Abstract
Background
Many observational studies have reported correlations between postoperative complications and prognosis after radical gastrectomy but the results are controversial. This meta-analysis was performed to investigate whether there is a correlation between postoperative complications and prognosis after radical gastrectomy.
Methods
Literature searches were performed in PubMed, EMBASE, and the Cochrane Library. Studies that investigated the correlations between any postoperative complications and prognosis after radical gastrectomy were included. The pooled hazard ratio (HR) with 95% confidence interval (CI) for postoperative complications regarding overall survival (OS) or recurrence-free survival (RFS) was calculated by using RevMan 5.3.5. Subgroup analyses were performed within pathological stages I, II, and III.
Results
Sixteen retrospective studies comprising 12,065 patients were included. The pooled HR (95% CI) for complications regarding OS was 1.79 (1.39, 2.30) and was 1.40 (1.06, 1.84) after excluding in-hospital mortality; the pooled HR (95% CI) for complications regarding RFS was 1.28 (1.10, 1.49). The pooled HR (95% CI) for infectious complications and leakage regarding OS was 1.86 (1.22, 2.83) and 2.02 (1.02, 4.00), respectively. The pooled HR (95% CI) for any reported postoperative complications regarding OS for stage I, II, and III diseases was 2.39 (0.77, 7.46), 4.35 (2.58, 7.35), and 2.84 (1.77, 4.56), respectively.
Conclusions
Postoperative complications correlate with poor prognosis after radical gastrectomy. Such correlations are found in stage II and III gastric cancer patients but remain to be determined in stage I gastric cancer patients.
Electronic supplementary material
The online version of this article (10.1186/s12957-019-1593-9) contains supplementary material, which is available to authorized users.
Keywords: Complications, Prognosis, Radical gastrectomy, Stomach neoplasms
Background
The incidence of postoperative complications after radical gastrectomy remains high [1–4], and the estimated incidence is 12.8 to 14% [5–7]. In addition to undermining the short-term survival, postoperative complications may also be correlated with long term prognosis. Currently, increasing numbers of observational studies have investigated the correlation between postoperative complications and long-term prognosis after radical gastrectomy. Although some reports have negative findings [8–12], other studies have demonstrated that overall postoperative complications, infectious complications, and gastrointestinal leakages are all correlated with poor overall survival (OS) and/or recurrence-free survival (RFS) [13–23]. Additionally, the correlations between postoperative complications and long-term prognosis in different stages are controversial and are based on subgroup analyses with small sample sizes [13, 18–20].
Given the prevalence of postoperative complications after radical gastrectomy, it is important to determine whether a correlation exists between postoperative complications and poor prognosis. The existence of that correlation may not only lead to a consideration of shortening follow-up interval and enforcing adjuvant chemotherapy in patient who have developed postoperative complications, but may also underline the necessity of neoadjuvant chemotherapy and stress control management in patients with high risk of developing postoperative complications to reduce the hazard for long term prognosis [9, 11, 21]. In the meta-analysis, the correlations between postoperative complications and prognosis after radical gastrectomy were assessed.
Methods
Search strategy and eligibility criteria
The PubMed, EMBASE, and Cochrane Library databases were searched from inception until February 24, 2019, for studies that assessed the relationship between postoperative complications and prognosis after radical gastrectomy. The following medical subject heading (MeSH) terms and keywords were used: “Stomach Neoplasms”, “Gastrectomy”, “Postoperative Complications”, and “Prognosis”. The search was restricted to studies on humans and to those that were published in the English language. The titles and abstracts were screened by two authors independently. The inclusion criterion was as follows: any study that compared the long-term prognosis between patients with and without postoperative complications after radical gastrectomy for gastric cancer. The exclusion criteria were as follows: (1) data of other neoplasms other than gastric cancer were included in the survival analysis; (2) data of palliative surgery were included in the survival analysis; (3) studies that describe the same patient population; (4) hazard ratio (HR) cannot be estimated; (5) describing complications without precise definitions; (6) letters, comments, or conference abstracts. When multiple studies describing the same patient population were identified, the most recent publication was used unless additional data were provided in the earlier work.
Data extraction
The following data were extracted: first author, year of publication, study design, number of subjects, adjuvant chemotherapy, tumor stage, types of complications, incidences of complications, HR of any postoperative complications, and 5-year OS and 5-year RFS for patients with and without postoperative complications, as well as whether in-hospital deaths were excluded in the survival analysis. Unreported data were requested through e-mail from corresponding authors of the included studies. If there was no response to the e-mails, the missing data were estimated from the figures in the published literatures using Engauge Digitizer 4.1 (Mark Mitchell, Baurzhan Muftakhidinov, and Tobias Winchen et al., “Engauge Digitizer Software.” Webpage: http://markummitchell.github.io/engauge-digitizer) and the HRs were estimated using the method of Tierney et al. [24].
Study quality assessment
The methodological quality of each observational study was assessed by the Newcastle-Ottawa Scale (NOS, ranging 0–9) [25]. In brief, each study was assessed for the following aspects: selection, comparability, and outcome or exposure. The comparability was primarily assessed for pathological stage and was also assessed for aspects of adjuvant chemotherapy and in-hospital death disposition in the survival analysis.
