Abstract
Glaucoma is one of the leading causes of blindness. Previous studies have indicated that the oxidative stress-induced apoptosis of trabecular meshwork cells (TMCs) may serve a key role in the pathogenesis of glaucoma, and that micro RNA(miR)-17-5p may be involved in this process. However, the specific mechanisms require further investigation. The aim of the present study was to investigate the effects of miR-17-5p on the proliferation and apoptosis of human TMCs (HTMCs) in response to oxidative stress. It was observed that exposure to H2O2 induced a significant decrease in the proliferation and a marked increase in the apoptosis of HTMCs. H2O2 exposure also suppressed the expression of miR-17-5p and promoted the expression of phosphatase and tensin homolog (PTEN). Furthermore, transient overexpression of miR-17-5p induced a significant increase in the proliferation and a significant decrease in the apoptosis of HTMCs by affecting the expression of PTEN, and the apoptosis-related proteins B-cell lymphoma-associated X protein (Bax), B-cell lymphoma-extra large (Bcl-xL) and B-cell lymphoma-2 (Bcl-2). However, knockdown of miR-17-5p demonstrated the opposite results. The results of a dual luciferase reporter assay demonstrated that PTEN may be a direct target of miR-17-5p. In conclusion, miR-17-5p was downregulated in HTMCs under oxidative conditions, and miR-17-5p may regulate the apoptosis of HTMCs by targeting PTEN. These results provide a novel theoretical basis and potential therapeutic target for the treatment of glaucoma.
Keywords: glaucoma, micro RNA-17-5p, phosphatase and tensin homolog, human trabecular meshwork cells, apoptosis
Introduction
Glaucoma is a common ophthalmic disease, and it is the second leading cause of blindness worldwide (1). It is estimated that by 2020, the number of patients with glaucoma will reach approximately 80 million (2). Glaucoma can be divided into open-angle glaucoma (OAG) and angle-closure glaucoma (ACG), and in Western countries, approximately 75% of glaucoma patients are diagnosed with OAG (3), while in Asian countries, ACG is more prevalent than OAG (4). In 2010, 86.5% of ACG patients were resident in Asia, and the largest number of patients affected by ACG was in China (47.5% of the total) (5). In recent years, increasing evidence has indicated that the oxidative stress-induced senescence of trabecular meshwork cells (TMCs) may play a key role in the occurrence and development of glaucoma (6,7); however, the specific mechanisms still require further investigation.
MicroRNAs (miRNAs) are short non-coding RNAs (length, ~22 nucleotides) that can bind to the 3′-UTR of target mRNAs and consequently suppress the expression of target genes. miRNAs can regulate a variety of biological activities, including embryonic development, angiogenesis, cell differentiation, proliferation and apoptosis (8–10). With the development of next-generation sequencing and bioinformatics methods, differentially expressed miRNAs that are potentially involved in the pathogenesis of diseases, including cancers (11,12) and glaucoma (13–15), have been successfully identified. The targets of these miRNAs, as well as related signaling pathways, have also been predicted. miR-17-5p is located on human chromosome 13q31, and it belongs to the miR-17-92 cluster (16). miR-17-5p is up-regulated in many tumor types and participates in the pathogenesis of different cancers as an oncogene (17–19). Besides cancer-related studies, miR-17-5p has been reported to promote oxidative stress-induced apoptosis of cardiomyocytes in ischemia/reperfusion-induced cardiac injury animal models (20). It has also been observed that knockdown of miR-17-5p can restore the expression of the very-low-density lipoprotein receptor and alleviate the symptoms of atherosclerosis in ApoE-/- mouse models (21). It has been observed that miR-17-5p is down-regulated in the trabecular meshwork under oxidative stress (22); however, the specific mechanisms have not yet been investigated.
In the present study, we focused on the roles of miR-17-5p in glaucoma and its related mechanisms. Human TMCs (HTMCs) were treated with H2O2 to induce oxidative stress, and the effects of miR-17-5p on the behaviors of HTMCs under oxidative stress were explored. We hypothesized that miR-17-5p would regulate the proliferation and apoptosis of HTMCs by targeting phosphatase and tensin homolog (PTEN). The present study may provide a theoretical basis and novel therapeutic target for the treatment of glaucoma.
Materials and methods
HTMC culture
HTMCs were purchased from Sciencell Research Laboratories (San Diego, CA, USA). According to the product information, HTMCs were isolated from the juxtacanalicular and corneoscleral regions of human eyes, and characterized by immunofluorescence methods using α-smooth muscle actin- and fibronectin-specific antibodies. Cells were cultured in a humidified incubator at 37°C with 5% CO2 using Dulbecco's modified Eagle's medium (DMEM; Gibco; Thermo Fisher Scientific, Inc., Waltham, MA, USA) supplied with 10% fetal bovine serum (Gibco; Thermo Fisher Scientific, Inc.) and 1% penicillin-streptomycin solution (Gibco; Thermo Fisher Scientific, Inc.). Oxidative stress was induced by treating HTMCs with DMEM that contained 300 µM H2O2 (Beyotime Institute of Biotechnology, Shanghai, China) for 3–7 h. Then, the cells were harvested for future analysis.
Cell transfection
The miR-17-5p inhibitor and miR-17-5p mimic oligonucleotides were synthesized by GenePharma Co., Ltd., (Shanghai, China). HTMCs were cultured until confluence, trypsinized and seeded onto new six-well plates at 100,000 cells/well. Cells were then transfected with either miR-17-5p inhibitor or miR-17-5p mimic using Lipofectamine® 3000 reagent (Invitrogen; Thermo Fisher Scientific, Inc.), according to the manufacturer's protocol. After transfection, cells were cultured for 48 h and harvested for future analysis.
