Abstract
Background
Only 5-fluorouracil (5-FU), cisplatin, and gemcitabine have been reimbursed for metastatic pancreatic cancer (mPC) treatment in Taiwan since 2003. It is uncertain whether the reimbursement of S-1 in June 2014 might change the treatment pattern and improve the survival of mPC patients in Taiwan.
Patients and methods
A total of 645 patients with newly diagnosed mPC who received palliative chemotherapy between 2010 and 2016 in Taiwan were analyzed retrospectively. Patients were stratified according to year at diagnosis of mPC for analysis of chemotherapeutic treatment pattern and survival.
Results
Overall, the most common chemotherapeutic agents used for the treatment of mPC were gemcitabine (94.8%), followed by cisplatin (52.4%), S-1 (38.1%), and 5-FU (29.7%). The percentage of patients treated with S-1 between 2010 and 2016 increased from 2.6% to 74.0% (P<0.001), while the percentage of patients treated with 5-FU decreased from 31.6% to 21.2% (P<0.001). The percentage of patients treated with gemcitabine, cisplatin, etc. remained consistent. An increase in the number of lines of treatment was observed throughout the study period, with 27.6% of patients receiving two or more lines of treatment in 2010, compared with 50.0% of patients in 2016 (P=0.013). The 12-month survival rate increased from 11.8% in 2010 to 41.4% in 2016, corresponding to an adjusted average annual percent change of 13.6% (0.3–28.7, P<0.05).
Conclusion
Based on this multi-institute cohort study in Taiwan, the reimbursement of S-1 changed the clinical practice and is associated with an improvement in survival outcome of mPC patients.
Keywords: palliative chemotherapy, pancreatic cancer, S-1, survival outcome
Introduction
Pancreatic cancer (PC) was the twelfth most common cancer and the seventh leading cause of cancer-related death in the US in 2015.1 In Taiwan, PC is the twelfth most common cancer, with a steady increase in the incidence rate from 3.7 per 100,000 population in 1999 to 5.0 per 100,000 population in 2015, and an increase in mortality rate from 3.5 per 100,000 population in 1999 to 5.3 per 100,000 population in 2015.2 PC is the most lethal malignancy, with a 5-year survival rate of 8.5% in the US1 and 6.7% in Taiwan.2
Systemic chemotherapy is the standard treatment of choice for patients with metastatic PC (mPC). The active chemotherapeutic agents used for mPC treatment include 5-fluorouracil (5-FU),3 gemcitabine,4,5 cisplatin,6 oxaliplatin,7 irinotecan,8 nab-paclitaxel,9 and S-1.10 S-1 is an oral 5-FU derivative which has been widely used in Japan for various types of solid cancer since it was approved in 1999.11 Several phase II studies have showed that S-1 monotherapy is effective in the treatment of unresectable PC;12,13 in addition, combination S-1 with other agents, including gemcitabine,14 oxaliplatin plus irinotecan,15 or nab-paclitaxel,16 demonstrated better antitumor activity in mPC. The Gemcitabine and S-1 Trial (GEST) was a phase III study conducted in Japan and Taiwan to compare the clinical efficacy of S-1 vs gemcitabine vs gemcitabine and S-1 (GS) combination therapy as first-line treatment of unresectable PC.10 Although the result showed an insignificant difference in overall survival among the three treatment groups, the response rate was significantly higher in the S-1 group (21%) and GS group (29.3%) than in the gemcitabine group (13%), and progression-free survival was significantly increased in the GS group (5.7 months) compared with the gemcitabine group (4.1 months). More importantly, S-1 demonstrated a generally well-tolerated toxicity profile compared with gemcitabine in the GEST study.
In Taiwan, 5-FU, cisplatin, and gemcitabine have been widely used in mPC treatment for several decades. After the approval of gemcitabine in 2003, there were no other novel agents reimbursed for mPC in Taiwan until June 2014 when S-1 was reimbursed based on the result of the GEST study.10 Because of the easy administration as oral medication and good toxicity profile, reimbursement of S-1 might significantly change the chemotherapeutic treatment pattern of mPC in clinical practice in Taiwan. However, whether the availability of S-1 might improve the survival outcome of patients with mPC remains unknown. Therefore, this study aims to analyze the evolution of the chemotherapeutic landscape and its association with survival outcome in patients with mPC before and after S-1 reimbursement in Taiwan.
Patients and methods
Patient selection
A total of 645 consecutive patients with newly diagnosed mPC who received palliative chemotherapy between 2010 and 2016 at four institutes of Chang Gung Memorial Hospital (CGMH) in Taiwan were analyzed retrospectively. All patients had either pathologically or radiographically diagnosed metastatic disease. Patients were excluded from the study if they had other concurrent active malignancies or had previously received adjuvant chemotherapy for resected PC. All patients were categorized according to year at diagnosis of mPC for comparison of the chemotherapeutic treatment pattern and survival outcome. This study was approved by the institutional review boards of all the CGMH branches at November 22, 2017 (ethic code: 201701796B0), and was conducted in compliance with the Declaration of Helsinki (1996). The requirement of informed consent from participants was waived by the IRB because no protected health information was included in this retrospective study.