Statistical analysis
Statistical analysis was performed with RevMan (version 5.3.5.; Cochrane Collaboration). HRs and their 95% confidence intervals (CIs) were used to evaluate the association between postoperative complications and prognosis (OS and/or RFS). Subgroup analyses were performed to investigate the correlations between infectious complication, gastrointestinal leakage, and prognosis. Furthermore, correlations were investigated for each pathological stage when possible. Statistical heterogeneities among studies were assessed by the I2 statistic. The random effects model and the fixed effects model were used. If I2 was less than 40% (cutoff point), we used the fixed effect model, while if I2 was more than 40%, the random effects model was chosen. Sensitivity analysis, in which one study was removed at a time, was performed to evaluate the stability of the results. Descriptive techniques were used when clinical heterogeneity existed or when no data could be used in the pooling analysis. The assessment of publication bias was evaluated using the funnel plot.
We followed both the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) Statement [26], and the guidelines for Meta-analysis of Observational Studies in Epidemiology (MOOSE) in reporting this study [27]. All analyses were based on previously published studies, thus no ethical approval and patient consent are required.
Results
Literature searches and description of studies
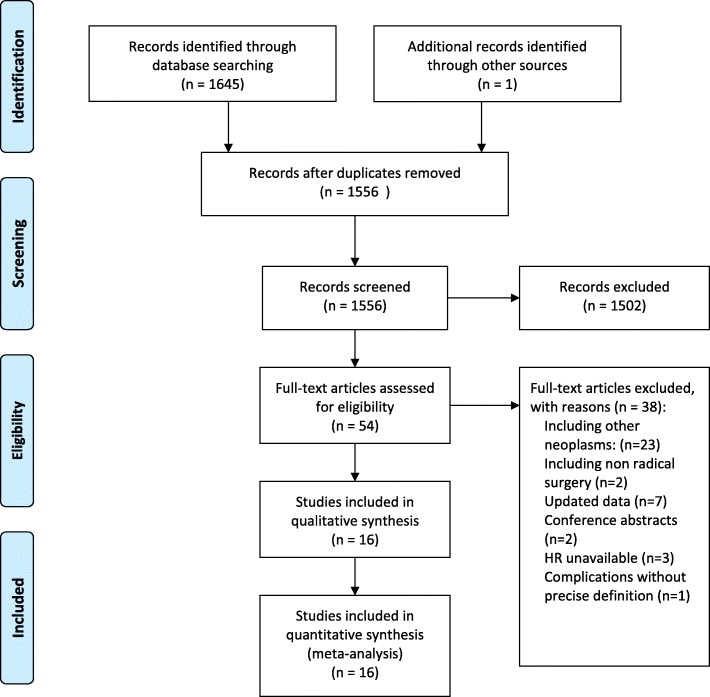
The flow diagram of the literature searches is shown in Fig. 1. The entire study sample size from the 16 included studies was 12,065 patients. The characteristics of the included studies are shown in Table 1. The quality of the included studies was analyzed, and the NOS scores of the included studies varied between 6 and 9 points (see Additional file 1: Table S1).
Fig. 1.
Flow chart of articles identified, included, and excluded
Table 1.
Study characteristics
| Author, year | NOS score | Country | Samplea size | Period | Complications type | Comparable | In-hospital death | 5-year RFSb | 5-year OSb | |
|---|---|---|---|---|---|---|---|---|---|---|
| Pathological stage | Chemotherapy | |||||||||
| Tsujimoto et al. 2009 [13] | 8 | Japan | 141/1191 | 1986–2005 | Infectious | Not | Not* | Excluded | NR | 57.9% vs. 78.1% |
| Sierzega et al. 2010 [14] | 7 | Poland | 41/649 | 1999–2004 | Leakage | NR | NR | Involved | NR | NR |
| Yoo et al. 2011 [15] | 6 | Korea | 32/446 | 2000–2005 | Leakage | Yes | NR | Involved | NR | 9.4% vs. 68.4% |
| Nagasako et al. 2012 [16] | 6 | Japan | 37/363 | 1997–2008 | Anastomotic | NR | NR | Involved | NR | 81% vs. 94.2% |