Cell proliferation analysis
An MTT assay was performed to determine the viability of HTMCs following different treatments. An MTT proliferation assay kit (Sigma-Aldrich; Merck KGaA, Darmstadt, Germany) was used, according to the manufacturer's protocol.
Cellular apoptosis analysis
For cellular apoptosis analysis, HTMCs subjected to different treatments were stained using an Annexin V-FITC apoptosis detection kit (Sigma-Aldrich; Merck KGaA). The apoptosis rate of the cells in different groups was detected and analyzed using a BD FACSVerse flow cytometer (BD Biosciences, San Jose, CA, USA), according to the manufacturer's protocol.
Reverse transcription-quantitative PCR (RT-qPCR)
Total RNA was extracted from HTMCs using TRIzol (Invitrogen; Thermo Fisher Scientific, Inc.), and reverse transcribed into cDNA using the PrimeScript™ RT Master Mix (Perfect Real Time) kit (Takara Biotechnology Co., Ltd., Dalian, China), then qPCR was performed using the SYBR ExScript RT-PCR kit (Takara Biotechnology Co., Ltd.) on an ABI 7300 Real-Time PCR System (Applied Biosystems; Thermo Fisher Scientific, Inc.), according to the manufacturer's protocol. The thermocycling profiles are shown in Table I. The relative expression of PTEN in each sample was normalized to the level of GAPDH using the 2−ΔΔCq method. The expression of miR-17-5p was examined using the Hairpin-it™ miRNAs qPCR Quantitation Kit (GenePharma Co., Ltd.) according to the manufacturer's protocol, and U6 (RNU6B; GenePharma Co., Ltd.) was used for normalization. Primers were synthesized by GenScript (Nanjing, China), and the sequences of the primers are shown in Table II.
Table I.
Thermocycling profiles for reverse transcription-quantitative polymerase chain reaction.
| Cycle no. | Temperature | Time |
|---|---|---|
| 1 | 95°C | 30 Sec |
| 40 | 95°C | 5 Sec |
| 60°C | 4 Sec |
Table II.
Sequences of the primers used in the present study.
| Gene name | Sequences of the primer |
|---|---|
| miR-17-5p | F: 5′-TCTAGATCCCGAGGACTG-3′ |
| R: 5′-ATCGTGACCTGAACC-3′ | |
| U6 | F: 5′-CTCGCTTTGGCAGCACA-3′ |
| R: 5′-AACGCTTCACGAATTTGCGT-3′ | |
| PTEN | F: 5′-CGACGGGAAGACAAGTTCAT-3′ |
| R: 5′-AGGTTTCCTCTGGTCCTGGT-3′ | |
| GAPDH | F: 5′-GACAGTCAGCCGCATCTTCT-3′ |
| R: 5′-TTAAAAGCAGCCCTGGTGAC-3′ |
PTEN, phosphatase and tensin homolog; F, forward; R, reverse.
Western blot analysis
Cells were lysed using RIPA buffer (Beyotime Institute of Biotechnology), and the concentration of total protein was measured using a BCA Kit (Beyotime Institute of Biotechnology). Next, SDS-PAGE was performed to separate the proteins, and the proteins were then transferred onto polyvinylidene fluoride membranes (EMD Millipore, Billerica, MA, USA). The membranes were blocked with 5% non-fat milk and incubated with primary antibodies (anti-Bax, anti-PTEN, anti-Bcl-2, anti-Bcl-xL and anti-GAPDH, all purchased from Abcam, Cambridge, MA, USA) overnight at 4°C. The following day, the membranes were incubated with HRP-conjugated secondary antibodies (purchased from Abcam, Cambridge, MA, USA), then washed and incubated with the enhanced chemiluminescence reagent (Beyotime Institute of Biotechnology). Finally, the signals were detected using the ChemiDoc™XRS+ imaging system (Bio-Rad Laboratories, Inc., Hercules, CA, USA).
Immunocytochemistry
HTMCs were fixed in 4% paraformaldehyde and permeabilized with 0.1% Triton X-100, then incubated with the anti-PTEN primary antibodies for 30 min. Cells were incubated with Alexa Fluor 488-conjugated secondary antibodies, then visualized using an Olympus fluorescent microscope (Olympus Corporation, Tokyo, Japan).
Computational methods
The target genes of miR-17-5p were predicted using online bioinformatics tools, TargetScan (http://www.targetscan.org) and miRanda (http://www.microrna.org).
Dual luciferase reporter assay
Fragments of either the wild-type PTEN 3′-UTR (PTEN-3′UTR) or mutant PTEN 3′-UTR (PTEN-MUT) region that contains the miR-17-5p binding site were synthesized and cloned into the pGL6-TA-reporter plasmid (Beyotime Institute of Biotechnology). The plasmids were transfected into 293 cells using Lipofectamine® 3000 for 48 h, and the activity of the luciferase in each group was examined using the dual-luciferase reporter system (Beyotime Institute of Biotechnology), according to the manufacturer's protocol.
Statistics
All statistical analyses were conducted using SPSS v.22.0 software (IBM Corp., Armonk, NY, USA). Data are presented as the mean ± standard deviation. Two independent sample t-tests were performed for comparisons between two groups. One-way analysis of variance followed by Dunnett's post-hoc test was performed for comparisons among multiple groups. P<0.05 was considered to indicate a statistically significant difference.