Data collection
The patient’s demographic data, including age, sex, body mass index, Eastern Cooperative Oncology Group performance status (ECOG PS), smoking history, pre-existing comorbidities by modified Charlson comorbidity index (CCI),17 anatomic location of the primary cancer, clinical T- and N- classification according to the seventh edition of the American Joint Committee on Cancer (AJCC),18 metastatic organ, and chemotherapeutic agents for mPC were recorded by the primary care physician using a prospectively formulated electronic data form from our previous study.19 Distant lymph nodes were defined as those other than the regional lymph nodes according to the AJCC staging system for the exocrine pancreatic cancer.18 The chemotherapeutic agent, dosage, and treatment schedule were determined by the primary care physician. Line of treatment was defined as the number of different chemotherapeutic regimens used in sequence to treat the patient. A break in the original treatment or the adoption of a new chemotherapeutic agent was defined as a different line of treatment. Overall survival (OS) was calculated from the time of initiation of chemotherapy to the date of death from any cause. All included patients were followed until death or December 31, 2017. All dates of death were obtained from either the Institutional Cancer Registry or the National Registry of Death in Taiwan.
Statistical analysis
Basic demographic data were summarized as n (%) for categorical variables and median with range for continuous variables. All patients were stratified by year of diagnosis of mPC for survival comparison. Survival time was analyzed using the Kaplan–Meier method. Log-rank tests were used to determine the significance of differences between the survival curves. Statistically significant changes in the use of chemotherapeutic agents among patients throughout the study period (P for trend) were examined using the Cochran-Armitage test.20 Annual percent change (APC) was calculated by joinpoint analysis to characterize trends in survival rates over time.21 We adjusted for sex, age, CCI, ECOG PS, metastatic organ, smoking history, and Institute of treatment to calculate trends in survival rate between 2010 and 2016. SPSS 17.0 software (SPSS Inc., Chicago, IL, USA) was used for statistical analysis. All statistical assessments were two-sided, and a P-value <0.05 was considered statistically significant.
Results
Basic patient characteristics
The patient characteristics are summarized in Table 1. Of the 654 patients, the median age was 62 years (range=25–88 years), and 60.1% were men. The majority of patients (68.8%) had a good performance status with an ECOG PS of 0–1, and 27.1% of the patients had no comorbidities. Overall, the three most common metastatic sites were the liver (67.0%), peritoneum (36.5%), and distant lymph nodes (22.9%). The most common antitumor agent received by our patient cohort was gemcitabine (94.8%), followed by cisplatin (52.4%), S-1 (38.1%), and 5-FU (29.7%). Less than 3% of our patients had been treated with erlotinib, irinotecan, or nab-paclitaxel for mPC because of a lack of insurance support.
Table 1.
Baseline characteristics of pancreatic cancer patients (n=654)
| Characteristics | Value (%) |
|---|---|
|
| |
| Median age, year (range) | 62 (25–88) |
| Sex | |
| Male | 393 (60.1) |
| Female | 261 (39.9) |
| Body mass index, Kg/m2 (range) | 22.4 (13.0–36.2) |
| Smoking history | 249 (38.1) |
| ECOG performance scale | |
| 0–1 | 450 (68.8) |
| 2 | 174 (26.6) |
| 3 | 30 (4.6) |
| Charlson comorbidity index | |
| 0 | 177 (27.1) |
| 1 | 227 (34.7) |
| 2 | 148 (22.6) |
| 3 | 79 (12.1) |
| 4 | 18 (2.8) |
| 5 | 5 (0.8) |
| Tumor size, cm (range) | 4.5 (1.4–14.0) |
| Primary tumor site | |
| Head | 224 (34.3) |
| Body | 121 (18.5) |
| Tail | 162 (24.8) |
| Overlapping | 147 (22.5) |
| T-classification | |
| 1 | 13 (2) |
| 2 | 82 (12.5) |
| 3 | 188 (28.7) |
| 4 | 371 (56.7) |
| N-classification | |
| 0 | 134 (20.5) |
| 1 | 520 (79.5) |
| Site of metastases | |
| Liver | 438 (67.0) |
| Peritoneum | 239 (36.5) |
| Distant lymph nodes | 150 (22.9) |
| Lung | 98 (15.0) |
| Others | 51 (7.8) |
| Chemotherapy agent | |
| Gemcitabine | 620 (94.8) |
| Cisplatin | 343 (52.4) |
| S-1 | 249 (38.1) |
| 5-FU | 194 (29.7) |
| Irinotecan | 15 (2.3) |
| Erlotinib | 12 (1.8) |
| Nab-paclitaxel | 9 (1.4) |
| Others | 72 (12.8) |
Abbreviation: ECOG, Eastern Cooperative Oncology Group.