| Li et al. 2013 [17] | 7 | China | 51/378 | 2005–2006 | Overall | NR | NR | Involved | NR | 21.8%vs. 39.9% |
| Tokunaga et al. 2013 [18] | 6 | Japan | 81/684 | 2002–2006 | Intra-abdominal infectious | Not | None | Involved | 64.9% vs. 84.5% | 66.4% vs. 86.8% |
| Kubota et al. 2014 [19] | 7 | Japan | 207/981 | 2005–2008 | Overall CD ≥ 2 | Not | NR | Excluded | NR | 84.1% vs. 93.1% |
| Hayashi et al. 2015 [20] | 7 | Japan | 52/450 | 2000–2005 | Infectious CD ≥ 2 | Not | None | None | NR | 58% vs. 83% |
| Kim et al. 2015 [8] | 7 | Korea | 72/3755 | 2003–2012 | Leakage | NR | NR | None | NR | 70.8% vs. 79.3% |
| Saito al. 2015 [9] | 6 | Japan | 86/219 | 2001–2012 | Overall CD ≥ 2 | NR | NR | NR | 53.4% vs. 70.5% | NR |
| Jin et al. 2016 [21] | 6 | U.S.A | 336/488 | 2000–2012 | Overall | Not | Not | Excluded | 23% vs. 40% | 27% vs. 43% |
| Abdul Kader et al. 2016 [22] | 7 | Japan | 38/227 | 1991–2010 | Intra-abdominal | Yes | Yes | Involved | NR | 24.6% vs. 69.2% |
| Climent et al. 2016 [10] | 8 | Spain | 162/109 | 1990–2009 | Overall CD ≥ 2/infectious | Yes | Yes | Excluded | 46.9% vs. 54.1% | 48.1% vs. 56.9% |
| Li et al. 2018 [23] | 8 | China | 86/172 | 2008–2015 | Overall CD > 2 | Yes | NA | Excluded | NA | 46.3% vs. 65.9% |
| Eto et al. 2018 [11] | 9 | Japan | 35/66 | 2005–2015 | Overall CD ≥ 2 | Yes | Yes | None | 41.7% vs. 43.9% | 58.2% vs. 56.3% |
| Watanabe et al. 2018 [12] | 7 | Japan | 134/296 | 1992–2010 | Overall CD > 2 | Not | Yes | Involved | 46.9% vs. 45.0% | 51.3% vs. 47.6% |
CD Clavien–Dindo classification of surgical complications, NOS Newcastle-Ottawa Scale, NA not available, NR not reported, OS overall survival, RFS recurrence-free survival
aPatients number with and without concerned complications
bComplications group vs. control group
*More patients in the complication group received adjuvant chemotherapy
Studies on postoperative complications and OS
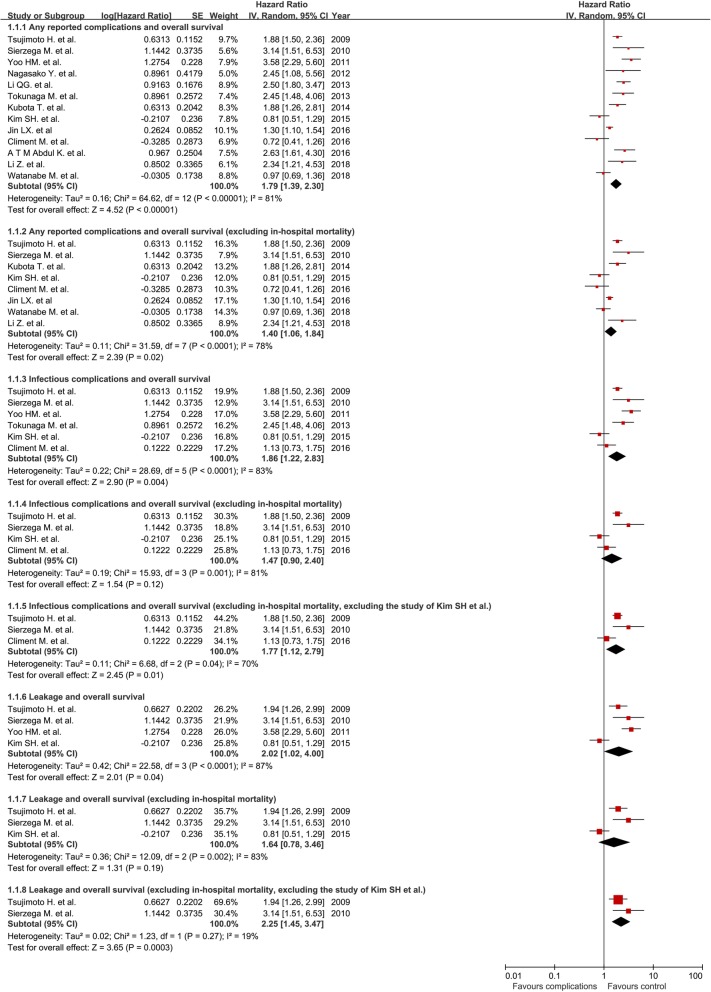
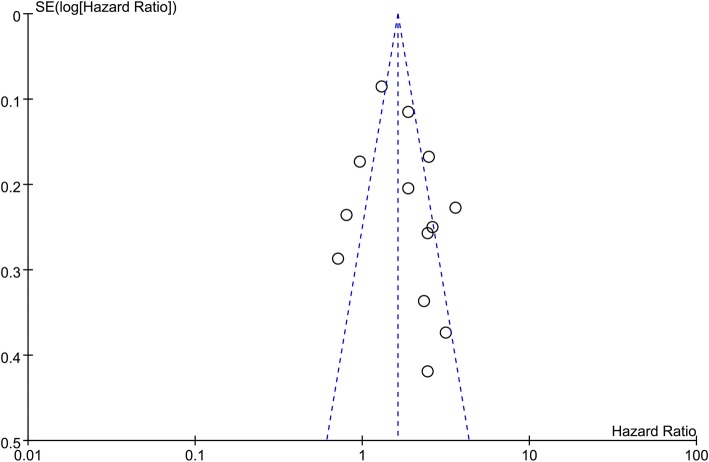
Thirteen studies were included in the analysis of correlation between any reported postoperative complications and OS [8, 10, 12–19, 21–23]. Of the included studies, eight excluded influences from in-hospital death in the survival analysis [8, 10, 12–14, 19, 21, 23]. The pooled HR (95% CI) of postoperative complications for OS was 1.79 (1.39, 2.30) and was 1.40 (1.06, 1.84) after excluding the in-hospital mortality (Fig. 2). The sensitivity analysis demonstrated that no individual study significantly influenced the overall effect of the HRs. Publication bias was examined by the funnel plot and there was no evidence of publication bias among these comparisons (Fig. 3).
