Abstract
Background
Epithelial-mesenchymal transition (EMT) is a vital process in cancer progression and metastasis. Yanggan Jiedu Sanjie (YGJDSJ) is Traditional Chinese Medicine formulation for liver cancer treatment. In the present study, we evaluated the effects of YGJDSJ on TGF-β1-induced EMT in hepatocellular carcinoma Bel-7402 cells.
Methods
Bel-7402 cells were treated with TGF-β1 and YGJDSJ. EMT was identified by morphological changes and expression of marker proteins. Cell morphology was observed under a microscope. Protein expression and phosphorylation was detected by western blotting. Cell migration was measured by the scratch assay. Cell adhesion and invasion was detected by a commercial kit.
Results
YGJDSJ reversed TGF-β1-induced morphological changes, as well as the expression of the EMT markers E-cadherin and N-cadherin in Bel-7402 cells. YGJDSJ also inhibited TGF-β1 up-regulated Smad3 phosphorylation and Snail expression in Bel-7402 cells. Moreover, YGJDSJ inhibited TGF-β1-induced cell adhesion, migration and invasion in Bel-7402 cells.
Conclusions
YGJDSJ inhibited TGF-β1-induced EMT and mediated metastatic potential of Bel-7402 cells, which may be related to down-regulation of Smad3 phosphorylation and Snail expression. The present study provides a new basis for application of this herbal formula for prevention of liver cancer metastasis.
Keywords: Hepatocellular carcinoma, Epithelial-mesenchymal transition, Chinese herbal medicine, Metastasis
Background
Liver cancer ranks sixth in incidence and fourth in mortality among all cancers globally [1]. Hepatocellular carcinoma (HCC) is the most common liver cancer (75–85%) [1]. Surgery is the only curative treatment option for early HCC [2, 3]. HCC is prone to metastasize to lung (39.5–53.8%), lymph node (33.8–34.2%), bone (25.4–38.5%) and other sites through the lymph or blood circulation [4, 5]. The efficacy of current treatment for metastatic HCC, including radiotherapy, chemotherapy and targeted therapy, is very poor [6, 7]. Therefore, prevention of HCC metastasis is particularly important.
HCC metastasis is closely related to epithelial-mesenchymal transition (EMT) [8, 9]. EMT is a biological process in which epithelial cells lose their epithelial characteristics and acquire the phenotype of mesenchymal cells, including changes in cell morphology and expression of marker proteins, such as down-regulation of epithelial marker gene E-cadherin and up-regulation of mesenchymal marker gene N-cadherin [8, 9]. Tumor cells with EMT have increased motility and invasiveness, which facilitate their metastasis. Suppression of EMT could inhibit HCC metastasis [10, 11].
Yanggan Jiedu Sanjie (YGJDSJ) is a modern Chinese herbal formula developed by us for the treatment of HCC [12]. YGJDSJ composed of multiple herbs. Some herbs in YGJDSJ have demonstrated inhibitory effects on EMT in cancer cells. Solanum nigrum L. (Long-Kui) and its ingredient α-Solanine, polysaccharide of Scutellaria barbata D. Don (Ban-Zhi-Lian), Duchesnea indica (Andr.) Focke (She-Mei), and β-elemene (a compound of Curcuma wenyujin Y.H. Chen et C. Ling (Yü-Jin)) can inhibit EMT in various cancer cells [13–17]. These findings suggested that YGJDSJ may also have a similar effect on EMT. In this study, we evaluated the effect of YGJDSJ on transforming growth factor-β1 (TGF-β1)-induced EMT in human HCC Bel-7402 cells.
Materials and methods
Chemicals and reagents
DMEM medium and fetal bovine serum were purchased from Thermo Fisher Scientific (Waltham, MA). TGF-β1 was obtained from PeproTech (Rocky Hill, NJ). Antibodies against Smad3, p-Smad3 (Ser423/425), Snail and GAPDH were from Cell Signaling Technology (Danvers, MA). E-cadherin and N-cadherin antibodies were bought from Santa Cruz Biotechnology (Santa Cruz, CA). CytoSelect™ 48-Well Cell Adhesion Assay and CytoSelect™ 24-Well Cell Invasion Assay kits were produced by Cell Biolabs (San Diego, CA).
YGJDSJ extraction
The herbs in YGJDSJ formula (Chinese patent No. ZL201110145109.0) are the fruits of Ligustrum lucidum Ait. (Nü-zhen-zi) 12 g, D. indica (Andr.) Focke (She-Mei) 15 g, S. nigrum L. (Long-Kui) 15 g, S. barbata D. Don (Ban-Zhi-Lian) 30 g, Euphorbia helioscopia L. (Ze-Qi) 15 g, the root of Ranunculus ternatus Thunb. (Mao-Zhua-Cao) 15 g, the root of C. wenyujin Y. H. Chen et C. Ling (Yü-Jin) 15 g and the root of Polygonum cuspidatum Sieb. et Zucc. (Hu-Zhang) 15 g. All herbs were obtained from the dispensary of Chinese medicine of Longhua Hospital and identified by Professor Liwen Xu from Shanghai University of Traditional Chinese Medicine, Shanghai, China. Voucher specimen is deposited in Institute of Traditional Chinese Medicine in Oncology, Longhua Hospital, Shanghai University of Traditional Chinese Medicine, Shanghai, China (specimen number: TCM-HCC-001). YGJDSJ extraction has been described previously [12]. YGJDSJ extract were dissolved in PBS and stored at − 20 °C until further use.