Survival outcome
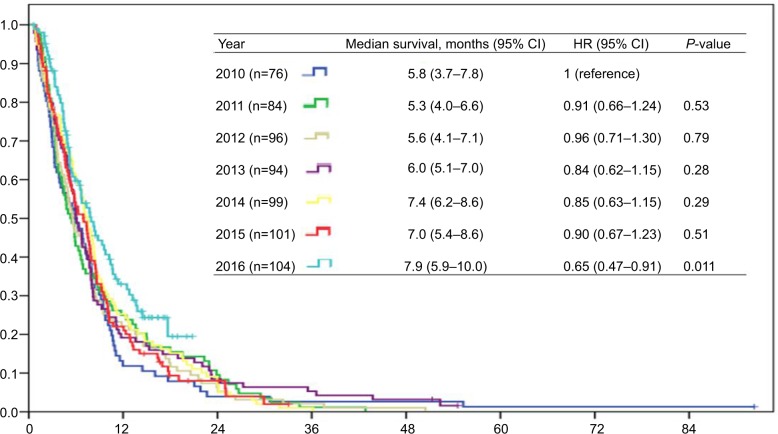
The median survival time in the overall cohort was 6.5 months (range=5.9–7.2), and 607 patients (92.8%) had died by the end of study. The Kaplan–Meier survival time of mPC patients based on the different years of diagnosis is shown in Figure 1. The median survival time was 5.8 months (95% CI=3.7–7.8) among patients diagnosed with mPC in 2010, and increased gradually to 7.9 months (95% CI=5.9–10.0) in 2016. Only patients diagnosed with mPC in 2016 had a statistically significant difference in survival time compared with patients diagnosed with mPC in 2010 (adjusted hazard ratio=0.62, 95% CI=0.44–0.87, P=0.006), while patients diagnosed with mPC in 2011–2015 did not have a significant survival difference compared with patients diagnosed with mPC in 2010 using log-rank tests.
Figure 1.
Survival outcome of patients with metastatic pancreatic cancer according to year of diagnosis.
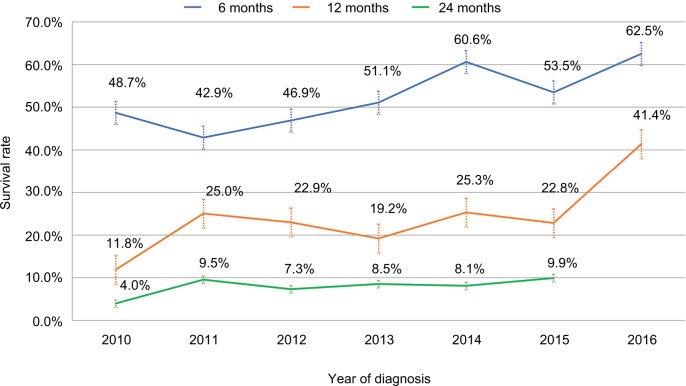
Figure 2 shows observed crude survival rates of mPC patients in 2010–2016. The 6- and 12-month survival rates were 48.7% and 11.8% in 2010, respectively, and increased gradually to 62.5% and 41.4% in 2016, respectively; this corresponded to an adjusted average annual percent change (AAPC) of 5.5% (95% CI=1.2–9.9, P<0.05) for the 6-month survival rate, and 13.6% (95% CI=0.3–28.7, P<0.05) for the 12-month survival rate using joinpoint analysis from 2010 to 2016 (defined as trend 1) (Table 2). Similarly, the 24-month survival rate also increased gradually with an adjusted AAPC of 25.1% (95% CI, 6.1–42.3, P<0.05) from 2010 to 2015. The joinpoint analysis of the trend for the survival rate was further stratified into 2010–2013 (defined as trend 2) and 2013–2016 (defined as trend 3), which represented the time period before and after the introduction of S-1 (June 2014) in Taiwan. Increases of the adjusted AAPC in the 6-, 12-, and 24-month survival rates of the trend 2 and trend 3 study periods were observed; however, the changes in the AAPC did not reach statistical significance.
Figure 2.
Crude 6-, 12-, and 24-month survival rates of pancreatic cancer patients, 2010–2016.
Table 2.
Joinpoint analysis of trend for pancreatic cancer survival rate, 2010–2016
| Survival rate | Trend 1 | Trend 2 | Trend 3 | |||
|---|---|---|---|---|---|---|
|
| ||||||
| Years | Adjusted AAPCa (95% CI) | Years | Adjusted AAPCa (95% CI) | Years | Adjusted AAPCa (95% CI) | |
|
| ||||||
| 6 months | 2010–2016 | 5.5% (1.2–9.9)* | 2010–2013 | 2.6% (−11.9–19.5) | 2013–2016 | 4.8% (−12.2–25.1) |
| 12 months | 2010–2016 | 13.6% (0.3–28.7)* | 2010–2013 | 7.0% (−46.6–114.2) | 2013–2016 | 27.6% (−12.8–86.9) |
| 24 months | 2010–2015 | 25.1% (6.1–42.3)* | 2010–2013 | 12.5% (−44.4–127.8) | 2013–2015 | 5.5% (−2.6–24.6) |
Notes:
Adjusted for sex, age, Charlson comorbidity, ECOG PS, site of metastatic organ, smoking history, and institute of treatment.