Fig. 2.
The association of postoperative complications with overall survival
Fig. 3.
Funnel plots for visual inspection of publication bias
Six studies reported the correlation between infectious complications and OS [8, 10, 13–15, 18]. Four of the studies excluded the in-hospital mortality [8, 10, 13, 14]. The pooled HR of postoperative infectious complications for OS was 1.86 (1.22, 2.83) and was 1.47 (0.90, 2.40) after excluding the in-hospital mortality (Fig. 2). Sensitivity analysis demonstrated that the study form Kim et al. caused high heterogeneity. After excluding the study, the corresponding pooled HR (95% CI) of infectious complications (in-hospital mortality excluded) changed from 1.47 (0.90, 2.40) to 1.77 (1.12, 2.79).
Four studies reported the relationship between gastrointestinal leakages and OS [8, 13–15]. Three studies excluded the in-hospital mortality [8, 13, 14]. The pooled HR of gastrointestinal leakages for OS was 2.02 (1.02, 4.00) and was 1.64 (0.78, 3.46) after excluding the in-hospital mortality (Fig. 2). Sensitivity analysis demonstrated that the study form Kim et al. caused high heterogeneity. After excluding the study, the corresponding pooled HR (95% CI) of leakage (in-hospital mortality excluded) changed from 1.64 (0.78, 3.46) to 2.25 (1.45, 3.47).
Studies on postoperative complications and RFS
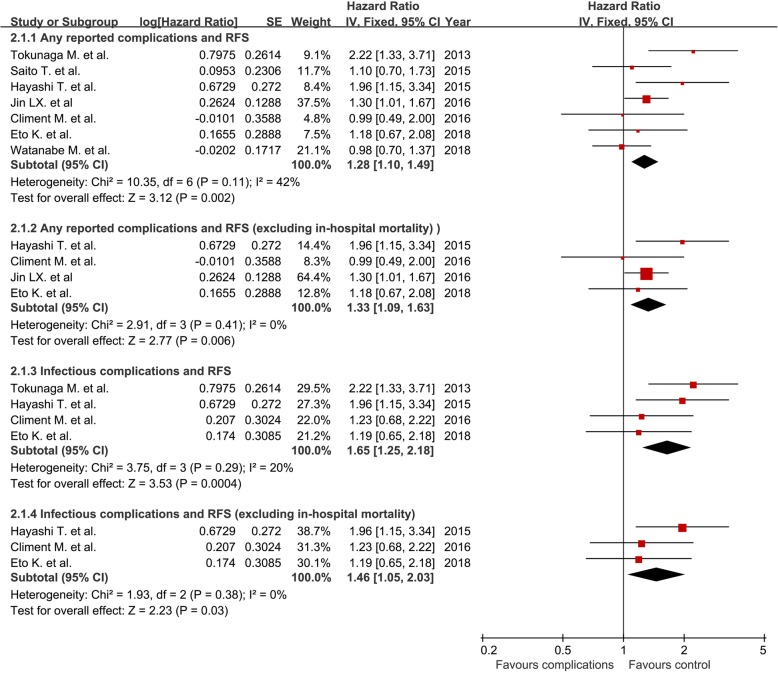
Seven studies were included in the analysis of correlation between any reported postoperative complications and RFS [9–12, 18, 20, 21]. Four studies excluded the in-hospital mortality [10, 11, 20, 21]. The pooled HR for RFS is 1.28 (1.10, 1.49) and was 1.33 (1.09, 1.63) after excluding the in-hospital death (Fig. 4).
Fig. 4.
The association of postoperative complications with recurrence-free survival
Four studies investigated the correlation between infectious complications and RFS [10, 11, 18, 20], and three of them excluded the in-hospital mortality in the analysis [10, 11, 20]. The pooled HR for the RFS in the infectious complications group was 1.65 (1.25, 2.18) and was 1.46 (1.05, 2.03) after excluding the in-hospital mortality (Fig. 4). The results of the sensitivity analysis demonstrated that no individual study significantly influenced the overall effect of HRs.
Studies on postoperative complications and survival in separated pathological stages
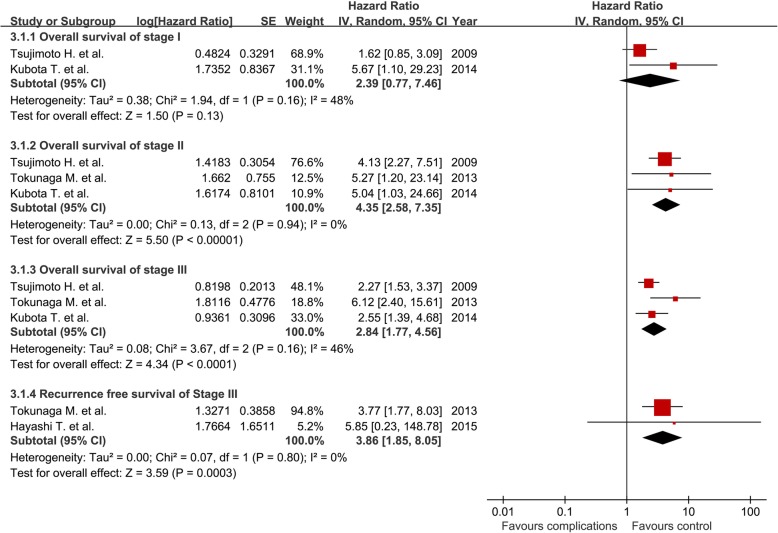
Three studies analyzed the correlations between postoperative complications and prognosis in stage I gastric cancer [13, 18, 19]. One study reported a nonsignificant correlation between postoperative complications and OS but did not present any detailed data or figures in the published report [18]. Therefore, two studies with available data were included in the analysis [13, 19]. The pooled HR (95% CI) of postoperative complications for OS in patients with stage I gastric cancer was 2.39 (0.77, 7.46) (Fig. 5).