Results
Effect of oxidative stress on the proliferation and apoptosis of HTMCs in vitro
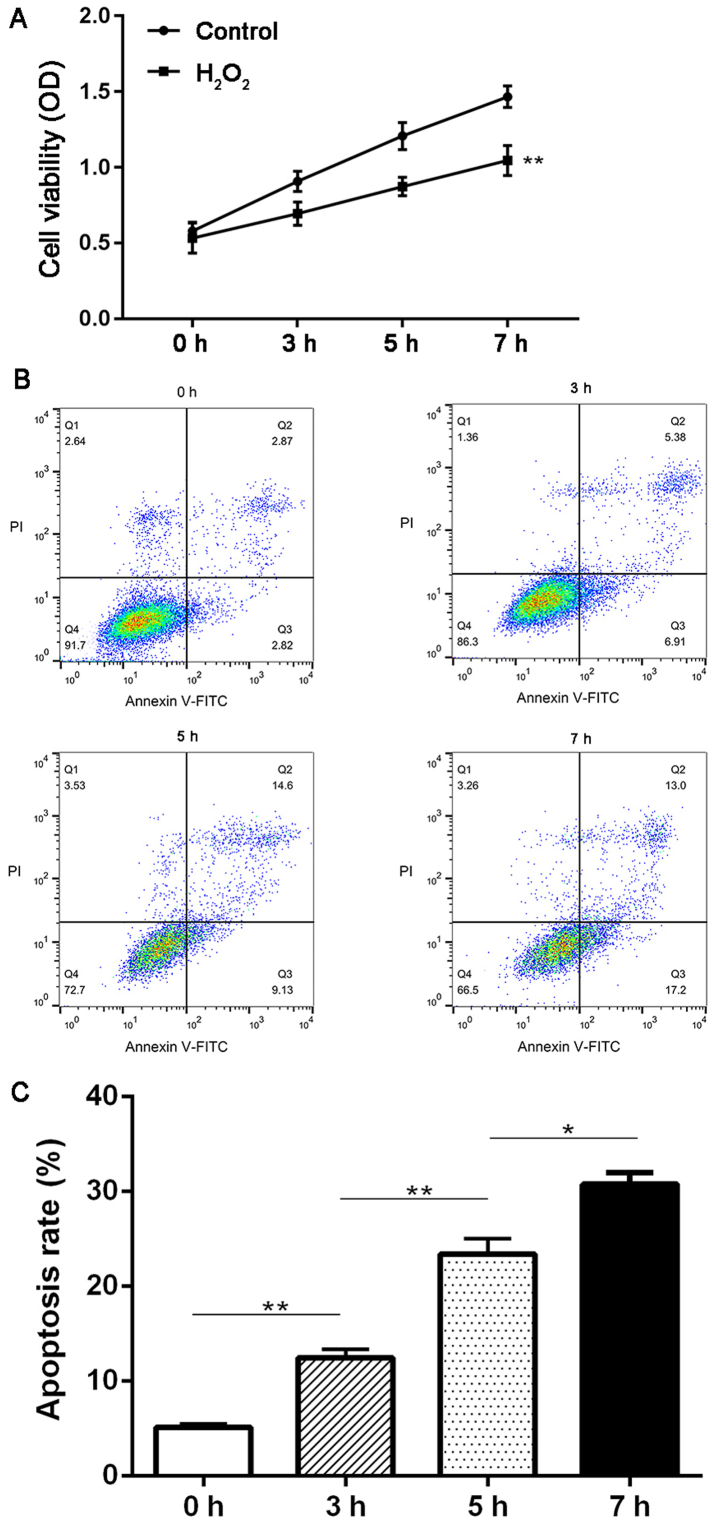
First, we investigated the effect of H2O2 treatment on the proliferation and apoptosis of HTMCs using MTT and flow cytometry methods. It was observed that the cell viability was significantly decreased in cells treated with H2O2 compared with the control group at 3, 5 and 7 h (P<0.01; Fig. 1A). Moreover, H2O2 treatment also induced a continuous increase in the apoptosis rate of HTMCs from 0 to 7 h (P<0.05; Fig. 1B and C).
Figure 1.
Effect of oxidative stress on the proliferation and apoptosis of HTMCs in vitro. (A) Proliferation rate of HTMCs in different groups by MTT assay, **P<0.01 vs. the control group; (B) Apoptosis rate of HTMCs in different groups by flow cytometry method. (C) Quantification of results in (B). **P<0.01 vs. the 3 h group, *P<0.05 vs. the 5 h group.
miR-17-5p was down-regulated in HTMCs under oxidative stress in vitro
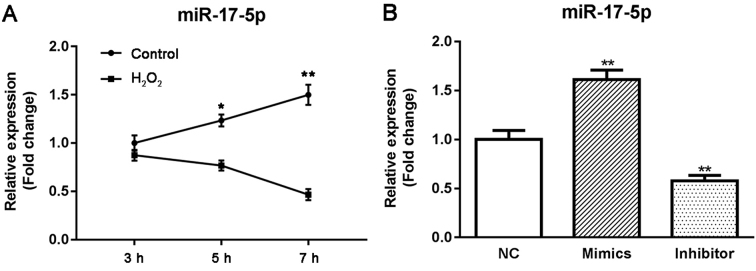
In order to explore the roles of miR-17-5p in HTMCs under oxidative stress, we compared the expression of miR-17-5p in control and H2O2 groups at different time points using RT-qPCR methods. As shown in Fig. 2A, H2O2 induced a significant decrease in the expression of miR-17-5p at 5 and 7 h (P<0.05; Fig. 2A).
Figure 2.