P<0.05.
Abbreviations: AAPC, average annual percent change; ECOG PS, European Cooperative Oncology Group Performance Status.
Chemotherapeutic landscape according to year of diagnosis of mPC
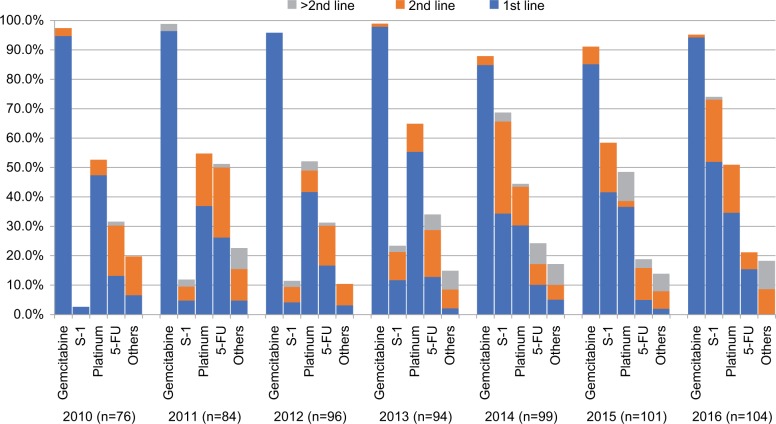
The use of chemotherapeutic agents for patients with mPC according to the year of diagnosis is presented in Figure 3. Gemcitabine was the most commonly used chemotherapy agent and the backbone in the first-line setting throughout the study periods. Cisplatin was the second most common agent used for treating mPC patients in 2010–2013, and it was replaced by S-1 as the second most commonly used agent in 2014–2016. The percentage of patients treated with gemcitabine remained steady from 97.4% to 95.2% between 2010 and 2016 (P for trend=0.40). The percentage of patients treated with S-1 between 2010 and 2016 increased from 2.6% to 74.0% (P for trend<0.001), while the percentage of patients treated with 5-FU decreased from 31.6% to 21.2% (P for trend<0.001) in the same time period. The percentage of patients treated with cisplatin (52.6–51.0%, P for trend=0.16), and the other agents (19.7–18.3%, P for trend=0.38) between 2010 and 2016 remained constant.
Figure 3.
Chemotherapy agents received according to year of diagnosis.
Note: The subtraction of each single agent with different colors, blue, orange, and gray, indicating each used in the first, second, or third and above line of treatment settings, respectively.
The percentage of patients treated with different lines of chemotherapy according to year at diagnosis is shown in Figure S1. An increase in the number of lines of treatment was observed throughout the study period, with 27.6% of patients receiving two or more lines of treatment in 2010, compared with 50.0% of patients in 2016 (P for trend=0.013). We summarized different regimens that patients received as first-line treatment at different periods in Figure S2. Gem-citabine monotherapy or gemcitabine+platinum were the most commonly used regimens to treat mPC patients between 2010 and 2015. Beginning in 2016, they were replaced by gemcitabine+S-1. The number of patients who received gemcitabine+S-1 increased significantly from 2.6% in 2010 to 35.6% in 2016 (P for trend<0.001). At the same time, the number of patients who received gemcitabine+platinum dropped from 40% to 23.1% (P for trend<0.001).
Impact of chemotherapeutic agent on survival outcome
The impact of each chemotherapeutic agent on survival outcome is presented in Table 3. In univariate analysis, OS was significantly better in patients treated with gemcitabine (vs never treated with gemcitabine: median survival=6.7 months vs 3.3 months; HR=0.40; 95% CI=0.28–0.57, P<0.001), treated with S-1 (vs never treated with S-1: median survival=8.6 months vs 5.2 months; HR=0.55; 95% CI=0.46–0.65, P<0.001), treated with cisplatin (vs never treated with cisplatin: median survival=8.2 months vs 4.5 months; HR=0.50; 95% CI=0.43–0.59, P<0.001), treated with 5-FU (vs never treated with 5-FU: median survival=9.0 months vs 5.5 months; HR=0.58; 95% CI=0.49–0.69, P<0.001), and treated with the others agents (vs never treated with the other agents: median survival=10.3 months vs 5.7 months; HR=0.58; 95% CI=0.43–0.75, P<0.001). After adjusting for other variables, being treated with any one of the chemotherapeutic agents, including gemcitabine, S-1, cisplatin, 5-FU, or the other agents, remained as a significant predictive factor for a better survival outcome.
Table 3.