Fig. 5.
The association of postoperative complications with overall survival and recurrence-free survival within varied pathological stages
Three studies analyzed the correlation between postoperative complications and OS in stage II gastric cancer [13, 18, 19]. The pooled HR of postoperative complications for OS in patients with stage II gastric cancer was 4.35 (2.58, 7.35) (Fig. 5).
Three studies analyzed the correlation between postoperative complications and OS in patients with stage III gastric cancer [13, 18, 19], and two studies reported the RFS [18, 20]. The pooled HR of postoperative complications for OS in patients with stage III gastric cancer from was 2.84 (1.77, 4.56), and the pooled HR (95% CI) for RFS was 3.86 (1.85, 8.05) (Fig. 5).
Discussion
The present study undertook a comprehensive review and meta-analysis of the literatures to assess the relationship between postoperative complications and patient prognosis. The results demonstrated that, although the correlation was not found by several studies, the pooled results showed that postoperative complications correlated with poor prognosis.
Several reasons may contribute to the divergences. First, the negative findings in some studies may be ascribed to the interfered application of adjuvant chemotherapy. Jin et al. demonstrated lower proportion of adjuvant chemotherapy in the complication group (47% vs. 61%), and the combination of postoperative complications and receiving no adjuvant therapy significantly increased the hazard of death and recurrence. Furthermore, decreased OS and RFS were not observed in patients who experienced complications but received adjuvant therapy [21]. Another study demonstrated that the adjuvant chemotherapy was postponed in patients with intra-abdominal complications (55.3 ± 34.7 vs. 26.6 ± 11.9 days) [22], and the postponed chemotherapy is correlated with poorer survival in patients with gastric cancer [28]. Second, the application of prophylactic neoadjuvant chemotherapy may abolish the poor prognosis induced by postoperative complications. In a cohort with 101 patients who underwent curative gastrectomy after receiving neoadjuvant chemotherapy, Eto et al. demonstrated a comparable RFS between patients with and without postoperative complications, and the 5-year RFS was 41.7% and 43.9%, respectively [11]. Third, the varied perioperative stress level may be an additional reason for the negative finding. Saito et al. demonstrated that the postoperative inflammation degree (reflected by the CRP level), rather than the postoperative complication itself, is related to the recurrence and poor prognosis [9]. Besides, Watanabe et al. also demonstrated comparable prognosis between patients with and without postoperative complications [12]. Their patients underwent total gastrectomy with splenectomy for the treatment of proximal advanced gastric cancer. The extensive resection might lead to an excessive surgical stress in both groups and that may lead to a deteriorated prognosis in patients without postoperative complications [29].
Accordingly, the results of the present study may highlight the importance of both adjuvant and neoadjuvant chemotherapy in patients with postoperative complications or with a high risk of developing postoperative complications. The results of the present study may have also highlighted the stress control management during the perioperative period. However, whether a decreased stress level will result to an improved prognosis remains to be determined. Additionally, any other methods that decrease the postoperative complications may also indirectly improve the prognosis. The intraoperative manipulation, such as the choice of reconstruction or the less invasive approach, may play a role in decreasing the postoperative complications and thereby improve the prognosis indirectly. For instance, recent studies demonstrated that BI reconstruction method significantly reduced the postoperative complications after laparoscopic distal gastrectomy [30, 31]. Therefore, patients may benefit more from that approach with low risk of postoperative complications.
In the analysis of the relationship between infectious complications or gastrointestinal leakages and OS, the study from Kim et al. demonstrated high heterogeneity. Kim et al. found that gastrointestinal leakage was not associated with decreased survival. There are some possible reasons for the negative results [8]. First, the effect of leakage may be diluted by the effect of other complications occurred in the control group. That is to say, other complications other than gastrointestinal leakage may also contribute to the poor prognosis and that may cause an underestimated effect of leakage on prognosis. Second, the sample size may not be adequate to detect the significant correlation because their Kaplan-Meier curve demonstrated a trend of poor OS in the leakage group (p = 0.076) [8].