Expressions of miR-17-5p in HTMCs by RT-qPCR methods. (A) Effect of oxidative stress on the expression of miR-17-5p in HTMCs. (B) Effect of transfection of miR-17-5p mimics or inhibitor on the expression of miR-17-5p in HTMCs. *P<0.05 and **P<0.01 vs. the control group. HTMCs, human trabecular meshwork cells.
miR-17-5p can regulate the proliferation and apoptosis of HTMCs in vitro
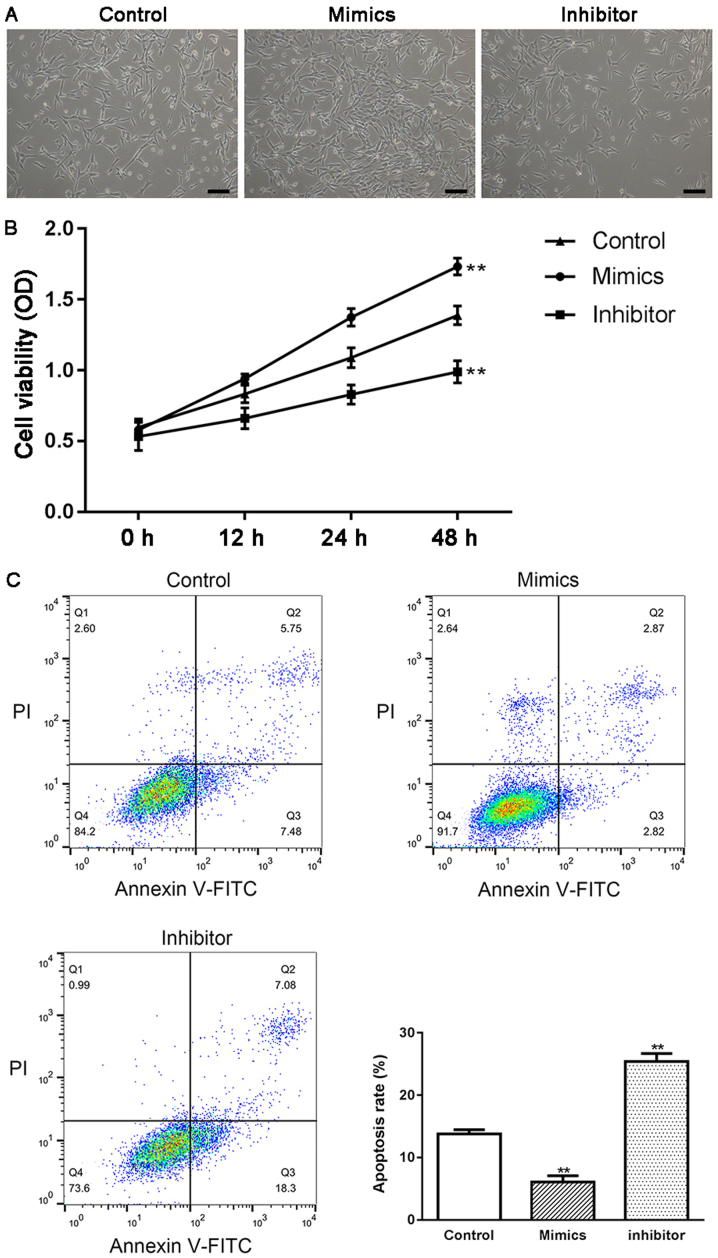
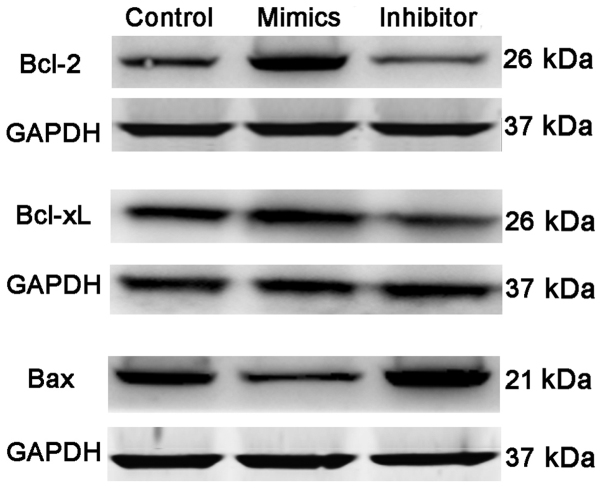
HTMCs were transfected with either miR-17-5p mimics or miR-17-5p inhibitor, and the effect of miR-17-5p on the proliferation and apoptosis of HTMCs was examined using MTT and flow cytometry methods. As shown in Fig. 2B, the expression of miR-17-5p was significantly increased in miR-17-5p mimic-transfected HTMCs and markedly decreased in miR-17-5p inhibitor-transfected HTMCs, suggesting that transfection had been successfully performed. Moreover, transient overexpression of miR-17-5p induced a significant increase in the proliferation (P<0.01; Fig. 3A and B) and a significant decrease in the apoptosis of HTMCs (P<0.01; Fig. 3C). Knockdown of miR-17-5p exhibited the opposite results. In addition, transfection of miR-17-5p mimics also induced a significant increase in the expression of Bcl-2 and Bcl-xL, and a marked decrease in the expression of Bax, while knockdown of miR-17-5p exhibited the opposite results (Fig. 4).
Figure 3.
Effect of miR-17-5p on the proliferation and apoptosis of HTMCs in vitro. (A) Morphological feature of HTMCs in different groups by inverted microscope (scale bar, 100 µm); (B) Proliferation rate of HTMCs in different groups by MTT assay; (C) Apoptosis rate of HTMCs in different groups by flow cytometry method. **P<0.01 vs. the control group. Control, un-transfected cells; Inhibitor, miR-17-5p inhibitor transfected cells; Mimics, miR-17-5p mimics transfected cells. HTMCs, human trabecular meshwork cells.
Figure 4.