Survival outcome of pancreatic cancer patients according to chemotherapy agents
| Chemotherapy agents | N | Median survival time, months (95% CI) | HR (95% CI) | P-value | Adjusted HRa (95% CI) | P-value | |
|---|---|---|---|---|---|---|---|
|
| |||||||
| Gemcitabine-treated | No | 34 | 3.3 (1.8–4.8) | 1 | 1 | ||
| Yes | 620 | 6.7 (6.1–7.4) | 0.40 (0.28–0.57) | <0.001 | 0.63 (0.43–0.91) | 0.014 | |
| S-1-treated | No | 405 | 5.2 (4.8–5.7) | 1 | 1 | ||
| Yes | 249 | 8.6 (7.6–9.6) | 0.55 (0.46–0.65) | <0.001 | 0.56 (0.480.67) | <0.001 | |
| Cisplatin-treated | No | 311 | 4.5 (4.0–5.1) | 1 | 1 | ||
| Yes | 343 | 8.2 (7.6–8.7) | 0.50 (0.43–0.59) | <0.001 | 0.61 (0.51–0.73) | <0.001 | |
| 5-FU-treated | No | 460 | 5.5 (5.0–6.0) | 1 | 1 | ||
| Yes | 194 | 9.0 (7.6–10.5) | 0.58 (0.49–0.69) | <0.001 | 0.65 (0.54–0.78) | <0.001 | |
| Other agents-treatedb | No | 546 | 5.7 (5.1–6.3) | 1 | 1 | ||
| Yes | 108 | 10.3 (9.2–11.4) | 0.58 (0.43–0.75) | <0.001 | 0.64 (0.51–0.80) | <0.001 | |
Notes:
Adjusted for sex, age, Charlson comorbidity, ECOG PS, site of metastatic organ, and smoking history.
Other agents included chemotherapeutic agents with irinotecan, oxaliplatin, nab-paclitaxel, and erlotinib.
Abbreviation: ECOG PS, European Cooperative Oncology Group performance status.
Discussion
Our study demonstrated the evolving therapeutic landscape of mPC in Taiwan between the period of 2010–2016. The most common antitumor agent used for the treatment of mPC was gemcitabine (94.8%), followed by cisplatin (52.4%), S-1 (38.1%), and 5-FU (29.7%). The percentage of patients treated with S-1 between 2010 and 2016 significantly increased, while the percentage of patients treated with 5-FU dramatically decreased, whereas the percentage of patients treated with gemcitabine, cisplatin, and the other agents remained constant. An increase in the number of lines of treatment was observed throughout the study period, with <30% of patients receiving two or more lines of treatment in 2010 compared with 50% of patients receiving two or more lines of treatments in 2016. The absolute difference in median survival time was 2.1 months from 2010 (5.8 months) to 2016 (7.9 months) in our study. We observed increases in the 6-, 12-, and 24-months survival rates with an adjusted average annual percent change of 5.5%, 13.6%, and 25.1% throughout the study period, respectively.
Systemic chemotherapy with gemcitabine+Nab-paclitaxel or the FOLFIRINOX (folinic acid, 5-fluorouracil, irinotecan, oxaliplatin) is the standard treatment for mPC in Western Countries. However, the median survival time under these regimens remains 8–11 months.8–10 The reasons for under-using these regimens in Taiwan were partly related to their toxicity profile as well as the cost-benefit concerns.8,9 Before the reimbursement of S-1 in 2014, gemcitabine, cisplatin, and 5-FU were the backbone of chemotherapeutic agents in the treatment of mPC in Taiwan. Both 5-FU and gemcitabine showed their efficacy to improve survival outcome compared with best supportive care in mPC in a meta-analysis.22 However, 5-FU alone offered a very limited response rate of roughly 0–9% and a median survival time of approximately 3–6 months in patients with mPC.3,4,23,24 Although gemcitabine provided a better survival outcome than 5-FU in a phase III study,4 the tumor response rate slightly increased to 6–11% and the median OS to 5.6–8.8 months.8–10 Gemcitabine in combination with either 5-FU25–27 or cisplatin6 has been reported to be more effective than gemcitabine monotherapy in prolonging progression-free survival; however, all randomized trials failed to demonstrate a significant survival difference with combination treatment compared with gemcitabine alone.6,25–29 The survival outcome observed in our patient cohort in 2010–2013 (ranging from 5.3–6.0 months) was consistent with those observed in patients receiving 5-FU, gemcitabine, or cisplatin in previous studies.4,6–9 Our results indicated the clinical limitations of 5-FU, gemcitabine, and cisplatin treatments, irrespective of whether they were administered as monotherapy or combination treatment, for mPC in daily practice.