The present study had some limitations. First, five of the included studies did not exclude in-hospital death in the survival analysis [15–18, 22]. It is well acknowledged that in-hospital mortality would be higher in patients with postoperative complications and would decrease the OS accordingly. Therefore, a subgroup analysis with the eight reports that excluded in-hospital death or have no in-hospital death was performed and a similar result was found (HR 1.40, 95% CI 1.06–1.84). Second, more preoperative comorbidity, a higher ASA or ECOG score, and older age were frequently observed in the complication group, as shown in Additional file 1: Table S1, and such characteristics are correlated with a shorter life expectancy after surgery. As a result, we analyzed the data from seven studies that reported RFS [9–12, 18, 20, 21], and the HR demonstrated a positive correlation between postoperative complications and reduced RFS (HR 1. 28, 95% CI 1.10–1.49). The correlation between postoperative complications and poor RFS still exist after the in-hospital mortality were excluded (HR 1.33, 95% CI 1.09–1.63). Third, patients in the complication group frequently had more advanced disease. Eleven of the studies demonstrated the proportion of each stage, and six of the studies reported comparable stages between the two groups [10–12, 15, 22, 23]. Such a bias may cause an overestimated correlation of postoperative complications with long-term prognosis. To avoid the influence of unbalanced tumor stages, the correlations between complications and prognosis were analyzed in separate stages based on the data from four studies [13, 18–20]. In addition to the correlation between postoperative complications and decreased OS and RFS in stage II and III patients, attention should be paid to stage I patients with postoperative complications because of the undetermined result (Fig. 5). If such a correlation did exist, the application of adjuvant chemotherapy might be expanded to stage I patients who have developed postoperative complications. However, a limited number of studies were included in the subgroup analysis of separated pathological stages and the confounders cannot be avoided in the subgroup analysis. More solid evidence from studies with larger sample sizes is warranted, and RFS analysis should also be considered in further studies.
Conclusions
In summary, there is good evidence to support the correlations between postoperative complications and poor prognosis after radical gastrectomy. The influence of postoperative complications on prognosis is also demonstrated in patients with stage II and III gastric cancer but remains to be determined in patients with stage I gastric cancer. To reduce the negative impact of postoperative complications on the long term prognosis, neoadjuvant chemotherapy may be considered in patients with high risk of developing postoperative complications and adjuvant chemotherapy should be enforced in patients who have developed postoperative complications. Additionally, perioperative stress control management might be beneficial for improving the long term prognosis after radical gastrectomy.
Additional file
Table S1. NOS of Cohort studies. NOS of Case-control studies. (DOCX 27 kb)
Acknowledgements
Not applicable
Funding
This work was supported by the National Natural Science Foundation of China (grant number 81400662) and Social Development Fund of Shaanxi Province (grant number 2018SF-271)
Availability of data and materials
All data generated or analyzed during this study are included in the published articles which were listed in Table 1.
Abbreviations
- HR
Hazard ratio
- OS
Overall survival
- RFS
Recurrence free survival
Authors’ contributions
SW and JY contributed to the study concept and design. LX, QW, JL, ZL, XW, PY, BB, and XL contributed to the acquisition of data. SW, JY, JL, ZL, LX, and QW contributed to the analysis and interpretation of data. LX, SW, JL, XW, and PY contributed to the drafting of the manuscript. QW, JY, BB, and XL contributed to the critical revision of the manuscript for important intellectual content. SW, QW, JL, and ZL contributed to the statistical analysis. SW and JY obtained funding. All authors read and approved the final manuscript.
Ethics approval and consent to participate
All analyses were based on previous published studies, thus no ethical approval and patient consent are required.
Consent for publication
Not applicable
Competing interests
The authors declare that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Shiqi Wang, Phone: 862984771533, Email: wsqfmmu@126.com.
Lei Xu, Email: xulfmmu@126.com.
Quan Wang, Email: wangquan2013@126.com.
Jipeng Li, Email: hy1977@fmmu.edu.cn.
Bin Bai, Email: baibin513127@163.com.
Zhengyan Li, Email: 597886939@qq.com.
Xiaoyong Wu, Email: 15035150329@163.com.
Pengfei Yu, Email: yupengfei8989@163.com.
Xuzhao Li, Email: 251170583@qq.com.
Jichao Yin, Phone: 862989626312, Email: 83797727@163.com.