Effect of miR-17-5p on the expression of Bcl-2, Bcl-xL and Bax in HTMCs by western blot methods. Control, un-transfected cells; Inhibitor, miR-17-5p inhibitor transfected cells; Mimics, miR-17-5p mimics transfected cells. Bcl-2, B-cell lymphoma-2; Bcl-xL, B-cell lymphoma extra-large; BAX, Bcl-2-associated X protein; HTMCs, human trabecular meshwork cells; GAPDH, glyceraldehyde-3-phosphate dehydrogenase.
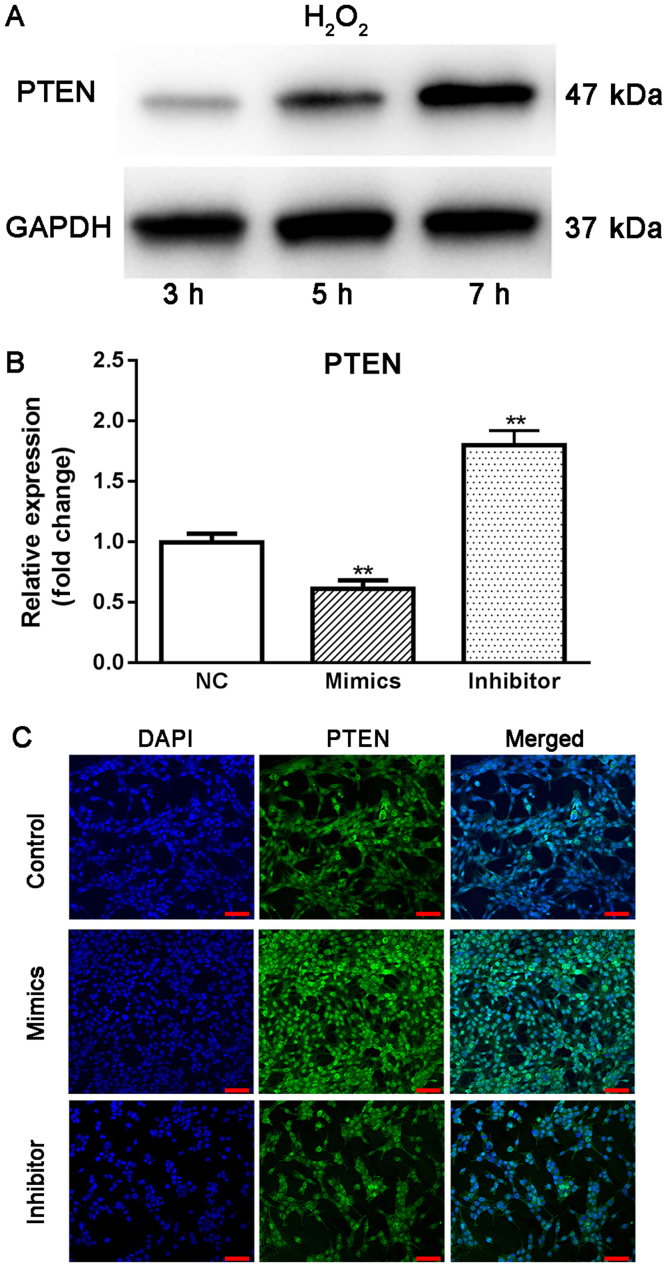
PTEN is a direct target of miR-17-5p in HTMCs
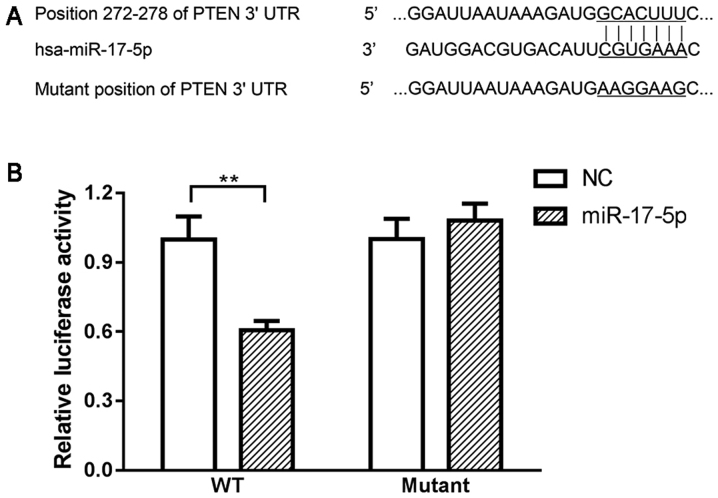
Using online bioinformatics tools, PTEN was predicted to be a direct target of miR-17-5p. Using western blot analysis, it was observed that the expression of PTEN continuously increased from 3 to 7 h under oxidative stress (Fig. 5A), suggesting that increased PTEN may be associated with the pathogenesis of glaucoma. Moreover, transient overexpression of miR-17-5p in HTMCs induced a significant decrease in the expression of PTEN, while knockdown of miR-17-5p promoted the expression of PTEN in HTMCs (Fig. 5B and C). Finally, the results from the dual luciferase reporter assay indicated that co-transfection of miR-17-5p mimics and wild-type PTEN-3′UTR plasmids significantly decreased the activity of the luciferases. Co-transfection of miR-17-5p mimic and PTEN-MUT exhibited no effect on the activity of the luciferases (Fig. 6), suggesting that PTEN is a direct target of miR-17-5p.
Figure 5.
Roles of PTEN in the pathogenesis of glaucoma. (A) Expression of PTEN in HTMCs under oxidative stress by western blot method; (B) Effect of overexpression and knockdown of miR-17-5p on the mRNA expression of PTEN in HTMCs by RT-qPCR method; (C) Effect of overexpression and knockdown of miR-17-5p on the protein expression of PTEN in HTMCs using ICC method (scale bar, 100 µm). **P<0.01 vs. NC. NC, un-transfected cells; Inhibitor, miR-17-5p inhibitor transfected cells; Mimics, miR-17-5p mimics transfected cells. PTEN, Phosphatase and tensin homolog; HTMCs, human trabecular meshwork cells; GAPDH, glyceraldehyde-3-phosphate dehydrogenase.