Cell culture
Bel-7402 cells were obtained from The Cell Bank of Type Culture Collection of Chinese Academy of Sciences (CBTCCCAS) and checked by CBTCCCAS. The cells were cultured in DMEM medium containing 10% FBS and 1% Pen-Strep, and maintained at 37 °C in a humidified atmosphere with 5% CO2.
EMT induction
Bel-7402 cells (5 × 105) in logarithmic growth phase were inoculated in 6-well plates and cultured in serum free DMEM and allowed to attach for 24 h before treatment. The cells were then treated with TGF-β1 (10 ng/mL) and YGJDSJ (100 μg/mL) or same volume of PBS for 48 h. The morphology of the cells was observed under a microscope.
Scratch / migration assay
Cell migration was measured by the scratch assay [18, 19]. Bel-7402 cells (1 × 106) were incubated in 6-well plates and cultured to 95% confluency. Then the cells were scratched by a sterile pipette tip, and washed three times with PBS. Fresh medium was added and the cells were treated with TGF-β1 (10 ng/mL) and YGJDSJ (100 μg/mL) or equal volume of PBS for 48 h. The cell migration was observed by microscopy.
Cell adhesion assay
Cell adhesion was detected by a commercial kit according to the manufacturer’s manual. Briefly, 1 × 105 TGF-β1 and YGJDSJ treated or untreated Bel-7402 cells were added to a 48 well plate. TGF-β1 (10 ng/mL), YGJDSJ (100 μg/mL) or same volume of PBS was also added to the wells. Cells were incubated for 90 min at 37 °C and stained with staining solution for 10 min at room temperature. After aspirating the staining solution, the plate was gently washed three times with 500 μl deionized water and air dried. 200 μL of extraction solution was then added to the wells and incubated for 10 min. The optical density of each well was measured at OD 560 nm by a plate reader.
Cell invasion assay
Cell invasion was detected by a commercial kit according to the manufacturer’s protocol. Briefly, 3 × 105 TGF-β1 and YGJDSJ treated or untreated Bel-7402 cells were added to the inner side of cell insert, and 500 μL of DMEM media with 10% FBS was added to the lower well of the invasion plate. TGF-β1 (10 ng/mL), YGJDSJ (100 μg/mL) or same volume of PBS was also added to the wells. The cells were incubated for 12 h at 37 °C. After removing the non-invasive cells, the inserts were stained with staining solution for 10 min at room temperature, and observed under a microscope. 200 μL of extraction solution was then added to the wells and incubated for 10 min. The optical density of each well was measured at OD 560 nm by a plate reader.
Western blot
Western blotting was performed as previously described [20, 21]. Briefly, the cells were lysed and subjected to 8–12% SDS-PAGE electrophoresis, and then transferred onto a nitrocellulose membrane (Bio-Rad, Richmond, CA). The membrane was blocked with 5% non-fat milk, washed, and probed with antibodies against E-cadherin (1:200), N-cadherin (1:200), Smad3 (1:1000), p-Smad3 (1:1000), Snail (1:1000) or GAPDH (1:2000) at 4 °C overnight. The blots were then washed and incubated with secondary antibodies (1:4000), developed by ECL substrates and exposed by ChemiDoc™ Touch Imaging System (Bio-Rad, Hercules, CA). Proteins expression was quantified by the Quantity One software (Bio-Rad, Hercules, CA).
Statistical analyses
Results are expressed as means ± standard deviation of at least two independent experiments. Differences between different treatment groups were analyzed by one-way ANOVA. Differences were considered significant at p-values < 0.05.
Results
YGJDSJ reversed TGF-β1-induced morphological changes
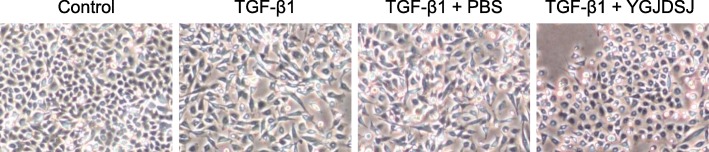
After TGF-β1 treatment, the morphology of Bel-7402 cells changed from round or oval to spindle shaped. The distribution of the cells was looser and they exhibited mesenchymal morphology. After YGJDSJ treatment, Bel-7402 cells were reverted to epithelial morphology, which was round or oval (Fig. 1).
Fig. 1.
Effects of YGJDSJ on TGF-β1-induced morphological changes. Bel-7402 cells were treated with TGF-β1 and YGJDSJ or PBS for 48 h, and cell morphology was observed under a microscope (× 200)
YGJDSJ reversed TGF-β1-induced expression change of EMT marker proteins
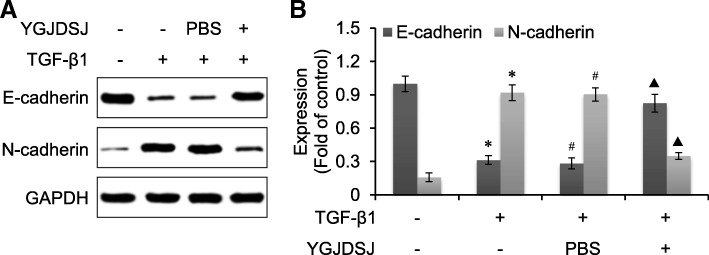
Western blot analysis showed that the expression of epithelial marker gene E-cadherin was down-regulated, while the mesenchymal marker gene N-cadherin was up-regulated after TGF-β1 treatment. YGJDSJ increased E-cadherin expression and inhibited N-cadherin expression (p < 0.01) (Fig. 2). These observations suggested that YGJDSJ inhibited TGF-β1-induced EMT in Bel-7402 cells.