References
- 1.Bonenkamp JJ, Songun I, Hermans J, Sasako M, Welvaart K, Plukker JT, van Elk P, Obertop H, Gouma DJ, Taat CW, et al. Randomised comparison of morbidity after D1 and D2 dissection for gastric cancer in 996 Dutch patients. Lancet. 1995;345:745–748. doi: 10.1016/S0140-6736(95)90637-1. [DOI] [PubMed] [Google Scholar]
- 2.Cuschieri A, Fayers P, Fielding J, Craven J, Bancewicz J, Joypaul V, Cook P. Postoperative morbidity and mortality after D1 and D2 resections for gastric cancer: preliminary results of the MRC randomised controlled surgical trial. The Surgical Cooperative Group. Lancet. 1996;347:995–999. doi: 10.1016/S0140-6736(96)90144-0. [DOI] [PubMed] [Google Scholar]
- 3.Kodera Y, Sasako M, Yamamoto S, Sano T, Nashimoto A, Kurita A, Gastric Cancer surgery study Group of Japan Clinical Oncology G Identification of risk factors for the development of complications following extended and superextended lymphadenectomies for gastric cancer. Br J Surg. 2005;92:1103–1109. doi: 10.1002/bjs.4979. [DOI] [PubMed] [Google Scholar]
- 4.Park DJ, Lee HJ, Kim HH, Yang HK, Lee KU, Choe KJ. Predictors of operative morbidity and mortality in gastric cancer surgery. Br J Surg. 2005;92:1099–1102. doi: 10.1002/bjs.4952. [DOI] [PubMed] [Google Scholar]
- 5.Yasunaga H, Horiguchi H, Kuwabara K, Matsuda S, Fushimi K, Hashimoto H, Ayanian JZ. Outcomes after laparoscopic or open distal gastrectomy for early-stage gastric cancer: a propensity-matched analysis. Ann Surg. 2013;257:640–646. doi: 10.1097/SLA.0b013e31826fd541. [DOI] [PubMed] [Google Scholar]
- 6.Kim HH, Han SU, Kim MC, Hyung WJ, Kim W, Lee HJ, Ryu SW, Cho GS, Song KY, Ryu SY. Long-term results of laparoscopic gastrectomy for gastric cancer: a large-scale case-control and case-matched Korean multicenter study. J Clin Oncol. 2014;32:627–633. doi: 10.1200/JCO.2013.48.8551. [DOI] [PubMed] [Google Scholar]
- 7.Yu J, Hu J, Huang C, Ying M, Peng X, Wei H, Jiang Z, Du X, Liu Z, Liu H, et al. The impact of age and comorbidity on postoperative complications in patients with advanced gastric cancer after laparoscopic D2 gastrectomy: results from the Chinese laparoscropic gastrointestinal surgery study (CLASS) group. Eur J Surg Oncol. 2013;39:1144–1149. doi: 10.1016/j.ejso.2013.06.021. [DOI] [PubMed] [Google Scholar]
- 8.Kim SH, Son SY, Park YS, Ahn SH, Park DJ, Kim HH. Risk factors for anastomotic leakage: a retrospective cohort study in a single gastric surgical unit. J Gastric Cancer. 2015;15:167–175. doi: 10.5230/jgc.2015.15.3.167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Saito T, Kurokawa Y, Miyazaki Y, Makino T, Takahashi T, Yamasaki M, Nakajima K, Takiguchi S, Mori M, Doki Y. Which is a more reliable indicator of survival after gastric cancer surgery: postoperative complication occurrence or C-reactive protein elevation? J Surg Oncol. 2015;112:894–899. doi: 10.1002/jso.24067. [DOI] [PubMed] [Google Scholar]
- 10.Climent M, Hidalgo N, Vidal O, Puig S, Iglesias M, Cuatrecasas M, Ramon JM, Garcia-Albeniz X, Grande L, Pera M. Postoperative complications do not impact on recurrence and survival after curative resection of gastric cancer. Eur J Surg Oncol. 2016;42:132–139. doi: 10.1016/j.ejso.2015.08.163. [DOI] [PubMed] [Google Scholar]
- 11.Eto K, Hiki N, Kumagai K, Shoji Y, Tsuda Y, Kano Y, Yasufuku I, Okumura Y, Tsujiura M, Ida S, et al. Prophylactic effect of neoadjuvant chemotherapy in gastric cancer patients with postoperative complications. Gastric Cancer. 2018;21:703–709. doi: 10.1007/s10120-017-0781-y. [DOI] [PubMed] [Google Scholar]
- 12.Watanabe M, Kinoshita T, Tokunaga M, Kaito A, Sugita S. Complications and their correlation with prognosis in patients undergoing total gastrectomy with splenectomy for treatment of proximal advanced gastric cancer. Eur J Surg Oncol. 2018;44:1181–1185. doi: 10.1016/j.ejso.2018.03.013. [DOI] [PubMed] [Google Scholar]
- 13.Tsujimoto H, Ichikura T, Ono S, Sugasawa H, Hiraki S, Sakamoto N, Yaguchi Y, Yoshida K, Matsumoto Y, Hase K. Impact of postoperative infection on long-term survival after potentially curative resection for gastric cancer. Ann Surg Oncol. 2009;16:311–318. doi: 10.1245/s10434-008-0249-8. [DOI] [PubMed] [Google Scholar]
- 14.Sierzega M, Kolodziejczyk P, Kulig J. Impact of anastomotic leakage on long-term survival after total gastrectomy for carcinoma of the stomach. Br J Surg. 2010;97:1035–1042. doi: 10.1002/bjs.7038. [DOI] [PubMed] [Google Scholar]
- 15.Yoo HM, Lee HH, Shim JH, Jeon HM, Park CH, Song KY. Negative impact of leakage on survival of patients undergoing curative resection for advanced gastric cancer. J Surg Oncol. 2011;104:734–740. doi: 10.1002/jso.22045. [DOI] [PubMed] [Google Scholar]
- 16.Nagasako Y, Satoh S, Isogaki J, Inaba K, Taniguchi K, Uyama I. Impact of anastomotic complications on outcome after laparoscopic gastrectomy for early gastric cancer. Br J Surg. 2012;99:849–854. doi: 10.1002/bjs.8730. [DOI] [PubMed] [Google Scholar]