Figure 6.
PTEN is direct target of miR-17-5p. (A) Sequence alignment of the paired 3′-UTR of miR-17-5p and PTEN by bioinformatics tool Targetscan and miRanda; (B) Results of dual luciferase reporter assay. **P<0.01. WT, Fragments of wild-type PTEN 3′-UTR; Mutant, fragments of wild-type PTEN 3′-UTR; NC, miR-17-5p negative control; PTEN, Phosphatase and tensin homolog; GAPDH, glyceraldehyde-3-phosphate dehydrogenase.
Discussion
Glaucoma is a common ophthalmic disorder that can cause damage to the optic nerve and loss of vision. Increasing evidence has indicated that oxidative stress-induced senescence of HTMCs is involved in the pathogenesis of glaucoma (7). H2O2 is an important oxidant in the human eye. It has been demonstrated that oxidative levels significantly increase in the aqueous humor of patients with glaucoma, which means that TMCs will also be exposed to high concentrations of H2O2. Thus, in previous studies, a high concentration of H2O2 has been used to induce oxidative stress in HTMCs, and this has now become a widely applied in vitro model in glaucoma-related studies (23,24).
In recent years, the effects of miRNAs on TMCs have been discussed in many studies (25,26). Li et al (22), performed miRNA array analysis to examine alterations in the expression levels of microRNAs in oxidative stress-induced cellular senescence in HTMCs. It was observed that the expression of miR-17-5p was significantly decreased by treatment with a high concentration of H2O2. However, the specific mechanisms underlying this remain unclear. In the present study, we identified that compared with untreated cells, H2O2 treatment induced a significant decrease in the proliferation and a marked increase in the apoptosis of HTMCs at each time point examined (P<0.05). Moreover, H2O2 treatment also induced a significant decrease in the expression of miR-17-5p, which was consistent with the finding from Li et al. These results indicate that miR-17-5p is down-regulated in HTMCs under oxidative stress, and miR-17-5p may participate in the mechanism of H2O2-induced increase in apoptosis and decrease in the proliferation of HTMCs.
Next, we transfected HTMCs with either miR-17-5p mimics or miR-17-5p inhibitor and evaluated the effect of miR-17-5p on the proliferation and apoptosis of HTMCs. We observed that transient overexpression of miR-17-5p induced a significant increase in the proliferation and a significant decrease in the apoptosis of HTMCs (P<0.05), while knockdown of miR-17-5p exhibited the opposite results. Moreover, Bcl-2 and Bcl-xL are known to be anti-apoptotic proteins, and Bax is a pro-apoptotic protein (27,28). Our data indicated that transfection of miR-17-5p mimics promoted the expression of Bcl-2 and Bcl-xL, and suppressed the expression of Bax, while knockdown of miR-17-5p exhibited the opposite effects. Taken together, these results indicate that miR-17-5p can regulate the proliferation and apoptosis of HTMCs in vitro.
PTEN is known to be a tumor suppressor in the field of cancer studies (29). Knockdown of PTEN can lead to an increase in the proliferation and a decrease in the apoptosis rate of many cancer cell types, leading to the incidence and development of various diseases (30–32). However, in the case of glaucoma, PTEN may play a different role. It has been observed that increased expression of PTEN can lead to the degeneration of optic neurons, and knockdown of PTEN can significantly alleviate optic nerve damage in glaucoma. In a very recent study, Tellios et al (33) demonstrated that phosphorylation of PTEN is significantly increased in TMCs upon treatment with TGF-β (increased levels of TGF-β in the aqueous humor is one of the main causes of fibrosis in OAG), and an increase in the expression of PTEN may contribute to the fibrosis of the HTMCs in glaucoma. In the present study, we observed that the expression of PTEN was markedly increased in HTMCs following exposure to H2O2 (P<0.01), suggesting that increased PTEN may be involved in the pathogenesis of glaucoma. Moreover, transfection of miR-17-5p mimic or inhibitor induced a significant decrease or increase in the expression of PTEN, respectively. The results from the dual luciferase reporter assay confirmed that PTEN is a direct target of miR-17-5p. In summary, these data indicated that miR-17-5p can regulate the proliferation and apoptosis of HTMCs by targeting PTEN.
The present study has certain limitations. We only performed cellular experiments, and the expression levels of miR-17-5p in tissue samples from patients and normal controls should be evaluated to confirm our findings from the clinical perspective. However, due to ethical issues, it is difficult to obtain the trabecular meshwork of healthy volunteers; thus, in vivo studies using animal models could be a better option.
In conclusion, our data demonstrated that miR-17-5p is down-regulated and PTEN is up-regulated in HTMCs under oxidative conditions. We also identified that miR-17-5p can regulate the proliferation and apoptosis of HTMCs through targeting PTEN. Our data indicated that miR-17-5p has the potential to become a novel therapeutic target for the treatment of glaucoma.
Acknowledgements
Not applicable.
Funding
The persent study was sponsored by the funds from the project of the Department of Health, Heilongjiang Province (project no. 2011-005).
Availability of data and materials
The datasets used and/or analyzed during the present study are available from the corresponding author on reasonable request.
Authors' contributions
FZ designed the study and wrote the majority of the manuscript. XW performed most of the experiments and wrote part of the manuscript. ZL and JB performed some of the experiments. WS performed some of the statistical analysis.
