ, Bray SR, Mcgowan CL, Macdonald MJ, Mccartney N. Effects of isometric handgrip training among people medicated for hypertension: a multilevel analysis. Blood Press Monit. 2007;12(5):307–314. doi: 10.1097/MBP.0b013e3282cb05db. [DOI] [PubMed] [Google Scholar]
- 49.Schlaich MP, Lambert E, Kaye DM, Krozowski Z, Campbell DJ, Lambert G, Hastings J, Aggarwal A, Esler MD. Sympathetic augmentation in hypertension: role of nerve firing, norepinephrine reuptake, and Angiotensin neuromodulation. Hypertension. 2004;43(2):169–175. doi: 10.1161/01.HYP.0000103160.35395.9E. [DOI] [PubMed] [Google Scholar]
- 50.Kaushik RM, Mahajan SK, Rajesh V, Kaushik R. Stress profile in essential hypertension. Hypertens Res. 2004;27(9):619–624. doi: 10.1291/hypres.27.619. [DOI] [PubMed] [Google Scholar]
- 51.Pikilidou MI, Scuteri A, Morrell C, Lakatta EG. The burden of obesity on blood pressure is reduced in older persons: the SardiNIA study. Obesity (Silver Spring) 2013;21(1):E10–13. doi: 10.1002/oby.20010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Singh HG, Singh JS, Gupta V, Gurmanpreet Effect of Isometric Handgrip Training on Heart Rate and Arterial Pressure in Normotensive Individuals. Sch J App Med Sci. 2014;2(6A):2010–2015. [Google Scholar]
- 53.Mueller PJ. Exercise training and sympathetic nervous system activity: evidence for physical activity dependent neural plasticity. Clin Exp Pharmacol Physiol. 2007;34(4):377–384. doi: 10.1111/j.1440-1681.2007.04590.x. [DOI] [PubMed] [Google Scholar]
- 54.Mortimer J, Mckune AJ. Effect of short-term isometric handgrip training on blood pressure in middle-aged females. Cardiovasc J Afr. 2011;22(5):257–260. doi: 10.5830/CVJA-2010-090. [DOI] [PMC free article] [PubMed] [Google Scholar]