Our study observed a significant improvement in the survival outcome of patients with mPC in 2010–2016, and several reasons may have contributed to the result. First, the subsequent treatment after first-line chemotherapy might impact on survival as the number of patients receiving two or more lines of treatments increased substantially during the study period. A meta-analysis study reported that second-line chemotherapy significantly improved OS in pancreatic cancer refractory to gemcitabine-based therapy.30 More than 70% of the patients in the gemcitabine group received S-1 as second-line chemotherapy in the GEST study.10 Subsequent treatment with S-1 might partially explain the longer median survival time of the gemcitabine arm (8.8 months) in the GEST study10 than patients in the gemcitabine arm (ranging from 5.6–6.8 months) in other phase III studies from the Western countries.4,8,9 Second, it is speculated that the efficacy of S-1 in mPC might contribute the improvement of OS in our patient cohort. Because S-1 had moderate efficacy and a tolerable toxicity profile,10 it is widely used in combination with gemcitabine in first-line treatment or as subsequent treatment in gemcitabine-refractory patients in clinical studies,31–33 as well as in our clinical practice in Taiwan. Accordingly, the survival outcome in mPC improved gradually because more patients received S-1 treatment in our patient cohort.
For the first time, our study showed that the reimbursement of S-1 significantly changed the chemotherapeutic treatment pattern in clinical practice, and demonstrated a significant trend for improvement in the survival outcome of patients with mPC in 2010–2016 based on four institutes across Taiwan.
Limitations
However, this study had some limitations. First, this retrospective study was subject to patient selection bias favoring patients who were fit and eligible for the systemic treatment. Second, the decision of which chemotherapy regimen to administer to the patient might be confounded by different variables, including consideration of the patient’s performance status,8 different toxicity profiles of chemotherapy regimens,8–10 and physician’s preference; this study was unable to address the significance of all these factors. The follow-up duration was too short to represent the survival difference by joinpoint analysis,21 which might contribute to the lack of statistical difference in the survival rates of the trend 2 and trend 3 study periods in our study. Third, S-1 mono-therapy or gemcitabine+S-1 is promising in Japan and Taiwan based on the GEST study. A phase II study (NCT00429858) to evaluate the efficacy of gemcitabine+S-1 treating mPC in Western populations was early terminated because of the low accrual rate.34 While gemcitabine+Nab-paclitaxel, or the FOLFIRINOX regimens are the standard first-line regimens in other countries, our healthcare coverage limited the use of them, resulting in <3% of the patients receiving Nab-paclitaxel or FOLFIRINOX regimen. Therefore, the use of S-1 in different ethnicity group remains to be further studied. Last, but not least, the change in survival outcome across the 7-year study period was multifactorial rather than because of the different chemotherapy regimens. Therefore, we adjusted for other important prognostic factors, including age, sex, performance status, comorbidity, metastatic organ, smoking history, and Institute of treatment, with the aim of minimizing the bias for survival analysis.
Conclusion
Our study demonstrated the evolution of the therapeutic landscape, including an upward trend in S-1 and a downward trend in 5-FU utilization, and an increased percentage of mPC patients who received multiple lines of chemotherapy in Taiwan in 2010–2016. Based on this multi-institute cohort study in Taiwan, the reimbursement of S-1 changed the clinical practice and is associated with an improvement in survival outcome of mPC patients.
Supplementary materials
Lines of chemotherapy treatment received by patients according to year of diagnosis.
Chemotherapy regimens that patients received as first-line treatment in 2010–2016.
Acknowledgments
The authors thank Ms Vengi Ho and all the members of the Cancer Center, Chang Gung Memorial Hospital, for assisting them with data collection. This research was not funded by any public, commercial, or nonprofit agency.
Footnotes
Disclosure
The authors report no conflicts of interest in this work.