Ethics approval and consent to participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Zhao R, Yin D, Wang E, Si B. The effect of MTHFR ala222val polymorphism on open-angle glaucoma: a meta-analysis. Ophthalmic Genet. 2015;36:27–30. doi: 10.3109/13816810.2014.969379. [DOI] [PubMed] [Google Scholar]
- 2.Tham YC, Li X, Wong TY, Quigley HA, Aung T, Cheng CY. Global prevalence of glaucoma and projections of glaucoma burden through 2040: A systematic review and meta-analysis. Ophthalmology. 2014;121:2081–2090. doi: 10.1016/j.ophtha.2014.05.013. [DOI] [PubMed] [Google Scholar]
- 3.Bailey JN, Loomis SJ, Kang JH, Allingham RR, Gharahkhani P, Khor CC, Burdon KP, Aschard H, Chasman DI, Igo RP, Jr, et al. Genome-wide association analysis identifies TXNRD2, ATXN2 and FOXC1 as susceptibility loci for primary open-angle glaucoma. Nat Genet. 2016;48:189–194. doi: 10.1038/ng.3482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Liang YB, Wang NL, Rong SS, Thomas R. Initial treatment for primary angle-closure glaucoma in China. J Glaucoma. 2015;24:469–473. doi: 10.1097/IJG.0000000000000075. [DOI] [PubMed] [Google Scholar]
- 5.Nongpiur ME, Khor CC, Jia H, Cornes BK, Chen LJ, Qiao C, Nair KS, Cheng CY, Xu L, George R, et al. ABCC5, a gene that influences the anterior chamber depth, is associated with primary angle closure glaucoma. PLoS Genet. 2014;10:e1004089. doi: 10.1371/journal.pgen.1004089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Shen W, Han Y, Huang B, Qi Y, Xu L, Guo R, Wang X, Wang J. MicroRNA-483-3p inhibits extracellular matrix production by targeting Smad4 in human trabecular meshwork cells. Invest Ophthalmol Vis Sci. 2015;56:8419–8427. doi: 10.1167/iovs.15-18036. [DOI] [PubMed] [Google Scholar]
- 7.Xu L, Zhang Y, Guo R, Shen W, Qi Y, Wang Q, Guo Z, Qi C, Yin H, Wang J. HES1 promotes extracellular matrix protein expression and inhibits proliferation and migration in human trabecular meshwork cells under oxidative stress. Oncotarget. 2017;8:21818–21833. doi: 10.18632/oncotarget.15631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Feng R, Sang Q, Zhu Y, Fu W, Liu M, Xu Y, Shi H, Xu Y, Qu R, Chai R, et al. MiRNA-320 in the human follicular fluid is associated with embryo quality in vivo and affects mouse embryonic development in vitro. Sci Rep. 2015;5:8689. doi: 10.1038/srep08689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Li Y, Cai B, Shen L, Dong Y, Lu Q, Sun S, Liu S, Ma S, Ma PX, Chen J. MiRNA-29b suppresses tumor growth through simultaneously inhibiting angiogenesis and tumorigenesis by targeting Akt3. Cancer Lett. 2017;397:111–119. doi: 10.1016/j.canlet.2017.03.032. [DOI] [PubMed] [Google Scholar]
- 10.Qun L, Wenda X, Weihong S, Jianyang M, Wei C, Fangzhou L, Zhenyao X, Pingjin G. miRNA-27b modulates endothelial cell angiogenesis by directly targeting Naa15 in atherogenesis. Atherosclerosis. 2016;254:184–192. doi: 10.1016/j.atherosclerosis.2016.10.007. [DOI] [PubMed] [Google Scholar]
- 11.Falzone L, Candido S, Salemi R, Basile MS, Scalisi A, McCubrey JA, Torino F, Signorelli SS, Montella M, Libra M. Computational identification of microRNAs associated to both epithelial to mesenchymal transition and NGAL/MMP-9 pathways in bladder cancer. Oncotarget. 2016;7:72758–72766. doi: 10.18632/oncotarget.11805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Bagnoli M, De Cecco L, Granata A, Nicoletti R, Marchesi E, Alberti P, Valeri B, Libra M, Barbareschi M, Raspagliesi F, et al. Identification of a chrXq27.3 microRNA cluster associated with early relapse in advanced stage ovarian cancer patients. Oncotarget. 2011;2:1265–1278. doi: 10.18632/oncotarget.401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Guo R, Shen W, Su C, Jiang S, Wang J. Relationship between the pathogenesis of glaucoma and miRNA. Ophthalmic Res. 2017;57:194–199. doi: 10.1159/000450957. [DOI] [PubMed] [Google Scholar]
- 14.Molasy M, Walczak A, Szaflik J, Szaflik JP, Majsterek I. MicroRNAs in glaucoma and neurodegenerative diseases. J Hum Genet. 2017;62:105–112. doi: 10.1038/jhg.2016.91. [DOI] [PubMed] [Google Scholar]
- 15.Paylakhi SH, Yazdani S, April C, Fan JB, Moazzeni H, Ronaghi M, Elahi E. Non-housekeeping genes expressed in human trabecular meshwork cell cultures. Mol Vis. 2012;18:241–254. [PMC free article] [PubMed] [Google Scholar]
- 16.Tsuchida A, Ohno S, Wu W, Borjigin N, Fujita K, Aoki T, Ueda S, Takanashi M, Kuroda M. miR-92 is a key oncogenic component of the miR-17-92 cluster in colon cancer. Cancer Sci. 2011;102:2264–2271. doi: 10.1111/j.1349-7006.2011.02081.x. [DOI] [PubMed] [Google Scholar]