- 17.Li QG, Li P, Tang D, Chen J, Wang DR. Impact of postoperative complications on long-term survival after radical resection for gastric cancer. World J Gastroenterol. 2013;19:4060–4065. doi: 10.3748/wjg.v19.i25.4060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Tokunaga M, Tanizawa Y, Bando E, Kawamura T, Terashima M. Poor survival rate in patients with postoperative intra-abdominal infectious complications following curative gastrectomy for gastric cancer. Ann Surg Oncol. 2013;20:1575–1583. doi: 10.1245/s10434-012-2720-9. [DOI] [PubMed] [Google Scholar]
- 19.Kubota T, Hiki N, Sano T, Nomura S, Nunobe S, Kumagai K, Aikou S, Watanabe R, Kosuga T, Yamaguchi T. Prognostic significance of complications after curative surgery for gastric cancer. Ann Surg Oncol. 2014;21:891–898. doi: 10.1245/s10434-013-3384-9. [DOI] [PubMed] [Google Scholar]
- 20.Hayashi T, Yoshikawa T, Aoyama T, Hasegawa S, Yamada T, Tsuchida K, Fujikawa H, Sato T, Ogata T, Cho H, et al. Impact of infectious complications on gastric cancer recurrence. Gastric Cancer. 2015;18:368–374. doi: 10.1007/s10120-014-0361-3. [DOI] [PubMed] [Google Scholar]
- 21.Jin LX, Sanford DE, Squires MH, 3rd, Moses LE, Yan Y, Poultsides GA, Votanopoulos KI, Weber SM, Bloomston M, Pawlik TM, et al. Interaction of postoperative morbidity and receipt of adjuvant therapy on long-term survival after resection for gastric adenocarcinoma: results from the U.S. Gastric Cancer Collaborative. Ann Surg Oncol. 2016;23:2398–2408. doi: 10.1245/s10434-016-5121-7. [DOI] [PubMed] [Google Scholar]
- 22.Abdul Kader ATM, Murakami Y, Yoshimoto M, Onishi K, Kuroda H, Matsunaga T, Fukumoto Y, Takano S, Tokuyasu N, Osaki T, et al. Intra-abdominal complications after curative gastrectomies worsen prognoses of patients with stage II–III gastric cancer. Yonago Acta Medica. 2016;59:210–216. [PMC free article] [PubMed] [Google Scholar]
- 23.Li Z, Bai B, Zhao Y, Yu D, Lian B, Liu Y, Zhao Q. Severity of complications and long-term survival after laparoscopic total gastrectomy with D2 lymph node dissection for advanced gastric cancer: a propensity score-matched, case-control study. Int J Surg. 2018;54:62–69. doi: 10.1016/j.ijsu.2018.04.034. [DOI] [PubMed] [Google Scholar]
- 24.Tierney JF, Stewart LA, Ghersi D, Burdett S, Sydes MR. Practical methods for incorporating summary time-to-event data into meta-analysis. Trials. 2007;8:16. doi: 10.1186/1745-6215-8-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.The Newcastle-Ottawa Scale (NOS) for assessing the quality of nonrandomised studies in meta-analyses. [http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp].
- 26.Moher D, Liberati A, Tetzlaff J, Altman DG, Group P Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Ann Intern Med. 2009;151:264–269. doi: 10.7326/0003-4819-151-4-200908180-00135. [DOI] [PubMed] [Google Scholar]
- 27.Stroup DF, Berlin JA, Morton SC, Olkin I, Williamson GD, Rennie D, Moher D, Becker BJ, Sipe TA, Thacker SB. Meta-analysis of observational studies in epidemiology: a proposal for reporting. Meta-analysis Of Observational Studies in Epidemiology (MOOSE) group. JAMA. 2000;283:2008–2012. doi: 10.1001/jama.283.15.2008. [DOI] [PubMed] [Google Scholar]
- 28.Qu JL, Qu XJ, Li X, Zhang JD, Teng YE, Jin B, Zhao MF, Yu P, Liu J, Li DY, Liu YP. Early initiation of fluorouracil-based adjuvant chemotherapy improves survival in patients with resectable gastric cancer. J BUON. 2015;20:800–807. [PubMed] [Google Scholar]
- 29.O'Leary DP, Wang JH, Cotter TG, Redmond HP. Less stress, more success? Oncological implications of surgery-induced oxidative stress. Gut. 2013;62:461–470. doi: 10.1136/gutjnl-2011-300948. [DOI] [PubMed] [Google Scholar]
- 30.Kim MS, Kwon Y, Park EP, An L, Park H, Park S. Revisiting laparoscopic reconstruction for Billroth 1 versus Billroth 2 versus Roux-en-Y after distal gastrectomy: a systematic review and meta-analysis in the modern era. World J Surg. 2019. 10.1007/s00268-019-04943-x. [Epub ahead of print] PMID: 30756163. [DOI] [PubMed]
- 31.Watanabe Y, Watanabe M, Suehara N, Saimura M, Mizuuchi Y, Nishihara K, Iwashita T, Nakano T. Billroth-I reconstruction using an overlap method in totally laparoscopic distal gastrectomy: propensity score matched cohort study of short- and long-term outcomes compared with Roux-en-Y reconstruction. Surg Endosc. 2019. 10.1007/s00464-019-06688-z. [Epub ahead of print]PMID: 30758666 [DOI] [PubMed]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Table S1. NOS of Cohort studies. NOS of Case-control studies. (DOCX 27 kb)
Data Availability Statement
All data generated or analyzed during this study are included in the published articles which were listed in Table 1.