Fig. 2.
Effects of YGJDSJ on TGF-β1-induced expression change of EMT marker proteins. a, Bel-7402 cells were treated with TGF-β1 and YGJDSJ or PBS for 48 h, and subjected to western blotting using indicated antibodies. b, protein expression was quantified by the Quantity One software. *p < 0.01, versus control group; #p > 0.05, versus TGF-β1 group; ▲p < 0.01, versus PBS group
YGJDSJ inhibited TGF-β1-induced Smad3 phosphorylation
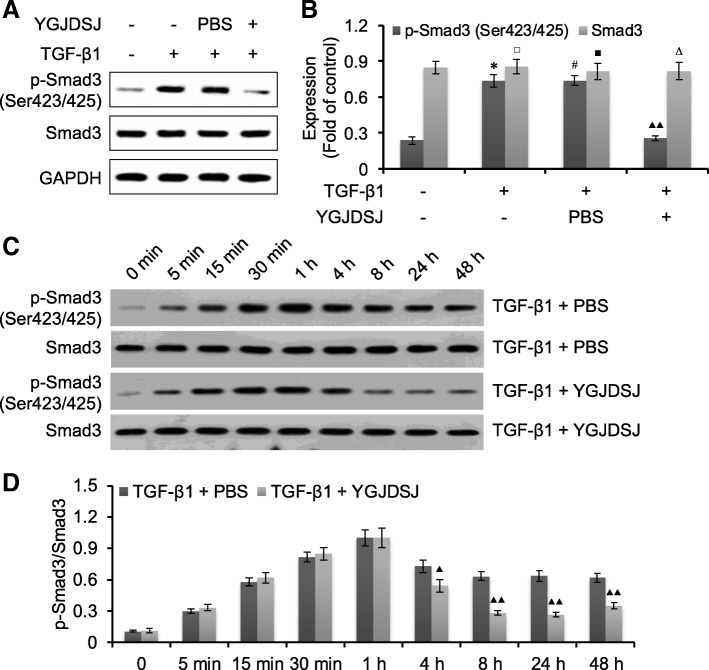
It has been reported that Smad3 is involved in TGF-β1 induced EMT [22, 23]. We observed Smad3 expression and phosphorylation by western blot. The results showed that the expression of Smad3 did not change significantly; but the phosphorylation levels of Smad3 were increased after TGF-β1 treatment. YGJDSJ could inhibit TGF-β1-induced phosphorylation of Smad3 (p < 0.01) (Fig. 3).
Fig. 3.
Effects of YGJDSJ on TGF-β1-induced Smad3 phosphorylation. a, Bel-7402 cells were treated with TGF-β1 and YGJDSJ or PBS for 48 h, and subjected to western blotting using indicated antibodies. c, Bel-7402 cells were treated with TGF-β1 and YGJDSJ or PBS for indicated times, and subjected to western blotting using Samd3 and p-Smad3. b and d, protein expression was quantified by the Quantity One software. *p < 0.01, versus control group; #p > 0.05, versus TGF-β1 group; ▲p < 0.05, versus PBS group; ▲▲p < 0.01, versus PBS group; □p > 0.05, versus control group; ■p > 0.05, versus TGF-β1 group; ∆p > 0.05, versus PBS group
YGJDSJ inhibited TGF-β1-induced Snail expression
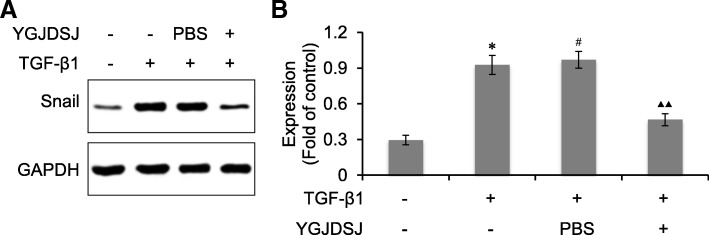
Snail is an important transcriptional regulator of EMT that can be up-regulated by TGF-β1 [24, 25]. We also detected the expression of Snail. The results showed that the expression of Snail in Bel-7402 cells was up-regulated by TGF-β1. YGJDSJ could inhibit the expression of Snail induced by TGF-β1 (p < 0.01) (Fig. 4).
Fig. 4.
Effects of YGJDSJ on TGF-β1-induced Snail expression. a, Bel-7402 cells were treated with TGF-β1 and YGJDSJ or PBS for 48 h, and subjected to western blotting using indicated antibodies. b, protein expression was quantified by the Quantity One software. *p < 0.01, versus control group; #p > 0.05, versus TGF-β1 group; ▲▲p < 0.01, versus PBS group
YGJDSJ inhibited TGF-β1-induced cell adhesion
Cell adhesion is an important biological process in tumor metastasis, and EMT can promote tumor cell adhesion [26, 27]. The results showed that the adhesion ability of Bel-7402 cells was enhanced by TGF-β1. YGJDSJ could inhibit TGF-β1-induced cell adhesion in Bel-7402 cells (p < 0.01) (Fig. 5).
Fig. 5.