- 17.Zhu Y, Gu J, Li Y, Peng C, Shi M, Wang X, Wei G, Ge O, Wang D, Zhang B, et al. MiR-17-5p enhances pancreatic cancer proliferation by altering cell cycle profiles via disruption of RBL2/E2F4-repressing complexes. Cancer Lett. 2018;412:59–68. doi: 10.1016/j.canlet.2017.09.044. [DOI] [PubMed] [Google Scholar]
- 18.Liao XH, Xiang Y, Yu CX, Li JP, Li H, Nie Q, Hu P, Zhou J, Zhang TC. STAT3 is required for MiR-17-5p-mediated sensitization to chemotherapy-induced apoptosis in breast cancer cells. Oncotarget. 2017;8:15763–15774. doi: 10.18632/oncotarget.15000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Jin T, Wu X, Yang H, Liu M, He Y, He X, Shi X, Wang F, Du S, Ma Y, et al. Association of the miR-17-5p variants with susceptibility to cervical cancer in a Chinese population. Oncotarget. 2016;7:76647–76655. doi: 10.18632/oncotarget.12299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Shi J, Bei Y, Kong X, Liu X, Lei Z, Xu T, Wang H, Xuan Q, Chen P, Xu J, et al. miR-17-3p contributes to exercise-induced cardiac growth and protects against myocardial ischemia-reperfusion injury. Theranostics. 2017;7:664–676. doi: 10.7150/thno.15162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Tan L, Meng L, Shi X, Yu B. Knockdown of microRNA-17-5p ameliorates atherosclerotic lesions in ApoE-/- mice and restores the expression of very low density lipoprotein receptor. Biotechnol Lett. 2017;39:967–976. doi: 10.1007/s10529-017-2337-y. [DOI] [PubMed] [Google Scholar]
- 22.Li G, Luna C, Qiu J, Epstein DL, Gonzalez P. Alterations in microRNA expression in stress-induced cellular senescence. Mech Ageing Dev. 2009;130:731–741. doi: 10.1016/j.mad.2009.09.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Yu AL, Fuchshofer R, Kampik A, Welge-Lüssen U. Effects of oxidative stress in trabecular meshwork cells are reduced by prostaglandin analogues. Invest Ophthalmol Vis Sci. 2008;49:4872–4880. doi: 10.1167/iovs.07-0984. [DOI] [PubMed] [Google Scholar]
- 24.Chen M, Liu B, Gao Q, Zhuo Y, Ge J. Mitochondria-targeted peptide MTP-131 alleviates mitochondrial dysfunction and oxidative damage in human trabecular meshwork cells. Invest Ophthalmol Vis Sci. 2011;52:7027–7037. doi: 10.1167/iovs.11-7524. [DOI] [PubMed] [Google Scholar]
- 25.Wang Y, Li F, Wang S. MicroRNA-93 is overexpressed and induces apoptosis in glaucoma trabecular meshwork cells. Mol Med Rep. 2016;14:5746–5750. doi: 10.3892/mmr.2016.5938. [DOI] [PubMed] [Google Scholar]
- 26.Luna C, Li G, Qiu J, Epstein DL, Gonzalez P. MicroRNA-24 regulates the processing of latent TGFβ1 during cyclic mechanical stress in human trabecular meshwork cells through direct targeting of FURIN. J Cell Physiol. 2011;226:1407–1414. doi: 10.1002/jcp.22476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Reshi L, Wang HV, Hui CF, Su YC, Hong JR. Anti-apoptotic genes Bcl-2 and Bcl-xL overexpression can block iridovirus serine/threonine kinase-induced Bax/mitochondria-mediated cell death in GF-1 cells. Fish Shellfish Immunol. 2017;61:120–129. doi: 10.1016/j.fsi.2016.12.026. [DOI] [PubMed] [Google Scholar]
- 28.Yang J, Yao S. JNK-Bcl-2/Bcl-xL-Bax/Bak pathway mediates the crosstalk between matrine-induced autophagy and apoptosis via interplay with Beclin 1. Int J Mol Sci. 2015;16:25744–25758. doi: 10.3390/ijms161025744. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Liu H, Pan Y, Han X, Liu J, Li R. MicroRNA-216a promotes the metastasis and epithelial-mesenchymal transition of ovarian cancer by suppressing the PTEN/AKT pathway. Onco Targets Ther. 2017;10:2701–2709. doi: 10.2147/OTT.S114318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Ronen S, Abbott DW, Kravtsov O, Abdelkader A, Xu Y, Banerjee A, Iczkowski KA. PTEN loss and p27 loss differ among morphologic patterns of prostate cancer, including cribriform. Hum Pathol. 2017;65:85–91. doi: 10.1016/j.humpath.2017.04.024. [DOI] [PubMed] [Google Scholar]
- 31.Feng Y, Zou W, Hu C, Li G, Zhou S, He Y, Ma F, Deng C, Sun L. Modulation of CASC2/miR-21/PTEN pathway sensitizes cervical cancer to cisplatin. Arch Biochem Biophys. 2017;623-624:20–30. doi: 10.1016/j.abb.2017.05.001. [DOI] [PubMed] [Google Scholar]
- 32.Li S, Shen Y, Wang M, Yang J, Lv M, Li P, Chen Z, Yang J. Loss of PTEN expression in breast cancer: Association with clinicopathological characteristics and prognosis. Oncotarget. 2017;8:32043–32054. doi: 10.18632/oncotarget.16761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Tellios N, Belrose JC, Tokarewicz AC, Hutnik C, Liu H, Leask A, Motolko M, Iijima M, Parapuram SK. TGF-β induces phosphorylation of phosphatase and tensin homolog: Implications for fibrosis of the trabecular meshwork tissue in glaucoma. Sci Rep. 2017;7:812. doi: 10.1038/s41598-017-00845-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and/or analyzed during the present study are available from the corresponding author on reasonable request.