References
- 1.Siegel RL, Miller KD, Jemal A. Cancer statistics, 2018. CA Cancer J Clin. 2018;68:7–30. doi: 10.3322/caac.21442. [DOI] [PubMed] [Google Scholar]
- 2.Health Registry Annual Report 2013. Republic of China. Republic of China: Republic of China: Bureau of health promotion, department of health, executive Yuan; 2015. [Accessed September 20, 2018]. Available from: http://www.hpa.gov.tw/BHPNet/Web/Stat/Statistics.aspx. [Google Scholar]
- 3.van Rijswijk RE, Jeziorski K, Wagener DJ, et al. Weekly high-dose 5-fluorouracil and folinic acid in metastatic pancreatic carcinoma: a phase II study of the EORTC gastrointestinal tract Cancer Cooperative Group. Eur J Cancer. 2004;40(14):2077–2081. doi: 10.1016/j.ejca.2004.06.012. [DOI] [PubMed] [Google Scholar]
- 4.Burris HA, Moore MJ, Andersen J, et al. Improvements in survival and clinical benefit with gemcitabine as first-line therapy for patients with advanced pancreas cancer: a randomized trial. J Clin Oncol. 1997;15(6):2403–2413. doi: 10.1200/JCO.1997.15.6.2403. [DOI] [PubMed] [Google Scholar]
- 5.Moore MJ, Goldstein D, Hamm J, et al. Erlotinib plus gemcitabine compared with gemcitabine alone in patients with advanced pancreatic cancer: a phase III trial of the National Cancer Institute of Canada Clinical Trials Group. J Clin Oncol. 2007;25(15):1960–1966. doi: 10.1200/JCO.2006.07.9525. [DOI] [PubMed] [Google Scholar]
- 6.Colucci G, Giuliani F, Gebbia V, et al. Gemcitabine alone or with cisplatin for the treatment of patients with locally advanced and/or metastatic pancreatic carcinoma: a prospective, randomized phase III study of the Gruppo Oncologia dell’Italia meridionale. Cancer. 2002;94:902–910. [PubMed] [Google Scholar]
- 7.Pelzer U, Schwaner I, Stieler J, et al. Best supportive care (BSC) versus oxaliplatin, folinic acid and 5-fluorouracil (off) plus BSC in patients for second-line advanced pancreatic cancer: a phase III-study from the German CONKO-study group. Eur J Cancer. 2011;47(11):1676–1681. doi: 10.1016/j.ejca.2011.04.011. [DOI] [PubMed] [Google Scholar]
- 8.Conroy T, Desseigne F, Ychou M, et al. Folfirinox versus gemcitabine for metastatic pancreatic cancer. N Engl J Med. 2011;364(19):1817–1825. doi: 10.1056/NEJMoa1011923. [DOI] [PubMed] [Google Scholar]
- 9.von Hoff DD, Ervin T, Arena FP, et al. Increased survival in pancreatic cancer with nab-paclitaxel plus gemcitabine. N Engl J Med. 2013;369(18):1691–1703. doi: 10.1056/NEJMoa1304369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ueno H, Ioka T, Ikeda M, et al. Randomized phase III study of gemcitabine plus S-1, S-1 alone, or gemcitabine alone in patients with locally advanced and metastatic pancreatic cancer in Japan and Taiwan: Gest study. JCO. 2013;31(13):1640–1648. doi: 10.1200/JCO.2012.43.3680. [DOI] [PubMed] [Google Scholar]
- 11.Shirasaka T, Shimamato Y, Ohshimo H, et al. Development of a novel form of an oral 5-fluorouracil derivative (S-1) directed to the potentiation of the tumor selective cytotoxicity of 5-fluorouracil by two biochemical modulators. Anticancer Drugs. 1996;7(5):548–557. doi: 10.1097/00001813-199607000-00010. [DOI] [PubMed] [Google Scholar]
- 12.Ueno H, Okusaka T, Ikeda M, Takezako Y, Morizane C. An early phase II study of S-1 in patients with metastatic pancreatic cancer. Oncology. 2005;68(2–3):171–178. doi: 10.1159/000086771. [DOI] [PubMed] [Google Scholar]
- 13.Okusaka T, Funakoshi A, Furuse J, et al. A late phase II study of S-1 for metastatic pancreatic cancer. Cancer Chemother Pharmacol. 2008;61(4):615–621. doi: 10.1007/s00280-007-0514-8. [DOI] [PubMed] [Google Scholar]
- 14.Nakamura K, Yamaguchi T, Ishihara T, Sudo K, Kato H, Saisho H. Phase II trial of oral S-1 combined with gemcitabine in metastatic pancreatic cancer. Br J Cancer. 2006;94(11):1575–1579. doi: 10.1038/sj.bjc.6603168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Yanagimoto H, Satoi S, Sho M, et al. Phase I study assessing the feasibility of the triple combination chemotherapy of SOXIRI (S-1/oxaliplatin/irinotecan) in patients with unresectable pancreatic ductal adenocarcinoma. Cancer Chemother Pharmacol. 2016;77(1):35–41. doi: 10.1007/s00280-015-2928-z. [DOI] [PubMed] [Google Scholar]
- 16.Zhang W, du C, Sun Y, et al. Nab-paclitaxel plus S-1 as first-line followed by S-1 maintenance for advanced pancreatic adenocarcinoma: a single-arm phase II trial. Cancer Chemother Pharmacol. 2018;82(4):655–660. doi: 10.1007/s00280-018-3650-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Quan H, Sundararajan V, Halfon P, et al. Coding algorithms for defining comorbidities in ICD-9-CM and ICD-10 administrative data. Med Care. 2005;43(11):1130–1139. doi: 10.1097/01.mlr.0000182534.19832.83. [DOI] [PubMed] [Google Scholar]
- 18.UICC TNM . Classification of Malignant Tumours. 7th ed. New York: Wiley & Liss; 2009. [Google Scholar]
- 19.Chou WC, Wang F, Cheng YF, et al. A simple risk stratification model that predicts 1-year postoperative mortality rate in patients with solid-organ cancer. Cancer Med. 2015;4(11):1687–1696. doi: 10.1002/cam4.518. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Armitage P. Tests for linear trends in proportions and frequencies. Biometrics. 1955;11(3):375–386. [Google Scholar]
- 21.National Cancer Institute [homepage on the Internet] Annual percent change (APC) and confidence interval. [Accessed September 20, 2018]. Available from: https://surveillance.cancer.gov/help/joinpoint/setting-parameters/method-and-parameters-tab/apc-aapc-tau-confidence-intervals/estimate-average-percent-change-apc-and-confidence-interval.