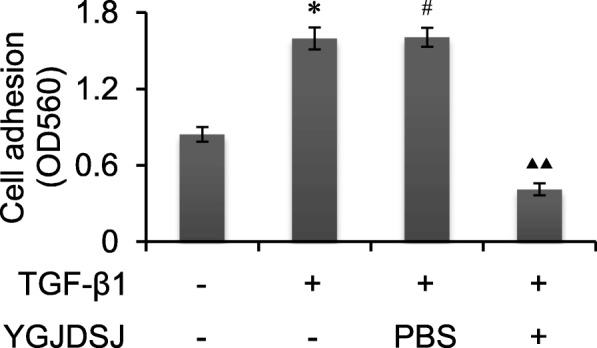
Effects of YGJDSJ on TGF-β1-induced cell adhesion. Bel-7402 cells were treated with TGF-β1 and YGJDSJ or PBS for 48 h, and subjected to cell adhesion assay using a commercial kit according to the manufacturer’s manual. *p < 0.01, versus control group; #p > 0.05, versus TGF-β1 group; ▲▲p < 0.01, versus PBS group
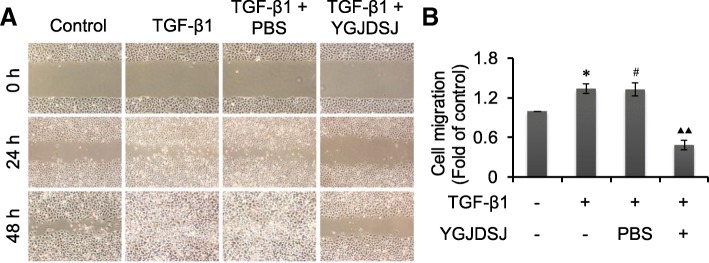
YGJDSJ inhibited TGF-β1-induced cell migration
Scratch assay is a simple and convenient method to detect cell migration [18, 19]. Results from scratch assay showed that the migration ability of Bel-7402 cells was enhanced by TGF-β1; but YGJDSJ inhibited the cell migration induced by TGF-β1 (Fig. 6).
Fig. 6.
Effects of YGJDSJ on TGF-β1-induced cell migration. a, Bel-7402 cells were scratched by a sterile pipette tip and treated with TGF-β1 and YGJDSJ or PBS for 48 h, cell migration was observed under a microscope (× 40). b, Cell migration distance at 24 h were measured by electronic ruler and expressed as fold of control. *p < 0.01, versus control group; #p > 0.05, versus TGF-β1 group; ▲▲p < 0.01, versus PBS group
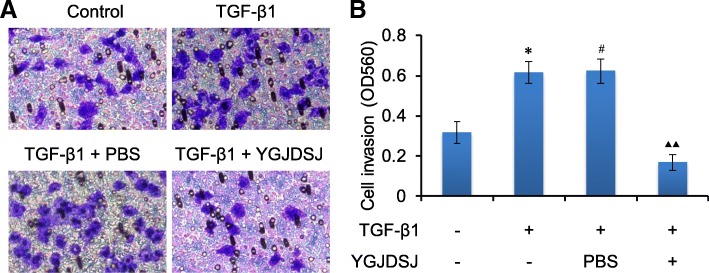
YGJDSJ inhibited TGF-β1-induced cell invasion
Cell invasion is an important process in tumor metastasis, and EMT can promote the invasion of tumor cells [28, 29]. Transwell assay showed that the invasion ability of Bel-7402 cells was enhanced by TGF-β1. YGJDSJ could inhibit the invasion of Bel-7402 cells induced by TGF-β1 (p < 0.01) (Fig. 7).
Fig. 7.
Effects of YGJDSJ on TGF-β1-induced cell invasion. Bel-7402 cells were treated with TGF-β1 and YGJDSJ or PBS for 48 h, and subjected to cell invasion assay using a commercial kit according to the manufacturer’s protocol. Cell invasion was observed under a microscope (× 200) (a) and measured at OD 560 nm by a plate reader (b). *p < 0.01, versus control group; #p > 0.05, versus TGF-β1 group; ▲▲p < 0.01, versus PBS group
Discussion
YGJDSJ is composed of several Chinese herbs with anticancer effects. The fruits of L. lucidum Ait. (Nü-Zhen-Zi) inhibits proliferation, as well as promotes apoptosis and senescence in HCC cells [30]. S. barbata D. Don (Ban-Zhi-Lian) promotes apoptosis of HCC cells via mitochondrial pathway [31]. S. nigrum L. inhibits proliferation, induces apoptosis, and arrests cell cycle at G2/M phase in HCC cells [32]. D. indica (Andr.) Focke (She-Mei) inhibits proliferation, induces apoptosis, and arrests cell cycle in cervical cancer cells [33].
E. helioscopia L. (Ze-Qi) inhibits tumor growth, induces apoptosis, and inhibits metastasis in HCC [34]. R. ternatus Thunb. (Mao-Zhao-Cao) inhibits proliferation and tumor growth in HCC [35, 36]. C. wenyujin Y.H. Chen et C. Ling (Yü-Jin) has a wide range of anticancer effects, its main components include β-elemene and curcumin [37–39]. P. cuspidatum Sieb. et Zucc (Hu-Zhang) exhibits anticancer effect on various tumors and resveratrol is one of its important ingredients [39, 40]. Thus, YGJDSJ is a modern anticancer herbal formula.
Some herbs or compounds in YGJDSJ have showed inhibitory effects on EMT. S. barbata (Ban-Zhi-Lian) polysaccharide inhibits EMT in colon cancer cells [15]. D. indica (She-Mei) extract inhibits migration and EMT in lung adenocarcinoma cells [16]. S. nigrum (Long-Kui) suppresses EMT in breast cancer cells [13]. β-elemene, a compound from C. wenyujin Y.H. Chen et C. Ling (Yü-Jin), inhibits EMT in glioblastoma and breast cancer cells [17, 41]. Resveratrol, a component of P. cuspidatum Sieb. et Zucc. (Hu-Zhang), inhibits EMT in multiple cancer cells [42–44].
TGF-β1 is a cytokine that is associated with multiple bioprocess, such as cell proliferation, differentiation, death and EMT, and is frequently used for EMT induction [8, 22, 23, 27, 42]. In present study, we observed that upon TGF-β1 treatment, the morphology of Bel-7402 cells changed from oval epithelial morphology to spindle mesenchymal morphology with down-regulation of E-cadherin and up-regulation of N-cadherin, indicating the occurrence of EMT. On the other hand, YGJDSJ reversed TGF-β1-induced morphological changes, as well as the expression of the EMT markers E-cadherin and N-cadherin in Bel-7402 cells, suggesting that YGJDSJ could inhibit EMT induced by TGF-β1.
TGF-β1 binding to its receptor could recruit and phosphorylate Smads, and up-regulate Snail, thus leading to EMT [8, 45–47]. Snail is an EMT-related transcriptional repressor, which regulate the transcription of target genes, such as E-cadherin, and is related to cancer metastasis [24, 25]. Snail is required for TGF-β1-induced EMT [48, 49]. Natural products, such as Jianpi Huayu Decoction, Nobiletin and Resveratrol, could inhibit EMT by inhibiting TGF-β1/Smad signaling and Snail [50–52]. Our results showed that Smad3 phosphorylation and Snail expression were increased in Bel-7402 cells by TGF-β1, suggesting that EMT induced by TGF-β1 was associated with Smad3 and Snail. YGJDSJ could inhibit TGF-β1-induced EMT, and decrease Smad3 phosphorylation and Snail expression, suggesting that Smad3 and Snail were involved in the effect of YGJDSJ on EMT.
EMT causes cancer cells to acquire mesenchymal phenotype, enhance their adhesion, migration and invasion capacities, thus promoting tumor metastasis [27–29, 53]. The present study showed that TGF-β1-induced EMT could enhance cell adhesion, migration and invasion of Bel-7402 cells. YGJDSJ inhibited cell adhesion, migration and invasion induced by TGF-β1, suggesting that YGJDSJ can inhibit metastatic potential of Bel-7402 cells by inhibiting EMT.
Conclusions
In conclusion, YGJDSJ inhibits TGF-β1-induced EMT and cell adhesion, migration and invasion in Bel-7402 cells, which is related to down-regulation of Smad3 phosphorylation and Snail expression. The present study provides a new basis for the application of YGJDSJ for the prevention and treatment of HCC metastasis, which is worthy of further study.
Acknowledgements
Not applicable.
Ethic approval and consent to participate
Not Applicable.
Funding
This study was supported by Program from Science & Technology Commission of Shanghai Municipality (09JC1413600 and 16401902500).
Availability of data and materials
The datasets used and/or analyzed during the current study available from the corresponding author on reasonable request.
Abbreviations
- ANOVA
Analysis of variance
- CBTCCCAS
The Cell Bank of Type Culture Collection of Chinese Academy of Sciences
- DMEM
Dulbecco’s modified Eagle’s medium
- ECL
Enhanced chemiluminence
- EMT
Epithelial-mesenchymal transition
- FBS
Fetal bovine serum
- GAPDH
Glyceraldehyde-3-phosphate dehydrogenase
- HCC
Hepatocellular carcinoma
- PBS
Phosphate-buffered saline
- SDS-PAGE
Sodium dodecyl sulfate-polyacrylamide gel electrophoresis
- TGF-β1
Transforming growth factor-β1
- YGJDSJ
Yanggan Jiedu Sanjie herbal formula
Authors’ contributions
BH conceived and designed the study. BH, HMA, XY, JLZ, XWH and ML carried out the experiment. HMA analyzed the data. BH drafted and revised the manuscript. All authors read and approved the final manuscript.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Bing Hu, Email: beearhu@hotmail.com.
Hong-Mei An, Email: lhsoatp@163.com.
Xia Yan, Email: gracemihe@163.com.
Jia-Lu Zheng, Email: zhengjialu19851206@126.com.
Xiao-Wei Huang, Email: 616278620@qq.com.
Miao Li, Email: elviraphoenix@hotmail.com.