- 22.Sultana A, Smith CT, Cunningham D, Starling N, Neoptolemos JP, Ghaneh P. Meta-analyses of chemotherapy for locally advanced and metastatic pancreatic cancer. J Clin Oncol. 2007;25(18):2607–2615. doi: 10.1200/JCO.2006.09.2551. [DOI] [PubMed] [Google Scholar]
- 23.Crown J, Casper ES, Botet J, Murray P, Kelsen DP. Lack of efficacy of high-dose leucovorin and fluorouracil in patients with advanced pancreatic adenocarcinoma. J Clin Oncol. 1991;9(9):1682–1686. doi: 10.1200/JCO.1991.9.9.1682. [DOI] [PubMed] [Google Scholar]
- 24.DeCaprio JA, Mayer RJ, Gonin R, Arbuck SG. Fluorouracil and high-dose leucovorin in previously untreated patients with advanced adenocarcinoma of the pancreas: results of a phase II trial. J Clin Oncol. 1991;9(12):2128–2133. doi: 10.1200/JCO.1991.9.12.2128. [DOI] [PubMed] [Google Scholar]
- 25.Di Costanzo F, Carlini P, Doni L, et al. Gemcitabine with or without continuous infusion 5-FU in advanced pancreatic cancer: a randomised phase II trial of the Italian oncology group for clinical research (GOIRC) Br J Cancer. 2005;93(2):185–189. doi: 10.1038/sj.bjc.6602640. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Hess V, Salzberg M, Borner M, et al. Combining capecitabine and gemcitabine in patients with advanced pancreatic carcinoma: a phase I/II trial. J Clin Oncol. 2003;21(1):66–68. doi: 10.1200/JCO.2003.04.029. [DOI] [PubMed] [Google Scholar]
- 27.Scheithauer W, Schüll B, Ulrich-Pur H, et al. Biweekly high-dose gemcitabine alone or in combination with capecitabine in patients with metastatic pancreatic adenocarcinoma: a randomized phase II trial. Ann Oncol. 2003;14(1):97–104. doi: 10.1093/annonc/mdg029. [DOI] [PubMed] [Google Scholar]
- 28.Colucci G, Labianca R, di Costanzo F, et al. Randomized phase III trial of gemcitabine plus cisplatin compared with single-agent gemcitabine as first-line treatment of patients with advanced pancreatic cancer: the GIP-1 study. J Clin Oncol. 2010;28(10):1645–1651. doi: 10.1200/JCO.2009.25.4433. [DOI] [PubMed] [Google Scholar]
- 29.Heinemann V, Quietzsch D, Gieseler F, et al. Randomized phase III trial of gemcitabine plus cisplatin compared with gemcitabine alone in advanced pancreatic cancer. J Clin Oncol. 2006;24(24):3946–3952. doi: 10.1200/JCO.2005.05.1490. [DOI] [PubMed] [Google Scholar]
- 30.Zhang YD, Yang Q, Jiang ZM, Ma W, Zhou SW, Xie DR. Overall survival of patients with advanced pancreatic cancer improved with an increase in second-line chemotherapy after gemcitabine-based therapy. JOP. 2011;12(2):131–137. [PubMed] [Google Scholar]
- 31.Ioka T, Komatsu Y, Mizuno N, et al. Randomised phase II trial of irinotecan plus S-1 in patients with gemcitabine-refractory pancreatic cancer. Br J Cancer. 2017;116(4):464–471. doi: 10.1038/bjc.2016.436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Ohkawa S, Okusaka T, Isayama H, et al. Randomised phase II trial of S-1 plus oxaliplatin vs S-1 in patients with gemcitabine-refractory pancreatic cancer. Br J Cancer. 2015;112(9):1428–1434. doi: 10.1038/bjc.2015.103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Takahara N, Isayama H, Nakai Y, et al. A retrospective study of S-1 and oxaliplatin combination chemotherapy in patients with refractory pancreatic cancer. Cancer Chemother Pharmacol. 2013;72(5):985–990. doi: 10.1007/s00280-013-2278-7. [DOI] [PubMed] [Google Scholar]
- 34.ClinicalTrial.gov [webpage on the Internet] NCT00429858. Gem-citabine and S-1 for locally advanced unresectable or metastatic pancreatic cancer. [Accessed October 20, 2018]. Available from: https://clinicaltrials.gov/ct2/show/NCT00429858?term=s-1&cond=Pancreatic+Cancer+Non-resectable&rank=4.
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Lines of chemotherapy treatment received by patients according to year of diagnosis.
Chemotherapy regimens that patients received as first-line treatment in 2010–2016.