References
- 1.Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018 Sep 12. 10.3322/caac.21492. [DOI] [PubMed]
- 2.Hartke J, Johnson M, Ghabril M. The diagnosis and treatment of hepatocellular carcinoma. Semin Diagn Pathol. 2017;34(2):153–159. doi: 10.1053/j.semdp.2016.12.011. [DOI] [PubMed] [Google Scholar]
- 3.Testino G, Leone S, Patussi V, Scafato E, Borro P. Hepatocellular carcinoma: diagnosis and proposal of treatment. Minerva Med. 2016;107(6):413–426. [PubMed] [Google Scholar]
- 4.Uchino K, Tateishi R, Shiina S, Kanda M, Masuzaki R, Kondo Y, et al. Hepatocellular carcinoma with extrahepatic metastasis: clinical features and prognostic factors. Cancer. 2011;117(19):4475–4483. doi: 10.1002/cncr.25960. [DOI] [PubMed] [Google Scholar]
- 5.Natsuizaka M, Omura T, Akaike T, Kuwata Y, Yamazaki K, Sato T, et al. Clinical features of hepatocellular carcinoma with extrahepatic metastases. J Gastroenterol Hepatol. 2005;20(11):1781–1787. doi: 10.1111/j.1440-1746.2005.03919.x. [DOI] [PubMed] [Google Scholar]
- 6.Desai JR, Ochoa S, Prins PA, He AR. Systemic therapy for advanced hepatocellular carcinoma: an update. J Gastrointest Oncol. 2017;8(2):243–255. doi: 10.21037/jgo.2017.02.01. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Dutta R, Mahato RI. Recent advances in hepatocellular carcinoma therapy. Pharmacol Ther. 2017;173:106–117. doi: 10.1016/j.pharmthera.2017.02.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Reichl P, Haider C, Grubinger M, Mikulits W. TGF-β in epithelial to mesenchymal transition and metastasis of liver carcinoma. Curr Pharm Des. 2012;18(27):4135–4147. doi: 10.2174/138161212802430477. [DOI] [PubMed] [Google Scholar]
- 9.Giannelli G, Koudelkova P, Dituri F, Mikulits W. Role of epithelial to mesenchymal transition in hepatocellular carcinoma. J Hepatol. 2016;65(4):798–808. doi: 10.1016/j.jhep.2016.05.007. [DOI] [PubMed] [Google Scholar]
- 10.Cao J, Liu J, Long J, Fu J, Huang L, Li J, et al. microRNA-23b suppresses epithelial-mesenchymal transition (EMT) and metastasis in hepatocellular carcinoma via targeting Pyk2. Biomed Pharmacother. 2017;89:642–650. doi: 10.1016/j.biopha.2017.02.030. [DOI] [PubMed] [Google Scholar]
- 11.Qin CD, Ma DN, Ren ZG, Zhu XD, Wang CH, Wang YC, et al. Astragaloside IV inhibits metastasis in hepatoma cells through the suppression of epithelial-mesenchymal transition via the Akt/GSK-3β/β-catenin pathway. Oncol Rep. 2017;37(3):1725–1735. doi: 10.3892/or.2017.5389. [DOI] [PubMed] [Google Scholar]
- 12.Hu B, Zhang T, An HM, Zheng JL, Yan X, Huang XW. Herbal formula YGJDSJ inhibits anchorage-independent growth and induces anoikis in hepatocellular carcinoma Bel-7402 cells. BMC Complement Altern Med. 2018;18(1):17. doi: 10.1186/s12906-018-2083-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Lai YJ, Tai CJ, Wang CW, Choong CY, Lee BH, Shi YC, et al. Anti-cancer activity of Solanum nigrum (AESN) through suppression of mitochondrial function and epithelial-mesenchymal transition (EMT) in breast cancer cells. Molecules. 2016;21(5):553. doi: 10.3390/molecules21050553. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Shen KH, Liao AC, Hung JH, Lee WJ, Hu KC, Lin PT, et al. α-Solanine inhibits invasion of human prostate cancer cell by suppressing epithelial-mesenchymal transition and MMPs expression. Molecules. 2014;19(8):11896–11914. doi: 10.3390/molecules190811896. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Sun P, Sun D, Wang X. Effects of Scutellaria barbata polysaccharide on the proliferation, apoptosis and EMT of human colon cancer HT29 cells. Carbohydr Polym. 2017;167:90–96. doi: 10.1016/j.carbpol.2017.03.022. [DOI] [PubMed] [Google Scholar]
- 16.Chen PN, Yang SF, Yu CC, Lin CY, Huang SH, Chu SC, et al. Duchesnea indica extract suppresses the migration of human lung adenocarcinoma cells by inhibiting epithelial-mesenchymal transition. Environ Toxicol. 2017;2(8):2053–2063. doi: 10.1002/tox.22420. [DOI] [PubMed] [Google Scholar]
- 17.Zhu T, Li X, Luo L, Wang X, Li Z, Xie P, et al. Reversion of malignant phenotypes of human glioblastoma cells by β-elemene through β-catenin-mediated regulation of stemness-, differentiation- and epithelial-to-mesenchymal transition-related molecules. J Transl Med. 2015;13:356. doi: 10.1186/s12967-015-0727-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Yarrow JC, Perlman ZE, Westwood NJ, Mitchison TJ. A high-throughput cell migration assay using scratch wound healing, a comparison of image-based readout methods. BMC Biotechnol. 2004;4:21. doi: 10.1186/1472-6750-4-21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Liang CC, Park AY, Guan JL. In vitro scratch assay: a convenient and inexpensive method for analysis of cell migration in vitro. Nat Protoc. 2007;2(2):329–333. doi: 10.1038/nprot.2007.30. [DOI] [PubMed] [Google Scholar]
- 20.Hu B, An HM, Shen KP, Song HY, Deng S. Polygonum cuspidatum extract induces anoikis in hepatocarcinoma cells associated with generation of reactive oxygen species and downregulation of focal adhesion kinase. Evid Based Complement Alternat Med. 2012;2012:607675. doi: 10.1155/2012/607675. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.An HM, Xue YF, Shen YL, Du Q, Hu B. Sodium valproate induces cell senescence in human hepatocarcinoma cells. Molecules. 2013;18(12):14935–14947. doi: 10.3390/molecules181214935. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Boldbaatar A, Lee S, Han S, Jeong AL, Ka HI, Buyanravjikh S, et al. Eupatolide inhibits the TGF-β1-induced migration of breast cancer cells via downregulation of SMAD3 phosphorylation and transcriptional repression of ALK5. Oncol Lett 2017;14(5):6031–6039. [DOI] [PMC free article] [PubMed]
- 23.Lou C, Zhang F, Yang M, Zhao J, Zeng W, Fang X, et al. Naringenin decreases invasiveness and metastasis by inhibiting TGF-β-induced epithelial to mesenchymal transition in pancreatic cancer cells. PLoS One. 2012;7(12):e50956. doi: 10.1371/journal.pone.0050956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Nishioka R, Itoh S, Gui T, Gai Z, Oikawa K, Kawai M, et al. SNAIL induces epithelial-to-mesenchymal transition in a human pancreatic cancer cell line (BxPC3) and promotes distant metastasis and invasiveness in vivo. Exp Mol Pathol. 2010;89(2):149–157. doi: 10.1016/j.yexmp.2010.05.008. [DOI] [PubMed] [Google Scholar]
- 25.Wang Y, Shi J, Chai K, Ying X, Zhou BP. The role of Snail in EMT and tumorigenesis. Curr Cancer Drug Targets. 2013;13(9):963–972. doi: 10.2174/15680096113136660102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Niit M, Hoskin V, Carefoot E, Geletu M, Arulanandam R, Elliott B, et al. Cell-cell and cell-matrix adhesion in survival and metastasis: Stat3 versus Akt. Biomol Concepts. 2015;6(5–6):383–399. doi: 10.1515/bmc-2015-0022. [DOI] [PubMed] [Google Scholar]
- 27.Xu Z, Shen MX, Ma DZ, Wang LY, Zha XL. TGF-beta1-promoted epithelial-to-mesenchymal transformation and cell adhesion contribute to TGF-beta1-enhanced cell migration in SMMC-7721 cells. Cell Res. 2003;13(5):343–350. doi: 10.1038/sj.cr.7290179. [DOI] [PubMed] [Google Scholar]
- 28.Gavert N, Ben-Ze'ev A. Epithelial-mesenchymal transition and the invasive potential of tumors. Trends Mol Med. 2008;14(5):199–209. doi: 10.1016/j.molmed.2008.03.004. [DOI] [PubMed] [Google Scholar]
- 29.Ding W, You H, Dang H, LeBlanc F, Galicia V, Lu SC, et al. Epithelial-to-mesenchymal transition of murine liver tumor cells promotes invasion. Hepatology. 2010;52(3):945–953. doi: 10.1002/hep.23748. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Hu B, Du Q, Deng S, An HM, Pan CF, Shen KP, et al. Ligustrum lucidum Ait. Fruit extract induces apoptosis and cell senescence in human hepatocellular carcinoma cells through upregulation of p21. Oncol Rep. 2014;32(3):1037–1042. doi: 10.3892/or.2014.3312. [DOI] [PubMed] [Google Scholar]
- 31.Dai ZJ, Wang XJ, Li ZF, Ji ZZ, Ren HT, Tang W, et al. Scutellaria barbate extract induces apoptosis of hepatoma H22 cells via the mitochondrial pathway involving caspase-3. World J Gastroenterol. 2008;14(48):7321–7328. doi: 10.3748/wjg.14.7321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Wang HC, Chung PJ, Wu CH, Lan KP, Yang MY, Wang CJ. Solanum nigrum L. polyphenolic extract inhibits hepatocarcinoma cell growth by inducing G2/M phase arrest and apoptosis. J Sci Food Agric. 2011;91(1):178–185. doi: 10.1002/jsfa.4170. [DOI] [PubMed] [Google Scholar]
- 33.Peng B, Hu Q, Liu X, Wang L, Chang Q, Li J, et al. Duchesnea phenolic fraction inhibits in vitro and in vivo growth of cervical cancer through induction of apoptosis and cell cycle arrest. Exp Biol Med (Maywood) 2009;234(1):74–83. doi: 10.3181/0806-RM-204. [DOI] [PubMed] [Google Scholar]
- 34.Cheng J, Han W, Wang Z, Shao Y, Wang Y, Zhang Y, et al. Hepatocellular carcinoma growth is inhibited by Euphorbia helioscopia L. extract in nude mice xenografts. Biomed Res Int. 2015;2015:601015. doi: 10.1155/2015/601015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Wang AW, Yuan H, Sun PY, Yuan JR, Geng H. Antitumor effect of different extracts from Radix Ranunculus ternate in H22 hepatoma. Chin J New Drugs. 2006;15(12):971–974. [Google Scholar]
- 36.Chen X, Tong Y, Yang F, Dai G, Ren Z, Wang B. The study on effects of Radix Ranunculus ternate saponins on the activity of human hepatocarcinoma cell line HepG2. Shandong Med J. 2013;51(10):3–5. [Google Scholar]
- 37.Jiang S, Ling C, Li W, Jiang H, Zhi Q, Jiang M. Molecular mechanisms of anti-cancer activities of β-elemene: targeting hallmarks of cancer. Anti Cancer Agents Med Chem. 2016;16(11):1426–1434. doi: 10.2174/1871520616666160211123424. [DOI] [PubMed] [Google Scholar]
- 38.Kasi PD, Tamilselvam R, Skalicka-Woźniak K, Nabavi SF, Daglia M, Bishayee A, et al. Molecular targets of curcumin for cancer therapy: an updated review. Tumour Biol. 2016;37(10):13017–13028. doi: 10.1007/s13277-016-5183-y. [DOI] [PubMed] [Google Scholar]
- 39.Du Q, Hu B, An HM, Shen KP, Xu L, Deng S, et al. Synergistic anticancer effects of curcumin and resveratrol in Hepa1-6 hepatocellular carcinoma cells. Oncol Rep. 2013;29(5):1851–1858. doi: 10.3892/or.2013.2310. [DOI] [PubMed] [Google Scholar]
- 40.Peng W, Qin R, Li X, Zhou H. Botany, phytochemistry, pharmacology, and potential application of Polygonum cuspidatum sieb.Et Zucc.: a review. J Ethnopharmacol. 2013;148(3):729–745. doi: 10.1016/j.jep.2013.05.007. [DOI] [PubMed] [Google Scholar]
- 41.Zhang X, Li Y, Zhang Y, Song J, Wang Q. Zheng L,et al. Beta-elemene blocks epithelial-mesenchymal transition in human breast cancer cell line MCF-7 through Smad3-mediated down-regulation of nuclear transcription factors. PLoS One. 2013;8(3):e58719. doi: 10.1371/journal.pone.0058719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Wang H, Zhang H, Tang L, Chen H, Wu C, Zhao M, et al. Resveratrol inhibits TGF-β1-induced epithelial-to-mesenchymal transition and suppresses lung cancer invasion and metastasis. Toxicology. 2013;303:139–146. doi: 10.1016/j.tox.2012.09.017. [DOI] [PubMed] [Google Scholar]
- 43.Baribeau S, Chaudhry P, Parent S, Asselin É. Resveratrol inhibits cisplatin-induced epithelial-to-mesenchymal transition in ovarian cancer cell lines. PLoS One. 2014;9(1):e86987. doi: 10.1371/journal.pone.0086987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Sun Y, Wang H, Liu M, Lin F, Hua J. Resveratrol abrogates the effects of hypoxia on cell proliferation, invasion and EMT in osteosarcoma cells through downregulation of the HIF-1α protein. Mol Med Rep. 2015;11(3):1975–1981. doi: 10.3892/mmr.2014.2913. [DOI] [PubMed] [Google Scholar]
- 45.Zhou SJ, Liu FY, Zhang AH, Liang HF, Wang Y, Ma R, et al. MicroRNA-199b-5p attenuates TGF-β1-induced epithelial-mesenchymal transition in hepatocellular carcinoma. Br J Cancer. 2017;117(2):233–244. doi: 10.1038/bjc.2017.164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Macias MJ, Martin-Malpartida P, Massagué J. Structural determinants of Smad function in TGF-β signaling. Trends Biochem Sci. 2015;40(6):296–308. doi: 10.1016/j.tibs.2015.03.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Aomatsu K, Arao T, Sugioka K, Matsumoto K, Tamura D, Kudo K, et al. TGF-β induces sustained upregulation of SNAI1 and SNAI2 through Smad and non-Smad pathways in a human corneal epithelial cell line. Invest Ophthalmol Vis Sci. 2011;52(5):2437–2443. doi: 10.1167/iovs.10-5635. [DOI] [PubMed] [Google Scholar]
- 48.Cho HJ, Baek KE, Saika S, Jeong MJ, Yoo J. Snail is required for transforming growth factor-beta-induced epithelial-mesenchymal transition by activating PI3 kinase/Akt signal pathway. Biochem Biophys Res Commun. 2007;353(2):337–343. doi: 10.1016/j.bbrc.2006.12.035. [DOI] [PubMed] [Google Scholar]
- 49.Li H, Wang H, Wang F, Gu Q, Xu X. Snail involves in the transforming growth factor β1-mediated epithelial-mesenchymal transition of retinal pigment epithelial cells. PLoS One. 2011;6(8):e23322. doi: 10.1371/journal.pone.0023322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Zhong C, Zhang YF, Huang JH, Wang ZY, Chen QY, Su LT, et al. The Chinese medicine, Jianpi Huayu decoction, inhibits the epithelial mesenchymal transition via the regulation of the Smad3/Smad7 cascade. Am J Transl Res. 2017;9(6):2694–2711. [PMC free article] [PubMed] [Google Scholar]
- 51.Ji Q, Liu X, Han Z, Zhou L, Sui H, Yan L, et al. Resveratrol suppresses epithelial-to-mesenchymal transition in colorectal cancer through TGF-β1/Smads signaling pathway mediated Snail/E-cadherin expression. BMC Cancer. 2015;15:97. doi: 10.1186/s12885-015-1119-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Da C, Liu Y, Zhan Y, Liu K, Wang R. Nobiletin inhibits epithelial-mesenchymal transition of human non-small cell lung cancer cells by antagonizing the TGF-β1/Smad3 signaling pathway. Oncol Rep. 2016;35(5):2767–2774. doi: 10.3892/or.2016.4661. [DOI] [PubMed] [Google Scholar]
- 53.Yeung KT, Yang J. Epithelial-mesenchymal transition in tumor metastasis. Mol Oncol. 2017;11(1):28–39. doi: 10.1002/1878-0261.12017. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and/or analyzed during the current study available from the corresponding author on reasonable request.